![]() Kevin T. Torgersen1
Kevin T. Torgersen1 ![]() ,
, ![]() Jessé M. Figueiredo-Filho1,
Jessé M. Figueiredo-Filho1, ![]() Eric B. Haddad1,
Eric B. Haddad1, ![]() Patricia Torres-Pineda2,
Patricia Torres-Pineda2, ![]() Hernán López-Fernández2 and
Hernán López-Fernández2 and ![]() James S. Albert1
James S. Albert1
PDF: Download Here | Supplementary: S1 S2 S3 | Cite this article
Associate Editor: ![]() William Crampton
William Crampton
Section Editor: ![]() William Crampton
William Crampton
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Um novo gênero é descrito para a espécie rara “Eigenmannia” guchereauae, originalmente descrita em Distocyclus, mas posteriormente transferida para Eigenmannia, apesar de sua morfologia ser incongruente com aquele gênero. A descrição é baseada em material recentemente coletado no alto rio Tapanahony no Suriname, parte da bacia do Maroni à qual a espécie é endêmica. Características osteológicas e merísticas, juntamente com análises filogenéticas multilocos (COI, CytB, Rag1), sustentam o reconhecimento desta linhagem como distinta e grupo-irmão de Archolaemus. O novo gênero parece morfologicamente mais similar a Archolaemus, mas é distinguido principalmente pela ausência de margem orbital livre. Este reconhecimento expande o número de gêneros de Sternopygidae para oito e destaca o Escudo das Guianas como um centro de endemismo de Gymnotiformes. Nossos resultados ressaltam a importância da exploração contínua de regiões pouco estudadas como o alto Tapanahony, que continua a revelar diversidade inesperada e complexidade biogeográfica em peixes elétricos neotropicais.
Palavras-chave: Água doce, Guiana Francesa, Suriname, Taxonomia.
Introduction
The Neotropical electric knifefishes of the order Gymnotiformes constitute a major component of the lowland freshwater fauna from Mexico to Argentina, with 277 valid species (Albert, 2001; Eschmeyer et al., 2025). These fishes exhibit remarkable regional species and phenotypic diversity across varied aquatic habitats, ranging from deep river channels, river rapids, floodplain lakes, swamps, mangrove estuaries, and terra firme streams (Albert, 2001; Crampton, 1996, 2011; Crampton et al., 2016; de Santana, Vari, 2010; Torgersen et al., 2023a; Torgersen et al.,2026), and often compose a large proportion of the vertebrate biomass in some habitats (e.g., large river channels; Crampton, 1996; Lundberg et al., 2013; Duarte et al., 2019). These fishes are unique among Neotropical fishes for their ability to generate and detect weak electric signals that aid in navigation, location of food, and communication (Albert, Crampton, 2005).
Despite substantial recent progress in documenting gymnotiform diversity, significant knowledge gaps persist in regions that remain logistically challenging to access. Among these, portions of the Guiana Shield stand out as particularly underexplored for gymnotiform fishes, despite the region’s complex geological and hydrological history. Addressing these gaps is essential not only for accurate species inventories but also for understanding broader patterns of diversification and endemism within Gymnotiformes.
Among the major lineages of Gymnotiformes, the family Sternopygidae Cope, 1871 is particularly diverse and ecologically widespread across both cis- and trans-Andean river basins, from Panama to Argentina. Currently, Sternopygidae includes seven extant genera with 64 recognized species distributed throughout the humid Neotropics, with species richness peaking in Greater Amazonia. The family also includes one extinct genus, †Humboldtichthys, from the Miocene of Bolivia (Albert, Fink, 2007). Within Sternopygidae, diversity is concentrated in the subfamily Eigenmanniinae Mago-Leccia, 1978, commonly known as the glass knifefishes (Tab. 1). This group accounts for most of the species diversity of the family, with 52 recognized species, 17 of which have been described in the past decade (Eschmeyer et al., 2025).
TABLE 1 | Genera of the subfamily Eigenmanniinae before the publication of this article, with taxonomic authority and species-level diversity for each.
Genus | Author | Valid species |
Archolaemus | Korringa, 1970 | 6 |
Distocyclus | Eigenmann & Allen, 1942 | 1 |
Eigenmannia | Jordan & Evermann, 1896 | 33 |
Japigny | Meunier, Jégu & Keith, 2011 | 1 |
Rhabdolichops | Eigenmann & Allen, 1942 | 10 |
Rhinosternarchus | Dutra, Peixoto, Abrahão, Wosiacki, Menezes & de Santana, 2021 | 1 |
The Guiana Shield in northern South America harbors a large but incompletely known fish diversity, with 1.168 species listed by Vari et al. (2009). Many additional species have been described since then, and even more are known to need scientific description (e.g., Mol, 2012, Mol et al., 2012; Alofs et al., 2014; Borstein et al., 2022; Taphorn et al., 2022; Torgersen, Albert, 2022). More recent estimates provide a minimum of 469 described species for Suriname (Mol, 2012), 659 for Guyana (Taphorn et al., 2022), and 416 for French Guiana (Le Bail et al., 2012), with all accounts highlighting that further field exploration and study of existing collections will reveal even more.
While not among the most diverse regions for gymnotiforms, the Guiana Shield is home to a growing number of known endemic gymnotiform taxa of significant interest in clarifying the systematics and biogeography of Neotropical freshwater fishes. The recently described genus Akawaio Maldonado-Ocampo, López-Fernández, Taphorn, Bernard, Crampton & Lovejoy, 2014, an endemic of the upper Mazaruni River in Guyana, represents the sister lineage to all other Hypopomidae (Maldonado-Ocampo et al., 2014). Likewise, the monotypic sternopygid Japigny kirschbaum Meunier, Jégu & Keith, 2011, described from the Approuage, Mana, and Maroni rivers in French Guiana (Meunier et al., 2011), is the sister lineage to all other species of the Eigenmanniinae (Dutra et al., 2021). In contrast, although the once monotypic gymnotid genus Electrophorus Linnaeus, 1776 was recently split into several additional species, the Guiana Shield endemic E. electricus Linnaeus, 1776 is not the sister taxon to its congeners (de Santana et al., 2019). Recent and ongoing exploration continues to reveal previously unrecognized species and genera across the Guiana Shield, underscoring the region’s role in preserving several deeply divergent and previously overlooked gymnotiform lineages.
The Tapanahony River is the largest tributary of the Maroni (known as Marowijne in Suriname), which is shared by Suriname and French Guiana, and is currently known to harbor at least 318 species of fish, with 28 of them considered endemic (Mol, 2012; Mol et al., 2012, 2025; Borstein et al., 2022). The upper reaches of the Tapanahony river originate on the northern slopes of the Toemoek-Hoemak (or Tumucunaque in Brazil) Mountains, which are thought to serve as headwater areas of connection with northern Amazonian tributaries, especially through the Jari river basin (Nijssen, 1970; Kullander, Nijssen, 1989; Cardoso, Montoya‐Burgos, 2009; Fisch-Muller et al., 2018; Lujan et al., 2018) as well as providing possible dispersal routes between the eastern and western lobes of the Guiana Shield (Kullander, Nijssen, 1989; Lehmberg et al., 2018). Despite its potential biogeographic relevance in understanding the history of Guiana Shield diversity and its possible relations to both the Amazon and other Guiana Shield basins, the farthest reaches of the upper Tapanahony remain incompletely explored, with the most recent sampling occurring at or below the Paloemeu River (Mol, Wan Tong You, 2013).
One taxonomically puzzling lineage of the Gymnotiformes from this region is the species “Eigenmannia” guchereauae (Meunier, Jegu & Keith, 2014), which has generated confusion regarding its proper generic assignment. This species is apparently rare and is known from only a handful of specimens in natural history collections. Originally described in 2014 based on six specimens collected in French Guiana, the species was initially assigned to Distocyclus Eigenmann & Allen, 1942 by the authors based on the presence of a conical snout with a terminal mouth (Meunier et al., 2014). The species was subsequently transferred to Eigenmannia Jordan & Evermann, 1896, as the putative sister taxon of E. oradens Dutra, Peixoto, de Santana & Wosiacki, 2018, by Dutra et al. (2021), based on their morphological examination. However, this reallocation has since been called into question by additional data derived from morphology and molecular phylogenetics, which we discuss here.
Additional specimens of “Eigenmannia” guchereauae were collected during a 2020 expedition by the University of Michigan Museum of Zoology and Conservation International Suriname in the upper Tapanahony upstream of the Blackawatra River (Borstein et al., 2022). Here, we describe a new genus for “Eigenmannia” guchereauae, based largely on these recently collected specimens and data provided in the original species description. This work contributes to a more complete understanding of diversity within Eigenmanniinae, restores the monophyly of Eigenmannia, and provides new insight into the evolutionary history of Sternopygidae.
