![]() Josiane Rodrigues Rocha da Silva1
Josiane Rodrigues Rocha da Silva1 ![]() ,
, ![]() Cleiton Campos da Silva2,
Cleiton Campos da Silva2, ![]() Layon Zafra-Lemos3,
Layon Zafra-Lemos3, ![]() Ana Luiza de Brito Portela-Castro1,4 and
Ana Luiza de Brito Portela-Castro1,4 and ![]() Carlos Alexandre Fernandes1,4
Carlos Alexandre Fernandes1,4
PDF: EN XML: EN | Supplementary: S1 S2 | Cite this article
Abstract
Os efluentes têxteis possuem uma composição complexa que inclui elementos físicos, químicos e biológicos, e podem apresentar propriedades mutagênicas, genotóxicas e carcinogênicas. Uma parte dos corantes utilizados na indústria têxtil é perdida durante o processo de tingimento, contaminando ambientes aquáticos quando liberados em corpos d’água. Assim, o objetivo deste estudo foi avaliar os efeitos cito/genotóxicos dos efluentes da produção têxtil em espécimes de Astyanax lacustris antes e depois do tratamento com um sistema de wetland construído de fluxo vertical (WCFV). Examinamos 60 peixes (divididos em cinco condições de tratamento, com quatro peixes por condição, em triplicata). Os peixes foram expostos aos contaminantes por um período de sete dias. Os métodos de análise empregados incluíram avaliação de biomarcadores, teste de micronúcleo (MN), análise de alterações morfológicas celulares (AMC) e ensaio cometa. Peixes expostos a 5% v/v de efluente têxtil bruto e os efluentes que passaram por tratamento WCFV exibiram danos significativos em comparação ao grupo controle. A biorremediação do efluente realizada pelo sistema WCFV mostrou-se eficiente, conforme comprovado por análises físico-químicas. Concluímos que a biodegradação de efluentes de produção têxtil pode ser alcançada com sucesso por meio do uso de WCFV.
Palavras-chave: Alterações morfológicas celulares, Ensaio cometa, Fitorremediação, Método sustentável, Teste de micronúcleo.
Introduction
The indiscriminate disposal of contaminated waste into the environment is a regrettable reality that has resulted in irreparable consequences for all living organisms. Among the industries contributing to environmental degradation is the textile sector, which not only consumes significant amounts of water during the dyeing process but also releases a portion of the dyes into aquatic systems (Yanto, Tachibana, 2014). These dyes are noted for their toxicity and vibrant colors, which adversely affect ecosystems due to their complex composition and challenging degradation (Kumar, Gunasundari, 2018).
Furthermore, according to Chandanshive et al. (2017), textile effluents contain metals, which further amplify their toxicity. Azo dyes, the most prevalent group used in the textile industry, are distinguished by their characteristic azo bonds linking aromatic rings (Popli, Patel, 2015). These dyes adversely affect the concentration of total organic carbon, as well as biological and chemical oxygen demand (Saratale et al., 2011), and are recognized as carcinogenic and/or mutagenic to humans, animals, and plants (Almeida et al., 2019).
Various biotechnological methods have been proposed to reduce the harmful effects of chemical contaminants in aquatic environments, primarily by eliminating these pollutants. Among these methods, phytoremediation has garnered considerable attention due to its natural selectivity and resistance manifested by certain plant species in response to specific xenobiotic agents (Pires et al., 2003). This approach can mitigate or remediate the residual impacts of various contaminants in soil or water, either by completely eliminating them or by reducing their toxicity (Krutz et al., 2009).
In the context of treating contaminated wastewater, a technology known as Constructed Wetlands (CW) has gained significant recognition. It is regarded as a comprehensive and effective alternative for phytoremediation, as its mechanisms involve a combination of physical processes (such as filtration and sedimentation), chemical actions (including adsorption and precipitation), and biological processes (like biodegradation and phytoremediation). This multi-faceted approach effectively meets current needs for the pre-treatment of effluents before they are discharged into water bodies (Hassan et al., 2021; Kiflay et al., 2021). Furthermore, the CW consist of ponds or shallow artificial channels with a filtering bed that accommodates aquatic plants, which play a vital role in attenuating the various contaminants present in the effluent (Dotro et al., 2017).
The CW biotechnology is an environmentally friendly and sustainable method for remediating polluted water with varying characteristics. It offers several advantages over conventional treatment techniques, including lower installation and operational costs, simplicity in process, and effective removal of pollutants such as total suspended solids, organic loads, and soluble nutrients (Sezerino et al., 2018).
There are various configurations of constructed wetlands designed specifically for treating industrial effluents. The selection of substrate material for the treatment bed occurs in the project’s initial phase, with options including conventional materials like gravel and sand, as well as alternative materials such as gravel, slag, or bamboo that can be repurposed. Furthermore, the classification of CW configurations is generally based on the direction of effluent flow within each treatment unit (either horizontal or vertical), the types of plants used and their arrangement (either floating or submerged), and the system’s hydrological regime (surface or subsurface water) (Khan, Faisal, 2018; Sezerino et al., 2018).
Among the various plants suitable for phytoremediation in CW, macrophytes are the most commonly employed due to their high tolerance for the toxicity of various contaminants and their capacity for purification (Guittonny-Philippe et al., 2015). In Brazil, the native plant species most frequently utilized for CW establishment belong to the Typhaceae and Poaceae families, notably including Typha spp. and Cynodon spp. Additionally, Eleocharis spp. and Zizaniopsis spp. are also considered, though to a lesser extent (Sezerino et al., 2015; Machado et al., 2017).
Fish serve as crucial biological models in ecotoxicological research (Bu, 2013). Specimens of Astyanax lacustris (Lütken, 1875) have demonstrated sensitivity to various chemical contaminants found in polluted waters, often exposed to mutagens or carcinogens that can induce alterations in the DNA molecule (Tincani et al., 2019). Therefore, measuring biomarkers in fish can provide essential data on the environmental damage inflicted by toxic agents, such as textile effluents, and assist in monitoring aquatic ecosystems. Several biotechnological methods capable of detecting and verifying genetic alterations include the comet assay, the micronucleus test, and the analysis of cellular morphological changes in erythrocytes. The present study aims to evaluate the cyto/genotoxic effects of textile production effluents on A. lacustris, both before and after treatment using a vertical flow constructed wetland system (VFCW). The assays employed include biomarker analysis, the micronucleus (MN) test, cellular morphological change (CMC) assessment, the comet assay, and the measurement of the primary physicochemical characteristics of both raw and treated effluents.
