![]() Luiz Fernando Caserta Tencatt1
Luiz Fernando Caserta Tencatt1 ![]() ,
, ![]() Vandergleison de Carvalho2,
Vandergleison de Carvalho2, ![]() Hugmar Pains da Silva3 and
Hugmar Pains da Silva3 and ![]() Marcelo Ribeiro de Britto2
Marcelo Ribeiro de Britto2
PDF: EN XML: EN | Cite this article
Abstract
Uma espécie nova de Osteogaster é descrita das bacias dos rios Aripuanã e Juruena, sistema do rio Amazonas, se diferenciando de suas congêneres por apresentar a seguinte combinação de seguintes características: (I) uma faixa dorsolateral escura, oblíqua e conspícua nos flancos, da primeira placa corporal dorsolateral até a porção média da base da nadadeira caudal, variavelmente se estendendo difusa no lobo ventral da nadadeira caudal, e (II) porção posterolateral do escapulocoracóide moderadamente desenvolvido, não expandido mesialmente. À luz dos novos dados trazidos em uma publicação recente liderada pelo primeiro autor, Osteogaster é aqui diagnosticado pela seguinte combinação de características: (I) mesetmóide de tamanho moderado, (II) margem posterior do espinho da nadadeira peitoral com todas ou quase todas as serrilhas direcionadas para a extremidade do espinho ou perpendicularmente a ele, (III) expansão laminar posterior do infraorbital 2 em contato com o pterótico-extrascapular, (IV) margem anterodorsal do infraorbital 2 expandida anteriormente, e (V) coloração geral do corpo variando de laranja-amarelado a laranja-avermelhado, apresentando apenas uma única e grande mancha escura nas laterais; todas as nadadeiras desprovidas de manchas escuras. A distribuição e a exclusividade das principais características morfológicas presentes nos distintos gêneros de Corydoradinae são discutidas. Além disso, é fornecida uma chave de identificação para as espécies deOsteogaster.
Palavras-chave: Corydoradinae, Corydoras sp. CW68, Rio Aripuanã, Rio Juruena, Taxonomia.
Introduction
Callichthyidae armored catfishes are unique among Siluriformes in possessing two longitudinal series of dermal plates along their flanks, and encompass nearly 230 valid species distributed into two subfamilies, Callichthyinae and Corydoradinae (Reis, 2003; Fricke et al., 2025). Until recently, Corydoradinae harbored three genera, namely: Aspidoras Ihering, 1907, Corydoras Lacépède, 1803 and Scleromystax Günther, 1864, following the classification proposed by Britto (2003). Despite not proposing any changes on Corydoradinae classification, the phylogenetic study (mostly based on mitochondrial DNA) by Alexandrou et al. (2011) established nine lineages of species, which were widely used as the main reference to recognize groups of species and/or diagnose species within Corydoras sensu lato (e.g., Tencatt et al., 2013, 2016, 2019, 2020, 2021, 2022a, 2023a,b; Tencatt, Pavanelli, 2015; Tencatt, Britto, 2016; Tencatt, Evers, 2016; Tencatt, Ohara, 2016a,b; Bono et al., 2019; Bentley et al., 2021).
Although most of the lineages proposed in Alexandrou et al. (2011) are supported by morphological features, it was never possible to provide a solid diagnosis between lineages 4 and 5 (Tencatt, Pavanelli, 2015; Bono et al., 2019), and lineages 6 and 9 (Tencatt, Ohara, 2016a; Tencatt et al., 2022a, 2023b). Contrary to the case involving lineages 4 and 5, which form a monophyletic clade, lineages 6 and 9 appeared as clearly different clades in Alexandrou et al. (2011). The remarkable morphological similarity between the species from the lineages 6 and 9 when contrasted with the fact that they appeared as clearly different clades in the phylogenetic hypothesis of Alexandrou et al. (2011), was surely one of the main reasons why no changes on the classification of Corydoradinae were proposed at that time. In 2018, a nuclear-based phylogenetic hypotheses using restriction-site-associated DNA markers showed, with strong support for all branches, that the lineages 6 and 9 form a monophyletic group (Marburger et al., 2018).