Material and methods
Two specimens of “Eigenmannia” guchereauae were takenon March 4, 2020, at Jawi Sula (rapids 7; 02º59.684’N 55º51.809’W) in the upper Tapanahony River by H. López-Fernández, S. Borstein, O. Lucanus, and K. Gajapersad. The newly collected specimens were tissue-sampled in the field and then µCT-scanned and measured. Morphometric and meristic data collected from these specimens were supplemented by data from the original species description (Meunier et al., 2014). Specimens from the type-series, deposited at the Muséum national d’Histoire naturelle (MNHN), Paris, were not available for our examination. However, the morphometric and meristic values we observed from the specimens collected in the Tapanahony coincide with the species diagnosis and fall within the reported range of morphometric and meristic values reported by Meunier et al. (2014). Institutional abbreviations throughout the text and supplemental materials follow Sabaj, 2020.
The following measurements were recorded for each specimen following Torgersen et al. (2023b) and Peixoto et al. (2015): 1) length to the end of the anal fin (LEA) measured from the tip of the snout (anterior margin of upper jaw at mid-axis of body) to the extent of the last anal-fin ray; 2) anal-fin length (AFL), measured from the origin of the anal fin at the isthmus to the end of the fin towards the tail; 3) caudal appendage (CA), measured as the distance from the last anal-fin ray to the distal end of the caudal filament. Note: the CA in sternopygid fishes is often damaged, entirely missing, or in a variable state of regeneration. Therefore, the values reported here are not considered to have diagnostic value; 4) caudal appendage depth (CD) measured dorsal to ventral depth of caudal filament immediately posterior to insertion of last anal-fin ray; 5) caudal appendage width (CW), measured across caudal filament immediately posterior to insertion of last anal-fin ray; 6) body depth (BD), measured as a vertical distance from the origin of the anal fin to the dorsal body border; 7) body width (BW), measured as body width at the origin of the anal-fin; 8) head length (HL), measured from the posterior margin of the bony opercle to the tip of the snout; 9) postorbital head length (PO), measured from the posterior margin of the bony opercle to posterior rim of free orbital margin of eye; 10) preorbital head length (PR), measured from the anterior rim of the orbital free margin to tip of snout; 11) eye diameter (ED), measured as the horizontal distance between the anterior and posterior rims of the free orbital margin; 12) prepectoral distance (PP), measured from tip of snout to insertion of first pectoral-fin ray; 13) interorbital length (IO), measured between the dorsomedial margins of the free orbital margin; 14) inter-narial distance (NN), measured from the posterior margin of the anterior nares to the anterior margin of the posterior nares; 15) posterior nares to orbit (PNO), measured from posterior margin of posterior naris to anterior margin of orbit; 16) mouth width (MW), measured as the horizontal distance of the gape at the rictus; 17) branchial opening (BO) measured as the distance from the posterodorsal to the anteroventral extent of the skin fold of the branchial opening along the anterior margin; 18) head depth (HD), measured as the vertical distance at the nape to ventral body border with the lateral line held horizontal; 19) head width (HW) measured as the width at nape; 20) suborbital depth (SD), measured from ventral margin of orbit to ventral margin of head; 21) snout to anus (SA), measures from tip of snout to anterior margin of anus; 22) preanal-fin distance (PRA), measured from tip of snout to insertion of first anal-fin ray; 23) preanal distance (PA), measured from the origin of the anal fin to the posterior margin of anus; 24) pectoral-fin length (P1), measured from the dorsal border of fin base where it contacts the cleithrum to the tip of the longest ray. Morphometric data were standardized for size by reporting values as a percent of HL, except in HL%, BD%, BW%, CA%, and PRA%, which are reported as a percent of LEA.
Meristic counts follow Torgersen et al. (2023b) and include: 1) anal-fin rays (AFR); 2) pectoral-fin rays (P1R) including all branched and unbranched rays; 3) precaudal vertebrae (PCV) including the four vertebrae that compose the Weberian apparatus; 4) scales above the lateral line (SAL) counted along a vertical line at the end of the body cavity; 5) scales below the lateral line (SBL) from the same point as SAL to the base of the anal-fin pterygiophores; 6) scales over the pterygiophores (SOP) counted from the same point as SAL at the base of the anal-fin pterygiophores to the anal-fin ventral border. Tooth counts are reported unilaterally, for a single premaxilla or dentary, rather than as totals across paired elements.
DNA was extracted from muscle tissues or fin clips preserved in a high-salt buffer (0.26% 0.5 M EDTA, 5.2 pH) or 100% ethanol, using the DNeasy Blood and Tissue Kit (Qiagen) following the manufacturer’s instructions. Additionally, we added 5 μL of RNase A (100 mg/mL; Qiagen) during lysis. Partial fragment sequences for three molecular markers, two mitochondrial: cytochrome oxidase subunit 1 (COI) and cytochrome b (CytB); and one nuclear: recombination-activating gene 1 (Rag1) of eight voucher specimens were amplified by polymerase chain reaction (PCR) with the primers described in Tab. S1.
PCR reactions for DNA amplification were performed in a total volume of 25 μl with 12.5 μl GoTaq® DNA Polymerase (Promega Corporation), 0.25 μl each of reverse and forward primers at 10 μM, and 12 μl of DNA + ddH2O. PCR cycles were performed following Oliveira et al. (2011). The PCR reactions consisted of 35 cycles of denaturation at 95ºC for 30 s, annealing at 50-54ºC for 45-60 s (according to primers; see Tab. S1), and extension at 72ºC for 90 s. Nested-PCR was used to amplify Rag1, with same PCR conditions for both cycles. The PCR product was purified with ExoSAP-IT™ PCR Product Cleanup (Applied Biosystems) following manufacturer’s instructions. Sanger sequencing was conducted by Eurofins Genomic LLC, Louisville, KY, USA.
We generated 20 specimen sequences and supplemented them with 31 sequences downloaded from GenBank for Sternopygidae (Tabs. S2, S3), including two mitochondrial markers (COI, CytB) and one nuclear marker (Rag1). Consensus sequences were obtained, manually edited, and trimmed in Geneious Prime v. 2024.0.7 (Kearse et al., 2012). Each gene was aligned independently using the MUSCLE algorithm (Edgar, 2004) under default parameters, and the alignments were then concatenated into a three-gene matrix. The final alignment included 32 sequences with 2,795 sites, of which 875 were distinct patterns, 623 parsimony-informative, 203 singleton sites, and 1,969 constant sites. Phylogenetic reconstructions were performed under Bayesian inference in MrBayes v. 3.2.7 (Ronquist et al., 2012). The best-fit model selected was TVM+F+I+G4 by PartitionFinder2 (Lanfear et al., 2017). Each run was comprised of 104 generations with sample frequency every 10 generations. Effective sample size values were inspected in Tracer v. 1.7.2 (Rambaut et al., 2018). 25% of the initial topologies were discarded in burn-in, and then, 25% of those remaining trees were summarized on a majority-rule consensus tree. Apteronotus bonapartii (Castelnau, 1855) was used as the outgroup, and the resulting tree with posterior probabilities was visualized and adjusted for presentation in FigTree v. 1.4.4 (http://tree.bio.ed.ac.uk/software/figtree/).
Micro-computed tomography (µCT) scans were made of the two specimens from the Tapanahony using a Bruker SkyScan1273 with settings optimized for the scanning of small vertebrates. Osteological observations were made from 3D renderings of the µCT scans in 3D Slicer (Fedorov et al., 2012). Precaudal vertebrae were counted from X-rays. Osteological descriptions presented below represent conditions in mature female specimens. The map of the geographic range of the new genus was generated in QGIS v. 3.38.3 (QGIS Development Team, 2025).
Results
Pararcholaemus, new genus
urn:lsid:zoobank.org:act:83521147-B850-45AF-BADF-F3B0E3EC9A13
(Figs. 1–4; Tab. 2)
Type-species. Pararcholaemus guchereauae (Meunier, Jegu & Keith, 2014), by monotypy.
Diagnosis. The new genus is unequivocally assigned to the subfamily Eigenmanniinae and diagnosed from all species of Sternopygus based on the following combination of characters: anal-fin rays branched, scapular foramen present, and the post-temporal fused with the supracleithrum. It further differs from Sternopygus by the lack of a free orbital margin, and from all other species of the Eigenmanniinae except Archolaemus and Eigenmannia oradens by having the teeth of the anteriormost row of the premaxilla attached only along their anterobasal margins (vs. anteriormost teeth completely attached to the premaxilla in Eigenmannia, Distocyclus, Japigny, Rhabdolichops, and Rhinosternarchus). The new genus differs from Archolaemus by the lack of a free orbital margin and by having proportionally larger parapophyses of the 2nd and 4th vertebrae of the Weberian apparatus (extending ventrally 2–3x the height of V1 vs. <1–1x the height of V1). Pararcholaemus differs from Eigenmannia, Japigny, and Rhabdolichops by having a longer head length (14.6–15.2% vs. 10.1–14.3%), and from Rhinosternarchus and Distocyclus in having longitudinal stripes extending across the body and tail. It further differs from Distocyclus, Rhabdolichops caviceps, and R. troscheli by having teeth attached to the ventral surface of the endopterygoid. It is also diagnosed from Japigny by the absence of 6–8 dark vertical pigment bars along the length of the body, and from Distocyclus in having a triangular snout (vs. conical snout) and hyaline anal fin (vs. darkly pigmented anal-fin margin).
