![]() Josiane Rodrigues Rocha da Silva1
Josiane Rodrigues Rocha da Silva1 ![]() ,
, ![]() Camila Oliveira de Andrade1,
Camila Oliveira de Andrade1, ![]() Patrícia Daniele Silva dos Santos2,
Patrícia Daniele Silva dos Santos2, ![]() Oscar Oliveira Santos2,
Oscar Oliveira Santos2, ![]() Roney Peterson Pereira3 and
Roney Peterson Pereira3 and ![]() Carlos Alexandre Fernandes1,4
Carlos Alexandre Fernandes1,4
PDF: Download Here | Supplementary: S1 | Cite this article
Associate Editor: ![]() Claudia Martinez
Claudia Martinez
Section Editor: ![]() Bernardo Baldisserotto
Bernardo Baldisserotto
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
O cloridrato de fluoxetina (FLX) é um antidepressivo classificado como um inibidor seletivo da recaptação da serotonina. É comumente encontrado em ambientes aquáticos devido à excreção humana e práticas inadequadas de descarte. A presença de FLX levanta preocupações, pois está ligada a alterações comportamentais, fisiológicas e genéticas em organismos aquáticos não alvos. Este estudo investigou os efeitos tóxicos do FLX em Astyanax altiparanae, com foco nas respostas sanguíneas e branquiais, e avaliou a eficiência de um wetland construído de fluxo vertical (WCFV) como uma solução de tratamento sustentável. Cinco grupos foram estabelecidos: um controle, três expostos a concentrações crescentes de FLX (0.01, 0.1 e 1 mg L-1) e um exposto a 1 mg L-1 de efluente tratado por WCFV. Após 96 horas, os espécimes expostos ao FLX mostraram um aumento significativo na frequência de micronúcleos (MN), alterações morfológicas celulares (AMC), danos ao DNA e lesões histopatológicas nas brânquias. Em contraste, nossos resultados indicaram que mais de 99% do FLX foi removido no WCFV, com cerca de 89% de células sem danos ao DNA, uma redução significativa em MN, AMC e lesões histopatológicas. Esses resultados demonstram a eficiência do sistema na remoção do FLX e na mitigação de seus efeitos adversos em organismos aquáticos.
Palavras-chave: Biorremediação, Ecotoxicologia, Fármacos, Método sustentável, Tecnologias verdes.
Introduction
Pharmaceutical compounds have consistently been released into aquatic environments (Correia et al., 2023). Among these, antidepressants have gained significant attention due to their presence in various ecosystems, including soil surfaces and near wastewater treatment facilities. Such contaminants often infiltrate water resources (Duarte et al., 2019), primarily through sewage from various sources, including hospitals, wastewater treatment plants, and pharmaceutical manufacturing sites. Consequently, this leads to the contamination and toxicity of natural water sources (Nałęcz-Jawecki et al., 2020).
Fluoxetine hydrochloride (FLX), an antidepressant of the selective serotonin reuptake inhibitor class, is widely used in the treatment of psychiatric disorders but can cause significant environmental impacts. The FLX is primarily excreted from the human body through the urinary system, with approximately 10% being eliminated as the parent compound (FLX) and the remainder as norfluoxetine (Hiemke, Härtter, 2000; Brooks et al., 2003).
Alongside the parent compound, norfluoxetine, the primary metabolite, exhibits pharmacological activity and notable persistence, with concentrations often equal to or exceeding those of FLX in the environment (Nałęcz-Jawecki et al., 2020). This raises significant concerns about potential ecotoxicological effects, as both the drug and its metabolites may impact aquatic organisms. Both compounds can reach wastewater treatment facilities through human excretion and the improper disposal of unused medication (Brodin et al., 2013).
The FLX is hydrolytically and photolytically stable, with an environmental half-life of 14–28 days depending on light and temperature, which hinders its removal during conventional wastewater treatment (Kwon, Armbrust, 2006). However, its stability is highly dependent on environmental conditions, and Pan et al. (2022) demonstrated that FLX can be almost completely degraded within five days under UV irradiation, underscoring the variability of its environmental persistence. Consequently, environmental concentrations of this drug are often detected at levels ranging from 0.012 to 1.4μg/L-1 (Calisto, Esteves, 2009; Togunde et al., 2012; Gomes, Gomes, 2024), raising concerns about its potential effects on aquatic biota (Daughton, Ternes, 1999).
The FLX has the potential to act as an endocrine disruptor in aquatic ecosystems, interfering with the hormonal regulation and reproductive capacities of aquatic organisms (Fong, 1998). It also impacts fish behavior, altering feeding activity, locomotion, and escape responses, which may ultimately compromise survival (Orozco-Hernández et al., 2022). Prolonged exposure has been linked to stress, hematological changes, and damage to vital organs, including the liver and kidneys, as well as the nervous system (Correia et al., 2023). Moreover, exposure to FLX can hinder feeding, growth, reproduction, and overall population success in fish, as evidenced by studies on Cichlasoma dimerus subjected to intraperitoneal injection (Dorelle et al., 2020), highlighting the compound’s significant ecotoxicological implications.
Although FLX is water-soluble, solubility alone does not dictate its potential for bioaccumulation. Its moderate lipophilicity (log Kow ≈ 4.05) promotes interactions with biological membranes and plasma proteins, resulting in accumulation in tissues such as the liver, gills, and brain (Pan et al., 2018; Duarte et al., 2020). Additionally, FLX has a relatively long half-life in organisms (4–6 days in humans and up to 16 days for norfluoxetine), which further enhances its bioaccumulative potential. Furthermore, the physiological characteristics of the organisms exposed, along with various environmental factors, can significantly influence absorption, distribution, and metabolism (Yamindago et al., 2021).
In traditional wastewater treatment facilities, the primary processes consist of coagulation/flocculation, sedimentation, filtration, and disinfection. Although these methods are effective in eliminating suspended solids and organic matter, they are less effective at removing micropollutants, such as pharmaceuticals and hormones. This limitation accounts for the persistent presence of these substances in treated effluents (Kaushik, Thomas, 2019; Reis et al., 2019). In response, Constructed Wetlands (CWs) have emerged as a promising and sustainable technology for the phytoremediation of contaminated wastewater. These systems typically consist of shallow ponds or canals that contain filter beds supporting aquatic vegetation. Contaminated wastewater is routed through these filter beds, where a combination of physical, chemical, and biological processes — such as filtration, sedimentation, adsorption, precipitation, and biodegradation — takes place. These processes effectively remove a wide range of contaminants from effluents (Dotro et al., 2021; Hassan et al., 2021; Kiflay et al., 2021). Consequently, CWs have garnered significant recognition as a viable option for treating contaminated wastewater across various contexts.
The CWs offer an ecologically sustainable solution for water remediation, presenting several advantages over traditional treatment methods, such as lower costs and simpler operation (Sezerino et al., 2018). These systems are effective at removing suspended solids, organic matter, and soluble nutrients, making them particularly suitable for protecting aquatic communities and enabling non-potable water reuse, like irrigation. However, their removal efficiency is influenced by factors such as the type of contaminant, hydraulic retention time, and the physicochemical characteristics of the influent. It’s important to note that CWs are not recommended for direct human consumption without additional potabilization steps (Dotro et al., 2021; Hassan et al., 2021). Fish serve as effective bioindicators due to their sensitivity to contaminants and their capacity to accumulate pollutants, which leads to observable physiological, biochemical, and histological responses (Monteiro et al., 2010; Tincani et al., 2019). Astyanax altiparanae Garutti & Britski, 2000, commonly found in Brazilian freshwater systems, is noted for its tolerance to a variety of environmental conditions and its ecological significance, making it an ideal model for ecotoxicological research (Schulz, Martins-Junior, 2001; Ostrensky et al., 2016). Due to the scarcity of studies examining the cyto/genotoxic potential of FLX in aquatic environments, this research aimed to assess the toxicological effects of FLX on the blood and gills of A. altiparanae. Additionally, it sought to evaluate the effectiveness of a sustainable treatment approach using a vertical flow constructed wetland (VFCW) system to manage this contaminant.
Material and methods
Fluoxetine hydrochloride. The pharmaceutical fluoxetine hydrochloride (TEUTO® Brazilian Laboratory S/A, FLX, batch: 2561244, CAS no: 54910–89–3) was acquired from a local commercial supplier for experimental purposes. A stock solution was prepared by dissolving an accurate quantity of FLX in distilled water, achieving a concentration of 10 mg L⁻¹. This solution was stored at 4 °C. Experimental concentrations were derived by diluting the stock solution in specific proportions with dechlorinated water.
The drug was administered at varying concentrations for each group: Group 1 served as the negative control and received only dechlorinated water; Group 2 was exposed to a concentration of 0.01 mg L⁻¹; Group 3 to 0.1 mg L⁻¹; Group 4 to 1 mg L⁻¹; and Group 5, also at 1 mg L⁻¹, but with prior treatment through the VFCW (Vertical Flow Constructed Wetland) system utilizing phytoremediation. The concentrations were selected based on Vijitkul et al. (2022), who evaluated the effects of FLX on fish.
Fluoxetine hydrochloride analysis in water
Sample preparation. For the chromatographic analysis of FLX, a solution with a concentration of 1 mg L⁻¹ was prepared. The sample underwent centrifugation at 10,000 rpm for 20 min at 4 °C, after which the supernatant was filtered through a 0.22 µm syringe filter. Identification of fluoxetine hydrochloride was achieved by comparing the retention time of the peak in the sample with that of a standard solution.
