![]() Katiane Mara Ferreira1,
Katiane Mara Ferreira1, ![]() Alexandre Cunha Ribeiro1 and
Alexandre Cunha Ribeiro1 and ![]() Fernando Rogério Carvalho2
Fernando Rogério Carvalho2 ![]()
PDF: Download Here | Cite this article
Associate Editor: ![]() George Mattox
George Mattox
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Uma espécie nova de Hemigrammus é descrita de afluentes do rio Cuiabazinho, bacia do alto rio Paraguai, Mato Grosso, Brasil. Ela pode ser prontamente distinguida de todas as suas congêneres por uma combinação única de caracteres, incluindo um padrão distinto de coloração corporal sem manchas, o número de escamas perfuradas na linha lateral (12–15), o número de raios ramificados na nadadeira anal (21–25) e a presença de pequenos ganchos ósseos nas extremidades das lepidotriquias do segundo ao quarto raios ramificados das nadadeiras pélvicas, os quais estão ausentes na nadadeira anal. Fornecemos também uma breve discussão sobre a recente reconfiguração taxonômica de Hemigrammus.
Palavras-chave: Peixes de água doce, Província Serrana de Mato Grosso, Rio Cuiabá, Serra Azul, Taxonomia.
Introduction
Hemigrammus Gill, 1858 is a rich genus in Acestrorhamphidae Eigenmann, 1907, with 57 valid species (Fricke et al., 2025), widely distributed across the Neotropical cis-Andean basins, including the basins of the Amazonas, Orinoco, La Plata, and São Francisco rivers, as well as those in Guyana, Suriname, French Guiana, and northeastern Brazil, with higher species diversity in the Amazon basin (Lima et al., 2009; Carvalho et al., 2010; Mendonça, Wosiacki, 2011; Ota et al., 2014; Lima et al., 2020; Toledo-Piza et al., 2024; Fricke et al., 2025). Recent studies based on molecular phylogenies (Melo et al., 2024), morphological phylogenies (Terán et al., 2020), and combined analyses of morphological and molecular data (Mirande, 2019) consistently demonstrate and reinforce that the genus is not a monophyletic clade. Its classification has been revised over the past few decades, with the most recent proposal (Melo et al., 2024), based on molecular data from ultraconserved elements (UCEs), housing Hemigrammus in the Pristellinae Géry & Boutière, 1964, within Acestrorhamphidae. However, a specific morphological diagnosis for the genus, in this new approach, was not provided in this reclassification.
In view of this, Hemigrammus is still being diagnosed by a combination of non-exclusive morphological traits, as proposed by Durbin (in Eigenmann, 1918) and Géry (1977), including: an incomplete pored lateral line, teeth on the premaxilla arranged in two rows, with the inner row of the premaxilla containing five teeth, few or no maxillary teeth, and scales extending to the basal region of the caudal-fin lobes.
In the absence of an updated diagnosis based on synapomorphy(ies), herein we adopt the morphological definition proposed by Durbin (in Eigenmann, 1918) and Géry (1977), for the proposed new species. This taxon was recently discovered during field expeditions in the headwaters of the upper rio Paraguai basin, specifically in the SESC Serra Azul region, municipality of Rosário Oeste, Mato Grosso State. The description of this new species aims to contribute to the taxonomy of Hemigrammus and to reduce the Linnean shortfall (Hortal et al., 2015; Reis et al., 2024), at a time marked by profound climatic changes and extensive anthropogenic impacts on ecosystems.
Material and methods
Counts and measurements follow Fink, Weitzman (1974) and Menezes, Weitzman (1990), except for counts of the horizontal scale rows below the lateral line counted to the pelvic-fin insertion. In the description, counts in parentheses refer to the number of specimens. Asterisks indicate value of the holotype. Counts of vertebrae and supraneurals were taken from cleared and stained (cs) paratypes; total vertebral counts include the four vertebrae of the Weberian apparatus and the terminal centrum. Specimens were cleared and stained following the method of Taylor, Van Dyke (1985). Osteological terminology follows Weitzman (1962) with the modifications listed by Vari, Harold (2001). Comparisons in Hemigrammus were based mainly on the analysis of type-specimens, including those in Carvalho et al. (2010). Institutional abbreviations follow Sabaj (2020).
Results
Hemigrammus serrazul,new species
urn:lsid:zoobank.org:act:A31D2F88-1D59-4DEC-BE7A-BED1C97C7293
(Figs. 1–5; Tab. 1)
Hemigrammus sp. n. —Ribeiro, Ferreira, 2023:8, 90 (listed).
Holotype. CPUFMT 8654, 36.7 mm SL, Brazil, Mato Grosso State, Rosário Oeste municipality, SESC Serra Azul Park, unnamed stream, rio Cuiabazinho tributary, upper rio Paraguai basin, 14°29’43.74”S 55°43’59.96”W, 280 m a.s.l., 28 Mar 2023, K. M. Ferreira & A. C. Ribeiro.
