![]() Jean Carlos Dantas de Oliveira1
Jean Carlos Dantas de Oliveira1 ![]() ,
, ![]() Jônnata Fernandes de Oliveira2,
Jônnata Fernandes de Oliveira2, ![]() Alexandre de Oliveira Marques3,
Alexandre de Oliveira Marques3, ![]() Danielle Peretti1,
Danielle Peretti1, ![]() Rodrigo S. da Costa Goldbaum4 and
Rodrigo S. da Costa Goldbaum4 and ![]() José Luís Costa Novaes4
José Luís Costa Novaes4
PDF: Download Here | Supplementary: S1 | Cite this article
Associate Editor: ![]() Paulo Pompeu
Paulo Pompeu
Section Editor: ![]() Fernando Pelicice
Fernando Pelicice
Editor-in-chief: ![]() Carla Pavanelli
Carla Pavanelli
Abstract
Investigamos os padrões espaciais de distribuição e abundância de peixes nativos (migradores e sedentários) e não nativos em um sistema de reservatórios em cascata localizado no semiárido brasileiro. Nossa hipótese era de que espécies sedentárias e não nativas dominariam os reservatórios do sistema em termos de abundância. Além disso, esperávamos observar um aumento na abundância de espécies, acompanhado por mudanças na composição das assembleias ao longo da cascata. A amostragem foi realizada com redes de espera em quatro reservatórios distribuídos ao longo do eixo longitudinal do rio Apodi-Mossoró. Os dados foram analisados por meio de ordenação não métrica (MDS) e Modelos Aditivos Generalizados para Localização, Escala e Forma (GAMLSS). No total, foram capturados 13.787 espécimes pertencentes a 22 espécies. A abundância média das espécies (CPUEn) apresentou um aumento em direção ao reservatório localizado no médio curso do rio, seguido de uma redução acentuada no reservatório do baixo curso. A hipótese do estudo foi confirmada uma vez que as espécies sedentárias apresentaram maior abundância ao longo do sistema em cascata, enquanto a abundância de espécies não nativas superou os migratórios. A variação espacial observada nas assembleias de peixes do rio Apodi-Mossoró parece estar relacionada a múltiplos fatores, especialmente ao tamanho dos reservatórios e à sua posição na cascata.
Palavras-chave: Bacia hidrográfica, Conservação, Espécies não nativas, Migração, Rio intermitente.
Introduction
The hydrographic basins of the semiarid region of Brazil exhibit distinctive features, with intermittent river regimes prevailing (Nascimento, 2014; Felix, Paz, 2016). The phenomenon of intermittent rivers arises from the short duration of the rainy season, low annual rainfall indices, irregular precipitation patterns, and high evaporation rates (Marques et al., 2020). Given these issues, reservoirs have been constructed along river courses to store water to meet diverse human needs (Novais et al., 2022), including water supply, irrigation, agriculture, and industrial activities. However, these constructions are not without impacts on the functioning of local ecosystems and biodiversity.
Dams act as barriers, obstructing the natural movement of fish and exerting direct influence on their distribution and abundance (Zacardi et al., 2021). This disruption in movement patterns can precipitate declines in population sizes, particularly impacting migratory species (Santos et al., 2018). Paradoxically, while these barriers pose challenges to native migratory species, they create favorable conditions for native sedentary species and non-native species, often leading to their proliferation (Jo et al., 2019). The presence of dams within river systems can significantly alter the composition and dynamics of fish communities, particularly in cascade reservoirs. This is because fluvial connectivity is essential for the dynamics of aquatic systems, encompassing interactions across multiple scales and dimensions, including longitudinal, lateral, vertical, and temporal (Fullerton et al., 2010).
Although the impacts of dams on large river systems such as altered hydrological dynamics and reduced migratory fish populations are well documented (Foubert et al., 2018), there is a gap in understanding these effects in smaller river basins, particularly in the semiarid region of Brazil. The fragmentation of aquatic ecosystems due to dam construction is a growing global concern, causing significant alterations in rivers and fish assemblages (Terêncio et al., 2021). However, the specific impact of this fragmentation on reservoir cascade systems along small rivers in the semiarid region of Brazil remains largely unexplored (Oliveira et al., 2021).
In Brazil, dam construction has played a crucial role in modulating the distribution and abundance of fish in reservoir cascade systems. For example, in the Paranapanema River, one of the main tributaries of the upper Paraná River basin, studies have revealed a prevalence of sedentary fish species, a low abundance of migratory species, and a high number of non-native species (Pelicice et al., 2018). Similarly, at the international scale, dams in the Columbia River basin in the United States have had negative impacts on migratory fish populations (Leonard et al., 2015), and reservoirs associated with hydropower plants in Serbia have influenced the distribution of fish, particularly sedentary species (Simonović et al., 2021).
The central question guiding our research was as follows: what is the distribution and abundance of fish in a cascade reservoir system of a small river in the semiarid region of Brazil? We hypothesize that sedentary and non-native fish species will dominate the reservoirs of the cascade system in terms of abundance. Additionally, we expect to observe an increase in species abundance, accompanied by changes in species composition throughout the cascade. In this study, data on the distribution and abundance of fish in the reservoir cascade system along the Apodi-Mossoró River, which is situated in the semiarid region of Brazil, were collected. Our primary objective was to analyze the fish communities present in these reservoirs and explore the spatial.
Material and methods
Study region. The study was conducted in the Atlantic Northeast Oriental Hydrographic Region (Lima et al., 2022), which is located in the Brazilian semiarid region and is characterized by medium-sized to small river basins. This hydrographic area is classified as medium-sized, with a perennial reach (Miranda, Meirelles Filho, 2016), covering 14,276 km². Its extent is impacted by the presence of approximately 618 reservoirs (IGARN, 2019). The Apodi-Mossoró River is one of the main watercourses of the Rio Grande do Norte State, exhibiting a predominantly intermittent regime typical of the Brazilian semiarid region. Its flow is directly dependent on rainfall, leading to periods of highdischarge during the rainy season interspersed with prolonged droughts, during which manysections experience reduced flow or dry up completely. The climate is semiarid, with annualprecipitation ranging between 400 and 800 mm (Costa et al., 2021) that is concentrated betweenFebruary and May. The vegetation consists of arboreal and shrubby Caatinga biome, characterized bythorny plants and cacti. Currently, this watershed supports the regional economy, which is driven byoil extraction, agriculture and irrigated fruit farming, extensive livestock farming, and limestone mining (Carvalho et al., 2011).
