![]() Jefferson Luan Crispim1,2
Jefferson Luan Crispim1,2 ![]() ,
, ![]() Pablo Argüello3,
Pablo Argüello3, ![]() Gabriel de Souza da Costa e Silva1,
Gabriel de Souza da Costa e Silva1, ![]() Claudio Oliveira1,
Claudio Oliveira1, ![]() Kyle R. Luckenbill2 and
Kyle R. Luckenbill2 and ![]() Mark H. Sabaj2
Mark H. Sabaj2
PDF: Download Here | Cite this article
Associate Editor: ![]() Marcelo Britto
Marcelo Britto
Section Editor: ![]() William Crampton
William Crampton
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Pterobunocephalus compreende atualmente duas espécies válidas: P. depressus descrito para a bacia do rio Guaporé e P. dolichurus do rio Trombetas em confluência com o rio Amazonas. Uma terceira espécie nominal, Bunocephalus albofasciatus, descrita para a bacia do alto Rio Mamoré, é atualmente considerada sinônimo júnior de P. depressus. Baseado em uma análise morfológica, nós descrevemos aqui uma nova espécie de Pterobunocephalus do alto da bacia Amazônica (rios Napo, Pastaza e Putumayo) no Equador. A nova espécie se distingue de seus congeneres pela seguinte combinação de caracteres: (1) olhos grandes; (2) margem anterior do mesetmóide relativamente larga, superficialmente côncava e afundada entre as margens anteriores dos pré-maxilares; (3) número total de raios da nadadeira anal 10–11; e (4) parapófise da quinta vértebra em forma de lâmina de machado de pastor com porção distal visivelmente expandida.
Palavras-chave: Bagre banjo, CT Scan, Morfologia, Taxonomia.
Introduction
Fowler (1943) proposed Pterobunocephalus as a monotypic subgenus of Bunocephalus Kner, 1855 based on the unique holotype of his new species Bunocephalus (Pterobunocephalus) albifasciatus and distinguished by its longer and more anteriorly placed anal fin. Böhlke (1959) later established the monotypic genus Petacara for Bunocephalus dolichurus Delsman, 1941. Friel (1994, 2003) subsequently elevated Pterobunocephalus to generic status, with Petacara as its junior synonym, and recognized two valid species, P. depressus (Haseman, 1911) and P. dolichurus (Delsman, 1941), while treating Fowler’s B. (P.) albifasciatus as a synonym of the former.
Pterobunocephalus is distributed throughout South America, including the Amazon, Orinoco, and Paraguay river basins (Friel, 1994, 2003; Sleen, Albert, 2017; Dagosta, de Pinna, 2019). According to Friel (1994), Pterobunocephalus is differentiated from all other Aspredinidae by the following combination of characters: (1) head and body highly depressed; (2) medial notch in the upper jaw frequently present and distinct; (3) skull ornamentation reduced or absent; (4) laminar process of pterotic shaped like the tip of an arrow and projecting at a right angle; (5) anal-fin rays 10–20; and (6) females bearing embryos directly attached to the body.
The placement of Pterobunocephalus within Aspredinidae varies between hypotheses based on morphological (Friel, 1994; Cardoso, 2008) and molecular (Carvalho et al., 2018) studies. Friel (1994) supported Pterobunocephalus as sister to Aspredininae, a clade easily identified by having more than 50 anal-fin rays and occurring in both freshwater and marine waters between the Orinoco and Amazon deltas, composed of the genera Platystacus Bloch, 1794, Aspredo Scopoli, 1777 and Aspredinichthys Bleeker, 1858. Cardoso’s (2008), supported Pterobunocephalus as sister to a clade composed of Bunocephalus sister to Amaralia Fowler, 1954 + Acanthobunocephalus Friel, 1995. More recently, a molecular phylogeny based on two mitochondrial (16S and COI) and three nuclear (RAG1, SH3PX3 and MyH6) markers, divided Aspredinidae into four major lineages including three subfamilies: (1) the monotypic subfamily Pseudobunocephalinae was sister to a clade composed of (2) monotypic genus Acanthobunocephalus sister to remaining aspredinids in which (3) Hoplomyzontinae (Dupouyichthys Schultz, 1944, Ernstichthys Fernández-Yépez, 1953, Hoplomyzon Myers, 1942 and Micromyzon Friel & Lundberg, 1996), was sister to (4) Aspredininae, a subfamily composed of “Bunocephalus” chamaizelus sister to Aspredinini (Platystacus, Aspredo and Aspredinichthys) plus Bunocephalini (Amaralia, Bunocephalus, Pterobunocephalus, and Xyliphius Eigenmann, 1912). Within the Bunocephalini, Xyliphius was sister to a clade composed of Pterobunocephalus, sister to Amaralia + Bunocephalus (Carvalho et al., 2018).
During the 1960s, Saul (1975) collected and preserved over 100 species of fishes from the upper Aguarico River (Napo drainage) in the vicinity of Santa Cecília, northern Ecuador (approximately 00°03’46”N 76°59’58”W). His sampling included four specimens from the Río Conejo, which Saul (1975:115) identified as Bunocephalus aleuropsis Cope, 1870, a species currently considered valid in Bunocephalus. These specimens are here re-identified and described as a new species of Pterobunocephalus. Additional material belonging to the new species was collected in the Pastaza and Napo river basins and is preserved in the Museo de Historia Natural “Gustavo Orcés V.”, Departamento de Biología, Escuela Politécnica Nacional (MEPN), Quito. The present study describes this new species based on analyses of external morphology and osteological features examined, in alcohol and cleared and stained specimens as well as 2D and 3D (nano-CT) X-rays.
Material and methods
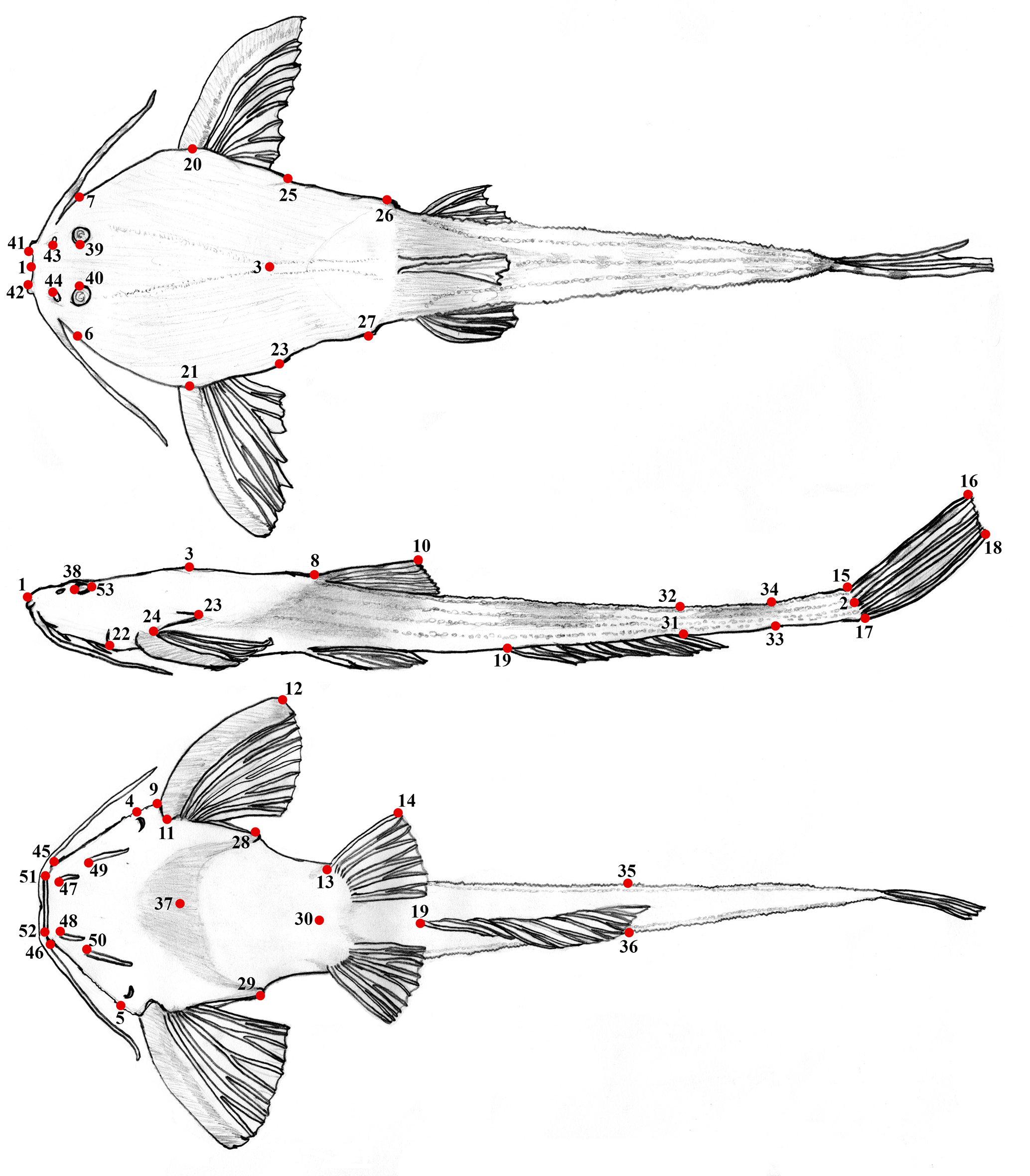
Morphological measurements were taken point-to-point using digital calipers (to 0.01 mm) and expressed as percentages of standard length and head length. Landmarks for 36 measurements (Tab. 1; Fig. 1) were adapted from Friel (1994), Cardoso (2010), and Carvalho et al. (2015), and are defined as follows: 1) Standard length (SL) measured laterally from snout tip to caudal-fin origin (end of hypural plates); 2) Head length (HL) measured between snout tip and posterior tip of supraoccipital bone; 3) Head width measured ventrally as transverse distance between contours of head taken across lateralmost ends of opercular openings); 4) Head width at eyes measured dorsally as transverse distance between lateral contours of head taken at same level as interorbital distance); 5) Predorsal length measured between snout tip and posterior margin of middle nuchal plate (i.e., anterior origin of dorsal-fin spine or, if present, dorsal-locking spine); 6) Prepectoral length measured ventrally between tip of snout and anterior margin of pectoral-spine insertion; 7) Length of unbranched dorsal-fin ray measured from its anterior margin of insertion to its distal tip with fin erected to approximately 45°; 8) Pectoral-fin spine length measured ventrally from middle base of spine (i.e., deepest point of cleithral notch) to distalmost bony tip; 9) Length of unbranched pelvic-fin ray measured ventrally, between its anterior margin of insertion and its distal tip (i.e., length of individual ray); 10–11) lengths of uppermost and lowermost unbranched principal caudal-fin rays measured between its insertion and its distal tip (i.e., length of individual ray); 12) Pectoral-pelvic distance (abdominal length sensu Cardoso, 2010) measured ventrally, between deepest point of cleithral notch and anteriormost