![]() Beatriz Cristina de Paula de Souza1,
Beatriz Cristina de Paula de Souza1, ![]() Luana Caroliny Possamai2,
Luana Caroliny Possamai2, ![]() Katia Yasuko Yofukuji3,
Katia Yasuko Yofukuji3, ![]() Taise Miranda Lopes2,4,
Taise Miranda Lopes2,4, ![]() Luiz Fernando Esser2,
Luiz Fernando Esser2, ![]() José Hilário Delconte Ferreira5,
José Hilário Delconte Ferreira5, ![]() Reginaldo Ré5,
Reginaldo Ré5, ![]() Weferson Júnio da Graça2,6,
Weferson Júnio da Graça2,6, ![]() Ana Francisca Gomes da Silva1,
Ana Francisca Gomes da Silva1, ![]() Jefferson Matheus Barros Ozório7,
Jefferson Matheus Barros Ozório7, ![]() Dayani Bailly2,6
Dayani Bailly2,6 ![]() and
and ![]() Valéria Flávia Batista-Silva1
Valéria Flávia Batista-Silva1
PDF: Download Here | Cite this article
Associate Editor: ![]() Franco Teixeira de Mello
Franco Teixeira de Mello
Section Editor: ![]() Fernando Pelicice
Fernando Pelicice
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
As mudanças climáticas representam uma crescente ameaça aos ecossistemas de água doce. Diante desse cenário, este estudo investigou os impactos dessas mudanças sobre a distribuição de Megaleporinus obtusidens, uma espécie migradora de longa distância da bacia Paraná-Paraguai, e identificou os principais atributos climático-ambientais associados à sua ocorrência. Para isso, empregou-se uma abordagem de projeção combinada com múltiplos algoritmos para estimar a adequabilidade ambiental e a distribuição da espécie, bem como identificar refúgios climáticos sob diferentes cenários futuros. A amplitude térmica e a ordem do rio foram os atributos com maior contribuição para o modelo. Embora grande parte da bacia atualmente ofereça condições favoráveis para M. obtusidens, é prevista uma redução de 46,9% nas áreas climaticamente adequadas até 2050 e de 88,0% até 2090, sob um cenário pessimista. No final do século, os refúgios climáticos estarão concentrados nos trechos superiores dos tributários da margem esquerda do alto rio Paraná e no canal principal desse rio. A perda de áreas adequadas supera os ganhos, que se restringem ao alto rio Paraná. Esses resultados destacam a vulnerabilidade de M. obtusidens às mudanças climáticas, ressaltando a necessidade de planos de conservação focados na proteção desses refúgios para assegurar a persistência da espécie na bacia.
Palavras-chave: Adequabilidade climático-ambiental, Bacia Paraná-Paraguai, Ecossistemas de água doce, Modelos de nicho ecológico, Refúgio climático.
Introduction
Climate change, driven by global warming, has been widely discussed in recent decades (Abbass et al.,2022; IPCC, 2023). The global average temperature has already increased by 1.1°C, and future projections are even more alarming, with expected increases ranging from 1.4°C to 4.4°C, depending on greenhouse gas emission levels (Nobre et al.,2019; IPCC, 2023). This thermal increase may cause considerable impacts on both terrestrial (Field et al.,2007; Sales et al.,2021; Bernardinis et al.,2023; IPCC, 2023) and aquatic ecosystems (Woodward et al.,2010; Lopes et al.,2017; Conceição et al.,2023; IPCC, 2023), affecting all levels of biological organization, from individuals to ecosystems (Woodward et al.,2010; Bellard et al., 2012). Among the main threats are local extinctions and the spatial redistribution of species (Cahill et al.,2013; Urban, 2015; Bailly et al., 2021; Sales et al.,2021).
Freshwater environments are among the most sensitive to the effects of climate change, and despite their importance, these environments are increasingly vulnerable to anthropogenic impacts, such as global warming and growing habitat fragmentation (Dudgeon, 2014). The rise in temperatures triggers several changes in the natural dynamics of freshwater systems, altering oxygen concentration, water temperature, pH, water flow, and the frequency and intensity of floods and droughts (Ficke et al., 2007; Döll, Zhang, 2010; Phillips et al.,2015). In this context of growing vulnerability, climate change is expected to affect fish, particularly migratory species (Ruaro et al., 2019; Bailly et al., 2021).
The life cycle of migratory species is strongly synchronized with the hydrological cycle, with the onset of rainfall acting as a trigger for shoal formation and the initiation of migrations, while flood peaks serve as stimuli for spawning (Suzuki et al., 2009; Bailly et al., 2008; Oliveira et al., 2015). Furthermore, gonadal development and oocyte maturation are regulated by factors such as temperature and photoperiod (Suzuki et al., 2009). Consequently, changes in these environmental variables imposed by climate change may compromise the reproduction of Neotropical migratory freshwater fishes. Aggravating this situation, the construction of dams for energy generation can negatively impact the life cycle of these organisms by hindering the upstream reproductive migrations of adults and the passive dispersal of eggs and larvae (Agostinho et al., 2008; Pelicice et al., 2014).
Among the migratory Neotropical fish species in the Paraná-Paraguay basin is Megaleporinus obtusidens (Valenciennes, 1837), commonly known as “piava” or “piapara” (Agostinho et al., 2003; Copatti, Amaral, 2009). Native to South America (Britski et al.,2012), M. obtusidens is widely distributed across the Uruguay, São Francisco, and Paraná-Paraguay River basins (Agostinho et al., 2003; Zaniboni-Filho, Schulz, 2003; Britski et al.,2012), but is considered a non-native species in the Iguaçu River sub-basin (Baumgartner et al., 2012; Reis et al., 2020). This migratory fish can reach up to 49.3 cm in length (Agostinho et al., 2003) and weigh up to 6 kg (Resende, 2003), characteristics that, combined with the quality of its meat, make it a species of high economic value. In the Paraná-Paraguay basin, it is one of the main targets of both commercial and sport fishing (Agostinho et al., 2007; Barletta et al., 2015). However, intense fishing pressure and habitat fragmentation, coupled with the increasing effects of climate change, represent serious threats to the dynamic of their populations, requiring special attention to the management and conservation actions for their habitats (Agostinho et al., 2007; Quirós et al., 2007; Peluso et al., 2022).
Given the rapid pace of environmental changes, understanding how species will respond to climate change is a central challenge for conservation management. Recent studies have increasingly applied Ecological Niche Models (ENMs) to freshwater environments, particularly for fish species, to assess how climate change and other anthropogenic drivers may alter their distributions. In riverine systems, ENMs have been used to evaluate the loss of suitable habitats (Lopes et al., 2017; Ruaro et al., 2019), the impacts of hydrological alterations such as dam construction (Peluso et al., 2022), and to identify potential climate refugia and priority areas for conservation (Bailly et al., 2021). These applications highlight the relevance of ENMs as tools not only for projecting future distributions but also for guiding management and conservation actions in freshwater ecosystems (Peterson et al., 2011; Brun et al., 2020). Therefore, the present study investigated the impacts of climate change on the potential distribution of M. obtusidens, a long-distance migratory species in the Paraná-Paraguay basin. Specifically, we examined its potential distribution under current climate conditions and future scenarios, considering both moderate and pessimistic greenhouse gas emission trajectories. We then performed spatial predictions to identify the main climate refuge areas for M. obtusidens and the climatic-environmental attributes associated with its current occurrence. Understanding the current and future potential distribution of M. obtusidens will provide valuable insights for developing effective conservation strategies for this species in the Paraná-Paraguay basin.
Material and methods
Study area. The Paraná-Paraguay basin drains about 2,800,000 km² of South American territory. In addition tooccupying large parts of the Southwestern and Southeastern regions of Brazil, it covers parts of Argentina, eastern Bolivia, and Paraguay (Graça et al., 2024; Fig. 1). The basin is mainly formed by Paraná and Paraguay Rivers that supports a high biodiversity and distinct landscapes. The Paraná River, with a length of 4,695 km, is formed by the junction of the Grande and Paranaíba rivers, and flows through southern Brazil, Paraguay, and Argentina. It ranks among the top ten rivers globally in terms of water volume (Brea, Zucol, 2011; Abrial et al., 2021). In the upper reaches of the Paraná River, there is a large floodplain, a dynamic and highly heterogeneous ecosystem that plays an essential role in preserving the biodiversity of this basin (Thomaz et al., 2004). The Paraguay River, stretching 2,550 km, is the fifth largest waterway in South America, originating in the Serra dos Parecis and flowing into the right bank of the Paraná River (Resende, 2003). The upper Paraguay River flows through the Chaco-Pantanal biome, one of the largest continuous alluvial plains in the world, extending across regions of Brazil, Bolivia, and Paraguay. This region is widely recognized for its rich biodiversity, encompassing a mosaic of habitats including temporary and permanent lakes, alluvial forests, and seasonally inundated savannas (Paranhos-Filho et al., 2013).
FIGURE 1| Observed occurrence records of Megaleporinus obtusidens in the Paraná-Paraguay basin.