Paratypes. All from Brazil, Mato Grosso State, Municipality of Rosário Oeste, SESC Serra Azul Park, rio Cuiabazinho tributaries, upper rio Paraguai basin. CPUFMT 7894, 50, 16.2–34.7 mm SL, 2 cs, 19.4–24.6 mm SL, no name stream, 14°29’43.74”S 55°43’59.96”W, 25 Nov 2021, A. C. Ribeiro & K. M. Ferreira; CPUFMT 8005, 1, 26.2 mm SL, mouth of ribeirão Serra Azul, 14°28’24.37”S 55°44’12.69”W, 28 Sep 2021, A. C. Ribeiro; CPUFMT 8015, 3, 30.9–34.4 mm SL, ribeirão Serra Azul, below the waterfall, 14°29’23.33”S 55°42’24.97”W, 27 Sep 2021, A. C. Ribeiro; CPUFMT 8030, 2, 23.3–25.7 mm SL, ribeirão Serra Azul, near the bridge, 14°29’07”S 55°42’56”W, 25 Nov 2021, A. C. Ribeiro, K. M. Ferreira; CPUFMT 8059, 14, 29.7–35.1 mm SL, ribeirão Serra Azul, near the bridge, 14°29’07”S 55°42’56”W, 1 Jan 2022, A. C. Ribeiro & R. Anastácio; CPUFMT 8097, 1, 31.8 mm SL, ribeirão Serra Azul, near the bridge, 14°29’07”S 55°42’56”W, 27 Oct 2022, A. C. Ribeiro & K. M. Ferreira; CPUFMT 8123, 1, 28.8 mm SL, lagoon in front of the SESC Serra Azul restaurant, 14°28’28”S 55°41’58”W, 27 Oct 2022, A. C. Ribeiro & K. M. Ferreira; CPUFMT 8136, 69, 24.7–36.8 mm SL, 3 cs, 30.7–37.9 mm SL cs, collected with holotype; CPUFMT 8161, 58, 20.6–33.87 mm SL, unnamed stream, 14°29’43.74”S 55°43’59.96”W, 22 Sep 2023, A. C. Ribeiro & K. M. Ferreira; CPUFMT 8162, 18, 25.2–35.2 mm SL, alcohol fixed material, collected with holotype; CPUFMT 8187, 65, 11.9–35.2 mm SL, unnamed stream, 14°29’43.74”S 55°43’59.96”W, 21 Nov 2023, A. C. Ribeiro & K. M. Ferreira; CPUFMT 8218, 14, 21.5–30.8 mm SL, córrego Cocal, 14°28’10.99”S 55°40’15.99”W, 24 May 2023, A. C. Ribeiro & K. M. Ferreira; CITL 1556, 10, 22.8–34.9 mm SL, collected with CPUFMT 8161; DZSJRP 25257, 2, 28.4–29.1 mm SL, MZUSP 131458, 2, 26.8–30.2 mm SL, ZUEC 18563, 4, 21.6–27.6 mm SL, all collected with holotype.
FIGURE 1| Hemigrammus serrazul, holotype, CPUFMT 8654, 36.7 mm SL, unnamed stream, rio Cuiabazinho drainage, upper rio Paraguai basin, Rosário Oeste, Mato Grosso State, Brazil.
Diagnosis. Hemigrammus serrazul can be distinguished from most congeners (except of H. apiaka (Esguícero& Castro, 2017), H. erythrozonus Durbin, 1909, H. gracilis (Lütken, 1875), H. mimus Böhlke, 1955, and H. tupebas (Esguícero& Castro, 2017) by flank uniformly colored, without spots, i.e., no humeral and caudal spots, just a tiny and narrow longitudinal stripe (vs. species with spots in humeral and/or caudal peduncle). From H. apiaka, H. erythrozonus, H. gracilis, H. mimus, and H. tupebas, H. serrazul differs by premaxillary teeth in two rows: outer with three to five tricuspid teeth, inner with five tetra- pentacuspid teeth (vs. premaxillary teeth in one row in H. apiaka, H. tupebas, and just one tooth on outer row in H. mimus, with 7–10 conical or tricuspid teeth in inner/unique row); a narrow silvery/yellowish longitudinal stripe on flank, with slightly blackish upper eye portion in in vivo specimens (vs. conspicuous reddish longitudinal stripe on flank extending to upper eye portion in H. erythrozonus); pored lateral line scales 12–15 (mode 15) [vs. 8–11 (mode 9) in H. gracilis] and branched anal-fin rays 21–25 (mode 22) [vs. 18–20 (mode 18) in H. apiaka; 17–18 (mode 17) in H. erythrozonus; 18–20 (mode 19) in H. gracilis; 14 in H. mimus, and 19–20 (mode 19) in H. tupebas)]. Additionally, H. serrazul can be distinguished from its congeners by the presence of small bony hooks on the tip of lepidotrichia of the second to fourth branched pelvic-fins rays, these absent from the anal fin.
Description. Morphometric and meristic data for holotype and paratypes in Tab 1. Body laterally compressed. Greatest body depth anterior to vertical through dorsal-fin origin. Dorsal profile of head convex from tip of upper jaw to vertical through anterior nostril; straight or slightly convex from that point to tip of supraoccipital spine. Dorsal body profile slightly convex from tip of supraoccipital spine to dorsal-fin origin; straight along dorsal-fin base; slightly convex from terminus of dorsal-fin base to adipose-fin insertion, and concave posteriorly from that point to anteriormost procurrent caudal-fin ray. Ventral profile of head and body convex from tip of lower jaw to pelvic-fin insertion, straight from that point to anal-fin origin, straight along anal-fin base and concave along caudal peduncle.