Material and methods
Textile effluent. The textile effluent was collected from the stabilization ponds of a knitting factory in Campo Mourão, Paraná, Brazil. After collection, the effluent was transported in a plastic container to the laboratory and kept at room temperature.
Experimental design. Adult individuals (males and females) of A. lacustris, with a mean weight of 9±12 g and length of 8±12 cm, were obtained from a local breeding facility. The fish were acclimatized in aquaria with dechlorinated water at room temperature, constant aeration, a natural photoperiod (12:12 h light/dark cycle), and fed with specific small fish feed once daily (Basic Alcon® Fish Food, Camboriú/SC, Brazil) for 10 days in the Sectorial Vivarium for Fish Keeping and Experimentation at the Universidade Estadual de Maringá (UEM) in Maringá, Paraná State, Brazil.
Five groups/conditions were tested, with four fish per group, and all experiments were performed in triplicate; in total, 60 fish were used in the study. The number of individuals per aquarium was established according to the body mass/water volume ratio, which should not exceed 0.5–2 g/L (CONCEA Guide, annex I, fish). The control group (Group 1) fish were maintained in dechlorinated water; Group 2 – raw textile effluent at 5% v/v concentration (i.e., 500 ml effluent diluted to 10 L with dechlorinated water); Group 3 and 4 – the effluent treated by phytoremediation(Typha domingensis) through the VFCW system, used at concentrations of 25% v/v and 50% v/v (i.e., 2.5 L and 5 L of the treated effluent diluted in 10 L of dechlorinated water, respectively); Group 5 – with 100% v/v of the textile effluent treated by phytoremediation through the VFCW system. In all cases, the total aquarium volume was 10 L.
The percentages of textile effluent used were defined based on preliminary tests, in which it was observed that concentrations higher than 5% of the raw effluent caused mortality in A. lacustris. The concentrations of textile effluent at 25%, 50%, and 100% were subjected to treatment through the VFCW system to evaluate its efficiency in mitigating contaminants. Additionally, the study aimed to assess the potential cyto/genotoxic effects of the treated effluent.
Dissection and sampling. After 7 days of exposure, the fish were removed from the aquarium (one at a time) and bathed in an anesthetic solution: clove oil, 5 ml, diluted with ethyl alcohol, 20 ml, as per Inoue et al. (2005). Then, 1 ml of this solution was added per L of water. The animal was only manipulated after it failed to respond to physical stimulus, denoting death from an anesthetic overdose (Ferraro et al., 2004). After euthanasia, blood was collected to analyze micronucleus tests, cellular morphological changes, and comet assay in erythrocytes, as described below. Finally, the animals were fixed in commercial absolute alcohol, recorded with number and origin, and stored in glass vials from the UEM/NUPELIA laboratory. Voucher specimens were deposited in the Fish Collection of the Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (NUPELIA), Universidade Estadual de Maringá, municipality of Maringá, Paraná State, Brazil, as Astyanax lacustris (NUP 25442).
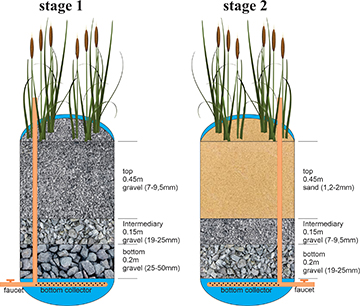
Construction of experimental built wetland treatment units with the vertical flow (VFCW). The making of the VFCW units followed some suggestions from the manual by Sezerino et al. (2018). This study made them from two cylindrical high-density polyethylene (HDPE) containers (0.80 m in height x 0.55 m in diameter). The filter mass of the first stage was composed of 0.2m of crushed stone (25–50 mm), 0.15m of crushed stone (19–25 mm), and 0.45 m of crushed stone (7–9.5 mm) (gravel). The second stage consisted of 0.2 m of gravel (19–25 mm), 0.15 m of gravel (7–9.5 mm), and 0.45 m of sand (1.2–2 mm). The adduction system consisted of 25 mm diameter polyvinyl chloride pipes and connections. The effluent was drained through an adductor system with perforations of 8.0 mm in diameter distributed along its entire extremity. The drainage pipe was positioned horizontally at the bottom of the bed, extending across the whole diameter of the units. The system contains a faucet for outputting the treated effluent at the bottom of the reservoir, installed 10 cm from the bottom (Fig. 1).
FIGURE 1| Scheme of an experimental vertical flow constructed wetland system (VFCW) unit.
The beds were populated with Typha domingensis at 16 plants per square meter density. The propagules were collected manually in a naturally flooded area located on a rural property in the municipality of Cianorte at the beginning of December 2021. The collection was carried out so that the rhizomes were preserved, carefully transported, and transplanted in the experimental units. The residence time of the effluent in the treatment system was 96 h, 48 h in the first stage, and 48 h in the second stage.
Physical-chemical analysis of the effluent. The physical-chemical analysis was conducted on the raw effluent sourced directly from the textile industry, as well as on the same raw effluent (at 100% concentration) after treatment by the VFCW system. The raw and treated effluent were stored in a thermal box, kept at ± 5ºC, and taken to the laboratory for physical-chemical characterization. The analyses were conducted in the Departamento de Engenharia Civil, Laboratório de Qualidade da Água e Controle da Poluição – Saneamento Ambiental of the UEM. Six physicochemical parameters of this textile effluent – pH, apparent color, BOD, COD, total suspended solids, and electrical conductivity were determined using standard methods (Rice et al., 2012). The physical-chemical parameters were defined according to CONAMA Resolution No 430/2011.