The samples were introduced into an Acquity UPLC® system (Milford, MA, USA) coupled with an Acquity TQDTM triple quadrupole mass spectrometer (Milford, MA, USA), which was equipped with a Waters Zspray™ (ESI) ionization source (Milford, MA, USA). The mobile phases comprised ultrapure water acidified with 0.1% formic acid (A) and acetonitrile (B). The mass spectrometer operated in positive ion mode with the following settings: capillary voltage of 3.0 kV, cone voltage adjusted based on the analyte, desolvation gas temperature of 500 °C with a gas flow rate of 400 L h⁻¹, and collision energy set at 12 V. The separation of analytes was performed on an Acquity UPLC® CSH C18 column (100 mm × 2.1 mm, 1.8 µm).
The gradient elution program utilized a flow rate of 0.20 mL min⁻¹ with the following solvent ratios: 65A:35B from 0 to 2.0 min, 20A:80B from 2.0 to 4.0 min, and 65A:35B maintained until the total run time of 8.0 min. The column temperature was kept at 40 °C, and the injection volume was 2.0 µL. Data processing was carried out using MassLynx™ 4.1 software (Milford, MA, USA), and results were reported in ppm.
Experimental design. Adult individuals (both males and females) of Astyanax altiparanae were used, with an average weight of 9 ± 12 g and length of 8 ± 12 cm, obtained from a local fish farm. The fish were acclimated in aquaria with dechlorinated water at room temperature, under constant aeration, a natural photoperiod (12:12 h light/dark cycle), and daily feeding with specific commercial feed for small fish (Basic Alcon® Fish Food, Camboriú, SC, Brazil) for 10 days at the Sectoral Vivarium for Fish Maintenance and Experimentation at the Universidade Estadual de Maringá (UEM), in Maringá, Paraná, Brazil.
A total of five experimental groups were tested, each consisting of four fish, and all experiments were carried out in triplicate, resulting in a total of 60 fish. The aquarium housed two fish per unit, which was determined based on the body mass-to-water volume ratio, adhering to the recommended limit of 0.5–2 g/L as outlined in the CONCEA Guide (Annex I – Fish). In the control group (Group 1), fish were maintained in dechlorinated water. In the remaining experimental groups (Groups 2 to 4), fish were exposed to FLX at concentrations of 0.01, 0.1, and 1 mg L⁻¹, respectively. In Group 5, the 1 mg L⁻¹ concentration of FLX was previously subjected to phytoremediation using the VFCW system, and the fish were subsequently exposed to the resulting treated effluent. Each group was exposed to FLX dissolved in 10 L of water for 96 h, characterizing an acute test. All samples were coded and analyzed under blind conditions.
Dissection and sampling. After four days of exposure, the fish were removed from the aquarium (one at a time) and bathed in an anesthetic solution: clove oil, 5 ml, diluted with ethyl alcohol, 20 ml, as per Inoue et al. (2005). Then, 1 ml of this solution was added per L of water. The animal was only manipulated after it failed to respond to physical stimulus, denoting death from anesthetic overdose (Svobodová, Vykusová, 1991). After euthanasia, the blood was collected to perform the micronucleus tests, cellular morphological changes, and comet assay in erythrocytes, as well as, the gills of the animals were collected and stored for histopathological analysis, as described below. Finally, the animals were fixed in commercial absolute alcohol, recorded with number and origin, and stored in glass vials from the Universidade Estadual de Maringá – UEM/NUPELIA laboratory. Voucher specimens were deposited in the Fish Collection of the Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (NUPELIA), Universidade Estadual de Maringá, municipality of Maringá, Paraná State, Brazil, as Astyanax altiparanae (NUP 25442).
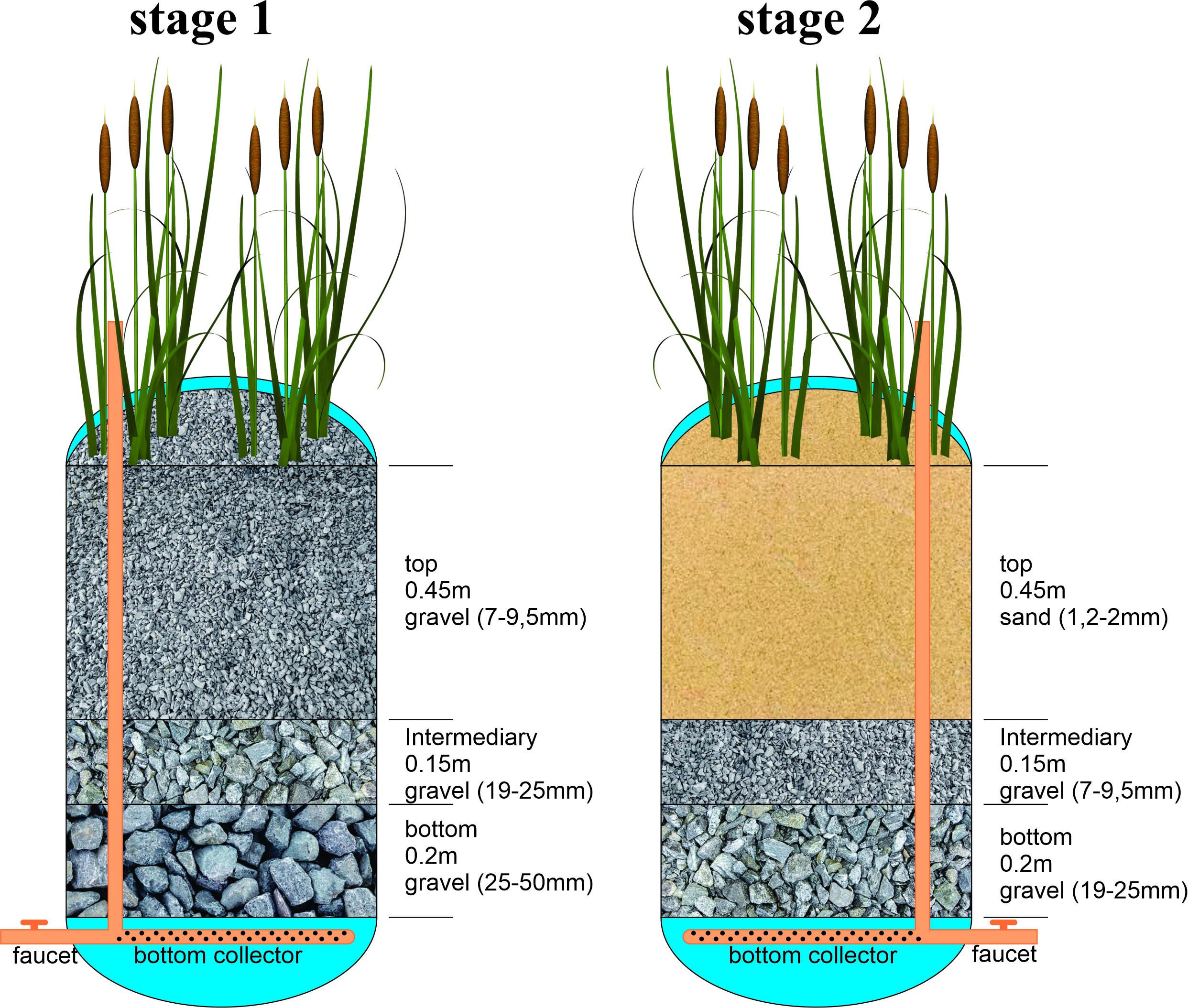
Construction of experimental built wetland treatment units with the vertical flow (VFCW). The making of the VFCW units followed some suggestions from the manual by Sezerino et al. (2018), with adaptations according to Silva et al. (2024). This study made them from two cylindrical high-density polyethylene (HDPE) containers (0.80 m in height X 0.55 m in diameter). The filter mass of the first stage was composed of 0.2 m of crushed stone (25–50 mm), 0.15 m of crushed stone (19–25 mm), and 0.45m of crushed stone (7–9.5 mm) (gravel). The second stage consisted of 0.2 m of gravel (19–25mm), 0.15m of gravel (7–9.5 mm), and 0.45 m of sand (1.2–2 mm). The adduction system consisted of 25 mm diameter polyvinyl chloride pipes and connections. The effluent was drained through an adductor system with perforations of 8.0 mm in diameter distributed along its entire extremity. The drainage pipe was positioned horizontally at the bottom of the bed, extending across the whole diameter of the units. The system contains a faucet for outputting the treated effluent at the bottom of the reservoir, installed 10 cm from the bottom (Fig. 1).
FIGURE 1| Scheme of an experimental vertical flow constructed wetland system (VFCW) unit.
The beds were populated with Typha domingensis at 16 plants per square meter density. The propagules were collected manually in a naturally flooded area located on a rural property in the municipality of Cianorte at the beginning of December 2021. The collection was carried out so that the rhizomes were preserved, carefully transported, and transplanted in the experimental units. The residence time of the effluent in the treatment system was 96 h, 48 h in the first stage, and 48 h in the second stage.
Micronucleus test and cellular morphological changes in erythrocytes. The micronucleus (MN) and cellular morphological changes (CMC) in the erythrocytes test were performed based on the description by Hooftman, Raat (1982). After the anesthesia of the animals, blood was extracted from the caudal vein with a heparinized syringe. A sample (approximately 10 μl) was dripped onto a sanitized glass slide, and the smear was performed with the aid of another slide. The dripped slide was kept at room temperature, drying for at least 12 h. Afterward, they were fixed with absolute ethanol for 20 min. Staining was performed for 10 min with 5% Giemsa solution diluted in phosphate buffer (pH 6.8). Then, the slides were washed in distilled water, left to dry naturally, and kept in closed boxes until analysis under microscopy. The slides were analyzed by optical microscopy under 1000 × magnification. The MN count and the study of CMC were performed on 2,000 erythrocytes per fish.
