![]() Francisco de Paulo Tavares Ribeiro1
Francisco de Paulo Tavares Ribeiro1 ![]() ,
, ![]() Bruno da Silveira Prudente1 and
Bruno da Silveira Prudente1 and ![]() Tiago Magalhães da Silva Freitas2
Tiago Magalhães da Silva Freitas2
PDF: Download Here | Supplementary: S1 | Cite this article
Associate Editor: ![]() Lilian Casatti
Lilian Casatti
Section Editor: ![]() Fernando Pelicice
Fernando Pelicice
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Este estudo investigou a influência de fatores ambientais sobre as características tróficas e as interações entre duas espécies de peixes simpátricas e filogeneticamente relacionadas, Copella arnoldi e Pyrrhulina capim, em riachos da Amazônia Oriental. As coletas ocorreram de março de 2019 a fevereiro de 2020. Nossa hipótese foi que a variabilidade ambiental modula os atributos tróficos e as interações entre essas espécies, ocasionando mudanças na composição da dieta, amplitude de nicho, intensidade alimentar e grau de sobreposição alimentar. Modelos Lineares Generalizados (GLMs) relacionaram as características tróficas às variáveis ambientais, e um modelo nulo testou a significância da sobreposição observada. Foram analisados 239 exemplares de C. arnoldi e 409 de P. capim, ambas onívoras com tendência à insetivoria. Copella arnoldi consumiu principalmente Formicidae, Diptera e Material vegetal, enquanto P. capim alimentou-se de Coleoptera, Diptera e Formicidae. Os GLMs indicaram que as categorias tróficas foram influenciadas por velocidade da água, pH, banco de folhas, temperatura e cobertura do dossel. A amplitude de nicho e a intensidade alimentar variaram conforme temperatura, velocidade da água, largura do riacho, cobertura do dossel e oxigênio dissolvido. A sobreposição trófica foi alta e superou os valores simulados, mas não foi influenciada pelas variáveis ambientais. Nossos achados ampliam a compreensão das interações tróficas entre peixes simpátricos e sua modulação por variações ambientais em riachos neotropicais.
Palavras-chave: Coexistência, Peixes, Precipitação, Recursos alimentares, Sobreposição trófica.
Introduction
Studies on the trophic ecology of fish in the Neotropical region are fundamental for understanding ecological relationships, both inter- and intraspecific, within aquatic ecosystems (Guglielmetti et al., 2019; Benone et al., 2020; Soares et al., 2020; Esteves et al., 2021). Despite their importance, significant knowledge gaps still exist, particularly those related to the Eltonian shortfalls (Hortal et al., 2015), which concerns our limited understanding of species interactions such as predation, competition, and resource sharing. Although substantial progress has been made in recent decades regarding the feeding habits of Amazonian fish (Silva et al., 2016; Barros et al., 2024), such studies remain scarce in the Amazon, a region that harbors the greatest fish biodiversity on Earth, with approximately 2,500 valid species (Fricke et al., 2024). This remarkable biodiversity is largely associated with streams, which represent a considerable portion of the region’s river basins (Silva et al., 2016; de Carvalho et al., 2017), reinforcing the need for further investigation into trophic relationships in these environments.
Among the vast ichthyofauna found in Amazonian streams, fish from the family Lebiasinidae (Characiformes) stand out. This group comprises around 75 small species, characterized by fusiform bodies, terminal mouths, and striking coloration, commonly known as pencil fish (Dagosta, de Pinna, 2019). These species exhibit omnivorous diets, with a predominance of arthropod consumption (Silva et al., 2016; Soares et al., 2020). They are also popular in the aquarium trade due to their small size, social behavior, and vibrant colors (Abe et al., 2015; de Moraes et al., 2019). Among them, Copella arnoldi (Regan, 1912) and Pyrrhulina capim Vieira & Netto-Ferreira, 2019 generally occur in sympatry in streams of the eastern Amazon (Montag et al., 2008; Benone, 2012; Barros et al., 2020; Buckup, 2021; Roussel et al., 2021).
Streams are small watercourses with low primary productivity, dependent on riparian vegetation for the input of organic matter and for preventing siltation (Graziano et al., 2022). Marginal vegetation also provides shade, helping to regulate water temperature and reduce evaporation, thereby creating a stable environment for fish (Teresa, Casatti, 2013; Dala-Corte et al., 2020). The pronounced rainfall variability in the Amazon is considered one of the main drivers of the structural dynamics of streams, adjacent vegetation, and the physical and chemical properties of the water (Espírito-Santo et al., 2009; Zeni, Casatti, 2014).
During the rainy season (from November to April), the increased quantity and diversity of resources in streams, resulting from the transport of organic matter from surrounding vegetation, provides many fish species with greater food availability (Borba et al., 2008). During this period, species tend to focus their consumption on items with higher energetic value, thereby reducing their trophic niche breadth (Barros et al., 2024). In contrast, during the dry season (from May to October), the decline in resource availability and diversity forces many species to reduce their feeding intensity and adjust a more generalist diet to meet their basic energy requirements (Barros et al., 2024). These trophic adjustments often depend on trophic strategy and are most common among generalist species that feed on a wide range of invertebrates and detritus (Soares et al., 2020).
Due to these characteristics, stream fish in tropical regions often do not exhibit trophic specializations, showing instead significant dietary flexibility, a pattern observed across various freshwater ecosystems (Pini et al., 2019; Esteves et al., 2021; Neves et al., 2021). This flexibility in resource use, influenced by behavioral and physiological traits, affects trophic interactions and enables ecologically similar species to coexist in the same space and time through trophic sympatry (Dias et al., 2022). This condition, where species share resources but display distinct feeding habits or resource-use strategies, reduce the likelihood of intense competition (Blois et al., 2014; Worischka et al., 2015; Baldasso et al., 2019; Esteves et al., 2021).
Such coexistence patterns are not random but are strongly influenced by environmental conditions, especially habitat structure and resource availability, which shape trophic interactions through both intra- and interspecific dynamics (Manna, Rezende, 2021). These interactions involve complex mechanisms that include both positive and negative aspects of relationships between populations (Blois et al., 2014; Barros et al., 2017). The “Limiting Similarity Theory” (MacArthur, Levins, 1967) posits that coexisting species must differ sufficiently in morphology or behavior to reduce competition. These ecological trade-offs, often involving resource specialization, may promote stable coexistence (McPeek,1996). This notion aligns with the “Optimal Foraging Theory” (OFT) (MacArthur, Pianka, 1966), which suggests that individuals adopt foraging strategies that maximize energetic efficiency, thereby reducing interspecific niche overlap. However, the relative effect of these processes largely dependent on food resource availability in the environment (MacArthur, Pianka, 1966; MacArthur, Levins, 1967). In the Amazon, for example, small fish species adopt different foraging strategies to maximize energetic efficiency, using microhabitats that rage from less turbulent areas, such as roots and submerged litter, to the open water column (Silva et al., 2016; Benone et al., 2020; Manna, Rezende, 2021).
According to OFT, an increase in the quantity, heterogeneity, and availability of resources in streams during the rainy season may lead organisms to exhibit a broader niche breadth, as the greater variety of available items allows for a wider range of resource use (Borba et al., 2008). Conversely, under low resource availability during the dry season, niche breadth tends to decrease as organisms focus their foraging on the most energetically profitable items, reducing dietary diversity (da Silva et al., 2012). However, this pattern may shift depending on the balance between resource availability and consumer density (Costa, Nunes 2022). For instance, if the number of competitors increases faster than resource availability, broader niches may emerge even under high-resource conditions, making competition theory more predictive in such scenarios.
The lebiasinids, C. arnoldi and P. capim are nektonic species that inhabit backwater environments, where they display active and opportunistic foraging behavior (Brejão et al., 2013; Soares et al., 2020). Their mobility and ecological versatility allow them to exploit a wide range of microhabitats and food resources, making them valuable model organisms for investigating trophic dynamics, behavioral adaptations, and interspecific interactions within Amazonian aquatic ecosystems.
In this context, this study aims to evaluate the effects of hydrological variation and environmental factors on diet composition, trophic niche breadth, feeding intensity and trophic overlap of C. arnoldi and P. capim in streams of the upper Guamá River basin, eastern Amazon (Pará, Brazil). We anticipate that temporal variations in precipitation, and their associated effects on the physical and chemical structure of Amazonian streams, exert significant influence on the feeding patterns of these species. Specifically, we expect that during the rainy months, both species will exhibit narrower trophic niche and higher feeding intensities, resulting in lower trophic overlap. In contrast, during dry periods, we predict that both species will broaden their trophic niches and reduce feeding intensity, which would lead to greater trophic overlap between P. capim and C. arnoldi.
