![]() Matheus Henrique Hamann1,
Matheus Henrique Hamann1, ![]() Matheus Oliveira Freitas1,
Matheus Oliveira Freitas1, ![]() Maurício Osvaldo Moura2,
Maurício Osvaldo Moura2, ![]() Luis Roberto Bueno3 and
Luis Roberto Bueno3 and ![]() Vinicius Abilhoa4
Vinicius Abilhoa4 ![]()
PDF: Download Here | Cite this article
Associate Editor: ![]() Fernando Carvalho
Fernando Carvalho
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Espécies do complexo Hoplias malabaricus estão amplamente distribuídas na maioria das drenagens neotropicais. Algumas espécies são semelhantes e difíceis de identificar e delimitar porque muitos caracteres morfológicos são altamente homoplásticos e variáveis. Como a morfologia dos otólitos tem se mostrado particularmente útil como um caráter adicional de valor diagnóstico, análises de contorno dos otólitos lagenares de indivíduos tentativamente identificados como Hoplias malabaricus foram realizadas, com o objetivo de fornecer evidências sobre a identidade deste morfotipo em alguns sistemas hidrográficos no sul do Brasil. Os otólitos asteriscus analisados apresentaram morfologia similar, entretanto diferenças intraespecíficas revelaram a provável existência de quatro distintos, de acordo com as bacias: (1) alto rio Paraná e baixo rio Iguaçu, (2) rio Jordão, (3) alto rio Iguaçu e (4) bacia do Atlântico Sul e rio Ribeira de Iguape. As diferenças observadas foram atribuídas à região da pseudoexcisura, além de pequenas modificações na forma do antirostrum, pseudoantirostrum e lobus major. Os resultados indicaram a existência de diversificação geográfica na forma dos otólitos devido à divergência alopátrica entre algumas populações isoladas de traíras identificadas como Hoplias cf. malabaricus no sul do Brasil. A potencial existência de novas espécies dentro deste grupo deve incentivar estudos com diferentes metodologias para descrever e diferenciar as espécies de Hoplias.
Palavras-chave: Coeficientes Wavelet, Morfologia, Traíras.
Introduction
The Neotropical region hosts the richest taxonomic and functional diversity of freshwater fishes in the world (Toussaint et al., 2016), and several families are endemic to this realm (Malabarba, Malabarba, 2020). The family Erythrinidae, which have broad geographic distribution throughout South and Central America (Nelson et al., 2016), comprises three extant genera of wolf fish: Hoplerythrinus (Gill, 1896), Erythrinus (Scopoli, 1777), and Hoplias (Gill, 1903). Hoplias is the most species-rich of these clades and one of the most widespread genera of Characiforms in South America, comprising 15 nominal valid species (Fricke et al., 2025). However, as in other diverse and widely distributed genera of Neotropical fish (Nascimento et al., 2023; Costa et al., 2024), genetic analyses (including DNA Barcoding) indicate the existence of cryptic lineages and potential undescribed taxa (Cardoso et al., 2018; Jacobina et al., 2018).
The species of Hoplias are characterized by the remarkably uniform external morphology and by the fact that they can be formally arranged into groups based on the configuration of the medial margin of dentaries and by the presence or absence of tooth-bearing plates on dorsal surface of the basihyal and basibranchials (Oyakawa, Mattox, 2009). The species of the ‘H. malabaricus’ group, regionally known as thrairas, show the medial margins of dentary converging toward the mandibular symphysis, forming a V-shaped margin in ventral view, four pores of laterosensory system on each side of the dentary and tooth plates present on the basihial and basibranchials (Oyakawa, Mattox, 2009).
Since the seminal cytogenetic and cytotaxonomic study of Bertollo et al. (2000), which recognized a conspicuous karyotype diversity and arranged ‘H. malabaricus’ in seven differentiated karyomorphs (A, B, C, D, E, F, and G), molecular (Santos et al., 2009; Pereira et al., 2012; Marques et al., 2013; Cardoso et al., 2018) and morphological (Baumgartner et al., 2012; Bifi, 2013; Ota et al., 2018) analyses reveal that ‘Hoplias malabaricus’ is a complex of cryptic species morphologically similar, with some independent evolutionary units found in sympatry (Lemos et al., 2002; Pazza, Júlio Júnior, 2003).
In a revisionary study of the ‘Hoplias malabaricus’group from the La Plata River basin, Bifi (2013) proposed diagnostic features based on external morphological characteristics, albeit with minimal divergence, and delimited the occurrence of five morphotypes in the region: Hoplias sp. A, Hoplias sp. B, Hoplias sp. C, Hoplias sp. D, and Hoplias cf. malabaricus. The morphotype Hoplias sp. A (cytotype C of Bertollo et al., 2000) is distributed in the Paraguay and Paraná drainages, whereas Hoplias sp. B (cytotype D of Bertollo et al., 2000) seems to be endemic to the upper rio Paraná basin. Hoplias sp. C was registered in the rio Iguaçu and rio Uruguay and Hoplias sp. D in the Paraguay, Uruguay and lower Paraná drainages. The morphotype Hoplias cf. malabaricus encompassed part of the karyomorph named cytotype A by Bertollo et al. (2000), and was registered in the Paraná, Iguaçu and Uruguay Rivers. Unfortunately, no mention on cytotype B of Bertollo et al. (2000) was made in Bifi (2013), even though this karyomorph was detected in the Iguaçu River basin (Lemos et al., 2002).
Recently, two of these lineages proposed by Bifi (2013) were recognized as valid species: Hoplias sp. A was described as H. mbigua Azpelicueta, Benítez, Aichino & Mendes, 2015, and Hoplias sp. D as H. misioneira Rosso, Mabragaña, González-Castro, Delpiani, Avigliano, Schenone & Díaz de Astarloa, 2016. Both species were considered non-natives in the upper rio Paraná by Pazza, Júlio Júnior (2003) and Ota et al. (2018). However, Cardoso et al. (2018), using mitochondrial (mt) DNA barcoding, expanded the known native geographic distribution of H. misionera in the Neotropical Region,including Paraguay (Argentina), upper and lower Paraná and Uruguay Rivers. They also suggested the occurrence of H. misionera in the Amazon and the Pilcomayo River basins in Bolivia.
A third species, the deep-bodied H. argentinensis Rosso, González-Castro, Bogan, Cardoso, Mabragaña, Delpiani & Díaz de Astarloa, 2018, originally described for the lower La Plata River basin, was recorded for the dos Patos-Mirim Lagoon (Cardoso et al., 2018) and recently included in the list of native species of the upper rio Paraná according to the compilation of digital records from ichthyological collections (Reis et al., 2020) and the comprehensive inventory of Dagosta et al. (2024). Currently, there is insufficient molecular and morphological evidence to decide whether its occurrence is natural in the upper rio Paraná or related to human-assisted introduction.