Comet assay. The alkaline Comet assay was performed according to Speit, Hartmann (1999), without species-specific modifications. Analyses were conducted in a blinded manner to assess conditions under an epifluorescence microscope, with images captured at 400× magnification. DNA damage was evaluated by visually classifying 100 nucleoids per fish into five categories (0–4), where class 0 indicates no damage and class 4 represents maximum damage. This assessment allowed for the derivation of two complementary parameters (Collins et al., 2023): (i) the DNA Damage Index (DI), representing the intensity of damage, calculated as the sum of the nucleoids in each class multiplied by the corresponding class value, with a range from 0 (no damage) to 400 arbitrary units (maximum damage); and (ii) the DNA Damage Frequency (DF), which reflects the proportion of nucleoids exhibiting some degree of damage (classes 1–4), expressed as a percentage of the total analyzed. The DI provides a measure of the average severity of DNA strand breaks, whereas the DF indicates the percentage of affected cells within each experimental group.
Histological analysis. After their gills were removed and washed in 0.9% saline solution and fixed in Bouin’s aqueous solution for 12 h (Behmer et al., 1976), subsequently stored in 70% alcohol. For histological processing, the material was dehydrated in an ascending series of alcohol (80%, 90% and 100%), clarified in xylene and embedded in paraffin. Semi-serial cross-sections of 5 μm thickness were obtained using a LEICA rotary microtome at the Animal Histotechnology Laboratory of the Department of Morphological Sciences at the Universidade Estadual de Maringá. The slides were stained by the Hematoxylin-Eosin (H.E.) and Periodic Acid-Schiff (PAS) methods (Behmer et al., 1976). An optical microscope (Olympus CX31RBSFA) was used to analyze the changes.
Quantitative analysis of gill changes. For morphological analysis and changes in the gills, 30 random fields per animal were evaluated under an optical microscope with a total magnification of 40x (Olympus CX31RBSFA) according to the semiquantitative method proposed by Schwaiger et al. (1997). Graduated Histological Change Index (HAI) scale, depending on the severity of the lesions, as described by Mallat (1985) with adaptation: Damage 0 = no histological changes; Damage 1: small changes; Damage 2: moderate and specific changes; Damage 3: moderate and extensive changes; Damage 4: severe, extensive and irreparable changes, due to secondary lamellar fusion, hypertrophy and hyperplasia of lamellar epithelial cells. For telangiectasia and aneurysm, 30 random fields per animal were evaluated under an optical microscope at 40x total magnification (Olympus CX31RBSFA), and the quantity per field was counted.
Statistical analysis. Statistical analyses were performed separately using the Kolmogorov-Smirnov normality test. Micronucleus (MN) and cellular morphological changes (CMC) data were obtained by the One-way ANOVA test followed by Tukey’s post-test. Data were received using the one-way ANOVA test for the comet assay. For analyses Gills histopathological, statistical analyses of the collected data were conducted to assess normality using the Kolmogorov-Smirnov test. Levene’s test was used to assess the homogeneity of variances between the experimental groups. The results indicated that the variances between the groups were comprehensive (p > 0.05), allowing the application of ANOVA for data analysis. A one-way analysis of variance (ANOVA) was performed, accompanied by Tukey’s post-test. A significance level of 5% was set, and the results were reported as mean ± standard error.
For the analysis of the frequency distributions of damage in the alterations of secondary lamellar fusion, hyperplasia, and hypertrophy, between the experimental groups and the control group, two statistical tests were performed: the Chi-square test and the Fisher’s exact test (p > 0.05).
Results
Analysis of MN and CMC in erythrocytes. The exposure of Astyanax altiparanae specimens to FLX resulted in various morphological alterations in erythrocytes. The sublethal concentrations of the compound used in this study induced a significantly higher number of micronuclei (MN) and other nuclear abnormalities (CMC) in erythrocytes compared to the control group.
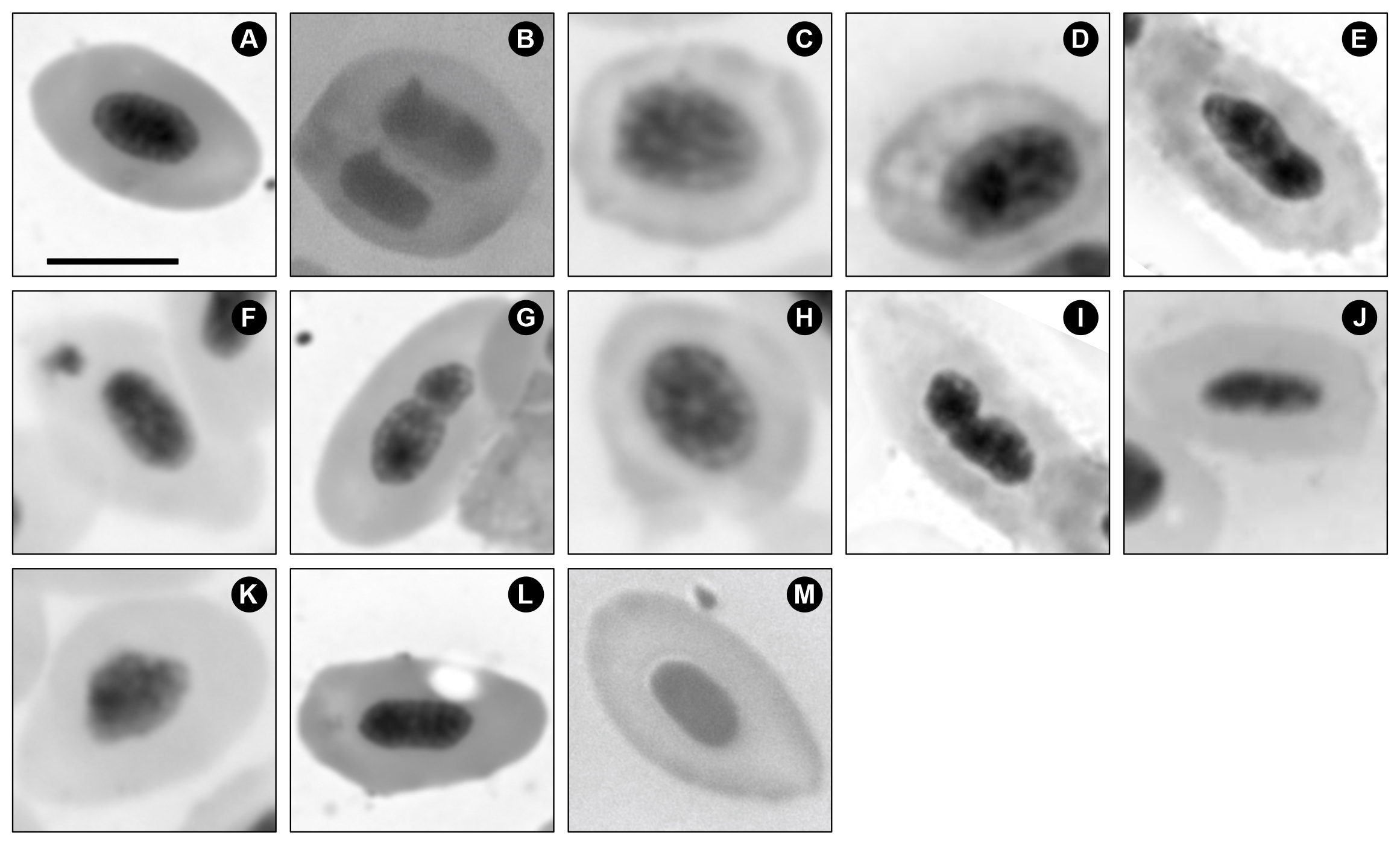
A total of 120,000 erythrocytes from 60 A. altiparanae specimens were analyzed, revealing 13 micronucleated cells and 1,428 morphological cellular changes (Tab. 1). The observed alterations included: micronucleus (12), notched nucleus (143), binucleated cell (57), macronucleus (63), fragmented nucleus (217), ellipsoid erythrocyte (160), blebbed nucleus (30), lobed nucleus (30), nuclear constriction (65), crenated erythrocyte (197), cytoplasmic vacuole (311), and tear-shaped erythrocyte (143). Some of these alterations can be observed in Fig. 2.
TABLE 1 | Absolute numbers for alteration in Astyanax altiparanae erythrocytes. For each fish, 2000 cells were analyzed. *Significantly different to control group < 0.05; MN = micronuclei; CMC = Cellular morphological changes.
Group | MN | Erythrocytes with CMC | Mean ± standard deviation of MN and CMC numbers |
Control | 0 | 46 | 3.7 ± 2.1 |
0.01 mg/L-1 | 1 | 282 | 23.5 ± 4.6* |
0.1 mg/L-1 | 2 | 339 | 27.5 ± 9.5* |
1 mg/L-1 | 8 | 663 | 56.0 ± 14.8* |
Treated VFCW | 1 | 86 | 7.2 ± 2.2 |
FIGURE 2| Photomicrograph of Astyanax altiparanae erythrocytes exposed to fluoxetine hydrochloride and stained with Giemsa. A. Normal Erythrocyte; B. Binucleate; C. Crenated Erythrocyte; D. Macronucleus; E. Blebbed; F. Micronuclei; G. Nuclear Constriction; H. Fragmented Nucleus; I. Lobed; J. Elliptocyte; K. Notched; L. Cytoplasmic Vacuole; M. Tear-Drop. Scale bar = 5 µm.
No significant differences in the frequency of MN and CMC were observed between the control group and the FLX group treated via the VFCW system (p > 0.05 in Tab. 1). However, the other exposure concentrations showed statistically significant differences when compared to both the control and the VFCW-treated fluoxetine group (p < 0.05 in Tab. 1).
