![]() Benton L. Fry1,2
Benton L. Fry1,2 ![]() ,
, ![]() Calvin J. Young1,
Calvin J. Young1, ![]() Carmen G. Montaña3,
Carmen G. Montaña3, ![]() Leslie C. Kelso Winemiller1 and
Leslie C. Kelso Winemiller1 and ![]() Kirk O. Winemiller1,2
Kirk O. Winemiller1,2
PDF: Download Here | Supplementary: S1 S2 | Cite this article
Associate Editor: ![]() Caroline Arantes
Caroline Arantes
Section Editor: ![]() Fernando Pelicice
Fernando Pelicice
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
AMost tropical rivers have a seasonal flood pulse that connects channel and floodplain habitats and facilitates entrance of allochthonous resources into the aquatic food web. Dry season isolation of floodplain habitats should result in greater spatial variation in production sources supporting aquatic food webs and greater input of autochthonous sources. We performed stable isotope analysis on tissue samples from fishes and basal production sources from the channel and three floodplain ponds of the Rupununi River, Guyana, to estimate assimilation of material originating from basal sources by five trophic guilds. Seston was the principal source assimilated by fish trophic guilds at each location, with the only exceptions being invertivores and piscivores inhabiting the river channel. Terrestrial plants were estimated to be the most important basal source supporting piscivores, possibly reflecting a time lag for basal source materials to pass several steps up food chains. Between-pond variation in proportions of basal resources assimilated by guilds was associated with differences in water depth and density of aquatic macrophytes. Findings further support the importance of autochthonous production sources for aquatic food webs of tropical rivers during the dry season and the role of floodplain heterogeneity in creating spatial variation in food web dynamics.
Keywords: Food web, Hydrologic connectivity, Neotropics, Stable isotope analysis.
Introduction
Tropical rivers generally have strongly seasonal flow regimes, with an annual rainy season producing a sustained flood pulse and hydrologic connectivity between the active channel and floodplains (Lowe-McConnell, 1964; Junk et al., 1989; Winemiller, Jepsen, 1998). During the dry season, water levels recede, creating isolated bodies of water in floodplains that vary in physicochemical and biotic features (Rodriguez, Lewis, 1997; Pouilly et al., 2004; Scarabotti et al., 2011). Given the effects of seasonal hydrology on habitat connectivity, primary production, and fish reproduction and dispersal, tropical river food webs are temporally dynamic and spatially variable (Winemiller, 1990, 1996). Decreasing water levels increases fish densities and rates of encounter between predators and their prey. As the dry season progresses, predation mortality reduces prey populations, and predators may switch to consuming sub-optimal prey in accordance with the functional response (Jepsen et al., 1997, Montaña et al., 2011). During high-water periods, herbivores and omnivores have access to and exploit allochthonous food resources in flooded riparian areas (Junk et al., 1989; Correa, Winemiller, 2018; Arantes et al., 2019). Conversely, during low-water periods, herbivores and omnivores rely more on autochthonous basal production (Roach et al., 2009b; Roach, Winemiller, 2015).
The present study compares within-guild and between-guild variation in basal resources assimilation during the dry season among fish inhabiting the channel and floodplain habitats of the Rupununi River, a meandering lowland river draining the Rupununi Savanna in Southwestern Guyana. The Rupununi has a predictable seasonal hydrology, with an annual flood pulse that starts in May and continues through September. Prior research in this region focused on the seasonal hydrologic connection between the Branco/Amazon and Rupununi/Essequibo basins via seasonally flooded savannas, i.e., the so-called Rupununi Portal (Souza et al., 2012, 2020). Other studies have produced comprehensive species lists of fishes from the Rupununi River and other regions of Guyana (Watkins et al., 2005; Taphorn et al., 2022). Research on fish trophic ecology in the Rupununi region was conducted by the pioneering fish ecologist Rosemary Lowe-McConnell (1964), who analyzed gut contents of fishes from isolated floodplain ponds during the dry season and the flooded savannas during the wet season. She found that fish fed consistently throughout the wet season, but many guts were empty when confined to ponds during the dry season. We aim to further understand food web dynamics in the Rupununi River and its associated floodplain ponds by estimating fish assimilation of material originating from basal production sources by analyzing stable isotope ratios of carbon and nitrogen.
Analysis of stable isotope ratios of carbon and nitrogen (δ13C, δ15N) in consumer tissues and food resources are commonly used to reveal aspects of trophic structure and food web dynamics (Boecklen et al., 2011; Layman et al., 2012). When basal production sources have sufficiently distinct isotopic ratios, δ13C in particular, mixing models can be used to estimate source contributions to consumer biomass (Hopkins, Ferguson, 2012). The vertical trophic position of consumers can be estimated from their tissue δ15N, and this is because the ratio increases (15N enrichment) as food is consumed and assimilated by animals (Post, 2002; Stephan et al., 2023). Several studies have employed stable isotope analysis and mixing models to estimate sources of production supporting fishes in tropical floodplain rivers. For example, fishes in a floodplain lake of the Cuiabá River, Brazil, were found to assimilate high proportions of material originating from terrestrial C3 plants during the flood pulse, with a decline in the vertical trophic positions of omnivorous fishes (Wantzen et al., 2002). During the dry season, there was a decline in assimilation of material from terrestrial sources, and estimated trophic positions of omnivorous fishes shifted to higher values. Roach et al. (2008b) analyzed isotopic signatures of fishes in the main channel and a connected floodplain lake of the Cinaruco River, Venezuela, during the dry season. Algae were inferred to be the predominant basal resource assimilated by fishes in all trophic guilds, and the food web structure was consistent between the river and floodplain. Neither of those studies examined basal resource assimilation in relation to floodplain heterogeneity or fish trophic guilds, which is the focus of the present study.
Here, we analyzed isotopic ratios of carbon and nitrogen in fish muscle tissue to estimate the relative importance of the most common basal production sources supporting fish biomass in the channel and floodplain ponds of the Rupununi River. We hypothesized that isotopic ratios of fishes within the same trophic guild would differ between channel and floodplain ponds. This is because the predominate basal production sources are different in lotic and lentic habitats (Roach et al., 2009b). In most tropical floodplain lakes, phytoplankton and aquatic macrophyte production appear to dominate (Roach et al., 2009b), while the main channel is supported almost exclusively by phytoplankton (Cotner et al., 2006). We further hypothesized that basal resources assimilated by fish guilds would differ among floodplain ponds due to variation in geomorphological, physicochemical, and hydrological features. Although autochthonous production is predicted to be the dominant carbon source assimilated in all habitat types during the dry season, fishes from larger ponds with more persistent hydrologic connections to the active channel should have different isotopic signatures than fishes from the same guild from smaller and more isolated ponds.
