![]() Fernando M. Assega1,
Fernando M. Assega1, ![]() Alexander Claro-García1,
Alexander Claro-García1, ![]() José L. O. Birindelli1 and
José L. O. Birindelli1 and ![]() Oscar A. Shibatta1
Oscar A. Shibatta1 ![]()
PDF: Download Here | Supplementary: S1 | Cite this article
Associate Editor: ![]() George Mattox
George Mattox
Section Editor: ![]() William Crampton
William Crampton
Editor-in-chief: ![]() Carla Pavanelli
Carla Pavanelli
Abstract
A ontogenia do esqueleto caudal em Lophiosilurus alexandri, um pseudopimelodídeo endêmico da bacia do rio São Francisco, foi analisada para esclarecer o desenvolvimento e a homologia dos elementos caudais. Foram analisados 26 exemplares de larvas recém-eclodidas à adultos, que foram diafanizados, radiografados ou preparados como esqueleto seco. A nadadeira caudal surge como uma prega, com ossificação gradual dos raios principais e procurrentes. Os hipurais 1–5 formam-se como elementos cartilaginosos e fusões posteriores originam o centro terminal composto (PU1+U1+2) e duas placas hipurais (PH+H1+2 e H3–5). O epural ossifica tardiamente nos juvenis e desenvolve um processo proximal em forma de gancho. Os uroneurais fundem-se ao centro terminal durante a transição juvenil–adulto, formando o pleuróstilo. Um U3 separado forma-se precocemente e, depois, funde-se à PU1+U1+2, condição típica de Siluriformes e considerada plesiomórfica para L. alexandri e Pseudopimelodidae. A variação ontogenética na quantidade de raios caudais provavelmente reflete demandas funcionais. Os resultados descrevem a sequência de desenvolvimento e as homologias dos elementos caudais, oferecendo uma base comparativa para a evolução da nadadeira caudal em Siluriformes.
Palavras-chave: Actinopterygii, Esqueleto axial, Hipurais, Ontogenia, Filogenia.
Introduction
Pseudopimelodidae is a catfish family widely distributed in trans- and cis-Andean South America (Shibatta, 2003; Nelson et al., 2016), currently encompassing 59 valid species in six genera (Fricke et al., 2025). Lophiosilurus Steindachner, 1876 comprises five species, and its phylogenetic position has been investigated in several morphological studies (Shibatta, 2003; Ortega-Lara, Lehmann, 2006; Birindelli, Shibatta, 2011; Shibatta, Vari, 2017; Shibatta et al., 2021). Despite some conflicting results, all supported Lophiosilurus as the sister group to Cephalosilurus. Shibatta et al. (2021) proposed the synonymy of Cephalosilurus with Lophiosilurus, which was also corroborated by molecular data (Silva et al., 2021).
Lophiosilurus alexandri, endemic to the São Francisco River basin (Britski et al., 1988; Shibatta, 2003), is the most studied pseudopimelodid species due to its threatened status with extinction (MMA, 2022) and has been cultivated for conservation purposes (Bazzoli, Godinho, 1997). It is the largest species of the family (up to 720 mm TL), inhabits calm waters, uses an ambush strategy, and has reproductive traits that include large adhesive eggs (3.1–3.6 mm) and male parental care (Sato et al., 2003).
This species exhibits the most pronounced morphological modifications within the family, making it a key model for evolutionary studies (Assega et al., 2016). During ontogeny, significant changes in body shape occur, particularly during the post-flexion stage (Assega et al., 2016). Adults exhibit a depressed body, dorsally positioned eyes, a prognathous mouth, robust fin spines, and extensive skeletal fusions, which complicate homology assessments (Shibatta, 1998). Ontogenetic analyses have already revealed unique character states relevant to phylogenetic interpretation, including unusual hypural fusions that motivated this study.
The caudal fin and its supporting skeleton are among the most extensively studied osteological complexes in teleosts, providing key evidence for higher-level taxonomy and phylogenetic hypotheses (Arratia, 2003, 2018; Schultze, Arratia, 2013). In Siluriformes, their diversity was first detailed by Lundberg, Baskin (1969), who identified diagnostic patterns for monophyletic groups. Ontogenetic studies are essential for establishing homology of caudal elements (Schultze, Arratia, 1988, 1989; Arratia, 1991; Hilton, Johnson, 2007; Britz, Johnson, 2012; Schultze, Arratia, 2013; Doosey, Wiley, 2014; Wiley et al., 2015), since the structures undergo significant modifications during development that affect identification and interpretation (Nybelin, 1963; Monod, 1968).
This study aims to describe the development of the caudal skeleton of L. alexandri and evaluate the homology of its elements with those of other catfishes, thereby contributing to a better understanding of the phylogeny of Pseudopimelodidae.
Material and methods
Specimens of Lophiosilurus alexandri were obtained from the Estação de Piscicultura de Três Marias (CODEVASF), São Francisco River basin, Minas Gerais, between August 1994 and January 1996. Immediately after collection, they were fixed in 4% formaldehyde buffered with calcium carbonate.
Developmental periods were defined following Ahlstrom, Ball (1954), and Kendall et al. (1984), with modifications by Nakatani et al. (2001). The larval period was subdivided into yolk-sac, flexion, and post-flexion stages. Juveniles were recognized by the presence of the full complement of fin rays and segmentation (Nakatani et al., 2001). Specimens ≥ 290.0 mm SL were considered adults.
A total of 26 specimens were examined: 19 larvae (four yolk-sac, one flexion, 14 post-flexion), four juveniles, and three adults. Larvae and juveniles were cleared and double-stained according to Dingerkus, Uhler (1977). The precise differentiation of mineralization in the middle layer of the notochord (the chordacentrum) and ossification in the outer layer of the notochord sheath (the autocentrum) regions was not possible with the preparations; however, the alizarin crimson coloration was interpreted as indicative of ossification. Adults were prepared as dried skeletons, as described by Bemis et al. (2004). Additional X-ray examinations were conducted at the Smithsonian Institution’s National Museum of Natural History, Washington (NMNH) and at the Museu de Zoologia da Universidade Estadual de Londrina, Londrina (MZUEL). Dissections followed Cailliet et al. (1986).
Osteological nomenclature follows Lundberg, Baskin (1969), Fujita (1989), Arratia, Schultze (1992), de Pinna, Ng (2004), Grünbaum, Cloutier (2010), Schultze, Arratia (2013, 2018), and Cumplido et al. (2020). Counts of caudal-fin elements follow Schultze, Arratia (1989), Arratia (2008), and Reimchen, MacAdams (2014), and patterns of caudal fusion follow Lundberg, Baskin (1969), and Cumplido et al. (2020). Pleurostyle was identified since its development, as described by Cumplido et al. (2020). The anterodorsal process formed by the fusion of the neural arch of preural centrum 1 (naPU1) and the neural arch of ural centrum 1 (naU1) was referred to as “specialized neural process” (SNP) following Hollister (1936). Additional comparative data on Pseudopimelodidae (ural plate fusions, numbers of principal and procurrent caudal-fin rays) are provided in Tab. S1.
Morphometric data on larvae were obtained using an ocular micrometer attached to a stereomicroscope; juvenile measurements were taken with a digital caliper. Photographs were prepared using a Leica M205A stereomicroscope and LAS software (v. 4.1.0). Institutional abbreviations follow Sabaj (2025).
Results
TABLE 1 |
Five developmental stages were identified: (1) yolk-sac, from 5.7 to 8.0 mm SL (Figs. 1A, B); (2) flexion, from 8.1 to 8.3 mm SL (Fig. 1C); (3) post-flexion, from 8.6 to 15.4 mm SL (Figs. 1D, 2A–D); (4) juvenile, from 15.9 to 28.1 mm SL (Figs. 3A–D); and (5) adult, ≥290.0 mm SL (Figs. 4A, B).