Material and methods
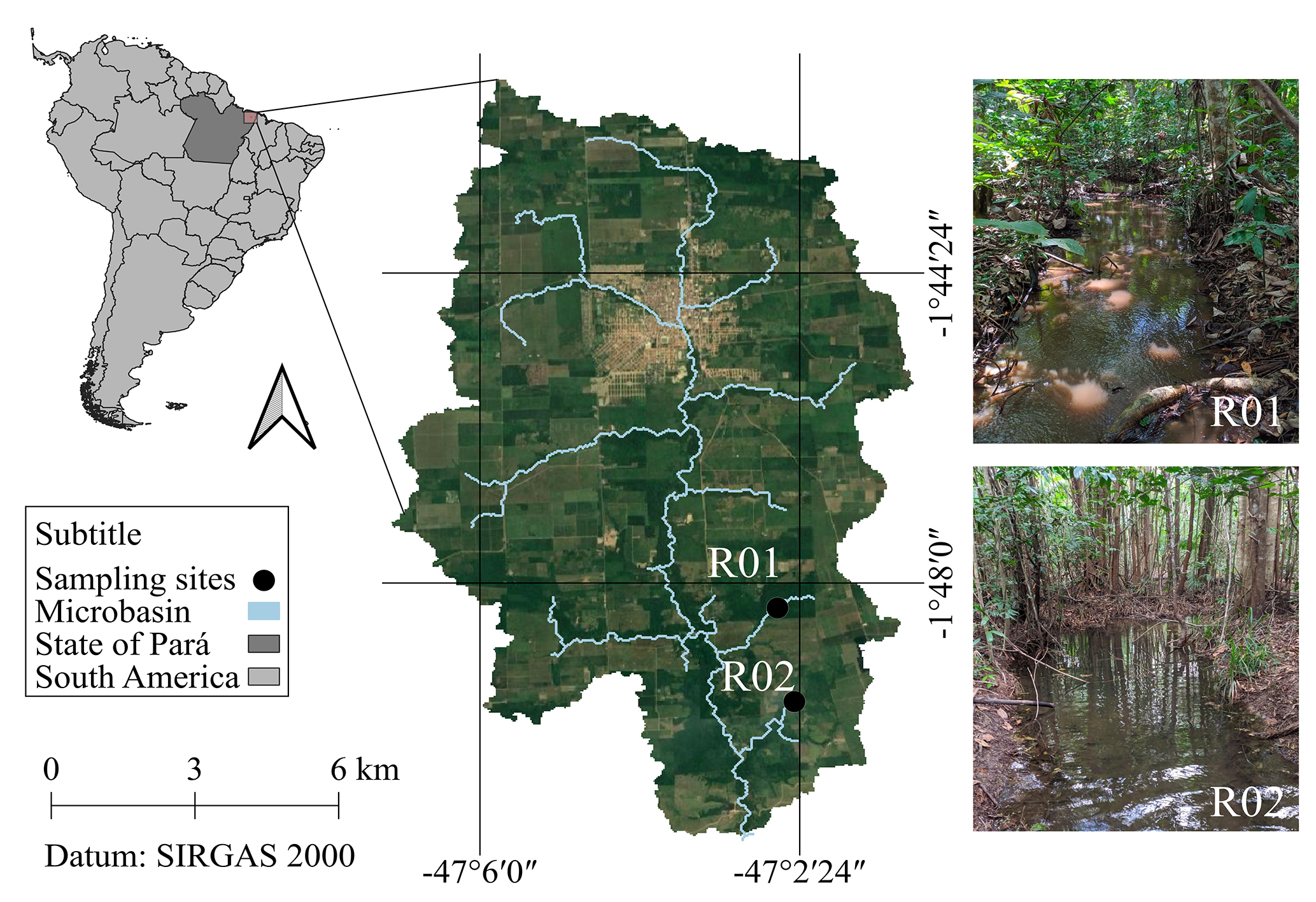
Sampling sites. The study was conducted in a microbasin located on the left bank of the upper Guamá River (01°44’47”S 47°03’57”W; Fig. 1), within the municipality of Capitão Poço, Pará, Brazil. The region’s climate is classified as Af, where A indicates a tropical climate e f indicates that rainfall occurs throughout most of the year, according to the Köppen system (adapted by Peel et al., 2007). The area experiences an average annual rainfall of 2,370 mm³ (INMET, 2020) and a mean temperature of 26 ºC (Schwartz, 2007). The driest period occurs between September and November, while the wettest months are between March and May (Pacheco, Bastos, 2001). The region’s topography is predominantly flat with gentle undulations and is primarily covered by Plateau Dense Latifoliate Forest (Silva et al., 1999). Although this description reflects the regional landscape, the surroundings of the streams currently show signs of anthropogenic disturbance, such as pasture, agriculture and livestock farming, with small patches of secondary forests and strips of Permanent Preservation Areas (APPs), as regulated by Article 30 of the Brazilian Forest Code (Law No 12651/12) (IDESP, 2011; Rodrigues et al., 2016; Silva et al., 2016).
FIGURE 1| Location of streams R01 and R02 (black circles) within the Capitão Poço River microbasin, Eastern Amazon, Pará, Brazil.
Environmental characterization of the streams and fish sampling were carried out monthly from March 2019 to February 2020 in two first-order streams, according to Strahler (1957) classification. These streams share similar physical habitat structure, with riparian vegetation forming a strip approximately 40 m wide, comprising large, medium and small trees, as described by Maciel et al. (2003). A 50 m sampling section was delimited in each stream and subdivided into six transverse segments (A to F) in a downstream-to-upstream direction, resulting in five longitudinal sections of 10 meters each. Monthly measurements included physical and chemical water parameters, habitat assessment, and fish sampling (López-Rodríguez et al., 2021; Oliveira et al., 2023).
The streams were evaluated for hydrogen potential (pH), electrical conductivity (μS/cm-1), temperature (ºC), and dissolved oxygen (%), measured using an AK88 multiparameter probe at three equidistant points along each 50 m sampling section. At each cross-section, we measured the wetted channel width (m) and thalweg depth (cm). In addition, we visually assessed substrate composition, considering categories such as sand, leaf banks, coarse gravel, fine gravel, submerged logs (wood), roots, and organic matter. However, only leaf banks and wood were selected for statistical analyses due to their ecological relevance to nektonic fish in backwater areas (Stegmann et al., 2019; Seabra et al., 2023).
The average current velocity (m/s) was determined by measuring the travel time of a floating object over a 1.5 m distance at three equidistant points along the stream section. Streamflow (m³/s) was calculated using the formula: Q = A * Vm, where Q is mean discharge, Vm is mean current velocity, and A is average cross-sectional area. Canopy cover was measured using photographs taken at three points (left margin, center and right margin) along each cross-section. The images were converted to grayscale, and the percentage of black pixels was determined using ImageJ software to estimate canopy closure. Finally, the average monthly accumulated precipitation (mm³) for the study area was obtained from the Instituto Nacional de Meteorologia (INMET- Station A248).
Specimens were collected using rectangular sieves (60 x 80 cm; 2 mm mesh), operated by three collectors. Sampling was conducted for 12 min per longitudinal section, totaling 60 min of sampling per month in each stream. Additionally, a trawl net measuring 3 m in length, 1.5 m in height, and 5 mm mesh was used, with two trawls passes per longitudinal section. Captured specimens were anesthetized by immersion in a eugenol solution (6 ml/3 L of water), fixed in 10% formaldehyde for 48 h, and subsequently preserved in 70% ethanol. Voucher specimens of Copella arnoldi were deposited in the Coleção de História Natural do Curso de Museologia (RTM) under vouchers number RTM.II.2.0277, while specimens of Pyrrhulina capim were deposited in the Ichthyological Collection of the Grupo de Ecologia Aquática (GEA) under catalog number GEA.ICT 12065, both housed at the Universidade Federal do Pará, Belém, Brazil.
Laboratory analysis. Specimens were measured for total mass (Mt; in grams) using an analytical balance with a precision of 0.0001 g, and for standard length (SI; in millimeters) using a digital caliper with a precision of 0.01 mm. To minimize ontogenetic effects, we selected only individuals with a SI > 9.00 mm for both species (Taguti et al., 2009; Cordeiro et al., 2023). Selected individuals were eviscerated through a longitudinal incision in the ventral region to remove the stomach, which was then weighed (Me; in grams) and preserved in Eppendorf tubes containing 70% alcohol for subsequent analysis.
Stomach contents were examined under a stereomicroscope (Olympus, Model CX21; with 52x magnification) using a Petri dish placed on graph paper with 1 mm² precision (Hellawell, Abel, 1971), following a modified version of Hyslop (1980). Stomach items were sorted and identified to the lowest possible taxonomic level using specialized literature (Costa, Simonka, 2006; Hamada, Ferreira-Keppler, 2012) and with expert assistance.
Data analysis. Food items were categorized by origin (autochthonous, allochthonous or undetermined). Items lacking sufficient information were excluded from the analysis. Properly categorized items were evaluated for relative Volume (Vo%), by measuring the proportion of graph paper squares occupied by each item relative to the total stomach content, using the equation: Vo% = (Vi/Vt) x 100, where Vi represents the number of squares occupied by item i, and Vt represents the total number of squares occupied by all food items. Items were also evaluated for Frequency of Occurrence (FOi%) (Hyslop, 1980), calculated as: FOi = (ni/N) x 100, where ni is the number of stomachs containing item i, and N is the total number of stomachs analyzed with at least one identifiable item. Both Vo% and FOi% were combined to calculate the Alimentary index (AI%) of each food item (Kawakami, Vazzoler, 1980), obtained from:
AI% = (FOi% x Vo%/ ∑FOi% x Vo%) x 100.
The AI% was calculated individually for each species, taking into account both food items and their origin. Calculations were performed for each sampling month as well as cumulatively for the entire study period.
Trophic niche breadth for each species was assessed using the standardized Levins Index (modified by Hurlbert, 1978), obtained from: Ba = [(∑jP2ij)]-1(n-1)-1, where Ba is the standardized trophic niche breadth, Pij is the proportion of food items j in species i’s diet, calculated from the Alimentary Index (AI%), and n is the total number of feeding categories. The index ranges from 0, when a species consumes a single food item (specialized diet), to 1, when a species consumes several food items similarly (generalized diet). For each species, Ba was calculated monthly and for the entire sampling period. To ensure data reliability, only stomachs containing at least one identifiable food item were included in analyses.
Feeding intensity was evaluated using the Stomach Repletion Index (SRI%), calculated as: SRI% = Me/Mt x 100, where Me is stomach mass and Mt is the individual’s total mass (Zavala-Camin, 1996). This index was calculated for each specimen, excluding empty stomachs, to assess the proportion of ingested food relative to body mass. Temporal feeding patterns were analyzed using monthly mean SRI% values.
Trophic niche overlap between the two species was calculated monthly and for the entire sampling period using Pianka’s Index (1973):
Ojk = ∑ni∑Pijx Pik / √ ∑ni∑Pijx Pik2,
where Ojk represents the niche overlap between species j and k, Pij and Pik are the proportions of food items i in the diets of species j and k, respectively. The index ranges from 0, when there is no trophic niche overlap, to 1, when there is total niche overlap between species. Overlap levels were interpreted as high (> 0.6), intermediate (0.4 to 0.6), or low (< 0.4) following Grossman (1986).
The effects of environmental variables on diet composition, trophic niche breadth, feeding intensity, and trophic niche overlap were evaluated using Generalized Linear Models (GLMs) with Gaussian distribution. For each species, models were constructed using the AI% values (diet composition) of the three most relevant trophic categories. Additionally, we modeled the relationship between environmental variables and the standardized Levins Index (niche breadth), the Stomach Repletion Index (SRI%; feeding intensity), and the Pianka Index (niche overlap). The Akaike Information Criterion (AIC) was used to select the best-fitting models, and only variables included in the best models were interpreted biologically.