Etymology. The generic name Pararcholaemus (masculine) is derived from the Greek prefix para– (παρά), meaning “beside” or “near,” combined with Archolaemus, the name of a related sternopygid genus stemming from archós (Greek ἀρχός), anus, and laimós (Greek λαιμός), throat, in reference to the position of the vent under the eye, a feature often highlighted in gymnotiform etymologies. The new genus name emphasizes the strong resemblance of this taxon to species of Archolaemus, from which it can be most readily distinguished by the absence of a free orbital margin.
Pararcholaemus guchereauae(Meunier, Jegu & Keith, 2014), new combination
Distocyclus guchereauaeMeunier et al., 2014:223–230 (original description). —Papa et al., 2020:13, tab. 1. (barcoding).
Eigenmannia guchereauae. —Dutra et al., 2021:3, 38–39, 42(assignment to Eigenmannia).
Eigenmannia sp. 2 aff. virescens.—Borstein et al., 2022:16 (lot UMMZ 252561 in species inventory).
Material examined. Suriname. UMMZ 252561, 250–266 mm TL (181–204 LEA), Sipaliwini, Jawi Sula (rapids 7), 02º59.684’N 55º51.809’W, 4 Mar 2020, H. López-Fernández, S. Borstein, O. Lucanus & K. Gajapersad. MHNG 2717.028 (photograph only), Sipaliwini, Wayu Camp, Paloemeu River, 03º11.933’N, 55º24.429’W, 28 Oct 2008, R. Covain, J. I. Montoya-Burgos, J. Mol & K. Wan Tong You.
Diagnosis. As for genus.
Description. Head and body in Fig. 1. Morphometric and meristic data in Tab. 2. Body elongate, laterally compressed, more laterally compressed in post-coelomic body region; body widest immediately behind head. Body covered with small cycloid scales except head and fins. Head long with blunt triangular snout; mouth small, slightly subterminal, lower jaw slightly shorter than upper. Eye large, covered by layer of skin. Anterior nares slightly tubular; posterior nares removed from eye approximately one eye diameter from edge of orbit. Dorsal and pelvic fins absent. Pectoral fins with 18–21 total rays, first two rays usually unbranched. Anal-fin origin at or near vertical with pectoral fin base; anal fin runs length of body, with 178–187 branched rays in specimens with undamaged tails. Tail as a slender caudal filament tapering to point with no anal fin. Dark stripe runs along lateral line to tail.
FIGURE 1| Head and body of Pararcholaemus guchereauae, UMMZ 252561, 204 mm LEA, gravid female. Note the lack of a free orbital margin. Scale bars = 1 cm.
TABLE 2 | Morphometric and meristic data for two specimens of Pararcholaemus guchereauae (UMMZ 252561) collected from the Tapanahony River, Suriname, supplemented with data from the original species description (Meunier et al., 2014). Abbreviations defined in Methods. TL, LEA, AFL, and CA given in mm, all others as proportions. Dashes represent missing data not reported in the original description.
| TL | LEA | AFL | CA |
Tapanahony specimens | 250–266 | 181–204 | 155–173 | 62–69 |
Type-series (from description) | 271–331 | 194–287 | 160–233 | 40–94 |
| CD% | CW% | BD% | BW% |
Tapanahony specimens | 10.7–10.8 | 5.2–6.1 | 15.8–16.0 | 6.2–6.5 |
Type-series (from description) | – | – | 16.1–16.5 | – |
| HL% | PO% | PR% | ED% |
Tapanahony specimens | 14.6–15.2 | 46.7–47.6 | 37.6–38.2 | 15.4–16.9 |
Type-series (from description) | 15.9–24.7 | – | – | 10.6–16.1 |
| PP% | IO% | NN% | PNO% |
Tapanahony specimens | 17.1–17.1 | 21.8–23.3 | 9.5–9.6 | 17.2–18.3 |
Type-series (from description) | 17.0–18.1 | 16.5–21.9 | 8.1–12.5 | 12.5–20.9 |
| MW% | BO% | HD% | HW% |
Tapanahony specimens | 21.5–21.9 | 36.4–40.8 | 73.5–80.0 | 52.3–53.2 |
Type-series (from description) | 15.3–30.0 | 30.5–41.6 | 61.8–72.0 | – |
| SD% | SA% | PRA% | PA% |
Tapanahony specimens | 32.3–40.0 | 55.4–64.2 | 19.0–19.3 | 68.7–72.7 |
Type-series (from description) | – | – | 16.5–18.9 | – |
| P1% | P1R | AFR | SAL |
Tapanahony specimens | 11.5–11.6 | 19–20 | 178–185 | 13–14 |
Type-series (from description) | 67.0–77.7 | 18–21 | 178–187 | – |
| SBL | SOP | PCV |
|
Tapanahony specimens | 7–7 | 11–12 | 13–13 |
|
Type-series (from description) | – | – | 13–14 |
|
Neurocranium in dorsal, lateral, and ventral views in Fig. 2A. Preorbital region of neurocranium dorsal margin straight and slightly decurved in the mesethmoid region. Neurocranium relatively well-ossified compared to other gymnotiforms, all bones of braincase ossified to their peripheral margins with little or no intervening cartilaginous plates, and little to no reticulations. Interorbital distance intermediate, sphenoid region of neurocranium broad, about as wide between anterior frontal processes as between posterior frontal processes (as in Archolaemus), with a single large opening in the supraorbital canals (as in all sternopygids). Supraorbital canal mostly fused to frontal. Lateral ethmoid cartilage well ossified, its ventral margin extending lateral to vomer, with a large, recurved process extending towards frontal (as in other Eigenmanniinae). Sphenotic lateral process does not extend to lateral margin of pterotic. Gap between parasphenoid, pterosphenoid, and orbitosphenoid about as deep as basioccipital articulating surface with first vertebrae. Pterosphenoid does not contact parasphenoid, while orbitosphenoid does contact. Bones of ethmoid region (e.g., lateral ethmoid, ventral ethmoid) relatively well-ossified as compared to other sternopygids. Anterior and posterior cranial fontanelles large, separated by narrow interorbital bridge, posterior fontanelle extending posterior to about vertical with anterior margin of exoccipital. Antorbital and postorbital processes of frontal robust, tapering to distal tips. Parasphenoid ventral margin straight in lateral view with little change in angle of ventral margin anterior and posterior to lateral parasphenoid wings. Parasphenoid anterior portion wider than posterior portion; parasphenoid ventral margin flat, without longitudinal ridges on lateral margins. Supraoccipital crest relatively small compared to Sternopygus, similar to other Eigenmanniinae.
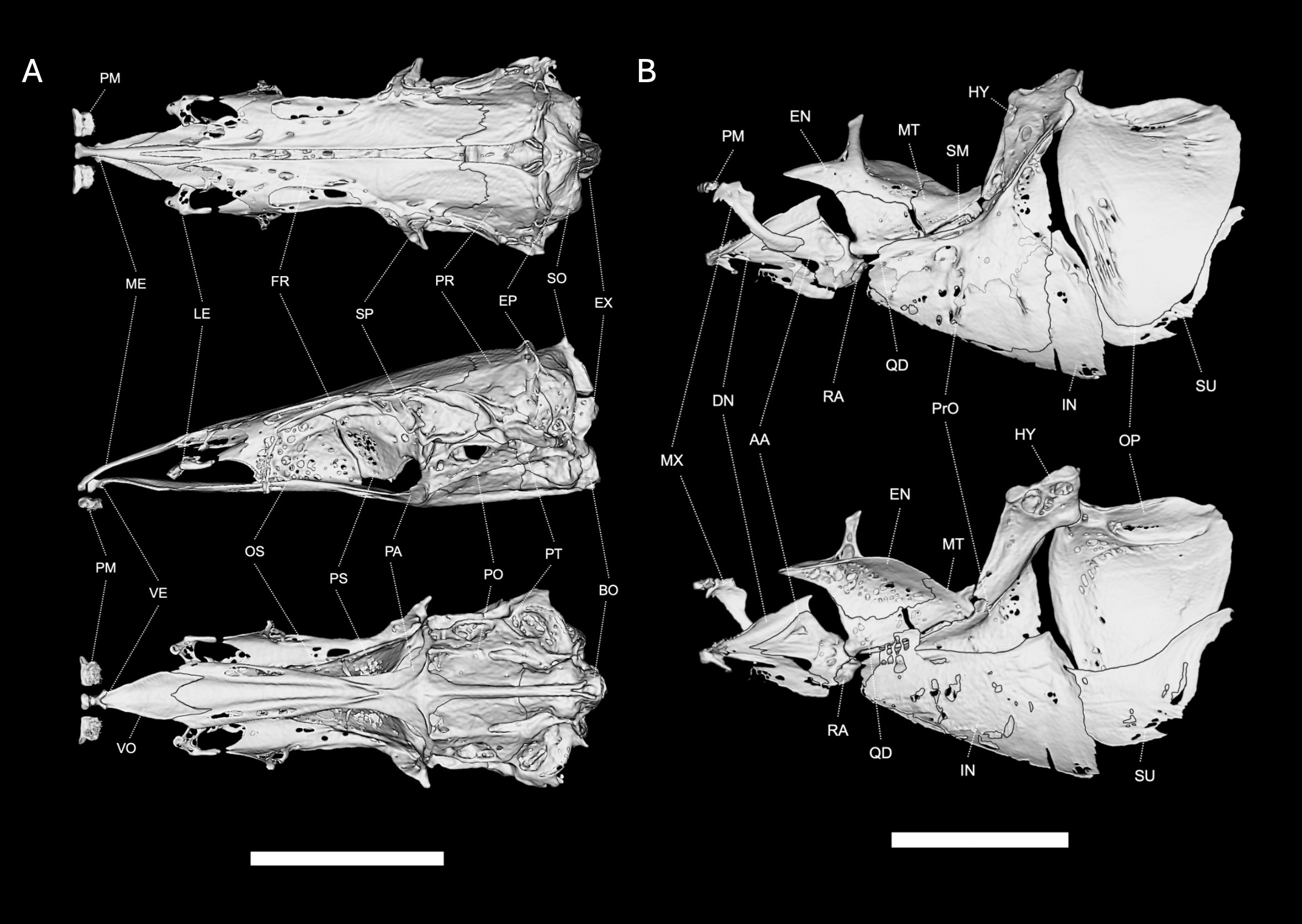
FIGURE 2| Neurocranium and suspensorium of Pararcholaemus guchereauae, UMMZ 252561, 204 mm LEA, based on µCT imaging. A. Neurocranium in dorsal, lateral and ventral views Abbreviations: PM = premaxilla, ME = mesethmoid, LE = lateral ethmoid, FR = frontal, PR = parietal, EP = epioccipital, BO = basioccipital, SO = supraoccipital, EX = exoccipital, VE = ventral ethmoid, VO = vomer, OS = orbitosphenoid, PT = pterotic, PO = prootic, PA = parasphenoid, PS = pterosphenoid. B. Suspensorium in lateral and medial views. Abbreviations: PM = premaxilla, MX = maxilla, DN = dentary, AA = angulo-articular, RA = retroarticular, EN = endopterygoid, MT = metapterygoid, QD = quadrate, SM = symplectic, PrO = preopercle, IN = interopercle, HY = hyomandibula, OP = opercle, SU = subopercle. Scale bars = 1 cm.