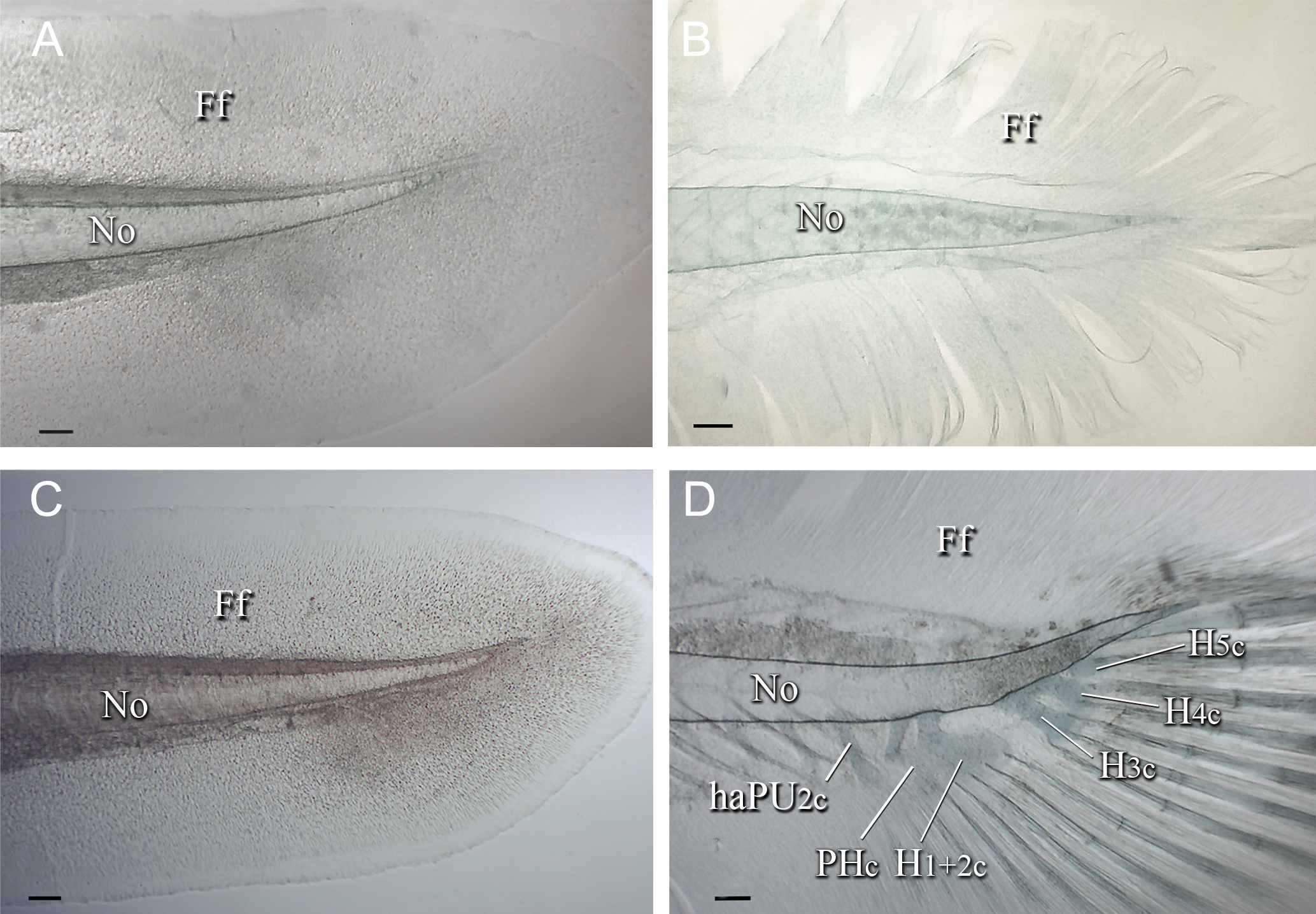
FIGURE 1| Caudal skeleton of Lophiosilurus alexandri; (A) MZUEL 5681, yolk sac stage, 7.9 mm SL; (B) MZUEL 5702, yolk sac stage, 7.5 mm SL; (C) MZUEL 5703, flexion stage, 8.2 mm SL; (D) MZUEL 5682, early postflexion stage, 9.1 mm SL. Ff: finfold; haPU2c: haemal arch of pre-ural 2 cartilage; H3c: hypural 3 cartilage; H4c: hypural 4 cartilage; H5c: hypural 5 cartilage; No: notochord; PH+H1 +2c: parhypural plus hypural 1 and hypural 2 cartilages. Scale bars = 100.0 µm.
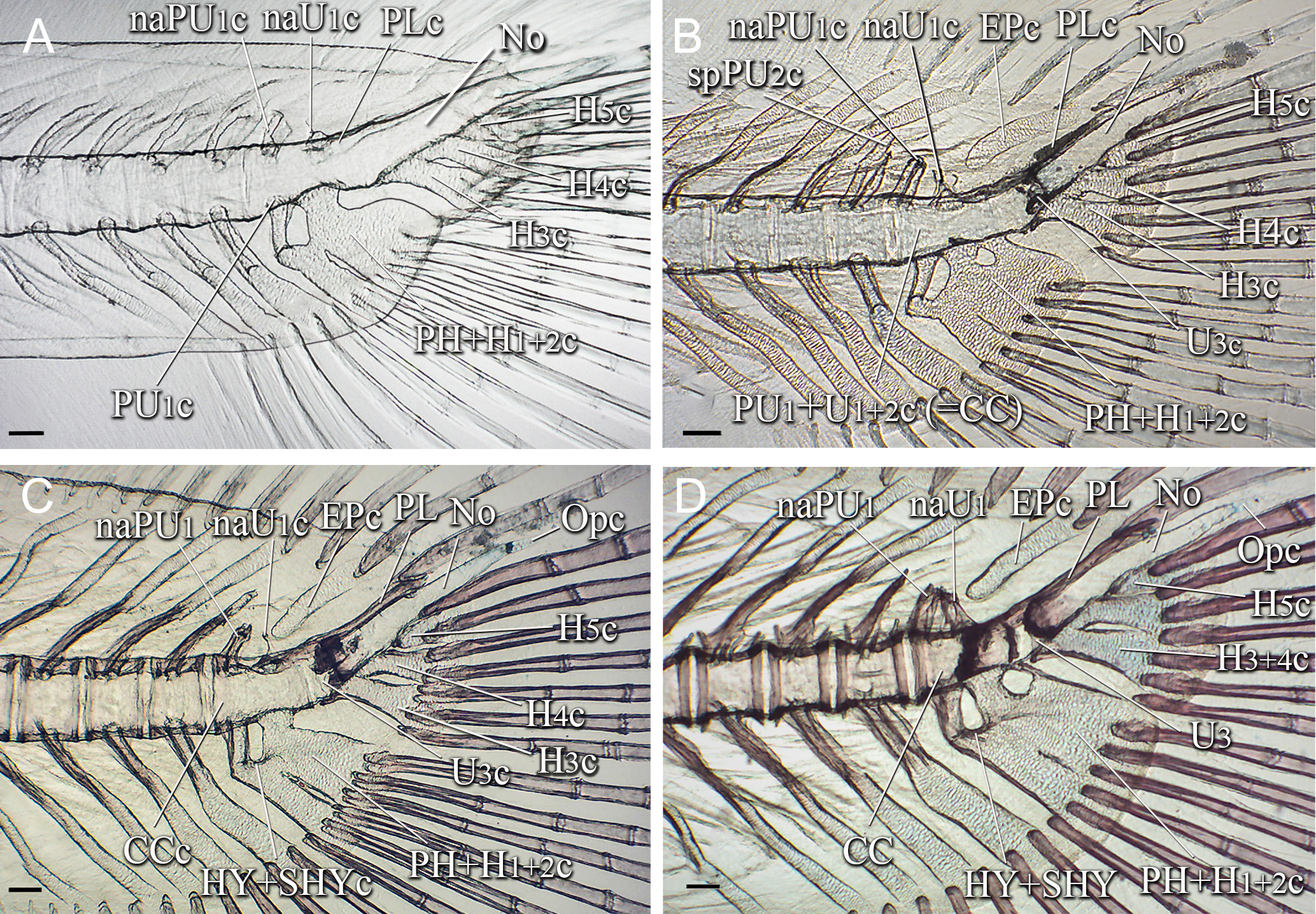
FIGURE 2| Caudal skeleton of Lophiosilurus alexandri in post-flexion stage; (A) MZUEL 5684, 11.1 mm SL; (B) MZUEL 5685, 11.4 mm SL; (C) MZUEL 5689, 14.2 mm SL; (D) MZUEL 5690, 14.9 mm SL. CC: compound centrum; EP: Epural; H3+4: hypural 3 plus hypural 4; H5: hypural 5; HY+SHYc: hypurapophysis plus secondary hypurapophysis cartilage; naPU1: neural arch of preural centrum 1; naU1: neural arch of ural centrum 1 cartilage; No: notochord; Opc: opisthural cartilage; PH+H1+2: parhypural plus hypural 1 and hypural 2; PL: pleurostyle; PU1+U1+2: preural centrum 1 plus ural centrum 1 and ural centrum 2; U3: ural centrum 3. Terminal “c” in abbreviations referring to cartilaginous structures. Scale bars = 100.0 µm.