At present, Hoplias malabaricus group is represented by nine valid species: H. malabaricus (Bloch, 1794), H. microlepis (Günther, 1864), H. teres (Valenciennes, 1847), H. mbigua, H. misionera, H. argentinensis, H. auri Guimarães, Rosso, González-Castro, Souza, Díaz de Astarloa & Rodrigues, 2021, H. cazumba Abreu, Pedroza, Oyakawa, Melo, Tchaicka & Piorski, 2025, and H. maranhensis Guimarães, Rosso, González-Castro, do Nascimento Andrade, Brito, Guimarães, Díaz de Astarloa & Rodrigues, 2025. However, molecular evidence suggests the presence of several independent new lineages (Marques et al., 2013; Cardoso et al., 2018; Jacobina et al., 2018), despite minimal morphological differences among populations, making the identification and delimitation of new species a cumbersome process.
In recent collecting efforts in reservoirs located in the rio Tibagi (upper rio Paraná basin), rio Passaúna (upper rio Iguaçu basin), two sites in lower stretch of rio Iguaçu, rio Jordão (rio Iguaçu basin), rio Capivari (rio Ribeira de Iguape basin), rio Arraial, and rio São João (South Atlantic basin) in Southern Brazil, several individuals of Hoplias with dentaries abruptly converging towards the mandibular symphysis and with bony tooth plates on the basihyal were collected. They were unambiguously recognized as belonging to the H. malabaricus group, and identified as the morphotype Hoplias cf. malabaricus by Bifi (2013). This morphotype, also called as Hoplias sp. 3 by Ota et al. (2018) and Hoplias sp. 1 by Baumgartner et al. (2012), has the series of scales on the base of the caudal-fin rays forming a straight margin and 38 to 41 perforated scales in the lateral line, plus one to three unperforated scales located anteriorly, under the opercle membrane. Previous studies in the rio Iguaçu basin showed that this karyomorph is the most widely distributed in the basin (Lemos et al., 2002; Vicari et al., 2006).
Given the considerable difficulties for the identification and delimitation of Hoplias cf. malabaricus and the potential existence of cryptic species in some hydrographic systems in Southern Brazil, the purpose of this study was to determine whether the shape of the lagenar otolith, the largest “ear stone” in most ostariophysians, varies among populations of Hoplias cf. malabaricus. Otolith morphology has proven to be a powerful diagnostic tool, especially for discriminating cryptic species and populations (Adams, 1940; Tuset et al., 2012; Deng et al., 2013; Paul et al., 2013; Mereles et al., 2021; Arroyo-Zúñiga et al., 2022; Takahashi et al., 2023), although the morphology and shape of otoliths can also show high levels of differentiation according to sex (Maciel et al., 2019), growth (Lombarte, Castellón, 1991), geography (Dalcin, Abilhoa, 2024), and environmental factors (Lombarte, Lleonart, 1993).
Considering that a morphometric frameworks have already been proven useful for species discrimination (Mereles et al., 2021; Park et al., 2023; Pattanayak et al., 2024), the specific aims of our study were (a) evaluate the utility of the lagenar otolith shape for identifying Hoplias cf. malabaricus morphotypes, and (b) investigate geographic diversification in otolith shape among isolated populations in southern Brazil, to provide insights for future taxonomic studies.
Material and methods
Data collection. Individuals of the morphotype Hoplias cf. malabaricus were sampled in reservoirs from six hydrographic systems in Southern Brazil: rio Tibagi (upper rio Paraná basin), rio Passaúna (upper rio Iguaçu basin), two sites in lower stretch of rio Iguaçu, rio Jordão (rio Iguaçu basin), rio Capivari (rio Ribeira de Iguape basin), rio Arraial and rio São João (South Atlantic basin), using gill nets (3, 4, 6, 8, 12, and 16 cm of mesh size) between January 2024 to March 2025 (Fig. 1). All specimens were identified using the morphological-based diagnostic features provided by Bifi (2013), Baumgartner et al. (2012), and Ota et al. (2018). Specimens were deposited as vouchers in the fish collection of the Museu de História Natural Capão da Imbuia (MHNCI). The list of the hydrographic systems, sampling sites, number of otoliths examined, and the range of total length (TL cm) are shown in Tab. 1.
FIGURE 1| Locations where Hoplias cf. malabaricus populations were sampled in southern Brazil.
TABLE 1 | Hydrographic systems, sampling sites, number (N) of examined otoliths, and the range of total length (TL cm) of Hoplias cf. malabaricus populations sampled in Southern Brazil.
Hydrographic system | River basin | Sampling site | N | Range | Voucher |
Upper rio Paraná basin | Tibagi River, tributary of the Paranapanema River basin | Mauá Hydroelectric Plant | 15 | 33.0–57.0 | MHNCI 13128 |
Rio Jordão basin | Jordão River, tributary of the Iguaçu River basin | Fundão-Santa Clara Energetic Complex | 35 | 14.0–61.0 | MHNCI 13129 |
Lower rio Iguaçu basin | Iguaçu River (main channel) | Salto Caxias Hydroelectric Plant | 12 | 26.0–40.0 | MHNCI 13127 |
Cavernoso River | Cavernoso Hydroelectric Plant | 9 | 10.0–40.0 | MHNCI 13126 | |
Upper rio Iguaçu basin | Passaúna River | Passaúna Dam | 19 | 31.0–45.0 | MHNCI 13133 |
Rio Ribeira de Iguape basin | Capivari River | Capivari-Cachoeira Hydroelectric Plant | 21 | 31.0–44.0 | MHNCI 13130 |
South Atlantic basin | Arraial River | Guaricana Hydroelectric Plant | 20 | 21.0–46.0 | MHNCI 13131 |
São João River | Vossoroca Hydroelectric Plant | 5 | 32.0–35.0 | MHNCI 13132 |
The lagenar otolith of 136 specimens were extracted and clean with distilled water and 70% ethanol to eliminate the otolithic membrane and macula adhered to them. Under a stereoscopic microscope, asteriscus otoliths were cleaned carefully using needles to remove the remaining organic material adhered and then stored dry before photographs and shape analyses. Lagenar otoliths with calcified structures in perfect condition were used. For the morphological analyses, the right asteriscus otolith were placed with the fossa acustica facing up (inner surface), and with the anterior extreme of the pseudoexcisura pointing left. Two-dimensional digital images were recorded using a Biofocus® ECZ-BLACK-TRI-45-L-BI trinocular stereomicroscope with 0.9x objective and 10x ocular lens, coupled with an Eakins® 48MP digital microscope camera of 1920 x 1080 resolution (magnification 45x), employing the S-EYE 2.0 software using reflected light with a dark background containing a reference for scaling. Otolith’s descriptive nomenclature and terminology followed Assis (2003).
