![]() Francisco Javier Luque1
Francisco Javier Luque1 ![]() and
and ![]() Gabriel J. Colorado Z.2
Gabriel J. Colorado Z.2
PDF: EN XML: EN | Supplementary: S1 | Cite this article
Abstract
Este estudio evaluó los patrones ecomorfológicos de los ensamblajes de peces en los ríos oligotróficos de cabecera al noroeste de la Amazonía colombiana. Nuestros objetivos fueron: 1) identificar correlaciones entre las variables morfológicas de los peces y el uso de hábitat, estructura trófica y distancia al río principal; 2) examinar patrones ecomorfológicos que podrían reflejar aspectos ecológicos de la locomoción, uso de hábitat y ecología trófica. Un total de 34 especies fueron analizadas usando 27 índices ecomorfológicos para probar si estos estaban relacionados con el ensamblaje de peces. Se encontraron correlaciones significativas entre rasgos anatómicos y gremios tróficos y uso de hábitat controlando las relaciones filogenéticas. Dos tendencias fueron identificadas dentro del espacio ecomorfológico. Primera, nectónicos insectívoros-piscívoros con cuerpos lateralmente comprimidos, ojos grandes, boca terminal-superior sin barbicelos y aletas anales grandes asociados con especies de nado continuo y uso de visión para capturar presas. Segunda, bénticos perifitovoros-invertívoros con cuerpos deprimidos, ojos pequeños, boca ventral o inferior con presencia de barbicelos y aletas pectorales desarrolladas asociadas con especies que explotan el sustrato de los ríos. Este estudio evidencia que los ensamblajes de peces en los ríos oligotróficos de cabecera se encuentran estructurados por los gremios tróficos y el uso de hábitat. Estos resultados resaltan la importancia del uso de la ecomorfología como una herramienta para predecir patrones ecológicos en estos ríos.
Palabras clave: Filogenia, Gremios tróficos, Guaviare, Morfología, Uso de hábitat.
Introduction
Ecomorphology analyzes the relationships between morphological traits, ecology, and how they relate to the environment (Bock, 1994; Wainwright, Richard, 1995). The main premise of ecomorphology suggests that species with similar morphology share similar resources (Fernández et al., 2012). Under this conceptual basis, organismal morphology allows the identification of biological roles, the environment where the species live in (Wootton, 1990), how species exploit and uses resources (Wainwright, Richard, 1995; Hugueny, Pouilly, 1999), the niche they occupy and, in general, how species assemblage is structured (Conde-Saldaña et al., 2017) through species anatomy (Gatz Jr., 1979). Thus, ecomorphological analysis provides a framework to understand and predict ecological patterns for the use of spatial and trophic resources.
Nevertheless, several studies have shown a lack of consensus on whether morphology is correlated to ecology (Bhat, 2005; Oliveira et al., 2010). This disagreement has extended to the paraphyletic group of fishes, the most taxonomically diverse vertebrate group on earth, for which ecomorphology represents a cornerstone discipline of research given their variety of forms and functions. For example, evidence suggests that depressed bodies exploit benthonic habitats, and that compressed bodies inhabit structured habitats (Gatz Jr., 1979; Cochran-Biederman, Winemiller, 2010; Soares et al., 2013) or that long digestive tracts are strongly related to herbivory and, in turn, head and mouth size is related to prey size (Ferreira, 2007; Cochran-Biederman, Winemiller, 2010; Oliveira et al., 2010). Nevertheless, other studies differed in these findings by suggesting a weak relationship between the morphology of the organism and its environment (Motta et al., 1995; Casatti, Castro, 2006; Conde-Saldaña et al., 2017). This discrepancy has been associated with phylogenetic influence (Douglas, Matthews, 1992; Motta et al., 1995), diverse statistical approaches to test hypothesis (Ricklefs, Miles, 1994), varied and inadequate selection of anatomical traits reliable to predict diet and habitat (Oliveira et al., 2010), and the influence of environmental and habitat variables (Lombarte et al., 2012; Oikonomou et al., 2018; Luiz et al., 2022).
From headwaters to downstream, streams increase in size, discharge, number of habitats and a myriad of physical, chemical, and ecological parameters along the longitudinal gradient within the dendritic stream network (Bistoni, Hued, 2002; Grenouillet et al., 2004). Some studies have investigated how fish assemblages and ecomorphological patterns respond to altitudinal changes (Conde-Saldaña et al., 2017), current velocity (Casatti, Castro, 2006), hydrological gradient (Luiz et al., 2022), and habitat features (Oliveira et al., 2010; Prado et al., 2016; Oikonomou et al., 2018). However, the effect of distance from source on ecomorphological patterns have received little attention, some studies have found that richness, trophic guild proportions and fish assemblages changed along the position of tributaries along the longitudinal continuum of tropical and temperate streams (Rahel, Hubert, 1991; Oberdorff et al., 1993; Fialho et al., 2007). Indeed, distance to large rivers has shown to be relevant in explaining the diversity and structure of fish assemblages at different spatial scales (e.g., Stegmann et al., 2019). For example, some adventitious streams may have richer fish communities than similar-size streams located further up in the network due to migration from larger rivers (Schaefer, Kerfoot, 2004).
Despite the vast diversity and endemism of fishes in tropical freshwater ecosystems, most ecomorphological studies have been carried out on temperate freshwater ecosystems (Bhat, 2005). However, some ecomorphological research have aimed to describe these types of relationships in different Amazonian aquatic environments such as white waters and its floodplains ( Pouilly et al., 2003; Ibañez et al., 2007; Mérona et al., 2008), but a few had addressed such studies on oligotrophic headwater streams (Ramírez et al., 2015). These are small, nutrient poor, dark water acidic streams that support a diverse and rich fish fauna, highly dependable on allochthonous inputs and abiotic conditions such as heavy rains that dramatically change water levels (Correa, Winemiller, 2018). Therefore, oligotrophic headwater streams represent an opportunity to test ecomorphological hypotheses encompassing an understudied geographical area.
In this study we examined ecomorphological patterns of fish assemblages in oligotrophic streams. Our aims were to: 1) Identify and assess correlations between morphological variables of fish species with habitat use, trophic guilds, and distance to the Guaviare River while controlling for phylogenetic relationships, and 2) describe possible ecomorphological patterns that reflect ecological aspects of locomotion, habitat use, and trophic ecology. We hypothesized that species with similar morphology will share both habitat and trophic guild, as well as exhibit similar functional roles, feeding, and locomotion behaviors. Additionally, fish assemblages close to the Guaviare River will present higher phylogenetic and functional richness than those closer to headwater (Stegmann et al., 2019). In this sense, the morphology of the species would differ among trophic guild by resource use, whereas it would differ in habitat use by physical, chemical, and environmental characteristics (Oliveira et al., 2010).
Material and methods
Study area. This study was conducted in headwater oligotrophic streams in the Guaviare River basin near the municipality of San José del Guaviare, Guaviare Department, northwestern part of the Colombian Amazon. The sampling area is located within the Guiana shield region, corresponding to a transition zone between the Amazon rain forest and the Orinoco grasslands (Huber, 2006; Medina-Rangel et al., 2019). The area has a tropical rainforest regime, with an annual precipitation average of 2,800 mm and a monomodal regime (dry period from November to February). Average temperature and humidity are 25.7 oC and 78–90%, correspondingly (Cárdenas-López et al., 2008). Streams headwaters are located within the Serranía de la Lindosa, a protected area with an extension of 12,000 ha. According to Lasso (2014), these streams are part of a system of white waters, affluents to the Guaviare River and part of the Orinoco River basin. Vegetation is composed by riparian forests, rocky outcrops shrubs and forests, white sand stands, and grasslands (Atuesta-Ibargüen, 2019).