Recently, a comprehensive phylogenetic study of the Corydoradinae based on Ultraconserved Elements was carried out by Dias et al. (2024), establishing seven lineages within the subfamily with support of both molecular and morphological data. Similarly to the study by Marburger et al. (2018), the results presented by Dias et al. (2024) are overall similar to Alexandrou et al. (2011), differing from it by recovering the species within the lineages 6 and 9 sensu Alexandrou et al. (2011) in a monophyletic clade. As previously demonstrated (Alexandrou et al., 2011; Marburger et al., 2018), Dias et al. (2024) recovered Corydoras as paraphyletic, with the clade harboring C. geoffroy Lacépède, 1803, type-species of the genus, as a sister group to a large clade composed by six subclades including the remaining Corydoradinae, two of which the Aspidoras and Scleromystax clades, and the other four composed by species previously allocated in Corydoras, all of them with available generic names. Therefore, in order to reestablish the monophyly of Corydoras, Dias et al. (2024) proposed the resurrection of four genera previously in the synonymy of Corydoras, namely: Brochis Cope, 1871, Gastrodermus Cope, 1878, Hoplisoma Swainson, 1838, and Osteogaster Cope, 1894.
Osteogaster is a relatively small clade composed by O. aenea (Gill, 1858) and its relatives, which can be distinguished from the remaining genera in Corydoradinaeby having the following combination of features: mesethmoid ranging from short to moderate in size (vs. conspicuously short in Gastrodermus; long in Corydoras; moderate-sized to extremely large in Brochis); posterior margin of pectoral-fin spine with all or nearly all serrations directed towards the tip of the spine or perpendicularly directed (vs. mostly directed towards the origin of spine in Brochis, Corydoras, Gastrodermus, except G. hastatus and G. pygmaeus, and Scleromystax); posterior laminar expansion of infraorbital 2 contacting pterotic-extrascapular (vs. laminar expansion typically not in contact with pterotic-extrascapular in Aspidoras and Hoplisoma; not contacting in Scleromystax); and ground color of body in shades of orange, with only a single, large dark patch on flanks; all fins devoid of dark spots (vs. ground color of body pale yellow, brownish yellow or grayish yellow, typically with small dark spots at least in some part of the body and/or more than one large, dark patch on body in most species of remaining genera) (Dias et al., 2024).
Currently, Osteogaster harbors seven species, namely: O. aenea, O. eques (Steindachner, 1876), O. melanotaenia (Regan, 1912), O. rabauti (LaMonte, 1941), O. zygata (Eigenmann & Allen, 1942), O. maclurei (Tencatt, Gomes & Evers, 2023), and O. hephaestus (Ohara, Tencatt & Britto, 2016) (Tencatt et al., 2023a; Dias et al., 2024). Additionally, 15 putatively undescribed species are known from the aquarium trade, namely: Osteogaster sp. CW7, CW9, CW10, CW14, CW23, CW26, CW41, CW43, CW68, CW78, CW84, CW93, CW97, CW139, and CW194, suggesting that the currently known diversity of the genus is underestimated.
Among the coded species stands out Osteogaster sp. CW68, considered to occur in the rio Aripuanã basin, a tributary of the rio Madeira basin, especially by its peculiar color pattern, which is characterized by an oblique dorsolateral longitudinal dark stripe on flank, resembling the color pattern of O. rabauti and O. zygata. The analysis of specimens from the rio Aripuanã basin allowed us to confirm an undescribed Osteogaster compatible with Osteogaster sp. CW68, which is formally described herein. Additionally, specimens from the rio Juruena basin, a tributary of the rio Tapajós, are considered herein as representatives of this new species. An identification key to the species of Osteogaster, as well as an updated diagnosis to the genus, are provided.
Material and methods
The generic placement of the new species was based on the morphological diagnoses compiled in Dias et al. (2024), with modifications of Tencatt et al. (2025); the classification for Corydoradinae follows Dias et al. (2024). Measurements were obtained using digital calipers to the nearest tenth of millimeter. Morphometric and meristic data were taken following Tencatt et al. (2022b) and Reis (1997), respectively. Morphometrics are reported as percent of standard length (SL) or head length (HL). Terminology of barbels follows Britto, Lima (2003). For the osteological analysis, some specimens were cleared and stained (c&s) according to the protocol of Taylor, Van Dyke (1985). Osteological terminology was based on Reis (1998), except for the use of parieto-supraoccipital instead of supraoccipital (Arratia, Gayet, 1995), pterotic-extrascapular instead of pterotic-supracleithrum (Slobodian, Pastana, 2018), and scapulocoracoid instead of coracoid (Lundberg, 1970). Additionally, the ischiac process of the basipterygium is further divided into a dorsal and a ventral process following Huysentruyt, Adriaens (2005). Nomenclature of the laterosensory canals and preopercular pores are according to Schaefer, Aquino (2000) and Schaefer (1988), respectively. The supra-preopercle sensu Huysentruyt, Adriaens (2005) was treated here as a part of the hyomandibula according to Vera-Alcaraz (2013). To determine the development degree of the anterior laminar expansion of infraorbital 1 in relation to the nasal capsule, the specimen was positioned to maintain the largest diameter of the nasal capsule horizontally. The width of frontal bone was obtained at the same point as the least interorbital width. Vertebral counts include only free centra (i.e., post-Weberian complex), with the compound caudal centrum (preural 1+ ural 1) counted as a single element. The last two dorsal-fin rays were counted as distinct elements. Pharyngeal teeth were counted in both sides of the branchial arches.