insertion of unbranched pelvic-fin ray; 13) Pelvic-anal distance measured from anteriormost insertion of unbranched pelvic-fin ray to anteriormost insertion of first unbranched anal-fin ray; 14) Cleithral width measured dorsally as the maximum width across lateral surfaces of contralateral cleithra; 15) Cleithrum length measured laterally from anteriormost margin of cleithrum to tip of posterior cleithral process; 16) Posterior-cleithral process length measured dorsally from posterior margin of pectoral-spine insertion (erected in approximately 45°) to tip of posterior cleithral process); 17) Distance between posterior cleithral processes measured dorsally as transverse distance between outer margins of tips of posterior cleithral processes); 18) Width across parapophysis of fifth vertebrae measured dorsally as transverse distance between outer margins of parapophysis of fifth vertebrae (measured only when parapophysis reach external contour of body); 19) Distance between coracoid processes measured ventrally as transverse distance between outer margins of tips of posterior coracoid processes; 20) Length of coracoid process measured ventrally from cleithral notch to tip of posterior coracoid process; 21) Body depth at dorsal-fin origin measured at vertical through anterior origin of dorsal-fin spine (or, if present, dorsal-locking spine) to ventral body surface (calipers held orthogonal the ventral surface of specimen); 22) Caudal-peduncle length measured laterally from posterior insertion of anal-fin to terminus of SL measurement; 23) Maximum caudal-peduncle depth measured vertically from posterior insertion of anal-fin to dorsal body surface (calipers held orthogonal the dorsal surface of specimen); 24) Least caudal-peduncle depth measured vertically with calipers orthogonal to specimen; 25) Caudal-peduncle width measured dorsally as transverse distance between outer margins of body at insertion of last anal-fin ray; 26) Anal-fin base length measured from anterior base of first conspicuous unbranched anal-fin ray to posterior base of last anal-fin ray; 27) Head depth as vertical distance between posterior margin of supraoccipital and ventral body surface (calipers held orthogonal to ventral surface of specimen; 28) Snout length measured between snout tip and anteriormost bony margin of orbit (i.e., anteriormost margin of eye); 29) Interorbital width measured as least distance between bony orbits; 30) Distance between anterior nostrils measured dorsally as distance between center of openings; 31) Distance between posterior nostrils measured as least distance between medial margins of posterior nostrils; 32) Distance between maxillary barbels measured ventrally from point (axil) at which ventral medial base of maxillary barbel meets lateral contour of head; 33) Distance between anterior mental barbels is least distance between margins of base of anterior mental barbels; 34) Distance between posterior mental barbels is least distance between margins of base of posterior mental barbels; 35) Mouth width is greatest transverse measure across mouth gape; 36) Eye diameter measured between its anteriormost and posteriormost margins (i.e., iris). Eye diameter is a relevant character to diagnose the new species, and the values reported were taken of the type series (n = 14) and all specimens listed in Comparative Material examined (n = 29).Descriptions and comparisons of osteological features were based on two paratypes (ANSP 130605, 46.4 mm SL; ANSP 130606, ca. 48.0 mm SL) cleared and stained following the protocol of Taylor, Van Dyke (1985), and on nano-CT scans of one alcohol-preserved paratype (ANSP 130605, 64.1 mm SL) using a Zeiss Xradia 620 Versa X-ray microscope (Nano-CT) of the Materials Characterization Core in the Bossone Research Enterprise Center at Drexel University, Philadelphia, USA. High-resolution X-ray computed tomography (HRXCT) images were rendered in VG Studio Max and edited in Adobe Photoshop CS6. Osteological descriptions were based on cleared and stained, radiographed, and CT-scanned specimens. Osteological nomenclature follows Friel (1994). Counts of vertebrae, ribs and pectoral-spine serrations were obtained from radiographs of two paratypes (ANSP 130605, 48.2–64.1 mm SL); c&s paratype ANSP 130606 (vertebrae only) and 28 specimens of congeners (Tab. 2); radiographs taken with a Thermo Scientific PXS10-16W X-ray system, using an exposure of 40 kV and 300 µA via the Kevex X-Ray Source Control Interface 6.2.0. Counts of fin rays are reported as spines (I), unbranched anterior soft rays (i), or branched rays (Arabic numerals) for 14 type specimens and all Comparative Material examined. Vertebral counts include the Weberian complex as five vertebrae, with the first post-Weberian vertebra considered the sixth, and the complex caudal vertebra (PU1 + U1) counted as one. Interpretation of caudal skeleton based on Lundberg, Baskin (1969) and Sabaj, Arce (2021). The distribution map was prepared using QGIS v. 3.8.2 (QGIS Development Team, 2025). Museum codes follow Sabaj (2020).
TABLE 1 | Measurements and landmarks adapted from Friel (1994), Cardoso (2010), and Carvalho et al. (2015).
Measurement | Landmarks range |
|
1 | 1–2 | Standard length (SL) |
2 | 1–3 | Head length |
3 | 4–5 | Head width |
4 | 6–7 | Head width at eyes |
5 | 1–8 | Predorsal length |
6 | 1–9 | Prepectoral length |
7 | 8–10 | Length of unbranched dorsal-fin ray |
8 | 11–12 | Pectoral-fin spine length |
9 | 13–14 | Length of unbranched pelvic-fin ray |
10 | 15–16 | Length of uppermost unbranched (principal) caudal-fin ray |
11 | 17–18 | Length of lowermost unbranched (principal) caudal-fin ray |
12 | 11–13 | Pectoral-pelvic distance (abdominal length of Cardoso, 2010) |
13 | 13–19 | Pelvic-anal-fin distance |
14 | 20–21 | Cleithral width |
15 | 22–23 | Cleithrum length (cleithral process length of Carvalho et al., 2015) |
16 | 23–24 | Posterior-cleithral process length |
17 | 23–25 | Distance between posterior-cleithral processes |
18 | 26–27 | Width across parapophysis of fifth vertebrae |
19 | 28–29 | Distance between coracoid processes |
20 | 11–28 | Length of coracoid process |
21 | 8–30 | Body depth at dorsal-fin origin |
22 | 31–2 | Caudal-peduncle length |
23 | 31–32 | Caudal-peduncle depth (maximum) |
24 | 33–34 | Caudal-peduncle depth (least) |
25 | 35–36 | Caudal-peduncle width |
26 | 19–31 | Anal-fin base length |
27 | 3–37 | Head depth |
28 | 1–38 | Snout length |
29 | 39–40 | Interorbital width |
30 | 41–42 | Distance between anterior nostrils |
31 | 43–44 | Distance between posterior nostrils |
32 | 45–46 | Distance between maxillary barbels |
33 | 47–48 | Distance between anterior mental barbels |
34 | 49–50 | Distance between posterior mental barbels |
35 | 51–52 | Mouth width |
36 | 38–53 | Eye diameter |
FIGURE 1| Landmarks used to obtain measurements, based on previous studies (Friel, 1994; Cardoso, 2010; Carvalho et al., 2015) with adaptations to indicate precisely the landmarks. See Tab. 1 for name of distances.
TABLE 2 | Specimens of Pterobunocephalus radiographed for meristic counts. n = Number of specimens counted; asterisk denote holotype.
