![]() César Santificetur1
César Santificetur1 ![]() ,
, ![]() Pollyana Christine Gomes Roque1,2,3,
Pollyana Christine Gomes Roque1,2,3, ![]() Sara de Castro Loebens2,4,
Sara de Castro Loebens2,4, ![]() Alessandra Maria Advíncula Pires2,
Alessandra Maria Advíncula Pires2, ![]() Danielle de Lima Viana2 and
Danielle de Lima Viana2 and ![]() Paulo Guilherme Vasconcelos de Oliveira2
Paulo Guilherme Vasconcelos de Oliveira2
PDF: Download Here | Cite this article
Associate Editor: ![]() Michael Mincarone
Michael Mincarone
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Os sagittae são amplamente utilizados como marcadores naturais em estudos taxonômicos e ecológicos de peixes, especialmente quando os caracteres morfológicos externos são conservadores ou ambíguos. Analisamos a variação morfológica entre duas espécies congêneres de Physiculus: P. cirm, endêmica do Arquipélago de São Pedro e São Paulo, e P. kaupi, distribuída ao longo da margem continental brasileira. Cinquenta espécimes adultos (25 por espécie) foram examinados utilizando descrições qualitativas, morfometria com índices de forma, e decomposição de contorno por wavelets de 4ª e 5ª ordens. Este trabalho representa a primeira descrição e análise morfométrica de seus otólitos sagitta. Ambas as espécies apresentaram otólitos sagitiformes com perfil plano-convexo e ostium elíptico, entretanto a região anterior é mais arredondada em P. cirm e mais pontiaguda em P. kaupi, enquanto a região posterior é lanceolada a arredondada em P. cirm, mas exclusivamente lanceolada em P. kaupi. Foram encontradas diferenças estatisticamente significativas nas razões altura/comprimento do otólito (OH/OL), espessura/comprimento do otólito (OT/OL), área do sulcus acusticus/ área total do otólito (SA/OA) e comprimento do otólito/ comprimento total do peixe (OL/TL). A classificação utilizando índices morfométricos e wavelets de 5ª ordem alcançou 98% de acurácia, destacando forte potencial discriminativo. Essas diferenças morfológicas provavelmente refletem divergência taxonômica e adaptação a habitats de águas profundas, apoiando o uso da morfologia dos otólitos para identificação de espécies e inferências ecológicas em peixes de águas profundas.
Palavras-chave: Atlântico Sudoeste, Morfologia, Morfometria, Wavelets.
Introduction
The inner ear of teleost fishes, essential for maintaining balance and sensory perception, is composed of three semicircular canals and three otic capsules: the sacculus, lagena, and utriculus. Each capsule contains a specific otolith: the sagitta, asteriscus, and lapillus, respectively (Ladich, Popper, 2004; Collin, 2007; Popper, Fay, 2011). These otoliths are metabolically inert structures, forming part of the mechanoreceptor system, primarily composed of aragonite (95%) and a protein matrix (5%), with trace elements (Campana, 2004; Volpedo, Vaz-dos-Santos, 2015; Schulz-Mirbach et al., 2019).
Among the three otolith pairs, sagittae are typically the most morphologically diverse and extensively used in various scientific applications (Assis, 2000; Volpedo, Vaz-dos-Santos, 2015). Otolith morphology varies considerably among species and even within the same species, influenced by physiological factors such as growth, sex, gonadal maturation, and diet, as well as by environmental conditions like depth and salinity, which affect biomineralization processes and morphometric patterns (Gagliano, McCormick, 2004; Popper, Fay, 2011; Avigliano et al., 2014). This variability renders sagittae valuable tools for species identification (Rossi-Wongtschowski et al., 2014; Moore et al., 2022; Haimovici et al., 2023), population differentiation (Bose et al., 2017; Vaz-dos-Santos et al., 2023; Almeida et al., 2024), and stock assessment (Soeth et al., 2019; Sbiba et al., 2024; Santificetur et al., 2025). Given the taxonomic complexity and broad distribution of many deep-sea fish groups, the interspecific morphological diversity of sagittae otoliths has proven particularly useful.
Among the diverse families of deep-sea fishes, Moridae Moreau, 1881, commonly known as “morid cods”, is notable for its richness, comprising 19 genera and 112 valid species (Fricke et al., 2025). Within this family, Physiculus Kaup, 1858 is the most species-rich genus. In the Atlantic Ocean, 12 Physiculus species have been recorded, four of which occur in Brazilian waters: P. cirm Carvalho-Filho & Pires, 2019, endemic to the Saint Peter and Saint Paul Archipelago; P. fulvus Bean, 1884, distributed in the northern region; P. karrerae Paulin, 1989, occurring along the southeastern and southern coasts; and P. kaupi Poey, 1865, which has a broader range extending from the northeastern to the southern Brazilian coast (Paulin, 1989; Oliveira et al., 2015; Pires et al., 2019; Lemes, Melo, 2025).
This study aims to distinguish P. cirm and P. kaupi based on the morphology and morphometry of sagittae, including contour analyses using 4th and 5th order wavelet decomposition. Given the limited knowledge of the genus Physiculus, particularly in the South Atlantic, otolith-based approaches represent valuable tools for improving taxonomic resolution, understanding population structure, and supporting fisheries monitoring of deep-sea fish species.