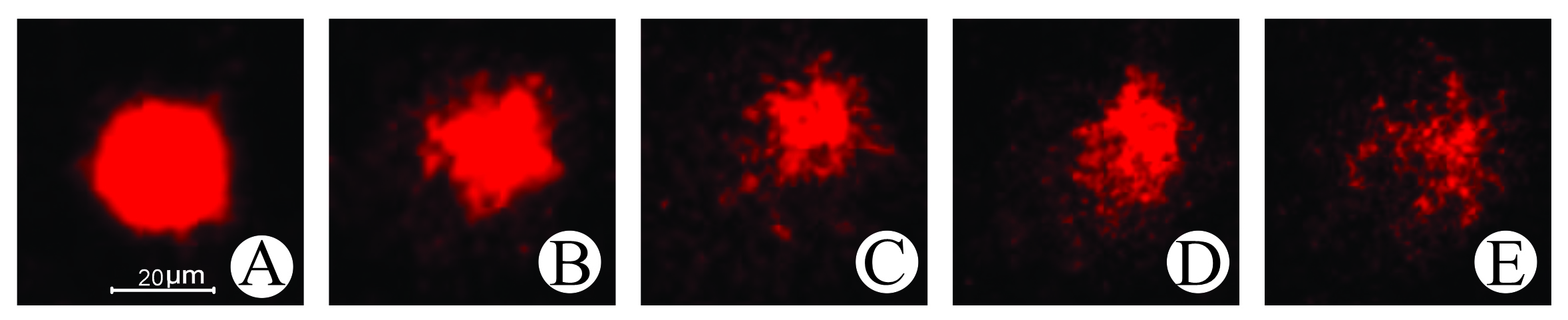
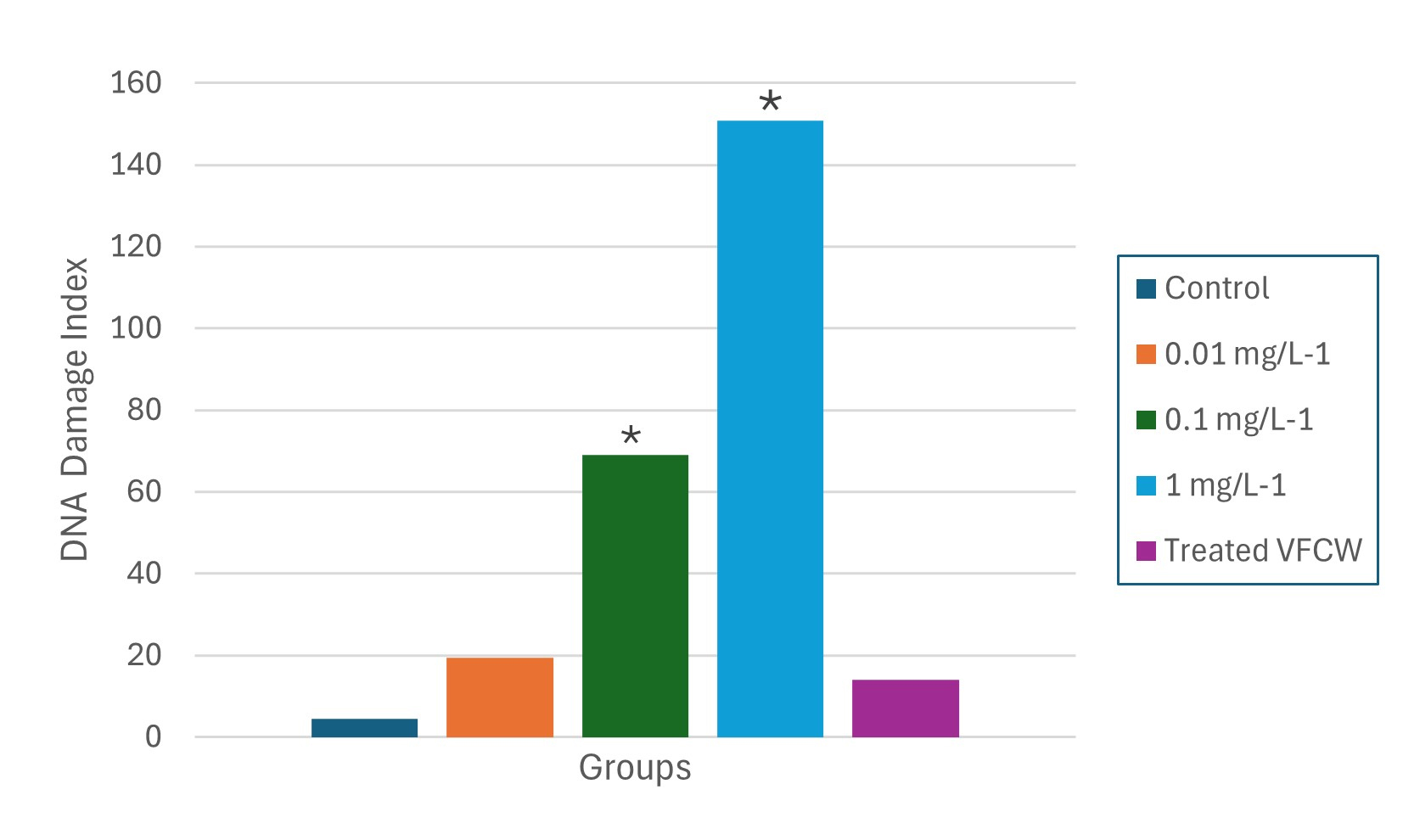
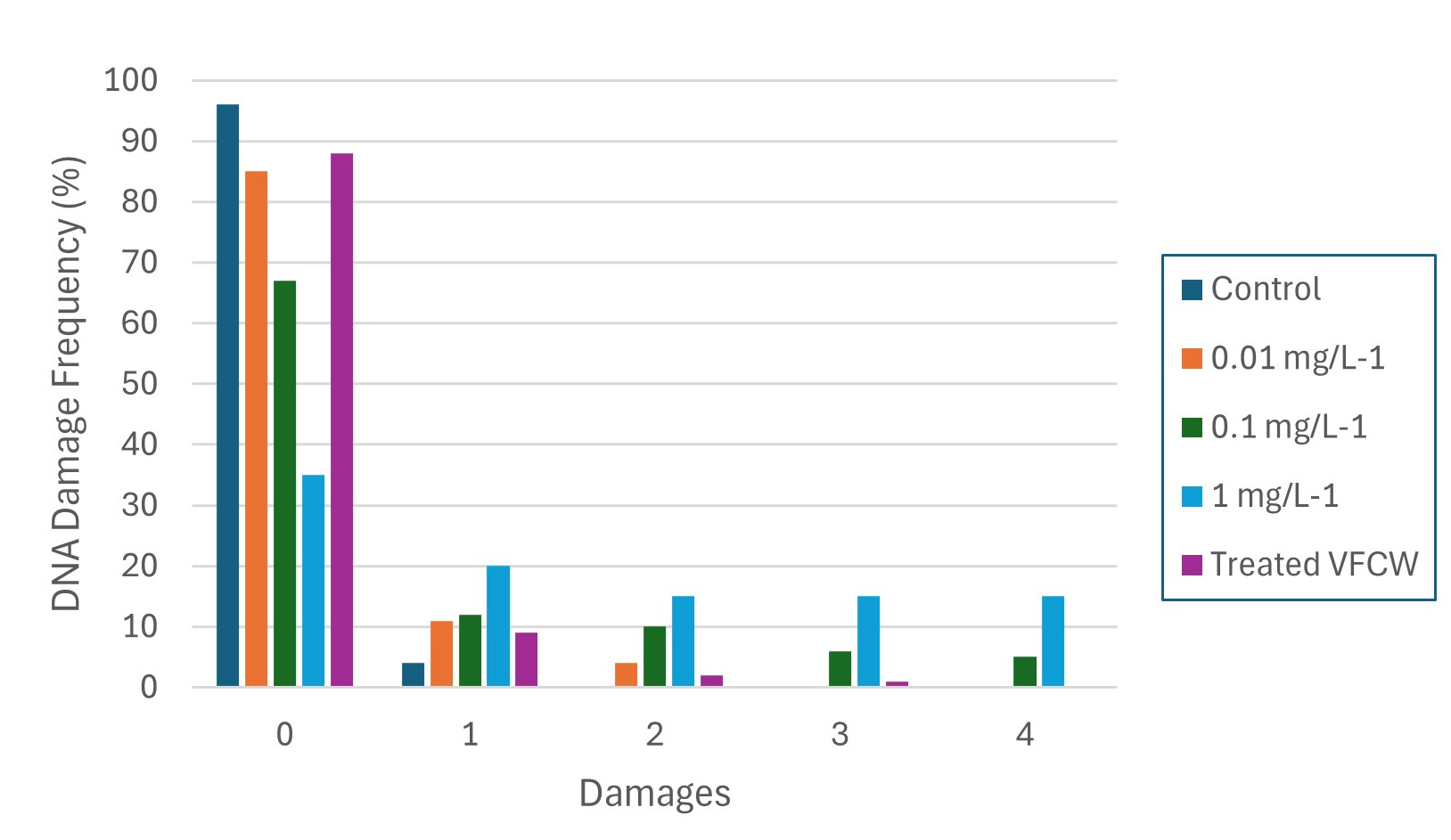
Comet assay. DNA damage classes 1, 2, 3 and 4 were detected through the comet assay only in the groups exposed to 0.1 and 1 mg L⁻¹ (Fig. 3). An apparent increase in DNA damage index (DI) was observed, with the highest values recorded at concentration 1 mg L⁻¹ of FLX. In contrast, the group treated by VCFW showed a DI close to that of the control group (Fig. 4).
FIGURE 3| Damage to erythrocytes from Astyanax altiparanae specimens from the control group and following exposure to FLX was measured by the comet assay (Magnification 400x). A. Normal cell – damage 0; B. Damage 1; C. Damage 2; D. Damage 3; E. Damage 4. Scale bar = 20 µm.