Material and methods
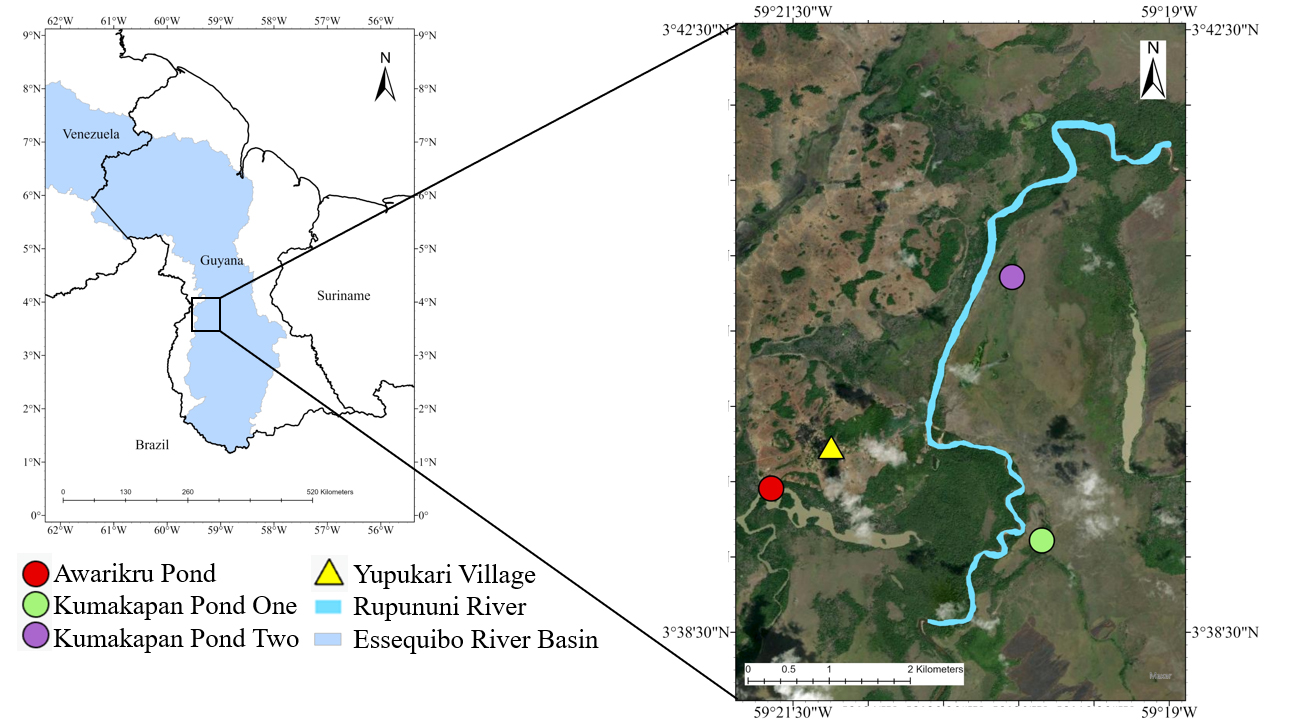
Sampling sites. This study was conducted in the Rupununi River, a lowland river with a meandering channel and a wide floodplain in the Rupununi Savanna region of Guyana (Taphorn et al., 2022) (Fig. 1). Habitats in the main channel consist of sand bars, backwaters, and deeper areas in the mainstem containing submerged woody debris and stronger current velocities. During the dry season, the water of the main channel is clear with pH of 6.4–6.9, whereas water in floodplain ponds varies from clear to slightly turbid due to suspended particles (Lowe-McConnell, 1964). We collected fishes and basal production sources from multiple sites within a 10-km reach of the active river channel (from 03°38’36”N 59°20’20”W to 03°41’43.9”N 59°19’01.5”W) and three floodplain ponds along the same reach. Awarikru Pond and Kumakapan Pond 1 are relatively large oxbow lakes with moderate dissolved oxygen levels and relatively low abundance of submerged aquatic macrophytes. These ponds have substrates of sand and clay overlain with submerged leaf litter, which was especially abundant in the latter. Kumakapan Pond 2 is a shallow depression with a mud substrate rich in organic material and an abundance of emergent and floating aquatic macrophytes. Samples were collected at the end of the dry season, prior to and just as water levels began to rise, when the ponds were disconnected from the main river channel. This sampling time interval afforded the longest possible time for habitat spatial heterogeneity to influence the composition of local fish assemblages as well as fish diets and assimilation of material into muscle tissue.
FIGURE 1| Map depicting sampling sites, three floodplain ponds and a location in the mainstem channel of the Rupununi River within the Essequibo River Basin, Guyana.
Sampling. Organisms and tissue samples were collected from May 10–25, 2023, from the river channel and floodplain ponds, using a variety of fishing gears that included a seine net (6 x 1.8 m, 6 mm mesh), cast nets (1.8 m diameter, 2 cm mesh), dip nets (40 x 30 cm, 6 mm mesh), and angling with lures and baited hooks. Tissues of common allochthonous and autochthonous production sources were collected from areas within and adjacent to the river channel and ponds. Macrophyte samples included submerged C3 plants, C3 terrestrial plants, and C4 terrestrial grasses. Decaying leaf matter (detritus) was collected from the bottom of the river and ponds. Seston was filtered from the water column using a hand pump and 1.2 µm GF/F filters (Tab. S2). Collected fishes were euthanized with an overdose of clove oil (Fernandes et al., 2016), and specimens were identified using taxonomic keys and descriptions (e.g., van der Sleen, Albert, 2018; Taphorn et al., 2022). Each species was assigned to one of five feeding guilds using published literature in the trophic ecology of Neotropical fishes (e.g., Saul, 1975; Goulding, 1988). Algivores/detritivores (alg/detritivore) consume variable fractions of material from macrophytes, algae, and detritus, which may include associated microbial decomposers (e.g., Aphanotorulus emarginatus). Omnivores consume variable fractions of plant and animal matter (especially invertebrates) (e.g., Amblydoras affinis). Invertivores consume mostly aquatic and/or terrestrial invertebrates (e.g., Geophagus surinamensis). Piscivore/invertivore (pisc/invertivore) consume variable fractions of invertebrates and fish (e.g., Saxatilia saxatilis). Piscivores feed nearly exclusively on fish (e.g., Cichla ocellaris). Muscle tissue was obtained from the dorsolateral region of specimens. Tissue samples were placed in individual labeled plastic bags, preserved with salt (NaCl), and transported to the laboratory, where they were stored in a freezer until analysis.
Samples were processed according to methods described in Arrington, Winemiller (2002) and Winemiller et al. (2011). Tissue samples were soaked and rinsed with distilled water to ensure the salt was completely removed. Each tissue sample was then placed in a labeled scintillation glass tube and dried in a drying oven at 60 ºC for 48 hours. The dried samples were individually ground into a homogenized powder using a mortar and pestle, and then packed into 4 x 6 mm tin capsules (Costech) in portions ranging from 1.5 to 3.0 mg. The subsamples were sent to the Stable Isotope Ecology Laboratory at the University of Georgia, USA, to obtain stable isotope ratios of C and N via mass spectrometry. Results are reported as parts per thousand (‰) change from the standard material (Vienna Pee Dee Belemnite and atmospheric N2):
δX = [ Rsample=Rstandard – 1] × 103, where R = 13C/12C or 15N/14N.
We collected a total of 505 fish tissue samples for stable isotope analysis, representing five feeding guilds: 41 algivore/detritivores, 150 omnivores, 157 invertivores, 31 piscivore/invertivores, and 125 piscivores (Fig. 2). A full list of sampled species and their assigned guilds appears in Tab. 1 (Fricke et al., 2026). The mean and standard deviation of δ13C and δ15N of each feeding guild from each of the four water bodies appear in Tab. S1.
TABLE 1 | Species list and trophic guild assignment for the Rupununi River and three floodplain ponds. The taxonomic classification follows Fricke et al. (2026).
Waterbody | Order | Family | Genus and species | Trophic guild | N | References |
Rupununi River | Characiformes | Acestrorhamphidae | Moenkhausia ceros | Omnivore | 1 | Winemiller (1990) |
|
|
| Moenkhausia copei | Omnivore | 5 | Winemiller (1990) |
|
|
| Moenkhausia lepidura | Omnivore | 6 | Winemiller (1990) |
|
|
| Moenkhausia shideleri | Omnivore | 5 | Winemiller (1990) |
|
|
| Moenkhausia sp. | Omnivore | 4 |
Winemiller
(1990), |
|
| Anostomidae | Leporinus brunneus | Omnivore | 2 |
Lowe-McConnell
(1987), |
|
|
| Leporinus sp. | Omnivore | 2 |
Lowe-McConnell
(1987), |
|
| Characidae | Aphyocharax erythrurus | Omnivore | 4 | Winemiller (1990) |
|
|
| Hemigrammus vorderwinkleri | Omnivore | 8 |
Winemiller
(1990), |
|
|
| Microschemobrycon casiquiare | Invertivore | 4 |
Winemiller
(1990), |
|
|
| Odonstostilbe sp. | Omnivore | 1 | Winemiller (1990) |
|
|
| Odontostilbe gracilis | Omnivore | 10 | Winemiller (1990) |
|
|
| Poptella sp. | Omnivore | 3 | Lowe-McConnel (1987) |
Rupununi River | Characiformes | Characidae | Roeboides affinis | Pisc/Invert | 2 | Albrecht et al. (2013) |
|
|
| Tetragonopterus argenteus | Omnivore | 1 | Winemiller (1990) |
|
|
| Tetragonopterus chalceus | Omnivore | 1 | Winemiller (1990) |
|
| Crenuchidae | Characidium catenatum | Invertivore | 1 | Casatti (2002) |
|
|
| Characidium zebra | Invertivore | 1 | Casatti (2002) |
|
| Cynodontidae | Hydrolycus armatus | Piscivore | 32 | Layman et al. (2005) |
|
| Erythrinidae | Hoplerythrinus unitaeniatus | Pisc/Invert | 1 |
Belger, |
|
|
| Hoplias malabaricus | Piscivore | 2 |
Belger, |
|
| Serrasalmidae | Pristobrycon striolatus | Piscivore | 1 | Sá-Oliveira et al. (2017) |