FIGURE 3| Caudal skeleton of Lophiosilurus alexandri in juvenile stage; (A) MZUEL 5691, 17.3 mm SL; (B) MZUEL 5692, 18.5 mm SL; (C) MZUEL 5693, 19.3 mm SL; (D) MZUEL 5694, 26.6 mm SL. CC: compound centrum; EP: epural; H3+4: hypural 3 plus hypural 4; H5: hypural 5; HY+SHY: hypurapophysis plus secondary hypurapophysis; naPU1: neural arch of preural centrum 1; naU1: neural arch of ural centrum 1; No: notochord; Opc: opisthural cartilage; PH+H1+2: parhypural plus hypural 1 and hypural 2; PL: pleurostyle; U3: ural centrum 3. Terminal “c” in abbreviations referring to cartilaginous structures. Scale bars = 100.0 µm.
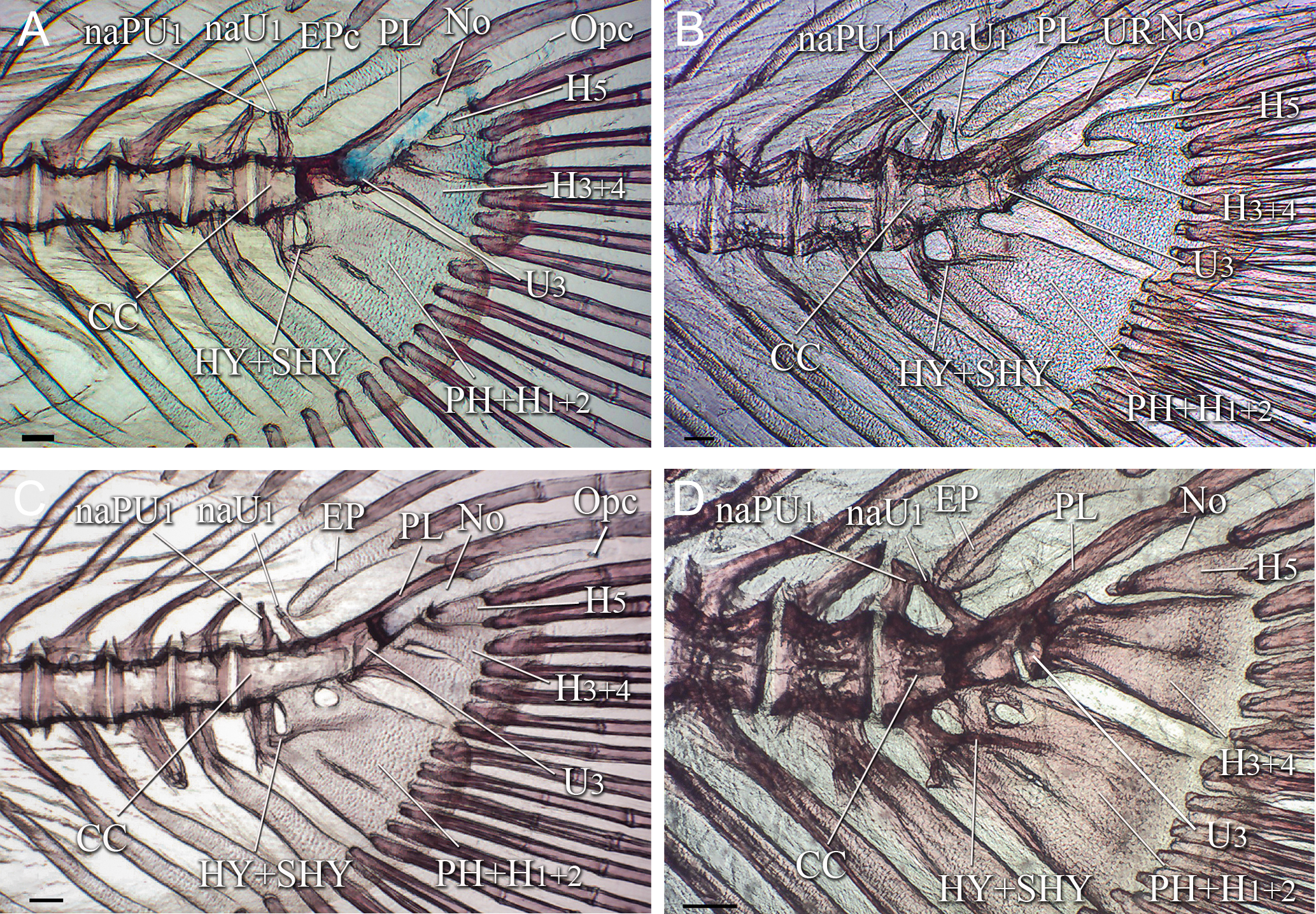
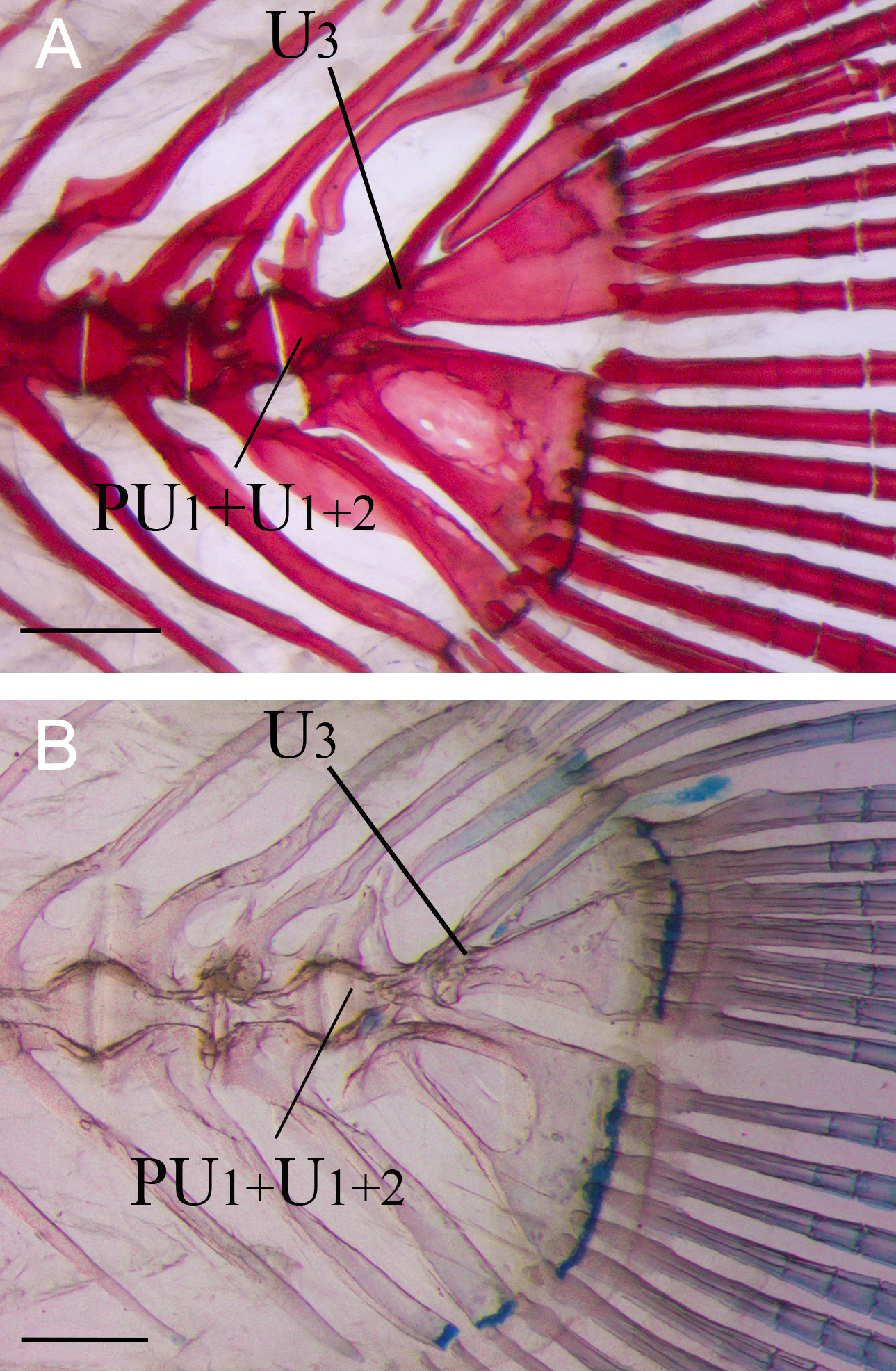
FIGURE 4| Caudal skeleton of Lophiosilurus alexandri in adult stage; (A) MZUEL 16469, 330.4 mm SL; (B) MZUSP 40223, 440.0 mm SL. CC+PH+H1-2+PL: Compound terminal centrum plus parhypural plus hypurals 1-2 and pleurostyle; CC+PH+H1-5+PL: Compound terminal centrum plus parhypural plus hypurals 1-5 and pleurostyle; EP: Epural; H3+4: Hypural 3 plus hypural 4; H5: Hypural 5; HY+SHY: hypurapophysis plus secondary hypurapophysis; PrR: Procurrent caudal-fin rays; PL: Pleurostyle; SNP: Specialized neural process. Scale bars = 10.0 mm.
