![]() Fernando Cesar Paiva Dagosta1,2,
Fernando Cesar Paiva Dagosta1,2, ![]() Willian Massaharu Ohara3 and
Willian Massaharu Ohara3 and ![]() Thomaz Jefrey Seren1,2
Thomaz Jefrey Seren1,2 ![]()
PDF: Download Here | Cite this article
Associate Editor: ![]() George Mattox
George Mattox
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Uma nova espécie é descrita das bacias dos rios Juruena e Guaporé, na Chapada dos Parecis, estado de Mato Grosso, Brasil. A nova espécie pode ser distinguida de todos os seus congêneres pela ausência de mancha umeral e de mancha no pedúnculo caudal, além de possuir nadadeira anal com 15–18 raios ramificados. As populações das bacias dos rios Juruena e Guaporé apresentam diferenças sutis nas proporções corporais, coloração e dentição; entretanto, em nossa avaliação, essas variações são insuficientes para justificar o reconhecimento como espécies distintas. As possíveis relações evolutivas e a biogeografia histórica da espécie são discutidas.
Palavras-chave: Captura de cabeceira, Chapada dos Parecis, Hyphessobryconinae, Tetra, Variação intraespecífica.
Introduction
Hyphessobrycon Durbin, 1908 is one of the most species-rich genera of Neotropical freshwater fishes, comprising nearly 150 valid species (Toledo-Piza et al., 2024; Fricke et al., 2025; Lima et al., 2025). The genus is diagnosed by the following combination of morphological characters: two teeth rows on the premaxilla with the innermost row with five teeth, incomplete lateral line, and caudal fin not scaled (Eigenmann, 1918). Although broadly distributed across the Neotropical realm, the majority of its diversity is concentrated within the Amazon Basin (Ohara, Lima, 2015; Dagosta, de Pinna, 2019; Toledo-Piza et al., 2024). In addition to its taxonomic richness, the genus exhibits remarkable morphological variation. Among all characids (sensu Mirande, 2019) and related groups, Hyphessobrycon stands out as one of the least morphologically homogeneous groups, particularly with respect to coloration patterns. Several species display dark lateral stripes, which vary from narrow and sharply defined (e.g., Dagosta et al., 2016; Ohara et al., 2017) to broad and diffuse (e.g., Ohara, Lima, 2015; Ohara et al., 2019). Others are vividly pigmented, exhibiting intense orange to red hues (e.g., Carvalho et al., 2014; Marinho et al., 2016; Camelier et al., 2018). Many species are sexually dimorphic, with males possessing bony hooks on fins (e.g., Vieira et al., 2016) and modified fin shapes, filamentous extensions, and/or distinct coloration compared to females (e.g., Lima, Gerhard, 2001; Moreira et al., 2002; Dagosta et al., 2014; Pastana et al., 2017). A unifying trait among species of Hyphessobrycon is their small body size. While some species may reach up to 60 mm in standard length (SL), the majority remain significantly smaller, with many recognized as miniature species (Weitzman, Vari, 1988; Toledo-Piza et al., 2014; Pastana et al., 2017).
Due to their vibrant coloration, peaceful behavior, and small size, several species of Hyphessobrycon possess traits highly valued by aquarium enthusiasts. Consequently, dozens of species within the genus have gained worldwide popularity in the ornamental fish trade, with interest continuing to grow as new graceful species are described each year. In fact, some species enter the aquarium market even prior to formal scientific description (Marinho et al., 2016), which is precisely the case for the species described herein. Recent ichthyofaunal surveys conducted in central Brazil, particularly in the Chapada dos Parecis region and adjacent areas drained by the upper rio Juruena and upper rio Guaporé, have revealed populations of a yet-undescribed Hyphessobrycon species already present in the aquarium trade. In this study, we formally describe this new species and provide brief remarks on its phylogenetic relationships and biogeographic context.
Material and methods
Counts and measurements follow Fink, Weitzman (1974), except for the number of horizontal scale rows below lateral line, which are counted to the pelvic-fin origin, not including the small scale at pelvic-fin origin and with the addition of distance from pelvic-fin origin to anal-fin origin. Standard length (SL) is given in millimeters and all other measurements are expressed as percentage of SL, or of head length (HL) for head subunits. In the description, the frequency of each count is provided in parentheses after the respective count, with the count of the holotype indicated by an asterisk. Number of cusps, small dentary teeth, supraneurals, branchiostegal rays, gill rakers, vertebrae, unbranched anal-fin rays and procurrent caudal-fin rays are obtained only from cleared and stained specimens (c&s) prepared according to Taylor, Van Dyke (1985). Radii and circuli counts were taken from the scale row immediately above the lateral line. Vertebrae of the Weberian apparatus are counted as four elements and the compound caudal centra (PU1+U1) as one element. The sex of specimens was confirmed in ten specimens by dissection and direct examination of the gonads. In the list of types and comparative material, catalog numbers are followed by the number of specimens in alcohol, their SL range, and if any, the number of c&s specimens and their respective SL range. Institutional abbreviations follow Fricke, Eschmeyer (2025). Lot MZUEL 17771 was not designated as paratype because it originated from the aquarium trade and lacks a known locality.
Results
Hyphessobrycon plumbellus, new species
urn:lsid:zoobank.org:act:4D8108CF-75AB-4442-9F2D-DCE0AA920660
(Figs. 1–4, 5A, B; Tab. 1)
Holotype. MZUSP 112070, 23.6 mm SL, Brazil, Mato Grosso State, Comodoro, tributary from rio Juína near of BR-364, 13°47’59.33”S 59°29’17.69”W, 21 Oct 2018, F. C. P. Dagosta, A. Ferreria & H. Lenza.
Paratypes. All from Brazil, Mato Grosso State, Comodoro Municipality. Rio Juruena basin: MUBIO 699, 558, 10.2–29.1 mm SL; MZUSP 124997, 229, 11.6–25.1 mm SL, 4 dna (MZICT 5207, 5208, 5209, 5210), 16.7–22.8 mm SL; INPA 62046, 20, 16.7–23 mm SL, MZUEL 24462, 20, 13.5–28.1; CITL 1571, 20, 14.8–25.8 mm SL, collected with the holotype. MZUSP 115691, 23, 17.6–25.1 mm SL, rio Juína, tributary of the rio Juruena, recreational site of the rio Juína, between Comodoro and Campos de Júlio, 13°47’41.2”S 59°27’19.6”W, 29 Sep 2012, O. T. Oyakawa, F. C. P. Dagosta, M. M. Marinho, P. Camelier. Rio Guaporé basin: MUBIO 698, 65, 12.2–26.2 mm SL, Comodoro Municipality, tributary of the rio Piolho, at the bridge on road BR-174, 13 km from Comodoro, upper rio Guaporé, rio Madeira basin, 13°46’50.5”S 59°46’10.3”W, 28 Oct 2022, F. C. P. Dagosta, T. J. Seren, B. A. Nagamatsu & J. Damasceno; UFRO 12313, 72, 17.8–37.6 mm SL, 5 c&s, 24.9–26.1 mm SL, 11 Dec 2012, W. M. Ohara, F. G. Vieira, J. A. Filho; INPA 61816, 40, 19.7–31.4 mm SL, 3 Aug 2018, W. M. Ohara.
Non-type material. MZUEL17771, 2, 21.1–22.2 mm SL, Brazil, Mato Grosso State, Campo Novo dos Parecis, uncertain locality, H. Lenza.
Diagnosis. Hyphessobrycon plumbellus differs from all congeners, except H. amandae Géry & Uj, 1987, H. axelrodi (Travassos, 1959), H. bayleyi Lima, Bastos, Rapp Py-Daniel & Ota, 2022, H. brumado Zanata & Camelier, 2010, H. compressus (Meek, 1904), H. diancistrus Weitzman 1977, H. eilyos Lima & Moreira, 2003, H. eos Durbin, 1909 H. gracilior Géry, 1964, H. georgettae Géry, 1961, H. juruna Faria, Lima & Bastos, 2018, H. minimus Durbin, 1909, H. parvellus Ellis, 1911, H. piabinhas Fowler, 1941, H. procerus Mahnert & Géry, 1987, H. saizi Géry, 1964, H. scutulatus Lucena, 2003, H. taurocephalus Ellis, 1911, and H. tukunai Géry, 1965 by the absence of a dark humeral blotch (vs. presence of a dark humeral spot and/or presence of a dark longitudinal stripe covering the humeral region). It can be distinguished from H. amandae, H. axelrodi, H. compressus, H. georgettae, and H. milleri by the absence of concentration of dark chromatophores in the dorsal fin forming a blotch (vs. presence). From all the remaining species, except H. bayleyi, H. diancistrus, H. eilyos, H. gracilior, H. otrynus and H. procerus by not having a delimited caudal-peduncle blotch (vs. presence of concentration of chromatophores forming a distinct caudal-peduncle blotch). The new species differs from H. bayleyi, H. diancistrus and H. otrynus by the caudal-fin pigmentation (hyaline vs. two symmetrical dark blotches at the distal portion of the caudal-fin lobes in H. bayleyi and black at distal third of their length in H. otrynus), from H. gracilior and H. procerus by having fewer branched anal-fin rays (15–18 vs. 18–20 in H. gracilior; 22–26 in H. procerus). It can be further distinguished from H. gracilior by the presence of inner premaxillary, maxillary, and dentary teeth with five to seven cusps (vs. conical to three). It differs from H. eilyos by the presence of a midlateral stripe extending from the middle of the body to the middle caudal-fin rays (vs. uniformly dense body pigmentation without the formation of a midlateral stripe). Additionally, H. plumbellus can be diagnosed from its congeners by having four (rarely five) teeth in inner row of premaxillary. A further useful diagnostic feature is the presence of inner premaxillary, maxillary, and dentary teeth with up to seven cusps (vs. up to five cusps in most congeners).