|
|
| Pygocentrus nattereri | Piscivore | 1 | Sá-Oliveira et al. (2017) |
|
|
| Serrasalmus altispinis | Piscivore | 1 | Taphorn et al. (1997) |
|
|
| Serrasalmus eigenmanni | Piscivore | 1 | Taphorn et al. (1997) |
|
|
| Serrasalmus rhombeus | Piscivore | 13 | Taphorn et al. (1997) |
| Cichliformes | Cichlidae | Apistogramma rupununi | Invertivore | 4 | Winemiller (1990) |
|
|
| Biotodoma cupido | Invertivore | 1 | Winemiller (1990) |
|
|
| Geophagus surinamensis | Invertivore | 1 | Duque-Correa et al. (2024) |
|
|
| Guianacara sp. | Invertivore | 1 | Lujan et al. (2011) |
| Clupeiformes | Engraulidae | Anchovia sp. | Invertivore | 1 |
Duque, |
|
|
| Anchoviella sp. | Invertivore | 3 | Silva (2018) |
|
|
| Lycengraulis batesii | Invertivore | 5 | Planquette et al. (1996) |
|
|
| Lycengraulis sp. | Invertivore | 1 | Planquette et al. (1996) |
| Gymnotiformes | Rhamphichthyidae | Gymnorhamphichthys sp. | Invertivore | 2 | Evans et al. (2019) |
|
| Sternopygidae | Eigenmannia macrops | Invertivore | 1 | Evans et al. (2019) |
|
|
| Rhabdolichops sp. | Invertivore | 1 | Evans et al. (2019) |
| Pleuronectiformes | Achiridae | Apionichthys finis | Invertivore | 1 | Duarte et al. (2003) |
|
|
| Hypoclinemus mentalis | Pisc/Invert | 4 | Duarte et al. (2003) |
| Siluriformes | Auchenipteridae | Ageneiosus cf. ucayalensis | Piscivore | 1 | Dary et al. (2017) |
|
|
| Ageneiosus inermis | Piscivore | 16 | Dary et al. (2017) |
|
| Callichthyidae | Corydoras sp. | Invertivore | 4 | Sazima (1986) |
|
|
| Megalechis thoracata | Omnivore | 1 | Sá-Oliveira et al. (2017) |
|
| Doradidae | Amblydoras affinis | Omnivore | 1 |
Arce, |
|
|
| Doras sp. | Invertivore | 4 |
Arce, |
|
|
| Oxydoras morei (Hemidoras) | Invertivore | 4 |
Arce, |
|
|
| Oxydoras niger | Omnivore | 2 |
Arce, |
|
|
| Platydoras costatus | Omnivore | 2 |
Arce, |
|
|
| Platydoras hancocki | Omnivore | 5 |
Arce, |
Rupununi River | Siluriformes | Loricariidae | Platydoras nattereri | Omnivore | 1 |
Arce, |
|
|
| Aphanotolurus emarginatus | Detrit/Algiv | 7 | Dary et al. (2017) |
|
|
| Farlowella nattereri | Detrit/Algiv | 1 | Dary et al. (2017) |
|
|
| Limatulichthys griseus | Detrit/Algiv | 1 | Dary et al. (2017) |
|
|
| Rineloricaria sp. | Detrit/Algiv | 2 | Dary et al. (2017) |
|
| Pimelodidae | Pimelodella cristata | Invertivore | 3 | Lowe-McConnell (1987) |
|
|
| Pimelodus blochii | Invertivore | 10 | Lima et al. (2023) |
|
|
| Pseudoplatystoma fasciatum | Piscivore | 9 | Dary et al. (2017) |
|
|
| Sorubim sp. | Piscivore | 11 | Dary et al. (2017) |
|
| Pseudopimelodidae | Hemisorubim platyrhynchos | Pisc/Invert | 7 | Erard et al. (2002) |
Awarikru Pond | Acanthuriformes | Sciaenidae | Pachypops sp. | Pisc/Invert | 1 | Lowe-McConnell (1987) |
|
|
| Plagioscion squamosissimus | Pisc/Invert | 2 | Neves et al. (2015) |
| Characiformes | Acestrorhamphidae | Moenkhausia copei | Omnivore | 5 | Winemiller (1990) |
|
|
| Moenkhausia sp. | Omnivore | 3 |
Winemiller
(1990), |
|
| Acestrorhynchidae | Acestrorhynchus falcirostris | Piscivore | 1 |
Dary
et
al. (2017), |
|
|
| Acestrorhynchus microlepis | Piscivore | 1 | Dary et al. (2017) |
|
| Anostomidae | Leporinus ortomaculatus | Omnivore | 1 |
Vari
(1992), |
|
|
| Pseudanos sp. | Omnivore | 1 |
Vari
(1992), |
|
| Characidae | Aphyocharax erythrurus | Omnivore | 1 | Winemiller (1990) |
|
| Chilodontidae | Caenotropus maculosus | Detrit/Algiv | 2 | Vari et al. (1986) |
|
| Curimatidae | Curimata sp. | Detrit/Algiv | 1 |
Castro, |
|
|
| Psectrogaster sp. | Detrit/Algiv | 1 | Correa (2015) |
|
| Prochilodontidae | Prochilodus rubrotaeniatus | Detrit/Algiv | 1 |
Duque-Correa
et
al.
(2024), |
|
| Serrasalmidae | Catoprion mento | Piscivore | 3 | Kolmann et al. (2018) |
|
|
| Serrasalmus eigenmanni | Piscivore | 1 | Taphorn et al. (1997) |
| Cichliformes | Cichlidae | Biotodoma cupido | Invertivore | 7 | Winemiller (1990) |
|
|
| Cichla ocellaris | Piscivore | 6 | Jepsen et al. (1997) |
|
|
| Lugubria acutirostris | Pisc/Invert | 1 | Winemiller (1990) |
|
|
| Lugubria lugubris | Pisc/Invert | 3 | Winemiller (1990) |
|
|
| Mesonauta insignis | Omnivore | 1 | Winemiller (1990) |
|
|
| Satanoperca leucostica | Invertivore | 3 | Winemiller (1990) |
| Clupeiformes | Engraulidae | Amazonsprattus scintilla | Invertivore | 1 | Silva (2018) |
|
|
| Anchoviella sp. | Invertivore | 1 | Silva (2018) |
| Siluriformes | Auchenipteridae | Trachelyopterus galeatus | Invertivore | 1 | Dary et al. (2017) |
|
| Doradidae | Platydoras costatus | Omnivore | 1 |
Arce, |
|
|
| Platydoras nattereri | Omnivore | 1 |
Arce, |
|
| Loricariidae | Pseudacanthicus sp. | Omnivore | 1 | Dary et al. (2017) |
|
|
| Rineloricaria sp. | Detrit/Algiv | 1 | Dary et al. (2017) |
|
| Pimelodidae | Pimelodus blochii | Pisc/Invert | 3 | Lima et al. (2023) |
|
|
| Pseudoplatystoma fasciatum | Piscivore | 2 | Dary et al. (2017) |
Kumakapan Pond One | Characiformes | Acestrorhamphidae | Moenkhausia sp. | Omnivore | 5 |
Winemiller
(1990), |
|
| Characidae | Aphyocharax erythrurus | Omnivore | 5 | Winemiller (1990) |
|
|
| Ctenobrychon spilurus | Omnivore | 4 | Winemiller (1990) |
|
|
| Hemigrammus sp. | Omnivore | 1 |
Winemiller
(1990), |
|
|
| Hemigrammus vorderwinkleri | Omnivore | 5 |
Winemiller
(1990), |
|
|
| Moenkhausia sp. | Omnivore | 5 |
Winemiller
(1990), |
|
|
| Tetragonopterus argenteus | Omnivore | 1 | Winemiller (1990) |
|
| Curimatidae | Curimatella immaculata | Detrit/Algiv | 7 |
Duque-Correa
et
al.
(2024), |
|
| Erythrinidae | Hoplias malabaricus | Piscivore | 2 | Belger et al. (2006) |
|
| Serrasalmidae | Pygopristis denticulatus | Piscivore | 1 | Sá-Oliveira et al. (2017) |
|
|
| Pygopristis sp. | Piscivore | 1 | Sá-Oliveira et al. (2017) |
| Cichliformes | Cichlidae | Aequidens tetramerus | Omnivore | 1 | Winemiller (1990) |
| Gymnotiformes | Hypopomidae | Brachyhypopomus sp. | Invertivore | 4 | Evans et al. (2019) |
| Siluriformes | Callichthyidae | Callichthys callichthys | Omnivore | 3 | Sazima (1986) |
|
|
| Corydoras sp. | Invertivore | 1 | Sazima (1986) |
|
|
| Corydoras sp. | Invertivore | 1 | Sazima (1986) |
|
| Doradidae | Amblydoras affinis | Omnivore | 10 |
Arce, |
|
|
| Farlowella sp. | Detrit/Algiv | 1 |
Arce, |
|
| Loricariidae | Hypostomus sp. | Detrit/Algiv | 2 | Power (1984) |
|
|
| Loricaria sp. | Detrit/Algiv | 7 | Power (1984) |
|
|
| Loricariichthys sp. | Omnivore | 1 | Power (1984) |
|
|
| Rineloricaria sp. | Detrit/Algiv | 2 | Power (1984) |
|
| Pimelodidae | Pimelodella sp. | Invertivore | 1 | Dary et al. (2017) |
|
|
| Pseudoplatystoma fasciatum | Piscivore | 1 | Dary et al. (2017) |
Kumakapan Pond Two | Characiformes | Characidae | Aphyocharax erythrurus | Omnivore | 5 | Winemiller (1990) |
|
|
| Ctenobrycon spilurus | Omnivore | 5 | Winemiller (1990) |
|
|
| Hemigrammus sp. | Omnivore | 5 |
Winemiller
(1990), |
|
|
| Hyphessobrycon cf. minor | Omnivore | 5 | Winemiller (1990) |
Kumakapan Pond Two | Characiformes | Curimatidae | Curimatella immaculata | Detrit/Algiv | 1 |
Duque-Correa
et
al.