TABLE 1 | Morphometric data of Hemigrammus serrazul. SD = Standard deviation; N = 30 specimens, including holotype.
| Holotype | Paratypes | ||
Range | Mean | SD | ||
Standard length | 36.7 | 21.0–37.4 | 29.8 | – |
Percentages of standard length | ||||
Body depth | 34.8 | 31.4–36.9 | 34.3 | 1.5 |
Snout to dorsal-fin origin | 51.1 | 46.2–53.8 | 49.9 | 1.7 |
Snout to pectoral-fin origin | 26.5 | 21.6–28.2 | 26.3 | 1.2 |
Snout to pelvic-fin origin | 46.5 | 43.5–50.4 | 46.0 | 1.5 |
Snout to anal-fin origin | 62.0 | 57.5–63.5 | 60.8 | 1.4 |
Pelvic-fin insertion to anal-fin origin | 14.4 | 12.6–18.2 | 15.3 | 1.0 |
Caudal-peduncle depth | 9.3 | 9.0–11.7 | 10.4 | 0.6 |
Caudal-peduncle length | 10.7 | 10.0–13.9 | 12.2 | 1.1 |
Dorsal-fin origin to caudal-fin base | 47.9 | 47.8–53.7 | 50.7 | 1.4 |
Dorsal-fin base length | 14.4 | 10.0–14.8 | 13.2 | 1.0 |
Anal-fin base length | 32.2 | 28.2–33.5 | 31.2 | 1.4 |
Pectoral-fin length | 23.5 | 18.7–26.3 | 22.1 | 1.5 |
Pelvic-fin length | 18.9 | 16.7–23,6 | 18.7 | 1.6 |
Dorsal-fin length | 31.1 | 25.9–34.9 | 29.6 | 1.6 |
Anal-fin length | 22.7 | 19.3–26.2 | 21.6 | 1.4 |
Bony head length | 26.7 | 23.7–27.9 | 25.9 | 1.1 |
Head depth | 24.2 | 21.8–25.9 | 23.7 | 1.1 |
Percentages of head length | ||||
Horizontal eye diameter | 42.4 | 41.1–45.8 | 43.4 | 1.3 |
Snout length | 23.4 | 22.3–28.0 | 24.7 | 1.3 |
Least interorbital distance | 31.6 | 28.3–33.5 | 31.7 | 1.2 |
Upper jaw length | 47.9 | 45.9–49.7 | 47.7 | 1.1 |
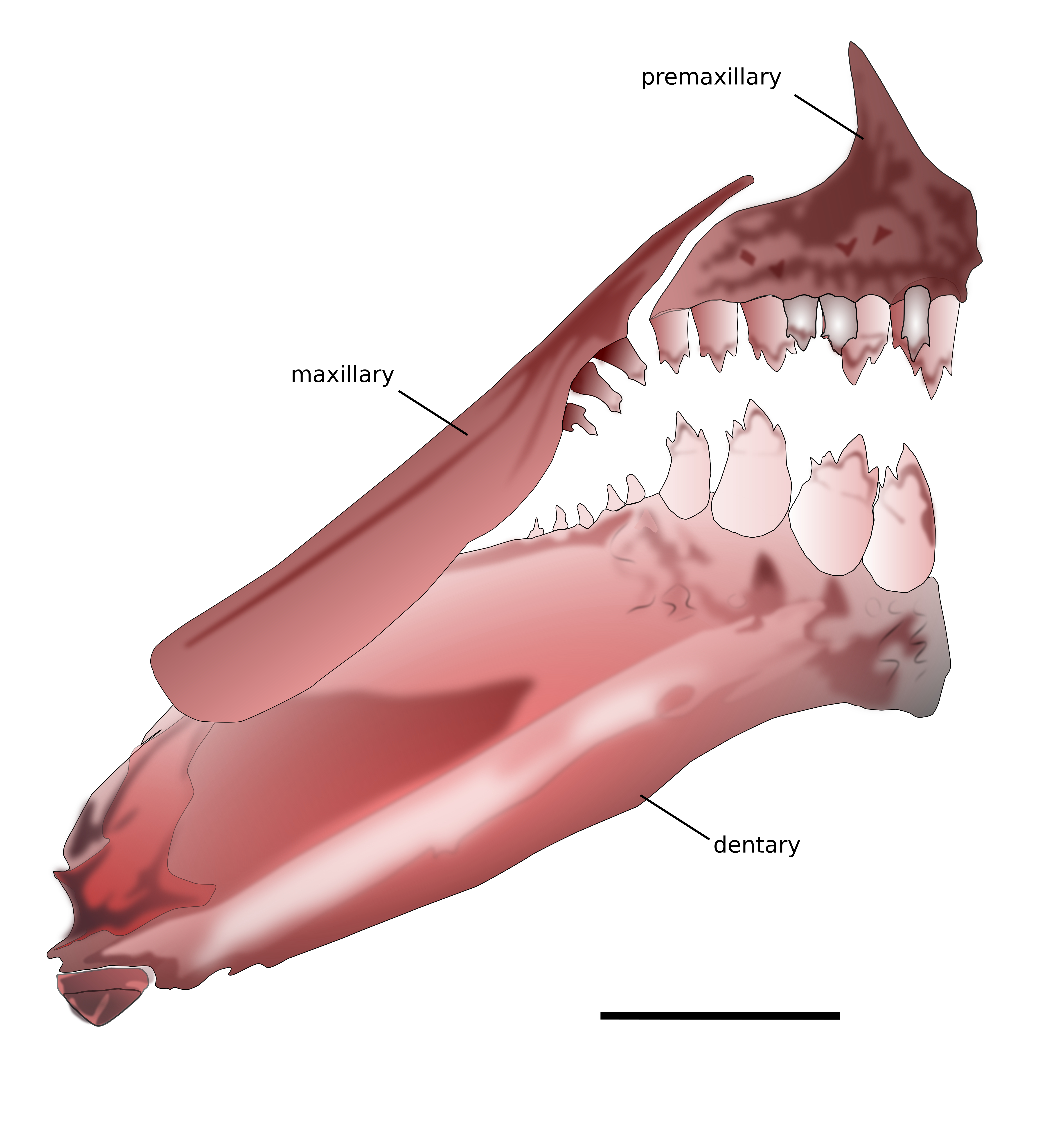
Mouth terminal. Posterior terminus of maxilla reaching vertical through anterior third of eye. Premaxilla with two rows of teeth (Fig. 2). Outer tooth row aligned, with 3(10), 4*(6) or 5(14) tricuspid teeth, with median cusps more developed. Inner row with five teeth larger than those of outer series; symphyseal tooth with five cusps, remaining teeth with tri- to pentacuspid. Maxilla with 1(2), 2(19) or 3*(9) tricuspid teeth. Dentary with 4 anteriormost teeth larger tri- to pentacuspid, gradually decreasing in size laterally, followed by a series of five to eight smaller unicuspid teeth.