Data analysis. Otolith image analyses were performed in the R environment (R Development Core Team, 2025) within R Studio (R Studio Team, 2025). First, brightness, contrast and sharpness of all images were enhanced with the magick package (Ooms, 2025) to ensure high resolution and to better distinguish the otolith’s shape from the surrounding background. Next, through the shapeR package (Libungan, Pálsson, 2015) the pixel noise around the contour was removed and otolith outlines were automatically extracted. All areas defined by the contours were normalized to 1. Polar axis and coordinates (from 0º to 360º angle) were drawn from the centroid of the contour and all otoliths were rotated and positioned horizontally with the anterior extreme of the pseudoexcisura pointing left. Outlines were decomposed in several wavelet functions to compute wavelet coefficients using a non-decimated Discrete Wavelet Transform (DWT).
The DWT method treats the outline of the otolith as a signal wave, utilizing signal processing techniques to approximate a series of functions that increasingly capture the topographic variation on the otolith outline (Gençay et al., 2001; Vetterli, Kovačević, 1995). All details not captured by each function are stored as numerical values, called wavelet coefficients. This series expands until there is nothing more to capture, that is, until the last coefficient is zero. Traditionally, the Fourier Series Transform has been used alongside the DWT method for generalized descriptions of shape in contour analysis (e.g., Gençay et al., 2001; Santos et al., 2017; Mereles et al., 2021). However, given the contour complexity of the lagenar otoliths of Hoplias cf. malabarius, the calculated Fourier harmonics didn’t converge for our shape data, maintaining a high deviation from the mean, even with the increase of the number of harmonics (Fig. 2).
FIGURE 2| Plots of the quality of Wavelet and Fourier outline reconstruction. The red lines indicate the level of wavelet and number of Fourier harmonics needed for a 95% accuracy.
For this reason and the well-known advantages of the DWT method over other contour analyses (Tuset et al., 2021; Vasconcelos et al., 2025), we decided to calculate only the wavelet coefficients because of the better quality of outline reconstruction. The DWT is effective for identifying shape differences in specific regions, such at a given angle on the otolith outline. Also, it has been proven to be a reliable method to detect inter and intraspecific variation while allowing a graphical reconstruction of shapes (Libungan, Pálsson, 2015; Tuset et al., 2021). Additionally, to account for the allometric relationships of contour and fish size, shape coefficients were normalized to remove the influence of size on the phenotypic variation of the otolith’s outline during growth. This was done by using a linear regression on fish length (a measure of overall size) and removal of significantly size-dependent correlated coefficients. This technique ensures that the observed shape differences were not merely a reflection of variation in fish size.
The proportion of variation within populations was summarized with intraclass correlation (ICC) analysis to inspect how the variation in the Wavelet coefficients is dependent on the position (angle) along the outline. Differences in the otolith contour among hydrographic systems were visualized with a plot of the mean shape of each population built through the reconstructed outlines of the normalized Wavelet coefficients.
Using the Momocs package (Bonhomme et al., 2014), a Principal Components Analysis (PCA) was performed on shape variables, creating new and independent variables (principal components) with combinations of Wavelet coefficients, while maintaining as much variation as possible (Wold et al., 1987; Yang et al., 2024). A Multivariate Analysis of Variance (Manova), with hydrographic region as factor (independent variable) and all Principal Components (PCs) as response variables, was used to test whether shape varies between and among populations (explanatory variables). As multiple comparisons increase the type I error, all p-values were adjusted based on the False Discovery Rate calculated using the Benjamini-Hochberg (Legendre, Legendre, 1998). Finally, to classify the phenotypic differences (PCs) between and within sampling sites, a Linear Discriminant Analysis (LDA) was used. As graphic representations, the first two PCs form a morphospace where the variation and covariation patterns of the shape coefficients of each otolith can be observed through the resulting orientation and clustering of observations, while the first two Linear Discriminant axis (LDs) maximize the distance between predefined groups when building linear combinations of PCs variables (Zelditch et al., 2012). Observations belonging to the same hydrographic basin were grouped with 95% confidence interval ellipses around the centroid and colored according to the hydrogeographic region.
Afterwards, to describe the main differences in the otolith outline of each population we found the nearest point to the population centroid using the machine-learning k Nearest Neighbor algorithm (Beygelzimer et al., 2024). Since the computed centroid represents an estimated average of the outline (Demir et al., 2018), taking the closest individual to the centroid as the group’s mean shape maintains all the intrinsic contour variation of an actual otolith and indicates the closest mathematical representation of the mean shape. For visualization purposes, we find it to be a more interesting way of displaying group differences against the global mean, since real variation is shown and it is more pronounced.
Results
Despite the conspicuous variation in head morphology and coloration (Fig. 3) and in the pigmentation pattern of the lower jaw (Fig. 4), all individuals collected in reservoirs in southern Brazil showed dentaries abruptly converging towards the mandibular symphysis, four pores of laterosensory system on each side of the dentary and the presence of tooth plates on tongue. The series of scales on the base of the caudal-fin rays forming a straight margin (Fig. 5), the dorsal profile of head straight, the head depth (50.8–60.1% of head length), the large orbital diameter (15.1–21.8% of head length), the number of perforated scales in the lateral line (41 or lower, mostly 40) and the number of scales around the caudal peduncle (20) diagnosed all individuals as the morphotype named Hoplias cf. malabaricus by Bifi (2013).
FIGURE 3| Lateral view of head of Hoplias cf. malabaricus populations sampled in southern Brazil. Scale bar: 20 mm.
FIGURE 4| Ventral view of head of Hoplias cf. malabaricus populations sampled in southern Brazil. Scale bar: 20 mm.
FIGURE 5| Pattern of last vertical series of scales on base of caudal-fin rays in Hoplias cf. malabaricus populations sampled in southern Brazil. Scale bar: 20 mm.