Species | Locality |
Bunocephalus albifasciatus | Beni River basin (ANSP 69193*, n = 1) |
Pterobunocephalus depressus | Beni River basin (FMNH 54338*, n = 1; INHS 150454, n = 1), Tamshiyacu River (INHS 54703, n = 1), Nanay River basin (INHS 36907, n = 1; INHS 46603, n = 1; INHS 43895, n = 1), upper Amazonas River basin (INHS 39126, n = 1; INHS 40167, n = 1) |
Pterobunocephalus dolichurus | Amazon River basin (ANSP 192609, n = 1; ANSP 207926, n = 1; ANSP 207936, n = 1; ANSP 139244, n = 4; ANSP 191481, n = 1; ANSP 207898, n = 1; ANSP 207851, n = 1), Nanay River basin (ANSP 182745, n = 1; ANSP 84943, n = 3; INHS 42141, n = 1; ANSP 43962, n = 1; INHS 141699, n = 1) |
Pterobunocephalus sp. ‘Guyana’ | Rupununi River basin (ANSP 180019, n = 2) |
Results
Pterobunocephalus carvalhoi, new species
urn:lsid:zoobank.org:act:B38B5EEC-B75E-4EE1-86C6-CF2C4B12210F
(Fig. 2; Tab. 3)
Bunocephalus aleuropsis. —Saul, 1975:115 (Ecological study of fishes; Upper Amazonian Ecuador).
Pterobunocephalus sp. —Friel, 1994:143 (tab. 4, comparative materials examined; phylogenetic relationships of Aspredinidae).
Pterobunocephalus depressus. —Barriga, 2012:110 (List of species; Ecuador).
Holotype. MEPN-I 20199, 69.9 mm SL, Ecuador, unnamed river 300 m from Sani Isla North oil well platform, headwaters of the Pañayacu River, Napo River basin, 00°27’53”S 76°19’09”W, 22 Oct 2001, R. Barriga & S. Greffa.
Paratypes. All from Ecuador. Napo River basin:MEPN-I 20147, 3, 43.7–69.8 mm SL, collected with holotype. MEPN-I 19931, 1, 43.6 mm SL, Orellana province, Rumiyacu River, 00°39’21”S 76°48’53”W, 8 Aug 2023, R. Hualpa. MEPN-I 20146, 2, 48.7–51.0 mm SL, Sucumbios province, Jivino River, near to the San Pedro de los Cofanes community, 00°08’35”S 76°52’08”W, 8 Mar 2002, R. Barriga, L. Rodriguez & L. Guamán. MEPN-I 20198, 1, 42.0 mm SL, Orellana province, unnamed stream tributary of the Rumiyacu River, 00°41’12”S 76°46’30”W, 7 Aug 2023, R. Hualpa. MEPN-I 19992, 2, 29.6–31.7 mm SL, Orellana province, unnamed stream tributary of the Rumiyacu River, 00°41’12”S 76°46’30”W, 7 Aug 2023, R. Hualpa. MEPN-I 19961, 1, 52.9 mm SL, Orellana province, tributary of the Rumiyacu River, eight km east of Dayuma village, 00°40’28”S 76°48’25”W, 10 Aug 2023. Pastaza River basin:MEPN-I 15406, 1, 74.3 mm SL, Ecuador, Pastaza province, Bobonaza River near to Moretecocha locality, 02°13’22”S 76°48’39”W, 17 Feb 1975, M. Olalla. Putumayo River basin:ANSP 130605, 2, 48.2–64.1 mm SL, 1 c&s, 46.4 mm SL, río Conejo [Blanco], tributary to río San Miguel, vicinity of Santa Cecília, inferred as 00°04’19”N 76°59’48”W, 15 Jun 1967, W. G. Saul & J. D. Lynch. ANSP 130606, 1 c&s, ca. 48.0 mm SL, same data as ANSP 130605.
FIGURE 2| Holotype of Pterobunocephalus carvalhoi from the Napo River basin, Ecuador, coordinates 00°27’53”S 76°19’09”W, MEPN-I 20199, 69.9 mm SL. Scale bar = 10 mm. Photo by P. Argüello.
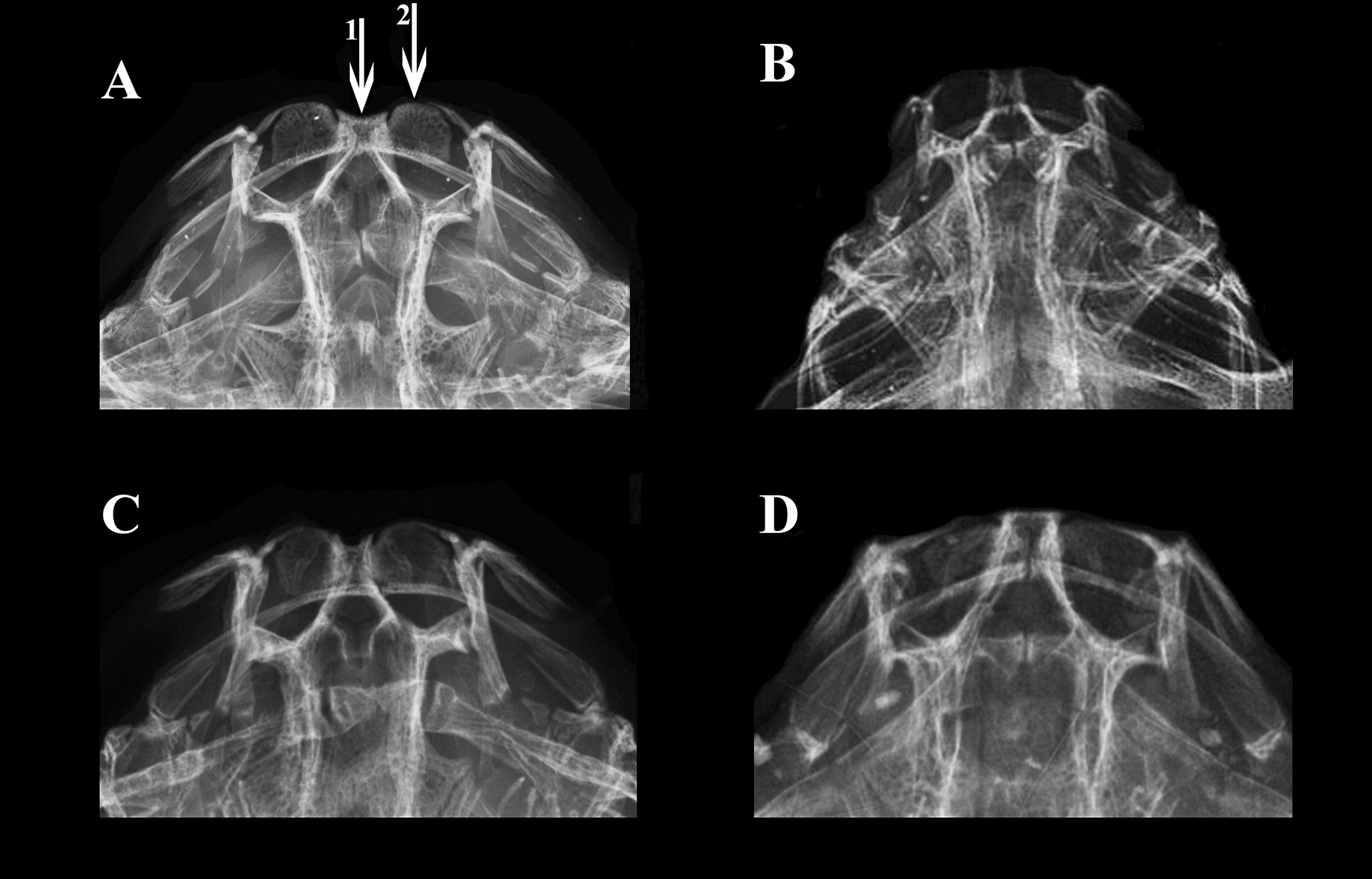
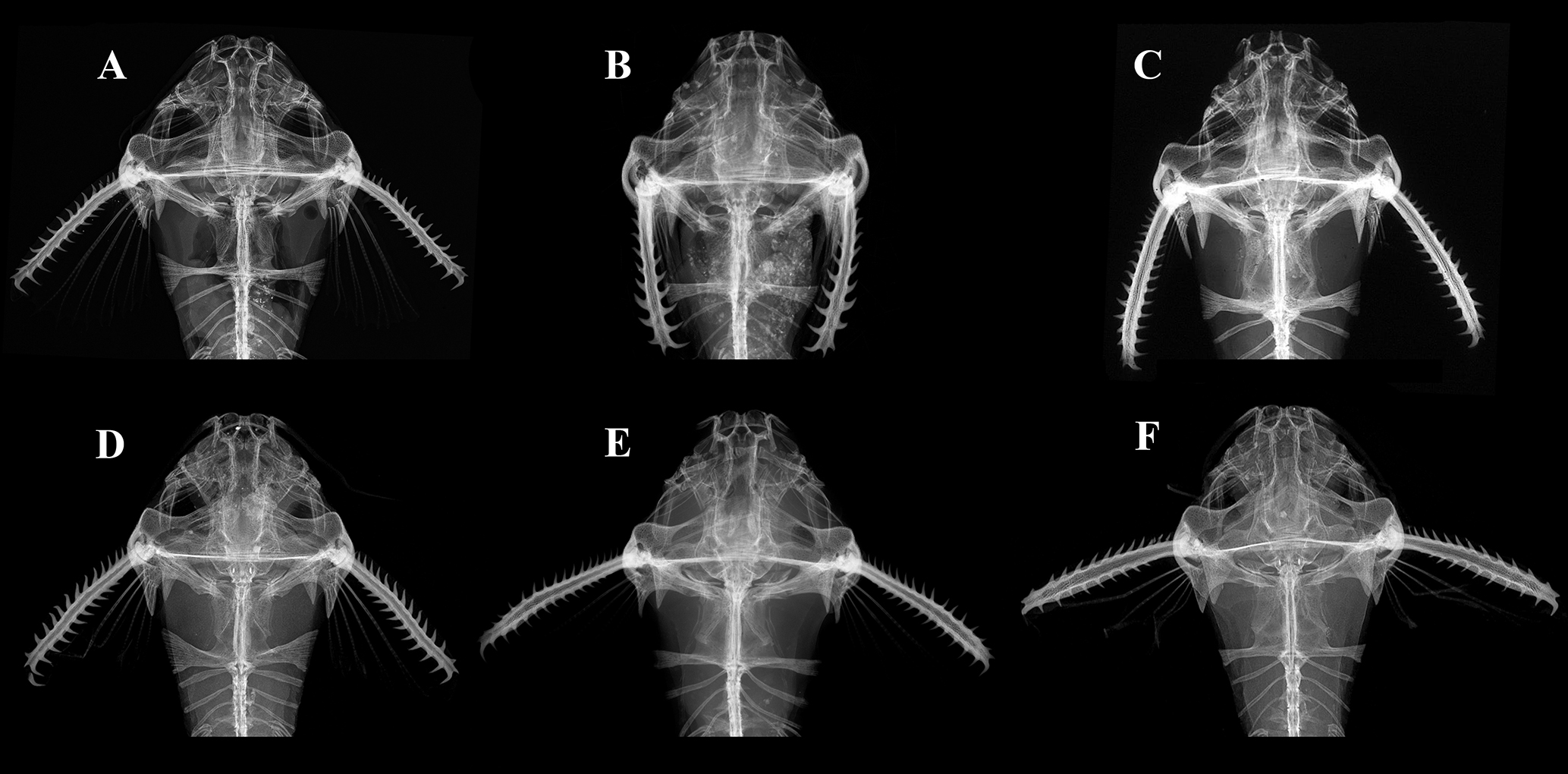
Diagnosis. Pterobunocephalus carvalhoi is diagnosed from all congeners by its larger eye diameter, 11.2–13.9% of HL (vs. 6.0–8.5% of HL in P. dolichurus and 6.1–9.1% of HL in P. depressus). It further differs from P. depressus by having, in dorsal view, anterior bony margin of mesethmoid sunken between (posterior to) those of premaxillae (Fig. 3A) vs. anterior bony margins of mesethmoid and premaxillae approximately level (or mesethmoid finishing more anteriorly than premaxillae) in P. depressus (Figs. 3B, D). The new species further differs from P. dolichurus in having a lower total number of anal-fin rays (10–11 vs. 15–16) and fifth vertebra with parapophysis slender proximally and conspicuously expanded distally, shaped like the blade of a shepherd’s axe in dorsal view (Figs. 4A, 6) vs. distal expansion lacking in P. dolichurus (Figs. 4E, F).