Oral jaws, suspensorium, and opercular series presented in Fig. 2B. Mouth terminal to slightly inferior, anterior margin of mesethmoid extending slightly anterior to anterior margin of dentary; premaxilla roughly cuboidal, slightly wider than long, with large anterolateral process, and with 26–43 large, straight conical teeth in five rows. Maxilla toothless, with a relatively broad and straight main axis for most of its length, and a broad, decurved distal tip, and a small decurved process on the anterior dorsal margin. No ossified anterior shelf as seen in many species of Apteronotidae. Dentary of intermediate length, oral margin about as long as distance from mandibular symphysis to dentary-retroarticular articulation, with a recurved process at the mental symphysis. Dentary with 26–52 small, conical teeth on the anterior portion arranged in three rows near mental symphysis and tapering to a single row posteriorly. Mandibular portion of preopercular-mandibular canal incompletely ossified with no ovoid foramina visible from lateral aspect (as in other Eigenmanniinae and contrasted with more complete ossification in Sternopygus). Suspensorium foreshortened as in other Eigenmanniinae (except Archolaemus) vs more elongate in Sternopygus, with the foreshortening occurring in the palatoquadrate region. All elements of suspensorium well-ossified with some pits and small holes in both dermal and endochondral bones. Palatine unossified cartilage contacting dorsoposterior margin of maxilla. Endopterygoid broad, with about 10–15 pointed teeth on anterior margin of medial surface, medial margin not contacting other contralateral endopterygoid at midline of palate, endopterygoid ascending process robustly ossified, tapering dorsally, connected by thin tendon to ventral surface of frontal. Hyomandibula with a diagnostic shape, size, and orientation for the new genus, relatively large as in other Eigenmanniinae, with a long and tapering descending limb, slightly more gracile than Sternopygus, its descending margin more tapered and more obliquely oriented from vertical, with large foramina on dorsomedial surface. Symplectic well ossified. Quadrate well-ossified and abutting endopterygoid but with cartilaginous margin with metapterygoid. Metapterygoid well ossified. Preopercle well ossified, posterior, ventral, and anterior margins approximating right angle, anterior margin ossified. Opercle well ossified with a relatively flat dorsal margin. Anterior articulating process large and horn-shaped, lateral opercular surface mostly smooth with ridges and small lacunae in anteroventral region.
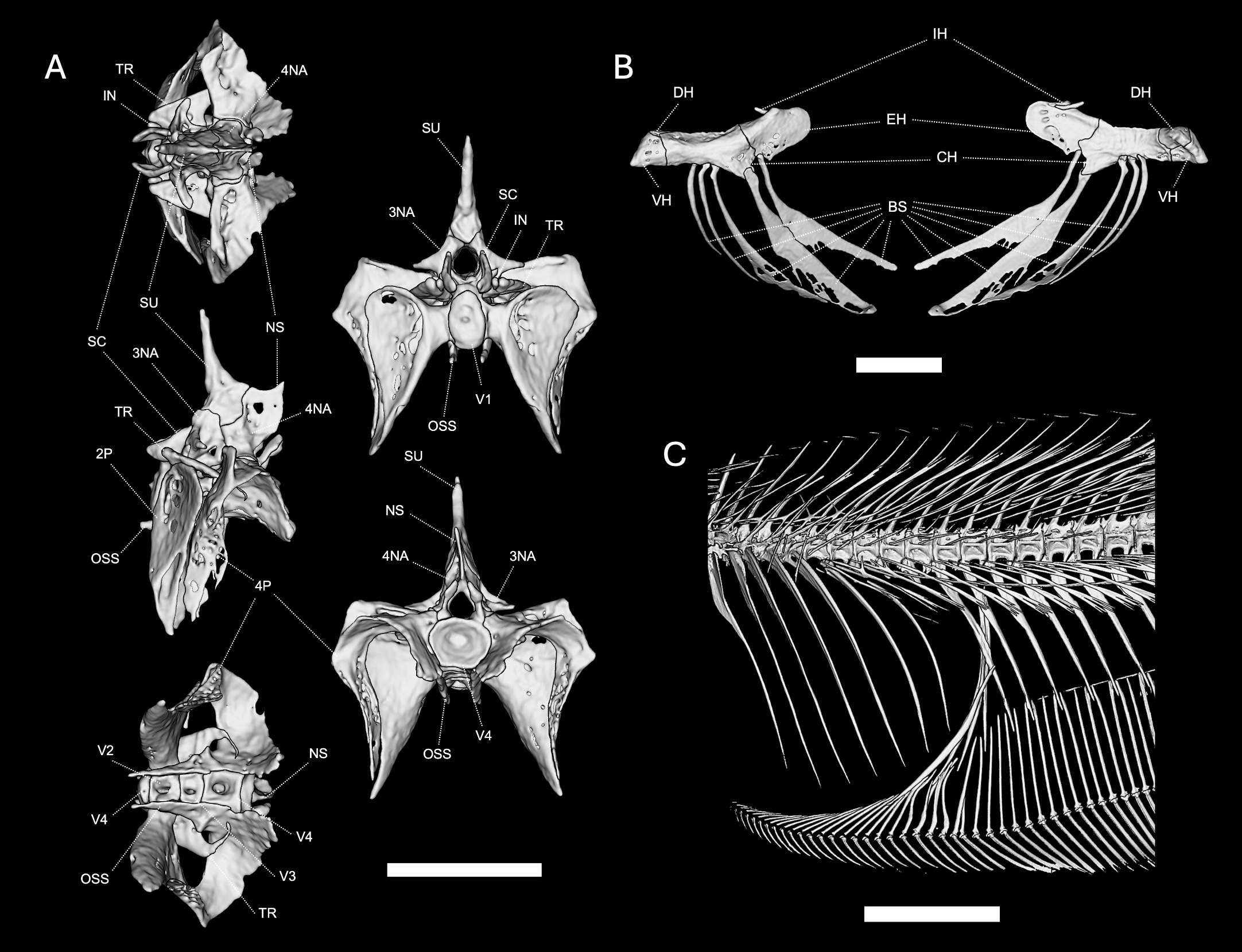
Weberian Apparatus, composed of first four vertebrae and accessory articulating elements, presented in Fig. 3A. Overall shape and configuration similar to other Eigenmanniinae in being axially compressed with large parapophyses of the second and fourth vertebrae involved in sound transduction from the swim bladder. Scaphium roughly triangular in lateral aspect. Supraneural slender and straight, pointing dorsally. Neural spine of 4th neural arch small and blunt. Os suspensorium robust.
