![]() Marcelle Fernanda de Oliveira Barbosa1,2,
Marcelle Fernanda de Oliveira Barbosa1,2, ![]() Raquel Santos dos Santos3,
Raquel Santos dos Santos3, ![]() Felipe Fernando Silva Siqueira4,
Felipe Fernando Silva Siqueira4, ![]() Eduardo Silva do Nascimento Albuquerque2,
Eduardo Silva do Nascimento Albuquerque2, ![]() Natalia Rodrigues da Silva2,
Natalia Rodrigues da Silva2, ![]() Sara Ferreira Pedrosa2,
Sara Ferreira Pedrosa2, ![]() Paulo de Souza Jesus2,
Paulo de Souza Jesus2, ![]() Otávio Antunes Lacerda1,2,
Otávio Antunes Lacerda1,2, ![]() Marcelo Ândrade5,
Marcelo Ândrade5, ![]() Maria Auxiliadora Pantoja Ferreira6,
Maria Auxiliadora Pantoja Ferreira6, ![]() Caio Maximino7 and
Caio Maximino7 and ![]() Diógenes Henrique Siqueira-Silva1,2
Diógenes Henrique Siqueira-Silva1,2 ![]()
PDF: Download Here | Cite this article
Associate Editor: ![]() Francisco Araújo
Francisco Araújo
Section Editor: ![]() Fernando Pelicice
Fernando Pelicice
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
A reprodução de peixes em rios amazônicos pode ser afetada pela antropização ambiental e mudanças no regime hidrológico. Thoracocharax stellatus é uma espécie ecologicamente importante, porém seu ciclo reprodutivo ainda é pouco compreendido. Seus padrões reprodutivos foram avaliados na Amazônia Oriental por quatro estações hidrológicas em áreas com diferentes níveis de impacto ambiental. Realizaram-se amostragens mensais entre fevereiro de 2023 e janeiro de 2024, em duas áreas: uma com baixo impacto (LI) e outra com alto impacto ambiental (HI). 232 espécimes foram coletados. Análises biométricas e histológicas determinaram o ciclo reprodutivo e fases de maturação gonadal (Imaturo, Em Desenvolvimento, Aptos à Reprodução, Aptos à desova, Regressão, Regeneração). Thorachocarax stellatus apresenta desova parcelada sincronizada com a estação chuvosa, com pico durante a cheia. Fêmeas na área de baixo impacto amadureceram em tamanhos maiores, com maior investimento reprodutivo. No ambiente de alto impacto apresentaram maturação precoce e menor atividade reprodutiva, sugerindo que a degradação ambiental afeta negativamente a reprodução. O IGS teve pico durante a cheia, e a Análise de Redundância destacou o pH, temperatura e sazonalidade como os principais fatores que influenciam a reprodução. A integridade ambiental é crucial para a reprodução de T. stellatus, e a degradação do habitat, intensificada pelo aquecimento global, ameaça a sustentabilidade populacional. Estratégias de conservação são necessárias para mitigar impactos antropogênicos nos ecossistemas aquáticos amazônicos.
Palavras-chave: Ciclo reprodutivo, Influência ambiental, Peixe de água doce, Sazonalidade, Variação hidrológica.
Introduction
Studies on the reproductive biology of teleost fishes have shown a wide variety of life cycle strategies, influenced by adaptations to environmental factors such as temperature, photoperiod, rainfall, and flood pulses (Agostinho et al., 2004; Suzuki, 2004; Alvarenga et al., 2006; Guerrero et al., 2009; Godinho et al., 2010; Bayley et al., 2018). Because of this, understanding the adaptive mechanisms of a population can offer valuable insights into how the species interacts with its habitat, as well as help develop effective measures for conserving and managing natural stocks (Nikolsky, 1969; Gomes et al., 2025).
Farias et al. (2025) demonstrated that the spawning period of the splash tetra Copella arnoldi is synchronized with the rainy season. This strategy ensures that the eggs deposited in plant sheets outside the water do not dehydrate. Another Amazonian tetra fish, Astyanax bimaculatus, exhibits only two reproductive phases throughout its entire reproductive cycle: Developing and Spawning Capable. As soon as the rainfall indices increase, the fish rapidly transition to the Spawning Capable phase, ensuring reproductive success in an unpredictable environment characterized by extended periods of drought (Cordeiro et al., 2019). Queiroz et al. (2010) also reported that the reproductive activity of Red-bellied Piranha (Pygocentrus nattereri) is closely linked to increased rainfall, presenting two reproductive peaks in the flooded Amazon forests of the Mamirauá Reserve. This pattern contrasts with other studies that reported only a single reproductive peak for the species.
Those are only a few examples of the fish’s adaptability to the conditions in the Amazon rivers and streams. The Amazon hosts a high diversity of freshwater fish species, with approximately 2,406 species, representing 15% of all described freshwater species worldwide, of which 1,402 are endemic to this basin (Jézéquel et al., 2020). Among them are members of the Gasteropelecidae family, which differ from other Characiformes by having a smaller head relative to the rest of the body and a well-developed keel-shaped abdomen, composed of well-developed muscles and bones (Netto-Ferreira et al., 2007). These fish are divided into the genera Gasteropelecus, Carnegiella, and Thoracocharax, with the latter comprising two species: Thoracocharax securis De Filippi, 1853, and Thoracocharax stellatus Kner, 1858 (Toledo-Piza et al., 2024).
The spotfin hatchetfish, Thoracocharax stellatus, is commercialized as an ornamental fish (Ladislau et al., 2020) and is widely distributed in Latin America (Toledo-Piza et al., 2024). It is a small species, reaching up to 6 cm in length as an adult (Weitzman and Palmer, 2003). It has a silver coloration with a dark spot on the first rays of the dorsal fin, in addition to a small pelvic fin and a well-developed anal fin, distinguishing it from other members of its family (Lourenço da Silva et al., 2009; Abe et al., 2013; Toledo-Piza et al., 2024). It is defined by Rauber et al. (2021) as a sedentary species with no significant migratory reproductive behavior. Its diet consists primarily of live insects from riparian forests (Netto-Ferreira et al., 2007). Therefore, the availability of food resources in riparian forest areas is crucial for its reproductive activities. The forest removal and reduction of river connectivity to these areas could make the species susceptible to population imbalances (Netto-Ferreira et al., 2007).
The Amazon forest has lost 11,590 km2 of its area due to wildfires, which reached their highest peak in decades in the last triennium (2022–2024). Additionally, irregular activity involving the suppression of native forests for human occupation and pasture expansion has occurred in this area (INPE, 2024). Sensitivity is much higher in areas near rivers (Alves et al., 2021) due to the decreased connectivity with riparian forests, which in turn affects the availability of shelter and food for aquatic organisms (Leite et al.,2015), ultimately impacting fish reproductive activity in these areas (Juen et al., 2016).
Studies on the reproductive biology of Amazonian fish species are a key factor for understanding how environmental and biological factors influence fish reproduction. In the case of T. stellatus, although the presence of a ZZ/ZW chromosomal sex-determination system has been reported (Carvalho et al., 2002), its reproductive biology has not yet been fully described in the literature. Addressing this knowledge gap is essential to support management and conservation strategies and to contribute to the development of public policies aimed at protecting areas sensitive to anthropogenic pressures (Mylonas et al., 2010; Pelicice et al., 2021).
Therefore, the present study aimed to investigate the regulatory mechanisms underlying the reproduction of T. stellatus in the Tauarizinho River, Western Amazon. To achieve this, we analyzed key reproductive aspects, including the gonadosomatic index (GSI) and hepatosomatic index (HSI), length-weight relationship, size at first sexual maturation, and reproductive cycle, as well as their interactions with environmental conditions. The research was conducted in two areas of the river that differ in their levels of environmental integrity, allowing a comparative assessment of how habitat quality influences reproductive activity.
Material and methods
Voucher specimens. Voucher specimens of Thoracocharax stellatus (GEA 13000) were deposited in the Laboratório de Ictiologia do Grupo de Ecologia Aquática (GEA), Universidade Federal do Pará (UFPA), Pará State, Brazil.
Study area. Belonging to the Tocantins River basin, the Tauarizinho is a 103.45 km long River, flowing through the southeastern region of Pará, with its source in the Sororó Indigenous Land, flowing out in the Tocantins River, in Marabá, Pará State. Its hydrological regime follows the seasonal cycle of the Tocantins-Araguaia basin. The River level ranges from 2.59 m to 3.71 m, with an average of 3.21 m, reflecting periods of flood and drought. Its water temperature varies between 25°C and 26°C, while the average air temperature in the region is 27°C. It has a semi-humid climate, with an average annual rainfall of 1,900 mm. The measured physicochemical parameters indicate good oxygenation (9.5–10.5 ppm) and slightly acidic pH (6.4–6.7). Toxic ammonia concentrations remain stable (0.25 ppm), while nitrite increases during the dry and rising-water seasons (up to 1.0 ppm), suggesting the influence of decomposition processes and anthropogenic pressures. Tauarizinho River features sinuous stretches, straight channels, and abrupt course changes, which create different aquatic microhabitats. Its margins, in natural areas, maintain riparian vegetation and stability, whereas in impacted stretches, they are unstable, narrow, and show signs of erosion.
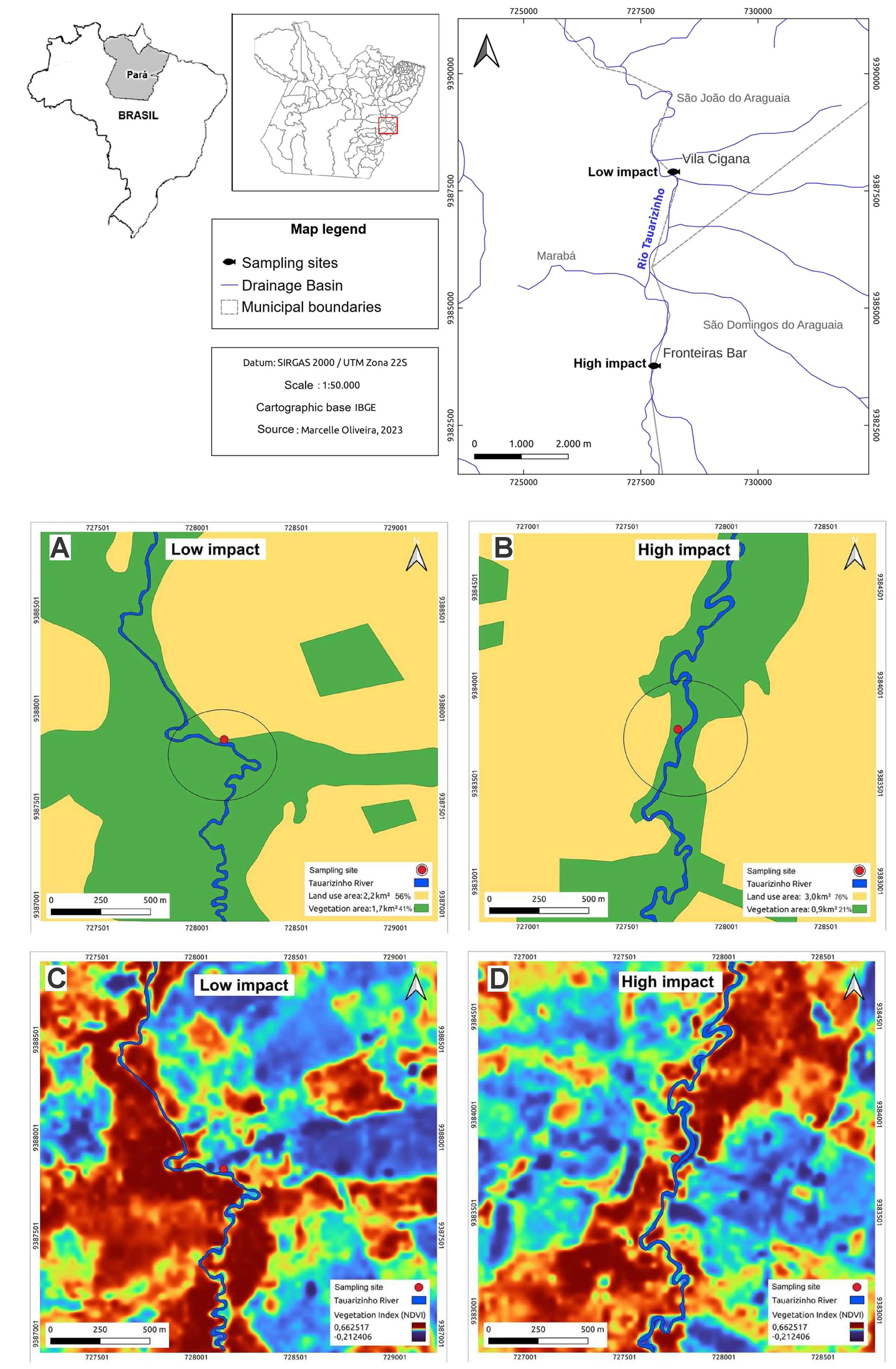
The Tauarizinho River faces varying degrees of intense anthropogenic pressures, such as the expansion of livestock farming and the disorderly occupation of its banks along its entire length (Costa et al., 2023). Two distinct sampling points were chosen for this study: one in the city of São João do Araguaia, Pará (05°32’02.3”S 48°56’24.2”W) and the other in another city, São Domingos do Araguaia, Pará (05°34’17.1”S 48°56’37.1”W) (Fig. 1).
