![]() Pedro L. C. Uzeda1
Pedro L. C. Uzeda1 ![]() ,
, ![]() Bruno H. M. Stabile2,
Bruno H. M. Stabile2, ![]() Renan B. dos Reis2,
Renan B. dos Reis2, ![]() Luana J. Sartori3,
Luana J. Sartori3, ![]() Carla S. Pavanelli2,4,5,
Carla S. Pavanelli2,4,5, ![]() Vladimir P. Margarido6 and
Vladimir P. Margarido6 and ![]() Weferson J. da Graça2,4,5
Weferson J. da Graça2,4,5
PDF: Download Here | Supplementary: S1 | Cite this article
Associate Editor: ![]() George Mattox
George Mattox
Section Editor: ![]() William Crampton
William Crampton
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
A new species of Characidium is described from the upper rio Paraná basin, Brazil, based on morphological and molecular data. It is distinguished from congeners by its unique red body coloration in adults, a naked isthmus with scaled chest, absence of a skin flap around the posterior naris, and a reduced foramen for the ophthalmic nerve. Juveniles (<35.0 mm SL) display a pattern of dark spots resembling members of the C. lauroi group, although not closely related. Molecular analyses of the mitochondrial COI marker place the species within a clade comprehending lineages related to C. fasciatum and C. gomesi. A novel secondary sexual dimorphism is reported for the genus: females have a slender snout, while males possess a more robust one. The species is restricted to the upper rio Grande sub-basin, inhabiting deep pools with submerged wood debris. Divergence from its sister lineage occurred around 3 million years ago, likely associated with hydrographic rearrangements between the upper Paraná and São Francisco basins. This discovery reveals underestimated diversity within the upper rio Paraná basin and emphasizes the importance of continued ichthyofaunal surveys in undersampled tributaries.
Keywords: Characidiinae, Freshwater fish, La Plata basin, Species complex, Species delimitation.
Introduction
Characidium Reinhardt, 1867 is the most species-rich genus within Crenuchidae, currently comprising 90 valid species (Fricke et al., 2026) distributed from Central America to Patagonia (Buckup, 2004). Members of this genus exhibit morphological adaptations to a wide range of habitats, such as depressed bodies and robust pectoral fins in rapids and riffles, or deep bodies and slender pectoral fins in sandy banks (Leal et al., 2011; Souza, Pompeu, 2020). Additionally, Characidium species display distinct coloration patterns of bars and blotches, which have been useful for diagnostic purposes (Buckup, Reis, 1997; Graça, Pavanelli, 2008), and even phylogenetic indicators, such as the presence of midlateral spots in the C. lauroi group (Melo, 2001; Melo et al., 2016; Oliveira-Silva et al., 2024).
The most common coloration pattern in Characidium consists of several dark vertical bars on a brownish background along the body (Buckup, 1993a). Consequently, species with distinct coloration patterns can be easily diagnosed, such as the green body of Characidium iaquira Zanata, Ohara, Oyakawa & Dagosta, 2020, the deep yellow fins of Characidium xanthopterum Silveira, Langeani, da Graça, Pavanelli & Buckup, 2008 (see live coloration in Melo et al. (2021)), or the purple hue of Characidium purpuratum Steindachner, 1882, Characidium boavistae Steindachner, 1915, and Characidium bolivianum Pearson, 1924. These coloration traits may be associated with sexual dimorphism (Melo, Oyakawa, 2015; Agudelo-Zamora et al., 2020) or ontogenetic variation (Zanata et al., 2020).
Recent expeditions to the headwaters of rio Grande, in the upper rio Paraná basin, have revealed the existence of a Characidium species exhibiting remarkable ontogenetic color changes and sexual dimorphism. By integrating morphological and molecular data, we hypothesize its phylogenetic relationship among congeners and formally describe it as a new species.
Material and methods
Sample collection. Samplings were conducted using sieves during the dry season. After capture, specimens were anesthetized in 0.5 mL/L eugenol solution, photographed, and preserved. Specimens designated for morphological analysis were fixed in 10% formalin solution for seven days and then transferred to 70% ethanol for long-term storage and handling. Those intended for molecular analysis (identified with “mol.” in paratypes list) were fixed in 99.2% ethanol and stored at -22ºC until muscle tissue sampling. Institutional abbreviations follow Fricke, Eschmeyer (2026).
Morphological data. Measurements and counts were taken from the left side of specimens using a digital caliper with a precision of 0.1 mm, following Buckup (1993a) and Melo, Oyakawa (2015). The measures are presented as percentages of standard length (SL), except for subunits of the head, which are given as percentages of head length (HL). In cases where scales were lost, certain counts could not be recorded or were taken from the right side instead. Sex determination was performed by dissecting six specimens under a stereomicroscope. Four specimens were cleared and stained (c&s) for osteological analysis following the protocol of Taylor, Van Dyke (1985). Osteological nomenclature follows Weitzman (1962). Vertebral counts included the first five elements modified into the Weberian apparatus, while the caudal centrum was counted as a single element. In the Diagnosis section, character states for C. boavistae, C. bolivianum, and C. purpuratum were taken from the morphological matrix provided by Buckup (1993b). In the Description section, each meristic character is followed by the number of specimens examined in parentheses, and the counts of the holotype marked by an asterisk. By ‘chest’ we refer to the ventral area of the body located between the bases of contralateral pectoral fins.
Molecular data. DNA was extracted from muscle tissue using the Wizard Genomic DNA Purification Kit (Promega) following the manufacturer’s protocol. After quantification with a NanoDrop™ Lite Spectrophotometer, a partial region of the cytochrome c oxidase subunit I (COI) was amplified using the specific primers FishF1 5’-TCAACCAACCACAAAGACATTCGGCAC-3’ (Ward et al., 2005) and FR1d 5’-CACCTCAGGGTGTCCGAARAAYCARAA-3’ (Ivanova et al., 2007). The polymerase chain reaction (PCR) was performed in a mix containing Tris-KCl 1x reaction buffer [20 mM Tris-HCl (pH 8.4), 50 mM KCl], MgCl2 (1.5 mM), primers (2.5 μM each), dNTPs (0.1 mM each), 1U of Taq DNA polymerase, 10 ng of template DNA, and ultrapure water to fill 25-μL. The PCR conditions consisted of an initial denaturation at 95°C for 5 min, followed by 35 cycles at 94°C for 30 s, 52°C for 40 s, and 72°C for 1 min, with a final extension at 72°C for 10 min (adapted from Ivanova et al., 2007). PCR products were purified with polyethylene glycol 8000 (Rosenthal et al., 1993) and sequenced on an ABI™ Sanger Sequencing 3500 Genetic Analyzer. We obtained four sequences of the new species (one haplotype), as well three sequences from topotypes of C. gomesi (one haplotype). Access to the genetic heritage of this species was authorized by the National System for Management of Genetic Heritage and Associated Traditional Knowledge (SISGEN, registration A18C108). All sequences used in this study are listed in Tab. S1.
Phylogenetic reconstruction and time calibration. The generated sequences were edited using BioEdit (Hall, 1999). Additional COIsequences of the most closely related Characidium species, the clade D sensu Oliveira-Silva et al. (2024) as identified by BLASTn tool (Johnson et al., 2008), were obtained from GenBank. We selected Crenuchus spilurus Günther, 1863, Poecilocharax weitzmani Géry, 1965, and Melanocharacidium blennioides (Eigenmann, 1909) as outgroups for the phylogenetic reconstruction. The dataset was aligned by ClustalW (Thompson et al., 1994) algorithm in MEGA X (Kumar et al., 2018). The best nucleotide substitution model for Bayesian Inference (BI) analysis was estimated in jModelTest v. 2.1.10 (Darriba et al., 2012).
The BI tree was constructed in BEAST v. 1.10.4 (Suchard et al., 2018) with the HKY substitution model with gamma distribution plus invariable sites, an uncorrelated molecular clock with lognormal distribution, and the birth-death process as tree prior. Parameters were set to 20,000,000 runs, 1,000 generations, and trees sampled every 1,000 generations. To estimate the divergence time between the new species and its closest congeners, in the absence of crenuchid fossil records, we calibrated our tree based on the estimates provided by Melo et al. (2022). In that study, based on 57 UCE loci (17,400 bp) and six characoid fossils from five families, the authors estimated the divergence between the Crenuchinae (Crenuchus Günther, 1863 + Poecilocharax Eigenmann, 1909) and the Characidiinae (herein represented by M. blennioides and Characidium spp.) approximately 78 million years ago. This constraint was set to a normal distribution with a standard deviation of 1.0. Chain convergence was analyzed using Tracer v. 1.10.4 to determine the stationary phase, with an effective sample size exceeding 200 (Rambaut et al., 2018). Ten percent of the chain was discarded as a burn-in procedure in TreeAnnotator v. 1.10.4 to obtain the Maximum Clade Credibility Tree (MCC) (Suchard et al., 2018), which was edited using the Interactive Tree Of Life (iTOL) (Letunic, Bork, 2024).
Species delimitation. To further assess the support of the new species based on molecular data, we performed three species delimitation methods: the distance-based Assemble Species by Automatic Partitioning (ASAP, Puillandre et al., 2021); and the coalescent-based methods General Mixed Yule-Coalescent (GMYC, Pons et al., 2006; Fujisawa, Barraclough, 2013), and the Poisson Tree Process in its Bayesian implementation (bPTP, Zhang et al., 2013). To optimize the delimitations to the evolutionary context of the new species, our ingroup consisted of only sequences recovered in the same subclade as the new taxon in the phylogenetic analysis (clade E2).
The ASAP analysis was conducted by directly inputting the aligned sequences on the ASAP web server (https://bioinfo.mnhn.fr/abi/public/asap/asapweb.html), using the Kimura 2-parameter (K80; 2.0) substitution model and all other parameters set to default. For the GMYC analysis, an ultrametric tree was constructed in BEAST from unique haplotypes of our ingroup, with the HKY substitution model with gamma distribution plus invariable sites, a lognormal relaxed molecular clock, and the Yule process as tree prior. Parameters were set to 20,000,000 runs, 1,000 generations, and trees sampled every 1,000 generations. The MCC was obtained as described above, and the Newick file was submitted as input on the GMYC web server (https://species.h-its.org/gmyc/), using the single-threshold method. For the bPTP analyses, the previously constructed BI tree was used as an input on the PTP web server (https://species.h-its.org/ptp/), using 100,000 Markov chain Monte Carlo generations and a 0.1 burn-in rate.
Conservation status. The conservation status assessment of the new species was conducted following the criteria and recommendations of the International Union for Conservation of Nature (IUCN Standards and Petitions Subcommittee, 2024). The extent of occurrence was calculated using QGIS v.3.30.2 (QGIS, 2025), defined as the area of the smallest sub-basin encompassing the entire known distribution of the new species, based on the HydroBASINS database (Lehner, Grill, 2013).
Results
Characidium urucum,new species
urn:lsid:zoobank.org:act:9CCDFB22-BFA5-41DB-9DAF-27765FDFC2D1
(Figs. 1–8; Tab. 1)
Characidium sp. —Pompeu et al., 2009:663 (tab. 2).
Characidium sp. —Silva-Sene et al., 2024:4 (tab. 1, ichthyofauna inventory).
Holotype. NUP 25755, male, 44.1 mm SL, Brazil, State of Minas Gerais, Carrancas, upper rio Paraná basin, rio Grande sub-basin, rio Capivari, bridge on Estrada Real, 21°30’45”S 44°38’57”W, 970 m a.s.l., 10 Aug 2023, P. L. C. Uzeda & L. Sartori.