Material and methods
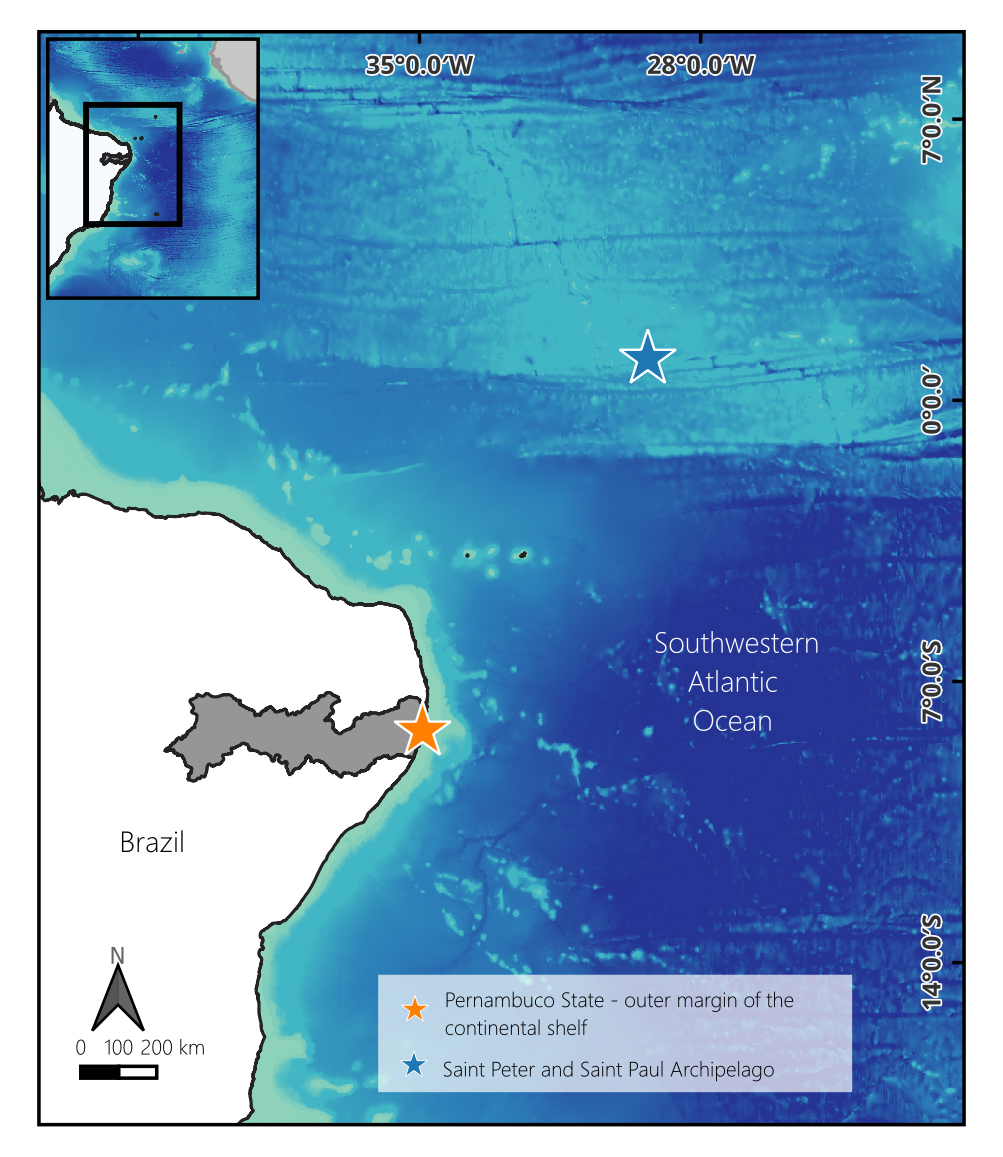
Study area. This study was conducted concurrently at two sites in the Southwestern Atlantic Ocean (Fig. 1). The first site is located along the outer margin of the continental shelf off the state of Pernambuco (PE; 07°50’S 34°27’W to 08°52’S 34°47’W). This region is characterized by a gently sloping sedimentary surface with a narrow shelf width (Manso et al., 2003). The shelf break lies between 50 and 80 m in depth, followed by a steep upper slope that gradually smooths out beyond 500 m (Kempf, 1970; Araújo et al., 2004; Camargo et al., 2015).
FIGURE 1| Map of Physiculus sampling locations from the Sothwestern Atlantic Ocean. Specimens from P. kaupi were collected along the outer margin of the continental shelf off the State of Pernambuco (orange star) and P. cirm were collected around the Saint Peter and Saint Paul Archipelago (blue star). Geographic Coordenate System. Datum: Sirgas 2000. Cartographic base: IBGE (2021/2022); SGB (2013); MapasAcademicos (2025); GEBCO (2024).
The second site is located around the Saint Peter and Saint Paul Archipelago (SPSPA; 00°55’10”N 29°20’33”W), situated on the equatorial Mid-Atlantic Ridge, approximately 1,060 km off the northeastern coast of Brazil. The archipelago represents the summit of a large sigmoidal submarine ridge rising 3,500 m above the seafloor and comprises five main islets (Belmonte, Southeast, Northeast, Northwest, and South) and five smaller rocks (Campos, 2009).
Data collection. A total of 100 Physiculus (48 P. cirm and 52 P. kaupi)specimens were collected through experimental fisheries using bottom-traps, between October 2014 and October 2018, at depths ranging from 200 to 750 m. Immediately after the capture, the specimens were cooled and, as far as possible, labelled at the Fisheries Oceanography Laboratory of Universidade Federal Rural de Pernambuco. The total length (TL, mm) of each fish was measured. Seven specimens were deposited to the Museu de Oceanografia Prof. Petrônio Alves Coelho, Universidade Federal de Pernambuco (two specimens of Physiculus cirm: MOCH 1521; five specimens of Physiculus kaupi: MOCH 1517). Only mature specimens were recorded in the samples.
The sagittae were extracted from the ear cavity and washed to remove any remaining macula and vestibule tissue. Images of 25 left and right sagittae from each species were taken with the inner face facing upwards and in profile view, using a Zeiss Discovery V12 stereomicroscope with reflected light at 8× magnification. The microscope was equipped with a 5 MP AxioCam MRc5 digital camera, connected via USB. Photographs were taken against a dark background to enhance contrast and facilitate visualization of relevant morphological structures. After the images were captured, left otoliths were used for morphology, morphometry and shape indices, wavelet-based otolith contour decomposition and statistical analysis.
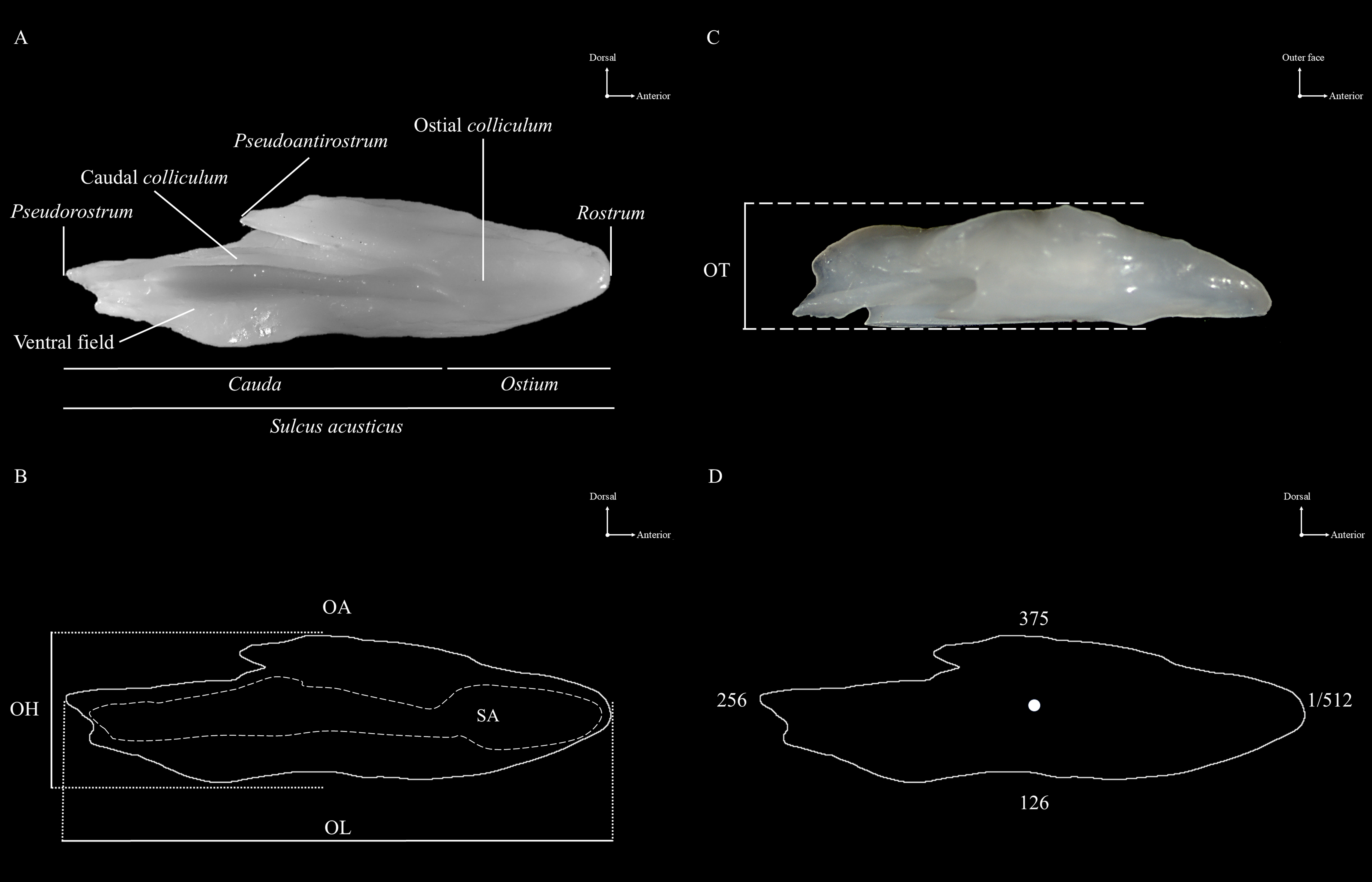
Morphology. Morphological analysis of otoliths considered both external and sulcus acusticus characteristics. For the external features, the following traits were evaluated: shape, anterior region, posterior region, edges, rostrum, antirostrum, pseudorostrum, pseudoantirostrum, and profile. The analysis of the sulcus acusticus focused on its position, orientation, opening, morphology, colliculum, ostium, and cauda (Fig. 2A). These morphological characteristics were assessed following the criteria established by Tuset et al. (2008) and Rossi-Wongtschowski et al. (2014).
