![]() Yanis Cruz-Quintana1,2,
Yanis Cruz-Quintana1,2, ![]() Ana María Santana-Piñeros1,2
Ana María Santana-Piñeros1,2 ![]() ,
, ![]() Byron Manuel Reyes-Mero1,
Byron Manuel Reyes-Mero1, ![]() Leonela Griselda Muñoz-Chumo1,
Leonela Griselda Muñoz-Chumo1, ![]() Juan Carlos Vélez-Chica1,
Juan Carlos Vélez-Chica1, ![]() Carles Alcaraz3 and
Carles Alcaraz3 and ![]() Neil Duncan2
Neil Duncan2
PDF: Download Here | Cite this article
Associate Editor: ![]() Fernando Pelicice
Fernando Pelicice
Section Editor: ![]() Fernando Pelicice
Fernando Pelicice
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
El dormilón del Pacífico, Dormitator latifrons es un pez nativo del Pacífico centro oriental que se encuentra bajo una creciente presión humana. La recolección de adultos para consumo y de juveniles para acuicultura resalta la urgente necesidad de comprender su biología para fines de manejo. Este estudio desarrolló una escala de desarrollo gonadal macroscópica e histológica, describió la morfología de la papila genital y evaluó la proporción sexual, el tamaño de madurez y el desarrollo de ovocitos. Entre diciembre de 2017 y agosto de 2018, se recolectaron 295 especímenes del río Carrizal-Chone, Ecuador. Se registró la longitud total, el peso y el sexo, y se analizaron los órganos reproductivos. La proporción sexual de machos a hembras fue de 1,05:1. La papila genital varió en tamaño y apariencia con el desarrollo gonadal. Una escala histológica universal de cinco fases identificó con precisión las fases reproductivas: inmadura, en desarrollo, capaz de reproducir, en regresión y en regeneración. El desarrollo ovárico fue sincrónico en grupo, lo que sugiere una estrategia de desove total. Los ovocitos no fertilizados fueron esféricos, con una cápsula delgada, de 4 a 8 gotitas de aceite y estrías longitudinales. La distribución del tamaño de los ovocitos reveló poblaciones maduras e inmaduras. La longitud al 50% de madurez (L50) fue de 13,26 cm para las hembras y de 12,09 cm para los machos. Estos hallazgos permiten una comprensión más precisa de la historia de vida de D. latifrons, y pueden contribuir a la conservación y la gestión sostenible de los recursos.
Palabras clave: Biología reproductiva de peces, Especies anfídromas, Histología, Papila genital.
Introduction
The study of reproductive biology in fish is critical for the effective and sustainable management of fisheries and aquaculture (Connaughton, Aida; 1999; Le Gac, Loir, 1999). Within the Gobiiformes, reproductive traits are notably diverse, reflecting their broad ecological distribution and the variety of reproductive strategies that have evolved in response to environmental pressures such as salinity, habitat type, and seasonality (Brockmann, 1974; Nordlie, 1981; Keith, Lord, 2011; Violante-González et al., 2016). These strategies range from semelparity, where individuals spawn once and subsequently die, to iteroparity, in which multiple spawning events occur throughout and individual’s lifespan (Wootton, 1990; Teletchea et al., 2009; Yamasaki et al., 2011). Iteroparous species may exhibit either synchronous ovarian development, leading to a single spawning event per cycle, or asynchronous development, characterized by the release of several batches of oocytes within a single reproductive season, i.e., fractional spawning (Tyler, Sumpter, 1996; Plaza et al., 2007). Among gobiid fishes, iteroparity is the predominant strategy and is typically associated with substrate spawning, adhesive eggs, and male parental care, particularly nest guarding and aeration of the eggs (Dinh et al., 2016; Miller, 1984).
Within the Gobiiformes, the family Eleotridae, commonly known as sleepers or gudgeons, is the second most species-rich lineage, comprising approximately 139 species across 22 genera distributed throughout tropical and subtropical regions of the Neotropics, Africa, and the Indo-Pacific (Nelson et al., 2016). Most eleotrid species inhabit freshwater and brackish environments, and many exhibit amphidromous life cycles, spawning in marine or estuarine habitats and returning to freshwater as juveniles (Maitland, 2003). Despite their ecological diversity and wide distribution, detailed information on fecundity, sexual differentiation, gonadal development, and spawning periodicity remains scarce, particularly among Neotropical species. While histological and morphological analyses have been conducted in some gobioid taxa, e.g., Tridentiger trigonocephalus (Cho et al., 2014), such studies remain limited within the Eleotridae (see Teixeira, 1994; Maeda et al., 2008; Nordlie, 1981, 2012), thus constraining our understanding of reproductive strategies in this group.
Among the Eleotrids, Dormitator latifrons, commonly known as Pacific fat sleeper, is an amphidromous species found along the Pacific coast of Central and South America (southern California, USA to Peru) including the Galapagos Islands (Massay, Mosquera, 1992; McDowall, 2009; Nordlie, 2012; Vega-Villasante et al., 2021). This species demonstrates remarkable environmental tolerance, thriving in salinities up to 40 UPS, temperatures between 21–30 °C and oxygen levels as low as 0.4 ppm (Chang, 1984; FAO, 2010). Adults inhabit estuarine and mangrove ecosystems during the breeding season and migrate to freshwater habitats during the non-reproductive period (Chang, Navas, 1984; Graham et al., 1985; Massay, Mosquera, 1992; Santana-Piñeros et al., 2024). Nonetheless, reproductive activity has also been observed in both freshwater and marine systems (Ruiz-Campos, 2012; Jiménez-Prado et al., 2015).
Economically, D. latifrons holds significant value in coastal ecosystems from Ecuador, where it is prefered over tilapia (Castro-Rivera et al., 2005; Bermúdez-Medranda et al., 2025). Aquaculture production currently relies on capturing wild juveniles for fattening (FAO, 2010), a practice that exerts pressure on natural population. In 2019, Ecuador produced 261 tons of D. latifrons (US$1,001,000) compared to 9,200 tons of tilapia ($23,552,000) (FAO, 2020). The species is also gaining aquaculture interest in countries like Mexico and Nicaragua, due to its rapid growth, high-quality flesh, and low dietary protein requirements, which position it as a native alternative to tilapia and a tool for mitigating ecological impacts from exotic species (Vega-Villasante et al., 2021; Aréchiga-Palomera et al., 2024).
Despite its ecological and economic importance, fundamental aspects of D. latifrons reproduction remain poorly unknown (Vega-Villasante et al., 2021). Reported data on reproductive mode, spawning seasonality, oocyte structure, fecundity, sex ratio, and gonadal development are fragmented. Previous studies have described an empirical, macroscopic, eight-stage maturation scale for females (Tresierra Aguilar et al., 2002) and reported the size at first sexual maturity for males (15.5 cm total length (TL); 60 g body weight (BW)) and females (20.5 cm TL; 105 g BW) in Ecuador (Chang, Navas, 1984), as well as females (~13.2 cm TL) in Mexico (Violante-González et al., 2016). However, macroscopic methods are often subjective and may misclassify maturation stages (Jakobsen et al., 2009), reinforcing the need for histological validation (Brown-Peterson et al., 2011). We hypothesize that the integration of macroscopic and histological analyses will allow for a more precise and comprehensive characterization of gonadal development and reproductive traits in D. latifrons populations from Ecuador. This knowledge is essential for informing sustainable management strategies and advancing aquaculture development for this native species. Specifically, this study aims to: (1) characterize the macroscopic and microscopic stages of gonadal development in both sexes; (2) describe the morphology of the genital papilla and oocytes; and (3) estimate key reproductive parameters, including sex ratio, size at sexual maturity, and batch fecundity. These findings will contribute with valuable insights to enhance resource management and support the implementation of sustainable aquaculture practices.
Material and methods
Sampling. This study was conducted in the Carrizal-Chone River system, in the area between the upper part of the Chone River estuary (00°42’04.15”S 80°13’40.35”W) and the La Segua Wetland (00°42’33.00”S 80°11’54.43”W), belonging to Manabí province, Ecuador. Nine samplings were performed, five in the rainy (January-May 2018) and four in the dry (December 2017 and June-August 2018) season, collecting 295 specimens (151 males and 144 females) of D. latifrons. During samplings, the animals were captured randomly by fishermen using cast nets and transported alive to the laboratory, where they were analysed. Although the specimens examined in this study were discarded, they were previously identified with taxonomic keys (Ruiz-Campos, 2012; Jiménez-Prado et al., 2015; Froese, Pauly, 2024) and compared with reference specimens deposited in the Ichthyology Collection of the Laboratório de Biologia e Genética de Peixes (LBP), Universidade Estadual Paulista, Botucatu, Brazil (LBP 29080), and in the Ichthyology Collection of the Universidad Técnica de Machala, Ecuador (UTMACH 365, 372, 373, 377, 378, 381, 383, 394, 395) (Paim et al., 2020).
Sampling procedures and data analysis. The animals were anesthetized with 170 mg L-1 of Eugenol (Zambrano-Bermúdez et al., 2023) prior to be euthanized using cerebral puncture following bioethical norms. Subsequently, a dissection of reproductive organs (gonads) was performed. Total length (TL in cm), total body weight (W in g) and gonadal weight (GW in g) were recorded for each animal. Sex was determined based on the external morphological characteristics of the genital papilla (male: triangular and female: subrectangular) and gonads (Vega-Villasante et al., 2021). The length and width of the genital papillae and gonads of both sexes were measured (cm) with a Vernier calliper. The gonadosomatic index (GSI) was calculated as:
GSI = (GW/W) x 100
Where GW = gonad weight (g) and W = total body weight (g)
Fragments of gonad tissue were fixed in 10% neutral buffered formalin for histological analysis. The histological samples were processed using the paraffin inclusion technique, sectioned at 5 µm, stained with haematoxylin and eosin and mounted on slides with toluene (Wijayanti et al., 2017). The slides obtained were analysed using an Olympus BX53 optical microscope (Olympus America Inc., USA). The images were taken with the help of an 18 MP AmScope MU108 camera (United Scope LLC., USA) coupled to an optical microscope. For interpreting germ cell stages and phases of gonadal development, we used the standardised scale for teleost fish proposed by Brown-Peterson et al. (2011) for females and males; briefly: I “Immature”, II “Developing, III “Spawning capable”, IV “Regressing” and V “Regenerating”. A total of 250 oocytes per female, from gonad sections of 20 mature females, were randomly measured using the ImageJ program (v. 1.45). A total of 30 unfertilised eggs were also measured.