FIGURE 3| Radiography of the anterior portion of head in species of Pterobunocephalus. A. P. carvalhoi, ANSP 130605, 64.1 mm SL; B. P. depressus, holotype, FMNH 54338; C. P. dolichurus, from Upper Amazon, ANSP 84943, 56.1 mm SL; D. Holotype of Bunocephalus albifasciatus, ANSP 69193, 31.9 mm SL, synonym of P. depressus. Arrows represents anterior margin of (1) mesethmoid and (2) premaxilla. Images by J. P. Friel (B) and J. L. Crispim (A, C, D).
FIGURE 4| Radiographs of anterior body in species of Pterobunocephalus. A. P. carvalhoi, ANSP 130605, 64.1 mm SL; B. Holotype of Bunocephalus albifasciatus, ANSP 69193, 31.9 mm SL, synonym of P. depressus; C. P. depressus, holotype, FMNH 54338; D. Pterobunocephalus sp. from Rupununi River, ANSP 180019, 54.0 mm SL; E. P. dolichurus from Negro River, ANSP 207936, 43.2 mm SL; F. P. dolichurus from Upper Amazon, ANSP 84943, 56.1 mm SL. Images by J. P. Friel (C) and J. L. Crispim (A, B, D, E, F).
Description. Morphometric and meristic data for Pterobunocephalus carvalhoi summarized in Tab. 3. In lateral view, head dorsoventrally compressed; dorsal profile scarcely ascending from tip of snout to tip of posteriorly-directed spine-like protuberance associated with dorsal crest of Weberian complex (protuberance elevated above surrounding contours); dorsal profile descending, scarcely convex, from this point to posterior insertion of dorsal fin, then straight to scarcely concave to caudal fin. Ventral profile gradually descending and gently convex from tip of snout to pelvic-fin origin, then more or less straight to anal-fin origin, finishing straight to scarcely concave to base of caudal fin.
TABLE 3 | Morphometric and meristic data of the holotype, 13 alcohol preserved paratypes, and two c&s preserved paratypes of Pterobunocephalus carvalhoi. SD = Standard deviation. Morphometric range values include holotype.
Measurements | Holotype | Range | Mean | SD |
Standard length | 69.9 | 41.5–74.3 | – | – |
Percentage of standard length |
|
|
|
|
Head length | 18.7 | 18.5–19.8 | 19.2 | 0.4 |
Head width | 19.7 | 19.2–22.8 | 20.5 | 1.1 |
Head width at eyes | 13.4 | 11.5–15.2 | 13.7 | 1.2 |
Predorsal length | 35.2 | 33.6–36.9 | 35.2 | 1.1 |
Prepectoral length | 18.7 | 18.7–20.4 | 19.4 | 0.6 |
Length of unbranched dorsal-fin ray | 12.7 | 12.7–15.4 | 13.8 | 1.0 |
Pectoral-fin spine length | 18.2 | 16.7–19.6 | 18.4 | 1.0 |
Length of unbranched pelvic-fin ray | 12.4 | 11.8–13.0 | 12.5 | 0.4 |
Length of uppermost unbranched (principal) caudal-fin ray | 17.3 | 17.3–19.9 | 18.6 | 1.0 |
Length of lowermost unbranched (principal) caudal-fin ray | 14.7 | 14.1–17.7 | 15.8 | 1.2 |
Pectoral-pelvic distance (abdominal length of Cardoso, 2010) | 22.6 | 20.2–23.2 | 22.1 | 0.8 |
Pelvic-anal-fin distance | 17.5 | 17.1–19.5 | 18.0 | 0.8 |
Cleithral width | 27.0 | 24.7–29.0 | 27.0 | 1.4 |
Cleithrum length | 12.5 | 10.1–13.6 | 12.0 | 1.0 |
Posterior-cleithral process length | 7.1 | 4.5–9.3 | 6.8 | 1.2 |
Distance between posterior-cleithral processes | 22.0 | 19.9–23.9 | 21.6 | 1.2 |
Width across parapophysis of fifth vertebrae | 17.5 | 14.3–19.9 | 16.7 | 1.8 |
Distance between coracoid processes | 18.9 | 17.9–20.5 | 19.1 | 0.8 |
Length of coracoid process | 7.3 | 5.6–7.9 | 6.6 | 0.8 |
Body depth at dorsal-fin origin | 6.2 | 5.8–7.6 | 6.4 | 0.6 |
Caudal-peduncle length | 22.3 | 19.2–24.7 | 21.1 | 1.8 |
Caudal-peduncle depth (maximum) | 2.7 | 2.4–3.3 | 2.8 | 0.3 |
Caudal-peduncle depth (least) | 2.3 | 2.1–2.8 | 2.4 | 0.3 |
Caudal-peduncle width | 3.9 | 2.7–5.8 | 4.0 | 1.0 |
Anal-fin base length | 26.1 | 23.3–27.3 | 25.7 | 1.2 |
Head depth | 9.1 | 8.1–9.9 | 9.1 | 0.5 |