FIGURE 2| Hemigrammus serrazul, paratype, CPUFMT 8136, 37.9 mm SL. Upper and lower jaws, left side, lateral view. Scale bar = 1 mm.
Scales cycloid, with two to six radii diverging from focus to posterior portion of scale; circuli weakly marked. Lateral line incomplete, with 12*(6), 13(4), 14(2), 15(9), 16(5), 17(3), or 18(2) perforated scales, longitudinal scale series including pored scale, 32*(4), 33(9), 34(9), or 35(6). Scales rows between dorsal-fin origin and lateral line 5*(30). Scales rows between lateral line and pelvic-fin 3(3) or 4*(27). Predorsal scales 9(2), 10*(13), 11(10), 12(4), or 13(1). Circumpeduncular scales 14*(30). Single row of 4–5 scales covering base of anal-fin rays. Base of caudal-fin scaled; squamation slightly more developed on lower caudal fin lobe, covering at least one-third of lobe. Scales on the caudal fin smaller that the scales covering the body (Fig. 3).
FIGURE 3| Hemigrammus serrazul, paratype, CPUFMT 8187, 33.4 mm SL. Left lateral view of caudal-fin squamation, stained specimen.
Dorsal-fin rays ii, 9*(20). Dorsal-fin origin at middle of standard length or slightly behind this point. First dorsal-fin pterygiophore located between neural spines of 8th and 9th(2) vertebrae. First unbranched ray about half the length of second ray; small ossification anterior to first unbranched ray present in the two cs specimens examined. Adipose fin present, its origin in vertical through last or next to last anal-fin ray insertion. Pectoral-fin rays i,11*(20) or 12(10). Tip of adpressed pectoral fin reaching pelvic-fin insertion. Pelvic-fin rays i,7*(30). Tip of adpressed pelvic fin reaching anteriormost anal-fin rays. Anal-fin rays iv, 21(7), 22(15), 23*(9), 24(1), or 25(1). First anal-fin pterygiophore located between hemal spine of 15th and 16th(2) vertebrae. Caudal fin forked, lobes similar in size; principal caudal-fin rays i,9/8,i*(20).
First gill arch with 10(4), 11*(14), or 12(9) gill rakers on the hypobranchial and ceratobranchial, 6(9), 7*(6), or 8(11) on the epibranchial, and 1 on cartilage between ceratobranchial and epibranchial. Branchiostegal rays 4(1), 3(2) on anterior ceratohyal, and 1(2) on posterior ceratohyal. Vertebrae 33(2). Supraneurals 4(2).
Coloration in alcohol. Ground color light beige. Snout and dorsal portion of head light brown, with high concentration of scattered melanophores. Dark background color over predorsal scales. Small melanophores present on maxilla and lower jaw, scattered melanophores on fifth to sixth infraorbitals and upper half of the opercle. Guanine on opercle, infraorbitals, and sides of body. Humeral and caudal peduncle spots absent. Slight and narrow longitudinal dark stripe along midline of body, originating at five to seven scales after opercle, covering less than one scale. Scales from the laterodorsal portion of the body with a high concentration of melanophores near the posterior margin, with an overall darker coloration to dorsal portion of body. Abdominal region slightly pigmented, some scattered melanophores in region of anal-fin base. Dorsal, adipose, pectoral, pelvic, anal and caudal fins hyaline, with scattered melanophores on interradial membranes. Melanophores outline the rays and form horizontal, narrow lines on the fins.
Coloration in life. Based on three specimens observed in the field (Figs. 4–5): general coloration of the body can vary from yellowish/reddish in males (Fig. 4A) and silver in females (Fig. 4B). Upper portion of eye dark. Caudal, anal, and dorsal fins reddish (Fig. 4A) or hyaline (Fig. 4B). Midlateral longitudinal stripe silvery-yellow. Melanophores as in preserved specimens (Fig. 1).
FIGURE 4| Color pattern variations in live specimens of Hemigrammus serrazul: A. Male; B. Female. Photo by Alexandre C. Ribeiro.
FIGURE 5| Hemigrammus serrazul, live specimen in aquarium, photographed just after capture in córrego Cocal (CPUFMT 8218), upper rio Paraguai basin, municipality of Rosário Oeste, Mato Grosso State, Brazil. Inserir: Photo by Alexandre C. Ribeiro.
Sexual dimorphism. Bony hooks present only on pelvic-fin rays of males larger than 29.2 mm SL; anal-fin without hooks. Pelvic fin of males with small hooks along margin of second to fourth branched rays; one small and weakly developed hook per distal ray segment. Females without hooks. Distal border of anal fin approximately straight in males (Fig. 4A), and slightly concave (falcated) in females (Fig. 4B). Color in life yellowish to reddish in males (Fig. 4A) and silver in females (Fig. 4B).