Data and statistical comparisons. Differences on TL and W between sex; length and width papillae among gonad development phase, and GSI among months were analyzed using one-way ANOVA. Statistica v. 7 software was used for statistical processing. Analysis of covariance (ANCOVA) was also used to compare the condition (weight-length relationship) and gonadal weight between sex categories using fish total length as the covariate. Quantitative variables were log-transformed, when necessary, for parametric analyses. The results are expressed as average and standard deviation. Size-at-maturity (L50) of a population is the total length at which 50% of the individuals are mature, and it was estimated by fitting a logistic regression for each sex, using maturation phases as the qualitative response variable (mature vs. immature) and fish total length as quantitative predictor (Fontoura et al., 2009; Barzotto, Mateus, 2017). This model allows the estimation of the probability of a fish to be mature according to its total length. Logistic models were fitted using SPSS 27.0.
Results
In total, males and females comprised 51% and 49% of D. latifrons caught, respectively. The male:female ratio was not significantly different (Chi-square test, χ2 = 0.11, p = 0.74, n = 295) and the sex ratio was 1.05:1. The mean total length of males (21.04 ± 6.07 cm, n = 151) and the mean body weight (20.00 ± 5.69 g, n = 144) did not show significant differences (F1,295 = 3.15, p = 0.077) between sex.
Morphological characteristics of the genital papilla. The length and width of the genital papillae were significantly smaller in females than in males (F1,254 = 10.02, p = 0.002, F1,254 = 4.08, p = 0.044; respectively) (Tab. 1). Additionally, both length and width showed significant differences among phases of gonadal development (F4,254 = 11.47, p = 0.001, F4,254 = 10.06, p = 0.001; respectively). Post-hoc analyses revealed that papillae were significantly smaller in phases I and II compared to phases III, IV and V.
TABLE 1 | Morphometric data of genital papilla on Dormitator latifrons according to gonad development phase. Mean length (cm) and mean width (cm), with the range shown in parenthesis. n = number of fish sampled. I: Immature, II: Developing, III: Spawning capable, IV: Regressing, V: Regenerating.
| Females | Males | ||||
Phase | n | Length | Width | n | Length | Width |
I | 18 | 0.40 (0.10–0.60) | 0.26 (0.10–0.50) | 10 | 0.54 (0.10–0.90) | 0.31 (0.10–0.60) |
II | 6 | 0.67 (0.30–0.90) | 0.45 (0.20–0.70) | 13 | 0.67 (0.40–0.90) | 0.39 (0.30–0.70) |
III | 109 | 0.86 (0.20–1.40) | 0.59 (0.20–1.00) | 114 | 0.89 (0.30–1.80) | 0.55 (0.20–1.80) |
IV | 6 | 0.67 (0.30–1.20) | 0.46 (0.10–0.80) | 5 | 0.77 (0.60–0.90) | 0.43 (0.30–0.50) |
V | 5 | 0.55 (0.30–0.80) | 0.35 (0.20–0.50) | 9 | 1.02 (0.80–1.40) | 0.64 (0.50–0.90) |
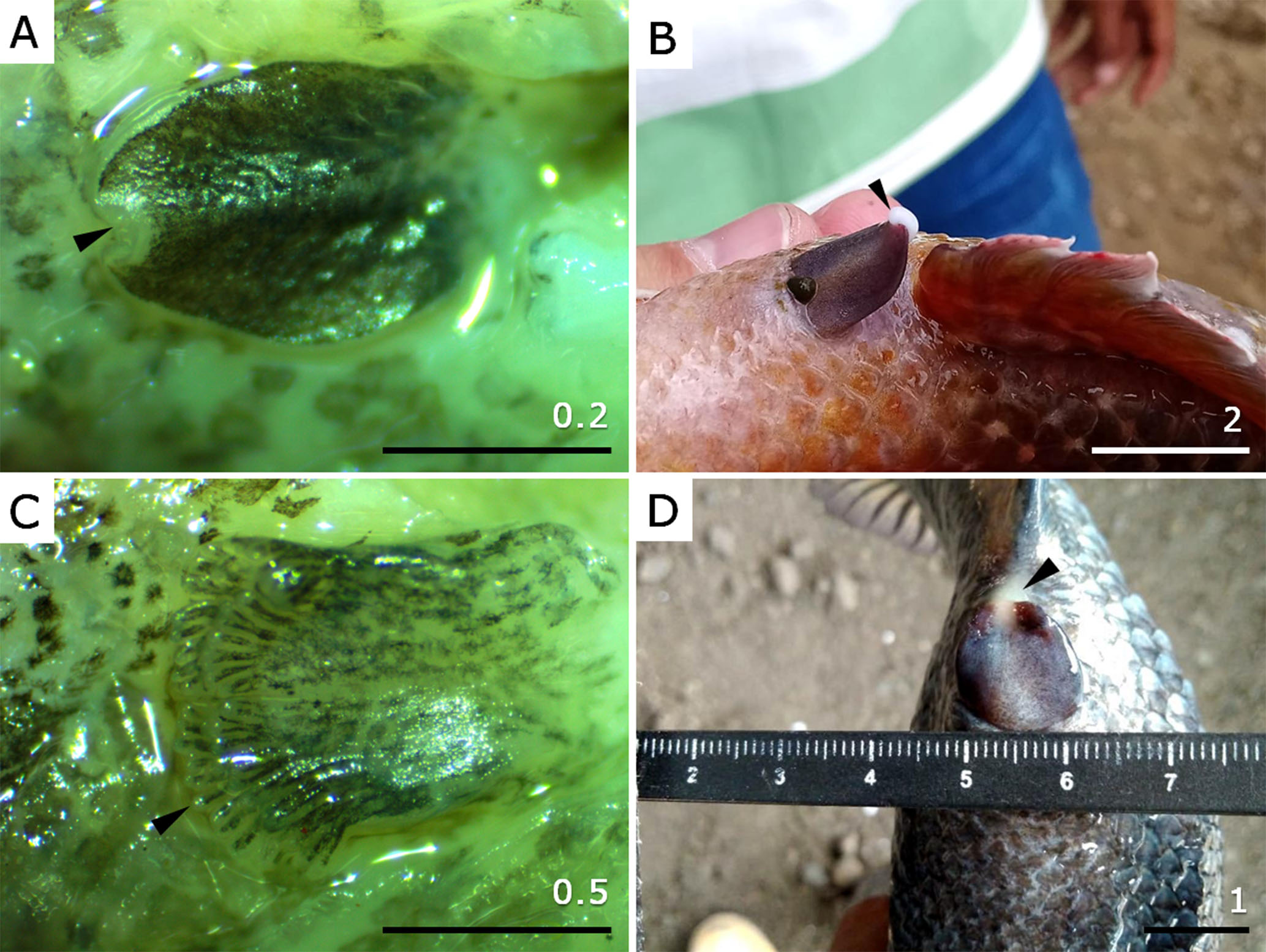
Morphologically, the genital papillae changed noticeably throughout gonadal maturation. In males at phases I and II of gonad development, the papilla is flat with sharp edges and oval appearance and a black-spotted colouration pattern (Fig. 1A). As male progress to mature stages, the papilla elongates and assumes a well-defined triangular shape. In mature males (phases III-V), it becomes oedematous with homogeneous colouration that transitions from predominantly yellowish at maturation to violet-yellow in the centre and dark wine on the edges (Fig. 1B). In females at phases I and II of gonad development, the papilla is flattened and sub-rectangular in shape; and also, presents a black-spotted colouration pattern, including digitiform villi (Fig. 1C). Upon maturation, the papilla elongates while maintaining its sub-rectangular form but with more rounded edges. It also becomes oedematous and changes colour, shifting from predominantly pale pink at maturation to dark violet, mainly at the edges, in mature (phases III-V) females (Fig. 1D). The villi show the most pronounced change, increasing in size and acquiring a wine coloration.
FIGURE 1| Genital papillae in immature and mature Dormitator latifrons males and females. A. Immature male with oval, flattened papilla, black-spotted pigmentation pattern, and white sphincter (arrowhead). B. Mature male showing elongated, triangular papilla with homogeneous violet-yellow colouration and presence of semen (arrowhead) at the genital pore. C. Immature female with flattened, sub-rectangular papilla and black-spotted pigmentation, including the digitiform villi (arrowhead). D. Mature female with oedematous, sub-rectangular papilla, and dark violet coloration; egg mass visible at the genital pore (arrowhead). Scale in cm.
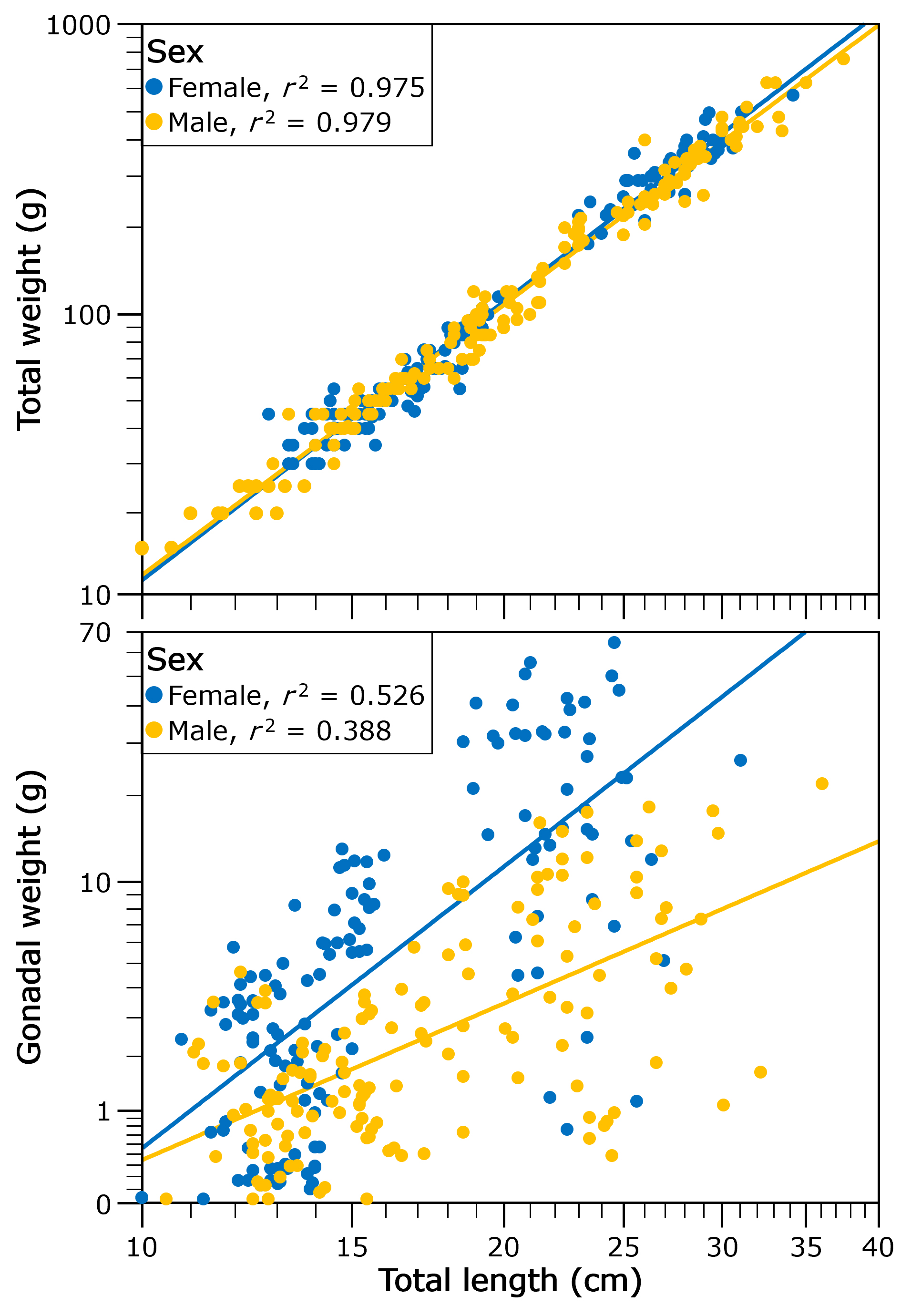
Morphological characteristics of the gonads. The gonads showed a significant increase in weight in phase III (F4,282 = 8.09, p = 0.001), while the length of both gonads (Fright gonad 4,279 = 12.30, p = 0.001, Fleft gonad 4,279 = 11.95, p = 0.001) showed a gradual increase during phases I to III, and subsequently a significant decrease in length in phases IV and V (Tab. 2). The width of the right (F4,284 = 15.43, p = 0.001) and left (F4,284 = 14.21, p = 0.001) gonads showed significant differences between phases of gonadal development, showing a significant increase during phases I to III and a decrease during phases IV and V. After controlling total length, weight did not show significant differences between sexes (ANCOVA, F1, 289 = 2.29 p = 0.13; Fig. 2A) but gonadal weight was heavier in females than males for a given total length (F1, 281 = 4.96, p = 0.27; Fig. 2B).