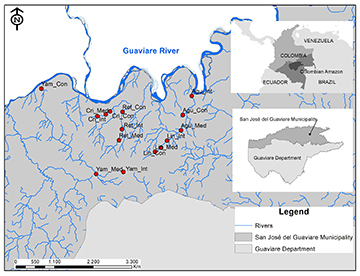
Field sampling. 15 sampling stations were established from ~1 to 30 km (mean ± SD: 14.53 km ± 7.60) from the main stream (Guaviare River), ranging from 185 to 285 m in elevation (Tab. 1). Station selection was based on the following criteria: 1) landowner access permission; 2) physical security; and 3) good accessibility to ensure repeatability. At each station, a reach of 350–400 m was established. Minimum distance between reaches was 3 km, maximizing independence among study reaches. Each reach was divided into three sections (i.e., Upstream, Midstream, and Downstream) following the methods used by (Torres-Bejarano et al., 2022) (Fig. 1).
TABLE 1 | Physical characteristics in 15 sampling stations in oligotrophic headwater streams of northwestern Amazonia, Guaviare Department, Colombia.
Sampling Station | Distance from river (km) | Elevation (m) | Habitat type | Mean depth (m) | Mean flow velocity (m/s) | Mean width (m) | Mean flow (m³/s) | Geographical coordinates | |
N | W | ||||||||
Yam_Med | 23.17 | 235 | Riffle | 0.50 ± 0.18 | 0.31 ± 0.16 | 5.58 ± 0.79 | 0.8649 | 2°27’13.70"S | 72°45’17.80"W |
Yam_Int | 29.68 | 281 | Riffle | 0.36 ± 0.04 | 0.44 ± 0.05 | 5.37 ± 1.45 | 0.850608 | 2°27’25.90"S | 72°42’40.30"W |
Yam_Con | 1.17 | 200 | Pool | 0.80 ± 0.64 | 0.06 ± 0.61 | 11.58 ± 4.83 | 0.55584 | 2°35’26.70"S | 72°50’34.20"W |
Ret_Med | 20.95 | 219 | Pool | 0.60 ± 0.18 | 0.05 ± 0.07 | 9.48 ± 0.54 | 0.2844 | 2°30’29.00"S | 72°43’5.00"W |
Ret_Int | 17.98 | 242 | Riffle | 0.48 ± 0.26 | 0.33 ± 0.18 | 5.34 ± 4.72 | 0.845856 | 2°31’32.70"S | 72°42’48.80"W |
Ret_Con | 14.4 | 212 | Pool | 0.60 ± 0.32 | 0.11 ± 0.15 | 5.51 ± 2.28 | 0.36366 | 2°33’13.10"S | 72°42’44.90"W |
Lin_Med | 19.19 | 219 | Pool | 0.83 ± 1.11 | 0.08 ± 0.16 | 7.45 ± 3.46 | 0.49468 | 2°29’21.71"S | 72°39’37.92"W |
Lin_Int | 12.65 | 203 | Pool | 0.96 ± 0.08 | 0.02 ± 0.17 | 20.57 ± 2.22 | 0.394944 | 2°30’26.40"S | 72°38’27.10"W |
Lin_Con | 16.65 | 208 | Riffle | 0.40 ± 0.02 | 0.17 ± 0.1 | 14.96 ± 6.88 | 1.01728 | 2°29’45.60"S | 72°39’2.20"W |
Agu_Med | 2.43 | 189 | Pool | 1.07 ± 0.46 | 0.02 ± 0.07 | 23.48 ± 2.42 | 0.502472 | 2°31’25.80"S | 72°37’7.50"W |
Agu_Int | 7.23 | 185 | Pool | 0.73 ± 1.03 | 0.03 ± 0.54 | 14.46 ± 12.2 | 0.316674 | 2°34’45.50"S | 72°36’4.90"W |
Agu_Con | 11.04 | 200 | Pool | 1.08 ± 0.17 | 0.03 ± 0.11 | 8.86 ± 1.83 | 0.287064 | 2°32’55.00"S | 72°36’57.30"W |
Cri_Med | 17.94 | 196 | Riffle | 0.26 ± 0.09 | 0.38 ± 0.11 | 2.1 ± 0.85 | 0.20748 | 2°32’47.80"S | 72°45’10.70"W |
Cri_Int | 13.27 | 203 | Riffle | 0.43 ± 0.30 | 0.5 ± 0.13 | 2.94 ± 0.71 | 0.6321 | 2°32’58.10"S | 72°44’21.70"W |
Cri_Con | 10.27 | 200 | Riffle | 0.54 ± 0.36 | 0.26 ± 0.08 | 3.97 ± 0.98 | 0.557388 | 2°33’15.50"S | 72°43’56.60"W |
FIGURE 1| Study area with sampling station location in oligotrophic headwater streams of northwestern Amazonia, Guaviare Department, Colombia.
Environmental variables. Channel width was considered as the distance between stream margins and measured with a measuring tape. Stream velocity was calculated using a FlowWatch (JDC electronic SA, Switzerland) and stream flow by multiplying mean width (m), mean depth (m), and mean velocity (m/s). Two habitats were evaluated, riffles and pools. Riffles were characterized by fast moving water and shallower depth, whereas pools showed slower flows and deeper waters (Tab. 2).
TABLE 2 | Physical characteristics of the evaluated habitats.
Habitats | Characteristics | |||
| Mean depth (m) | Mean width (m) | Mean flow (m3/s) | Mean velocity (m/s) |
Riffles | 0.44 | 4.78 | 0.7 | 0.34 |
Pools | 0.83 | 8.85 | 0.39 | 0.04 |
Fish collection and morphological data. Fish were collected at study reaches once during the wet and once during the dry season using active seining (30 m x 2 m with 2.5 cm mesh) from downstream to upstream and passive gill netting (15 m x 3 m with 2.5 cm mesh) at both ends of the reach closing it and preventing the entry and exit of fish. Our protocols followed those proposed by Anjos, Zuanon (2007) and Torres-Bejarano et al. (2022), which support that ~240 m is sufficient to adequately represent fish richness in small Amazonian streams. Our sampling efforts included 70–90 min collections over ~300 m2 at each study reach. The fish collected were anesthetized and euthanized with MS–222 (200 mg/L), and frozen for posterior analysis in the laboratory. All individuals were identified to species using taxonomic keys (Géry, 1977; Gálvis et al., 2006, 2007; Kullander, 2006; Urbano-Bonilla et al., 2017), and expert assistance when necessary. Species were classified into trophic guilds (i.e., detritivores, insectivores, invertivores, periphytivores, and piscivores) using published information (Gálvis et al., 2006,2007; van der Sleen, Albert, 2018; Froese, Pauly, 2023).
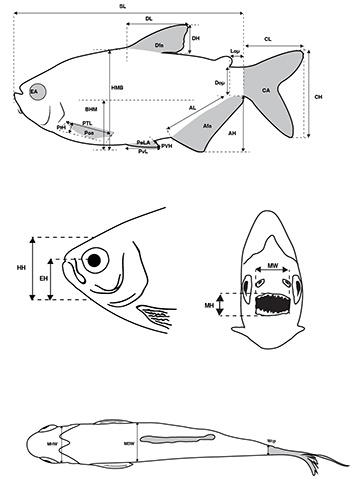
Twenty three linear morphometric measurements and six areas were taken on the left side of the individuals using a digital caliper (0.01 mm precision), whereas areas of eyes and fins were obtained from photographs, and calculated using AutoCAD software (Fig. 2). Only species with more than five adult individuals were measured, considering that rare species could reflect non-interactive components of main community (Winemiller, 1991), small samples limit the detection of specific morphological variations (Oliveira et al., 2010), and ontogenetic growth can promote significant changes in body shape (Bower, Piller, 2015), habitat use (Sagnes et al., 1997) and feeding habits (Oliveira et al., 2010; Prado et al., 2016). After all measurements were taken, specimens were fixed with 10% concentration formalin and then transferred to 70% ethanol solution and deposited in the Ichthyological collection of Instituto de Ciencias Naturales (ICN) of Universidad Nacional de Colombia, Bogotá, Colombia.