FIGURE 1| Map of fish collection points in the municipalities of São Domingos do Araguaia and São João do Araguaia, State of Pará, Brazil. The collection sites are located along the Tauarizinho River, classified according to the environmental impact levels: Low Impact (LI) and High Impact (HI). Subfigures illustrate: Land use and land cover in the study areas for A. LI; B. HI; C. RGB vegetation index maps for LI; and D. HI areas. Source: IBGE Cartographic Base. Prepared by: MFOB.
The areas were selected based on the Rapid River Assessment Protocol (PARR), adapted from Bersot et al. (2015) and Silva, Nascimento (2017), which evaluates rivers and streams based on physical characteristics and the level of environmental impact resulting from anthropogenic actions. This protocol consists of 22 questions, assigning values from 0 to 5 points to parameters related to soil erosion levels, vegetation cover, the availability of aquatic plants along the banks, water flow characteristics, and the type of substrate on the riverbed (S1). A score between 0 and 40 represents areas considered to have “high impact”, while 41 to 60 indicates areas with “moderate impact”, and 61 to 100 represents areas with “low impact”.
According to the PARR, the area in São João do Araguaia was classified as an area with low environmental impact (LI) (75 points), with greater vegetation cover along the riverbanks. The relative bank stability contributes to the natural maintenance of flow and water quality characteristics, with lower anthropogenic impact and higher ecological carrying capacity. The total vegetation in this region amounts to 1.7 km2 (Figs. 1A, C). The area located in the São Domingos do Araguaia region was classified as an area with high environmental impact (HI) with a score of 34 points, showing significant erosion along the riverbanks, sedimentation in the riverbed, the absence of riparian vegetation and aquatic plants, with the total vegetation in this area recorded as 0.9 km2 (Figs. 1B, D).
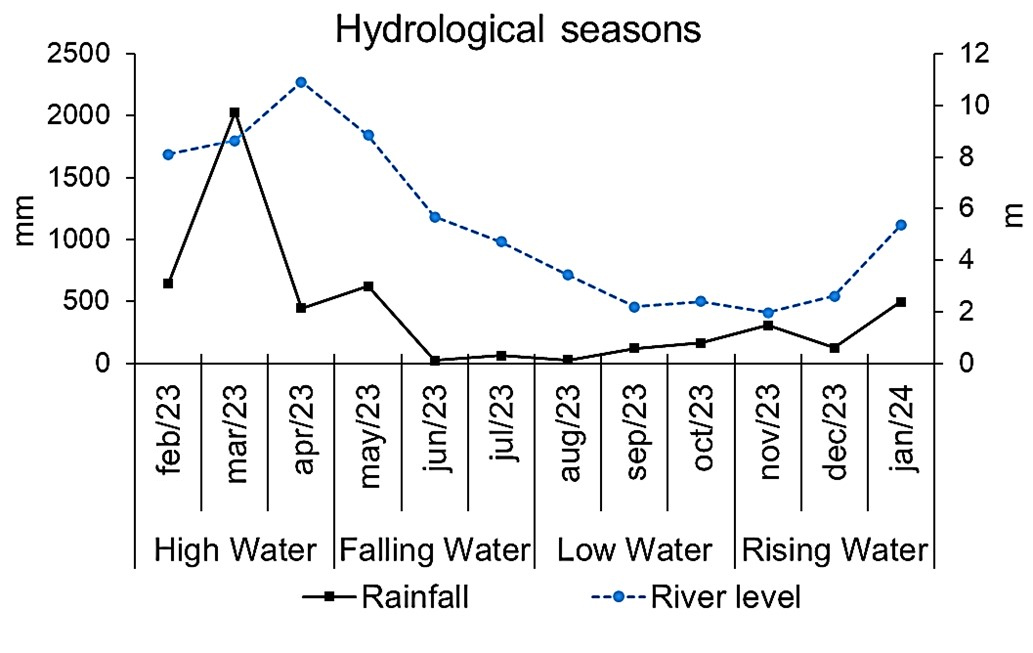
Hydrological seasons and physical and chemical parameters of water. The hydrological seasons for the Tocantins-Araguaia river basin, which includes the Tauarizinho River, were defined based on fluviometric data obtained from hydrological bulletins available on the platform of the Secretaria Estadual do Meio Ambiente e Sustentabilidade of Pará (Semas/PA), in collaboration with the Agência Nacional das Águas (ANA), whilst pluviometry data (monthly rainfall accumulation) were obtained from the platform of the Centro Nacional de Monitoramento e Alerta de Desastres Naturais (CEMADEN). Four hydrological seasons were recorded: High water (February to April); Falling water (May to July); Low water (August to October); and Rising water (November to January). Data are summarized in Fig. 2 and Tab. 1.
FIGURE 2| Average water level (in meters) of the Tauarizinho River and accumulated rainfall (in millimeters) recorded monthly across hydrological seasons. Sources: ANA (2024) and CEMADEN (2024).
TABLE 1 | Average data for rainfall, river levels, and physical and chemical water parameters of the Tauarizinho River, located within the Tocantins-Araguaia Hydrographic Basin, across the various hydrological seasons (2023–2024). The data include temperature, pH, dissolved oxygen, and toxic ammonia, differentiated by Low Impact (LI) and High Impact (HI) areas. Sources: ANA (2024) and CEMADEN (2024). Asterisks indicate statistically significant values.
Hydrological seasons | ||||||||
| High water | Falling water | Low water | Rising water | ||||
River level (m) | 9.2 | 6.4 | 2.7 | 3.3 | ||||
Rainfall (mm) | 104 | 237 | 105 | 310 | ||||
Area | LI | HI | LI | HI | LI | HI | LI | HI |
Temperature (°C) | 24±0.1 | 25±0.05 | 24±0.2 | 25±0.2 | 25±0.2* | 26±0.1* | 25±0.1 | 26±0.1 |
pH | 6.5±0.01 | 6.3±0.03 | 6.5±0.1 | 6.8 ±0.2 | 7±0.1 | 6.3±0.1 | 7.2±0.2* | 6.2±0.2* |
Dissoved Oxigen (ppm) | 10±0.02 | 11±0.03 | 8±0.2 | 11 ±0.02 | 11±0.1* | 8±0.3* | 11±0,1 | 9±0.1 |
Toxic ammonia (ppm) | 0.2±0.2 | 0.1±0.1 | 0.1±0.03 | 0.1 ±0.2 | 0.12±0.1 | 0.4±0.5 | 0.2±0.3 | 0.2±0.2 |
Animal collection. Animal collections were conducted once a month, always in the morning, from February 2023 to January 2024. Thoracocharax stellatus specimens (Fig. 3) were captured using five drift nets measuring 10 m x 1 m with a mesh size of 1 mm, specifically designed for capturing small-sized fish. The nets were cast upstream, near the vegetation along the banks, with a 5-hour sampling time on average. The captured specimens were placed in 30-liter plastic bags with aerators and transported to the Laboratory of Neurosciences and Behavior (LANEC/Unifesspa).
FIGURE 3| Spotfin hatchetfish fish: Thoracocharax stellatus. Scale bar = 1 cm.
Histological characterization. In the laboratory, the specimens were separated, identified, and euthanized in a container with 600 mL of water and a lethal dose of Eugenol solution (20 mL of Eugenol in 180 mL of 100% ethanol, BIODINÂMICA). Subsequently, biometric data (weight (g) and total length (cm)) were recorded. A ventrolateral incision was made, and the gonads and liver were removed, weighed for the calculation of the Gonadosomatic Index (GSI) and Hepatosomatic Index (HSI), and fixed in glutaraldehyde solution (2.5% glutaraldehyde in bibasic phosphate buffer, pH 7.2) for a minimum period of 24 hours. The fixed samples were dehydrated through successive passages in ethanol solutions of increasing concentrations (70%, 80%, 95%, and 100%). They were then embedded in Historesin. The material was sectioned at 3 μm using a manual microtome (LUPETEC, MRP, 2015) equipped with a steel blade. Histological slides were stained with Hematoxylin and Eosin, analyzed under an optical microscope (Nikon Eclipse Ei), and a smartphone’s camera (Samsung A23/ Android 12.0. Resolutions 50 MP) mounted on a universal smartphone adapter attached to the microscope lens (Siqueira-Silva et al., 2025)
Data analysis. The variation in the mean GSI and HSI across the hydrological seasons was determined for females and males using the following equations: GSI = Wg/Wt100 (Wg = gonad weight; Wt = total fish weight) and HSI = Wl/Wt100 (Wl = liver weight; Wt = total fish weight). The definition of the testicular maturation phases of T. stellatus followed Siqueira-Silva et al. (2013). The ovarian maturation phases and oogenesis analysis were defined according to Quagio-Grassiotto et al. (2011) and Heins, Brown‐Peterson (2022). The species’ reproductive cycle was determined based on the relative frequency (%) of the distribution of the gonadal maturation phases. Data were organized into contingency tables for each hydrological season and maturation phase. Subsequently, Fisher’s test was performed to assess possible significant differences (p>0.05) in the distribution of gonadal maturation phases between areas with low and high environmental impact.
The sex ratio was calculated using the G-test, which is recommended for small sample sizes. The number of males (Om) and females (Of) was recorded, resulting in a total sample size of N = Om + Of. Assuming an expected 1:1 sex ratio, the expected frequencies for males (Em) and females (Ef) were calculated as Em = Ef = N / 2. The G statistic was computed using the formula:
G = 2 [Om x In(Om/Em) + Of x In (Ef/of)]
The characterization of reproductive biology and the length-weight relationship for females and males in both areas was determined using the potential equation: Wt = φLsθ, where Wt represents weight and Ls represents total length. In this equation, φ is the condition factor coefficient, and θ is the angular or growth coefficient, which determines the type of species growth based on the following reference values: θ > 3 indicates positive allometric growth; θ < 3 indicates negative allometric growth; and θ = 3 indicates isometry (Dikou, 2023). To visualize the size distribution patterns between sexes across hydrological seasons, histograms were constructed for both body weight (g) and total length (cm). Data were grouped by sex (male and female) and stratified by four hydrological seasons. Density curves were superimposed to illustrate distribution profiles.
The size of the first sexual maturation (L50) was determined for males and females, considering juveniles and adults and their total length. Sexual maturation was established based on gonadal maturation phases (Immature, In Development, Able to Reproduce (for males), Spawning capable (for females), Regression, Regeneration), grouped into length classes (mm) with 20 mm intervals, using a logistic function to calculate the size at first sexual maturation: P = 1/(1 + exp [- r (L – L50)]). Where P is the proportion of adults (or sexually mature individuals) at a given total length, L is the total length (TL) of the individual, L50 is the length at which 50% of individuals are sexually mature (the inflection point of the logistic curve) and r is the slope parameter, indicating the rate of transition from juvenile to adult; higher values of r reflect a more abrupt transition.
Relative fecundity, defined as the number of oocytes released by females per spawning event, was estimated in seven sexually mature females. Gonads were removed and weighed, after which the medial region was dissected and weighed separately. Vitellogenic oocytes from this region were isolated and placed in a Petri dish for counting under a stereomicroscope. The resulting value was used to calculate relative fecundity using the formula AF = (NO × Wmr) / O, where NO is the number of counted oocytes, Wmr is the weight of the medial gonadal region (g), and O is the total ovary weight, as described by Silva et al. (2023).