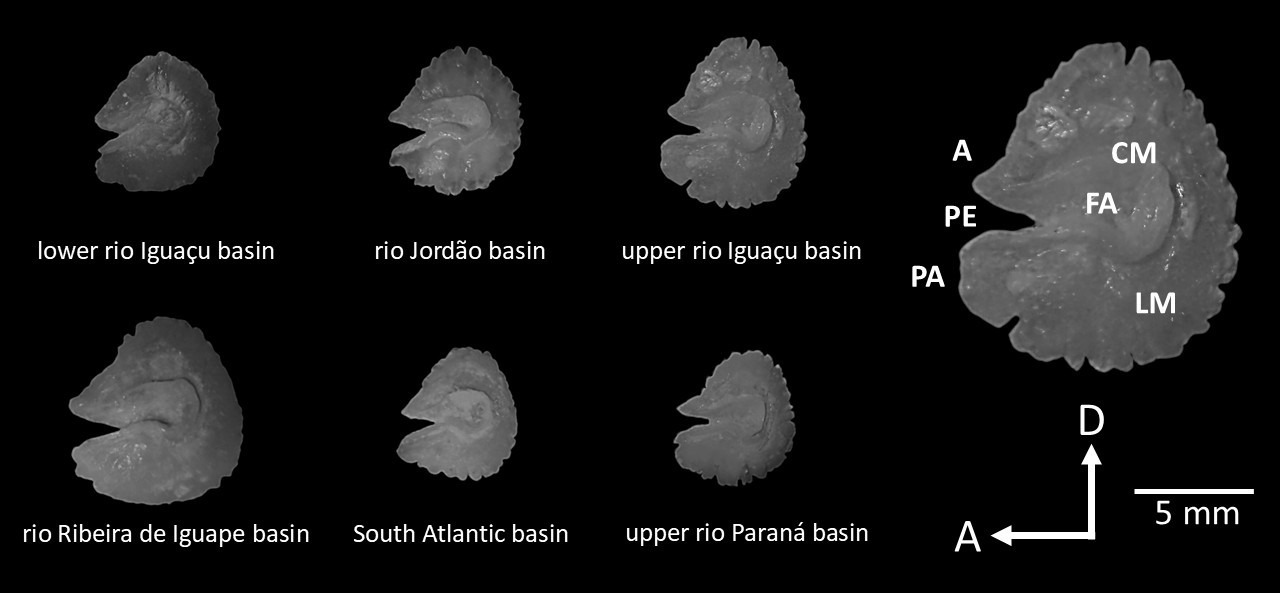
The lagenar otolith of Hoplias cf. malabaricus populations display an oval (auricular) shape and a broad, almost straight, depression in its central region (fossa acustica) surrounded by an elevation (crista). The lobus major is well-developed. Antirostrum and pseudoantirostrum are located on the anterior part of the otolith, separated by a conspicuous notch between them (pseudoexcisura) (Fig. 6).
FIGURE 6| Lateral view of the right lagenar otoliths (asteriscus) of Hoplias cf. malabaricus sampled in southern Brazil. Terminology for the main features displayed in the otolith according to Assis (2003): FA (fossa acustica), LM (lobus major), CM (crista medial), A (antirostrum), PE (pseudoexcisura), PA (pseudoantirostrum). D (dorsal) and A (anterior) directions.
Most of the variation in the Wavelet coefficients within populations was observed in the 0-30° interval, which is localized at the ventralmost part of the lobus major, as well as in the 170-210° interval, which corresponds to the dorsal part of the otolith. In contrast, the region spanning angles 310-340º, corresponding roughly to the antirostrum, pseudoexcisura and pseudoantirostrum areas, showed the highest values of means and standard deviations of wavelet coefficients, capturing the most significant shape variation among different Hoplias cf. malabaricus populations. These results point towards different phenotypical variation patterns between populations (Fig. 7).
FIGURE 7| A. Mean shapes of otolith contours. B. Mean (circles) and standard deviation (sd, vertical bars) of the wavelet coefficients for all lagenar otoliths (asteriscus) of Hoplias cf. malabaricus sampled in southern Brazil and the proportion of variance among populations – intraclass correlation (ICC) (black line). The horizontal axis shows angle in degrees based on the polar coordinates of mean shapes of the otolith outlines.
The first two principal components of the PCA analysis explained 53.3% of the total variation and revealed a high overlap in the lagenar otolith morphology among the hydrographic regions (Fig. 8A). Although the main axes of variation are similar, there are shape differences in the otoliths among some populations (Tab. 2). These significant differences in the shape of otoliths occurred mainly among samples from lower rio Iguaçu, rio Jordão and South Atlantic basin (Tab. 2), as shown in the first two axes of the LDA plot (Fig. 8B).
FIGURE 8| A. Principal component analysis plot (PC1 vs. PC2) of the lagenar otolith contours of Hoplias cf. malabaricus populations from Southern Brazil applied to Wavelet coefficients, with 95% confidence interval ellipses. B. Linear Discriminant Analysis of the lagenar otolith contours of Hoplias cf. malabaricus populations from Southern Brazil applied to Wavelet coefficients, with 95% confidence interval ellipses. Symbols and ellipses were colored according to the hydrogeographic region. Ellipses represent the average dispersion of those points around the centroid (open circle).
TABLE 2 | Multivariate Analysis of variance (MANOVA) for Wavelet coefficients of the lagenar otolith shape among Hoplias cf. malabaricus populations sampled in Southern Brazil. Bold numbers denote significant differences.
Factors | Df | Pillai | approx F | num Df | den Df | Pr(>F) |
MANOVA | ||||||
Hydrogeographic regions | 5 | 1.536 | 1.621 | 145 | 530 | 0.001 |
Residuals | 130 |
|
|
|
|
|
Pairwise pos hoc test | ||||||
Lower rio Iguaçu basin vs. rio Jordão basin | 1 | 0.287 | 1.267 | 13 | 41 | 0.289 |
Lower rio Iguaçu basin vs. rio Ribeira de Iguape basin | 1 | 0.744 | 2.525 | 15 | 26 | 0.001 |
Lower rio Iguaçu basin vs. South Atlantic basin | 1 | 0.581 | 3.308 | 13 | 31 | 0.006 |
Lower rio Iguaçu basin vs. upper rio Iguaçu basin | 1 | 0.668 | 3.710 | 13 | 24 | 0.006 |
Lower rio Iguaçu basin vs. upper rio Paraná basin | 1 | 0.440 | 1.148 | 13 | 19 | 0.382 |
Rio Jordão basin vs. rio Ribeira de Iguape basin | 1 | 0.607 | 4.878 | 13 | 41 | 0.001 |
Rio Jordão basin vs. South Atlantic basin | 1 | 0.325 | 1.706 | 13 | 46 | 0.105 |
Rio Jordão basin vs. upper rio Iguaçu basin | 1 | 0.681 | 6.415 | 13 | 39 | 0.001 |
Rio Jordão basin vs. upper rio Paraná basin | 1 | 0.526 | 2.898 | 13 | 34 | 0.011 |
Rio Ribeira de Iguape basin vs. South Atlantic basin | 1 | 0.459 | 2.021 | 13 | 31 | 0.066 |
Rio Ribeira de Iguape basin vs. upper rio Iguaçu basin | 1 | 0.550 | 2.253 | 13 | 24 | 0.055 |
Rio Ribeira de Iguape basin vs. upper rio Paraná basin | 1 | 0.752 | 4.438 | 13 | 19 | 0.005 |
South Atlantic basin vs. upper rio Iguaçu basin | 1 | 0.540 | 2.616 | 13 | 29 | 0.024 |
South Atlantic basin vs. upper rio Paraná basin | 1 | 0.740 | 5.268 | 13 | 24 | 0.001 |
Upper rio Iguaçu basin vs. upper rio Paraná basin | 1 | 0.682 | 2.803 | 13 | 17 | 0.035 |
The high overlap in the PCA suggests general shape similarity, while the significant MANOVA results and segregation in the LDA indicate consistent, albeit subtle, shape differences that are amplified when group structure is imposed. The most remarkable variations in the auricular shape of the lagenar otoliths occurred in the anterior region, and were attributed mainly to the position and shape of the pseudoexcisura, in addition to minor modifications in the antirostrum and pseudoantirostrum structures (Fig. 9).