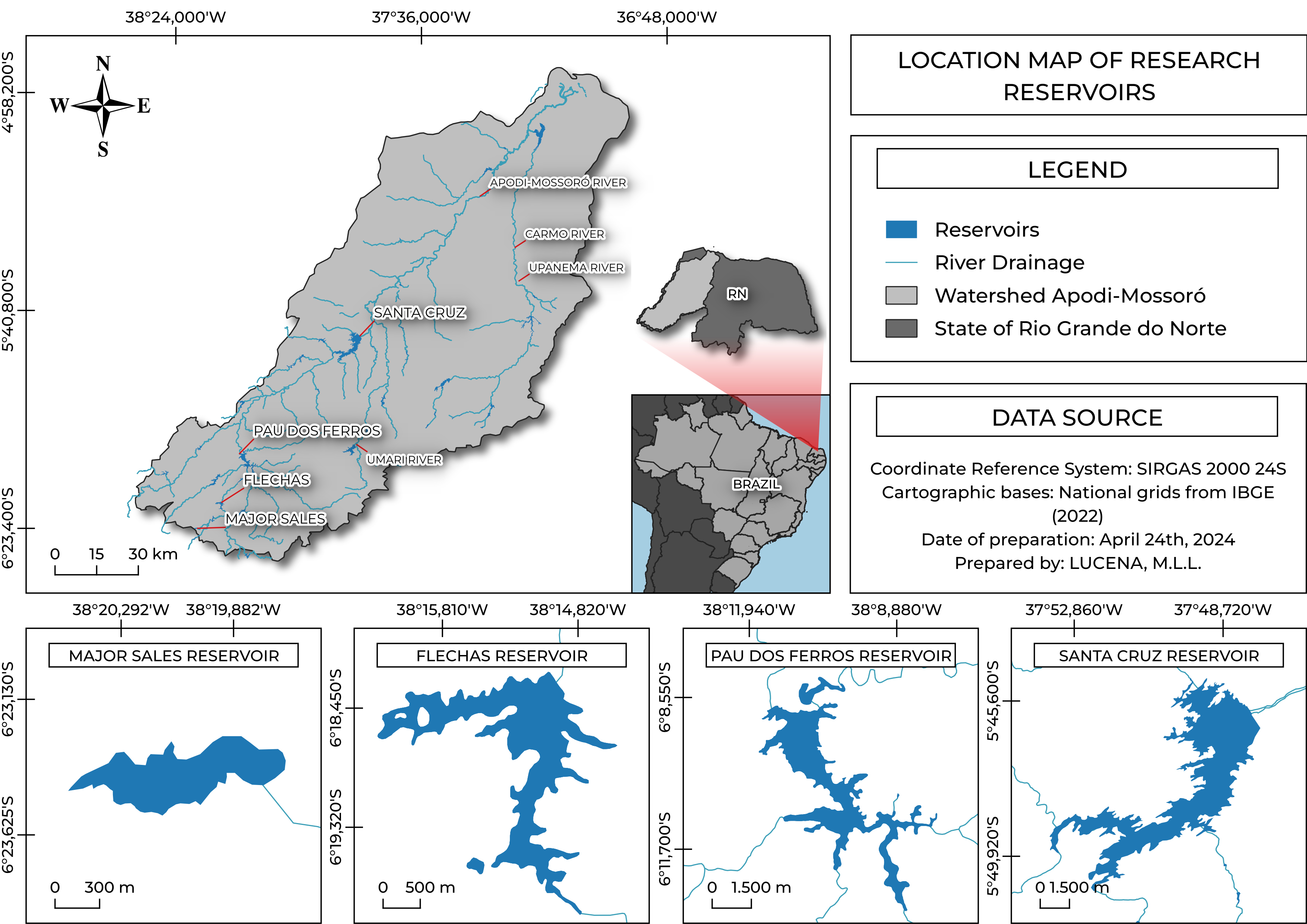
Sampling. Sampling was conducted quarterly in February, May, August, and November. Collections were carried out in 2011 and 2012 in Pau dos Ferros Reservoir (PF), as funding was available specifically for that period, and in Major Sales (MS), Flechas (FL), and Santa Cruz (SC) Reservoirs in 2015 and 2016 (Fig. 1; Tab. 1).
FIGURE 1| Reservoir cascade system along the Apodi-Mossoró River in the semiarid region of Rio Grande do Norte State, Brazil.
TABLE 1 | Reservoirs forming the cascade system along the main channel of the Apodi-Mossoró River, Rio Grande do Norte State, Brazil. Information about the reservoirs: river course region, coordinates, conclusion year, area (km²), volume (m³), sampling period, number of sampling sites and Sampled areas.
General information | Major Sales | Flechas | Pau dos Ferros | Santa Cruz |
Position in the watershed | Upper | Upper | Medium | Lower |
Coordinates |
06º23’24”S
|
06º18’18”S
|
06°08’48”S
|
05°45’45”S
|
Started of operation | 1985 | 1983 | 1967 | 2002 |
Area (km²) | 0.06 | 2.59 | 11.65 | 34.13 |
Maximum volume (m³) | 2,641,850 | 8,949,675 | 50,846,000 | 599,712,000 |
Sampling period | 2015–2016 | 2015–2016 | 2011–2012 | 2015–2016 |
Number of samplings sites | 2 | 2 | 4 | 8 |
Sampled areas | Near the dam, transition areas between reservoir and river | Near the dam, transition areas between reservoir and river | Near the dam, transition areas between reservoir and river | Near the dam, transition areas between reservoir and river |
Standardized procedures were implemented at each sampling site within the reservoirs (Tab. 1), utilizing a set of eleven mesh gillnets with sizes ranging from 12 to 70 mm (measured between adjacent knots). Each gillnet measured 15 m in length and had a height varying between 1.8 and 2.0 m.
The gillnets were positioned parallel to the reservoir shores, prioritizing areas with greater depth. The gillnets were exposed for 12 hours, spanning from 5:00 p.m. to 5:00 a.m. the following day, with two fish removal operations per sampling site at 10:00 p.m. and 5:00 a.m. The captured specimens were carefully placed in plastic bags, preserved on ice, and transported to the Fish Ecology and Continental Fisheries Laboratory at UFERSA. Species identification was subsequently confirmed by taxonomists from the Universidade Federal da Paraíba (UFPB), where specimens were deposited in the ichthyology collection (voucher numbers UFPB 8934–8986 in Tab. S1).
Data analysis. To evaluate the relative abundance of fish species in behavioral groups and reservoirs and the interactions between these factors, we computed the catch per unit effort in numbers (CPUEn = C/f), where ‘C’ represents the catch in numbers and ‘f’ is the effort, which is defined as a²/t, wher ‘a’ is the net area (301.8 m²) and ‘t’ is the exposure time of the fishing nets (12 h) (Orsi et al., 2014). CPUEn data were standardized to capture number per 1,000 m² over 24 h. A rarefaction curve by reservoir was generated using the iNext package (Hsieh et al., 2022) to assess sampling sufficiency.