Yolk-sac stage. At this stage, the caudal fin is represented by a continuous finfold (Figs. 1A, B). The notochord is straight, with no flexion or constrictions. Hypural cartilages are already formed by the end of this stage; as a result, a pre-flexion stage is not present in this species. No formation of principal caudal-fin rays, procurrent rays, preural and ural centra, or parhypural is evident.
Flexion stage. The notochord bends dorsally, marking the onset of caudal flexion. The opisthural cartilage is not yet formed. The caudal fin remains as a finfold (Fig. 1C), and both principal caudal-fin rays and procurrent rays are absent. No differentiation of preural centrum 1 (PU1), ural centrum 1, 2 (U1, 2), or the parhypural (PH) has yet been observed.
Post-flexion stage. During this stage, the caudal skeleton undergoes a sequence of great differentiation and ossification. Hypurals 1 to 5 cartilages are identifiable from early post-flexion stages, at 9.1 mm SL. H1 and H2 are fused (H1+2) since their formation. H3 and H4 are fused (H3+4) at the base and at the distal tip, with an elongated gap between them (Fig. 1d), remaining recognizable during the stage. H1+2 and H3+4 form broad, trapezoidal-shaped plates. H5 cartilage seems to develop as a detached structure, oval-shaped, shorter than the other hypurals. Hypurals begin perichondral ossification at their base, completing at the distalmost portion (Figs. 2D, 3C). Ossification of hypurals begins around 14.9 mm SL (Fig. 2B). By 19.3 mm SL, H1+2 are almost entirely ossified (Fig. 3C). H6 is never formed.
The parhypural (PH) develops early and by 9.1 mm SL, its dorsal region is slightly anteriorly directed (Fig. 1D) and separated from the dorsal region of H1+2 cartilage by an elongated gap between them. Ventrally, the parhypural is broad and partially fused to H1+2 cartilage, leaving a small gap between them. Perichondral ossification begins around 14.9 mm SL, starting at its base and progressing distally (Figs. 2C, 3C). Complete ossification is attained by 19.3 mm SL (Fig. 2D). Still, the small gap between PH and H1+2 remains.
The first evidence of preural centra formation occurs by 11.1 mm SL, when the development of preural centrum 1 (PU1) initiates at the base of the cartilaginous parhypural (PH). Also, the notochord has constrictions where the conical region of the centra will be formed (Fig. 2A).
By 11.4 mm SL, development initiated as a fused U1+,U2 and PU1, forming the compound centrum (CC). The first signs of ossification of the neural arch of PU1 (naPU1) are visible by 14.2 mm SL (Fig. 2C). It is completed by 14.9 mm SL (Fig. 2D). An area with higher density is visible in some preparations between PU1 and U1+2 (e.g., 14.9 mm SL; Fig. 2D), which is interpreted as the limits of each part.
Around 11.4 mm SL, ural centrum 3 (U3) is conspicuous and articulated with the base of the cartilaginous H3 and posteriorly with the remaining notochord (Fig. 2B). At this stage, U3 is separated from CC. U3 grows dorsally around 14.9 mm SL (Fig. 2D).
Haemal arch cartilages of pre-ural centrum 2 (hePU2) and 3 (hePU3) begin forming by 9.1 mm SL (Fig. 1D), with cartilaginous spines evident by 11.1 mm SL. Haemal arches begin to ossify perichondrally at 14.2 mm SL from base to distal region, and by 19.3 mm SL, they are almost completely ossified. Neural arch cartilages of PU1 (naPU1) and U1 (naU1) are also visible (Fig. 2A) by 11.1 mm SL. The cartilaginous pre-ural spines start to form by 11.4 mm SL and perichondrally ossify from the base to the distal region around 14.9 mm SL.
The epural is an elongated, broad structure with a rounded proximal region and a wider and almost straight distal region. It develops as a cartilage located posterodorsally to naU1 at 11.4 mm SL (Fig. 2B), and begins to ossify perichondrally around 14.9 mm SL (Fig. 2D).
The pleurostyle is formed as an elongate structure on the dorsal region of the notochord by 11.1 mm SL, when it appears to be anteriorly separated from NaU1c (Fig. 2A); however, by 11.4 mm SL, it has fused to NaU1c. It becomes laminar and shows membranous ossification by 14.2 mm SL (Fig. 2C). Around 14.9 mm SL, the pleurostyle is fused to the ossified CC.
The hypurapophysis cartilage (HYc) becomes evident by 14.2 mm SL as a lateral outgrowth of the parhypural arch and H1+2 (type C; Fig. 2C). Around 14.9 mm SL, the hypurapophysis starts to ossify anteriorly. The opisthural cartilage appears posteriorly and is articulated to the notochord at 14.2 mm SL.
The development of caudal-fin rays parallels the differentiation of other skeletal elements. At 9.1 to 10.2 mm SL, specimens display seven principal rays in the dorsal lobe and eight in the ventral lobe (7/8). At this stage, the first unbranched ventral ray articulates on the haemal spine of PU2, whereas the first unbranched dorsal ray inserts on H5, ventral to the notochord (Fig. 1D). As development advances, a shift in the first dorsal unbranched ray occurs, with the definitive ray articulating with the uroneurals by 11.4 mm SL (Fig. 2B). In the preparations, ossification of caudal-fin rays were evident at 11.8 mm SL, when one third of the rays are red colored. By 14.2 mm SL, the anterior halves of the rays are colored, and by 14.9 mm SL, approximately two-thirds of their length is stained. Three dermal procurrent rays are visible in the ventral lobe at 11.1 mm SL (Fig. 2A). Procurrent rays are added anteriorly during the post-flexion stage, and alizarin staining proceeds progressively from the base toward the distal tips.
Juvenile stage. By 17.3 mm SL, the fusion of the distal portions of H3, H4, and H5 becomes more evident (Fig. 3A) than in the post-flexion stage. The gap between H3 and H4 began to close by 19.3 mm SL (Fig. 3C) and is almost closed by 26.6 mm SL (Fig. 3D), when this structure, PH, and the remaining hypurals are more ossified. At this size, the separation of H5 from H3+4 increases distally, but a narrow gap remains between PH and H1+2. The HY and secondary hypurapophysis (SHY) are ossified as ridges or processes on the parhypural arch and H1+2 by 18.5 mm SL.
The CC is extensively ossified by 17.3 mm SL, and U3 is partially ossified (Fig. 3A). By 18.5 mm SL, U3 is ossified and articulated with H3 (Fig. 3B). U3 persists as an independent element of CC during the juvenile stage. The ring-shaped specialized neural process (SNP) is formed by 26.6 mm SL, and the pleurostyle becomes deeper (Fig. 3D).
The epural is nearly fully ossified by 19.3 mm SL, but its proximal region is still rounded. By the size of 26.6 mm SL, this region begins to enlarge by adding a keel anteriorly and posteriorly. The principal caudal fin rays are completely ossified by 17.3 mm SL, but they are not branched. At this size, eight dorsal and eight ventral principal rays are counted. Procurrent rays exhibit high variability in number, ranging from 6 to 20 in the ventral lobe and 8 to 15 in the dorsal lobe, which are supported by six to eight preural vertebrae. By 17.3 mm SL, procurrent rays are fully ossified.