FIGURE 3| Weberian apparatus, branchiostegal apparatus, and body cavity of Pararcholaemus guchereauae, UMMZ 252561, 204 mm LEA, based on µCT imaging. A. Weberian apparatus in multiple views. Abbreviations: V1 = vertebrae 1, V2 = vertebrae 2, V3 = vertebrae 3, V4 = vertebrae 4, TR = tripus, SN = supraneural, SC = scaphium, NS = neural spine, IN = intercalarium, 3NA = 3rd neural arch, 4NA = 4th neural arch, 2P = 2nd parapophysis, 4P = 4th parapophysis, OSS = os suspensorium. Scale bar = 5 mm. B. Branchiostegal apparatus. Abbreviations: IH = interhyal, BS = branchiostegal rays, EH = epihyal, CH = ceratohyal, DH = dorsal hypohyal, VH = ventral hypohyal. Scale bar = 5 mm. C. Displaced hemal spines at the end of the body cavity. Scale bar = 1 cm.
Hyoid Arch presented in Fig. 3B.Dorsal and ventral hypohyals partially fused. Ceratohyal larger than posterior ceratohyal with a broad midsection and ossified dorsal and ventral margins. First two branchiostegal rays narrow and spine-like, with subsequent three rays more paddle-like and incompletely ossified. Interhyal independently ossified.
Body cavity presented in Fig. 3C. One thin anterior DHS and three thin posterior DHS at the end of the body cavity; 13–14 PCV.
Coloration in life. In life, specimens of Pararcholaemus are light bluish-gray in overall appearance with paler coloration below the lateral line (Fig. 4). Anal and pectoral fins hyaline. Black mark at base of each anal-fin pterygiophore. Darker pigmentation along body at lateral line.
FIGURE 4| Live specimen of Pararcholaemus guchereauae collected and photographed at Weyu (Wayu) Rapid, Paloemeu River, Suriname, 03º11.933’N 55º24.429’W, by Karsten Schönherr. Specimen not preserved. Note that the dark spots visible on the dorsum of the specimen are nematode cysts, a condition we have observed in other gymnotiform specimens from the region.
Coloration in alcohol. In preservative, specimens of Pararcholaemus are yellow throughout the body (Fig. 1). Head and dorsum are more darkly pigmented and brown in color. Anal and pectoral fins hyaline. Black mark at base of each anal-fin pterygiophore. Darker pigmentation along body at lateral line.
Sexual dimorphism. Both specimens examined from the Tapanahony River were determined to be gravid females. A photograph of a putative male specimen (MHNG 2717.028) from the Paloemeu River (tributary of Tapanahony) shows a darkly pigmented head, a condition also reported in sexually dimorphic males of some species of Archolaemus (Vari et al., 2012).
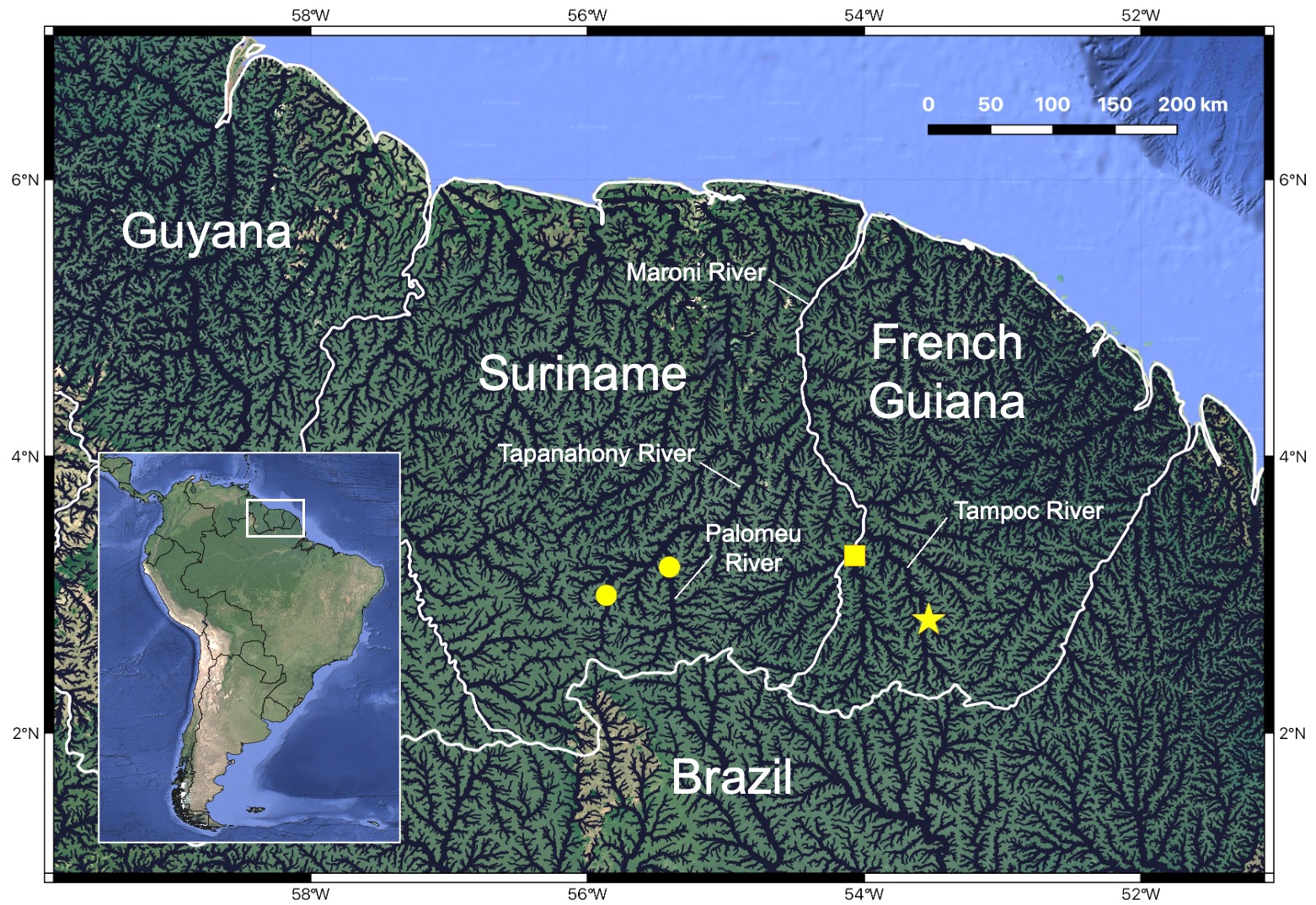
Geographical distribution. Pararcholaemus guchereauae is known from the upper Maroni River basin of French Guiana and Suriname, where it has been collected in the Litany, Paloemeu, Tampoc, and Tapanahony rivers (Fig. 5).
FIGURE 5| The known geographic distribution of Pararcholaemus guchereauae is indicated by yellow symbols. The star indicates the type-locality of the species. The square indicates the collection locality of paratypes not collected with the holotype. The circles indicate new records of this species from the upper Tapanahony and Paloemeu rivers, which expands the species’ known range into Suriname.
Ecological notes. Two specimens were collected from sandbanks and backwaters proximal to the rapids at the bottom of Jawi Sula in the upper Tapanahony (Fig. 6), suggesting a similar ecology to species of Archolaemus that are known to inhabit rapids and fast-flowing rivers in Brazil (Schwassmann, Carvalho, 1985; Vari et al., 2012). Water quality measurements taken at this locality at the time of collection include: Temperature = 26.3 ºC; Dissolved Oxygen =94%/7.58 mg/L; Conductivity = 17.6uS/cm; Total Dissolved Solids = 11.13 mg/L; pH ~6.4; Turbidity = 3.3 FNU; Total Suspended Solids = 0 mg/L; and Current Velocity 0-1.6 m/s.
FIGURE 6| Photograph of the collection locality of the specimens from the upper Tapanahony River at Jawi Sula, rapids 7; 02º59.684’N 55º51.809’W. Photo: H. López-Fernández.
Conservation status. Pararcholaemus guchereauae has been evaluated on the IUCN Red List under the name Distocyclus guchereauae and is currently listed as Near Threatened (NT) (Torgersen, 2023).