The assemblages of the reservoirs were compared using a permutational multivariate analysis of variance (PERMANOVA) followed by visualization using nonmetric multidimensional scaling (NMDS). Prior to these analyses, the biotic data matrix was standardized using the Hellinger method. The PERMANOVA procedure was conducted with 9,999 permutations, and the p values were adjusted using the Bonferroni correction. Data standardization was performed using the “decostand” function of the “vegan” package (Oksanen et al., 2022), a step recommended as a preliminary treatment for linear tests (Legendre, Legendre, 2012).
The relative abundance of the fish species (CPUEn) was calculated in relation to spatial variables (reservoirs), behavioral groups, and their interaction using Generalized Additive Models for Location, Scale, and Shape (GAMLSS). In preparation for statistical analysis, the abundance data were transformed by squaring the values. We subsequently utilized the “fitDist” function from the “gamlss” package (Rigby, Stasinopoulos, 2005) to determine the distribution of the response variable (CPUE²), which was modeled as a reverse Gumbel distribution. Model selection was carried out using the “stepGAICAll.A” function from the “gamlss” package, with selection based on the generalized Akaike information criterion (GAIC), choosing the final model with the lowest value (Rigby, Stasinopoulos, 2005). We conducted pairwise post hoc analysis of each factor using the “emmeans” function (Lenth, 2022). The resulting statistical model underwent visual validation through worm plots and inspection of the residual distribution. All analyses were performed using R software (R Development Core Team, 2021).
The data analysis was performed using GAMLSS, a semiparametric univariate regression suitable for a wide range of potential distributions of the response variable (Stasinopoulos et al., 2018). We used the GAMLSS model, which includes multiple distribution parameters, such as the mean, variance, skewness, and kurtosis, providing a better fit for complex data (Rigby, Stasinopoulos, 2005). The use of multiple coefficients reflects the ability of GAMLSS to separately capture the effects of individual variables on various aspects of the distribution (Rigby et al., 2019). This flexibility enables the fitting of models that are better suited to complex data, where the assumption of homoscedasticity or symmetric distribution may not be realistic.
Results
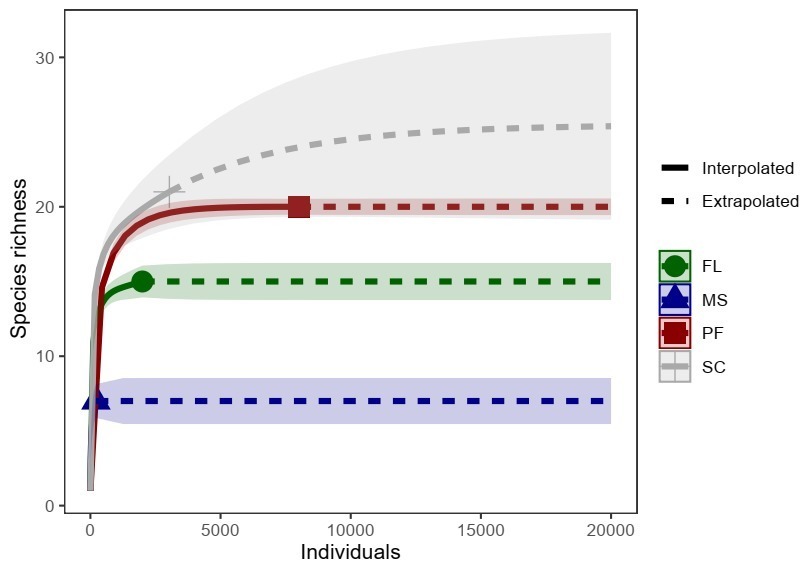
During the study, a total of 13,787 individuals fish representing 22 species, were collected from four reservoirs in the semiarid region of northeastern Brazil. The rarefaction curve of the reservoir tended to stabilize, demonstrating sufficiency sampling of the fish assemblage at each site (Fig. 2).
FIGURE 2| Species accumulation curve based on sampling effort in the cascade reservoir system along the Apodi-Mossoró River, semiarid region, Rio Grande do Norte State. Major Sales (MS), Flechas (FL), Pau dos Ferros (PF), and Santa Cruz (SC).
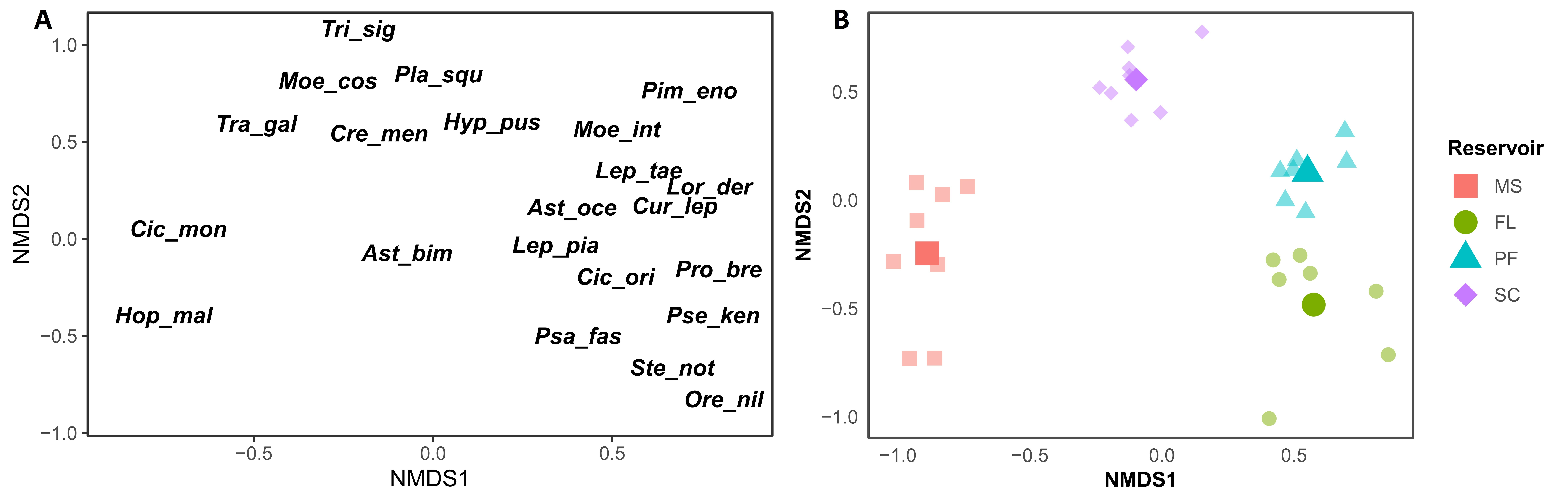
The most abundant species was Astyanax aff. bimaculatus (Linnaeus, 1758), a native sedentary species found in all sampled reservoirs. Additionally, five other species were present in all reservoirs: Psalidodon fasciatus (Cuvier, 1819), Saxatilia brasiliensis (Bloch, 1792), Hoplias aff. malabaricus (Bloch, 1794), Hypostomus pusarum (Starks, 1913), and Leporinus piau Fowler, 1941. The highest fish abundance was recorded in the upper (FL) and middle (PF) reservoirs, where sedentary species predominated. Migratory species were also more abundant in these reservoirs, whereas nonnative species were more prevalent in the lower reservoir (SC). Therefore, the composition of the fish assemblage differed among reservoirs (PERMANOVA; F = 21.992; R² = 71.73%; p < 0.0001; Fig. 3; Tab. 2).