Adult stage. In large adults (≥440 mm SL), H5 fuses to H3+4, forming a single hypural plate. This terminal complex is fully developed, and the hypural diastema is reduced, persisting only as a small triangular space. The parhypural is completely fused with H1+2, forming a consolidated element in the caudal-fin skeleton. The epural is strongly ossified, robust, and hook-shaped. The hypurapophysis and secondary hypurapophysis have a strong lateral keel, ranging from parhypural arch to H1+2. The SNP is strongly ossified, wider anteriorly than posteriorly, and its opening is reduced. U3, which is separated from U1+2 during larval and juvenile stages, becomes fused with CC (Fig. 4A) in adults.
The caudal fin bears eight fully ossified principal rays in both lobes (8+8), all branched, except the upper and lowermost rays. It is asymmetrically rounded, with the dorsal lobe being slightly longer than the ventral lobe. The number of procurrent rays varied among the adults examined, with 12 to 15 ventral and 15 to 20 dorsal rays (Fig. 4B).
Material examined. Pseudopimelodidae. Lophiosilurus alexandri: All specimens (except MZUSP 40223 and MZUEL 16469) from Estação de Piscicultura de Três Marias, CODEVASF, São Francisco River, Minas Gerais, Brazil. MZUEL 5542, 1, 290 mm SL; MZUEL 5681, 3, 7.6–8.0 mm SL; MZUEL 5682, 37, 8.7–9.5 mm SL; MZUEL 5683, 21, 9.7–10.4 mm SL; MZUEL 5684, 23, 10.8–11.3 mm SL; MZUEL 5685, 20, 11.0–11.7 mm SL; MZUEL 5686, 25, 10.6–11.7 mm SL; MZUEL 5687, 19, 10.7–11.7 mm SL; MZUEL 5688, 13, 11.4–12.3 mm SL; MZUEL 5689, 6, 13.3–15.2 mm SL; MZUEL 5690, 7, 14.4–15.4 mm SL; MZUEL 5691, 7, 16.9–18.3 mm SL; MZUEL 5692, 8, 15.9–21.2 mm SL; MZUEL 5693, 6, 18.4–20.0 mm SL; MZUEL 5694, 3, 25.1–28.1 mm SL; MZUEL 5702, 3, 7.4–7.5 mm SL; MZUEL 5703, 3, 8.1–8.3 mm SL; MZUEL 5709, 3, 10.6–11.0 mm SL; MZUEL 5714, 3, 10.5–10.8 mm SL; MZUEL 5715, 2, 10.5–11.2 mm SL; MZUEL 5718, 3, 10.6–11.0 mm SL; MZUEL 5720, 3, 10.4–10.8 mm SL; MZUEL 5724, 3, 5.7–6.5 mm SL; MZUEL 5725, 3, 6.0–6.6 mm SL. MZUEL 16469, 1, 300.4 mm SL, São Francisco River, Minas Gerais, Brazil. MZUSP 40223, 1, 440.0 mm SL, no data collection. Lophiosilurus albomarginatus: ROM 61336, 1, 34.5–91.3 mm SL, Tukeit Falls, Potaro River, Essequibo basin, Guyana; ROM 61482, 2, 52.0–81.0 mm SL, Essequibo River, Guyana. Lophiosilurus apurensis: MBUCV-V-15379, 1, 165.2 mm SL, Río Apure, in front of Apurito island, Apure, Venezuela. Lophiosilurus fowleri: MZUEL 12095, 1, 157.0 mm SL, São Francisco River, Minas Gerais, Brazil. Batrochoglanis raninus: MZUSP 23407, 24, 21.7–74.4 mm SL, igarapé Tucuxi, Ati-Paraná, NW of Fonte Boa, Amazonas, Brazil. Batrochoglanis villosus: USNM 226137, 1, 101.6 mm SL, Suriname; Nickerie District; Mataway Creek, approximately 8 km from its intersection with Corantijn River. Cruciglanis pacifici: IMCN 113, 1, 91.6 mm SL, Río San Cipriano when it crosses San Cipriano village, Río Dagua basin, Buenaventura, Colombia. Microglanis carlae: MZUEL 5021, 1, 30.4 mm SL, Arroyo Yacare, tributary to left margin of río Tebicuary, lower Río Paraguay basin, Mburika, Ñeembucú, Paraguay. Microglanis cottoides: MZUSP 68198, 1, 44.6 mm SL, Ribeirão Poço Grande, under bridge of SP-79, km 204, Juquiá, São Paulo, Brazil. Microglanis garavelloi: MZUEL 1677, 1, 30.2 mm SL, Ribeirão Taquari, Jataizinho, Paraná, Brazil; MZUEL 3288, 1, 27.8 mm SL, Ribeirão Pau D’álho, tributary to reservatório de Canoas II, Ibirarema, São Paulo, Brazil. Microglanis lundbergi: INPA 18774, paratypes, 3, 22.3–24.7 mm SL, Solimões River, Costas das Capivaras, Alvarães, Tefé, Amazonas, Brazil. Microglanis parahybae: MNRJ 15989, 2, 28.3–35.8 mm SL, Dois Rios River, Paraíba do Sul River basin, Rio de Janeiro, Brazil. Pseudopimelodus mangurus: MZUEL 12937, 1, 217.9 mm SL, Mogi-Guaçu River, São Paulo, Brazil. Pseudopimelodus schultzi: DUF 920, 1, 200.9 mm SL, Río Magdalena, Colombia. Rhyacoglanis paranensis: MCP 14442, 1, 57.6 mm SL, Tejuco River, Salto Morais, Ituiutaba, Minas Gerais, Brazil. Pimelodidae. Hemisorubim platyrhynchos: MZUEL 3372, 1, 288.8 mm SL, Miranda River, Bonito, Mato Grosso do Sul, Brazil; MZUEL 6688, 1, 268.4 mm SL, Feira do Ceasa, Manaus, Amazonas, Brazil; MZUEL 6701, 1, 329.7 mm SL, Feira do Ceasa, Manaus, Amazonas, Brazil. Pimelodus britski: FMNH 105098, 1, 297.0 mm SL, Iguaçu River, União da Vitória, Paraná, Brazil. Pimelodus pohli: MCP 38775, 1, 150.0 mm SL, Pará River, São Francisco River drainage, Minas Gerais, Brazil. Pimelodus tetramerus: MZUSP 85809, holotype, 112.9 mm, rio Palmeiras, Tocantins River drainage, Ponte Alta do Bom Jesus, Tocantins, Brazil. Steindachneridion melanodermatum: MZUEL 5227, 10, 93.8–111.5 mm SL, Estação Experimental de Estudos Ictiológicos de Salto Segredo, Candói, Paraná, Brazil. Steindachneridion parahybae: MZUEL 14552, 12, 113.6–132.1 mm SL, Estação de Piscicultura CESP, Paraibuna, São Paulo, Brazil. Steindachneridion scriptum: MZUEL 1482, 1, 165.0 mm SL, Tibagi River, Telêmaco Borba, Paraná, Brazil. MZUEL 866, 1, 602.3 mm SL, rio Tibagi, Sapopema, Paraná, Brazil. Heptapteridae. Acentronichthys leptos: UFRJ 289, 1, 83.3 mm SL, São Roque River, Parati, Rio de Janeiro, Brazil. Taunayia bifasciata: MZUSP 45903, 2, 53.1 to 58.7 mm SL, Paranapiacaba, São Paulo, Brazil. Rhamdiopsis moreirai: MCP 14513, 3, 34.1–39.3 mm SL, Negro River, Paraná, Brazil. Rhamdiopsis microcephala: MZUSP 49244, 2, 56.5–69.3 mm SL, Pardo River, Cajuru, São Paulo, Brazil. Phreatobius cisternarum: MNRJ 11569, 1, 55.0 mm SL, Amazonas River, Macapá, Amapá, Brazil. Pimelodella heteropleura: USNM 226105, 73, 26.8–36.1 mm SL, Sisa Creek, “Camp Geologie”, Nickerie, Suriname. Pimelodella marthae: USNM 305613, 34, 24.3–36.0 mm SL, Río Matos, Província Ballivia, Departamento Beni, Bolivia. Pimelodella sp.: UFRJ 2294, 3, 27.3–34.6 mm SL, Javaés River, Tocantins, Brazil. Nemuroglanis pauciradiatus: MZUSP 45908, 2, 28.4–28.5 mm SL, Sanabani River, Silves, Amazonas, Brazil.