(2024), |
|
| Erythrinidae | Hoplias malabaricus | Piscivore | 10 | Belger et al. (2006) |
|
| Serrasalmidae | Megalechis thoracata | Omnivore | 1 | Sá-Oliveira et al. (2017) |
|
|
| Mesonauta insignis | Omnivore | 2 | Sá-Oliveira et al. (2017), |
| Cichliformes | Cichlidae | Acaronia nassa | Pisc/Invert | 5 | Lujan et al. (2011) |
|
|
| Apistogramma sp. | Invertivore | 5 | Winemiller (1990) |
|
|
| Cichlasoma bimaculatum | Omnivore | 5 | Winemiller (1990) |
|
|
| Saxatilia saxatilis | Pisc/Invert | 1 | Winemiller (1990) |
| Gymnotiformes | Hypopomidae | Brachyhypopomus sp. | Invertivore | 26 | Evans et al. (2019) |
|
|
| Hypopygus sp. | Invertivore | 2 | Evans et al. (2019) |
| Osteoglossiformes | Osteoglossidae | Osteoglossum bicirrhosum | Pisc/Invert | 4 | Lowe-McConnel (1987) |
| Siluriformes | Loricariidae | Hypoptopoma guianense | Detrit/Algiv | 3 |
Arce, |
|
|
| Hypoptopoma sp. | Detrit/Algiv | 1 |
Arce, |
|
| Pseudopimelodidae | Microglanis poecilus | Invertivore | 1 | Winemiller (1990) |
| Synbranchiformes | Synbranchidae | Synbranchus marmoratus | Invertivore | 3 | Galvis et al. (1997) |
FIGURE 2| Biplot of δ13C and δ15N ‰ isotopic ratios for fishes and basal resources collected in May 2023 from Rupununi River and three floodplain ponds near the Yupukari Village, Guyana.
Statistical analyses. We employed Bayesian mixing models using the MixSIAR package (Stock et al., 2018; v. 3.1.12) in R to estimate the proportional contributions of basal sources to consumer biomass. Model inputs included δ¹³C and δ¹⁵N values from fish tissue and representative basal sources at each site. Basal sources were assigned to functional groups to reduce the total number of sources and improve model resolution and interpretability (Moore, Semmens, 2008; Arantes et al., 2019) (Tab. S2). A conservative estimate of trophic enrichment for primary consumers is approximately 3.4‰ for δ¹⁵N (Post, 2002; Stephans et al., 2023). The highest δ¹⁵N value observed for a primary consumer in this study was 12.19‰; therefore, any basal sources with δ¹⁵N signatures greater than 8.79‰ were excluded from the mixing model because their δ¹⁵N signatures were too high to be assimilated in consumers. Similarly, C4-derived enrichment averages about 1.0‰ for δ¹³C, and all C4 grasses were excluded because their δ¹³C values were more than 10‰ heavier than the lightest primary consumer (Fig. 2). Several studies have shown that C4 macrophytes contribute minimally to fish biomass in Neotropical rivers (Benedito-Cecilio, 2000; Jepsen, Winemiller, 2002, 2007; Arantes et al., 2019).
Previous studies have emphasized the importance of periphyton (e.g., microphytobenthos and biofilm) for tropical river food webs during low-water periods (Roach et al., 2014; Roach, Winemiller 2015; Ou, Winemiller, 2016), we were unable to find sufficient periphyton biomass (biofilm) for analysis, possibly because stocks had been grazed down by consumers (Winemiller et al., 2006, 2014). After removing isotopic outliers and aggregating basal sources based on functional similarity, we included three primary basal resources in mixing models for fishes from each of the three ponds. These included C3 aquatic macrophytes (mean δ¹³C: -31.9 ± 1.9‰; δ¹⁵N: 5.02 ± 0.01‰), C3 terrestrial plants with detritus (coarse particulate organic matter, CPOM) included due to isotopic similarity (δ¹³C: -31.0 ± 1.83‰; δ¹⁵N: 3.59 ± 1.53‰), and seston, which is assumed to be a mixture of phytoplankton and fine particulate organic matter (FPOM) (δ¹³C: -35.9 ± 1.96‰; δ¹⁵N: 5.9 ± 0.45‰). For the river channel site, only two basal resources were included in the model: C3 terrestrial plants (δ¹³C: -30.2 ± 1.49‰; δ¹⁵N: 3.85 ± 2.1‰) and seston (δ¹³C: -30.6 ± 2.86‰; δ¹⁵N: 5.8 ± 0.31‰). C3 aquatic macrophytes were not included in river models because they were not encountered at any river locations during our field work.
To account for isotopic fractionation between trophic levels, we applied trophic discrimination factors (TDFs) appropriate to each guild. For algivore/detritivores, we used a TDF of 3.4‰ for δ¹⁵N and 1.0‰ for δ¹³C (Post, 2002). For carnivores (invertivores, piscivore/invertivores, and piscivores), we used TDFs of 5.0‰ for δ¹⁵N and 2.0‰ for δ¹³C, reflecting the average δ¹⁵N values across two trophic transfers (the TDF for δ¹⁵N was 3.4‰ from basal resource to primary consumer, and was 1.6‰ from primary consumer to secondary consumer; the TDF for δ¹³C was assumed to be constant between resource and consumer at each trophic level) (Madigan et al., 2012; Hussey et al., 2014). Omnivores were assigned intermediate TDF values of 4.25 ± 0.33‰ for δ¹⁵N and 1.5 ± 0.33‰ for δ¹³C, with variability included to account for varying basal resource and primary consumer assimilation (Kopf et al., 2025). We used uninformative priors, included only process error, and ran three Markov chains of 100,000 iterations each, with a burn-in of 50,000 and a thinning interval of 50. Model convergence was assessed using Gelman-Rubin and Geweke diagnostics. Final estimates of source contributions to species and each feeding guild were summarized as the mode of the posterior distributions (Kaymak et al., 2023).
Results
Assimilation of basal production sources. Assimilation of material originating from alternative basal production sources varied among trophic guilds, but there were differences between the river channel and ponds in the floodplain (Tab. 2). In the river channel, seston was the most important basal source supporting algivore/detritivores and omnivores (mean estimate from mixing model probability distribution >95%). Terrestrial plants were increasingly important for fishes at higher trophic levels. Invertivores assimilated approximately 56% material originating from seston and 44% from C3 terrestrial plants. Piscivore/invertivores predominantly assimilated material originating from C3 terrestrial plants (~78%), and most piscivores appeared to exploit food chains with C3 terrestrial plants at the base (>97%).
TABLE 2 | Mixing model estimates for proportions of basal production sources assimilated (mean ±1 SD) by fish trophic guilds in the Rupununi River and three floodplain ponds.