Percentage of head length |
|
|
|
|
Snout length | 20.5 | 19.6–23.0 | 21.3 | 1.2 |
Interorbital width | 18.5 | 17.5–21.3 | 19.9 | 1.2 |
Distance between anterior nostrils | 16.9 | 15.8–19.3 | 17.5 | 1.1 |
Distance between posterior nostrils | 29.0 | 25.9–33.7 | 29.7 | 2.3 |
Distance between maxillary barbels | 47.7 | 47.4–50.9 | 48.3 | 1.0 |
Distance between anterior mental barbels | 33.4 | 32.0–40.2 | 35.5 | 2.4 |
Distance between posterior mental barbels | 55.3 | 52.7–59.7 | 56.0 | 2.5 |
Mouth width | 42.2 | 24.7–45.7 | 40.6 | 2.8 |
Eye diameter | 11.5 | 11.2–13.9 | 12.1 | 0.9 |
Meristic data |
|
|
|
|
Dorsal-fin rays | I,4*(14) | |||
Pectoral-fin rays | I,5*(14) | |||
Pelvic-fin rays | i,5*(14) | |||
Principal caudal-fin rays | i,8,i*(14) | |||
Anal-fin rays | 10(3), 11*(11) | |||
Vertebrae counts | 38(3) | |||
Ribs | 4+4(2) | |||
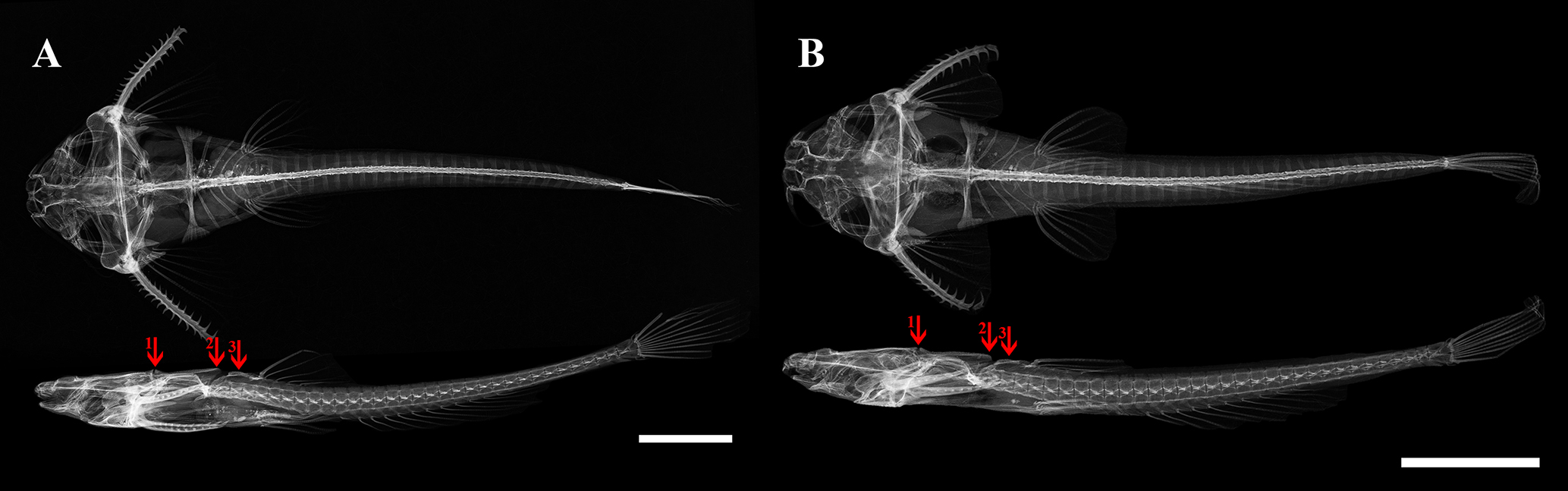
Skull ornamentation includes three dorsomedial bony protuberances (Fig. 5): first formed by posterodorsal tip of supraoccipital and anterior end of superficial lamina associated with dorsal crest of Weberian complex (arrow 1); second and most prominent at the posterior end of superficial lamina of Weberian complex dorsal crest (arrow 2); third and most discrete on dorsal face of middle nuchal plate (arrow 3).
FIGURE 5| Radiographs of paratype specimens of Pterobunocephalus carvalhoi. A. ANSP 130605 64.1 mm SL; B. ANSP 130605 48.2 mm SL. Scale bars = 10 mm. Arrows represents dorsomedial bony protuberances.
Eye small, without free orbital margin, located dorsally in anterior portion of head. Anterior nostril tubular with dorsal portion slightly enlarged, finishing near anterior margin of snout. Posterior nostril with larger opening, located near anterior margin of orbit. All barbels simple (unbranched). Maxillary barbel surpassing posterior tip of cleithral process. Anterior (inner) mental barbel shorter than posterior (outer) mental barbel, its tip slightly surpassing insertion of posterior mental barbel. Posterior mental barbel reaching gill opening. Mouth subterminal, upper lip more prominent than lower lip. In dorsal view, anterior margin of snout with conspicuous medial notch formed by anterior margin of mesethmoid less prominent than premaxillae (Figs. 4A, 6). Gill opening small, short slit restricted to ventral surface and finishing before lateral margin of head; covered by skin flap. Urogenital region with small papillae just posterior to anus. Skin covered with unculiferous tubercles; enlarged tubercles on body longitudinally aligned, forming multiple distinct rows on caudal-peduncle; one middorsal, two above lateral-line, one associated with lateral line, and two below lateral line.
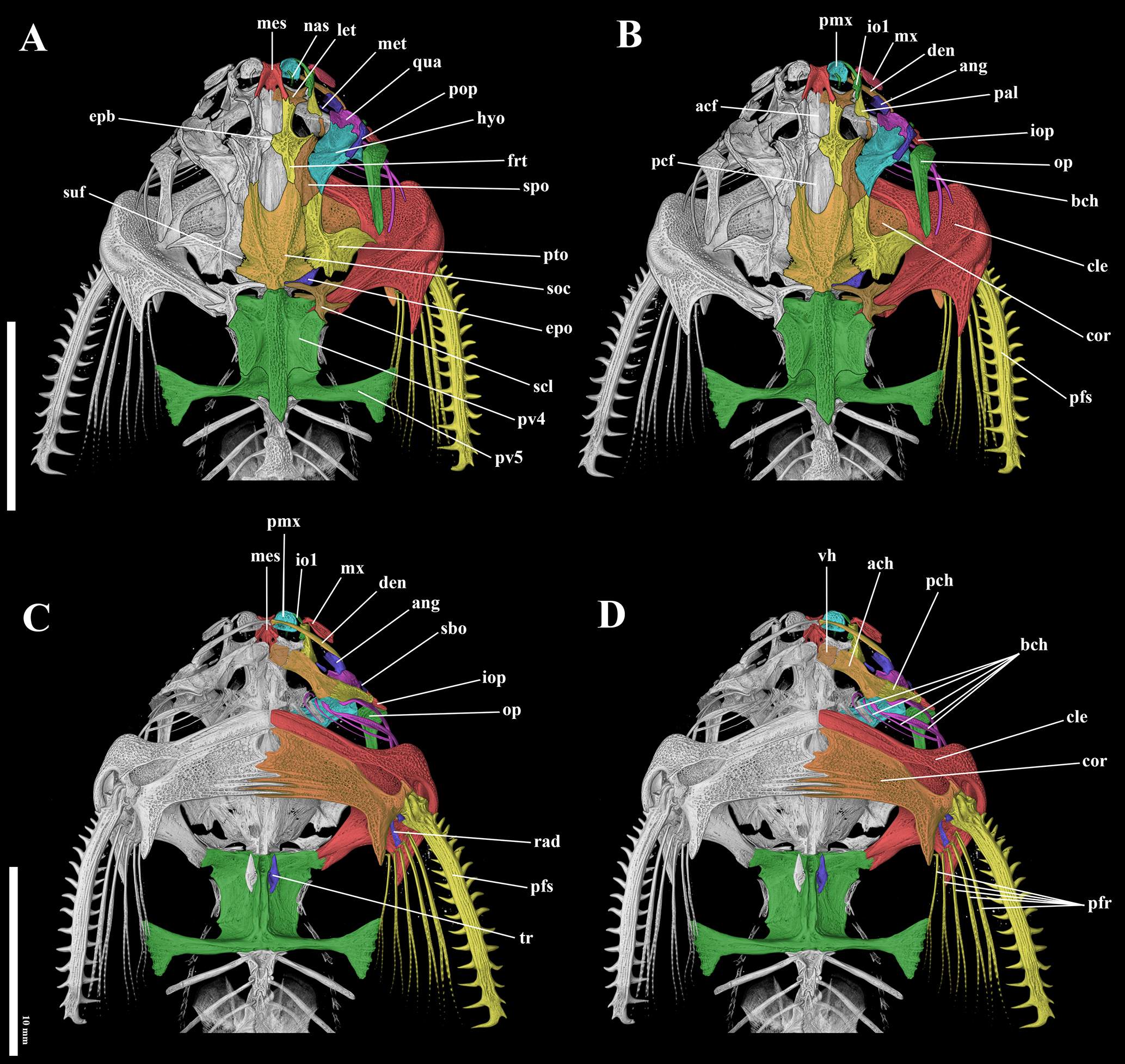
FIGURE 6| Computed tomography images of the holotype of Pterobunocephalus carvalhoi, ANSP 130605, 64.1 mm SL. A-B. Dorsal view; C-D. Ventral view. Abbreviations: acf: anterior cranial fontanel; ach: anterior ceratohyal; ang: anguloarticular; bch: branchiostegal rays; cle: cleithrum; cor: coracoid; den: dentary; epb: epiphyseal bar; epo: epioccipital; frt: frontal; hyo: hyomandibular; io1: infraorbitals; iop: interopercle; let: lateral ethmoid; mes: mesethmoid; met: metapterygoid; mx: maxilla; nas: nasal; op: opercle; pal: palatine; pcf: posterior cranial fontanel; pch: posterior ceratohyal; pfr: pectoral-fin rays; pfs: pectoral fin spine; pmx: premaxilla; pop: preopercle; pto: pterotic; pv4: parapophysis of fourth vertebra; pv5: parapophysis of fifth vertebra; qua: quadrate; rad: pectoral-fin radial; sbo: subpreopercle; scl: supracleithrum; soc: supraoccipital; spo: sphenotic; suf: supratemporal fossa; tr: tripus; vh: ventral hypohyal. Scale bars = 10 mm.