Paratypes. All from Minas Gerais State, rio Grande sub-basin, upper rio Paraná basin. Rio Capivari: CI-UFLA 2177, 11, 30.5–45.9 mm SL, NUP 25703, 14, 30.3–44.5 mm SL, NUP 25800, 3, 31.4–32.3 mm SL, mol., collected with holotype. CI-UFLA 3782, 1, 38.9 mm SL, C. P. Porto, G. S. do Vale, T. S. T. Rodrigues, V. A. Urbano & P. L. C. Uzeda, 17 Aug 2023, same locality as holotype. CI-UFLA 3783, 3, 34.4–40.9 mm SL, CI-UFLA 3784, 3, 32.2–42.3 mm SL, CI-UFLA 4005, 4 c&s, 38.3–41.8 mm SL, DZSJRP 25172, 5, 32.4–44.8 mm SL, MCNIP 5449, 4, 31.8–40.5 mm SL, MZUFV 13331, 4, 34.9–49.6 mm SL, collected with CI-UFLA 3782. Córrego dos Cabritos, affluent to rio Capivari: CI-UFLA 0007, 8, 25.0–42.7 mm SL, 21°32’42”S 44°43’16”W, 979 m a.s.l., 7 Oct 2007, P. S. Pompeu & Lab. Eco. Peixes team. Ribeirão da Cachoeira, affluent to rio Ingaí: CI-UFLA 3168, 2, 44.2–45.1 mm SL, 21°31’47”S 44°52’06”W, 1000 m a.s.l., Jun 2010, P. H. Borrego & Lab. Eco. Peixes team. CI-UFLA 3179, 2, 47.6–51.8 mm SL, 21°31’47”S 44°52’06”W, 1.000 m a.s.l., Jun 2010, P. H. Borrego & Lab. Eco. Peixes team. Riacho da Vargem Grande, affluent to rio Capivari: UFMG-ICT 4030, 5, 30.8–39.1 mm SL, 21º28’25”S 44º42’38”W, 945 m a.s.l., 12 Jul 2025, P. L. C. Uzeda & V. P. Margarido. Rio Grande, downstream from Cachoeira do rio Grande:CI-UFLA 1548, 4, 35.0–53.0 mm SL, 22°13’8”S 44°32’21”W, 1.232 m a.s.l., 10 Jun 2023, P. L. C. Uzeda & L. J. Sartori.Ribeirão Lambari, affluent to rio Jacaré:CI-UFLA 3110, 1, 45.4 mm SL, CI-UFLA 3112, 1, 44.9 mm SL, municipality of Oliveira, coordinates unavailable, 24 Apr 2018, S. Saraiva & Lab. Eco. Peixes team. Córrego Corguinho, affluent to rio Aiuruoca: CI-UFLA 150, 1, 65.6 mm SL, 22°10’24”S 44°37’07”W, 1.120 m a.s.l., 2 Apr 2010, R. Casarim & Lab. Eco. Peixes team.
FIGURE 1| Dorsal, lateral, and ventral views of Characidium urucum, holotype, NUP 25755, male, 44.1 mm SL, Brazil, State of Minas Gerais, Carrancas, rio Grande sub-basin, rio Capivari, bridge on Estrada Real. Scale bar = 10 mm.
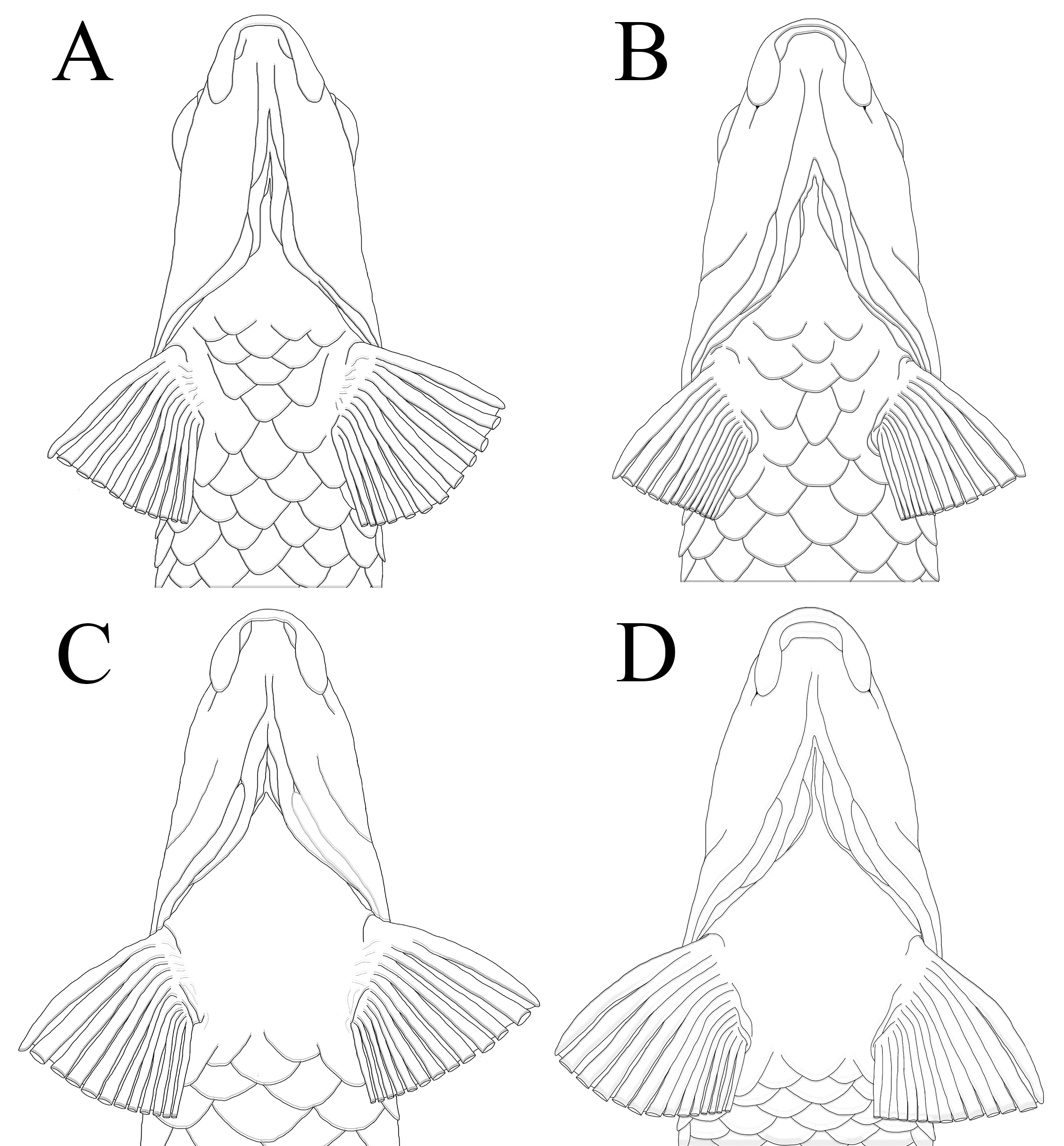
Diagnosis. Characidium urucum differs from most congeners, except for C. alipioi Travassos, 1955, C. amaila Lujan, Agudelo-Zamora, Taphorn, Booth & López-Fernández, 2013, C. boavistae, C. bolivianum, C. crandellii Steindachner, 1915, C. cricarense Malanski, Sarmento-Soares, Silva-Malanski, Lopes, Ingenito & Buckup, 2019, C. declivirostre Steindachner, 1915, C. duplicatum Armbruster, Lujan & Bloom, 2021, C. fasciatum Reinhardt, 1867, C. gomesi Travassos, 1956, C. grajahuense Travassos, 1944, C. hasemani Steindachner, 1915, C. helmeri Zanata, Sarmento-Soares & Martins-Pinheiro, 2015, C. iaquira, C. japuhybense Travassos, 1949, C. kalunga Melo, Bouquerel, Masumoto, França & Netto-Ferreira, 2021, C. kamakan Zanata & Camelier, 2015, C. krenak Oliveira-Silva, Santos, Lopes & Zanata, 2022, C. lanei Travassos, 1967, C. lauroi Travassos, 1949, C. nambiquara Zanata & Ohara, 2020, C. oiticicai Travassos, 1967, C. pterostictum, C. purpuratum, C. schubarti Travassos, 1955, C. summus Zanata & Ohara, 2015, C. tatama, C. timbuiense, C. travassosi Melo, Buckup & Oyakawa, 2016, C. tupi Zanata & Oliveira-Silva, 2025, C. vidali, and C. wangyapoik Armbruster, Lujan & Bloom, 2021 by having a naked isthmus (vs. isthmus scaled). Characidium urucum can be diagnosed from all aforementioned species, except for members of the C. lauroi species group (i.e., C. lauroi, C. japuhybense, C. oiticicai, C. schubarti, C. travassosi and C. tupi), by lacking scales on the area between the anterior limit of the isthmus and anterior margin of the cleithrum, and a large area surrounding the bases of the pectoral fins (Figs. 2A–B) (vs. lacking scales only in the area between the anterior limit of the isthmus and anterior margin of the cleithrum in C. alipioi, C. cricarense, C. hasemani, C. helmeri, C. kalunga, C. kamakan, C. krenak, C. lanei, C. pterostictum, C. purpuratum, C. summum and C. vidali; chest completely naked in C. amaila, C. boavistae, C. bolivianum, C. fasciatum, C. gomesi, C. grajahuense, C. nambiquara, C. tatama and C. timbuiense (Figs. 2C–D); naked area extending slightly posterior to bases of contralateral pectoral fins in C. iaquira, and posteriorly to bases of pelvic fins in C. crandellii, C. declivirostre, C. duplicatum and C. wangyapoik). The new species can be diagnosed from members of the C. lauroi species group by the presence of the parietal branch of the supraorbital sensory canal (vs. parietal branch absent), and by the absence of numerous darks spots along the flank on adult specimens (longer than 35.0 mm SL) (vs. members of the C. lauroi species group with numerous dark spots along the flank on juveniles and adult specimens), and from C. tupi by the absence of a series of rounded or squared dark blotches along lateral line scales (vs. presence), an by the absence of a 3-shaped dark band on caudal fin (vs. presence).
FIGURE 2| Schematic view of ventral scaling patterns in Characidium species with naked isthmus. A. Characidium urucum, paratype, male, MZUFV 13331, 41.7 mm SL and B. Characidium oiticicai, CI-UFLA 3652, 48.7 mm SL, both with naked area restricted to the region between branchiostegal membranes and surrounding the bases of pectoral fins. C. Characidium fasciatum, CI-UFLA 3290, 64.1 mm SL and D. Characidium gomesi, CI-UFLA 3828, 58.2 mm SL, both with naked area extending over the entire pectoral region and posterior to bases of pectoral fins.
