![]() Ana Clara S. Athayde1,
Ana Clara S. Athayde1, ![]() Fernanda A. Rolim1,
Fernanda A. Rolim1, ![]() Otto B. F. Gadig2,
Otto B. F. Gadig2, ![]() Caio R. Pimentel1,
Caio R. Pimentel1, ![]() Rafael R. Munhoz1,
Rafael R. Munhoz1, ![]() Maisha Gragnolati1,
Maisha Gragnolati1, ![]() Fernando Z. Gibran3,
Fernando Z. Gibran3, ![]() Leonardo M. Neves4,
Leonardo M. Neves4, ![]() Domingos Garrone-Neto5,
Domingos Garrone-Neto5, ![]() Gabriel R. S. Souza5,
Gabriel R. S. Souza5, ![]() Luiza D. Chelotti1,
Luiza D. Chelotti1, ![]() Guilherme H. Pereira‑Filho1 and
Guilherme H. Pereira‑Filho1 and ![]() Fábio S. Motta1
Fábio S. Motta1 ![]()
PDF: Download Here | Supplementary: S1 S2 | Cite this article
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Os recifes subtropicais brasileiros são predominantemente habitats rochosos localizados em uma zona de transição do Atlântico Sudoeste, que sustentam uma ictiofauna rica. A recente descoberta do recife de coral mais austral do Atlântico, associado a um banco de rodolitos nessa região, revelou uma estrutura carbonática que cessou seu crescimento há aproximadamente 2.400 anos. Essa descoberta levantou questionamentos sobre o papel ecológico e a função de um recife de coral mesmo após atingir sua senescência geológica. Este estudo apresenta uma lista compilada e detalhada da fauna de peixes associada ao recife de coral mais austral do Atlântico conhecido e áreas adjacentes, por meio da combinação de Censos Visuais Subaquáticos Estacionários (UVC), desembarques da pesca com rede de emalhe, Censos Visuais Subaquáticos Estacionários (BRUV), programas de monitoramento por ciência cidadã, espécimes-voucher depositados em coleções científicas e revisão da literatura. Um total de 212 táxons pertencentes a 71 famílias foi registrado, distribuídos em 184 Actinopterygii (85,38%) e 31 Chondrichthyes (14,62%). Essa elevada riqueza de peixes recifais indica a importância da heterogeneidade dos habitats recifais para a biodiversidade local, apesar de a área de estudo estar sob influência de múltiplos usos (por exemplo, pesca comercial, pesca recreativa embarcada, pesca submarina e mergulho recreativo). Assim, ações de manejo devem ser direcionadas para permitir a persistência dessa rica fauna de peixes.
Palavras-chave: Censo visual subaquático, Espécies ameaçadas de extinção, Peixes de recife, Subtropical, Vídeo subaquático remoto com isca.
Introduction
The southeastern coast of Brazil is located in a subtropical transition zone of the southwestern Atlantic Ocean, characterized by a high richness of fishes (e.g., Floeter et al., 2001; Cord et al., 2022) and benthic organisms (e.g., Aued et al., 2018), as well as an extensive marine biodiversity, including tropical and temperate organisms as also endemic species (Eschmeyer et al., 2010; Pinheiro et al., 2018; Quimbayo et al., 2021). The subtropical Brazilian reefs are composed mainly of rocky reefs (Floeter et al., 2006; Pinheiro et al., 2018), which are the main habitat of reef fish species (Ferreira et al., 2001) and occur extensively between 19ºS and 29ºS (Anderson et al., 2019). Although rocky reefs generally present less topographic complexity than biogenic reefs, these environments support a rich fish fauna (Ferreira et al., 2001, Luiz et al., 2008; Gibran, Moura, 2012; Pinheiro et al., 2018; Souza et al., 2018; Motta et al., 2021; Rolim et al., 2017, 2019; Hoff et al., 2023), in addition to being important marine ecosystems worldwide for providing an array of ecosystem services, including food security, the survival of marine species and the maintenance of biodiversity, as well as the contribution to human well-being by providing leisure and recreation (Bevilaqcua et al., 2021; Riofrío-Lazo et al., 2022). In contrast, more structurally complex and biodiverse biogenic subtropical reefs are restricted to a few areas within the subtropical Brazilian region, including both rhodolith beds and coral reefs (Pereira-Filho et al., 2019).
Recently mapped, a coral reef associated with a rhodolith bed in the subtropical waters of São Paulo state, southeastern Brazil (around the Queimada Grande Island – QGI; 24°29’S 46°41’W), expanded the southern limit of the Atlantic coral reefs from 17ºS to 24°S (Pereira-Filho et al., 2019, 2021). Structurally complex coral reefs can enhance the abundance and richness of reef fishes (Darling et al., 2017), playing a key role in the trophic and non-trophic interactions between fish and benthic communities (Pereira et al., 2014; Longo et al., 2019). In this regard, the QGI has a high diversity of reef fishes, which may be associated with the three distinct reef habitats present in the region (i.e., rocky reefs, high coral cover, and rhodolith bed) (Pereira-Filho et al., 2019; Grillo et al., 2021; Becker et al., 2024). In addition to the QGI, located 16 km away, the coastal island of Queimada Pequena – QPI (24º22’S 46º48’W) is part of the Tupiniquins Ecological Station (ESEC, IUCN Category Ia: Strict Nature Reserve). Despite being a small no-take area (0.12 km2), the proximity of QPI to the QGI makes it particularly relevant for studying reef fish assemblages, as the short distance can allow ecological connectivity between these islands. The QPI has an exclusion radius of 1 km for marine protection, with only scientific research permitted, providing an important comparison to QGI and helping to understand how local protection and habitat diversity influence reef fish communities (e.g., Gragnolati et al., 2024).
The Brazilian subtropical coast has the highest number of threatened marine species in Brazil (Magris et al., 2021). In this context, coastal islands are important for biodiversity conservation, particularly in areas facing cumulative impacts, where high species diversity coincides with intense human activities. Coastal islands are also often associated with tourism and recreation due to their proximity to the coast (Ghermandi, Nunes, 2013; Freire et al., 2016; Marconi et al., 2020; Santos et al., 2023). However, the context of broad interests and multiple uses of these areas present a challenge to effective management, contributing to adverse environmental impacts and influencing the local economy (Tratalos, Austin, 2001; Coleman et al., 2004; Marconi et al., 2020). This reality results in an alarming scenario, given that Brazilian reef fishes play key ecological roles and their fisheries account for up to 15% of the total marine catches in Brazil (Eggertsen et al., 2024).
Considering the socioecological relevance of the southernmost Atlantic coral reef (see Marconi et al., 2020; Motta et al., 2022; Niz et al., 2023) and the limited number of studies on the fish fauna associated with rocky reefs in the subtropical Brazilian region (e.g., Luiz et al., 2008; Gibran, Moura, 2012; Souza et al., 2018; Motta et al., 2021), the aim of this study is to provide a comprehensive checklist of fish species (actinopterygians and chondrichthyans) occurring in the coastal areas around two nearby islands, Queimada Grande and Queimada Pequena, in southeastern Brazil. We also include information on the conservation status, geographic distribution, and trophic category of the recorded species. Altogether, this dataset provides an important baseline for conservation planning, monitoring, and management.
Material and methods
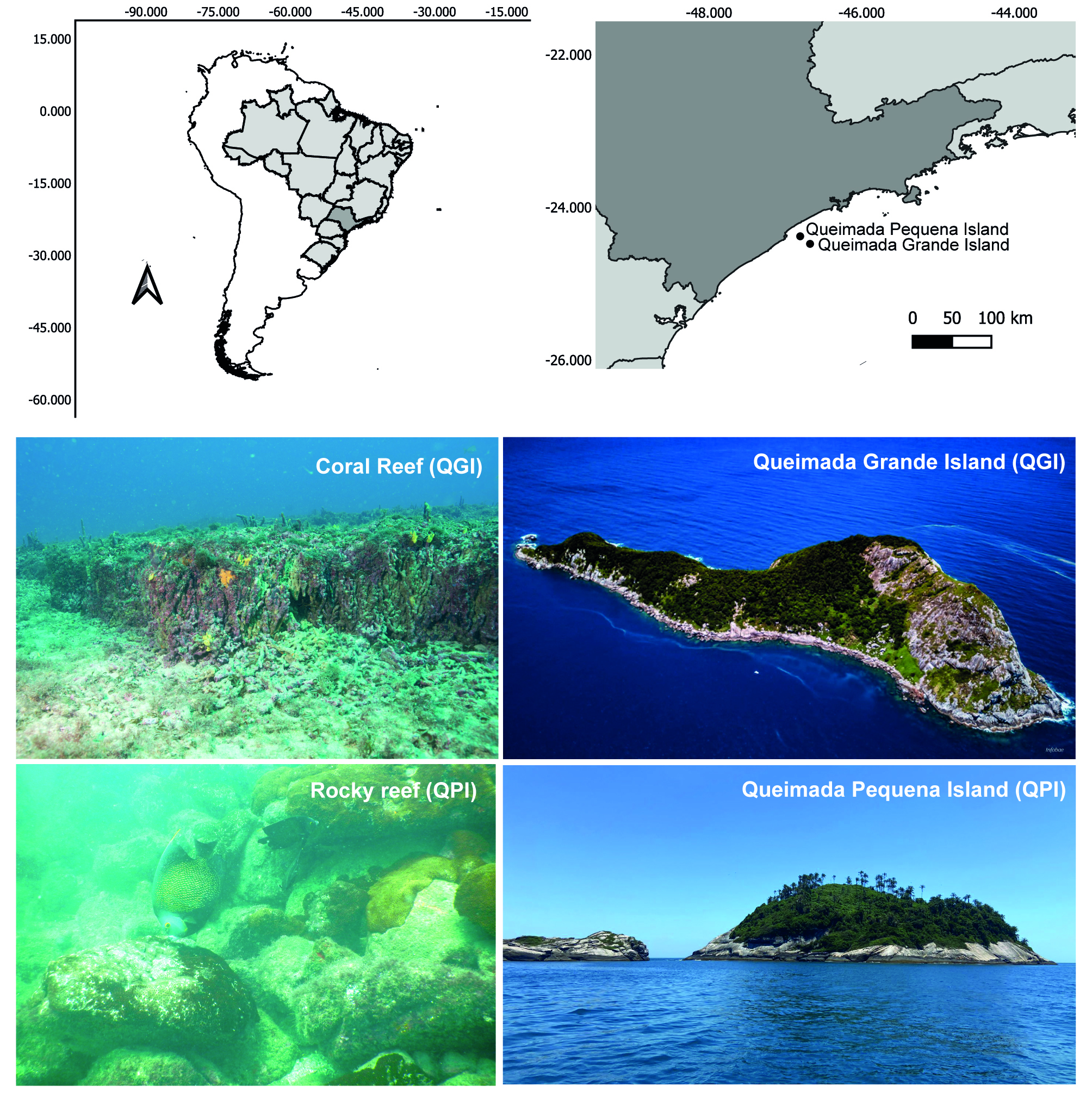
Study sites. Sampling was carried out in Queimada Grande (QGI) and Queimada Pequena (QPI) islands (Fig. 1). The QGI (24°29’S 46°41’W) is located off the state of São Paulo, southeastern Brazil, at a distance of 34.8 km from the coast. The QGI has an emerging area of 0.78 km² and its marine surroundings are part of the Environmental Protected Area of the central coast of São Paulo State (EPA, category V IUCN), a multiple-use Marine Protected Area (MPA). The terrestrial area of the island belongs to the Area of Relevant Ecological Interest (ARIE, category IV IUCN) of Queimada Grande and Queimada Pequena islands. Tourism and fishing are only allowed within the EPA, with some restrictions. For instance, in 2021, through the EPA management plan a zone of the 19 km2 around the QGI was established aiming to making recreational uses (e.g., fishing and scuba diving) compatible with biodiversity conservation (Motta et al., 2021; Niz et al., 2023). Despite this recent zoning, QGI is a historically targeted area for commercial and recreational fisheries, including spearfishing (Niz et al., 2023). QGI also is an important destination for recreational scuba diving (Marconi et al., 2020) and despite these multiple uses, the conservation of local marine biodiversity is compromised due to the lack of effective enforcement and fishing management initiatives (Marconi et al., 2020).