Discussion
In Lophiosilurus alexandri, the caudal fin is the first to differentiate from the finfold, although the caudal-fin rays remain unbranched throughout larval and juvenile periods. In teleosts, the caudal fin typically develops earlier than other fins (Kohno et al., 1996; Omori et al., 1996; Sfakianakis et al., 2004), likely reflecting its role as the primary propulsive structure (Faustino, Power, 1998; Koumoundouros et al., 2001). Active swimming in search of food has been reported in teleost larvae (Blaxter, 1988; Gozlan et al., 1999), including in L. alexandri (Chang, 2013).
The ontogenetic sequence of Lophiosilurus alexandri reveals a progressive consolidation of the caudal skeleton, marked by early fusion events and later reorganization of its elements. The parhypural and the ventral hypurals (H1+2) fuse early in development, followed by the coalescence of H3 and H4. H5 exhibits a more complex trajectory, initially appearing as an independent element in the early stages, transiently fusing with H3+4 during the juvenile phase, and finally fusing into a single plate in large adults (approximately 440.0 mm SL). In the mature condition, two major plates (PH+H1+2 and H3–5) are fused to the compound terminal vertebrae (PU1+U1+U2), producing a compact caudal skeleton with a narrow hypural diastema that diminishes as growth proceeds. Unlike Ictalurus punctatus, which retains H6 (Kubicek, 2022), L. alexandri lacks this element, as in most catfishes (Arratia, 1987) and other pseudopimelodids. Reduction of hypural plates by loss or fusion during ontogeny is a common evolutionary trend in catfishes and other teleosts (Lundberg, Baskin, 1969; Wiley, Johnson, 2010). In large adults of L. alexandri, the complete fusion of hypural plates obscures their individual origins. However, it is a condition unique within Pseudopimelodidae, since H5 remains individualized in other species. The shallow hypural diastema and the late fusion of H5 to H3+4 are thus interpreted as autapomorphies of L. alexandri. Similarly, the recurved epural observed in large adults is shared only with L. fowleri, providing additional support for their close relationship (Shibatta, 1998; Birindelli, Shibatta, 2011; Shibatta et al., 2021; Silva et al., 2021).
The limits between the first preural centrum (PU1) and the first and second ural centra (U1+2) are indistinguishable in L. alexandri from the postflexion stage onward, as these elements develop as a single unit forming the complex centrum PU1+U1+2. The third ural centrum (U3) arises early in postflexion larvae as an independent element but fuses to the PU1+U1+2 complex in adults. The homology of U1, U2, and U3 remains uncertain; however, studies on Teleostei suggest that the element usually identified as U1 may correspond to U1+2, whereas U2 may represent U3 or even U3-5 (Schultze, Arratia, 1988; Hilton, 2002; Cumplido et al., 2020). Evolutionarily, Teleostei exhibit a consistent trend toward a reduction in the number of ural centra (Greenwood, 1970; Schultze, Arratia, 1989; 2013; Arratia, 1991; de Pinna, Ng, 2004). Groups such as Elopomorpha and Osteoglossomorpha retain several ural centra (Schultze, Arratia, 1988, 2013; Arratia, Schultze, 1992; de Pinna, Ng, 2004), whereas most Ostariophysi have only one (Fink, Fink, 1981). In Gymnotiformes, Characiformes, and Cypriniformes, the typical condition is the absence of a well-developed U2 (de Pinna, Ng, 2004).
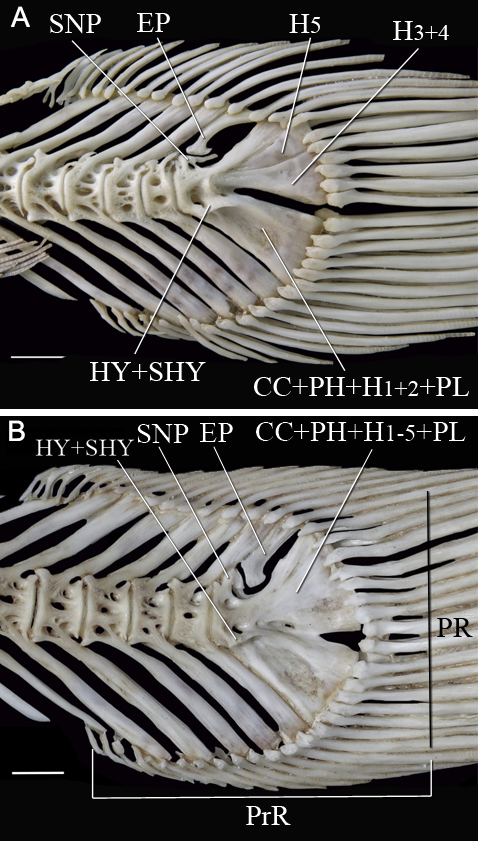
In Lophiosilurus alexandri, U3 is a conspicuous element in juveniles. Still, it later fuses with the CC in adults, representing the plesiomorphic condition for Pseudopimelodidae and aligning with the broader evolutionary trend in Siluriformes toward reduction of ural centra. The same condition occurs in adults of Pseudopimelodus mangurus and Batrochoglanis villosus, in which U3 is fused with the CC as described by Lundberg, Baskin (1969). In contrast, all Microglanis species examined (M. carlae, M. cottoides, M. garavelloi, M. parahybae, and M. lundbergi) retain a well-developed U3 in adults (Fig. 5), despite previous reports suggesting its absence in the genus (de Pinna, Ng, 2004). However, the same authors report the occurrence of a separate U3 in Batrochoglanis raninus (previously identified as Pseudopimelodus raninus) and draw attention to a possible autapomorphy of this species or a synapomorphy to a subset of Pseudopimelodus species. Persistence of U3 in adults of Microglanis species may therefore represent pedomorphosis and a synapomorphy of Microglanis and B. raninus within Pseudopimelodidae. At the same time, the fusion of U3 with CC seems to reflect the ancestral state for the family. Among Siluriformes, the most common pattern involves a not evident U2 (=U3) fused to the bases of H3+4, while a separate U2 (=U3) fused only to H3 is rare (Lundberg, Baskin, 1969). Adult catfishes with an individualized U3 are restricted to Helogeninae (Cetopsidae), Aspredinidae, Amblyceps (Amblycipitidae), some Chiloglanis (Mochokidae), and Pseudopimelodidae (Lundberg, Baskin, 1969; de Pinna, Ng, 2004). This contrast between retention and fusion highlights the phylogenetic significance of U3, suggesting that ontogenetic and adult variation offer valuable insights for distinguishing the evolutionary conditions in the caudal skeleton of Siluriformes.
FIGURE 5| Caudal skeleton of adults of (A) Microglanis garavelloi, MZUEL 3288, 27.8 mm SL; (B) Microglanis carlae, MZUEL 5021, 30.4 mm SL. PU1+U1+2: Preural 1 plus ural centrum 1 and ural centrum 2; U3: Ural centrum 3. Scale bars = 1.0 mm.
The number of procurrent rays in L. alexandri varied considerably in adults (12 to 15 ventral and 15 to 20 dorsal), encompassing the variation reported for other Pseudopimelodidae (Tab. S1) and overlapping with ranges observed in other Siluriformes such as Ictalurus punctatus (Kubicek, 2022). Although the evolutionary significance of this variation is uncertain, procurrent ray counts may aid in species-level diagnoses. Likewise, the number of preural vertebrae supporting procurrent rays varies among pseudopimelodids, ranging from six to eight in L. alexandri, in contrast to four to five in Microglanis spp. and up to nine in L. apurensis.
The ontogenetic transformations of the caudal skeleton in L. alexandri reveal how developmental processes underpin evolutionary patterns within Pseudopimelodidae. The progressive fusion of elements such as the hypurals, preural, and ural centra illustrates a developmental trajectory toward structural simplification. The persistence of intermediate stages during ontogeny provides valuable insight into the sequence of morphological modifications that have shaped the caudal complex of the family. These observations underscore the importance of ontogenetic evidence in identifying plesiomorphic and apomorphic conditions, highlighting that developmental data are crucial for comprehending the evolution and functional diversification of the caudal skeleton in catfishes.