FIGURE 2| Linear morphometric measurements and calculated areas: standard length (SL), dorsal-fin length (DL), caudal peduncle length (Lcp), caudal fin length (CL), pectoral-fin length (PTL), pelvic-fin length (PvL), anal-fin length (AL), mouth width (MW), maximum body height (HMB), midbody height (BHM), dorsal fin height (DH), caudal peduncle depth (Dcp), caudal fin height (CH), pectoral fin height (Pth), pelvic fin height (PVH), anal fin height (AH), head height (HH), eye height (EH), mouth width (MW), mouth height (MH), maximum head width (MHW), maximum body width (MBW), caudal peduncle width (WCP), eye area (EA), dorsal fin area (Dfa), caudal fin area (CA), anal fin area (Afa), pelvic fin area (PeLA), and pectoral fin area (Pca). Based on a specimen of Astyanax bimaculatus.
Based on these criteria, 34 of 62 species captured were measured and considered for further analysis (Tab. 3). Linear measurements and areas were used to calculate 27 ecomorphological indices related to locomotion, habitat use, and trophic ecology (Tab. 4). Additionally, two ecological traits that do not involve measurements were considered for the analysis: presence of barbels and mouth position. Both traits are related to trophic guild and habitat use (Tab. 5). These ecomorphological traits are unitless ratios, so they are not correlated with body size (Oikonomou et al., 2018), allowing comparisons between species or individuals of different sizes and focus on function and ecological aspects of morphology while minimizing the confounding effects of body size (Bower, Piller, 2015) .
TABLE 3 | Fish species of the oligotrophic headwater streams in northwestern Amazonia with trophic guilds, habitat association and distance from source group.
Order/Family/Species | Code | Measured specimens | Habitat type | Distance to river (km) | Trophic guild |
Characiformes | |||||
Acestrorhynchidae | |||||
Acestrorhynchus falcatus (Bloch, 1794) | Ac_fal | 5 | Pool | 0 – 5 | Piscivore |
Bryconidae | |||||
Brycon whitei Myers & Weitzman, 1960 | Br_whi | 11 | Pool | 6 – 10 | Piscivore |
Acestrorhamphidae | |||||
Astyanax integer Myers, 1930 | As_int | 20 | Pool | >15 | Insectivore |
Astyanax bimaculatus (Linnaeus, 1758) | As_bim | 20 | Pool | 0 – 5 | Omnivore |
Astyanax sp.2 | A_sp2 | 20 | Pool | 6 – 10 | Insectivore |
Bario oligolepis (Günther, 1864) | Mo_oli | 17 | Pool | 6 – 10 | Insectivore |
Hemigrammus barrigonae Eigenmann & Henn, 1914 | He_bar | 20 | Riffle | >15 | Omnivore |
Hyphessobrycon acaciae García-Alzate, Román-Valencia & Prada-Pedreros, 2010 | Hy_aca | 20 | Riffle | 0 – 5 | Omnivore |
Hyphessobrycon taguae García-Alzate, Román-Valencia & Taphorn, 2010 | Hy_tag | 20 | Riffle | 11 – 15 | Omnivore |
Characidae | |||||
Charax metae Eigenmann, 1922 | Ch_met | 10 | Pool | 11 – 15 | Piscivore |
Stevardiidae | |||||
Creagrutus calai Vari & harold, 2001 | Cr_cal | 20 | Riffle | >15 | Insectivore |
Moenkhausia comma Eigenmann, 1908 | Mo_com | 15 | Pool | 0 – 5 | Insectivore |
Moenkhausia mikia Marinho & Langeani, 2010 | Mo_mik | 16 | Pool | 0 – 5 | Insectivore |
Tyttocharax metae Román-Valencia, García-Alzate, Ruiz-C. & Taphorn, 2012 | Ty_met | 5 | Riffle | 6 -10 | Omnivore |
Curimatidae | |||||
Cyphocharax oenas Vari, 1992 | Cy_one | 5 | Pool | 6 – 10 | Detritivore |
Steindachnerina argentea (Gill, 1858) | St_arg | 6 | Pool | 0 – 5 | Detritivore |
Erythrinidae | |||||
Hoplerythrinus unitaeniatus (Spix & Agassiz, 1829) | Ho_uni | 5 | Pool | 6 – 10 | Piscivore |
Hoplias malabaricus (Bloch, 1794) | Ho_mal | 8 | Pool | 6 – 10 | Piscivore |
Iguanodectidae | |||||
Bryconops giacopinii (Fernández-Yépez, 1950) | Br_gia | 20 | Pool | 11 – 15 | Insectivore |
Lebiasinidae | |||||
Copella arnoldi (Regan, 1912) | Co_arn | 20 | Pool | 11 – 15 | Omnivore |
Pyrrhulina lugubris Eigenmann, 1922 | Py_lug | 20 | Pool | 11 – 15 | Insectivore |
Cichliformes | |||||
Cichlidae | |||||
Aequidens tetramerus (Heckel, 1840) | Ae_tet | 18 | Pool | 0 – 5 | Invertivore |
Apistogramma hoignei Meinken, 1965 | Ap_hoi | 16 | Pool | 11 – 15 | Invertivore |
Bujurquina mariae (Eigenmann, 1922) | Bu_mar | 15 | Pool | 0 – 5 | Omnivore |
Satanoperca mapiritensis (Fernández-Yépez, 950) | Sa_map | 7 | Pool | 0 – 5 | Invertivore |
Saxatilia alta (Eigenmann, 1912) | Cr_alt | 8 | Pool | 6 – 10 | Piscivore |
Cyprinodontiformes | |||||
Rivulidae | |||||
Anoblepsoides sp. | An_sp1. | 19 | Pool | 6 – 10 | Omnivore |
Siluriformes | |||||
Auchenipteridae | |||||
Duringlanis romani (Mees, 1988) | Ce_rom | 7 | Riffle | 0 – 5 | Invertivore |
Callichthyidae | |||||
Hoplisoma melini (Lönnberg & Rendhal, 1930) | Co_mel | 6 | Riffle | 0 – 5 | Invertivore |
Loricariidae | |||||
Ancistrus triradiatus Eigenmann, 1918 | An_tri | 6 | Riffle | >15 | Detritivore |
Farlowella vittata Myers, 1942 | Fa_vit | 17 | Riffle | 0 – 5 | Detritivore |
Rineloricaria eigenmanni (Pellegrin, 1908) | Ri_eig | 18 | Riffle | 0 – 5 | Detritivore |
Heptapteridae | |||||
Pimelodella metae Eigenmann, 1917 | Pi_met | 15 | Pool | 0 – 5 | Invertivore |
Trichomycteridae | |||||
Ituglanis metae (Eigenmann, 1917) | It_met | 17 | Riffle | 0 – 5 | Insectivore |
TABLE 4 | Ecomorphological indices used in the present study and their respective code, formulae, ecological relation, and biological interpretation.