Occurrence data of the species. The georeferenced occurrence records of M. obtusidens in the Paraná-Paraguay basin were obtained through four databases: SpeciesLink (http://splink.cria.org.br), Global Biodiversity Information Facility (GBIF; http://www.gbif.org/), Base de Datos de Peces de Águas Continentales de Argentina (http://www.pecesargentina.com.ar/; Liotta et al., 2020), and Ictioplata Database (https://app.sedoo.fr/freshwaterfishdata/#/amazonfish; Jézéquel et al., 2020). Occurrence data were obtained from these databases up until September 2025. Additionally, occurrences from the literature were included by searching the Web of Science (Thomson Institute for Scientific Information, ISI; www.isiknowledge.com), Scopus (https://www.scopus.com/) and Google Scholar (https://www.googlescholar.com/), using the terms “Leporinus obtusidens” and “Megaleporinus obtusidens”, encompassing all historical and current taxonomic designations for the species. After compiling all these sources, a total of 1,267 georeferenced occurrence records were obtained. Only records from the native range of the species were considered. Records from introduced populations, such as those from the Iguaçu River basin, were excluded, since M. obtusidens is regarded as non-native in that basin (Baumgartner et al., 2012; Reis et al., 2020).
Subsequently, these occurrence records underwent a rigorous data cleaning process using the R package CoordinateCleaner (Zizka et al., 2019), which tested them against several potential spatial biases, including the coordinates of capital cities, country centroids, equal latitude and longitude, research institution locations, and invalid coordinates. We also excluded records outside the Paraná-Paraguay River basin, which could bias models by introducing different configurations of predictor variables that are not present in our study area. This allows us to make predictions based solely on the arrangements faced by the natural populations within the basin, since there is no migration or gene flow between Paraná-Paraguay and other basins where the species is currently found. After these steps, we tested different grid sizes and selected the 10 km resolution, which maximized record retention while maintaining adequate spatial detail (for more details, please see Seo et al., 2009). The cleaned dataset (941 occurrence records) was then mapped on a regular geographical grid, with a spatial resolution of 10 km, comprising 15,617 cells, covering the river network in the Paraná-Paraguay basin. After this process, 209 occurrence records of M. obtusidens remained (Fig. 1). The grid was constructed in EPSG 6933, an equivalent spatial reference system that maintains a constant area.
Bioclimatic and hydrological variables. The distribution of the species was modeled as a function of bioclimatic and hydrological variables covering the Paraná-Paraguay basin. Present bioclimatic variables (1970–2000; representing 30 years temporal average) and future variables (2050 and 2090) were extracted from the WorldClim 2.1 database (http://www.worldclim.org/; Fick, Hijmans, 2017), with 2.5 arc minutes of spatial resolution. Future climate scenarios and general circulation models available in WorldClim were provided by the Intergovernmental Panel on Climate Change, Sixth Assessment Report (IPCC-AR6; https://www.ipcc.ch/assessment-report/ar6/) considering the new Coupled Model Inter‐comparison Project phase 6 (CMIP6). CMIP6 aims to better understand climate changes arising from natural, unforced variability or in response to changes in radiative forcing in a multi-model context (Eyring et al.,2016). Thus, for future forecasts, we used two Shared Socio-economic Pathways (SSPs), moderate (SSP2–4.5) and pessimistic (SSP5–8.5). The SSPs not only consider carbon emission scenarios but also socioeconomic factors that might change in the next few years, such as population size, education, economic growth, development of new technologies, and urbanization (Riahi et al., 2017). Two atmospheric-ocean general circulation models (AOGCMs) were used: CNRM-CM6-1-HR (Centre National de Recherches Météorologiques) and INM-CM4-8 (Institute of Numerical Mathematics). The Global Circulation Models (GCM) selection was made based on the chooseGCM package (Esser et al., 2025).
After filtering bioclimatic variables by excluding those with higher chance of environmental discontinuities (Booth, 2022), we kept 11 bioclimatic variables available in WorldClim with potential causation effect on species occurrence. We also used seven hydrological variables obtained from HydroSHEDS database (https://www.hydrosheds.org/products/hydrorivers; Lehner, Grill, 2013) aiming for variables with causation effect on species occurrence (Tab. 1). Bioclimatic and hydrological variables were rescaled using a 10 km geographical grid to obtain the climatic-environmental layers for both present and future times. Hydrological variables were considered temporally stationary for future forecasts.
TABLE 1 | Bioclimatic and hydrological variables used in the predictor selection for the ecological niche modeling of Megaleporinus obtusidens in the Paraná-Paraguay basin.
Variable | Description | Source |
BIO1 | Annual Mean Temperature | WorldClim |
BIO2 | Mean Diurnal Range | WorldClim |
BIO3 | Isothermality | WorldClim |
BIO4 | Temperature Seasonality | WorldClim |
BIO5 | Max Temperature of Warmest Month | WorldClim |
BIO6 | Min Temperature of Coldest Month | WorldClim |
BIO7 | Temperature Annual Range | WorldClim |
BIO12 | Annual Precipitation | WorldClim |
BIO13 | Precipitation of Wettest Month | WorldClim |
BIO14 | Precipitation of Driest Month | WorldClim |
BIO15 | Precipitation Seasonality | WorldClim |
LENGTH_KM | Length of the river reach segment (km) | HydroSHEDS |
DIST_DN_KM | Distance from the most downstream pixel of the reach, to the final downstream location along the river network (km) | HydroSHEDS |
DIST_UP_KM | Distance from the most downstream pixel of the reach, to the most upstream location along the river network (km) | HydroSHEDS |
CATCH_KM | Area of the catchment that contributes directly to the individual reach (km²) | HydroSHEDS |
UPLAND_KM | Total upstream area (km²) calculated from the headwaters to the most downstream pixel of the river reach | HydroSHEDS |
DIS_AV_CMS | Average long-term discharge estimate for river reach (m³/s) | HydroSHEDS |
ORD_STRA | Indicator of river order following the Strahler (1957) ordering system | HydroSHEDS |
The Variance Inflation Factor (VIF) was used to evaluate multicollinearity problems in our set of variables. Using the usdm package in R (Naimi, Araújo, 2016), variables were examined for pairwise linear correlations, and in each interaction, the variable exhibiting the highest VIF was excluded. The procedure continued until all variable pairs fell below the collinearity threshold (here 0.5). Thus, the following variables were kept: BIO2, BIO5, BIO14, DIST_DN_KM, CATCH_KM and ORD_STRA (Tab. 2).
TABLE 2 | Variance Inflation Factor (VIF) values for the predictor variables retained in the ecological niche modeling of Megaleporinus obtusidens. All pairwise correlations were below the collinearity threshold (r < 0.5).
Variables | VIF |
BIO2 | 1.3334 |
BIO5 | 1.1601 |
BIO14 | 1.0348 |
DIST_DN_KM | 1.3035 |
CATCH_KM | 1.3497 |
ORD_STRA | 1.4435 |
Generation of pseudo-absence data. We generated pseudo-absences to contrast environmental information and allow the use of machine learning-based algorithms. We randomly obtained n pseudo-absences within the study area, where n is equal to the number of occurrence records of the species (209). This procedure was necessary to avoid imbalance issues (Japkowicz, Stephen, 2002). We built a machine learning approach to obtain occurrence probabilities using algorithms that perform their functions using pseudo-absence data, not background data (see Sillero, Barbosa, 2021). In order to build distinguishable pseudo-absences from true absences and avoid biases that rise with randomness, we randomly selected 10 sets of 209 pseudo-absences outside an environmental envelope generated with all occurrence records and selected predictor variables at these locations (Nix, 1986; Busby, 1991). This procedure guarantees that pseudo-absences are geographically and environmentally distinct from presences (see Lobo et al.,2006). Thus, a binary matrix of presence (1) and pseudo-absence (0) was created to serve as input for the niche models.
Species distribution modeling. Binary matrices representing the presence and pseudo-absence of M. obtusidens, and environmental layers corresponding to the set of non-collinear variables based on VIF results for the species were used to calibrate models. The different algorithms provide distinct predictions of the species distribution area, generating uncertainty about which model best represents the geographic distribution of the species (Diniz-Filho et al., 2009). To overcome this uncertainty and minimize errors, the ensemble forecasting approach was used since it provides a consensus projection among multiple algorithms (Araújo, New, 2007). By obtaining a consensus model, errors that affect each algorithm differently tend to cancel each other out, resulting in a reliable and moderate solution (Terribile et al., 2010). In this context, we used seven algorithms, as follows: Support Vector Machine with Radial Kernel (svmRadial), Random Forest (RF), General Linear Models (GLM), Multiple Discriminant Analysis (MDA), Neural Networks (NNET), K-Nearest Neighbors (KNN), and Naive Bayes (Naive_Bayes) to predict the environmental suitability and potential distribution of M. obtusidens in Paraná-Paraguay basin.