Micronucleus test and cellular morphological changes in erythrocytes. The Micronucleus (MN) and cellular morphological changes (CMC) in the erythrocytes test were performed based on the description by Hooftman, Raat (1982). After the anesthesia of the animals, blood was extracted from the caudal vein with a heparinized syringe. A sample (approximately 10 ìL) was dripped onto a sanitized glass slide, and the smear was performed with the aid of another slide. The dripped slide was kept at room temperature, drying for at least 12 h. Afterward, they were fixed with immersion in absolute ethanol for 20 min. Staining was performed for 10 min with 5% Giemsa solution diluted in phosphate buffer (pH 6.8). Then, the slides were washed in distilled water, left to dry naturally, and kept in closed boxes until analysis under microscopy. The slides were analyzed by optical microscopy under 1000 ´ magnification. The MN count and the study of CMC were performed on 2,000 red blood cells per fish.
Comet assay. The comet assay was conducted according to Speit, Hartmann (1999) with modifications according to Ferraro et al. (2004). A sample (approximately 10 ìL) of blood was collected from the tail vein of each fish with a heparinized syringe and diluted in fetal calf serum, 1 ml. Slides were prepared for microscopy using this cell suspension, 10 ìL, added to low-melting-point agarose at 37EC, 120 ìL, followed by incubation in lysis solution (1 ml of Triton X-100, 10 mL of DMSO, and 89 ml of stock lysis solution consisting of 2.5 M NaCl, 100 mM EDTA, 10 mM Tris, pH 10, 2.5 M NaOH and 1% N-lauryl sarcosinate). The slides were then dark for 1 h at 8EC.
After lysis, slides were placed in the electrophoresis tank, covered in buffer solution (0.3 N NaOH, 1 mM EDTA, pH > 13) in a cold environment at 8EC for 20 min to unwind the DNA. Electrophoresis was performed for 20 min at 25 V, 300 mA, field strength 1V/cm; slides were neutralized (0.4 M Tris) for 10 min, fixed in absolute ethanol for 10 min, and then stained with ethidium bromide (2 ìg/ml), and covered with coverslips for analysis, after drying for approximately 12 h. Analyses were performed blind to test conditions under an epifluorescence microscope, with image capture at 400´ magnification, counting 100 nucleoids per fish. DNA damage was visually classified into four classes based on DNA migration: 0 (no apparent damage), 1 (slight damage), 2 (medium damage), 3 (large damage), and 4 (maximum damage).
Statistical analysis. Biomarkers (MN test, CMC assay, and comet assay) were analyzed using the Kruskal-Wallis test, a nonparametric analysis of variance, with Dunn’s post hoc test for multiple comparisons, performed in GraphPad Prism v. 5. The level of significance for all studies was p < 0.05.
Results
MN test and CMC in erythrocytes. In a detailed analysis of 120,000 erythrocytes from 60 specimens of A. lacustris, MN were found in 54 erythrocytes. Furthermore, a total of 3,826 CMC were recorded (Tab. 1; Fig. 2). The specimens exposed to a concentration of 5% v/v of raw textile effluent and the 50% v/v and 100% v/v concentrations of VFCW-treated textile effluents showed a significantly higher frequency of CMC compared to the control (p < 0.05 in Tab. 1).
TABLE 1 | Absolute numbers and percentage for alteration in Astyanax lacustris erythrocytes. For each fish, 2000 cells were analyzed. *Significantly different to control group (p < 0.05); MN = micronuclei; CMC = cellular morphological changes.
Group | MN | % MN |
Mean
± standard | CMC | %CMC |
Mean
± standard deviation |
Control | 0 | 0% | 0.0 ±0.0 | 212 | 0.88% | 17.66 ± 5.61 |
Raw
textile effluent | 33 | 0.14% | 2.75±1.65* | 2046 | 8.53% | 170.5 ± 31.88* |
Effluent
treated by VFCW | 1 | 0.004% | 0.08±2.88 | 276 | 1.15% | 23.08 ± 9.41 |
Effluent
treated by VFCW | 10 | 0.042% | 0.8±1.02 | 474 | 1.98% | 39.5± 9.60* |
Effluent
treated by VFCW | 14 | 0.058% | 1.16±1.46 | 814 | 3.39% | 67.8 ± 11.51* |
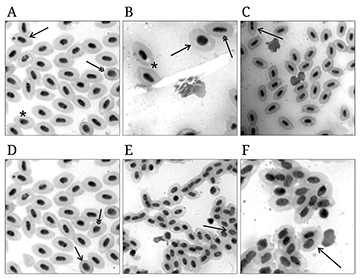
FIGURE 2| Erythrocytes of Astyanax lacustris from the control group and after exposure to raw and treated textile effluent using a vertical flow constructed wetland system (VFCW). (A) Normal erythrocytes (asterisk), Micronucleus (single arrow), Crenate erythrocyte (double arrow), (B) Cytoplasmic vacuole (asterisk), Immature erythrocyte (single arrow), Elliptocyte (double arrow), (C) Nuclear constriction (single arrow), (D) and nuclear fragmentation (single arrow), Notched (double arrow), (E) Blebbed (single arrow), (F) Binucleated (single arrow). Magnification: 1000X.
On the other hand, there was no significant difference in CMC in the comparison between 25% v/v of textile effluent treated by VFCW and the control (p > 0.05 in Tab. 1). Furthermore, there were no significant differences in MN across all groups when compared to the control (p > 0.05 in Tab. 1).
Comet assay. The comet assay demonstrated higher levels of DNA damage (Fig. 3) recorded in the 5% v/v concentration of raw textile effluent compared to the control (p < 0.05 in Tab. 2). There was no significant difference in the effluent treated by VFCW at a concentration of 25% v/v when compared with the control (p > 0.05 in Tab. 2). On the other hand, the concentrations of 50% v/v and 100% v/v of effluent treated by VFCW differed significantly from the control (p < 0.05 in Tab. 2).
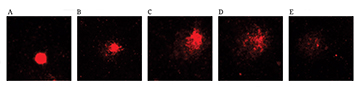
FIGURE 3| Damage to erythrocytes from Astyanax lacustris specimens from the control group and following exposure to raw and treated textile effluent using a vertical flow constructed wetland system (VFCW) was measured by the comet assay (Magnification 400´). A) normal nucleus, B) damage 1, C) damage 2, D) damage 3, E) damage 4.