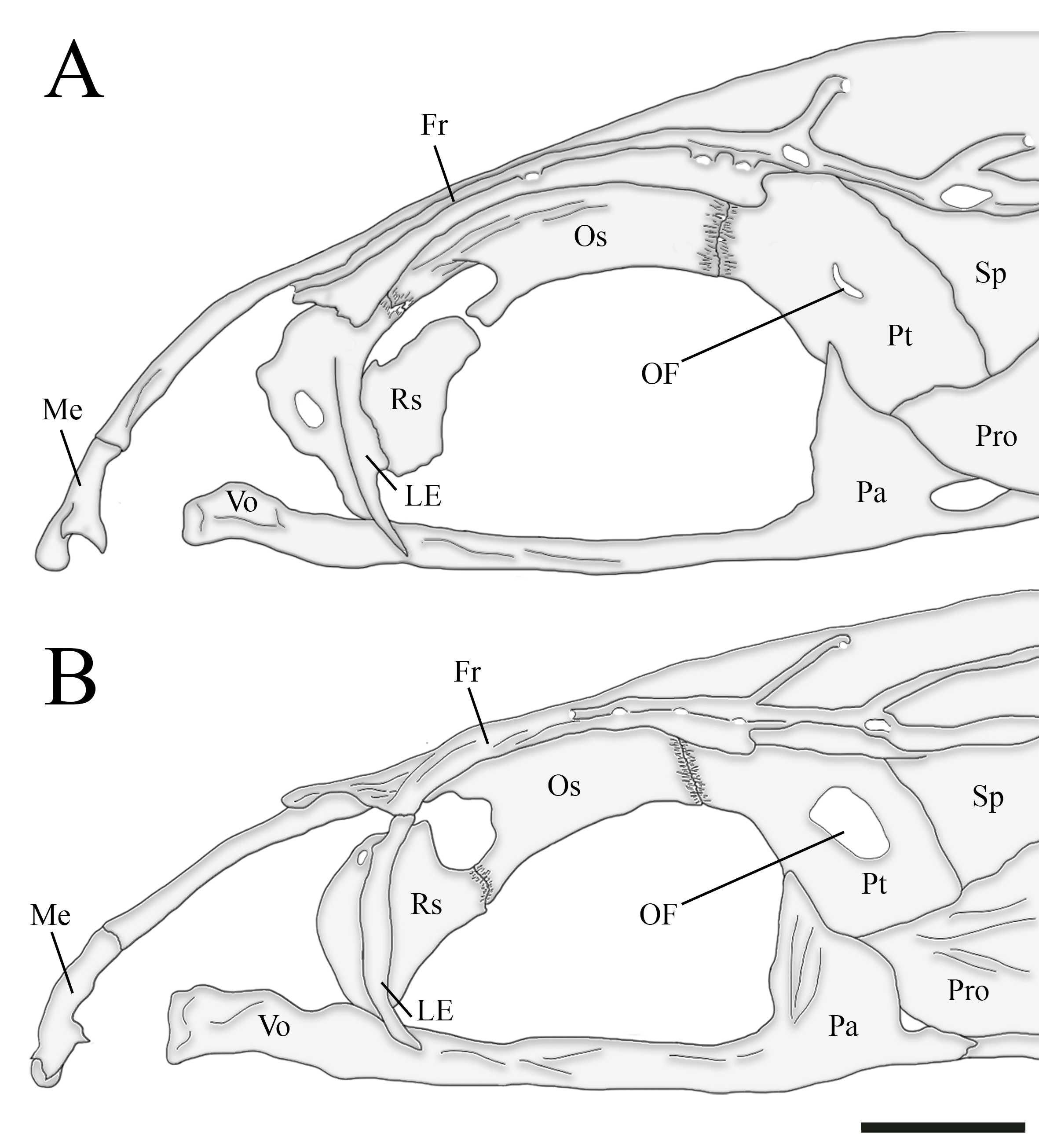
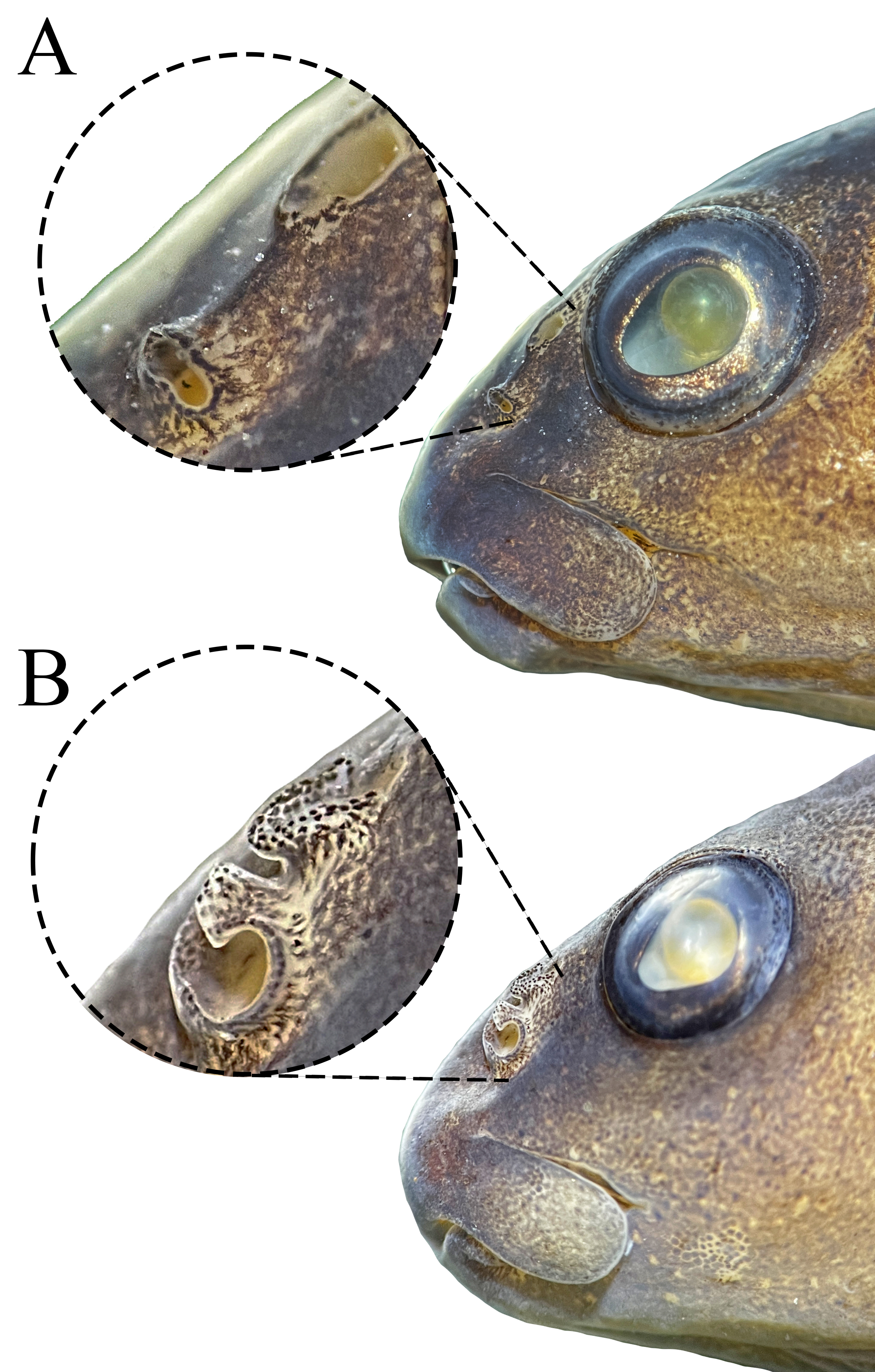
Characidium urucum is herein recovered as part of a clade comprising C. bolivianum, C. fasciatum, C. gomesi and C. purpuratum (E2 clade sensu Oliveira-Silva et al., 2024, and part of C2 clade sensu Buckup, 1993b), and can be diagnosed from these species by having the pterosphenoid foramen for the ophthalmic nerve comma-shaped and reduced in diameter, in such a way that the brain cavity is not visible through its opening (Fig. 3A) (vs. pterosphenoid foramen for the ophthalmic nerve rounded and exceptionally large; brain cavity visible through its opening; Fig. 3B). Additionally, the new species can be easily diagnosed from C. bolivianum, C. fasciatum and C. gomesi by having a naked isthmus with scaled chest (Fig. 2A) (vs. isthmus and chest completely naked, Figs. 2C–D); from the closely related C. fasciatum and C. gomesi by having the skin flap surrounding the posterior naris poorly developed or absent (Fig. 4A) (vs. skin flap surrounding the posterior naris well developed, Fig. 4B); from C. gomesi by the absence of conspicuous dark bars on fins (vs. presence of conspicuous dark bars on dorsal, adipose and anal fins, and a 3-shaped bar at caudal fin); and from C. purpuratum by having 33 to 36 lateral-line scales (vs. 32 or less).
FIGURE 3| Lateral view of the neurocranium of two Characidium species. A. Characidium urucum, CI-UFLA 4005, 39.8 mm SL, c&s paratype, showing the small, comma-shaped foramen of the ophthalmic nerve (OF). B. Characidium fasciatum, CI-UFLA 2385, 53.1 mm SL, showing the large and rounded foramen of the ophthalmic nerve (OF). Bones are represented as Fr (frontal), LE (lateral ethmoid), Me (mesethmoid), Os (orbitosphenoid), Pa (parasphenoid), Pro (prootic), Pt (pterosphenoid), Rs (rhinosphenoid), Sp (sphenotic), and Vo (vomer). Scale bar = 1 mm.
FIGURE 4| Detail of nares exhibited by Characidium species. A. Characidium urucum, CI-UFLA 1548, 52.9 mm SL, paratype, female, showing the absence of the skin flap surrounding the posterior naris. B. Characidium fasciatum, CI-UFLA 3290, 49.3 mm SL, showing the prominent skin flap surrounding the posterior naris.
Description. General external morphology illustrated in Figs. 1, 7–8. Morphometric data in Tab. 1. In lateral view, body elongated and robust. Head rounded; mouth subterminal. Body profile pronouncedly ascending from snout tip to interorbital region, gradually ascending to vertical through middle of pectoral fin, and gently descending to end of caudal peduncle. Body deepest at vertical through middle of pectoral fin. Ventral profile of body slightly rounded and convex from tip of lower lip to anal-fin origin; straightly ascending at anal-fin base, and horizontally continuing to end of caudal peduncle. In dorsal view, body elongated and fusiform; snout tip squared or roughly rounded; body width increasing to tip of operculum, and progressively decreasing to end of caudal peduncle. Pectoral fin long and arched, posteriorly curved; pelvic fin triangular. In ventral view, body elongated and fusiform; snout tip squared or roughly rounded. Body width increasing from dentary tip to operculum, and progressively decreasing to end of caudal peduncle. Eyes protruding out of head, their edges visible in ventral view. Nares separated; posterior naris slightly closer to orbit than to anterior naris. Margins of anterior naris margins raised, forming circular rim. Posterior naris without semicircular skin flap around margins; when present, flap minute and restricted to anterior margin of naris.
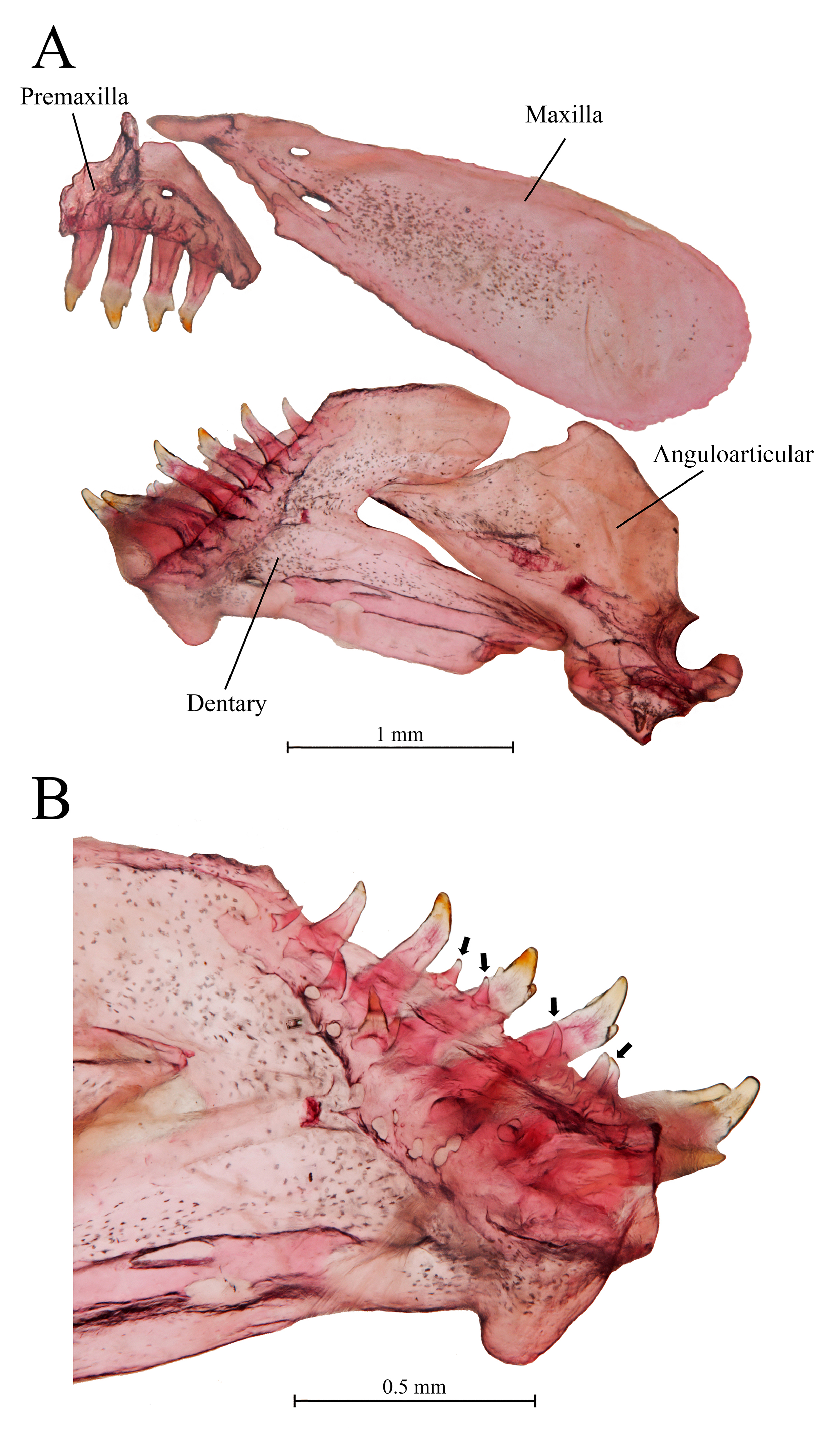
FIGURE 5| Jaws and teeth of Characidium urucum, paratype, CI-UFLA 4005. A. Lateral view of upper and lower jaws. B. Medial view of dentary bone. Black arrows indicate dentary teeth from the inner series.