FIGURE 1| Study area in the Southwestern Atlantic. Brazil in light gray and São Paulo State in dark gray on the maps above. Queimada Grande Island (QGI) and Queimada Pequena Island (QPI) represented by black points. Below are the Queimada Grande and Queimada Pequena islands, and the coral and rocky reefs that occur in the area, respectively.
The QPI marine surroundings are protected by the Tupiniquins Ecological Station (Tupiniquins ESEC), a no-take MPA (IUCN category Ia) that covers a radius of 1 km from the coast of the island. QPI is located about 22 km from the coast (24°22’S 46°48’W) with an area of approximately 0.12 km2. Visitation and recreational diving in this area are prohibited, with only scientific research allowed. The proximity to the coast exposes the island to various disturbances, mainly due to the easy access for artisanal fishers and recreational anglers (Giglio et al., 2022).
Data compilation. The checklist was compiled from data collected between 1999 to 2025, composed by: Censos Visuais Subaquáticos Estacionários (UVC), protocol adapted from Minte-Vera et al. (2008); landings of gillnet fisheries from the Cação Project; Baited Remote Underwater Video (BRUV), method widely used to evaluate fish assemblages (Cappo et al., 2006; Mallet, Pelletier, 2014; Goetze et al., 2015); personal observation; photographic; and scientific collection vouchers (Queimada Grande Island, NUPEC Collection – Research and Study Center in Chondrichthyes – 984); citizen science monitoring program from the “Viu Raia? Project”, and literature (LIT; Moura, 2002; Moura et al., 2003; Pivetta et al., 2012).
The method by which species were recorded and documented: UVC = Underwater Visual Census; BRUV = Baited Remote Underwater Video; CP = landings of gillnet fishing (Cação Project); PHO = Photographs; OBS = Personal Observation; VOU = scientific collection vouchers of the Center for Research and Study in Chondrichthyes (NUPEC); Moura et al., 2002, 2003; Pivetta et al., 2012; and Anderson et al., 2020. The record types were categorized in two types of font: LIT = Literature and Present Study (PS) (Tabs. 1, S1;Fig. S2).
TABLE 1 | Record type, method and font. Literature (LIT) and Present Study (PS). Underwater Visual Census (UVC), Baited Remote Underwater Video (BRUV), landings of gillnet fishing (Cação Project), Photographs (PHO), Personal observation (DGN; OBFG; ACSA) and scientific collection vouchers (NUPEC).
Record type | Method | Sampling effort | Font |
Underwater Visual Census (2015) | UVC | 196 visual census | PS |
Baited Remote Underwater Video (2015) | BRUV | 6 deployments | PS |
Baited Remote Underwater Video (2017) | BRUV | 26 deployments | PS |
Baited Remote Underwater Video (2020–2021) | BRUV | 33 deployments | PS |
Landings of gillnet fishing (1996–2005 and 2012–2017) | Cação Project | 326 landing samples | PS |
Photographs (2019–2021) | PHO | 83 photographs | PS |
Domingos Garrone-Neto – DGN (2017–2024) | Personal observation | 25 years of field experience | PS |
Otto B. F. Gadig – OBFG (1996–2017) | Personal observation | 40 years of field experience | PS |
Ana Clara S. Athayde – ACSA (2024) | PHO | 10 dives | PS |
Center for Research and Study in Chondrichthyes (NUPEC) | Scientific Collection Vouchers | 1 species | PS |
Recreational Scuba Diver sightings (2023–2024) | “Viu Raia? Project” | 58 diving operations | PS |
Moura (2002) | UVC | 29 visual census | LIT |
Moura et al. (2003) | UVC | 62 visual census | LIT |
Pivetta et al. (2012) | UVC | 30 visual census | LIT |
Fishes were grouped and listed in the phylogenetic order of families, following Eschmeyer’s Catalog of Fishes (Fricke et al., 2026). Species were identified according to Menezes, Figueiredo (1980, 1985), Figueiredo, Menezes (1980), Carvalho-Filho (1999, 2024), Rolim et al. (2017) and Floeter et al. (2023).
The general diet of species, the geographical distributions and endemism of Southwestern Atlantic were adapted from the literature (Pinheiro et al., 2018; Floeter et al., 2023). Trophic group was categorized as follows: MINV: mobile invertebrate feeders; MCAR: macro carnivores; HERB: herbivores; PLAN: planktivores; OMNI: omnivores; and SINV: sessile invertebrate feeders. About geographical distributions: WA, Western Atlantic; OIB, Oceanic Islands of Brazil; CT, Circumtropical; EA, Eastern Atlantic; BR, Brazilian Coast Endemics; MAR, Mid Atlantic Ridge and SA, Southwestern Atlantic.
The conservation category was based on the International Red List of Threatened Species (International Union for Conservation of Nature – IUCN, 2021), and the National Red List of Threatened Species (ICMBio, 2018; MMA, 2022, 2023).
Results
We compiled a total of 212 taxa belonging to 71 families and 209 species (202 species belonging to 71 families and 89 species belonging to 44 families were recorded in Queimada Grande and Queimada Pequena, respectively). Five species were only recorded in QPI but not recorded in QGI, which were Scomberomorus brasiliensis, Alectis ciliaris, Bothus robinsi, Caranx lugubris, and Selene vomer (Tab. 2). A total of 184 genus (85.38%) included in 54 families was associated to actinopterygians, and 31 genus (14.62%) of 17 families to chondrichthyans (Figs. 2–4). We also recorded a Holocephali (chimaera) for the first time in these areas. The families of actinopterygians with the highest species richness were Carangidae (n = 21), followed by Labridae (n = 19), Epinephelidae (n = 9) and Pomacentridae (n = 9). Regarding the chondrichthyans, the richest families were Carcharhinidae, Dasyatidae and Mobulidae (n = 7, n = 4 and n = 3, respectively).
TABLE 2 | Checklist of the fish species recorded at the Queimada Grande and Queimada Pequena Islands. IUCN: CR = Critically Endangered; EN = Endangered; VU = Vulnerable; NT = Near Threatened; LC = Least Concern; and DD = Data Deficient. National Category – ICMBio: CR = Critically Endangered; EN = Endangered; VU = Vulnerable; NT = Near Threatened; LC = Least Concern; and DD = Data Deficient. Geographic Range: WA = Western Atlantic; OIB = Oceanic Islands of Brazil; CT = Circumtropical; EA = Eastern Atlantic; BR = Brazilian Province Endemics; MAR = Mid Atlantic Ridge; and SA = Southwestern Atlantic. Trophic group: MINV = mobile invertebrate feeders; MCAR = macro-carnivores; HERB = herbivores; PLAN = planktivores; OMNI = omnivores; and SINV = sessile invertebrate feeders. Font record type: Present Study (PS); Literature (LIT). Vouchers used as reference: Diplectrum radiale ACF74110003, Haemulon atlanticus ACF77110001, Halichoeres bathyphilus ACF76060026, Caranx latus ACF78040101, Acanthurus sp. ACF78040101, Gymnothorax ocellatus HEM57022001, Serranus baldwini RLO91092003, Synodus foetens RLO91112303, Bothus sp. RLO91112303, Scorpaena isthmensis RLO93050203, Sphoeroides camila RLO93050203, Canthigaster figueiredoi RLO93050203, Coryphopterus glaucofrenum RLO91061503, Emblemariopsis signifer RLO91061503, Apogon pseudomaculatus RLO91061503, Stegastes pictus RLO91061503, Chromis jubauna RLO91121503, Azurina multilineata RLO92040501, Trachinotus falcatus RPL66120001, Squatina guggenheim IQG001.