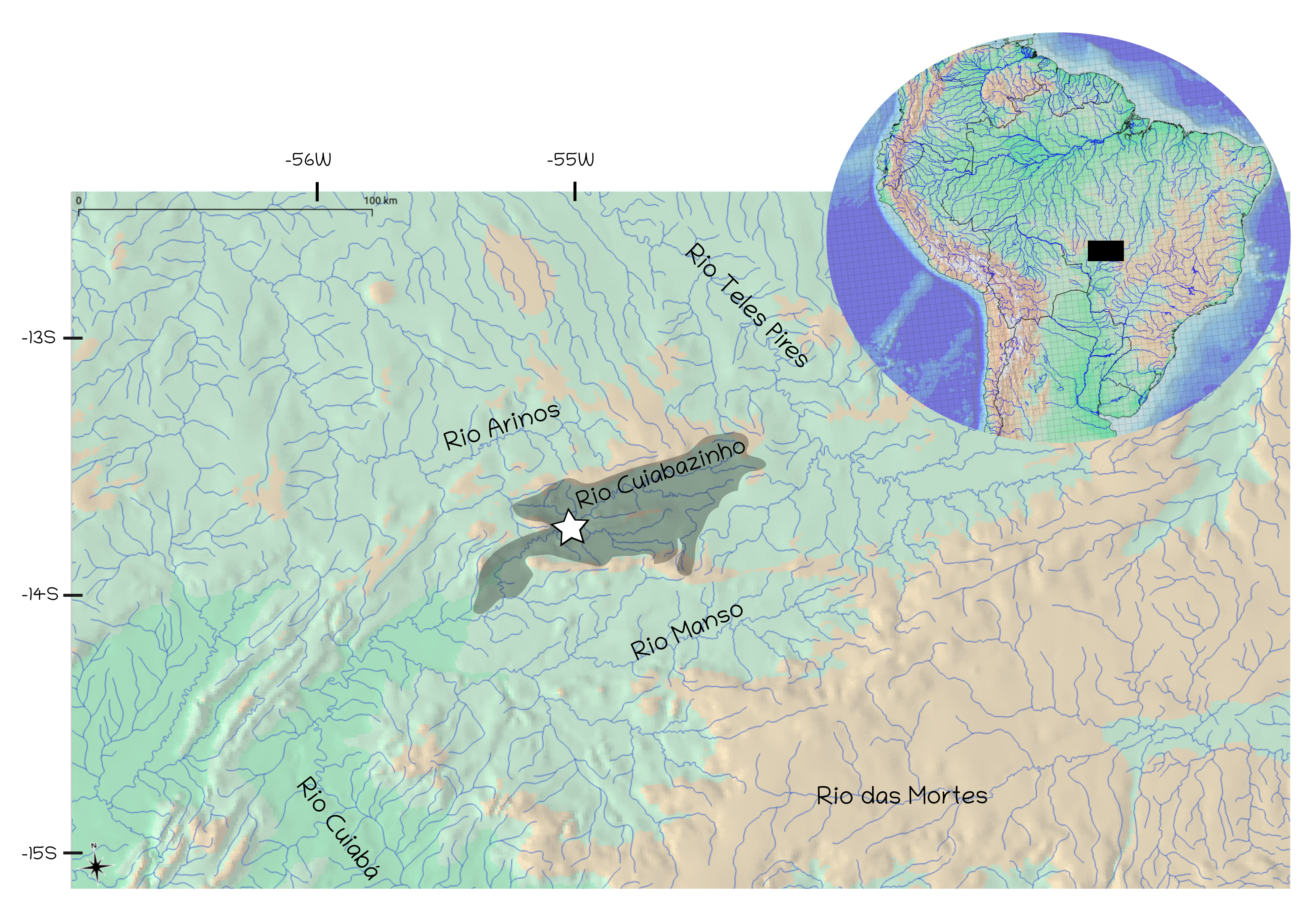
Geographical distribution. Hemigrammus serrazul is known from the rio Cuiabazinho (upper rio Cuiabá basin) and its headwater tributaries within the SESC Serra Azul Park, in the municipality of Rosário Oeste, Mato Grosso State, Brazil (Fig. 6).
FIGURE 6| Map of South American hydrography highlighting the upper rio Paraguai basin (dark rectangle). The inset shows the rio Cuiabazinho basin (highlighted in light gray), with the white star indicating the type locality of Hemigrammus serrazul within the rio Cuiabazinho basin.
Ecological notes. Hemigrammus serrazul was collected along the banks of the rio Cuiabazinho and in three of its tributaries, all within the SESC Serra Azul Park area. Among the sampled sites, the stream that showed the highest abundance and consistency of H. serrazul collections is a first-order stream and a direct tributary of the left bank of the rio Cuiabazinho. This stream is shallow (15–50 cm), with lentic waters, a muddy substrate, and abundant macrophytes (Fig. 7). The six stomach contents analyzed revealed the presence of ants and aquatic insect larvae.
FIGURE 7| Type locality of Hemigrammus serrazul, unnamed stream, rio Cuiabazinho drainage, upper rio Paraguai basin, municipality of Rosário Oeste, Mato Grosso State, Brazil. Photo by Katiane M. Ferreira.
Etymology. The specific epithet serrazul refers to the type locality where the new species was collected, the Serra Azul, the local portion of the so-called Serrana Province of Mato Grosso, a range of escarped relief that serves as a watershed divide between the rio Cuiabá, Paraguai, and Arinos basins. A noun in apposition.