Description. Morphometric data for the holotype and paratypes presented in Tab. 1. Body compressed, moderately short and deep. Greatest body depth slightly anterior to dorsal-fin origin. Dorsal profile of head convex from tip of upper jaw to vertical through posterior nostril; straight to slightly concave from that point to tip of supraoccipital spine. Dorsal profile of body convex from supraoccipital spine tip to dorsal-fin origin; straight to slightly concave along dorsal-fin base; straight to slightly concave from terminus of dorsal-fin to adipose-fin origin and concave between adipose-fin origin, and concave along caudal peduncle. Ventral profile of head and body convex from tip of lower lip to pelvic-fin origin; slightly convex from that point to anal-fin origin; straight and posterodorsally inclined along anal-fin base, and concave along caudal peduncle.
TABLE 1 | Morphometric data of the holotype and paratypes of Hyphessobrycon plumbellus. Range not includes the values of the holotype. Number of specimens: rio Guaporé n = 42; rio Juruena n = 12; SD = Standard deviation.
| Holotype | Rio Juruena basin | Rio Guaporé basin | ||||
Range | Mean | SD | Range | Mean | SD | ||
Standard length (mm) | 23.6 | 21.1–29.0 | 24.3 | – | 17.3–37.6 | 27.2 | – |
Percentage of standard length | |||||||
Depth at dorsal-fin origin | 38.7 | 37.9–42.3 | 40.3 | 1.3 | 31.7–38.1 | 35.0 | 1.8 |
Snout to dorsal-fin origin | 54.8 | 54.1–59.5 | 56.2 | 1.8 | 51.0–56.8 | 53.1 | 1.2 |
Snout to pectoral-fin origin | 27.6 | 27.6–31.3 | 29.5 | 1.2 | 25.8–30.7 | 27.9 | 1.3 |
Snout to pelvic-fin origin | 53.2 | 53.0–55.5 | 54.2 | 0.9 | 48.4–55.0 | 51.1 | 1.4 |
Snout to anal-fin origin | 67.7 | 67.1–70.4 | 68.7 | 1.1 | 64.0–71.5 | 66.6 | 1.4 |
Caudal peduncle depth | 13.0 | 12.3–14.4 | 13.5 | 0.7 | 11.5–14.7 | 12.8 | 0.7 |
Caudal peduncle length | 12.2 | 9.8–12.6 | 11.0 | 0.9 | 7.9–15.4 | 13.2 | 1.9 |
Pectoral-fin length | 24.0 | 19.6–24.0 | 21.6 | 1.4 | 18.0–21.9 | 20.5 | 0.9 |
Pelvic-fin length | 20.6 | 17.3–20.6 | 18.7 | 1.1 | 16.0–18.4 | 17.2 | 0.6 |
Dorsal-fin base length | 16.3 | 13.9–18.1 | 16.4 | 1.1 | 13.2–18.5 | 15.3 | 1.4 |
Dorsal-fin length | 31.0 | 28.1–33.3 | 31.1 | 1.8 | 26.3–33.0 | 29.6 | 1.7 |
Anal-fin base length | 24.7 | 24.4–28.2 | 26.6 | 1.2 | 22.9–29.8 | 25.5 | 1.6 |
Anal-fin length | 19.5 | 17.6–22.3 | 20.1 | 1.5 | 15.0–21.1 | 18.0 | 1.5 |
Head length | 26.8 | 26.7–28.6 | 27.8 | 0.6 | 24.1–29.4 | 26.3 | 1.2 |
Percentage of head length | |||||||
Horizontal length eye | 38.9 | 35.9–41.7 | 38.5 | 1.7 | 32.3–42.6 | 37.9 | 2.6 |
Snout length | 23.5 | 21.6–26.0 | 24.0 | 1.6 | 19.2–26.1 | 23.1 | 1.6 |
Least interorbital width | 34.9 | 29.7–34.9 | 32.3 | 1.6 | 27.8–35.3 | 32.3 | 1.9 |
Upper jaw length | 46.2 | 39.9–48.3 | 44.5 | 2.0 | 37.7–45.8 | 42.4 | 1.8 |
Jaws equal, mouth terminal. Posterior terminus of maxilla reaching vertical through anterior margin of pupil. Maxilla approximately at 45 degree angle relative to longitudinal axis of body. Nostrils close to each other, anterior opening circular, posterior opening crescent-shaped. Nostrils separated by narrow flap of skin.
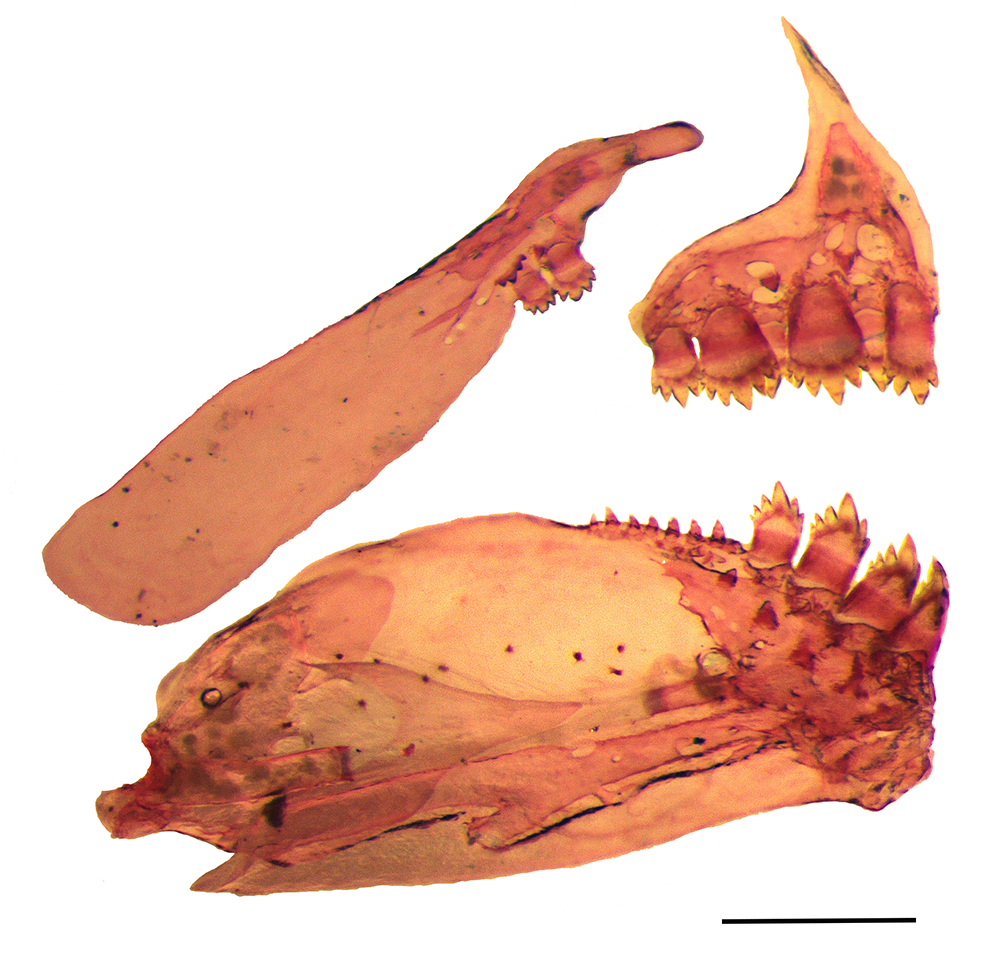
Premaxillary teeth in two rows; outer teeth row with 2(22) or 3*(31) tricuspid teeth; inner row with 4(50) or 5*(4) teeth with five to seven cusps, symphyseal tooth of inner series narrower and laterally asymmetric. Maxilla with 1(12), 2*(38), or 3(4) teeth with five to seven cusps (Fig. 2); anteriormost tooth larger. Dentary with 4(40) or 5*(35) penta- to heptacuspid larger teeth, followed by series of 4(1), 6(3), or 7(1) small conical or tricuspid teeth. Central cusp of all teeth longer than lateral cusps. Branchiostegal rays 4(5). First gill arch with 2(5) gill rakers on hypobranchial, 8(5) on ceratobranchial, 1(5) on intermediate cartilage, and 5(3) or 6(2) rakers on epibranchial.
FIGURE 1| Hyphessobrycon plumbellus, MZUSP 112070, holotype, 23.6 mm SL, from the upper rio Juruena, rio Tapajós basin, Comodoro Municipality, Mato Grosso State, Brazil.
FIGURE 2| Medial view of left side of the jaws of Hyphessobrycon plumbellus, UFRO 12313, paratype, 25.3 mm SL. Scale bar = 200 m.
Scales cycloid, circuli distributed over whole area of scales with two to eight radii diverging nearly from anterior margin to posterior border. Perforated scales of lateral line 5(1), 6(5), 7*(10), 8(21), 9(14), 10(3), 12(2), or 16(1); longitudinal scales series including pored scales 30(3), 31(10), 32*(31), or 33(2). Longitudinal scale rows between dorsal-fin origin and lateral line 5*(57). Longitudinal scale rows between lateral line and pelvic-fin origin 4*(57). Scales in median series between tip of supraoccipital spine and dorsal-fin origin 8(4), 9(15), 10*(25), or 11(2). Horizontal scale rows around caudal peduncle 13(5) or 14*(45). Single row of 2(2), 3*(17), or 4(10) scales covering base of anterior most anal-fin rays. Caudal fin not scaled.