Family/Species | QGI | QPI | IUCN | National | Geographic range | Trophic category | Font |
CHONDRICHTHYES | |||||||
Rhincodontidae | |||||||
Rhincodon typus Smith, 1828 | x |
| EN | VU | CT, OIB | PLAN | PS |
Carchariidae | |||||||
Carcharias taurus Rafinesque, 1810 | x | x | CR | CR | – | MCAR | PS |
Lamnidae | |||||||
Isurus oxyrinchus Rafinesque, 1810 | x |
| EN | CR | – | MCAR | PS |
Carcharhinidae | |||||||
Carcharhinus brevipinna (Valenciennes, 1839) | x | x | VU | VU | – | MCAR | PS |
Carcharhinus falciformis (Bibron, 1839) | x | x | VU | CR | – | MCAR | PS |
Carcharhinus limbatus (Valenciennes, 1839) | x | x | VU | NT | – | MCAR | PS |
Carcharhinus obscurus (Lesueur, 1818) | x | x | EN | EN | OIB | MCAR | PS |
Carcharhinus plumbeus (Nardo, 1827) | x | x | EN | CR | OIB | MCAR | PS |
Rhizoprionodon lalandii (Valenciennes, 1839) | x | x | VU | NT | WA | MCAR | PS |
Rhizoprionodon porosus (Poey, 1861) | x | x | VU | DD | WA | MCAR | PS |
Galeocerdidae | |||||||
Galeocerdo cuvier (Péron & Lesueur, 1822) | x | x | NT | NT | CT | MCAR | PS |
Sphyrnidae | |||||||
Sphyrna lewini (Griffith & Smith, 1834) | x | x | CR | CR | OIB | MCAR | PS |
Sphyrna zygaena (Linnaeus, 1758) | x | x | VU | CR | OIB | MCAR | PS |
Squatinidae | |||||||
Squatina guggenheim Marini, 1936 | x | x | EN | CR | SA | – | PS |
Narcinidae | |||||||
Narcine brasiliensis (Olfers, 1831) | x | x | NT | VU | SA | MINV | PS |
Trygonorrhinidae | |||||||
Zapteryx brevirostris (Müller & Henle, 1841) | x | x | EN | VU | SA | MINV | PS |
Rhinobatidae | |||||||
Pseudobatos horkelii (Müller & Henle, 1841) | x | x | CR | CR | SA | MCAR | PS |
Pseudobatos percellens (Walbaum, 1792) | x | x | EN | VU | WA | MCAR | PS |
Dasyatidae | |||||||
Bathytoshia centroura (Mitchill, 1815) | x | x | VU | CR | WA, OIB, EA | MCAR | PS |
Dasyatis hypostigma Santos & Carvalho, 2004 | x | x | EN | DD | SA | MCAR | PS |
Hypanus berthalutzae Petean, Naylor & Lima, 2020 | x | x | VU | – | WA, OIB | MCAR | PS |
Hypanus guttatus (Bloch & Scheider, 1801) | x | x | NT | LC | WA | MCAR | PS |
Gymnuridae | |||||||
Gymnura altavela (Linnaeus, 1758) | x | x | EN | CR | WA, EA | MCAR | PS |
Aetobatidae | |||||||
Aetobatus narinari (Euphrasen, 1790) | x | x | EN | DD | WA, EA | MCAR | PS, LIT |
Myliobatidae | |||||||
Myliobatis freminvillei Lesueur, 1824 | x | x | VU | EN | WA | – | PS |
Rhinopteridae | |||||||
Rhinoptera bonasus (Mitchill, 1815) | x | x | VU | DD | WA | – | PS |
Rhinoptera brasiliensis Müller, 1836 | x | x | VU | CR | WA | – | PS |
Mobulidae | |||||||
Mobula birostris (Walbaum, 1792) | x |
| EN | VU | CT, OIB | PLAN | LIT |
Mobula hypostoma (Bancroft, 1831) | x | x | EN | – | WA, OIB | – | LIT |
Mobula thurstoni (Lloyd, 1908) | x | x | EN | VU | CT, OIB | – | PS |
Callorhinchidae | |||||||
Callorhinchus callorynchus (Linnaeus, 1758) | x | x | VU | DD | SA | – | PS |
ACTINOPTERYGII | |||||||
Elopidae | |||||||
Elops smithi McBride, Rocha, Ruiz-Carus, & Bowen, 2010 | x |
| DD | – | – | MCAR | PS |
Megalopidae | |||||||
Megalops atlanticus Valenciennes, 1847 | x |
| VU | VU | WA, EA, EP | MCAR | PS |
Muraenidae | |||||||
Gymnothorax funebris Ranzani, 1839 | x |
| LC | DD | WA, OIB | MCAR | PS, LIT |
Gymnothorax moringa (Cuvier, 1829) | x | x | LC | DD | WA, OIB, MAR | MCAR | PS, LIT |
Gymnothorax ocellatus Agassiz, 1831 | x |
| LC | DD | WA | MCAR | PS, LIT |
Gymnothorax vicinus (Castelnau, 1855) | x |
| LC | DD | WA, OIB, MAR | MCAR | PS, LIT |
Ophichthidae | |||||||
Myrichthys breviceps (Richardson, 1848) | x |
| LC | LC | WA, OIB | MINV | LIT |
Myrichthys ocellatus (Lesueur, 1825) | x |
| LC | LC | WA, OIB | MINV | PS, LIT |
Ophichthus ophis (Linnaeus, 1758) | x |
| LC | LC | WA, OIB, EA | MCAR | LIT |
Clupeidae | |||||||
Sardinella aurita Valenciennes, 1847 | x |
| DD | DD | WA, EA | PLAN | PS |
Synodontidae | |||||||
Synodus foetens (Linnaeus, 1766) | x |
| LC | LC | WA | MCAR | PS, LIT |
Synodus intermedius (Spix & Agassiz, 1829) | x |
| LC | LC | WA | MCAR | PS, LIT |
Synodus synodus (Linnaeus, 1758) | x |
| LC | LC | WA, OIB, MAR | MCAR | PS |
Holocentridae | |||||||
Holocentrus adscensionis (Osbeck, 1765) | x | x | LC | LC | WA, OIB, MAR | MINV | PS, LIT |
Myripristis jacobus Cuvier, 1829 | x |
| LC | LC | WA, OIB, MAR | MINV | PS, LIT |
Plectrypops retrospinis (Guichenot, 1853) | x |
| LC | LC | WA, OIB | MINV | PS |
Apogonidae | |||||||
Apogon americanus Castelnau, 1855 | x |
| – | LC | BR, OIB | PLAN | PS, LIT |
Apogon quadrisquamatus Longley, 1934 | x |
| LC | DD | WA | PLAN | LIT |
Apogon pseudomaculatus Longley, 1932 | x |
| LC | LC | WA, EA | PLAN | LIT |
Gobiidae | |||||||
Bathygobius soporator (Valenciennes, 1837) | x |
| LC | LC | BR | MINV | LIT |
Coryphopterus dicrus Böhlke & Robins, 1960 | x | x | LC | LC | WA | MINV | LIT |
Coryphopterus glaucofraenum Gill, 1863 | x |
| LC | LC | WA | MINV | PS, LIT |
Ctenogobius saepepallens (Gilbert & Randall, 1968) | x |
| LC | LC | WA | MINV | LIT |
Elacatinus figaro Sazima, Moura & Rosa, 1997 | x | x | – | VU | BR | MINV | PS, LIT |
Gnatholepis thompsoni Jordan, 1904 | x |
| LC | LC | WA | MINV | LIT |
Lythrypnus brasiliensis Greenfield, 1988 | x |
| – | LC | BR, OIB | MINV | LIT |
Ptereleotris randalli Gasparini, Rocha & Floeter, 2001 | x |
| LC | LC | BR | PLAN | PS, LIT |
Dactylopteridae | |||||||
Dactylopterus volitans (Linnaeus, 1758) | x |
| LC | LC | WA, OIB, EA | MINV | LIT |
Mullidae | |||||||
Pseudupeneus maculatus (Bloch, 1793) | x | x | LC | LC | WA, OIB | MINV | PS, LIT |
Callionymidae | |||||||
Callionymus bairdi Jordan, 1888 | x |
| LC | LC | WA, OIB, MAR | MINV | PS |
Syngnathidae | |||||||
Hippocampus erectus Perry, 1810 | x |
| VU | VU | WA, MAR | MINV | LIT |
Hippocampus reidi Ginsburg, 1933 | x |
| NT | VU | WA, OIB | MINV | LIT |
Pomatomidae | |||||||
Pomatomus saltatrix (Linnaeus, 1766) | x |
| VU | NT | CT | MCAR | PS |
Scombridae | |||||||
Katsuwonus pelamis (Linnaeus, 1758) | x |
| LC | LC | CT | MCAR | PS |
Scomberomorus brasiliensis Collette, Russo & Zavala-Camin, 1978 |
| x | LC | LC | WA | MCAR | PS |
Trichiuridae | |||||||
Trichiurus lepturus Linnaeus, 1758 | x | x | LC | LC | CT | MCAR | PS |
Centropomidae | |||||||
Centropomus undecimalis (Bloch, 1792) | x |
| LC | LC | WA | MCAR | PS |
Centropomus spp. | x |
| LC | LC | WA | MCAR | LIT |
Sphyraenidae | |||||||
Sphyraena barracuda (Edwards, 1771) | x |
| LC | LC | CT | MCAR | LIT |
Sphyraena guachancho Cuvier, 1829 | x |
| LC | LC | WA, OIB, EA | MCAR | PS, LIT |
Bothidae | |||||||
Bothus lunatus (Linnaeus, 1758) | x |
| LC | LC | WA, OIB, MAR | MCAR | LIT |
Bothus ocellatus (Agassiz, 1831) | x |
| LC | LC | WA | MINV | PS, LIT |
Bothus robinsi Topp & Hoff, 1972 |
| x | LC | LC | WA | MINV | PS |
Xiphiidae | |||||||
Xiphias gladius Linnaeus, 1758 | x |
| LC | NT | CT | MCAR | PS |
Carangidae | |||||||
Alectis ciliaris (Bloch, 1787) |
| x | LC | LC | CT | MCAR | PS |
Caranx bartholomaei Cuvier, 1833 | x | x | LC | LC | WA, OIB, EA | MCAR | PS, LIT |
Caranx crysos (Mitchill, 1815) | x | x | LC | LC | WA, OIB, MAR | MCAR | PS, LIT |
Caranx hippos (Linnaeus, 1766) | x | x | LC | LC | WA, EA | MCAR | PS |
