![]() Maria Cecília Feitoza Gomes1,
Maria Cecília Feitoza Gomes1, ![]() Patricia Charvet1,
Patricia Charvet1, ![]() Leonardo Mesquita Pinto2,
Leonardo Mesquita Pinto2, ![]() Ronaldo César Gurgel-Lourenço2,
Ronaldo César Gurgel-Lourenço2, ![]() Telton Pedro Anselmo Ramos3,
Telton Pedro Anselmo Ramos3, ![]() Sergio Maia Queiroz Lima4,
Sergio Maia Queiroz Lima4, ![]() Carlos Alberto de Sousa Rodrigues-Filho5,6,
Carlos Alberto de Sousa Rodrigues-Filho5,6, ![]() Danielle Sequeira Garcez7 and
Danielle Sequeira Garcez7 and ![]() Jorge Iván Sánchez-Botero8
Jorge Iván Sánchez-Botero8 ![]()
PDF: EN XML: EN | Supplementary: S1 | Cite this article
Abstract
Listas vermelhas, que avaliam o risco de extinção das espécies, são fundamentais na formulação de políticas públicas de gestão dos recursos naturais e mitigação de impactos. O objetivo desse estudo foi caracterizar o estado de conservação da ictiofauna continental do estado do Ceará, com dados de ocorrências ictiológicas e critérios da União Internacional para Conservação da Natureza. Oitenta e cinco espécies de peixes pertencentes a 59 gêneros, 25 famílias e oito ordens foram registradas no Ceará. Quarenta e três espécies foram consideradas menos preocupantes, quatro quase ameaçadas, 17 ameaçadas de extinção, 15 com dados insuficientes e seis espécies não foram avaliadas. Dentre as 17 espécies consideradas ameaçadas, cinco foram classificadas como Vulneráveis, cinco Em Perigo e sete Criticamente em Perigo. As bacias Metropolitana e Salgado apresentaram a maior riqueza (44 e 39 espécies, respectivamente), enquanto as bacias da Serra de Ibiapaba (6) e Curu (17) apresentaram as menores riquezas, indicando ampla variabilidade entre as bacias avaliadas. As bacias com maior número de espécies de peixes ameaçadas foram a Metropolitana, Litoral e baixo Jaguaribe. Esses resultados destacam a urgente necessidade de ações de conservação para salvaguardar a diversidade de peixes de água doce do Ceará, particularmente nas bacias com mais espécies ameaçadas.
Palavras-chave: Bacias hidrográficas, Biologia da conservação, Lista vermelha de espécies de peixes ameaçadas, Nordeste do Brasil, Peixes da Caatinga.
Introduction
Assessing the conservation status of species is essential for formulating public policies aimed at natural resource management and impact mitigation. Including freshwater fish as a target group for protection actions is particularly significant for aquatic ecosystems, as these species have rarely been prioritized in establishing conservation measures (see Frederico et al., 2021; Leal et al., 2021). Freshwater fish, in particular, are considered vulnerable and have historically been neglected in the northeastern region of Brazil (Berbel-Filho et al., 2018). The State of Ceará is located within the Mid-Northeastern Caatinga Ecoregion (MNCE), one of the least-known regions in the world regarding ichthyofaunal biodiversity (Berbel-Filho et al., 2018). Nonetheless, this region has important records of fish species (Rosa et al., 2003; Rodrigues-Filho et al., 2016; Lima et al., 2017). A notable increase in studies has challenged the notion that Ceará’s freshwater biodiversity might be underestimated. Information on population characteristics and species distribution in this region is crucial for developing extinction risk assessments or a Red List of Threatened Fish Species of Ceará, a fundamental tool for conservation initiatives.
The “Inventário da Fauna do Ceará” was launched by the “Programa Cientista Chefe”of the Government of the Ceará State (Law 17,378, January 4, 2021), containing 1,275 vertebrate species, including 502 fish species (400 marine and 102 freshwater), 140 mammals (25 marine and 115 continental), 133 reptiles, 57 amphibians, and 443 birds (SEMA, 2022). The list of freshwater fish of Ceará, compiled by Sánchez-Botero et al. (2021), identified 84 native species and 18 introduced. Of the total number of species in the ichthyofauna of Ceará, four were listed as threatened according to the Red List of the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio). Four were assessed as threatened in the International Union for Conservation of Nature (IUCN) Red List of Threatened Species (hereinafter IUCN Red List). However, the number of freshwater fish species considered threatened with extinction in Ceará may be underestimated.
State-level Red Lists are crucial for more regional assessments, which could foster better local conservation efforts with more specific biases for targeted issues depending on the region of the country. Red lists of threatened species of freshwater fish have been published in the states of Pará (the only one from North Brazil) (COEMA, 2007), Bahia (the only one from Northeast Brazil) (SEMA, 2017; Silva et al., 2020a), Minas Gerais (COPAM, 2010), Espírito Santo (IEMA, 2022), Rio de Janeiro (SEMA, 1998), and São Paulo (SMA, 2018) (Southeast Brazil), Paraná (Paraná, 2018), Santa Catarina (FATMA, 2011), and Rio Grande do Sul (SEMA, 2014) (South Brazil). Thus, most Brazilian states (about 65%) still do not have assessments regarding their threatened fish biodiversity, including the Ceará State.
Regarding the distribution of ichthyofauna in the river basins of Ceará State, it is noteworthy that only the Jaguaribe and Mundaú river basins have comprehensive information on the composition of this faunal group throughout most of their courses (Lima et al., 2017; Teixeira et al., 2017; Rodrigues-Filho et al., 2019; Silva et al., 2020b). The other river basins in the Ceará State (nine in total) have sporadic information on ichthyofauna and lack continuous monitoring, requiring greater sampling efforts to understand their composition. Among the most neglected sections of the state’s river basins in terms of ichthyofauna knowledge are the headwaters and streams. These areas remain poorly studied and are under constant threat from anthropogenic activities such as mining, removal of riparian vegetation, and the perennialization of water flow, among others (Chiu et al., 2017; Sánchez-Botero et al., 2017; Silva et al., 2020; Vorste et al., 2020; Terra et al., 2021).
Currently, there are serious risks to the maintenance of the biodiversity of freshwater fish in the Ceará State, due to projects with significant potential to impact the main river basins: pollution derived from mineral extraction, fragmentation of ecosystems due to dam construction, and the introduction of non-native species from the transposition of the São Francisco River. In light of this reality, the objective of this study is to assess the conservation status of freshwater fish species occurring in Ceará State.
Material and methods
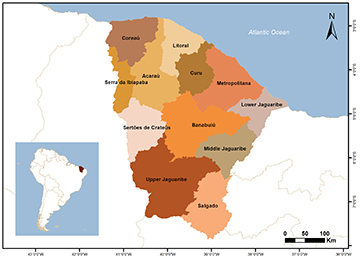
The continental regions of the State of Ceará, located within the Mid-Northeastern Caatinga Ecoregion (MNCE), comprise intermittent rivers and streams, cave streams, temporary pools, and marginal lagoons that harbor native and non-native ichthyofauna (Lima et al., 2017; Sánchez-Botero et al., 2021; Terra et al., 2021). Furthermore, water use management strategies implemented by the human population in the region have led to the development of anthropogenic features such as reservoirs (Morais, Pinheiro, 2011). The territory is mostly located in the semi-arid northeastern region, but it also includes humid and sub-humid areas where elevated mountain ranges create exception islands (Moro et al., 2015). According to the Companhia de Gestão dos Recursos Hídricos (COGERH, 2022), a total of 12 Hydrographic Regions were defined within the scope of the State Water Resources Policy, linked to the Secretaria dos Recursos Hídricos (SRH). These include the basins of Coreaú, Acaraú, Litoral, Curu, Jaguaribe (which contains the sub-basins of lower Jaguaribe, middle Jaguaribe, upper Jaguaribe, Banabuiú, and Salgado), Metropolitana, Serra da Ibiapaba, and Sertões de Crateús (Fig. 1).
FIGURE 1| Location of the hydrographic basins of the Ceará State, Brazil.
The list of native ichthyofauna in the Ceará State (Sánchez-Botero et al., 2021), available on the Secretaria do Meio Ambiente e Mudança do Clima (SEMA) website, was updated based on a review of the literature on the composition of freshwater fish species in the Mid-Northeastern Caatinga Ecoregion (MNCE), and records from the ichthyological collections of the Universidade Federal do Rio Grande do Norte (UFRN) and Universidade Federal da Paraíba (UFPB) and queries on SpeciesLink network (CRIA, 2024). Other institutions follow Sabaj (2022). The database of the ichthyofauna composition and occurrences gathered from the literature review was based on publications in taxonomy, ecology, biogeography, and ethnoichthyology from the following basins: Acaraú (Gurgel-Lourenço et al., 2013, 2015; Batista et al., 2016; Faustino, Terra, 2019; Oliveira et al., 2023; Mapurunga et al., 2024; Oliveira et al., 2024), Coreaú (Rodrigues-Filho et al., 2016, 2018a; Gurgel-Lourenço et al., 2017; Sánchez-Botero et al., 2017; Faustino, Terra, 2019), Curu (Silva et al., 2005; Bezerra et al., 2013; Manna et al., 2017, 2018), lower Jaguaribe (Abrantes et al., 2020; Silva et al., 2020), middle Jaguaribe (Abrantes et al., 2020), upper Jaguaribe (Rodrigues-Filho et al., 2016, 2018b, 2019; Gurgel-Lourenço et al., 2017; Berbel-Filho et al., 2018; Silva et al., 2020; Ramos et al., 2020), Banabuiú (Abrantes et al., 2020), Salgado (Rodrigues-Filho et al., 2016; Gurgel-Lourenço et al., 2017), Litoral (Teixeira et al., 2017; Berbel-Filho et al., 2018; Faustino, Terra, 2019; Abrantes et al., 2020; Gonçalves-Silva et al., 2022), Metropolitana (Rosa, Groth, 2004; Chaves et al., 2013; Sánchez-Botero et al., 2014a; Bezerra et al., 2017; Berbel-Filho et al., 2018; Abrantes et al., 2020, 2023), Sertões de Crateús (Rodrigues-Filho et al., 2016; Berbel-Filho et al., 2018), and Serra da Ibiapaba (Sánchez-Botero et al., 2014b; Rodrigues-Filho et al., 2016; Gurgel-Lourenço et al., 2017).
Based on geographic distribution information, we calculated each species’ Extent of Occurrence (EOO; the area contained within the shortest continuous imaginary boundary, which can be drawn to encompass all the points of occurrence of a species) and Area of Occupancy (AOO; a scaled metric that represents the area of suitable habitat currently occupied by the taxon) (IUCN, 2024). Information regarding the classification of ichthyofauna concerning endemism in the Caatinga and MNCE was sourced from Rosa et al. (2003), Gurgel-Lourenço et al. (2013), Ramos et al. (2014), Rodrigues-Filho et al. (2016), Bezerra et al. (2017), Lima et al. (2017), Berbel-Filho et al. (2018), Silva et al. (2020), Abrantes et al. (2020, 2023), and Lustosa-Costa et al. (2024).
Following IUCN recommendations for freshwater fish species, the Minimum Convex Polygon (MCP) was used to calculate the Extent of Occurrence (EOO), encompassing all occurrence points of the species (Brazil; IUCN, 2024). The information indicates whether EOO is < 20,000 km2, < 5,000 km2, or < 100 km2 for each species. It also assesses the conditions of the Area of Occupancy (AOO), determining the qualitative degree of fragmentation and the number of locations where the fish species is found. It notes if there is a continued decline in EOO and/or AOO, in addition to habitat quality conditions and/or number of mature individuals, where available. AOO is noted if it is < 2,000 km2, < 500 km2, or < 10 km2. The thresholds of EOO and AOO areas assessed here were used, along with other criteria, to assess the species’ extinction risk and, when applicable, categorize them as Vulnerable, Endangered, and Critically Endangered (IUCN, 2024).
Occurrence points registered in Ceará State, AOO, and EOO were obtained for each species. Calculation of AOO and EOO values in km2 was performed using QGIS software (QGIS, 2024). Concurrently, maps of species richness and the percentage of threatened species per basin in Ceará were developed based on ichthyofauna occurrence data.
To complement occurrence data for fish not documented in scientific literature, historical documents from the Departamento Nacional de Obras Contra as Secas (DNOCS) (Ihering, 1933; Menezes, 1956; DNOCS, 1963; Peixoto, Gurgel, 1978; Gurgel, 1979), Environmental Impact Studies and Environmental Impact Reports provided by the Superintendência Estadual de Meio Ambiente (SEMACE), and the Instituto Brasileiro do Meio Ambiente e Recursos Naturais Renováveis (IBAMA) were consulted.
Set of information related to the number of occurrences of fish species recorded in the basins of Ceará, AOO, and EOO, spatial distribution, population characteristics, occurrence habitats, potential threats, as well as information regarding their conservation status in global (IUCN) and national (ICMBio) red lists, were applied the criteria and categories of the IUCN Red List. This has resulted in the unprecedented determination of extinction risk for continental fish species in the state.
Results
A total of 1,423 records from ichthyological collections with geographic coordinates of species distributed in Ceará State were evaluated. Based on available literature and collection records, 85 species of native fish were recorded across twelve continental basins in Ceará State, with 43 (51%) being endemic to the Caatinga, according to Lima et al. (2017) (Tab. 1). Characiformes accounted for 47% (40) and Siluriformes for 32% (27) of the species; the remaining six orders comprised approximately 20%. Regarding families, Loricariidae represented 15% (13), Acestrorhamphidae 13% (11), and Rivulidae 8% (7) of the species; other families constituted 64%.