Water body | Feeding guild | C3 Aquatic plants | C3 Terrestrial plants | Seston |
Rupununi River | Algil/ Detritivore |
| 0.04 ± 0.11 | 0.96 ± 0.11 |
Omnivores |
| 0.00 ± 0.01 | 1.00 ± 0.01 | |
Invertivores |
| 0.44 ± 0.06 | 0.56 ± 0.06 | |
Pisc/Invert |
| 0.78 ± 0.11 | 0.22 ± 0.11 | |
Piscivores |
| 0.98 ± 0.05 | 0.02 ± 0.05 | |
Awarikru Pond | Algil/ Detritivore | 0.01 ± 0.05 | 0.01 ± 0.02 | 0.98 ± 0.05 |
Omnivores | 0.64 ± 0.20 | 0.01 ± 0.03 | 0.35 ± 0.18 | |
Invertivores | 0.13 ± 0.12 | 0.64 ± 0.14 | 0.23 ± 0.15 | |
Pisc/Invert | 0.01 ± 0.01 | 0.04 ± 0.05 | 0.86 ± 0.10 | |
Piscivores | 0.09 ± 0.08 | 0.03 ± 0.04 | 0.89 ± 0.08 | |
Kumakapan 1 | Algil/ Detritivore | 0.01 ± 0.04 | 0.01 ± 0.05 | 0.98 ± 0.07 |
Omnivores | 0.90 ± 0.10 | 0.00 ± 0.01 | 0.10 ± 0.10 | |
Invertivores | 0.11 ± 0.12 | 0.50 ± 0.14 | 0.34 ± 0.17 | |
Pisc/Invert |
|
|
| |
Piscivores | 0.04 ± 0.06 | 0.01 ± 0.02 | 0.94 ± 0.07 | |
Kumakapan 2 | Algil/ Detritivore | 0.01 ± 0.04 | 0.01 ± 0.03 | 0.98 ± 0.05 |
Omnivores | 0.80 ± 0.12 | 0.01 ± 0.03 | 0.19 ± 0.11 | |
Invertivores | 0.37 ± 0.16 | 0.52 ± 0.12 | 0.10 ± 0.08 | |
Pisc/Invert | 0.42 ± 0.14 | 0.05 ± 0.06 | 0.53 ± 0.12 | |
Piscivores | 0.41 ± 0.15 | 0.03 ± 0.04 | 0.56 ± 0.14 |
Estimated proportions of material from basal production sources assimilated by trophic guilds differed not only between ponds and the river channel, but in some cases varied among ponds (Tab. 2). In all three ponds and similar to the river channel, algivore/detritivores assimilated material from seston almost exclusively (mean estimate 98%). Omnivores primarily assimilated material from C3 aquatic macrophytes, though estimated proportions varied among ponds. In Kumakapan Pond 1, omnivores assimilated material from C3 aquatic plants almost exclusively (~90%). In Awarikru and Kumakapan Pond 2, material from seston made larger contributions to omnivore biomass (~35% and ~19%, respectively) compared to omnivores in Kumakapan Pond 1. C3 terrestrial plants contributed between 50% and 67% of the material assimilated by invertivores in ponds. The estimated importance of other basal production sources for invertivores varied by pond; seston was relatively important in Awarikru and Kumakapan Pond 1, and C3 aquatic plants were relatively more important in Kumakapan Pond 2. Piscivores/invertivores and invertivores showed similar proportional assimilation estimates within each pond, but these estimates varied among ponds. In Awarikru and Kumakapan Pond 1, these two guilds were estimated to assimilate material originating from seston almost exclusively (>85%). In contrast, in Kumakapan Pond 2, piscivore/invertivores and piscivores assimilated nearly equal proportions of material from C3 aquatic plants and seston (~41% and ~55%, respectively).
Discussion
Seston was estimated to be the primary carbon assimilated by about half of the trophic guilds across the habitats surveyed, consistent with previous research in tropical floodplain rivers (Araujo-Lima et al., 1986; Benedito-Cecilio et al., 2000; Lewis et al., 2001), especially during the dry season (Roach et al., 2009b; Ou, Winemiller, 2016). Our study was conducted at the end of the dry season at the start of annual rains, the period associated with lowest water levels. At these low water levels, water body shorelines have receded away from riparian vegetation, which presumably reduces inputs of allochthonous food resources (plant material and invertebrates) to aquatic food webs. The quality of available allochthonous resources also may be reduced during the dry season, if many plants are not producing flowers, fruits or seeds. In the Cinaruco River, Venezuela, aquatic ecosystem primary production was highest during the annual low-water period in both the river channel and floodplain lakes (Cotner et al., 2006; Montoya et al., 2006). Other studies in Neotropical rivers have also documented the importance of phytoplankton biomass within floodplain lakes during low-water periods (Rai, Hill, 1984; Putz, Junk, 1997). However, isotopic signatures of seston do not necessarily reflect those of phytoplankton in rivers and floodplains (Arantes et al., 2019), and seston (suspended fine particulate organic matter) in these systems likely contains variable fractions of detritus, microbial decomposers, and phytoplankton). In the river channel and the three floodplain ponds, algivore/detritivores assimilated material almost entirely from seston, as did omnivores captured from the active channel. Much of this material likely was ingested directly by benthivorous fishes that graze FPOM (seston) that settles from the water column and accumulates on substrates (Montoya et al., 2006; Winemiller et al., 2006). Seston was estimated to be of relatively low importance for omnivorous fishes in Kumakapan ponds 1 and 2, where material originating from aquatic macrophytes had greater importance in supporting their biomass during the dry season. Seston was estimated to have relatively low importance supporting biomass of invertivores in all three ponds. Terrestrial plants were the most important basal production source supporting invertivore biomass in ponds. This was likely due to consumption of terrestrial invertebrates by certain invertivorous fishes, such as anchovies (Engraulidae) as well as benthivorous invertivores (e.g., Apistogramma, Characidium, Geophagus, Satanoperca, Pimelodella) consuming aquatic insects that assimilated detritus of allochthonous origin. Invertivous fishes in the river channel assimilated material from terrestrial plants and seston in similar proportions.
Piscivore/invertivores tended to reflect basal source assimilation proportions of piscivores more closely than invertivores. In the river channel, both of these trophic guilds were estimated to have assimilated much larger fractions of terrestrial sources compared to guilds at lower trophic levels. This disparity could result from a time lag for assimilation of material originating from basal production sources to pass through several steps along food chains (Tieszen et al.,1983; O’Reilly et al., 2004; Thomas, Crowther, 2015). At the time of our study, the floodplain ponds had been isolated for several months, presumably sufficient time for fishes to assimilate in situ basal production sources. During the wet season, many Neotropical fishes enter flooded riparian areas to feed directly on allochthonous food resources, such as seeds, fruits, flowers, leaves, and terrestrial invertebrates (Goulding, 1980; Correa, Winemiller, 2018). If one assumes an elemental turnover rate of approximately 1–2 months for fish muscle tissue (C half-life ~ 26 days, N half-life ~ 35 days; Mont’Alverne et al., 2016), then it might take 3–6 months for a significant amount of material from basal production sources to pass through 2–3 trophic levels to become assimilated within piscivore biomass. In contrast, herbivores and omnivores at low trophic positions should reflect a history of feeding and growth over the previous few weeks, which in our study would fall entirely within the dry season. Although tissue elemental turnover rate is affected by several factors, including body mass and temperature (Thomas, Crowther, 2015), our findings for the river channel are consistent with this time-lag hypothesis; estimates for assimilated material derived from terrestrial plants were greater for fishes at higher trophic levels. Interestingly, the same pattern was not found for piscivores in ponds, where seston was estimated to be most important, along with aquatic macrophytes in Kumakapan Pond 2, a shallow pond with high density of aquatic macrophytes.
Our study design carries three assumptions: 1) fish samples reliably represented local assemblages, 2) fishes were resident in the habitat long enough for their isotopic signatures to reflect assimilation of material from local resources, and 3) samples were obtained for all important basal sources with sufficient isotopic discrimination for estimating proportional assimilation. With regards to the first two assumptions, our study included several species known to be migratory,such as Hydrolycus scomberoides, thatcould have recently moved into the channel location from an upstream or downstream reach. This seems plausible with respect to the river channel, but seems unlikely for ponds that had been isolated from the channel for several months before we sampled them.
Our third assumption has greater uncertainty. A limitation of isotopic mixing models is that the inclusion of multiple sources reduces the ability to discriminate their contributions to consumer biomass with precision. This problem is compounded when sources overlap in their isotopic ratios (Moore, Semmons, 2008). To limit the number of sources used as inputs for the models, we excluded C4 grasses because their δ13C was much higher than any of the fishes, and previous research has shown that few Neotropical fishes appear to assimilate appreciable amounts of material derived from these grasses (Benedito-Cecilio, 2000; Jepsen, Winemiller, 2002, 2007; Arantes et al., 2019). We attempted to obtain samples of common basal production sources at each survey site, and our assessments were based on visual inspection of submerged substrates, vegetation, leaf litter and other forms of detritus in littoral and riparian zones. Nonetheless, some important sources could have been missed. Other studies have revealed evidence for the importance of microphytobenthos (biofilms), periphyton, and particulate organic matter (POM) in tropical river food webs (Winemiller, 1990; Wantzen et al., 2002; Roach, Winemiller, 2015; Ou, Winemiller, 2016; Arantes et al., 2019). Visible stands of microphytobenthos or periphyton were not present in the habitats we sampled, and POM in our seston samples could have included variable fractions of phytoplankton and detritus of either aquatic or terrestrial origin. Interestingly, a few of the fish specimens we collected had extremely low δ13C values (<-40‰), values commonly associated with methanogenesis in microbial communities (Qin et al., 2020). Six of these fishes were algivore/detritivores in the families Curimatidae and Loricariidae, and three others were planktivorous anchovies (Amazonsprattus, Anchoviella) with high trophic positions (high δ15N) that likely fed from food chains that include methanogenic bacteria at their base. We also cannot rule out fractions of phytoplankton with low δ13C in our seston samples, and the relative influence of phytoplankton vs detritus on isotopic signatures of bulk seston samples is vexing problem (Marty, Planas, 2008; Karlsson et al., 2014).