Dorsal-fin rays ii,8(2) or ii,9*(52). Dorsal-fin origin at middle of body and slightly posterior to vertical through pelvic-fin origin. First unbranched dorsal-fin ray shorter than second ray. First dorsal-fin pterygiophore inserted posterior to neural spine of 9th(3) or 10th(2) vertebra. Pectoral-fin rays i,9*(23), 10(35), or 11(1), not reaching pelvic-fin origin. Pelvic-fin rays i,6*(47) or i,7(6). Tip of pelvic fin reaching anterior portion of anal-fin. Adipose fin present. Anal-fin with iv(4) or v(1), 15(4), 16*(24), 17(23), or 18(6) rays. First anal-fin pterygiophore inserted posterior to haemal spine of 16th(3) or 17th(2) vertebra. Principal caudal-fin rays i,9,8,i*(51) or i,8,8,i(3). Caudal-fin forked, lobes somewhat pointed and of similar size. Dorsal procurrent caudal-fin rays 9(3) or 10(2); ventral procurrent caudal-fin rays 8(3) or 9(2). Total vertebrae 32(4) or 33(1): precaudal vertebrae 15(1) or 16(4) and caudal vertebrae 16(3) or 17(2). Supraneurals 4(1) or 5(4).
Coloration in alcohol. Overall ground coloration of body pale yellow. Dorsal portion of head and dorsal midline of body darker. Anterior portion of jaws, infraorbitals 1–2, snout and dorsal portion of head with concentration of small dark chromatophores. Opercle series and infraorbitals 2–4 with scattered dark chromatophores. Opercular series with guanine retention in some specimens. Humeral blotch absent. Muscle reduction at humeral region rendering darker coloration to the area. Narrow subjacent longitudinal dark line extending along horizontal septum, from vertical through middle pectoral fin to end of caudal peduncle. Superficially, a concentration of dark chromatophores is present along this dark line, beginning approximately at the vertical through the origin of the pelvic fin. This concentration becomes progressively denser posteriorly, reaching its greatest intensity on the caudal peduncle region, though without forming a distinct caudal-peduncle blotch. Dark midlateral longitudinal stripe narrower anteriorly and broader posteriorly, extending to tip of middle caudal-fin rays formed by concentration of dark chromatophores. Dorsal region with concentration of dark chromatophores. Abdominal region lighter with few small dark chromatophores; guanine retention in some individuals. Faint dark chromatophores on lower portion of body arranged along myosepta of the hypaxial muscle bundles from area above anal-fin end to caudal peduncle. Unpaired fins with concentration of dark chromatophores on interradial membranes, especially in males (see Sexual Dimorphism section). Paired and adipose fins with small, scattered chromatophores.
Coloration in life. Dorsal ground coloration sexually dimorphic (Fig. 3): blueish to greyish in males, greyish to olive in females. Abdominal region silvery; ventral portion occasionally light orange in mature males from rio Guaporé basin (Fig. 3A). Dorsal portion of eye red to orange. Head olive dorsally; silvery in infraorbital, jaws, gular, and opercular regions. Fin coloration sexually dimorphic: grayish in males, with yellowish portions in females (Figs. 3B, D). Midlateral golden stripe extending from head to caudal peduncle, more conspicuous in specimens from rio Guaporé basin.
FIGURE 3| Live specimens of Hyphessobrycon plumbellus from the rio Guaporé basin (MUBIO 698, A. Male; B. Female) and from the rio Juruena basin (MUBIO 689, C. Male; D. Female). Specimens not measured.
Sexual dimorphism. Characteristics based on examination of fixed specimens, field observations and photographs of live specimens (Fig. 3). Although several morphological traits differentiate males from females, individuals at the extremes are more readily sexed. In many cases, however, sex identification is more challenging and may require gonadal examination for confirmation. Well-preserved specimens, particularly those with intact fins, greatly facilitate accurate sexual determination. Males with more conspicuous grayish pigmentation, extending over the dark midlateral stripe, which is more pronounced in females (Fig. 3). Males proportionally with deeper body than females (Figs. 6B, C). Live males with overall more vivid coloration; eyes with more intense reddish pigmentation. Presence of sexual dichromatism: dorsal region grayish to bluish in males, olive-colored in females (Fig. 3). Dorsal, anal, pelvic and caudal fins with denser dark pigmentation in males; less intense in females (Figs. 3–4). Branched anal-fin rays proportionally longer in males (Figs. 3–4). The distal profile of the anal fin is straighter in males and more concave in females, forming a more prominent anterior lobe (Figs. 3–4). Dorsal fin somewhat more rounded in males than in females (Figs. 3–4). Adipose and base of caudal fin grayish in males; yellowish in females (Fig. 3). Pelvic-fin rays proportionally longer in males.
FIGURE 4| Preserved specimens of Hyphessobrycon plumbellus from the rio Guaporé basin (UFRO 12313, A. Male, 31.4 mm SL; B. Female, 31.8 mm SL) and from the rio Juruena basin (MUBIO 699, C. Male, 21.7 mm SL; D. Female, 25.6 mm SL).
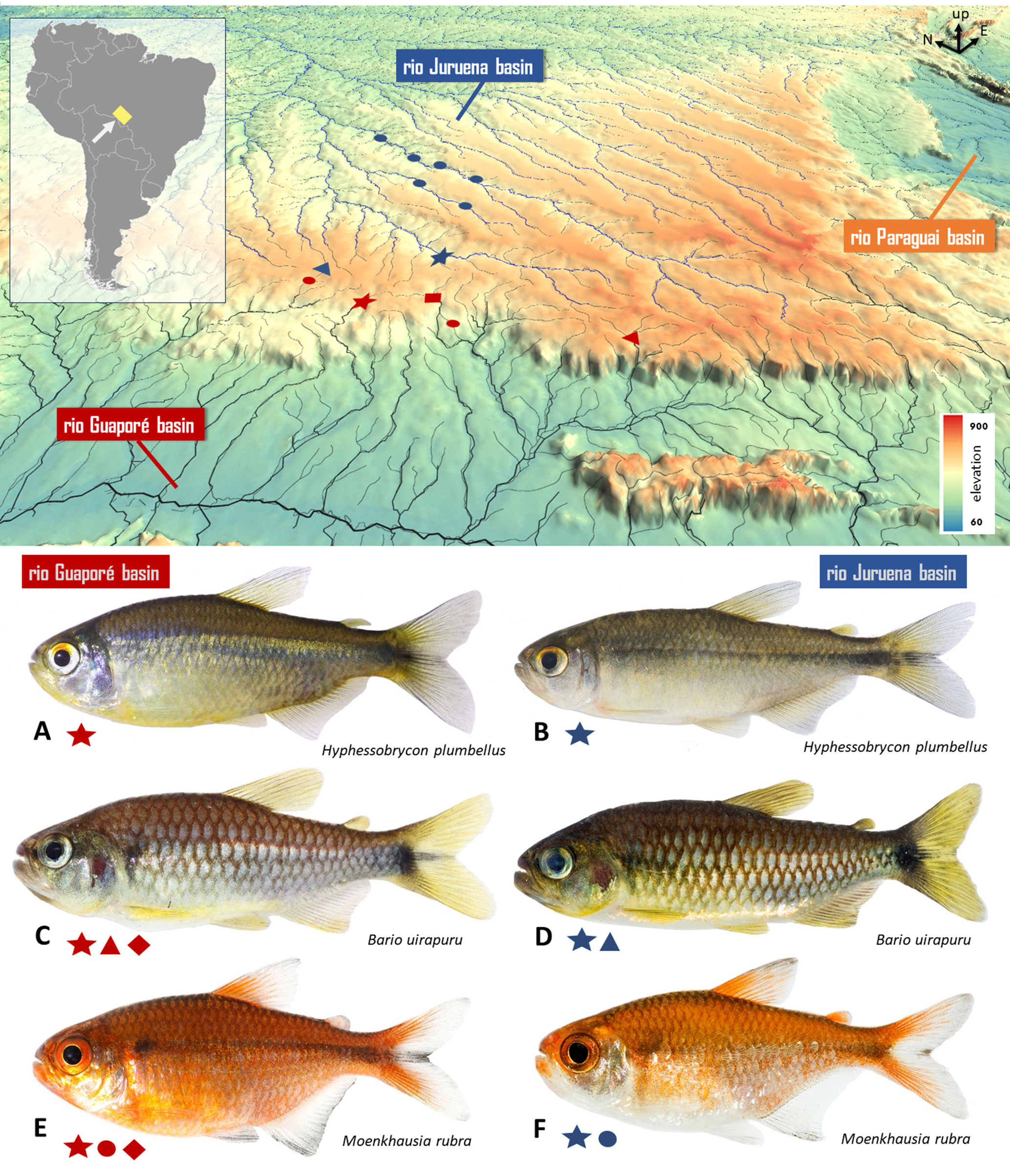
Geographical distribution. The new species is known from three localities: a tributary of the rio Piolho in the upper rio Guaporé, rio Madeira basin, and two nearby sites in the rio Juína basin, upper rio Juruena, rio Tapajós basin. All localities are situated in the Chapada dos Parecis, Comodoro Municipality, Mato Grosso State, Brazil (Fig. 5).
FIGURE 5| Map of the Chapada dos Parecis, viewed from above and highlighting its western region. Stars indicate the distribution of Hyphessobrycon plumbellus, and the blue star representing the type-locality. Blue symbols represent collection sites within the rio Juruena basin, while red symbols correspond to localities in the rio Guaporé basin. Symbols may represent more than one collection event. Each locality on the map has its own ichthyofaunal composition, represented by different symbols (star, diamond, or triangle). These symbols are placed below each species to indicate their occurrence across the respective localities. Representative fish species shared between adjacent basins: A. H. plumbellus female (rio Guaporé basin); B. H. plumbellus female (rio Juruena basin); C. Bario uirapuru (rio Guaporé basin); D. B. uirapuru (rio Juruena basin); E. Moenkhausia rubra (rio Guaporé basin); F. M. rubra (rio Juruena basin).