TABLE 1 | Species occurrence by basin, voucher numbers. AC = Acaraú, UJ = upper Jaguaribe, MJ = middle Jaguaribe, LJ = lower Jaguaribe, BA = Banabuiú, CO = Coreaú, CU = Curu, LI = Litoral, ME = Metropolitana, AS = Salgado, SI = Serra da Ibiapaba, SC = Sertões de Crateús.
Species | Occurrences (basins) | Vouchers/References |
Anablepsoides cearensis | LI, ME | LI: UFRN 2576, UFRN 2530, UFRN 2657, UFPB 11017, UFPB 11931//ME: UFRN 2673, UFRN 5847, UFPB 11930 |
Ancistrus damasceni | SC | UFPB 9282 |
Apareiodon davisi | SA | UFPB 9139 |
Aspidoras carvalhoi | ME | UFRN 4840 |
Aspidoras raimundi | AC, BA CO, SA, SI | AC: UFRN 2895, UFRN 2897, UFRN 3726, UFRN 3735, UFRN 3745, UFRN 4903, UFPB 7064, UFPB 7616//BA: UFPB 7644//CO: UFRN 1466, UFRN 2662, UFPB 11947, UFPB 9247, UFPB 9251//SA: UFPB 9427, UFRN 1521, UFRN 1580, UFRN 1521, UFRN 1885, UFRN 2096, UFRN 4392, UFPB 9427, UFPB 12005//SI: UFPB 9415 |
Aspidoras rochai | ME | MZUSP 5300 |
Astyanax bimaculatus | AC, AJ, BA BJ, CO, CU, LI, ME, MJ, SA, SC | AC: UFRN 897, UFRN 5316, UFRN 5447, UFRN 5468, UFRN 5470, UFRN 4904, UFRN 5452, UFPB 7056, UFPB 7059, UFPB 7598, UFPB 7614, UFPB 7622, UFPB 7623, UFPB 12088, UFPB 12090, UFPB 12105, UFPB 12107//AJ: UFRN 331, UFRN 349, UFRN 358, UFRN 360, UFRN 372, UFRN 375, UFRN 382, UFRN 383, UFRN 403, UFRN 1190, UFRN 1263, UFRN 1276, UFRN 1515, UFRN 1591, UFRN 2238, UFRN 3228, UFRN 3807, UFPB 6844, UFPB 11829//BA: UFRN 3242, UFRN 3422, UFRN 3527, UFRN 3657, UFPB 6846, UFPB 7607, UFPB 7626, UFPB 7627, UFPB 7638, UFPB 7639, UFPB 11752, UFPB 11769, UFPB 11773, UFPB 11779, UFPB 11788, UFPB 11796, UFPB 11803, UFPB 12089, UFPB 12106 //BJ: UFRN 3224, UFRN 4323, UFRN 4341, UFRN 2905, UFRN 2914, UFRN 5681//CO: UFRN 2675, UFRN 3826, UFRN 5336, UFRN 5347, UFRN 5349, UFRN 5351, UFRN 5352, UFRN 5353, UFRN 5355, UFRN 5357, UFRN 5361, UFRN 5362, UFRN 5364, UFRN 5365, UFRN 5368, UFRN 5397, UFRN 5421, UFRN 5429, UFRN 5474, UFRN 5516, UFRN 5517, UFRN 5526, UFRN 5529, UFRN 5531, UFRN 5532, UFRN 5535, UFRN 5537, UFRN 5542, UFRN 5616, UFRN 5617, UFPB 9254// CU: UFRN 4616, UFPB 7070// LI: UFRN 1420, UFRN 1428, UFRN 1441, UFRN 1445, UFRN 2560, UFRN 2567, UFRN 2573, UFRN 2595, UFRN 2634, UFRN 2651, UFRN 5384, UFPB 11025, UFPB 11030, UFPB 11164//ME: UFRN 4763, UFRN 4821, UFRN 4838, UFRN 4890, UFRN 4899, UFPB 6847, UFPB 6848, UFPB 6849, UFPB 6850, UFPB 9157, UFPB 9172, UFPB 11714, UFPB 11740, UFPB 12951//MJ: UFRN 3232, UFRN 3240, UFPB 9146, UFPB 9190//SA: UFRN 320, UFRN 381, UFRN 1171, UFRN 1195, UFRN 1200, UFRN 1209, UFRN 1339, UFRN 1504, UFRN 1518, UFRN 1876, UFRN 1886, UFRN 1888, UFRN 2027, UFRN 2116, UFRN 3235, UFRN 3808, UFRN 2919, UFRN 3827, UFRN 5069, UFPB 6735, UFPB 6842, UFPB 9132, UFPB 11809, UFPB 11824, UFPB 12013//SC: UFRN 5081, UFPB 9283 |
Awaous tajasica | BJ, LI, ME | BJ: OBIS_BR 266//LI: Teixeira et al. (2017)//ME: ANSP 84174 |
Callichthys callichthys | LI, ME | LI: UFRN 2607, UFRN 2617//ME: UFPB 13111, UFRN 1875 |
Characidium bimaculatum | AC, AJ, BA, CO, LI, ME, MJ, SA, SI | AC: UFPB 7060, UFPB 7619//AJ: UFRN 387, UFPB 11799, UFPB 11806//BA: UFPB 7608, UFPB 7635//CO: UFRN 2686, UFPB 9256//LI: UFRN 1439, UFRN 2636//ME: UFPB 6868, UFPB 6869, UFPB 11791//MJ: UFRN 2680, UFRN 3233//SA: UFRN 1320, UFRN 1503, UFRN 1512, UFRN 1673, UFRN 1675, UFRN 1676, UFRN 1890, UFRN 2021, UFRN 2197, UFPB 6731, UFPB 6866//SI: UFPB 11307 |
Cheirodon jaguaribensis | BA, BJ, CO, LI, ME, MJ, SA | BA: UFRN 3244, UFPB 9105, UFPB 11724, UFPB 11728, UFRN 3588//BJ: UFRN 3247, UFRN 3810, UFRN 3829//CO: UFPB 11965//LI: UFRN 1433, UFRN 3777, UFPB 11035, UFPB 11165//ME: UFPB 11617//MJ: UFRN 3239, UFRN 3825//SA: UFRN 1652 |
Cichlasoma orientale | AC, AJ, BA, BJ, CO, CU, LI, ME, MJ, SA, SC, SI | AC: UFRN 918, UFRN 919, UFRN 2899, UFRN 5313, UFRN 5318, UFRN 5324, UFRN 5440, UFRN 5455, UFRN 5456, UFRN 5459, UFRN 5463, UFPB 7595, UFPB 7613, UFPB 12097, UFPB 12116, UFPB 12118//AJ: UFRN 333, UFRN 340, UFRN 353, UFRN 361, UFRN 367, UFRN 369, UFRN 379, URFN 676, URFN 677, UFRN 678, UFRN 679, UFRN 680, UFRN 681, UFRN 1165, UFRN 1205, UFRN 1316, UFRN 1332, UFRN 1334, UFRN 1517, UFRN 1566, UFRN 1844, UFRN 1847, UFRN 2244, UFRN 2289, UFRN 3211, UFPB 6872//BA: UFRN 3243, UFRN 3524, UFRN 3525, UFPB 6873, UFPB 6874, UFPB 7603, UFPB 7630, UFPB 11622, UFPB 11719, UFPB 11794, UFPB 11800, UFPB 11819, UFPB 11823, UFPB 11827, UFPB 12098, UFPB 12117//BJ: UFRN 2912, UFRN 3227, UFRN 4337, UFRN 5684//CO: UFRN 939, UFRN 940, UFRN 941, UFRN 942, UFRN 1186, UFRN 1187, UFRN 2683, UFRN 5334, UFRN 5358, UFRN 5370, UFRN 5393, UFRN 5419, UFRN 5420, UFRN 5431, UFRN 5432, UFRN 5499, UFRN 5502, UFRN 5515, UFRN 5522, UFRN 5527, UFRN 5533, UFRN 5541, UFPB 9249//CU: UFRN 1011, UFRN 1012, UFRN 1013, UFPB 7067, UFPB 7081, UFPB 11707//LI: UFRN 1426, UFRN 1427, UFRN 2520, UFRN 2528, UFRN 2534, UFRN 2536, UFRN 2578, UFRN 2585, UFRN 2586, UFRN 2593, UFRN 2601, UFRN 2603, UFRN 2614, UFRN 2641, UFRN 2656, UFRN 5383, UFRN 5388, UFRN 5475, UFRN 5476, UFRN 5497, UFPB 11019, UFPB 11024, UFPB 11031, UFPB 11038, UFPB 11109, UFPB 11113, UFPB 11161//ME: UFRN 2671, UFRN 2882, UFRN 4836, UFRN 4837, UFRN 4892, UFPB 6876, UFPB 6877, UFPB 6878, UFPB 6879, UFPB 9150, UFPB 10344, UFPB 11821, UFPB 11873//MJ: UFRN 2682, UFRN 3214, UFRN 3215, UFPB 9185//SA: UFRN 0407, UFRN 1162, UFRN 1194, UFRN 1279, UFRN 1324, UFRN 1327, UFRN 1330, UFRN 2026, UFRN 2921, UFPB 6870, UFPB 6871, UFPB 9138, UFPB 11753, UFPB 11833, UFPB 12011//SC: UFRN 5079//SI: UFPB 13250 |
Cichlasoma sanctifranciscense | AJ, SC, SI | AJ: UFRN 1845//SC: UFPB 9260, UFPB 12185, UFPB 13248//SI: UFPB 12940, UFPB 13183 |
Compsura heterura | AC, BA, CO, CU, LI, ME, SC | AC: UFRN 5453, UFPB 7618//BA: UFRN 3205, UFRN 5113, UFPB 11715, UFPB 11802//CO: UFRN 5343, UFRN 5345// CU: UFPB 7077//LI: UFRN 1430, UFRN 2561, UFRN 2638, UFRN 2644, UFRN 3198, UFRN 5424//ME: UFPB 6862, UFPB 6863, UFPB 6864, UFPB 6865, UFPB 11730, UFPB 11775//SC: UFPB 10612 |
Ctenobrycon kennedyi | AJ, BA, BJ, SA | AJ: UFPB 11734//BA: UFPB 11798//BJ: UFRN 2906, UFRN 4340//SA: UFPB 12012 |
Ctenobrycon spilurus | BA, BJ | BA: UFRN 3209//BJ: UFRN 3230 |
Cynolebias microphthalmus | BJ | UFRJ 4860, UFRJ 4878 |
Geophagus brasiliensis | AJ, SA | AJ: UFRN 362, UFRN 589, UFRN 590, UFRN 1622//SA: UFRN 414 |
Hemigrammus guyanensis | LI, ME | LI: UFRN 2533, UFRN 2599, UFPB 11023, UFPB 11032//ME: UFRN 2693, UFPB 10337 |
Hemigrammus marginatus | AJ, BA, BJ, LI, MJ, SA | AJ: UFRN 1270, UFRN 1277//BA: UFPB 11736// BJ: UFRN 2915, UFRN 4325//LI: UFRN 5425//MJ: UFPB 9476//SA: UFRN 1173, UFRN 1280, UFRN 1336, UFPB 6898 |
Hemigrammus rodwayi | AC, AJ, BA, BJ, CO, CU, LI, ME, SA, SC | AC: UFPB 7048, UFPB 7055//AJ: UFRN 3820, UFPB 11722, UFPB 11766//BA: UFPB 9110, UFPB 11748, UFPB 6899//BJ: UFRN 2911, UFRN 4338// CO: UFRN 3778, UFPB0011952//CU: UFPB 7069//LI: UFRN 2521, UFRN 2572, UFRN 2592, UFRN 2598, UFRN 2628, UFRN 2640, UFRN 2525, UFRN 2637, UFRN 2653, UFRN 2658, UFPB 11036//ME: UFRN 2700, UFPB 9471, UFPB 10335//SA: UFPB 11832/ SC: UFPB 10611 |
Hemiodus parnaguae | SC | UFRN 5090, UFPB 9270, UFPB 9271 |
Hoplerythrinus unitaeniatus | CO, ME, SI | CO: UFPB 11959//ME: UFPB 10343, UFPB 12058//SI: UFPB 10614 |
Hoplias malabaricus | AC, AJ, BA, BJ, CO, CU, LI, ME, SA, SC | AC: UFRN 931, UFRN 932, UFRN 933, UFRN 3741, UFRN 5321, UFRN 5322, UFRN 5323, UFRN 5325, UFRN 5326, UFRN 5328, UFRN 5443, UFRN 5444, UFRN 5445, UFRN 5460, UFRN 5462, UFRN 5464, UFRN 5465, UFRN 5471, UFPB 7049, UFPB 7594, UFPB 7621, UFPB 12093, UFPB 12112, UFPB 12114//AJ: UFRN 335, UFRN 341, UFRN 364, UFRN 373, UFRN 405, UFRN 603, UFRN 605, UFRN 1208, UFRN 1315, UFRN 1526, UFRN 1617, UFRN 1846, UFRN 3213, UFPB0011792//BA: UFRN 3220, UFRN 3428, UFRN 3656, UFPB 6886, UFPB 7604, UFPB 7628, UFPB 7641, UFPB 11742, UFPB 11778, UFPB 12113//BJ: UFRN 2907, UFRN 5677//CO: UFRN 2684, UFRN 5367, UFRN 5391, UFRN 5423, UFRN 5427//CU: UFPB 7071//LI: UFRN 2582, UFRN 2589, UFRN 2605, UFRN 2609, UFRN 2633, UFRN 2652, UFRN 5385, UFRN 5478, UFPB 11018, UFPB 11026, UFPB 11163//ME: UFRN 4834, UFPB 6887, UFPB 6888, UFPB 6889, UFPB 9153, UFPB 9155, UFPB 10342, UFPB 11762, UFPB 11782, UFPB 11815//SA: UFRN 0397, UFRN 1325, UFRN 1328, UFRN 1615, UFRN 1794, UFRN 1795, UFRN 1799, UFRN 2194, UFRN 2918, UFPB 6732, UFPB 6883, UFPB 6884, UFPB 11836, UFPB 12006//SC: UFRN 3429, UFPB 9275, UFPB 9276, UFPB 9277 |