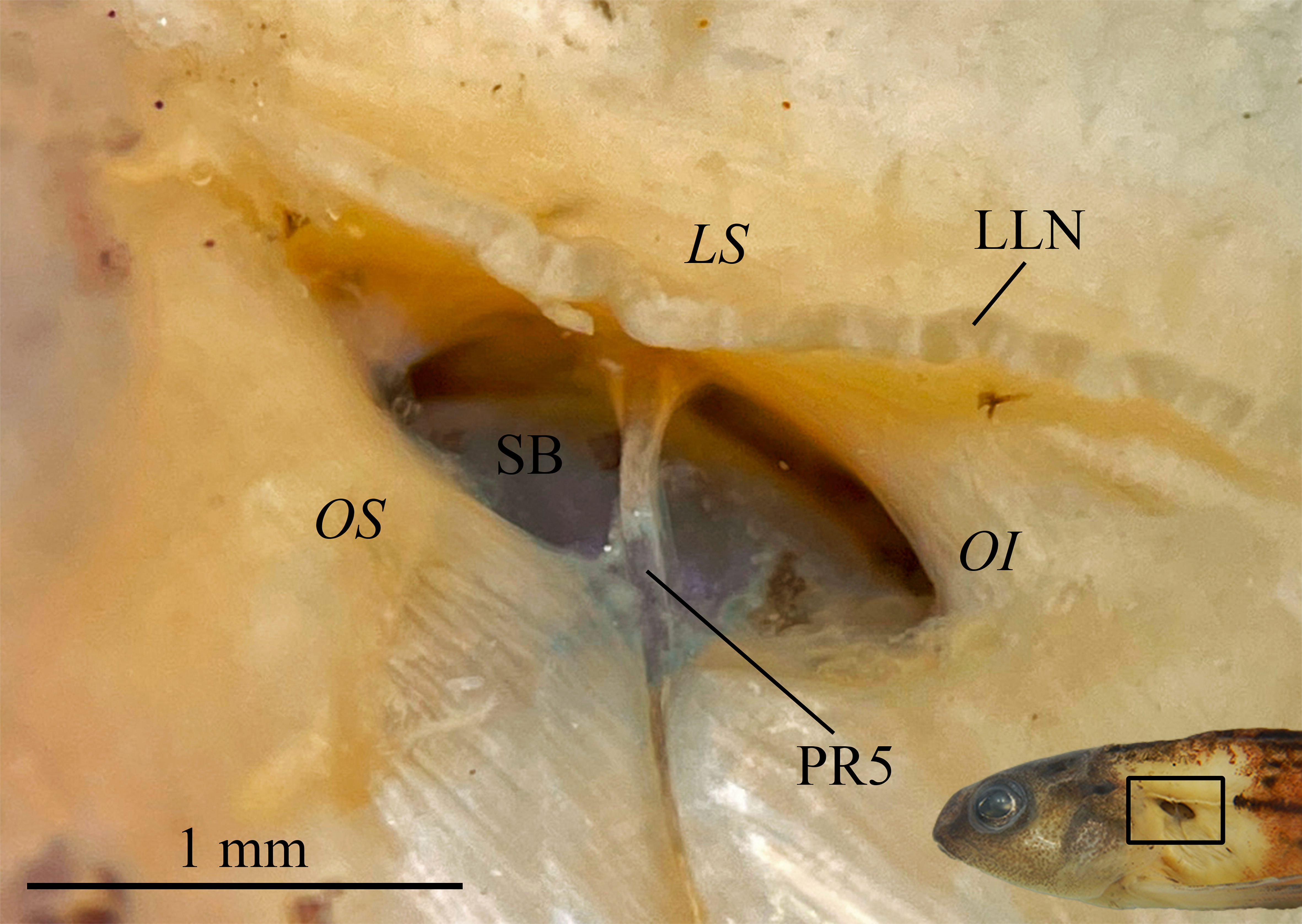
FIGURE 6| Pseudotympanum of Characidium urucum, paratype, male, CI-UFLA 3782, 38.9 mm SL. Right side, reversed; anterior to left. Anatomical structures are represented as LLN (lateral line nerve); LS (lateralis superficialis); OI (obliquus inferioris); OS (obliquus superioris); PR5 (pleural rib of fifth vertebra); and SB (swimbladder).
FIGURE 7| Ontogenetic variation of coloration pattern in paratypes of Characidium urucum; females to left (CI-UFLA 3783) and males to right (CI-UFLA 3784). A. 34.4 mm SL. B. 36.6 mm SL. C. 40.9 mm SL. D. 32.2 mm SL. E. 37.1 mm SL. F. 42.3 mm SL.
FIGURE 8| Live coloration of Characidium urucum, NUP 25703, 44.5 mm SL, paratype, female.
TABLE 1 | Morphometric data of holotype and paratypes of Characidium urucum (n = 26), range includes the holotype. SD = standard deviation.
| Holotype | Range | Mean | SD |
Total length (mm) | 53.6 | 36.8–55.2 | – | – |
Standard length (mm) | 44.0 | 30.3–45.9 | – | – |
Percentage of standard length | ||||
Head length | 23.9 | 22.8–25.8 | 24.6 | 0.8 |
Prepectoral distance | 21.8 | 20.4–23.2 | 21.7 | 0.7 |
Predorsal distance | 46.4 | 45.3–50.6 | 47.1 | 1.1 |
Pectoral-fin height | 19.6 | 16.4–22.3 | 19.4 | 1.4 |
Dorsal-fin height | 16.9 | 15–18.8 | 16.9 | 1.1 |
Dorsal-fin base | 14.6 | 13.7–16.7 | 14.6 | 0.7 |
Prepelvic distance | 50.7 | 47.8–52.2 | 50.3 | 1 |
Pelvic-fin height | 13.5 | 11.8–14.9 | 13.4 | 0.7 |
Preanal distance | 71.0 | 69.7–74.6 | 72.1 | 1.3 |
Anal-apex distance | 92.1 | 90.1–96.7 | 92.0 | 1.3 |
Anus to anal-fin distance | 5.5 | 3.2–6.7 | 5.1 | 1 |
Anal-fin height | 15.7 | 12.7–16.4 | 14.9 | 0.9 |
Anal-fin base | 8.8 | 6.3–9.1 | 7.6 | 0.7 |
Adipose-fin height | 3.6 | 3.2–5.9 | 4.4 | 0.6 |
Peduncle length | 22.5 | 19.5–23.9 | 21.4 | 1.2 |
Body depth at dorsal-fin origin | 21.3 | 18.1–24.3 | 21.3 | 1.3 |
Body depth at anal-fin origin | 17.6 | 14.7–18.2 | 16.4 | 0.9 |
Body depth at caudal peduncle | 11.4 | 10–12.1 | 11.2 | 0.6 |
Body width | 9.6 | 7.4–10.5 | 9.5 | 0.7 |
Percentage of head length | ||||
Snout length | 22.1 | 18.7–24.2 | 22.1 | 1.2 |
Snout-maxillary tip | 25.7 | 22.6–27.8 | 24.1 | 1.3 |
Anterior naris-orbit | 10.3 | 6.7–10.6 | 8.7 | 0.9 |
Posterior naris-orbit | 8.6 | 6.6–10.3 | 8.6 | 1.1 |
Cheeck depth | 7.7 | 6.3–10.0 | 8.2 | 1 |
Orbital diameter | 26.2 | 23.2–28.8 | 26.2 | 1.3 |
Interorbital distance | 14.8 | 9.3–14.9 | 11.6 | 1.3 |
Scales cycloid. Isthmus naked between anterior limit of isthmus and anterior margin of cleithrum; chest scaled, except for lateral areas surrounding bases of pectoral fins. Lateral line completely pored with 33(1), 34(15), 35*(3) scales from supracleithrum to hypural plate; one specimen with lateral line incomplete on left side (29 scales) and complete on right side (36 scales). Predorsal series aligned or unaligned, with 10(1), 11(14), 12*(9), or 13(1) scales between posterior tip of supraoccipital process and first dorsal-fin ray. Four*(25) to 5(1) scale series between lateral series and predorsal series. Four*(25) to 5(1) scale series between lateral series and midventral line. Three (3), 4*(22), or 5(1) scales between anus and first anal-fin ray. Scales series around caudal peduncle 12(8), 13(1) or 14*(15).
Premaxilla right-triangle shaped; ascending process very slender, about as deep as main body of premaxillary bone, articulating with anterior tip of mesethmoid through small notch. One series of 4(2), 5*(10), 6(12), or 7(2) premaxillary teeth, progressively decreasing in size posteriorly. All teeth tricuspid; lateral cusps poorly developed, visible as small lateral bulks at base of each crown (Fig. 5A). Maxilla edentulous, drop shaped and elongated in lateral view; ascending process slender and curved anteriorly, lying on dorsal surface of premaxilla and shaped like the dorsal profile of the latter. Two foramina present at base of ascending process of maxilla (Fig. 3A). Dentary teeth disposed in two rows; outer row with 4*(3), 5(8), 6(5), 7(7), or 9(3) tricuspid teeth, progressively decreasing in size from symphysis; inner row only visible in c&s specimens, composed of five (2) to eleven (2) minute conical teeth (Fig. 5B).
Dorsal-fin ii,8(2), ii,9(23), iii,8*(1); distal margin of dorsal fin convex. Adipose-fin well developed. Pectoral fin iii,7(1), iii,8(15), iii,8,i*(1), iii,9(6), iv,7(1), or iv,8(1), with its tip surpassing dorsal-fin origin in vertical when adpressed; second branched pectoral-fin rays usually longest. Pelvic-fin i,6,i*(21), i,7(2), or i,7,i(1); second to third branched pelvic-fin rays longest. Anal-fin i,4,i*(1), ii,4(1), ii,5(1), ii,5,i(4), ii,6(17); posterior margin of anal-fin convex, with first and second branched rays usually longest. Caudal fin i,9,7,i(1) or i,9,8,i*(25).
Total vertebrae 35(4); 15(1) to 16(3) caudal vertebrae, 19(3) to 20(1) precaudal vertebrae. Pleural ribs 14(3) or 15(1). Six (4) supraneurals; 26(2) to 28(2) epineurals; 15(1), 16(2) or 17(1) epipleurals. Dorsal-fin origin above 14th(4) vertebral centrum; first pterygiophore inserted between neural spines of 11th-12th(4) vertebral centra. Adipose-fin base above 28th(1) or 29th(3) vertebral centra. Pelvic-fin origin between 15th–16th(1) or 16th–17th(3) vertebral centra in vertical. Anal-fin origin below 25th(2) or 26th(2) vertebral centra. First anal-fin pterygiophore inserted between haemal spines of 21–22nd(1) or 22–23rd(3) vertebral centra. Eight (3) or nine (1) dorsal procurrent caudal-fin rays, and 6(3) or 7(1) ventral procurrent caudal-fin rays.