Although no conspicuous morphological differences were found between the populations from the rivers Aripuanã and Tapajós, we designated only the specimens from the rio Aripuanã basin, where the new species was discovered, as paratypes. This conservative decision is justified by the fact that these populations occur in quite distinct drainages, and future analyses may eventually show that they represent different species. In the description, numbers in parentheses represent the total number of specimens with those counts. Numbers with an asterisk refer to the counts of the holotype. Institutional abbreviations follow Sabaj (2025). As for political entities, the geographic information of the type-specimens was provided in the following order: country, state, and municipality. This study was based on museum specimens, and no collecting permit was necessary. The conservation status of the new species was suggested using the categories and criteria of the International Union for Conservation of Nature guidelines (IUCN Standards and Petitions Subcommittee, 2024).
Results
Osteogaster oharai, new species
urn:lsid:zoobank.org:act:ED4C6156-E673-4ACA-986C-964760E6BF04
(Fig. 1)
Corydoras aff. rabauti. —Silva et al., 2019:615 (tab. 3; list of species).
Holotype. MNRJ 55884, 43.4 mm SL, Brazil, Mato Grosso, Aripuanã, stream tributary of the rio Aripuanã, upstream Salto das Andorinhas and Dardanelos waterfalls, rio Madeira basin, 10°16’46”S 59°33’08”W, 23 Jul 2014, H. P. Silva.
Paratypes. All from Brazil, Mato Grosso, Aripuanã (except when noted), rio Aripuanã basin upstream Salto das Andorinhas and Dardanelos waterfalls, rio Madeira basin. CPUFMT 4889, 2 c&s, 35.8–44.7 mm SL, stream tributary of the rio Aripuanã, 10°20’29”S 59°28’23”W, 20 Jul 2014, H. P. Silva. CPUFMT 4934, 5, 29.9–39.9 mm SL, stream tributary of the rio Aripuanã, 10°23’35”S 59°28’01”W, 24 Jul 2014, H. P. Silva. CPUFMT 5015, 2, 34.4-35.4 mm SL, stream tributary of the rio Aripuanã, 10°21’32”S 59°33’06”W, 24 Jul 2014, H. P. Silva. CPUFMT 5032, 1, 26.2 mm SL, stream tributary of the rio Aripuanã, 10°13’49”S 59°24’08”W, 18 Jul 2014, H. P. Silva. CPUFMT 5106, 2, 25.0–36.7 mm SL, stream tributary of the rio Aripuanã, 10°16’36”S 59°26’53”W, 20 Jul 2014, H. P. Silva. INPA 11590, 1, 33.1 mm SL; INPA 11721, 1, 34.4 mm SL; INPA 12130, 1, 33.3 mm SL; INPA 12131, 3, 29.7 – 30.1 mm SL, stream tributary of the rio Aripuanã, 10°10’00”S 59°27’34”W, 9 Nov 1976, INPA staff. UFRO-ICT 21340, 5 of 11, 34.1–42.8 mm SL, stream tributary of the rio Aripuanã, 10°10’42”S 59°27’26”W, 20 Jul 2013, D. Hungria. UFRO-ICT 21931, 5 of 10, 32.1–42.5 mm SL, stream tributary of the rio Aripuanã, 10°11’43”S 59°26’58”W, 19 Jul 2013, W. M. Ohara. UFRO-ICT 22240, 3, 24.5–31.3 mm SL, igarapé do Areião, 10°27’26”S 59°22’08”W, 21 Jul 2013, W. M. Ohara. UFRO-ICT 22456, 1, 41.7 mm SL, Juína, stream tributary of the rio Aripuanã, 10°53’12”S 59°11’37”W, 23 Jul 2013, B. Barros. CPUFMT 5077, 1, 40.9 mm SL, collected with the holotype.