Hoplosternum littorale | ME, MJ, SA | ME: UFRN 4814, UFPB 9151//MJ: UFRN 4460//SA: UFPB 12085 |
Hyphessobrycon piabinhas | AC, AJ, BA, BJ, LI, ME, SA | AC: UFPB 7617, UFPB 9073//AJ: UFRN 3824//BA: UFPB 7633, UFPB 11729, UFPB 9521, UFPB 7634//BJ: UFRN 3813//LI: UFPB 11162, UFRN 2562, UFRN 2602, UFRN 2635, UFRN 2643, UFRN 2648, UFRN 2522, UFRN 2531//ME: UFPB 10336//SA: UFPB 6739, UFRN 2562, UFRN 2602, UFRN 2635, UFRN 2643, UFRN 2648, UFRN 2522, UFRN 2531 |
Hypostomus johnii | SC | UFPB 9266, UFPB 9267, UFPB 9268 |
Hypostomus pusarum | AC, AJ, BA, CO, CU, LI, ME, MJ, SA | AC: UFRN 891, UFRN 892, UFRN 893, UFRN 894, UFRN 4902, UFRN 5436, UFPB 9121, UFRN 3734, UFRN 2868, UFPB 7050, UFPB 7062, UFPB 7615, UFPB 12101, UFPB 12122, UFRN 3734, UFRN 2868//AJ: UFRN 352, UFRN 359, UFRN 370, UFRN 607, UFRN 608, UFRN 1509, UFRN 1790, UFRN 1834, UFRN 3815, UFRN 4466, UFPB 11759, UFRN 377, UFRN 393, UFRN 1499, UFRN 1624, UFRN 1813, UFRN 5061, UFRN 5076, UFPB 7700//BA: UFPB 9226, UFPB 9222, UFPB 6896, UFPB 11781, UFPB 11804//CO: UFRN 5392, UFPB 11960// CU: UFPB 7068//LI: UFRN 2584, UFRN 2608//ME: UFPB 11765, UFPB 11835, UFRN 2870, UFRN 4888, UFPB 7697, UFPB 9223, UFPB 6892, UFPB 6893, UFPB 6894, UFPB 7692, UFPB 11939, UFPB 11943, UFPB 7698, UFPB 9220, UFPB 11765, UFPB 11835//MJ: UFRN 4463, UFPB 7699//SA: UFRN 1810, UFRN 2017, UFPB 12009, UFRN 377, UFRN 393, UFRN 1168, UFRN 1499, UFRN 1506, UFRN 1624, UFRN 1811, UFRN 1813, UFRN 2189, UFRN 5061, UFRN 5076, UFPB 6890, UFPB 6891, UFPB 6895, UFPB 6897, UFPB 9225, UFPB 7700, UFRN 608 |
Hypostomus sertanejo | ME, MJ, SA | ME: UFRN 4887, UFRN 4896, UFRN 4898, UFPB 7694//MJ: UFRN 4462, UFPB 7696// SA: UFRN 5062, UFRN 5070, UFPB 7691, UFPB 9224 |
Hypsolebias antenori | BA, BJ | BA: MZUSP 14755//BJ: UFRN 5842, UFRN 5848, UFPB 14125, UFPB 14134, UFPB 14146, UFPB 14149, UFPB 14155, UFPB 14743, UFPB 14900, MNRJ 19465, MNRJ 4542, UFRJ 4860, UFRJ 4864 |
Hypsolebias gongobira | ME | UFRN 5847, UFRN 6029, MNRJ 54900, MZUSP 129607 |
Hypsolebias longignatus | ME | ME: UFRN 5845, UFPB 12084, UFPB 12676, UFRJ 8764 |
Hypsolebias martinsi | LI | LI: UFRN 5843, UFRN 5844, UFRN 5414, UFRN 5413, ZUEC 10791 |
Knodus victoria | SI | UFPB 10615 |
Kryptolebias hermaphroditus | CO | CO: UFRN 5231, UFRN 5232 |
Leporinus piau | AC, AJ, BA, CO, CU, LI, ME, MJ, SA, SC | AC: UFRN 920, UFRN 923, UFRN 924, UFRN 925, UFRN 927, UFPB 7052, UFPB 7596, UFPB 12115//AJ: UFRN 599, UFRN 600, UFRN 3814, UFRN 3821// BA: UFPB 11732, UFPB 11822, UFPB 11826//CO: UFPB 11961//CU: UFPB 7080//LI: UFRN 1421, UFRN 1440//ME: UFRN 3661, UFRN 4841, UFRN 4893, UFPB 9156, UFPB 9176// MJ: UFPB 9145//SA: UFRN 0601, UFRN 5063, UFRN 5066, UFPB 6743//SC: UFRN 5085, UFPB 9259 |
Leporinus taeniatus | AC, AJ, BA, MJ, SA | AC: UFRN 5441//AJ: UFRN 596, UFRN 597, UFRN 598, UFRN 1273, UFRN 1510, UFRN 1828, UFRN 1840, UFRN 3222//BA: UFRN 3199, UFPB 7605, UFPB 11777// MJ: UFPB 9144, UFPB 9184//SA: UFRN 595, UFRN 1458, UFRN 1836, UFRN 5073, UFPB 9133, UFPB 11834, UFPB 12325 |
Loricariichthys derbyi | AJ, BJ, ME, MJ, SA | AJ: UFRN 586, UFRN 587//BJ: UFRN 3223//ME: UFRN 4453, UFRN 4894, UFPB 7659//MJ: UFPB 7793, UFRN 4451//SA: UFRN 1837, UFRN 5059, UFRN 5071 |
Megalamphodus bentosi | ME | UFRN 2691, UFPB 10338 |
Megalechis thoracata | ME | UFPB 11874 |
Moenkhausia costae | AJ, ME, SA | AJ: UFRN 1278//ME: UFRN 4567, UFRN 4694, UFRN 4851, UFPB 9174//SA: UFRN 1283, UFRN 1337, UFRN 1623 |
Moenkhausia intermedia | AJ, BA, ME, MJ, SA | AJ: UFPB 11739//BA: UFPB 6908, UFPB 11737, UFPB 11774, UFPB 11790// ME: UFPB 9173//MJ: UFPB 9142, UFPB 9188// SA: UFPB 6907, UFPB 11757 |
Nannostomus beckfordi | LI, ME | LI: UFRN 2535, UFRN 2591, UFRN 2600, UFRN 2523, UFRN 2659, UFPB 11021, UFPB 11037, UFPB 11168//ME: UFRN 2672, UFRN 2936, UFRN 2937 |
Parotocinclus cearensis | AC, BA, CO, LI, ME, SA, SC | AC: UFRN 2698, UFRN 2699, UFRN 2884, UFRN 2885, UFRN 2886, UFRN 2887, UFPB 7610, UFPB 12123//BA: UFRN 3426, UFPB 7637, UFPB 7643//CO: UFRN 1120, UFRN 1473, UFRN 5434, UFPB 9248, UFPB 11948//LI: UFRN 1422, UFRN 1423//ME: UFRN 4839, UFPB 6919, UFPB 11717, UFPB 11892, UFPB 11941// SA: UFRN 388, UFRN 404, UFRN 1183, UFRN 1242, UFRN 1260, UFRN 1295, UFRN 1505, UFRN 1581, UFRN 1873, UFRN 1881, UFRN 2024, UFRN 2087, UFRN 2258, UFRN 5058, UFRN 5088, UFRN 3216//SC: UFPB 7033 |
Parotocinclus haroldoi | CO | UFRN 1294, UFRN 5726, UFPB 11880, UFPB 11955, UFPB 11962 |
Parotocinclus jumbo | AJ, BA, MJ | AJ: UFRN 350//BA: UFPB 11869//MJ: UFPB 12003 |
Parotocinclus spilurus | AJ, SA | AJ: UFRN 345, UFRN 3806, UFRN 3830, UFPB 12077//SA: UFRN 321, UFRN 376, UFRN 389, UFRN 1248, UFRN 1252, UFRN 1253, UFRN 1255, UFRN 1300, UFRN 5057 |
Phenacogaster calverti | AC, CO, LI, ME, SC | AC: UFRN 5467, UFPB 7047, UFPB 7053//CO: UFRN 2511, UFRN 5333, UFRN 5519, UFRN 5528, UFRN 5539, UFRN 5543, UFPB 11950, UFPB 11963//LI: UFRN 2557, UFRN 2569, UFRN 2580, UFRN 2646, UFRN 5426, UFRN 5496, UFPB 11033//ME: UFPB 6915, UFPB 9170, UFPB 11818, UFPB 11942//SC: UFPB 9262 |
Pimelodella dorseyi | AJ, SA | AJ: UFRN 1821//SA: UFRN 409, UFRN 5064, UFRN 5072, UFPB 6911, UFPB 6912, UFPB 9136 |
Pimelodella parnahybae | SC | UFRN 5086, UFPB 9279 |
Pimelodella witmeri | AJ, BA, MJ | AJ: UFRN 588//BA: UFPB 6913//MJ: UFPB 9147 |
Pimelodella wolfi | ME | ME: ANSP 69388, ANSP 69389 |
Platydoras brachylecis | SC | UFPB 9272 |
Poecilia sarrafae | AC, LI | AC: UFPB 14646//LI: UFRN 2574, UFRN 2575, UFRN 2579, UFRN 2529, UFRN 2654, UFPB 11022, UFPB 11034, UFPB 11116, UFPB 11167, UFPB 14645 |
Poecilia vivipara | AC, AJ, BA, BJ, CO, CU, LI, ME, MJ, SA, SC | AC: UFRN 5439, UFRN 5472, UFPB 7602, UFPB 7611, UFPB 12104, UFPB 12127//AJ: UFRN 332, UFRN 337, UFRN 365, UFRN 371, UFRN 394, UFRN 396, UFRN 399, UFRN 1314, UFRN 2243, UFRN 2594, UFPB 6927, UFPB 11621, UFPB 11750//BA: UFRN 2910, UFRN 3203, UFRN 4336//BJ: UFRN 3210, UFRN 3423, UFRN 3529, UFRN 3659, UFPB 6928, UFPB 6929, UFPB 7631, UFPB 11747, UFPB 11754, UFPB 11763, UFPB 11795//CO: UFRN 5233, UFRN 5240, UFRN 5340, UFRN 5346, UFRN 5354, UFRN 5356, UFRN 5360, UFRN 5363, UFRN 5369, UFRN 5371, UFRN 5394, UFPB 9250, UFPB 9252, UFPB 9253, UFPB 9255, UFPB 11956//CU: UFRN 1073, UFRN 1075, UFRN 1076, UFRN 4644, UFPB 7073//LI: UFRN 1419, UFRN 1437, UFRN 2564, UFRN 2568, UFRN 2621, UFRN 2649, UFRN 5374, UFRN 5508, UFPB 11020, UFPB 11028, UFRN 2594//ME: UFRN 3421, UFRN 4857, UFRN 5165, UFRN 5185, UFRN 5187, UFRN 5188, UFPB 6931, UFPB 6932, UFPB 6933, UFPB 6934, UFPB 11725, UFPB 11731, UFPB 11745, UFPB 11772, UFPB 11879//MJ: UFRN 3238//SA: UFRN 400, UFRN 1172, UFRN 1262, UFRN 1323, UFRN 1326, UFRN 1456, UFRN 3202, UFPB 6740, UFPB 6924, UFPB 6925, UFPB 11785//SC: UFRN 5087 |
Prochilodus brevis | AC, AJ, BA, CU, LI, ME, SA | AC: UFRN 895, UFRN 896, UFRN 4905, UFRN 5437, UFRN 5457, UFPB 12094, UFRN 895, UFRN 896//AJ: UFRN 593, UFRN 594, UFRN 1792, UFRN 1801, UFRN 1804, UFRN 3819//BA: UFPB 6917, UFPB 11767, UFPB 11814, UFPB 11817//CU: UFPB 6918, UFPB 7078//LI: UFRN 3530, UFRN 5386//ME: UFRN 3425, UFRN 4897, UFPB 9149, UFPB 9154, UFPB 9179, UFPB 12064//SA: UFRN 5149, UFPB 6734, UFPB 6916, UFPB 9141, UFPB 11811, UFPB 11830 |
Prochilodus lacustris | SC | UFPB 9258 |