Pseudotympanum present, fusiform and obliquely oriented in lateral view. Humeral hiatus divided into anterior and posterior chambers by pleural rib of fifth vertebra; anterior chamber elongated, about same length as posterior chamber. Humeral hiatus dorsally delimited by lateralis superficialis, posteriorly delimited by obliquus inferioris, and anteroventrally by obliquus superioris. Anterior chamber of swimbladder visible underneath opening of humeral hiatus (Fig. 6).
Coloration in alcohol. Ontogenetic variation observed in the species (Fig. 7). Juveniles (< 35.0 mm SL) possess a yellowish body background with several dark brown spots, approximately one to three scales in diameter, randomly scattered across the flank. As body size increases, these spots progressively fade, becoming inconspicuous or absent in adult specimens, while yellow background coloration is replaced by red hue. Melanophores distributed over the entire dorsal surface of the head, concentrated at the occipital region and absent in the fontanel region. Melanophores present around orbit and iris. Longitudinal dark brown stripe on lateral surface of body well delimited and conspicuous in specimens of all sizes; stripe slender between snout and anterior margin of orbit (preorbital stripe), and diffuse between posterior margin of orbit and branchial slit; stripe slightly enlarged over pseudotympanum, and slender from above pectoral fin to end of caudal peduncle, not fused to basicaudal spot. Humeral spot conspicuous, dark brown, and rounded. Two to four dorsalmost scales series with dark brown reticulate pattern on margins of each scale. Fins with diffuse, dusky brown pigmentation on rays, interradial membranes hyaline. Adipose fin hyaline. Basicaudal spot present.
Coloration in life. Similar to coloration in alcohol, except for an inconspicuous longitudinal dark stripe on the lateral surface of the body, and intense red hue on the body and head. Fins translucent (Fig. 8).
Sexual dimorphism. Males exhibit longer paired fins, with the tip of pectoral fin surpassing the origin of pelvic fin, and the tip of pelvic fin surpassing the origin of anal fin (fins of females not reaching the origin of subsequent fin). Furthermore, males exhibit a more robust and rounded snout compared to females, which have a slenderer snout (Fig. 9).
FIGURE 9| Sexual dimorphism in snout shape of Characidium urucum. A. Female, lateral view, paratype, CI-UFLA 3783, 40.9 mm SL. B. Male, lateral view, paratype, CI-UFLA 3784, 42.3 mm SL. C. Same female, dorsal view. D. Same male, dorsal view.
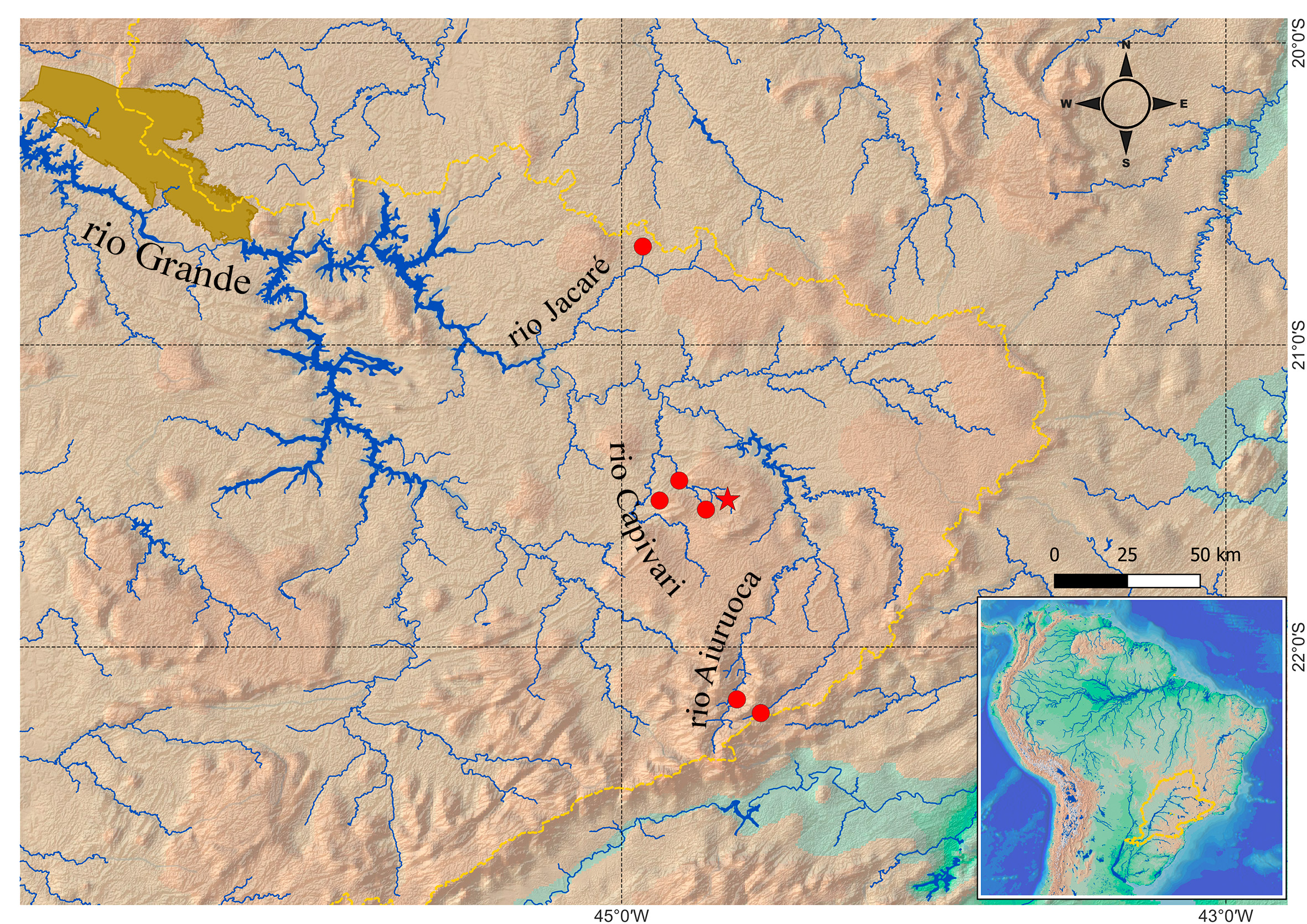
Geographical distribution. Characidium urucum is known from the upper rio Grande sub-basin, occurring at the Aiuruoca, Capivari, and Jacaré drainages, as well as the headwaters of the rio Grande (Fig. 10).
FIGURE 10| Distribution map of Characidium urucum. The red star represents the type-locality, and red circles represent the other known localities. The yellow dashed line indicates the limits of the upper rio Paraná basin. The brown blot represents Serra da Canastra.
Ecological notes. Around the type-locality, the rio Capivari is classified as a third-order stream (sensu Strahler, 1957), varying in width from four to eight meters, and in depth from 20 to 150 cm. The substrate primarily consists of sand, with the presence of pebbles (1–4 cm) and cobbles (4–15 cm) in riffle sections. Characidium urucum was exclusively captured in the deepest pools, associated with wood debris near the riverbanks (Fig. 11). The species was not found in riffles or river stretches with an open canopy, suggesting a strong habitat specificity. The gut contents of six specimens were predominantly composed of Chironomidae larvae, although remnants of digested arthropod exoskeletons were also observed, indicating that the species is primarily invertivorous. Characidium urucum was collected in sympatry with C. gomesi, Cetopsorhamdia iheringi Schubart & Gomes, 1959, Geophagus iporangensis Haseman, 1911, Hasemania cf. nana (Lütken, 1875), Hypostomus nigromaculatus (Schubart, 1964), Pareiorhina carrancas Bockmann & Ribeiro, 2003, Psalidodon cf. paranae (Eigenmann, 1914), Trichomycterus pirabitira Barbosa & Azevedo-Santos, 2012, T. piratymbara Katz, Barbosa & Costa, 2013 and T. ingaiensis Katz & Costa, 2021.
FIGURE 11| Type-locality of Characidium urucum in the rio Capivari, Carrancas, rio Grande sub-basin, upper rio Paraná basin, Brazil.
Conservation status. Characidium urucum has an extent of occurrence (EOO) estimated as 26,432 km², corresponding to the area of the upper rio Grande sub-basin (HydroBasins level 6). Although the species has not yet been recorded within protected areas, most of its localities remain pristine or lightly impacted, and there are no identifiable threats to the viability of its population at present or in the foreseeable future. Therefore, we recommend that Characidium urucum should be categorized as Least Concern (LC) according to the IUCN criteria.
Etymology. The specific epithet “urucum” is the common name for Bixa orellana L., derived from the Tupi language of South America (Navarro, 2013). “Urucum” is a plant native to tropical America, widely known for the intense red hue of its seeds. These seeds have traditionally been used by South American indigenous peoples as condiments, pigments, and body paint, and their use remains prevalent in Brazilian culture today. A noun in apposition, in allusion to the distinctive red coloration observed in adult individuals of the new species.
Common name. Characidium urucum is known as ‘mocinha’ or ‘canivete’, common names widely used in Portuguese for members of Characidiinae. Regarding the remarkably red coloration of the new species, we suggest the common name ‘mocinha-vermelha’, meaning ‘red mocinha’ in Portuguese.
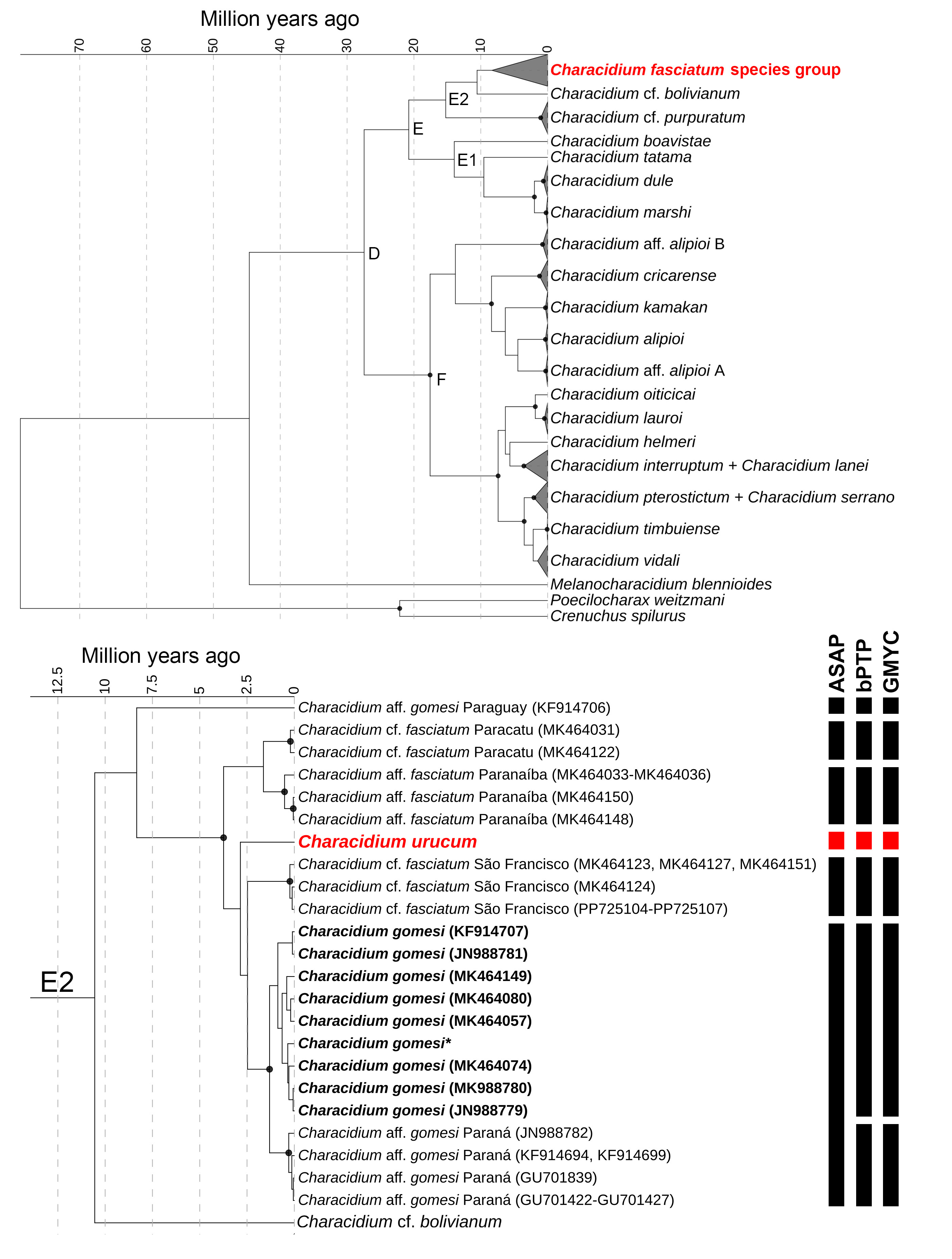
Phylogenetic reconstruction. The final alignment of 106 sequences and 666 bp exhibited a nucleotide composition of A (25.1%), C (25.5%), T (32.0%), and G (17.3%), of which 237 were variable and 208 parsimony informative. Our molecular analysis recovered Characidium urucum within the E2 clade sensu Oliveira-Silva et al. (2024), composed of C. cf. bolivianum, C. gomesi, C. fasciatum, and C. cf. purpuratum (Fig. 12A). In this clade, C. urucum is part of the C. fasciatum species group, along with C. fasciatum, C. gomesi, and similar forms; the new species is sister to a subclade comprehending C. cf. fasciatum from the rio São Francisco River basin + C. gomesi and C. aff. gomesi from the upper Paraná River basin (Fig. 12B). Within the C. fasciatum species group, C. urucum presented the lowest K2P genetic distance to C. gomesi from the upper rio Paraná basin (2.64%), and the highest genetic distance to C. aff. gomesi from the rio Paraguay basin (8.31%) (Tab. 2).