Acknowledgments
Special thanks to Gloria Arratia for her critical analysis of the manuscript, and also to Mark Sabaj (ANSP), Ralf Britz (NHM), George Mattox (UFSCar), and Fernando Jerep (UEL) for suggestions to this study; to Andréa Bialetzki (UEM), Yoshimi. Sato, and Elizabeth Cardoso Lomelino (CODEVASF, Três Marias) for providing most of the specimens used in the study; to Osvaldo Oyakawa (MZUSP) for the loan of a dried skeletonized specimen; and to Edson Santana and Aparecido de Souza (UEL) for help with laboratory procedures.
References
Ahlstrom EH, Ball OP. Description of eggs and larvae of jack mackerel (Trachurus symmetricus) and distribution and abundance of larvae in 1950 and 1951. Fish B-NOAA. 1954; 56(1):209–45.
Arratia G. Description of the primitive family Diplomystidae (Siluriformes, Teleostei, Pisces): morphology, taxonomy and phylogenetic implications. Bonn Zool Monogr. 1987; 24:1–120.
Arratia G. The caudal skeleton of Jurassic teleosts; a phylogenetic analysis. In: Chang M-M, Liu YH, Zhang G-R, editors. Early vertebrates and related problems in evolutionary biology. Beijing: Science Press; 1991. p.249–340.
Arratia G. The siluriform postcranial skeleton. An overview. In: Arratia G, Kapoor BG, Chardon M, Diogo R, editors. Catfishes. Enfield: Science Publishers; 2003. p.121–57.
Arratia G. Actinopterygian postcranial skeleton with special reference to the diversity of fin ray elements, and the problem of identifying homologies. In: Arratia G, Schultze HP, Wilson MVH, editors. Mesozoic fishes 4 – Homology and phylogeny. Munchen: Verlag Dr. Friedrich Pfeil; 2008. p.49–101.
Arratia G. Otomorphs (= otocephalans or ostarioclupeomorphs) revision. Neotrop Ichthyol. 2018; 16(3):e180079. https//doi.org/10.1590/1982-0224-20180079
Arratia G, Schultze HP. Reevaluation of the caudal skeleton of certain actinopterygian fishes: III. Salmonidae. Homologization of caudal skeletal structures. J Morphol. 1992; 214:187–249.
Assega FM, Birindelli JLO, Bialetzki A, Shibatta OA. External morphology of Lophiosilurus alexandri Steindachner, 1876 during early stages of development, and its implications for the evolution of Pseudopimelodidae (Siluriformes). PLoS ONE. 2016; 11(4):e0153123. https//doi.org/10.1371/journal.pone.0153123
Bazzoli N, Godinho HP. Ovócitos vitelogênicos do surubim Pseudoplatystoma corruscans e do pacamã Lophiosilurus alexandri. In: Miranda MOT, editor. Surubim. Belo Horizonte: IBAMA; 1997. p.81–90.
Bemis WE, Hilton EJ, Brown B, Arrindell R, Richmond AM, Little CD et al. Methods for preparing dry, partially articulated skeletons of osteichthyans, with notes on making ridewood dissections of the cranial skeleton. Copeia.2004;2004(3):603–09. https://doi.org/10.1643/CI-03-054R1
Birindelli JLO, Shibatta OA. Morphology of the gas bladder in bumblebee catfishes (Siluriformes, Pseudopimelodidae). J Morphol. 2011; 272(7):890–96. https://doi.org/10.1002/jmor.10961
Blaxter JS. Pattern and variety in development. In: Hoar WS, Randall DJ, editors. Fish physiology. London: Academic Press; 1988. p.1–58.
Britz R, Johnson GD. Ontogeny and homology of the skeletal elements that form the sucking disc of remoras (Teleostei, Echeneoidei, Echeneidae). J Morphol. 2012; 273(12):1353–66. https://doi.org/10.1002/jmor.20063
Britski HA, Sato Y, Rosa ABS. Manual de identificação de peixes da região de Três Marias (com chaves de identificação para os peixes da Bacia do São Francisco). 3ª ed. Brasília: CODEVASF; 1988.
Cailliet GM, Love MS, Ebeling AW. Fishes: a field and laboratory manual on their structure, identification and natural history. Belmont: Wadsworth Publishing Company; 1986.
Chang I. Breeding success with the pac-man catfish, Lophiosilurus alexandri. Amazonas. 2013; 2:74–78.
Cumplido N, Allende ML, Arratia G. From devo to evo: patterning, fusion and evolution of the zebrafish terminal vertebra. Front Zool. 2020; 17:18. https://doi.org/10.1186/s12983-020-00364-y
Dingerkus G, Uhler LD. Enzyme clearing of alcian blue stained whole small vertebrates for demonstration of cartilage. Stain Technol. 1977; 52(4):229–32. https://doi.org/10.3109/10520297709116780
Doosey M, Wiley EO. Epural bones in teleost fishes: a problem of phylogenetic homology. Ichthyol Res. 2014; 62(2):131–44.
Faustino M, Power DM. Development of osteological structures in the sea bream: vertebral column and caudal fin complex. J Fish Biol. 1998; 52(1):11–22. https://doi.org/10.1111/j.1095-8649.1998.tb01548.x
Fink SV, Fink WL. Interrelationships of the ostariophysan fishes (Teleostei). Zool J Linn Soc-Lond. 1981; 72(4):297–353. https://doi.org/10.1111/j.1096-3642.1981.tb01575.x
Fricke R, Eschmeyer WN, Fong JD. Species by family/subfamily. [Internet]. San Francisco: California Academy of Science; 2025. Available from:http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Fujita K. Nomenclature of cartilaginous elements in the caudal skeleton of teleostean fishes. Jpn J Ichthyol. 1989; 36(1):22–29. https://doi.org/10.1007/bf02905669
Gozlan RE, Copp GH, Tourenq JN. Early development of the sofie, Chondrostoma toxostoma. Environ Biol Fish. 1999; 56:67–77. https://doi.org/10.1023/A:1007576222907
Greenwood PH. On the genus Lycoptera and its relationships with the family Hiodontidae (Pisces, Osteoglossomorpha). B Brit Mus Nat Hist-Zool. 1970; 19:259–85. https://doi.org/10.5962/p.314188
Grünbaum T, Cloutier R. Ontogeny, variation, and homology in Salvelinus alpinus caudal skeleton (Teleostei: Salmonidae). J Morphol. 2010; 271(1):12–24. https://doi.org/10.1002/jmor.10775
Hilton EJ. Osteology of the extant North American fishes of the genus Hiodon Lesueur, 1818 (Teleostei: Osteoglossomorpha: Hiodontiformes). Fieldiana Zool. 2002; 100:1–142. https://doi.org/10.5962/bhl.title.2666
Hilton EJ, Johnson GD. When two equals three: developmental osteology and homology of the caudal skeleton in carangid fishes (Perciformes: Carangidae). Evol Dev. 2007; 9(2):178–89. https://doi.org/10.1111/j.1525-142X.2007.00148.x
Hollister G. Caudal skeleton of Bermuda shallow water fishes. Order Isospondyli: Elopidae, Megalopidae, Albulidae, Clupeidae, Dussumieriidae, Engraulidae. Zoologica. 1936; 21(8):257–90.
Kendall Jr. AW, Ahlstrom EH, Moser HG. Early life history stages of fishes and their characters. In: Kendall Jr. AW, Ahlstrom EH, Richards WJ, Cohen DM, Faray MP, Richardson SL et al., editors. Ontogeny and systematics of fishes. La Jolla: American Society of Ichthyologists and Herpetologists; 1984. p.11–22.
Kohno H, Ordonio-Aguilar R, Ohno A, Taki Y. Morphological aspects of feeding and improvement in feeding ability in early stage larvae of the milkfish, Chanos chanos. Ichthyol Res. 1996; 43(2):133–40.
Koumoundouros G, Sfakianakis D, Maingot E, Divanachn P, Kentouri M. Osteological development of the vertebral column and of the fins in Diplodus sargus (Teleostei: Perciformes: Sparidae). Mar Biol. 2001; 139:853–62. https://doi.org/10.1007/s002270100645
Kubicek KL. Developmental osteology of Ictalurus punctatus and Noturus gyrinus (Siluriformes: Ictaluridae) with a discussion of siluriform bone homologies. Vert Zool. 2022; 72:661–727. https://doi.org/10.3897/vz.72.e85144
Lundberg J, Baskin J. The caudal skeleton of the catfishes, order Siluriformes. Am Mus Novit. 1969; 2398:1–50.