FIGURE 2| External features and otolith morphometry of Physiculus species. A. Shows the inner face of the left sagitta otolith highlighting external anatomical features; B. Presents the otolith outline with morphometric measurements including otolith length (OL), otolith height (OH), otolith area (OA, solid line), and sulcus acusticus area (SA, dotted line); C. Shows the otolith profile indicating its thickness (OT); D. Displays the otolith outline with the centroid and selected points along the 512 point digitized contour used for wavelet-based shape analysis.
Morphometry and shape indices. The variables included otolith length (OL, mm), otolith height (OH, mm), otolith thickness (OT, mm), otolith area (OA, mm2), and sulcus acusticus area (SA, mm2) (Figs. 2B–C) (Lombarte, Morales‐Nin, 1995; Tuset et al., 2003; Rossi-Wongtschowski et al., 2014). A paired sample t-test was used to assess whether there were significant differences between left and right otoliths, based on the mean values of the measured variables. From these measurements, the following morphometric indices were calculated: the ratio of otolith height to otolith length (OH/OL), the ratio of otolith thickness to otolith length (OT/OL), the ratio of sulcus acusticus area to otolith area (SA/OA), and the ratio of otolith length to total length of specimen (OL/TL) (Volpedo, Echeverría, 2003; Avigliano et al., 2015; Souza et al., 2019). These indices provide a detailed geometric characterization of the otoliths.
Wavelet-based otolith contour decomposition. The morphological analysis of otolith contours was conducted through wavelet-based signal decomposition, following the methodologies outlined by Parisi-Baradad et al. (2010) and Sadighzadeh et al. (2014). Wavelet transforms provide a multiscale framework for characterizing local shape variations by decomposing the contour signal into a series of scaled and translated versions of a mother function. The wavelet transform applied follows the general form Ψs(x) = (1/s)Ψ(φ/s), where Ψ is a compactly supported function, φ denotes a step filter, and s is a scale parameter controlling frequency and amplitude modulation (Mallat, 1991).
Each otolith was represented by 512 equidistant points extracted along the perimeter, beginning at the rostrum (Fig. 2D). The resulting contour coordinates were processed using wavelet transforms of both 4th and 5th orders. These specific orders were selected based on their proven sensitivity in capturing fine-scale morphological features, with 5th wavelets showing enhanced resolution for detecting subtle variations relevant to stock and population discrimination (Sadighzadeh et al., 2014; Tuset et al., 2015).
Contour extraction and preprocessing were performed using AFORO (http://aforo.cmima.csic.es), a dedicated software platform for the acquisition and analysis of otolith morphometric data (Parisi-Baradad et al., 2010). The tool enabled standardized digitization of the contours and facilitated the subsequent application of wavelet-based analytical procedures.
Statistical analysis. The nonparametric Mann-Whitney test was used to compare left and right otoliths and between males and females to evaluate bilateral symmetry and sex-related effects on otolith morphology. As no significant differences were found, subsequent analyses were conducted using only left otoliths.
The Mann-Whitney test was also applied to each shape index individually to detect significant morphometric differences among species. Before multivariate analyses, data were mean-centered to eliminate absolute effects and allow for consistent group comparisons (Legendre, Legendre, 1998). For all datasets, including shape indices and wavelet coefficients of 4th and 5th orders, linear models were used to correct for ontogenetic effects associated with body size. Residuals from these models were used in subsequent analyses (Lleonart et al., 2000).
Principal Component Analysis (PCA) was performed on the variance-covariance matrices of each dataset to reduce dimensionality while preserving axes that explain the most variance (Tuset et al., 2015). The calculated morphometric indices for the two Physiculus species were subjected to PCA, performed separately for each index.
Principal components (PCs) were retained based on the broken-stick criterion (Gauldie, Crampton, 2002), ensuring that only the most informative variables were included. These PCs were then used as predictor variables in a Linear Discriminant Analysis (LDA) to assess the discriminatory power of otolith morphology for classifying species into their respective trophic guilds (Stransky, MacLellan, 2005). Model performance was validated using both k-fold cross-validation and the jackknife method, providing robust statistical support and minimizing overfitting risks (Sokal, Rohlf, 1995). The LDA results were evaluated using a confusion matrix, with accuracy calculated.
Finally, differences in otolith morphology among trophic guilds were tested using Permutational Multivariate Analysis of Variance (PERMANOVA), based on Euclidean distances of the adjusted data. All analyses were performed in PAST software v. 4.03 (Hammer et al., 2001).
Results
Morphology. The otoliths of Physiculus cirm and P. kaupi exhibit considerable morphological similarities, most notably a common sagittiform shape (Fig. 3). Nevertheless, the anterior region is more rounded in P. cirm and more peaked in P. kaupi, whereas the posterior region is lanceolate to rounded in P. cirm and lanceolate in P. kaupi. The dorsal margin in P. cirm is entire and slightly elevated at the center, while in P. kaupi it is entire with uniformly low relief. These characteristics result in a broader and more rounded contour in P. cirm,contrasting with the narrower and more peaked contour found in P. kaupi. All other morphological characteristics are similar between the two species. The Tab. 1 presents the detailed description of the morphological characteristics of both species.
FIGURE 3| Specimen and respective left sagitta otoliths of Physiculus cirm (A1–A4) and P. kaupi (B1–B4), with total length (TL) and otolith length (OL) as follows: A2: 204 mm TL, 10.94 mm OL; A3: 249 mm TL, 11.20 mm OL; A4: 279 mm TL, 12.56 mm OL; B2: 210 mm TL, 9.03 mm OL; B3: 235 mm TL, 10.25 mm OL; B4: TL 274 mm TL, 12.38 mm OL. Scale bars = 1 mm.
TABLE 1 | Comparative morphological characteristics of the sagittae otoliths of Physiculus cirm and P. kaupi.