To assess trophic overlap significance, we implemented a null model with 999 randomizations, using the ra3 randomization algorithm, which shuffles zero and nonzero values within rows to generate a simulated matrix. Comparing the simulated matrix and observed matrices provided statistical support for the observed overlap values. All analyzes were conducted in R software (R Development Core Team, 2020) using the packages EcoSimR (Gotelli et al., 2015), MuMin (Kamil, Barton, 2020), vegan (Oksanen et al., 2020) and car (Fox, Weisberg, 2019), with 999 permutations and a significance level of 5%.
Results
Over the 12-month sampling period, 239 specimens of C. arnoldi and 409 specimens of P. capim were analyzed. The standard length (SI) of C. arnoldi individuals ranged from 9.24 mm to 38.07 mm (21.32 ± 6.41 mm), while for P. capim, SI ranged from 10.02 mm to 53.07 mm (24.01 ± 10.02 mm).
For C. arnoldi, items were grouped into 12 categories: autochthonous (Coleoptera larvae, Copepoda, Diptera larvae, Hemiptera larvae, Hymenoptera larvae, Megaloptera larvae, Plecoptera larvae, and Thysanoptera larvae), allochthonous (Formicidae, Gastropoda (Hygrophila-Planorbidae), and plant material), and undetermined (exoskeleton fragments). For P. capim, 17 food items were identified: autochthonous (filamentous algae, Coleoptera larvae, Copepoda, Diptera larvae, Ephemeroptera larvae, Hemiptera larvae, Odonata larvae, Thysanoptera larvae and Trichoptera larvae), allochthonous (Arachnida, Formicidae, Gastropoda (Hygrophila-Planorbidae), Isoptera, Lepidoptera, Plant material, and Neuroptera), and undetermined (exoskeleton fragments).
The most important food item for C. arnoldi was Diptera larvae (26.1%), followed by plant material (23.7%) and Formicidae (19.5%). The remaining items collectively accounted for approximately 9.0% of the dietary importance (Tab. 1). For P. capim, the main food items were Diptera larvae (26.1%), Formicidae (21.3%), and Coleoptera larvae (17.8%), with the remaining categories contributing less than 1% to the overall dietary importance (Tab. 2).
TABLE 1 | Alimentary Index (AI%) for the feeding categories comprising the diet of Copella arnoldi. The table shows the analyzed months, AI% values for each feeding category, and the origin of the items (autochthonous, allochthonous, and undefined) highlighted in bold. *Indicates the most relevant feeding categories. N = number of individuals with stomachs containing identifiable items.
Category | 2019 | 2020 | |||||||||||
Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec | Jan | Feb | Total | |
(N = 20) | (N = 13) | (N = 27) | (N = 23) | (N = 24) | (N = 22) | (N = 20) | (N = 8) | (N = 19) | (N = 15) | (N = 15) | (N = 8) | (N = 214) | |
Autochthonous | 82.62 | 56.17 | 52.07 | 54.62 | 56.05 | 36.35 | 54.68 | 21.94 | 85.66 | 67.1 | 40.95 | 80.88 | 59.99 |
Copepoda | 1.4 | 1.04 | – | – | 1.32 | – | 0.4 | – | – | – | – | 3.64 | 0.18 |
Coleoptera (immature) | 24.91 | 4.19 | 12.33 | 0.34 | 5.22 | 2.37 | 16.31 | 5.81 | 19.83 | 2.94 | 7.42 | 18.22 | 9.18 |
Diptera (larvae) | 17.75 | 26.57 | 9.81 | 43.96 | 17.00 | 26.92 | 14.68 | 1.16 | 26.2 | 47.63 | 18.47 | 1.21 | 26.11* |
Hemiptera (immature) | 15.78 | 5.87 | 14.78 | 0.34 | 7.00 | – | 0.32 | 1.16 | 25.63 | 2.78 | – | 17.13 | 5.27 |
Hymenoptera (immature) | 1.09 | 2.09 | 0.27 | 0.32 | 2.52 | 1.09 | 1.04 | 5.42 | 0.11 | 3.74 | 1.73 | 8.26 | 1.73 |
Megaloptera (immature) | 1.09 | 0.11 | 0.05 | – | 0.08 | – | – | – | 0.05 | – | 0.14 | 7.29 | 0.14 |
Plecoptera (immature) | – | – | – | 0.69 | – | 0.52 | 1.54 | – | 0.93 | 5.81 | – | – | 0.39 |
Thysonoptera (immature) | 1.09 | – | 0.13 | 0.11 | – | – | – | – | 0.31 | – | 0.42 | 7.29 | 0.16 |
Allochthonous | 8.95 | 30.58 | 38.81 | 45.37 | 19.69 | 62.73 | 35.19 | 77.64 | 12.08 | 31.59 | 43.78 | 15.35 | 34.14 |
Gastropoda (Hygrophila-Planorbidae) | 0.54 | 0.41 | 0.13 | – | 0.46 | 0.21 | 0.09 | 0.32 | – | 0.28 | – | – | 0.16 |
Formicidae | 11.76 | 26.39 | 30.68 | 0.81 | 9.48 | 16.09 | 23.42 | 15.99 | 18.53 | 19.69 | 29.65 | 23.32 | 19.51* |
Plant material | 0.73 | 6.29 | 13.59 | 53.38 | 9.65 | 51.27 | 22.17 | 69.45 | 3.27 | 14.21 | 14.68 | 2.91 | 23.71* |
Undefined | 8.41 | 13.23 | 9.1 | – | 24.24 | 0.9 | 10.12 | 0.4 | 2.25 | 1.3 | 15.25 | 3.75 | 5.85 |
Insect fragments | 23.79 | 26.98 | 18.18 | – | 47.22 | 1.51 | 19.98 | 0.64 | 5.09 | 2.88 | 27.45 | 10.69 | 13.4 |
Average precipitation (mm³) | 370.0 | 445.6 | 301.0 | 234.2 | 137.0 | 101.4 | 34.0 | 75.0 | 47.2 | 68.0 | 369.2 | 511.6 | 2694.2 |
TABLE 2 | Alimentary Index (AI%) for the feeding categories comprising the diet of Pyrrhulina capim. The table shows the analyzed months, AI% values for each feeding category, and the origin of the items (autochthonous, allochthonous, and undefined) highlighted in bold. *Indicates the most relevant feeding categories. N = number of individuals with stomachs containing identifiable items.
Category | 2019 | 2020 | |||||||||||
Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec | Jan | Feb | Total | |
(N = 13) | (N = 23) | (N = 22) | (N = 25) | (N = 23) | (N = 36) | (N = 34) | (N = 35) | (N = 49) | (N = 27) | (N = 30) | (N = 18) | (N = 335) | |
Autochthonous | 73 | 66.30 | 55.09 | 76.08 | 23.02 | 66.29 | 62.12 | 75.99 | 59.3 | 65.15 | 54.14 | 63.46 | 62.77 |
Filamentous algae | 6.09 | – | 0.55 | – | – | 1.84 | 0.22 | – | 0.42 | 0.31 | 1.01 | 0.77 | 0.38 |
Copepoda | – | 0.03 | – | 0.03 | 0.11 | 0.23 | – | – | – | 0.02 | – | – | 0.02 |
Coleoptera (immature) | 19.16 | 18.88 | 17.67 | 6.96 | 11.39 | 14.35 | 22.12 | 12.9 | 15.94 | 19.75 | 25.42 | 24.15 | 17.75* |
Diptera (larvae) | 20.73 | 36.53 | 18.7 | 57.02 | 4.38 | 25.04 | 15.66 | 52.7 | 25.45 | 31.30 | 7.78 | 23.08 | 26.07* |
Ephemenoptera (immature) | – | 0.06 | – | – | 0.03 | – | – | 0.03 | 0.01 | 0.04 | – | 0.11 | 0.01 |
Hemiptera (immature) | 2.36 | 0.27 | 0.09 | 0.11 | – | 0.65 | 0.11 | 0.94 | 0.44 | 0.48 | 0.12 | 1.18 | 0.39 |
Odonata (immature) | 0.84 | – | – | 0.35 | – | – | – | – | – | – | – | – | 0.01 |
Thysanoptera (immature) | – | – | – | 0.05 | – | 0.03 | 0.51 | 0.56 | – | – | – | – | 0.03 |
Tricoptera (immature) | – | 0.39 | 0.09 | 1.81 | – | 0.37 | 1.13 | 0.39 | 0.20 | 0.30 | 0.24 | – | 0.33 |
Allochthonous | 5.43 | 18.80 | 26.76 | 15.24 | 45.35 | 8.93 | 13.90 | 17.22 | 16.65 | 15.03 | 14.79 | 10.24 | 16.64 |
Gastropoda (Hygrophila-Planorbidae) | – | – | – | 0.19 | – | 0.12 | – | 1.51 | – | – | – | – | 0.03 |
Arachnida | – | 0.01 | – | 0.01 | 0.59 | – | – | – | – | 0.01 | – | – | 0.01 |
Formicidae | 10.22 | 20.89 | 37.48 | 19.12 | 45.36 | 11.30 | 19.70 | 18.06 | 20.67 | 17.16 | 21.10 | 9.33 | 21.33* |
Isoptera | – | – | – | – | 0.09 | 0.04 | 0.29 | 0.11 | 0.05 | – | – | 0.15 | 0.03 |
Lepdoptera | – | 0.07 | – | 0.06 | – | – | – | – | 0.01 | 0.05 | – | – | 0.01 |
Neuroptera | – | 0.14 | – | – | – | – | – | 0.08 | 0.10 | 0.10 | – | 0.25 | 0.03 |
Plant material | – | 0.23 | – | 0.19 | – | 0.07 | – | – | – | 0.17 | – | – | 0.01 |
Undefined | 21.56 | 14.88 | 18.13 | 8.66 | 31.62 | 24.76 | 23.97 | 6.78 | 24.03 | 19.80 | 31.06 | 26.29 | 20.57 |
Insect fragments | 40.57 | 22.45 | 25.39 | 14.05 | 38.00 | 45.89 | 40.23 | 12.67 | 36.64 | 30.23 | 44.30 | 40.94 | 33.50 |
Average precipitation (mm³) | 370.0 | 445.6 | 301.0 | 234.2 | 137.0 | 101.4 | 34.0 | 75.0 | 47.2 | 68.0 | 369.2 | 511.6 | 2694.2 |
For C. arnoldi, Formicidae consumption was significantly influenced by pH (positive effect) and temperature (negative effect), which together explained 68.2% of the variation (R² = 0.682, p = 0.007; p = 0.001). The importance of plant material in the diet was positively associated with leaf litter availability, accounting for 41.5% of the variation (R² = 0.415; p = 0.010). In contrast, average current velocity was selected as the best predictor for Diptera consumption according to the AIC criterion, explaining 28.1% of the variation (R² = 0.281), although this relationship was not statistically significant (p = 0.076) (Tab. 3).