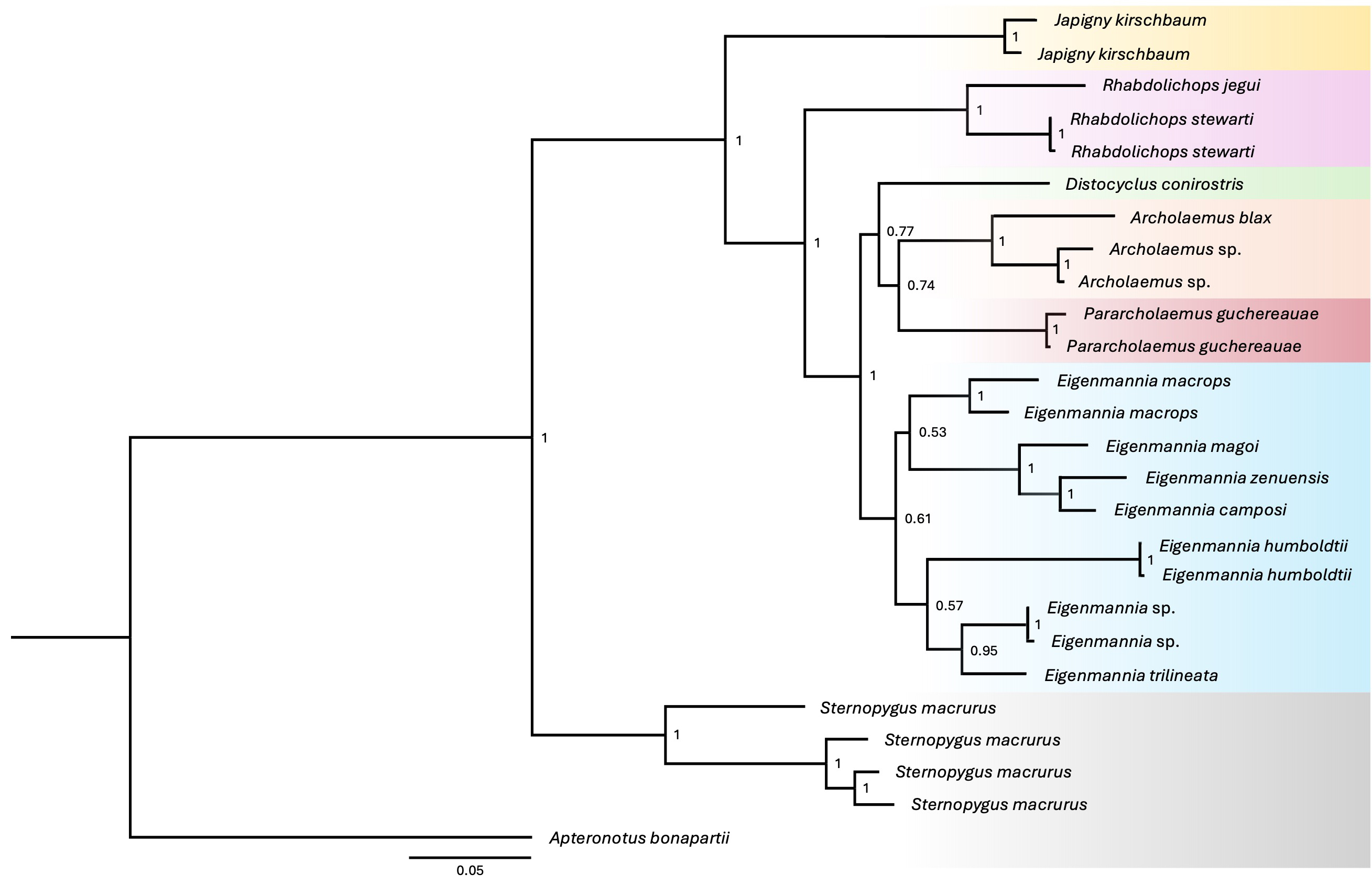
Phylogenetic relationships of Eigenmanniinae. Our multilocus Bayesian phylogenetic analyses, based on concatenated sequences of COI, CytB, and Rag1, recover Eigenmanniinae as a monophyletic group, with Sternopygus as its sister lineage (Fig. 7). At the genus level, the resulting topology is broadly concordant with the recent morphology-based phylogeny of Dutra et al. (2021). Within Eigenmanniinae, Japigny kirschbaum is resolved as the earliest diverging lineage, forming the sister group to all other genera. Among the principal clades, Rhabdolichops is recovered as sister to a clade comprising Archolaemus, Distocyclus, Eigenmannia, and Pararcholaemus guchereauae. Within this clade, Pararcholaemus guchereauae is sister to Archolaemus, and together they form the sister group to Distocyclus, which in turn is recovered as sister to Eigenmannia.
FIGURE 7| Bayesian phylogeny of Sternopygidae based on concatenated sequences of COI, CytB, and Rag1. Posterior probabilities are shown at each node.
Discussion
Taxonomic justification and phylogenetic position. The recognition of Pararcholaemus as a distinct genus represents a refinement in our understanding of eigenmanniine diversity and phylogenetic relationships. Originally described as Distocyclus guchereauae by Meunier et al. (2014) based on the possession of a conical snout with a terminal mouth, the species was subsequently transferred to Eigenmannia by Dutra et al. (2021) as the putative sister taxon of E. oradens. However, our combined morphological and molecular analyses reveal that this species occupies a unique phylogenetic position as the sister lineage to Archolaemus, together forming a clade that is sister to Distocyclus. Molecular support for the monophyly of Pararcholaemus in our phylogeny is moderate (PP = 0.74; Fig. 7). Consequently, the phylogenetic evidence should be interpreted cautiously, particularly given the limited number of genes and taxa included in this analysis.
Despite these limitations, the distinctive combination of salient morphological characters supports recognition at the generic level. These include the absence of a free orbital margin in adults, and teeth of the anteriormost premaxillary row attached only along their anterobasal margins (shared only with Archolaemus and E. oradens among eigenmanniines). Placement in Archolaemus at present would necessitate expansion of that genus’s diagnosis to accommodate a species lacking the free orbital margin, which is a defining feature of adult Archolaemus. We consider recognition of morphologically diagnosable lineages as distinct genera to be more conservative than lumping taxa based on phylogenetic hypotheses derived from limited molecular datasets. The molecular data serve as corroborative evidence for the distinctiveness of this lineage; however, erection of Pararcholaemus is predicated principally on the unique combination of morphological characters that readily diagnose it from all other genera of Eigenmanniinae.
Guiana Shield endemism. Pararcholaemus is one of two sternopygid genera endemic to waters draining the Guiana Shield (along with Japigny kirschbaum), and one of five gymnotiform genera from the region, including the hypopomids Akawaio penak, Hypopomus artedi Kaup, 1856, and Racenisia fimbriipinna Mago-Leccia, 1994. Vari, Ferraris (2009) report a total of 1,168 fish species in the Guiana Shield, of which about 700 species (60%) are endemic, and represent 376 genera, of which 10 genera (2.6%) are endemic. These taxa include the crenuchid Skiotocharax Presswell, Weitzman & Bergquist, 2000, the characid Acanthocharax Eigenmann, 1912, the loricariids Corymbophanes Eigenmann, 1909 and Yaluwak Lujan, Armbruster & Werneke, 2020, and the cichlids Guianacara Kullander & Nijssen, 1989 and Mazarunia Kullander, 1990 (López-Fernández et al., 2012; Lemopoulos, Covain, 2019; Taphorn et al., 2022; Morgan, 2023). These endemic genera emphasize the long-standing isolation and persistent endemism characteristic of many South American fish lineages on the Guiana Shield (Alofs et al., 2014; Dagosta, de Pinna, 2019; Albert et al., 2020; Borstein et al., 2022; Torgersen, Albert, 2022; Akin et al., 2024; Conde‐Saldaña et al., 2025).
Limitations of our phylogeny. A phylogeny estimated using only three genes and limited taxon sampling faces several inherent limitations in both accuracy and interpretability. With such a small genetic dataset, the resulting tree may not capture the full evolutionary signal, leaving it vulnerable to stochastic error, incomplete lineage sorting, and biases introduced by gene-specific evolutionary histories. Different genes can evolve at different rates or be subject to selection pressures, meaning that relying on just three markers may produce a tree that reflects gene histories rather than the true species history. Limited taxon sampling further compounds these problems by reducing the ability to break up long branches, detect homoplasy, or resolve rapid radiations, often leading to misleading topologies or inflated support values. For these reasons, our phylogeny should only be viewed as a preliminary hypothesis. Nevertheless, when combined with morphometric data, it provides additional support for the recognition of a new endemic genus until more comprehensive genomic analyses can be completed.
Comparative material examined. Brazil: Archolaemus blax:INPA 5064, 1, 380 mm TL. MCP 15897, 1, 252 mm TL. Archolaemus janeae:ANSP 194705, 2, 214.0–252 mm TL. ANSP 198161, 1, 229 mm TL. Distocyclus conirostris: AMNH 221185, 3, 164–222 mm TL. USNM 301988, 4, 192–282 mm TL. Eigenmannia desantanai: NUP 3470, 3, 175–188 mm TL, paratypes. Sternopygus branco: MCP 32246, 3, 393–440 mm TL, paratypes. Guyana: Archolaemus ferreirai: ROM 96107, 7, 163–320 mm TL. Eigenmannia macrops: BMNH 1897.8.6.1, holotype, 227 mm TL. Japigny kirschbaum:BMNH 1972.10.17.519, 1, 173.0 mm TL. FMNH 50185, 5, 93.0–182.0 mm TL. FMNH 94511, 1, 162.0 mm TL. FMNH 94512, 1, 158.0 mm TL. Rhabdolichops sp.: ROM 97152, 5, 220–252 mm TL. Paraguay: Eigenmannia virescens: NRM 32532, 2, 246–260 mm TL. Suriname: Rhabdolichops jegui: ROM 100958, 7, 155–321 mm TL. Sternopygus sabaji: ANSP 208090, holotype, 374.0 mm TL. ANSP 189018, 17 paratypes, 46.0–356.0 mm TL. FMNH 146152, 7 paratypes, 98.0–372.0 mm TL. Venezuela: Eigenmannia nigra: ANSP 162130, 2 paratypes, 296–321 mm TL. Rhinosternarchus goajira: USNM 121596, holotype, 503 mm TL. USNM 121597, 1 paratype, 423 mm TL. Sternopygus astrabes: ANSP 162128, 1 paratype, 104.0 mm TL. ANSP 162663, 4, 75.0–237.0 mm TL. INHS 61503, 10, 104.0–226.0 mm TL. Sternopygus macrurus: ANSP 162298, 6, 143–403 mm TL.