TABLE 2 | Morphometric data of Dormitator latifrons ovary and testicle based on gonadal development phase. Mean length (cm), mean width (cm) and mean weight (g) are shown, with the range in parenthesis. n= fish sampled. I: Immature, II: Developing, III: Spawning capable, IV: Regressing, V: Regenerating.
Phase | Ovary | Testicle | ||||||
n | Length | Width | Weight | n | Length | Width | Weight | |
I | 18 | 3.13 (1.70–5.45) | 0.27 (0.10–0.65) | 0.30 (0.03–0.68) | 10 | 3.76 (1.80–4.95) | 0.34 (0.10–0.75) | 0.32 (0.02–1.08) |
II | 6 | 3.68 (2.75–5.25) | 0.57 (0.30–1.25) | 1.35 (0.70–5.41) | 13 | 5.26 (4.20–9.15) | 0.57 (0.80–1.05) | 0.92 (0.80–3.12) |
III | 109 | 6.90 (2.50–13.80) | 1.42 (0.15–3.70) | 13.52 (0.50–3.62) | 114 | 6.58 (2.65–12.50) | 1.31 (0.35–2.85) | 4.76 (0.30–21.68) |
IV | 6 | 5.13 (2.45–7.80) | 0.77 (0.30–1.25) | 2.75 (0.17–6.79) | 5 | 5.78 (4.35–8.35) | 0.53 (0.30–0.90) | 0.71 (0.16–1.11) |
V | 5 | 4.48 (2.15–6.80) | 0.40 (0.30–0.50) | 0.63 (0.14–1.11) | 9 | 8.51 (6.55–10.15) | 0.61 (0.40–0.85) | 2.17 (0.60–1.82) |
FIGURE 2| Relationship of total weight (A) and gonad weight (B) of both sexes with the total length of Dormitator latifrons.
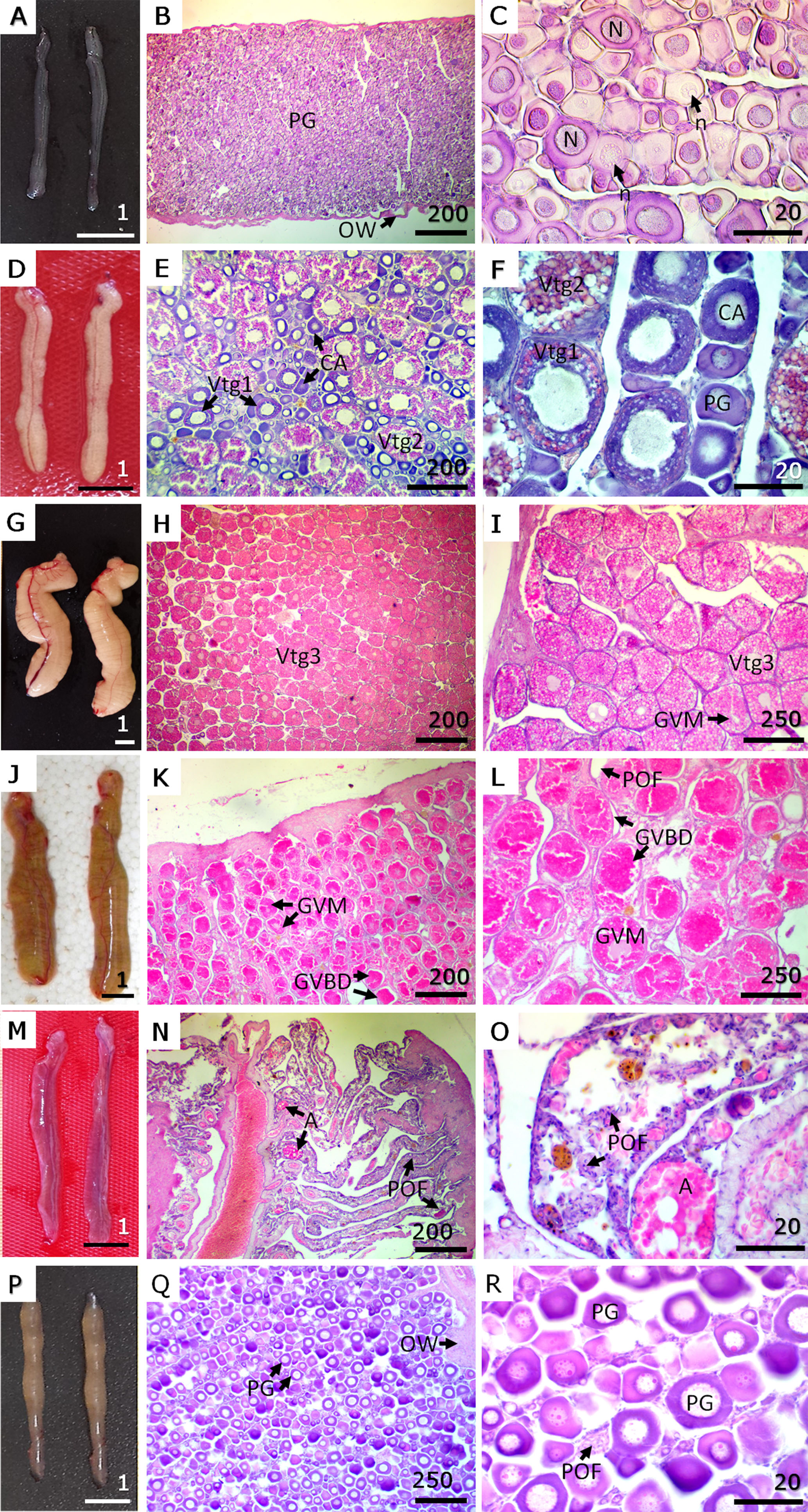
Oogenesis. In Phase I, ovaries were small, smooth, translucent to slightly whitish, and lacked visible blood vessels (Fig. 3A). Histologically, only oogonia and primary growth oocytes were observed, tightly packed without visible intercellular spaces. The ovarian wall was thin (~25 µm), and no postovulatory follicles or atretic oocytes were detected (Figs. 3B, C). In Phase II, ovaries appeared enlarged and cream-coloured, with a slightly granular texture, irregular edges, and more visible blood vessels (Fig. 3D). Microscopically, cortical alveolar oocytes and primary (Vtg1) and secondary vitellogenic oocytes (Vtg2) predominated. Primary growth oocytes and some atretic oocytes were also present, but no mature oocytes or postovulatory follicles were observed (Figs. 3E, F).
In Phase III, ovaries were large, turgid, and yellow to slightly orange, with irregular edges and well-defined blood vessels (Fig. 3G). Histological examination revealed a predominance of tertiary vitellogenic oocytes (Vtg3) and the onset of germinal vesicle migration (Figs. 3H, I). In a more advanced subphase of this stage, the active spawning subphase, ovaries appeared reddish and slightly flaccid (Fig. 3J). Histologically, this subphase showed the presence of mature oocytes (MGV, BDGV) and postovulatory follicles, with abundant atretic oocytes, while cortical alveolar and primary growth oocytes were scarce (Figs. 3K, L). In Phase IV, ovaries were smooth, flaccid, reddish, and translucent, with prominent blood vessels (Fig. 3M). Histologically, postovulatory follicles and atretic oocytes predominated, and blood vessels appeared congested. Primary growth oocytes were still present, and the ovarian wall was thickened (Figs. 3N, O). Finally, in Phase V, ovaries were thin, whitish translucent, and showed reduced vascularisation (Fig. 3P). Histologically, primary growth oocytes, atretic oocytes, and postovulatory follicles were present, and the ovarian wall was markedly thick (~76 µm) (Figs. 3Q, R).