TABLE 3 | Model selection for diet composition of Copella arnoldi and Pyrrhulina capim based on Generalized Linear Models (GLMs). The response variables were the three categories with the highest AI% values for each species.
Species | Diet Composition | Environmental variables | β | SE of β | t | p |
C. arnoldi | Diptera larva (R² = 0.281; F(1, 10) = 3.92; AIC: 102.22) | Average Speed | -7.691 | 3.887 | -1.979 | 0.076 |
Formicidae (R² = 0.682; F(2, 90) = 9.68; AIC: 84.89) | Hydrogen Potential | 9.742 | 2.824 | 3.450 | 0.007 | |
Temperature | -12.406 | 2.824 | -4.394 | 0.001 | ||
Plant material (R² = 0.415; F(1, 10) = 1.28; AIC: 110.88) | Leaf Bank | 14.849 | 5.575 | 2.663 | 0.010 | |
P. capim | Coleoptera larva (R² = 0.520; F(1, 10) = 10.48; AIC: 73.62) | Temperature | -3.891 | 1.181 | -3.296 | 0.008 |
Diptera larva (R² = 0.352; F(2,9) = 2.080; AIC: 90.70) | Canopy | -7.149 | 1.537 | -4.652 | 0.046 | |
Leaf Bank | 13.492 | 2.924 | 4.614 | 0.049 | ||
Formicidae (R² = 0.224; F(1,10) = 2.890; AIC: 89.84) | Hydrogen Potential | 0.289 | 0.126 | 2.284 | 0.045 |
For P. capim, Coleoptera consumption showed a negative association with temperature, explaining 52.0% of the variation (R² = 0.520; p = 0.008). Diptera consumption was influenced by canopy cover (negative effect) and leaf litter availability (positive effect), which together explained 35.2% of the variation (R² = 0.352; p = 0.046; p = 0.049). Finally, Formicidae consumption exhibited a significant positive association with pH, explaining 22.4% of the variation (R² = 0.224; p = 0.045) (Tab. 3).
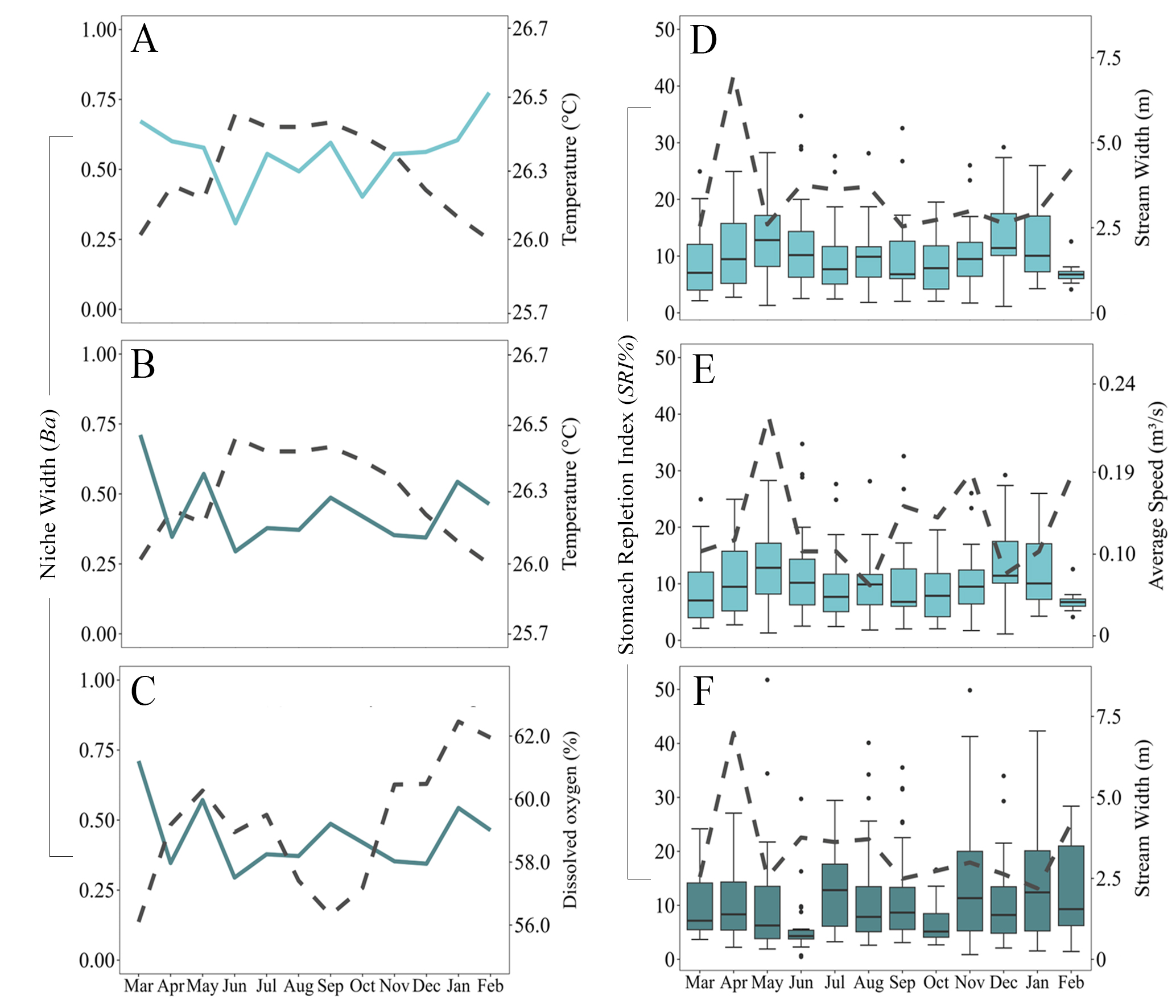
The widest trophic niche of C. arnoldi occurred in March (Ba = 0.67), January (Ba = 0.57), and February (Ba = 0.77), while the lowest values were observed in June (Ba = 0.30), August (Ba = 0.49), and October (Ba = 0.40). These fluctuations were associated with slight decreases in average water temperature, which accounted for 59.0% of the observed variation (R² = 0.593; p = 0.0485) (Fig. 2A; Tab. 4). Conversely, P. capim exhibited its highest trophic niche breadth in March (Ba = 0.71), May (Ba = 0.51), and January (Ba = 0.54), with the lowest values in April (Ba = 0.34), June (Ba = 0.29), and December (Ba = 0.34). Overall, the species’ trophic niche breadth showed high sensitivity to environmental changes, with 64.0% of the variation explained (R² = 0.642; p = 0.019; p = 0.024). This variation was mainly associated with lower water temperature (Fig. 2B) and reduced dissolved oxygen levels (Fig. 2C; Tab. 4). The monthly mean values of Ba, SRI% and Pianka for both species are provided in Tab. S1.
FIGURE 2| Monthly mean values of trophic Niche Breadth (Ba) and Stomach Repletion Index (SRI%) for Copella arnoldi (light blue) and Pyrrhulina capim (dark blue). The black dashed line indicates the environmental parameter and its monthly variation from March 2019 to February 2020 in the Capitão Poço River microbasin, Eastern Amazon, Pará, Brazil.
TABLE 4 | Selection of GLM models evaluating the relationship between the Standardized Levins Index (Ba), Stomach Repletion Index (SRI%), and trophic niche overlap (Pianka’s Index) for Copella arnoldi and Pyrrhulina capim.
Species | Index | Environmental variables | β | SE of β | t | p |
C. arnoldi | Levins Index (R² = 0.593; F(1,22) = 4.09; AIC: 19.806) | Temperature | -0.091 | 0.024 | -3.822 | 0.048 |
Stomach Repletion Index (R² = 0.626; F(3,20) = 3.39; AIC: 39.150) | Average Speed | -1.71 | 0.566 | -3.021 | 0.027 | |
Stream Width | -3.868 | 1.334 | -2.899 | 0.032 | ||
Canopy | 2.285 | 1.231 | 1.855 | 0.056 | ||
P. capim | Levins Index (R² = 0.642; F(2,21) = 1.74; AIC: 16.421) | Dissolved oxygen | -0.066 | 0.0267 | -2.506 | 0.019 |
Temperature | -0.104 | 0.026 | -3.929 | 0.024 | ||
Stomach Repletion Index (R² = 0.299; F(1,22) = 2.38; AIC: 38.716) | Stream Width | -0.784 | 0.379 | -2.069 | 0.049 | |
Trophic overlap | Pianka Index (R² = 0.263; F(1,22) = 3.50; AIC: 7.701) | Average Speed | -0.119 | 0.063 | -1.891 | 0.069 |
Among the C. arnoldi stomachs, ten (4.2%) were empty, and 15 (7.3%) contained only digested material, which were included solely in the feeding intensity evaluation. The highest mean SRI% values occurred in May (13.27%), June (10.78%), and December (12.41%), while the lowest values occurred in March (9.02%), September (7.12%), and February (6.06%). This variation was explained by environmental factors, which accounted for 62.0% of the observed variation, considering the combined influence of average channel velocity, stream width, and canopy cover (R² = 0.626; p = 0.027; p = 0.032) (Figs. 2D, E). Both channel velocity and stream width had positive and significant effects, whereas canopy cover had a negative but non-significant effect (p = 0.056) (Tab. 4).