Ecomorphological index | Code | Code | Formula | Related to | Biological interpretation |
Compression index | Com_ind | Ci | MBH/MBW | Habitat use | High values indicates that fish is laterally compressed, which could be related to the exploit of slow water velocity habitats (Watson, Balon, 19984) |
Depression index | De_ind | Di | BMH/MBH | Habitat use | Low values indicates that fish presents a depressed body, which could be related to the exploit of bottom water column (Watson, Balon, 1984) |
Relative depth | Re_dep | Rd | MBH/SL | Habitat use | Low values could indicate fish occupy high velocity habitats (Gatz Jr., 1979; Watson, Balon, 1984) |
Eye position | Eye_pos | EP | EH/Hd | Habitat use | High values indicate dorsally positioned eyes, often found on benthic species. Low values indicates laterally positioned eyes, found on nektonic species (Poully et al., 2003; Watson, Balon, 1984) |
Relative area of pelvic fin | Re_area_pfin | Rap | PeL/(SL)2 | Habitat use | High values are related to benthic species whereas lower values to nektonic species (Breda, 2005; Gatz Jr., 1979) |
Aspect ratio of pelvic fin | As_ra_pfin | Arp | (PeL)2/PeA | Habitat use | High values are related to nektonic habits whereas lower values to benthic habits since fins are use as support on the substrate (Breda, 2005; Gatz Jr., 1979) |
Caudal peduncle trottle | Ca_pen_ro | Cpt | Cfd/Cpd | Locomotion | High values are related to caudal propulsion efficiency for fast movements through a reduction in drag (Luiz et al., 2022) |
Aspect ratio caudal fin | As_ra_cfin | Arc | Cfd/Cfa | Locomotion | High values are present in species have active and continuous swimming habits (Balon et al., 1986) |
Relative area caudal fin | Re_area_cfin | Rac | Cfa/(SL)2 | Locomotion | Larger values are associated with active and continuous swimming habits (Balon et al., 1986) |
Caudal peduncle compression index | ca_pen_comin | Cpc |
| Locomotion | High values are present in species with low swimming activity and low manuverability (Gatz Jr., 1979; Watson, Balon, 1984) |
Relative length caudal peduncle | Re_len_capend | Rlc | Lcp/SL | Locomotion | Larger values are related to species with good swimming capacity and fish that inhabit fast water flow habitats (Breda , 2005; Watson, Balon, 1984; Winemiller, 1991) |
Relative height caudal peduncle | Rehig_capend | Rhc | Hcp/MBH | Locomotion | Low values are associated to species withhigh manuvelavility capacity (Oliveira et al., 2010; Winemiller, 1991) |
Relative width caudal peduncle | Re_wid_capend | Rwc | Wcp/MBW | Locomotion | Low values areassociated to species with less capacity for continuous swimming (Winemiller, 1991) |
Relative area dorsal fin | Re_area_dfin | Rad | Dfa/(SL)2 | Locomotion | High values provide greater stabilization (Breda, 2005; Gosline, 1971) |
Relative area anal fin | Re_area_afin | Raa | Afa/(SL)2 | Locomotion | High values provide greatar stabilization and movement capacity (Breda, 2005; Wolff, 2008) |
Aspect ratio anal fin | As_ra_afin | Ara | Afl/Afa | Locomotion | High values are related to the capacity to perfrm fast forward and backwards movement (Breda, 2005; Wolff, 2008) |
Relative area pectoral fin | Re_area_pecfin | Rape | Pca/(SL)2 | Locomotion / habitat use | Low values are present in species that inhabit slow flowing waters. High values are present in slow swiming species or species with benthic habitats that deflect water to stay as close as possible to the substrate (Watson, Balon, 1984) |
Aspect ratio pectoral fin | As_ra_pecfin | Arpe | (Pcl)2/Pca | Locomotion | High values are present in species that swim continuously, can reach higher speeds or migrate (Breda, 2005; Watson, Balon, 1984) |
Relative eye size | Rel_eye_si | Res | Ea/(SL)2 | Trophic | Use as an aprroximation for vision capacity. Larger eyes are related to food detection and its use for predatory activity (Poully et al., 2003) |
Relative height of head | Re_hehead | Rhh | MHH/MBH | Trophic | Related to predatory capacity. Larger values are related to species that feed on larger preys (Wolff, 2008) |
Relative width of head | Re_wihead | Rwh | MHW/MBW | Trophic | Related to predatory capacity. Larger values are related to species that feed on larger preys (Soares et al., 2013) |
Relative height of mouth | Re_hemou | Rhm | MMH/MBH | Trophic | Related to predatory capacity, indicating the size of prey items. High values are indicate species that feed on larger preys (Watson, Balon, 1984) |
Relative width of mouth | Re_wimou | Rwm | MWM/MBW | Trophic | Related to predatory capacity, indicating the size of prey items. High values are indicate species that feed on larger preys (Watson, Balon, 1984) |
Oral gap size | O_gap_si | Ogs | (Mmw*Mmh)/(SL)2 | Trophic | Related to predatory capacity, indicating the size of prey items. High values are indicate species that feed on larger preys (Luiz et al., 2022) |
Oral gap shape | O_gap_sh | Osgh | Mmh/Mmw | Trophic | Related to predatory capacity, indicating the size of prey items. High values are indicate species that feed on larger preys (Luiz et al., 2022) |
Barbels | Barb | Bar | 0 = ausent 1 = present | Trophic / habitat use | Presence of barbels are related to species that primarly feed and inhabit the bottom of the water column (Hugueny, Poully, 1999; Conde-Saldaña et al., 2017) |
Mouth position | Mo_pos | Mp | 1 = superior 2 = terminal 3 = subterminal 4 = inferior 5 = ventral | Trophic / habitat use | Position of mouth indicates the location in the water column where the species obtain its food. Inferior and vental mouths are present in benthic species where as terminal and superior mouth are present in nektonic species (Gatz Jr., 1979; Conde-Saldaña et al., 2017) |
TABLE 5 | Contribution of the ecomorphological indices in the first three axes of the Principal Components Analysis (PCA). Indices with higher correlations values for each principal component are shown in bold and were selected for interpretation. See Tab. 3 for the list ecomorphological indices abbreviations.
Ecomorphological index | PC 1 | PC 2 | PC 3 |
Com_ind | -0.799 | -0.11 | 0.441 |
De_ind | -0.934 | -0.065 | -0.127 |
Re_dep | -0.884 | -0.071 | 0.008 |
Rel_eye_si | -0.913 | 0.053 | -0.302 |
Eye_pos | 0.948 | 0.088 | -0.093 |
Re_area_pfin | 0.287 | -0.87 | -0.214 |
As_ra_pfin | -0.749 | 0.431 | 0.388 |
Ca_pen_tro | -0.438 | -0.345 | -0.207 |
As_ra_cfin | -0.559 | -0.423 | 0.278 |
Re_area_cfin | -0.248 | -0.88 | -0.057 |
Ca_pen_comin | -0.59 | -0.357 | 0.596 |
Re_len_capend | 0.942 | 0.099 | -0.083 |
Re_hig_Capend | 0.736 | 0.267 | -0.07 |
Re_wid_capend | -0.537 | -0.16 | 0.374 |
Re_area_dfin | -0.07 | -0.66 | -0.123 |
Re_area_afin | -0.849 | 0.044 | -0.034 |
As_ra_afin | -0.682 | 0.272 | 0.184 |
Re_area_pecfin | 0.334 | -0.819 | -0.121 |
As_ra_pecfin | -0.316 | 0.727 | 0.28 |
Re_hehead | -0.601 | -0.222 | -0.571 |
Re_wihead | -0.16 | 0.373 | -0.774 |
Re_hemou | -0.649 | 0.082 | -0.592 |
Re_wimou | -0.485 | 0.303 | -0.755 |
O_gap_si | -0.383 | -0.026 | -0.847 |
O_gap_sh | -0.807 | 0.144 | -0.029 |
Barb | 0.955 | 0.026 | -0.073 |
Mo_pos | 0.814 | 0.021 | -0.172 |
Eigenvalue | 7.25 | 5.35 | 4.42 |
Explained variance (%) | 44.9 | 16.6 | 11.4 |
Accumulated variance (%) | 44.9 | 61.5 | 72.9 |
Broken-stick percentage | 14.41 | 10.7 | 8.85 |
Data analyses. Ecomorphological structure. To test the hypothesis that species with similar morphology will share both habitat and trophic guild, independent of phylogeny (taxonomic), a Mantel test analysis was performed. This test assesses the correlation between two distance matrices and the statistical significance is obtained by n randomizations of the input distance matrices. Thus, the analysis seeks to test the hypothesis that the matrices are not linearly or monotonically related (Legendre, Legendre, 2012). To conduct this analysis, we generated a distance matrix containing the morphological information of the fish assemblage (i.e., morphological matrix) and compared it against a set of generated matrices comprising phylogenetic, habitat use, trophic guilds, and distance to river information, as follows:
Morphological matrix was calculated using a Euclidean distance matrix made for each pair of species and based on their ecomorphological indices. This matrix was generated using the average of each species and the decostand function of Vegan package on Rstudio software (R Development Core Team, 2011).
Phylogenetic matrix was constructed following Douglas, Matthews (1992) and Conde-Saldaña et al. (2017). A value of one was attributed to the pairs of congeneric species; a value of 1.5 to those in different genera but same subfamily; a value of two to those in different subfamily but same family; a value of three to those in different families but in the same order; a value of 4 to those of different order but same superorder; and a value of 5 in for those of different superorder. We used this approach as a surrogate of phylogenetic relationships since a complete phylogenetic tree of Colombian amazon freshwater fish species is not currently available.