FIGURE 3| Representation of the NMDS ordination showing the variation in fish species along the Apodi-Mossoró River reservoir cascade system, Rio Grande do Norte State. Reservoir positions: upper section, Major Sales (MS) and Flechas (FL); middle section, Pau dos Ferros (PF); and lower section, Santa Cruz (SC). A. Distribution of species along the first two NMDS axes. B. Clustering of samples according to the analyzed reservoirs.
TABLE 2 | Taxonomic classification and CPUE (standardized for catch numbers per 1000 m² and 24 hours) for sedentary, non-native, and migratory species in the reservoir cascade system along the Apodi-Mossoró River, semiarid region, Rio Grande do Norte State, Brazil. The bold numbers indicate the most abundant species in the Apodi-Mossoró River reservoir cascade system.
Group | Species | Major Sales | Flechas | Pau dos Ferros | Santa Cruz |
Sedentary (SEDNA) | CHARACIFORMES |
|
|
|
|
Acestrorhamphidae |
|
|
|
| |
Astyanax aff. bimaculatus (Linnaeus, 1758) | 42.13 | 16.17 | 35.33 | 14.67 | |
Psalidodon fasciatus (Cuvier, 1819) | 0.46 | 9.03 | 3.06 | 0.16 | |
Moenkhausia costae (Steindachner, 1907) |
|
| 0.12 | 20.46 | |
Moenkhausia cf. intermedia Eigenmann, 1908 |
| 0.34 | 14.54 | 6.95 | |
Ctenobrycon kennedyi (Eigenmann, 1903) |
| 1.21 | 0.29 | 0.03 | |
Anostomidae |
|
|
|
| |
Leporinus piau Fowler, 1941 | 1.39 | 3.86 | 6.94 | 0.84 | |
Curimatidae |
|
|
|
| |
Curimatella lepidura (Eigenmann & Eigenmann, 1889) |
| 14.38 | 26.51 | 7.49 | |
Steindachnerina notonota (Miranda-Ribeiro, 1937) |
| 43.48 | 0.24 |
| |
Erythrinidae |
|
|
|
| |
Hoplias aff. malabaricus (Bloch, 1794) | 44.44 | 2.99 | 1.52 | 4.79 | |
Triportheidae |
|
|
| 8.43 | |
Triportheus signatus (Garman, 1890) |
|
|
|
| |
SILURIFORMES |
|
|
| 8.43 | |
Auchenipteridae |
|
|
|
| |
Trachelyopterus galeatus (Linnaeus, 1766) | 2.31 |
| 0.07 | 8.33 | |
Heptapteridae |
|
|
|
| |
Pimelodella enochi Fowler, 1941 |
|
| 0.04 | 0.03 | |
Loricariidae |
|
|
|
| |
Hypostomus pusarum (Starks, 1913) | 1.39 | 0.24 | 0.32 | 6.92 | |
Loricariichthys derbyi Fowler, 1915 |
|
| 5.25 | 0.06 | |
CICHLIFORMES |
|
|
|
| |
Cichlidae |
|
|
|
| |
Cichlasoma orientale Kullander, 1983 |
| 1.69 | 0.40 | 0.39 | |
Saxatilia brasiliensis (Bloch, 1792) | 0.93 | 0.29 | 0.09 | 2.96 | |
Migratory (MIG) | CHARACIFORMES |
|
|
|
|
Anostomidae |
|
|
|
| |
Leporinus taeniatus Lütken, 1875 |
| 0.72 | 2.34 | 0.29 | |
Prochilodontidae |
|
|
|
| |
Prochilodus brevis Steindachner, 1875 |
| 4.01 | 2.50 | 0.77 | |
Non-native (NNSSED) | CICHLIFORMES |
|
|
|
|
Cichlidae |
|
|
|
| |
Astronotus ocellatus (Agassiz, 1831) |
| 0.05 |
| 0.03 | |
Oreochromis niloticus (Linnaeus, 1758) |
| 1.54 | 0.07 | 0.26 | |
Cichla kelberi Kullander & Ferreira, 2006 | 6.94 |
| 0.12 | 0.93 | |
ACANTHURIFORMES |
|
|
|
| |
Sciaenidae |
|
|
|
| |
Plagioscion squamosissimus (Heckel, 1840) |
|
| 0.24 | 15.21 | |
Total number of species per reservoir | 8 | 15 | 20 | 21 | |
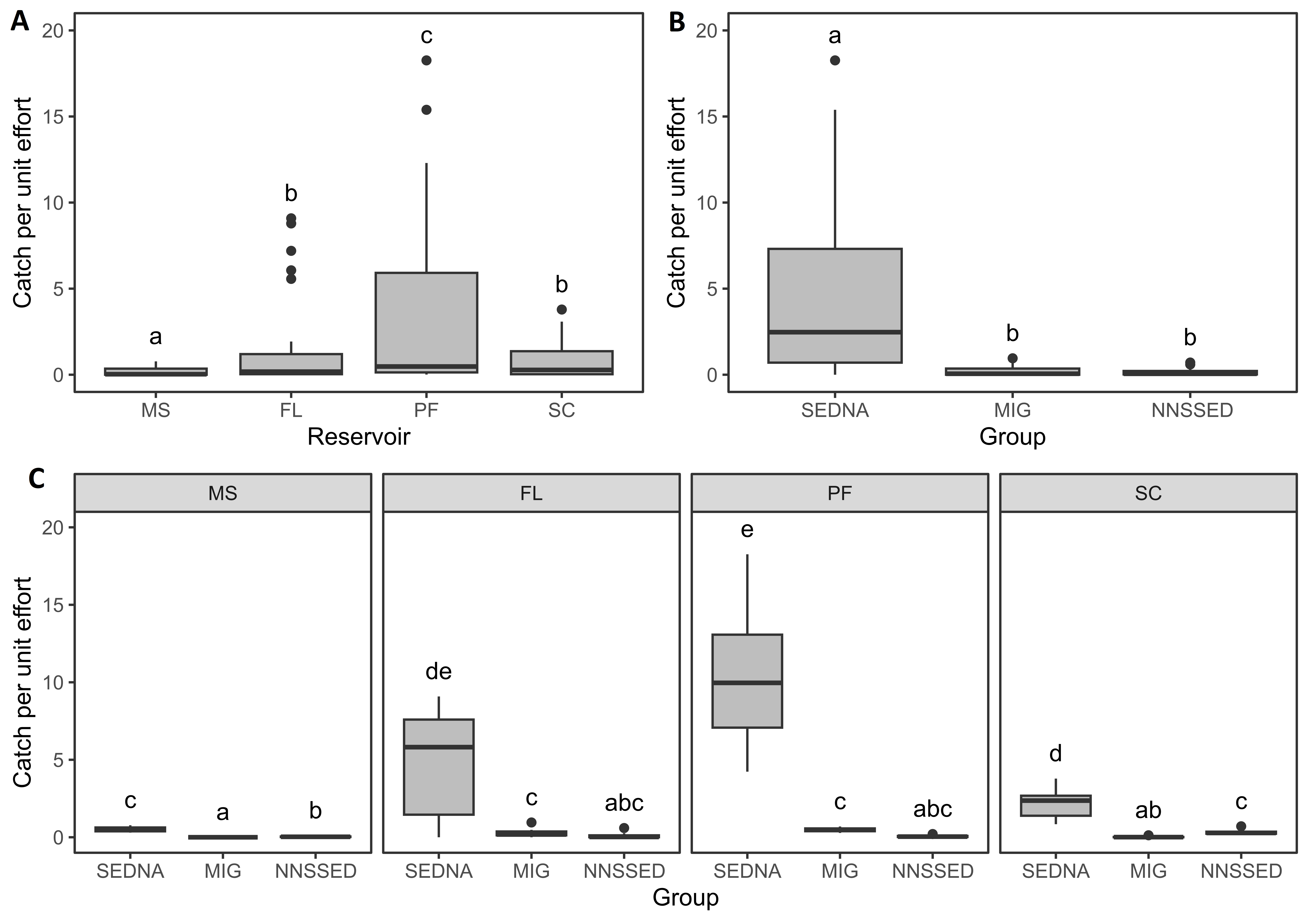