In conclusion, stable isotope analysis revealed variation in estimates of assimilated material from basal production sources both between-guilds within a habitat and within-guilds among habitats of the Rupununi River during the dry season. Our findings support previous research that autochthonous primary production broadly supports tropical river food webs during the low-water phase of the seasonal flood cycle. For some guilds within certain habitats, terrestrial or aquatic vegetation was estimated to more important basal sources supporting fish biomass, however, the influence of time lags for element assimilation in muscle tissue cannot be discounted for fishes at high trophic levels. Isotopic differences of individual trophic guilds between floodplain ponds were associated with differences in floodplain pond water depth and the density of aquatic vegetation. Future research on food webs of tropical rivers should further examine the relationship between floodplain heterogeneity and community trophic and food web structures.
Acknowledgments
The authors thank Elford Liverpool at the University of Guyana for logistical support. We also thank Ashley Holland, Megan Beeksma, Sophie Scott, Kendall Hastings, Sam Grinstead, Akeem Sinclair, Chelbie Gilkes, Takiya Jackson, Lily Williams, James Vankley, and Donald Burt for assistance during field sampling and sample processing.
References
Albrecht MP, Reis VCS, Caramaschi EP. Resource use by the facultative lepidophage Roeboides affinis (Günther, 1868): a comparison of size classes, seasons and environment types related to impoundment. Neotrop Ichthyol. 2013; 11(2):387–94. https://doi.org/10.1590/S1679-62252013005000007
Arantes CC, Winemiller KO, Petrere M, Freitas CEC. Spatial variation in aquatic food webs in the Amazon River floodplain. Freshw Sci. 2019; 38(1):213–28. https://doi.org/10.1086/701841
Arce H M, Birindelli JLO. The state of the art of the Doradidae. In: Arratia G, Reis RE, editors. Catfishes, a highly diversified group: evolution and phylogeny. Vol. 2. United Kingdom: Routledge; 2025.
Arrington DA, Winemiller KO. Preservation effects on stable isotope analysis of fish muscle. T Am Fish Soc. 2002; 131(2):337–42. https://doi.org/10.1577/1548-8659(2002)131%3C0337:PEOSIA%3E2.0.CO;2
Baldasso MC, Wolff LL, Neves MP, Delariva RL. Ecomorphological variations and food supply drive trophic relationships in the fish fauna of a pristine Neotropical stream. Environ Biol Fishes. 2019; 102:783–800. https://doi.org/10.1007/s10641-019-00871-w
Belger L, Forsberg BR. Factors controlling mercury levels in two predatory fish species from the Amazon. Sci Total Environ. 2006; 367(1):451–59. https://doi.org/10.1016/j.scitotenv.2006.03.033
Benedito-Cecilio E, Araujo-Lima CARM, Forsberg BR, Bittencourt MM, Martinelli LC. Carbon sources of Amazonian fisheries. Fish Manag Ecol. 2000; 7(4):305–14. https://doi.org/10.1046/j.1365-2400.2000.007004305.x
Boecklen WJ, Yarnes CT, Cook BA, James AC. On the use of stable isotopes in trophic ecology. Annu Rev Ecol Evol Syst. 2011; 42:411–40. https://doi.org/10.1146/annurev-ecolsys-102209-144726
Bowen SH. Detritivory in neotropical fish communities. Environ Biol Fishes. 1983; 9(2):137–44. https://doi.org/10.1007/BF00690858
Casatti L. Alimentação dos peixes em um riacho do Parque Estadual Morro do Diabo, bacia do Alto Rio Paraná, sudeste do Brasil. Biota Neotrop. 2002; 2(2):1–14. https://doi.org/10.1590/S1676-06032002000200012
Castro RMC, Vari RP. Detritivores of the South American fish family Prochilodontidae (Teleostei: Ostariophysi: Characiformes): a phylogenetic and revisionary study. Smithson Contrib Zool. 2004; 622. http://dx.doi.org/10.5479/si.00810282.622
Correa SB, Winemiller K. Terrestrial-aquatic trophic linkages support fish production in a tropical oligotrophic river. Oecologia. 2018; 186(4):1069–78. https://doi.org/10.1007/s00442-018-4093-7
Correa SB, Costa-Pereira R, Fleming T, Goulding M, Anderson JT. Neotropical fish-fruit interactions: eco-evolutionary dynamics and conservation. Biol Rev. 2015; 90(4):1263–78. https://doi.org/10.1111/brv.12153
Cotner JB, Montoya JV, Roelke DL, Winemiller KO. Seasonally variable riverine production in the Venezuelan llanos. J N Am Benthol Soc. 2006; 25(1):171–84. https://doi.org/10.1899/0887-3593(2006)25[171:SVRPIT]2.0.CO;2
Dary EP, Ferreira E, Zuanon J, Röpke CP. Diet and trophic structure of the fish assemblage in the mid-course of the Teles Pires River, Tapajós River basin, Brazil. Neotrop Icthyol. 2017; 15(4):e160173. https://doi.org/10.1590/1982-0224-20160173
Duarte GAS, Andreata JV. Hábito alimentar das espécies de Achiridae e Cynoglossidae que ocorrem na Baía da Ribeira, Angra dos Reis, Rio de Janeiro, Brasil. Bioikos. 2003; 17(1–2):39–48. Available from: https://periodicos.puc-campinas.edu.br/bioikos/article/view/893
Duque G, Acero AP. Food habitats of Anchovia clupeoides (Pisces: Engraulidae) in the Ciénaga Grande de Santa Marta, Colombian Caribbean. Gulf Mex Sci. 2003; 21(1):1–09. https://doi.org/10.18785/goms.2101.01
Duque-Correa MJ, Clements KD, Meloro C, Ronco F, Boila A, Indermaur A et al. Diet and habitat as determinants of intestine length in fishes. Rev Fish Biol Fish. 2024; 34:1017–34. https://link.springer.com/article/10.1007/s11160-024-09853-3
Erard C, Le Bail KP, Planquette P. Atlas des poissons d’eau douce de Guyane. Tome 2, fascicule I. Batrachoidiformes, Mugiliformes, Beloniformes, Cyprinodontif ormes, Synbranchifor-mes, Percif ormes, Pleuronectiformes, Tetraodontiformes. Patrimoines naturels, 43 (I). Paris, Muséum national d’Histoire naturelle, Service du Patrimoine naturel; 2000.
Evans KM, Kim LY, Schubert BA, Albert JS. Ecomorphology of Neotropical electric fishes: an integrative approach to testing the relationships between form, function, and trophic ecology. Integr Org Biol. 2019; 1(1):obz015. https://doi.org/10.1093/iob/obz015
Fernandes IM, Bastos YF, Barreto DS, Lourenço LS, Penha JM. The efficacy of clove oil as an anesthetic and in euthanasia procedures for small-sized tropical fishes. Braz J Biol. 2016; 77(3):444–50. https://doi.org/10.1590/1519-6984.15015
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2026. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Galvis G, Mojica JI, Camargo M. Peces del Catatumbo. Asociación Cravo Norte; 1997.
Goulding M. The fishes and the forest: explorations in Amazonian Natural History. Berkeley: University of California Press; 1980.
Hopkins JB, Ferguson JM. Estimating the diets of animals using stable isotopes and a comprehensive Bayesian mixing model. PLoS ONE. 2012; 7(1):e28478. https://doi.org/10.1371/journal.pone.0028478
Hussey NE, MacNeil MA, McMeans BC, Olin JA, Dudley SFJ, Cliff G et al. Rescaling the trophic structure of marine food webs. Ecol Lett. 2014; 17(2):239–50. https://doi.org/10.1111/ele.12226
Jepsen DB, Winemiller KO. Structure of tropical river food webs revealed by stable isotope ratios. Oikos. 2002; 96(1):46–55. https://doi.org/10.1034/j.1600-0706.2002.960105.x
Jepsen DB, Winemiller KO. Basin geochemistry and isotopic ratios of fishes and basal production sources in four neotropical rivers. Ecol Freshw Fish. 2007; 16(3):267–81. https://doi.org/10.1111/j.1600-0633.2006.00218.x
Jepsen DB, Winemiller KO, Taphorn DC. Temporal patterns of resource partitioning among Cichla species in a Venezuelan blackwater river. J Fish Biol. 1997; 51(6):1085–108. https://doi.org/10.1111/j.1095-8649.1997.tb01129.x
Junk WJ, Bayley PB, Sparks RE. The flood pulse concept in river-floodplain systems. In: Dodge DP, editor. Proceedings of the international large rivers symposium. Can J Fish Aquat Sci. 1989; 106:110–27.