For each algorithm used, the M. obtusidens occurrence data (presences and pseudo-absences) were randomly divided into two datasets, one comprising 75% of the data for model calibration and the other with 25% for model evaluation. This process was repeated 10 times using a cross-validation system of subsampling K-folds, with K = 4 (totalizing 40 repetitions). The K-fold validation divides the data into K subsets of approximately equal size and adjusts the models K times. Each time a subset is used as test data, the remaining K-1 subsets are used as training data (Naimi, Araújo, 2016). Iterations of the cross-validation were held to each set of pseudo-absences. In each repetition, each algorithm was also submitted to an automated fine-tuning with a grid search pattern of size 10, and optimizing the Area Under the Receiver Operating Characteristic Curve (AUC) values. A total of 40 climatic-environmental suitability projections were generated for each algorithm. Each projection was then binarized into presence and absence data for every grid cell using the threshold that maximizes the sum of sensitivity and specificity (Liu et al., 2016). We summed the binary outputs, generating frequency outputs that represent comparable suitability among different models. Thus, for each cell, the comparable suitability (i.e., the frequency, referred to as climatic-environment suitability) was calculated, ranging between 0 and 1 (1 represents the most suitable conditions and 0 the least suitable). Climatic-environmental suitability was then converted in presence and absence to obtain the species distribution, where values higher than 0.5 represented presences, and values lower than or equal to 0.5 represented absences.
The predictive performance, for each algorithm, was evaluated using the Receiver Operating Characteristic (ROC) method, which generates a curve in a bivariate space by plotting the true positive rate (sensitivity) against the false positive rate (1 – true negative rate or 1 – specificity). The AUC was calculated as a measure of the predictive performance of models, independent of the decision threshold (Manel et al., 2001; Liu et al., 2005). AUC values of 0.5 indicate models that perform no better than random selection, while values close to 1 indicate good performance with higher true-positive rates and lower false-positive rates. Models with AUC > 0.9 were combined to obtain a consensus prediction (Araújo, New, 2007). To do that, we used the majority consensus rule (adapted from Diniz-Filho et al., 2009) to obtain the consensus models, considering species presence only in cells where more than 50% of the ENMs retained in the ensemble indicated the species as being present. The climatic-environmental suitability of the consensus model was obtained by averaging the climatic-environmental suitability of the models retained in the consensus. The modeling protocol produced a total of 28,000 projections for the current time (7 algorithms x 40 repetitions x 10 fine-tuning iterations x 10 pseudoabsence datasets) and a total of 224,000 projections for the future time (7 algorithms x 40 repetitions x 2 climate models x 2 SSPs x 2 future times x 10 fine-tuning iterations x 10 pseudoabsence datasets).
The importance of variables was assessed through a “filter” method, where the models are built with and without a given variable. The change in AUC is used as a metric of variable importance: if the value does not change, the variable has minimum influence on the model. Calibration, testing, fine-tuning and ensembling were performed in caretSDM R package (https://github.com/luizesser/caretSDM). Maps were produced using QGIS v. 3.40.1.
To identify potential areas within the Paraná-Paraguay basin where M. obtusidens may gain, lose, or maintain its potential distribution in the future, we used the results of the consensus model of presence-absence. The current distribution of the species was then compared with projections for 2050 and 2090 under moderate and pessimistic climate scenarios. By overlaying these maps, the areas were classified into four categories: (1) stable, where the species remains present across projections; (2) susceptible to loss, where the species is currently present but is predicted to contract its potential range in the future; (3) susceptible to gain, where the species may expand its distribution to currently unoccupied areas; and (4) unsuitable areas, where conditions remain unsuitable in both the present and future.
Results
The models retained for the consensus (AUC > 0.9), included MDA, Naive Bayes, RF, and svmRadial, demonstrated high predictive performance for the occurrence of M. obtusidens in the Paraná-Paraguay basin (mean AUC of 0.96 ± 0.03). The environmental-climatic suitability gradients, generated by the consensus model, indicated that nearly half of the area of the Paraná-Paraguay basin offers favorable conditions for the occurrence of M. obtusidens currently. Extensive and contiguous regions of the Paraná River basin were predicted as suitable for the species, favoring its wide distribution throughout the basin. The main channel of the Paraná River, across nearly its entire length, along with the tributaries of its upper and lower stretches showed the highest environmental-climatic suitability. In contrast, in the Paraguay River basin, high environmental suitability was restricted to the large tributaries of the upper region, mainly in the Pantanal floodplain, and in the tributaries on the left bank of the lower Paraguay River, configuring a fragmented distribution pattern amidst predominantly unfavorable conditions (Figs. 2A–B).
FIGURE 2| Environmental-climatic suitability (A) and presence (red) / absence (gray) (B) estimated for Megaleporinus obtusidens in the Paraná-Paraguay basin for the present. Estimates are based on the results of the consensus models. Suitability values equal to 1 correspond to ideal environmental conditions and values equal to 0 correspond to suboptimum environmental conditions.
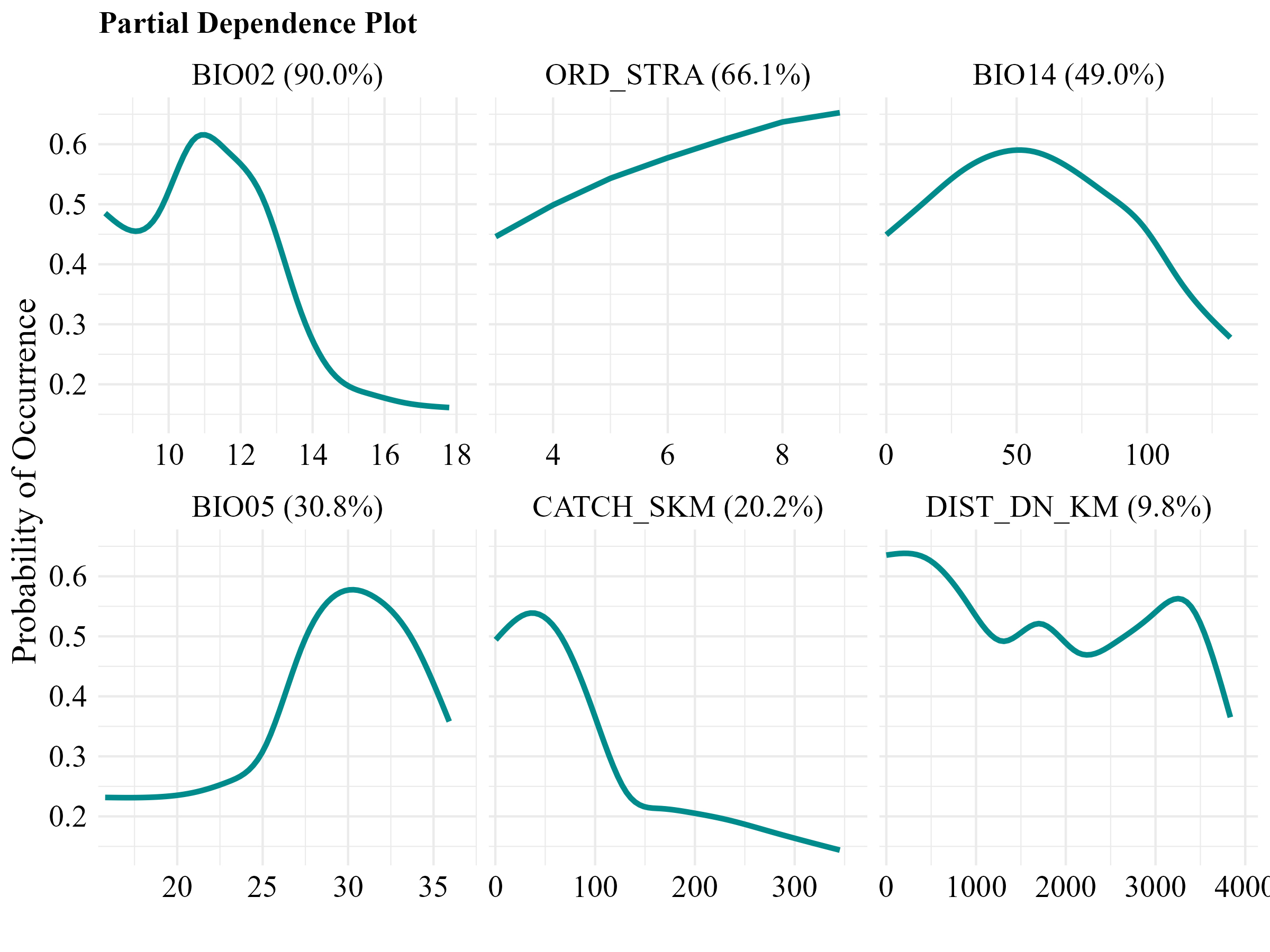
The environmental variables that most contributed to explaining the distribution of M. obtusidens in the Paraná-Paraguay basin were the mean diurnal temperature range (BIO2) and stream order (ORD_STRA) (Fig. 3). The analysis of partial dependence curves revealed that the probability of species occurrence increases in areas with a daily temperature range between approximately 9.8°C and 12.2°C, and in rivers of 4th order or higher (Fig. 4).
FIGURE 3| Relative importance of bioclimatic and hydrological variables for the current distribution of Megaleporinus obtusidens.
FIGURE 4| Response curves of variables used in the ecological niche modeling of Megaleporinus obtusidens in the Paraná-Paraguay basin. Values in parentheses indicate the relative importance of each variable.