Non-type specimens. All from Brazil, Mato Grosso, Cotriguaçu (except when noted), rio Juruena basin, rio Tapajós basin. UFRO-ICT 21258, 3, 26.4–37.2 mm SL, Juruena, 10°21’02”S 58°30’08”W, 22 Jul 2013, D. Hungria. UFRO-ICT 22449, 4 of 7, 23.6–27.7 mm SL, Juína, igarapé do Boi Preto, 11°13’06”S 58°54’53”W, 11 Sep 2013, W. M. Ohara. CITL 1504, 16 of 19, 25.8–37.7 mm SL, 3 c&s of 19, 38.3–41.1 mm SL, stream with unknown name, 09°51’36”S 58°28’34”W, 28 Oct 2010, I. M. Fernandes. CITL 1505, 6, 26.0–38.9 mm SL, stream with unknown name, 09°49’14”S 58°27’50”W, 28 Oct 2010, I. M. Fernandes. CITL 1506, 12, 24.4–37.3 mm SL, stream with unknown name, 09°52’09”S 58°28’39”W, 28 Oct 2010, I. M. Fernandes. CITL 1507, 2, 28.3–37.0 mm SL, stream with unknown name, 09°51’36”S 58°28’34”W, 29 Oct 2010, I. M. Fernandes. CITL 1508, 6, 26.7–37.8 mm SL, stream with unknown name, 09°52’09”S 58°28’39”W, 29 Oct 2010, I. M. Fernandes. CPUFMT 8621, 633, 19.7–43.2 mm SL; MNRJ 55728, 57 of 60, 21.6–39.7 mm SL, 3 c&s of 60, 35.6–41.1 mm SL, stream with unknown name, 09°48’43”S 58°15’50”W, Oct 2010, T. Izzo. MZUSP 130834, 11, 21.9–37.3 mm SL, stream with unknown name, 09°51’36”S 58°28’34”W, 27 Oct 2010, I. M. Fernandes. NUP 25764, 27, 22.4–39.7 mm SL, stream with unknown name, 09°52’09”S 58°28’39”W, 27 Oct 2010, I. M. Fernandes.
FIGURE 1| Osteogaster oharai, holotype, MNRJ 55884, 43.4 mm SL, Aripuanã, Mato Grosso, Brazil, stream tributary of the rio Aripuanã, upstream Salto das Andorinhas and Dardanelos waterfalls, rio Madeira basin.
Diagnosis. Osteogaster oharai can be distinguished from its congeners, except O. eques, O. rabauti, and O. zygata by having an oblique dorsolateral longitudinal dark stripe on flanks, starting at first dorsolateral body plate and gradually descending towards middle portion of caudal-fin base, variably extending diffuse on ventral lobe of caudal fin (vs. anterior portion of flank with large, conspicuous roughly rounded to longitudinally elliptical dark brown or black blotch, variably extending towards caudal-fin base, forming a somewhat mace-shaped longitudinal patch running parallel to longitudinal axis of the body or slightly inclined upwards anteriorly, with wider portion on anterior half of trunk, gradually becoming narrower posteriorly in O. aenea; almost entirely dark brown or black in O. hephaestus; a large, conspicuous dark brown or black blotch on anterodorsal portion of flank; blotch somewhat rounded to roughly diamond shaped in O. maclurei; a longitudinal dark stripe almost entirely covering dorsal and middle portion of flank, running in parallel to longitudinal axis of the body or slightly inclined upwards anteriorly, with anterior portion slightly wider, smoothly becoming narrower posteriorly in O. melanotaenia). The new species can be distinguished from O. eques, O. rabauti and O. zygata, by having moderately-developed posterolateral portion of scapulocoracoid, not expanded mesially (vs. strongly well-developed posterolateral portion of scapulocoracoid, conspicuously expanded mesially, contacting its counterpart and almost entirely covering ventral surface of trunk), and by having dorsolateral stripe not extending anteriorly and meeting its counterpart on head (vs. dorsolateral stripe extending anteriorly on head, converging mesially and meeting its counterpart on parieto-supraoccipital, forming a V-shaped pattern in dorsal view, with anteriormost portion variably reaching the frontals); it can be further distinguished from O. eques by having conspicuous dorsolateral dark stripe on flanks (vs. dorsolateral stripe indistinct from dark patch almost entirely covering flanks, conspicuous only on predorsal region of body).
Description. Morphometric data in Tab. 1. Head laterally compressed with convex dorsal profile, roughly triangular in dorsal view. Snout short to intermediate long, ranging from smoothly rounded to somewhat straight, variably with smoothly concave area on region of mesethmoid. Head profile convex from tip of snout to anterior nares, ascending nearly straight or slightly convex from this point to dorsal-fin origin; region of frontal fontanel variably slightly concave. Profile nearly straight to slightly convex along dorsal-fin base. Postdorsal-fin body profile slightly concave to adipose-fin spine, slightly concave from this point to caudal-fin base; region between dorsal and preadipose platelets typically ranging from smoothly concave to roughly straight. Ventral profile of body nearly straight or slightly convex from isthmus to pectoral girdle, and slightly convex from this point until pelvic girdle. Profile nearly straight to slightly convex from pelvic girdle to base of first anal-fin ray, ascending slightly concave until caudal-fin base. Body roughly elliptical in cross section at pectoral girdle, gradually becoming more compressed toward caudal fin. Highest body depth at vertical through anterior origin of dorsal fin.