Features | Physiculus cirm | Physiculus kaupi |
Shape | Sagitiform | Sagitiform |
Anterior region | Round to Peaked | Peaked |
Posterior region | Round to Lanceolated | Lanceolated |
Dorsal edge | Slightly elevated at center | Low relief throughout |
Ventral edge | Entire | Entire |
Rostrum | Developed | Developed |
Antirostrum | Absent | Absent |
Pseudorostrum | Developed | Developed |
Pseudoantirostrum | Developed | Developed |
Profile | Plane-convex | Plane-convex |
Sulcus acusticus position | Median | Median |
Sulcus acusticus orientation | Horizontal | Horizontal |
Sulcus acusticus opening | Caudal | Caudal |
Sulcus acusticus morphology | Heterosulcoid | Heterosulcoid |
Colliculum | Heteromorphic | Heteromorphic |
Ostium | Elliptic | Elliptic |
Cauda | Funnel-like | Funnel-like |
Morphometry and shape indices. Means of the raw measurements and derived shape indices for both Physiculus cirm and P. kaupi are presented in Tab. 2. Morphological indices OH/OL, OT/OL, SA/OA, and OL/TL were calculated and compared between species. All indices showed statistically significant differences according to the Mann-Whitney U test: OH/OL (U = 22; p < 0.0001), OT/OL (U = 78; p < 0.0001), SA/OA (U = 68; p < 0.0001), and OL/TL (U = 109; p < 0.0001).
TABLE 2 | Minimum and maximum values, range of means, and standard deviation (SD) for morphometric variables and shape indices of the left sagitta otolith of Physiculus kaupi and P. cirm. N = number of otolith; TL = fish total length (mm); OL = otolith length (mm); OH = otolith height (mm); OT = otolith thickness (mm); OA = otolith area (mm2); SA = sulcus acusticus area (mm2).
| Physiculus cirm | Physiculus kaupi | ||||||
Min | Max | Mean | SD | Min | Max | Mean | SD | |
N | 25 | 25 | ||||||
TL | 174 | 315 | 253 | 40 | 206 | 274 | 231 | 19 |
OL | 9.45 | 14.43 | 11.98 | 1.38 | 8.02 | 12.38 | 9.85 | 0.95 |
OH | 3.03 | 4.28 | 3.7 | 0.35 | 2.12 | 3.02 | 2.64 | 0.21 |
OT | 2.9 | 3.90 | 3.41 | 0.40 | 2.01 | 3.10 | 2.35 | 0.32 |
OA | 18.69 | 38.54 | 28.27 | 5.68 | 10.5 | 24.31 | 16.67 | 2.72 |
SA | 7.43 | 15.89 | 11.66 | 2.59 | 3.26 | 8.14 | 5.91 | 0.93 |
OH/OL | 27.56 | 37.04 | 31.03 | 1.87 | 23.4 | 29.77 | 26.85 | 1.69 |
OT/OL | 23.86 | 32.91 | 28.73 | 2.50 | 17.8 | 29.77 | 24.01 | 2.94 |
SA/OA | 34.43 | 45.48 | 41.1 | 2.60 | 29.2 | 42.96 | 35.59 | 3.57 |
OL/TL | 4.21 | 6.64 | 4.77 | 0.52 | 3.52 | 4.87 | 4.27 | 0.33 |
The calculated morphometric indices revealed distinct patterns of morphological variation between species. Four principal components (PCs) were retained; PC1 explained 66% of the variance and PC2 accounted for 17%. A confusion matrix indicated an overall classification accuracy of 98%, with correct classification rates of 100% for P. cirm and 96% for P. kaupi (Tab. 3A). PERMANOVA analysis applied to the shape indices showed statistically significant differences between species (F = 42.23; p < 0.0001).
Wavelet-based otolith contour decomposition. Mean wavelet coefficients of the 4th and 5th orders were calculated for the two Physiculus species (Fig. 4). Separate Principal Component Analyses (PCA) for each wavelet order revealed distinct morphological variation patterns between the species. For the 4th order coefficients, PC1 explained 43% of the total variance and PC2 accounted for 14%. Based on the Broken Stick criterion, 35 principal components were retained for Linear Discriminant Analysis (LDA). The resulting confusion matrix (Tab. 3B) showed an overall classification accuracy of 90%, with correct classification rates of 92% for P. cirm and 88% for P. kaupi.
FIGURE 4| The graphs illustrate the average shape variation captured by the 4th order (A) and 5th order (B) wavelet transformations for Physiculus cirm (blue line) and P. kaupi (orange line). The lines represent each species mean wavelet decomposition patterns of otolith contours
TABLE 3 | Jackknife reclassification matrix for two Physiculus species based on different otolith analysis approaches: (A) morphometric indices, (B) 4th order wavelet coefficients, and (C) 5th order wavelet coefficients. The actual species, P. cirm and P. kaupi, are listed in rows, while the predicted classifications are shown in columns. The last column (%) indicates the classification accuracy for each species. The overall accuracy for each method is provided in the last row.
Real species | Predicted species | ||
A | Physiculus cirm | Physiculus kaupi | % |
Physiculus cirm | 25 | 0 | 100 |
Physiculus kaupi | 1 | 24 | 96 |
Total | 26 | 24 | 98 |
B | Physiculus cirm | Physiculus kaupi | % |
Physiculus cirm | 23 | 2 | 92 |
Physiculus kaupi | 3 | 22 | 88 |
Total | 26 | 24 | 90 |
C | Physiculus cirm | Physiculus kaupi | % |
Physiculus cirm | 25 | 0 | 100 |
Physiculus kaupi | 1 | 24 | 96 |
Total | 26 | 24 | 98 |
In contrast, PCA on the 5th order coefficients revealed that PC1 explained 32% and PC2 19% of the variance. Forty-four components were retained for LDA, which achieved an overall classification accuracy of 98% (Tab. 3C), with P. cirm correctly classified in 100% of cases and P. kaupi in 96%.
PERMANOVA analysis applied to the wavelet coefficients of both orders detected statistically significant differences between the species, with greater differentiation for the 5th order coefficients (F = 3.533, p = 0.0073 for 4th order; F = 5.257, p < 0.0001 for 5th order).
Discussion
The family Moridae can be subdivided into three main morphological groups based on otolith characteristics: Mora group, Pseudophycis group, and Physiculus group (Karrer, 1971; Fitch, Barker, 1972; Schwarzhans, 2019). The Physiculus group, which includes the species analyzed in this study, comprises the genera Gadella Lowe, 1843; Laemonema Günther, 1862; Momonatira Paulin, 1986; Paralaemonema Trunov, 1990; Physiculus, Salilota Günther, 1887; and Tripterophycis Boulenger, 1902 (Schwarzhans, 2019).
Otoliths in this group are characterized by being thick and elongated, with a plane-convex profile, a flat inner face, and a sulcus acusticus extends over the entire inner face, with a short, elliptical ostium. In contrast, otoliths from the Mora and Pseudophycis groups tend to be less compact, thinner, and exhibit more pronounced internal curvatures (Schwarzhans, 2019).