Dorsal fin located in anterior half of body; rays I,4* (14), spinelet absent; last dorsal-fin ray adnate to body by membrane for less than half its length. Adipose-fin absent. Anal fin rays 10 (3) or 11* (11); its origin just posterior to posterior tip of compressed dorsal fin; last anal-fin ray adnate to body by membrane for less than half its length. Pectoral-fin origin just posterior to gill opening, adpressed tip reaching pelvic-fin origin in adults and shorter in small individuals; rays I,5* (14) (Fig. 6D – “pfr”); pectoral spine gently curved with large retrorse to erect serrations along anterior (13–15 serrations) and posterior (9–12) margins; serrations increasing in number according body size (Figs. 6B, C – “pfs”). Pelvic-fin origin at vertical through dorsal-fin origin; rays i,5* (14). Principal caudal-fin rays i,8,i* (14); caudal-fin distal margin profile convex with upper and lower principal caudal-fin rays shorter than branched rays.
Osteology. The following descriptions mostly refer to Fig. 6. Mesethmoid with slightly concave anterior margin and small anterolateral projections. Anterior cranial fontanel wide; anterior extent poorly delimited; posterior margin obtusely triangular, finishing almost level with transverse through posterior margin of orbit. Posterior cranial fontanel scarcely wider than anterior one, anterior half bordered by frontals; posterior half formed by deep U-shaped concavity in supraoccipital bone. Epiphyseal bar wide with contact/suture evident between contralateral halves. Frontal with concavity extended by acute lateral projection forming dorsal and posterior margin of orbit; anterior arm of frontal contacting mesethmoid and lateral ethmoid; posterior arm sharing long suture laterally with sphenotic and short suture posteriorly with supraoccipital; medial margin forming posterior rim of anterior cranial fontanel, epiphyseal bar and anterior rim of posterior cranial fontanel (Fig. 6A – “frt”). Sphenotic with anterior portion angled laterally, sutured to lateral projection of frontal; posterior portion wider, contacting pterotic and supraoccipital (Fig. 6A – “spo”). Pterotic with anterior arm sutured medially to supraoccipital and anteriorly to sphenotic and briefly to hyomandibula; posterior portion with broad aliform lateral expansion, the distal tip of which lies above (but does not contact) supracleithrum. Posterolateral margin of supraoccipital sutured to epioccipital and briefly contacting medial arm of supracleithrum; posterior tip close but not contacting superficial lamina associated with dorsal crest of Weberian complex (Fig. 6A – “soc”). Supratemporal fossa closed, located at posterior portion of contact between pterotic and supraoccipital (Fig. 6A – “suf”).
Parasphenoid narrow anteriorly and expanded posteriorly; posterior region contacting prootic, exoccipital and basioccipital. Lateral ethmoid small, compact, extended laterally in the shape of apaving stone. Infraorbital 1 gently sickle shaped in dorsal view with posterior end contacting lateral ethmoid. Prootic rectangular, located ventrally on neurocranium, contacting posterior half of parasphenoid and pterotic. Exoccipital located ventrally on neurocranium with anterior region narrow and expanded posteriorly; shares deeply interdigitating sutures with parasphenoid. Premaxilla compact with convex anterior margin and straight posterior margin. Dentary teeth present but extremely small, conical and arranged in two rows on dorsal surface of long, slender dentary.
Weberian apparatus with dorsal crest supporting broad lateral expansions; anterior portion fused to the supracleithrum dorsally and basioccipital ventrally; anterolateral portion contacting cleithrum and supracleithrum; posterior surface contacting the 6th vertebrae. Parapophysis of fifth vertebra expanded distally forming the shape of a shepherd’s axe. Tripus bone ear shaped, visible on anteroventral surface of Weberian apparatus.
Metapterygoid elongate, posteriorly connected to quadrate by cartilage (Figs. 6A – “met”). Hyomandibula well developed, tightly sutured anteriorly with quadrate, medially contacting sphenotic and laterally associated with preopercle; posteriormost extremity briefly sutured to pterotic. Quadrate with concave in anterior face for receiving anguloarticular. Dentary long and slender, gently bowed (Figs. 6B, C – “den”). Maxilla elongated with lateral laminar portion. Palatine tubular, articulating anteriorly with proximal head of maxilla and infraorbital 1, and medially with lateral ethmoid. Nasal bone free and small, tubular. Ventral hypohyal trapezoidal with rounded corners, broadly contacting anterior ceratohyal. Anterior ceratohyal elongated with gently concave free margins; expanded posterior end broadly contacting posterior ceratohyal. Posterior ceratohyal wider anteriorly, posteriorly contacting interopercle (Fig. 6D – “pch”). Interopercle small and posteriorly contacting opercle. Opercle elongated, wider anteriorly (club-shaped). Subpreopercle present; small and tubular, weakly ossified (Fig. 6C – “sbo”). Five pairs of slender branchiostegal rays: first one in contact with posterior ceratohyal. Urohyal triangular, like the tip of an arrow; posterior margin with medial notch.
Centra six to nine each supporting one pair of ribs (four pairs total). Lateral-line canal nearly complete, last pore located near base of caudal skeleton; formed by simple ossified tubes. Total vertebrae 38(3), first five fused into Weberian complex; 33 post Weberian vertebrae including compound PU1+U1 counted as one. Caudal skeleton with two triangular plates distally separated by partial diastema. Lower plate fused to compound caudal centrum (PU1+U1) and composed of fused parhypural and hypurals one and two (PH+HY1+2); proximal portion of lower plate with lateral bony shelf formed by fusion of primary and secondary hypurapophyses supported, respectively, by parhypural and hypural portions of plate; shelf thin but expansive longitudinally and transversely with contralateral shelves outlining shield shape in dorsal view (tapered posteriorly). Upper plate composed of fused hypurals, presumably three through five (HY3+4+5). Pleurostyle interpreted as element with proximal portion sharing intervertebral joint with compound caudal centrum (PU1+U1) and thus incorporating remnant of second ural centrum (U2); pleurostyle widens distally and appears wedged between (not fused to) upper hypural plate posteroventrally and neural ossifications of caudal centra anterodorsally. Epural not clearly independent from other neural ossifications of caudal centra; epural interpreted as semi-elliptical element (convex ventrally) occupying much of concave space between neural arch of second preural central (PU2) and pleurostyle; epural partially sutured or fused to modified neural ossifications of the compound caudal (PU1+U1) and second ural (U2) centra with distinct oval gap separating ventralmost margin of epural from surrounding ossifications. One upper and one lower small caudal procurrent rays.
Coloration in alcohol. Ground color dorsally brown (head) or beige (posterior body), ventrally beige to pale yellow. Dorsal surface with conspicuous dark brown to black saddle centered on dorsal-fin insertion; anterodorsal margin of saddle concave, posterodorsal margin irregular; dark saddle expanded laterally and ventrally onto sides of body with dark pigmentation continuous anteriorly to pectoral girdle and posteriorly along ventral half of body to caudal fin, effecting bicolored appearance to posterior body (pale above, dark below). One to three additional saddles evident on posterior body, but smaller, more irregular and lacking the dark intensity of the main saddle. Dorsal surface of head mottled with dark blotches variable in size and intensity, some coalesced into irregular darker bars, stripes or larger blotches associated with anterior nostrils, region ventrolateral to orbit as well as check and opercle, and middorsally on posterior head and nuchal region.
In six specimens of the type series (including the holotype), a pattern of four spots was observed: one small, irregular blotch near the occipital tip; two somewhat oval blotches at the posterior edge of the pectoral-fin base; and one larger, somewhat rectangular blotch over the parapophysis of the fourth vertebra. Dorsal fin more or less uniformly dark brown except for pale distal margin. Pectoral fin mostly dark brown except for pale tips of spine and branched rays. Pelvic fins hyaline with dark brown spots, sometimes forming a mottled pattern. Anal fin hyaline dark brown spots, sometimes forming a mottled pattern, with distal tip of rays pale. Caudal fin hyaline with small brown blotches effecting faint banding pattern. Barbels light brown with dark brown blotches (Fig. 7).
FIGURE 7| Holotype, MEPN-I 20199, 69.9 mm SL (left), and paratype, ANSP 130605, 64.1 mm SL (right) of Pterobunocephalus carvalhoi showing variation in color pattern.
Etymology. The specific epithet “carvalhoi” honors Brazilian ichthyologist Tiago Pinto Carvalho, Professor at the Universidad Nacional de Colombia, Bogotá, in recognition of his diverse and far-reaching contributions to the study of Neotropical fishes, particularly the taxonomy and systematics of the family Aspredinidae. A noun in a genitive case.