Caranx latus Agassiz, 1831 | x | x | LC | LC | WA, OIB, MAR | MCAR | PS, LIT |
Caranx lugubris Poey, 1860 |
| x | LC | LC | CT | MCAR | PS |
Caranx ruber (Bloch, 1793) | x |
| LC | LC | WA, OIB, MAR | MCAR | LIT |
Chloroscombrus chrysurus (Linnaeus, 1766) | x | x | LC | LC | WA, EA | PLAN | PS, LIT |
Decapterus macarellus (Cuvier, 1833) | x |
| LC | LC | CT | PLAN | PS |
Decapterus punctatus (Cuvier,1829) | x |
| LC | LC | WA, MAR, EA | PLAN | PS |
Elagatis bipinnulata (Quoy & Gaimard, 1825) | x |
| LC | LC | CT | MCAR | PS |
Oligoplites spp. | x |
| LC | LC | WA | MCAR | PS |
Pseudocaranx dentex (Bloch & Schneider, 1801) | x | x | LC | LC | CT | MINV | PS, LIT |
Selene vomer (Linnaeus, 1758) |
| x | LC | LC | WA | PLAN | PS |
Seriola dumerili (Risso, 1810) | x | x | LC | LC | CT | MCAR | PS, LIT |
Seriola fasciata (Bloch, 1793) | x |
| LC | DD | WA | MCAR | LIT |
Seriola lalandi Valenciennes, 1833 | x |
| LC | LC | CT | MCAR | PS, LIT |
Seriola rivoliana Valenciennes, 1833 | x |
| LC | LC | CT | MCAR | PS, LIT |
Trachinotus carolinus (Linnaeus, 1766) | x |
| LC | LC | WA | MINV | PS |
Trachinotus falcatus (Linnaeus, 1758) | x |
| LC | LC | WA, OIB | MINV | PS |
Trachinotus goodei Jordan & Evermann, 1896 | x | x | LC | LC | WA | MINV | PS, LIT |
Rachycentridae | |||||||
Rachycentron canadum (Linnaeus, 1766) | x | x | LC | LC | CT | MCAR | PS, LIT |
Coryphaenidae | |||||||
Coryphaena equiselis Linnaeus, 1758 | x |
| LC | LC | CT | MCAR | PS |
Coryphaena hippurus Linnaeus, 1758 | x |
| LC | LC | CT | MCAR | PS |
Belonidae | |||||||
Tylosurus acus (Lacepède, 1803) | x |
| LC | LC | WA, EA | MCAR | PS |
Mugilidae | |||||||
Mugil spp. | x |
| – | – | – | HERB | LIT |
Pomacentridae | |||||||
Abudefduf saxatilis (Linnaeus, 1758) | x | x | LC | LC | WA, OIB, MAR | OMNI | PS, LIT |
Azurina multilineata (Guichenot, 1853) | x | x | LC | LC | WA, OIB, MAR | PLAN | PS, LIT |
Chromis flavicauda (Günther, 1880) | x |
| DD | LC | BR, OIB | PLAN | LIT |
Chromis jubauna Moura, 1995 | x |
| – | LC | BR, OIB | PLAN | PS, LIT |
Chromis limbata (Valenciennes, 1833) | x |
| LC | – | SA, EA | PLAN | PS, LIT |
Chromis vanbebberae McFarland, Baldwin, Robertson, Rocha & Tornabene, 2020 | x |
| LC | LC | BR, OIB | PLAN | LIT |
Stegastes fuscus (Cuvier, 1830) | x | x | LC | LC | BR, OIB | HERB | PS, LIT |
Stegastes pictus (Castelnau, 1855) | x | x | – | LC | BR, WA, OIB | OMNI | PS, LIT |
Stegastes variabilis (Castelnau, 1855) | x | x | – | LC | BR | HERB | PS, LIT |
Tripterygiidae | |||||||
Enneanectes altivelis Rosenblatt, 1960 | x |
| LC | LC | WA, OIB | MINV | LIT |
Blenniidae | |||||||
Hypleurochilus fissicornis (Quoy & Gaimard, 1824) | x |
| LC | LC | SA, EA | SINV | LIT |
Hypsoblennius invemar Smith-Vaniz & Acero, 1980 | x |
| LC | LC | WA | MINV | LIT |
Parablennius marmoreus (Poey, 1876) | x | x | LC | LC | WA | OMNI | PS, LIT |
Parablennius pilicornis (Cuvier, 1829) | x |
| LC | LC | SA, EA | OMNI | PS LIT |
Scartella cristata (Linnaeus, 1758) | x |
| LC | LC | WA, EA | HERB | LIT |
Labrisomidae | |||||||
Gobioclinus kalisherae (Jordan, 1904) | x | x | LC | LC | WA, OIB | MINV | LIT |
Labrisomus nuchipinnis (Quoy & Gaimard, 1824) | x | x | LC | LC | WA, OIB, EA | MCAR | PS, LIT |
Malacoctenus delalandii (Valenciennes, 1836) | x | x | LC | LC | WA | MINV | LIT |
Malacoctenus zaluari Carvalho-Filho, Gasparini & Sazima, 2020 | x |
| LC | LC | – | – | LIT |
Starksia brasiliensis (Gilbert, 1900) | x |
| LC | LC | BR | MINV | LIT |
Chaenopsidae | |||||||
Emblemariopsis signifer (Ginsburg, 1942) | x | x | LC | LC | BR | MINV | LIT |
Serranidae | |||||||
Diplectrum formosum (Linnaeus, 1766) | x |
| LC | LC | WA, OIB | MCAR | PS |
Diplectrum radiale (Quoy & Gaimard, 1824) | x |
| LC | LC | WA | MCAR | PS, LIT |
Serranus atrobranchus (Cuvier, 1829) | x |
| LC | LC | WA | MCAR | PS |
Serranus baldwini (Evermann & Marsh, 1899) | x | x | LC | LC | WA, OIB | MCAR | PS, LIT |
Serranus flaviventris (Cuvier, 1829) | x | x | LC | LC | WA | MCAR | PS, LIT |
Epinephelidae | |||||||
Cephalopholis furcifer (Valenciennes, 1828) | x |
| LC | LC | WA, OIB, MAR | PLAN | PS, LIT |
Epinephelus itajara (Lichtenstein, 1822) | x |
| VU | CR | WA, OIB, EA | MCAR | LIT |
Epinephelus marginatus (Lowe, 1834) | x | x | VU | VU | SA, EA, WA | MCAR | PS, LIT |
Epinephelus morio (Valenciennes, 1828) | x |
| VU | VU | WA | MCAR | LIT |
Hyporthodus niveatus (Valenciennes, 1828) | x |
| VU | VU | WA | MCAR | PS, LIT |
Mycteroperca acutirostris (Valenciennes, 1828) | x | x | LC | DD | WA | MCAR | PS, LIT |
Mycteroperca bonaci (Poey, 1860) | x |
| NT | VU | WA, OIB | MCAR | PS, LIT |
Mycteroperca interstitialis (Poey, 1860) | x |
| VU | VU | WA, OIB | MCAR | LIT |
Mycteroperca microlepis (Goode & Bean, 1879) | x |
| VU | DD | WA | MCAR | LIT |
Scorpaenidae | |||||||
Scorpaena isthmensis Meek & Hildebrand, 1928 | x |
| LC | LC | WA, OIB | MCAR | LIT |
Scorpaena plumieri Bloch, 1789 | x |
| LC | LC | WA, OIB, MAR | MCAR | PS |
Labridae | |||||||
Bodianus pulchellus (Poey, 1860) | x |
| LC | LC | WA, OIB, EA | MINV | PS, LIT |
Bodianus rufus (Linnaeus, 1758) | x | x | LC | LC | WA, OIB | MINV | PS, LIT |
Clepticus brasiliensis Heiser, Moura & Robertson, 2000 | x |
| LC | LC | BR, OIB | PLAN | PS, LIT |
Cryptotomus roseus Cope, 1871 | x |
| LC | LC | WA, OIB, MAR | HERB | PS, LIT |
Doratonotus megalepis Günther, 1862 | x |
| LC | LC | WA, OIB | MINV | LIT |
Halichoeres brasiliensis (Bloch, 1791) | x | x | DD | LC | BR, OIB | MINV | PS, LIT |
Halichoeres dimidiatus (Agassiz, 1831) | x | x | LC | LC | BR, OIB | MINV | PS, LIT |
Halichoeres poeyi (Steindachner, 1867) | x | x | LC | LC | WA, OIB | MINV | PS, LIT |
Halichoeres sazimai Luiz, Ferreira & Rocha, 2009 | x | x | – | LC | BR | MINV | PS, LIT |
Nicholsina usta (Valenciennes, 1840) | x |
| LC | LC | WA | HERB | LIT |
Scarus trispinosus Valenciennes, 1840 | x |
| EN | EN | BR | HERB | LIT |
Scarus zelindae Moura, Figueiredo & Sazima, 2001 | x |
| DD | VU | BR, OIB | HERB | PS, LIT |
Sparisoma amplum (Ranzani, 1841) | x |
| LC | NT | BR, OIB | HERB | PS, LIT |
Sparisoma axillare (Steindachner, 1878) | x | x | DD | VU | BR, OIB | HERB | PS, LIT |
Sparisoma frondosum (Agassiz, 1831) | x | x | DD | VU | BR, OIB | HERB | PS, LIT |
Sparisoma radians (Valenciennes, 1840) | x |
| LC | LC | WA | HERB | LIT |
Sparisoma tuiupiranga Gasparini, Joyeux & Floeter, 2003 | x | x | LC | LC | BR, OIB | HERB | PS |
Thalassoma noronhanum (Boulenger, 1890) | x |
| LC | LC | BR, OIB | MINV | LIT |
Xyrichthys novacula (Linnaeus, 1758) | x |
| LC | LC | WA, OIB, EA | MINV | LIT |
Kyphosidae | |||||||
Kyphosus sectatrix (Linnaeus, 1758) | x | x | LC | – | WA, OIB, MAR | HERB | PS, LIT |
Kyphosus vaigiensis (Quoy & Gaimard, 1825) | x |
| – | – | WA, EA | HERB | LIT |
Pempheridae | |||||||
Pempheris schomburgkii Müller & Troschel, 1848 | x | x | LC | LC | WA, OIB | PLAN | PS, LIT |
Gerreidae | |||||||
Eucinostomus melanopterus (Bleeker, 1863) | x |
| LC | LC | WA, EA | MINV | LIT |
Ephippidae | |||||||
Chaetodipterus faber (Broussonet, 1782) | x | x | LC | LC | WA | OMNI | PS, LIT |
Sciaenidae | |||||||
Cynoscion acoupa (Lacepède, 1801) | x |
| VU | NT | – | – | PS |
Cynoscion leiarchus (Cuvier, 1830) | x |