FIGURE 9| Main differences in the otolith outline of each population of Hoplias cf. malabaricus sampled in southern Brazil. Outlines show the main deviations (colored according to the hydrogeographic region) of a representative otolith from each population compared to the overall mean shape (in black).
Discussion
The asteriscus otolith of Hoplias cf. malabaricus has an overall morphology that can be effectively assigned to the lagenar otolith of most characins (Boyle, Herrel, 2018; Costa et al., 2018) and ostariophysan taxa (Frost, 1925), despite the great diversity and phenotypic plasticity of Characiformes (Melo et al., 2022). The lagenar otolith analyzed in the present study corresponds to the gyro type proposed by Assis (2003) based on lobe shape and orientation. This morphological (auricular) type of otolith is large and typically robust, and can be easily extracted and manipulated in characins (Mélotte et al., 2018). It is characterized by a well-developed lobus major, a reduced lobus minor (usually fused to one of the margins of the pseudoexcisura), a curved fossa acustica and a conspicuous anterior pseudoexcisura (Assis, 2003).
Notwithstanding that all asteriscus otoliths analyzed showed similar morphology, the linear discriminant analysis (LDA) of otolith shape and the results from Manova tests indicated the existence of diversification in some isolated populations of Hoplias cf. malabaricus. The wavelet-based methods effectively captured subtle morphological differences in the lagenar otolith outlines, particularly in the anterior margin and structures (pseudoexcisura, antirostrum, and pseudoantirostrum), which contributed to the separation of populations. The observed intraspecific morphological differences in the asteriscus otoliths revealed the putative existence of four distinct groups, according to the basins: (1) upper rio Paraná, lower rio Iguaçu, (2) rio Jordão, (3) upper rio Iguaçu, and (4) South Atlantic basin and rio Ribeira de Iguape.
Individuals from the upper rio Paraná and lower rio Iguaçu basins have an otolith shape different from populations of other hydrographic systems, indicating that the morphotype proposed by Bifi (2013) based on material from the Paraná River (including the lower rio Iguaçu) and Uruguay River basins may correspond to a single taxonomic unit, despite the geographical isolation of the ichthyofauna of the rio Iguaçu caused by the Iguaçu falls. The otolith shape of population from the right-bank tributary of the lower stretch of the rio Iguaçu (rio Jordão)was rather similar to the material analyzed from the lower rio Iguaçu and South Atlantic basin. However, it has a very distinct asteriscus shape compared to the remaining populations, which suggests that the population of the rio Jordão may represent a distinct naturally isolated lineage within Hoplias.
The individuals from the upper rio Iguaçu also showed an otolith shape different from populations of other hydrographic systems, except from rio Ribeira de Iguape. This is probably the result of the biogeographic history shared by these two drainages, associated with the geomorphological activity of the Ponta Grossa Arch formation, which resulted in faunistic exchanges between the upper rio Iguaçu and neighboring drainages (Dagosta et al., 2024). This distributional feature was recognized by Ribeiro (2006) as “Pattern C”. However, to clarify this scenario, taxonomic information about the cytotype B described by Bertollo et al. (2000), registered in the upper rio Iguaçu, rio Ribeira de Iguape, and rio Doce (Bertollo et al., 2000; Lemos et al., 2002; Vicari et al., 2005), is needed. Otoliths with a similar outline were also found among individuals of Hoplias cf. malabaricus from the South Atlantic basin and rio Ribeira de Iguape. This result is congruent with the Southeastern haplogroup of H. malabaricus from the Brazilian coast (Pereira et al., 2012), a region isolated by the largest structural discontinuities of the South American Platform, the Serra do Mar Rift System (Ribeiro, 2006).
Phenotypic plasticity of otoliths are related to differences in hearing/balance capacities and associated mechanisms (Popper, Fay, 2011) and can be influenced by several aspects such as ontogeny (Lombarte, Castellón, 1991), sexual dimorphism (Maciel et al., 2019), environmental factors (Lombarte, Lleonart, 1993) and biogeographical processes (Tuset et al., 2016; Dalcin, Abilhoa, 2024). However, the factors that determine otolith shapes are not fully understood and considering our framework they are difficult to interpret because almost all Hoplias individuals were adult and collected in lentic environments (reservoirs). Even considering the synergistic action of several factors, the distinct evolutionary histories caused by the natural isolation among Hoplias cf. malabaricus populations are likely the main driver of the observed otolith shape differentiation. Similar processes have been suggested to explain inter-population divergences observed within the cytotype A of Bertollo et al. (2000), with populations from the São Francisco, Araguaia/Tocantins, and Xingu Rivers in Brazil showing clear genetic differentiation, likely caused by historical isolation (Blanco et al., 2010).
An evaluation of the identity and delimitation of Hoplias cf. malabaricus is beyond the scope of this paper and the application of otolith shape analysis alone is insufficient for the recognition of individual species. However, otolith shape constitutes a versatile taxonomic and low-cost resource when analyzing closely related and cryptic teleost species (Bani et al., 2013; Wakefield et al., 2014; Zhuang et al., 2015; Zischke et al., 2016), indicating that the geographic variation in otolith shape observed by us is consistent with the hypothesis of allopatric divergence among isolated populations of trahiras in southern Brazil, which may represent new distinct biological entities within the historically confused H. malabaricus group.
Our study demonstrates that otolith shape analysis, particularly through Discrete Wavelet Transform, is a valuable tool for revealing geographic diversification within the taxonomically challenging Hoplias cf. malabaricus morphotype. The evidence of allopatric divergence supports the potential existence of distinct evolutionary lineages in southern Brazilian basins. Although molecular approaches are unveiling the hidden diversity within this group of Hoplias (Marques et al., 2013; Cardoso et al., 2018; Jacobina et al., 2018; Rosso et al., 2018), including material from some hydrographic systems studied by us, morphological circumscription through a deep integrative taxonomic review is still needed to delimit species in a practical sense. A holistic approach that combines genetic and morphometric analyses would improve our understanding of H. cf. malabaricus delimitation and distribution, paving the way for evolutionary studies and to understand biogeographical patterns of South American freshwater fishes.
Acknowledgments
We thank the Meros do Brasil project (sponsored by Petrobras) for the logistic support and Museu de História Natural do Capão da Imbuia (MHNCI) for the infrastructure support. We are grateful to Adriano Hauer for the aid in field collections. Part of the otoliths were obtained from fish collected during the monitoring program of the Companhia Paranaense de Energia (Copel) performed by Cerne Consultoria e Projetos Ltda.