FIGURE 12| Time-calibrated Bayesian Inference gene tree constructed using partial fragment of COI sequences with HKY+G+I nucleotide substitution model. Circles on nodes represent posterior probabilities >97. A. Molecular relationship among Characidium species from Clade D (sensu Oliveira-Silva et al., 2024). B. Pruned tree of Characidium fasciatum species group with new species C. urucum highlighted in red. Sequences of C. gomesi stricto sensu are highlighted in bold; topotypes with asterisk. Delimitation results from the three species delimitation methods are shown as solid black blocks.
TABLE 2| Mean genetic distances (and standard deviation) between lineages of the Characidium fasciatum species group, based on the COI marker and the K2P nucleotide substitution model.
| C. urucum | C. gomesi |
C.
aff. gomesi |
C.
cf. fasciatum
|
C.
aff. fasciatum
|
C.
cf. fasciatum
|
C. gomesi | 2.74 + 0.10 |
|
|
|
|
|
C. aff.gomesi Paraná | 3.17 + 0.10 | 0.74 + 0.37 |
|
|
|
|
C. cf. fasciatum Paracatu | 3.07 | 2.66 + 0.25 | 3.07 + 0.16 |
|
|
|
C. aff. fasciatum Paranaíba | 3.83 + 0.10 | 3.46 + 0.39 | 3.78 + 0.29 | 1.84 + 0.26 |
|
|
C. cf. fasciatum São Francisco | 3.71 | 3.41 + 0.37 | 3.38 + 0.25 | 4.31 + 0.18 | 4.62 + 0.13 |
|
C. aff. gomesi Paraguay | 8.31 | 8.72 + 0.14 | 9.33 + 0.50 | 7.64 | 8.51 + 0.14 | 9.70 + 0.13 |
We recovered the cis-Andean clade E2 diverging from the trans-Andean clade E1 about 20 million years ago (mya). Within clade E2, C. cf. purpuratum diverged from C. cf. bolivianum + C. fasciatum species group about 15 mya, and C. cf. bolivianum from the C. fasciatum species group about 10 mya. We recovered a rapid radiation within the C. fasciatum species group, whereas the most recent common ancestor of all lineages from the upper Paraná and São Francisco basins dates back from 3.7 mya. We found a median divergence time estimate of 2.85 (5.7–1.06) million years ago between C. urucum and its sister clade.
Species delimitation. Our species delimitation methods recovered high diversity within the clade composed of C. fasciatum, C. gomesi,and C. urucum, as ASAP delimited six lineages, them being: C. aff. gomesi from the rio Paraguay basin; C. cf. fasciatum ‘Paracatu’ and C. cf. fasciatum ‘São Francisco’ from the middle rio São Francisco basin; C. aff. fasciatum ‘Paranaíba’, C. gomesi, and C. urucum from the upper Rio Paraná basin (Fig. 12B). The bPTP and GMYC methods recovered seven equally-delimited lineages with major congruence with ASAP, with the exception of C. gomesi, which is split into two lineages according to these methods (C. gomesi, and the herein called ‘C. aff. gomesi Paraná’) (Fig. 12B). Characidium urucum is equally supported as a distinct lineage by all three delimitation methods (Fig. 12B).
Discussion
Characidium urucum is the 10th species of the genus described from the upper rio Paraná basin. The last four species (C. heirmostigmata da Graça & Pavanelli, 2008, C. xanthopterum Silveira, Langeani, da Graça, Pavanelli & Buckup, 2008, C. onca Melo, Brito, Ribeiro & Lima, 2021, and C. itarare Stabile, Reis, Frota, da Graça & Oliveira, 2024) were described within the last 16 years, following a period of over four decades without any new descriptions of Characidium species from this basin. These findings support the hypothesis proposed by Graça, Pavanelli (2008) regarding the undiscovered diversity in the upper rio Paraná basin. Furthermore, they position Characidium alongside Melanorivulus, Trichomycterus, Hypostomus,and Neoplecostomus as the only genera with at least ten species in this basin, which exhibits a low frequency of sympatry among related lineages (Dagosta et al., 2024).
Among the 89 valid species of Characidium, only 22 exhibit traits of secondary sexual dimorphism, such as bony processes on fin rays, variations in the size and shape of the anal and pelvic fins, and sexual dichromatism in body and fin coloration (Teixeira, Melo, 2021; Zanata et al., 2023; Oliveira-Silva et al., 2025). In certain species, such as C. satoi Melo & Oyakawa, 2015, only the males display dark coloration during the breeding season, in addition to possessing hooks on the pelvic-fin rays (Melo, Oyakawa, 2015). The combination of these characteristics, along with the size and shape of the anal and pelvic fins, allows the identification of three distinct types of secondary sexual characters within the Characidium species (see review in Teixeira, Melo, 2021). Characidium urucum not only exhibits differences in fin size between sexes, but also introduces a novel secondary sexual character for the genus: the shape of the head, where females possess a slender snout, while males have a more robust and curved snout (Fig. 8).
In addition to the character states listed in the Diagnosis section, adult specimens of C. urucum can be readily distinguished from congeners by the presence of an intense red hue in body coloration, particularly in live individuals (vs. yellowish, pale brown, dark green, or purple in other species). However, because this remarkable coloration is restricted to adults and may fade in preserved material, we opted not to propose it as a formal diagnostic character, but rather to report its occurrence as supplementary diagnostic information.
Characidium urucum also exhibits a remarkable ontogenetic variation in its coloration pattern. In juvenile specimens, the typical red coloration of this species is absent; instead, the flank is adorned by numerous dark spots randomly scattered over a yellowish background, resembling the pattern observed in the C. lauroi species group (Melo, 2001; Melo, Buckup & Oyakawa, 2016). However, C. urucum is not closely related to these species (Fig. 11A), indicating that the presence of dark spots on the flank is a homoplastic character state. Characidium urucum can also be distinguished from species of the C. lauroi group by the presence of the parietal branch of the supraorbital laterosensory canal, which is absent in those species (Melo, 2001; Melo et al., 2016).
Given that C. urucum is part of the C2 clade (sensu Buckup, 1993b), its coloration pattern appears to be a derived characteristic of this clade (Buckup, 1993b). This is further supported by the observation that other members of the C2 clade exhibit distinct coloration patterns. For instance, C. fasciatum and C. gomesi can display a brown coloration, while C. purpuratum can be recognized for its purple coloration. This suggests that the coloration pattern observed in C. urucum is a unique trait that evolved within this clade, distinguishing it from its congeners.
The phylogenetic placement of C. urucum as a sister species toa clade comprehending C. cf. fasciatum from the São Francisco River, and C. gomesi + C. aff. gomesi from the upper rio Paraná basin supports hypotheses of recent drainage rearrangement between the upper rio Grande and the São Francisco river basins (Rezende et al., 2018; Cassemiro et al., 2024). According to Cassemiro et al. (2024), the rio Grande was part of the rio São Francisco basin until about six million years ago, when hydrographic changes around the Serra da Canastra started detouring the rio Grande to the rio Paraná basin. This rearrangement provided faunal exchanges that ended about two million years ago, when the upper rio Grande became part of the rio Paraná basin and completely isolated from the rio São Francisco basin. These events could have caused the cladogenesis that gave origin to C. urucum in the upper rio Grande, about 2.85 million years ago, and posteriorly to C. cf. fasciatum in the São Francisco River basin, and C. gomesi and C. aff. gomesi in the upper Paraná River basin, between 2.47 and 1.31 million years ago, in our phylogenetic reconstruction.
The description of this new species highlights the diversity in a region characterized by limited sampling efforts, suggesting the potential for discovering additional new species (Silva-Sene et al., 2024). This basin consists of multiple sub-basins identified as areas of endemism due to the presence of sympatric species with restricted distributions and well-defined geographic boundaries (Costa et al., 2023; Dagosta et al., 2024). Notable endemic taxa within this basin include several species of Trichomycterus from the T. reinhardti species group, Heptapterus carmelitanorum Azevedo-Santos, Deprá, Aguilera, Faustino-Fuster & Katz, 2022, Neoplecostomus altimontanus Uzeda, Paiola, Siqueira-Cesar, Okubo, Marques-Frisoni, Andrade & Langeani, 2024, N. canastra Roxo, Silva, Zawadzki & Oliveira, 2017, N. sapucai Andrade, Uzeda, Paiola, Siqueira-Cesar, Okubo, Marques-Frisoni & Langeani, 2024, Pareiorhaphis togoroi Oliveira & Oyakawa, 2019, Pareiorhina carrancas, and P. pelicicei Azevedo-Santos & Roxo, 2015 (Bockmann, Ribeiro, 2003; Azevedo-Santos, Roxo, 2015; Roxo et al., 2017; Oliveira, Oyakawa, 2019; Costa, Katz, 2021; Deprá et al., 2022; Costa et al., 2023; Uzeda et al., 2024). Characidium urucum is now also recognized as part of this assemblage, and given its restricted distribution and potential threats to the integrity of local aquatic ecosystems (see Costa et al., 2023), concerns arise regarding the future conservation of these species.
Comments on Characidium fasciatum and C. gomesi. Characidium fasciatum and C. gomesi do not form species-exclusive groups in our phylogenetic analysis, which may be mainly attributed to the uncertainties in the taxonomic history of these species (Travassos, 1956; Buckup, 1992) and the generalization of characters used for their identification, which are briefly discussed below.