Overall, future projections indicated a reduction in climatically suitable areas for M. obtusidens in the Paraná-Paraguay basin. By 2050, under a moderate scenario, a decrease in climatic suitability is expected, especially in the tributaries of the Upper Paraguay River, while suitable areas are projected to persist in the Lower Paraguay River and in most tributaries of the upper and lower stretches of the Paraná River (Fig. 5A). However, under the pessimistic scenario, the extent of these suitable areas is expected to decline, particularly in the tributaries located on the right bank of the upper Paraná River (Fig. 5B). For 2090, the moderate scenario follows the same trend for 2050, with a progressive decline in suitable areas but persistence of relatively favorable conditions along major tributaries of the Paraná River (Fig. 5C). In contrast, under the pessimistic scenario, a considerable reduction in climatically suitable areas is projected, limiting the favorable regions for M. obtusidens to a few segments of left-bank tributaries of the upper Paraná River and to small isolated patches in the southern portion of the basin (Fig. 5D).
FIGURE 5| Environmental-climatic suitability estimated for Megaleporinus obtusidens in the Paraná-Paraguay basin under future scenarios: A–B for 2050, and C–D for 2090, under moderate (SSP2–4.5) and pessimistic (SSP5–8.5) emission scenarios, respectively. Estimates are based on the results of the consensus models. Suitability values equal to 1 correspond to ideal environmental conditions and values equal to 0 correspond to suboptimum environmental conditions.
The potential distribution of M. obtusidens in the Paraná-Paraguay basin projected for 2050 under the moderate scenario indicates a loss of 38.1% of the currently suitable areas. Nevertheless, the lower stretches of the Paraguay River and extensive regions of the Paraná River basin are expected to serve as an important climatic refuge for the species (Fig. 6A). In the pessimistic scenario, a reduction of 46.9% of these areas is projected, especially in the tributaries of the upper Paraná River (Fig. 6B). The decrease in suitable areas becomes even more pronounced by 2090, both in the moderate (63.6%) (Fig. 6C) and, more intensively, in the pessimistic scenario (88.0%) (Fig. 6D) resulting in a greater contraction in the potential distribution of M. obtusidens.
In the pessimistic scenario for 2090, climate refugia are restricted to reduced stretches of the upper reaches of the left-bank tributaries of the upper Paraná River (Piquiri, Ivaí, Paranapanema, Tietê, Grande, and Paranaíba rivers), and to segments of the main channel of the Paraná river in its lower reach (Fig. 6D). Overall, the losses in suitable cells due to climate change greatly outweigh the gains, which are mainly limited to the upper stretches of tributaries of the upper Paraná River (Figs. 6A–D).
FIGURE 6| Predicted stable (blue), gain (green), loss (red) and unsuitable (grey) areas for Megaleporinus obtusidens in the Paraná-Paraguay: A–B for 2050, and C–D for 2090, under moderate (SSP2–4.5) and pessimistic (SSP5–8.5) emission scenarios, respectively. Estimates are based on the results of the consensus models.
Discussion
In this study, we highlight that the eastern portion of the Paraná-Paraguay basin along the main river axis exhibits a broad gradient of environmental climatic suitability, favoring a wide potential distribution of M. obtusidens in the present day. This species is recognized as a long-distance migratory fish, a characteristic that allows for a wider home range (Griffiths, 2010). Tagging studies in the Uruguay River basin have shown that the species can swim more than 500 km in a few days (Espinach Ros, 1999). In the Paraná-Paraguay basin, similar movements along the main rivers channel promote the dispersal of early life stages and the maintenance of adults, which supports the wide distribution observed in this study. Similar results were observed for other long-distance migratory fish in the region, such as the “curimba” (Prochilodus lineatus) (Agostinho et al., 1993) and the “dourado” (Salminus brasiliensis) (Ruaro et al., 2019). These movement patterns indicate that hydrological connectivity and the preservation of the integrity of these habitats are essential for the maintenance of M. obtusidens in the basin.
The distribution for M. obtusidens in the Paraná-Paraguay basin was largely explained by river order and thermal amplitude. Larger rivers (7th to 9th order), which provided greater water volume, habitat heterogeneity, and hydrological stability (Vannote et al., 1980; Ward, 1998; Zeiringer et al., 2018), showed the highest probabilities of species occurrence. These conditions favor feeding, shelter, and reproduction, as corroborated by the predominance of “piava” in the main channels of medium- to large-sized rivers channels (Palmuti et al., 2021). Consequently, maintaining longitudinal connectivity is critical for the maintenance of M. obtusidens populations, given their reproductive migratory behavior. Disruptions caused by dams and other anthropogenic barriers may fragment populations, reduce gene flow, and compromise the long-term viability (Carvajal-Quintero et al., 2019; Valenzuela-Aguayo et al., 2020). Nonetheless, it is important to mention that the importance of stream order may become even more prominent if projections that consider the influence of future climate and land use scenarios on this variable are available.
As ectothermic organisms, fish are sensitive to temperature variations (Vliet et al., 2013; Jonsson, 2023) that directly influence physiological processes such as reproduction, feeding, growth, and sensory sensitivity (Ficke et al., 2007; Wysocki et al., 2009; Li et al., 2022). In the case of M. obtusidens, experiments with juveniles showed that growth is optimized at temperatures between 26 °C and 30 °C, while lower temperatures reduce metabolic activity and higher temperatures, although they increase food consumption, do not promote significant growth (Piana et al., 2003). In our study, the most suitable areas for the species exhibited daily thermal amplitudes between 9.8°C and 12.2°C, suggesting that this range provides favorable conditions for development. Beyond its direct physiological implications, daily thermal amplitude may also serve as an indicator of environmental stability. Large fluctuations impose additional metabolic costs on ectothermic organisms, reducing the energy available for growth and reproduction (Ficke et al., 2007; Pörtner, Farrell, 2008), whereas intermediate ranges favor more stable habitats that are particularly important for migratory species such as M. obtusidens (Comte, Olden, 2017). As a result, these thermal conditions represent an essential component for the persistence of the species in the basin, especially under future climate change scenarios, which could alter the temperature ranges in areas currently suitable for its occurrence.
It is important to recognize that climatically suitable areas are not the only factor that supports the presence of M. obtusidens, as hydrology plays a decisive role in shaping its distribution. In this context, the restricted occurrence of the species in portions of the Paraná-Paraguay basin, particularly in the Chaco region (including northern Argentina and western Paraguay), likely reflects the structure of local drainage networks rather than climatic limitations. These regions, characterized by high thermal amplitude and more arid conditions, are dominated by intermittent or poorly connected river systems (Drago et al., 2008) that function as hydrological barriers and restrict the occurrence of the species even where climatic conditions remain favorable. For long-distance migratory fishes that depend on high-order rivers and prolonged flood pulses for reproduction (Agostinho et al., 2004), such landscapes become unsuitable, reinforcing the role of hydrological connectivity in determining species persistence and movement across the basin. In addition, future climate change may further alter hydrological regimes in the basin, potentially intensifying these constraints.
Despite this strong hydrological component, it is also important to consider that, in other areas of the basin, the absence of records may be related to sampling gaps rather than true absence. Regions of central and northern Argentina, particularly the Pilcomayo, Bermejo, and Salado rivers, as well as western Paraguay and the Bolivian portion of the Paraguay River, are widely recognized as under-sampled for freshwater fishes due to low population density, limited accessibility, and the lack of long-term monitoring programs in the Gran Chaco (Hablützel, Huanto, 2020). Historical ichthyological surveys from central Argentina provide additional support for this interpretation (Casciotta et al., 1989). Thus, the spatial distribution of available records may reflect this uneven sampling effort, which represents an inherent limitation of regional biodiversity databases including that of the present study.
Future projections indicate a concerning scenario for M. obtusidens across the basin, with losses of climatically suitable areas in nearly all regions that currently provide favorable conditions for the species. This trend has already been observed in previous studies, which warned about the impacts of these changes on M. obtusidens populations in the Paraná-Paraguay basin (Bailly et al., 2021; Peluso et al., 2022). These projected losses are particularly alarming because M. obtusidens as an omnivorous species, positioned at an intermediate trophic level, plays an important role in tropical freshwater ecosystems, influencing trophic interactions and contributing to nutrient cycling (Andrian et al., 1994; Vanni, 2002; Hahn et al., 2004). In addition, omnivorous fish contribute to the balance of zooplankton and phytoplankton communities, as well as in the biological control of invasive bivalves (Vadeboncoeur et al., 2005; Rosa et al., 2014; Salazar-Torres et al., 2015). These multiple ecological interactions illustrate not only the functional importance of M. obtusidens in the environment, but also highlight that biotic interactions contribute to shaping species distributions, influencing patterns of occurrence and local persistence.
While abiotic factors are traditionally considered the dominant drivers of species distribution at macroecological scales, considering biotic interactions can refine predictions of species responses to environmental changes, and may explain potential differences between modeled and observed distributions. Thus, the loss of suitable areas may, therefore, compromise the trophic dynamics and functionality of aquatic ecosystems in the Paraná-Paraguay basin, reinforcing the need for the conservation of M. obtusidens to ensure the maintenance of the basin’s ecological integrity.
Considering the importance of M. obtusidens for artisanal and recreational fishing (Barletta et al., 2010, 2015), the reduction of climatically suitable areas under future scenarios represents a significant loss of ecosystem services (e.g., provisioning and cultural), with potential economic impacts across various regions of the Paraná-Paraguay basin. One such region is the Paraguay River basin, which encompasses the “Pantanal”, a globally significant floodplain where fishing plays a crucial socio-economic role. In 2018, the average catch per unit of effort (CPUE) in this basin was estimated at 8,601 Kg per fisherman per day (6,606 Kg per fisherman per day in Mato Grosso and 14,685 Kg per fisherman per day in Mato Grosso do Sul) (ANA, 2020). Additionally, the total income of artisanal professional fishermen from the value of the first sale in the Paraguay River basin was estimated at R$ 69,820,920.29, with R$ 38,014,094.90 in Mato Grosso and R$ 31,788,825.39 in Mato Grosso do Sul (ANA, 2020).