| LC | LC | – | – | PS |
Odontoscion dentex (Cuvier, 1830) | x | x | LC | LC | WA | MCAR | PS, LIT |
Pareques lineatus (Cuvier, 1830) | x | x | LC | DD | WA | MINV | PS, LIT |
Pogonias cromis (Linnaeus, 1766) | x |
| LC | EN | – | – | PS |
Haemulidae | |||||||
Anisotremus surinamensis (Bloch, 1791) | x | x | DD | DD | WA, OIB | MINV | PS, LIT |
Anisotremus virginicus (Linnaeus, 1758) | x | x | LC | LC | WA | MINV | PS, LIT |
Haemulon atlanticus Carvalho, Marceniuk, Oliveira & Wosiacki, 2020 | x | x | LC | LC | WA | MINV | PS, LIT |
Haemulon aurolineatum Cuvier, 1830 | x | x | LC | LC | WA | MINV | PS, LIT |
Haemulon parra (Desmarest, 1823) | x | x | LC | LC | WA, OIB | MINV | PS |
Haemulon plumierii (Lacepède, 1801) | x |
| LC | DD | WA | MINV | PS, LIT |
Orthopristis rubra (Cuvier, 1830) | x |
| LC | LC | WA | MINV | PS |
Lobotidae | |||||||
Lobotes surinamensis (Bloch, 1790) | x |
| LC | LC | CT | MCAR | PS |
Lutjanidae | |||||||
Lutjanus analis (Cuvier, 1828) | x |
| NT | NT | WA | MCAR | PS, LIT |
Lutjanus cyanopterus (Cuvier, 1828) | x | x | VU | VU | WA | MCAR | PS, LIT |
Lutjanus jocu (Bloch & Schneider, 1801) | x |
| DD | NT | WA, OIB, MAR | MCAR | PS, LIT |
Lutjanus synagris (Linnaeus, 1758) | x |
| NT | NT | WA | MCAR | PS |
Ocyurus chrysurus (Bloch, 1791) | x |
| DD | NT | CT | MCAR | PS |
Pristipomoides freemani Anderson, 1966 | x |
| LC | LC | WA | MCAR | PS |
Rhomboplites aurorubens (Cuvier, 1829) | x | x | VU | NT | WA, OIB | MCAR | PS, LIT |
Malacanthidae | |||||||
Malacanthus plumieri (Bloch, 1786) | x |
| LC | LC | WA, OIB, MAR | MCAR | PS, LIT |
Pomacanthidae | |||||||
Centropyge aurantonotus Burgess, 1974 | x |
| LC | DD | BR, WA, OIB | HERB | LIT |
Holacanthus ciliaris (Linnaeus, 1758) | x |
| LC | DD | WA, OIB | SINV | LIT |
Holacanthus tricolor (Bloch, 1795) | x |
| LC | DD | WA, OIB | SINV | PS, LIT |
Pomacanthus arcuatus (Linnaeus, 1758) | x |
| LC | DD | WA, OIB | OMNI | LIT |
Pomacanthus paru (Bloch, 1787) | x | x | LC | DD | WA, OIB, MAR | OMNI | PS, LIT |
Chaetodontidae | |||||||
Chaetodon sedentarius Poey, 1860 | x |
| LC | LC | WA, EA | SINV | LIT |
Chaetodon striatus Linnaeus, 1758 | x | x | LC | LC | WA, OIB | SINV | PS, LIT |
Acanthuridae | |||||||
Acanthurus bahianus Castelnau, 1855 | x | x | LC | LC | BR, WA, OIB | HERB | PS, LIT |
Acanthurus chirurgus (Bloch, 1787) | x | x | LC | LC | WA, OIB, MAR | HERB | PS, LIT |
Acanthurus coeruleus Bloch & Schneider, 1801 | x | x | LC | LC | WA, OIB, MAR | HERB | LIT |
Sparidae | |||||||
Calamus penna (Valenciennes, 1830) | x |
| LC | LC | WA | MINV | PS, LIT |
Calamus pennatula Guichenot, 1868 | x |
| LC | LC | WA | MINV | PS, LIT |
Diplodus argenteus (Valenciennes, 1830) | x | x | LC | LC | BR, OIB | OMNI | PS, LIT |
Pagrus pagrus (Linnaeus, 1758) | x |
| LC | DD | WA, EA, OIB | MINV | PS, LIT |
Priacanthidae | |||||||
Heteropriacanthus cruentatus (Lacepède, 1801) | x |
| LC | LC | CT | MCAR | LIT |
Priacanthus arenatus Cuvier, 1829 | x |
| LC | LC | WA, OIB, EA | MCAR | PS, LIT |
Ogcocephalidae | |||||||
Ogcocephalus vespertilio (Linnaeus, 1758) | x | x | LC | LC | WA | MINV | PS, LIT |
Diodontidae | |||||||
Diodon holocanthus Linnaeus, 1758 | x |
| LC | LC | CT | MINV | LIT |
Diodon hystrix Linnaeus, 1758 | x |
| LC | LC | CT | MINV | LIT |
Tetraodontidae | |||||||
Canthigaster figueiredoi Moura & Castro, 2002 | x | x | LC | LC | BR, WA, OIB | OMNI | PS, LIT |
Sphoeroides camila Carvalho-Filho, Rotundo, Pitassy & Sazima, 2023 | x | x | LC | LC | WA, OIB, EA | MINV | PS, LIT |
Sphoeroides testudineus (Linnaeus, 1758) | x |
| LC | DD | WA | MINV | PS |
Ostraciidae | |||||||
Acanthostracion quadricornis (Linnaeus, 1758) | x |
| LC | LC | WA, OIB, EA | SINV | LIT |
Acanthostracion polygonius Poey, 1876 | x |
| LC | LC | WA, OIB | SINV | LIT |
Lactophrys trigonus (Linnaeus, 1758) | x |
| LC | LC | WA, OIB | OMNI | LIT |
Monacanthidae | |||||||
Aluterus scriptus (Osbeck, 1765) | x |
| LC | LC | CT | OMNI | LIT |
Cantherhines macrocerus (Hollard, 1843) | x |
| LC | LC | WA, OIB, EA | OMNI | PS |
Cantherhines pullus (Ranzani, 1842) | x |
| LC | LC | WA, OIB, EA | OMNI | PS, LIT |
Stephanolepis hispida (Linnaeus, 1766) | x |
| LC | LC | WA, OIB, EA | OMNI | LIT |
Balistidae | |||||||
Balistes vetula Linnaeus, 1758 | x |
| NT | NT | WA, OIB, MAR | MINV | PS, LIT |
FIGURE 2| Some reef fishes from Queimada Grande and Queimada Pequena islands. A. Gymnothorax moringa; B. Sparisoma tuiupiranga; C. Ptereleotris randalli; D. Myrichthys ocellatus; E. Epinephelus marginatus; F. Chromis limbata; G. Ogcocephalus vespertilio; H. Elacatinus figaro; I. Pomacanthus paru (juvenile); J. Parablennius marmoreus (yellow phase); K. Dasyatis hypostigma; L. Mobula birostris. Photos by Carlo L. Francini.
FIGURE 3| Some shark and chimaera species from Queimada Grande and Queimada Pequena islands. A. Sphyrna lewini; B. Sphyrna zygaena – 86 cm; C. Rhizoprionodon lalandii; D. Rhizoprionodon porosus; E. Carcharhinus brevipinna – 70 cm; F. Carcharhinus falciformis; G. Carcharhinus limbatus; H. Carcharhinus obscurus; I. Carcharhinus plumbeus – 87 cm; J. Carcharias taurus – 93.7 cm; K. Galeocerdo cuvier; L. Isurus oxyrinchus; M. Rhincodon typus; N. Squatina guggenheim; O. Callorhinchus callorynchus. Photos by Otto Bismarck F. Gadig, except photo M by C. L. Sampaio.
FIGURE 4| Some ray species from Queimada Grande and Queimada Pequena islands. A. Mobula thurstoni; B. Modula birostris; C. Mobula hypostoma; D. Narcine brasiliensis; E. Aetobatus narinari; F. Myliobatis freminvillei; G. Rhinoptera brasiliensis; H. Rhinoptera bonasus; I. Pseudobatos percellens; J. Pseudobatos horkelii; K. Zapteryx brevirostris; L. Gymnura altavela; M. Dasyatis hypostigma; N. Bathytoshia centroura; O. Hypanus guttatus; P. Hypanus berthalutzae. Photos by Otto Bismarck F. Gadig, except photo B by William White, F by C. E. Rangel, N by H. Santos, and P by F. Petean.
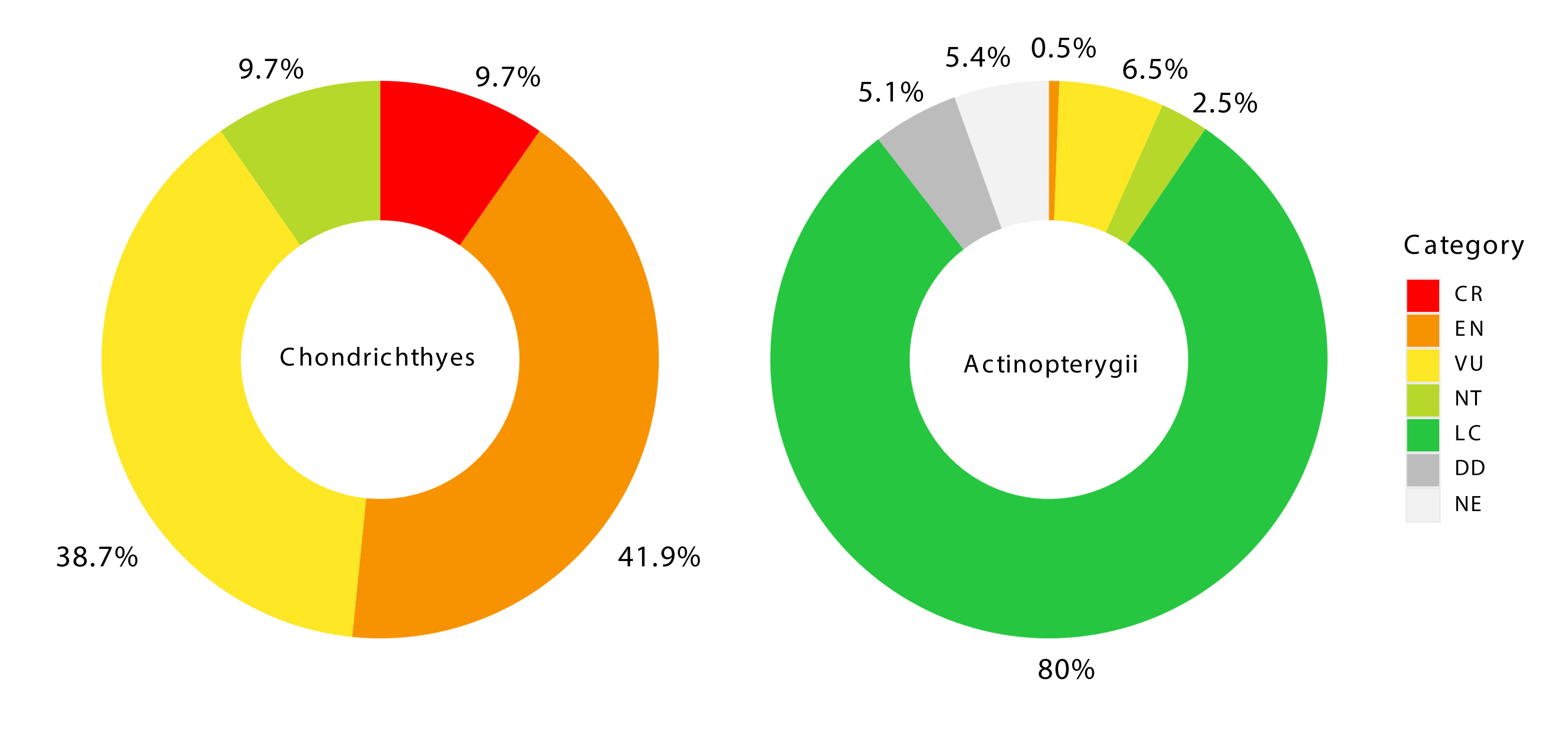
Regarding global conservation status, 42 species (~20% of the total) were classified in threatened categories (CR, EN, or VU; Tab. 2; Fig. 5). Three chondrichthyan species (~10% of this group of fish) were listed as Critically Endangered (CR) by the IUCN: Carcharias taurus, Sphyrna lewini, and Pseudobatos horkelii; all were recorded in both islands. Among the 14 Endangered (EN) species identified, 13 were chondrichthyans (42%) and one is an actinopterygian. Four species were recorded exclusively in QGI: Rhincodon typus, Isurus oxyrinchus, Mobula birostris, and Scarus trispinosus. The remaining species were observed on both islands. Twenty-four species were classified as Vulnerable (VU), including 12 chondrichthyans (38.7%) and 12 actinopterygians (~6.5%). Of these, 15 species were recorded in both islands, while nine were observed exclusively in QGI: Megalops atlanticus, Pomatomus saltatrix, Hippocampus erectus, Cynoscion acoupa, Epinephelus morio, E. itajara, Hyporthodus niveatus, Mycteroperca interstitialis, and M. microlepis. Regarding the national conservation status, ~6% of species were classified as Critically Endangered (CR), ~2% as Endangered (EN) and ~9% as Vulnerable (VU).