TABLE 2 | Damage recorded in the DNA of erythrocytes of Astyanax lacustris specimens, following exposure to raw and VFCW-treated textile effluent. One hundred nucleoids from each fish were analyzed. *Significantly different to control group (p < 0.05).
Groups | Specimens | Damage classes | Scores | Mean and standard error of scores | ||||
0 | 1 | 2 | 3 | 4 | ||||
Control | 1 | 88 | 9 | 3 | 0 | 0 | 15 | 13.33 ± 1.92 |
2 | 95 | 3 | 2 | 0 | 0 | 7 | ||
3 | 81 | 12 | 7 | 0 | 0 | 26 | ||
4 | 90 | 7 | 3 | 0 | 0 | 13 | ||
5 | 92 | 8 | 0 | 0 | 0 | 8 | ||
6 | 89 | 11 | 1 | 0 | 0 | 13 | ||
7 | 77 | 19 | 4 | 0 | 0 | 27 | ||
8 | 92 | 5 | 3 | 0 | 0 | 11 | ||
9 | 94 | 4 | 2 | 0 | 0 | 8 | ||
10 | 91 | 7 | 2 | 0 | 0 | 11 | ||
11 | 92 | 8 | 0 | 0 | 0 | 8 | ||
12 | 92 | 7 | 3 | 0 | 0 | 13 | ||
Raw textile effluent at 5% v/v concentration | 13 | 27 | 38 | 33 | 2 | 0 | 110 | 129.91 ± 3.09* |
14 | 27 | 32 | 28 | 13 | 0 | 127 | ||
15 | 21 | 39 | 27 | 10 | 3 | 135 | ||
16 | 29 | 33 | 26 | 12 | 0 | 121 | ||
17 | 25 | 36 | 31 | 6 | 2 | 124 | ||
18 | 24 | 34 | 21 | 18 | 3 | 142 | ||
19 | 16 | 40 | 34 | 8 | 2 | 140 | ||
20 | 23 | 37 | 28 | 12 | 0 | 129 | ||
21 | 23 | 32 | 41 | 2 | 2 | 128 | ||
22 | 29 | 36 | 19 | 16 | 0 | 122 | ||
23 | 24 | 25 | 31 | 17 | 3 | 150 | ||
24 | 23 | 38 | 24 | 15 | 0 | 131 | ||
Effluent treated by VFCW at 25% v/v concentration | 25 | 78 | 16 | 6 | 0 | 0 | 28 | 32.08 ± 4.44 |
26 | 67 | 20 | 9 | 4 | 0 | 50 | ||
27 | 73 | 14 | 10 | 3 | 0 | 43 | ||
28 | 69 | 12 | 15 | 4 | 0 | 54 | ||
29 | 76 | 20 | 4 | 0 | 0 | 28 | ||
30 | 82 | 9 | 6 | 3 | 0 | 30 | ||
31 | 85 | 8 | 5 | 2 | 0 | 24 | ||
32 | 66 | 25 | 5 | 4 | 0 | 47 | ||
33 | 78 | 7 | 10 | 3 | 2 | 44 | ||
34 | 91 | 5 | 3 | 1 | 0 | 14 | ||
35 | 93 | 7 | 0 | 0 | 0 | 7 | ||
36 | 87 | 10 | 3 | 0 | 0 | 16 | ||
Effluent treated by VFCW at 50% v/v concentration | 37 | 65 | 21 | 11 | 3 | 0 | 52 | 56.0 ± 4.81* |
38 | 59 | 24 | 13 | 4 | 0 | 62 | ||
39 | 53 | 34 | 10 | 3 | 0 | 63 | ||
40 | 59 | 19 | 17 | 4 | 1 | 69 | ||
41 | 68 | 28 | 4 | 0 | 0 | 36 | ||
42 | 62 | 22 | 5 | 6 | 5 | 70 | ||
43 | 55 | 42 | 3 | 0 | 0 | 48 | ||
44 | 71 | 24 | 5 | 0 | 0 | 34 | ||
45 | 76 | 13 | 7 | 4 | 0 | 39 | ||
46 | 78 | 12 | 5 | 2 | 3 | 40 | ||
47 | 47 | 35 | 12 | 4 | 2 | 79 | ||
48 | 55 | 23 | 12 | 7 | 3 | 80 | ||
Effluent treated by VFCW at 100% v/v concentration | 49 | 39 | 29 | 24 | 8 | 0 | 101 | 69.5 ± 7.85* |
50 | 57 | 22 | 13 | 8 | 0 | 72 | ||
51 | 50 | 29 | 12 | 9 | 0 | 80 | ||
52 | 47 | 28 | 14 | 10 | 1 | 90 | ||
53 | 69 | 13 | 8 | 0 | 10 | 69 | ||
54 | 82 | 13 | 5 | 0 | 0 | 23 | ||
55 | 71 | 23 | 6 | 0 | 0 | 35 | ||
56 | 34 | 39 | 12 | 8 | 7 | 115 | ||
57 | 67 | 21 | 2 | 5 | 4 | 56 | ||
58 | 58 | 34 | 3 | 5 | 0 | 55 | ||
59 | 67 | 21 | 8 | 4 | 0 | 49 | ||
60 | 35 | 52 | 6 | 3 | 4 | 89 | ||
Physical-chemical analysis of the effluent. The results of the physical-chemical analysis of the effluent are summarized in Tab. 3. The physical-chemical analyses conducted on both the raw and VFCW-treated textile effluent (coming directly from the textile industry, both without dilution) demonstrated a significant reduction across all evaluated parameters, notably in apparent color, which decreased by 99.29% of the treated textile effluent in comparasion with raw textile effluent. The Biochemical Oxygen Demand (BOD) also showed a remarkable reduction of 99.33%. Additionally, the Chemical Oxygen Demand (COD), a crucial parameter for assessing water pollution, as it accounts for the oxygen required to degrade both biodegradable organic matter and non-biodegradable organic materials derived from chemical processes, exhibited a reduction of 95.46%. Additionally, there was a decrease in electrical conductivity by 36.14% and total dissolved solids by 17.6%.