Statistical analyses. Data were processed for homogenization and outlier removal after verifying data normality using the Shapiro-Wilk test and homoscedasticity using Levene’s test. Data that did not exhibit normal distribution were subjected to the Kruskal-Wallis test to identify differences among groups across hydrological seasons. For normally distributed data, a two-way ANOVA was performed, followed by Bonferroni’s test for multiple comparison adjustments. The analyses were conducted using R (R-Studio, v. 2024.04.02), with a significance level set at p<0.05.
To assess the influence of environmental variables on reproductive indices (GSI and HSI) of males and females, Redundancy Analysis (RDA) was applied, as indicated by DCA testing for the linear nature of the data. Model significance, axes, and explanatory variables were tested using PERMANOVA (adonis2) with 999 permutations, allowing the comparison of effects between areas with different environmental impacts and across hydrological seasons.
Results
Specimen collection. A total of 232 specimens of T. stellatus were collected, comprising 127 males and 105 females. The sex ratio was 1:1 (p > 0.05). Between the areas, the proportion of males was higher in the low-impacted area, while the proportion of females was higher in the high-impacted area, during the flood season (p < 0.05) (Tab. 2).
TABLE 2 | The number of male and female Thoracocharax stellatus individuals collected across hydrological seasons in areas with low (LI) and high (HI) environmental impact. Asterisks indicate statistically significant values.
Hydrological seasons | ||||||
| High water | Falling water | Low water | Rising water | Total | |
LI | Male | 6 | 6 | 25 | 37 | 74 |
Female | 7 | 11 | 18 | 21 | 57 | |
G-test | 0.07 | 1.4 | 1.1 | 4.4 | 2.2 | |
p valor | >0.05 | >0.05 | >0.05 | <0.05* | >0.05 | |
HI | Male | 8 | 5 | 23 | 17 | 53 |
Female | 2 | 10 | 23 | 13 | 48 | |
G-test | 3.8* | 1.6 | 0 | 0.5 | 0.2 | |
p valor | <0.05* | >0.05 | >0.05 | >0.05 | >0.05 | |
Thoracocharax stellatus exhibited sexual dimorphism in size, with females being larger than males (p > 0.05). The total length and weight of females ranged from 3.0 to 6.9 cm (mean: 4.4 ± 0.72 cm) and from 0.1 to 6.4 g (mean: 1.4 ± 1.17 g), respectively. For males, total length ranged from 2.9 to 5.1 cm (mean: 4.4 ± 0.66 cm) and weight from 0.3 to 4.2 g (mean: 1.43 ± 0.72 g).
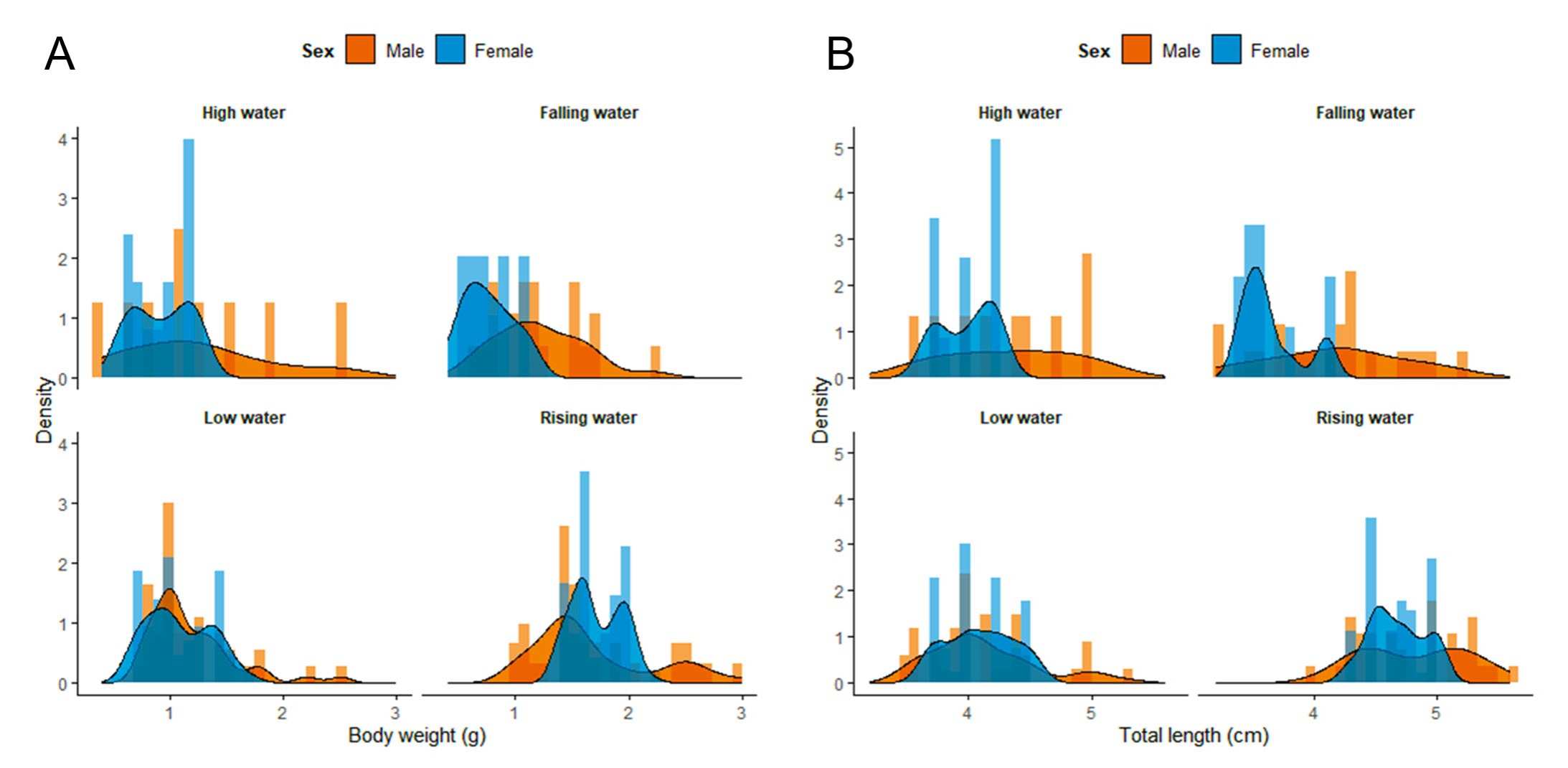
Significant differences between males and females of T. stellatus were observed in two of the four hydrological seasons. During the falling water season, females showed significantly greater total length and body weight (p = 0.004 and p = 0.006, respectively). In the rising water season, a significant difference was also observed in total length (p = 0.04), with females being larger. In the other seasons, no statistically significant differences were detected between sexes (Tab. 3). The frequency distribution of body length and weight for both sexes is presented in the density histograms (Figs. 4A, B), which illustrate the variation and overlap in size structure between males and females.
TABLE 3 | Average length (cm) and weight (g), standard deviation (SD), and p-values of male and female Thoracocharax stellatus individuals collected across hydrological seasons in areas with low (LI) and high (HI) environmental impact.
Length (cm) | |||||
Hydrological seasons | Male | SD | Female | SD | p-value |
High water | 0.9 | 0.3 | 1.3 | 0.65 | 0.1 |
Falling water | 0.7 | 0.21 | 1.1 | 0.35 | 0.004 |
Low water | 1.0 | 0.23 | 1.1 | 0.37 | 0.39 |
Rising water | 1.1 | 0.28 | 1.6 | 0.53 | 0.04 |
Weight (g) | |||||
| Male | SD | Female | SD | p-value |
High water | 3.6 | 0.2 | 4.3 | 0.51 | 0.1 |
Falling water | 3.6 | 0.5 | 4.1 | 0.53 | 0.006 |
Low water | 4.1 | 0.3 | 4.1 | 0.44 | 0.2 |
Rising water | 4.6 | 0.2 | 4.8 | 0.43 | 0.2 |
FIGURE 4| Density plots of total body weight (A) and length (B) for males and females of Thoracocharax stellatus across all hydrological seasons, highlighting sex-related differences through distribution patterns.
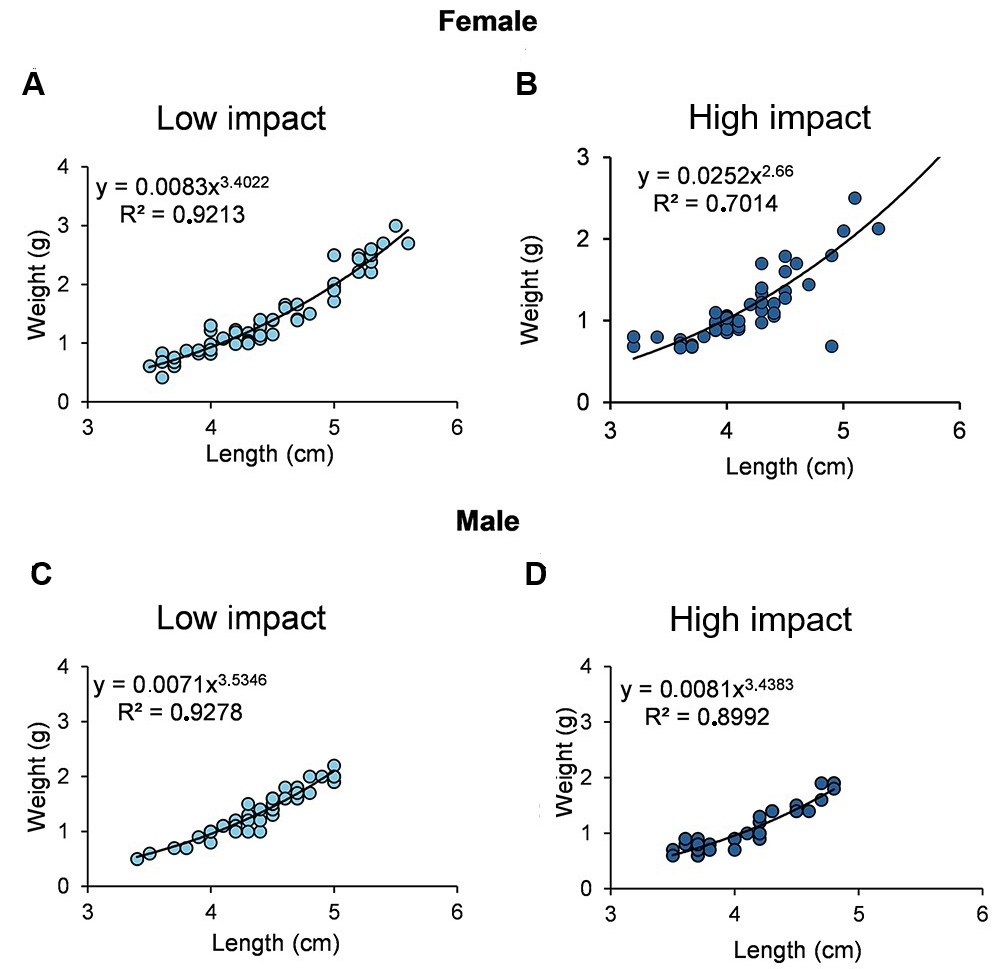
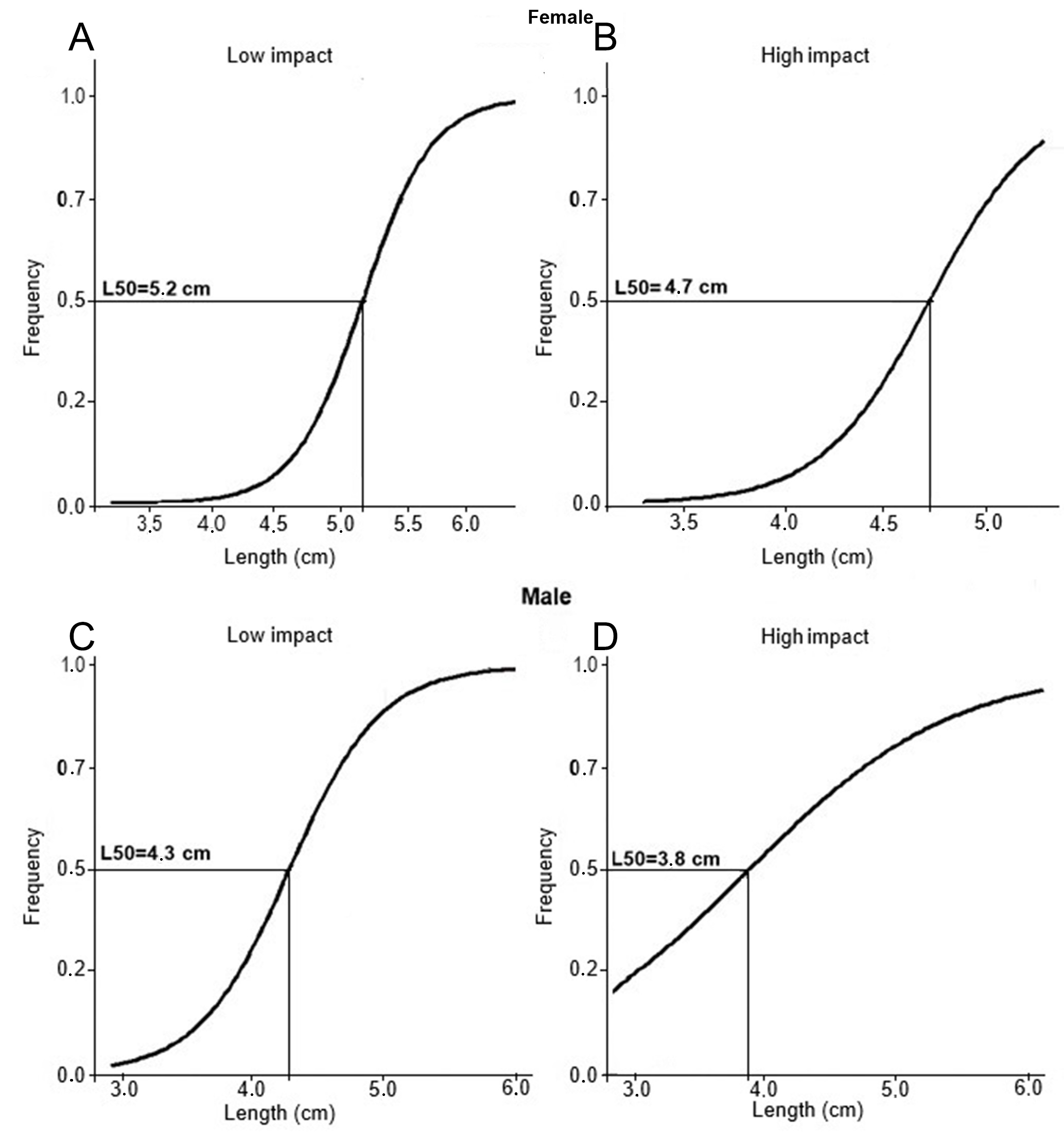
In the LI area, female growth was positively allometric (θ = 3.4; Fig. 5A), whereas in the HI area, it was negatively allometric (θ = 2.66; Fig. 5B). For males, growth was positively allometric in both areas (LI: θ = 3.5; HI: θ = 3.4; Figs. 5C, D). The size at first sexual maturation (L50) was larger in the LI area, with 5.2 ± 0.43 cm for females (Fig. 6A) and 4.3 ± 0.56 cm for males (Fig. 6C), whereas in the HI area, it was 4.7 ± 0.41 cm for females (Fig. 6B) and 3.8 ± 0.52 cm for males (Fig. 6D).