FIGURE 5| IUCN threatened species for chondrichthyans and actinopterygians registered in the Queimada Grande and Queimada Pequena Islands. Critically Endangered (CR); Endangered (EN); Vulnerable (VU), Near Threat (NT); Least Concern (LC); Data Deficient (DD) and Not Evaluated (NE).
Regarding to the trophic groups, most species recorded were macro carnivores (~40%; n = 85), mobile invertebrate feeders (~26.4%; n = 56), herbivores (~8.9%; n = 19) and planktivores (~8.9%; n = 19) (Tab. 1). Considering only actinopterygians, the proportion of species belonging to each trophic group were very similar between the studied islands (Tab. 1). Among to the chondrichthyes recorded, 64.5% of the species were macro carnivores (mostly Carcharhinidae), while 6.5% were mobile invertebrate feeders (batoids), and 6.5% are planktivores (e.g., Rhincodon typus and Mobula birostris). Most species (~25%; n = 53) are exclusively distributed across the Western Atlantic, followed by circumtropical distribution (~11%; n = 23) (Tab. 1). Furthermore, ~4% of the species (n = 8) are endemic to Brazil, and one non-native species, Chromis limbata, was also recorded.
Discussion
Here we demonstrated that the habitats associated with the southernmost Atlantic coral reef off QGI, along with the rocky reefs of QPI, support a remarkably high reef fish species richness (n = 208). Despite the relatively small reef area, we recorded nearly one third of the 733 reef fish species found along the 8,000 km of the Brazilian coast, seamounts and oceanic islands (Pinheiro et al., 2018), highlighting the significant socioecological and conservation values of these insular environments. The reef fish richness of QGI is comparable to many southeastern Atlantic no-take MPAs (Luiz et al., 2008; Rolim et al., 2017, 2019; Hoff et al., 2023). However, concerning biomass, QGI presents lower values than that observed in no-take MPAs located in São Paulo State coast, probably in the face of fishing effects (see Motta et al., 2021; Gragnolati et al., 2024).
The high fish species richness recorded in this relatively small study area can be explained by a mosaic of reef environments, including a unique condition of QGI at this subtropical region, with different types of complexity and sizes of submerged rocky reefs, a coral reef, rhodolith beds and sandy channels (Pereira-Filho et al., 2019, 2021; Grillo et al., 2021; Motta et al., 2022). Furthermore, seasonal (austral summer) upwellings may create favorable oceanographic conditions such as nutrient enrichment for the occurrence, spawning, and persistence of fish species (Katsuragawa et al., 2014; Motta et al., 2022). The disparity in fish species richness between QGI (202 species) and QPI (89 species) may be related to the larger reef area and higher habitat heterogeneity at QGI, in addition to having a 75,000 m² coral reef at 7–12 m depth, built mainly by M. decactis colonies cemented by crustose coralline algae (CCA) (Pereira-Filho et al., 2019). However, further studies are needed to confirm the influence of these habitat differences on fish richness.
Nearly 20% (40 species) of the recorded fish species are classified as threatened with extinction. Considering only the Chondrichthyes (31 species), this proportion increases to approximately 90% and 55%, according to global (IUCN) and national (ICMBio) assessments, respectively. These findings align with previous studies indicating that the coast of São Paulo harbors the highest number of threatened marine fish species along the Brazilian coastline (Ceretta et al., 2020; Magris et al., 2021). This aspect can be explained by two factors: the São Paulo coast is located in a faunal transition zone and is subject to high levels of cumulative impacts, especially overfishing (Magris et al., 2021). Moreover, these results reinforce, at a local scale, the high number of threatened species of Chondrichthyes in Brazil. For instance, approximately 33% of Chondrichthyes along the Brazilian coast are classified as threatened, similar to an estimated global rate of 32.6% (Dulvy et al., 2021).
Although most recorded actinopterygians species (81%) are categorized as Least Concern, some commercially important families include threatened species, such as Epinephelidae (e.g., Epinephelus itajara – CR; and E. marginatus – VU), the subfamily Scarinae (e.g., Scarus trispinosus – EN), and Lutjanidae represented by Lutjanus cyanopterus (Cuvier, 1828), classified as Vulnerable (VU). According to Niz et al. (2023), jacks, such as Seriola spp. and Alectis ciliaris, dusky grouper (Epinephelus marginatus) and cubera snapper (Lutjanus cyanopterus) are among the main targets of recreational fishing (onboard and spearfishing) around QGI. Regarding cubera snapper, the first in situ validation of a spawning aggregation of this species in subtropical waters off the southwestern Atlantic was recorded in close proximity to QGI and QPI (Motta et al., 2022), reinforcing the importance of local management and conservation efforts.
The high richness of macro carnivores (39.4%) and mobile invertebrate feeders (27.3%), as well as the predominance of species exclusively distributed in the Western Atlantic (24.7%), is consistent with previous studies conducted in the subtropical southwestern Atlantic (Ferreira et al., 2004; Luiz et al., 2008; Daros et al., 2012; Gibran, Moura, 2012). Still regarding the trophic groups, it is also worth highlighting the high richness of macro carnivore, which may be directly related to the various sources of information used which, in turn, include sampling through multiple techniques (e.g., UVC, BRUV, gillnet landings, citizen science) across different habitats (e.g., coral and rocky reef, water column, and sandy and rhodolith beds). By combining these methods and habitats, we detected species that are otherwise difficult to observe, such as large predatory fishes and elasmobranchs. Furthermore, this raises concerns about biodiversity conservation, as the group includes species under intense fishing pressure (Floeter et al., 2023), many of which are currently threatened with extinction, including most epinephelids, some lutjanids, and various species of sharks and rays.
Although the rocky reefs of the QPI are located within a no-take MPA, we observed fishing gear alongside different stages of benthic organism colonization during our field sampling trips, indicating long time frequent illegal fishery. Given the current low management effectiveness of the Tupiniquins ESEC, resulting from the lack of enforcement, the non-implementation of the management plan, and the inactivity of the management council (see Giglio et al., 2019), reactivating the effective implementation of this MPA, particularly through strengthened awareness and enforcement measures, is essential to safeguard the local fish fauna. Alternatively, recategorizing the area from an Ecological Station to a Park would permit public visitation such as recreational diving, which has been shown to help reduce the risk of illegal fishing in popular dive destinations (Steenbergen, 2013; Motta et al., 2021).
In 2003, a formal proposal for the establishment of a marine national park around Queimada Grande Island (QGI) was introduced by the Brazilian government. However, conflicts with the fishing sector, particularly the recreational fishing, impeded its advancement. Recreational activities at QGI have historical significance and support a substantial local economic chain. For example, onboard recreational fishing is estimated to generate at least USD 350,000 annually (Niz et al., 2023), while recreational scuba diving contributes approximately USD 160,000 per year to the regional economy (Agune, 2024). Additionally, the expansion of no-take zones of two coastal marine protected areas in São Paulo State (Laje de Santos Marine State Park and Alcatrazes Archipelago Wildlife Refuge) has increased the pressure for more effective management of the multiple-use MPA encompassing QGI. This culminated in the establishment of a tourism zoning regulation in 2021. In this context, Niz et al. (2023) developed spatial models to assess various risk scenarios to biodiversity, habitat integrity, and cultural ecosystem services (e.g., recreational activities) at QGI. Their findings suggest that the most effective management scenario is one that harmonizes conservation goals with recreational uses (Niz et al., 2023). Key management strategies highlighted include measures already outlined in the management plan of the multiple-use MPA (EPA), such as the prohibition of trawling within a 500-meter buffer zone around the island and anchoring on coral reef areas. Other important measures established under federal regulations, including catch quotas for recreational fishing, minimum and maximum catch sizes and prohibitions on the capture of threatened species (Niz et al., 2023).
Given the close relationship between fish populations and ecosystem services (Holmlund, Hammer, 1999), the removal of fish species can alter community structure and interactions, thereby affecting ecosystem functions (Luza et al., 2023; Eggertsen et al., 2024) and reducing both the productivity of recreational fisheries and the quality of recreational diving experience. Therefore, it is important to carry out systematic efforts to raise awareness among these users, as well as to develop and implement a tourism management plan to ensure sustainable use. In fact, these important subtropical reefs (QGI and QPI) still lack policies to safeguard their socioecological attributes, such as high fish richness. Here, we provide new information about the hidden biodiversity of the Brazilian subtropical reefs and highlight the need for measures to promote effective conservation and responsible use of the marine life in this singular mosaic of reef habitats.
Acknowledgments
We thank the management staff of the Tupiniquins Ecological Station (ESEC Tupiniquins) and Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) and Fundação Florestal by research permits; Bruno Oliveira, Lawrence Ikeda and Marcos Hamamura for field support and to share some fish records for the QGI.
References
Agune FC. Contribuição econômica do mergulho recreativo em áreas marinhas protegidas sujeitas a diferentes regimes e contextos de gestão: um estudo de caso para o sudeste do Brasil. [Master Dissertation]. Santos: Universidade Federal de São Paulo; 2024.