Both P. cirm and P. kaupi conform to the general otolith pattern of the Physiculus group, displaying robust sagittae with a plane-convex profile and a short, elliptical sulcus acusticus. However, notable morphological differences exist between the two species. Physiculus kaupi exhibits more elongated otoliths, with an OH/OL ratio of 26.85 ± 1.69 compared to 31.03 ± 1.87 in P. cirm. Schwarzhans (2019) listed P. kaupi among the species for which otoliths are not known. However, based on the characteristics observed in the present study, this species can be assigned to a cluster of very slender otoliths, characterized by low dorsal rims and occasional posteroventral expansions on the outer face. Species such as P. cyanostrophus Anderson, Tweddle, 2002; P. dalwigki Kaup, 1858; P. fulvus; P. grinnelli Jordan & Jordan, 1922; P. japonicus Hilgendorf, 1879; P. longicavis Parin, 1984; P. microbarbata Paulin & Matallanas, 1990; and P. nigripinnis Okamura, 1982 share this morphology and are widely distributed in tropical and subtropical regions of the Pacific and Indian Oceans.
Furthermore, based on the otolith characteristics observed in the present study, P. cirm, a species described by Pires et al. in 2019 and thus not previously analysed, can be assigned to the second morphological category proposed by Schwarzhans also in 2019, defined by moderately elongated otoliths with a slight expansion of the mid-dorsal rim, a broad concavity in the antero-dorsal region, and a subtly upward-curving ostium. Members of this assemblage include P. karrerae; P. luminosa Paulin, 1983; P. natalensis Gilchrist, 1922; P. nigrescens Smith & Radcliffe, 1912; and P. therosideros Paulin, 1987; which mainly inhabit tropical to temperate waters of the Atlantic and Indian Oceans.
A third distinct group described by Schwarzhans (2019) exhibits more compressed otoliths featuring pronounced mid-dorsal expansions and, occasionally, an additional post-dorsal extension on the outer face. This assemblage comprises species such as P. argyropastus Alcock, 1894; P. capensis Gilchrist, 1922; P. cynodon Sazonov, 1986; P. huloti Poll, 1953; P. marisrubri Brüss, 1986; P. nematopus Gilbert, 1890; P. rastrelliger Gilbert, 1890; and P. roseus Alcock, 1891; distributed across the Eastern Pacific, Southwestern Pacific, Indian Ocean, and Southwestern Atlantic. These differences illustrate the morphological diversity within the Physiculus group and highlight the value of otoliths as tools for both taxonomic and potentially ecological differentiation.
In Brazil, the only Moridae species with previously described otolith morphology are Gadella imberbis (Vaillant, 1888) and Laemonema goodebeanorum Meléndez & Markle, 1997 (Rossi-Wongtschowski et al., 2014). Both species exhibit typical features of the Physiculus group, such as a sagittiform shape, prominent pseudorostrum and pseudoantirostrum development, and a heterosulcoid sulcus acusticus with caudal opening. They also share a plane-convex profile and pronounced otolith thickness, distinguishing Moridae from other teleost families.
In addition to these morphological differences, both P. cirm and P. kaupi possess relatively large size of their sagittae (OL/TL). According to Paxton (2000), otoliths with OL/TL ratios above approximately 4–5% of standard length are considered large, a characteristic often observed in deep-water and luminous fish species, where enhanced auditory capabilities are beneficial. This trait aligns with patterns described for deep-sea fishes, where enlarged otoliths are often linked to an enhanced role of auditory perception and acoustic communication in low-light environments (Platt, Popper, 1981; Popper, Fay, 1993; Paxton, 2000). As suggested by Gauldie (1988) and Lychakov, Rebane (2000), otolith size can indicate the functional capacity of the teleost inner ear, particularly in relation to sound transduction efficiency. In deep-sea habitats, where visual cues are limited and ambient noise is low, larger otoliths may serve as functional adaptations that improve the detection of sound or vibration stimuli (Paxton, 2000; Lombarte et al., 2010).
Moreover, although environmental factors such as temperature and depth influence otolith development (Simkiss, 1974; Morales-Nin, 1987), they do not fully explain the magnitude of variation observed among species inhabiting similar bathymetric zones. This suggests that, beyond environmental influences, intrinsic factors such as evolutionary history and functional specialization play a key role in shaping otolith size (Lombarte, Lleonart, 1993; Losos, Miles, 1994). Thus, the shared otolithic features of P. cirm and P. kaupi reflect not only a phylogenetic link within the Physiculus group but also possible adaptive convergence related to the sensory demands of deep-sea environments.
The OH/OL and OT/OL ratios showed differences between the species analyzed, suggesting subtle yet consistent variations in otolith robustness. Physiculus cirm tends to exhibit proportionally taller and thicker otoliths compared to P. kaupi, which may reflect functional differences related to postural stability, vibration detection, and habitat use. According to Tuset et al. (2003), the OH/OL ratio is one of the most effective shape descriptors for species discrimination, reflecting both phylogenetic constraints and ecological adaptations. Volpedo, Echeverría (2003) noted that taller and thicker otoliths are often associated with species of lower mobility, which rely more heavily on equilibrium and positional perception. Lombarte et al. (2010) further emphasized that environmental and functional factors, such as depth, swimming behavior, and vertical position in the water column, shape these otolith features. Therefore, although the differences between the species are not pronounced in isolation, the combined morphometric patterns contribute to the distinction between P. cirm and P. kaupi, suggesting also a slight divergence in their sensory and ecological adaptations.
The ratio between the area of the sulcus acusticus and the total otolith area (SA/OA) has been widely used as a functional indicator of auditory sensitivity in teleost fishes (Gauldie, 1988; Lombarte, 1992; Arellano et al., 1995). Morphological variations of the sulcus acusticus among species reflect ecological differences, indicating adaptations to distinct habitats and behaviors (Lombarte, Popper, 2004; Taylor et al., 2020; Verocai et al., 2023). An increase in this ratio suggests a greater capacity to detect acoustic stimuli, which is associated with higher vertical activity, acoustically complex environments, or increased reliance on hearing over vision (Rogers, Cox, 1988; Aguirre, Lombarte, 1999). In the present study, Physiculus cirm exhibited a significantly higher SA/OA ratio than P. kaupi, indicating possible auditory specialization related to different habitat use patterns and trophic strategies.
Finally, the analysis of otolith morphology using wavelet transforms proved to be an effective tool for distinguishing P. cirm and P. kaupi. Although both 4th and 5th order wavelet decompositions captured relevant morphological variation, the 5th order decomposition demonstrated higher classification accuracy, standing out for its greater discriminatory power, similar to what was observed in coastal species (Carvalho et al., 2024). These results highlight the potential of high-resolution contour analysis for identifying morphologically similar taxa. The integration of traditional shape indices with wavelet-based approaches thus represents a robust and complementary strategy for taxonomic and morpho-functional studies (Sadighzadeh et al., 2014; Tuset et al., 2021; Charmpila et al, 2024). Future research should incorporate ontogenetic, genetic, and otolith microchemical data to further elucidate the evolutionary and ecological drivers of sagitta morphology.
The results of this study provide the first morphometric description and analysis of its sagittal otoliths and demonstrate that Physiculus cirm and P. kaupi can be reliably distinguished based on multiple otolith morphological features. Qualitative differences in the anterior and posterior regions, significant variation in shape indices, and distinct contours captured by 4th and 5th order wavelets consistently support the morphological separation between these two species. The significant differences in OH/OL and OT/OL ratios indicate variations in otolith morphology and robustness between the species. Additionally, the significantly higher SA/OA ratio in P. cirm suggests a difference in the relative size of the sensory area. These morphological disparities reflect taxonomic divergence and may be associated with distinct functional adaptations to their deep-sea habitats.