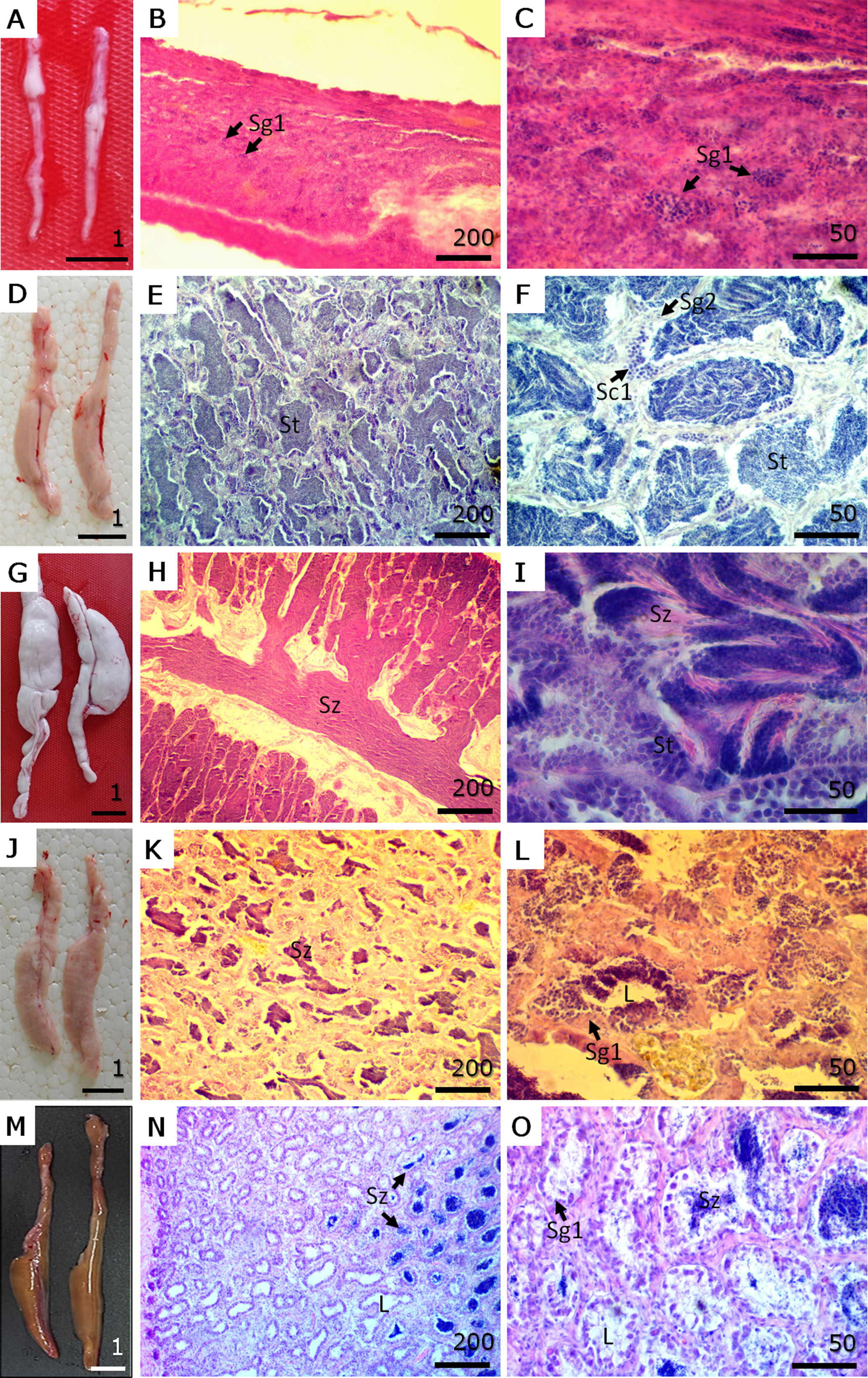
Spermatogenesis. In D. latifrons males, five reproductive phases were identified based on macroscopic and histological features. In Phase I (immature), testes were thin, elongated, translucent, and whitish (Fig. 4A). Histologically, the seminiferous tubules were poorly defined and lacked lumens, with only primary spermatogonia (Sg1) present (Figs. 4B, C). In Phase II (developing), testes increased in size, became creamy white, and showed visible vascularization (Fig. 4D). Microscopically, small but well-structured seminiferous tubules were evident, containing secondary spermatogonia (Sg2), spermatocytes, and sperm in various developmental stages, although spermatozoa were not yet observed in the lumen or sperm ducts (Figs. 4E, F). Phase III (spawning capable) was characterized by large, turgid, milky white testes with pronounced vascularization (Fig. 4G). Histologically, spermatogenesis was active, and abundant spermatozoa were found in the tubule lumens and sperm ducts (Figs. 4H, I). In Phase IV (regressing), the testes appeared smaller, opaque, flaccid, and pink to reddish (Fig. 4J), with halted spermatogenesis and a noticeable decrease in spermatocytes and spermatozoa within the ducts and lumens (Figs. 4K, L). Finally, in Phase V (regenerating), the testes were markedly reduced in size, flaccid, and reddish to brown (Fig. 4M). Primary spermatogonia (Sg1) reappeared, and residual spermatozoa (Ez) were observed in the tubule lumens and spermatic ducts, with most tubules now exhibiting a clear lumen (Figs. 4N, O).
FIGURE 3| Macroscopic view and histological sections of gonads in each phase of the reproductive cycle of female Dormitator latifrons. A. Phase I – Immature. B. Low magnification of immature ovary. C. High magnification of immature ovary. D. Phase II – Developing. E. Low magnification of developing ovary. F. High magnification of developing ovary. G. Phase III – Spawning capable. H. Low magnification of an ovary in spawning capable phase. I. High magnification of an ovary in spawning capable phase. J. Phase III – actively spawning subphase of the spawning capable reproductive phase. K. Low magnification of an ovarian in actively spawning subphase. L. High magnification of an ovarian in actively spawning subphase. M. Phase IV – Regressing. N. Low magnification of an ovary in regressing phase. O. High magnification of an ovary in regressing phase. P. Phase V – Regenerating. Q. Low magnification of an ovarian in regenerating phase. R. High magnification of an ovarian in regenerating phase. PG = primary growth oogonia; OW = ovarian wall; N = nucleus; n = nucleolus; CA = cortical alveolar oocytes; Vtg1 = primary vitellogenic oocyte; Vtg2 = secondary vitellogenic oocytes; Vtg3 = tertiary vitellogenic oocytes; GVM = germinal vesicle migration; GVBD = germinal vesicle breakdown; POF = postovulatory follicles; A = atretic oocytes. Macroscopic view (A, D, G, J, M, P) scale in cm. Microscopic view (Staining H-E) in µm.
FIGURE 4| Macroscopic view and histological sections of gonads in each phase of the reproductive cycle of male Dormitator latifrons. A. Phase I – Immature. B. Low magnification of immature testicle. C. High magnification of immature testicle. D. Phase II – Developing. E. Low magnification of developing testicle. F. High magnification of developing testicle. G. Phase III – Spawning capable. H. Low magnification of a testicle in spawning capable phase. I. High magnification of a testicle in spawning capable phase. J. Phase IV – Regressing. K. Low magnification of a testicle in regressing phase. L. High magnification of a testicle in regressing phase. M. Phase V – Regenerating. N. Low magnification of a testicle in regenerating phase. O. High magnification of a testicle in regenerating phase. Sg1 = primary spermatogonia; Sg2 = secondary spermatogonia; Sc1 = primary spermatocyte; St = permatid; Sz = spermatozoa; L = lumen of lobule. Macroscopic view (A, D, G, J, M) scale in cm. Microscopic view (Staining H-E) scale bars in µm.
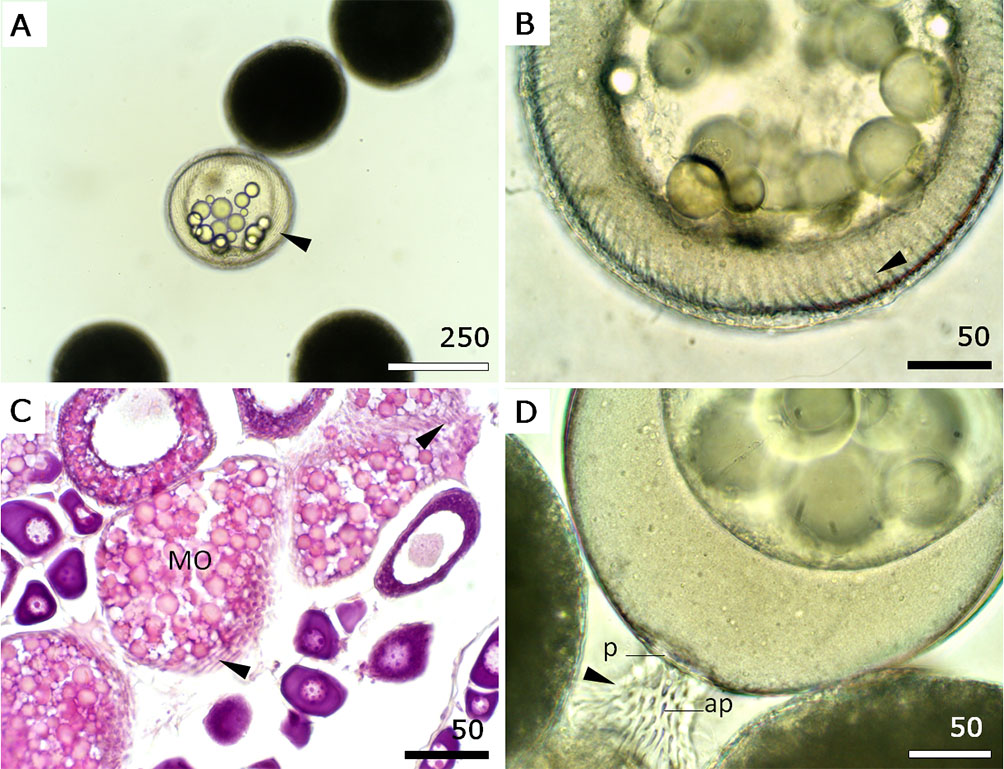
Unfertilised egg. Average diameters of oocytes in the primary growth, cortical alveoli, secondary vitellogenesis, tertiary vitellogenesis, mature (stages MGV and BDGV) and unfertilised egg stages were 39.16 ± 5.66, 76.00 ± 5.04, 106.19 ± 12.96, 163.67 ± 8.57, 183.83 ± 5.18, and 337.88 ± 43.72 µm, respectively. Unfertilised oocytes were round, with a thin capsule without perivitelline space, a variable number (4–8) of oil drops and longitudinal striations under the capsule (Figs. 5A, B). These striations, which can be also observed in histological sections of vitellogenic oocytes (Fig. 5C), are filaments forming a membrane project after spawning as part of an adhesion apparatus (Fig. 5D). The projected membrane has a hyaline aspect, with apertures (ap) that become larger as they move away from the stalk and end in a bundle filament (Fig. 5D).