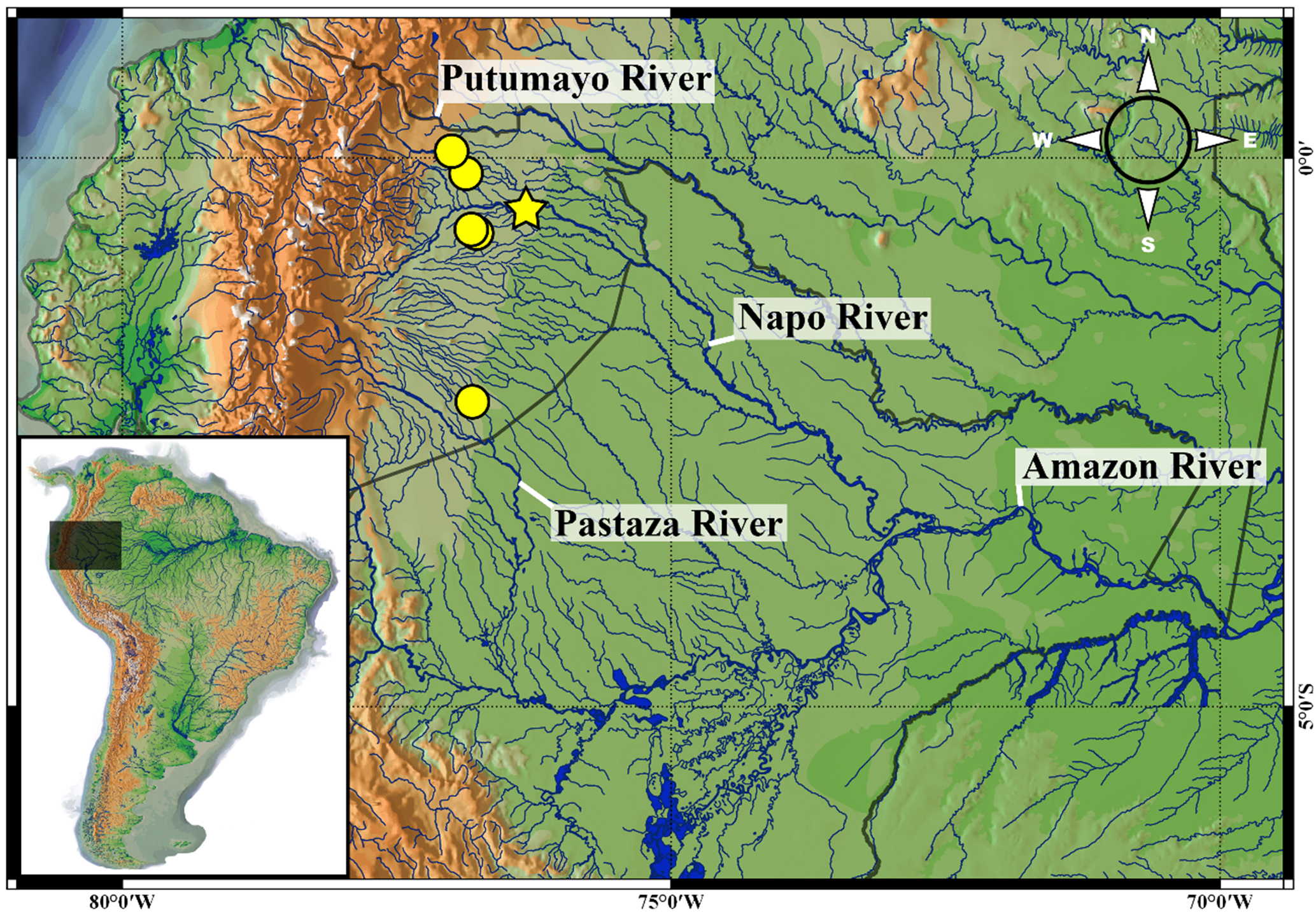
Geographical distribution. Pterobunocephalus carvalhoi is known from the upper reaches of left-bank tributaries to the upper Amazon River. The new species was recorded at six sites, including the main channel of the Napo River and three of its tributaries, one site in the Bobonaza River (a tributary of the Pastaza River) and one in the Blanco River, which flows into the Putumayo River basin and connects it to the Napo basin during high waters (Fig. 8).
FIGURE 8| Distribution map of the Pterobunocephalus carvalhoi, Napo, Pastaza and Putumayo River basins. Holotype locality: yellow star; paratypes: yellow dots.
Ecological notes. Aspredinids are commonly captured from the bottoms of rivers and streams, usually within accumulations of leaf litter (JLCR, MHS, and R. P. Ota, pers. obs.). Paratype specimens of Pterobunocephalus carvalhoi were found exclusively in the swift channel of the Conejo River, and associated with substrates of solid sand, clay, and bedrock. Saul (1975:99) described the Conejo channel as ~6 m wide and concealed by overhanging vegetation. Its water varied from light, yellow-brown during the high-water season to olive or tea color during dry periods. During the summer of 1967, water temperature was ~22.5°C, pH was ~6.5, and dissolved oxygen was ~6.5 ppm. Paratypes of the new species collected in the Jivino River (MEPN-I 20146), a tributary of the Napo River, were found in a stretch similarly characterized by dense riparian vegetation (Fig. 9). The main components of their diet included insects, plant debris, and fly larvae (Chironomidae) (Saul, 1975).
FIGURE 9| Stretch of the Jivino River, Napo River basin, ~300 m from the sampling site of the type series of Pterobunocephalus carvalhoi, MEPN-I 20146. Photo by Pablo Argüello.
Conservation status. According to IUCN criteria and categories, we recommend categorizing Pterobunocephalus carvalhoi as Least Concern (LC), even though only 15 specimens have been recorded. Aspredinids are generally rare in scientific collections, likely because they belong to a taxonomic group that is difficult to collect. We documented the distribution of this species at six sites across three hydrographic basins (Napo, Pastaza, and Putumayo) in the Upper Amazon. Specimens have also been reported from the Blanco River, which connects the Aguarico (Napo River basin) with the San Miguel River (Putumayo River basin). Although these specimens were collected near the Aguarico River, this channel ultimately flows into the Putumayo River basin, indicating the species’ occurrence in the third hydrographic basin of the Upper Amazon.
Discussion
The genus Pterobunocephalus is widely distributed across South America, including the Amazon, Essequibo, Orinoco, and Paraná-Paraguay river systems (Sleen, Albert, 2017; Dagosta, de Pinna, 2019; Friel, 2003). In addition to the three species recognized here, hidden diversity may exist in this genus and a comprehensive taxonomic revision is needed. The generic diagnosis provided by Friel (1994) remains valid for the most part, although we did not observe a notch in the upper jaw of P. depressus. Also, it is not known if females of the new species carry embryos, a maternal behavior reported in several other aspredinid genera (Burgess, 1989; Friel, 1994).
Radiographs of widely distributed specimens of Pterobunocephalus, including the Amazon, Nanay, Tamshiyacu, Solimões, Takutu, Rupununi, and Beni River basins, revealed two osteological features that are important for evaluating variation: (i) the shape of the parapophysis of the fifth vertebra: expanded distally in P. carvalhoi (Figs. 4A, 5, 6) and P. depressus (Fig. 4C) versus not expanded distally in P. dolichurus (Figs. 4E, F); and (ii) upper jaw (anterior margin of mesethmoid) with medial concavity in P. carvalhoi (Figs. 3A, 6) and P. dolichurus (Fig. 3C), versus straight in P. depressus (Fig. 3B).
In the new species, the parapophysis of the fifth vertebra is narrow proximally and expanded distally into a blade-like shape that resembles a shepherd’s axe in dorsal view (Figs. 4A, 5, 6). This condition is similar to that observed in the holotype of P. depressus (Fig. 4C), but somewhat lacking in the holotype of its junior synonym, Bunocephalus albifasciatus (Fig. 4B), which is a juvenile specimen (31.9 mm SL). In Pterobunocephalus sp. from the Rupununi River, the parapophysis of the fifth vertebra expands more continuously from its base (Fig. 4D), whereas in P. dolichurus, the width of the parapophysis is more uniform, scarcely expanded distally (Figs. 4E, F).
The coordinates reported by Saul (1975:94), 00°06’N 76°51’W, point to an area east of the vicinity of Santa Cecilia and do not correspond precisely to the collection site of paratype specimens ANSP 130605 and 130606. Saul (1975:115) noted that all aspredinids were collected in the Conejo River, which corresponds to the Río Blanco, a tributary to the San Miguel River (Putumayo basin) connecting it to the Aguarico River (Napo basin). Considering his map of the study site (Saul, 1975:95, fig. 2), we propose new coordinates to more accurately locate the paratype sampling site: río Blanco [Conejo], 00°04’19”N 76°59’48”W.
Thus, our study contributes to the knowledge of the aspredinid genus Pterobunocephalus, extending its distribution to the extreme northwestern lowlands of the Amazon basin and providing detailed descriptions of its morphological diversity. This genus has not yet undergone a comprehensive revision, which is necessary to better understand its hidden diversity and the phylogenetic relationships among its lineages.