FIGURE 4| DNA Damage Index (DI, mean ± SE) in erythrocytes of Astyanax altiparanae after 96 h of exposure to FLX (0.01, 0.1, 1 mg L-1) and VFCW-treated effluent, compared to the control group. *Significantly different to control group < 0.05.
The distribution of damage classes (DF) indicated that the control group demonstrated approximately 96% of erythrocytes without alterations, with only 4% exhibiting some degree of damage. In contrast, the group exposed to 1 mg L⁻¹ showed a significant decrease in intact cells (36%) and an increase in damaged cells to 64%. Intermediate concentrations revealed about 15% of damaged cells at 0.01 mg L⁻¹ and 33% at 0.1 mg L⁻¹. Conversely, the group exposed to VFCW-treated effluent displayed around 89% of cells without damage, a result comparable to the control group (p > 0.05) (Fig. 5). Statistically significant differences (p < 0.05) were observed between the groups exposed to concentrations of 0.1 and 1 mg L⁻¹ of FLX and both the control and VFCW-treated groups (Fig. 4).
FIGURE 5| DNA Damage Frequency (DF, %, mean ± SE) in erythrocytes of Astyanax altiparanae after 96 h exposure to FLX (0.01, 0.1, 1 mg L-1) and VFCW-treated effluent. Class 0 = no damage; Classes 1–4 = increasing DNA damage.
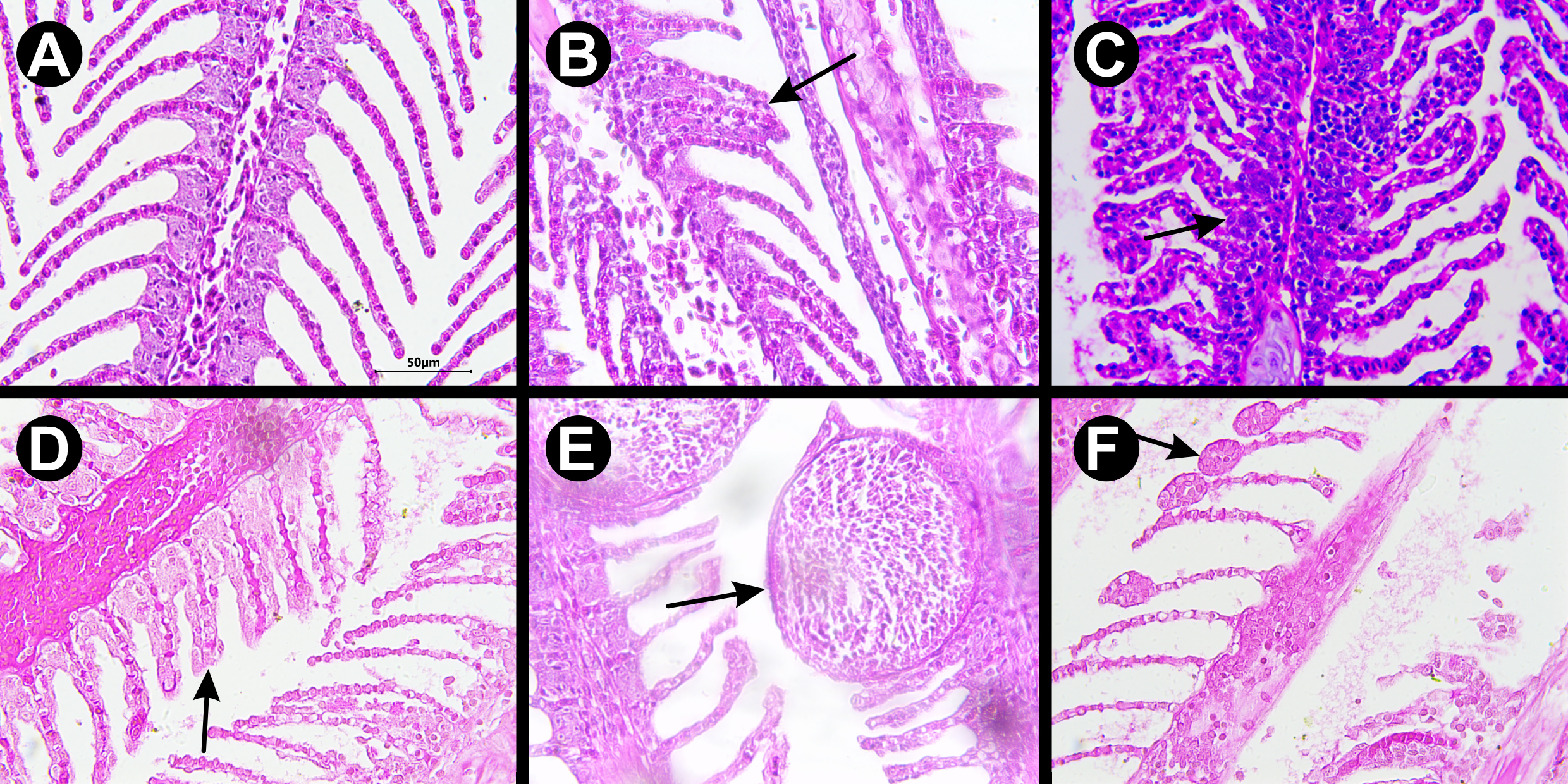
Histological analysis of gills. Normal gills exhibited the expected structure, with well-defined and organized lamellae, and no signs of damage or abnormalities. However, several other pathological alterations were observed in samples exposed to FLX (Fig. 6), with severity and frequency increasing proportionally to the concentration.
FIGURE 6| Photomicrograph of Astyanax altiparanae gills exposed to different concentrations of fluoxetine hydrochloride and stained with hematoxylin and eosin (HE). A. Control; B. Secondary lamellar fusion; C. Hypertrophy; D. Hyperplasia; E. Telangiectasia; F. Aneurysm. Scale bar = 50 µm.
For instance, secondary lamellar fusion was observed with 30 mild alterations in the control group, whereas the group exposed to 1 mg L⁻¹ presented 64 severe, extensive, and/or irreversible alterations (Tab. 2). Regarding lamellar epithelial hyperplasia, the control group showed only 18 mild changes, while the 1 mg L⁻¹ group exhibited 61 severe, extensive, and/or irreversible changes. Similarly, in epithelial cell hypertrophy, the control group showed only 14 mild alterations, while the group exposed to 1 mg L⁻¹ showed 53 severe alterations (Tab. 2).
TABLE 2 | Levels of histopathological damage in the gills of Astyanax altiparanae, consolidating lamellar fusion, hyperplasia, and hypertrophy. Damage 0 = no histological alteration; Damage 1 = mild alterations; Damage 2 = moderate and specific alterations; Damage 3 = moderate and extensive alterations; Damage 4 = severe, extensive, and/or irreversible alterations. *Significantly different to control group < 0.05.
Group | Secondary Lamellar Fusion | Hyperplasia | Hypertrophy | ||||||||||||
Damage | 0 | 1 | 2 | 3 | 4 | 0 | 1 | 2 | 3 | 4 | 0 | 1 | 2 | 3 | 4 |
Control | 120 | 30 | 0 | 0 | 0 | 122 | 18 | 10 | 0 | 0 | 130 | 14 | 6 | 0 | 0 |
0.01 mg/L-1 * | 62 | 42 | 30 | 11 | 5 | 52 | 68 | 30 | 0 | 0 | 86 | 32 | 21 | 11 | 0 |
0.1 mg/L-1 * | 21 | 30 | 38 | 44 | 17 | 0 | 18 | 61 | 57 | 14 | 49 | 77 | 11 | 13 | 0 |
1 mg/L-1 * | 3 | 20 | 31 | 32 | 64 | 0 | 21 | 33 | 35 | 61 | 0 | 19 | 38 | 40 | 53 |
Treated (VFCW) | 72 | 52 | 16 | 10 | 0 | 68 | 46 | 27 | 9 | 0 | 110 | 22 | 10 | 8 | 0 |
The results for telangiectasia at the 0.01 mg L⁻¹ concentration (P = 0.1861) and in the group treated with WCFV-treated fluoxetine showed no significant differences compared to the control group (p > 0.05 in Tab. 3). In the aneurysm analysis, only the group exposed to VFCW-treated fluoxetine did not differ significantly from the control (p > 0.05 in Tab. 3), while all other concentrations presented statistically significant differences from the control. Both telangiectasia and aneurysms showed dose-dependent responses.
TABLE 3 | Whole values representing the total number of aneurysms and telangiectasias observed in the gills of Astyanax altiparanae. Data correspond to the sum of lesions recorded in five individuals per experimental group. *Significantly different to control group < 0.05.
FLX Concentration | Total Aneurysms | Total Telangiectasia |
Control | 41 | 24 |
0.01 mg/L1 | 132* | 53 |
0.1 mg/L1 | 268* | 111* |
1 mg/L1 | 387* | 143* |
Treated (VFCW) | 81 | 48* |
Fluoxetine hydrochloride analysis in water. Water analysis was performed using the UPLC-MS/MS technique, which yielded a calibration curve for FLX with an R² value of 0.9948, represented by the equation y = 125547x – 4320 (Fig. S1). The water sample containing 1 mg L⁻¹ of FLX, after being treated through the WCFV system, presented a concentration below the detection limit, indicating that the fluoxetine concentration in the sample was less than 0.01 mg L⁻¹.
Discussion
The cyto/genotoxic effects of FLX in A. altiparanae were observed starting from the lowest dose, with significant changes evident from a concentration of 0.1 mg L-1. Although the toxic effects tended to increase with FLX concentration, no statistically significant differences were observed between the intermediate doses (0.01 and 0.1 mg L⁻¹). Thus, our results demonstrate the occurrence of adverse effects starting from a threshold concentration, but not a strictly dose-dependent response. However, the increase in damage related to increasing concentrations is in line with the findings of other studies conducted in several organisms and tissues, including Schmidtea mediterranea (Ofoegbu et al., 2019), Drosophila melanogaster (Öz et al., 2024), rat C6 glioma cells (Slamon et al., 2001), and hamster ovary cells (Lemos et al., 2005).