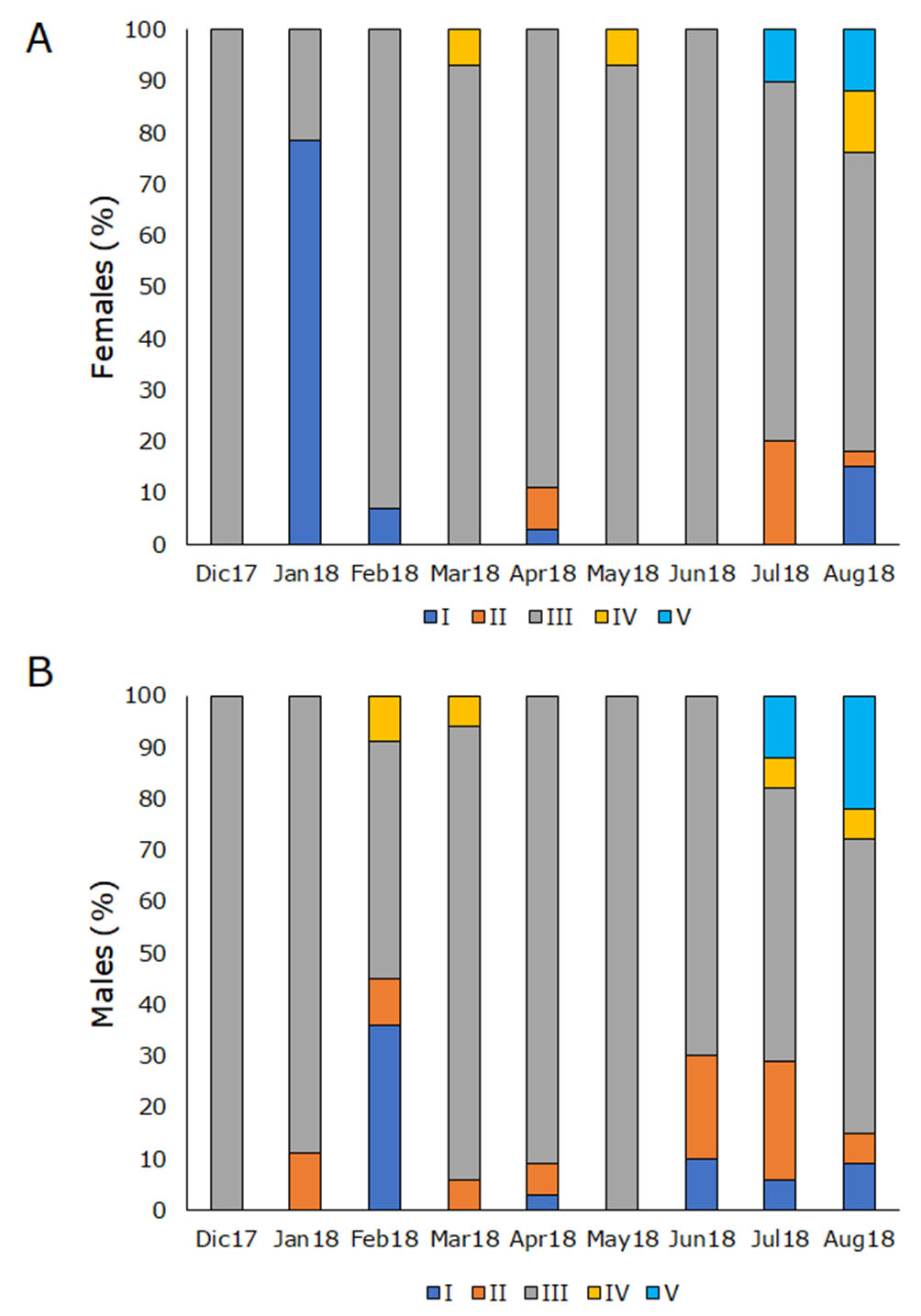
Reproductive parameters. Mature females and males (phase III) were observed in high proportion (55–100%) throughout the whole sampling time (Fig. 6). Gonads of females at phase I were mostly recorded in January while males were observed in February (Fig. 6). Phases IV and V in both sexes were observed in the dry season (July-August) (Fig. 6).
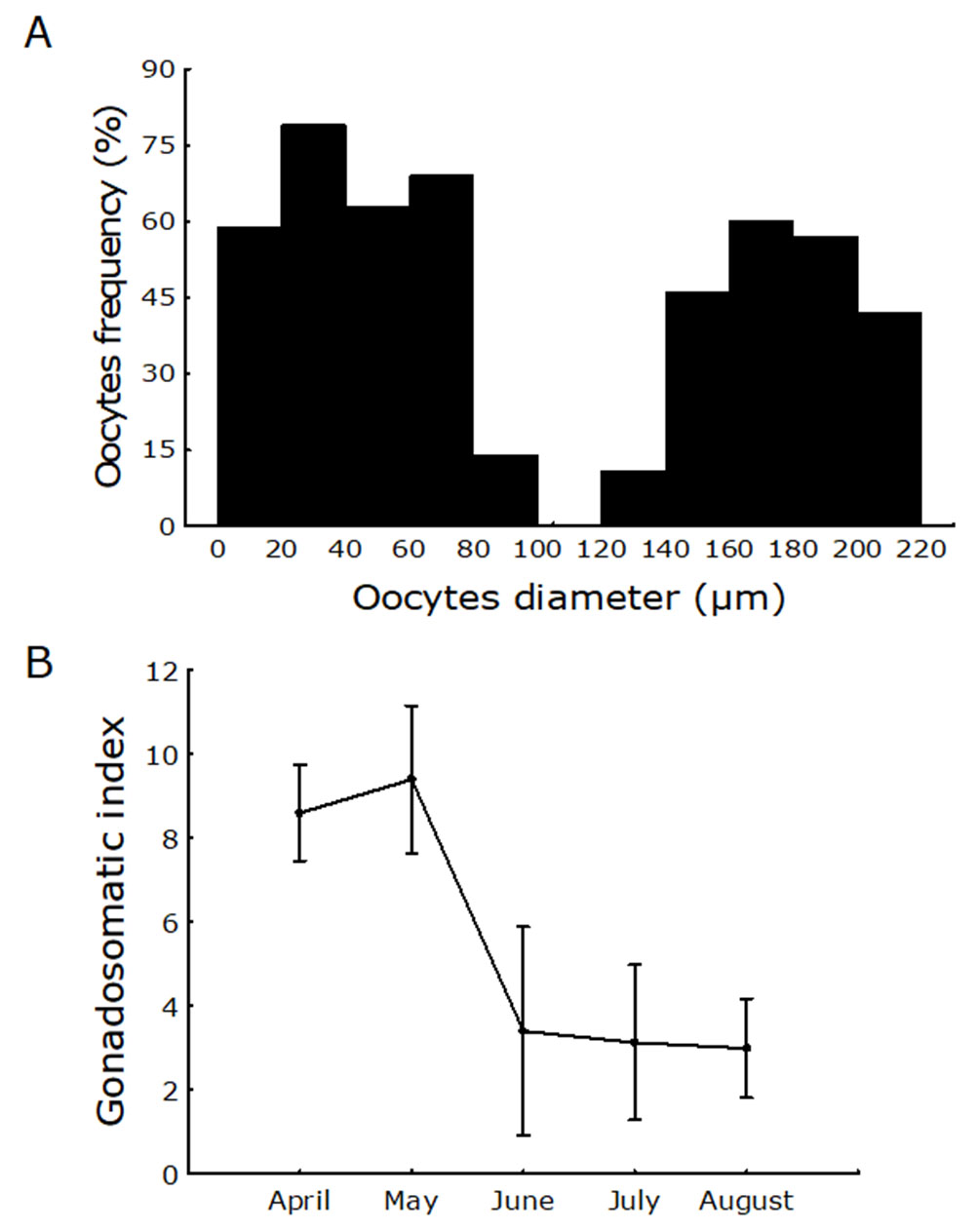
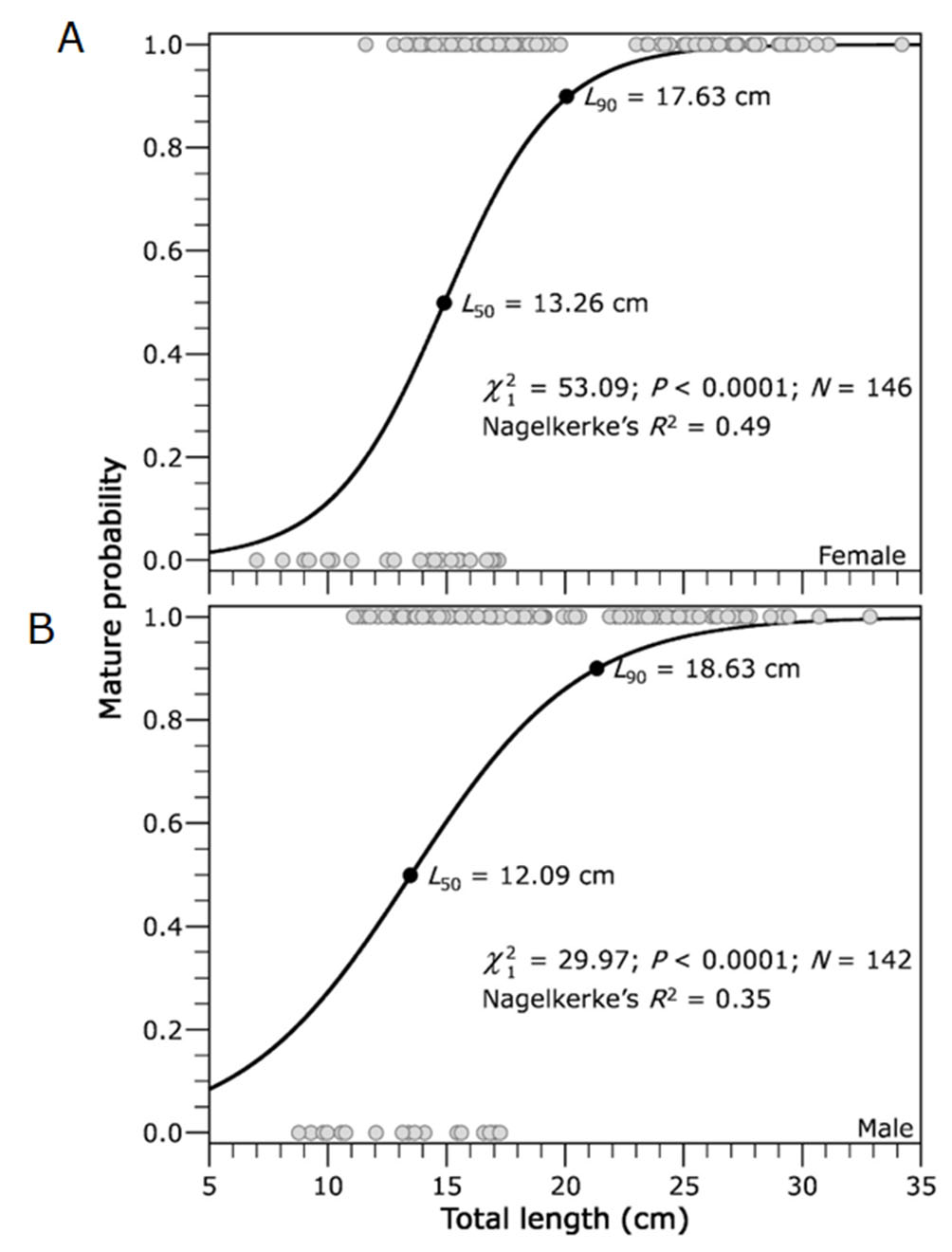
The size frequency distribution of oocytes showed two populations of oocytes in the ovary at the same time, a population of mature oocytes and a population of smaller immature oocytes (Fig. 7A). The GSI was significantly higher during the months of April and May (rainy season) than in the months of June through August (dry season) (F4,204 = 18.30, p=0.001) (Fig. 7B). Females begin to mature with smaller sizes (7 cm) than males (8.7 cm); The L50 for both females and males was 13.26 cm and 12.09 cm, respectively (Fig. 8).