FIGURE 5| Length-weight relationship for females (A-B) and males (C-D) of Thoracocharax stellatus in the two study areas.
FIGURE 6| Size at first sexual maturation: Logistic curve and frequency of sexually mature animals by length and size class at first sexual maturation (L50) of Thoracocharax stellatus, by study area. A-B. Females; C-D. Males.
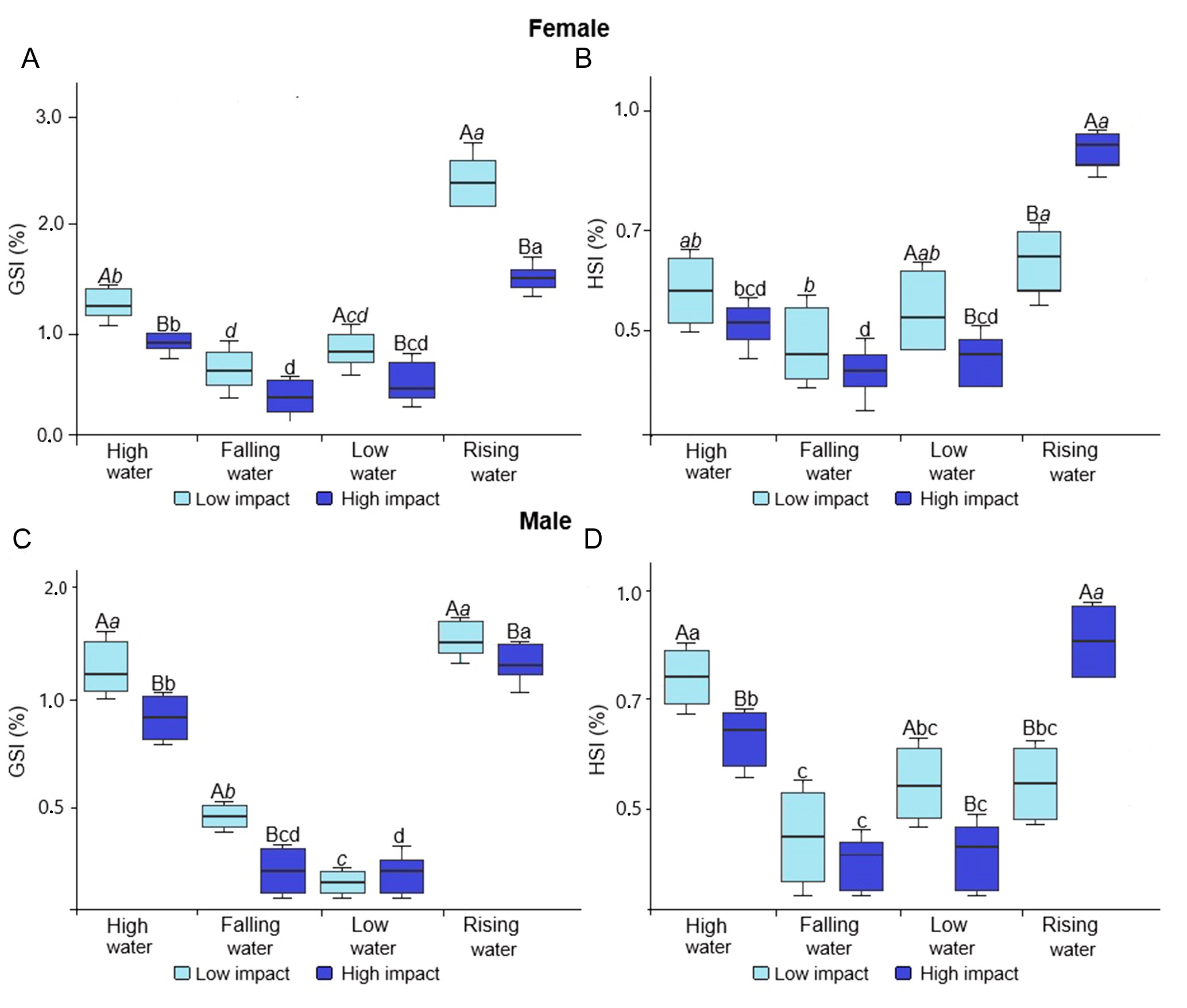
The GSI of T. stellatus females was higher in the LI area across all seasons compared to the HI area and varied significantly throughout the hydrological seasons (p > 0.05), with the highest values recorded during high water and the lowest during falling water (Fig. 7A). The HSI differed between areas during rising water (p < 0.05), being higher in the HI area and lower in the LI area (Fig. 7B). During the low water season, the highest HSI values were recorded in the LI area, and the lowest value in the HI area.
For males, GSI was higher in the LI area during the high water, falling water, and rising water seasons (p < 0.05; Fig. 7C). HSI also showed significant differences during high water (p < 0.05), with the highest values coinciding with the reproductive period (Fig. 7D).
FIGURE 7| Mean Gonadosomatic Index (GSI) and Hepatosomatic Index (HSI) of Thoracocharax stellatus across the hydrological seasons in Low Impact and High Impact areas. A-B. Females; C-D. Males. Lowercase letters indicate statistical differences between hydrological seasons; uppercase letters indicate differences between areas (p > 0.05)
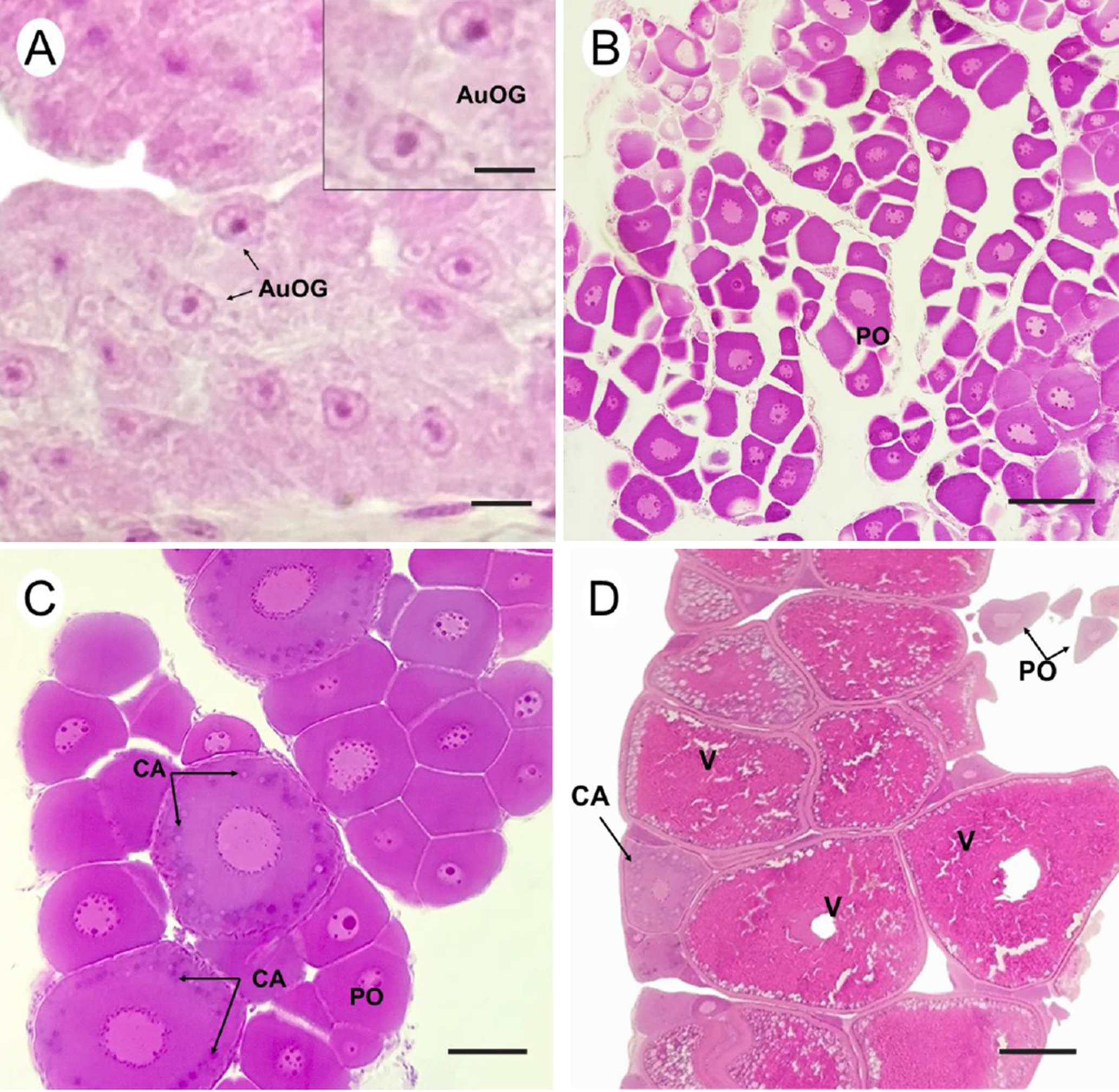
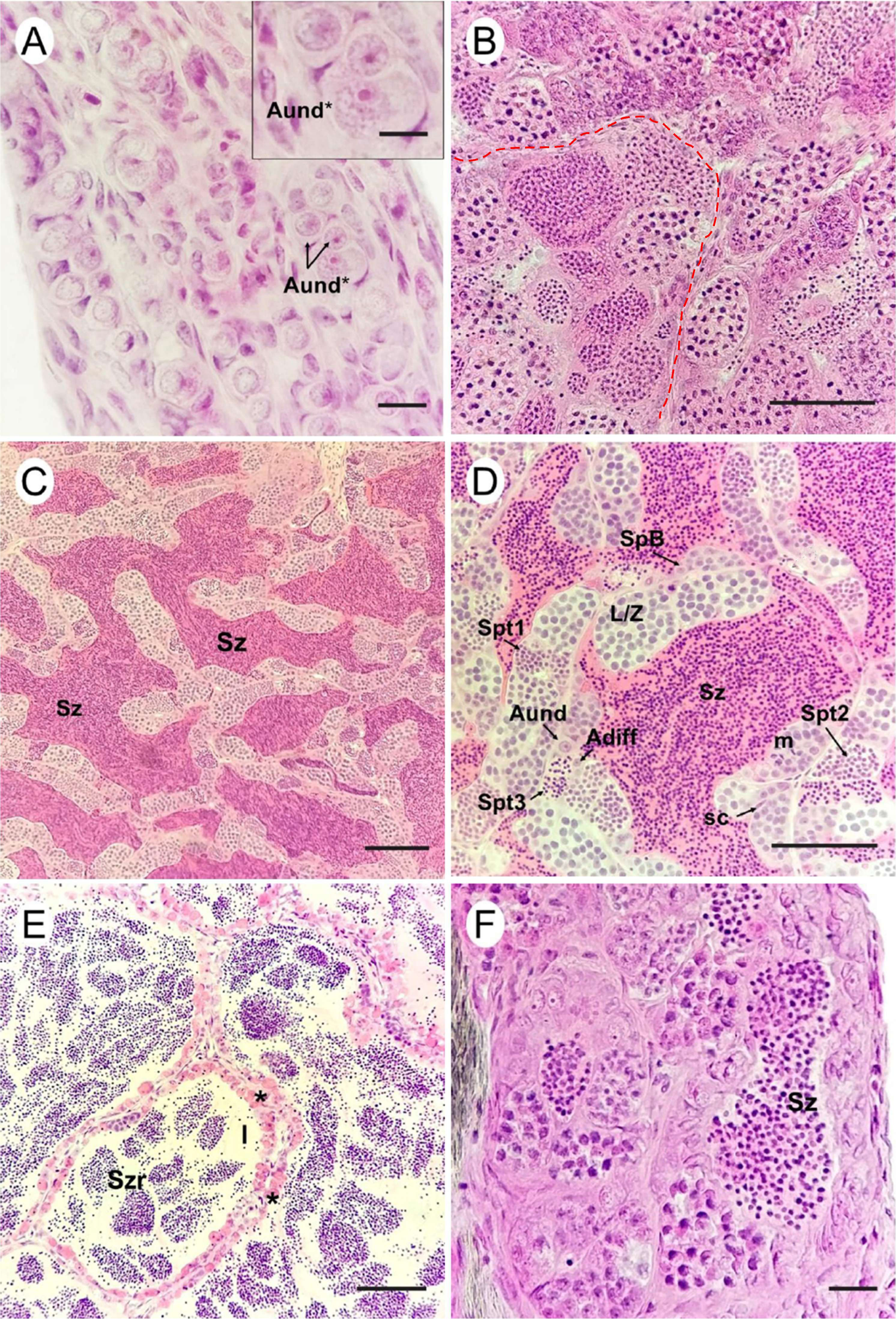
Histological characterization. The gonads of T. stellatus are paired, elongated organs located in the coelomic cavity, parallel to the digestive system organs (Fig. 8). Absolute fecundity ranged from 421 to 6,052 oocytes, with a mean of 2,024.5 ± 2,056.5 oocytes. Relative fecundity values ranged from 12.23 to 33.21 oocytes/g, with a mean of 19.74 ± 7.62 oocytes/g. The mean diameter of mature oocytes was 0.7 ± 0.3 mm, ranging from 0.6 to 0.9 mm. Three gonadal maturation phases were observed in females (Fig. 9; Tab. 4), while five gonadal maturation phases were identified in males (Fig. 10; Tab. 5) of T. stellatus.
FIGURE 8| Thoracocharax stellatus gonads: A. Detailed view of ovaries (g), alongside digestive system organs (sd). up = urogenital papilla; B. Ovaries; C. Testes.
FIGURE 9| Ovary maturation in Thoracocharax stellatus. A. Immature female: presence of undifferentiated type-A oogonia (AuOG); B-C. In Development: Perinucleolar oocytes, exhibiting basophilic nucleoli (PO), cortical alveolus oocytes (CA); D. Spawning Capable: presence of vitellogenic oocytes, perinucleolar oocytes, and cortical alveolus oocytes at the periphery of the ovary. Scale bars: A. 20 µm, B–D. 50 µm. Staining: Hematoxylin and eosin.
TABLE 4 | Phases of ovarian maturation in Thoracocharax stellatus.
Phases | Description |
Immature | The ovaries are small, translucent, and filamentous, with the presence of oogonia (AuOG) (Fig. 9A). |
In Development | Ovaries contain perinucleolar oocytes attached to the lamellar epithelium, with basophilic cytoplasm and nucleoli positioned at the periphery of the nucleus (Fig. 9B). As this phase progresses, cortical alveoli are deposited at the periphery of the oocytes (Fig. 9C), gradually progressing to secondary growth stages. |