Karlsson J, Berggren M, Ask J, Byström P, Jonsson A, Laudon H et al. Response to comment: terrestrial support of pelagic consumers in unproductive lakes-uncertainty and potential in assessments using stable isotopes. Limnol Oceanogr. 2014; 59(5):1800–03. https://doi.org/10.4319/lo.2014.59.5.1800
Kaymak N, Emre N, Yalim FB, Toslak C, Emre Y, Akin Ş. Seasonal variation in trophic niches and niche overlap between native and introduced cyprinid fishes. Spectrosc Lett. 2023; 56(4):227–37. https://doi.org/10.1080/00387010.2023.2202229
Kolmann MA, Huie JM, Evans K, Summers AP. Specialized specialists and the narrow niche fallacy: a tale of scale-feeding fishes. R Soc Open Sci. 2018; 5(1):171581. https://doi.org/10.1098/rsos.171581
Kopf RK, McPhan L, Mclnerney PJ, Zampatti B, Thiem J, Koster W et al. Intraspecific body size determines isotopic trophic structure of a large river fish community. J Anim Ecol. 2025; 94(7):1435–48. https://doi.org/10.1111/1365-2656.70069
Layman CA, Araujo MS, Boucek R, Hammerschlag-Peyer CM, Harrison E, Jud ZR et al. Applying stable isotopes to examine food-web structure: an overview of analytical tools. Biol Rev. 2012; 87(3):545–62. https://doi.org/10.1111/j.1469-185X.2011.00208.x
Layman CA, Winemiller KO. Patterns of habitat segregation among large fishes in a tropical floodplain river. Neotrop Ichthyol. 2005; 3(1):103–09. https://doi.org/10.1590/S1679-62252005000100007
Lewis WM, Hamilton SK, Rodríguez MA, Saunders JF, Lasi MA. Foodweb analysis of the Orinoco floodplain based on production estimates and stable isotope data. J N Am Benthol Soc. 2001; 20(2):241–54. https://doi.org/10.2307/1468319
Lima MS, Martins SS, Montag LFA, Freitas TMS. Dietary shift of a pimelodid catfish in response to the flood pulse in the Xingu River. Neotrop Ichthyol. 2023; 21(4):e230097. https://doi.org/10.1590/1982-0224-2023-0097
Lowe-McConnell RH. Ecological studies in tropical fish communities. Cambridge: Cambridge Univ Press; 1987.
Lowe-McConnell RH. The fishes of the Rupununi Savana district of British Guiana, South America. Part 1. Ecological groupings of fish species and effects of the seasonal cycle on the fish. Zool J Linn Soc-Lond. 1964; 45(304):103–44.
Lubich C, Aguiar-Santos J, Corrêa F, Freitas CEC, Siqueira-Souza FK. Trophic ecology of Acestrorhynchus falcirostris in island lakes on the lower Solimões. Braz J Biol. 2022; 84:e252062. https://doi.org/10.1590/1519-6984.253852
Lujan NK, Winemiller KO, Arbour J. Ecomorphology of Guianacara spp. Environ Biol Fishes. 2011; 90:367–80. https://doi.org/10.1007/s10641-010-9751-8
Madigan DJ, Litvin SY, Popp BN, Carlisle AB, Farwell CJ, Block BA. Tissue turnover rates and isotopic trophic discrimination factors in the endothermic Teleost, Pacific Bluefin Tuna (Thunnus orientalis). PLoS ONE. 2012; 7(11):e49220. https://doi.org/10.1371/journal.pone.0049220
Marty J, Planas D. Comparison of methods to determine algal d13C in freshwater. Limnol Oceanogr Methods. 2008; 6:51–63. Available from: https://aslopubs.onlinelibrary.wiley.com/doi/pdf/10.4319/lom.2008.6.51
Montaña CG, Layman CA, Winemiller KO. Gape size influences seasonal patterns of piscivore diets in three Neotropical rivers. Neotrop Ichthyol. 2011; 9(3):647–55. https://doi.org/10.1590/S1679-62252011005000028
Mont’Alverne R, Jardine TD, Pereyra PER, Oliveira MCLM, Medeiros RS, Sampaio LA et al. Elemental turnover rates and isotopic discrimination in a euryhaline fish reared under different salinities: implications for movement studies. J Exp Mar Biol Ecol. 2016; 480:36–44. https://doi.org/10.1016/j.jembe.2016.03.021
Montoya JV, Roelke DL, Winemiller KO, Cotner JB, Snider JA. Hydrological seasonality and benthic algal biomass in a Neotropical floodplain river. J N Am Benthol Soc. 2006; 25(1):157–70. https://doi.org/10.1899/0887-3593(2006)25[157:HSABAB]2.0.CO;2
Moore JW, Semmens BX. Incorporating uncertainty and prior information into stable isotope mixing models. Ecol Lett. 2008; 11(5):470–80. https://doi.org/10.1111/j.1461-0248.2008.01163.x
Neves MP, Delariva RL, Guimarães ATB, Sanches PV. Carnivory during ontogeny of Plagioscion squamosissimus: a successful non-native fish lentic environment of the upper Paraná River basin. PLoS ONE. 2015; 10(11):e0141651. https://doi.org/10.1371/journal.pone.0141651
Ou C, Winemiller KO. Seasonal hydrology shifts in production sources supporting fishes in rivers of the lower Mekong basin. Can J Fish Aquat Sci. 2016; 73(9):1342–62. https://doi.org/10.1139/cjfas-2015-0214
O’Reilly CM, Verburg P, Hecky RE, Plisnier P-D, Cohen AS. Food web dynamics in stable isotope ecology: time integration of different trophic levels. In: Seuront L, Sutton P, editors. Handbook of scaling methods in aquatic ecology: measurement, analysis, simulation. CRC Press, Boca Raton, Florida; 2004. p.125–34.
Planquette P, Keith P, Le Bail P-Y. Atlas des poissons d’eau douce de Guyane (tome 1). Collection du Patrimoine naturel. 1996; 22:154.
Post DM. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology. 2002; 83(3):703–18. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2
Pouilly M, Yunoki T, Rosales C, Torres L. Trophic structure of fish assemblages from Mamoré River floodplain lakes (Bolivia). Ecol Freshw Fish. 2004; 13(4):245–57. https://doi.org/10.1111/j.1600-0633.2004.00055.x
Power ME. Depth distributions of armored catfish: predator-induced resource avoidance? Ecology. 1984; 65(2):523–28. https://doi.org/10.2307/1941414
Putz R, Junk WJ. Phytoplankton and Periphyton. In: Junk WJ, editor. The Central Amazon floodplain: ecology of a pulsing system. Berlin, Heidelberg: Springer; 1997. p.207–22.
Qin X, Li Y, Wan Y, Fan M, Liao Y, Li Y et al. Multiple stable isotopic signatures corroborate the predominance of acetoclastic methanogenesis during CH4 formation in agricultural river networks. Agric Ecosyst Environ. 2020; 296:e106930. https://doi.org/10.1016/j.agee.2020.106930
Rai H, Hill G. Primary production in the Amazonian aquatic ecosystem. In: The Amazon: limnology and landscape ecology of a mighty tropical river and its basin. Dordrecht: Springer Netherlands; 1984. p.311–35.