TABLE 3 | Values of physicochemical parameter rs before and after treatment of textile effluent treated by vertical flow constructed wetland system (VFCW).
Assay | Raw textile effluent | VFCW-treated textile effluent |
Electric conductivity (μS/cm) | 3090 | 1973 |
Apparent color (AC) | 983.4 | 6.9 |
Biochemical oxygen demand (BOD) (mg L-1) | 96.59 | < 0.64 |
Chemical oxygen demand (COD) (mg L-1) | 253.51 | < 11.5 |
pH | 7.25 | 7.25 |
Total dissolved solids (mg L-1) | 1764 | 1453 |
Discussion
Our data indicated considerable damage to the erythrocytes of A. lacustris both before and after treatment with the VFCW system. While the use of phytoremediation reduced these damages, it did not completely eliminate them. Similar changes have been documented in studies involving fish exposed to textile effluents, which are known for their toxic, mutagenic, or carcinogenic properties (Çavaș, Ergene-Gözükara, 2003; Hemachandra, Pathiratne, 2016; Parmar, Barot, 2016; Silva et al., 2023).
The VFCW system, particularly at a concentration of 25% v/v, demonstrated effectiveness, as statistical analyses for MN test, CMC, and the comet assay did not show significant differences compared to the control. Furthermore, during the tests, the exposure of specimens to wastewater treated with VFCW at 25%, 50%, and 100% v/v did not lead to fish mortality. This finding highlights that, despite the observed cellular alterations, the system contributed to the detoxification of the effluent. It not only reduced toxicity but also mitigated potential harmful metabolites, avoiding any toxic effects.
In alignment with our findings, Hussain et al. (2018) conducted tests with both vegetated and non-vegetated VFCWs, utilizing a consortium of plants and endophytic bacteria. Their study evaluated the toxicity of treated textile effluents by VFCWs in fish from 12 h to 72 h, utilizing wastewater collected from the equalization tank at Interloop Limited in Khurrianwala, Faisalabad, Pakistan. The findings confirmed the effectiveness of the treatment, with even more promising results obtained through the use of a plant consortium.
In the present study, exposures to raw textile effluent concentrations exceeding 5% v/v resulted in fish mortality, consistent with the findings of Zhang et al. (2012), who evaluated acute toxicity in Danio rerio using water samples collected during textile industry processing. Notably, the maximum non-lethal dose was also identified as 5% v/v of the raw effluent. While the cell damage observed from effluent treated by the VFCW system was significantly lower than that from raw textile effluent at the 5% v/v concentration, statistical analysis of the comet assay revealed substantial differences in DNA damage between the control and the 50% v/v and 100% v/v concentrations of effluent treated by the VFCW system. Furthermore, comparisons between the raw effluent at 5% v/v and the treated effluents showed significant differences, indicating that the treatments were statistically distinct in both the control and raw effluent groups (Tab. 3). This can be attributed to the high sensitivity of the comet assay, which has been documented in several studies; even low concentrations of contaminants can elicit a genotoxic response (Mitchelmore, Chipman, 1998; Bücker et al., 2006; Frenzilli et al., 2009), demonstrating concentration-dependent damage (Zhang et al., 2012).
The expressive decline of the BOD observed in the present study is closely linked to the presence of biodegradable organic matter, which necessitates oxygen for its degradation (Menezes et al., 2009). Elevated BOD levels in raw textile effluent can be detrimental to aquatic environments, as they may lead to the complete depletion of dissolved oxygen, ultimately threatening various forms of aquatic life at the discharge site (Matos et al., 2014).
Research utilizing constructed wetlands for the treatment of textile effluents has yielded promising results. For instance, Ong et al. (2009b) reported that a constructed wetland featuring an ascending flow and populated with Phragmites australis achieved an impressive 86% reduction in COD and a 96% efficiency rate in removing the azo dye Acid Orange 7. Similarly, Ong et al. (2009a) demonstrated that combining aerobic and anaerobic processes in constructed wetlands resulted in a 96% reduction of NH4-N and aromatic amines, thereby enhancing treatment performance for textile effluents. In Tanzania, Mbuligwe (2005) conducted a comparative analysis of wastewater treatment rich in dyes using vegetated wetland systems with various plant species, achieving average reductions exceeding 70% across several parameters, with the coco yam plant exhibiting the best performance.
In a prototype system featuring built-up-flow wetlands designed for treating effluent from the textile industry, Chandanshive et al. (2017) conducted tests using Typha angustifolia, Paspalum scrobiculatum, and their co-plantation (consortium). The study found that the in situ treatment of effluent by T. angustifolia, P. scrobiculatum, and the consortium resulted in the removal of color and metals — such as arsenic, cadmium, chromium, and lead — by up to 77% following phytoremediation. T. angustifolia and P. scrobiculatum demonstrated a removal efficiency of up to 80% for Congo Red dye (100 mg L-1), while the consortium achieved a remarkable 94% discoloration. Similarly, research by Shehzadi et al. (2014) utilized the Typha domingensis plant in vertical flow constructed wetlands, highlighting its potential to degrade textile effluent. The introduction of endophytic bacteria significantly enhanced the degradation process, leading to a reduction in values of BOD, COD, and apparent color compared to non-inoculated plants. Collectively, these studies, along with our own, affirm the effectiveness of CW systems in treatment applications, contingent on the level of effluent contamination. These findings emphasize the necessity for further investigations aimed at mitigating cyto/genotoxic damage to A. lacustris specimens through consortia of plants and endophytic bacteria, as well as between different plant species, to achieve even greater results.
Numerous studies have demonstrated the effectiveness of CW systems in treating textile effluents and various other pollutants. According to Domingos et al. (2011), the CW systems are capable of efficiently treating a wide range of effluents, including those from textiles, dairy production, swine farms, septic tank dewatering, sanitary landfill leachate, inorganic industrial discharges, and tannery operations, among others. In their research, Domingos and his colleagues examined the performance of vertical flow constructed wetlands in treating effluents from a chemical and fertilizer industry in Australia, with a focus on removing nitrogen and metals. They observed impressive results, achieving a 90% removal rate of ammonia.