Acknowledgments
We thank Karsten Schönherr for providing a photograph of a living specimen collected in the Paloemeu River, Suriname, and Guilherme Dutra for sharing a photograph of the single specimen in lot MHNG 2717.028. We also thank Kory Evans (Rice University) for allowing us to CT scan specimens at his lab and Ryan Hou for his support in lab work at the Biodiversity Lab (BDL) at the University of Michigan. We thank Claudio Oliveira and Daniel Akin for assistance in uploading the sequence files to NCBI. HLF thanks K. Gajapersad, S. Borstein, and O. Lucanus for their contributions to collecting the specimens described herein, and Conservation International Suriname, the Amazon Conservation Team, and the community of Pelelu Tepu for their support in the field.
References
Akin DR, Weyand CA, Buehler MD, Armbruster JW. Phylogenetics and biogeography of the Guiana Shield pencil catfishes, genus Trichomycterus. Ichthyol Herpetol. 2024; 112(4):600–14. https://doi.org/10.1643/i2024002
Albert JS. Species diversity and phylogenetic systematics of American knifefishes (Gymnotiformes, Teleostei). Misc Publ Mus Zool Univ Michigan. 2001; (190):1–129.
Albert JS, Crampton WGR. Diversity and phylogeny of Neotropical electric fishes (Gymnotiformes). In: Bullock TH, Hopkins CD, Popper AN, Fay RR, editors. Electroreception. New York: Springer; 2005. p.360–409. https://doi.org/10.1007/0-387-28275-0_13
Albert JS, Fink WL. Phylogenetic relationships of fossil Neotropical electric fishes (Osteichthyes: Gymnotiformes) from the Upper Miocene of Bolivia. J Vertebr Paleontol. 2007; 27(1):17–25. https://doi.org/10.1671/0272-4634(2007)27[17:PROFNE]2.0.CO;2
Albert JS, Tagliacollo VA, Dagosta F. Diversification of Neotropical freshwater fishes. Annu Rev Ecol Evol Syst. 2020; 51(1):27–53. https://doi.org/10.1146/annurev-ecolsys-011620-031032
Alofs K, Liverpool E, Taphorn D, Bernard C, López-Fernández H. Mind the (information) gap: the importance of exploration and discovery for assessing conservation priorities for freshwater fish. Divers Distrib. 2014; 20(1):107–13. https://doi.org/10.1111/ddi.12127
Borstein SR, Lucanus O, Gajapersad K, Singer R, Mol JH, López-Fernández H. Fish diversity of the upper Tapanahony River, Suriname. Misc Publ Mus Zool Univ Michigan. 2022; (211):1–20. https://dx.doi.org/10.7302/4816
Cardoso YP, Montoya-Burgos JI. Unexpected diversity in the catfish Pseudancistrus brevispinis reveals dispersal routes in a Neotropical center of endemism: the Guyanas Region. Mol Ecol. 2009; 18(5):947–64. https://doi.org/10.1111/j.1365-294X.2008.04068.x
Conde-Saldaña CC, Melo BF, Roxo FF, Tagliacollo VA, Oliveira C, Albert JS. Landscape evolution drives continental diversification in Neotropical freshwater fishes of the family Erythrinidae (Teleostei, Characiformes). J Biogeogr. 2025; 52(6):e15129. https://doi.org/10.1111/jbi.15129
Crampton WGR. Gymnotiform fish: an important component of Amazonian fioodplain fish communities. J Fish Biol. 1996; 48(2):298–301. https://doi.org/10.1111/j.1095-8649.1996.tb01122.x
Crampton WGR. An ecological perspective on diversity and distributions. In: Albert JS, Reis RE, editors. Historical biogeography of neotropical freshwater fishes. California: University of California Press, Berkeley; 2011. p.165–89. https://doi.org/10.1525/california/9780520268685.003.0010
Crampton WGR, de Santana CD, Waddell JC, Lovejoy NR. A taxonomic revision of the Neotropical electric fish genus Brachyhypopomus (Ostariophysi: Gymnotiformes: Hypopomidae), with descriptions of 15 new species. Neotrop Ichthyol. 2016; 14(04):e150146. https://doi.org/10.1590/1982-0224-20150146
Dagosta FCP, de Pinna M. The fishes of the Amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 2019(431):1–163. https://doi.org/10.1206/0003-0090.431.1.1
Duarte C, Espírito-Santo HMV, Zuanon J, Rapp Py-Daniel L, Deus CP. What happens in the darkness? Seasonal variations in tropical benthic fish assemblages. Mar Freshw Res. 2019; 71(4):419–31. https://doi.org/10.1071/MF19038
Dutra GM, Peixoto LAW, Abrahão VP, Wosiacki WB, Menezes NA, de Santana CD. Morphology-based phylogeny of Eigenmanniinae Mago-Leccia, 1978 (Teleostei: Gymnotiformes: Sternopygidae), with a new classification. J Zool Syst Evol Res. 2021; 59(8):2010–59. https://doi.org/10.1111/jzs.12535
Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004; 32(5):1792–97. https://doi.org/10.1093/nar/gkh340
Eschmeyer WN, Fricke R, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Sciences; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, Pujol S et al. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging. 2012; 30(9):1323–41. https://doi.org/10.1016/j.mri.2012.05.001
Fisch-Muller S, Mol JH, Covain R. An integrative framework to reevaluate the Neotropical catfish genus Guyanancistrus (Siluriformes: Loricariidae) with particular emphasis on the Guyanancistrus brevispinis complex. PLoS ONE. 2018; 13(1):e0189789. https://doi.org/10.1371/journal.pone.0189789
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics. 2012; 28(12):1647–49. https://doi.org/10.1093/bioinformatics/bts199
Kullander SO, Nijssen H. The cichlids of Surinam: Teleostei, Labroidei. Leiden: Brill; 1989.
Lanfear R, Frandsen PB, Wright AM, Senfeld T, Calcott B. PartitionFinder 2: new methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol Biol Evol. 2017; 34(3):772–73. https://doi.org/10.1093/molbev/msw260
Le Bail PY, Covain R, Jégu M, Fisch-Muller S, Vigouroux R, Keith P. Updated checklist of the freshwater and estuarine fishes of French Guiana. Cybium. 2012; 36(1):293–319.
Lehmberg ES, Elbassiouny AA, Bloom DD, López-Fernández H, Crampton WGR, Lovejoy NR. Fish biogeography in the “Lost World” of the Guiana Shield: Phylogeography of the weakly electric knifefish Gymnotus carapo (Teleostei: Gymnotidae). J Biogeogr. 2018; 45(4):815–25. https://doi.org/10.1111/jbi.13177
Lemopoulos A, Covain R. Biogeography of the freshwater fishes of the Guianas using a partitioned parsimony analysis of endemicity with reappraisal of ecoregional boundaries. Cladistics. 2019; 35(1):106–24. https://doi.org/10.1111/cla.12341
López-Fernández H, Taphorn DC, Liverpool EA. Phylogenetic diagnosis and expanded description of the genus Mazarunia Kullander, 1990 (Teleostei: Cichlidae) from the upper Mazaruni River, Guyana, with description of two new species. Neotrop Ichthyol. 2012; 10(3):465–86. https://doi.org/10.1590/S1679-62252012000300001
Lujan NK, Armbruster JW, Lovejoy NR. Multilocus phylogeny, diagnosis and generic revision of the Guiana Shield endemic suckermouth armoured catfish tribe Lithoxini (Loricariidae: Hypostominae). Zool J Linn Soc. 2018;184(4):1169–86. https://doi.org/10.1093/zoolinnean/zly025
Lundberg JG, Cox Fernandes C, Campos-da-Paz R, Sullivan JP. Sternarchella calhamazon n. sp., the Amazon’s most abundant species of apteronotid electric fish, with a note on the taxonomic status of Sternarchus capanemae Steindachner, 1868 (Gymnotiformes: Apteronotidae). Proc Acad Nat Sci Phila. 2013; 162(1):157–73. https://doi.org/10.1635/053.162.0110
Maldonado-Ocampo JA, López-Fernández H, Taphorn DC, Bernard CR, Crampton WGR, Lovejoy NR. Akawaio penak, a new genus and species of Neotropical electric fish (Gymnotiformes, Hypopomidae) endemic to the upper Mazaruni River in the Guiana Shield. Zool Scr. 2014; 43(1):24–33. https://doi.org/10.1111/zsc.12029
Meunier FJ, Jégu M, Keith P. A new genus and species of Neotropical electric fish, Japigny kirschbaum (Gymnotiformes: Sternopygidae), from French Guiana. Cybium. 2011; 35(1):47–53.