References
Adams LA. Some characteristic otoliths of American Ostariophysi. J Morphol. 1940; 66(3):497–527. https://doi.org/10.1002/jmor.1050660307
Arroyo-Zúñiga KI, Pacheco-Ovando R, Granados-Amores E, Granados-Amores J, González Ramírez J, Díaz-Santana-Iturrios M. Lapillus otolith shape, a useful taxonomic feature for the identification of sea catfishes (Ariidae: Siluriformes) from the north-eastern Pacific. J Fish Biol. 2022; 101(5):1262–69. https://doi.org/10.1111/jfb.15198
Assis CA. The lagenar otoliths of teleosts: their morphology and its application in species identification, phylogeny and systematics. J Fish Biol. 2003; 62(6):1268–95. https://doi.org/10.1046/j.1095-8649.2003.00106.x
Bani A, Poursaeid S, Tuset VM. Comparative morphology of the sagittal otolith in three species of south Caspian gobies. J Fish Biol. 2013; 82(4):1321–32. https://doi.org/10.1111/jfb.12073
Baumgartner G, Pavanelli CS, Baumgartner D, Bifi AG, Debona T, Frana VA. Peixes do baixo rio Iguaçu. Maringá: Eduem, Maringá; 2012.
Bertollo LA, Born GG, Dergam JA, Fenocchio AS, Moreira-Filho O. A biodiversity approach in the neotropical erythrinidae fish, Hoplias malabaricus. Karyotypic survey, geographic distribution of cytotypes and cytotaxonomic considerations. Chromosome Res. 2000; 8(7):603–13. https://doi.org/10.1023/a:1009233907558
Beygelzimer A, Kakadet S, Langford J, Arya S, Mount D, LiS. FNN: Fast Nearest Neighbor Search – algorithms and applications. 2024. https://doi.org/10.32614/CRAN.package.FNN
Bifi AG. Revisão taxonômica das espécies do grupo Hoplias malabaricus (Bloch, 1794) (Characiformes: Erythrinidae) da bacia do rio da Prata. [PhD Thesis]. Maringá: Universidade Estadual de Maringá; 2013. Avaliable from: http://nou-rau.uem.br/nou-rau/document/?code=vtls000205331
Blanco DR, Lui RL, Bertollo LAC, Margarido VP, Moreira-Filho O. Karyotypic diversity between allopatric populations of the group Hoplias malabaricus (Characiformes: Erythrinidae): evolutionary and biogeographic considerations. Neotrop Ichthyol. 2010; 8(2):361–68. https://doi.org/10.1590/S1679-62252010000200015
Bonhomme V, Picq S, Gaucherel C, Claude J. Momocs: outline analysis using R. J Stat Softw. 2014; 56(13):1–24. https://doi.org/10.18637/jss.v056.i13
Boyle KS, Herrel A. Relative size variation of the otoliths, swim bladder, and Weberian apparatus structures in piranhas and pacus (Characiformes: Serrasalmidae) with different ecologies and its implications for the detection of sound stimuli. J Morphol. 2018; 279:1849–71. https://doi.org/10.1002/jmor.20908
Cardoso YP, Rosso JJ, Mabragaña E, González-Castro M, Delpiani M, Avigliano E et al. A continental-wide molecular approach unraveling mtDNA diversity and geographic distribution of the Neotropical genus Hoplias. PLoS ONE. 2018; 13(8):e0202024. https://doi.org/10.1371/journal.pone.0202024
Costa RMR, Fabré NN, Amadio SA, Tuset VM. Plasticity in the shape and growth pattern of asteriscus otolith of black prochilodus Prochilodus nigricans (Teleostei: Characiformes: Prochilodontidae) freshwater Neotropical migratory fish. Neotrop Ichthyol. 2018; 16(4):e180051. https://doi.org/10.1590/1982-0224-20180051
Costa WJEM, Feltrin CRM, Mattos JLO, Katz AM. Top mountain areas of subtropical southern Brazil sheltering four new small-ranged catfishes (Siluriformes, Trichomycteridae): relationships and taxonomy. Evol Syst. 2024; 8(2):199–218. https://doi.org/10.3897/evolsyst.8.126393
Dagosta FCP, Monção MS, Nagamatsu BA, Pavanelli CS, Carvalho FR, Lima FCT et al. Fishes of the upper rio Paraná basin: diversity, biogeography and conservation. Neotrop Ichthyol. 2024; 22(1):e230066. https://doi.org/10.1590/1982-0224-2023-0066
Dalcin RH, Abilhoa V. Discriminating Scleromystax barbatus (Siluriformes: Callichthyidae) populations from Atlantic Rainforest streams employing otolith shape. Neotrop Ichthyol. 2024; 22(1):e230129. https://doi.org/10.1590/1982-0224-2023-0129
Demir B, Sayıncı B, Çetin N, Yaman M, Çömlek R, Aydın Y et al. Elliptic Fourier based analysis and multivariate approaches for size and shape distinctions of walnut (Juglans regia L.) cultivars. Grasas y Aceites. 2018; 69(4):e271. https://doi.org/10.3989/gya.0104181
Deng X, Wagner H, Popper AN. Interspecific variations of inner ear structure in the deep‐sea fish family Melamphaidae. Anat Rec. 2013; 296(7):1064–82. https://doi.org/10.1002/ar.22703
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Frost GA. LXI – A comparative study of the otoliths of the Neopterygian fishes (continued). Ann Mag Nat Hist. 1925; 15(89):553–61. https://doi.org/10.1080/00222932508633246
Gençay R, Selçuk F, Whitcher B. Diferentiating intraday seasonalities through wavelet multi-scaling. Phys A Stat Mech Appl. 2001; 289:543–56. https://doi.org/10.1016/S0378-4371(00)00463-5
Jacobina UP, Lima SMQ, Maia DG, Souza G, Batalha-Filho H, Torres RA. DNA barcode sheds light on systematics and evolution of neotropical freshwater trahiras. Genetica. 2018; 146(6):505–15. https://doi.org/10.1007/s10709-018-0043-x
Legendre P, Legendre L. Numerical Ecology. 2nd English ed. Amsterdam: Elsevier; 1998.