Ecological notes. The population of Hyphessobrycon plumbellus from the rio Guaporé basin inhabits both a small clearwater tributary, 3–5 m wide and 0.5–2 m deep, characterized by swift current and a substrate composed of rocks, sand, and submerged vegetation, as well as an upstream reservoir formed by a hydroelectric power plant. The reservoir features transparent water, an average depth of 1.5 m, and sparse aquatic vegetation. The species is by far the most abundant species in this region. Downstream of the hydroelectric plant, the species was collected syntopically with Saxatilia lepidota (Heckel, 1840), Crenicichla sp., Characidium nambiquara Zanata & Ohara, 2020, Hoplias malabaricus (Bloch, 1794) and Moenkhausia rubra Pastana & Dagosta, 2014. In contrast, only Aequidens sp. and S. lepidota were recorded in the reservoir. The analysis of the stomach contents of five cleared and stained paratypes revealed mostly filamentous algae, and smaller quantities of ants, spider, Odonata, larvae of Diptera (Chironomidae), nematoids, Ephemeroptera, unidentified insect fragments, and sand. Some individuals of H. plumbellus possess a bulge on the side of the body, caused by a nematode endoparasite.
Conservation status. The species is known only from the municipality of Comodoro, Mato Grosso State, Brazil, in three localities: one in a tributary of the rio Piolho, upper rio Guaporé, rio Madeira basin, and two closely situated sites in the rio Juína basin, upper rio Juruena, rio Tapajós basin. In the Guaporé locality, specimens were collected both immediately downstream and upstream of a small hydroelectric power plant. The species appears to have adapted well to the small reservoir formed by the dam, where it is by far the most abundant fish. Thus, the species shows high adaptability to such altered environment. Another potential threat is collection for the ornamental fish trade. However, according to ornamental fishers, the species currently has low commercial value due to its successful captive breeding by aquarists, and there is little remaining demand for wild-caught specimens. Additionally, the species occurs in the main channel of the rio Juína, a large river that enters the Nambikwara Indigenous Territory just over 10 km downstream from the collection site, where it likely finds pristine habitat conditions. Therefore, Hyphessobrycon plumbellus should be classified as Least Concern (LC) under the categories and criteria of the International Union for Conservation of Nature (IUCN, 2024).
Etymology. The specific name plumbellus is derived from the Latin plumbeus, meaning “leaden,” in reference to the grayish-blue coloration of the males, which has led to the species being popularly known in the aquarium trade as the “tetra chumbinho” in portuguese (= “little lead tetra in english”).The suffix -ellus is a Latin diminutive indicating a reduced expression of a quality, here referring to the subtle plumbaceous coloration. In addition, the diminutive may also allude to the small body size of the species. An adjective.
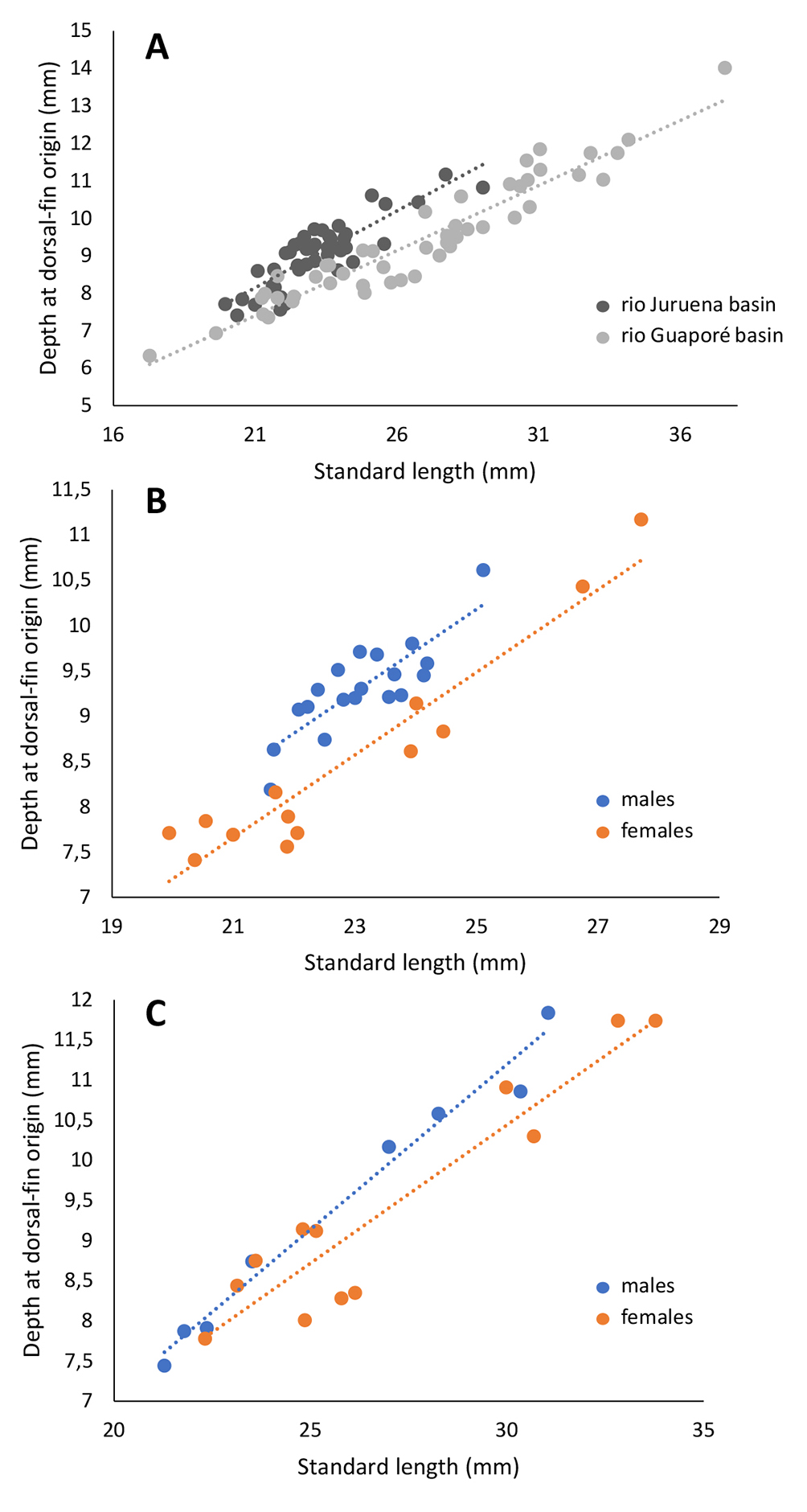
Intraspecific variation. The golden midlateral stripe is markedly more conspicuous in specimens from the rio Guaporé population. Specimens from the rio Juruena population have proportionally deeper bodies: depth at dorsal-fin origin 37.9–42.3% SL (mean 40.3% SL) vs. 31.7–38.1% SL (mean 35.0% SL) (Fig. 6). Specimens from the rio Guaporé reach larger body sizes, up to 37.6 mm SL, whereas those from the rio Juruena attain only up to 29.1 mm SL. A meristic difference is also observed. Most specimens from rio Guaporé basin possess four large dentary teeth (n = 40), and only two with five, while all individuals from Juruena (n = 33) exhibit five large dentary teeth. Sexual dimorphism, although present in both populations, is more pronounced in specimens from the rio Juruena basin.
FIGURE 6| Comparative regression analysis of body depth at the dorsal-fin origin (mm) as a function of standard length (SL, mm). A. Regression comparison between specimens from the rio Juruena (R² = 0.7314) and rio Guaporé (R² = 0.906) basins, with individuals of both sexes pooled. B. Sexual dimorphism within the rio Juruena population: females (n = 13, R² = 0.9087) and males (n = 19, R² = 0.692). C. Sexual dimorphism within the rio Guaporé population: females (n = 12, R² = 0.8776) and males (n = 8, R² = 0.9774).
Discussion
According to Durbin (1908), Hyphessobrycon differs from Hemigrammus Gill, 1858 solely by the absence of scales on the caudal-fin lobes, a definition that has remained in use to this day due to ongoing phylogenetic uncertainties within these groups. This character, however, is highly homoplastic among characids, and scales are easily lost through friction, even in recently collected specimens (Carvalho et al., 2010). As such, a formal diagnosis distinguishing the new species from all species of Hemigrammus is required. Hyphessobrycon plumbellus can be readily distinguished from all species of Hemigrammus, except He. boesemani Géry, 1959, He. brevis Ellis, 1911, He. diagonicus Mendonça & Wosiacki, 2011, He. durbinae Ota, Lima & Pavanelli, 2015, He. filamentosus Zarske, 2011, He. hyanuary Durbin, 1918, He. mahnerti Uj & Géry, 1989, He. marginatus Ellis, 1911, He. matei Eigenmann, 1918, He. micropterus Meek, 1907, He. parana Marinho, Carvalho, Langeani & Tatsumi, 2008, He. rodwayi Durbin, 1909, He. silimoni Britski & Lima, 2008, and He. tridens Eigenmann, 1907 by the absence of a humeral spot (vs. presence of a dark humeral spot and/or presence of a dark longitudinal stripe covering the humeral region). Beyond the lack of scales covering the caudal-fin lobes, Hy. plumbellus can be diagnosed from all aforementioned species, except He. levis Durbin, 1908, He. mahnerti, He. rodwayi, He. silimoni, and He. tridens by having 15–18 branched anal-fin rays (vs. 14 or less in He. hyanuary and He. rhodostomus (Ahl, 1924), 19 or more in other species). It differs from He. levis, He. mahnerti, He. rodwayi, and He. tridens by not having a dark well-delimited caudal-peduncle blotch (vs. presence) and from He. silimoni by the absence of an ocellated caudal-fin blotch on the dorsal caudal-fin lobe (vs. presence).