Habitat use matrix was constructed following Conde-Saldaña et al. (2017), using abundance as a predictor of relationship between fish and their habitats (Rosenfeld, 2003). Since abundance did not differ between dry and wet seasons (non-parametric Kruskall-Wallis, H (1) = 0.0348; p = 0.851), the data was pooled to establish the species association in each habitat type (riffle and pools). The indicator value method (IndVal) Dufrene, Legendre (1997) was used to establish species habitat association; thus, each species was assigned to a type of habitat according to the results of the maximum indicator value for each type of habitat (maxcls). This analysis was performed using the IndVal function of the labdsv package of Rstudio (R Development Team, 2011). A value of one was given to the pair of species that belong to the same habitat type and a value of two for species that did not fulfill this condition (Soares et al., 2013).
Trophic guild matrix was generated using the trophic guild assign to each species; a value of one was attributed to the pair of species belonging to the same guild and a value of two for species belonging to different guilds (Soares et al., 2013).
Distance to river matrixwas constructed following Conde-Saldaña et al. (2017). Four groups with different distances to river were generated by taking the square root of the number of sampling locations (Tab. 2). Abundance data in each sampling site was used to perform the IndVal analysis and classification in each distance to river group was done using the maxcls option from the IndVal function of the labdsv package of Rstudio (R Development Team, 2011). The matrix was constructed giving a value of one to species belonging to the same distance to river group whereas a value of two was given to the pairs of species that did not satisfy this condition (Soares et al., 2013).
To avoid false correlations between morphology and trophic guild, habitat use, and distance to river structures due to phylogenetic effects (Winemiller, 1991; Douglas, Matthews, 1992; Neves et al., 2015), we assessed partial correlations between two matrices (i.e., morphological vs. habitat use, morphological vs. trophic guilds, and morphological vs. distance to river) while controlling for the effect of a third matrix (phylogenetic) using a partial Mantel test. Mantel and partial Mantel tests were carried out using mantel and partial.mantel functions of vegan package Rstudio (R Develompent Core Team, 2011).
Ecomorphological patterns. In order to examine and identify patterns of fish assemblages distribution in the multivariate ecomorphological space and seek for interspecific differences in ecomorphological traits (i.e., locomotion, habitat use, and trophic ecology), we used a Principal Components Analysis (PCA). PCA was applied on the correlation matrix composed by the mean values of the 27 ecomorphological indices previously transformed with log(x+1). Components retained for interpretation were chosen according to the broken-stick model, in which only the components exceeding the pattern of expected eigenvalues were considered significant (Jackson, 1993). Mouth position and barbels were used as categorical variables. These analyzes were performed in Rstudio using the BiodiversityR package (R Development Core Team, 2011).
Results
A total of 3,598 specimens belonging to 62 fish species, 27 families, and five orders were captured (Tab. S1). Of the 34 non-rare species retained for the analysis, we measured a total 472 individuals, where Characiformes was the dominant order followed by Siluriformes, Cichliformes, and Cyprinodontiformes, respectively. With respect to trophic guilds, nine species were classified as insectivores, eight omnivores, six piscivores, six invertivores, and five detritivores (Tab. 3).
Mantel test showed that the morphological pattern of the fish assemblage had a strong association with its phylogeny structure (r = 0.49; p = 0.001), habitat use (r = 0.18; p = 0.003) and trophic guilds (r = 0.296; p = 0.001), and a lack of association with distance to river (r = -0.05; p = 0.835). After controlling for phylogenetic relationships, partial Mantel tests showed that fish morphology was a good predictor of both trophic structure (r = 0.227; p = 0.001) and habitat structure (r = 0.24; p = 0.025) in oligotrophic headwater streams of northwestern Amazonia.
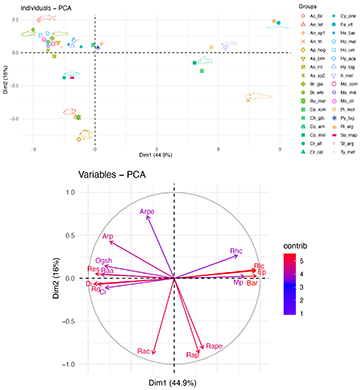
The first two components of PCA explained 60.9 % of total variation in the ecomorphospace (Fig. 3). The ecomorphological traits with greater influence in morphospace were related to feeding and locomotion (PC1, 44.9% of the variation) and habitat use (PC2, 16%). PC1 shows a gradient, with positive loads corresponding depressed bodies, dorsal eyes, small eyes, ventral mouth position, presence of barbels, scores located towards the positive side. These characteristics categorized species as benthic consumers of periphyton or invertebrates; this group was mostly composed by the order of Siluriformes and families such as Trichomycteridae, Heptapteridae, and Loricariidae. Whereas negative scores were associated to laterally compressed bodies, larger anal fins, larger eyes, lateral eyes, and superior and subterminal mouths. These characteristics are related to higher maneuverability and mobility, nektonic habits, and tendency to chase prey such as fish and feed on insects and invertebrates adrift in the water column. These traits are characteristics of characids, cichlids, and cyprinids (Fig. 3). Likewise, PC2 shows a gradient as well. Positive scores were related to the development of pectoral fins, characteristics of benthic species whereas negative scores related to the development of dorsal and caudal fins, characteristics of active swimmers or species that perform explosive movements.
FIGURE 3| Distribution of 34 fish species in the multivariate ecomorphological space generated by the first two PCA axes (above) and the corresponding contribution of ecomorphological indices according to their scores (below). See Tabs. 2–3 for the list of species and ecomorphological indices abbreviations, correspondingly.
Discussion
Ecomorphology theory states that species morphological attributes are correlated with their ecology and environmental factors, therefore it can act as predictor of how species use several resources such as habitat (e.g., occupation in water column), and diet (e.g., foraging behavior) (Casatti, Castro, 2006; Soares et al., 2013). However, few studies have evaluated the predictive ability of this theory in oligotrophic headwater streams of northwestern Amazonia. In this study, habitat use and trophic structure attributes showed a significant correlation with species morphology, while controlling for phylogenetic effects. This suggests that fish assemblage in these oligotrophic headwater streams is ecomorphologically structured, allowing to predict information about habitat and trophic guild based on morphological features, e.g., if two species with similar morphology were sampled, independently of their phylogeny, there is a great probability that they would share same space (habitat) and food resources (trophic guild) (Oliveira et al., 2010). Additionally, it indicates the high capacity of the used indices to describe the trophic differences between morphologically similar species.
Studies evidencing the correlation between trophic ecology and morphology are ambiguous. On one hand, Pouilly et al. (2003), Ward-Campbell et al. (2005), Ferreira (2007;), Oliveira et al. (2010), found significant relationships between morphological traits and diet, whereas Casatti, Castro (2006), Soares et al. (2013), Conde-Saldaña et al. (2017) did not find any significance on these associations. This ambiguity might result from the lack of capacity from several ecomorphological indices to reflect diet and trophic differences among morphological similar species, trophic differences between morphological similar species (Conde-Saldaña et al., 2017), and lack of standardized indices protocols which resulted in researchers measuring same indices in different ways (Villéger et al., 2017). Our results showed significant relationships between anatomy and trophic structure and habitat use, indicating that fish assemblage is not defined by phylogenetic relationships among species nor is randomly structured. However, we highlight that due to our species selection criteria, our analyses only retained nearly half of the species found in the sampled streams (34 out of 62) which, in turn, could have influenced the obtained results. For instance, the input of morphological and ecological different species captured but excluded from our analysis such as Potamorrhaphis guianensis (Jardine, 1843), Eigenmannia virescens (Valenciennes, 1836), Otocinclus huaorani Schaefer, 1997, and Electrophorus electricus (Linnaeus, 1766) might help to better elucidate and strengthen or reject the patterns found.