FIGURE 5| Morphology of unfertilised Dormitator latifrons eggs. A. An unfertilised egg showing round shape and striation (arrowhead) under the capsule. B. Detailed striations (arrowhead) from A and C. C. Histological section of the ovary showing striations (arrowhead) in mature oocytes (MO). H&E stain. D. Unfertilised eggs showing the adhesion apparatus in the pedicle (P). Note the apertures (ap) in the hyaline membrane (arrowhead). Scale bars: A = 250 µm; B–D = 50 µm.
FIGURE 6| Relative frequencies of gonadal development stages of Dormitator latifrons. A. Females and B. Males. I: Immature; II: Developing; III: Spawning capable; IV: Regressing and V: Regenerating.
FIGURE 7| A. Frequency distribution of oocyte size in mature females of Dormitator latifrons. B. The gonadosomatic index (GSI) of D. latifrons from April to August of 2018.
FIGURE 8| Relationship between maturity stage of both female (A) and male (B) of Dormitator latifrons with total length (0: immature and 1: mature).
Discussion
The reproductive biology of D. latifrons shows strong affinities with other Eleotrids and Gobiiformes, characterized by complex gonadal development and marked sexual dimorphism in genital morphology and reproductive behaviour. In the present study, analysis of 259 individuals revealed a sex ratio of 1.05:1 (males:females), closely matching the 1:1 ratio reported by Chang, Navas (1984) for the same area more than three decades ago. Despite long-standing fishing pressure in the Carrizal-Chone River System, where population declines have been documented (Stram et al., 2005; Gonzalez-Martinez et al., 2020), this balanced sex ratio suggests that current artisanal fishing practices, lacking sexual selectivity, have not yet disrupted the natural sexual structure of the population. Moreover, a near 1:1 sex ratio can be considered a positive indicator of the specie´ persistence in the region, as it ensures the presence of reproductive partners even under fishing pressure. Nevertheless, the persistent harvest of wild juveniles for aquaculture fattening (FAO, 2010), coupled with the unregulated capture of subadults and adults for consumption (Vega-Villasante et al., 2021), poses a potential threat to long-term reproductive output. To safeguard population sustainability, management measures such as enforcing seasonal closures during peak spawning, implementing minimum size limits, and discouraging the capture of reproductive individuals during the rainy season should be considered. In parallel, the development of government-supported breeding programs to complete the species’ reproductive cycle in captivity could provide juveniles for aquaculture without depleting wild stocks.
Beyond population structure, our findings also reveal marked morphological differences between sexes, providing key insights into the reproductive biology of the species. The morphology of the genital papilla in D. latifrons exhibits clear sexual dimorphism: males possess smooth, triangular papillae, whereas females display a sub-rectangular structure with prominent finger-like villi. Additionally, our observations indicate that these external structures vary in size and coloration across reproductive phases, suggesting their potential as reliable and non-invasive indicators of sexual maturity. This is particularly advantageous in D. latifrons, where conventional ovarian biopsy is hindered by the reduced size and anatomical inaccessibility of the genital pore (Rodríguez-Montes de Oca et al., 2012), and where invasive methods may induce significant stress or physical harm to the specimens. Similarly, in the bigmouth sleeper (Gobiomorus dormitor), papilla erection and reddish coloration indicate imminent spawning readiness, suggesting that external morphology may serve as a stress-reducing signal and determine reproductive timing (Harris et al., 2011).
The variability observed in the genital papilla of D. latifrons fits within a broader pattern documented in other gobioids, where morphological changes are closely linked to gonadal status and hormonal regulation. In bidirectional sex-changing gobies such as Trimma okinawae, the urogenital papillae and associated genital ducts undergo morphological changes during sexual transition, a process influenced by sex steroid hormones, particularly androgens, suggesting a physiological link between papilla morphology and gonadal status (Kobayashi et al., 2012). Adopting genital papilla morphology as a favorable indicator for field breeding can significantly improve broodstock selection for D. latifrons, minimizing management stress and aligning reproductive interventions with fish welfare and sustainable management objectives. In this context, we propose a methodological approach for future research and management initiatives that integrates the use of our sexual maturity scale, coupled with precise morphometric analysis of the genital papilla. This strategy could serve not only as a practical tool for broodstock selection in aquaculture programs but also as a non-invasive biomonitoring indicator of reproductive dynamics in natural populations, especially under changing environmental conditions.
Histologically, D. latifrons exhibits a group-synchronous ovarian development, characterized by two distinct oocyte cohorts: one composed of mature oocytes at the germinal vesicle stage and another of smaller, less-developed oocytes. Our findings suggest that females may follow a total spawning strategy, releasing all mature oocytes in a single reproductive event over a short period. This hypothesis is supported by the absence of late vitellogenic oocytes in ovaries during the regression phase, indicating complete spawning before ovarian regression. Similar patterns of group-synchronous development have been reported in other tropical gobioid species, including Cotylopus acutipinnis (Teichert et al., 2015), Sicyopterus lagocephalus (Teichert et al., 2014), and Gobius paganellus (Louiz et al., 2013), and are considered adaptive responses to seasonal environments. In these cases, oocyte maturation occurs in advance, allowing females to spawn synchronously once environmental conditions, such as rainfall or hydrological shifts, become favourable. Thus, while reproductive activation in D. latifrons appears to be environmentally triggered, the ovarian structure reflects a preparatory strategy that enables rapid spawning in response to external cues. Further population-level validation is needed to confirm this reproductive pattern. By reducing the need to sacrifice specimens, this histological approach also supports more ethical and sustainable research practices. Ultimately, our findings offer a baseline for refining management actions, such as seasonal harvest bans covering the spawning period, post-spawning harvest bans or spatial protections, to safeguard reproductive potential and ensure the long-term sustainability of D. latifrons populations.
This pattern in females contrasts with the gonadal organization observed in males, which exhibits distinctive histological traits that also influence the species’ reproductive strategy. In males of D. latifrons, the testes exhibit a lobular-type structure at the histological level, with seminiferous tubules arranged radially from the dorsal and lateral walls toward a central lumen, an organization similar to that described in tilapia (Patimar et al., 2011). In teleost fishes, this lobular organization is associated with cystic spermatogenesis, wherein germ cells develop within discrete cysts that release mature spermatozoa synchronously into the lobular lumen (Caspers, 1984; Pecio et al., 2001; Uribe et al., 2014). Such morphology likely facilitates coordinated release of large quantities of sperm, which is advantageous during mass spawning events or tightly timed reproductive periods. From an applied standpoint, understanding this structural pattern enables targeted and ethical broodstock management: monitoring reproductive phases through histological or non-invasive assessments allows aquaculturists to optimize the timing for sperm collection and induce spawning under controlled conditions. Concurrently, recognizing phases of regression and regeneration can inform the establishment of post-spawning rest periods, minimizing overexploitation and enhancing stock sustainability. These biological insights thus bridge anatomical knowledge with practical strategies for both aquaculture development and the sustainable management of wild D. latifrons populations.
These adaptations in sperm production and release are complemented by oocyte characteristics that optimize fertilization success and recruitment under variable environmental conditions. In D. latifrons, the newly spawned oocytes averaged 337.9 ± 43.7 µm in diameter, substantially smaller than those of other eleotrid species such as D. maculatus (Flores-Coto, Garcia, 1983). These small, spherical eggs equipped with a filamentous adhesive apparatus facilitate substrate attachment, a trait fundamental for demersal spawning strategies. While larger eggs generally confer higher survival rates (Einum, Fleming, 2000), the production of numerous small eggs, as observed in D. latifrons and other gobioids like Hypseleotris sp., Eleotris spp., and E. fusca (Lake, 1967; Anderson et al., 1971; Auty, 1978; Maeda et al., 2008) supports an opportunistic strategy that maximizes reproductive output under fluctuating environmental conditions (Miller, 1984). Furthermore, producing many small eggs should be more advantageous in terms of future recruitment than producing a smaller number of large eggs that can be more easily detected and preyed upon (Connaughton, Aida, 1999). For aquaculture, these findings suggest that high fecundity and efficient seed production could be achieved with minimal broodstock, given the moderate egg size and adhesive properties that ease handling and incubation. In wild management, understanding that D. latifrons invests in quantity over per-egg quality can guide conservation efforts, populations may be vulnerable to recruitment bottlenecks if larval substrate quality or availability is compromised. Maintaining optimal habitat complexity (e.g., rocky substrates or structured vegetation) is therefore crucial to ensure larval survival and sustain wild stocks. Additionally, contamination from plastic waste (including micro- and nanoplastics) and siltation can degrade substrate quality, obstruct larval settlement, cause physiological stress, reduce oxygenation at the substrate surface, or physically smother benthic surfaces. Studies have shown that microplastics accumulate in sediments and on benthic surfaces, where they negatively affect organismal health and reproduction (Osuna-Laveaga et al., 2023). Consequently, conservation strategies should also include measures to reduce plastic pollution and control sediment runoff in the habitats of D. latifrons.
The combination of high fecundity and egg adhesion ensures that reproductive success is synchronized with the most favourable environmental windows, as reflected in the species’ annual cycle. Our data show that the reproductive cycle of D. latifrons is nearly continuous, with mature individuals recorded year-round and clear peaks in gonadal activity during the rainy season (April-May), as indicated by elevated gonadosomatic index (GSI) values. Such seasonal reproductive patterns, synchronized with environmental cues like rainfall and hydrological fluctuations, are common among tropical freshwater fishes and have been documented in multiple gobioid taxa and broader fish assemblages (Chang, Navas, 1984; Dinh et al., 2016, 2022; Nordlie, 1981). These results suggest that in our study area, spawning events in D. latifrons are stimulated by hydrological changes during rainy season, particularly the rise in freshwater levels and the downstream displacement of the saline plume, which improve spawning and nursery conditions in the estuary. However, our dataset was obtained from a limited number of samples and a relatively short observation period. Therefore, while our findings provide initial insights into reproductive periodicity, broader studies encompassing larger sample sizes and longer temporal scales are necessary to confirm these patterns at the population level. From a management standpoint, our results support the potential implementation of seasonal bans to protect reproductive aggregations and enhance recruitment potential. Moreover, aligning broodstock collection with natural reproductive cycles may improve aquaculture success while minimizing pressure on wild stocks.
Understanding reproductive seasonality becomes even more relevant when accurately determining size at first maturity, a key parameter for fishery regulation and sustainable management. Sexual maturity data for D. latifrons have been remarkably scarce, as highlighted by Froese, Pauly (2021), underscoring the limited knowledge of the species’ reproductive parameters. Our analysis revealed that the smallest mature females and males measured 7 cm and 8.7 cm in total length, respectively. These values are considerably lower than the 13.2 cm TL reported by Violante-González et al. (2016) for females in Laguna Tres Palos, Guerrero state, Mexico. However, their study did not explicitly define the criteria for determining sexual maturity, which may have led to an underestimation of the smallest mature females. Furthermore, our results are significantly below the 20.5 cm TL reported by Chang, Navas (1984) for the Carrizal-Chone system, Manabí, Ecuador. Their methodology was based on abdominal massage, a less precise technique compared to the histological analysis employed in our study, which provides a more accurate assessment of reproductive capacity. The presence of oocytes at vitellogenic stage 3, indicative of readiness to spawn, validates that 50% of mature females and males of 13.26 cm and 12.09 cm TL, respectively, are capable of reproducing during the current reproductive period. This histological validation represents a crucial update and serves as a reference for future biological-fishery studies on D. latifrons, to establish maturation values based on L50 or higher to guide sustainable management practices.