Comparative material examined. Bolívia: Bunocephalus albifasciatus: ANSP 69193, 1, holotype, 31.86 mm SL, Chapare River. Pterobunocephalus depressus: INHS 150454, 1, 37.96 mm SL, Beni River. AMNH 37014, 1, 45.46 mm SL, Beni River. Brazil: Pterobunocephalus depressus: ANSP 182774, 1, 44.66 mm SL, Solimões River. Pterobunocephalus dolichurus: ANSP 192609, 1, 44.12 mm SL, Amazon River basin. ANSP 207926, 1, 41.91 mm SL, Amazon River basin. ANSP 207936, 1, 43.19 mm SL, Amazon River basin. ANSP 139244, 4, 34.87 – 42.38 mm SL, Amazon River basin. ANSP 191481, 1, 30.75 mm SL, Amazon River basin. ANSP 207898, 1, 39.2 mm SL, Amazon River basin. ANSP 207851, 1, 36.04 mm SL, Amazon River basin. Guyana: Pterobunocephalus sp.: ANSP 180019, 2, 51.39–53.97 mm SL, Rupununi River. Peru: Pterobunocephalus depressus: INHS 54703, 1, 44.3 mm SL, Tamshiyacu River. INHS 36907, 1, 36.71 mm SL, Nanay River. INHS 46603, 1, 37.43 mm SL, Nanay River. INHS 39126, 1, 58.86 mm SL, Amazonas River. INHS 40167, 1, 81.14 mm SL, Orosa River. INHS 43895, 1, 45.15 mm SL, trib Nanay River. IIAP 5951, 1, 73.48 mm SL. IIAP 5892, 1, 64.45 mm SL. IIAP 5996, 2, 46.59–47.97 mm SL. IIAP 2679, 2, 46.37–46.59 mm SL. IIAP 1401, 1, 54.74 mm SL. Pterobunocephalus dolichurus: ANSP 182745, 1, 69.62 mm SL, Nanay River. ANSP 84943, 3, 52.07–56.11 mm SL, Nanay River. INHS 42141, 1, 62.11 mm SL, Nanay River. ANSP 43962, 1, 60.52 mm SL, Nanay River. INHS 141699, 1, 35.57 mm SL, Nanay River.
Acknowledgments
We are grateful to the following people for the loan of specimens: David Werneke and Jonathan Armbruster, from Auburn University Museum Fish Collection (AUM); and Enrique Santoyo Brito, from Illinois Natural History Survey (INHS). We also thank Bruno F. Melo and Ryan Thoni for welcoming us at the American Museum of Natural History (AMNH), and Junior Chuctaya and Morgan Ruiz-Tafur for welcoming us at the Instituto de Investigaciones de la Amazonia Peruana (IIAP). We also thank to Rafaela P. Ota, Buno F. Melo, José L. O. Birindelli and Cristhian C. Conde-Saldaña for the suggestions for the manuscript; and Ramiro Barriga and Ronnie Hualpa, for the collection and management of the MEPN specimens used in this study.
References
Barriga R. Lista de peces de agua dulce e intermareales del Ecuador. Politécnica. 2012; 30(3):83–119.
Böhlke JE. Results of the Catherwood Foundation Peruvian Amazon Expedition. Petacara, a new genus for the bunocephalid catfish, Bunocephalus dolichurus Delsman. Not Nat Acad Nat Sci Phila. 1959; 318:1–06.
Burgess WE. An atlas of freshwater and marine catfishes: a preliminary survey of the Siluriformes. Tfh Pubns Inc; 1989.
Cardoso AR. Filogenia da família Aspredinidae adams, 1854 e revisão taxonômica de Bunocephalinae Eigenmann & Eigenmann, 1888 (Teleostei: Siluriformes: Aspredinidae). [PhD Dissertation]. Porto Alegre: Pontifícia Universidade Católica do Rio Grande do Sul; 2008.
Cardoso AR. Bunocephalus erondinae, a new species of banjo catfish from southern Brazil (Siluriformes: Aspredinidae). Neotrop Ichthyol. 2010; 8(3):607–13. https://doi.org/10.1590/S1679-62252010000300005
Carvalho TP, Arce H M, Reis RE, Sabaj Pérez MH. Molecular phylogeny of banjo catfishes (Ostaryophisi: Siluriformes: Aspredinidae): a continental radiation in South American freshwaters. Mol Phylogenet Evol. 2018; 127:459–67. https://doi.org/10.1016/j.ympev.2018.04.039
Carvalho TP, Cardoso AR, Friel JP, Reis RE. Two new species of the banjo catfish Bunocephalus Kner (Siluriformes: Aspredinidae) from the upper and middle rio São Francisco basins, Brazil. Neotrop Ichthyol. 2015; 13(3):499–512. https://doi.org/10.1590/1982-0224-20140152
Dagosta FCP, de Pinna M. The fishes of the Amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 431:1–163. https://doi.org/10.1206/0003-0090.431.1.1
Fowler HW. Zoological results of the second Bolivian expedition for the Academy of Natural Sciences of Philadelphia, 1936-1937. Part II. Additional new fishes. Not Nat Acad Nat Sci Phila. 1943; 120:1–07.
Friel JP. A phylogenetic study of the Neotropical banjo catfishes (Teleostei: Siluriformes: Aspredinidae). [PhD Dissertation]. Ann Arbor: Duke University; 1994.
Friel JP, Carvalho TP. Family Aspredinidae (Banjo catfishes). In: Reis R, Kullander SO, Ferraris Jr. CJ, editors. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.261–67.
Lundberg JG, Baskin JN. The caudal skeleton of the catfishes, order Siluriformes. Am Mus Novit. 1969; 2398:1–49.
QGIS Development Team. QGIS Geographic Information System. Version 3.8.2. Open Source Geospatial Foundation Project. 2025. Available from: https://qgis.org
Sabaj MH. Codes for natural history collections in ichthyology and herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
Sabaj MH, Arce H M. Towards a complete classification of the Neotropical thorny catfishes (Siluriformes: Doradidae). Neotrop Ichthyol. 2021; 19(4):e210064. https://doi.org/10.1590/1982-0224-2021-0064
Saul WG. An ecological study of fishes at a site in upper Amazonian Ecuador. Proc Acad Nat Sci Phila. 1975; 127:93–134.
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Sleen P, Albert JS. Field guide to the fishes of the Amazon, Orinoco, and Guianas. Princeton: Princeton University Press; 2017.
Authors
![]() Jefferson Luan Crispim1,2
Jefferson Luan Crispim1,2 ![]() ,
, ![]() Pablo Argüello3,
Pablo Argüello3, ![]() Gabriel de Souza da Costa e Silva1,
Gabriel de Souza da Costa e Silva1, ![]() Claudio Oliveira1,
Claudio Oliveira1, ![]() Kyle R. Luckenbill2 and
Kyle R. Luckenbill2 and ![]() Mark H. Sabaj2
Mark H. Sabaj2
[1] Laboratório de Ecologia e Conservação da Amazônia (LABECA), Universidade Federal Rural da Amazônia, campus Capitão Poço, Rua Professora Antônia Cunha de Oliveira, Vila Nova, 68650-000, Capitão Poço, PA, Brazil. (FPTR) pauloribeiro995@gmail.com (corresponding author), (BSP) brunoprudente8@gmail.com.
[2] Laboratório de Zoologia (LabZoo), Universidade Federal do Pará, Campus Universitário do Marajó-Breves, Alameda IV, 3418, 68000-000, Breves, PA, Brazil. (TMSF) freitastms@gmail.com.
Authors’ Contribution 

Jefferson Luan Crispim: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Validation, Visualization, Writing-original draft, Writing-review and editing.
Pablo Argüello: Data curation, Investigation, Methodology, Validation, Writing-review and editing.
Gabriel de Souza da Costa e Silva: Investigation, Methodology, Supervision, Validation, Writing-review and editing.
Claudio Oliveira: Data curation, Funding acquisition, Investigation, Project administration, Resources, Supervision, Validation, Writing-review and editing.
Kyle R. Luckenbill: Data curation, Methodology, Software, Supervision, Validation, Visualization, Writing-review and editing.
Mark H. Sabaj: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
This research was funded by the Fundação de Amparo à Pesquisa no Estado de São Paulo (FAPESP processes 2024/07526–2 and 2023/01862–8 to JLCR). This research was also funded by the James E. Böhlke and Eugenia B. Böhlke Memorial Endowment Fund (ANSP). CO received financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP grant 2020/13433–6), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq proc. 306054/2006–0 and 441128/2020–3), and Pro-Reitoria de Pesquisa da Universidade Estadual Paulista Júlio de Mesquita Filho (Prope-UNESP). X-ray nanoCT scanner at Drexel University acquired via grant from the National Science Foundation Major Research Instrumentation Program (NSF-MRI CMMI-2216175; PI Antonios Konstos and Antonios Zavaliangos; Senior Personnel MHS).
How to cite this article
Crispim JL, Argüello P, Silva GSC, Oliveira C, Luckenbill KR, Sabaj MH. After eight decades: a new species of Pterobunocephalus (Siluriformes: Aspredinidae) from the upper Putumayo, Napo and Pastaza rivers, Ecuador. Neotrop Ichthyol. 2026; 24(1):e250162. https://doi.org/10.1590/1982-0224-2025-0162
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 22, 2025
Accepted December 22, 2025
![]() Submitted September 17, 2025
Submitted September 17, 2025
![]() Epub April 17, 2026
Epub April 17, 2026