Our results failed to detect a relationship between the functional patterns (e.g., morphology and trophic guild) of the fish assemblage and distance to large, source rivers. Several studies have found that species richness increase with distance from source, which in turn increase trophic structure complexity, whereas headwater communities showed simpler structures (Vannote et al., 1980; Bistoni, Hued, 2002; Fialho et al., 2007) due mainly to the lack of habitat and resources diversity that increase as streams increases their size. Our finding could result from the short distances these streams flow until they reach the main river, Guaviare River, limiting the turnover of fish species (Stegmann et al., 2019). The furthest sample point, Yam_int (Tab. 1), was only 30 km from the Guaviare River with an altitudinal change of 81 m, and only displayed changes in river substratum when compared to the other two sites downstream (FJL, pers. obs.). Most fish assemblages were reported to change after 150 km or when streams surpasses fourth order (Grenouillet et al., 2004; Fialho et al., 2007). Further studies including landscape position of samples point, habitat features, and other environmental variables are needed to fully understand how these oligotrophic species rich and diverse streams change along longitudinal gradient and within the dendritic network.
In this research, we identified two ecomorphological diversification patterns of headwater streams of northwestern Amazonia based on interspecific differences in ecomorphological traits. The first one is associated with feeding and habitat use. In this context, two groups were observed in the ecomorphological space: 1) piscivorous, omnivores, and insectivores, nektonic species with compressed bodies and 2) detritivores and invertivores, benthic species with depressed bodies. Species of the first group were associated with high maneuverability, larger lateral eyes, developed pectoral fins, larger anal fins large mouths and heads, and compressed bodies. These species use their vision to capture their prey and are good swimmers capable of stabilization of movement (Watson, Balon, 1984; Breda et al., 2005; Conde-Saldaña et al., 2017). For instance, omnivores and insectivores such as Anablepsoides sp., A. bimaculatus and B. giacopinii have small dorsal fins, large eyes and pelvic fins which allowed them to move throughout the water column, use different habitats and consume diverse food items such as insects, fish, leaves, seeds, and fruits (Casatti, Castro, 2006; Conde-Saldaña et al., 2017). Piscivores such as H. malabaricus and A. falcatus,had fusiform bodies that allow high speed and larger heads and mouths, typical of ambush predators that wait in marginal areas and dart to capture their prey (Breda et al., 2005; Ferreira, 2007). The second group, detritivores and invertivores, have developed pectoral fins and caudal peduncles, both fundamental for body stability on streams substrate and short distance movements. Within this group, catfishes from families Loricariidae, Auchenipteridae, and Trichomycteridae were most abundant and had depressed bodies, inferior sucker mouths and dorsal eyes, typical of fishes that exploit intense current habitats (Ferreira, 2007; Oliveira et al., 2010; Prado et al., 2016). In turn, Cichliformes exhibit smaller pectoral fins which provide better stability (Breda et al., 2005) for the use of structurally complex habitats with slow moving waters such as macrophyte stands, roots and backwaters (Breda et al., 2005; Oliveira et al., 2010; Conde-Saldaña et al., 2017).
The second ecomorphological diversification pattern was associated with locomotion. This trend showed a gradient influenced by dorsal fin. Dorsal fin orientate motion as it helps to avoid the rotation of fish along its longitudinal axis (Watson, Balon, 1984; Breda et al., 2005). On one hand, loricariids use this fin as a keel and, together with their developed pectoral fins, helps to keep the body on the bottom, whereas characids use their small dorsal fin and elongated pectoral fins for continuous swimming throughout the water column (Casatti, Castro, 2006). On the other hand, the large two dorsal fins of cichlids greatly improve maneuverability (Breda et al., 2005).
Ecomorphology is a tool that can be used to explore intraspecific and interspecific relationships and variations required to comprehend and predict complex relationships between morphology and ecological specializations and generalizations (Soares et al., 2013; Prado et al., 2016). Ecomorpholgy can be use to understand how fish assemblages are affected by environmental changes such as logging, inflow change, and habitat degradation (Lombarte et al., 2012; Jacob et al., 2021). This is particularly necessary in the northwestern Amazonia as anthropogenic threats, including deforestation and expansion of the agricultural frontier (Armenteras et al., 2013, 2019; Armenteras, Rodríguez Eraso, 2014), cattle ranching (Etter et al., 2006a,b), illicit crops (Dávalos et al., 2011), have increased since peace agreements ended armed conflict in Colombia (Bogota-Gregory et al., 2024). These threats can pose strong deleterious effects on fish fauna by altering streams integrity (Barletta et al., 2010; Pelicice et al., 2017). Our results showed that fish assemblages in headwater oligotrophic streams of northwestern Amazonia are spatially and trophically structured, meaning assemblages could be negatively affected by changes in habitat structure along the streams. Thus, future studies in these systems must incorporate the effect of anthropogenic disturbance on the structure and functionality of fish assemblages.
This study provides novel information that can be used for effective management and conservation efforts in this highly threatened region of the Amazonia, since it contributes to understanding how fish assemblages are structured and how they can be affected by current anthropogenic activities. Also, our results highlighted the use of morphological traits as a tool to understand fish assemblage patterns and the ecological interactions that affect them and their dynamics (Lombarte et al., 2012). Further studies should include more species; analyze new possible ecomorphological patterns that provide links between global changes, functional diversity and ecosystems services (e.g., mediation of nutrient cycling through excretion and egestion, results that can be associated to body mass using allometric coefficients). Additionally, the development of a phylogenetic tree to better differentiate species phylogenetic relatedness overcoming the weakness that might render the approach used in this research. Finally, we recommend that ecomorphology should be incorporated into the research of fish communities and advocate for a standardized protocols to avoid measuring same indices in different ways, promoting consistency, and strengthen ecomorphology hypotheses.
Acknowledgments
We thank Tania González, Juan David González, and Fabian Arias for their suggestions and help with the graphical output, and Marco Melo and Edilberto Pachon for their help during fieldwork. Funding was provided by the Cohort I Bicentennial Doctoral Excellence Scholarship from The Ministry of Science, Technology, and Innovation of Colombia (MINCIENCIAS) and the General Royalties System (Guaviare Department), The Rufford foundation small grant (36352–1). We wish to thank IdeaWild for the equipment donation. We express our gratitude to Universidad Nacional de Colombia. Finally, we thank the reviewers for their comments and suggestions that greatly improved our manuscript.
References
Anjos MB, Zuanon J. Sampling effort and fish species richness in small terra firme forest streams of central Amazonia, Brazil. Neotrop Ichthyol. 2007; 5(1):45–52. https://doi.org/10.1590/S1679-62252007000100006
Armenteras D, Cabrera E, Rodríguez N, Retana J. National and regional determinants of tropical deforestation in Colombia. Reg Environ Chang. 2013; 13(6):1181–93. https://doi.org/10.1007/s10113-013-0433-7
Armenteras D, Murcia U, González TM, Barón OJ, Arias JE. Scenarios of land use and land cover change for NW Amazonia: impact on forest intactness. Glob Ecol Conserv. 2019; 17:e00567. https://doi.org/10.1016/j.gecco.2019.e00567
Armenteras D, Rodríguez Eraso N. Forest deforestation dynamics and drivers in Latin America: a review since 1990. Colomb For. 2014; 17(2):233–46. https://doi.org/10.14483/udistrital.jour.colomb.for.2014.2.a07
Atuesta-Ibargüen DJ. Composición florística y formas de vida de las macrofítas acuáticas de la serranía de La Lindosa (Guaviare), Guayana Colombiana. Caldasia. 2019; 41(2):1–27. https://doi.org/10.15446/caldasia.v41n2.71615
Barletta M, Jaureguizar AJ, Baigun C, Fontoura NF, Agostinho AA, Almeida-Val VMF et al. Fish and aquatic habitat conservation in South America: a continental overview with emphasis on neotropical systems. J Fish Biol. 2010; 76(9):2118–76. https://doi.org/10.1111/j.1095-8649.2010.02684.x
Bhat A. Ecomorphological correlates in tropical stream fishes of southern India. Environ Biol Fishes. 2005; 73(2):211–25. https://doi.org/10.1007/s10641-005-0561-0
Bistoni MA, Hued AC. Patterns of fish species richness in rivers of the central region of Argentina. Braz J Biol. 2002; 62(4):753–64. https://doi.org/10.1590/S1519-69842002000500004
Bock WJ. Concepts and methods in ecomorphology. J Biosci. 1994; 19(4):403–13. https://doi.org/10.1007/BF02703177
Bogota-Gregory JD, Jenkins DG, Acosta-Santos A, Agudelo Córdoba E. Fish diversity of Colombian Andes-Amazon streams at the end of conflict is a reference for conservation before increased land use. Ecol Evol. 2024; 14(3):e11046. https://doi.org/10.1002/ece3.11046
Bower LM, Piller KR. Shaping up: a geometric morphometric approach to assemblage ecomorphology. J Fish Biol. 2015; 87(3):691–714. https://doi.org/10.1111/jfb.12752
Breda L, Oliveira EF, Goulart E. Ecomorfologia de locomoção de peixes com enfoque para espécies neotropicais. Acta Sci Biol Sci. 2005; 27(4):371–81. https://doi.org/10.4025/actascibiolsci.v27i4.1271
Cárdenas-López D, Castaño-Arboleda N, Zubieta-Vega M, Jaramillo-Echeverry M. Flora de las formaciones rocosas de la Serranía de La Lindosa. Bogotá, D.C.: Instituto Amazónico de Investigaciones Científicas, SINCHI; 2008.