TABLE 1 | Morphometric data of the holotype, 16 paratypes and 19 non-type specimens of Osteogaster oharai. SD = Standard deviation.
| Holotype | Low–High | Mean±SD |
Standard length (mm) | 43.4 | 25.0–43.4 | 34.5±4.2 |
Percentage of standard length | |||
Depth of body | 36.8 | 35.4–45.4 | 39.0±2.4 |
Predorsal distance | 52.3 | 46.1–60.7 | 54.3±2.9 |
Prepelvic distance | 44.6 | 44.6–54.2 | 48.4±2.4 |
Preanal distance | 75.3 | 75.3–83.5 | 80.0±2.1 |
Preadipose distance | 81.7 | 81.7–90.3 | 85.9±2.1 |
Length of dorsal spine | 15.4 | 13.5–23.1 | 16.9±2.1 |
Length of pectoral spine | 20.7 | 20.7–30.0 | 24.7±2.0 |
Length of adipose-fin spine | 6.4 | 5.6–11.4 | 7.9±1.3 |
Depth of caudal peduncle | 14.9 | 14.4–18.7 | 16.3±0.9 |
Length of dorsal-fin base | 18.1 | 16.0–21.6 | 18.4±1.4 |
Dorsal to adipose distance | 22.6 | 17.1–25.2 | 21.7±1.9 |
Maximum cleithral width | 25.8 | 23.8–29.2 | 26.1±1.3 |
Head length | 42.4 | 41.5–51.4 | 44.8±2.0 |
Length of maxillary barbel | 13.1 | 9.0–19.7 | 14.6±2.3 |
Percentage of head length | |||
Head depth | 82.5 | 76.5–98.3 | 81.8±3.8 |
Least interorbital distance | 32.2 | 29.4–40.9 | 34.3±2.8 |
Horizontal orbit diameter | 18.3 | 16.2–20.4 | 18.5±1.1 |
Snout length | 41.1 | 34.0–41.1 | 38.1±1.9 |
Least internarial distance | 17.5 | 13.0–18.9 | 16.6±1.3 |
Eye rounded, located dorsolaterally on head. Orbit delimited anteriorly by lateral ethmoid, anterodorsally by frontal, posterodorsally by sphenotic, posteroventrally by infraorbital 2, and anteroventrally by infraorbital 1 (Fig. 2). Anterior and posterior nares close to each other, only separated by flap of skin. Anterior naris tubular. Posterior naris close to anterodorsal margin of orbit, separated from it by distance typically similar to naris diameter; slightly larger than naris diameter in some specimens. Mouth small, subterminal, width similar to bony orbit diameter. Maxillary barbel ranging from relatively short, not reaching posteroventral edge of opercle, to relatively long in size, slightly surpassing anteroventral limit of gill opening. Outer mental barbel typically slightly longer than maxillary barbel; outer mental barbel variably with similar size, or slightly shorter than maxillary barbel, apparently due to barbel damage in some specimens. Inner mental barbel fleshy, base of each counterpart slightly separated from each other. Area at mouth corner, ventral to maxillary barbel, typically with small wrinkle of skin. Small rounded papillae covering entire surface of all barbels, upper and lower lips, snout and isthmus.
FIGURE 2| Head osteological pattern of Osteogaster oharai, showing the general morphology in lateral view of (A) CPUFMT 4889, 44.7 mm SL, and (B) CITL 1504, 41.1 mm SL. Abbreviations: f: frontal, io1–2: infraorbital 1 and 2, iop: interopercle, le: lateral ethmoid, n: nasal, me: mesethmoid, op: opercle, pes: pterotic-extrascapular, pop: preopercle, prh: posterodorsal ridge of hyomandibula, pso: parieto-supraoccipital, sph: sphenotic. Area where the illustrated bones are located in fish’s body marked in red in the miniature photo of an uncatalogued specimen in life (lateral view). Scale bars = 1 mm.