The findings of this study suggest that exposure to FLX can lead to CMC in the erythrocytes of A. altiparanae. Data analysis indicated a notable increase in the frequency of erythrocyte alterations among the group exposed to the highest concentration of FLX (1 mg L-1) when compared to the control group. A similar pattern was observed in the study by Vijitkul et al. (2022), which reported that after 96 hours of exposure to FLX, concentrations exceeding 100 μg L-1 resulted in alterations in the erythrocytes of Oreochromis niloticus.
An apparent increase in DNA damage was observed, with the highest values recorded at a concentration of 1 mg L⁻¹ of FLX, as measured by the DNA Damage Index (DI) and DNA Damage Frequency (DF). These parameters facilitated a comprehensive evaluation, revealing a significant intensity and extent of DNA damage in this group compared to those with lower concentrations of FLX. Specifically, group 2 (0.01 mg L⁻¹) exhibited up to approximately 10% of cells showing DNA damage classified as classes 1 and 2, with no cells categorized in classes 3 and 4. In contrast, group 3 (0.1 mg L⁻¹) showed up to around 10% of cells with DNA damage across all classes from 1 to 4.
Significant results were observed in the comparison between groups 4 and 5, both of which were treated at the same concentration (1 mg L-1). Notably, group 5, which underwent treatment in VFCW, achieved average outcomes comparable to those of the control group, while the untreated group displayed a substantially higher average in changes. This highlights the effectiveness of the VFWC treatment in removing FLX compounds. Previous studies have already established the efficacy of VFCW in eliminating contaminants, as evidenced by its cytotoxic effects after treatment with various xenobiotics, along with similar findings concerning textile effluent (Silva et al., 2023), herbicides (Andrade et al., 2024), and textile dyes (Silva et al., 2024). Utilizing the ULPC-MS/MS technique, our study revealed that the VFCW system achieved an impressive contaminant removal rate of 99.99% at a dosage of 1 mg L-1. This finding aligns with the research conducted by Ilyas et al. (2020), which examined 34 widely studied pharmaceuticals and reported an average removal efficiency of up to 93% in constructed wetlands.
The significant reduction of FLX in VFCW (>99%) resulted in the display of around 89% of cells without DNA damage, a significant reduction in MN, CMC, and histopathological lesions in A. altiparanae, a result comparable to the control (p > 0.05), reinforcing the efficiency of the system in mitigating the cyto/genotoxic effects of FLX. Similar high efficiencies for drugs have been reported in other CWs (Ilyas et al., 2020; Chen et al., 2016). However, previous studies indicate that removal performance may be less consistent at environmentally very low concentrations (<1 µg L⁻¹) or in the presence of complex mixtures of micropollutants, which represents an important limitation to be considered in practical applications (Matamoros, Bayona, 2006; Verlicchi, Zambello, 2014).
Similarly, the histopathological changes observed in this study were significant following exposure to FLX, particularly at the highest concentration. However, the alterations in the gills were notably less pronounced in group 5, which was treated with the VFCW system. These changes are frequently linked to adaptive responses to environmental stressors in aquatic ecosystems (Pramanik, Biswas, 2024). Similar findings were reported by Rezaeipour et al. (2024), who studied Danio rerio exposed to FLX concentrations ranging from 0.1 to 100 μg L⁻¹ over 96 h. Their observations included histopathological alterations such as epithelial hyperplasia, lamellar fusion, aneurysm formation, and lamellar clubbing. These results support our findings in A. altiparanae, emphasizing that even environmentally relevant concentrations, significantly lower than those tested here, can lead to notable gill lesions. This underscores the ecological necessity of removing FLX from aquatic systems.
While some of these changes may be reversible in the short term, prolonged exposure results in detrimental effects such as lamellae obstruction, epithelial thickening, and a reduced water contact area, as previously observed in fish exposed to antibiotic oxytetracycline and Metformin hydrochloride (Rodrigues et al., 2017; Barbieri et al., 2022). Therefore, despite the initial protective role of gill modifications, this process ultimately impairs the efficiency of gas exchange, jeopardizing tissue oxygenation and, as a result, the overall health of the organism (Kumar et al., 2019).
The vascular lesions identified in this study, including telangiectasias and aneurysms, may be linked to hemodynamic changes in the branchial lamellae due to increased blood flow, which can lead to the rupture of pillar cells and capillaries (Ahmed et al., 2013). These lesions are indicative of significant damage, potentially resulting in internal hemorrhages and respiratory dysfunction (Azadbakht et al., 2019; Hasan et al., 2022). The presence of telangiectasias further underscores the impact of FLX on the branchial microvasculature, as noted by Strzyżewska-Worotyńska et al. (2017).
Fish gills serve as the primary organs for respiration, excretion, gas exchange, and osmoregulation, making them vital indicators of population health and valuable tools for aquatic biomonitoring. In this study, the gill histology of Astyanax altiparanae exposed to FLX revealed several alterations, including lamellar fusion, epithelial hyperplasia, hypertrophy, aneurysms, and telangiectasia. Changes in lamellar structure and epithelial tissue are recognized as primary pathological indicators in fish (Thophon et al., 2003) that can reduce respiratory efficiency and may become irreversible with prolonged exposure (Hesni et al., 2011; Mauryaa et al., 2019). Additionally, vascular lesions can lead to hemorrhage and significant impairment (Ahmed et al., 2013).
Numerous studies support these findings, indicating that fish exposed to various contaminants, including pesticides, pharmaceuticals, and industrial effluents, exhibit similar patterns of gill damage, irrespective of the pollutant’s chemical nature (Kumar et al., 2016; Nowakowska et al., 2020; Ogunwole et al., 2021; Rezaeipour et al., 2024). Overall, the observed changes reflect the fish’s defense mechanisms against contaminant entry but underscore the ecological risk posed by FLX as it compromises respiratory and osmoregulatory functions in fish.
Moreover, existing literature suggests that FLX is not metabolized extensively in fish, which contributes to its bioaccumulation in various tissues, such as the liver, brain, muscles, and gills (Pan et al., 2018; Duarte et al., 2020). Yan et al. (2020) found that both FLX and its active metabolite, norfluoxetine, were present in all tissues of Carassius auratus, with concentrations increasing over the duration of exposure. Likewise, Vaclavik et al. (2022) note that the gills serve as a primary entry route for FLX into the body and are among the most structurally impacted tissues.
We emphasize that the VFCW effectively removed FLX to non-detectable levels and substantially reduced cytogenotoxic and histopathological outcomes in A. altiparanae. By reducing dissolved FLX through substrate sorption, microbial degradation, and phytoremediation, the VFCW decreases branchial exposure, which in turn mitigates epithelial and vascular lesions. These results, consistent with reports of high pharmaceutical removal in constructed wetlands (Chen et al., 2016; Ilyas et al., 2020; Dotro et al., 2021), validate previous evidence and reinforce that FLX exposure, even at environmentally relevant concentrations, can seriously compromise gill integrity and fish homeostasis. This underscores the ecological relevance of prioritizing FLX monitoring and implementing sustainable treatment technologies, such as VFCWs, to mitigate the impact of pharmaceutical effluents on aquatic systems.
Acknowledgments
We thank the support of the Programa de Pós-Graduação em Biotecnologia Ambiental da Universidade Estadual de Maringá for all support.
References
Ahmed MdK, Habibullah-Al-Mamun Md, Parvin E, Akter MS, Khan MS. Arsenic induced toxicity and histopathological changes in gill and liver tissue of freshwater fish, tilapia (Oreochromis mossambicus). Exp Toxicol Patholo. 2013; 65(6):903–09. https://doi.org/10.1016/j.etp.2013.01.003
Andrade CO, Silva JRR, Barbieri PA, Borin-Carvalho LA, Portela-Castro ALB, Fernandes CA. The effect of acute exposure of yellowtail tetra fish Astyanax lacustris (Lütken, 1875) to the glyphosate-based herbicide Templo®. Mutat Res Genet Toxicol Environ Mutagen. 2024; 897:503771. https://doi.org/10.1016/j.mrgentox.2024.503771
Azadbakht F, Shirali S, Ronagh MT, Zamani I. Assessment of gill pathological responses in yellowfin sea bream (Acanthopagrus Latus) under Aeromonas Hydrophila exposure. Arch Razi Inst. 2019; 74(1):83–89. https://doi.org/10.22092/ari.2017.114702.1139
Barbieri PA, Mari-Ribeiro IP, Lupepsa L, Gigliolli AAS, Paupitz BR, Melo RF et al. Metformin-induced alterations in gills of the freshwater fish Astyanax lacustris (Lütken, 1875) detected by histological and scanning electron microscopy. Ecotoxicology. 2022; 31(8):1205–16. https://doi.org/10.1007/s10646-022-02580-0
Behmer OA, Tolosa EMC, Neto AGF. Manual de técnicas para histologia normal e patológica. Vol. 1. 1st ed. São Paulo: EDART Livraria Editora LTDA; 1976.