The effectiveness of CW systems in eliminating commonly used pharmaceuticals globally has been well-documented, with removal efficiencies exceeding 90% (Ranieri et al., 2011; Ávila et al., 2014; Chen et al., 2016; Santos et al., 2019). Brazil research on CW targets the treatment of domestic sewage, focusing on the removal of carbonaceous organic matter, as indicated by COD and total suspended solids, as well as essential nutrients like nitrogen (N), phosphorus (P), and potassium (K) (Machado et al., 2017; Pelissari et al., 2018; Santos et al., 2018; Melo Júnior et al., 2019; Mazucato et al., 2020).
In this study, the biological evaluation using fish erythrocyte biomarkers proved to be effective for environmental biomonitoring. Similarly, the VFCW system demonstrated positive outcomes concerning the physicochemical parameters of the treated effluent and did not show lethal toxicity to the specimens. However, the cyto/genotoxic damage observed in samples of A. lacustris indicates that the VFCW system warrants further investigation and optimization regarding the use of aquatic macrophytes, their co-planting, and the integration of microorganisms. This approach may enhance the efficiency of wastewater treatment. Further research is essential to identify key species of plants and bacteria and to explore their combined application on a larger scale for improved degradation. Additionally, various types of constructed wetlands should be tested to assess their efficacy in conjunction with different plants, microorganisms, and a range of contaminants.
Acknowledgments
We thank Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), through the Dean of the Departamento de Pós-Graduação e Pesquisa of the Universidade Estadual de Maringá, UEM-PPG), for the master’s scholarship granted to JRRS. We would also like to thank the Textile Industry, Sajamas Malhas, for the effluent granted for carrying out the tests.
References
Almeida EJR, Mazzeo DEC, Sommaggio LRD, Marin-Morales MA, Andrade AR, Corso CR. Azo dyes degradation and mutagenicity evaluation with a combination of microbiological and oxidative discoloration treatments. Ecotoxicol Environ Saf. 2019; 183:109484. https://doi.org/10.1016/j.ecoenv.2019.109484
Ávila C, Nivala J, Olsson L, Kassa K, Headley T, Mueller RA et al. Emerging organic contaminants in vertical subsurface flow constructed wetlands: influence of media size, loading frequency and use of active aeration. Sci Total Environ. 2014; 494–95:211–17. https://doi.org/10.1016/j.scitotenv.2014.06.128
Bu A. Fish ecogenotoxicology: an emerging science, an emerging tool for environmental monitoring and risk assessment. J Environ Anal Toxicol. 2012; 3(1):1000165. https://doi.org/10.4172/2161-0525.1000165
Bücker A, Carvalho W, Alves-Gomes JA. Avaliation of mutagenicity and gentotoxicity in Eigenmannia virescens (Teleostei: Gymnotiformes) exposed to benzene. Acta Amaz. 2006; 36(3):357–64. https://doi.org/10.1590/S0044-59672006000300011
Çavas T, Ergene-Gözükara S. Micronuclei, nuclear lesions and interphase silver-stained nucleolar regions (AgNORs) as cytogenotoxicity indicators in Oreochromis niloticus exposed to textile mill effluent. Mutat Res. 2003; 538(1–2):81–91. https://doi.org/10.1016/S1383-5718(03)00091-3
Chandanshive VV, Rane NR, Tamboli AS, Gholave AR, Khandare RV, Govindwar SP. Co-plantation of aquatic macrophytes Typha angustifolia and Paspalum scrobiculatum for effective treatment of textile industry effluent. J Hazard Mater. 2017; 338:47–56. https://doi.org/10.1016/j.jhazmat.2017.05.021
Chen Y, Vymazal J, Bøezinová T, Koželuh M, Kule L, Huang J et al. Occurrence, removal and environmental risk assessment of pharmaceuticals and personal care products in rural wastewater treatment wetlands. Sci Total Environ. 2016; 566–567:1660–69. https://doi.org/10.1016/j.scitotenv.2016.06.069
Conselho Nacional do Meio Ambiente (CONAMA). Resolução nº 430, de 13 de maio de 2011. Dispõe sobre condições e padrões de lançamento de efluentes. Diário Oficial da União: seção 1, Brasília, DF, 16 maio 2011.
Domingos SS, Dallas S, Skillman L, Felstead S, Ho G. Nitrogen removal and ammonia-oxidising bacteria in a vertical flow constructed wetland treating inorganic wastewater. WST. 2011; 64(3):587–94. https://doi.org/10.2166/wst.2011.565
Dotro G, Molle P, Nivala J. Biological Wastewater Treatment Series. Vol. 7. Treatment Wetlands: London; 2017.