Mesethmoid moderate-sized, its length slightly smaller than frontal length; anterior tip roughly straight in lateral view, relatively long, with size similar to or slightly larger than 50% of bone length; lateral cornua variably present; posterior margin relatively narrow, its width smaller than maximum width of posterior portion of bone; base typically relatively long, lateral posterodorsal expansion with relatively wide roughly triangular or trapezoid external projection, emerging relatively distant to distal point of suture with frontal; relatively short base, emerging relatively close to distal point of suture with frontal, or relatively narrow external projection in some specimens; external projection typically relatively long in size, distal tip slightly surpassing outermost margin of nasal bone, or variably short, not surpassing outermost margin of nasal bone; posterodorsal portion typically partially exposed and bearing small odontodes, variably almost entirely covered by thick skin layer, with tiny exposed portion; posterior ventrolateral expansion partially visible in dorsal view, emerging slightly anteriorly to external projection of lateral posterodorsal expansion (Fig. 3). Upper and lower jaws edentulous; premaxilla overall funnel-like shaped, with anteroventral surface roughly square-, triangular- or rectangular-shaped in frontal view; anteroventral margin irregular; posterodorsal portion with conspicuous pointed process, mesially set in frontal view (Fig. 4A); maxilla elongated, relatively slender and roughly hatchet shaped in frontal view, proximal half with roughly trapezoid to rounded laminar process on posterolateral portion (Fig. 4A); dentary relatively slender, with roughly trapezoid to triangular expansion on anteroventral portion, in ventrolateral perspective, smoothly bent anteriorly, with distal edge variably smoothly curved posteriorly; roughly triangular process on its posterodorsal portion, bent posteriorly; angulo-articular moderately deep in middle portion, with roughly triangular to rounded dorsal laminar expansion, deepest area slightly posterior to posterodorsal margin of dentary; posteroventral portion with roughly triangular process, in lateral view, bent posteriorly (Fig. 4B). Palatine longitudinally elongated, moderate-sized, with roughly triangular to rounded or trapezoid dorsolateral laminar expansion on its posterior portion, articulating with lateroventral expansion of posterior portion of mesethmoid; dorsolateral laminar expansion roughly falciform, curved anteriorly, in right side of specimen CPUFMT 4889, 35.8 mm SL, apparently due to malformation; posterolateral process well developed, extending posteriorly in parallel to dorsal margin of anterior laminar expansion of metapterygoid (Fig. 4C).
FIGURE 3| Cranial osteology of Osteogaster oharai, showing the detail of the mesethmoid in dorsal (A) and lateral (B) views (CPUFMT 4889, 44.7 mm SL; paratype), and (C) the anterior portion of neurocranium in dorsal view (MNRJ 55728, 40.9 mm SL; non-type specimen). Abbreviations: eplp: external projection of lateral posterodorsal expansion of mesethmoid, f: frontal, io1: infraorbital 1, lc: lateral cornua, le: lateral ethmoid, me: mesethmoid, n: nasal, pes: pterotic-extrascapular, pve: ventrolateral expansion of posterior portion of mesethmoid, sph: sphenotic. Area where the illustrated bones in (A) and (B) are located in fish’s body marked in red in the miniature photo of an uncatalogued specimen in life (lateral view), and in (C) in the miniature photo of the preserved holotype (MNRJ 55884, 43.4 mm SL) in dorsal view. Scale bars = 1 mm.
FIGURE 4| Cranial osteology in a c&s paratype of Osteogaster oharai (CPUFMT 4889, 44.7 mm SL), showing (A) the upper jaw in frontal view (= dorsal view considering everted mouth), (B) the suspensorium plus operculum in lateral view, and (C) the palatine in dorsal view. Abbreviations: aa: angulo-articular, d: dentary, hym: hyomandibula, iop: interopercle, mp: metapterygoid, mx: maxilla, op: opercle, pmx: premaxilla, pop: preopercle, prh: posterodorsal ridge of hyomandibula, q: quadrate. Red arrow indicates laminar process on posterolateral portion of maxilla; yellow and green arrows indicate anteroventral expansion and posterodorsal process of dentary, respectively; orange and blue arrows indicate dorsal laminar expansion and posteroventral process of anguloarticular, respectively. Dotted line in (C) outlining dorsolateral longitudinal laminar expansion of palatine. Area where the illustrated bones are located in fish’s body marked in red in the miniature photo of an uncatalogued specimen in life (lateral view). Scale bars = 1 mm.