Generic allocation, evolutionary relationships and biogeography. Despite clear evidence that Hyphessobrycon is not a monophyletic group (Javonillo et al., 2010; Oliveira et al., 2011; Mirande, 2019; Melo et al., 2024), the genus continues to be defined according to the characters originally proposed by Durbin in Eigenmann (1908) and later followed by Eigenmann (1917, 1918). This persistence is largely due to the inability of available phylogenetic hypotheses to provide practical diagnostic characters for the clades recovered in their analyses. As a result, these clades cannot yet be translated into workable taxonomic units. This limitation reflects the extremely high complexity of the group, which is not only species-rich, but also exhibits a high degree of homoplasy.
According to the most recent phylogenetic hypothesis (Melo et al., 2024), Hyphessobrycon is highly polyphyletic, although most species are placed within the subfamily Hyphessobryconinae. The species of the genus that are endemic to the Chapada dos Parecis and included in the analysis of Melo et al. (2024, fig. 6) form a closely related group within a clade that also includes the genera Erythrocharax Netto-Ferreira, Birindelli, Sousa, Mariguela & Oliveira, 2013, Dinotopterygium Frainer, Carvalho, Bertaco & Malabarba, 2021, Phycocharax Ohara, Mirande & Lima, 2017, together with additional species of Hyphessobrycon. Notably, members of this clade exhibit teeth with up to seven cusps (except Hyphessobrycon psittacus Dagosta, Marinho, Camelier & Lima, 2016) and lack bony hooks on fins (except in species of Dinotopterygium), both of which are conditions present in H. plumbellus. This clade is sister to another composed mainly of lowland Amazonian species such as H. heterorhabdus (Ulrey, 1894) and H. agulha Fowler, 1913, and together these two clades form the sister group to the lineage that contains the type species of the genus, H. compressus. Although species of Hyphessobrycon are scattered across several unrelated lineages (see Melo et al., 2024), the clade to which H. plumbellus is most likely to belong is closely related to the type species of the genus. Given this proximity, and despite the availability of more recently described generic names such as Dinotopterygium, Erythrocharax, and Phycocharax that are more closely related, we adopt a conservative approach and retain the species within Hyphessobrycon.
In the absence of a formal hypothesis regarding the phylogenetic relationships of the species, we are limited to highlighting certain morphological traits that stand out and may carry evolutionary significance. These traits should ultimately be evaluated in the context of future phylogenetic analyses. Although it may be premature to propose a relationship between two species without analytical support, the aim here is merely to point out morphological, ecological, and biogeographical similarities that may suggest a potential close relationship.
Among all known species of Hyphessobrycon, the one that most closely resembles H. plumbellus is H. myrmex Pastana, Dagosta & Esguícero, 2017, particularly the females (see Fig. 7). This similarity is marked by a set of relatively uncommon traits among Hyphessobrycon species shared between the two species, which deserve special attention. Both exhibit a distinct black midlateral stripe accompanied dorsally by an orange band, a feature also observed in other speciesof Hyphessobrycon (e.g., H. vilmae Géry, 1966, species of the H. heterorhabdus group sensu Faria et al. (2021)). Both species exhibit a midlateral dark stripe that is narrower anteriorly and becomes progressively broader posteriorly, without forming a distinctly delimited caudal-peduncle blotch. Other traits are related to sexual dimorphism. Both H. plumbellus and H. myrmex display sexual dichromatism, although the pigments involved differ. In H. plumbellus, coloration is based on black and blue pigments, whereas in H. myrmex, it results from black and carotenoid-based pigments (Pastana et al., 2017). In both species, males exhibit significantly denser pigmentation in the anal fin compared to females, making the anal-fin rays more prominent. The shape of the anal fin is also sexually dimorphic, with females having a well-defined anterior lobe, while in males the distal profile of the fin is proportionally straighter and the anterior lobe is poorly developed or indistinct. Another shared feature is the reddish pigmentation on the dorsal portion of the eye, which is consistently more intense in males than in females. Notably, both species have become highly abundant in impounded environments formed by small hydroelectric power plants (PCHs). In many of these sites, they are the dominant or even the only species consistently recorded.
FIGURE 7| Live coloration pattern of Hyphessobrycon myrmex,MZUSP 118672, paratypes: A. Male, 21.6 mm SL; B. Female, 19.5 mm SL.
Biogeographic evidence for this relationship includes the observation that several tetra species endemic to the Chapada dos Parecis have sister taxa or closely related congeners distributed across neighboring basins, never occurring in sympatry. In the case of H. plumbellus and H. myrmex, the former occurs in the rio Juína basin, with a secondary presence in the rio Guaporé basin, while the latter inhabits the adjacent rio Formiga basin. Secondary occurrences likely represent recent range expansions due to headwater capture events along the Chapada escarpment and are discussed in the Biogeography section.
This pattern is not unique to H. plumbellus and H. myrmex. For example, H. wadai Marinho, Dagosta, Camelier & Oyakawa, 2016 is endemic to the rio do Sangue (Marinho et al., 2016), whereas its sister species H. cyanotaenia Zarske & Géry, 2006 (Melo et al., 2024) occurs in the rio Juruena basin, with secondary presence in the rio Guaporé (Dagosta et al., 2016). Similarly, Bario uirapuru (Ohara & Lima, 2015) inhabits the rio Juína basin with secondary presence in the rio Guaporé basin (Dagosta, de Pinna, 2019), and B. cosmops (Lima, Britski & Machado, 2007) occurs in the rio Juruena and rio Papagaio basins, with secondary presence in the Paraguay and Guaporé basins (Lima et al., 2007; Dagosta, de Pinna, 2019). The sister group to this clade is Bario skolioplatus (Bertaco & Carvalho, 2005) (Melo et al., 2024), endemic to the rio Mutum and rio Doze de Outubro basins.
Other morphologically similar pairs for which phylogenetic hypotheses are lacking follow the same trend. Hyphessobrycon melanostichos Carvalho & Bertaco, 2006 and H. comodoro Dagosta, Seren, Ferreira & Marinho, 2022 are found in adjacent basins (rio Doze de Outubro and rio Mutum, respectively) and are often traded under the same name in the ornamental fish market (Dagosta et al., 2022). Likewise, Moenkhausia rubra Pastana & Dagosta, 2014 inhabits the rio Juína and rio Juruena basins, with secondary occurrence in the rio Guaporé, whereas M. nigromarginata Costa, 1994 is recorded in the rio Papagaio and rio Sangue basins (Dagosta, de Pinna, 2019).
The two known populations of H. plumbellus do not constitute a metapopulation, as there is no gene flow between them. They inhabit separate drainage basins, and this distribution holds important implications for historical biogeography. It is crucial here to distinguish between dispersion and dispersal in the biogeographic sense (Platnick, 1976). Dispersion refers to the movement of individuals within their normal range as part of their life cycle (Platnick, 1976; Dagosta, de Pinna, 2017), whereas dispersal denotes the crossing of a pre-existing geographic barrier. Since freshwater fishes are confined to aquatic environments, river basin divides represent strict biogeographic barriers that lie outside their suitable range. Therefore, dispersion cannot account for the presence of freshwater fishes in separate basins; only dispersal or vicariance can explain such a distribution. The distinction between these two processes lies in the relative age of the geographic barrier and the taxa involved: if dispersal is invoked, the barrier must predate the divergence of the taxa; if vicariance is the explanation, then the barrier is as old as resulting taxa.
For freshwater fishes, which are incapable of aerial or terrestrial movement (Myers, 1947), dispersal is often considered an ad hoc explanation. As Nelson (1978) noted, dispersal is “the science of the rare, the mysterious, and the miraculous”. Any distributional pattern can be attributed to dispersal, but such explanations typically lack mechanistic detail, predictive power, and falsifiability. Dispersal hypotheses are inherently species-specific, depending on the physiological or ecological traits of individual taxa, and thus do not account for broader biogeographic patterns (Croizat et al., 1974; Nelson, Platnick, 1981). As Rosen (1976) emphasized, while dispersal may enable a species to cross a barrier, it does not generate repeatable patterns across taxa. In contrast, as Croizat (1958) argued, vicariant events generate shared biogeographic patterns, while dispersal tends to obscure them. Dispersal hypotheses for freshwater fishes invoke mechanisms such as fish eggs attaching to (epizoochory or ectozoochory) or egestion from (endozoochory) waterbirds (Hirsch et al., 2018; Silva et al., 2019; Lovas-Kiss et al., 2020, 2024; Green et al., 2023; Yao et al., 2024) or fishes being transported by windstorms during extreme weather events. The scientific literature provides little convincing evidence that such events play a significant role in facilitating fish dispersal across drainage divides, and current explanations remain largely speculative, with little support from rigorous empirical data.
In contrast, multiple unrelated freshwater taxa, including Bario uirapuru, Hyphessobrycon plumbellus, and Moenkhausia rubra (Fig. 5), display the same disjunct distribution across the Guaporé and Juruena basins. Other tetras, such as Hyphessobrycon cyanotaenia, H. hexastichos Bertaco & Carvalho, 2005, H. psittacus, and Bario cosmops, are likewise confined to these two basins in other parts of the Chapada dos Parecis (Dagosta, de Pinna, 2019). Moreover, a putatively monophyletic species pair, Melanorivulus modestus (Costa, 1991)(rio Juruena basin) and M. melanopterus Nielsen & Ohara, 2024 (rio Guaporé basin), is also confined to these basins (Nielsen, Ohara, 2024). In this case, headwater capture is likely to have driven allopatric speciation.
The repeated pattern of species or lineages shared exclusively between the same drainages strongly suggests a common underlying mechanism. In historical biogeography, it is vicariance, rather than random species-specific dispersal, that gives rise to shared distributional patterns across taxa. Therefore, the occurrence of H. plumbellus in both the rio Guaporé and rio Juruena basins is best explained by a vicariant event. As previously discussed, because freshwater fishes are confined to aquatic corridors, their current biogeographic distributions must be interpreted in light of past drainage reorganizations that allowed for temporary hydrological connectivity, followed by subsequent isolation. Indeed, time-series remote sensing imagery, topographic analysis, and erosion rates show that drainage divides are not static but highly dynamic (Dahlquist et al., 2018; Scheingross et al., 2020; He et al., 2024).