In conclusion, this study offers the first comprehensive, histologically validated account of the reproductive biology of D. latifrons, integrating gonadal development, genital morphology, and reproductive periodicity. The marked sexual dimorphism in genital papilla morphology, combined with its size and colour changes across reproductive phases, emerges as a practical, non-invasive indicator of sexual maturity, reducing the need for harmful sampling. Histological assessment confirmed a lobular testicular organization and group-synchronous ovarian development, with evidence supporting a highly coordinated spawning strategy, likely involving synchronized gamete release during environmentally triggered reproductive peaks. These traits, together with smaller adhesive eggs and high fecundity, suggest an opportunistic reproductive strategy adapted to fluctuating freshwater environments. From an applied perspective, our results provide essential reference points for broodstock selection, optimal timing of gamete collection, and the design of seasonal closures, minimum size limits, and habitat protection measures. By linking detailed anatomical and histological insights with actionable management recommendations, this work establishes a foundation for both sustainable aquaculture development and the conservation of wild D. latifrons populations.
Acknowledgments
We are grateful artisanal fishermen from La Segua wetland and Network LARVAplus (ref. 117RT0521).
References
Anderson JR, Lake JS, Mackay NJ. Notes on reproductive behaviour and ontogeny in two species of Hypseleotris (Carassiops) (Gobiidae: Teleostei). Mar Freshw Res. 1971; 22(2):139–46.
Aréchiga-Palomera MA, Vega-Villasante F, Nieves-Rodríguez KN, Carrillo-Farnés O, Nolasco-Soria H. Characterization of the digestive enzymes of juvenile Dormitator latifrons (Richardson, 1844). Fish Physiol Biochem. 2024; 50(6):2535–50. https://doi.org/10.1007/s10695-024-01400-y
Auty EH. Reproductive behaviour and early development of the empire fish Hypseleotris compressus (Eleotridae). Mar Freshw Res. 1978; 29(5):585–97. https://doi.org/10.1071/MF9780585
Barzotto E, Mateus L. Reproductive biology of the migratory freshwater fish Salminus brasiliensis (Cuvier, 1816) in the Cuiabá river basin, Brazil. J Appl Ichthyol. 2017; 33(3):415–22. https://doi.org/10.1111/jai.13262
Bermúdez-Medranda AE, Ureña-Peralta MO, Cruz-Quintana Y, Lucas AR. Nutritional composition of Dormitator latifrons (Richardson, 1844, Perciformes, Eleotridae) and quality index method (QIM) scheme for determining its shelf life. J Food Sci Technol. 2025; 62:1383–91. https://doi.org/10.1007/s13197-024-06113-x
Brockmann FW. Seasonality of fishes in a South Florida brackish canal. Fla Sci. 1974; 37(2):65–70. https://www.jstor.org/stable/24319581
Brown-Peterson NJ, Wyanski DM, Saborido-Rey F, Macewicz BJ, Lowerre-Barbieri SK. A standardized terminology for describing reproductive development in fishes. Mar Coast Fish. 2011; 3(1):52–70. https://doi.org/10.1080/19425120.2011.555724
Caspers H. Takashi Hibiya (Editor): an atlas of fish histology. Normal and Pathological Features. Stuttgart/New York: Gustav Fischer Verlag; 1982. Int Rev Ges Hydrobiol Hydrogr. 1984; 69(3):406. https://doi.org/10.1002/iroh.19840690307
Castro-Rivera R, Aguilar GB, Girón Hernández JP. Conversión alimenticia en engordas puras y mixtas de popoyote (Dormitator latifrons Richardson) en estanques de cemento. Revista AquaTIC. 2005; 23:45–52.
Chang BD. Tolerances to salinity and air exposure of Dormitator latifrons (Pisces: Eleotridae). Rev Biol Trop. 1984; 32(1):155–57.
Chang BD, Navas W. Seasonal variations in growth, condition and gonads of Dormitator latifrons (Richardson) in the Chone River basin, Ecuador. J Fish Biol. 1984; 24(6):637–48. https://doi.org/10.1111/j.1095-8649.1984.tb04834.x
Connaughton MA, Aida K. Female reproductive system, fish. In: Knobil E, Neil JD, editors. Encyclopedia of reproduction. New York: Academic Press; 1999. p.193–204.
Cho HC, Hwang IJ, Baek HJ. Histological Analysis of early gonadal development and sex differentiation in chameleon goby, Tridentiger trigonocephalus. Dev Reprod. 2014; 18(1):51–56. https://doi.org/10.12717/DR.2014.18.1.051
Dinh QM, Nguyen THD, Truong NT, Doan DX, Nguyen TTK. Ovarian development, spawning season, size at maturity and fecundity of Acentrogobius viridipunctatus (Valenciennes, 1837) in the Vietnamese Mekong Delta. PeerJ. 2022; 10:e14077. https://doi.org/10.7717/peerj.14077
Dinh QM, Qin J, Dittmann S, Tran DD. Reproductive biology of the burrow dwelling goby Parapocryptes serperaster. Ichthyol Res. 2016; 63(3):324–32. https://doi.org/10.1007/s10228-015-0502-7
Einum S, Fleming IA. Highly fecund mothers sacrifice offspring survival to maximize fitness. Nature. 2000; 405(6786):565–67. https://doi.org/10.1038/35014600
Food and Agriculture Organization of the United Nations (FAO). Peces nativos de agua dulce de América del Sur de interés para la acuicultura: Una síntesis del estado de desarrollo tecnológico de su cultivo. Ser Acuic En Latinoamérica: Numero 1. FAO; 2010.
Food and Agriculture Organization of the United Nations (FAO). FishStatJ – Software for fishery and aquaculture statistical time series. Statistics Team (NFISS), Fisheries and Aquaculture Division (NFI), FAO; 2020. Available from: https://www.fao.org/fishery/en/topic/166235/en
Flores-Coto C, Garcia FZ. Descripcion de huevos y larvas de Dormitator maculatus (Bloch) de la Laguna de Alvarado, Veracruz. (Pisces: Gobiidae). An Inst Cienc Mar Limnol. 1983; 91:127–40.
Fontoura NF, Braun AS, Milani PC. Estimating size at first maturity (L50) from gonadossomatic index (GSI) data. Neotrop Ichthyol. 2009; 7(2):217–22. https://doi.org/10.1590/S1679-62252009000200013
Froese R, Pauly D, editors. Dormitator latifrons: Pacific fat sleeper. Fishbase [Internet]; 2024. Available from: https://www.fishbase.se/summary/Dormitator-latifrons
Gonzalez-Martinez A, Lopez M, Molero HM, Rodriguez J, González M, Barba C et al. Morphometric and meristic characterization of native chame fish (Dormitator latifrons) in Ecuador using multivariate analysis. Animals. 2020; 10(10):1805. https://doi.org/10.3390/ani10101805
Graham MS, Haedrich RL, Fletcher GL. Hematology of three deep-sea fishes: A reflection of low metabolic rates. Comp Biochem Physiol A. 1985; 80(1):79–84. https://doi.org/10.1016/0300-9629(85)90682-6
Harris NJ, Neal JW, Perschbacher PW, Mace CE, Muñoz-Hincapié M. Notes on hatchery spawning methods for bigmouth sleeper Gobiomorus dormitor. Aquac Res. 2011; 42(8):1145–52. https://doi.org/10.1111/j.1365-2109.2010.02702.x
Jakobsen T, Fogarty MJ, Megrey BA, Moksness E. Introduction. In: Jakobsen T, Fogarty MJ, Megrey BA, Moksness E, editors. Fish reproductive biology: implications for assessment and management. Blackwell Publishing Ltd; 2009. p.1–08. https://doi.org/10.1002/9781444312133.ch
Jiménez-Prado P, Aguirre WE, Laaz Moncayo ED, Navarrete R, Nugra FI, Rebolledo Monsalve ER et al. Guia de peces para aguas continentales en la vertiente occidental del Ecuador. Pontificia Universidad Católica del Ecuador Sede Esmeraldas; 2015.
Keith P, Lord C. Tropical freshwater gobies: Amphidromy as a life cycle. In: Patzner R, Van Tassell JL, Kovacic M, Kapoos BG, editors. The biology of gobies. Boca Raton: CRC Press; 2011. p.243–77.
Kobayashi Y, Usami T, Sunobe T, Manabe H, Nagahama Y, Nakamura M. Histological observation of the urogenital papillae in the bi-directional sex-changing gobiid fish, Trimma okinawae. Zool Sci. 2012; 29(2):121–26. https://doi.org/10.2108/zsj.29.121
Lake JS. Rearing experiments with five species of Australian freshwater fishes. II. Morphogenesis and ontogeny. Aust J Mar Freshw Res. 1967; 18(2):155–76. https://doi.org/10.1071/MF9670155
Le Gac F, Loir M. Male reproductive system, fish. In: Knobil E, Neil JD, editors. Encyclopedia of reproduction. New York: Academic Press; 1999. p.20–30.
Louiz I, Mossadok B A, Ben Hassine OK. Some aspects of reproductive biology of Gobius paganellus (Gobiidae) on the northeastern coasts of Tunisia (Bizerta Lagoon). J Mar Biol Assoc United Kingdom (1000 Impact Factor). 2013; 93(8):2235–46. https://doi.org/10.1017/S0025315413000982
Maeda K, Yamasaki N, Kondo M, Tachihara K. Reproductive biology and early development of two species of sleeper, Eleotris acanthopoma and Eleotris fusca (Teleostei: Eleotridae). Pac Sci. 2008; 62(3):327–40. https://doi.org/10.2984/1534-6188(2008)62[327:RBAEDO]2.0.CO;2
Maitland PS. Freshwater fish distribution, by Tim M. Berra. Academic Press, San Diego, 2001. Aquat Conserv Freshw Ecosyst. 2003; 13(4):372. https://doi.org/10.1002/aqc.548
Massay S, Mosquera R. Presence of Chame Dormitator latifrom (Richardson, 1844) (Pisces: Eleotridae) in the Galapagos Islands, Ecuador. J Fish Biol. 1992; 40(5):815–16. https://doi.org/10.1111/j.1095-8649.1992.tb02627.x
McDowall RM. Early hatch: A strategy for safe downstream larval transport in amphidromous gobies. Rev Fish Biol Fish. 2009; 19:1–08. https://doi.org/10.1007/s11160-008-9085-y
Miller PJ. The tokology of gobioid fishes. In: Potts GW, Wootton RJ, editors. Fish reproduction: strategies and tactics. Academic Press; 1984. p.119–53.