Both species conform to the general otolith morphology of the Physiculus group, which is characterized by thick, plane-convex sagittae with a short sulcus acusticus and elliptical ostium. However, consistent interspecific differences, especially those captured by 5th order wavelet decomposition, allowed for high classification accuracy. This reinforces the value of high-resolution contour analysis as a powerful tool for distinguishing morphologically conservative taxa. In this context, sagittae of Physiculus species emerge not only as taxonomic markers but also as integrative structures that encapsulate functional and ecological information, offering a robust framework for systematics, evolutionary inference, and biodiversity assessment in deep-sea environments.
Acknowledgments
This paper contributes to Ciências do Mar II CAPES project: “Ecologia e pesca de espécies pelágicas oceânicas e demersais de profundidade na costa nordeste e ilhas oceânicas do Brasil”. We gratefully acknowledge the institutional support provide by the Brazilian Navy. Special thanks to the crew of R/V Sinuelo for their assistance during field surveys, and to the fishers from Transmar for their collaboration and essential knowledge of local marine environments. We also thank all collaborators involved in data collection, processing and fieldwork. We are especially grateful to Professor Fábio Hazin (in memoriam) whose mentorship and profound knowledge influenced this work. His dedication to marine science continues to inspire us.
References
Aguirre H, Lombarte A. Ecomorphological comparisons of sagittae in Mullus barbatus and M. surmuletus. J Fish Biol. 1999; 55(1):105–14. https://doi.org/10.1016/S0378-5955(00)00026-5
Almeida PRC, Costa MR, Ribeiro ATR, Almeida A, Azevedo R, Monteiro-Neto C et al. Population structure and habitat connectivity of Pogonias courbina (Perciformes, Sciaenidae) in two Brazilian lagoon systems on south-east coast of Rio de Janeiro, Brazil, inferred from otolith shape and elemental signatures. J Sea Res. 2024; 199:102500. https://doi.org/10.1016/j.seares.2024.102500
Araújo TCM, Seoane JCS, Coutinho PN. Geomorfologia da plataforma continental de Pernambuco. Oceanografia: um cenário tropical. 2004; 39–57.
Arellano RV, Hamerlynck O, Vincx M, Mees J, Hostens K, Gijselinck W. Changes in the ratio of the sulcus acusticus area to the sagitta area of Pomatoschistus minutus and P. lozanoi (Pisces, Gobiidae). Mar Biol. 1995; 122(3):355–60. https://doi.org/10.1007/BF00350868
Assis CADS. Estudo morfológico dos otólitos Sagitta, Asteriscus e Lapillus de teleósteos (Actinopterygii, teleostei) de Portugal Continental (Ph.D. Dissertation). Portugal: Universidade de Lisboa; 2000.
Avigliano E, Martinez CFR, Volpedo AV. Combined use of otolith microchemistry and morphometry as indicators of the habitat of the silverside (Odontesthes bonariensis) in a freshwater-estuarine environment. Fish Res. 2014; 149:55–60. https://doi.org/10.1016/j.fishres.2013.09.013
Avigliano E, Villatarco P, Volpedo AV. Otolith Sr: Ca ratio and morphometry as indicators of habitat of a euryhaline species: the case of the silverside Odontesthes bonariensis. 2015; Ciênc Mar. 41(3):189–202. https://doi.org/10.7773/cm.v41i3.2464
Bose AP, Adragna JB, Balshine S. Otolith morphology varies between populations, sexes and male alternative reproductive tactics in a vocal toadfish Porichthys notatus. 2017; J Fish Biol. 90(1):311–25. https://doi.org/10.1111/jfb.13187
Camargo JMR, Araújo TCM, Ferreira BP, Maida M. Topographic features related to recent sea level history in a sediment-starved tropical shelf: linking the past, present and future. Reg Stud Mar Sci. 2015; 2:203–11. https://doi.org/10.1016/j.rsma.2015.10.009
Campana SE. Photographic atlas of fish otoliths of the northwest Atlantic Ocean. Canadian Science Publishing eBooks: Canadian Science Publishing; 2004. https://doi.org/10.1139/9780660191089
Campos G. Posição ímpar do Arquipélago de São Pedro e São Paulo na diversidade geológica da Terra. In: Viana D, Hazin F, Carvalho M, editors. O Arquipélago de São Pedro e São Paulo: 10 anos de Estação Científica. Brasília, DF: SECIRM; 2009.
Carvalho BMD, Bot RL, Schwarz R, Spach HL, Volpedo AV. Otolith as a tool to differentiate juveniles of two species Centropomidae. Pap Avulsos Zool. 2024; 64:e202464007. https://doi.org/10.11606/1807-0205/2024.64.007
Charmpila EA, Azad Teimori, Reichenbacher B. Otolith-based species identification in the killifish Aphaniops (Teleostei; Cyprinodontiformes; Aphaniidae) using both morphometry and wavelet analysis. Acta Zool. 2024; 106(3):291–308. https://doi.org/10.1111/azo.12518
Collin SP. Nervous and sensory systems. In: McKenzie DJ, Farrel AP, Brauner CJ, editors. Primitive fishes. Academic Press; 2007. p.121–79. https://doi.org/10.1016/S1546-5098(07)26003-0
Fitch JE, Barker LW. The fish family Moridae in the eastern North Pacific with notes on morid otoliths, caudal skeletons, and the fossil record. Fish Bull. 1972; 70(3):565–84.
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Gagliano M, McCormick M. Feeding history influences otolith shape in tropical fish. Mar Ecol Progr Ser. 2004; 278:291–96. https://doi.org/10.3354/meps278291
Gauldie RW. Function, form and time-keeping properties of fish otoliths. Comp Biochem Physiol Part A Physiol. 1988; 91(2):395–402. https://doi.org/10.1016/0300-9629(88)90436-7
Gauldie RW, Crampton JS. An eco-morphological explanation of individual variability in the shape of the fish otolith: comparison of the otolith of Hoplostethus atlanticus with other species by depth. J Fish Biol. 2002; 60(5):1204–21. https://doi.org/10.1111/j.1095-8649.2002.tb01715.x
Haimovici M, Rodrigues LS, Lucato SHB, Freire MA, Fischer LG, Cardoso LG. Otolith atlas for marine fishes of the southwestern Atlantic occurring along southern Brazil (28 S-34 S). MAFIS. 2023; 37(1):53–207. https://doi.org/10.47193/mafis.3712024010101
Hammer Ø, Harper DAT, Ryan PD. Paleontological statistics software package for education and data analysis. Palaeontol Electr. 2001; 4(1):1–09.
Karrer C. Die otolithen der Moridae (Teleostei, Gadiformes) und ihre systematische Bedeutung. Zool Jb Syst. 1971; 98:153–204.
Kempf M. A plataforma continental de Pernambuco (Brasil): nota preliminar sobre a natureza do fundo. Trab Oceanogr UFPE. 1970; 9(11):111–24.
Ladich F, Popper AN. Parallel evolution in fish hearing organs. In: Manley GA, Fay RR, Popper AN, editors. Evolution of the vertebrate auditory system. New York: Springer; 2004. p.95–127. https://doi.org/10.1007/978-1-4419-8957-4_4
Legendre P, Legendre L. Numerical ecology: second English edition Elsevier. Amsterdam: Science BV; 1998.