Meunier FJ, Jégu M, Keith P. Distocyclus guchereauae a new species of Neotropical electric fish (Gymnotiformes: Sternopygidae) from French Guiana. Cybium. 2014; 38(3):223–30.
Mol JH. The freshwater fishes of Suriname. Leiden: Brill; 2012.
Mol JH, Covain R, Willink PW. Rivers of the North Atlantic. In: Graça MAS, Callisto M, Teixeira de Mello F, Rodríguez-Olarte D, editors. Rivers of South America. Amsterdam: Elsevier; 2025. p.153–211. https://doi.org/10.1016/B978-0-12-823429-7.00013-6
Mol JH, Vari RP, Covain R, Willink PW, Fisch-Muller S. Annotated checklist of the freshwater fishes of Suriname. Cybium. 2012; 36(1):263–92.
Mol JH, Wan Tong You K. Fishes of the Palumeu River, Suriname. In: Alonso LE, Larsen TH, editors. A rapid biological assessment of the upper Palumeu River watershed (Grensgebergte and Kasikasima) of Southeastern Suriname. RAP Bull Biol Assess 67. Arlington, VA: Conservation International; 2013. p.119–30. https://doi.org/10.1896/054.067.0116
Morgan T. Cichlids model the role of riverine connectivity in shaping the biogeography, diversification, and population structure of fishes in the Guiana Shield, South America. [PhD Dissertation] Ann Arbor, Michigan: University of Michigan; 2023. Available from: https://doi.org/10.7302/22998
Nijssen H. Revision of the Surinam catfishes of the genus Corydoras Lacépède, 1803 (Pisces: Siluriformes: Callichthyidae). Beaufortia. 1970; 18(230):1–75.
Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G et al. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol. 2011; 11:275. https://doi.org/10.1186/1471-2148-11-275
Papa Y, Le Bail PY, Covain R. Genetic landscape clustering of a large DNA barcoding dataset reveals shared patterns of genetic divergence among freshwater fishes of the Maroni Basin. Mol Ecol Resour. 2021; 21(6):2109–24. https://doi.org/10.1111/1755-0998.13402
Peixoto LAW, Dutra GM, Wosiacki WB. The electric glass knifefishes of the Eigenmannia trilineata species-group (Gymnotiformes: Sternopygidae): monophyly and description of seven new species. Zool J Linn Soc. 2015; 175(2):384–414. https://doi.org/10.1111/zoj.12274
QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project; 2025. Available from: https://www.qgis.org
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA. Posterior summarisation in Bayesian phylogenetics using Tracer 1.7. Syst Biol. 2018; 67(5):901–04. https://doi.org/10.1093/sysbio/syy032
Ronquist F, Teslenko M, Van der Mark P, Ayres DL, Darling A, Höhna S et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol. 2012; 61(3):539–42. https://doi.org/10.1093/sysbio/sys029
Sabaj MH. Codes for Natural History Collections in Ichthyology and Herpetology. Ichthyol Herpetol. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
de Santana CD, Crampton WGR, Dillman CB, Frederico RG, Sabaj MH, Covain R et al. Unexpected species diversity in electric eels with a description of the strongest living bioelectricity generator. Nat Commun. 2019; 10(4000):1–10. https://doi.org/10.1038/s41467-019-11690-z
de Santana CD, Vari RP. Electric fishes of the genus Sternarchorhynchus (Teleostei, Ostariophysi, Gymnotiformes); phylogenetic and revisionary studies. Zool J Linn Soc. 2010; 159(1):223–371. https://doi.org/10.1111/j.1096-3642.2009.00588.x
Schwassmann HO, Carvalho ML. Archolaemus blax Korringa (Pisces, Gymnotiformes, Sternopygidae); a redescription with notes on ecology. Spixiana. 1985; 8(3):231–40.
Taphorn DC, Liverpool E, Lujan NK, DoNascimiento C, Hemraj DD, Crampton WGR et al. Annotated checklist of the primarily freshwater fishes of Guyana. Proc Acad Nat Sci Phila. 2022; 168(1):1–95. https://doi.org/10.1635/053.168.0101
Torgersen KT. Distocyclus guchereauae. The IUCN Red List of Threatened Species. 2023:e.T177521398A177521402. https://doi.org/10.2305/IUCN.UK.2023-1.RLTS.T177521398A177521402.en
Torgersen KT, Albert JS. A new species of Sternopygus (Gymnotiformes: Sternopygidae) from the Atlantic coast of the Guiana Shield. Ichthyol Herpetol. 2022; 110(4):714–27. https://doi.org/10.1643/i2022013
Torgersen KT, Bogotá-Gregory JD, Acosta-Santos A, Potosí-Chuña JJ, Agudelo-Cordoba E, Albert JS. Gymnotiform electric fishes of the Tres Fronteras region of the western Amazon. Check List. 2023a; 19(5):767–90. https://doi.org/10.15560/19.5.767
Torgersen KT, Galindo-Cuervo AM, Ortega-Lara A, Lujan NK, Reis RE, Albert JS. Towards deconstruction of the Sternopygus macrurus species complex (Gymnotiformes: Sternopygidae): description of a new species from the Pacific Coast of Colombia. Zootaxa. 2026; 5748(4):539–560
Torgersen KT, Galindo-Cuervo AM, Reis RE, Albert JS. A new species of barred Sternopygus (Gymnotiformes: Sternopygidae) from the Orinoco River. Neotrop Ichthyol. 2023b; 21(1):e220088. https://doi.org/10.1590/1982-0224-2022-0088
Vari RP, Ferraris CJ Jr., Radosavljevic A, Funk VA. Checklist of the freshwater fishes of the Guiana Shield. Proc Biol Soc Wash. 2009; 17(1):1–95. https://doi.org/10.2988/0097-0298-17.1.i
Vari RP, de Santana CD, Wosiacki WB. South American electric knifefishes of the genus Archolaemus (Ostariophysi: Gymnotiformes): undetected diversity in a clade of rheophiles. Zool J Linn Soc. 2012; 165(3):670–99. https://doi.org/10.1111/j.1096-3642.2012.00827.x
Authors
![]() Kevin T. Torgersen1
Kevin T. Torgersen1 ![]() ,
, ![]() Jessé M. Figueiredo-Filho1,
Jessé M. Figueiredo-Filho1, ![]() Eric B. Haddad1,
Eric B. Haddad1, ![]() Patricia Torres-Pineda2,
Patricia Torres-Pineda2, ![]() Hernán López-Fernández2 and
Hernán López-Fernández2 and ![]() James S. Albert1
James S. Albert1
[1] School of Biological Sciences, University of Louisiana at Lafayette, Lafayette, LA 70503, USA. (KTT) kevintorgersen@gmail.com (corresponding author), (JMFF) jessefilho3@gmail.com, (EBH) eric.bruschi@hotmail.com, (JSA) james.albert@louisiana.edu.
[2] Department of Ecology and Evolutionary Biology and Museum of Zoology, University of Michigan, Ann Arbor, MI 48109, USA. (PTP) ptorresp@umich.edu, (HLF) hlopezf@umich.edu.
Authors’ Contribution 

Kevin T. Torgersen: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Jessé M. Figueiredo-Filho: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Eric B. Haddad: Data curation, Formal analysis, Investigation, Methodology, Software, Validation, Visualization, Writing-original draft, Writing-review and editing.
Patricia Torres-Pineda: Data curation, Investigation, Methodology, Resources, Validation, Writing-original draft, Writing-review and editing.
Hernán López-Fernández: Conceptualization, Funding acquisition, Project administration, Resources, Supervision, Writing-original draft, Writing-review and editing.
James S. Albert: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
All collections of specimens related to this project were made with HLF as Principal Investigator under Animal Care Protocol number PRO00008267 approved by the Institutional Animal Care and Use Committee at the University of Michigan in compliance with University Guidelines, State, and Federal Regulations, and the standards of the “Guide for the Care and Use of Laboratory Animals”.
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article and the accompanying supplementary material.
AI statement
Portions of the manuscript text were reviewed using ChatGPT (OpenAI) solely to improve grammar, clarity, and wording. No scientific content, data analysis, interpretation, or conclusions were generated by artificial intelligence, and all final text was written, reviewed, and approved by the authors.
Funding
HLF was funded by the University of Michigan. KTT received funding support from a Linnean Society & Systematics Association LinnéSys: Systematics Research Fund Award and a UL Lafayette Doctoral Research and Travel Grant.
Supplementary Material
Supplementary material S1
Supplementary material S2
Supplementary material S3
How to cite this article
Torgersen KT, Figueiredo-Filho JM, Haddad EB, Torres-Pineda P, López-Fernández H, Albert JS. A new endemic genus of glass knifefish (Gymnotiformes: Sternopygidae) for the rare species Distocyclus guchereauae from the Maroni basin. Neotrop Ichthyol. 2026; 24(1):e250167. https://doi.org/10.1590/1982-0224-2025-0167
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 11, 2025
Accepted December 11, 2025
![]() Submitted September 22, 2025
Submitted September 22, 2025
![]() Epub March 23, 2025
Epub March 23, 2025