Climate refugia can be defined as geographically stable areas where projected future climate conditions overlap with current or historical conditions, providing suitable habitats for the persistence of species in a climate change scenario (Keppel et al., 2012; Graham et al., 2019). The identification of these areas has become a key strategy for the management and conservation of species (Gavin et al., 2014; Sobral-Souza et al., 2018; Graham et al., 2019). In this context, our study showed that by mid-century, the areas of climate stability for M. obtusidens are expected to concentrate in the main channels of the Paraná and Paraguay Rivers, as well as in the tributaries of the Paraná River. However, by the end of the century, an expressive contraction of these areas is projected, resulting in the spatial fragmentation of refugia into small isolated segments of the Paraná-Paraguay basin, compromising the connectivity between essential habitats for the species.
The loss of climatic-environmental suitable connectivity between refugia regions may lead to the local extirpation of M. obtusidens across extensive portions of the basin, as the species undertakes long-distance migratory routes to complete its life cycle. Habitat fragmentation, intensified by the presence of numerous dams in the basin (Agostinho et al., 2008), further exacerbates this scenario in areas recognized as potential refugia, such as the Piquiri, Ivaí, Tietê, Paranapanema, Grande and Paranaíba rivers, which are left-bank tributaries of the Paraná-Paraguai basin draining the Serra do Mar and Serra da Mantiqueira. These barriers block migratory routes, restrict dispersal, and interrupt connectivity between spawning and recruitment areas (Mounic-Silva et al., 2019). Although the Ivaí and Piquiri rivers are currently dam-free climatic refuges, both have planned and inventoried hydroelectric plants (Affonso et al., 2015; ANEEL, 2019). The implementation of these projects represents an additional threat to the species’ survival, reinforcing the urgency of adopting effective conservation measures aimed at maintaining these climate refugia. It is further emphasized that the upper stretch of the Tietê River sub-basin is highly eutrophicated due to the discharge of domestic and industrial effluents (CETESB, 2023), a condition that exacerbates the threats to the species survival and further compromises the functionality of these refuges.
Paradoxically, climate change may also transform previously unsuitable areas into environmentally favorable habitats (see Williams, Jackson, 2007; Parmesan, Yohe, 2003). The ability of species to track these changes will depend on their ability to colonize new environments and establish viable populations. The speed of these processes will be decisive for the occupation of newly suitable environmental areas (Angert et al., 2011). In the case of M. obtusidens, although there is potential for a range expansion in the upper stretches of the left bank tributaries of the upper Paraná River, this process may be severely limited by habitat fragmentation, the absence of climate-environmental corridors (as discussed earlier), and by specific characteristics of the species’ life history.
In our study, climate projections indicated that the distribution of M. obtusidens in the Paraná-Paraguay basin will be severely affected by the end of this century, with potential negative consequences for the ecosystem services provided by this species. Mitigating these impacts requires long-term strategic conservation planning (Groves et al., 2012; Stein et al., 2014; Wang et al., 2024). However, studies show that the currently existing protected areas in the basin do not provide adequate protection for migratory species like M. obtusidens, a gap that is likely to worsen with the projected climate changes (Bailly et al., 2021). In light of this scenario, it becomes imperative to implement preventive measures, such as the preservation of free-flowing rivers, which maintain longitudinal connectivity, and protecting floodplains and wetlands, which safeguards lateral connectivity. These environments provide environmental stability, act as natural climate refugia, and serve as nurseries for the reproduction and juvenile development of M. obtusidens (Nakatani et al.,2004; Thomaz et al., 2007). Their inclusion in protected area systems is essential to ensure the integrity of their ecological characteristics, thereby promoting the resilience of aquatic ecosystems and the conservation of fish in the face of ongoing climate change.
Acknowledgments
The authors thank the Universidade Estadual de Mato Grosso do Sul (UEMS) for the physical and institutional infrastructure provided.
References
Abbass K, Qasim MZ, Song H, Murshed M, Mahmood H, Younis I. A review of the global climate change impacts, adaptation, and sustainable mitigation measures. Environ Sci Pollut Res. 2022; 29:42539–59. https://doi.org/10.1007/s11356-022-19718-6
Abrial E, Lorenzón RE, Rabuffetti AP, Blettler MC, Espínola LA. Hydroecological implication of long-term flow variations in the middle Paraná River floodplain. J Hydrol. 2021; 603:126957. https://doi.org/10.1016/j.jhydrol.2021.126957
Affonso IP, Azevedo RF, Santos NLCS, Dias RM, Agostinho AA, Gomes LC. Pulling the plug: strategies to preclude expansion of dams in Brazilian rivers with high-priority for conservation. Nat Conserv. 2015; 13(2):199–203. https://doi.org/10.1016/j.ncon.2015.11.008
Agência Nacional de Águas e Saneamento Básico (ANA). Plano de recursos hídricos da região hidrográfica do rio Paraguai [Internet]. Brasília; 2020. Available from: https://www.gov.br/ana/pt-br/assuntos/gestao-das-aguas/planos-e-estudos-sobre-rec-hidricos/plano-de-recursos-hidricos-rio-paraguai
Agência Nacional de Energia Elétrica (ANEEL). Nota Técnica nº 167/2019-SCG/ANEEL [Internet]. Brasília; 2019. Available from: https://www.aneel.gov.br/cedoc/ndsp2019778.pdf
Agostinho AA, Gomes LC, Suzuki HI, Júlio-Jr HF. Migratory fishes of the Paraná River basin, Brazil. In: Carolsfeld J, Harvey B, Ross C, Baer A, editors. Migratory fishes of South America:biology, fisheries and conservation status. Victoria: World Fisheries Trust; 2003. p.19–98.
Agostinho AA, Gomes LC, Veríssimo S, Okada EK. Flood regime, dam regulation and fish in the Upper Paraná River: effects on assemblage attributes, reproduction and recruitment. Rev Fish Biol Fish. 2004; 14:11–19. https://doi.org/10.1007/s11160-004-3551-y
Agostinho AA, Pelicice FM, Petry AC, Gomes LC, Júlio-Jr HF. Fish diversity in the upper Paraná River basin: habitats, fisheries, management and conservation. Aquat Ecosyst Health Manag. 2007; 10(2):174–86. https://doi.org/10.1080/14634980701341719
Agostinho AA, Pelicice FM, Gomes LC. Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz J Biol. 2008; 68(4):1119–32. https://doi.org/10.1590/S1519-69842008000500019
Agostinho AA, Vazzoler AEAM, Gomes LC, Okada EK. Estratificación espacial y comportamiento de Prochilodus scrofa en distintas fases del ciclo de vida en la planicie de inundación del alto rio Paraná y embalse de Itaipu, Paraná, Brasil. Rev Hydrobiol Trop. 1993; 26(1):79–90.
Andrian IF, Dória CR, Torrente G, Ferreti CML. Espectro alimentar e similaridade na composição da dieta de quatro espécies de Leporinus (Characiformes, Anostomidae) do rio Paraná, Brasil. Rev UNIMAR. 1994; 16(3):97–106.
Angert AL, Crozier LG, Rissler LJ, Gilman SE, Tewksbury JJ, Chunco AJ. Do species’ traits predict recent shifts at expanding range edges? Ecol Lett. 2011; 14(7):677–89. https://doi.org/10.1111/j.1461-0248.2011.01620.x
Araújo MB, New M. Ensemble forecasting of species distributions. Trends Ecol Evol. 2007; 22(1):42–47. https://doi.org/10.1016/j.tree.2006.09.010
Bailly D, Agostinho AA, Suzuki HI. Influence of the flood regime on the reproduction of fish species with different reproductive strategies in the Cuiabá River, Upper Pantanal, Brazil. River Res Appl. 2008; 24(9):1218–29. https://doi.org/10.1002/rra.1147
Bailly D, Batista-Silva VF, Cassemiro FAZ, Lemes P, Graça WJ, Oliveira AG et al. The conservation of migratory fishes in the second largest river basin of South America depends on the creation of new protected areas. Aquat Conserv. 2021; 31(9):2515–32. https://doi.org/10.1002/aqc.3594
Barletta M, Cussac VE, Agostinho AA, Baigun C, Catella A, Fontoura NF et al. Fishery ecology in South American river basins. In: Craig JF, editor. Freshwater fisheries ecology. Oxford: John Wiley & Sons Ltd; 2015. p.311–48.
Barletta M, Jaureguizar AJ, Baigun C, Fontoura NF, Agostinho AA, Almeida-Val VMF et al. Fish and aquatic habitat conservation in South America: a continental overview with emphasis on neotropical systems. J Fish Biol. 2010; 76(9):2118–76. https://doi.org/10.1111/j.1095-8649.2010.02684.x
Baumgartner G, Pavanelli CS, Baumgartner D, Bifi AG, Debona T, Frana VA. Peixes do baixo rio Iguaçu. Maringá: Eduem; 2012.