Psalidodon fasciatus | AC, AJ, BA, CO, CU, ME, SA,SC | AC: UFPB 7054, UFPB 7061, UFPB 7599, UFPB 12091, UFPB 12108, UFPB 12109//AJ: UFPB 11756//BA: UFPB 6853, UFPB 11624, UFPB 11721//CO: UFPB 11949, UFPB 11964//CU: UFPB 7074// ME: UFPB 6854, UFPB 6855, UFPB 6856, UFPB 9171, UFPB 11716, UFPB 11738, UFPB 11933//SA: UFPB 6736, UFPB 9137, UFPB 11812, UFPB 12010//SC: UFPB 9261 |
Psectrogaster rhomboides | AC, AJ, SA, SC | AC: UFRN 903, UFRN 904, UFRN 905//AJ: UFRN 4465// SA: UFPB 6923, UFPB 11764, UFPB 11831//SC: UFPB 9284 |
Psectrogaster saguiru | AC, CU, BJ | AC: USNM 267318//CU:USNM 220009//BJ: ANSP 88590 |
Pseudancistrus genisetiger | AJ, MJ, SA | AJ: UFRN 1793, UFRN 1812//MJ: UFPB 9183//SA: UFRN 0411, UFRN 1477, UFRN 5060, UFRN 5068, UFPB 6910 |
Pseudancistrus papariae | AJ | AJ: ANSP 69442 |
Pterengraulis atherinoides | CU, BJ | CU: Silva et al. (2005)//BJ: ANSP 69370 |
Pterygoplichthys parnaibae | SC | UFPB 9281 |
Pygocentrus nattereri | AC, AJ, SA, SC | AJ: UFRN 1839//SA: UFRN 5074//SC: UFPB 10613 |
Rhamdia quelen | SA | UFRN 1201, UFRN 1388, UFRN 1927, UFRN 3234, UFRN 1472, UFRN 1893 |
Saxatilia brasiliensis | AC, CO, CU, MJ, SA, BA LI, ME, SC | AC: UFRN 5314, UFRN 5319, UFRN 5320, UFRN 5438, UFRN 5442, UFRN 5454, UFRN 5466, UFPB 7063//CO:UFRN 5332, UFRN 5428, UFRN 5430//CU:UFPB 7076//AJ: UFPB0012007, UFPB 9135//MJ: UFPB 9182, UFPB 9287//SA: UFPB 6880//BA: UFPB 6882//LI: UFRN 5373, UFRN 5389, UFPB 11027, UFPB 11039, UFPB 11166//ME: UFPB 11940//SC:UFPB 12900, UFPB 12954, UFPB 13072, UFPB 9278 |
Schizodon dissimilis | ME, SC | ME: UFPB 9180, UFPB 11708//SC: UFPB 9274 |
Serrapinnus heterodon | AC, AJ, BA, BJ, CO, CU, LI, ME, MJ, SA | AC: UFRN 5331, UFPB 7057, UFPB 7597, UFPB 7612, UFPB 12110//AJ: UFRN 330, UFRN 336, UFRN 366, UFRN 374, UFRN 395, UFRN 1264, UFRN 1275, UFRN 1705, UFRN 2241, UFRN 3249, UFRN 3812, UFRN 3823, UFPB 11720, UFPB 11751//BA: UFRN 3520, UFRN 3528, UFRN 3660, UFPB 7606, UFPB 7629, UFPB 7642, UFPB 11735, UFPB 11746, UFPB 11805, UFPB 11807, UFPB 6942, UFPB 6943//BJ: UFRN 3246, UFRN 5674, UFRN 5676, UFRN 5754//CO: UFRN 5344, UFRN 5372, UFRN 5545, UFRN 5619, UFRN 5622, UFRN 5536, UFRN 5530//CU: UFRN 935//LI: UFRN 1431, UFRN 1435, UFRN 1436, UFRN 2558, UFRN 2566, UFRN 2624, UFRN 2627, UFRN 2629, UFRN 2632, UFRN 2639, UFRN 5473//ME: UFPB 11758, UFPB 6944, UFPB 6945, UFPB 6946// MJ: UFPB 9189//SA: UFRN 1167, UFRN 1338, UFRN 1653, UFRN 1677, UFRN 1878, UFRN 1883, UFRN 4345, UFPB 11749, UFPB 11825, UFPB 6737, UFPB 6939, UFPB 6940, UFRN 1894 |
Serrapinnus piaba | AC, AJ, BA, BJ, CO, CU, LI, ME, MJ, SA | AC: UFRN 5450, UFRN 5451, UFRN 5458, UFPB 7620//AJ: UFRN 1202, UFRN 1272, UFRN 1672, UFRN 3817, UFPB0011723//BA: UFRN 3206, UFRN 3522//BJ: UFRN 3816, UFRN 5675//CO: UFRN 2677, UFRN 5341, UFRN 5342, UFRN 5621, UFRN 5524, UFPB 11951//CU: UFRN 934, UFPB 7075//LI: UFRN 1429, UFRN 2563, UFRN 2571, UFRN 2596, UFRN 2604, UFRN 2623, UFRN 2625, UFRN 2631, UFRN 2655, UFRN 2526, UFRN 2645//ME: UFPB 6949, UFPB 6950, UFPB 6951, UFPB 6952, UFPB 11801, UFPB 11938, UFPB 11945//MJ: UFRN 3248, UFRN 3818// SA: UFRN 1247, UFRN 1285, UFRN 1877, UFRN 2920, UFRN 2922, UFPB0011623, UFPB 6738, UFPB 6947 |
Serrasalmus brandtii | AJ, ME, MJ, SA | AJ: UFRN 1524, UFRN 2240, UFRN 4472, UFRN 5075//ME: UFRN 4895, UFPB 9178//MJ: UFRN 4464//SA: UFRN 5075 |
Serrasalmus rhombeus | AJ, BA, BJ, ME, SA, SC | AJ: UFRN 328, UFRN 343, UFRN 1257, UFRN 1274, UFRN 1480, UFRN 1498, UFRN 1857, UFRN 4470//BA: UFRN 3221//BJ: UFRN 2908, UFRN 3245, UFRN 4334//ME: UFRN 4590, UFRN 4860//SA: UFRN 380, UFRN 398, UFRN 1501, UFRN 1502, UFRN 1858//SC: UFPB 9269 |
Steindachnerina notonota | AC, AJ, BA, CO, LI, ME, SA, SC | AC: UFRN 902, UFRN 5315, UFRN 5317, UFRN 5327, UFPB 7051, UFPB 7609, UFPB0012092, UFPB0012111//AJ: UFRN 357, UFRN 0363, UFRN 401, UFPB 11784, UFPB 11808//BA: UFRN 3219, UFRN 3420, UFRN 3424, UFPB 6956, UFPB 7625, UFPB 7640, UFPB 11755, UFPB0011771//CO: UFRN 1593, UFRN 5335, UFRN 5433, UFRN 5523, UFPB 11957//LI: UFRN 1443, UFRN 1444, UFRN 2570, UFRN 2587, UFRN 2642, UFRN 2660, UFRN 5387//ME: UFRN 2869, UFRN 3748//SA: UFRN 1884, UFRN 3225, UFPB 6742, UFPB 6953, UFPB 6954, UFPB 11761, UFPB 12008//SC: UFRN 5083 |
Sternopygus macrurus | SC | UFPB 9273 |
Synbranchus marmoratus | AC, BA, CO, LI, ME, SA, SC | AC: UFRN 3748, UFRN 2869//BA: UFRN 3204//CO: UFRN 1184, UFRN 2513, UFPB 9257//LI: UFRN 3534//ME: UFRN 2674, UFRN 4889, UFRN 5170, UFPB 6938//SA: UFRN 1471, UFRN 2917//SC: UFPB 9280 |
Tetragonopterus argenteus | AJ, BJ | UFRN 3809, UFRN 4471//BJ: ANSP 84416, ANSP 84412 |
Trachelyopterus cratensis | AJ | UFRN 356 |
Trachelyopterus galeatus | AC, AJ, BA, CO, LI, ME, SA, SC | AC: UFRN 909, UFPB 7046, UFPB0012126/AJ: UFRN 592, UFRN 1798, UFRN 4469//BA: UFPB 11718//CO: UFRN 5366, UFPB 11958//UFRN 3531//ME: UFPB 9181, UFPB 11946//SA: UFRN 1887, UFRN 3201, UFPB 6730// SC: UFPB 9286 |
Triportheus signatus | AC, AJ, BA, CU, ME, MJ, SA, SC | AC: UFRN 906, UFRN 907, UFPB 7600, UFPB0012095//AJ: UFRN 591, UFRN 1291, UFRN 1494, UFRN 3207, UFRN 4473, UFRN 5067, UFPB 9140// BA: UFRN 3518, UFPB 6960, UFPB 6961, UFPB 11820, UFPB 11828//CU: UFRN 4420, UFPB 7079// ME: UFPB 6962, UFPB 9175, UFPB 11793//MJ: UFPB 9148, UFPB 9187//SA: UFRN 5067, UFPB 6958, UFPB 6959, UFPB 9140, UFPB 11816//SC: UFRN 5080, UFPB 9285 |
A total of 25 species (29%) had localized records, occurring in only one hydrographic basin. Another 42 species (49%) had a wide distribution, occurring in at least three hydrographic basins (Tab. 1). Three species (Erythrinus erythrinus (Bloch & Schneider, 1801), Curimatella lepidura (Eigenmann & Eigenmann, 1889), Gymnotus carapo Linnaeus, 1758) lacked information about their hydrographic basins in the ichthyological collection records, only noting their occurrence in the Ceará State. Six species (7%) had some taxonomic issues: Hemiodus parnaguae Eigenmann & Henn, 1916, Parotocinclus jumbo Britski & Garavello, 2002, Pimelodella witmeri Fowler, 1941, P. wolfi (Fowler, 1941), Pseudancistrus papariae Fowler, 1941 and Trachelyopterus cratensis (Miranda Ribeiro, 1937).
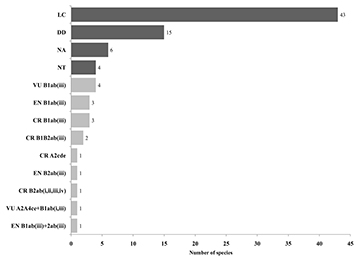
Of the 85 species assessed, 43 were classified as Least Concern (LC), four species as Near Threatened (NT), 17 species were considered threatened under one of the categories (Vulnerable – VU, Endangered – EN, or Critically Endangered – CR), and 15 species having insufficient data to be assessed (Data Deficient – DD). Among the 17 species classified as threatened, four species were categorized as VU B1ab(iii); three species as EN B1ab(iii), three species as CR B1ab(iii), two species as CR B1B2ab(iii), one species as CR A2cde, one species as EN B2ab(iii), one species as CR B2ab(i, ii, iii, iv), one species as VU A2A4ce+B1ab(i,iii), and one species as ENB1ab(iii)+2ab(iii) (Fig. 2; Tab. 2; S1).
FIGURE 2| Number of freshwater fish species in the Ceará State and categories based on IUCN criteria. Legend: LC – Least Concern; DD – Data Deficient; NT – Near Threatened; NE – Not Evaluated; VU – Vulnerable; EN – Endangered; CR – Critically Endangered. The meanings of the codes are detailed in S1.Number of freshwater fish species in the Ceará State and categories based on IUCN criteria. Legend: LC – Least Concern; DD – Data Deficient; NT – Near Threatened; NE – Not Evaluated; VU – Vulnerable; EN – Endangered; CR – Critically Endangered. The meanings of the codes are detailed in S1.
TABLE 2 | Conservation status of freshwater fish species in Ceará State. Taxonomic sequence according to Fricke et al. (2024). OCC = Occurrence Coordinates; AOO = Area of Occupancy; EOO = Extent of Occurrence; LC = Least Concern; NT = Near Threatened; CR = Critically Endangered; EN = Endangered; VU = Vulnerable; DD = Data deficient; NE = Not Evaluated. *Species exclusive to Ceará.