Conservation status. The new species has been identified at four nearby localities within SESC Serra Azul Park, which spans 5,700 hectares of relatively well-preserved Cerrado. Hemigrammus serrazul is abundant at these sites, and we did not identify any imminent threats to the species within its range. Therefore, we suggest that H. serrazul be categorized as Least Concern (LC) according to the categories and criteria of the International Union for Conservation of Nature (IUCN Standards and Petitions Subcommittee, 2024).
Discussion
Géry (1977) conducted an artificial classification of Hemigrammus species based only on coloration patterns. In this approach, Hemigrammus serrazul is part of the group “a”, called “the He. gracilis-group” which is characterized by the absence of black spots on the body of the species. In addition to H. gracilis (Fig. 8B), from the rio São Francisco basin, Géry (1977) included H. erythrozonus (Fig. 8A) and H. mimus (Fig. 8C) in the group “a”. Hemigrammus mimus is a poorly known species from the the Amazon basin and H. erythrozonus from the río Essequibo basin in Guyana. Hemigrammus serrazul differs from the species of Géry’s group “a” as outlined in the diagnosis.
FIGURE 8| Hemigrammus species without black markings sensu Géry (1977): A. Hemigrammus erythrozonus, FMNH 53546, holotype, 24.0 mm SL; B. Tetragonopterus gracilis, NMW 68066, syntype, 27.5 mm SL; C. Hemigrammus mimus, CAS 47759, holotype, 27.8 mm SL. Credits: A. © Field Museum of Natural History – CC BY-NC 4.0; B. Fernando R. Carvalho; C. © 2001 California Academy of Sciences.
Hemigrammus serrazul represents the sixth species of the genus recorded for rio Paraguai basin. Among current species of Hemigrammus recorded in this basin, H. flava (Britzke, Troy, Oliveira & Benine, 2018), originally described as a Moenkhausia species from tributaries of the rio Sepotuba, above the Salto das Nuvens and Salto Maciel waterfalls, is restricted to the upper rio Paraguai basin. The other species, H. lunatus Durbin, 1918, H. machadoi Ota, Lima & Pavanelli, 2014, H. tridens Eigenmann, 1907, and H. ulreyi (Boulenger, 1895) also occur in adjacent basins. Hemigrammus flava can be easily distinguished from H. serrazul by several morphological features, including the presence of a complete lateral line (vs. lateral line incomplete), presence of a humeral spot (vs. absence), six scale rows above the lateral line (vs. five). Ota et al. (2014) proposed a putative monophyletic group within the genus Hemigrammus, based on general body morphology and coloration patterns, termed the “H. lunatus group”. This group includes four species, three of which (H. machadoi, H. lunatus, and H. ulreyi) have records in the rio Paraguai basin. Hemigrammus serrazul can be readily distinguished from these three species by the absence of a conspicuous dark horizontal stripe across the eye (vs. presence). Additionally, H. serrazul differs from H. machadoi and H. lunatus in lacking a humeral spot (vs. presence). And finally, H. tridens is easily differentiated from H. serrazul by the presence of a conspicuous spot on the caudal peduncle (vs. absence).
The Serra Azul region, located in the Serrana Province of Mato Grosso, encompasses the Raizama, Araras, and Diamantinha Formations and is embedded in a geomorphological context marked by steep escarpments resulting from neotectonic activity (Ribeiro, Ferreira, 2023). Such conditions favor the isolation of biological lineages, particularly small-bodied taxa (≤ 150 mm SL sensu Castro, 1999). The discovery of a new taxon in this area highlights its biogeographical relevance and underscores the need for conservation actions on the plateau of the upper rio Paraguai basin. In this context, the SESC Serra Azul Park plays a central role in safeguarding the region’s biodiversity.