Bishop (1995) identifies three mechanisms of drainage reorganization: Capture (bottom‐up river capture), Diversion (top‐down river capture), and Beheading. Bottom-up capture occurs when a stream at a lower elevation with a higher rate of erosion intercepts the flow of an adjacent stream at a higher elevation. This process is driven by asymmetric erosion, where one side of a drainage divide erodes more rapidly than the other. In tectonically inactive regions, drainage divides tend to migrate toward areas with lower erosion rates (Mudd, Furbish, 2005; He et al., 2024; Hoskins et al., 2024). Streams with steeper gradients and higher stream power are generally more erosive, carving deeper into the landscape and gradually shifting the divide in the direction of the less erosive basin. This shift is a key mechanism of headward erosion, through which a more energetic stream progressively expands into the neighboring basin, effectively ‘stealing’ drainage area. When this headward erosion reaches a tributary of the adjacent basin, a river capture event occurs: the intercepted stream is diverted, and its flow is rerouted into the capturing river (Prince et al., 2011; Stokes et al., 2018; He et al., 2024; Hoskins et al., 2024). This process fundamentally alters the hydrological connectivity between basins and may facilitate the exchange of aquatic biota across previously isolated drainage system. It also may promote allopatric speciation by fragmenting populations (e.g., Wilkinson et al., 2006; Albert, Crampton, 2010; Albert, Reis, 2011; Albert et al., 2020; He et al., 2024).
Another form of drainage reorganization is diversion, or top-down river capture, in which a stream at higher elevation redirects its flow into a lower-elevation drainage system by breaching the divide (Bishop, 1995; Shugar et al., 2017; He et al., 2024; Hoskins et al., 2024). In this case, the initiative comes from the stream that is eventually captured. Due to lateral erosion, channel instability, or avulsion, the stream gradually alters its course and begins to flow toward a neighboring valley. This redirection may naturally lead to a new connection with an adjacent drainage network. Unlike bottom-up capture, this process does not involve direct headward erosion from a neighboring basin; instead, the stream effectively abandons its original catchment and integrates into another. It redirects its flow to a different basin, driven by internal dynamics rather than external erosive forces (e.g., Bishop, 1995; Shugar et al., 2017; Hoskins et al., 2024).
Finally, drainage reorganization may also occur through beheading, or divide migration, in which drainage area is gradually transferred from one basin to another without the preservation of original channels or flow paths (Bishop, 1995; Stokes et al., 2018; Hoskins et al., 2024). Unlike capture (bottom-up) or diversion (top-down), beheading does not involve the direct transfer of river channels. Instead, it results in the loss of drainage area from one catchment to another, without the migration of active stream courses. Another key distinction lies in the nature and tempo of these processes: capture and diversion typically occur over short timescales and are often considered abrupt, discrete events of drainage reorganization (Hoskins et al., 2024). In contrast, beheading is a gradual and continuous process, driven by the long-term shift of the drainage divide, often as a result of differential erosion or tectonic uplift (Hoskins et al., 2024). As the divide shifts, portions of the landscape are incrementally annexed by the expanding basin, while the neighboring basin progressively loses drainage area, particularly in its headwaters, without any direct re-routing of stream channels (e.g., Willett et al., 2014; Stokes et al., 2018; He et al., 2024; Hoskins et al., 2024).
The relative importance of drainage capture versus divide migration in driving drainage reorganization remains debated (Hoskins et al., 2024). From a biogeographic perspective, however, bottom-up and top-down river captures are far more relevant than divide migration for aquatic organisms, as only the former two involve shared connections between basins, which allow for faunal exchange. In contrast, divide migration typically does not preserve channel continuity, and thus does not enable passage for aquatic biota. Although identifying the specific mechanism responsible for a given instance of drainage reorganization can be challenging, biological evidence can offer valuable clues.
The presence of H. plumbellus in both the Guaporé and Juruena basins provides strong evidence for a historical headwater connection between these systems, indicating that one or more river capture events likely occurred. Faster erosion on one side of a drainage divide suggests that the divide migrates toward the side with slower erosion (Mudd, Furbish, 2005; He et al., 2024; Hoskins et al., 2024). In this region, the exposed escarpment indicates erosion is advancing from the lower-elevation Guaporé basin toward the higher-elevation Juruena basin. These observations support the hypothesis that bottom-up river captures account not only for the distribution of H. plumbellus across both basins but also for the shared occurrence of other tetra species (see Fig. 5).
The Fig. 5 shows that the northern segment of the western escarpment of the Chapada dos Parecis (left side of the image) is considerably more eroded, providing clearer geomorphological evidence of river capture from the rio Juruena by the rio Guaporé. In contrast, the southern segment remains comparatively intact. Species shared between the Guaporé and Juruena basins are more frequently recorded in the more eroded northern portion, reflecting this pattern of drainage reorganization. This suggests a long-term trend in which an increasing number of Juruena headwaters are progressively incorporated into the Guaporé basin. Consequently, the Guaporé may continue to capture sister populations from the Juruena, which over time could undergo genetic divergence and potentially speciation, generating additional biogeographic and evolutionary outcomes across the region.
The occurrence of H. plumbellus and other tetra species across multiple, unconnected river basins is not an unusual pattern. As discussed above, numerous studies have shown that drainage divides are not static but highly dynamic (Dahlquist et al., 2018; Scheingross et al., 2020; He et al., 2024). As a result, they are often poor predictors of freshwater fish distribution (Dagosta, de Pinna, 2017), since these boundaries are frequently breached by headwater capture events (Albert, Crampton, 2010; Albert, Reis, 2011; Lima, Ribeiro, 2011; Albert et al., 2020). Such events facilitate ichthyofaunal exchange between adjacent basins, resulting in hybrid drainages in both their species composition and biogeographic history (Dagosta, de Pinna, 2017). The rio Guaporé basin also fits this pattern: its ichthyofauna is of mixed origin, including several species that are exclusively shared with the rio Juruena basin (Fig. 5), among other sources such as the Amazonian lowlands (Ohara, 2018).
As discussed above, geological evidence strongly supports that the allopatric populations of H. plumbellus in the rio Guaporé and rio Juruena basins are sister lineages, separated by a vicariant event: capture of Juruena headwaters by the Guaporé drainage. As detailed in the “Intraspecific Variation” section, these populations exhibit differences expected from isolated groups, which diverge over time in the absence of gene flow. Determining whether they represent separate species requires careful consideration.
Debating the definition of a species, or precisely where species boundaries begin and end, falls outside the scope of this manuscript. Evolutionary processes give rise to a continuum of living organisms across space and time, and species represent segments of this continuum rather than universally discrete taxonomic entities. Nonetheless, it is well recognized that divergence times vary substantially among different lineages of sister taxa, as does the degree of phenotypic and genotypic differentiation observed. The diagrams in de Queiroz (2005, fig. 2; 2007, fig. 1) effectively illustrate this: in the early stages of divergence, populations may be indistinguishable, while over time they accumulate differences until a consensus emerges that they constitute separate species. Between these endpoints lies what de Queiroz termed the “gray zone”: a phase during which diverging lineages become increasingly differentiated but have not yet reached clear diagnosability. This gray zone appears to characterize the relationship between the H. plumbellus populations occurring in distinct drainage basins.
Although the populations show measurable differences, none are fixed or exclusive; traits vary in frequency or intensity rather than kind. No combination of traits unambiguously separates all individuals of one population from the other, failing to meet the criterion of the smallest diagnosable units (sensu Nelson, Platnick, 1981; Cracraft, 1983; Nixon, Wheeler, 1990). This weakens the case for recognizing them as distinct species. Nevertheless, the populations are on independent evolutionary trajectories, and the absence of fixed traits does not preclude eventual species-level divergence. The shared disjunct distribution pattern in other tetras suggests that the river capture event may be relatively recent, insufficient for complete speciation.
A further critical point concerns the potential impact of taxonomic inflation (Isaac et al., 2004). The recognition of geographically isolated populations as distinct species can lead to an artificial inflation of species counts and a distorted understanding of evolutionary processes. Delimiting species based solely on minor geographic variation risks overestimating biodiversity, a practice with far-reaching implications, especially for conservation. Conservation strategies are typically grounded in species-based frameworks, where the legal status of threatened species is formally defined and substantial funding is allocated by governments and international agencies. Consequently, species counts often serve as a foundation for conservation assessments and management plans (Myers et al., 2000).
Landscapes evolve over time, reshaping watersheds and altering drainage divides (Bishop, 1995; He et al., 2024; Hoskins et al., 2024). Fish are participants in this dynamic history, and their populations inevitably reflect these geological changes. Morphological variation will always exist to some degree. Over-splitting taxa based on minor morphological differences risks overemphasizing the terminal branches of the tree of life, highlighting distinctions that are virtually imperceptible without detailed examination. Morphological evidence alone already indicates population-level differences, but regardless of what genetic analyses may uncover, diagnosing both populations as distinct species in practice would remain challenging, as they lack conspicuous morphological traits that would justify such separation. That difficulty in consistently separating these populations based on discrete traits should guide our taxonomic judgment.
Comparative material examined. Hasemania negodagua, MZUSP 54589, 111, paratypes, 19.4–29.1 mm SL, rio Paraguaçu drainage, Bahia State. Hemigrammus silimoni, all from rio Tapajós basin, Mato Grosso State. MZUSP 93559, holotype, 29.7 mm SL; MZUSP 93560, 1 of 2, paratypes, 32.7 mm SL. MZUSP 115874, 21, 16.3–25.1 mm SL. Hyphessobrycon rutiliflavidus, MUSP 92438, 4, paratypes, 35.5–37.1 mm SL, rio Paraguai, Mato Grosso State.