Nasal capsule delimited posteriorly and dorsally by frontal, anteriorly by mesethmoid, and ventrally and posteriorly by lateral ethmoid (Figs. 2‒3). Nasal slender, laterally curved, inner margin typically with poorly-developed laminar expansion, contacting only frontal or frontal and mesethmoid; outer margin with strongly reduced laminar expansion, nearly imperceptible in some specimens (Figs. 2B, 3C, 5A). Lateral ethmoid ranging from slender to moderately deep in lateral view, moderately expanded anteroventrally, with anterodorsal expansion clearly separated from nasal, and anterior margin contacting external projection of lateral posterodorsal expansion of mesethmoid (Fig. 3). Frontal elongated, typically narrow, width less than half of entire length, variably relatively wide, width equal to or slightly larger than half of entire length; anterior projection ranging from short, size smaller than nasal length, to moderate, size equal to nasal length (Fig. 6); left side of specimen CITL 1504, 41.1 mm SL, not considered due to malformation of nasal (Fig. 6B). Frontal fontanel large, slender, and somewhat ellipsoid; posterior tip extension slightly surpassing anterior margin of parieto-supraoccipital (Figs. 6A, B, D). Sphenotic somewhat trapezoid, contacting parieto-supraoccipital dorsally, pterotic-extrascapular posteriorly, second infraorbital posteroventrally and frontal anteriorly (Fig. 2). Pterotic-extrascapular roughly pipe-shaped, with posteriormost portion contacting first lateral-line ossicle, posteroventral margin contacting cleithrum, and anteroventral margin contacting opercle and infraorbital 2; posterior expansion almost entirely covering lateral opening of swimbladder capsule, leaving slender area on its dorsal margin covered only by thick layer of skin (Fig. 2). Parieto-supraoccipital wide, posterior process long, variably contacting nuchal plate; close but not in contact with nuchal plate in paratype CPUFMT 4889, 35.8 mm SL (Fig. 6A); posterior process apparently not in contact with nuchal plate externally, but in contact under thick layer of skin in non-type specimen CITL 1504, 41.1 mm SL (Fig. 6B); posterior process in direct contact with nuchal plate externally in paratype CPUFMT 4889, 44.7 mm SL (Fig. 6C); posterior process relatively distant from nuchal plate externally, first dorsolateral body plate contacting its counterpart between both structures in a non-type specimen (MNRJ 55728, 35.6 mm SL; Fig. 6D; first dorsolateral body plate fragmented in one side of specimen, possibly due to malformation); region between posterior process and nuchal plate covered by thick layer of skin (Fig. 6).
FIGURE 5| Cranial osteology in a c&s paratype of Osteogaster oharai (CPUFMT 4889, 44.7 mm SL), showing (A) the nasal bone in dorsal view, and the infraorbital series in lateral (B) and dorsal (C) views. Abbreviations: ade: anterodorsal edge of infraorbirtal 2, io1–2: infraorbital 1 and 2. Red arrows indicate the inner laminar expansion of both infraorbitals; yellow and blue arrows indicate posterodorsal and posterior-middle expansions of infraorbital 2, respectively. Area where the illustrated bones in (A) are located in fish’s body marked in red in the miniature photo of the preserved holotype (MNRJ 55884, 43.4 mm SL) in dorsal view, and in (B) plus (C) in the miniature photo of an uncatalogued specimen in life (lateral view). Scale bars = 1 mm.
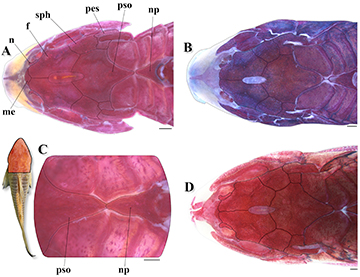
FIGURE 6| Top of head and predorsal region of trunk of Osteogaster oharai in dorsal view, showing the variations related to the contact between the parieto-supraoccipital and nuchal plate. A. Parieto-supraoccipital close but not in contact with nuchal plate in a c&s paratype (CPUFMT 4889, 35.8 mm SL); B. Posterior process apparently not in contact with nuchal plate externally, but in contact under thick layer of skin in a c&s non-type specimen (CITL 1504, 41.1 mm SL); C. Posterior process in direct contact with nuchal plate externally in a c&s paratype (CPUFMT 4889, 44.7 mm SL); D. Posterior process relatively distant from the nuchal plate externally, first dorsolateral body plate contacting its counterpart between both structures in a c&s non-type specimen (MNRJ 55728, 35.6 mm SL). Abbreviations: f: frontal, n: nasal, np: nuchal plate, me: mesethmoid, pes: pterotic-extrascapular, pso: parieto-supraoccipital, sph: sphenotic. Area where the illustrated bones are located in fish’s body marked in red in the miniature photo of the preserved holotype (MNRJ 55884, 43.4 mm SL) in dorsal view. Scale bars = 1 mm.