Comparative material examined. Besides the material presented in Carvalho et al. (2010), the comparative material examined include: type material (analysed by FRC). Bolivia: Holopristis neptunus: MTD F 25536, 31.3 mm SL, holotype of Hemigrammus neptunus Zarske & Géry, 2002; MTD F 25537–25545, 9, 24.8–28.6 mm SL, paratypes of H. neptunus. Brazil: Hemigrammus bellottii: NMW 57253, 14, 13.0–24.1 mm SL, NMW 57254, 4, 24.2–30.0 mm SL, syntypes of Tetragonopterus bellottii Steindachner, 1882. Hemigrammus collettii: NMW 57379, 3, 39.2–49.9 mm SL, NMW 57382, 5, 27.4–31.4 mm SL, syntypes of Tetragonopterus collettii Steindachner, 1882. Hemigrammus elegans: NMW 57390, 8, 21.6–24.5 mm SL, syntypes of Tetragonopterus elegans Steindachner, 1882. Hemigrammus filamentosus Zarske, 2011: MTD F 32438, 36.2 mm SL, holotype. Hemigrammus gracilis: NMW 68066, 5, 23.4–27.9 mm SL, MNHN 9583, 27.3 mm SL, syntypes of Tetragonopterus gracilis. Hemigrammus pretoensis Géry, 1965: SMF 7237, 45.3 mm SL, holotype. Hemigrammus schmardae: NMW 57979, 3, 26.1–26.2 mm SL, syntypes of Tetragonopterus schmardae Steindachner, 1882. Hemigrammus mimus: CAS 147759, 27.8 mm SL, holotype. Hemigrammus vorderwinkleri Géry, 1963: MHNG 2179.043, 2, 21.9–29.5 mm SL, ZMA 113.852, 26.6 mm SL, paratypes. French Guiana: Hemigrammus aereus Géry, 1959: MHNG 2181.86, 23.7 mm SL, holotype; MHNG 2181.087, 17.2 mm SL, paratype. Hemigrammus boesemani: MHNG 2181.80, 25.6 mm SL, holotype of Hemigrammus micropterus boesemani Géry, 1959. Hemigrammus unilineatus: MHNG 2179.061, 36.4 mm SL, holotype of Hemigrammus unilineatus cayennensis Géry, 1959; MHNG 2179.055, 21.2 mm SL, MHNG 2179.063, 28.8 mm SL, SMF 5385, 26.2 mm SL, ZMA 101049, 19.8 mm SL, all paratypes of H. u. cayennensis. Holopristis guyanensis: MHNG 2181.023, 23.2 mm SL, holotype of Hemigrammus guyanensis Géry, 1959. Guyana: Hemigrammus analis Durbin, 1909: MHNG 2182.040, 2, 21.4–24.3 mm SL, paratypes; NMW 69498, 20.2 mm SL, paratype. Hemigrammus erythrozonus: FMNH 53546, 24.0 mm SL, holotype; ZMA 101032, 16.6 mm SL, paratype. Hemigrammus orthus Durbin, 1909: MHNG 2180.032, 21.0 mm SL, paratype; ZMA 100.977, 14.9 mm SL, paratype. Non-type specimens (analyzed by KMF and ACR). All from Mato Grosso, Brazil: Cyanogaster geisleri: CPUFMT 2676, 49, 20.5–22.3 mm SL, rio São Benedito, rio Teles Pires basin. Hemigrammus bellottii: CPUFMT 4908, 3, 19.2–34.4 mm SL, downstream of Dardanelos and Andorinhas waterfalls, rio Aripuanã, rio Madeira basin. Hemigrammus lunatus: CPUFMT 552, 4, 28.4–32.3 mm SL, rio Cuiabá, upper rio Paraguai basin. Hemigrammus machadoi: CPUFMT 4309, 3, 21.4–30.7 mm SL, rio Mutum, upper rio Paraguai basin. Hemigrammus cf. microstomus Durbin, 1918: CPUFMT 4245, 2, 18.9–19.9 mm SL, rio Verde, rio Teles Pires basin. Hemigrammus neptunus: CPUFMT 7683, 1, 22.4 mm SL, rio Aricá-Açu, upper rio Paraguai basin. Hemigrammus ora Zarske, Le Bail & Géry, 2006: CPUFMT 913, 15, 23.1–28.0 mm SL, rio Capão Bonito, rio Correntes basin, upper rio Paraguai basin. Hemigrammus parana Marinho, Carvalho, Langeani & Tatsumi, 2008: CPUFMT 2955, 24, 18.9–25.8 mm SL, córrego Covas, rio dos Mortos, Araguaia basin. Hemigrammus silimoni Britski & Lima, 2008: CPUFMT 5034, 5, 21.4–30.7 mm SL, upstream of Dardanelos and Andorinhas waterfalls, rio Aripuanã, rio Madeira basin. Hemigrammus tridens: CPUFMT 491, 699, 11.7–13.7 mm SL, lagoa 54, rio Cuiabá basin, upper rio Paraguai basin. Hemigrammus ulreyi: CPUFMT 4325, 1, 32.9 mm SL, rio Mutum, upper rio Paraguai basin.
Acknowledgments
The authors are grateful to the team of the Polo Socioambiental do SESC Pantanal for the logistical support provided during the project. We also thank Anderson Florencio for his assistance at the SESC Serra Azul Park. For museum and technical support and/or loan of material we are indebted to David Catania, Jon Fong, Tomio Iwamoto (CAS), Mary Anne Rogers, Kevin Swagel, Philip Willink, Susan Mochel (FMNH), Axel Zarske (MTD F), Sonia Fisch-Muller, Raphael Covain (MHNG), Patrice Pruvost (MNHN), Wellendorf Helmut (NMW), Ronald de Ruiter (SMF and ZMA).
References
Castro RMC. Evolução da ictiofauna de riachos sul-americanos: padrões gerais e possíveis processos causais. Oecol Aust. 1999; 6:139–55.
Carvalho FR, Bertaco VA, Jerep FC. Hemigrammus tocantinsi: a new species from the upper rio Tocantins basin, Central Brazil (Characiformes: Characidae). Neotrop Ichthyol. 2010; 8(2):247–54. https://doi.org/10.1590/S1679-62252010000200002
Eigenmann CH. The American Characidae. Part 2. Mem Mus Comp Zool. 1918; 43:103–208.
Fink WL, Weitzman SH. The so-called cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Smithson Contrib Zool. 1974; 172:1–46.
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Géry J. Characoids of the world. New Jersey: T.F.H. Publications; 1977.