Taxa | OCC | AOO (km2) | EOO (km2) | Status | Endemic to Caatinga |
CLUPEIFORMES | |||||
Engraulidae | |||||
Pterengraulis atherinoides (Linnaeus, 1766) | 2 | Undefined AOO | Undefined EOO | DD |
|
CHARACIFORMES | |||||
Crenuchidae | |||||
Characidium bimaculatum Fowler, 1941 | 28 | 20064.32 | 106096.93 | LC | X |
Erythrinidae | |||||
Erythrinus erythrinus (Bloch & Schneider, 1801) | Undefined coordinates | Undefined AOO | Undefined EOO | DD |
|
Hoplerythrinus unitaeniatus (Spix & Agassiz, 1829) | 4 | 2277.94 | 33719.61 | LC |
|
Hoplias malabaricus (Bloch, 1794) | 97 | 137930.12 | 35752.89 | LC |
|
Parodontidae | |||||
Apareiodon davisi (Fowler, 1941) | 1 | Undefined AOO | Undefined EOO | CR B1ab (iii) | X |
Serrasalmidae | |||||
Metynnis lippincottianus (Cope, 1870) | 1 | Undefined AOO | Undefined EOO | DD |
|
Pygocentrus nattereri Kner, 1858 | 3 | 2738.68 | 17007.44 | LC |
|
Serrasalmus brandtii Lütken, 1875 | 7 | 7304.86 | 41737.26 | LC |
|
Serrasalmus rhombeus (Linnaeus, 1766) | 20 | 8288.72 | 79510.42 | LC |
|
Hemiodontidae | |||||
Hemiodus parnaguae Eigenmann & Henn, 1916 | 3 | 1094.81 | 1323.32 | NT | X |
Anostomidae | |||||
Leporinus piau Fowler, 1941 | 33 | 20961.65 | 131713.42 | LC | X |
Leporinus taeniatus Lütken, 1875 | 21 | 15495.62 | 69971.70 | NT | X |
Schizodon dissimilis (Garman, 1890) | 3 | 2280.69 | 14490.76 | VU B1ab(iii) | X |
Curimatidae | |||||
Curimatella lepidura (Eigenmann & Eigenmann, 1889) | Undefined coordinates | Undefined AOO | Undefined EOO | DD | X |
Psectrogaster rhomboides Eigenmann & Eigenmann, 1889 | 10 | 2849.28 | 41251.15 | LC |
|
Psectrogaster saguiru (Fowler, 1941) | 3 | 192.49 | 18170.00 | DD | X |
Steindachnerina notonota (Miranda Ribeiro, 1937) | 47 | 26061.79 | 134972.28 | LC | X |
Prochilodontidae | |||||
Prochilodus brevis Steindachner, 1875 | 34 | 19959.44 | 108993.99 | LC | X |
Prochilodus lacustris Steindachner, 1907 | 1 | Undefined AOO | Undefined EOO | NT | X |
Lebiasinidae | |||||
Nannostomus beckfordi Günther, 1872 | 11 | 2933.09 | 6746.11 | DD |
|
Triportheidae | |||||
Triportheus signatus (Garman, 1890) | 30 | 22501.30 | 92572.38 | LC | X |
Bryconidae | |||||
Salminus hilarii (Valenciennes,1849) | Undefined coordinates | Undefined AOO | Undefined EOO | CR A2cde |
|
Stevardiidae | |||||
Knodus victoria (Steindachner, 1907) | 1 | Undefined AOO | Undefined EOO | LC |
|
Characidae | |||||
‘Cheirodon’ jaguaribensis Fowler, 1941 | 17 | 12868.25 | 85863.05 | LC | X |
Compsura heterura Eigenmann, 1915 | 22 | 11376.08 | 69523.58 | LC | X |
Phenacogaster calverti (Fowler, 1941) | 24 | 10360.61 | 61705.05 | LC | X |
Serrapinnus heterodon (Eigenmann, 1915) | 72 | 30699.22 | 122551.64 | LC |
|
Serrapinnus piaba (Lütken, 1875) | 49 | 20636.71 | 112661.82 | LC |
|
Tetragonopterus argenteus Cuvier, 1816 | 4 | 1747.21 | 13579.53 | DD |
|
Acestrorhamphidae | |||||
Astyanax bimaculatus (Linnaeus, 1758) | 159 | 47608.23 | 144343.27 | LC |
|
Ctenobrycon kennedyi (Eigenmann, 1903) | 5 | 4329.83 | 30565.71 | LC |
|
Ctenobrycon spilurus (Valenciennes, 1850) | 2 | Undefined AOO | Undefined EOO | DD |
|
Hemigrammus guyanensis Géry, 1959 | 6 | 2933.09 | 6746.11 | VU B1ab (iii) |
|
Hemigrammus marginatus Ellis, 1911 | 11 | 8127.05 | 57526.24 | LC |
|
Hemigrammus rodwayi Durbin, 1909 | 29 | 15413.27 | 110787.91 | LC |
|
Hyphessobrycon piabinhas Fowler, 1941 | 18 | 10308.63 | 81627.31 | NE | X |
Megalamphodus bentosi (Durbin, 1908) | 2 | Undefined AOO | Undefined EOO | DD |
|
Moenkhausia costae (Steindachner, 1907) | 8 | 5804.99 | 30581.85 | LC | X |
Moenkhausia intermedia Eigenmann, 1908 | 10 | 9662.85 | 36578.47 | LC |
|
Psalidodon fasciatus (Cuvier, 1819) | 27 | 17516.64 | 112147.80 | LC |
|
GYMNOTIFORMES | |||||
Gymnotidae |
|
|
|
|
|
Gymnotus carapo Linnaeus, 1758 | Undefined coordinates | Undefined AOO | Undefined EOO | DD |
|
Sternopygidae |
|
|
|
|
|
Sternopygus macrurus (Bloch & Schneider, 1801) | 1 | Undefined AOO | Undefined EOO | DD |
|
SILURIFORMES | |||||
Callichthyidae | |||||
Aspidoras carvalhoi (Nijssen & Isbrücker, 1976) (*) |
| Undefined AOO | Undefined EOO | CR B1ab (iii) | X |
Aspidoras raimundi (Steindachner, 1907) | 22 | 7316.27 | 44447.36 | LC | X |
Aspidoras rochai Ihering, 1907* | 1 | Undefined AOO | Undefined EOO | CRB1ab(iii) | X |
Callichthys callichthys (Linnaeus, 1758) | 4 | 3549.99 | 7791.03 | LC |
|
Hoplosternum littorale (Hancock, 1828) | 4 | 4280.86 | 32618.09 | LC |
|
Megalechis thoracata (Valenciennes, 1840) | 1 | Undefined AOO | Undefined EOO | LC |
|
Doradidae | |||||
Platydoras brachylecis Piorski, Garavello, Arce & Sabaj Pérez, 2008 | 1 | Undefined AOO | Undefined EOO | DD | X |
Loricariidae | |||||
Ancistrus damasceni (Steindachner, 1907) | 1 | Undefined AOO | Undefined EOO | DD | X |
Aphanotorulus gomesi (Fowler, 1942) | Undefined coordinates | Undefined AOO | Undefined EOO | NE | X |
Hypostomus johnii (Steindachner, 1877) | 3 | 1094.81 | 1323.32 | EN B1ab (iii) |
|
Hypostomus pusarum (Starks, 1913) | 73 | 34326.73 | 128520.23 | LC | X |
Hypostomus sertanejo Zawadzki, Ramos & Sabaj, 2017* | 10 | 5515.26 | 25324.83 | LC | X |
Loricariichthys derbyi Fowler, 1915 | 10 | 6034.92 | 46181.21 | LC | X |
Parotocinclus cearensis Garavello, 1977 | 42 | 14045.31 | 94347.54 | LC | X |
Parotocinclus haroldoi Garavello, 1988 | 5 | 1112.72 | 2869.13 | VU B1ab (iii) | X |
Parotocinclus jumbo Britski & Garavello, 2002 | 3 | 5058.39 | 24743.89 | LC | X |
Parotocinclus spilurus (Fowler, 1941)* | 13 | 5681.38 | 9981.14 | EN B2ab(iii) | X |
Pseudancistrus genisetiger Fowler, 1941 | 8 | 4712.03 | 25156.84 | NT | X |
Pseudancistrus papariae Fowler, 1941 | 1 | Undefined AOO | Undefined EOO | NE |
|
Pterygoplichthys parnaibae (Weber, 1991) | 1 | Undefined AOO | Undefined EOO | DD | X |
Auchenipteridae | |||||
Trachelyopterus cratensis (Miranda Ribeiro, 1937) | 1 | Undefined AOO | Undefined EOO | NE | X |
Trachelyopterus galeatus (Linnaeus, 1766) | 18 | 15682.47 | 123425.50 | LC |
|
Heptapteridae | |||||
Pimelodella dorseyi Fowler, 1941 | 7 | 3331.25 | 4402.34 | EN B1ab(iii) | X |
Pimelodella parnahybae Fowler, 1941 | 2 | Undefined AOO | Undefined EOO | LC | X |
Pimelodella witmeri Fowler, 1941 | 3 | 5629.66 | 19319.51 | NE |
|
Pimelodella wolfi (Fowler, 1941) | 2 | Undefined AOO | Undefined EOO | NE |
|
Rhamdia quelen (Quoy & Gaimard, 1824) | 6 | 3754.51 | 8710.23 | LC |
|
GOBIIFORMES | |||||
Gobiidae | |||||
Awaous tajasica (Lichtenstein, 1822) | 4 | 4685.21 | 18108.16 | LC |
|
SYNBRANCHIFORMES | |||||
Synbranchidae | |||||
Synbranchus marmoratus Bloch, 1795 | 14 | 10182.81 | 107661.25 | LC |
|
CICHLIFORMES | |||||
Cichlidae | |||||
Cichlasoma orientale Kullander, 1983 | 155 | 43453.77 | 139561.89 | LC | X |
Cichlasoma sanctifranciscense Kullander, 1983 | 6 | 3665.52 | 19417.35 | LC | X |
Saxatilia brasiliensis (Bloch, 1792) | 30 | 16209.75 | 97258.99 | LC |
|
Geophagus brasiliensis (Quoy & Gaimard, 1824) | 5 | 3046.80 | 8661.91 | LC |
|
CYPRINODONTIFORMES | |||||
Rivulidae |
|
|
|
|
|
Anablepsoides cearensis (Costa & Vono, 2009)* | 8 | 2933.09 | 6746.11 | CR B2ab(i, ii, iii, iv) | X |
Cynolebias microphthalmus Costa & Brasil, 1995 | 1 | Undefined AOO | Undefined EOO |
VU
A2A4ce+ | X |
Hypsolebias antenori (Tulipano, 1973)* | 15 | 4455.01 | 6574.10 | VU B1ab (iii) | X |
Hypsolebias gongobira (Abrantes, Ramos, Bento & Lima, 2023)* | 1 | Undefined AOO | Undefined EOO | CR B1B2ab(iii) | X |
Hypsolebias longignatus (Costa, 2008)* | 4 | 898.54 |
1608.60 | CR B1B2ab(iii) | X |
Hypsolebias martinsi Britzke, Nielsen & Oliveira, 2016 | 4 |
3027.97 |
3655.60 | EN B1ab(iii) + 2ab(iii) | X |
Kryptolebias hermaphroditus Costa, 2011 | 2 | Undefined AOO | Undefined EOO | DD |
|
Poeciliidae | |||||
Poecilia sarrafae Bragança & Costa, 2011 | 11 | 4580.35 | 7331.94 | EN B1ab(iii) |
|
Poecilia vivipara Bloch & Schneider, 1801 | 92 | 37835.05 | 142687.29 | LC |
|
Additionally, six species were categorized as Not Evaluated (NE) because they were classified as inquirenda based on Lima et al. (2017) and/or Berbel-Filho et al. (2028): Trachelyopterus cratensis, Pimelodella witmeri, Pimelodella wolfi, Aphanotorulus gomesi (Fowler, 1942), Pseudancistrus papariae .
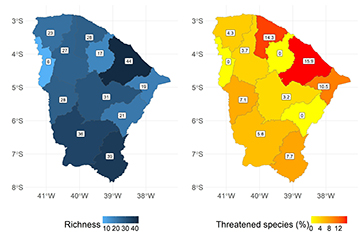
We observed a considerable variability in the number of species among basins (mean 25.1 ± 10.3). The Metropolitana and Salgado basins exhibited the highest species richness (44 and 39 species, respectively). The Serra de Ibiapaba and Curu basins showed the lowest richness (6 and 17 species, respectively) (Fig. 3A). The basins with the highest percentage of threatened fish species were Metropolitana (15.9%), Litoral (14.3%) and lower Jaguaribe (10.5%) (Fig. 3B).
FIGURE 3| Freshwater fish by hydrographic basin in the Ceará State. A. Species richness; B. Percentage of threatened species.
Discussion
Previous records available from federal agencies regarding the freshwater fish fauna occurring in the Ceará State primarily indicated the use of freshwater fish for human consumption, eradication of species considered ‘pests’ (e.g., piranhas), and stocking activities with both native and non-native species (Ihering, 1933; Menezes, 1956; DNOCS, 1963; Peixoto, Gurgel, 1978, 1979). These documents do not properly include cataloged lists of native fish species in collections nor report actions for the conservation of native freshwater fish. From an academic perspective, the earliest records of native ichthyofauna in Ceará’s freshwater environments (Ihering, 1907; Fowler, 1915) indicated the faunistic influence of the Atlantic Forest and Amazon biomes on the current species composition and distribution. More recently, taxonomic and phylogeographic studies discussed the influences of these biomes and geomorphological processes that have shaped the current freshwater fish diversity (Rosa et al., 2003; Rosa, Groth, 2004; Berbel-Filho et al., 2015; Lima et al., 2017; Rodrigues-Filho et al., 2018a).
Out of the 85 fish species in Ceará State, four are listed in the ICMBio Red List of Threatened Fauna (MMA, 2022) and four are listed in the IUCN Red List. Anablepsoides cearensis (Costa & Vono, 2009), Hypsolebias longignatus (Costa, 2008), and Parotocinclus spilurus (Fowler, 1941) are listed in both lists. Apareiodon davisi (Fowler, 1941) and Hypsolebias martinsi Britzke, Nielsen & Oliveira, 2016 are exclusively listed in the ICMBio and IUCN lists, respectively. Six species were categorized as DD (Data Deficient) by ICMBio and seven by the IUCN Red List. Two species were not assessed by ICMBio (Hemigrammus rodwayi Durbin, 1909 and Hypsolebias gongobira (Abrantes, Ramos, Bento & Lima, 2023), and 31 species were not evaluated by the IUCN Red List. These results highlight a significant number of fish species not evaluated by the IUCN Red List (36%), underscoring the need for updating both national and global official lists.
The high endemism recorded (51%) indicated that almost half of the continental fish species have restricted geographic distribution to the Caatinga (Lima et al., 2017), with eight exclusive to Ceará State: A. cearensis, Hypsolebias antenori (Tulipano, 1973), H. longignatus, H. gongobira, Parotocinclus spilurus, Aspidoras carvalhoi (Nijssen & Isbrücker, 1976), Aspidoras rochai Ihering, 1907, and Hypostomus sertanejo Zawadzki, Ramos & Sabaj, 2017. On the other hand, about one-third of the native fish species in the state are widely distributed, with greater survival potential in the face of natural and anthropogenic disturbances occurring in Ceará’s hydrographic basins. Conversely, the three species without records in the state’s basins highlight the lack of information related to Area of Occupancy (AOO), Extent of Occurrence (EOO), and their actual distributions, indicating the need for further studies. It is noteworthy that some widely distributed species may represent species complexes, with some potentially endemic to Ceará’s basins (e.g., Parotocinclus Eigenmann & Eigenmann, 1889), and others purportedly endemic (e.g., Hypostomus, Pseudancistrus Bleeker, 1862, and Pimelodella Eigenmann & Eigenmann, 1888) (Lima et al., 2017; Berbel-Filho et al., 2018). These taxonomically undefined species underscore the need for further taxonomic and molecular studies to resolve uncertainties, as in the recent study by Lustosa-Costa et al. (2024), which clarifies the species complex of Hypostomus pusarum (Starks, 1913) using integrative taxonomy. Indeed, Lima et al. (2017), Berbel-Filho et al. (2018), and Terra et al. (2021) highlight the need for more studies on Caatinga ichthyofauna to understand biological aspects.