Spawning Capable | Distinct presence of vitellogenic oocytes. Presence of perinucleolar oocytes and cortical alveoli at the periphery of the ovary (Fig. 9D). |
FIGURE 10| Testes maturation in Thoracocharax stellatus. A. Immature. Presence of undifferentiated spermatogonia (Aund*); B. In Development: increased number of spermatocytes, continuous epithelium along seminiferous tubules (dashed line); C-D. Able to Reproduce: lumen of the tubules dilated, filled with spermatozoa; E. Regression: dilated lumen of the tubules containing residual spermatozoa; F. Regeneration: intense spermatogonia proliferation. Lumen in some regions containing few residual spermatozoa. Aund* – A undifferentiated spermatogonia *; Aund – A undifferentiated spermatogonia. Adiff – differentiated spermatogonia type A; SpB – Type B spermatogonia; Sc – Sertoli cell; L/Z – spermatocyte, leptotene zygotene; St1 – initial spermatid cysts; St2 – intermediate spermatid cysts; St3 – final spermatid cysts; Sz – spermatozoa; Szr – residual spermatozoa; m – metaphase; l – lumen; (*) – melanomacrophage centers. Scale bars: A. 20 µm, B–F. 50 µm. Staining: Hematoxylin and eosin.
TABLE 5 | Phases of testicular maturation in Thoracocharax stellatus. Aund* = undifferentiated spermatogonia.
Phases | Description |
Immature | The immature testis is filamentous and transparent, showing only spermatogonia (Aund* and Aund) cells, which are precursors of spermatogenesis (Fig. 10A). |
In Development | The testes show continuous germinal epithelium. Through mitosis, undifferentiated spermatogonia (Aund*) and (Aund) differentiate and proliferate into differentiated spermatogonia types A (Adiff) and B. As the phase progresses, spermatogenic cysts are observed along the germinal epithelium, with cells at various stages. There is a gradual increase in the lumen and a small amount of seminal fluid containing spermatozoa (Fig. 10B). |
Able to Reproduce | In this phase, the testes appear milky and more voluminous. This phase is marked by the advanced discontinuity of the germinal epithelium, with cysts rupturing after the differentiation of germ cells into spermatozoa, releasing a large volume of spermatozoa into the lumen (Figs. 10C, D). |
Regression | In this phase, the testes appear flaccid and reduced in size. The germinal epithelium is completely discontinuous, with residual spermatozoa in the lumen. Presence of Sertoli cells and the emergence of melanomacrophage clusters (Fig. 10E). |
Regeneration | This phase is characterized by the proliferation of germ cell cysts in all stages. There is a significant reduction in testicular size, and residual spermatozoa are occasionally observed in the lumen, now more discreet (Fig. 10F). |
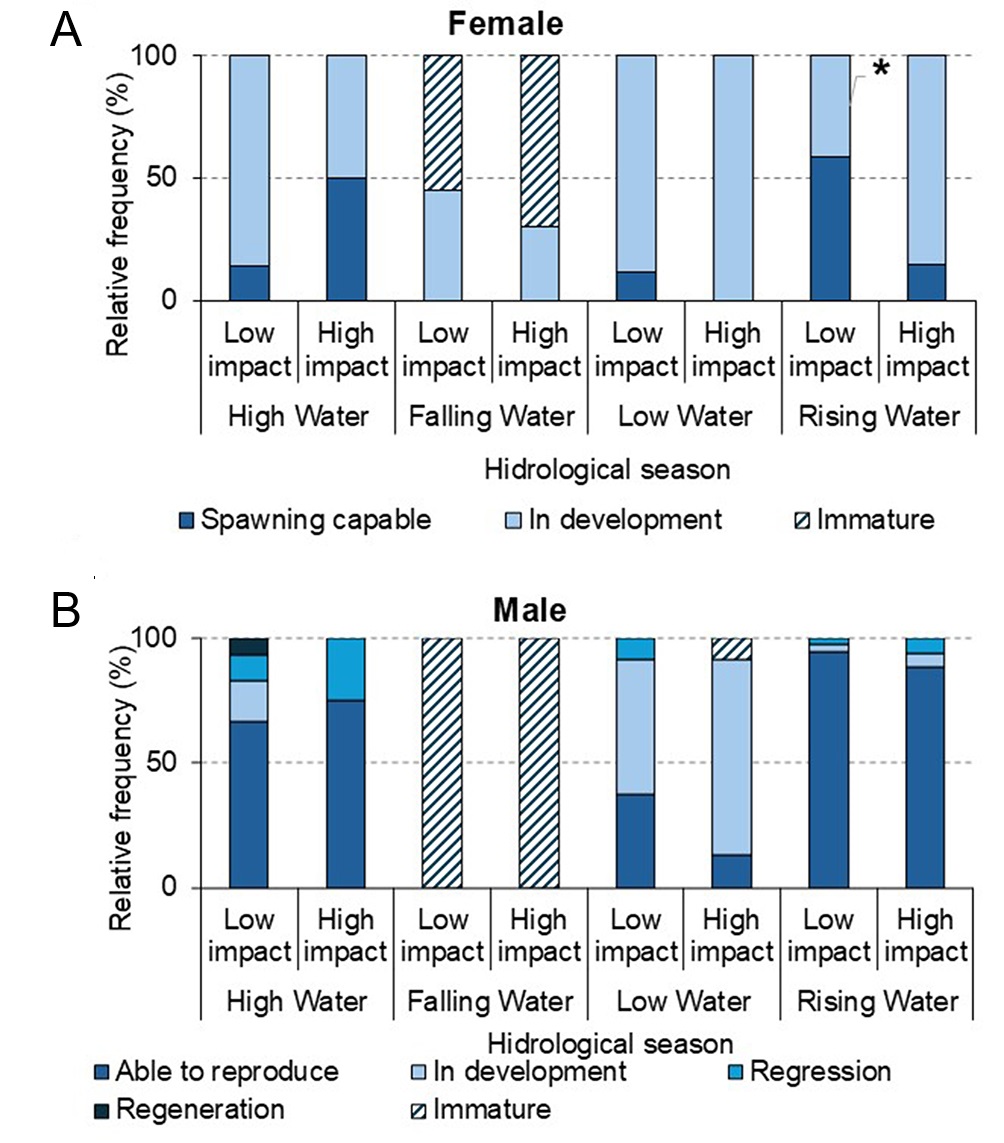
The frequencies of T. stellatus gonadal maturation phases (Fig. 11) varied across hydrological seasons and impact levels. During the high water season, the frequency of Spawning Capable females was higher in the HI area (50%) compared to the LI area (14%). In the falling water season, most females were Immature in both areas (LI: 55%; HI: 70%), while in the low water season, females In Development predominated in both areas (LI: 88%; HI: 100%). The greatest difference occurred during the rising-water season, with 59% of Spawning Capable females in the LI area and 15% in the HI area (p < 0.05; odds ratio = 7.47).