Brodin T, Fick J, Jonsson M, Klaminder J. Dilute concentrations of a psychiatric drug alter behavior of fish from natural populations. Science. 2013; 339(6121):814–15. https://doi.org/10.1126/science.1226850
Brooks BW, Foran CM, Richards SM, Weston J, Turner PK, Stanley JK et al. Aquatic ecotoxicology of fluoxetine. Toxicol Lett. 2003; 142(3):169–83. https://doi.org/10.1016/S0378-4274(03)00066-3
Calisto V, Esteves VI. Psychiatric pharmaceuticals in the environment. Chemosphere. 2009; 77(10):1257–74. https://doi.org/10.1016/j.chemosphere.2009.09.021
Chen Y, Vymazal J, Březinová T, Koželuh M, Kule L, Huang J et al. Occurrence, removal and environmental risk assessment of pharmaceuticals and personal care products in rural wastewater treatment wetlands. Sci Total Environ. 2016; 566–567:1660–69. https://doi.org/10.1016/j.scitotenv.2016.06.069
Collins A, Møller P, Gajski G, Vodenková S, Abdulwahed A, Anderson D et al. 2023. Measuring DNA modifications with the comet assay: a compendium of protocols. Nat Protoc. 2023; 18(3):929–89. https://doi.org/10.1038/s41596-022-00754-y
Correia D, Domingues I, Faria M, Oliveira M. Effects of fluoxetine on fish: what do we know and where should we focus our efforts in the future? Sci Total Environ. 2023; 857:159486. https://doi.org/10.1016/j.scitotenv.2022.159486
Daughton CG, Ternes TA. Pharmaceuticals and personal care products in the environment: agents of subtle change? Environ Health Perspect. 1999; 107(suppl 6):907–38. https://doi.org/10.1289/ehp.99107s6907
Dorelle LS, Cuña RH, Sganga DE, Rey Vázquez G, López Greco L, Nostro FL. Fluoxetine exposure disrupts food intake and energy storage in the cichlid fish Cichlasoma dimerus (Teleostei, Cichliformes). Chemosphere. 2020; 238:124609. https://doi.org/10.1016/j.chemosphere.2019.124609
Dotro G, Langergraber G, Molle P, Nivala J, Puigagut J, Stein O et al. Biological wastewater treatment series – treatment wetlands. IWA Publishing; 2021. https://doi.org/10.2166/9781780408774
Duarte IA, Pais MP, Reis-Santos P, Cabral HN, Fonseca VF. Biomarker and behavioural responses of an estuarine fish following acute exposure to fluoxetine. Mar Environ Res. 2019; 147:24–31. https://doi.org/10.1016/j.marenvres.2019.04.002
Duarte IA, Reis-Santos P, Novais SC, Rato LD, Lemos MFL, Freitas A et al. Depressed, hypertense and sore: long-term effects of fluoxetine, propranolol and diclofenac exposure in a top predator fish. Sci Total Environ. 2020; 712:136564. https://doi.org/10.1016/j.scitotenv.2020.136564
Fong PP. Zebra mussel spawning is induced in low concentrations of putative serotonin reuptake inhibitors. Biol Bull. 1998; 194(2):143–49. https://doi.org/10.2307/1543044
Gomes MP, Gomes LP. Tracking the surge of psychiatric pharmaceuticals in urban rivers of Curitiba amidst and beyond the SARS-CoV-2 pandemic. Sci Total Environ. 2024; 954:176685. https://doi.org/10.1016/j.scitotenv.2024.176685
Hasan J, Ferdous SR, Rabiya SBA, Hossain MF, Hasan AM, Shahjahan M. Histopathological responses and recovery in gills and liver of Nile tilapia (Oreochromis niloticus) exposed to diesel oil. Toxicol Rep. 2022; 9:1863–68. https://doi.org/10.1016/j.toxrep.2022.10.005
Hassan I, Chowdhury SR, Prihartato PK, Razzak SA. Wastewater treatment using constructed wetland: current trends and future potential. Processes. 2021; 9(11):1917. https://doi.org/10.3390/pr9111917
Hesni MA, Savari A, Mortazavi MS, Sohrab AD. Gill histopathological changes in milkfish (Chanos chanos) exposed to acute toxicity of diesel oil. World Appl Sci J. 2011; 14(10):1487–92.
Hiemke C, Härtter S. Pharmacokinetics of selective serotonin reuptake inhibitors. Pharmacol Ther. 2000; 85(1):11–28. https://doi.org/10.1016/S0163-7258(99)00048-0
Hooftman RN, Raat WK. Induction of nuclear anomalies (micronuclei) in the peripheral blood erythrocytes of the eastern mudminnow Umbra pygmaea by ethyl methanesulphonate. Mut Res Lett. 1982; 104(1–3):147–52. https://doi.org/10.1016/0165-7992(82)90136-1
Ilyas H, Masih I, Van Hullebusch ED. Pharmaceuticals’ removal by constructed wetlands: a critical evaluation and meta-analysis on performance, risk reduction, and role of physicochemical properties on removal mechanisms. J Water Health. 2020; 18(3):253–91. https://doi.org/10.2166/wh.2020.213
Inoue LAKA, Afonso LOB, Iwama GK, Moraes G. Effects of clove oil on the stress response of matrinxã (Brycon cephalus) subjected to transport. Acta Amazon. 2005; 35(2):289–95. https://doi.org/10.1590/S0044-59672005000200018
Kaushik G, Thomas MA. The potential association of psychoactive pharmaceuticals in the environment with human neurological disorders. Sustain Chem Pharm. 2019; 13:100148. https://doi.org/10.1016/j.scp.2019.100148
Kiflay E, Selemani J, Njau K. Integrated constructed wetlands treating industrial wastewater from seed production. Water Pract Technol. 2021; 16(2):504–15. https://doi.org/10.2166/wpt.2021.008
Kumar Maurya P, Malik DS, Kumar Yadav K, Gupta N, Kumar S. Haematological and histological changes in fish Heteropneustes fossilis exposed to pesticides from industrial waste water. Hum Ecol Risk Assess. 2019; 25(5):1251–78. https://doi.org/10.1080/10807039.2018.1482736
Kumar R, Pande V, Singh L, Sharma L, Saxena N, Thakuria D et al. Pathological findings of experimental Aeromonas hydrophila infection in golden mahseer (Tor putitora). Fish Aquac J. 2016; 7(1):160. https://doi.org/10.4172/2150-3508.1000160
Kwon J-W, Armbrust KL. Laboratory persistence and fate of fluoxetine in aquatic environments. Environ Toxicol Chem. 2006; 25(10):2561–68. https://doi.org/10.1897/05-613R.1
Lemos NG, Mantovani MS, Vicentini VEP. Avaliação do efeito genotóxico do Prozac (fluoxetina), sem e com adição de vitaminas A e C, através do teste do cometa em cultura de células CHO-K1. Semin Ciênc Biol Saúde. 2005; 26(2):95. https://doi.org/10.5433/1679-0367.2005v26n2p95
Matamoros V, Bayona JM. Elimination of pharmaceuticals and personal care products in subsurface flow constructed wetlands. Environ Sci Technol. 2006; 40(18):5811–16. https://doi.org/10.1021/es0607741
Monteiro DA, Rantin FT, Kalinin AL. Inorganic mercury exposure: toxicological effects, oxidative stress biomarkers and bioaccumulation in the tropical freshwater fish matrinxã, Brycon amazonicus (Spix and Agassiz, 1829). Ecotoxicology. 2010; 19(1):105–23. https://doi.org/10.1007/s10646-009-0395-1
Maurya PK, Malik DS, Yadav KK, Kumar A, Kumar S, Kamyab H. Bioaccumulation and potential sources of heavy metal contamination in fish species in River Ganga basin: possible human health risks evaluation. Toxicol Report. 2019; 6(1):472–81. https://doi.org/10.1016/j.toxrep.2019.05.012
Nałęcz-Jawecki G, Wawryniuk M, Giebułtowicz J, Olkowski A, Drobniewska A. Influence of selected antidepressants on the ciliated protozoan Spirostomum ambiguum: toxicity, bioaccumulation, and biotransformation products. Molecules. 2020; 25(7):1476. https://doi.org/10.3390/molecules25071476
Nowakowska K, Giebułtowicz J, Kamaszewski M, Adamski A, Szudrowicz H, Ostaszewska T et al. Acute exposure of zebrafish (Danio rerio) larvae to environmental concentrations of selected antidepressants: bioaccumulation, physiological and histological changes. CBPC. 2020; 229:108670. https://doi.org/10.1016/j.cbpc.2019.108670
Ofoegbu PU, Lourenço J, Mendo S, Soares AMVM, Pestana JLT. Effects of low concentrations of psychiatric drugs (carbamazepine and fluoxetine) on the freshwater planarian, Schmidtea mediterranea. Chemosphere. 2019; 217:542–49. https://doi.org/10.1016/j.chemosphere.2018.10.198
Ogunwole GA, Saliu JK, Osuala FI, Odunjo FO. Chronic levels of ibuprofen induces haematoxic and histopathology damage in the gills, liver, and kidney of the African sharptooth catfish (Clarias gariepinus). Environ Sci Poll Res. 2021; 28(20):25603–13. https://doi.org/10.1007/s11356-020-12286-7
Orozco-Hernández JM, Gómez-Oliván LM, Elizalde-Velázquez GA, Rosales-Pérez KE, Cardoso-Vera JD, Heredia-García G et al. Fluoxetine-induced neurotoxicity at environmentally relevant concentrations in adult zebrafish Danio rerio. Neurotoxicology. 2022; 90:121–29. https://doi.org/10.1016/j.neuro.2022.03.007
Ostrensky A, Pedrazzani AS, Vicente AL. Use of MS-222 (tricaine methanesulfonate) and propofol (2,6-diisopropylphenol) as anaesthetics for the tetra Astyanax altiparanae (Teleostei, Characidae). Aquac Res. 2016; 47(11):3477–88. https://doi.org/10.1111/are.12797
Öz S, Sarıkaya ZN, Larçın Ö, Sarıkaya R. Investigation of the genotoxic effect of fluoxetine hydrochloride in Drosophila melanogaster. KSU Tarım ve Doğa Dergisi. 2024; 27(2):316–24. https://doi.org/10.18016/ksutarimdoga.vi.1310729
Pan C, Yang M, Xu H, Xu B, Jiang L, Wu M. Tissue bioconcentration and effects of fluoxetine in zebrafish (Danio rerio) and red crucian cap (Carassius auratus) after short-term and long-term exposure. Chemosphere. 2018; 205:8–14. https://doi.org/10.1016/j.chemosphere.2018.04.082
Pan C, Zhu F, Wu M, Jiang L, Zhao X, Yang M. Degradation and toxicity of the antidepressant fluoxetine in an aqueous system by UV irradiation. Chemosphere. 2022; 287(4):132434. https://doi.org/10.1016/j.chemosphere.2021.132434
Pramanik S, Biswas JK. Histopathological fingerprints and biochemical changes as multi-stress biomarkers in fish confronting concurrent pollution and parasitization. IScience. 2024; 27(12):111432. https://doi.org/10.1016/j.isci.2024.111432
Reis EO, Foureaux AFS, Rodrigues JS, Moreira VR, Lebron YAR, Santos LVS et al. Occurrence, removal and seasonal variation of pharmaceuticals in Brasilian drinking water treatment plants. Environ Poll. 2019; 250:773–81. https://doi.org/10.1016/j.envpol.2019.04.102
Rezaeipour A, Kazempoor R, Mortazavi P, Ghorbanzadeh A. Histopathological alterations in adult zebrafish (Danio rerio) under acute fluoxetine exposure. INJVR. 2024; 4(2):21–33.