Lemes PCR, Melo MRS. Moridae in Catálogo Taxonômico da Fauna do Brasil [Internet]. 2025. Available from: http://fauna.jbrj.gov.br/fauna/faunadobrasil/5930
Lleonart J, Salat J, Torres GJ. Removing allometric effects of body size in morphological analysis. J Theor Biol. 2000; 205(1):85–93. https://doi.org/10.1006/jtbi.2000.2043
Lombarte A. Changes in otolith area: sensory area ratio with body size and depth. Environ Biol Fishes. 1992; 33(4):405–10. https://doi.org/10.1007/BF00010955
Lombarte A, Lleonart J. Otolith size changes related with body growth, habitat depth and temperature. Environ Biol Fishes. 1993; 37(3):297–306. https://doi.org/10.1007/BF00004637
Lombarte A, Morales-Nin B. Morphology and ultrastructure of saccular otoliths from five species of the genus Coelorinchus (Gadiformes: Macrouridae) from the Southeast Atlantic. J Morphol. 1995; 225(2):179–92. https://doi.org/10.1002/jmor.1052250204
Lombarte A, Palmer M, Matallanas J, Gómez-Zurita J, Morales-Nin B. Ecomorphological trends and phylogenetic inertia of otolith sagittae in Nototheniidae. Environ Biol Fishes. 2010; 89(3–4):607–18. https://doi.org/10.1007/s10641-010-9673-2
Lombarte A, Popper AN. Quantitative changes in the otolithic organs of the inner ear during the settlement period in European hake Merluccius merluccius. Mar Ecol Prog Ser. 2004; 267:233–40. https://doi.org/10.3354/meps/267233
Losos JB, Miles DB. Adaptation, constraint, and the comparative method: phylogenetic issues and methods. In: Wainwright PC, Reilly S, editors. Ecological morphology: integrative organismal biology. 1994; p.60–98.
Lychakov DV, Rebane YT. Otolith regularities. Hear Res. 2000; 143(1–2):83–102. https://doi.org/10.1016/S0378-5955(00)00026-5
Mallat S. Zero-crossings of a wavelet transform. IEEE. 1991; 37(4):1019–33. https://doi.org/10.1109/18.86995
Manso VDAV, Correa ICS, Guerra N. Morfologia e sedimentologia da plataforma continental interna entre as Praias Porto de Galinhas e Campos-Litoral Sul de Pernambuco, Brasil. Pesqui Geociênc. 2003; 30(2):17–25. https://doi.org/10.22456/1807-9806.19587
Moore BR, Parker SJ, Pinkerton MH. Otolith shape as a tool for species identification of the grenadiers Macrourus caml and M. whitsoni. Fish Res. 2022; 253:106370. https://doi.org/10.1016/j.fishres.2022.106370
Morales-Nin BYO. The influence of environmental factors on microstructure of otoliths of three demersal fish species caught off Namibia. S Afr J Mar Sci. 1987; 5(1):255–62. https://doi.org/10.2989/025776187784522207
Oliveira JEL, Nóbrega MF, Garcia Jr. J, Sampaio CLS, Di Dario F, Fischer LG et al. Biodiversidade marinha da Bacia Potiguar/RN. Peixes do Talude Continental. Rio de Janeiro: Museu Nacional do Rio de Janeiro, Série Livros; 2015.
Parisi-Baradad V, Manjabacas A, Lombarte A, Olivella R, Chic Ò, Piera J et al. Automated taxon identification of Teleost fishes using an otolith online database – AFORO. Fish Res. 2010; 105(1):13–20. https://doi.org/10.1016/j.fishres.2010.02.005
Paulin CD. Moridae: overview. In: Cohen DM, editor. Papers on the systematics of gadiform fishes. Nat Hist Mus Los Angeles County Sci Ser. 1989; 243–50.
Paxton JR. Fish otoliths: do sizes correlate with taxonomic group, habitat and/or luminescence? Philos Trans R Soc Lond B Biol Sci. 2000; 355(1401):1299–303. https://doi.org/10.1098/rstb.2000.0688
Pires AM, Carvalho-Filho A, Ferreira RC, Viana D, Nunes D, Hazin FH. Review of the Brazilian species of Physiculus (Gadiformes: Moridae), with description of a new species from Saint Peter and Saint Paul Archipelago, equatorial Atlantic. Zootaxa. 2019; 4671(1):67–80. https://doi.org/10.11646/zootaxa.4671.1.5
Platt C, Popper AN. Otolith organ receptor morphology in herring-like fishes. In: Gualtierotti T, editor. The vestibular system: function and morphology. New York: Springer; 1981. p.64–76. https://doi.org/10.1007/978-1-4612-5902-2_3
Popper AN, Fay RR. Sound detection and processing by fish: critical review and major research questions (part 2). Brain Behav Evol. 1993; 41(1):26–38.
Popper AN, Fay RR. Rethinking sound detection by fishes. Hear Res. 2011; 273(1–2):25–36. https://doi.org/10.1016/j.heares.2009.12.023
Rogers PH, Cox M. Underwater sound as a biological stimulus. In: Atema J, Fay RR, Popper AN, Tavolga WN, editors. Sensory biology of aquatic animals. New York: Springer; 1998. p.131–49. https://doi.org/10.1007/978-1-4612-3714-3_5
Rossi-Wongtschowski CLDB, Siliprandi CC, Brenha MR, Gonsales SDA, Santificetur C, Santos AMVD. Atlas of marine bony fish otoliths (Sagittae) of Southeastern-Southern Brazil Part I: Gadiformes (Macrouridae, Moridae, Bregmacerotidae, Phycidae and Merlucciidae); Part II: Perciformes (Carangidae, Sciaenidae, Scombridae and Serranidae). Braz J Oceanogr. 2014; 62:1–103. https://doi.org/10.1590/S1679-875920140637062sp1
Sadighzadeh Z, Valinassab T, Vosugi G, Motallebi AA, Fatemi MR, Lombarte A et al. Use of otolith shape for stock identification of John’s snapper, Lutjanus johnii (Pisces: Lutjanidae), from the Persian Gulf and the Oman Sea. Fish Res. 2014; 155:59–63. https://doi.org/10.1016/j.fishres.2014.02.024
Santificetur C, Rossi-Wongtschowski CLDB, Ruperti A, Almeida A, Pinto E, Correia AT. Stock structure of the gulf hake Urophycis cirrata (Teleostei: Phycidae) in South-Western Atlantic using otolith shape and elemental analyses. Fishes. 2025; 10(2):63. https://doi.org/10.3390/fishes10020063
Sbiba SE, Correia AT, Nikolioudakis N, Bazairi H, Chlaida M. Insights into the stock structure of Scomber colias Gmelin, 1789 along the Northwest African coast inferred from otolith shape analysis. Fish Res. 2024; 272:106955. https://doi.org/10.1016/j.fishres.2024.106955
Schulz-Mirbach T, Ladich F, Plath M, Heß M. Enigmatic ear stones: what we know about the functional role and evolution of fish otoliths. Biol Rev. 2019; 94(2):457–82. https://doi.org/10.1111/brv.12463
Schwarzhans W. A comparative morphological study of recent otoliths of the Moridae (Gadiformes). Memorie della Società Italiana di Scienze Naturali e del Museo di Storia Naturale di Milano. 2019; 46:355–70.
Simkiss K. Calcium metabolism of fish in relation to ageing. In: Bagenal TB, editor. Ageing of fish. London: Unwin brothers; 1974. p.1–12.