FIGURE 11| Relative frequency of gonadal maturation phases in Low Impacted and High Impacted areas across hydrological seasons of Thoracocharax stellatus. A. Females; B. Males. The asterisk indicates statistically significant differences between areas (p > 0.05).
In males, during the high water season, the Able to Reproduce phase predominated (67%), followed by the In Development (17%), Regression (10%), and Regeneration (7%) phases in the LI area. In contrast, in the HI area, most individuals were in the In Development phase (75%), with a low frequency of Able to Reproduce individuals (25%). During the falling water season, all males were Immature in both areas. In the low water season, the In Development phase was the most frequent (54%), followed by Able to Reproduce (38%) and Regression (8%) in the LI area. During the low water season, in the HI area, 9% of males were Immature, 13% were Able to Reproduce, and 78% were In Development.
During the rising-water season, the LI area recorded a higher frequency of Able to Reproduce males (94%), along with 3% In Development and 3% Regression. In the HI area, the frequency of Able to Reproduce individuals was 88%, followed by 6% In Development and 6% in Regression. The frequencies of reproductive phases, associated with the GSI and HSI values of T. stellatus demonstrated that the species spawns from flood waters to the High waters period, during the entire rainy season (Fig. 11).
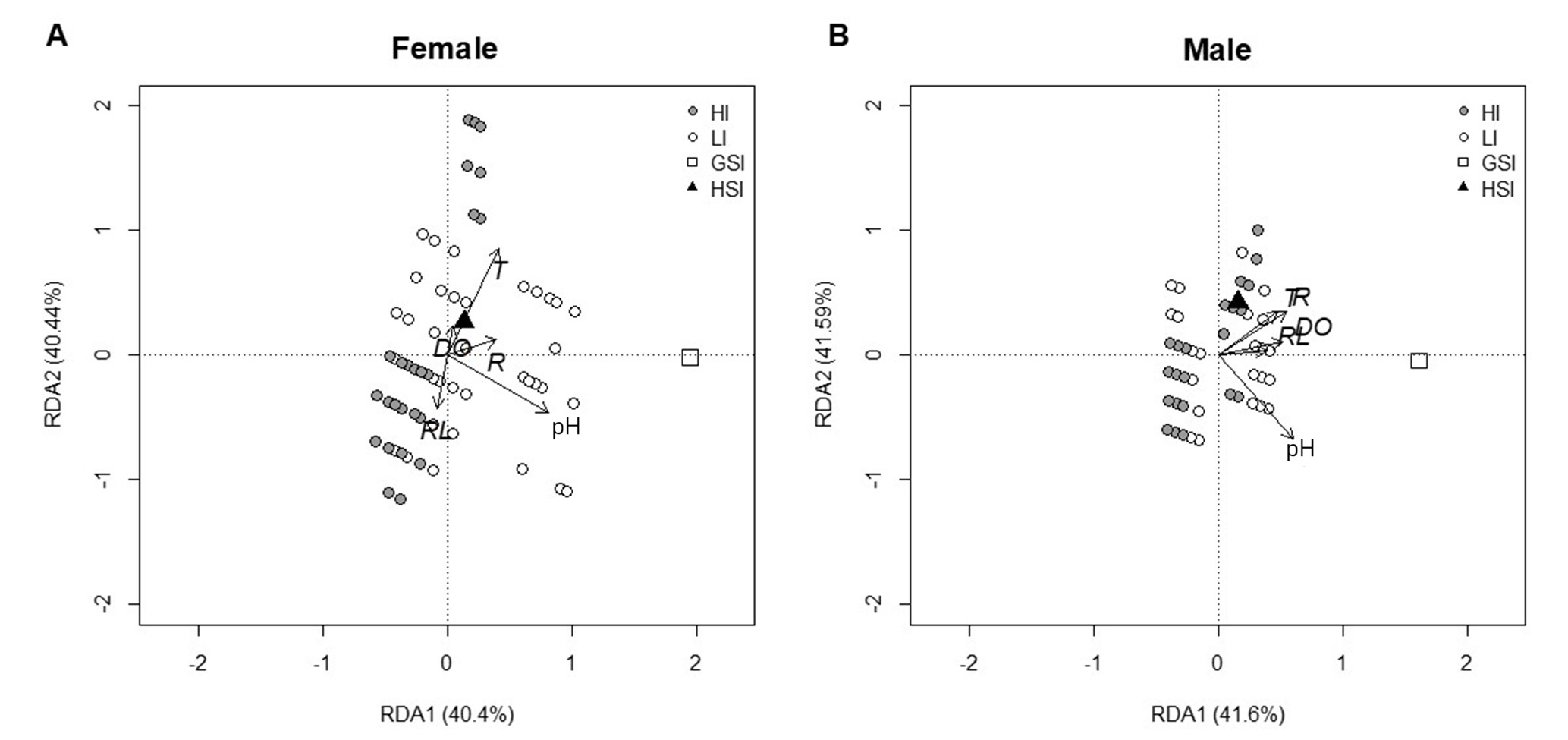
Redundancy analysis (RDA). Redundancy analysis (RDA) indicated that environmental factors explained approximately 41% of the variation in female reproductive indices (GSI and HSI), with the global model being statistically significant (Pr(>F) = 0.001). The first RDA axis accounted for 40.4% of the variation, while the second axis contributed 0.78%. Among the environmental variables evaluated, pH had the greatest influence on reproductive activity and hepatic condition, followed by temperature and rainfall, whereas dissolved oxygen and river level had no relevant effect. For males, environmental factors explained approximately 44.5% of the variation in reproductive indices, with global significance (Pr(>F) = 0.001), the first axis accounting for 41.6% and the second for 2.9% of the variation.
The most influential environmental variables for males were temperature, pH, dissolved oxygen, and rainfall, while river level had a less pronounced effect. Area-specific evaluation showed that individuals from the high environmental impact area (HI) had lower GSI and HSI values compared to those from the low impact area (LI), indicating reduced gonadal maturation and hepatic reserves, with differences more pronounced in females. These results demonstrate that both reproductive activity and hepatic condition were modulated by environmental conditions and local impact level, with the strongest effects observed in females (Fig. 12).
FIGURE 12| Redundancy Analysis (RDA) of the biological composition of females (A) and males (B) in relation to environmental variables. Points represent study areas: gray circles (HI) and white circles (LI). Response variables are indicated by symbols: white square (GSI) and black triangle (HSI). Arrows indicate the direction and strength of the association between environmental variables and biological composition. RDA1 and RDA2 axes show the percentage of variance explained by each component.
Discussion
The present study demonstrated how the life of T. stellatus is influenced by environmental conditions, which differed between a preserved area and a region under ecological impact. These differences directly affected the reproduction, and growth of T. stellatus, revealing adaptive strategies developed by the species in response to local challenges.
During the rising-water season, a predominance of males was observed in the low-impact area. This population composition may maximize reproductive success by increasing male competition for females (Emlen, Oring, 1977). Similar dynamics have been observed in Poecilia reticulata, where male interference has been found to favor reproductive success in male-biased populations (Jirotkul, 1999; Chechi et al., 2022), as well as in other Characiforms (Nunes et al., 2015; Maskill et al., 2017). In the high water season, the predominance of males in the high impacted area shows that environmental factors and local selectivity affect sex-differentiated survival (Baroiller et al., 2009), possibly due to the greater sensitivity of males to stressors such as pH, temperature, and predation (Guillante et al., 2023; Ashrafi et al., 2024).
Growth also reflected these environmental differences. Females showed positive allometric growth in the low-impacted area (θ = 3.4) and negative allometric growth in the high-impacted area (θ = 2.66), while males maintained positive allometric growth in both environments. These variations in the weight–length relationship are associated with seasonality, resource availability, and habitat quality (Freitas et al., 2017; Prestes et al., 2019).
Regarding sexual dimorphism, females were larger than males, a pattern consistent with other Characiforms (Martins-Queiroz et al., 2008; Silva et al., 2023). The significant difference in weight and length between the sexes in the falling water season can be explained by the captured males. Since all of them were immature, they are supposed to be lighter and smaller than females. On the other hand, significantly bigger females in the rising water season must be related to their fecundity, since larger females usually produce more oocytes (Domínguez-Petit et al., 2022), and at this time, more than 50% females were spawning capable. The variation in body size between areas, with larger and better-conditioned females in the low-impact site, suggests that more favorable environmental conditions, with greater resource availability, are essential for growth and reproductive development. For the species, this provides insights into fecundity. Franssen (2011) corroborates this idea, observing that habitat alterations caused by human activities can induce rapid changes in the morphology of Cyprinella lutrensis, including variations in body size.
Environmental differences also affected sexual maturity and reproductive frequency. In the high-impacted area, individuals matured earlier and at smaller sizes, a strategy that ensures reproduction under adverse conditions and limited resources (Keller et al., 2021). In addition, we found evidence of a lower frequency of reproductively active females, suggesting they prioritized survival over reproduction (Haque et al., 2019). In preserved areas, however, fish matured at larger sizes and exhibited a higher frequency of reproductively active females, showing greater investment in reproduction (Oliveira et al., 2020). Similar strategies have been observed in other Amazonian species, such as Triportheus trifurcatus (Martins-Queiroz et al., 2008) and Peckoltia oligospila (Molica et al., 2024).
Reproductive indices (GSI and HSI) also showed a strong relationship with environmental variables, including rainfall, temperature, pH, and dissolved oxygen. Redundancy analysis (RDA) revealed that in the low-impacted area, natural conditions favored reproduction, while in the high-impacted area, reproduction was negatively affected. Even small changes in these parameters may compromise reproductive performance (Gurgel et al., 2012; Waddell, Crampton, 2020). It is worth noting that during the falling-water season, the connectivity between the river and riparian vegetation is reduced, limiting the availability of shelter and food, and generating stressors for organisms (Röpke et al., 2022). This can reduce female fecundity and reflects the “capital breeders” theory, in which investment in current reproduction compromises future reproduction (Wright et al., 2017).
In impacted environments, individuals allocate more energy to physiological maintenance and foraging, showing compensatory responses to adverse conditions (Araújo et al., 2019; López-Rodríguez et al., 2021). In preserved areas, by contrast, GSI increases during the flood season, when environmental cues stimulate reproduction, and the availability of microhabitats and adequate resources for spawning and early juvenile development increases (McBride et al., 2015). The differences in reproductive activity of T. stellatus between areas reinforce that anthropogenic disturbances not only affect local environmental quality but also amplify the effects of natural hydrological variability (Costa et al., 2023).
Deforestation, another relevant factor in the studied areas, harms aquatic ecosystems by eliminating riparian vegetation, which is essential for water quality and thermal regulation (Jung et al., 2020). The removal of vegetation accelerates bank erosion and increases sedimentation in water bodies, directly affecting spawning habitats and impairing egg and larval development (Pusey, Arthington, 2003; Mello et al., 2018). It also reduces shading over the river, which may raise water temperatures (Fantin-Cruz et al., 2011). In species such as T. stellatus, which maintain a close association with riparian vegetation, deforestation compromises the availability and quality of essential reproductive resources, potentially leading to local population declines.
This study highlights the critical role of environmental conditions in the reproduction and growth of aquatic species, emphasizing the need for conservation and management strategies to mitigate habitat degradation and preserve the health of T. stellatus populations, as well as other freshwater fish species in the region.