Hortal J, Bello F, Diniz JAF, Lewinsohn TM, Lobo JM, Ladle RJ. Seven shortfalls that beset large-scale knowledge on biodiversity. Annu Rev Ecol Evol Syst. 2015; 46:523–49. https://doi.org/10.1146/annurev-ecolsys-112414-054400
International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for using the IUCN Red List categories and criteria. Version 16 [Internet]. Gland; 2024. Available from: http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf
Lima FCT, Wosiacki WB, Ramos CS. Hemigrammus arua, a new species of characid (Characiformes: Characidae) from the lower Amazon, Brazil. Neotrop Ichthyol. 2009; 7(2):153–60. https://doi.org/10.1590/S1679-62252009000200004
Lima FCT, Urbano-Bonilla A, Prada-Pedreros S. A new Hemigrammus from the upper Río Vaupés, Colombia (Characiformes: Characidae), with a discussion on the presence of an enlarged urogenital papilla in the family. J Fish Biol. 2020; 96:868–76. https://doi.org/10.1111/jfb.14267
Melo BF, Ota RP, Benine RC, Carvalho FR, Lima FCT, Mattox GMT et al. Phylogenomics of Characidae, a hyper-diverse Neotropical freshwater fish lineage, with a phylogenetic classification including four families (Teleostei: Characiformes). Zool J Linn Soc. 2024; 202:zlae101. https://doi.org/10.1093/zoolinnean/zlae101
Mendonça MB, Wosiacki WB. A new species of Hemigrammus from the lower Amazon floodplain (Characiformes: Characidae). Copeia. 2011; 2011(2):211–15. https://doi.org/10.1643/CI-09-214
Menezes NA, Weitzman SH. Two new species of Mimagoniates (Teleostei: Characidae: Glandulocaudinae), their phylogeny and biogeography and a key to the glandulocaudin fishes of Brazil and Paraguay. Proc Biol Soc Wash. 1990; 103:380–426. Available from: https://www.biodiversitylibrary.org/page/34591837#page/400/mode/1up
Mirande JM.Morphology, molecules and the phylogeny of Characidae (Teleostei: Characiformes). Cladistics. 2019; 35(3):282–300. https://doi.org/10.1111/cla.12345
Ota RP, Lima FCT, Pavanelli CS. A new species of Hemigrammus Gill, 1858 (Characiformes: Characidae) from the rio Madeira and rio Paraguai basins, with a redescription of H. lunatus. Neotrop Ichthyol. 2014; 12(2):265–79. https://doi.org/10.1590/1982-0224-20130176
Reis GS, Tejerina–Garro FL, Dagosta FCP, Teresa FB, Carvalho RA. Seeking for gaps in taxonomic descriptions of endemic fishes: a pathway to challenge the Linnean shortfall in a Neotropical basin. Neotrop Ichthyol. 2024; 22(2):e230128. https://doi.org/10.1590/1982-0224-2023-0128
Ribeiro AC, Ferreira KM. Peixes do Ribeirão Serra Azul. Cuiabá: Ed. dos Autores; 2023.
Taylor WR, Van Dyke GC. Revised procedures for clearing and staining small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Terán GE, Benitez MF, Mirande JM. Opening the Trojan horse: phylogeny of Astyanax, two new genera and resurrection of Psalidodon (Teleostei: Characidae). Zool J Linn Soc. 2020; 190(4):1217–34. https://doi.org/10.1093/zoolinnean/zlaa019
Toledo–Piza M, Baena EG, Dagosta FCP, Menezes NA, Ândrade M, Benine RC et al. Checklist of the species of the Order Characiformes (Teleostei: Ostariophysi). Neotrop Ichthyol. 2024; 22(1):e230086. https://doi.org/10.1590/1982-0224-2023-0086
Sabaj MH. Codes for natural history collections in ichthyology and herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
Vari RP, Harold AS. Phylogenetic study of the neotropical fish genera Creagrutus Günther and Piabina Reinhardt (Teleostei: Ostariophysi: Characiformes), with revision of the Cis-Andean species. Smithson Contrib Zool. 2001; 613:1–239. http://dx.doi.org/10.5479/si.00810282.613
Weitzman SH. The osteology of Brycon meeki, a generalized characid fish, with an osteological definition of the family. Stanford Ichthyol Bull. 1962; 8(1):1–77.
Authors
![]() Katiane Mara Ferreira1,
Katiane Mara Ferreira1, ![]() Alexandre Cunha Ribeiro1 and
Alexandre Cunha Ribeiro1 and ![]() Fernando Rogério Carvalho2
Fernando Rogério Carvalho2 ![]()
[1] Instituto de Biociências, Departamento de Biologia e Zoologia, Laboratório de Ictiologia da UFMT-CPUFMT, Universidade Federal de Mato Grosso – UFMT, Avenida Fernando Corrêa, 2367, Boa Esperança, 78060-900, Cuiabá, MT, Brazil. (KMF) kmferreira@gmail.com, (ACR) alexandrecunharibeiro@gmail.com.
[2] Câmpus de Três Lagoas, Laboratório de Ictiologia e Coleção Ictiológica de Três Lagoas (CITL), Universidade Federal de Mato Grosso do Sul – UFMS, Avenida Ranulpho Marques Leal, 3484, Distrito Industrial II, 79613-000, Três Lagoas, MS, Brazil. (FRC) carvalhofr@gmail.com (corresponding author).
Authors’ Contribution 

Katiane Mara Ferreira: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Writing-original draft, Writing-review and editing.
Alexandre Cunha Ribeiro: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Writing-original draft, Writing-review and editing.
Fernando Rogério Carvalho: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Writing-original draft, Writing-review and editing.
Ethical Statement
The fish were collected under permission issued by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio/SISBIO number 71172–3).
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
KMF and ACR were funded by Universidade Federal de Mato Grosso (Edital de Apoio à Pesquisa/2021); ACR was funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq #404831/2023–0); FRC was previously funded by CNPq (#420620/2018–4) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP #2012/03404–2).
How to cite this article
Ferreira KM, Ribeiro AC, Carvalho FR. A new Hemigrammus (Characiformes: Acestrorhamphidae) from the rio Cuiabazinho and its tributaries, upper rio Paraguai basin. Neotrop Ichthyol. 2026; 24(1):e250157. https://doi.org/10.1590/1982-0224-2025-0157
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted September 3, 2025
Accepted September 3, 2025
![]() Submitted October 21, 2025
Submitted October 21, 2025
![]() Epub March 16, 2026
Epub March 16, 2026