The average fish abundance (CPUEn) increased in the reservoirs from the upper to the middle course of the river (PF), followed by a sharp decline in the lower course reservoir (SC). The PF reservoir exhibited the highest total fish abundances, with mean CPUE value of 3.64. In contrast, the FL and SC reservoirs recorded significantly intermediate abundances, with values of 1.74 and 0.85, respectively. The MS reservoir presented the lowest CPUEn, with values of 0.13 (GAMLSS; p < 0.05; Tab. 3; Fig. 4A).
TABLE 3 | Generalized Additive Models for Location, Shape, and Scale (GAMLSS) results with the best-fit model for comparing fish abundance in three groups (Sedentary, Migratory, and Nonnative) across four reservoirs (Major Sales, Flechas, Pau dos Ferros, and Santa Cruz) within the main channel of the Apodi-Mossoró River in northeastern Brazil. Legend: R² – coefficient of determination; AIC – Akaike information criterion; Pr (>|t|) – significance level; df – degree of freedom; MIG – migratory; NNA – nonnative; FL – Flechas; PF – Pau dos Ferros; SC – Santa Cruz. Bold values indicate statistical significance of the variable (α < 0.05).
Model: CPUEn ~ Group + Reservoir + Group: Reservoir, sigma.formula =~ Group + Reservoir, Family = Normal, AIC = 23.251, R² = 99.55% | ||||
Mu coef. | Estimate | Std. Error | t value | Pr(>|t|) |
Intercept | 0.5144 | 0.0673 | 7.641 | 4.65e-11 |
Group (MIG) | -0.5144 | 0.0673 | -7.634 | 4.79e-11 |
Group (NNA) | -0.4771 | 0.0677 | -7.039 | 6.65e-10 |
Reservoir (FL) | 4.3190 | 1.5786 | 2.736 | 0.0077 |
Reservoir (PF) | 9.8726 | 1.1440 | 8.630 | 5.65e-13 |
Reservoir (SC) | 1.7033 | 0.3985 | 4.274 | 5.37e-05 |
Group (MIG): Reservoir (FL) | -4.0258 | 1.5801 | -2.548 | 0.0128 |
Group (NNA): Reservoir (FL) | -4.2345 | 1.5896 | -2.664 | 0.0093 |
Group (MIG): Reservoir (PF) | -9.3928 | 1.1450 | -8.203 | 3.81e-12 |
Group (NNA): Reservoir (PF) | -9.8411 | 1.1519 | -8.543 | 8.32e-13 |
Group (MIG): Reservoir (SC) | -1.6791 | 0.3988 | -4.210 | 6.78e-05 |
Group (NNA): Reservoir (SC) | -1.4189 | 0.4012 | -3.536 | 0.0006 |
Sigma coef. |
|
|
|
|
Intercept | -1.6586 | 0.2170 | -7.642 | 4.63e-11 |
Group (MIG) | -3.1540 | 0.1971 | -16.003 | <2e-16 |
Group (NNA) | -2.1379 | 0.2164 | -9.878 | 2.16e-15 |
Reservoir (FL) | 3.1540 | 0.2696 | 11.701 | <2e-16 |
Reservoir (PF) | 2.8311 | 0.2548 | 11.111 | <2e-16 |
Reservoir (SC) | 1.7638 | 0.2118 | 8.328 | 2.18e-12 |
FIGURE 4| Comparisons of fish abundance (capture per unit of effort²) according to reservoirs (A), behavioral groups (B), and the interaction of these factors (C) collected in the main channel of the Apodi-Mossoró River, Northeast Brazil. Legend: MS – Major Sales; FL – Flechas; PF – Pau dos Ferros; SC – Santa Cruz; SED – sedentary; MIG – migratory; NNS – nonnative (NNSSED). *Different letters indicate statistical significance (α < 0.05).
Sedentary species (SED) presented the highest average abundance (CPUE = 4.48), followed by migratory species (MIG) and non-native fish (NNA), which presented an average CPUEn of 0.19 and 0.13, respectively (GAMLSS; p < 0.05; Tab. 3; Fig. 4B). Analysis of the abundance of the three fish groups in each individual reservoir revealed a higher abundance of sedentary fish (SED). However, in the MS and SC reservoirs, the CPUE of non-native fish (NNA) surpassed that of migratory fish (MIG) (GAMLSS; p < 0.05; Tab. 3; Fig. 4C).