Bellard C, Bertelsmeier C, Leadley P, Thuiller W, Courchamp F. Impacts of climate change on the future of biodiversity. Ecol Lett. 2012; 15(4):365–77. https://doi.org/10.1111/j.1461-0248.2011.01736.x
Bernardinis GB, Cobos ME, Brum FT, Marques MCM, Peterson AT, Carlucci MB et al. Ecological restoration and protection of remnants are key to the survival of the critically endangered Araucaria tree under climate change. Glob Ecol Conserv. 2023; 47:e02668. https://doi.org/10.1016/j.gecco.2023.e02668
ecipitation data before their use in species distribution models. Austral Ecol. 2022; 47(7):1506–14. https://doi.org/10.1111/aec.13234
Brea M, Zucol A. The Paraná-Paraguay Basin: geology and paleoenvironments. In: Albert J, Reis R, editors. Historical biogeography of Neotropical freshwater fishes. Berkeley: University of California Press; 2011. p.69–88.
Britski HA, Birindelli JLO, Garavello JC. A new species of Leporinus Agassiz, 1829 from the upper Rio Paraná basin (Characiformes, Anostomidae) with redescription of L. elongatus Valenciennes, 1850 and L. obtusidens (Valenciennes, 1837). Pap Avulsos Zool. 2012; 52(37):441–75. https://doi.org/10.1590/S0031-10492012021700001
Brun P, Thuiller W, Chauvier Y, Pellissier L, Wüest RO, Wang Z, Zimmermann NE. Model complexity affects species distribution projections under climate change. J Biogeogr. 2020; 47(1):130 – 42. https://doi.org/10.1111/jbi.13734
Busby JR. BIOCLIM – a bioclimate analysis and prediction system. In: Margules CR, Austin MP, editors. Nature conservation: cost effective biological surveys and data analysis. Melbourne: CSIRO; 1991. p.64–68.
Cahill AE, Aiello-Lammens ME, Fisher-Reid MC, Hua X, Karanewsky CJ, Ryu HY et al. How does climate change cause extinction? Proc R Soc B. 2013; 280(1750):20121890. https://doi.org/10.1098/rspb.2012.1890
Carvajal-Quintero J, Villalobos F, Oberdorff T, Grenouillet G, Brosse S, Hugueny B et al. Drainage network position and historical connectivity explain global patterns in freshwater fishes’ range size. PNAS. 2019; 116(27):13434–39. https://doi.org/10.1073/pnas.1902484116
Casciotta JR, López HL, Menni RC, Miquelarena AM. The first fish fauna from the Salado River (central Argentina, South America) with additions to the Dulce River and limnological comments. Arch Hydrobiol. 1989; 115(4):603–12.
Companhia Ambiental do Estado de São Paulo (CETESB). Qualidade das águas interiores no estado de São Paulo [Internet]. São Paulo; 2023. Available from: https://cetesb.sp.gov.br/aguas-interiores/wp-content/uploads/sites/12/2024/11/RAI-2023-Relatorio-de-Qualidade-de-Aguas-Interiores-2023.pdf
Comte L, Olden JD. Climatic vulnerability of the world’s freshwater and marine fishes. Nat Clim Change. 2017; 7(10):718–22. https://doi.org/10.1038/nclimate3382
Conceição EO, Mantovano T, Campos R, Couto EV, Ferreira JHD, Rangel TF et al. Predicted changes in the distribution of Ostracoda (Crustacea) from river basins in the southern cone of South America, under two climate change scenarios. Hydrobiologia. 2023; 850:1443–60. https://doi.org/10.1007/s10750-023-05144-3
Copatti CE, Amaral R. Osmorregulação em juvenis de piava, Leporinus obtusidens (Characiformes: Anastomidae), durante trocas do pH da água. Bio Pampeana. 2009; 7(1):1–06.
Diniz-Filho JAF, Bini LM, Rangel TF, Loyola RD, Hof C, Nogués-Bravo D et al. Partitioning and mapping uncertainties in ensembles of forecasts of species turnover under climate change. Ecography. 2009; 32(6):897–906. https://doi.org/10.1111/j.1600-0587.2009.06196.x
Döll P, Zhang J. Impact of climate change on freshwater ecosystems: a global-scale analysis of ecologically relevant river flow alterations. Hydrol Earth Syst Sci. 2010; 14(5):783–99. https://doi.org/10.5194/hess-14-783-2010
Drago EC, Paira AR, Wantzen KM. Channel-floodplain geomorphology and connectivity of the Lower Paraguay hydrosystem. Ecohydrol Hydrobiol. 2008; 8(1):31–48. https://doi.org/10.2478/v10104-009-0003-2
Dudgeon D. Threats to freshwater biodiversity in a changing world. In: Freedman B, editor. Global environmental change. Dordrecht: Springer Netherlands; 2014, p.243–53.
Espinach Ros A. Migraciones de peces en el río Uruguay. In: Comisión Administradora del Río Uruguay, editor. Primeras jornadas sobre conservación de la fauna íctica en el río Uruguay. Paysandú: CARU; 1999. p.13–15.
Esser LF, Bailly D, Lima MR, Ré R. chooseGCM: a toolkit to select general circulation models in R. Glob Change Biol. 2025; 31(1):e70008. https://doi.org/10.1111/gcb.70008
Eyring V, Bony S, Meehl GA, Senior CA, Stevens B, Stouffer RJ et al. Overview of the Coupled Model Intercomparison Project Phase 6 (CMIP6) experimental design and organization. Geosci Model Dev. 2016; 9(5):1937–58. http://doi.org/10.5194/gmd-9-1937-2016
Ficke AD, Myrick CA, Hansen LJ. Potential impacts of global climate change on freshwater fisheries. Rev Fish Biol Fish. 2007; 17:581–613. https://doi.org/10.1007/s11160-007-9059-5
Fick SE, Hijmans RJ. WorldClim 2: new 1‐km spatial resolution climate surfaces for global land areas. Int J Climatol. 2017; 37(12):4302–15. https://doi.org/10.1111/2041-210X.13152
Field CB, Lobell DB, Peters HA, Chiariello NR. Feedbacks of Terrestrial Ecosystems to Climate Change. Ann Rev Env Resour. 2007; 32(1):1–29. https://doi.org/10.1146/annurev.energy.32.053006.141119
Gavin DG, Fitzpatrick MC, Gugger PF, Heath KD, Rodríguez-Sánchez F, Dobrowski SZ et al. Climate refugia: joint inference from fossil records, species distribution models and phylogeography. New Phytol. 2014; 204(1):37–54. https://doi.org/10.1111/nph.12929
Graça MA, Callisto M, Mello FT, Olarte DR, editors. Rivers of South America (1st ed.). Elsevier; 2024.
Graham V, Baumgartner JB, Beaumont LJ, Esperón-Rodríguez M, Grech A. Prioritizing the protection of climate refugia: designing a climate-ready protected area network. J Environ Plan Manag. 2019; 62(14):2588–606. https://doi.org/10.1080/09640568.2019.1573722
Griffiths D. Pattern and process in the distribution of North American freshwater fish. Biol J Linn Soc Lond. 2010; 100(1):46–61. https://doi.org/10.1111/j.1095-8312.2010.01404.x
Groves CR, Game ET, Anderson MG, Cross M, Enquist C, Ferdaña Z et al. Incorporating climate change into systematic conservation planning. Biodivers Conserv. 2012; 21:1651–71. https://doi.org/10.1007/s10531-012-0269-3
Hablützel PI, Huanto RB. Checklist and practical identification key for the cichlid fishes (Cichliformes: Cichlidae) of the La Plata drainage in Bolivia, including three new geographical records. Ecol Boliv. 2020; 55(1):46–61. https://doi.org/10.1101/785006
Hahn NS, Fugi R, Andrian IF. Trophic ecology of the fish assemblages. In: Thomaz SM, Agostinho AA, Hahn NS, editors. The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004. p.247–69.