Ferraro MVM, Fenocchio AS, Mantovani MS, Ribeiro CO, Cestari MM. Mutagenic effects of tributyltin and inorganic lead (Pb II) on the fish H. malabaricus as evaluated using the comet assay and the piscine micronucleus and chromosome aberration tests. Genet Mol Biol. 2004; 27(1):103–07. https://doi.org/10.1590/S1415-47572004000100017
Frenzilli G, Nigro M, Lyons BP. The Comet assay for the evaluation of genotoxic impact in aquatic environments. Mutat Res Rev Mutat Res. 2009; 681(1):80–92. https://doi.org/10.1016/j.mrrev.2008.03.001
Guittonny-Philippe A, Petit M-E, Masotti V, Monnier Y, Malleret L, Coulomb B et al. Selection of wild macrophytes for use in constructed wetlands for phytoremediation of contaminant mixtures. J Environ Manage. 2015; 147:108–23. https://doi.org/10.1016/j.jenvman.2014.09.009
Hassan I, Chowdhury SR, Prihartato PK, Razzak SA. Wastewater treatment using constructed wetland: current trends and future potential. Processes. 2021; 9(11):1917. https://doi.org/10.3390/pr9111917
Hemachandra CK, Pathiratne A. Combination of physico-chemical analysis, Allium cepa test system and Oreochromis niloticus erythrocyte based comet assay/nuclear abnormalities tests for cyto-genotoxicity assessments of treated effluents discharged from textile industries. Ecotoxicol Environ Saf. 2016; 131:54–64. https://doi.org/10.1016/j.ecoenv.2016.05.010
Hooftman RN, Raat WK. Induction of nuclear anomalies (micronuclei) in the peripheral blood erythrocytes of the eastern mudminnow Umbra pygmaea by ethyl methanesulphonate. Mutat Res. 1982; 104(1–3):147–52. https://doi.org/10.1016/0165-7992(82)90136-1
Hussain Z, Arslan M, Malik MH, Mohsin M, Iqbal S, Afzal M. Treatment of the textile industry effluent in a pilot-scale vertical flow constructed wetland system augmented with bacterial endophytes. Sci Total Environ. 2018; 645:966–73. https://doi.org/10.1016/j.scitotenv.2018.07.163
Inoue LAKA, Afonso LOB, Moraes GK, Moraes G. Effects of clove oil on the stress response of matrinxã (Brycon cephalus) subjected to transport. Acta Amaz. 2005; 35(2):289–95. https://doi.org/10.1590/s0044-59672005000200018
Khan HN, Faisal M. Phytoremediation of industrial wastewater by hydrophytes. In: Ansari A, Gill S, Gill R, Lanza G, Newman L, editors. Phytoremediation.Springe Cham; 2018. p.179–200. https://doi.org/10.1007/978-3-319-99651-6_8
Kiflay E, Selemani J, Njau K. Integrated constructed wetlands treating industrial wastewater from seed production. Water Pract Technol. 2021; 16(2):504–15. https://doi.org/10.2166/wpt.2021.008
Krutz LJ, Burke IC, Reddy KN, Zablotowicz RM, Price AJ. Enhanced Atrazine Degradation: evidence for reduced residual weed control and a method for identifying adapted soils and predicting herbicide persistence. Weed Sci. 2009; 57(4):427–34. https://doi.org/10.1614/WS-09-010.1
Kumar PS, Gunasundari E. Sustainable wet processing – an alternative source for detoxifying supply chain in textiles. In: Muthu S, editor. Detox fashion, textile science and clothing technology. Singapore: Springer; 2018. p.37–60. https://doi.org/10.1007/978-981-10-4876-0_2
Machado AI, Beretta M, Fragoso R, Duarte E. Overview of the state of the art of constructed wetlands for decentralized wastewater management in Brazil. J Environ Manage. 2017; 187:560–70. https://doi.org/10.1016/j.jenvman.2016.11.015
Matos MP, Borges AC, Matos AT, Silva EF, Martinez MA. Effect of time-temperature binomial in obtaining biochemical oxygen demand of different wastewaters. Eng Agric. 2014; 34(2):332–40. https://doi.org/10.1590/S0100-69162014000200014
Mazucato VSH, Santos VB, Cavalheri PS, Magalhães Filho FJC. Comportamento de wetland construído de fluxo vertical com fundo parcialmente saturado na remoção de matéria orgânica nitrogenada e fósforo. Braz J Dev. 2020; 6(8):56506–20. https://doi.org/10.34117/bjdv6n8-168
Mbuligwe SE. Comparative treatment of dye-rich wastewater in engineered wetland systems (EWSs) vegetated with different plants. Water Res. 2005; 39(2–3):271–80. https://doi.org/10.1016/j.watres.2004.09.022
Melo Júnior AS, Nascimento PC, Dias CJ, Ribeiro KA, Merij AC, Gama SM. Qualidade no tratamento de esgoto doméstico por wetland construídos. Int J Innov Res Sci Eng Technol. 2019; 7(1):20–39.
Menezes JM, Prado RB, Silva Júnior GC, Mansur KL, Oliveira ES. Qualidade da água e sua relação espacial com as fontes de contaminação antrópicas e naturais: bacia hidrográfica do rio São Domingos – RJ. Eng Agric. 2009; 29(4):687–98. https://doi.org/10.1590/S0100-69162009000400019
Mitchelmore CL, Chipman JK. DNA strand breakage in aquatic organisms and the potential value of the comet assay in environmental monitoring. Mut Res Fundam Mol. 1998; 399(2):135–47. https://doi.org/10.1016/S0027-5107(97)00252-2
Ong SA, Uchiyama K, Inadama D, Ishida Y, Yamagiwa K. Phytoremediation of industrial effluent containing azo dye by model up-flow constructed wetland. Chin Chem Lett. 2009a; 20(2):225–28. https://doi.org/10.1016/j.cclet.2008.10.028
Ong SA, Uchiyama K, Inadama D, Yamagiwa K. Simultaneous removal of color, organic compounds and nutrients in azo dye-containing wastewater using up-flow constructed wetland. J Hazard Mater. 2009b; 165(1–3):696–703. https://doi.org/10.1016/j.jhazmat.2008.10.071
Parmar A, Barot J. Determination of genotoxic effect of azo dye CI RR 120 on fish Catla. Biotechnol Res. 2016; 2(2):77–80.
Pelissari C, Guivernau M, Ávila C, Viñas M, García J, Sezerino PH. Influência de parâmetros operacionais sobre as populações oxidantes de amônia e desnitrificantes em wetland construído vertical. Tecno-Lógica. 2018; 22(1):1–05. https://doi.org/10.17058/tecnolog.v22i1.10479
Pires FR, Souza CM, Silva AA, Procópio SO, Ferreira LR. Fitorremediação de solos contaminados com herbicidas. Plan Dan. 2003; 21(2):335–41. https://doi.org/10.1590/S0100-83582003000200020
Popli S, Patel UD. Destruction of azo dyes by anaerobic–aerobic sequential biological treatment: a review. Int J Environ Sci Technol. 2015; 12(1):405–20. https://doi.org/10.1007/s13762-014-0499-x
Ranieri E, Verlicchi P, Young TM. Paracetamol removal in subsurface flow constructed wetlands. J Hydrol. 2011; 404(3–4):130–35. https://doi.org/10.1016/j.jhydrol.2011.03.015
Rice EW, Baird RB, Eaton AD, Lesceri LS. Standard methods for the examination of water and wastewater. 22nd ed. Denver, CO: American Water Works Association; 2012.