Among the 17 threatened continental fish species in the State of Ceará, the order Cyprinodontiformes stands out with seven species, followed by Siluriformes with six species and Characiformes with four species. Rivulidae and Loricariidae include six and three species, respectively, classified under some threat criteria. These species are predominantly small-sized and exhibit restricted geographical distributions. These two characteristics are correlated (Ripple et al., 2017) and have been considered predictors of extinction risk for freshwater fish species in global assessments (Olden et al., 2007; Kalinkat et al., 2017).
The majority of Rivulidae species (86%) are threatened in Ceará. In Brazil, this family is also among the freshwater fish groups at the highest risk of extinction, with 125 species classified under some threat level (Castro, Polaz, 2019). In addition to the intrinsic characteristics mentioned above, another critical factor for this group is habitat integrity. Most species within this family exhibit adaptations and seasonal life cycles that enable survival under the extreme environmental conditions of temporary pools (Berois et al., 2016). However, such habitats are often overlooked by conservation initiatives (Zacharias, Zamparas, 2010; Calhoun et al., 2017) and are increasingly threatened by various anthropogenic impacts, including road construction, agriculture, and sand extraction for the construction industry (Abrantes et al., 2020). This finding underscores the urgent need to implement protection policies for this group in the State, ensuring the preservation of permanent wetlands and seasonal pools in floodplain areas where they complete their life cycles (Teixeira et al., 2017; Abrantes et al., 2020).
The Metropolitana, Litoral, and lower Jaguaribe basins exhibit a higher percentage of threatened fish species (seven, four, and two species, respectively). This result possibly reflects the regional occupation pattern, which concentrates on activities such as industry and tourism along the coastal region (Pinheiro et al., 2016). Particularly in the Metropolitan Region, another impact source to be considered is urbanization. This factor can contribute to the decline in freshwater fish biodiversity as it is associated with processes such as loss of natural habitats (Walsh et al., 2005), increased pollutants and organic matter (Parr et al., 2016), and the introduction of non-native species (Ortega et al., 2021). The disparity in species richness among these basins, with higher values in the Metropolitana, likely indicates greater sampling effort in areas closer to research institutions. This bias is common in biodiversity data (Costa et al., 2010; Cardoso et al., 2024) and underscores the need to expand sampling efforts to enhance knowledge of fish distribution in the state.
Other state red lists have reported varying numbers of threatened continental fish species, with the states of Pará, Espírito Santo, and Paraná having the lowest numbers (nine, eleven, and fifteen species, respectively), and São Paulo having the highest number (66 species) (COEMA, 2007; Paraná, 2018; SMA, 2018; IEMA, 2022). Rivulidae is prominently represented with a high number of threatened species, being the most represented family in Bahia (14 species), Espírito Santo (three), and Rio Grande do Sul (27 species) (SEMA, 2014; SEMA, 2017; Silva et al., 2020a; IEMA, 2022), and the second most represented family in Minas Gerais (10), Rio de Janeiro (10), and Santa Catarina (seven species) (SEMA, 1998; COPAM, 2010; FATMA, 2011). Loricariidae is represented in the number of threatened species in São Paulo (14) and Minas Gerais (seven species) (COPAM, 2010; SMA 2018). Families not yet recorded in Ceará State, such as Pimelodidae and Trichomycteridae, also comprise relatively high numbers in other state lists (ranging from one to eight species, and one to twelve species, respectively), and potential occurrences in Ceará cannot be ruled out with increased sampling efforts.
The total number of fish species categorized as Least Concern (LC = 43), Data Deficient (DD = 15), Near Threatened (NT = 4), and Not Evaluated (NE = 6) accounts for 80% of the ichthyofauna documented in this study. The fish species classified as LC (51% of the total) are widely distributed across the hydrographic basins of Ceará, indicating no imminent threat of extinction. However, considering the historical introduction of non-native species, the construction of large dams impacting underlying ecosystems, and efforts to eradicate species deemed harmful such as piranhas, as documented by historical records from DNOCS, this value underscores a resilient and robust ichthyofauna. On the other hand, the aforementioned disturbances may have led to the extinction of species of fish that were never formally recorded.
The 15 fish species categorized as Data Deficient (DD) were justified in this category due to limited available information on population aspects, distribution, and taxonomic issues. Part of the species in this category (Erythrinus erythrinus (Bloch & Schneider, 1801), Curimatella lepidura (Eigenmann & Eigenmann, 1889), Gymnotus carapo Linnaeus, 1758, Aphanotorulus gomesi (Fowler, 1942)) do not have occurrence coordinates in the basins of Ceará, although their presence is known from other bibliographic sources (Rosa et al., 2003; Rosa, Groth, 2004; Silva et al., 2005, 2020b; Rodrigues-Filho et al., 2016; Berbel-Filho et al., 2018; Sánchez-Botero et al., 2021). Five species also classified in this category have only one occurrence coordinate (Sternopygus macrurus (Bloch & Schneider, 1801), Platydoras brachylecis Piorski, Garavello, Arce H. & Sabaj Pérez, 2008, Ancistrus damasceni (Steindachner, 1907), Pterygoplichthys parnaibae (Weber, 1991) and Metynnis lippincottianus (Cope, 1870) or only two (Ctenobrycon spilurus (Valenciennes, 1850), Megalamphodus bentosi (Durbin, 1908) and Kryptolebias hermaphroditus Costa, 2011). Therefore, greater collection efforts for these species are necessary to assess their conservation status and potential extinction threats. In addition to this, six other species have dubious taxonomy Hyphessobrycon piabinhas Fowler, 1941, Trachelyopterus cratensis, Pimelodella witmeri, P. wolfi, Aphanotorulus gomesi, and Pseudancistrus papariae.
Large-scale developments underway in Ceará State are likely to cause disturbances that will affect freshwater fish fauna. The Complexo Industrial e Portuário do Pecém (CIPP), through a public utility decree defining its coverage area (CEARÁ, 2007), plans development projects in areas containing stream springs as well as the infilling of lakes and lagoons, impacting local biodiversity (Meireles et al., 2012). The species Hemigrammus guyanensis Géry, 1959 and Anablepsoides cearensis, which occur in the CIPP region, are classified as threatened with extinction according to the assessment conducted in this study, with the latter having a restricted distribution (AOO <10 km2). Furthermore, the São Francisco River Transposition Project expected effects include the introduction of non-native species, potentially altering biotic interactions (competition, predation, and parasitism) and modifying natural habitats, thereby impacting native species (Silva et al., 2020b; Falkenberg et al., 2024). These disturbances are expected to particularly impact the threatened ichthyofauna found in the Jaguaribe River sub-basins, including Apareiodon davisi, Cynolebias microphthalmus Costa & Brasil, 1995, Hypsolebias antenori, Parotocinclus spilurus, and Pimelodella dorseyi Fowler, 1941. In addition to the aforementioned ongoing projects, other initiatives of concern to be considered in the medium- and long-term conservation strategies for freshwater fish in the State are the ongoing uranium and phosphate mining projects in the municipality of Santa Quitéria, in the licensing phase (EIA, 2021).
In the State of Ceará, dam construction has historically been adopted as a strategy to ensure water supply for the human population, addressing water scarcity and prolonged droughts typical of the semi-arid region (Gurgel-Lourenço et al., 2023). These structures are also crucial for providing water for livestock and supporting agricultural activities (Gaiser et al., 2003). However, such developments fragment riverine habitats, preventing species from moving between breeding, growth, and feeding areas, leading to population declines or even local extinctions (Barbarossa et al., 2020; Sun et al., 2023). Dams represent one of the main threats to migratory fish species, which rely on the natural flow of rivers to complete their life cycles (Thomé et al., 2005), leading to the reduction or extinction of populations, as observed in the case of Salminus hilarii (Valenciennes,1849) in the Jaguaribe River basin (in Ceará State, it occurs exclusively in this basin), a migratory species that was once among the most important fisheries resources, is now absent and possibly extinct (Menezes, 1953; Lima, 2022). The construction of new dams exacerbates conservation risks for this fauna, particularly for many migratory species, such as those from the genera Prochilodus Agassiz, 1829 and Leporinus Agassiz, 1829, which have traditionally been targeted by artisanal fishing in Ceará State (Batista et al., 2016; Costa et al., 2016). Additionally, Hemiodus parnaguae and Prochilodus lacustris Steindachner, 1907, classified as near-threatened and recorded in the State only within the Sertões de Crateús basin, could be negatively impacted by the ongoing construction of the Fronteiras Dam in Crateús-CE. Thus, implementing technologies such as fish ladders and waterways should increasingly be promoted and adopted as a mitigation strategy (Kemp, 2015), still with risks regarding how effective they can be for all fish species.
In conclusion, this study underscores the urgent need to implement impact mitigation programs for freshwater fish in Ceará. The species distribution information provided in this study should be considered and applied to assist in defining and prioritizing protected areas and management measures to ensure ichthyofauna biodiversity maintenance and protection of species facing some threat level. These areas should encompass hydrographic basins that shelter threatened fish species and exhibit higher species richness. This study provides a survey of information on the conservation status of the continental ichthyofauna of the Ceará State. However, the extinction risk assessment proposals presented here must undergo procedures established by the State to meet legal requirements and officially become the Red List of the State of Ceará.
Acknowledgments
The authors would like to thank the Universidade Federal do Ceará (UFC), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support through the MCTI/CNPq Program (Grant 28/2018, 423628/2018–6 and Grant 63/2022, 409354/2022–8) for the field and scholarship support to RCGL and LMP. We thank the Programa de Pós-Graduação em Sistemática, Uso e Conservação da Biodiversidade – PPGSIS/UFC for support to MCFG during magister research. We are grateful for the support of Instituto Nacional de Ciência e Tecnologia de Materiais na Interface Continente-Oceano (INCT-TMCOcean), based at UFC. SMQL thanks the CNPq for the productivity Grant (312066/2021–0). TPAR thanks the CNPq for the PDS grant (102460/2022–1). PC acknowledges the visiting researcher grant from FUNCAP (PVS–0215–00123.2.00/23) and a Save Our Seas Foundation Conservation Fellowship (SOSF 588).
References
Abrantes YG, Medeiros LS, Bennemann ABA, Bento DM, Teixeira FK, Rezende CF et al. Geographic distribution and conservation of seasonal killifishes (Cyprinodontiformes, Rivulidae) from the Mid-Northeastern Caatinga ecoregion, northeastern Brazil. Neotrop Biol Conserv. 2020; 15(3):301–15. https://doi.org/10.3897/neotropical.15.e51738
Abrantes YG, Ramos TPA, Bento DM, Lima SMQ. Molecular delimitation of the seasonal killifishes of the Hypsolebias antenori species group (Cyprinodontiformes, Rivulidae), with description of two new species from the Caatinga coastal basins, northeastern Brazil. Zootaxa. 2023; 5389(5):545–62. https://doi.org/10.11646/zootaxa.5389.5.2
Barbarossa V, Schmitt RJP, Huijbregts MAJ, Zarfl C, King H, Schipper AM. Impacts of current and future large dams on the geographic range connectivity of freshwater fish worldwide. PNAS. 2020; 117(7):3648–55. https://doi.org/10.1073/pnas.1912776117
Batista LPP, Sánchez-Botero JI, Paula EO, Silva EV. Etnotaxonomia e tabus alimentares dos pescadores artesanais nos açudes Araras e Edson Queiroz, bacia do Rio Acaraú, Ceará, Brasil. Entorno Geográfico. 2016; 12:34–49. https://doi.org/10.25100/eg.v0i12.3543
Berbel-Filho WM, Martinez PA, Ramos TPA, Torres RA, Lima SMQ. Inter- and intra-basin phenotypic variation in two riverine cichlids from northeastern Brazil: potential eco-evolutionary damages of São Francisco interbasin water transfer. Hydrobiologia. 2015; 766:43–56. https://doi.org/10.1007/s10750-015-2440-9
Berbel-Filho WM, Ramos TPA, Jacobina UP, Maia DJG, Torres RA, Lima SMQ. Updated checklist and DNA barcode-based species delimitations reveal taxonomic uncertainties among freshwater fishes from the mid-north-eastern Caatinga ecoregion, north-eastern Brazil. J Fish Biol. 2018; 93(2):311–23. https://doi.org/10.1111/jfb.13758
Berois N, García G, Sá RO. Annual fishes: life history strategy, diversity, and evolution. Boca Raton: CRC Press; 2016.
Bezerra LAV, Angelini R, Vitule JRV, Coll M, Sánchez-Botero JI. Food web changes associated with drought and invasive species in a tropical semiarid reservoir. Hydrobiologia. 2017; 817:475–89. https://doi.org/10.1007/s10750-017-3432-8
Bezerra LAV, Gurgel-Lourenço RC, Sánchez-Botero JI. Vertical segregation of Serrapinnus piaba and Serrapinnus heterodon (Characiformes: Characidae) in a river stretch in Northeastern Brazil.Sodebras. 2013; 8(86):81–85.
Calhoun AJK, Mushet DM, Bell KP, Boix D, Fitzsimons JA, Isselin-Nondedeu F. Temporary wetlands: challenges and solutions to conserving a ‘disappearing’ ecosystem. Biol Conserv. 2017; 21:3–11. https://doi.org/10.1016/j.biocon.2016.11.024
Cardoso MNM, Azevedo F, Dias A, Almeida ACS, Senna AR, Marques AC et al. Causes and effects of sampling bias on marine Western Atlantic biodiversity knowledge. Divers Distrib. 2024; 30(6):e13839. https://doi.org/10.1111/ddi.13839
Castro RMC, Polaz CNM. Small-sized fish: the largest and most threatened portion of the megadiverse neotropical freshwater fish fauna. Biota Neotrop. 2019; 20(1):e20180683. https://doi.org/10.1590/1676-0611-BN-2018-0683
CEARÁ. Decreto n° 28.883 de 18 de setembro de 2007. Diário Oficial do Estado: série 2, Fortaleza, CE, ano 10, n. 170, p. 1–3; 2007.