Ministério do Meio Ambiente (MMA). Portaria MMA no 148, de 7 de junho de 2022. Anexo 3 – Peixes. Brasília, Ministério do Meio Ambiente; 2022. Available from: https://www.icmbio.gov.br/cepsul/images/stories/legislacao/Portaria/2020/P_mma_148_2022_altera_anexos_P_mma_443_444_445_2014_atualiza_especies_ameacadas_extincao.pdf.
Monod T. Le complexe urophore des poissons téléostéens. Mém Inst Fondam Afrique Noire. 1968; 81:1–705.
Nakatani K. Ovos e larvas de peixes de água doce: desenvolvimento e manual de identificação. Maringá: Eduem; 2001.
Nelson JS, Grande T, Wilson MVH. Fishes of the World. 5th ed. Hoboken: John Wiley & Sons; 2016. Available from: https://doi.org/10.1002/9781119174844
Nybelin O. Zur morphologie un terminologie des schwanzkelletes der actinopterygier. Arkhiv Zool. 1963; 15:485–516.
Omori M, Sugawara Y, Honda H. Morphogenesis in hatchery-reared larvae of the black rockfish, Sebastes schlegeli, and its relationship to the development of swimming and feeding functions. Ichthyol Res. 1996; 43:267–82. https://doi.org/10.1007/BF02347599
Ortega-Lara A, Lehmann A P. Cruciglanis, a new genus of Pseudopimelodid catfish (Ostariophysi: Siluriformes) with description of a new species from the Colombian Pacific coast. Neotrop Ichthyol. 2006; 4(2):147–56. https://doi.org/10.1590/S1679-62252006000200002
de Pinna MCC, Ng HH. The second ural centrum in Siluriformes and its implication for the monophyly of superfamily Sisoroidea (Teleostei, Ostariophysi). Am Mus Novit. 2004; 2004(3437):1–23. https://doi.org/10.1206/0003-0082(2004)437<0001:TSUCIS>2.0.CO;2
Reimchen TE, MacAdams JC. Population, sex, and ontogenetic differences in the procurrent rays of the caudal fin in threespine sticklebacks (Gasterosteus aculeatus). Can J Zool. 2014; 92(8):675–79. https://doi.org/10.1139/cjz-2014-0055
Sabaj MH. Standard symbolic codes for institutional resource collections in herpetology and ichthyology: an online reference. Version 9.7 (3 March 2025). Washington, DC: American Society of Ichthyologists and Herpetologists; 2025. Available from: http://www.asih.org/
Sato Y, Fenerich-Verani N, Nuñer APO, Godinho HP, Verani JR. Padrões reprodutivos de peixes da bacia do São Francisco. In: Godinho HP, Godinho AL, editors. Águas, peixes e pescadores do São Francisco das Minas Gerais. Belo Horizonte: PUC Minas; 2003. p.229–74.
Schultze HP, Arratia G. Reevaluation of the caudal skeleton of actinopterygian: II. Hiodon, Elops and Albula. J Morphol. 1988; 195:257–303.
Schultze HP, Arratia G. The composition of the caudal skeleton of teleosts (Actinopterygii: Osteichthyes). Zool J Linn Soc. 1989; 97(3):189–231. https://doi.org/10.1111/j.1096-3642.1989.tb00547.x
Schultze HP, Arratia G. The caudal skeleton of basal teleosts, its conventions, and some of its major evolutionary novelties in a temporal dimension. In: Arratia G, Schultze HP, Wilson MH, editors. Mesozoic fishes 5 – Global diversity and evolution. München: Verlag Dr. F. Pfeil; 2013. p.187–246.
Sfakianakis DG, Koumoundouros G, Divanach P, Kentouri M. Osteological development of the vertebral column and of the fins in Pagellus erythrinus (L. 1758). Temperature effect on the developmental plasticity and morpho-anatomical abnormalities. Aquaculture. 2004; 232(1–4):407–24. https://doi.org/10.1016/j.aquaculture.2003.08.014
Shibatta OA. Sistemática e evolução da família Pseudopimelodidae (Ostariophysy, Siluriformes), com a revisão taxonômica do gênero Pseudopimelodus [PhD Thesis]. São Carlos: Universidade Federal de São Carlos; 1998.
Shibatta OA. Phylogeny and classification of Pimelodidae. In: Arratia G, Kapoor BG, Chardon M, Diogo R, editors. Catfishes. Enfield: Science Publishers Inc.; 2003. p.385–400.
Shibatta OA, Jarduli LR, Abrahão VP, Souza-Shibatta L. Phylogeny of the Neotropical Pacman catfish genus Lophiosilurus (Siluriformes: Pseudopimelodidae). Neotrop Ichthyol. 2021; 19(4):e210040. https://doi.org/10.1590/1982-0224-2021-0040
Shibatta OA, Vari RP. A new genus of Neotropical rheophilic catfishes, with four new species (Teleostei: Siluriformes: Pseudopimelodidae). Neotrop Ichthyol. 2017; 15(2):e160132. https://doi.org/10.1590/1982-0224-20160132
Silva GSC, Melo BF, Roxo FF, Ochoa LE, Shibatta OA, Sabaj MH, Oliveira C. Phylogenomics of the bumblebee catfishes (Siluriformes: Pseudopimelodidae) using ultraconserved elements. J Zool Syst Evol Res. 2021; 59(8):1662–72. https://doi.org/10.1111/jzs.12513
Wiley EO, Johnson GD. A teleost classification based on monophyletic groups. In: Nelson JS, Schultze HP, Wilson MVH, editors. Origin and phylogenetic interrelationships of teleosts. München: Verlag Dr. Friedrich Pfeil; 2010. p.123–82.
Wiley EO, Fuiten AM, Doosey MH, Looman BK, Merker C, Azuma M. The caudal skeleton of the zebra fish, Danio rerio, from a phylogenetic perspective: a polyural interpretation of homologous structures. Copeia. 2015; 103(4):740–50. https://doi.org/10.1643/CG-14-105
Authors
![]() Fernando M. Assega1,
Fernando M. Assega1, ![]() Alexander Claro-García1,
Alexander Claro-García1, ![]() José L. O. Birindelli1 and
José L. O. Birindelli1 and ![]() Oscar A. Shibatta1
Oscar A. Shibatta1 ![]()
[1] Programa de Pós-Graduação em Ciências Biológicas, Departamento de Biologia Animal e Vegetal, Centro de Ciências Biológicas, Universidade Estadual de Londrina, 86057-970, Londrina, PR, Brazil. (FMA) fernandoassega@hotmail.com, (ACG) alessclaro@ gmail.com, (JLOB) josebirindelli@uel.br, (OAS) shibatta@uel.br (corresponding author).
Authors’ Contribution 

Fernando M. Assega: Data curation, Formal analysis, Investigation, Methodology, Writing-original draft.
Alexander Claro-García: Formal analysis, Investigation, Writing-original draft.
José L. O. Birindelli: Validation, Writing-original draft, Writing-review and editing.
Oscar A. Shibatta: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
ChatGPT was used to detect information redundancy, writing coherence, English revision, and Abstract shortening.
Funding
This study is part of the MSc program of the first author, which was supported by a grant from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001 though the Programa de Pós-Graduação em Ciências Biológicas at Universidade Estadual de Londrina. JLOB (308846/2023–0) and OAS (307951/2021–9) are thankful to CNPq for their research grants, and to Fundação Araucária for funding of the Taxoline network (process NAPI 168/2024).
Supplementary Material
Supplementary material S1
How to cite this article
Assega FM, Claro-García A, Birindelli JLO, Shibatta OA. Development of the caudal fin and supporting skeleton in Lophiosilurus alexandri (Siluriformes: Pseudopimelodidae) and insights into its evolution in the family. Neotrop Ichthyol. 2026; 24(1):e250118. https://doi.org/10.1590/1982-0224-2025-0118
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted November 14, 2025
Accepted November 14, 2025
![]() Submitted July 4, 2025
Submitted July 4, 2025
![]() Epub March 16, 2026
Epub March 16, 2026