Characidium gomesi was described from “Córrego da Lazica”, a tributary to the headwaters of the Mogi-Guaçu River (rio Grande, upper rio Paraná basin), in the municipality of Ouro Fino, Minas Gerais State, southeast Brazil (Travassos, 1956). However, the name “Córrego da Lazica” is not cataloged in the Brazilian National Water Agency (ANA), and neither is it relatable by locals to any river, waterfall, county, farm, or person around the municipality of Ouro Fino (PLCU, pers. obs.), making the type-locality of C. gomesi currently inaccessible. However, we have managed to collect specimens of C. gomesi in another stream affluent to the Mogi-Guaçu River in the surroundings of the municipality of Ouro Fino: the Ribeirão do Capinzal, downstream from Cachoeira do Tabuão. We sequenced the COImarker of three specimens (Tab. S1) to serve as reference haplotypes for our molecular species delimitation methods, as well as future taxonomic studies involving the species.
In the original description of C. gomesi, Travassos diagnoses the species from “C. fasciatum” by having a naked chest, however, the author misidentified C. fasciatum as a scaled-isthmus species of the C. zebra species group (MNRJ 6125) collected at Cachoeira de Emas, upper rio Paraná basin (Travassos, 1956:7, figs. 6–7). As a consequence, the taxonomic limits between C. fasciatum and C. gomesi remain nebulous, especially considering that our molecular species delimitation methods recovered a consensual total of seven lineages previously hidden under these two names. Furthermore, these lineages are distributed in the upper Paraná, Paraguay and São Francisco river basins, neighbouring hydrographic basins with a recent history of rearrangements and faunal exchanges (Cassemiro et al., 2024).
As provided in the redescription of C. fasciatum by Buckup (1992), this species can be diagnosed from C. gomesi by the absence of dark bands on pelvic, anal, and caudal fins. Recently collected specimens of C. fasciatum from the rio das Velhas drainage and C. gomesi from the rio Mogi-Guaçu drainage confirm the observation of Buckup (1992), since adult specimens of C. fasciatum present a continuous dusky coloration over fin rays, especially on dorsal and caudal fins, in such a way that the fin rays are homogeneously pigmented from base to subterminal region (Fig. 13A). Contrastingly, specimens of C. gomesi present conspicuous dark bands on all fins, with the ones on dorsal and caudal fins being highly conspicuous since juveniles, especially the 3-shaped dark band on caudal fin (Fig. 13B).
FIGURE 13| Recently collected specimens of A. Characidium fasciatum from rio das Velhas drainage, CI-UFLA 3290, 64.1 mm SL, and B. Characidium gomesi from the headwaters of Mogi-Guaçu drainage, Ouro Fino, Minas Gerais, CI-UFLA 3828, 58.2 mm SL.
It is worth mentioning that all lineages delimited herein are exclusive to a single hydrographic basin, which stands against the hypothesis of Buckup (1992), who expands the distribution of C. fasciatum to the upper rio Paraná basin based on lots from the Paranaíba, Grande and Tietê drainages. None of the sequences from the upper rio Paraná basin used in our study grouped with those from the rio São Francisco basin; in fact, most of these sequences were either associated with C. gomesi or ‘C. aff. gomesi Paraná’. Additionally, one of the sequences used herein (KF914694) was collected in the same river as one of the lots analyzed by Buckup (1992) (MNRJ 11460), who identified specimens as C. fasciatum in the rio Alambari, rio Tietê drainage, upper rio Paraná basin, herein recovered as belonging to ‘C. aff. gomesi Paraná’. The same holds true for specimens identified as C. gomesi from the rio São Francisco basin, which are not closely related to C. gomesi “stricto sensu”. Therefore, it is possible that C. gomesi is restricted to the upper rio Paraná basin, and C. fasciatum to the rio São Francisco basin. However, definitive taxonomic arrangements within this clade would require a thorough, integrative analysis of populations used in these registers, especially considering the broad distribution of members of this species group.
Characidium urucum is herein recovered as part of a clade comprehending C. fasciatum, C. gomesi, and similar forms. However, the new species can be easily diagnosed from its closest congeners by several morphological character states listed in the Diagnosis section, and also represents an independent evolutionary unit, as shown by our molecular phylogenetic reconstruction and species delimitation methods.
Comparative material examined. Comparative material was obtained from the list of species provided by Stabile et al. (2024), with the addition of the following specimens. Brazil. Characidium iaquira: NUP 11699, 1, 49.8 mm SL, riacho Sumidouro, tributary to rio Preto, rio Tapajós basin, Diamantino, Mato Grosso. Characidium fasciatum: Minas Gerais, rio São Francisco basin, córrego Catana, tributary to rio das Velhas, Itabirito: CI-UFLA 3290, 4, 49.3–64.1 mm SL; rio das Velhas, Ouro Preto: CI-UFLA 2385, 3 c&s, 53.1–61.5 mm SL; NUP 25908, 1, 29.1 mm SL. Rio Pandeiros, Januária: NUP 25907, 2, 25.2–28.5 mm SL. Characidium gomesi: rio Grande sub-basin, upper rio Paraná basin. CI-UFLA 3828, 9, 41.9–58.2 mm SL; NUP 26174, 4, 41.7–43.9 mm SL, ribeirão do Capinzal, tributary to rio Mogi-Guaçu, upper rio Paraná basin, Ouro Fino, Minas Gerais. NUP 8584, 5, 25.0–48.0 mm SL, ribeirão da Boa Vista, tributary to rio Ingaí, upper rio Paraná basin, Ingaí, Minas Gerais. NUP 10365, 49, 31.7–70.8 mm SL, córrego Monjolo, tributary to rio Sapucaí, upper rio Paraná basin, São Bento do Sapucaí, São Paulo. NUP 11539, 6, 44.8–65.1 mm SL, córrego Monjolinho, tributary to rio Sapucaí Mirim, São Bento do Sapucaí, São Paulo. Characidium oiticicai: CI-UFLA 3652, 11, 32.1–48.7 mm SL, ribeirão Taiaçupeba Açu, tributary to rio Tietê, upper rio Paraná basin, Santo André, São Paulo.
Acknowledgments
We are grateful to Carina Porto, Gabriela Spuri, Thayanna Rodrigues and Vinícius Urbano for the assistance during fieldwork and collecting part of the type-series of the new species; to Elisabeth Henschel (Universidade Federal de Viçosa – MZUVF), Júllia Tôrres (Pontifícia Universidade Católica de Minas Gerais – MCNIP), Marli C. Campos (Nupélia/UEM), and Priscilla Silva (Universidade Federal de Minas Gerais – UFMG-ICT) for cataloging the vouchers and taking type specimens under their respective institutions; and to Rodrigo L. Ferreira from Centro de Estudos em Biologia Subterrânea (CEBS-UFLA) for sharing their Zeiss Axio-Zoom V16 stereomicroscope.
References
Agudelo-Zamora HD, Tavera J, Murillo YD, Armando Ortega-Lara. The unknown diversity of the genus Characidium (Characiformes: Crenuchidae) in the Chocó biogeographic region, Colombian Andes: two new species supported by morphological and molecular data. J Fish Biol. 2020; 97(6):1662–75. https://doi.org/10.1111/jfb.14527
Azevedo-Santos VM, Roxo FF. A new species of the genus Pareiorhina (Teleostei: Siluriformes: Loricariidae) from the upper rio Paraná basin, southeastern Brazil. Zootaxa. 2015; 3937(2):377–85. https://doi.org/10.11646/zootaxa.3937.2.8
Bockmann FA, Ribeiro AC. Description of a new suckermouth armored catfish of the genus Pareiorhina (Siluriformes: Loricariidae), from southeastern Brazil. Ichthyol Explor Freshw. 2003; 14(3):231–42.
Buckup PA. Redescription of Characidium fasciatum, type species of the Characidiinae (Teleostei, Characiformes). Copeia. 1992; 1992(4):1066–73. https://doi.org/10.2307/1446639
Buckup PA. Review of the characidiin fishes (Teleostei: Characiformes), with descriptions of four new genera and ten new species. Ichthyol Explor Freshw. 1993a; 4(2):97–154.
Buckup PA. Phylogenetic interrelationships and reductive evolution in neotropical Characidiin fishes (Characiformes, Ostariophysi). Cladistics. 1993b; 9(3):305–41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
Buckup PA. Family Crenuchidae (South American darters). In: Reis RE, Kullander SO, Ferraris CJ, Jr., editors. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2004. p.87–95.
Buckup PA, Reis RE. Characidiin genus Characidium (Teleostei, Characiformes) in Southern Brazil, with description of three new species. Copeia. 1997; 1997(3):531–48. https://doi.org/10.2307/1447557
Cassemiro F, Salgado AAR, Pinto RWP, Rezende EA. The timing of large drainage rearrangement in South America: a study based on morphological and ecological evidence. Geomorphology. 2024; 468:109457. https://doi.org/10.1016/j.geomorph.2024.109457
Costa WJEM, Katz AM. Integrative taxonomy supports high species diversity of south-eastern Brazilian mountain catfishes of the T. reinhardti group (Siluriformes: Trichomycteridae). Syst Biodivers. 2021; 19(6):601–21. https://doi.org/10.1080/14772000.2021.1900947
Costa WJEM, Azevedo-Santos VM, Mattos JLO, Katz AM. Molecular phylogeny, taxonomy and distribution patterns of Trichomycterine catfishes in the middle Rio Grande Drainage, South-Eastern Brazil (Siluriformes: Trichomycteridae). Fishes. 2023; 8(4):206. https://doi.org/10.3390/fishes8040206
Dagosta FCP, Monção MS, Nagamatsu BA, Pavanelli CS, Carvalho FR, Lima FCT et al. Fishes of the upper rio Paraná basin: diversity, biogeography and conservation. Neotrop Ichthyol. 2024; 22(1):e230066. https://doi.org/10.1590/1982-0224-2023-0066
Darriba D, Taboada GL, Doallo R, Posada D. jModelTest 2: more models, new heuristics and parallel computing. Nat Methods. 2012; 9(8):772. https://doi.org/10.1038/nmeth.2109
Deprá GC, Aguilera G, Faustino-Fuster DR, Katz AM, Azevedo-Santos VM. Redefinition of Heptapterus (Heptapteridae) and description of Heptapterus carmelitanorum, a new species from the upper Paraná River basin in Brazil. Zoosyst Evol. 2022; 98(2):327–43. https://doi.org/10.3897/zse.98.89413
Fricke R, Eschmeyer WN. Eschmeyer’s catalog of fishes: guide to fish collections [Internet]. San Francisco: California Academy of Science; 2026. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/collections.asp
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2026. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Fujisawa T, Barraclough TG. Delimiting species using single-locus data and the generalized mixed Yule Coalescent Approach: a revised method and evaluation on simulated data sets. Syst Biol. 2013; 62(5):707–24. https://doi.org/10.1093/sysbio/syt033
Graça WJ, Pavanelli CS. Characidium heirmostigmata, a new characidiin fish (Characiformes: Crenuchidae) from the upper rio Paraná basin, Brazil. Neotrop Ichthyol. 2008; 6(1):53–56. https://doi.org/10.1590/S1679-62252008000100006
Hall TA. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser. 1999; 41:95–98.
International Union for Conservation of Nature (IUCN). Standards and Petitions Committee. Guidelines for using the IUCN Red List Categories and Criteria. Version 16. 2024. Available from: https://www.iucnredlist.org/documents/RedListGuidelines.pdf
Ivanova NV, Zemlak TS, Hanner RH, Hebert PDN. Universal primer cocktails for fish DNA barcoding. Mol Ecol Notes. 2007; 7(4):544–48. https://doi.org/10.1111/j.1471-8286.2007.01748.x
Johnson M, Zaretskaya I, Raytselis Y, Merezhuk Y, McGinnis S, Madden TL. NCBI BLAST: a better web interface. Nucleic Acids Res. 2008; 36:5–09. https://doi.org/10.1093/nar/gkn201
Leal CG, Junqueira NT, Pompeu PS. Morphology and habitat use by fishes of the rio das Velhas basin in southeastern Brazil. Environ Biol Fish. 2011; 90:143–57. https://doi.org/10.1007/s10641-010-9726-6
Lehner B, Grill G. Global river hydrography and network routing: baseline data and new approaches to study the world’s large river systems. Hydrol Process. 2013; 27(15):2171–86. https://doi.org/10.1002/hyp.9740
Letunic I, Bork P. Interactive Tree of Life (iTOL) v6: recent updates to the phylogenetic tree display and annotation tool. Nucleic Acids Res. 2024; 52(1):78–82. https://doi.org/10.1093/nar/gkae268
Melo MRS. Sistemática, filogenia e biogeografia do grupo Characidium lauroi Travassos, 1949 (Characiformes, Crenuchidae) [PhD Dissertation]. Rio de Janeiro: UFRJ, Museu Nacional; 2001.