Anderson AB, Batista MB, Gibran FZ, Félix-Hackradt FC, Hackradt CW, García-Charton JA et al. Habitat use of five key species of reef fish in rocky reef systems of southern Brazil: evidences of MPA effectiveness. Mar Biodivers. 2019; 49:1027–36. https://doi.org/10.1007/s12526-018-0893-6
Anderson AB, Silva JP, Sorvilo R, Francini CLB, Floeter SR, Barreiros JP. Population expansion of the invasive Pomacentridae Chromis limbata (Valenciennes, 1833) in southern Brazilian coast: long-term monitoring, fundamental niche availability and new records. J Fish Biol. 2020; 97(2):362–73. https://doi.org/10.1111/jfb.14365
Aued AW, Smith F, Quimbayo JP, Candido DV, Longo GO, Ferreira CE et al. Large-scale patterns of benthic marine communities in the Brazilian Province. PLoS ONE. 2018; 13(6):e0198452. https://doi.org/10.1371/journal.pone.0198452
Becker ACF, Roos NC, Dias GM, Gibran FZ, Motta FS, Pereira-Filho GH. Predation effects on benthic communities are maximized by greater taxonomic richness and diversity in Subtropical SW Atlantic. Estuar Coast Shelf Sci. 2024; 302:108779. https://doi.org/10.1016/j.ecss.2024.108779
Bevilaqcua S, Airoldi L, Ballesteros E, Benedetti-Cecchi L, Boero F, Bulleri F et al. Chapter One – Mediterranean rocky reefs in the Anthropocene: present status and future concerns. Adv Mar Biol. 89:1–51. https://doi.org/10.1016/bs.amb.2021.08.001
Cappo M, Harvey E, Shortis M. Counting and measuring fish with baited video techniques – an overview. In: Lyle JM, Furlani DM, Buxton CD, editors. Australian Society for Fish Biology Workshop Proceedings. Tasmania: Australian Society for Fish Biology; 2006. p.101–14.
Carvalho-Filho A. Peixes: Costa Brasileira. São Paulo: Editora Melro; 1999.
Carvalho-Filho A. Peixes: Costa Brasileira. São Paulo: Literare Books; 2024.
Ceretta BF, Fogliarini CO, Giglio VJ, Maxwell MF, Waechter LS, Bender MG. Testing the accuracy of biological attributes in predicting extinction risk. Perspect Ecol Conserv. 2020; 18(1):12–18. https://doi.org/10.1016/j.pecon.2020.01.003
Coleman FC, Figueira WF, Ueland JS, Crowder LB. The impact of United States recreational fisheries on marine fish populations. Science. 2004; 305:1958–60. https://doi.org/10.1126/science.1100397
Cord I, Nunes LT, Barroso CX, Freire AS, Gadig OB, Gomes PB et al. Brazilian marine biogeography: a multi-taxa approach for outlining sectorization. Mar Biol. 2022; 169(5):61. https://doi.org/10.1007/s00227-022-04045-8
Darling ES, Graham NA, Januchowski-Hartley FA, Nash KL, Pratchett MS, Wilson SK. Relationships between structural complexity, coral traits, and reef fish assemblages. Coral Reefs. 2017; 36:561–75. https://doi.org/10.1007/s00338-017-1539-z
Daros FA, Bueno LS, Vilar CC, Passos AC, Spach HL. Checklist of rocky reef fishes from the Currais Archipelago and Itacolomis Island, Paraná State, Brazil. Check List. 2012; 8(3):349–54. https://doi.org/10.15560/8.3.349
Dulvy NK, Pacoureau N, Rigby CL, Pollom RA, Jabado RW, Ebert DA et al. 2021 Overfishing drives over one-third of all sharks and rays toward a global extinction crisis. Curr Biol. 2021; 31(21):4773-87. https://doi.org/10.1016/j.cub.2021.08.062
Eggertsen L, Luza AL, Cordeiro CAMM, Dambros C, Ferreira CEL, Floeter SR et al. Complexities of reef fisheries in Brazil: a retrospective and functional approach. Rev Fish Biol Fish. 2024; 34:511–38. https://doi.org/10.1007/s11160-023-09826-y
Ferreira CE, Gonçalves JEA, Coutinho R. Community structure of fishes and habitat complexity on a tropical rocky shore. Environ Biol Fishes. 2001; 61:353–69. https://doi.org/10.1023/A:1011609617330
Ferreira CEL, Floeter SR, Gasparini JL, Ferreira BP, Joyeux JC. Trophic structure patterns of Brazilian reef fishes: a latitudinal comparison. J Biogeogr. 2004; 31(7):1093–106. https://doi.org/10.1111/j.1365-2699.2004.01044.x
Figueiredo JL, Menezes NA. Manual de peixes marinhos do sudeste do Brasil: II. Teleostei (2). São Paulo: Museu de Zoologia da Universidade de São Paulo; 1980.
Floeter SR, Guimarães RZP, Rocha LA, Ferreira CEL, Rangel CA, Gasparini JL. Geographic variation in reef-fish assemblages along the Brazilian coast. Glob Ecol Biogeogr. 2001; 10(4):423–31. https://doi.org/10.1046/j.1466-822X.2001.00245.x
Floeter SR, Halpern BS, Ferreira CEL. Effects of fishing and protection on Brazilian reef fishes. Biol Conserv. 2006; 128(3):391–402. https://doi.org/10.1016/j.biocon.2005.10.005
Floeter SR, Krajewski JP, Fiuza TMJ, Rocha LA, Carvalho-Filho A. Peixes recifais brasileiros. Curitiba: CRV; 2023.
Freire KMF, Tubino RA, Monteiro-Neto C, Andrade-Tubino MF, Belruss CG, Tomás ARG et al. Brazilian recreational fisheries: current status, challenges and future direction. Fish Manag Ecol. 2016; 23:276–90. https://doi.org/10.1111/fme.12171
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2026. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Ghermandi A, Nunes PALD. A global map of coastal recreation values: results from a spatially explicit meta-analysis. Ecol Econ. 2013; 86:1–15. https://doi.org/10.1016/j.ecolecon.2012.11.006
Gibran FZ, Moura RL. The structure of rocky reef fish assemblages across a nearshore to coastal islands’ gradient in southeastern Brazil. Neotrop Ichthyol. 2012; 10(2):369–82. https://doi.org/10.1590/S1679-62252012005000013
Giglio VJ, Moura RL, Gibran FZ, Rossi LC, Banzato BM, Corsso JT et al. Do managers and stakeholders have congruent perceptions on marine protected area management effectiveness? Ocean Coast Manag. 2019; 179:104865. https://doi.org/10.1016/j.ocecoaman.2019.104865
Giglio VJ, Pereira-Filho GH, Marconi M, Rolim FA, Motta FS. Stakeholders’ perceptions on environmental quality and threats to subtropical marine reserves. Reg Stud Mar Sci. 2022; 56:102664. https://doi.org/10.1016/j.rsma.2022.102664
Goetze JS, Jupiter SD, Langlois TJ, Wilson SK, Harvey ES, Bond T et al. Diver operated video most accurately detects the impacts of fishing within periodically harvested closures. J Exp Mar Biol Ecol. 2015; 462:74–82. https://doi.org/10.1016/j.jembe.2014.10.004
Gragnolati M, Rolim FA, Pereira-Filho GH, Athayde ACS, Ciotti AM, Motta FS. Vertical structure of reef fish assemblages and light penetration reveal new boundaries of mesophotic ecosystems in the subtropical Southwestern Atlantic. Mar Environ Res. 2024; 198:106527. https://doi.org/10.1016/j.marenvres.2024.106527
Grillo AC, Candido CF, Giglio VJ, Longo GO. Unusual high coral cover in a Southwestern Atlantic subtropical reef. Mar Biodivers. 2021; 51:77. https://doi.org/10.1007/s12526-021-01221-9
Hoff NT, Silbiger HLN, Dias JF. The ichthyofauna of the Alcatrazes archipelago (São Sebastião, São Paulo, Brazil). Taxa. 2023; 1:ad23105. Available from: https://www.cappadociapublishing.com/index.php/taxa/article/view/13
Holmlund CM, Hammer M. Ecosystem services generated by fish populations. Ecol Econ. 1999; 29:253–68. https://doi.org/10.1016/S0921-8009(99)00015-4
Katsuragawa M, Dias JF, Harari J, Namiki C, Zani-Teixeira ML. Patterns in larval fish assemblages under the influence of the Brazil current. Cont Shelf Res. 2014; 89:103–17. https://doi.org/10.1016/j.csr.2014.04.024
Instituto Chico Mendes de Conservação da Biodiversidade (IMBio). Livro Vermelho da Fauna Brasileira Ameaçada de Extinção. Volume VI – Peixes. Brasília: ICMBio/MMA; 2018.
International Union for Conservation of Nature (IUCN). The IUCN Red List of Threatened Species. Version 2021–2 [Internet]. Gland; 2021. Available from: https://www.iucnredlist.org
Longo GO, Hay ME, Ferreira CEL, Floeter SR. Trophic interactions across 61 degrees of latitude in the Western Atlantic. Glob Ecol Biogeogr. 2019; 28(2):107–17. https://doi.org/10.1111/geb.12806
Luiz Jr. OJ, Carvalho-Filho A, Ferreira CEL, Floeter SR, Gasparini JL, Sazima I. The reef fish assemblage of the Laje de Santos Marine State Park, Southwestern Atlantic: annotated checklist with comments on abundance, distribution, trophic structure, symbiotic associations, and conservation. Zootaxa. 2008; 1807(1):1–25. https://doi.org/10.11646/zootaxa.1807.1.1
Luza AL, Aued AW, Barneche DR, Dias MS, Ferreira CEL, Floeter SR et al. Functional diversity patterns of reef fish, corals and algae in the Brazilian biogeographical province. J Biogeogr. 2023; 50(6):1163–76. https://doi.org/10.1111/jbi.14599
Magris RA, Costa MDP, Ferreira CEL, Vilar CC, Joyeux JC, Creed JC et al. A blueprint for securing Brazil’s marine biodiversity and supporting the achievement of global conservation goals. Divers Distrib. 2021; 27(2):198–215. https://doi.org/10.1111/ddi.13183
Mallet D, Pelletier D. Underwater video techniques for observing coastal marine biodiversity: A review of sixty years of publications (1952–2012). Fish Res. 2014; 154:44–62. https://doi.org/10.1016/j.fishres.2014.01.019
Marconi M, Giglio VJ, Pereira Filho GH, Motta FS. Does quality of scuba diving experience vary according to the context and management regime of marine protected areas? Ocean Coast Manag. 2020; 194:105246. https://doi.org/10.1016/j.ocecoaman.2020.105246
Menezes NA, Figueiredo JL. Manual de peixes marinhos do sudeste do Brasil. IV. Teleostei (3). São Paulo: Museu de Zoologia da Universidade de São Paulo; 1980.
Menezes NA, Figueiredo JL. Manual de peixes marinhos do sudeste do Brasil. V. Teleostei (4). São Paulo: Museu de Zoologia da Universidade de São Paulo; 1985.
Minte-Vera CV, Moura RL, Francini-Filho RB. Nested sampling: an improved visual-census technique for studying reef fish assemblages. Mar Ecol Prog Ser. 2008; 367:283–93. https://doi.org/10.3354/meps07511
Ministério do Meio Ambiente (MMA). Portaria MMA nº 148, de 7 de junho de 2022. Altera os Anexos da Portaria nº 443, de 17 de dezembro de 2014, da Portaria nº 444, de 17 de dezembro de 2014, e da Portaria nº 445, de 17 de dezembro de 2014, referentes à atualização da Lista Nacional de Espécies Ameaçadas de Extinção. Brasília: Diário Oficial da União; 2022.