Lemos PMM, Fenocchio AS, Bertollo LAC, Cestari MM. Karyotypic studies on two Hoplias malabaricus populations (Characiformes, Erythrinidae) of the 2n = 42 group, from the first plateau of the Iguaçu river basin (Paraná State, Brazil). Caryologia. 2002; 55:193–98. https://doi.org/10.1080/00087114.2002.10589277
Libungan LA, Pálsson S. ShapeR: an R package to study otolith shape variation among fish populations. PloS ONE. 2025; 10(3):e0121102. http://dx.doi.org/10.1371/journal.pone.0121102
Lombarte A, Castellón A. Interspecific and intraspecific otolith variability in the genus Merluccius as determined by image analysis. Can J Zool. 1991; 69(9):2442–49. https://doi.org/10.1139/z91-343
Lombarte A, Lleonart J. Otolith size changes related with body growth, habitat depth and temperature. Environ Biol Fishes. 1993; 37(3):297–306. https://doi.org/10.1007/BF00004637
Maciel TR, Vaz-dos-Santos AM, Barradas JRS, Vianna M. Sexual dimorphism in the catfish Genidens genidens (Siluriformes: Ariidae) based on otolith morphometry and relative growth. Neotrop Ichthyol. 2019; 17(1):e180101. https://doi.org/10.1590/1982-0224-20180101
Malabarba LR, Malabarba MC. Phylogeny and classification of Neotropical fish. In: Baldissetotto B, Urbinati E, Cyrino J, editors. Biology and physiology of freshwater Neotropical fish. Cambridge: Academic Press; 2020. p.1–19. https://doi.org/10.1016/B978-0-12-815872-2.00001-4
Marques DF, Santos FA, Silva SS, Sampaio I, Rodrigues LRR. Cytogenetic and DNA barcoding reveals high divergence within the trahira, Hoplias malabaricus (Characiformes: Erythrinidae) from the lower Amazon River. Neotrop Ichthyol. 2013; 11(2):459–66. https://doi.org/10.1590/S1679-62252013000200015
Melo BF, Sidlauskas BL, Near TJ, Roxo FF, Ghezelayagh A, Ochoa LE et al. Accelerated diversification explains the exceptional species richness of tropical characoid fishes. Syst Biol. 2022; 71(1):78–92. https://doi.org/10.1093/sysbio/syab040
Mélotte G, Parmentier E, Michel C, Herrel A, Boyle K. Hearing capacities and morphology of the auditory system in Serrasalmidae (Teleostei: Otophysi). Sci Rep. 2018; 8:1281. https://doi.org/10.1038/s41598-018-19812-1
Mereles MA, Sousa RGC, Barroco LSA, Campos CP, Pouilly M, Freitas CEC. Discrimination of species and populations of the genus Cichla (Cichliformes: Cichlidae) in rivers of the Amazon basin using otolithic morphometry. Neotrop Ichthyol. 2021; 19(4):e200149. https://doi.org/10.1590/1982-0224-2020-0149
Nascimento MHS, Aragão DG, Silva JLN, Lima RC, Birindelli JLO, Fraga EC et al. The DNA barcode reveals cryptic diversity and a new record for the genus Leporinus (Characiformes, Anostomidae) in the hydrographic basins of central northern Brazil. PeerJ. 2023; 11:e15184. https://doi.org/10.7717/peerj.15184
Nelson JS, Grande TC, Wilson MVH. Fishes of the World. New Jersey: John Wiley & Sons; 2016. https://doi.org/10.1002/9781119174844
Ooms J. magick: advanced graphics and image-processing in R. R package version 2.8.7. 2025. Available from: https://CRAN.R-project.org/package=magick
Ota RR, Deprá GC, Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: revised, annotated and updated. Neotrop Ichthyol. 2018; 16(2):e170094. https://doi.org/10.1590/1982-0224-20170094
Oyakawa OT, Mattox GMT. Revision of the Neotropical trahiras of the Hoplias lacerdae species-group (Ostariophysi: Characiformes: Erythrinidae) with descriptions of two new species. Neotrop Ichthyol. 2009; 7(2):117–40. https://doi.org/10.1590/S1679-62252009000200001
Park JM, Kang MG, Kim JH, Jawad LA, Majeed S. Otolith morphology as a tool for stock discrimination of three rockfish species in the East Sea of Korea. Front Mar Sci. 2023; 10:1301178. https://doi.org/10.3389/fmars.2023.1301178
Pattanayak M, Mallick N, Sahu P, Mohapatra SK, Roy S, Seth JK. Unveiling otolith shape variation: geometric morphometric analysis of three croaker species (Acanthuriformes: Sciaenidae). Proc Indian Natl Sci Acad. 2024; 1–08. https://doi.org/10.1007/s43538-024-00379-7
Paul K, Oeberst R, Hammer C. Evaluation of otolith shape analysis as a tool for discriminating adults of Baltic cod stocks. J Appl Ichthyol. 2013; 29(40):743–50. https://doi.org/10.1111/jai.12145
Pazza R, Júlio Júnior HF. Occurrence of three sympatric cytotypes of Hoplias malabaricus (Pisces, Erythrinidae) in the Upper Paraná River Foodplain (Brazil). Cytologia. 2003; 68(2):159–63. https://doi.org/10.1508/cytologia.68.159
Pereira TL, Santos U, Schaefer CE, Souza GO, Paiva SR, Malabarba LR et al. Dispersal and vicariance of Hoplias malabaricus (Bloch, 1794) (Teleostei, Erythrinidae) populations of the Brazilian continental margin. J Biogeogr. 2012; 40:905–14 https://doi.org/10.1111/jbi.12044
Popper AN, Fay RR. Rethinking sound detection by fishes. Hear Res. 2011; 273(1–2):25–36. https://doi.org/10.1016/j.heares.2009.12.023
R Development Core Team. R: A language and environment for statistical computing [Internet]. Vienna: R Foundation for Statistical Computing; 2025. Available from: https://www.R-project.org/
R Studio Team. R Studio: integrated development for R [Internet]. Boston: R Studio, PBC; 2025. Available from: http://www.rstudio.com/
Reis RB, Frota A, Deprá GC, Ota RR, Graça WJ. Freshwater fishes from Paraná State, Brazil: an annotated list, with comments on biogeographic patterns, threats, and future perspectives. Zootaxa. 2020; (4):451–94. https://doi.org/10.11646/zootaxa.4868.4.1
Ribeiro AC. Tectonic history and the biogeography of the freshwater fishes from the coastal drainages of eastern Brazil: an example of faunal evolution associated with a divergent continental margin. Neotrop Ichthyol. 2006; 4(2):225–46. https://doi.org/10.1590/S1679-62252006000200009
Rosso JJ, González-Castro M, Bogan S, Cardoso YP, Mabragaña E, Delpiani M et al. Integrative taxonomy reveals a new species of the Hoplias malabaricus species complex (Teleostei: Erythrinidae). Ichthyol Explor Freshw. 2018; 1–18. http://doi.org/10.23788/IEF-1076
Santos RS, Azevedo MCC, Albuquerque CQ, Araújo FG. Different sagitta otolith morphotypes for the whitemouth croaker Micropogonias furnieri in the Southwestern Atlantic coast. Fish Res. 2017; 195:222–09. https://doi.org/10.1016/j.fishres.2017.07.027