Melo MRS, Bouquerel BB, Masumoto FT, França RS, Netto-Ferreira AL. A new species of Characidium (Characiformes: Crenuchidae) from the Chapada dos Veadeiros, Goiás, Brazil. Neotrop Ichthyol. 2021; 19(2):e200152. https://doi.org/10.1590/1982-0224-2020-0152
Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná State, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. https://doi.org/10.1590/1982-0224-20160014
Melo MRS, Oyakawa OT. A new species of Characidium Reinhardt (Characiformes, Crenuchidae) with a distinctively dimorphic male. Copeia. 2015; 103(2):281–89. https://doi.org/10.1643/CI-14-073
Melo BF, Sidlauskas BL, Near TJ, Roxo FF, Ghezelayagh A, Ochoa LE et al. Accelerated diversification explains the exceptional species richness of tropical characoid fishes. Syst Biol. 2022; 71(1):78–92. https://doi.org/10.1093/sysbio/syab040
Navarro EA. Dicionário de tupi antigo: a língua indígena clássica do Brasil. São Paulo: Global Editora; 2013.
Oliveira JCD, Oyakawa OT. New loricariid fishes from headwaters on Serra da Mantiqueira and Complexo do Espinhaço, Minas Gerais State, Brazil (Teleostei: Siluriformes: Loricariidae). Zootaxa. 2019; 4586(3):401–24. https://doi.org/10.11646/zootaxa.4586.3.1
Oliveira-Silva L, Batalha-Filho H, Camelier P, Zanata AM. Underestimated diversity in Characidium (Characiformes: Crenuchidae) from Neotropical rivers revealed by an integrative approach. Syst Biodivers. 2024; 22(1):2346510. https://doi.org/10.1080/14772000.2024.2346510
Oliveira-Silva L, Britzke R, Meza-Vargas V, Hidalgo MH, Faustino-Fuster D, Oliveira C et al. Morphological and molecular evidence reveals a new species of Characidium from the Ucayali-Urubamba Piedmont, Peru, and novel molecular clades are proposed within the genus. J Fish Biol. 2025; 1–12. https://doi.org/10.1111/jfb.70261
Pompeu PS, Reis LS, Gandini CV, Souza RCR, Favero JM. The ichthyofauna of upper rio Capivari: defining conservation strategies based on the composition and distribution of fish species. Neotrop Ichthyol. 2009; 7(4):659–66. https://doi.org/10.1590/S1679-62252009000400015
Pons J, Barraclough TG, Gomez-Zurita J, Cardoso A, Duran DP, Hazell S et al. Sequence-based species delimitation for the DNA taxonomy of undescribed insects. Syst Biol. 2006; 55(4):595–609. https://doi.org/10.1080/10635150600852011
Puillandre N, Brouillet S, Achaz G. ASAP: assemble species by automatic partitioning. Mol Ecol Resour. 2021; 21(2):609–20. https://doi.org/10.1111/1755-0998.13281
QGIS Development Team – QGIS Geographic Information System. Open Source Geospatial Foundation. Version 3.30.2. 2025. Available from: https://qgis.org
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA. Posterior summarization in Bayesian phylogenetics using tracer 1.7. Syst Biol. 2018; 67(5):901–04. https://doi.org/10.1093/sysbio/syy032
Rezende EA, Salgado AAR, Castro PTA. Evolução da rede de drenagem e evidências de antigas conexões entre as bacias dos rios Grande e São Francisco no sudeste brasileiro. Rev Bras Geomorfol. 2018; 19(3). https://doi.org/10.20502/rbg.v19i3.1304
Rosenthal A, Coutelle O, Craxton M. Large-scale production of DNA sequencing templates by microtitre format PCR. Nucleic Acids Research. 1993; 21(1):173–74.
Roxo FF, Silva GSC, Zawadzki CH, Oliveira C. Neoplecostomus canastra, a new catfish (Teleostei: Siluriformes) species from upper rio Paraná basin. Zootaxa. 2017; 4294(2):226–40. https://doi.org/10.11646/zootaxa.4294.2.6
Silva-Sene AM, Prado IG, Uzeda PLDC, Casarim R, Campos Cruz L, Gomes JP et al. Fish fauna of the upper Grande River basin: checklist and distribution patterns. Stud Neotrop Fauna E. 2024:1–10. https://doi.org/10.1080/01650521.2024.2424136
Souza RCR, Pompeu PS. Ecological separation by ecomorphology and swimming performance between two congeneric fish species. Zoologia. 2020; 37:1–08. https://doi.org/10.3897/zoologia.37.e47223
Stabile BHM, Reis RB, Frota A, Graça WJ, Oliveira AV. Morphology and molecular evidence of a new species of Characidium (Characiformes: Crenuchidae) from the edges of a protected area at rio Itararé, upper rio Paraná, southern Brazil. J Fish Biol. 2024; 105(6):1850–61. https://doi.org/10.1111/jfb.15935
Suchard MA, Lemey P, Baele G, Ayres DL, Drummond AJ, Rambaut A. Bayesian phylogenetic and phylodynamic data integration using BEAST 1.10. Virus Evol. 2018; 4(1):vey016. https://doi.org/10.1093/ve/vey016
Strahler AN. Quantitative analysis of watershed geomorphology. Eos. 1957; 38(6):913–20. https://doi.org/10.1029/TR038i006p00913
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19. https://doi.org/10.26028/cybium/1985-92-001
Teixeira TF, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) from the Río Huallaga, Central Peruvian Amazon, with a review of secondary sexual characters in the genus. J Fish Biol. 2021; 98(1):178–88. https://doi.org/10.1111/jfb.14568
Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucl Acids Res. 1994; 22(22):4673–80. https://doi.org/10.1093/nar/22.22.4673
Travassos H. Ictiofauna de Pirassununga. II. Sôbre Characidiinae H. Travassos, 1952 (Cypriniformes – Characoidei). Bol Mus Nac Rio J Zool. 1956; 135:1–14.
Uzeda PLC, Paiola I, Cesar PS, Okubo VKN, Marques-Frisoni WJ, Andrade BN et al. Two new species of Neoplecostomus (Siluriformes: Loricariidae) from high altitudes of the upper rio Paraná basin, Brazil. Neotrop Ichthyol. 2024; 22(4):e240021. https://doi.org/10.1590/1982-0224-2024-0021
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PDN. DNA barcoding Australia’s fish species. Philos T Roy Soc B Biol Sci. 2005; 360(1462):1847–57. https://doi.org/10.1098/rstb.2005.1716
Weitzman SH. The osteology of Brycon meeki, a generalized characid fish, with an osteological definition of the family. Vol. 8. Stanford, California, U.S.A.: Stanford University, Division of Systematic Biology; 1962.
Zanata AM, Ohara WM, Oyakawa OT, Dagosta FCP. A new rheophilic South American darter (Crenuchidae: Characidium) from the rio Juruena basin, Brazil, with comments on morphological adaptations to life in fast-flowing waters. J Fish Biol. 2020; 97(5):1343–53. https://doi.org/10.1111/jfb.14485
Zanata AM, Oliveira-Silva L. A new South American darter (Crenuchidae: Characidium) from rivers draining the Northeastern Mata Atlantica Freshwater Ecoregion, Brazil: morphological andmolecular evidence. J Fish Biol. 2025; 1–13. https://doi.org/10.1111/jfb.70316
Zanata AM, Oliveira-Silva L, Ohara WM. A new sexually dichromatic miniature Characidium (Characiformes: Crenuchidae) from the rio Guaporé, rio Madeira basin, Brazil, with remarkable morphological novelties to the genus. Neotrop Ichthyol. 2023; 21(1):e220059. https://doi.org/10.1590/1982-0224-2022-0059
Zhang J, Kapli P, Pavlidis P, Stamatakis A. A general species delimitation method with applications to phylogenetic placements. Bioinformatics. 2013; 29(22):2869–76. https://doi.org/10.1093/bioinformatics/btt499
Authors
![]() Pedro L. C. Uzeda1
Pedro L. C. Uzeda1 ![]() ,
, ![]() Bruno H. M. Stabile2,
Bruno H. M. Stabile2, ![]() Renan B. dos Reis2,
Renan B. dos Reis2, ![]() Luana J. Sartori3,
Luana J. Sartori3, ![]() Carla S. Pavanelli2,4,5,
Carla S. Pavanelli2,4,5, ![]() Vladimir P. Margarido6 and
Vladimir P. Margarido6 and ![]() Weferson J. da Graça2,4,5
Weferson J. da Graça2,4,5
[1] Laboratório de Ecologia de Peixes, Departamento de Ecologia e Conservação, Universidade Federal de Lavras, Trevo Rotatório Professor Edmir Sá Santos, s/n, 37203-202, Lavras, MG, Brazil. (PLCU) pedro.lc.uzeda@gmail.com (corresponding author).
[2] Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais, Departamento de Biologia, Centro de Ciências Biológicas, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900, Maringá, PR, Brazil. (BHMS) bhmstabile@gmail.com, (CSP) carlasp@nupelia.uem.br, (RBR) reis.renanb@gmail.com, (WJG) weferson@nupelia.uem.br.
[3] Departamento de Biologia, Universidade Federal de Lavras, Trevo Rotatório Professor Edmir Sá Santos, s/n, 37203-202, Lavras, MG, Brazil. (LJS) luanasartori82@gmail.com.
[4] Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), Centro de Ciências Biológicas, Universidade Estadual de Maringá, PR, Brazil.
[5] Programa de Pós-Graduação em Biologia Comparada, Centro de Ciências Biológicas, Universidade Estadual de Maringá, PR, Brazil.
[6] Centro de Ciências Biológicas e da Saúde, Universidade Estadual do Oeste do Paraná, 85819-210, Cascavel, PR, Brazil. (VPM) vladimir.margarido@unioeste.br.
Authors’ Contribution 

Pedro L. C. Uzeda: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Bruno H. M. Stabile: Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Resources, Software, Validation, Writing-original draft, Writing-review and editing.
Renan B. dos Reis: Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Resources, Validation, Visualization, Writing-original draft, Writing-review and editing.
Luana J. Sartori: Conceptualization, Funding acquisition, Investigation, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Carla S. Pavanelli: Data curation, Funding acquisition, Investigation, Resources, Supervision, Validation, Visualization, Writing-review and editing.
Vladimir P. Margarido: Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Resources, Validation, Visualization, Writing-review and editing.
Weferson J. da Graça: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Project administration, Resources, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Specimens were collected under SISBIO sampling permit #74189 from Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) to Paulo S. Pompeu (Universidade Federal de Lavras).
Competing Interests
The author declares no competing interests.
Data availability statement
All sequences used in the present work are available in supporting information.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
PLCU and LJS were supported by scholarships from Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) under grants number 15385 and 15381, respectively. BHMS and RBR were supported by scholarships from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) process number: 88887.629037/2021–00 and 88887.629034/2021–00, respectively. CSP and WJG receive personal grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (CSP grant: 307124/2023–1, and WJG grants: 305200/2018–6, 307089/2021–5).
Supplementary Material
Supplementary material S1
Supplementary material S2
How to cite this article
Uzeda PLC, Stabile BHM, Reis RB, Sartori LJ, Pavanelli CS, Margarido VP, Graça WJ. Integrative taxonomy reveals a remarkably colored species of Characidium (Characiformes: Crenuchidae) from the upper rio Paraná basin, Brazil. Neotrop Ichthyol. 2026; 24(1):e250174. https://doi.org/10.1590/1982-0224-2025-0174
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 4, 2025
Accepted December 4, 2025
![]() Submitted February 19, 2025
Submitted February 19, 2025
![]() Epub March 23, 2026
Epub March 23, 2026