Acknowledgments
The authors are grateful to several colleagues for their invaluable support and assistance during fieldwork, including Anderson Ferreira (UFGD), Bárbara Nagamatsu (UFGD), Diogo Hungria (GIA), Fabiola Vieira, João Filho, and ornamental fishers João Damasceno and Humberto Lenza. We also thank Ângela Araujo and Mariluce (UFRO), as well as Osvaldo Oyakawa and Michel Gianeti (MZUSP), for their curatorial assistance and for facilitating access to specimens.
References
Albert JS, Crampton WGR. The geography and ecology of diversification in Neotropical freshwaters. Nat Educ Knowl. 2010; 3(10):13.
Albert JS, Reis RE. Introduction to Neotropical Freshwaters. In: Albert JS, Reis RE, editors. Historical biogeography of Neotropical freshwater fishes. Los Angeles: University of California Press; 2011. p.3–19.
Albert JS, Tagliacollo VA, Dagosta F. Diversification of Neotropical freshwater fishes. Annu Rev Ecol Evol Syst. 2020; 51:27–53. https://doi.org/10.1146/annurev-ecolsys-011620-031032
Bishop P. Drainage rearrangement by river capture, beheading and diversion. Prog Phys Geogr. 1995; 19:449–73.
Camelier P, Dagosta FCP, Marinho MMF. New remarkable sexually dimorphic miniature species of Hyphessobrycon (Characiformes: Characidae) from the upper rio Tapajós basin. J Fish Biol. 2018; 92(4):1149–62. https://doi.org/10.1111/jfb.13732
Carvalho FR, Bertaco VA, Jerep FC. Hemigrammus tocantinsi: a new species from the upper rio Tocantins basin, Central Brazil (Characiformes: Characidae). Neotrop Ichthyol. 2010; 8(2):247–54. https://doi.org/10.1590/S1679-62252010000200006
Carvalho FR, Jesus GC, Langeani F. Redescription of Hyphessobrycon flammeus Myers, 1924 (Ostariophysi: Characidae), a threatened species from Brazil. Neotrop Ichthyol. 2014; 12(2):247–56. https://doi.org/10.1590/1982-0224-20130120
Cracraft J. Species concepts and speciation analysis. Curr Ornithol. 1983; 1:159–87.
Croizat L. Panbiogeography or an introductory synthesis of zoogeography, phytogeography, and zoology. With notes on evolution, systematics, ecology, anthropology, etc. Vol. 1 – The New World. Vol. 2 – The Old World. Caracas: Published by the author; 1958.
Croizat L, Nelson G, Rosen DE. Centers of origin and related concepts. Syst Zool. 1974; 23(2):265–87.
Dagosta FCP, Marinho MMF, Camelier P. A new species of Hyphessobrycon Durbin (Characiformes: Characidae) from the middle rio São Francisco and upper and middle rio Tocantins basins, Brazil, with comments on its biogeographic history. Neotrop Ichthyol. 2014; 12(2):365–75. https://doi.org/10.1590/1982022420130179
Dagosta FCP, Marinho MMF, Camelier P, Lima FCT. A new species of Hyphessobrycon (Characiformes: Characidae) from the upper rio Juruena basin, Central Brazil, with a redescription of H. cyanotaenia. Copeia. 2016; 104(1):250–59. https://doiorg/101643/CI-15-243
Dagosta FCP, de Pinna M. Biogeography of Amazonian fishes: deconstructing river basins as biogeographic units. Neotrop Ichthyol. 2017; 15(3):e170034. https://doi.org/10.1590/1982-0224-20170034
Dagosta FCP, de Pinna M. The fishes of the Amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 2019(431):1–163. https://doi.org/10.1206/0003-0090.431.1.1
Dagosta FCP, Seren TJ, Ferreira A, Marinho MMF. The emerald green tetra: a new restrictedrange Hyphessobrycon (Characiformes: Characidae) from the upper rio Juruena, Chapada dos Parecis, Brazil. Neotrop Ichthyol. 2022; 20(1):e210119. https://doi.org/10.1590/1982-0224-2021-0119
Dahlquist MP, West AJ, Li G. Landslide-driven drainage divide migration. Geology. 2018; 46(5):403–06. https://doi.org/10.1130/G39916.1
Durbin ML. Preliminary descriptions of new genera and species of tetragonopterid characins -Zoölogical Results of the Thayer Brazilian expedition. Bull Mus Comp Zool. 1908; 52(6):91–106.
Eigenmann CH. Preliminary descriptions of new genera and species of tetragonopterid characins – Zoölogical Results of the Thayer Brazilian expedition. Bull Mus Comp Zool. 1908; 52(6):91–106.
Eigenmann CH. The American Characidae [Part 1]. Mem Mus Comp Zool. 1917; 43(pt 1):1–102.
Eigenmann CH. The American Characidae [Part 2]. Mem Mus Comp Zool. 1918; 43:103–208.
Faria TC, Guimarães KLA, Rodrigues LRR, Oliveira C, Lima FCT. A new Hyphessobrycon (Characiformes: Characidae) of the Hyphessobrycon heterorhabdus species-group from the lower Amazon basin, Brazil. Neotrop Ichthyol. 2021; 19(1):e200102. https://doi.org/10.1590/1982-0224-2020-0102
Fink WL, Weitzman SH. The so-called cheirodontin fishes of Central America with description of two new species (Pisces: Characidae). Smithson Contrib Zool. 1974; 172:1–46.
Fricke R, Eschmeyer WN. Eschmeyer’s Catalog of Fishes: guide to fish collections [Internet]. San Francisco: California Academy of Science; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Green AJ, Lovas-Kiss A, Reynolds C, Sebastián-González E, Silva GG, Van Leeuwen CHA et al. Dispersal of aquatic and terrestrial organisms by waterbirds: a review of current knowledge and future priorities. Freshw Biol. 2023; 68(2):173–90. https://doi.org/10.1111/fwb.14038
He C, Braun J, Tang H, Yuan X, Acevedo-Trejos E, Ott RF et al. Drainage divide migration and implications for climate and biodiversity. Nat Rev Earth Environ. 2024; 5(3):177–92. https://doi.org/10.1038/s43017-023-00511-z
Hirsch PE, N’Guyen A, Muller R, Adrian-Kalchhauser I, Burkhardt-Holm P. Colonizing islands of water on dry land – on the passive dispersal of fish eggs by birds. Fish Fish. 2018; 19(3):502–10. https://doi.org/10.1111/faf.12270
Hoskins AM, Attal M, Mudd SM, Castillo M. Catchment-averaged erosion rates reveal signals of divide migration and drainage capture. JGR Earth Surf. 2024; 129(9):e2024JF007701. https://doi.org/10.1029/2024JF007701
International Union for Conservation of Nature (IUCN). Standards and petitions committee. Guidelines for using the IUCN Red List categories and criteria. Version 16 [Internet]. Gland; 2024. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Isaac NJB, Mallet J, Mace GM. Taxonomic inflation: its influence on macroecology and conservation. Trends Ecol Evol. 2004; 19(9):464–69. https://doi.org/10.1016/j.tree.2004.06.004
Javonillo R, Malabarba LR, Weitzman SH, Burns JR. Relationships among major lineages of characid fishes (Teleostei: Ostariophysi: Characiformes), based on molecular sequence data. Mol Phylogenet Evol. 2010; 54(2):498–511. https://doi.org/10.1016/j.ympev.2009.08.026
Lima FCT, Gerhard P. A new Hyphessobrycon (Characiformes: Characidae) from Chapada Diamantina, Bahia, Brazil, with notes on its natural history. Ichthyol Explor Freshw. 2001; 12(2):105–14.
Lima FCT, Ribeiro AC. Continental-scale tectonic controls of biogeography and ecology. In: Albert, JS, Reis RE, editors. Historical biogeography of Neotropical freshwater fishes. Los Angeles: University of California Press; 2011. p.145–64.
Lima FCT, Britski HA, Machado FA. A new Moenkhausia (Characiformes: Characidae) from central Brazil, with comments on the area relationship between the upper rio Tapajós and upper rio Paraguai systems. Aqua Int J Ichthyol. 2007; 13(2):45–54.
Lima FCT, Silva‑Oliveira C, Oliveira C, Faria TCF. A new Hyphessobrycon (Characiformes: Acestrorhamphidae) from the Central Amazon basin, Brazil. Pap Avulsos Zool. 2025; 65:e202565011. https://doi.org/10.11606/1807-0205/2025.65.011
Lovas-Kiss A, Antal L, Mozsár A, Nyeste K, Somogyi D, Kiss B et al. Bird-mediated endozoochory as a potential dispersal mechanism of bony fishes. Ecography. 2024; 2024(6):e07124. https://doi.org/10.1111/ecog.07124
Lovas-Kiss A, Vincze O, Löki V, Pallér-Kapusi F, Halasi-Kovács B, Kovács G et al. Experimental evidence of dispersal of invasive cyprinid eggs inside migratory waterfowl. PNAS. 2020; 117(27):15397–99. https://doi.org/10.1073/pnas.2004805117
Marinho MMF, Dagosta FCP, Camelier P, Oyakawa OT. A name for the ‘blueberry tetra’, an aquarium trade popular species of Hyphessobrycon Durbin (Characiformes: Characidae), with comments on fish species descriptions lacking accurate type locality. J Fish Biol. 2016; 89(1):510–21. https://doiorg/101111/jfb12991
Melo BF, Ota RP, Benine RC, Carvalho FR, Lima FCT, Mattox GMT et al. Phylogenomics of Characidae, a hyper-diverse Neotropical freshwater fish lineage, with a phylogenetic classification including four families (Teleostei: Characiformes). Zool J Linn Soc. 2024; 202(1):1–37. https://doi.org/10.1093/zoolinnean/zlae101
Mirande JM. Morphology, molecules and the phylogeny of Characidae (Teleostei, Characiformes). Cladistics. 2019; 35(3):282–300. https://doiorg/101111/cla12345
Moreira CR, Landim MI, Costa WJEM. Hyphessobrycon heliacus: a new characid fish (Ostariophysi: Characiformes) from the upper rio Tapajós basin, central Brazil. Copeia. 2002; (2):428–32. https://www.jstor.org/stable/1448057
Mudd SM, Furbish DJ. Lateral migration of hillcrests in response to channel incision in soil-mantled landscapes. J Geophys Res. 2005; 110:F04026. https://doi.org/10.1029/2005JF000313
Myers GS. The Amazon and its fishes. Part 2. The Fishes. Aquar J. 1947; 18(7):13–20.