Roach KA, Thorp JH, Delong MD. Influence of lateral gradients of hydrologic connectivity on trophic positions of fishes in the upper Mississippi River. Freshw Biol. 2009a; 54(3):607–20. https://doi.org/10.1111/j.1365-2427.2008.02137.x
Roach KA, Winemiller KO, Davis SE. Autochthonous production in shallow littoral zones of five floodplain rivers: effects of flow, turbidity, and nutrients. Freshw Biol. 2014; 59(6):1278–93. https://doi.org/10.1111/fwb.12347
Roach KA, Winemiller KO, Layman CA, Zeug SC. Consistent trophic patterns among fishes in lagoon and channel habitats of a tropical floodplain river: evidence from stable isotopes. Acta Oecol. 2009b; 35(4):513–22. https://doi.org/10.1016/j.actao.2009.03.007
Roach KA, Winemiller KO. Hydrologic regime and turbidity influence entrance of terrestrial material into river food webs. Can J Fish Aquat Sci. 2015; 72(7):1099–112. https://doi.org/10.1139/cjfas-2014-0459
Rodriguez MA, Lewis WM Jr. Structure of fish assemblages along environmental gradients in floodplain lakes of the Orinoco River. Ecol Monogr. 1997; 67(1):109–28. https://doi.org/10.1890/0012-9615(1997)067[0109:SOFAAE]2.0.CO;2
Sá-Oliveira JC, Araujo FR, Souza AT, Freitas CPD. Resource partitioning between two piranhas in an Amazonian reservoir. J Fish Biol. 2017; 91(5):1405–16. https://doi.org/10.1111/jfb.13461
Saul WG. An ecological study of fishes at a site in the upper Amazonian Ecuador. PNAS. 1975; 127(12):93–134. Available from: http://www.jstor.org/stable/4064705
Sazima I. Similarities in feeding behavior between some marine and freshwater fishes in two tropical communities. J Fish Biol. 1986; 29(1):53–65. https://doi.org/10.1111/j.1095-8649.1986.tb04926.x
Scarabotti PA, López JA, Pouilly M. Flood pulse and the dynamics of fish assemblage structure from Neotropical floodplain lakes. Ecol Freshw Fish. 2011; 20(4):605–18. https://doi.org/10.1111/j.1600-0633.2011.00510.x
Silva EM. Dieta da pilombeta, Anchoviella lepidentostole (Fowler, 1911), (Actinopterygii: Engraulidae) no baixo São Francisco, Nordeste do Brasil. [TCC]. Penedo: Universidade Federal de Alagoas; 2018.
Souza LS, Ambruster JW, Werneke DC. The influence of the Rupununi Portal on distribution of freshwater fish in the Rupununi District, Guyana. Cybium. 2012; 36(1):31–43. https://doi.org/10.26028/CYBIUM/2012-361-004
Souza LS, Armbruster JW, Willink PW. Connectivity of Neotropical river basins in the Central Guiana Shield based on fish distributions. Front For Glob Change. 2020; 3:8. https://doi.org/10.3389/ffgc.2020.00008
Stephens RB, Shipley ON, Moll RJ. Meta-analysis and critical review of trophic discrimination factors (ΔC and ΔN): importance of tissue, trophic level and diet source. Funct Ecol. 2023; 37(9):2535–48. https://doi.org/10.1111/1365-2435.14403
Taphorn DC, Liverpool E, Lujan NK, DoNascimiento C, Hemraj DD, Crampton WGR et al. Annotated checklist of the primarily freshwater fishes of Guyana. PNAS. 2022; 168(1):1–95. https://doi.org/10.1635/053.168.0101
Taphorn DC, Winemiller KO, Lilyestrom CG. Food habits of piranhas in the low Llanos of Venezuela. Biotropica. 1997; 29(1):9–17.
Thomas SM, Crowther TW. Predicting rates of isotopic turnover across the animal kingdom: a synthesis of existing data. J Anim Ecol. 2015; 84(3):861–70. https://doi.org/10.1111/1365-2656.12326
Van der Sleen P, Albert JS, editors. Field guide to the fishes of the Amazon, Orinoco & Guianas. Princeton University Press, Princeton and Oxford; 2018.
Vander Zanden MJ, Rasmussen JB. Variation in δ15N and δ13C trophic fractionation: implications for aquatic food web studies. Limnol Oceanogr. 2001; 46(8):2061–66. https://doi.org/10.4319/lo.2001.46.8.2061
Vari RP. Systematics of the Neotropical Anostomid genus Leporinus with ecological notes. Smithson Contrib Zool. 1991; 507:1–69.
Vari RP. Systematics of Leporinus with notes on feeding (omnivory/detritivory). Smithson Contrib Zool. 1992; 529:1–48.
Vari RP, Ortega H. The fishes of the Peruvian Amazon: distribution and ecology. Smithson Contrib Zool. 1986; 430:1–25.
Wantzen KM, Machado FA, Voss M, Boriss H, Junk W. Seasonal isotopic shifts in fish of the Pantanal wetland, Brazil. Aquat Sci. 2002; 64:239–51. https://doi.org/10.1007/PL00013196
Watkins G, Saul W, Holm E, Watson C, Arjoon D, Bicknell J. The fish fauna of the Iwokrama Forest. PNAS. 2005; 154(1):39–53. https://doi.org/10.1635/0097-3157(2004)154[0039:TFFOTI]2.0.CO;2
Winemiller KO. Spatial and temporal variation in tropical fish trophic networks. Ecol Monogr. 1990; 60(3):331–67. https://doi.org/10.2307/1943061
Winemiller KO. Factors driving temporal and spatial variation in aquatic floodplain food webs. In: Polis GA, Winemiller KO, editors. Food webs: integration of patterns & dynamics. Springer US, Boston, Massachusetts; 1996. p.298–312. https://doi.org/10.1007/978-1-4615-7007-3_29
Winemiller KO, Hoeinghaus DJ, Pease AA, Esselman PC, Honeycutt RL, Gbanaador D et al. Stable isotope analysis reveals food web structure and watershed impacts along the fluvial gradient of a Mesoamerican coastal river. River Res App. 2011; 27(6):791–803. https://doi.org/10.1002/rra.1396
Winemiller KO, Jepsen DB. Effects of seasonality and fish movement on tropical river food webs. J Fish Biol. 1998; 53:267–96. https://doi.org/10.1111/j.1095-8649.1998.tb01032.x
Winemiller KO, Montaña CG, Roelke DL, Cotner JB, Montoya JV, Sanchez L et al. Pulsing hydrology determines topdown control of basal resources in a tropical riverfloodplain ecosystem. Ecol Monogr. 2014; 84(4):621–35. https://doi.org/10.1890/13-1822.1
Winemiller KO, Montoya JV, Layman CA, Roelke DL, Cotner JB. Seasonally varying impact of detritivorous fishes on the benthic ecology of tropical floodplain river. J North Am Benthol Soc. 2006; 25(1):250–62. https://doi.org/10.1899/0887-3593(2006)25[250:SVIODF]2.0.CO;2
Zeug SC, Winemiller KO. Evidence supporting the importance of terrestrial carbon in a large-river food web. Ecology. 2008; 89(6):1733–43. https://doi.org/10.1890/07-1064.1
Authors
![]() Benton L. Fry1,2
Benton L. Fry1,2 ![]() ,
, ![]() Calvin J. Young1,
Calvin J. Young1, ![]() Carmen G. Montaña3,
Carmen G. Montaña3, ![]() Leslie C. Kelso Winemiller1 and
Leslie C. Kelso Winemiller1 and ![]() Kirk O. Winemiller1,2
Kirk O. Winemiller1,2
[1] Ecology and Conservation Biology, Texas A&M University, 534 John Kimbrough Blvd. 77843 College Station, TX, USA. (BLF) bfry@ tamu.edu (corresponding author), (CJY) cyoung725@tamu.edu, (LCKW) leslie.winemiller@ag.tamu.edu, (KOW) k-winemiller@tamu.edu.
[2] Ecology and Evolutionary Biology, 534 John Kimbrough Blvd. 77843 College Station, TX, USA.
[3] Biology, Stephen F. Austin State University, 1901 North St. 75965 Nacogdoches, TX, USA. (CGM) montanascg@sfasu.edu.
Authors’ Contribution 

Benton L. Fry: Conceptualization, Formal analysis, Investigation, Methodology, Visualization, Writing-original draft, Writing-review and editing.
Calvin J. Young: Data curation, Formal analysis, Investigation, Writing-original draft.
Carmen G. Montaña: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Writing-review and editing.
Leslie C. Kelso Winemiller: Data curation, Funding acquisition, Investigation.
Kirk O. Winemiller: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
Fish were collected according to TAMU IACUC permit no 2022–0093 and Guyana EPA permit no 20221102 BR 016. Animal handling conformed to the Guidelines for Use of Fishes in Research (Joint Committee of the American Fisheries Society, American Institute of Fishery Research Biologists, 2014).
Competing Interests
The author declares no competing interests.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
Financial support was provided by the US National Science Foundation IRES program grant no. 2153453.
Supplementary Material
Supplementary material S1
Supplementary material S2
How to cite this article
Fry BL, Young CJ, Montaña CG, Winemiller LCK, Winemiller KO. Variation in basal production sources supporting fish trophic guilds in the channel and floodplain ponds of the Rupununi River, Guyana, during dry season isolation. Neotrop Ichthyol. 2026; 24(1):e250155. https://doi.org/10.1590/1982-0224-2025-0155
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 12, 2025
Accepted December 12, 2025
![]() Submitted September 2, 2025
Submitted September 2, 2025
![]() Epub April 27, 2026
Epub April 27, 2026