References
Abe KT, Mariguela TC, Avelino GS, Castro RMC, Oliveira C. Multilocus molecular phylogeny of Gasteropelecidae (Ostariophysi: Characiformes) reveals the existence of an unsuspected diversity. Mol Phylogenet Evol. 2013; 69(3):1209–14. https://doi.org/10.1016/j.ympev.2013.07.005
Agostinho AA, Gomes LC, Veríssimo S, Okada EK.Flood regime, dam regulation and fish in the Upper Paraná River: effects on assemblage attributes, reproduction and recruitment. Rev Fish Biol Fish. 2004; 14:11–19. https://doi.org/10.1007/s11160-004-3551-y
Alvarenga ER, Bazzoli N, Santos GB, Rizzo E. Reproductive biology and feeding of Curimatella lepidura (Eigenmann & Eigenmann) (Pisces, Curimatidae) in Juramento reservoir, Minas Gerais, Brazil. Rev Bras Zool. 2006; 23(2):314–22. https://doi.org/10.1590/S0101-81752006000200002
Alves JC, Andreotti GF, Agostinho AA, Gomes LC. Effects of the El Niño Southern Oscillation (ENSO) on fish assemblages in a Neotropical floodplain. Hydrobiologia. 2021; 848(8):1811–23. https://doi.org/10.1007/s10750-021-04555-4
Agência Nacional de Águas e Saneamento Básico (ANA). Boletins hidrológicos. [Internet]. Brasília; 2024. Available from: https://www.gov.br/ana/pt-br
Araújo FG, Nascimento AA, Gomes ID, Sales A, Oliveira BAC. Gonadal development and reproductive period of the characin Astyanax aff. bimaculatus (characiformes: Characidae) in a tropical reservoir in southeastern Brazil. Zoologia. 2019; 36:1–14. https://doi.org/10.3897/zoologia.36.e30610
Ashrafi R, Westermark A, Leppänen MT, Vehniäinen ER. Female-biased sex ratios and delayed puberty in two fish species with different Ecologies in an Anthropogenically affected urban lake. Environ Res. 2024. 262:119844. https://10.1016/j.envres.2024.119844
Bayley PB, Castello L, Batista VS, Fabré NN. Response of Prochilodus nigricans to flood pulse variation in the central Amazon. R Soc Open Sci. 2018; 5(6):172232. http://doi.org/10.1098/rsos.172232
Baroiller JF, D’Cotta H, Saillant E. Environmental effects on fish sex determination and differentiation. Sex Dev. 2009; 3(2–3):118–35. https://doi.org/10.1159/000223077
Bersot MROB, Menezes JM, Andrade SF. Application protocol rapid assessment of rivers (RAP) River Basin Imbé – RJ. Ambiência. 2015; 11(2):277–94. https://doi.org/10.5935/ambiencia.2015.02.01
Carvalho ML, Oliveira C, Foresti F. Description of a ZZ/ZW sex chromosome system in Thoracocharax cf. stellatus (Teleostei, Characiformes, Gasteropelecidae). Genet Mol Biol. 2002; 25(3):299–303. https://doi.org/10.1590/S1415-47572002000300008
Centro Nacional de Monitoramento e Alerta de Desastres Naturais (CEMADEN). Dados pluviométricos: acumulado mensal de chuva. [Internet]. São José dos Campos; 2024. Available from: http://www.cemaden.gov.br
Chechi TS, Narasimhan A, Biswas B, Prasad NG. Male mating success evolves in response to increased levels of male-male competition. Evolution. 2022; 76:1638–51. https://doi.org/10.1111/evo.14501
Cordeiro JG, Rodrigues J, Rodrigues MS, Bertolini RM, Nóbrega RH, Yasui GS et al. Reproductive cycle of the tetra Astyanax bimaculatus (Characiformes: Characidae) collected in Amazonian streams. Zygote. 2019; 28(1):37–44. https://doi.org/10.1017/S0967199419000601
Costa IMC, Knoechelmann CM, Siqueira FFS. Effect of habitat quality on the biodiversity of ant genera and functional groups in a riparian forest area of the Tauarizinho River in Eastern Amazonia. Res Soc Dev. 2023; 12(3):e19712340636. https://doi.org/10.33448/rsd-v12i3.40636
Dikou A. Weight-length relationship in fish populations reflects environmental regulation on growth. Hydrobiologia. 2023; 850(2):335–46. https://doi.org/10.1007/s10750-022-05072-8
Domínguez-Petit R, García-Fernández C, Leonarduzzi E, Rodrigues K, Macchi GJ. Parental effects and reproductive potential of fish and marine invertebrates: cross-generational impact of environmental experiences. Fishes. 2022; 7(4):188. https://doi.org/10.3390/fishes7040188
Emlen ST, Oring LW. Ecology, sexual selection, and the evolution of mating systems. Science. 1977; 197(4300):215–23. https://doi.org/10.1126/science.327542
Farias RR, López-Rodríguez NC, Gonçalves LAB, Tavares CNSC, Rocha RM, Freitas TMS et al. Reproductive biology of Copella arnoldi (Characiformes: Lebiasinidae), a terrestrial-spawning fish from the Amazon. Neotrop Ichthyol. 2025; 23(1):e240054. https://doi.org/10.1590/1982-0224-2024-0054
Fantin-Cruz I, Tondato KK, Motta-Marques D. Regime térmico em águas correntes e sua importância na estrutura do habitat e na biologia de organismos aquáticos. Caminhos Geogr. 2011; 11(36):295–307. https://doi.org/10.14393/rcg113616291
Franssen NR. Anthropogenic habitat alteration induces rapid morphological divergence in a native stream fish. Evol Appl. 2011; 4(6):791–804. https://10.1111/j.1752-4571.2011.00200.x
Freitas TMS, Souza JBS, Prudente BS, Montag LFA. Length-weight relationship in ten species from the Nhamundá River, the Amazon basin, Brazil. Acta Amazon. 2017; 47(1):75–78. https://doi.org/10.1590/1809-4392201601272
Godinho AL, Lamas IR, Godinho HP. Reproductive ecology of Brazilian freshwater fishes. Environ Biol Fish. 2010; 87:143–62. https://doi.org/10.1007/s10641-009-9574-4
Gomes MCF, Charvet P, Pinto LM, Gurgel-Lourenço RC, Ramos TPA, Lima SMQ et al. Conservation status of the freshwater fish species from Ceará State, Brazil. Neotrop Ichthyol. 2025; 23(2):e240084. https://doi.org/10.1590/1982-0224-2024-0084
Guerrero HY, Cardillo E, Poleo G, Marcano D. Reproductive biology of freshwater fishes from the Venezuelan floodplains. Fish Physiol Biochem. 2009; 35(1):189–96. https://doi.org/10.1007/s10695-008-9249-7
Guillante T, Fonseca JDS, Costa PG, Bianchini A, Robaldo RB, Zebral YD. Sex-biased response of pollution biomarkers in fish: insights from the killifish Poecilia vivipara. Aquat Toxicol. 2023; 261:106613. https://10.1016/j.aquatox.2023.106613
Gurgel LL, Verani JR, Chellappa S.Reproductive ecology of Prochilodus brevis an endemic fish from the semiarid region of Brazil. Sci World. 2012; 2012(1):810532. https://doi.org/10.1100/2012/810532
Haque R, Singha K, Karmakar S. Environmental stressors on fish and it’s adaptation physiology. Aqua Int. 2019; 46–52.
Heins DC, Brown‐Peterson NJ. The reproductive biology of small fishes and the clutch concept: combining macroscopic and histological approaches. Aquac Fish Fish. 2022; 2(4):253–64. https://doi.org/10.1002/aff2.49
Instituto Nacional de Pesquisas Espaciais (INPE). Estimativa de desmatamento na Amazônia Legal para 2024. [Internet]. São José dos Campos: INPE; 2024. Available from: https://data.inpe.br/big/web/biomasbr/notas-tecnicas/estimativa-de-desmatamento-na-amazonia-legal-para/
Jézéquel C, Tedesco PA, Bigorne R, Maldonado-Ocampo JA, Ortega H, Hidalgo M etal. A database of freshwater fish species of the Amazon Basin. Sci Data. 2020; 7(1):96. https://doi.org/10.1038/s41597-020-0436-4
Jirotkul M. Operational sex ratio influences female preference and male–male competition in guppies. Anim Behav. 1999; 58(2):287–94. https://doi.org/10.1006/anbe.1999.1149
Juen L, Cunha EJ, Carvalho FG, Ferreira MC, Begot TO, Andrade AL et al. Effects of oil palm plantations on the habitat structure and biota of streams in eastern Amazon. River Res Appl. 2016; 32(10):2081–94. https://doi.org/10.1002/rra.3050
Jung EH, Brix KV, Richards JG, Val AL, Brauner CJ. Reduced hypoxia tolerance and survival at elevated temperatures may limit the ability of Amazonian fishes to survive in a warming world. Sci Total Environ. 2020; 748:141349. https://doi.org/10.1016/J.SCITOTENV.2020.141349
Keller A, Chattopadhyay S, Piniewski M. What evidence exists on the impacts of flow variability on fish and macroinvertebrates of temperate floodplain rivers in Central and Western Europe? A systematic map protocol. Environ Evid. 2021; 10(1):10. https://doi.org/10.1186/s13750-021-00225-z
Ladislau DS, Ribeiro MWS, Castro PDS, Aride PHR, Paiva AJV, Polese MF et al. Ornamental fishing in the region of barcelos, amazonas: Socioeconomic description and scenario of activity in the view of “piabeiros”. Braz J Biol. 2020; 80(3):544–56. https://doi.org/10.1590/1519-6984.215806
Leite GFM, Silva FTC, Gonçalves JFJ, Salles P. Effects of conservation status of the riparian vegetation on fish assemblage structure in neotropical headwater streams. Hydrobiologia. 2015; 762(1):223–38. https://doi.org/10.1007/s10750-015-2351-9
López-Rodríguez NC, Leão AHF, Rocha RM, Prudente BS, Montag LFA. Environmental influence on the reproductive strategy of Helogenes marmoratus (Siluriformes: Cetopsidae) in the Amazonian streams. Neotrop Ichthyol. 2021; 19(4):e210092. https://doi.org/10.1590/1982-0224-2021-0092
Martins-Queiroz MF, Mateus LAF, Garutti V, Venere PC. Reproductive biology of Triportheus trifurcatus (Castelnau, 1855) (Characiformes: Characidae) in the middle rio Araguaia, MT, Brazil. Neotrop ichthyol. 2008; 6(2):231–36. https://doi.org/10.1590/S1679-62252008000200010
Maskill PAC, Miller IR, Halvorson LJ, Treanor HB, Fraser CW, Webb MAH. Role of sex ratio and density on fertilization success of intensively cultured endangered woundfin. J Fish Wildl Manag. 2017; 8(1):249–54. https://doi.org/10.3996/062016-JFWM-048
McBride RS, Somarakis S, Fitzhugh GR, Albert A, Yaragina NA, Wuenschel MJ et al. Energy acquisition and allocation to egg production in relation to fish reproductive strategies. Fish Fish. 2015; 16(1):23–57. https://doi.org/10.1111/faf.12043
Mello K, Valente RA, Randhir TO, Santos ACA, Vettorazzi CA. Effects of land use and land cover on water quality of low-order streams in Southeastern Brazil: watershed versus riparian zone. Catena. 2018; 167:130–38. https://doi.org/10.1016/J.CATENA.2018.04.027
Molica JR, Mendes YA, Prudente BS, Oliveira RS, Pantoja JCD, Torres MF et al. Morphological and ecological approaches in reproduction of the endemic catfish of the eastern Amazon. J Fish Biol. 2024; 106(4):1036–46. https://doi.org/10.1111/jfb.16022
Mylonas CC, Fostier A, Zanuy S. Broodstock management and hormonal manipulations of fish reproduction. Gen Comp Endocrinol. 2010; 165(3):516–34. https://doi.org/10.1016/J.YGCEN.2009.03.007
Netto-Ferreira AL, Albrecht MP, Nessimian JL, Caramaschi EP. Feeding habits of Thoracocharax stellatus (Characiformes: Gasteropelecidae) in the upper river Tocantins, Brazil. Neotrop ichthyol. 2007; 5(1):69–74. https://doi.org/10.1590/S1679-62252007000100009
Nikolsky GV. Fish population dynamics: as the biological background for rational exploitation and management of fishery resources. Edinburgh: Oliver & Boyd; 1969.