Casatti L, Castro RMC. Testing the ecomorphological hypothesis in a headwater riffles fish assemblage of the rio São Francisco, southeastern Brazil. Neotrop Ichthyol. 2006; 4(2):203–14. https://doi.org/10.1590/s1679-62252006000200006
Cochran-Biederman JL, Winemiller KO. Relationships among habitat, ecomorphology and diets of cichlids in the Bladen River, Belize. Environ Biol Fishes. 2010; 88(2):143–52. https://doi.org/10.1007/s10641-010-9624-y
Conde-Saldaña CC, Albornoz-Garzón JG, López-Delgado EO, Villa-Navarro FA. Ecomorphological relationships of fish assemblages in a trans-andean drainage, Upper Magdalena River basin, Colombia. Neotrop Ichthyol. 2017; 15(4):e170037. https://doi.org/10.1590/1982-0224-20170037
Correa SB, Winemiller K. Terrestrial-aquatic trophic linkages support fish production in a tropical oligotrophic river. Oecologia. 2018; 186(4):1069–78. https://doi.org/10.1007/s00442-018-4093-7
Dávalos LM, Bejarano AC, Hall MA, Correa HL, Corthals A, Espejo OJ et al. Forests and drugs: coca-driven deforestation in tropical biodiversity hotspots. Environ Sci Technol. 2011; 45(45):1219–27. https://doi.org/10.1021/es102373d
Douglas ME, Matthews WJ. Does morphology predict ecology? Hypothesis testing within a freshwater stream fish assemblage. Oikos. 1992; 65(2):213–24. https://doi.org/10.2307/3545012
Dufrene M, Legendre P. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr. 1997; 67(3):345–66.
Etter A, McAlpine C, Pullar D, Possingham H. Modelling the conversion of Colombian lowland ecosystems since 1940: drivers, patterns and rates. J Environ Manage. 2006a; 79(1):74–87. https://doi.org/10.1016/j.jenvman.2005.05.017
Etter A, McAlpine C, Wilson K, Phinn S, Possingham H. Regional patterns of agricultural land use and deforestation in Colombia. Agric Ecosyst Environ. 2006b; 114(2–4):369–86. https://doi.org/10.1016/j.agee.2005.11.013
Fernández EM, Ferriz RA, Bentos CA, López GR. Dieta y ecomorfología de la ictiofauna del arroyo Manantiales, provincia de Buenos Aires, Argentina. Rev Mus Argent Cienc Nat. 2012; 14(1):1–13.
Ferreira KM. Biology and ecomorphology of stream fishes from the rio Mogi-Guaçu basin, Southeastern Brazil. Neotrop Ichthyol. 2007; 5(3):311–26. https://doi.org/10.1590/s1679-62252007000300012
Fialho AP, Oliveira LG, Tejerina-Garro FL, Gomes LC. Fish assemblage structure in tributaries of the Meia Ponte River, Goiás, Brazil. Neotrop Ichthyol. 2007; 5(1):53–60. https://doi.org/10.1590/s1679-62252007000100007
Froese R, Pauly D. Fishbase. World Wide Web electronic publication. 2023. Available from: https://www.fishbase.se
Gálvis G, Mojica JI, Provenzano F, Lasso CA, Taphorn D, Royero R et al. Peces de la Orinoquía con enfasis en especies de interés ornamental. 1st ed. Bogotá, D.C.: 2007.
Gálvis G, Mojiva J, Duque S, Castellanos C, Sánchez-Duarte P, Arce M et al. Peces del medio Amazonas. Región Leticia. Bogotá, D.C.: Panamericana; 2006.
Gatz Jr. AJ. Community organization in fishes as indicated by morphological features. Ecology. 1979; 60(4):711–18. https://doi.org/10.2307/1936608
Géry J. Characoids of the world. Neptune City: TFH Publications; 1977.
Grenouillet G, Pont D, Hérissé C. Within-basin fish assemblage structure: the relative influence of habitat versus stream spatial position on local species richness. Can J Fish Aquat Sci. 2004; 61(1):93–102. https://doi.org/10.1139/f03-145
Huber O. Herbaceous ecosystems on the Guiana Shield, a regional overview. J Biogeogr. 2006; 33(3):464–75. https://doi.org/10.1111/j.1365-2699.2005.01454.x
Hugueny B, Pouilly M. Morphological correlates of diet in an assemblage of West African freshwater fishes. J Fish Biol. 1999; 54(6):1310–25. https://doi.org/10.1006/jfbi.1999.0962
Ibañez C, Tedesco PA, Bigorne R, Hugueny B, Pouilly M, Zepita C et al. Dietary-morphological relationships in fish assemblages of small forested streams in the Bolivian Amazon. Aquat Living Resour. 2007; 20(2):131–42. https://doi.org/10.1051/alr:2007024
Jackson DA. Stopping rules in principal components analysis: a comparison of heuristical and statistical approaches. Ecology. 1993; 74(8):2204–14. https://doi.org/10.2307/1939574
Jacob LL, Prudente BS, Montag LFA, Silva RR. The effect of different logging regimes on the ecomorphological structure of stream fish assemblages in the Brazilian Amazon. Hydrobiologia. 2021; 848(5):1027–39. https://doi.org/10.1007/s10750-020-04508-3
Kullander S. A review of the South Americacn cichlid genus Cichla, with descripcion of nine new species (Teleostei: Cichlidae). Ichthyol Explor Freshw. 2006; 17(4):289–398.
Lasso CA. Tipología de aguas (blancas, claras, negras) y su relación con la identificación y caracterización de los humedales de la Orinoquia. In: Lasso Carlos A, Rial A, Colonello G, Machado-Allison A, Trujillo F, editors. Humedales de la Orinoquia. Bogotá, D.C.: Instituto de investigación de Recursos Biológicos Alexander von Humboldt; 2014. p.50–61.
Legendre P, Legendre LF. Numerical ecology. 3rd ed. Amsterdam: Elsevier; 2012.