Rodrigues S, Antunes SC, Nunes B, Correia AT. Histological alterations in gills and liver of rainbow trout (Oncorhynchus mykiss) after exposure to the antibiotic oxytetracycline. Environ Toxicol Pharmacol. 2017; 53:164–76. https://doi.org/10.1016/j.etap.2017.05.012
Schulz UH, Martins-Junior H. Astyanax fasciatus as bioindicator of water pollution of Rio dos Sinos, RS, Brazil. Braz J Biol. 2001; 61(4):615–22. https://doi.org/10.1590/S1519-69842001000400010
Schwaiger J, Wanke R, Adam S, Pawert M, Honnen W, Triebskorn R. The use of histopathological indicators to evaluate contaminant-related stress in fish. Vol. 6. Kluwer Academic Publishers; 1997.
Sezerino PH, Rousso BZ, Pelissari C, Santos MO, Freitas MN, Fechine VY et al. Wetlands construídos aplicados no tratamento de esgoto sanitário: recomendações para implantação e boas práticas de operação e manutenção. Tubarão: Fundação Nacional da Saúde – Funasa; 2018.
Silva JRR, Andrade CO, Ribeiro AC, Macruz PD, Bergamasco R, Fernandes CA. Cyto and genotoxicity induced by acute exposure to Novacron® Bold Deep Navy dye on Astyanax lacustris can be reduced after treatment through a vertical flow constructed wetland system. Environ Sci Poll Res. 2024; 31(59):66630–42. https://doi.org/10.1007/s11356-024-35687-4
Silva JRR, Gregorio A, Portela-Castro ALB, Fernandes CA. Genotoxicity and cytotoxicity of textile production effluents, before and after Bacillus subitilis bioremediation, in Astyanax lacustris (Pisces, Characidae). Mut Res Genet Toxicol Environ Mutagen. 2023; 886:503588. https://doi.org/10.1016/j.mrgentox.2023.503588
Slamon D, Ward T, Butler J, Pentreath V. Assessment of DNA damage in C6 glioma cells after antidepressant treatment using an alkaline comet assay. Arch Toxicol. 2001; 75(4):243–50. https://doi.org/10.1007/s002040100228
Speit G, Hartmann A. The comet assay (Single-Cell Gel Test): a sensitive genotoxicity test for the detection of DNA damage and repair. DNA Repair Protocols. New Jersey: Humana Press; 1999. p.203–12.
Strzyżewska-Worotyńska E, Szarek J, Babińska I, Gulda D. Gills as morphological biomarkers in extensive and intensive rainbow trout (Oncorhynchus mykiss, Walbaum 1792) production technologies. Environ Monit Assess. 2017; 189(12):611. https://doi.org/10.1007/s10661-017-6278-7
Svobodová Z, Vykusová B. Diagnostics, prevention and therapy of fish diseases and intoxications: manual for international training course on fresh-water fish diseases and intoxications: diagnostics, prophylaxis and therapy. Vol. 1. Vodnany, Czechoslovakia: Institute of Fish Culture and Hydrobiology; 1991.
Tincani FH, Santos GS, Azevedo ACB, Marques AEML, Pereira LS, Castellano GC et al. Climbing the taxonomic ladder: Could a genus be used as bioindicator? The ecotoxicological relationship between biomarkers of Astyanax altiparanae, Astyanax bifasciatus and Astyanax ribeirae. Ecol Indic. 2019; 106:105474. https://doi.org/10.1016/j.ecolind.2019.105474
Thophon S, Kruatrachue M, Upatham ES, Pokethitiyook P, Sahaphong S, Jaritkhuan S. Histopathological alterations of white seabass, Lates calcarifer, in acute and subchronic cadmium exposure. Environ Pollut. 2003; 121(3):307–20. https://doi.org/10.1016/S0269-7491(02)00270-1
Togunde OP, Oakes KD, Servos MR, Pawliszyn J. Determination of pharmaceutical residues in fish bile by solid-phase microextraction couple with liquid chromatography-tandem mass spectrometry (LC/MS/MS). Environ Sci Technol. 2012; 46(10):5302–09. https://doi.org/10.1021/es203758n
Vaclavik J, Sehonova P, Blahova J, Medkova D, Postulkova E, Maly O et al. Foodborne fluoxetine impacts the immune response in rainbow trout (Oncorhynchus mykkis). Environ Toxicol Pharmacol. 2022; 90:103818. https://doi.org/10.1016/j.etap.2022.103818
Verlicchi P, Zambello E. How efficient are constructed wetlands in removing pharmaceuticals from untreated and treated urban wastewaters? Sci Total Environ. 2014; 470–71:1281–306. https://doi.org/10.1016/j.scitotenv.2013.10.085
Vijitkul P, Kongsema M, Toommakorn T, Bullangpoti V. Investigation of genotoxicity, mutagenicity, and cytotoxicity in erythrocytes of Nile tilapia (Oreochromis niloticus) after fluoxetine exposure. Toxicol Rep. 2022; 9:588–96. https://doi.org/10.1016/j.toxrep.2022.03.031
Yamindago A, Lee Nayun, Lee Nayoung, Jo Y, Woo S, Yum S. Fluoxetine in the environment may interfere with the neurotransmission or endocrine systems of aquatic animals. Ecotoxicol Environ Saf. 2021; 227:112931. https://doi.org/10.1016/j.ecoenv.2021.112931
Yan Z, Zhang X, Bao X, Ling X, Yang H, Liu J et al. Influence of dissolved organic matter on the accumulation, metabolite production and multi-biological effects of environmentally relevant fluoxetine in crucian carp (Carassius auratus). Aquat Toxicol. 2020; 226:105581. https://doi.org/10.1016/j.aquatox.2020.105581
Authors
![]() Josiane Rodrigues Rocha da Silva1
Josiane Rodrigues Rocha da Silva1 ![]() ,
, ![]() Camila Oliveira de Andrade1,
Camila Oliveira de Andrade1, ![]() Patrícia Daniele Silva dos Santos2,
Patrícia Daniele Silva dos Santos2, ![]() Oscar Oliveira Santos2,
Oscar Oliveira Santos2, ![]() Roney Peterson Pereira3 and
Roney Peterson Pereira3 and ![]() Carlos Alexandre Fernandes1,4
Carlos Alexandre Fernandes1,4
[1] Programa de Pós-Graduação em Biotecnologia Ambiental, Departamento de Biotecnologia, Genética e Biologia Celular, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil. (JRRS) josianerrs@gmail.com (corresponding author), (COA) camilamedvet2019@gmail.com, (CAF) cafernandes@uem.br.
[2] Departamento de Química, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil. (PDSS) patriciadanieless@hotmail.com, (OOS) oosjunior@uem.br.
[3] Departamento de Meio Ambiente e Tecnologia, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil. (RPP) roney_peterson@hotmail.com.
[4] Núcleo de Pesquisa em Limnologia, Ictiologia e Aquicultura (NUPELIA), Centro de Ciências Biológicas, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil.
Authors’ Contribution 

Josiane Rodrigues Rocha da Silva: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Writing-original draft.
Camila Oliveira de Andrade: Visualization, Writing-original draft, Writing-review and editing.
Patrícia Daniele Silva dos Santos: Formal analysis, Investigation, Methodology, Software.
Oscar Oliveira Santos: Formal analysis, Investigation, Methodology.
Roney Peterson Pereira: Formal analysis, Software, Writing-review and editing.
Carlos Alexandre Fernandes: Project administration, Resources, Supervision, Validation, Visualization, Writing-review and editing.
Ethical Statement
All procedures involving the use of animals in research were approved by the Animal Ethics Committee (CEUA – UEM), under license number 3359040723.
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
The authors were funded by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001.
Supplementary Material
Supplementary material S1
How to cite this article
Silva JRR, Andrade CO, Santos PDS, Santos OO, Pereira RP, Fernandes CA. Reduced fluoxetine-induced cyto- and genotoxicity in yellowtail tetra fish Astyanax altiparanae (Characiformes: Acestrorhamphidae) after treatment through a constructed wetlands system. Neotrop Ichthyol. 2026; 24(1):e250111. https://doi.org/10.1590/1982-0224-2025-0111
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted October 27, 2025
Accepted October 27, 2025
![]() Submitted June 26, 2025
Submitted June 26, 2025
![]() Epub March 16, 2026
Epub March 16, 2026