Myers N, Mittermeier RA, Mittermeier CG, Fonseca GAB, Kent J. Biodiversity hotspots for conservation priorities. Nature. 2000; 403:853–58. https://doi.org/10.1038/35002501
Nelson G. From Candolle to Croizat: comments on the history of biogeography. J Hist Biol. 1978; 11:269–305.
Nelson G, Platnick NJ. Systematics and biogeography: cladistics and vicariance. New York: Columbia University Press; 1981.
Nielsen DTB, Ohara WM. A new restricted-range Melanorivulus (Cyprinodontiformes: Rivulidae) from the upper rio Guaporé, Chapada dos Parecis, Brazil. Zootaxa. 2024; 5496(3):332–42. https://doi.org/10.11646/zootaxa.5496.3.2
Nixon KC, Wheeler QD. An amplification of the phylogenetic species concept. Cladistics. 1990; 6(3):211–23. https://doi.org/10.1111/j.1096-0031.1990.tb00541.x
Ohara WM. Endemismo e análise biogeográfica dos peixes da bacia do rio Madeira. [PhD Thesis]. São Paulo: Universidade de São Paulo; 2018. Available from: https://teses.usp.br/teses/disponiveis/38/38131/tde-20052018-174432/publico/TeseoriginalWillianMassaharuOhara.pdf
Ohara WM, Lima FCT. Moenkhausia uirapuru, a new species from the upper rio Guaporé, Chapada dos Parecis, Mato Grosso, Brazil (Teleostei: Characidae). Ichthyol Explor Freshw. 2015; 26(2):159–70.
Ohara WM, Lima FCT, Barros BS. Hyphessobrycon petricolus, a new species of tetra (Characiformes: Characidae) from the rio Madeira basin, Mato Grosso, Brazil. Zootaxa. 2017; 4221(2):242–50. https://doi.org/10.11646/zootaxa.4221.2.8
Ohara WM, Teixeira TF, Albornoz-Garzón JG, Mirande JM, Lima FCT. Hyphessobrycon rheophilus, a new species from rapids of the Amazon and Orinoco river basins (Characiformes: Characidae: Stethaprioninae). Zootaxa. 2019; 4712(4):561–75. https://doi.org/10.11646/zootaxa.4712.4.5
Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Orti G et al. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol. 2011; 11:1–25. https://doi.org/10.1186/1471-2148-11-275
Pastana MNL, Dagosta FCP, Esguícero ALH. A new sexually dichromatic miniature Hyphessobrycon (Teleostei: Characiformes: Characidae) from the rio Formiga, upper rio Juruena basin, Mato Grosso, Brazil, with a review of sexual dichromatism in Characiformes. J Fish Biol. 2017; 91(5):1301–18. https://doi.org/10.1111/jfb.13449
Platnick NI. Concepts of dispersal in historical biogeography. Syst Zool. 1976; 25(4):294–95.
Prince PS, Spotila JA, Henika WS. Stream capture as driver of transient landscape evolution in a tectonically quiescent setting. Geology. 2011; 39(9):823–26. https://doi.org/10.1130/G32008.1
de Queiroz K. Ernst Mayr and the modern concept of species. PNAS. 2005; 102:6600–07. https://doi.org/10.1073/pnas.0502030102
de Queiroz K. Species concepts and species delimitation. Syst Biol. 2007; 56(6):879–86. https://doi.org/10.1080/10635150701701083
Rosen DE. A vicariance model of Caribbean biogeography. Syst Zool. 1976; 24(4):431–64.
Scheingross JS, Limaye AB, McCoy SW, Whittaker AC. The shaping of erosional landscapes by internal dynamics. Nat Rev Earth Environ. 2020; 1:661–76. https://doi.org/10.1038/s43017-020-0096-0
Shugar DH, Clague JJ, Best JL, Schoof C, Willis MJ, Copland L et al. River piracy and drainage basin reorganization led by climate-driven glacier retreat. Nat Geosci. 2017; 10(5):370–75. https://doi.org/10.1038/ngeo2932
Silva GG, Weber V, Green AJ, Hofmann P, Silva VS, Volcan MV et al. Killifish eggs can disperse via gut passage through waterfowl. Ecology. 2019; 100(11):e02778. https://doi.org/10.1002/ecy.2774
Stokes MF, Goldberg SL, Perron JT. Ongoing river capture in the Amazon. Geophys Res Lett. 2018; 45(11):5545–52. https://doi.org/10.1029/2018GL078129
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Toledo-Piza M, Baena EG, Dagosta FCP, Menezes NA, Ândrade M, Benine RC et al. Checklist of the species of the Order Characiformes (Teleostei: Ostariophysi). Neotrop Ichthyol. 2024; 22(1):1–548. https://doiorg/101590/1982-0224-2023-0086
Toledo-Piza M, Mattox GMT, Britz R. Priocharax nanus, a new miniature characid from the rio Negro, Amazon basin (Ostariophysi: Characiformes), with an updated list of miniature Neotropical freshwater fishes. Neotrop Ichthyol. 2014; 12(2):229–46. https://doi.org/10.1590/1982-0224-20130171
Vieira CS, Bartolette R, Brito MFG. Comparative morphology of bony hooks of the anal and pelvic fin in six neotropical characid fishes (Ostariophysi: Characiformes). Zool Anz. 2016; 260:57–62. https://doi.org/10.1016/j.jcz.2016.01.003
Weitzman SH, Vari RP. Miniaturization in South American freshwater fishes; an overview and discussion. Proc Biol Soc Wash. 1988; 101:444–65.
Wilkinson MJ, Marshall LG, Lundberg JG. River behavior on megafans and potential influences on diversification and distribution of aquatic organisms. J S Am Earth Sci. 2006; 21(1–2):151–72.
Willett SD, McCoy SW, Perron JT, Goren L, Chen CY. Dynamic reorganization of river basins. Science. 2014; 343(6175):1248765. https://doi.org/10.1126/science.1248765
Yao A, Mashiko M, Toquenaga Y. Passive dispersal potential of medaka eggs by attaching to waterbirds. Sci Nat. 2024; 111:53. https://doi.org/10.1007/s00114-024-01935-3
Authors
![]() Fernando Cesar Paiva Dagosta1,2,
Fernando Cesar Paiva Dagosta1,2, ![]() Willian Massaharu Ohara3 and
Willian Massaharu Ohara3 and ![]() Thomaz Jefrey Seren1,2
Thomaz Jefrey Seren1,2 ![]()
[1] Laboratório de Biogeografia e Sistemática de Peixes (LABISPE-FCBA), Universidade Federal da Grande Dourados, Rodovia Dourados/Itahum, km 12, 79804-970, Dourados, MS, Brazil. (FCPD) ferdagosta@gmail.com, (TJS) thomazjefrey@gmail.com (corresponding author).
[2] Museu da Biodiversidade (MUBIO), Universidade Federal da Grande Dourados, Rodovia Dourados/Itahum, km 12, 79804-970, Dourados, MS, Brazil.
[3] Departamento de Biologia, Universidade Federal do Amazonas, Av. Jauary Marinho, Setor Sul, 69077-000, Manaus, AM, Brazil. (WMO) ohara@ufam.edu.br.
Authors’ Contribution 

Fernando Cesar Paiva Dagosta: Conceptualization, Data curation, Formal analysis, Funding acquisition, Project administration, Writing-original draft, Writing-review and editing.
Willian Massaharu Ohara: Conceptualization, Data curation, Investigation, Methodology, Project administration, Writing-original draft, Writing-review and editing.
Thomaz Jefrey Seren: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Ethical Statement
Specimens were collected under IBAMA collection permits 83/2012 (May 2012 – September 2013) and 60634/2018.
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
AI statement
The authors used AI-assisted technology (ChatGPT 4 – OpenAI) for grammatical revision.
Funding
Part of the material analyzed in this study were obtained during the project “Monitoramento e Conservação da Ictiofauna do rio Madeira,” a collaborative initiative carried out between 2009–2012 by the Universidade Federal de Rondônia, Instituto de Estudos e Pesquisas do Agronegócio e Organizações Sustentáveis, and Santo Antônio Energia. Additional specimens were collected as part of the project “Miniature Characiformes (Teleostei: Ostariophysi): evolution, diversity, and biogeography,” funded by FAPESP (grant #2017/09321–5). This paper was supported by INCT–Peixes, funded by MCTIC/CNPq (proc. 405706/2022–7), and by CNPq (proc. 401459/2025-0). FCPD is supported by CNPq (proc. 304864/2024-2).
How to cite this article
Dagosta FCP, Ohara WM, Seren TJ. One species, two basins: a new Hyphessobrycon (Characiformes: Acestrorhamphidae) from the upper rio Juruena and rio Guaporé basins, Mato Grosso, Brazil. Neotrop Ichthyol. 2026; 24(1):e250125. https://doi.org/10.1590/1982-0224-2025-0125
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted October 11, 2025
Accepted October 11, 2025
![]() Submitted July 13, 2025
Submitted July 13, 2025
![]() Epub April 11, 2025
Epub April 11, 2025