Santos U, Völcker CM, Belei FA, Cioffi MB, Bertollo LA, Paiva SR et al. Molecular and karyotypic phylogeography in the Neotropical Hoplias malabaricus (Erythrinidae) fish in eastern Brazil. J Fish Biol. 2009; 75(9):2326–43. https://doi.org/10.1111/j.1095-8649.2009.02489.x
Takahashi M, Wakefield CB, Saunders BJ, Fairclough DF, Harvey ES, Newman SJ. Efficacy of otolith morphometry for rapid discrimination of cryptic fishes. Estuar Coast Shelf Sci. 2023; 295:108516. https://doi.org/10.1016/j.ecss.2023.108516
Toussaint A, Charpin N, Brosse S, Villéger S. Global functional diversity of freshwater fish is concentrated in the Neotropics while functional vulnerability is widespread. Sci Rep. 2016; 6:22125. https://doi.org/10.1038/srep22125
Tuset VM, Azzurro E, Lombarte A. Identification of Lessepsian fish species using the sagittal otolith. Sci Mar. 2012; 76(2):289–99. https://doi.org/10.3989/scimar.03420.18E
Tuset VM, Otero-Ferrer JL, Siliprandi C, Manjabacas A, Marti-Puig P, Lombarte A. Paradox of otolith shape indices: routine but overestimated use. Can J Fish Aquat Sci. 2021; 78(6):681–92. https://doi.org/10.1139/cjfas-2020-0369
Vasconcelos J, Otero-Ferrer JL, Lombarte A, Jurado-Ruzafa A, Manjabacas A, Tuset VM. Choosing wavelet methods for otolith contour studies. Rev Fish Biol Fish. 2025; 35:201–16. https://doi.org/10.1007/s11160-024-09896-6
Vetterli M, Kovačević J. Wavelets and Subband Coding. Englewood Cliffs, New Jersey: Prentice Hall PTR; 1995. Available from: http://waveletsandsubbandcoding.org/Repository/VetterliKovacevic95_Manuscript.pdf
Vicari MR, Artoni RF, Bertollo LAC. Comparative cytogenetics of Hoplias malabaricus (Pisces, Erythrinidae): a population analysis in adjacent hydrographic basins. Genet Mol Biol. 2005; 28(1):103–10. https://doi.org/10.1590/S1415-47572005000100018
Vicari MR, Pazza R, Artoni RF, Margarido VP, Bertollo LAC. Cytogenetics and biogeography: Considerations about the natural origin of Hoplias malabaricus (Characiformes, Erythrinidae) on the Iguaçu River. Braz Arch Biol Technol. 2006; 49:297–303. https://doi.org/10.1590/S1516-89132006000300015
Wakefield CB, Williams AJ, Newman SJ, Bunel M, Dowling CE, Armstrong CA et al. Rapid and reliable multivariate discrimination for two cryptic Eteline snappers using otolith morphometry. Fish Res. 2014; 151:100–06. https://doi.org/10.1016/j.fishres.2013.10.011
Wold S, Esbensen KH, Geladi P. Principal component analysis. Chemometr Intell Lab Syst. 1987; 2(1–3):37–52. https://doi.org/10.1016/0169-7439(87)80084-9
Yang T, Xiao P, Jiang X, Zhang Q, Zhao Y. Otolith morphometrics and variations between two populations of Sillago sinica (Perciformes, Sillaginidae) in the East China Sea and the Yellow Sea. Thalassas. 2024; 40(2):1007–17. https://doi.org/10.1007/s41208-024-00711-4
Zelditch M, Swiderski D, Sheets HD. Geometric morphometrics for biologists: a primer 2nd ed. Massachusetts: Cambridge; 2012.
Zischke MT, Litherland L, Tilyard BR, Stratford NJ, Jones EL, Wang YG. Otolith morphology of four mackerel species (Scomberomorus spp.) in Australia: Species differentiation and prediction for fisheries monitoring and assessment. Fish Res. 2016; 176:39–47. https://doi.org/10.1016/j.fishres.2015.12.003
Zhuang L, Ye Z, Zhang C. Application of otolith shape analysis to species separation in Sebastes spp. from the Bohai Sea and the Yellow Sea, northwest Pacific. Environ Biol Fishes. 2015; 98:547–58. http://dx.doi.org/10.1007/s10641-014-0286-z
Authors
![]() Matheus Henrique Hamann1,
Matheus Henrique Hamann1, ![]() Matheus Oliveira Freitas1,
Matheus Oliveira Freitas1, ![]() Maurício Osvaldo Moura2,
Maurício Osvaldo Moura2, ![]() Luis Roberto Bueno3 and
Luis Roberto Bueno3 and ![]() Vinicius Abilhoa4
Vinicius Abilhoa4 ![]()
[1] Instituto Meros do Brasil, Rua Benjamin Constant, 67, 80060-020, Curitiba, PR, Brazil. (MHH) mattyhams01@gmail.com, (MOF) serranidae@gmail.com.
[2] Laboratório de Dinâmicas Ecológicas, Departamento de Zoologia, Universidade Federal do Paraná, 81531-980, Curitiba, PR, Brazil. (MOM) mauricio.moura@ufpr.br.
[3] Programa de Pós-Graduação em Biologia Evolutiva, Universidade Estadual de Ponta Grossa, Av. Carlos Cavalcanti, 4748, 84030 900, Ponta Grossa, PR, Brazil. (LRB) luisbrbio@hotmail.com.
[4] Museu de História Natural Capão da Imbuia, MAPCF/SMMA, Prefeitura de Curitiba, Rua Professor Benedito Conceição, 407, 82810-080, Curitiba, PR, Brazil. (VA) vabilhoa@uol.com.br (corresponding author).
Authors’ Contribution 

Matheus Henrique Hamann: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-original draft, Writing-review and editing.
Matheus Oliveira Freitas: Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Validation, Visualization, Writing-original draft, Writing-review and editing.
Maurício Osvaldo Moura: Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-original draft.
Luis Roberto Bueno: Investigation, Methodology, Visualization, Writing-original draft.
Vinicius Abilhoa: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) (license number: SISBIO 10320–8) and Instituto Água e Terra (IAT) (license 36/2021, protocol 17.889.275–4) authorized the capture of fish.
Competing Interests
The author declares no competing interests.
Data availability statement
The data supporting the findings of this study are available from the corresponding author, upon reasonable request.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its tables and figures.
Funding
Meros do Brasil Project is sponsored by Petrobras.
How to cite this article
Hamann MH, Freitas MO, Moura MO, Bueno LR, Abilhoa V. Addressing challenges in Hoplias cf. malabaricus (Characiformes: Erythrinidae) taxonomy and distribution in Southern Brazil with otolith shape analysis. Neotrop Ichthyol. 2026; 24(1):e250129. https://doi.org/10.1590/1982-0224-2025-0129
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 24, 2025
Accepted December 24, 2025
![]() Submitted July 18, 2025
Submitted July 18, 2025
![]() Epub April 20, 2026
Epub April 20, 2026