For P. capim, 36 stomachs (11.2%) were empty and 38 (11.6%) contained only digested material, also used exclusively for the feeding intensity analysis. The highest average feeding intensity values were recorded in July (12.76%), November (11.25%), and January (12.26%), while lower SRI% values were recorded in May (8.30%), June (5.43%), and October (6.29%) (Fig. 3B). In contrast to C. arnoldi, P. capim showed lower sensitivity to environmental variation, with feeding intensity being negatively influenced only by stream width, which explained 29.0% of the variation (R² = 0.299; p = 0.049) (Fig. 2F; Tab. 4).
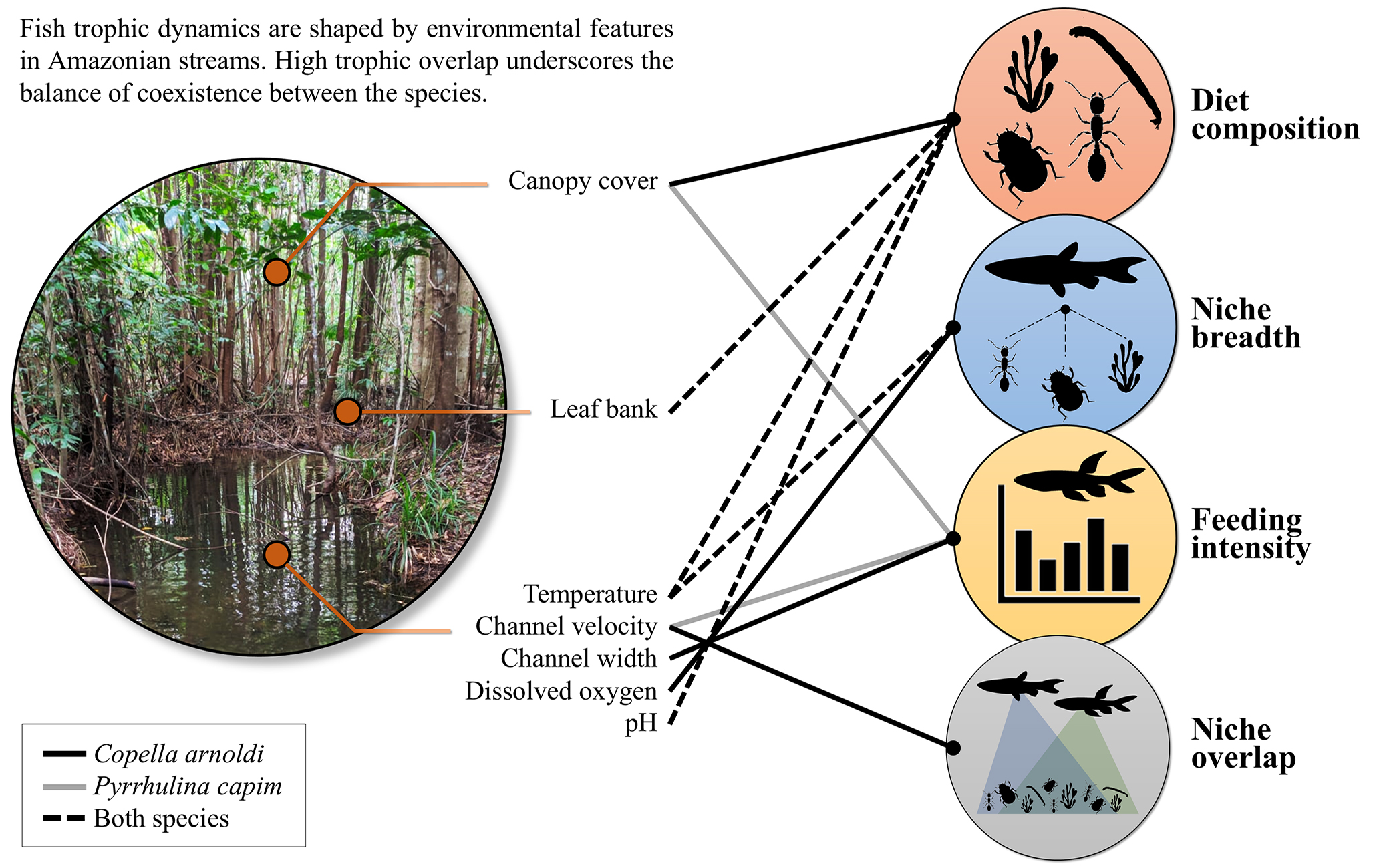
FIGURE 3| Infographic illustrating the relationship between environmental variables and trophic ecology of Copella arnoldi and Pyrrhulina capim in the Capitão Poço River microbasin, Eastern Amazon, Pará, Brazil.
The trophic niche overlap, calculated using Pianka’s Index, revealed a high degree of dietary overlap (Ojk = 0.79), significantly greater than expected by chance (simulated mean = 0.18; p = 0.042). The highest overlap occurred in October (Ojk = 1.00), while the lowest was observed in August (Ojk = 0.47). Both values were significantly higher than expected by chance (simulated means = 0.83; 0.38; p = 0.044).
Finally, based on the AIC selection criterion, average channel velocity was the only variable identified as a predictor of trophic niche overlap between C. arnoldi and P. capim, explaining 26.0% of the observed variation (R² = 0.263), although this association was not statistically significance (Tab. 4). A diagram summarizing the relationship between environmental variables and the trophic dynamics of both species is presented in Fig. 3.
Discussion
Both species, C. arnoldi and P. capim,exhibited omnivorous feeding habits with a predominance of animal-based items, indicating tendency toward insectivory, primarily consuming autochthonous resources. For C. arnoldi, the most important food items were Diptera larvae, Formicidae, and plant material, while P. capim consumed Coleoptera, Diptera larvae, and Formicidae. Although rainfall was included in the models, it was not selected as a significant predictor for any trophic metric, indicating that other environmental variables better explain the observed patterns for both species, namely: water current velocity, pH, temperature, the presence of leaf litter banks, and canopy cover. While C. arnoldi exhibited a more generalized diet only in the rainiest months, P. capim maintained a consistently generalist diet throughout the study period. Both species exhibited monthly variations in feeding intensity, but without a clear pattern associated with rainfall or hydrological periods. Although rainfall data were presented for contextual purposes, no direct relationship with feeding patterns was observed. Furthermore, trophic niche overlap between the species remained consistently high throughout the study, except during June and August, when it was intermediate.
The consumption of Diptera larvae, Formicidae, and plant material by C. arnoldi reflects the importance of these resources as food sources. Diptera larvae, abundant in aquatic environments, provide essential proteins, while Formicidae, though terrestrial, often fall into streams and serve as prey for aquatic fauna (Correa et al., 2007; Angulo et al., 2016; Nogales-Mérida et al., 2019; Limberger et al., 2021). Environmental variables such as channel velocity, pH, temperature, and the presence of leaf litter influenced the consumption of these items by C. arnoldi. Variation in these factors affect water quality and, consequently, the availability of food resources. For example, leaf litter in streambeds functions both as shelter and as feeding sites for Diptera larvae and Formicidae (Gibb, Hochuli, 2003; Angulo et al., 2016). Therefore, reliance on these resources not only reflects their importance as dietary components but also underscores their interconnectedness with environmental factors that shape stream ecological dynamics (Vannote et al., 1980).
The main food items of P. capim (Diptera larvae, Coleoptera, and Formicidae) are widely recognized components of aquatic food webs (Arslan et al., 2010; Angulo et al., 2016; Sousa et al., 2017). The presence of allochthonous items in the species’ diet, such as beetles and ants, suggests that these insects are likely captured when they fall into the water (Restrepo-Santamaria et al., 2022). However, the prevalence of Formicidae in the diet may also reflect the degree of urbanization near the studied streams, as many ant species are associated with altered riparian vegetation (Rocha et al., 2015). This underscores the need for further investigations into the effects of anthropogenic activities on the feeding ecology of Amazonian ichthyofauna, particularly regarding the role of adjacent vegetation, despite existing studies in other biomes (Zina et al., 2021; Reyes-Morales et al., 2023).
Environmental variables such as temperature, canopy cover, leaf litter presence, and pH significantly influenced the consumption of P. capim main food items, as indicated by the item-specific statistical analyses. The combined effects of canopy cover and leaf litter directly shape the microclimatic conditions of streams, altering light incidence, humidity, and water temperature, which in turn regulate the accumulation and decomposition of organic matter (Mendes et al., 2021). Leaf litter banks act as both food sources and microhabitats for beetles and Diptera larvae, supporting detritivore invertebrate communities (Zeus et al., 2024). Fluctuations in these environmental features may increase the exposure and availability of such invertebrates, facilitating their capture by fish (Fracalossi, Cyrino, 2012; Sousa et al., 2017). This dynamic likely explains the high importance of these food sources in the diet of P. capim.
Copella arnoldi exhibited a higher trophic breadth in February (rainfall peak) linked to the increased consumption of various invertebrates such as Diptera larvae. However, no consistent pattern was observed throughout the rainy months. This result aligns with our initial hypothesis, which predicted a more generalist feeding habit during months of higher rainfall, when increased resource availability theoretically allows more energetically advantageous choices (MacArthur, Pianka, 1966). Despite sharing a similar diet, P. capim consumed an even wider variety of items, including Diptera larvae, Formicidae, and Coleoptera larvae as the most frequent, demonstrating a more generalist feeding strategy unaffected by seasonality. This pattern may be linked to increases in invertebrate abundance, such as Diptera larvae, which tend to reach population peaks during wetter months (Inoue et al., 2012). Environmental factors like dissolved oxygen and water temperature, also affect feeding behavior, potentially causing P. capim to reduce activity or select less energetically costly prey under low-oxygen conditions, thereby limiting dietary diversity (Mendes et al., 2021). Therefore, both species expand their diets during the rainy season, but prey composition and environmental constraints shape their foraging strategies (MacArthur, Pianka, 1966).