Centro de Referência e Informação Ambiental (CRIA). SpeciesLink network; 2024. Available from: https://specieslink.net/
Chaves FDN, Sánchez-Botero JI, Garcez DS, Reis VC. Population features of Hoplosternum littorale (Hancock, 1828) (Siluriformes, Callichthyidae) at Santo Anastacio Reservoir, Brazil. Rev MVZ Cordoba. 2013; 18(3):3767–72.
Chiu M-C, Leigh C, Mazor R, Cid N, Resh V. Anthropogenic threats to intermittent rivers and ephemeral streams. In: Datry T, Bonada N, Boulton A, editors. Intermittent rivers and ephemeral streams. San Diego: Academic Press; 2017. p.433–54. https://doi.org/10.1016/B978-0-12-803835-2.00017-6
Conselho Estadual do Meio Ambiente (COEMA). Resolução COEMA 54, de 24 de outubro de 2007. Homologa a lista de espécies da flora e da fauna ameaçadas no Estado do Pará. 2007. [Internet]. Available from: https://www.semas.pa.gov.br/legislacao/files/pdf/375.pdf
Companhia de Gestão dos Recursos Hídricos (COGERH). Atlas dos Recursos Hídricos do Ceará [Internet]. 2002. Available from: http://atlas.cogerh.com.br/
Conselho Estadual de Política Ambiental (COPAM). Deliberação Normativa COPAM 147, de 30 de abril de 2010. Aprova a Lista de Espécies Ameaçadas de Extinção da Fauna do Estado de Minas Gerais. 2010. [Internet]. http://www.siam.mg.gov.br/sla/download.pdf?idNorma=13192
Costa GC, Nogueira C, Machado RB, Colli GR. Sampling bias and the use of ecological niche modeling in conservation planning: a field evaluation in a biodiversity hotspot. Biodivers Conserv. 2010; 19(3):883–99. https://doi.org/10.1007/s10531-009-9746-8
Costa RB, Farias JO, Sales RO. Common curimatã (Prochilodus cearaensis) in backwoodsman life in northeast Brazil. Rev Bras Hig Sanid Anim. 2016; 10(1):158–65. https://doi.org/10.5935/1981-2965.20160045
Departamento Nacional de Obras Contra as Secas (DNOCS). Pesca nos açudes públicos do Ceará e sua comercialização. Fortaleza: Imprensa Universitária do Ceará; 1963.
Estudo de Impacto Ambiental (EIA). Projeto Santa Quitéria. Volume II B. Consórcio Santa Quitéria. Tetra +. São Paulo, SP.2021. [Internet]. Available from: https://consorciosantaquiteria.com.br/eia-rima/
Falkenberg J, Lima V, Yamada F, Ramos T, Lacerda AC. Changes in parasite communities of fishes from an intermittent river in the Brazilian semi-arid, after a major interbasin water transfer. Aquat Ecol. 2024; 58:895–916. https://doi.org/10.1007/s10452-024-10112-7
Faustino ACQ, Terra BF. Ecological drivers of fish metacommunities: environmental and spatial factors surpass predation in structuring metacommunities of intermittent rivers.Ecol Freshw Fish. 2019; 29(1):145–55. https://doi.org/10.1111/eff.12502
Fundação do Meio Ambiente (FATMA). Resolução Consema 02, de 06 de dezembro de 2011. Lista Oficial de Espécies da Fauna Ameaçadas de Extinção no Estado de Santa Catarina. 2011. [Internet]. Available from: https://www.ima.sc.gov.br/index.php/biodiversidade/biodiversidade/fauna
Fowler HR. Cold-blooded vertebrates from Florida, the west INDIES, Costa Rica and eastern Brazil. PNAS. 1915; 67:244–69.
Frederico RG, Reis VCS, Polaz CNM. Conservação de peixes de riacho: planejamento e políticas públicas. Oecol Austr. 2021; 25(2):546–64. https://doi.org/10.4257/oeco.2021.2502.20
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2024. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Gaiser T, Krol M, Frischkorn H, Araújo JC, editors. Global change and regional impacts: water availability and vulnerability of ecosystems and society in the semiarid northeast of Brazil. New York: Springer Berlin Heidelberg; 2003. https://doi.org/10.1007/978-3-642-55659-3
Gonçalves-Silva M, Manna LR, Rodrigues-Filho CAS, Teixeira FK, Rezende CF. Effect of drying dynamics on the functional structure of a fish assemblage from an intermittent river network. Front Environ Sci. 2022; 10:903974. https://doi.org/10.3389/fenvs.2022.903974
Gurgel JJS. Pesca e piscicultura em águas represadas do polígono das secas. Fortaleza: Ministério do Interior – Departamento Nacional de Obras Contra as Secas (MINTER-DNOCS)/Diretoria de Pesca e Piscicultura; 1979.
Gurgel-Lourenço RC, Rodrigues-Filho CAS, Angelini R, Garcez DS, Sánchez-Botero JI. On the relation amongst limnological factors and fish abundance in reservoirs at semiarid region. Acta Limnol Bras. 2015; 27(1):24–38. http://doi.org/10.1590/S2179-975X2414
Gurgel-Lourenço RC, Rodrigues-Filho CAS, Bezerra LAV, Garcez DS, Sánchez-Botero JI. Length-weight relationships for freshwater fish species from humid forest enclaves at the Brazilian semiarid. J Appl Ichthyol. 2017; 33:1254–57. http://doi.org/10.1111/jai.13444
Gurgel-Lourenço, RC, Rodrigues-Filho CAS, Pinto LM, Sánchez-Botero JI. Prolonged drought influences the taxonomic and functional structure of fish assemblages in estuaries along the Brazilian semiarid coast. Hydrobiologia. 2023; 850(20):4443–66. https://doi.org/10.1007/s10750-022-05059-5
Gurgel-Lourenço RC, Sousa WA, Sánchez-Botero JI, Garcez DS. Ichthyofauna of two reservoirs in the middle Acaraú river basin, Ceará, Northeastern, Brazil. Check List. 2013; 9(6):1391–95. https://doi.org/10.15560/9.6.1391
Ihering RV. Comissão técnica de piscicultura do nordeste do Brasil. Ministério da Viação e Obras Públicas. Recife: Escola de Artes Gráficas de Pernambuco; 1933.
Instituto Estadual de Meio Ambiente e Recursos Hídricos (IEMA). Decreto Estadual 5237, de 25 de novembro de 2022. 2022. [Internet]. Available from: https://leisestaduais.com.br/es/decreto-n-5237-2022-espirito-santo-declara-as-especies-de-fauna-ameacadas-de-extincao-no-estado-do-espirito-santo-e-da-outras-providencias
International Union for Conservation of Nature (IUCN). Standards and petitions committee. Guidelines for using the IUCN Red List categories and criteria. Version 16 [Internet]. Gland; 2024. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Kalinkat G, Jähnig SC, Jeschke JM. Exceptional body size-extinction risk relations shed new light on the freshwater biodiversity crisis. PNAS. 2017; 114(48):e10263. https://doi.org/10.1073/pnas.1717087114
Kemp PS. Impoundments, barriers and abstractions: impact on fishes and fisheries, mitigation and future directions. In: Craig JF, editor. Freshwater fisheries ecology. Oxford: John Wiley & Sons, Ltd; 2015. p.717–69. https://doi.org/10.1002/9781118394380.ch52
Leal CG, Lennox GD, Ferraz SFB, Ferreira J, Gardner TA, Thomson JR et al. Integrated terrestrial-freshwater planning doubles conservation of tropical aquatic species. Science. 2021; 370(6512):117–21. https://doi.org/10.1126/science.aba7580
Lima FCT. Revision of the smaller-sized dorados (Salminus), with comments on the monophyly of the genus and its biogeography (Characiformes: Bryconidae). Zootaxa. 2022; 5226(1):1–66. https://doi.org/10.11646/zootaxa.5226.1.1
Lima SMQ, Ramos TPA, Silva MJ, Rosa RS. Diversity, distribution, and conservation of the Caatinga fishes: advances and challenges. In: Silva JMC, Leal IR, Tabarelli M, editors. Caatinga – The largest tropical dry forest region in South America. Cham: Springer; 2017. p.97–131. https://doi.org/10.1007/978-3-319-68339-3_4
Lustosa-Costa SY, Ramos TPA, Zawadzki CH, Jacobina UP, Lima SMQ. Integrative taxonomy clarifies the armoured catfish Hypostomus pusarum (Starks) species complex (Siluriformes: Loricariidae) and reveals a new species in the drainages of Northeastern Brazil. Zool J Linn Soc Lond. 2024; 201(3):zlae059. htps://doi.org/10.1093/zoolinnean/zlae059
Manna LR, Rezende CF, Mazzoni R. Effect of body size on microhabitat preferences in stream-dwelling fishes. J Appl Ichthyol. 2017; 33:193–202. htps://doi.org/10.1111/jai.13320
Manna LR, Villéger S, Rezende CF, Mazzoni R. High intraspecific variability in morphology and diet in tropical stream fish communities. Ecol Freshw Fish. 2018; 28(1):41–52. https://doi.org/10.1111/eff.12425
Mapurunga MER, Pessoa YVB, Maria AH, Oliveira JS, Terra BF. A influência de filtros ambientais locais ictiofauna de riachos intermitentes. RCGS. 2024; 26(2):134–45. https://doi.org/10.35701/rcgs.v26.1027
Meireles AJA, Brissac S, Schettino MP. O povo indígena Anacé e seu território tradicionalmente ocupado. Parecer Técnico N° 01/09 – Ministério Público Federal. Cadernos LEME. 2012; 4(1):115–235.
Menezes RS. A carpa: peixe flagelo que deve e precisa ser combatido. Ministério da Viação e Obras Públicas. Fortaleza: Departamento de Obras Contra as Secas. Serviço de Piscicultura; Publicação 171. Série I.C. 1956.
Menezes RS. Lista dos nomes vulgares de peixes de água doces e salobras da zona seca do nordeste e leste do Brasil. Arq Mus Nac. 1953; 42:343–88.
Ministério do Meio Ambiente (MMA). Portaria MMA Nº 148, de 7 de Junho de 2022. 2022. Ministério do Meio Ambiente. [Internet]. Available from: https://www.icmbio.gov.br/cepsul/images/stories/legislacao/Portaria/2020/P_mma_148_2022_altera_anexos_P_mma_443_444_445_2014_atualiza_especies_ameacadas_extincao.pdf
Morais JO, Pinheiro LS. The effect of semi-aridity and damming on sedimentary dynamics in estuaries – Northeastern region of Brazil. J Coastal Res. 2011; 64:1540–44.
Moro MF, Macêdo MB, Moura-Fé MM, Costa RC. Vegetação, unidades fitoecológicas e diversidade paisagística do estado do Ceará. Rodriguésia. 2015; 66(3):717–43. http://doi.org/10.1590/2175-7860201566305
Olden JD, Hogan ZS, Zanden MJV. Small fish, big fish, red fish, blue fish: size-biased extinction risk of the world’s freshwater and marine fishes. Global Ecol Biogeogr. 2007; 16(6):694–701. https://doi.org/10.1111/j.1466-8238.2007.00337.x
Oliveira JS, Hughes RM, Terra BF. Fish and macroinvertebrates respond differently to seasonal drying in tropical non-perennial streams. Austral Ecol. 2024; 49(7):e13558. https://doi.org/10.1111/aec.13558
Oliveira JS, Santos AMP, Santos VC, Xavier ALM, Conceição ALF, Terra BF. Biodiversidade de peixes em riachos intermitentes da região semiárida brasileira (Bacia do rio Acaraú, Brasil). Essentia. 2023; 24(1):1–10.
Ortega JCG, Bacani I, Dorado-Rodrigues TF, Strüssmann C, Fernandes IM, Morales J et al. Effects of urbanization and environmental heterogeneity on fish assemblages in small streams. Neotrop Ichthyol. 2021; 19(3):e210050. https://doi.org/10.1590/1982-0224-2021-0050
Paraná. Livro Vermelho da Fauna Ameaçada no Estado do Paraná. Conexão Ambiental [Internet]. 2018. Available from: https://www.conexaoambiental.pr.gov.br/Pagina/Livro-Vermelho-da-Fauna-Ameacada-no-Estado-do-Parana-0
Parr TB, Smucker NJ, Bentsen CN, Neale MW. Potential roles of past, present, and future urbanization characteristics in producing varied stream responses. Freshw Sci. 2016; 35(1):436–43. https://doi.org/10.1086/685030
Peixoto JT, Gurgel JJS. Produção e distribuição de alevinos pelas estações de piscicultura do DNOCS em 1976. Boletim Técnico do DNOCS. 1978; 36(1):5–15.