Discussion
The spatial variation observed in the average abundance of fish species, particularly in sedentary and migratory species, with an increase downstream in the cascade reservoir system of the Apodi-Mossoró River, was expected. This is because the reservoirs in the middle (PF) and lower (SC) courses of the river are the two largest reservoirs in the cascade system. According to Agostinho et al. (2007), there is a positive correlation between species abundance and flooded area in Neotropical reservoirs due to a greater variety of habitats and resources. However, we recognize that the sharp reduction in species abundance in the lower course reservoir (SC) may reflect the sampling design, as this reservoir is both the largest and the newest in the Apodi-Mossoró cascade system.
The high abundance of species in the impoundment of the middle region of the river (PF) was surprising, given that this reservoir began operating in 1967. In Neotropical reservoirs, there is a tendency for species abundance to decline in older reservoirs, with higher values observed in newer reservoirs (Agostinho et al., 2007). The high trophic status of the middle impoundment (Moura, Henry-Silva, 2018), compared with that of the other impoundments, may have influenced species abundance, as the high productivity of a lacustrine environment favors the establishment and abundance of small Acestrorhamphidae species (Agostinho et al., 1999; Monaghan et al., 2020).
The predominance of sedentary fish species in the reservoir cascade system of the Apodi-Mossoró River likely stems from the role of reservoirs in reshaping aquatic habitats and fish population dynamics (Agostinho et al., 2016). According to reservoir theory (Bem et al., 2021), dam construction and reservoir formation create new aquatic environments that may differ from natural river habitats, affecting the distribution and abundance of fish species, especially those with sedentary habits. In some cases, the construction of reservoirs can positively affect certain sedentary fish species by flooding previously dry areas, creating new aquatic habitats, and providing opportunities for colonization by species that previously could not inhabit these regions (Agostinho et al., 2007; Vitule et al., 2012). In the cascade system analyzed, all non-native fish species are classified as sedentary. It is also noteworthy that the system supports both sedentary and migratory native species.
As expected, migratory species exhibited low abundance in the cascade reservoir system of the Apodi-Mossoró River, with the main factor being the presence of dams, which interrupt the longitudinal connectivity of rivers and hinder fish movement (Pelicice, Agostinho, 2008; Petesse, Petrere Jr., 2012; Santos et al., 2017). The main factor responsible for the low abundance of migratory fish species in these systems is the presence of dams, which interrupt the longitudinal connectivity of rivers and hinder fish movement (Pelicice, Agostinho, 2008; Petesse, Petrere Jr., 2012; Santos et al., 2017). Additionally, the intermittency of fluvial systems a common feature in arid and semi-arid regions may also influence this dynamic, particularly in river stretches where some seasonal variation in the hydrological regime still occurs. In these environments, due to the short rainy season and prolonged dry periods, river segments remain disconnected for most of the year (Sarmento-Soares et al., 2017). As a result, migratory species generally have the opportunity to migrate only during the rainy season, typically initiating their movements at the beginning of this period to exploit newly flooded habitats as breeding grounds. It is noteworthy that, in some cases, reservoirs may mask this seasonal variation, rendering certain river stretches perennial, which can further alter the natural migration patterns.
The higher abundance of migratory species (MIG) recorded in the mid-river reservoirs aligns with diversity patterns commonly observed in reservoir cascade systems, especially when considering the effects of physical barriers, longitudinal position along the river, and hydrological connectivity factors that, collectively, are widely discussed in the scientific literature (Agostinho et al., 2007; Pompeu et al., 2012; Lima et al., 2016). Reservoirs located in these intermediate positions tend to maintain a certain degree of connectivity with upstream and downstream sections and receive direct influence from adjacent tributaries, which favors the occurrence and persistence of species with different ecological strategies.
The absence of migratory fish in the Major Sales Reservoir can be attributed to three main factors: (i) the geographic location of the reservoir in the upper course of the river, which presents low connectivity with downstream reaches (Maltchik, Medeiros, 2006). This limitation is associated with the presence of physical barriers, such as dams, which restrict the longitudinal connectivity of the system and hinder the arrival of migratory species inhabiting the middle and lower stretches, thus compromising their natural distribution; (ii) the typical environmental conditions of headwater areas in semiarid regions (Lima et al., 2016), characterized by lower water volume, increased hydrological seasonality, and low diversity of aquatic habitats; (iii) the cumulative effect of barriers imposed by the cascade reservoir system (Agostinho et al., 2007; Pompeu et al., 2012), which progressively reduces longitudinal connectivity along the river, impairing the reproductive migration of migratory species. Therefore, these factors emphasize the necessity of considering longitudinal connectivity and the preservation of functional habitats as fundamental elements for the conservation of migratory species in rivers of the Brazilian semiarid region.
In summary, the study’s hypothesis was confirmed: sedentary species were more abundant throughout the cascade reservoir system, while the abundance of non-native species exceeded that of migratory ones. The spatial variation observed in fish assemblages appears to be related to multiple factors, including reservoir size, position within the cascade, and local environmental characteristics. These results highlight the role of fragmentation in reservoir systems in the semiarid region in shaping ichthyofauna distribution, reinforcing the importance of connectivity between water bodies for maintaining community dynamics and biodiversity. These findings are consistent with those reported by de Oliveira et al. (2024), who analyzed the ichthyofauna in three reservoirs of the Apodi-Mossoró River prior to the São Francisco River transposition. Together, both studies complement each other by revealing similar patterns in fish community structure and emphasizing the combined influence of local variables and the spatial organization of reservoirs on the composition and abundance of regional ichthyofauna.
References
Agostinho AA, Gomes LC, Pelicice FM. Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil. Maringá: EDUEM; 2007.
Agostinho AA, Gomes LC, Santos NC, Ortega JC, Pelicice FM. Fish assemblages in Neotropical reservoirs: colonization patterns, impacts and management. Fish Res. 2016; 173(1):26–36. https://doi.org/10.1016/j.fishres.2015.04.006
Agostinho AA, Miranda LE, Bini LM, Gomes LC, Thomaz SM, Suzuki HI. Patterns of colonization in neotropical reservoirs, and prognoses on aging. In: Tundisi JG, Straskraba M, editors. Theoretical reservoir ecology and its applications. Leiden: Backhuys Publishers; 1999. p.227–65.