The higher feeding intensity observed in C. arnoldi during wetter months could be related to its morphological traits. Fish with slender, elongated bodies often exhibit greater swimming acceleration capacity, which facilitates exploration of diverse microhabitats and access to a broader prey spectrum (Buckup, 1999; Ponton, Mérigoux, 2000; Prado et al., 2016). Conversely, P. capim displayed greater feeding intensity during the drier months, an unexpected result. This pattern may relate to regional rainfall characteristics: reduced rainfall during the dry season decreases stream turbidity, improving prey visibility and detection (Monteiro, 2022). However, this inverse relationship in feeding activity between the two species warrants further investigation, as factors such as reproductive cycles, population fluctuations, and interspecific competition could also play considerable roles (Sánchez-Hernández et al., 2019).
The trophic niche overlap between C. arnoldi and P. capim was high and significantly different from what would be expected by chance. However, despite this high overlap, the current availability of food resources may minimize potential competitive impacts. Moreover, annual variability in precipitation alters stream conditions, providing distinct resources such as shelter, microhabitats, and, most notably, food. This allows species to exploit different resources or microhabitats at different times, thereby reducing direct competition and facilitating coexistence (Espírito-Santo et al., 2009; Zeni, Casatti, 2014). These dynamics conditions favor species with high trophic plasticity, enhancing coexistence (Silva et al., 2016). Previous studies have also observed variations in trophic overlap among fishes in response to hydrological changes across seasonal periods (Mazzoni et al., 2012; Silva et al., 2016, 2019; Neves et al., 2021).
The high dietary overlap observed in certain months may indicate either intensified competition due to limited resources or reflect the use of abundant shared food items that do not necessarily generate competition (Zeni, Casatti, 2014). In our study, total dietary overlap in October likely resulted from habitat changes or reduced food availability during the Amazon’s dry season, leading both species to rely on similar resources. However, without an explicit analysis of the correlation between niche overlap and niche breadth, it remains unclear whether this overlap stems from a contraction of niche breadth in both species or from other ecological factors. Neves et al. (2021) observed a similar pattern in the Atlantic Forest, suggesting that such seasonal dynamics may be common across tropical ecosystems facing comparable environmental pressures. Conversely, the intermediate overlap observed in August suggests dietary diversification as resource variety increases during the transitional period between seasons. These seasonal fluctuations are crucial for species coexistence, allowing flexible feeding strategies that optimize resource use (Zeni, Casatti, 2014) and promote survival within the complex Amazonian ecosystem.
These mechanisms reduce resource-use overlap and facilitate coexistence even in highly dynamic ecosystems (Neves et al., 2021). Such adaptations often involve diversified behavioral traits that mitigate competition, such as foraging in distinct microhabitats or during different periods of heightened feeding activity (Silva et al., 2016). The results compiled in this study help address part of the Eltonian shortfalls (Hortal et al., 2015), contributing to a better understanding of species interactions such as predation, competition, and resource sharing.
The study of species’ trophic characteristics represents a valuable tool for assessing stream environmental conditions, especially in the context of ongoing human activities. Logging, urbanization, and mining negatively impact the trophic ecology of local species, potentially driving habitat homogenization and biodiversity loss (Barros at al., 2024). Even in areas legally designated for permanent preservation, streams adjacent to anthropogenic activities (e.g., agriculture, pasture, and livestock farming) may experience degradation of riparian vegetation (Maracahipes-Santos et al., 2020). These environmental alterations directly influence the diet and feeding strategies of fish species inhabiting such systems. At this stage, our findings clarified part of these complex relationships, highlighting that complimentary results can be obtained through approaches such as stable isotope analysis, as it provides evidence of resource assimilation by individuals and reveals trophic pathways that traditional methods may overlook (Anderson, Cabana, 2007).
The findings presented here are fundamental for developing effective conservation and sustainable management strategies for stream fish populations. Copella arnoldi and Pyrrhulina capim exhibited omnivorous feeding habits with a tendency toward insectivory, demonstrating a strong reliance on resources derived from adjacent vegetation. Their trophic ecology, including diet composition, niche breadth, and feeding intensity, was shaped by local environmental variations. Although a high trophic overlap was observed between the two species, the environmental heterogeneity of the studied streams, such as heterogeneous leaf litter distribution, variable canopy cover, and seasonal changes in water flow and prey availability, appear to facilitate their coexistence by allowing dynamic adjustments in foraging behavior, habitat use, and resource access. Such variability in the physicochemical structure of streams directly influences interspecific interactions, particularly competition and resource partitioning. Thus, lebiasinids fishes appear to perform remarkably similar ecological roles but coexist through niche partitioning and other behavioral or morphological adaptations, including temporal variation in feeding intensity, exploitation of distinct microhabitats, and context-dependent shifts in dietary composition.
Acknowledgments
We extend our gratitude to the Universidade Federal do Pará (UFPA) and the Programa de Pós-Graduação em Zoologia (PPGZOOL) for the opportunity to conduct this research. Additionally, we are grateful to the Universidade Federal Rural da Amazônia (UFRA/Capitão Poço) and the Laboratório de Ecologia e Conservação da Amazônia (LABECA) for their authorization and support in providing essential facilities and equipment for this project.
References
Abe H, Dias J, Cordeiro C, Ramos F, Fujimoto R. Pyrrhulina brevis (Steindachner, 1876) como uma nova opção para a piscicultura ornamental nacional: Larviculture. Bol Inst Pesca. 2015; 41(1):113–22.
Anderson C, Cabana G. Estimating the trophic position of aquatic consumers in river food webs using stable nitrogen isotopes. J North Am Benthol Soc. 2007; 26(2):273–85. https://doi.org/10.1016/B978-0-323-99762-1.00044-9
Angulo E, Boulay R, Ruano F, Tinaut A, Cerdá X. Anthropogenic impacts in protected areas: assessing the efficiency of conservation efforts using Mediterranean ant communities. Peer J. 2016; 4:e2773. https://doi.org/10.7717/peerj.2773
Arslan N, Ayik Ö, Şahin Y. Diversity and structure of Chironomidae (Diptera) limnofauna of Lake Uluabat, a Ramsar Site of Turkey, and their relation to environmental variables. Turk J Fish Aquat Sci. 2010; 10(3):315–22. https://doi.org/10.4194/trjfas.2010.0303
Baldasso MC, Wolff LL, Neves MP, Delariva RL. Ecomorphological variations and food supply drive trophic relationships in the fish fauna of a pristine neotropical stream. Environ Biol Fishes. 2019; 102:783–800. https://doi.org/10.1007/s10641-019-00871-w
Barros BSF, Doria CRC, Rodrigues CG, Filho JAL. A ictiofauna de igarapés da microbacia do Belmont, um sistema hidrográfico degradado pela expansão urbana na Amazônia Sul Ocidental. Rev Bra Ciên Amaz. 2020; 9(3):120–43. https://doi:org/10.47209/2317-5729
Barros G, Zuanon J, Deus C. Effects of species co-occurrence on the trophic-niche breadth of characids in Amazon Forest streams. J Fish Biol. 2017; 90(1):326–40. https://doi.org/10.1111/jfb.13183
Barros HKS, Barbosa TAP, Prudente BS. Feeding ecology of Moenkhausia collettii (Steindachner, 1882) (Characiformes: Characidae) in streams in the eastern Amazon: Environmental factors and body size. J Fish Biol. 2024; 104(6):2008–21. https://doi.org/10.1111/jfb.15743
Benone NL, Lobato CMC, Soares BE, Montag LFA. Spatial and temporal variation of the diet of the flag tetra Hyphessobrycon heterorhabdus (Characiformes: Characidae) in streams of the Eastern Amazon. Neotrop Ichthyol. 2020; 18(4):e200078. https://doi.org/10.1590/1982-0224-2020-0078
Benone NL. Efeitos do espaço e do ambiente sobre assembleias de peixes de igarapés da Amazônia oriental. [Master’s dissertation]. Belém: Universidade Federal do Pará; 2012. Available from: http://www.repositorio.ufpa.br/jspui/handle/2011/3474
Blois JL, Gotelli NJ, Behrensmeyer AK, Tyler FJ, Lyons Sk, Williams JW et al. A framework for evaluating the influence of climate, dispersal limitation, and biotic interactions using fossil pollen associations across the late Quaternary. Ecography. 2014; 37(11):1095–08. https://doi.org/10.1111/ecog.00779
Borba CS, Fugi R, Agostinho AA, Novakowski GC. Dieta de Astyanax asuncionensis (Characiformes, Characidae), em riachos da bacia do rio Cuiabá, Estado do Mato Grosso. Acta Scient Biol Sci. 2008; 30(1):39–45. https://doi.org/10.4025/actascibiolsci.v30i1.1442
Brejão GL, Gerhard P, Zuanon J. Functional trophic composition of the ichthyofauna of forest streams in eastern Brazilian Amazon. Neotrop Ichthyol. 2013; 11(2):361–73. https://doi.org/10.1590/S1679-62252013005000006
Buckup PA. Sistemática e biogeografia de peixes de riachos. In: Caramaschi EP, Mazzoni R, Peres-Neto PR, editors. Ecologia de peixes de riachos. Oecol Bras. 1999; 6(1):91–138. https://doi.org/10.4257/oeco.1999.0601.03
Buckup PA. Taxonomia e filogenia de peixes de riachos brasileiros. Oeco Aust. 2021; 25(2):127–230. https://doi.org/10.4257/oeco.2021.2502.01
de Carvalho DR, Castro DMP, Callisto M, Moreira MZ, Pompeu PS. The trophic structure of fish communities from streams in the Brazilian Cerrado under different land uses: an approach using stable isotopes. Hydrobiologia. 2017; 795:199–217. https://doi.org/10.1007/s10750-017-3130-6
Cordeiro JG, Rodrigues J, Santos R, Rodrigues MS, Nóbrega RH, Siqueira FFS et al. Reproductive biology of the Amazonian amphibian fish the splash tetra Copella arnoldi with emphasis to histological characterization. Acta Zool. 2023; 104(3):444–57. https://doi.org/10.1111/azo.12430
Correa SB, Winemiller KO, Lopez-Fernandez H, Galetti M. Evolutionary perspectives on seed consumption and dispersal by fishes. BioScience. 2007; 57(9):748–56. https://doi.org/doi:10.1641/B570907
Costa CIS, Simonka CE, editors. Insetos imaturos: metamorfose e identificação. Ribeirão Preto: Editora Holos; 2006.