Pinheiro LS, Morais JO, Maia LP. The beaches of Ceará. In: Short AD, Klein AHF, editors. Brazilian beach systems. Cham: Springer; 2016. p.175–99. https://doi.org/10.1007/978-3-319-30394-9_7
QGIS Development Team. QGIS geographic information system. Chicago, IL: Open Source Geospatial Foundation Project; 2024. Available from: https://qgis.org/pt_BR/site/
Ramos TPA, Barros Neto LF, Ferreira KCF, Barbosa JEL. Redescription of Parotocinclus haroldoi Garavello, 1988 (Siluriformes: Loricariidae), Northeastern Brazil. Zootaxa. 2020; 4741(2):321–32. https://doi.org/10.11646/zootaxa.4751.2.7
Ramos TPA, Ramos RTC, Ramos SAQA. Ichthyofauna of the Parnaíba river basin, Northeastern Brazil. Biota Neotrop. 2014; 14(1):e20130039. https://doi.org/10.1590/S1676-06020140039
Ripple WJ, Wolf C, Newsome TM, Hoffmann M, Wirsing AJ, McCauley DJ. Extinction risk is most acute for the world’s largest and smallest vertebrates. PNAS. 2017; 114(40):10678–83. https://doi.org/10.1073/pnas.1702078114
Rodrigues-Filho CAS, Gurgel-Lourenço RC, Bezerra LAV, Oliveira EF, Leitão RP, Garcez DS et al. How are local fish communities structured in Brazilian semiarid headwater streams? Hydrobiologia. 2018b; 819:93–108. https://doi.org/10.1007/s10750-018-3650-8
Rodrigues-Filho CAS, Gurgel-Lourenço RC, Bezerra LAV, Sousa WA, Garcez DS, Lima SMQ et al. Ichthyofauna of the humid forest enclaves in the tablelands of Ibiapaba and Araripe, Northeastern Brazil. Biota Neotrop. 2016; 16(4):e20160273. http://doi.org/10.1590/1676-0611-BN-2016-0273
Rodrigues-Filho CAS, Gurgel-Lourenço RC, Lima SMQ, Oliveira EF, Sánchez-Botero JI. What governs the functional diversity patterns of fishes in the headwater streams of the humid forest enclaves: environmental conditions, taxonomic diversity or biotic interactions? Environ Biol Fish. 2017; 100:1023–32. http://doi.org/10.1007/s10641-017-0603-4
Rodrigues-Filho CAS, Gurgel-Lourenço RC, Ramos EA, Novaes JLC, Garcez DS, Costa RS et al. Metacommunity organization in an intermittent river in Brazil: the importance of riverine networks for regional biodiversity. Aquat Ecol. 2019; 54:145–61. https://doi.org/10.1007/s10452-019-09732-1
Rodrigues-Filho CAS, Leitão RP, Zuanon J, Sánchez-Botero JI, Baccaro FB. Historical stability promoted higher functional specialization and originality in Neotropical stream fish assemblages. J Biogeogr. 2018a; 45(6):1345–54. http://doi.org/10.1111/jbi.13205
Rosa RS, Groth F. Ictiofauna dos ecossistemas de brejos de altitude de Pernambuco e Paraíba. In: Pôrto KC, Cabral JJP, Tabarelli M, editors. Brejos de altitude em Pernambuco e Paraíba. Brasília: Ministério do Meio Ambiente; 2004. p.201–28.
Rosa RS, Menezes NA, Britski HA, Costa WJEM, Groth F. Diversidade, padrões de distribuição e conservação nos peixes da Caatinga. In: Leal IR, Tabarelli M, Silva JMC, editors. Ecologia e conservação da Caatinga. Recife: Editora Universitária da UFPE; 2003. p.135–80.
Sabaj MH. Codes for Natural History Collections in Ichthyology and Herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
Sánchez-Botero JI, Garcez DS, Cascon P, Lima SMQ, Brito LBM, Roberto IJ. Anfíbios e peixes do Parque Nacional de Ubajara e Entorno – Guia Ilustrado. Fortaleza: UFC/LABOMAR/NAVE; 2014b.
Sánchez-Botero JI, Gurgel-Lourenço RC, Pinto LM, Leitão RP, Novaes JLC, Ramos TPA et al. Peixes estuarinos da costa semiárida do Brasil: guia ilustrado. Fortaleza: Expressão Gráfica e Editora; 2023.
Sánchez-Botero JI, Lourenço RCG, Rodrigues-Filho CAS, Ramos TPA, Pinto LM, Garcez DS. Lista de peixes continentais do Ceará. Fortaleza: Secretaria do Meio Ambiente do Ceará [Internet]. 2021. Available from: https://www.sema.ce.gov.br/fauna-do-ceara/peixes
Sánchez-Botero JI, Reis VC, Nascimento FD, Garcez DS. Fish assemblage of the Santo Anastácio reservoir (Ceará State, Brazil). Bol Inst Pesca. 2014a; 40(1):1–15.
Sánchez-Botero JI, Rodrigues-Filho CAS, Gurgel-Lourenço RC, Bezerra LAV, Sousa WA, Chaves FDN et al. Ecomorfologia de peixes como ferramenta na avaliação da unidade de conservação Parque Nacional de Ubajara. In: Mantovani W, Monteiro RF, Anjos L, Cariello MO, editors. Pesquisas em unidades de conservação no domínio da Caatinga: subsídios à gestão. Fortaleza: Edições UFC; 2017. p.465–79.
Secretaria de Meio Ambiente (SMA). Decreto 63.853, de 27 de novembro de 2018. Declara as espécies da fauna silvestre no Estado de São Paulo regionalmente extintas, as ameaçadas de extinção, as quase ameaçadas e as com dados insuficientes para avaliação. 2018. Available from: https://www.al.sp.gov.br/repositorio/legislacao/decreto/2018/decreto-63853-27.11.2018.html
Secretaria do Meio Ambiente (SEMA). Decreto 51.797, de 8 de setembro de 2014. Declara as Espécies da Fauna Silvestre Ameaçadas de Extinção no Estado do Rio Grande do Sul. 2014. Available from: http://www.al.rs.gov.br/filerepository/replegis/arquivos/dec%2051.797.pdf
Secretaria do Meio Ambiente (SEMA). Fauna do Ceará. 2022. www.sema.ce.gov.br/faunadoceara
Secretaria do Meio Ambiente (SEMA). Portaria n° 37 de 15 de agosto de 2017. Lista Oficial das Espécies da Fauna Ameaçadas de Extinção do Estado da Bahia. 2017. Available from: https://dool.egba.ba.gov.br/ver-html/2765/#e:2765
Secretaria do Meio Ambiente (SEMA). Portaria SEMA 1, de 4 de junho de 1998. Lista Estadual da Fauna Ameaçada de Extinção do Estado do Rio de Janeiro. 1998. Available from: https://www.rcambiental.com.br/Atos/ver/PORT-SEMA-RJ-1-1998
Silva AC, Sampaio SS, Viana MSR. Biologia e pesca do timbiro, Pterengraulis atherinoides, do açude Pereira de Miranda (Pentecoste, Estado do Ceará, Brasil). Acta Sci. 2005; 27(4):513–21.
Silva JWB, Araújo FAG. Resultados do povoamento de dez açudes públicos do Ceará, Brasil, com espécies selecionadas de peixes. Cienc Agron. 1996; 27(1/2):23–28.
Silva AT, Chagas RJ, Santos ACDA, Zanata AM, Rodrigues BK, Polaz CNM et al. Freshwater fishes of the Bahia State, Northeastern Brazil. Biota Neotropica. 2020a; 20(4):e20200969. https://doi.org/10.1590/1676-0611-bn-2020-0969
Silva MJ, Ramos TPA, Carvalho FR, Brito MFG, Ramos RTC, Rosa RS et al. Freshwater fish richness baseline from the São Francisco Interbasin Water Transfer Project in the Brazilian Semiarid. Neotrop Ichthyol. 2020b; 18(4):e200063. https://doi.org/10.1590/1982-0224-2020-0063
Sun J, Du W, Lucas MC, Ding C, Chen J, Tao J et al. River fragmentation and barrier impacts on fishes have been greatly underestimated in the upper Mekong River. J Environ Manage. 2023; 327:116817. https://doi.org/10.1016/j.jenvman.2022.116817
Teixeira FK, Ramos TPA, Paiva REC, Távora MA, Lima SMQ, Rezende CF. Ichthyofauna of Mundaú river basin, Ceará State, Northeastern Brazil. Biota Neotrop. 2017; 17(1):e20160174. http://doi.org/10.1590/1676-0611-BN-2016-0174
Terra BF, Medeiros ESF, Sánchez-Botero JI, Novaes JLC, Rezende CF. Ecologia de peixes de riachos intermitentes. Oecol Austr. 2021; 25(2):605–19. https://doi.org/10.4257/oeco.2021.2502.23
Thomé RG, Bazzoli N, Rizzo E, Santos GB, Ratton TF. Reproductive biology of Leporinus taeniatus Lütken (Pisces, Anostomidae) in Juramento Reservoir, São Francisco River basin, Minas Gerais, Brazil. Rev Bras Zool. 2005; 22(3):565–70. https://doi.org/10.1590/S0101-81752005000300006
Vorste RV, Sarrajame R, Datry T. Intermittent rivers and ephemeral streams: a unique biome with important contributions to biodiversity and ecosystem services. In: Goldstein MI, DellaSala DA, editors. Encyclopedia of the world’s biomes. Berkeley: Elsevier; 2020. p.419–29. https://doi.org/10.1016/B978-0-12-409548-9.12054-8
Walsh CJ, Roy AH, Feminella JW, Cottingham PD, Groffman PM, Morgan RP. The urban stream syndrome: current knowledge and the search for a cure. J N Am Benthol Soc. 2005; 24(3):706–23. https://doi.org/10.1899/04-028.1
Zacharias I, Zamparas M. Mediterranean temporary ponds. A disappearing ecosystem. Biodivers Conserv. 2010; 19(14):3827–34. https://doi.org/10.1007/s10531-010-9933-7
Authors
![]() Maria Cecília Feitoza Gomes1,
Maria Cecília Feitoza Gomes1, ![]() Patricia Charvet1,
Patricia Charvet1, ![]() Leonardo Mesquita Pinto2,
Leonardo Mesquita Pinto2, ![]() Ronaldo César Gurgel-Lourenço2,
Ronaldo César Gurgel-Lourenço2, ![]() Telton Pedro Anselmo Ramos3,
Telton Pedro Anselmo Ramos3, ![]() Sergio Maia Queiroz Lima4,
Sergio Maia Queiroz Lima4, ![]() Carlos Alberto de Sousa Rodrigues-Filho5,6,
Carlos Alberto de Sousa Rodrigues-Filho5,6, ![]() Danielle Sequeira Garcez7 and
Danielle Sequeira Garcez7 and ![]() Jorge Iván Sánchez-Botero8
Jorge Iván Sánchez-Botero8 ![]()
[1] Programa de Pós-graduação em Sistemática, Uso e Conservação da Biodiversidade, Universidade Federal do Ceará, Departamento de Biologia, Av. Mister Hull s/n, 60455-760 Fortaleza, CE, Brazil. (MCFG) cecilia.feitozak@gmail.com, (PC) pchalm@gmail.com.
[2] Universidade Federal do Ceará, Campus do Pici, Departamento de Biologia, Laboratório de Ecologia Aquática e Conservação, Av. Mister Hull s/n, 60455-760 Fortaleza, CE, Brazil. (LMP) leopinto.ca@gmail.com, (RCGL) ronaldocgl@yahoo.com.br.
[3] Universidade Federal da Paraíba, Laboratório de Ecologia Aplicada e Conservação, 58051-900 João Pessoa, PB, Brazil. (TPAR) telton@gmail.com.
[4] Universidade Federal do Rio Grande do Norte, Laboratório de Ictiologia Sistemática e Evolutiva, 59078-970 Natal, RN, Brazil. (SMQL) sergio.lima@ufrn.br.
[5] Instituto Nacional de Pesquisas da Amazônia, Av. André Araújo, 2936, 69067-375 Manaus, AM, Brazil. (CASRF) carlosfilho918@gmail.com.
[6] Instituto de Desenvolvimento Sustentável Mamirauá (IDSM), Estrada do Bexiga, 2584, 69553-225 Tefé, AM, Brazil.
[7] Universidade Federal do Ceará, Instituto de Ciências do Mar – Labomar, Laboratório de Ecologia Pesqueira e Estudos Socioambientais, Av. da Abolição 3207, 60165-081 Fortaleza, CE, Brazil. (DSG) daniellegarcez@ufc.br.
[8] Universidade Federal do Ceará, Campus do Pici, Departamento de Biologia, Laboratório de Ecologia Aquática e Conservação, Av. Mister Hull s/n, 60455-760 Fortaleza, CE, Brazil. (JISB) jorgebotero.leac@ufc.br (corresponding author).
Authors’ Contribution 

Maria Cecília Feitoza Gomes: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Patricia Charvet: Conceptualization, Methodology, Supervision, Validation, Writing-review and editing.
Leonardo Mesquita Pinto: Conceptualization, Formal analysis, Investigation, Methodology, Writing-review and editing.
Ronaldo César Gurgel-Lourenço: Conceptualization, Formal analysis, Methodology, Writing-review and editing.
Telton Pedro Anselmo Ramos: Data curation, Methodology, Validation, Writing-review and editing.
Sergio Maia Queiroz Lima: Data curation, Methodology, Validation, Writing-review and editing.
Carlos Alberto de Sousa Rodrigues-Filho: Investigation, Methodology, Writing-review and editing.
Danielle Sequeira Garcez: Conceptualization, Project administration, Resources, Writing-review and editing.
Jorge Iván Sánchez-Botero: Conceptualization, Methodology, Project administration, Resources, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
How to cite this article
Gomes MCF, Charvet P, Pinto LM, Gurgel-Lourenço RC, Ramos TPA, Lima SMQ, Rodrigues-Filho CAS, Garcez DS, Sánchez-Botero JI. Conservation status of the freshwater fish species from Ceará State, Brazil. Neotrop Ichthyol. 2025; 23(2):e240084. https://doi.org/10.1590/1982-0224-2024-0084
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted March 7, 2025
Accepted March 7, 2025
![]() Submitted August 22, 2024
Submitted August 22, 2024
![]() Epub May 26, 2025
Epub May 26, 2025