Bem JD, Ribolli J, Röpke C, Winemiller KO, Zaniboni-Filho E. A cascade of dams affects fish spatial distributions and functional groups of local assemblages in a subtropical river. Neotrop Ichthyol. 2021; 19(3):e200133. https://doi.org/10.1590/1982-0224-2020-0133
Carvalho RG, Kelting FMS, Silva EV. Indicadores socioeconômicos e gestão ambiental nos municípios da bacia hidrográfica do rio Apodi-Mossoró, RN. Soc Nat. 2011; 23(1):143–59.
Costa AST, Bezerra AC, Araújo AMQ, Silva MF, Neto JFDC, Alves RM et al. Dinâmica espaço-temporal das anomalias de precipitação em uma região semiárida, Nordeste do Brasil. Rev Gest Água Am Lat. 2021; 18:1–15. https://dx.doi.org/10.21168/rega.v18e14
Felix VS, Paz AR. Representação dos processos hidrológicos em bacia hidrográfica do semiárido paraibano com modelagem hidrológica distribuída. RBRH. 2016; 21:556–69. https://doi.org/10.1590/2318-0331.011616009
Foubert A, Lecomte F, Legendre P, Cusson M. Spatial organisation of fish communities in the St. Lawrence River: a test for longitudinal gradients and spatial heterogeneities in a large river system. Hydrobiologia. 2018; 809:155–73. https://doi.org/10.1007/s10750-017-3457-z
Fullerton AH, Burnett KM, Steel EA, Flitcroft RL, Pess GR, Feist BE et al. Hydrological connectivity for riverine fish: measurement challenges and research opportunities. Freshw Biol. 2010; 55(11):2215–37. https://doi.org/10.1111/j.1365-2427.2010.02448.x
Hsieh TC, Ma KH, Anne C. iNEXT: iNterpolation and EXTrapolation for species diversity. R package version 3.0.0. 2022. Available from: http://chao.stat.nthu.edu.tw/wordpress/software-download/
Instituto de Gestão das Águas do Rio Grande do Norte (IGARN). Bacia Apodi/Mossoró. Available from: http://www.igarn.rn.gov.br/
Jo H, Jeppesen E, Ventura M, Buchaca T, Gim JS, Yoon JD et al. Responses of fish assemblage structure to large-scale weir construction in riverine ecosystems. Sci Total Environ. 2019; 657:1334–42. https://doi.org/10.1016/j.scitotenv.2018.11.446
Legendre P, Legendre L. Numerical ecology. Vol. 24. Amsterdam: Elsevier; 2012.
Lenth RV. Emmeans: estimated marginal means, aka least-squares means. R package version 1.7.2. 2022. Available from: https://cran.r-project.org
Leonard NJ, Fritsch MA, Ruff JD, Fazio JF, Harrison J, Grover T. The challenge of managing the Columbia River basin for energy and fish. Fish Manag Ecol. 2015; 22(1):88–98. https://doi.org/10.1111/fme.12106
Lima AC, Agostinho CS, Sayanda D, Pelicice FM, Soares AMVM, Monaghan KA. The rise and fall of fish diversity in a Neotropical river after impoundment. Hydrobiologia. 2016; 763(1):207–21. https://doi.org/10.1007/s10750-015-2377-z
Lima FV, Gonçalves RM, Montecino HD, Carvalho RA, Mutti PR. Observações geodésicas multissensoriais para caracterização da seca na Região Hidrográfica Atlântico Nordeste Oriental, Brasil. Sci Total Environ. 2022; 846:157426. https://doi.org/10.1016/j.scitotenv.2022.157426
Marques TV, Mendes K, Mutti P, Medeiros S, Silva L, Perez-Marin AM et al. Environmental and biophysical controls of evapotranspiration from Seasonally Dry Tropical Forests (Caatinga) in the Brazilian Semiarid. Agric For Meteorol. 2020; 287:107957. https://doi.org/10.1016/j.agrformet.2020.107957
Maltchik L, Medeiros ESF. Conservation importance of semi-arid streams in north-eastern Brazil: implications of hydrological disturbance and species diversity. Aquat Conserv Mar Freshw Ecosyst. 2006; 16(7):665–77. https://doi.org/10.1002/aqc.805
Miranda EE, Meirelles Filho J. Rios do Brasil – História & Cultura. Editora Metalivros; 2016.
Monaghan K, Agostinho CS, Pelicice FM, Soares AM. The impact of a hydroelectric dam on Neotropical fish communities: a spatio–temporal analysis of the Trophic Upsurge Hypothesis. Ecol Freshw Fish. 2020; 29(2):384–97. https://doi.org/10.1111/eff.12522
Moura RST, Henry-Silva GG. Is there a zonation pattern in aquatic macrophytes communities in the aquatic environments of the Brazilian semiarid? Braz J Bot. 2018; 41:665–74. https://doi.org/10.1007/s40415-018-0488-2
Nascimento FR. Bacias hidrográficas interminentes sazonais e potencialidades hidroambientais no nordeste setentrional brasileiro. GEOgraphia. 2014; 16(32):90–117.
Novais RP, Carvalho Júnior AP, Oliveira MA. The perennialization of rivers through the construction of dams in the Brazilian northeastern semiarid region. Geopauta. 2022; 6:e940. https://doi.org/10.22481/rg.v6.e2022.e9401
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D et al. Community ecology package. Package ‘vegan’. package version 1.7.2. 2022. Available from: https://cran.r-project.org/web/packages/vegan/index.html
Oliveira JF, Peretti D, Novaes JLC, Costa SR, Oliveira JCD, Fernandes RTV et al. Ictiofauna em três reservatórios do Rio Apodi-Mossoró, semiárido brasileiro, antes da transposição do Rio São Francisco. Rev Geociênc Nordeste. 2024; 10(1):37–44. https://doi.org/10.21680/2447-3359.2024v10n1ID34680
Oliveira JCD, Oliveira JF, Marques ADO, Peretti D, Costa RS, Novaes JLC. Trophic ecology of detritivorous fish along a reservoir cascade in a tropical semi–arid region. Ecol Freshw Fish. 2021; 30(2):234–43. https://doi.org/10.1111/eff.12579
Orsi ML, Britton JR. Long-term changes in the fish assemblage of a neotropical hydroelectric reservoir. J Fish Biol. 2014; 84(6):1964–70. https://doi.org/10.1111/jfb.12392
Pelicice FM, Agostinho AA. Fish-passage facilities as ecological traps in large Neotropical rivers. Conserv Biol. 2008; 1(22):180–88. https://doi.org/10.1111/j.1523-1739.2007.00849.x
Pelicice FM, Azevedo-Santos VM, Esguícero ALH, Agostinho AA, Arcifa MS. Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotrop Ichthyol. 2018; 16(2):e170150. https://doi.org/10.1590/1982-0224-20170150
Petesse ML, Petrere M Jr. Tendency towards homogenization in fish assemblages in the cascade reservoir system of the Tietê river basin, Brazil. Ecol Eng. 2012; 48:109–16. https://doi.org/10.1016/j.ecoleng.2011.06.033
Pompeu PS, Agostinho AA, Pelicice FM. Existing and future challenges: the concept of successful fish passage in South America. River Res Appl. 2012; 28(4):504–12. https://doi.org/10.1002/rra.1557
R Development Core Team. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2021. Available from: https://www.R-project.org
Rigby RA, Stasinopoulos DM, Heller GZ, Bastiani F. Distributions for modeling location, scale, and shape. Using GAMLSS in R. CRC Press. 2019.