Lombarte A, Gordoa A, Whitfield AK, James NC, Tuset VM. Ecomorphological analysis as a complementary tool to detect changes in fish communities following major perturbations in two South African estuarine systems. Environ Biol Fishes. 2012; 94(4):601–14. https://doi.org/10.1007/s10641-011-9966-0
Luiz OJ, Olden JD, Kennard MJ, Crook DA, Douglas MM, Saunders TM et al. Substantial intraspecific trait variation across a hydrological gradient in northern Australian fishes. Ecosphere. 2022; 13(7):e4169. https://doi.org/10.1002/ecs2.4169
Medina-Rangel GF, Méndez-Galeano MA, Calderón Espinosa ML. Herpetofauna of San José del Guaviare, Guaviare, Colombia. Biota Colomb. 2019; 20(1):75–90. https://doi.org/10.21068/c2019.v20n01a05
Mérona B, Hugueny B, Tejerina-garro FL, Gautheret E. Diet-morphology relationship in a fish assemblage from a medium-size river of French Guiana: the effect of speces taxonomic proximity. Aquat Living Resour. 2008; 21:171–84. https://doi.org/10.1051/alr:2008032
Motta PJ, Clifton KB, Hernandez P, Eggold BT. Ecomorphological correlates in ten species of subtropical seagrass fishes: diet and microhabitat utilization. Environ Biol Fishes. 1995; 44(1–3):37–60. https://doi.org/10.1007/BF00005906
Neves MP, Delariva RL, Wolff LL. Diet and ecomorphological relationships of an endemic, species-poor fish assemblage in a stream in the Iguaçu National Park. Neotrop Ichthyol. 2015; 13(1):245–54. https://doi.org/10.1590/1982-0224-20140124
Oberdorff T, Guilbert E, Lucchetta J-C. Patterns of fish species richness in the Seine River basin, France. Hydrobiologia. 1993; 259(3):157–67. https://doi.org/10.1007/BF00006595
Oikonomou A, Leprieur F, Leonardos ID. Ecomorphological diversity of freshwater fishes as a tool for conservation priority setting: a case study from a Balkan hotspot. Environ Biol Fishes. 2018; 101(7):1121–36. https://doi.org/10.1007/s10641-018-0759-6
Oliveira EF, Goulart E, Breda L, Minte-Vera CV, Paiva LRS, Vismara MR. Ecomorphological patterns of the fish assemblage in a tropical floodplain: effects of trophic, spatial and phylogenetic structures. Neotrop Ichthyol. 2010; 8(3):569–86. https://doi.org/10.1590/s1679-62252010000300002
Pelicice FM, Azevedo-Santos VM, Vitule JRS, Orsi ML, Lima Junior DP, Magalhães ALB et al. Neotropical freshwater fishes imperilled by unsustainable policies. Fish Fish. 2017; 18(6):1119–33. https://doi.org/10.1111/faf.12228
Pouilly M, Lino F, Bretenoux J-G, Rosales C. Dietary-morphological relationships in a fish assemblage of the Bolivian Amazonian floodplain. J Fish Biol. 2003; 62(5):1137–58. https://doi.org/10.1046/j.1095-8649.2003.00108.x
Prado AVR, Goulart E, Pagotto JPA. Ecomorphology and use of food resources: inter- and intraspecific relationships of fish fauna associated with macrophyte stands. Neotrop Ichthyol. 2016; 14(4):e150140. https://doi.org/10.1590/1982-0224-20150140
R Development Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2011. Available from: https://www.r-project.org/
Rahel FJ, Hubert WA. Fish assemblages and habitat gradients in a rocky mountain-great plains stream: biotic zonation and additive patterns of community change. Trans Am Fish Soc. 1991; 120(3):319–32. https://doi.org/10.1577/1548-8659(1991)120%3C0319:FAAHGI%3E2.3.CO;2
Ramírez F, Davenport TL, Mojica JI. Dietary-morphological relationships of nineteen fish species from an Amazonian terra firme blackwater stream in Colombia. Limnologica. 2015; 52:89–102. https://doi.org/10.1016/j.limno.2015.04.002
Ricklefs RE, Miles DB. Ecological and evolutionary inferences from morphology: an ecological perspective. In: Wainwright PC, Reilly SM, editors. Ecological morphology integrative organismal biology. Chicago: University of Chicago; 1994. p.13–41.
Rosenfeld J. Assessing the habitat requirements of stream fishes: an overview and evaluation of different approaches. Trans Am Fish Soc. 2003; 132(5):953–68. https://doi.org/10.1577/t01-126
Sagnes P, Gaudin P, Statzner B. Shifts in morphometrics and their relation to hydrodynamic potential and habitat use during grayling ontogenesis. J Fish Biol. 1997; 50(4):846–58. https://doi.org/10.1111/j.1095-8649.1997.tb01977.x
Schaefer JF, Kerfoot JR. Fish assemblage dynamics in an adventitious stream: a landscape perspective. Am Midl Nat. 2004; 151(1):134–45. https://doi.org/10.1674/0003-0031(2004)151[0134:FADIAA]2.0.CO;2
van der Sleen P, Albert J. Field guide to the fishes of the Amazon, Orinoco & Guianas. New Jersey: Princenton University Press; 2018.
Soares BE, Ruffeil TOB, Montag LFA. Ecomorphological patterns of the fishes inhabiting the tide pools of the Amazonian coastal zone, Brazil. Neotrop Ichthyol. 2013; 11(4):845–58. https://doi.org/10.1590/S1679-62252013000400013
Stegmann LF, Leitão RP, Zuanon J, Magnusson WE. Distance to large rivers affects fish diversity patterns in highly dynamic streams of Central Amazonia. PLoS ONE. 2019; 14(10):e0223880. https://doi.org/10.1371/journal.pone.0223880
Torres-Bejarano AM, Sullivan SMP, González-Daza W, Cáceres C, Colorado Z. GJ. Riparian vegetation structure and seasonality influence functional diversity more than taxonomic diversity of stream fish assemblages in the Colombian Amazon. Aquat Ecol. 2022; 56(1):153–72. https://doi.org/10.1007/s10452-021-09904-y
Urbano-Bonilla A, Souza L, Maldonado-Ocampo JA, Jhon Z. Peces de quebradas de cabecera de la cuenca alta del Río Inírida y quebradas de cabecera tributarios del Río Guaviare. F Museum. 2017:1–07.
Vannote RL, Minshall WG, Cummins KW, Sedell JR, Cushing CE. The river continuum concept. Can J Fish Aquat Sci. 1980; 37(1):130–37. https://doi.org/10.1139/f80-017
Wainwright PC, Richard BA. Predicting patterns of prey use from morphology of fishes. Environ Biol Fishes. 1995; 44(1–3):97–113. https://doi.org/10.1007/BF00005909
Watson DJ, Balon EK. Ecomorphological analysis of fish taxocenes in rainforest streams of northern Borneo. J Fish Biol. 1984; 25(3):371–84. https://doi.org/10.1111/j.1095-8649.1984.tb04885.x
Winemiller KO. Ecomorphological diversification in lowland freshwater fish assemblages from five biotic regions. Ecol Monogr. 1991; 61(4):343–65. https://doi.org/10.2307/2937046
Wootton RJ. Ecology of teleost fishes. London: Chapman & Hall; 1990.
Authors
![]() Francisco Javier Luque1
Francisco Javier Luque1 ![]() and
and ![]() Gabriel J. Colorado Z.2
Gabriel J. Colorado Z.2
[1] Grupo en Ecología y Conservación de Fauna y Flora Silvestre, Departamento de Biología, Universidad Nacional de Colombia. Carrera 45 # 26 – 85, 111321, Bogotá D.C, Colombia. fjluquem@unal.edu.co (corresponding author).
[2] Grupo en Ecología y Conservación de Fauna y Flora Silvestre, Departamento de Ciencias Forestales, Universidad Nacional de Colombia. Carrera 65 # 59a – 110, Medellín, Colombia. gjcoloradoz@unal.edu.co.
Authors’ Contribution 

Francisco Javier Luque: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Gabriel J. Colorado Z.: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Ethical Statement
This research was conducted under the scientific research permit No 0255, March 14, 2014, of the Autoridad Nacional de Licencias Ambientales (ANLA) and approved by the Ethical Committee of the Facultad de Ciencias of Universidad Nacional de Colombia, Bogotá.
Competing Interests
The author declares no competing interests.
How to cite this article
Luque FJ, Colorado Z. GJ. Spatial and trophic structure drive ecomorphological patterns of fish assemblages in oligotrophic headwater streams of northwestern Amazonia. Neotrop Ichthyol. 2025; 23(2):e230085. https://doi.org/10.1590/1982-0224-2023-0135
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted March 26, 2025
Accepted March 26, 2025
![]() Submitted December 13, 2023
Submitted December 13, 2023
![]() Epub June 06, 2025
Epub June 06, 2025