Navarro-Rodríguez MC, González Guevara LF, Flores-Vargas R, González Ruelas ME, Carrillo González FM. Composición y variabilidad del ictioplancton de la laguna El Quelele, Nayarit, México. Rev Biol Mar Oceanogr. 2006; 41(1):35–43. http://dx.doi.org/10.4067/S0718-19572006000100006
Nelson JS, Grande TC, Wilson MVH. Fishes of the world. John Wiley & Sons; 2016. https://doi.org/10.1002/9781119174844
Nordlie F. Life-history characteristics of eleotrid fishes of the western hemisphere, and perils of life in a vanishing environment. Rev Fish Biol Fish. 2012; 22(1):189–224. https://doi.org/10.1007/s11160-011-9229-3
Nordlie FG. Feeding and reproductive biology of eleotrid fishes in a tropical estuary. J Fish Biol. 1981; 18(1):97–110. https://doi.org/10.1111/j.1095-8649.1981.tb03764.x
Osuna-Laveaga DR, Ojeda-Castillo V, Flores-Payán V, Gutiérrez-Becerra A, Moreno-Medrano ED. Micro- and nanoplastics current status: legislation, gaps, limitations and socioeconomic prospects for future. Front Environ Sci. 2023: 11:1241939. https://doi.org/10.3389/fenvs.2023.1241939
Paim FG, Nirchio M, Oliveira C, Rossi AR. Sex chromosomes and internal telomeric sequences in Dormitator latifrons (Richardson 1844) (Eleotridae: Eleotrinae): an insight into their origin in the genus. Genes. 2020: 11(6):659. https://doi.org/10.3390/genes11060659
Patimar R, Amouei M, Langroudi SMM-A. New data on the biology of Cobitis cf. satunini from the southern Caspian basin (northern Iran). Folia Zool. 2011; 60(4):308–14. https://doi.org/10.25225/fozo.v60.i4.a7.2011
Pecio A, Lahnsteiner F, Rafiński J. Ultrastructure of the epithelial cells in the aspermatogenic part of the testis in Mimagoniates barberi (Teleostei: Characidae: Glandulocaudinae) and the role of their secretions in spermatozeugmata formation. Ann Anat. 2001; 183(5):427–35. https://doi.org/10.1016/S0940-9602(01)80197-7
Plaza G, Sakaji H, Honda H, Hirota Y, Nashida K. Spawning pattern and type of fecundity in relation to ovarian allometry in the round herring Etrumeus teres. Mar Biol. 2007; 152(5):1051–64. https://doi.org/10.1007/s00227-007-0756-3
Rodríguez-Montes de Oca GA, Medina-Hernández EA, Velázquez-Sandoval J, López-López VV, Román-Reyes JC, Dabrowski K et al. Production of “Chame’ (Dormitator latifrons, Pisces: Eleotridae) larvae using GnRHa and LHRHa. Rev Colomb Ciencias Pecu. 2012; 25(3):422–29.
Ruiz-Campos G. Catálogo de peces dulceacuícolas de Baja California Sur. México Instituto Nacional de Ecología (SEMARNAT); 2012.
Santana-Piñeros AM, Cruz-Quintana Y, Reyes-Mero BM, González-Solís D, Rodríguez-Canul R. Hematological parameters of the Pacific fat sleeper, Dormitator latifrons (Pisces: Eleotridae), under natural and cultured conditions. Egypt J Aquat Res. 2024; 50(1):162–67. https://doi.org/10.1016/j.ejar.2023.11.002
Stram DL, Kincaid CR, Campbell DE. Water quality modeling in the rio Chone estuary. J Coast Res. 2005; 2005(214):797–810. https://doi.org/10.2112/011-NIS.1
Teichert N, Pierre V, Fostier A, Grondin H, Gaudin P. Reproductive biology of an endemic amphidromous goby, Cotylopus acutipinnis, from La Réunion Island. Mar Freshw Res. 2015; 67(4):526–36. https://doi.org/10.1071/MF14264
Teichert N, Pierre V, Fostier A, Lagarde R, Gaudin P. Reproductive biology of an amphidromous goby, Sicyopterus lagocephalus, in La Réunion Island. Hydrobiologia. 2014; 726(1):123–41. https://doi.org/10.1007/s10750-013-1756-6
Teixeira RL. Abundance, reproductive period, and feeding habits of eleotrid fishes in estuarine habitats of north-east Brazil. J Fish Biol. 1994; 45(5):749–61. https://doi.org/10.1111/j.1095-8649.1994.tb00941.x
Teletchea F, Fostier A, Kamler E, Gardeur J-N, Le Bail P-Y, Jalabert B et al. Comparative analysis of reproductive traits in 65 freshwater fish species: Application to the domestication of new fish species. Rev Fish Biol Fish. 2009; 19(4):403–30. https://doi.org/10.1007/s11160-008-9102-1
Tresierra Aguilar AE, Culquichicón Malpica ZG, Veneros Urbina B. Biología reproductiva en peces de Perú. Lima: Editora Nuevo Norte S.A; 2002.
Tyler CR, Sumpter JP. Oocytes growth and development in teleosts. Rev Fish Biol Fish. 1996; 6(3):287–318.
Uribe MC, Grier HJ, Mejía-Roa V. Comparative testicular structure and spermatogenesis in bony fishes. Spermatogenesis. 2014; 4(3):e983400. https://doi.org/10.4161/21565562.2014.983400
Vega-Villasante F, Ruiz-González LE, Carillo-Chong O, Basto-Rosales MER, Palma-Cancino DJ, Tintos-Gómez A et al. Biology and use of the Pacific fat sleeper Dormitator latifrons (Richardson, 1844): state of the art review. Lat Am J Aquat Res. 2021; 49(3):391–403. http://dx.doi.org/10.3856/vol49-issue3-fulltext-2637
Violante-González J, Marquez-Silva NE, Monks S, García-Ibáñez S, Pulido-Flores G, Rojas-Herrera A et al. Population dynamics of the acanthocephalan Neoechinorhynchus brentnickoli (Neoechinorhynchidae) in Pacific fat sleeper, Dormitator latifrons, from Tres Palos Lagoon, Guerrero, Mexico. IRD. 2016; 61(1):34–40. https://doi.org/10.1080/07924259.2016.1267044
Wijayanti G, Setyawan P, Kurniawati I. A simple paraffin embedded protocol for fish egg, embryo, and larvae. Scr Biol. 2017; 4(2):85–89.
Wootton RJ. Ecology of teleost fishes. New York: Chapman and Hall; 1990.
Yamasaki N, Kondo M, Maeda K, Tachihara K. Reproductive biology of three amphidromous gobies, Sicyopterus japonicus, Awaous melanocephalus, and Stenogobius sp., on Okinawa Island. Cybium. 2011; 35(4):345–59.
Zambrano-Bermúdez D, Santana-Piñeros AM, Muñoz-Chumo LG, Velez-Chica JC, Cruz-Quintana Y. Eficacia del eugenol como anestésico en el chame Dormitator latifrons mantenidos a dos salinidades. Rev Investig Vet Del Perú. 2023; 34(4):e24587. https://doi.org/10.15381/rivep.v34i4.24587
Authors
![]() Yanis Cruz-Quintana1,2,
Yanis Cruz-Quintana1,2, ![]() Ana María Santana-Piñeros1,2
Ana María Santana-Piñeros1,2 ![]() ,
, ![]() Byron Manuel Reyes-Mero1,
Byron Manuel Reyes-Mero1, ![]() Leonela Griselda Muñoz-Chumo1,
Leonela Griselda Muñoz-Chumo1, ![]() Juan Carlos Vélez-Chica1,
Juan Carlos Vélez-Chica1, ![]() Carles Alcaraz3 and
Carles Alcaraz3 and ![]() Neil Duncan2
Neil Duncan2
[1] Grupo de Investigación en Sanidad Acuícola, Inocuidad y Salud Ambiental (SAISA), Departamento de Acuicultura, Pesca y Recursos Naturales Renovables, Facultad de Acuicultura y Ciencias del Mar, Universidad Técnica de Manabí, Calle Gonzalo Loor Velasco s/n, Ciudadela Universitaria, Bahía de Caráquez, Manabí, Ecuador. (YCQ) yanis.cruz@utm.edu.ec, (AMSP) ana.santana@utm.edu.ec (corresponding author), (BMRM) byron.reyes@utm.edu.ec, (LGMC) leonela.munoz@utm.edu.ec, (JCVC) juan.velez@utm.edu.ec.
[2] IRTA, Aquaculture, Centre de La Ràpita, 43540 La Ràpita, Catalonia, Spain. (ND) neil.duncan@irta.cat.
[3] IRTA, Marine and Continental Waters, 43540 La Ràpita, Catalonia, Spain. (CA) carles.alcaraz@irta.cat.
Authors’ Contribution 

Yanis Cruz-Quintana: Conceptualization, Funding acquisition, Investigation, Methodology, Resources, Supervision, Validation, Writing-original draft, Writing-review and editing.
Ana María Santana-Piñeros: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing-original draft, Writing-review and editing.
Byron Manuel Reyes-Mero: Data curation, Investigation, Methodology.
Leonela Griselda Muñoz-Chumo: Data curation, Investigation, Methodology.
Juan Carlos Vélez-Chica: Formal analysis, Methodology.
Carles Alcaraz: Formal analysis, Methodology, Writing-review and editing.
Neil Duncan: Validation, Writing-review and editing.
Ethical Statement
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.The specimens were collected under a collection permit (number 004–2019–DP–DPAM–MAE) issued by the Ministerio de Ambiente, Ecuador. The proposed methods, use of animals, and research practices (approval number: CBI–UTM–INT–24–11–12_AMSP) were examined and approved by the Animal Welfare and Ethics Committee at Universidad Técnica Manabí.
Competing Interests
The author declares no competing interests.
Data availability statement
The data sets created during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
This study was funded through the contribution of Universidad Técnica de Manabí, which provided financial support to the project “Biological aspects of the chame Dormitator latifrons from natural environment and production” (PYTBEC613–2018–FCV0012).
How to cite this article
Cruz-Quintana Y, Santana-Piñeros AM, Reyes-Mero BM, Muñoz-Chumo LG, Vélez-Chica JC, Alcaraz C, Duncan N. Gonadal development and reproductive traits of pacific flat sleeper Dormitator latifrons (Gobiiformes: Eleotridae): insights for sustainable management in Ecuador. Neotrop Ichthyol. 2026; 24(1):e250013. https://doi.org/10.1590/1982-0224-2025-0013
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted October 9, 2025
Accepted October 9, 2025
![]() Submitted January 29, 2025
Submitted January 29, 2025
![]() Epub March 06, 2026
Epub March 06, 2026