Intergovernmental Panel on Climate Change (IPCC). Climate Change 2023: Synthesis Report. Genebra: Core Writing Team [Internet]. Genebra; 2023. Available from: https://www.ipcc.ch/report/ar6/syr/
Japkowicz N, Stephen S. The class imbalance problem: a systematic study. Intel Data Anal. 2002; 6(5):429–49. http://dx.doi.org/10.3233/IDA-2002-6504
Jézéquel C, Bigorne R, Liotta J, Oberdorff T, Tedesco PA. Metadata description of the Ictioplata database: a fish distribution database for the La Plata drainage basin. Freshw Metadata J. 2020; 46:1–06. https://doi.org/10.15504/fmj.2020.46
Jonsson B. Thermal effects on ecological traits of salmonids. Fishes. 2023; 8(7):337. https://doi.org/10.3390/fishes8070337
Keppel G, Van Niel KP, Wardell-Johnson GW, Yates CJ, Byrne M, Mucina L et al. Refugia: identifying and understanding safe havens for biodiversity under climate change. Glob Ecol Biogeogr. 2012; 21(4):393–404. https://doi.org/10.1111/j.1466-8238.2011.00686.x
Lehner B, Grill G. Global river hydrography and network routing: baseline data and new approaches to study the world’s large river systems. Hydrol Process. 2013; 27(15):2171–86. https://doi.org/10.1002/hyp.9740
Li D, Dorber M, Barbarossa V, Verones F. Global characterization factors for quantifying the impacts of increasing water temperature on freshwater fish. Ecol Indic. 2022; 142:109201. https://doi.org/10.1016/j.ecolind.2022.109201
Liotta J. Geographical distribution database of freshwater fishes from Argentina. Freshw Metadata J. 2020; 47:1–05. https://doi.org/10.15504/fmj.2020.47
Liu C, Berry PM, Dawson TP, Pearson RG. Selecting thresholds of occurrence in the prediction of species distributions. Ecography. 2005; 28(3):385–93. https://doi.org/10.1111/j.0906-7590.2005.03957.x
Liu C, Newell G, White B. On the selection of thresholds for predicting species occurrence with presence-only data. Ecol Evol. 2016; 6(1):337–48. https://doi.org/10.1002/ece3.1878
Lobo JM, Verdú JR, Numa C. Environmental and geographical factors affecting the Iberian distribution of flightless Jekelius species (Coleoptera: Geotrupidae). Divers Distrib. 2006; 12(2):179–88. https://doi.org/10.1111/j.1366-9516.2005.00214.x
Lopes TM, Bailly D, Almeida BA, Santos NCL, Gimenez BCG, Landgraf GO et al. Two sides of a coin: effects of climate change on the native and non-native distribution of Colossoma macropomum in South America. PLoS ONE. 2017; 12(6):1–18. https://doi.org/10.1371/journal.pone.0179684
Manel S, Williams HC, Ormerod SJ. Evaluating presence-absence models in ecology: the need to account for prevalence. J Appl Ecol. 2001; 38(5):921–31. https://doi.org/10.1046/j.1365-2664.2001.00647.x
Mounic-Silva CE, Lopes CA, Porto-Ferreira LSB, Nunes ME, Reynalte-Tataje DA, Zaniboni-Filho E. Spawning and recruitment areas of migratory fish in the Uruguay river: Applying for rivers connectivity conservation in South America. Bol Inst Pesca. 2019; 45(3):e510. https://doi.org/10.20950/1678-2305.2019.45.3.510
Naimi J, Araújo MB. SDM: a reproducible and extensible R platform for species distribution modelling. Ecography. 2016; 39(4):368–75. https://doi.org/10.1111/ecog.01881
Nakatani K, Bialeztki A, Baumgartner G, Sanches PV, Makrakis MC. Temporal and spatial dynamics of fish eggs and larvae. In: Thomaz SM, Agostinho AA, Hahn NS, editors. The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004. p.293–308.
Nix H. A biogeographic analysis of Australian elapid snakes. In: Longmore R, editor. Snakes: atlas of elapid snakes of Australia. Canberra: Bureau of Flora and Fauna; 1986. p.4–10.
Nobre CA, Marengo JA, Soares WR. Climate change risks in Brazil. Cham: Springer; 2019.
Oliveira AG, Suzuki HI, Gomes LC, Agostinho AA. Interspecific variation in migratory fish recruitment in the Upper Paraná River: effects of the duration and timing of floods. Environ Biol Fish. 2015; 98:1327–37. https://doi.org/10.1007/s10641-014-0361-5
Palmuti R, Dagosta F, de Pinna M. Peixes do Brasil. 1. ed. São Paulo: Marte; 2021.
Paranhos-Filho AC, Nummer AR, Albrez EA, Ribeiro AA, Machado R. A study of structural lineaments in Pantanal (Brazil) using remote sensing data. An Acad Bras Ciênc. 2013; 85(3):913–22. https://doi.org/10.1590/s0001-37652013000300007
Parmesan C, Yohe G. A globally coherent fingerprint of climate change impacts across natural systems. Nature. 2003; 421(6918):37–42. https://doi.org/10.1038/nature01286
Pelicice FM, Pompeu PS, Agostinho AA. Large reservoirs as ecological barriers to downstream movements of Neotropical migratory fish. Fish Fish. 2014; 16(4):697–715. https://doi.org/10.1111/faf.12089
Peluso LM, Mateus L, Penha J, Bailly D, Cassemiro F, Suárez Y et al. Climate change negative effects on the Neotropical fishery resources may be exacerbated by hydroelectric dams. Sci Total Environ. 2022; 828:2–10. https://doi.org/10.1016/j.scitotenv.2022.154485
Peterson AT, Soberón J, Pearson RG, Anderson RP, Martínez-Meyer E, Nakamura M et al. Ecological niches and geographic distributions. Princeton: Princeton University Press; 2011.
Phillips JC, McKinley GA, Bennington V, Bootsma HA, Pilcher DJ, Sterner RH et al. The potential for CO₂-induced acidification in freshwater. Oceanography. 2015; 28(2):136–45. https://doi.org/10.5670/oceanog.2015.37
Piana PA, Baumgartner G, Gomes LC. Influência da temperatura sobre o desenvolvimento de juvenis de piapara (Leporinus cf. obtusidens). Acta Sci Biol Sci. 2003; 25(1):87–94. https://doi.org/10.4025/actascibiolsci.v25i1.2103
Pörtner HO, Farrell AP. Physiology and climate change.Science. 2008; 322(5902):690–92. https://doi.org/10.1126/science.1163156
Quirós R, Bechara JA, Resende EK. Fish diversity and ecology, habitats and fisheries for the un-dammed riverine axis Paraguay-Parana-Rio de la Plata (Southern South America). Aquat Ecosyst Health Manag. 2007; 10(2):187–200. https://doi.org/10.1080/14634980701354761
Reis RB, Frota A, Depra GDC, Ota RR, Graca WJ. Freshwater fishes from Paraná State, Brazil: an annotated list, with comments on biogeographic patterns, threats, and future perspectives. Zootaxa. 2020; 4868(4):451–94. https://doi.org/10.11646/zootaxa.4868.4.1
Resende EK. Migratory fishes of the Paraguay-Paraná Basin, excluding the upper Paraná Basin. In: Carolsfeld J, Harvey B, Ros C, Baer A, editors. Migratory fishes of South America: biology, fisheries and conservation status. Victoria: World Fisheries Trust; 2003. p.99–156.
Riahi K, Vuuren DPV, Kriegler E, Edmonds J, O’neill BC, Fujimori S et al. The Shared Socioeconomic Pathways and their energy, land use, and greenhouse gas emissions implications: an overview. Glob Environ Change. 2017; 42:153–68. https://doi.org/10.1016/j.gloenvcha.2016.05.009
Rosa DM, Santos GB, Gomes PLA, Campos MCS, Dias JHP. Occurrence of Limnoperna fortunei (Dunker, 1857) in the fish diet from a south-eastern Brazilian reservoir. J Appl Ichthyol. 2014; 31:188–91. http://dx.doi.org/10.1111/jai.12623
Ruaro R, Conceição EO, Silva JC, Cafofo EG, Angulo-Valencia MA, Mantovano T et al. Climate change will decrease the range of a keystone fish species in La Plata River Basin, South America. Hydrobiologia.2019; 836(1):1–19. https://doi.org/10.1007/s10750-019-3904-0
Salazar-Torres GL, Silva HS, Rangel LM, Attayde JL, Huszar VLM. Cyanobacteria are controlled by omnivorous filter-feeding fish (Nile tilapia) in a tropical eutrophic reservoir. Hydrobiologia. 2015; 765:115–29. https://doi.org/10.1007/s10750-015-2406-y
Sales LP, Rodrigues R, Masiero R. Climate change drives spatial mismatch and threatens the biotic interactions of the Brazil nut. Global Ecol Biogeogr. 2021; 30(1):117–27. https://doi.org/10.1111/geb.13200
Sillero N, Barbosa AM. Common mistakes in ecological niche models. Int J Geogr Inf Sci. 2021; 35(2):213–26. https://doi.org/10.1080/13658816.2020.1798968
Sobral-Souza T, Vancine MH, Ribeiro MC, Lima-Ribeiro MS. Efficiency of protected areas in Amazon and Atlantic Forest conservation: a spatio-temporal view. Acta Oecol. 2018; 87:1–07. https://doi.org/10.1016/j.actao.2018.01.001
Seo C, Thorne JH, Hannah L, Thuiller W. Scale effects in species distribution models: implications for conservation planning under climate change. Biol Lett. 2009; 5:39–43. https://doi.org/10.1098/rsbl.2008.0476
Stein BA, Glick P, Edelson N, Staudt A. Climate-smart conservation: putting adaptation principles into practice. Washington: National Wildlife Federation; 2014.
Suzuki HI, Agostinho AA, Bailly D, Gimenes MF, Júlio Jr. HF, Gomes LC. Inter-annual variations in the abundance of young-of-the-year of migratory fishes in the upper Paraná River floodplain: relations with hydrographic attributes. Braz J Biol. 2009; 69(2):649–60. https://doi.org/10.1590/s1519-69842009000300019
Terribile LC, Diniz-Filho JAF, Marco Jr. P. How many studies are necessary to compare niche-based models for geographic distributions? Inductive reasoning may fail at the end. Braz J Biol. 2010; 70(2):263–69. https://doi.org/10.1590/S1519-69842010000200005
Thomaz SM, Agostinho AA, Hahn NS. The upper Paraná River and its floodplain. Leiden: Backhuys; 2004.