Rigby RA, Stasinopoulos DM. Generalized additive models for location, scale and shape. J Roy Stat Soc Ser C Appl Stat. 2005; 54(3):507–54. https://doi.org/10.1111/j.1467-9876.2005.00510.x
Santos NCL, García-Berthou E, Dias JD, Lopes TM, Paiva AI, Severi W et al. Cumulative ecological effects of a Neotropical reservoir cascade across multiple assemblages. Hydrobiologia. 2018; 819:77–91. https://doi.org/10.1007/s10750-018-3630-z
Santos NCL, Santana HS, Ortega JCGA, Dias RM, Stegmann LF, Araújo IMS et al. Environmental filters predict the trait composition of fish communities in reservoir cascades. Hydrobiologia. 2017; 802:245–53. https://doi.org/10.1007/s10750-017-3274-4
Sarmento-Soares LM, Alves CBM, Melo FAG, Moraes LE, Lima SMQ, Ramos TPA. Ictiofauna das ecorregiões de água doce e marinhas do nordeste brasileiro. Bol Soc Bras Ictiol. 2017; 122:16–35.
Simonović P, Ristić R, Milčanović V, Polovina S, Malušević I, Radić B et al. Effects of run-of-river hydropower plants on fish communities in montane stream ecosystems in Serbia. River Res Appl. 2021; 37(5):722–31. https://doi.org/10.1002/rra.3795
Stasinopoulos MD, Rigby RA, Bastiani FD. GAMLSS: a distributional regression approach. Stat Model. 2018; 18(3–4):248–73. https://doi.org/10.1177/1471082X18759144
Terêncio DPS, Pacheco FAL, Fernandes LS, Cortes RMV. Is it safe to remove a dam at the risk of a sprawl by exotic fish species? Sci Total Environ. 2021; 771:144768. https://doi.org/10.1016/j.scitotenv.2020.144768
Vitule JRS. Ecology: preserve Brazil’s aquatic biodiversity. Nature. 2012; 485:309. https://doi.org/10.1038/485309c
Zacardi DM, Lima SMA, Oliveira CC, Oliveira LS, Cajado RA. O gradiente longitudinal (riobarragem) do reservatório de Curuá-Una afeta a composição das assembleias de larvas de peixes? Rev Bras Meio Amb. 2021; 9(1):99–112.
Authors
![]() Jean Carlos Dantas de Oliveira1
Jean Carlos Dantas de Oliveira1 ![]() ,
, ![]() Jônnata Fernandes de Oliveira2,
Jônnata Fernandes de Oliveira2, ![]() Alexandre de Oliveira Marques3,
Alexandre de Oliveira Marques3, ![]() Danielle Peretti1,
Danielle Peretti1, ![]() Rodrigo S. da Costa Goldbaum4 and
Rodrigo S. da Costa Goldbaum4 and ![]() José Luís Costa Novaes4
José Luís Costa Novaes4
[1] Universidade do Estado do Rio Grande do Norte – UERN, Câmpus Central, Programa de Pós-Graduação em Ciências Naturais, Rua Professor Antônio Campos, s/n, Bairro Costa e Silva, 59600-000, Mossoró, RN, Brazil. (JCDO) jeanceac2020@gmail.com (corresponding author), (DP) danielleperetti@uern.br.
[2] Instituto Federal do Rio Grande do Norte – IFRN, Campus Mossoró, Departamento de Ensino, Rua Raimundo Firmino de Oliveira, 400, Conjunto Ulrick Graff, 59628-330, Mossoró, RN, Brazil. (JFO) jonnata.oliveira@ifrn.edu.br.
[3] Núcleo de Ecologia Aquática e Pesca da Amazônia NEAP, Universidade Federal do Pará UFPA, Avenida Perimetral, 2651, 66077 530, Belém, PA, Brazil. (AOM) alexandremarques@ufpa.br.
[4] Universidade Federal Rural do Semi-Árido – UFERSA, Departamento de Biociências, 59610-210, Mossoró, RN, Brazil. (RSCG) rodrigo.goldbaum@ufersa.edu.br, (JLCN) novaes@ufersa.edu.br.
Authors’ Contribution 

Jean Carlos Dantas de Oliveira: Conceptualization, Data curation, Investigation, Writing-original draft, Writing-review and editing.
Jônnata Fernandes de Oliveira: Investigation.
Alexandre de Oliveira Marques: Data curation, Formal analysis.
Danielle Peretti: Investigation.
Rodrigo S. da Costa Goldbaum: Project administration.
José Luís Costa Novaes: Conceptualization, Investigation, Methodology.
Ethical Statement
Fish collection was conducted under the appropriate license issued by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio, process number 27046).
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
Artificial Intelligence (ChatGPT – GPT-5) was used for text adjustments and translation purposes.
Funding
We thank the Universidade Federal Rural do Semi-Árido (UFERSA) and the following institutions for their support in conducting this research: the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Fundação de Amparo e Promoção da Ciência, Tecnologia e Inovação do Rio Grande do Norte (FAPERN) for the DCR fellowship and research support granted to JLCN (process no 68.0025/2005–7); the financial support from CNPq (process no 479883/2012–2), CNPq/Instituto Nacional do Semi-Árido (INSA) (process no. 562525/2010–6), and CNPq (process no 309046/2015–7) for the research productivity fellowship awarded to JLCN. AOM is indebted to CNPq (PPBio – process 442421/2023–0) for their financial support.
Supplementary Material
Supplementary material S1
How to cite this article
Oliveira JCD, Oliveira JF, Marques AO, Peretti D, Goldbaum RSC, Novaes JLC. Diversity and abundance patterns of fish fauna in cascade reservoirs in the Brazilian semiarid tropical region. Neotrop Ichthyol. 2026; 24(1):e250092. https://doi.org/10.1590/1982-0224-2025-0092
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted September 12, 2025
Accepted September 12, 2025
![]() Submitted June 27, 2024
Submitted June 27, 2024
![]() Epub March 16, 2026
Epub March 16, 2026