Nunes DMF, Magalhães ALB, Weber AA, Gomes RZ, Normando FT, Santiago KB et al. Influence of a large dam and importance of an undammed tributary on the reproductive ecology of the threatened fish matrinxã Brycon orthotaenia Günther, 1864 (Characiformes: Bryconidae) in southeastern Brazil. Neotrop Ichthyol. 2015; 13(2):317–24. https://doi.org/10.1590/1982-0224-20140084
Oliveira MSB, Silva LMA, Prestes L, Tavares-Dias M. Length-weight relationship and condition factor for twelve fish species from the igarapé Fortaleza basin, a small tributary of the Amazonas River estuary. Acta Amazon. 2020; 50(1):8–11. https://doi.org/10.1590/1809-4392201900702
Pelicice FM, Agostinho AA, Akama A, Andrade Filho JD, Azevedo-Santos VM, Barbosa MVM et al. Large-scale degradation of the Tocantins-Araguaia River basin. Environ Manage. 2021; 68(4):445–52. https://doi.org/10.1007/s00267-021-01513-7
Prestes L, Oliveira MSB, Tavares-Dias M, Soares MGM, Cunha FC. Length-weight relationship and condition factor of eight fish species from the upper Araguari River, State of Amapá, Brazil. Acta Sci Biol Sci. 2019; 41(1):e46666. https://doi.org/10.4025/actascibiolsci.v41i1.46666
Pusey BJ, Arthington AH. Importance of the riparian zone to the conservation and management of freshwater fish: a review. Mar Freshw Res. 2003; 54(1):1–16. https://doi.org/10.1071/MF02041
Quagio-Grassiotto I, Grier H, Mazzoni TS, Nóbrega RH, Amorim JPA. Activity of the ovarian germinal epithelium in the freshwater catfish, Pimelodus maculatus (Teleostei: Ostariophysi: Siluriformes): germline cysts, follicle formation and oocyte development. J Morphol. 2011; 272(11):1290–306. https://doi.org/10.1002/jmor.10981
Queiroz HL, Sobanski MB, Magurran AE. Reproductive strategies of red-bellied piranha (Pygocentrus nattereri Kner, 1858) in the white waters of the Mamirauá flooded forest, central Brazilian Amazon. Environ Biol Fish. 2010; 89:11–19. https://doi.org/10.1007/s10641-010-9658-1
Rauber RG, Strictar L, Gomes LC, Suzuki HI, Agostinho AA. Spatial segregation in the reproductive activity of Neotropical fish species as an indicator of the migratory trait. J Fish Biol. 2021; 98(3):694–706. https://doi.org/10.1111/jfb.14614
Röpke C, Pires THS, Zuchi N, Zuanon J, Amadio S. Effects of climate-driven hydrological changes in the reproduction of Amazonian floodplain fishes. J Appl Ecol. 2022; 59(4):1134–45. https://doi.org/10.1111/1365-2664.14126
Silva EL, Centofante L, Miyazawa CS. Análise morfométrica em Thoracocharax stellatus (Kner, 1858) (Characiformes, Gasteropelecidae) proveniente de diferentes bacias hidrográficas Sul-americanas. Biota Neotrop. 2009; 9(2):71–76. https://doi.org/10.1590/S1676-06032009000200006
Silva K, Nascimento DTF. Aplicação do protocolo de avaliação rápida (PAR) para análise da degradação ambiental do córrego Tamanduá (Iporá-GO). In: Perez Filho A, Amorim RR, editors. Os desafios da geografia física na fronteira do conhecimento. Campinas: Instituto de Geociências, Unicamp; 2017.
Silva MCR, Rodrigues J, Silva HTL, Jesus PS, Cunha CV, Souza KNS et al. Reproductive cycle of the flying fish Hemiodus unimaculatus (Bloch, 1794) (Characiforms: Hemiodontidae) in the upstream area of the Tucurui hydroelectric plant, Pará, Brazil. Acta Zool. 2023; 105(4):439–49. https://doi.org/10.1111/azo.12481
Siqueira-Silva DH, Alberto Vicentini C, Ninhaus-Silveira A, Veríssimo-Silveira R. Reproductive cycle of the Neotropical cichlid yellow peacock bass Cichla kelberi: a novel pattern of testicular development. Neotrop Ichthyol. 2013; 11(3):587–96. https://doi.org/10.1590/S1679-62252013000300012
Siqueira-Silva DH, Rodrigues J, Braga RS, Lima B, Jesus P, Coimbra VC et al. Alternative protocol for the histological preparation of animal tissues. Tissue Cell. 2025; 95:102841. https://doi.org/10.1016/j.tice.2025.102841
Suzuki H I. Reproductive ecology of the fish assemblages In: Thomaz SM, Agostinho AA, Hahn NS, editors. The upper Paraná river and its floodplain: physical aspects, ecology and conservation. Leidein: Backhuys Publichers; 2004. p.271–91.
Toledo-Piza M, Baena EG, P Dagosta FC, Menezes NA, Ândrade M, Benine RC et al.Checklist of the species of the Order Characiformes (Teleostei: Ostariophysi). Neotrop Ichthyol. 2024; 22(1):e230086. https://doi.org/10.1590/1982-0224-2023-0086
Waddell JC, Crampton WG. Environmental correlates of circannual breeding periodicity in a multi-species assemblage of Amazonian electric fishes. Environ Biol Fish. 2020; 103:233–50. https://doi.org/10.1007/s10641-020-00950-3
Weitzman SH, Palmer L. Gasteropelecidae (Freshwater hatchetfishes). In Reis RE, Kullander SO, Ferraris, Jr. SJ, editors. Checklist of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.101–03.
Wright IJ, Dong N, Maire V, Prentice IC, Westoby M, Díaz S et al. Global climatic drivers of leaf size. Science. 2017; 357(6354):917–21. https://doi.org/10.1126/science.aal4760
Authors
![]() Marcelle Fernanda de Oliveira Barbosa1,2,
Marcelle Fernanda de Oliveira Barbosa1,2, ![]() Raquel Santos dos Santos3,
Raquel Santos dos Santos3, ![]() Felipe Fernando Silva Siqueira4,
Felipe Fernando Silva Siqueira4, ![]() Eduardo Silva do Nascimento Albuquerque2,
Eduardo Silva do Nascimento Albuquerque2, ![]() Natalia Rodrigues da Silva2,
Natalia Rodrigues da Silva2, ![]() Sara Ferreira Pedrosa2,
Sara Ferreira Pedrosa2, ![]() Paulo de Souza Jesus2,
Paulo de Souza Jesus2, ![]() Otávio Antunes Lacerda1,2,
Otávio Antunes Lacerda1,2, ![]() Marcelo Ândrade5,
Marcelo Ândrade5, ![]() Maria Auxiliadora Pantoja Ferreira6,
Maria Auxiliadora Pantoja Ferreira6, ![]() Caio Maximino7 and
Caio Maximino7 and ![]() Diógenes Henrique Siqueira-Silva1,2
Diógenes Henrique Siqueira-Silva1,2 ![]()
[1] Programa de Pós-Graduação em Reprodução Animal na Amazônia (ReproAmazon) do Instituto de Medicina Veterinária, Universidade Federal Rural da Amazonia (UFRA), Campus II, Castanhal, Rodovia BR-316, km 61, 68740-970, Castanhal, PA, Brazil. (MFOB) celle2035@gmail.com, (OAL) otaviolacerda30@gmail.com.
[2] Grupo de Estudos sobre a Reprodução de Peixes da Amazônia (GERPA), Laboratório de Neurociências e Comportamento (LANEC), Faculdade de Biologia (FACBIO), Universidade Federal do Sul e Sudeste do Pará (UNIFESSPA), Campus III, Avenida dos Ipês, s/n, 68500-000, Marabá, PA, Brazil. (ESNA) eduardoalbuquerque677@gmail.com, (NRS) rodriguesnatalia4002@gmail.com, (PSJ) paulodejesusj96@gmail.com, (SFP) saraferreirapedrosa@gmail.com, (CM) cmaximino@unifesspa.edu.br, (DHSS) diogenessilva@unifesspa.edu.br (corresponding author).
[3] Grupo de Pesquisa Produção e Conservação da Biodiversidade de Espécies Aquáticas (AQUAM), Programa de Pós-Graduação em Zootecnia, Universidade Federal do Rio Grande do Sul, Avenida Bento Gonçalves, 7712, 91540-000, Porto Alegre, RS, Brazil. (RSS) racheelhere@gmail.com.
[4] Grupo de Estudos em Ecologia e Conservação na Amazônia, Universidade Federal do Sul e Sudeste do Pará (UNIFESSPA), Faculdade de Biologia (FACBIO), Marabá, PA, Brazil. (FFSS) felipeuast@gmail.com.
[5] Núcleo de Ecologia Aquática e Pesca da Amazônia, Universidade Federal do Pará, 66077-830, Belém, PA, Brazil. (MA) andrademarcosta@gmail.com
[6] Laboratório de Pesquisa em Imuno-histoquímica e Biologia do Desenvolvimento, Universidade Federal do Pará (UFPA), Cidade Universitária Professor José Silveira Netto, Av. Perimetral, 2651, Terra Firme, 66077-830, Belém, PA, Brazil. (MAPF) auxi@ufpa.br.
[7] Laboratório de Neurociência e Comportamento “Frederico Guilherme Graeff” (LANEC), Universidade de Psicologia, Instituto de Estudos de Saúde e Biológicos, Universidade Federal do Sul e Sudeste do Pará, Av. dos Ipês, s/n, 68507-590, Marabá, PA, Brazil.
Authors’ Contribution 

Marcelle Fernanda de Oliveira Barbosa: Conceptualization, Investigation, Methodology, Project administration, Resources, Validation, Writing-original draft.
Raquel Santos dos Santos: Resources, Validation, Writing-original draft.
Felipe Fernando Silva Siqueira: Conceptualization, Investigation, Methodology, Supervision, Writing-original draft.
Eduardo Silva do Nascimento Albuquerque: Investigation, Resources.
Natalia Rodrigues da Silva: Methodology, Resources.
Sara Ferreira Pedrosa: Methodology, Resources.
Paulo de Souza Jesus: Formal analysis, Investigation, Methodology, Validation.
Otávio Antunes Lacerda: Formal analysis, Investigation, Resources.
Marcelo Ândrade: Data curation, Formal analysis, Methodology.
Maria Auxiliadora Pantoja Ferreira: Formal analysis, Investigation, Methodology, Resources, Validation, Writing-original draft.
Caio Maximino: Data curation, Funding acquisition, Writing-review and editing.
Diógenes Henrique Siqueira-Silva: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing-original draft.
Ethical Statement
Collection of the animals was authorized by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio), the Brazilian protected areas agency, through protocol SISBIO 88053–1, and all experimental procedures involving the animals were approved by the Animal Use Ethics Committee of the Universidade Federal do Sul e Sudeste do Pará (Unifesspa), (CEUA-Unifesspa, project number 23479.010692/2021–16). The procedures were consistent with the established guidelines of the Conselho Nacional para o Controle da Experimentação Animal (CONCEA). The project was also registered in the Sistema Nacional de Gestão do Patrimônio Genético e Conhecimentos Tradicionais Associados do Ministério do Meio Ambiente, Brazil (register n° AD00F28).
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures
Funding
The authors would like to thank the Fundação Amazonia de Amparo a Estudos e Pesquisas (FAPESPA), and Conselho Nacional de Desenvolvimento Científco e Tecnológico (CNPq) for granting the students’ scholarship through the intermediary of Pró-Reitoria de Pós-Graduação, Pesquisa e Inovação Tecnológica da Unifesspa. Research fellows DHSS (CNPq grants 313053/2022–7 and 442763/2023–9), MAPF (CNPq grant 308895/2022–3), and CNPq for funding project 300189/2022–2.
How to cite this article
Barbosa MFO, Santos RS, Siqueira FFS, Albuquerque ESN, Silva NR, Pedrosa SF, Jesus PS, Lacerda OA, Ândrade M, Ferreira MAP, Maximino C, Siqueira-Silva DH. Reproductive biology of the spotfin hatchetfish Thoracocharax stellatus (Characiformes: Gasteropelecidae) in the Western Amazon. Neotrop Ichthyol. 2026; 24(1):e250046. https://doi.org/10.1590/1982-0224-2025-0046
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted November 6, 2025
Accepted November 6, 2025
![]() Submitted March 15, 2025
Submitted March 15, 2025
![]() Epub April 17, 2025
Epub April 17, 2025