Thomaz SM, Bini LM, Bozelli RL. Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia. 2007; 579:1–13. https://doi.org/10.1007/s10750-006-0285-y
Urban MC. Accelerating extinction risk from climate change. Science. 2015; 348(6234):571–73. https://doi.org/10.1126/science.aaa4984
Vadeboncoeur Y, McCann KS, Vander-Zanden MJ, Rasmussen JB. Effects of multi-chain omnivory on the strength of trophic control in lakes. Ecosystems. 2005; 8(6):682–93. https://doi.org/10.1007/s10021-003-0149-5
Valenzuela-Aguayo F, McCracken GR, Manosalva A, Habit E, Ruzzante DE. Human-induced habitat fragmentation effects on connectivity, diversity, and population persistence of an endemic fish, Percilia irwini, in the Biobío River basin (Chile). Evol Appl. 2020; 13(4):794–807. https://doi.org/10.1111/eva.12901
Vanni MJ. Nutrient cycling by animals in freshwater ecosystems. Annu Rev Ecol Syst. 2002; 33:341–70. https://doi.org/10.1146/annurev.ecolsys.33.010802.150519
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE. The river continuum concept. Can J Fish Aquat Sci. 1980; 37(1):130–37. https://doi.org/10.1139/f80-017
Vliet MTV, Franssen WH, Yearsley JR, Ludwig F, Haddeland I, Lettenmaier DP et al. Global river discharge and water temperature under climate change. Glob Environ Change. 2013; 23(2):450–64. https://doi.org/10.1016/j.gloenvcha.2012.11.002
Wang Z, Wang T, Zhang X, Wang J, Yang Y, Sun Y et al. Biodiversity conservation in the context of climate change: facing challenges and management strategies. Sci Total Environ. 2024; 937:173377. https://doi.org/10.1016/j.scitotenv.2024.173377
Ward J. Riverine landscapes: biodiversity patterns, disturbance regimes, and aquatic conservation. Biol Conserv. 1998; 83(3):269–78. https://doi.org/10.1016/S0006-3207(97)00083-9
Williams JW, Jackson ST. Novel climates, no-analog communities, and ecological surprises. Front Ecol Environ. 2007; 5(9):475–82. https://doi.org/10.1890/070037
Woodward G, Perkins DM, Brown LE. Climate change and freshwater ecosystems: impacts across multiple levels of organization. Philos Trans R Soc B. 2010; 365(1549):2093–106. https://doi.org/10.1098/rstb.2010.0055
Wysocki LE, Montey K, Popper AN. The influence of ambient temperature and thermal acclimation on hearing in a eurythermal and a stenothermal otophysan fish. J Exp Biol. 2009; 212(19):3091–99. https://doi.org/10.1242/jeb.033274
Zaniboni-Filho E, Schulz UH. Migratory fishes of the Uruguay River. In: Carolsfeld J, Harvey B, Ross C, Baer A, editors. Migratory fishes of South America: biology, fisheries and conservation status. Victoria, Canada: World Fisheries Trust; International Development Research Centre; World Bank; 2003. p.161–98.
Zeiringer B, Seliger C, Greimel F, Schmutz S. River hydrology, flow alteration, and environmental flow. In: Petts G, Mesterhézy A, Moreira M, editors. Riverine ecosystem management: science for governing towards a sustainable future. Cham: Springer International Publishing, 2018. p.67–89.
Zizka A, Silvestro D, Andermann T, Azevedo J, Ritter CD, Edler D et al. CoordinateCleaner: standardized cleaning of occurrence records from biological collection databases. Methods Ecol Evol. 2019; 10(5):744–51. https://doi.org/10.1111/2041-210X.13152
Authors
![]() Beatriz Cristina de Paula de Souza1,
Beatriz Cristina de Paula de Souza1, ![]() Luana Caroliny Possamai2,
Luana Caroliny Possamai2, ![]() Katia Yasuko Yofukuji3,
Katia Yasuko Yofukuji3, ![]() Taise Miranda Lopes2,4,
Taise Miranda Lopes2,4, ![]() Luiz Fernando Esser2,
Luiz Fernando Esser2, ![]() José Hilário Delconte Ferreira5,
José Hilário Delconte Ferreira5, ![]() Reginaldo Ré5,
Reginaldo Ré5, ![]() Weferson Júnio da Graça2,6,
Weferson Júnio da Graça2,6, ![]() Ana Francisca Gomes da Silva1,
Ana Francisca Gomes da Silva1, ![]() Jefferson Matheus Barros Ozório7,
Jefferson Matheus Barros Ozório7, ![]() Dayani Bailly2,6
Dayani Bailly2,6 ![]() and
and ![]() Valéria Flávia Batista-Silva1
Valéria Flávia Batista-Silva1
[1] Universidade Estadual de Mato Grosso do Sul (UEMS), Programa de Pós-Graduação em Biodiversidade e Sustentabilidade Ambiental (PGBSA), Grupo de Estudo em Ciências Ambientais e Educação (GEAMBE), BR-163, 235, 79980-000, Mundo Novo, MS, Brazil. (BCPS) beatrizarnhorn@gmail.com, (AFGS) ana.francisca@uems.br, (VFBS) vfb_silva@uems.br.
[2] Universidade Estadual de Maringá (UEM), Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais (PEA), Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil. (LCP) luanacarolinypossamai@gmail.com, (TML) taisemlopes@gmail.com, (LFE) luizesser@gmail.com, (WJG) weferson@nupelia.uem.br, (DB) dayanibailly@nupelia.uem.br (corresponding author).
[3] CIMO, LA SusTEC, Instituto Politécnico de Bragança, Campus de Santa Apolónia, 5300-253, Bragança, Portugal. (KYY) kayofukuji96@gmail.com.
[4] Universidade Estadual do Oeste do Paraná (UNIOESTE), Campus Cascavel, Programa de Pós-Graduação em Conservação e Manejo de Recursos Naturais, Rua Universitária, 1619, 85819-110, Cascavel, PR, Brazil.
[5] Universidade Tecnológica Federal do Paraná (UTFPR), Via Rosalina Maria dos Santos, 1233, 87301-899, Campo Mourão, PR, Brazil. (JHDF) jhdferreira@gmail.com, (RR) reginaldo@utfpr.edu.br.
[6] Universidade Estadual de Maringá (UEM), Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil.
[7] Universidade Estadual de Mato Grosso do Sul (UEMS), Cidade Universitária de Dourados, 79804-970, Dourados, MS, Brazil. (JMBO) ozorio.jmb@outlook.com.
Authors’ Contribution 

Beatriz Cristina de Paula de Souza: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Luana Caroliny Possamai: Formal analysis, Visualization, Writing-review and editing.
Katia Yasuko Yofukuji: Data curation, Visualization, Writing-review and editing.
Taise Miranda Lopes: Formal analysis, Writing-review and editing.
Luiz Fernando Esser: Methodology, Software, Writing-review and editing.
José Hilário Delconte Ferreira: Software, Writing-review and editing.
Reginaldo Ré: Software, Writing-review and editing.
Weferson Júnio da Graça: Data curation, Validation, Writing-review and editing.
Ana Francisca Gomes da Silva: Visualization, Writing-review and editing.
Jefferson Matheus Barros Ozório: Formal analysis, Writing-review and editing.
Dayani Bailly: Conceptualization, Data curation, Formal analysis, Methodology, Writing-review and editing.
Valéria Flávia Batista-Silva: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
This manuscript does not require approval from the Ethical Committee for Animal Use in Experiments, since the data were obtained from publicly available online databases.
Competing Interests
The author declares no competing interests.
Data availability statement
The data supporting the findings of this study are available from the corresponding author, upon reasonable request.
AI statement
ChatGPT (OpenAI) was used for English language editing. All outputs were reviewed and approved by the authors.
Funding
This study was financed in part by the “Programa Institucional de Bolsas aos Alunos de Pós-Graduação” (PIBAP/UEMS – proc. 86891363000180/2024-1) which granted a master’s scholarship to BCPS, by the “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” (CAPES – proc. 88887.968266/2024-00) which granted a master’s scholarship to LCP, by the “Fundação de Ciência e Tecnologia” (FCT – MCTES/PIDDAC: CIMO, UIDB/00690/2020 and SusTEC – LA/P/0007/2020) which supported the postdoctoral fellowship of KYY, and by the “Fundação Araucária/SETI” which granted postdoctoral fellowships to LEF (proc. 683/2022) and TML (proc. 154/2021). WJG received funding from the “Conselho Nacional de Desenvolvimento Científico e Tecnológico” (CNPq – proc. 303571/2024-1) through the research productivity fellowship.
How to cite this article
Souza BCP, Possamai LC, Yofukuji KY, Lopes TM, Esser LF, Ferreira JHD, Ré R, Graça WJ, Silva AFG, Ozório JMB, Bailly D, Batista-Silva VF. Climate change affects the potential distribution of a Neotropical freshwater migratory fish. Neotrop Ichthyol. 2026; 24(1):e250091. https://doi.org/10.1590/1982-0224-2025-0091
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted January 23, 2026
Accepted January 23, 2026
![]() Submitted May 16, 2025
Submitted May 16, 2025
![]() Epub April 27. 2026
Epub April 27. 2026