Costa ID, Nunes NNS. Niche partitioning of two piscivorous fish species in a river in the western Brazilian Amazon. Iheringia Sér Zool. 2022; 112:e2022002. https://doi.org/10.1590/1678-4766e2022002
Dagosta FC, de Pinna M. The fishes of the Amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Amer Mus Nat Hist. 2019; 431(1):1–163. https://doi.org/10.1206/0003-0090.431.1.1
Dala-Corte RB, Melo AS, Siqueira T, Bini LM, Martins RT, Cunico AM et al. Thresholds of freshwater biodiversity in response to riparian vegetation loss in the Neotropical region. J Appl Ecol. 2020; 57(7):1391–402. https://doi.org/10.1111/1365-2664.13657
Dias RM, Tófoli RM, Silva JCB, Gomes LC, Agostinho AA. Effects of habitat complexity on trophic interactions of three congeneric fish species. Aquat Ecol. 2022; 56:877–89. https://doi.org/10.1007/s10452-022-09954-w
Espírito-Santo HMV, Magnusson WE, Zuanon J, Mendonça FP, Landeiro VL. Seasonal variation in the composition of fish assemblages in small Amazonian Forest streams: evidence for predictable changes. Freshw Biol. 2009; 54(3):536–48. https://doi.org/10.1111/j.1365-2427.2008.02129.x
Esteves KE, Aranha JMR, Albrecht MP. Ecologia trófica de peixes de riacho: uma releitura 20 anos depois. Oeco Aust. 2021; 25(2):266–82. https://doi.org/10.4257/oeco.2021.2502.04
Fox J, Weisberg S. An R companion to applied regression. Third Edition. Thousand Oaks: Sage; 2019.
Fracalossi DM, Cyrino JEP. Nutriaqua: nutrição e alimentação de espécies de interesse para a aquicultura brasileira. Florianópolis: AQUABIO; 2012.
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Natural Sciences; 2024. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Gibb H, Hochuli DF. Colonization by a dominant ant facilitated by anthropogenic disturbance: effects on ant assemblage composition, biomass and resource use. Oikos. 2003; 103(3):469–78. https://doi.org/10.1034/j.1600-0706.2003.12652.x
Gotelli NJ. EcoSimR: null model analysis for ecological data. R Package version 0.1.0. 2015. Available from: https://github.com/gotellilab/EcoSimR
Graziano MP, Deguire AK, Surasinghe TD. Riparian buffers as a critical landscape feature: Insights for riverscape conservation and policy renovations. Diversity. 2022; 14(3):172. https://doi.org/10.3390/d14030172
Grossman GD. Food resources partitioning in a rocky intertidal fish assemblage. J Zool. 1986; 1(2):317–55. https://doi.org/10.1111/j.1096-3642.1986.tb00642.x
Guglielmetti R, Silva MR, Higuti J, Fugi R. Diet of benthivorous fish and prey availability in streams of the Pirapó River basin-PR. Acta Limn Bras. 2019; 31:e7. https://doi.org/10.1590/S2179-975X4518
Hamada N, Ferreira-Keppler RL, editors. Guia ilustrado de insetos aquáticos e semiaquáticos da Reserva Florestal Ducke. Manaus: EDUA; 2012.
Hellawell J, Abel R. A rapid volumetric method for the analysis of the food of fishes. J Fish Biol. 1971; 3(1):29–37. https://doi.org/10.1111/j.1095-8649.1971.tb05903.x
Hortal J, Bello F, Diniz-Filho JAF, Thomas ML, Lobo JM, Ladle RJ. Seven shortfalls that beset large-scale knowledge of biodiversity. Annu Rev Ecol Evol Syst. 2015; 46:523–49. https://doi.org/10.1146/annurev-ecolsys-112414-054400
Hurlbert SH. The measurement of niche overlap and some relatives. Ecology. 1978; 59(1):67–77. https://doi.org/10.2307/1936632
Hyslop EJ. Stomach contents analysis – a review of methods and their application. J Fish Biol. 1980; 17(4):411–29. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Inoue M, Shinotou S, Maruo Y, Miyake Y. Input, retention, and invertebrate colonization of allochthonous litter in streams bordered by deciduous broadleaved forest, a conifer plantation, and a clear-cut site in southwestern Japan. Limnology. 2012; 13:207–19. https://doi.org/10.1007/s10201-011-0369-x
Instituto de Desenvolvimento Econômico, Social e Ambiental Do Pará(IDESP). Capitão Poço, Belém: Governo do Estado do Pará; 2011.
Instituto Nacional de Meteorologia (INMET). Mapas meteorológicos [Internet]. Brasília; 2019. Available from: https://mapas.inmet.gov.br/
Kamil B. MuMIn: Multi-Model Inference. R package version 1.43.17. 2020; https://CRAN.R-project.org/package=MuMIn
Kawakami E, Vazzoler G. Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Bol Inst Oceanogr. 1980; 29(2):205–07. https://doi.org/10.1590/S0373-55241980000200043
Limberger PM, Tataje DAR, Novicki D, Júnior LCM, Strieder MN. Spatio-temporal distribution of preimaginal black flies (Diptera: Simuliidae) in streams of a sub-basin of the Uruguay river basin, southern Brazil. Entomobrasilis. 2021; 14:e946. https://doi.org/10.12741/ebrasilis.v14.e946
López-Rodríguez NC, Leão AHF, Rocha RM, Prudente BS, Montag LFA. Environmental influence on the reproductive strategy of Helogenes marmoratus (Siluriformes: Cetopsidae) in the Amazonian streams. Neotrop Ichthyol. 2021; 19(4):e210092. https://doi.org/10.1590/1982-0224-2021-0092
MacArthur RH, Levins R. The limiting similarity, convergence and divergence of coexisting species. Am Nat. 1967; 101(921):377–85. https://doi.org/10.1086/282505
MacArthur RH, Pianka ER. On the optimal use of a patchy habitat. Am Nat. 1966; 100(916):603–09. https://doi.org/10.1086/28245
Maciel MNM, Watzlawick LF, Schoeninger ER, Yamaji FM. Classificação ecológica das espécies arbóreas. Rev Acad Ciênc Anim. 2003; 1(2):69–78. https://doi.org/10.7213/cienciaanimal.v1i2.14922
Manna L, Rezende C. Variação intraespecífica em peixes de riacho com ênfase na ecologia trófica. Oeco Aust. 2021; 25(2):301–22. https://doi.org/10.4257/oeco.2021.2502.06
Maracahipes-Santos L, Silvério DS, Macedo MN. Agricultural land-use change alters the structure and diversity of Amazon riparian forests. Biol Con. 2020; 252:e108862. https://doi.org/10.1016/j.biocon.2020.108862
McPeek MA. Trade-offs, food web structure, and the coexistence of habitat specialists and generalists. Am Nat. 1996; 148:124–38. https://doi.org/10.1086/285906
Mazzoni R, Marques PS, Rezende CF, Iglesias-Rios R. Niche enlargement as a consequence of co-existence: a case study. Braz J Biol. 2012; 72(2):267–74. https://doi.org/10.1590/S1519-69842012000200006
Mendes TP, Esposito MC, Filho FC, Juen L, Alvarado ST, Sousa JRP. Necrophagous flies (Diptera: Calliphoridae and Sarcophagidae) as indicators of the conservation or anthropization of environments in eastern Amazonia, Brazil. J Insect Conserv. 2021; 25(4):719–32. https://doi.org/10.1007/s10841-021-00338-3
Montag LFA, Freitas TMS, Wosiacki WB Barthem RB. Os peixes da Floresta Nacional de Caxiuanã (municípios de Melgaço e Portel, Pará, Brasil). 2008; 3(1):11–34. Available from: https://repositorio.museu-goeldi.br/handle/mgoeldi/1466
Monteiro LC. Estrutura populacional de espécies do gênero Cichla (Osteichthyes: Cichliformes) introduzidas em diferentes regiões brasileiras – uma revisão bibliográfica. Biodivers Bras. 2022; 12(2):1–11. https://doi.org/10.37002/biodiversidadebrasileira.v12i2.2192
de Moraes RLR, Sember A, Bertollo LAC, Oliveira EA, Rab P, Hatanaka T et al. Comparative cytogenetics and Neo-Y formation in small-sized fish species of the genus Pyrrhulina (Characiformes, Lebiasinidae). Front Genet. 2019; 10:678. https://doi.org/10.3389/fgene.2019.00678
Neves MP, Kratina P, Delariva RL, Jones J, Fialho CB. Seasonal feeding plasticity can facilitate coexistence of dominant omnivores in Neotropical streams. Rev Fish Biol Fish. 2021; 31(2):417–32. https://doi.org/10.1007/s11160-021-09648-w
Nogales-Mérida S, Gobbi P, Józefiak D, Mazurkiewicz J, Dudek K, Rawski M et al. Insect meals in fish nutrition. Rev Aquac. 2019; 11(4):1080–103. https://doi.org/10.1111/raq.12281
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, et al. Vegan: community ecology package. R package version 2. 5-7. 2020. https://CRAN.Rproject.org/package=vegan
Oliveira AEP, Montag LFA, Rocha RM, López-Rodríguez NC, Prudente BS. Environmental predictors of the life history of the flag tetra Hyphessobrycon heterorhabdus (Characiformes: Characidae) in streams of the Eastern Amazon. Neotrop Ichthyol. 2023; 21(4):e220094. https://doi.org/10.1590/1982-0224-2022-0094
Pacheco NA, Bastos TX. Caracterização climática do Município de Capitão Poço-PA. Documentos, 79. Belém: Embrapa Amazônia Oriental, 2001. Available from: https://www.infoteca.cnptia.embrapa.br/bitstream/doc/403420/1/OrientalDoc79.pdf
Peel MC, Finlayson BL, Mcmahon TA. Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci. 2007; 4(2):439–73. https://doi.org/10.5194/hess-11-1633-2007
Pianka ER. The structure of lizard communities. Annu Rev Ecol Syst. 1973; 4:53–74. http://www.jstor.org/stable/2096804
Pini SFR, Abelha MCF, Kashiwaqui EAL, Delariva RL, Makrakis S, Makrakis MC. Food resource partitioning among species of Astyanax (Characiformes: Characidae) in the Lower Iguaçu River and tributaries, Brazil. Neotrop Ichthyol. 2019; 17(4):e190028. https://doi.org/10.1590/1982-0224-20190028