Soeth M, Spach HL, Daros FA, Adelir-Alves J, Almeida ACO, Correia AT. Stock structure of Atlantic spadefish Chaetodipterus faber from Southwest Atlantic Ocean inferred from otolith elemental and shape signatures. Fish Res. 2019; 211:81–90. https://doi.org/10.1016/j.fishres.2018.11.003
Sokal RR, Rohlf FJ. Biometry: the principles and practice of statistics in biological research. 3rd Edition. New York: W.H. Freeman and Company; 1995.
Souza GM, Tubino RA, Monteiro-Neto C, Costa MR. Relationships between fish and otolith dimensions of Pomatomus saltatrix (Linnaeus, 1766) (Perciformes: Pomatomidae) in southeastern Brazil. Neotrop Ichthyol. 2019; 17(1):e180032. https://doi.org/10.1590/1982-0224-20180032
Stransky C, MacLellan SE. Separação de espécies e zoogeografia de cantarilhos e peixes-rocha (gênero Sebastes) por análise da forma do otólito. Can J Fish Aquat Sci. 2005; 62 (10):2265–76. https://doi.org/10.1139/f05-143
Taylor MD, Fowler AM, Suthers IM. Insights into fish auditory structure-function relationships from morphological and behavioural ontogeny in a maturing sciaenid. Mar Biol. 2020; 167(2):21. https://doi.org/10.1007/s00227-019-3619-9
Tuset VM, Lozano IJ, González JA, Pertusa JF, García-Díaz MM. Shape indices to identify regional differences in otolith morphology of comber, Serranus cabrilla (L., 1758). J Appl Ichthyol. 2003; 19(2):88–93. https://doi.org/10.1046/j.1439-0426.2003.00344.x
Tuset, VM, Lombarte A, Assis CA. Atlas otólito para o Mediterrâneo Ocidental, Atlântico Norte e Centro-Leste. Sci Mar. 2008; 72(S1):7–198. https://doi.org/10.3989/scimar.2008.72s17
Tuset VM, Imondi R, Aguado G, Otero-Ferrer JL, Santschi L, Lombarte A et al. Otolith patterns of rockfishes from the Northeastern Pacific. J Morphol. 2015; 276(4):458–69. https://doi.org/10.1002/jmor.20353
Tuset VM, Otero-Ferrer JL, Siliprandi C, Manjabacas A, Marti-Puig P, Lombarte A. Paradox of otolith shape indices: routine but overestimated use. Can J Fish Aquat Sci. 2021; 78(6):681–92. https://doi.org/10.1139/cjfas-2020-0369
Vaz-dos-Santos AM, Rautenberg KA, Augusto CG, Ballester ELC, Schwingel PR, Pinto E et al. Geographic variation in Opisthonema oglinum (Lesueur, 1818) in the Southeastern Brazilian Bight inferred from otolith shape and chemical signatures. Fishes. 2023; 8(5):234. https://doi.org/10.3390/fishes8050234
Verocai JE, Cabrera F, Lombarte A, Norbis W. Form function of sulcus acusticus of the sagittal otolith in seven Sciaenidae (Acanthuriformes) species using geometric morphometrics (southwestern Atlantic). J Fish Biol. 2023; 103(5):1199–213. https://doi.org/10.1111/jfb.15521
Volpedo AV, Echeverría DD. Ecomorphological patterns of the sagitta in fish on the continental shelf off Argentine. Fish Res. 2003; 60(2–3):551–60. https://doi.org/10.1016/S0165-7836(02)00170-4
Volpedo AV, Vaz-dos-Santos AM. Métodos de estudios con otolitos: principios y aplicaciones. INPA CONICET UBA: Ciudad Autónoma de Buenos Aires, Argentina; 2015.
Authors
![]() César Santificetur1
César Santificetur1 ![]() ,
, ![]() Pollyana Christine Gomes Roque1,2,3,
Pollyana Christine Gomes Roque1,2,3, ![]() Sara de Castro Loebens2,4,
Sara de Castro Loebens2,4, ![]() Alessandra Maria Advíncula Pires2,
Alessandra Maria Advíncula Pires2, ![]() Danielle de Lima Viana2 and
Danielle de Lima Viana2 and ![]() Paulo Guilherme Vasconcelos de Oliveira2
Paulo Guilherme Vasconcelos de Oliveira2
[1] Laboratório de Diversidade, Ecologia e Evolução de Peixes – DEEP Lab, Instituto Oceanográfico da Universidade de São Paulo (IOUSP), Praça do Oceanográfico 191, 05508-120, São Paulo, SP, Brazil. (CS) csantificetur@gmail.com (corresponding author), (PCGR) pollyana_cgr@hotmail.com.
[2] Laboratório de Etologia de Peixes – LEP, Universidade Federal Rural de Pernambuco (UFRPE), Rua Dom Manuel de Medeiros s/n, 51171-900, Recife, PE, Brazil. (AMAP) alessandraapires@outlook.com, (DLV) vianadl@yahoo.com, (PGVO) oliveirapg@hotmail.com.
[3] Laboratório de Ecologia de Peixes Marinhos (LEPMAR), DCAB/UFES, BR-101, km 60 – Litorâneo, São Mateus, ES 29932-540, Brazil.
[4] Laboratório de Estudos em Ecossistemas Oceânicos e Recifais – LECOR, Departamento de Oceanografia, Universidade Federal de Pernambuco, Av. Arquitetura, s/n, Cidade Universitária, 50740-550, Recife, PE, Brazil. (SCL) sara.loebens2@gmail.com.
Authors’ Contribution 

César Santificetur: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Pollyana Christine Gomes Roque: Conceptualization, Data curation, Formal analysis, Investigation, Writing-original draft, Writing-review and editing.
Sara de Castro Loebens: Data curation, Investigation, Writing-review and editing.
Alessandra Maria Advíncula Pires: Data curation, Visualization, Writing-review and editing.
Danielle de Lima Viana: Project administration, Writing-review and editing.
Paulo Guilherme Vasconcelos de Oliveira: Funding acquisition, Supervision, Writing-review and editing.
Ethical Statement
The data used in this research were obtained within the scope of the projects “Biodiversidade e Ecologia dos Peixes Demersais Marinhos em Águas Profundas no Estado de Pernambuco” and “Ecologia e Pesca de Espécies Pelágicas Oceânicas e Demersais de Profundidade na Costa Nordeste e Ilhas Oceânicas do Brasil”, both coordinated by Prof. Fábio Hazin (in memoriam), and were fully approved by the Brazilian Ministry of the Environment through the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio), under permit numbers 53702–3 and 39978–4. The animal capture methods were approved by the Commission of Ethics on the Use of Animals of the Universidade Federal Rural de Pernambuco (licenses 044/2016, protocol 23082.025800/2015; and 084/2018, protocol 23082.016332/2018–86).
Competing Interests
The author declares no competing interests.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
The data used in this study were generated with funding from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES: grant #43/2013) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq: grants #442884/2015–0 and #438961/2018–8).
How to cite this article
Santificetur C, Roque PCG, Loebens SC, Pires AMA, Viana DL, Oliveira PGV. Differences in sagittal otoliths between two morid species: insights into the genus Physiculus (Moridae: Gadiformes). Neotrop Ichthyol. 2026; 24(1):e250113. https://doi.org/10.1590/1982-0224-2025-0113
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted November 1, 2025
Accepted November 1, 2025
![]() Submitted June 26, 2025
Submitted June 26, 2025
![]() Epub March 06, 2026
Epub March 06, 2026