Ministério do Meio Ambiente (MMA). Portaria MMA nº 354, de 27 de janeiro de 2023. Revoga as Portarias MMA nº 299, de 13 de dezembro de 2022, e nº 300, de 13 de dezembro de 2022, e dá outras providências. Brasília: Diário Oficial da União; 2023.
Motta FS, Moura RL, Neves LM, Souza GRS, Gibran FZ, Francini CL et al. Effects of marine protected areas under different management regimes in a hot spot of biodiversity and cumulative impacts from SW Atlantic. Reg Stud Mar Sci. 2021; 47:101951. https://doi.org/10.1016/j.rsma.2021.101951
Motta FS, Freitas MO, Rolim FA, Abilhoa V, Pereira Filho GH. Direct evidence of a spawning aggregation of cubera snapper (Lutjanus cyanopterus) in southeastern Brazil and its management implications. Fish Res. 2022; 252:106339. https://doi.org/10.1016/j.fishres.2022.106339
Moura RL. Levantamento rápido das comunidades de peixes associados a fundos consolidados da Estação Ecológica dos Tupiniquins. ESEC Tupiniquins; 2002.
Moura RL, Francini-Filho RB, Menezes NA, Dutra GF, Cappel D, Comin EJ. Memorial descritivo do meio marinho da Ilha da Queimada Grande e proposta de ampliação e recategorização da ARIE. Conservation International Brasil Programa Marinho; 2003.
Niz WC, Laurino IR, de Freitas DM, Rolim FA, Motta FS, Pereira-Filho GH. Modeling risks in marine protected areas: mapping of habitats, biodiversity, and cultural ecosystem services in the southernmost Atlantic coral reef. JEM. 2023; 345:118855. https://doi.org/10.1016/j.jenvman.2023.118855
Pereira PHC, Moraes RL, Santos MVB, Lippi DL, Feitosa JLL, Pedrosa M. The influence of multiple factors upon reef fish abundance and species richness in a tropical coral complex. Ichthyol Res. 2014; 61:375–84. https://doi.org/10.1007/s10228-014-0409-8
Pereira-Filho GH, Mendes VR, Perry CT, Shintate GI, Niz WC, Sawakuchi AO et al. Growing at the limit: Reef growth sensitivity to climate and oceanographic changes in the South Western Atlantic. Glob Planet Change. 2021; 201:103479. https://doi.org/10.1016/j.gloplacha.2021.103479
Pereira-Filho GH, Shintate GSI, Kitahara MV, Moura RL, Amado-Filho GM, Bahia RG et al. The southernmost Atlantic coral reef is off the subtropical island of Queimada Grande (24°S), Brazil. Bull Mar Sci. 2019; 95(2):277–87. https://doi.org/10.5343/bms.2018.0056
Pinheiro HT, Rocha LA, Macieira RM, Carvalho-Filho A, Anderson AB, Bender MG et al. South-western Atlantic reef fishes: Zoogeographical patterns and ecological drivers reveal a secondary biodiversity centre in the Atlantic Ocean. Divers Distrib. 2018; 24(7):951–65. https://doi.org/10.1111/ddi.12729
Pivetta APD, Morais MM, Comin EJ. Caracterização preliminar quantitativa e qualitativa da estrutura das assembleias de peixes recifais da ilha da Queimada Grande – SP. Rev Ceciliana. 2012; 4:77–83.
Quimbayo JP, Silva FC, Mendes TC, Ferrari DS, Danielski SL, Bender MG et al. Life-history traits, geographical range, and conservation aspects of reef fishes from the Atlantic and Eastern Pacific. Ecology. 2021; 102(5):e03298. https://doi.org/10.1002/ecy.3298
Riofrío-Lazo M, Zetina-Rejón MJ, Vaca-Pita L, Murillo-Posada JC, Páez-Rosas D. Fish diversity patterns along coastal habitats of the southeastern Galapagos archipelago and their relationship with environmental variables. Sci Rep. 2022; 12(1):3604. https://doi.org/10.1038/s41598-022-07601-w
Rolim FA, Langlois T, Rodrigues PFC, Bond T, Motta FS, Neves LM et al. Network of small no-take marine reserves reveals greater abundance and body size of fisheries target species. PLoS ONE. 2019; 14(1):e0204970. https://doi.org/10.1371/journal.pone.0204970
Rolim FA, Rodrigues PFC, Gadig OBF. Peixes de recifes rochosos, estação ecológica de Tupinambás, São Paulo. São Paulo: Anolis Books; 2017.
Santos PRS, Klafke K, Gadig OBF, Garrone-Neto D. Retrieving records of a rare and threatened shark in a mosaic of marine-protected areas of southeastern Brazil. J Fish Biol. 2023; 102(5):1256–60. https://doi.org/10.1111/jfb.15357
Souza GRS, Gadig OBF, Motta FDS, Moura RLD, Francini-Filho RB, Garrone-Neto D. Reef fishes of the Anchieta Island State Park, Southwestern Atlantic, Brazil. Biota Neotrop. 2018; 18:e20170380. https://doi.org/10.1590/1676-0611-BN-2017-0380
Steenbergen DJ. The role of tourism in addressing illegal fishing: the case of a dive operator in Indonesia. CSEA. 2013; 35(2):188–214. https://doi.org/10.1355/cs35-2c
Tratalos JA, Austin TJ. Impacts of recreational SCUBA diving on coral communities of the Caribbean island of Grand Cayman. Biol Conserv. 2001; 102:67–75. https://doi.org/10.1016/S0006-3207(01)00085-4
Authors
![]() Ana Clara S. Athayde1,
Ana Clara S. Athayde1, ![]() Fernanda A. Rolim1,
Fernanda A. Rolim1, ![]() Otto B. F. Gadig2,
Otto B. F. Gadig2, ![]() Caio R. Pimentel1,
Caio R. Pimentel1, ![]() Rafael R. Munhoz1,
Rafael R. Munhoz1, ![]() Maisha Gragnolati1,
Maisha Gragnolati1, ![]() Fernando Z. Gibran3,
Fernando Z. Gibran3, ![]() Leonardo M. Neves4,
Leonardo M. Neves4, ![]() Domingos Garrone-Neto5,
Domingos Garrone-Neto5, ![]() Gabriel R. S. Souza5,
Gabriel R. S. Souza5, ![]() Luiza D. Chelotti1,
Luiza D. Chelotti1, ![]() Guilherme H. Pereira‑Filho1 and
Guilherme H. Pereira‑Filho1 and ![]() Fábio S. Motta1
Fábio S. Motta1 ![]()
[1] Laboratório de Ecologia e Conservação Marinha, Instituto do Mar, Universidade Federal de São Paulo (UNIFESP), Av. Dr. Carvalho de Mendonça, 144, Encruzilhada, 11070-100, Santos, SP, Brazil. (ACSA) anaclarathy@gmail.com, (FAR) fernandarolim2@gmail.com, (CRP) caiopimentelr@gmail.com, (RRM) raro.munhoz@gmail.com, (MG) mgragnolatifernandes@gmail.com, (LDC) luizadchelotti@gmail.com, (GHPF) pereira.filho@unifesp.br, (FSM) fmotta@unifesp.br (corresponding author).
[2] ELASMOBRASIL – Laboratório de Pesquisa em Elasmobrânquios, Instituto de Biociências, Universidade Estadual Paulista (UNESP), Campus do Litoral Paulista, Praça Infante Dom Henrique, s/n, Parque Bitaru, 11330-900, São Vicente, SP, Brazil. (OBFG) otto.gadig@unesp.br.
[3] Centro de Ciências Naturais e Humanas, Universidade Federal do ABC, Alameda da Universidade, s/n, Bairro Anchieta, 09606 045, São Bernardo do Campo, SP, Brazil. (FZG) fernando.gibran@ufabc.edu.br.
[4] Laboratório de Ecologia Marinha e Costeira, Universidade Federal Rural do Rio de Janeiro, BR-465, km 7, 23897-000, Seropédica, RJ, Brazil. (LMV) leonardomneves@gmail.com.
[5] Laboratório de Ictiologia e Conservação de Peixes Neotropicais, Faculdade de Ciências Agrárias do Vale do Ribeira, Universidade Estadual Paulista (UNESP), Campus Registro – Av. Saburo Kameyama, 375, Estrada Municipal RGT-265, Bairro Carapiranga, 11900-000, Registro, SP, Brazil. (DGN) domingos.garrone-neto@unesp.br, (GRSS) gabrielraposo.souza@gmail.com.
Authors’ Contribution 

Ana Clara S. Athayde: Conceptualization, Formal analysis, Investigation, Writing-original draft.
Fernanda A. Rolim: Investigation, Writing-review and editing.
Otto B. F. Gadig: Investigation, Writing-review and editing.
Caio R. Pimentel: Writing-review and editing.
Rafael R. Munhoz: Investigation, Writing-review and editing.
Maisha Gragnolati: Formal analysis, Investigation, Writing-review and editing.
Fernando Z. Gibran: Investigation, Writing-review and editing.
Leonardo M. Neves: Investigation, Writing-review and editing.
Domingos Garrone-Neto: Formal analysis, Investigation, Writing-review and editing.
Gabriel R. S. Souza: Investigation, Writing-review and editing.
Luiza D. Chelotti: Investigation, Writing-review and editing.
Guilherme H. Pereira‑Filho: Investigation, Writing-review and editing.
Fábio S. Motta: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Project administration, Supervision, Writing-original draft.
Ethical Statement
Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) and Fundação Florestal by research permits (SISBIO: 46206 and COTEC: 004.358/2020; 26108–009.754/2014).
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
The authors (ACSA, DGN, RRM, CRP, FAR) thank Petrobras S/A (Tripartite Agreement FapUnifesp-Unifesp-Petrobras number #23089.102938/2019–54) for the scholarships. Financial support was provided by FAPESP (Process numbers 2019/19423–5 and 2023/11845–3), Instituto Linha D’água and Fundação SOS Mata Atlântica. Finally, FSM, GHPF, OBFG acknowledge individual grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Supplementary Material
Supplementary material S1
Supplementary material S2
How to cite this article
Athayde ACS, Rolim FA, Gadig OBF, Pimentel CR, Munhoz RR, Gragnolati M, Gibran FZ, Neves LM, Garrone-Neto D, Souza GRS, Chelotti LD, Pereira‑Filho GH, Motta FS. High fish richness associated with the southernmost Atlantic coral reef and adjacencies: comments on conservation and management for sustainable use. Neotrop Ichthyol. 2026; 24(1):e250090. https://doi.org/10.1590/1982-0224-2025-0090
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted October 17, 2025
Accepted October 17, 2025
![]() Submitted May 13, 2025
Submitted May 13, 2025
![]() Epub March 23, 2026
Epub March 23, 2026