Ponton D, Mérigoux S. Comparative morphology and diet of young cichlids in the dammed Sinnamary River, French Guiana, South America. J Fish Biol. 2000; 56(1):87–102. https://doi.org/10.1111/j.1095-8649.2000.tb02088.x
Prado A V, Goulart E, Pagotto J. Ecomorphology and use of food resources: inter-and intraspecific relationships of fish fauna associated with macrophyte stands. Neotrop Ichthyol. 2016; 14(4):e150140. https://doi.org/10.1590/1982-0224-20150140
R Development Core Team. R: a language and environment for statistical computing. Vienna, Austria; 2020. Available from: http://www.r-project.org/
Regan CT. The classification of the Teleostean fishes of the order Pediculati. Ann Mag Nat Hist. 1912; 9(51):277–89. https://doi.org/10.1080/00222931208693132
Restrepo-Santamaria D, Navia AF, Palacios J, Jiménez-Segura LF. Feeding strategy of fish that colonize reservoirs in the Magdalena River basin. Univ Sci. 2022; 27(3):234–52. https://doi.org/10.11144/Javeriana.SC273.fsof
Reyes-Morales PS, Lara C, Castillo-Guevara C, Cuautle M. Does the ant functional group classification really work? A validation in two contrasting habitats from Central Mexico. Ekológia. 2023; 42(1):26–38. https://doi.org/10.2478/eko-2023-0004
Rocha WO, Dorval A, Filho OP, Vaez CA, Ribeiro ES. Formigas (Hymenoptera: Formicidae) bioindicadoras de degradação ambiental em Poxoréu, Mato Grosso, Brasil. Floresta Ambient. 2015; 22(1):88–98. https://doi.org/10.1590/2179-8087.0049
Rodrigues RSS, Fernandes L, Crispim DL, Vieira SA, Pessoa FCL. Caracterização morfométrica da bacia hidrográfica do Igarapé da Prata, Capitão Poço, Pará, Brasil. Rev Verde Agroecol Desenvolv Sustent. 2016; 11(3):143–50. https://doi.org/10.18378/rvads.v11i3.4313
Roussel JM, Covain R, Vigouroux R, Allard L, Treguier A, Papa Y et al. Fish communities critically depend on forest subsidies in small neotropical streams with high biodiversity value. Biotropica. 2021; 53(4):1096–108. https://doi.org/10.1111/btp.12949
Sánchez-Hernández J, Nunn AD, Adams CE, Amundsen PA. Causes and consequences of ontogenetic dietary shifts: a global synthesis using fish models. Biol Rev. 2019; 94(2):539–54. https://doi.org/10.1111/brv.12468
Schwartz G. Manejo sustentável de florestas secundárias: espécies potenciais no Nordeste do Pará, Brasil. Amazônia: Ci & Desenv. 2007; 3(5):125–47.
Seabra LB, Palheta GS, Benone NL, Ayres-Santos B, Montag LFA. Environmental filters explain the ecomorphological patterns of stream fish in the southern Amazon. Acta Amaz. 2023; 53(3):223–31. https://doi.org/10.1590/1809-4392202203192
Silva BNR, Silva LGT, Rocha AMA, Sampaio SMN. Interação biofísica e do uso da terra na dinâmica da paisagem do município de Capitão Poço-PA, em sistema de informação geográfica. Documentos, 10. Belém: Embrapa Amazônia Oriental; 1999. Available from: https://www.infoteca.cnptia.embrapa.br/bitstream/doc/377456/1/OrientalDoc10.pdf
Silva DMA, Oliveira WFS, Santos JP, Oliveira FJM. Comendo no mesmo prato? sobreposição de nicho trófico de duas espécies de peixes em riachos da bacia do alto rio Tocantins. Heringeriana. 2019; 13(2):29–39. https://doi.org/10.17648/heringeriana.v13i2.917889
Silva NCS, Costa AJL, Louvise J, Soares BE, Reis VCS, Albrecht MP et al. Resource partitioning and ecomorphological variation in two syntopic species of Lebiasinidae (Characiformes) in an Amazonian stream. Acta Amaz. 2016; 46(1):25–36. https://doi.org/10.1590/1809-4392201501024
da Silva DA, Pessoa EKR, Costa SAGL, Chellappa NT, Chellappa S. Ecologia alimentar de Astyanax lacustris (Osteichthyes: Characidae) na lagoa do Piató, Assu, Rio Grande do Norte, Brasil. Biota Amaz. 2012; 2(1):74–82.
Soares BE, Benone NL, Rosa DCO, Montag LFA. Do local environmental factors structure the trophic niche of the splash tetra, Copella arnoldi? A test in an Amazonian stream system. Acta Amaz. 2020; 50(1):54–60. https://doi.org/10.1590/1809-4392201802681
Sousa JIM, Oliveira JCD, Oliveira JF, Rebouças LGF, Novaes JLC, Costa RS et al. Variação temporal e espacial na dieta de Trachelyopterus galeatus (Siluriformes, Auchenipteridae) em dois reservatórios no semiárido Neotropical. Iheringia Sér Zool. 2017; 107:e2017040. https://doi.org/10.1590/1678-4766e2017040
Stegmann LF, Leitão RP, Zuanon J, Magnusson WE. Distance to large rivers affects fish diversity patterns in highly dynamic streams of Central Amazonia. PLoS ONE. 2019; 14(10):e0223880. https://doi.org/10.1371/journal.pone.0223880
Strahler AN. Quantitative analysis of watershed geomorphology. Eos Trans Am Geophys Union. 1957; 38(6):913–20. https://doi.org/10.1029/TR038i006p00913
Teresa FB, Casatti L. Development of habitat suitability criteria for Neotropical stream fishes and an assessment of their transferability to streams with different conservation status. Neotrop Ichthyol. 2013; 11(2):395–402. https://doi.org/10.1590/S1679-62252013005000009
Taguti TL, Kipper D, Bialetzki A, Sanches PV, Makrakis MC, Baumgartner G et al. Early development of Pyrrhulina australis Eigenmann & Kennedy, 1903 (Characiformes, Lebiasinidae). Biota Neotrop. 2009; 9(4):59–65. https://doi.org/10.1590/S1676-06032009000400006
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE. The river continuum concept. Can J Fish Aquat Sci. 1980; 37(1):130–37. https://doi.org/10.1139/f80-017
Vieira LS, Netto-Ferreira AL. New species of Pyrrhulina (Teleostei: Characiformes: Lebiasinidae) from the eastern Amazon, Pará, Brazil. Neotrop Ichthyol. 2019; 17(2):e190013. https://doi.org/10.1590/1982-0224-20190013
Worischka S, Schmidt SI, Hellmann C, Winkelmann C. Selective predation by benthivorous fish on stream macroinvertebrates – The role of prey traits and prey abundance. Limnologica. 2015; 52:41–50. https://doi.org/10.1016/j.limno.2015.03.004
Zavala-Camin LA. Introdução aos estudos sobre alimentação natural em peixes. 1st ed. Maringá: EDUEM; 1996.
Zeni JO, Casatti L. The influence of habitat homogenization on the trophic structure of fish fauna in tropical streams. Hydrobiologia. 2014; 726:259–70. https://doi.org/10.1007/s10750-013-1772-6
Zeus FC, Cerezo RG, Pagés PA, Martínez JM, Romero JB, Sánchez-Montoya MD. Disentangling responses of aquatic and terrestrial invertebrates to drying in saline streams and shallow lakes. Aquat Sci. 2024; 86:57. https://doi.org/10.1007/s00027-024-01072-z
Zina V, Ordeix M, Franco JC, Ferreira MT, Fernandes MR. Ants as bioindicators of riparian ecological health in Catalonian Rivers. Forests. 2021; 12(5):625. https://doi.org/10.3390/f12050625
Authors
![]() Francisco de Paulo Tavares Ribeiro1
Francisco de Paulo Tavares Ribeiro1 ![]() ,
, ![]() Bruno da Silveira Prudente1 and
Bruno da Silveira Prudente1 and ![]() Tiago Magalhães da Silva Freitas2
Tiago Magalhães da Silva Freitas2
[1] Laboratório de Ecologia e Conservação da Amazônia (LABECA), Universidade Federal Rural da Amazônia, campus Capitão Poço, Rua Professora Antônia Cunha de Oliveira, Vila Nova, 68650-000, Capitão Poço, PA, Brazil. (FPTR) pauloribeiro995@gmail.com (corresponding author), (BSP) brunoprudente8@gmail.com.
[2] Laboratório de Zoologia (LabZoo), Universidade Federal do Pará, Campus Universitário do Marajó-Breves, Alameda IV, 3418, 68000-000, Breves, PA, Brazil. (TMSF) freitastms@gmail.com.
Authors’ Contribution 

Francisco de Paulo Tavares Ribeiro: Conceptualization, Formal analysis, Investigation, Methodology, Resources, Software, Validation, Visualization, Writing-original draft, Writing-review and editing.
Bruno da Silveira Prudente: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Tiago Magalhães da Silva Freitas: Conceptualization, Funding acquisition, Methodology, Resources, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Collections were legally authorized by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) (Permit number 63603/SISBIO) and regulated by the Ethics Committee on the Use of Animals (Protocol number 054/2018 CEUA) of the Universidade Federal Rural da Amazônia, Capitão Poço, Brazil.
Competing Interests
The author declares no competing interests.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
AI statement
No artificial intelligence (AI) tools were used in the writing, editing, translation, or data analysis of this manuscript.
Funding
This research was funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), through a scholarship granted during the development of this project.
Supplementary Material
Supplementary material S1
How to cite this article
xxxxxxxx
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted October 23, 2025
Accepted October 23, 2025
![]() Submitted April 3, 2025
Submitted April 3, 2025
![]() Epub April 17, 2026
Epub April 17, 2026