Santos F, Almeida CMR, Ribeiro I, Mucha AP. Potential of constructed wetland for the removal of antibiotics and antibiotic resistant bacteria from livestock wastewater. Ecol Eng. 2019; 129:45–53. https://doi.org/10.1016/j.ecoleng.2019.01.007
Santos MO, Pelissari C, Rousso BZ, Freitas MN, Sezerino PH. Avaliação dos dois primeiros anos de operação de um wetland construído vertical de fundo saturado aplicado no tratamento de esgoto sanitário. Tecno-Lógica. 2018; 22(1):25–29. https://doi.org/10.17058/tecnolog.v22i1.10500
Saratale RG, Saratale GD, Chang JS, Govindwar SP. Bacterial decolorization and degradation of azo dyes: a review. J Taiwan Inst Chem Eng. 2011; 42(1):138–57. https://doi.org/10.1016/j.jtice.2010.06.006
Sezerino PH, Bento AP, Decezaro ST, Magri ME, Philippi LS. Experiências brasileiras com wetlands construídos aplicados ao tratamento de águas residuárias: parâmetros de projeto para sistemas horizontais. Eng San Ambient. 2015; 20(1):151–58. https://doi.org/10.1590/S1413-41522015020000096615
Sezerino PH, Rousso BZ, Pelissari C, Santos MO, Freitas MN, Fechine VY et al. Wetlands construídos aplicados no tratamento de esgoto sanitário: recomendações para implantação e boas práticas de operação e manutenção. Tubarão: Fundação Nacional da Saúde – Funasa; 2018.
Shehzadi M, Afzal M, Khan MU, Islam E, Mobin A, Anwar S et al. Enhanced degradation of textile effluent in constructed wetland system using Typha domingensis and textile effluent-degrading endophytic bacteria. Water Res. 2014; 58:152–59. https://doi.org/10.1016/j.watres.2014.03.064
Silva JRR, Gregório A, Portela-Castro ALB, Fernandes CA. Genotoxicity and cytotoxicity of textile production effluents, before and after Bacillus subitilis bioremediation, in Astyanax lacustris (Pisces, Characidae). Mut Res Genet Toxicol Environ Mutagen. 2023; 886:503588. https://doi.org/10.1016/j.mrgentox.2023.503588
Speit G, Hartmann A. The comet assay (Single-Cell Gel Test): a sensitive genotoxicity test for the detection of DNA damage and repair. DNA Repair Protocols. New Jersey: Humana Press; 1999. p.203–12. https://doi.org/10.1385/1-59259-675-4:203
Tincani FH, Santos GS, Azevedo ACB, Marques AEML, Pereira LS, Castellano GC et al. Climbing the taxonomic ladder: could a genus be used as bioindicator? The ecotoxicological relationship between biomarkers of Astyanax altiparanae, Astyanax bifasciatus and Astyanax ribeirae. Ecol Indic. 2019; 106:105474. https://doi.org/10.1016/j.ecolind.2019.105474
Yanto DHY, Tachibana S. Enhanced biodegradation of asphalt in the presence of Tween surfactants, Mn2+ and H2O2 by Pestalotiopsis sp. in liquid medium and soil. Chemosphere. 2014; 103:105–13. https://doi.org/10.1016/j.chemosphere.2013.11.044
Zhang W, Liu W, Zhang J, Zhao H, Zhang Y, Quan X et al. Characterization of acute toxicity, genotoxicity and oxidative stress posed by textile effluent on zebrafish. J Environ Sci. 2012; 24(11):2019–27. https://doi.org/10.1016/S1001-0742(11)61030-9
Authors
![]() Josiane Rodrigues Rocha da Silva1
Josiane Rodrigues Rocha da Silva1 ![]() ,
, ![]() Cleiton Campos da Silva2,
Cleiton Campos da Silva2, ![]() Layon Zafra-Lemos3,
Layon Zafra-Lemos3, ![]() Ana Luiza de Brito Portela-Castro1,4 and
Ana Luiza de Brito Portela-Castro1,4 and ![]() Carlos Alexandre Fernandes1,4
Carlos Alexandre Fernandes1,4
[1] Programa de Pós-Graduação em Biotecnologia Ambiental, Departamento de Biotecnologia, Genética e Biologia Celular, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil. (JRRS) josianerrs@hotmail.com (corresponding author), (ALBPC) albpcastro@nupelia.uem.br, (CAF) cafernandes@uem.br.
[2] Faculdade UMFG, Rodovia PR-082, km 468, 87200-000 Cianorte, PR, Brazil. (CCS) cleiton@ciacampos.com.br.
[3] Centro Universitário Unifatecie, Rua Getúlio Vargas, 333, 87709-000 Paranavaí, PR, Brazil. (LZL) layonzafra@hotmail.com.
[4] Núcleo de Pesquisa em Limnologia, Ictiologia e Aquicultura (NUPELIA), Centro de Ciências Biológicas, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil.
Authors’ Contribution 

Josiane Rodrigues Rocha da Silva: Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Writing-original draft.
Cleiton Campos da Silva: Data curation, Methodology, Visualization.
Layon Zafra-Lemos: Data curation, Software.
Ana Luiza de Brito Portela-Castro: Validation, Visualization, Writing-review and editing.
Carlos Alexandre Fernandes: Project administration, Supervision, Validation, Visualization, Writing-review and editing.
Ethical Statement
The Ethics Committee approved all procedures for using Animals in Research (CEUA – UEM), license No 3062160921.
Competing Interests
The author declares no competing interests.
How to cite this article
Silva JRR, Silva CC, Zafra-Lemos L, Portela-Castro ALB, Fernandes CA. Cyto and genotoxicity induced by exposure to textile production effluents on Astyanax lacustris (Pisces: Characidae) can be reduced after treatment through a vertical flow constructed wetland system. Neotrop Ichthyol. 2025; 23(2):e250007. https://doi.org/10.1590/1982-0224-2025-0007
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted April 24, 2025
Accepted April 24, 2025
![]() Submitted January 17, 2025
Submitted January 17, 2025
![]() Epub July 11, 2025
Epub July 11, 2025