Two laminar infraorbitals with minute odontodes. Infraorbital 1 large, ventral laminar expansion typically ranging from well-developed to extremely well developed, covering most of lateral surface of snout in some specimens (Figs. 2, 5B); some juvenile specimens (up to about 30.0 mm SL) with moderately-developed ventral expansion; anterior portion generally with conspicuously well-developed laminar expansion, reaching or surpassing anterior margin of nasal capsule; inner laminar expansion poorly developed (Fig. 5C). Infraorbital 2 small, relatively wide, with posterior laminar expansion strongly well developed (Figs. 2, 5B); posteroventral margin contacting posterodorsal ridge of hyomandibula, posterior margin contacting opercle, and posterodorsal edge contacting sphenotic and pterotic-extrascapular (Fig. 2); posterodorsal portion with roughly triangular to trapezoid expansion, and middle portion with moderately- to well-developed roughly triangular expansion, smoothly bent downwards (Figs. 2, 5B), with tip just above dorsal edge of posterodorsal ridge of hyomandibula (Fig. 2); anterodorsal edge expanded anteriorly, reaching about middle portion of ventral margin of sphenotic (Figs. 2, 5B); inner laminar expansion moderately developed (Fig. 5C). Posterodorsal ridge of hyomandibula close to its articulation with opercle slender, exposed, and bearing small odontodes (Figs. 2, 4B). Dorsal ridge of hyomandibula between pterotic-extrascapular and opercle typically covered by posterodorsal edge of infraorbital 2. Interopercle partially covered by thick layer of skin, with posterior portion exposed and bearing odontodes; subtriangular, anterior projection ranging from moderately to well developed (Figs. 2, 4B). Preopercle elongated, relatively slender; minute odontodes on external surface (Figs. 2, 4B). Opercle dorsoventrally elongated, relatively compact in shape, with width equal to or larger than half of entire length; free margin convex, without serrations and covered by small odontodes (Figs. 2, 4B).
Four branchiostegal rays decreasing in size posteriorly. Hypobranchial 1 deep; hypobranchial 2 somewhat triangular, tip ossified and directed towards anterior portion, posterior margin cartilaginous; ossified portion well developed, its size about twice cartilaginous portion. Five ceratobranchials with expansions increasing posteriorly; ceratobranchial 1 with small process on anterior margin of mesial portion; ceratobranchial 3 notched on postero-lateral margin, variably with continuous laminar expansion on postero-lateral margin; ceratobranchial 5 toothed on posterodorsal surface, with 36 to 42 (8) teeth aligned in one row. Four epibranchials with similar size; epibranchial 2 slightly larger than others, with small pointed process on laminar expansion of posterior margin; epibranchial 3 with roughly triangular uncinate process on laminar expansion of posterior margin, uncinate process variably mesially curved. Two wide pharyngobranchials (3 and 4); pharyngobranchial 3 typically with roughly triangular laminar expansion on posterior margin, variably roughly rounded; laminar expansion typically notched. Upper tooth plate roughly oval, 39 to 52 (8) teeth aligned in two rows on posteroventral surface; rows closely aligned.
Lateral-line canal reaching cephalic laterosensory system through pterotic-extrascapular, branching twice before reaching sphenotic: pterotic branch, with single pore, preoperculomandibular branch conspicuously reduced, with single pore opening at postotic main canal; postotic main canal widens just posterior to pterotic branch. Sensory canal continuing through pterotic-extrascapular, reaching sphenotic as temporal canal, which splits into two branches: one branch giving rise to infraorbital canal, other branch connecting to frontal through supraorbital canal, both with single pore. Supraorbital canal branched, running through nasal bone. Epiphyseal branch conspicuously reduced; pore opening close to supraorbital main canal, directed towards frontal fontanel. Nasal canal with three openings, first on posterior edge, second on posterolateral portion and variably fused with first pore, and third on anterior edge. Infraorbital canal running through entire infraorbital 2, extending to infraorbital 1 and typically opening into two pores. Preoperculomandibular branch giving rise to preoperculo-mandibular canal, which runs through almost entire preopercle with three openings, leading to pores 3, 4, and 5, respectively.
Dorsal fin subtriangular, typically located just posterior to second or third dorsolateral body plate. Dorsal-fin rays II,8* (35), II,9 (1), with first or second branched rays as longest fin element, both typically with similar size, remaining branched rays typically decreasing in size posteriorly, with first, second and third branched rays slightly longer than ossified portion of dorsal-fin spine, remaining rays with similar size or shorter than spine; first branched rays (variably up to fifth) similar-sized, and just slightly longer than ossified portion of spine in some specimens; posterior margin of dorsal-fin spine with five to 13 strongly reduced serrations; most serrations antrorse; some serrations variably perpendicularly directed (in relation to main axis of spine), especially on distal portion of spine; bifid serrations variably present; serrations absent close to origin of spine; small odontodes on anterior and lateral surfaces of spine (Fig. 7A). Nuchal plate moderately developed, almost entirely exposed, with minute odontodes. Spinelet short; spine ranging from poorly developed, with adpressed distal tip not reaching posterior origin of dorsal-fin base, to moderately developed, with adpressed distal tip reaching or slightly surpassing posterior origin of dorsal-fin base. Pectoral fin roughly triangular, its origin just posterior to gill opening. Pectoral-fin I,7 (2), I,7,i (3), I,8* (29), one specimen with I,6 on left pectoral fin, apparently due to malformation; first branched ray typically as longest fin element; branched rays decreasing in size posteriorly, with first and second, and variably third, branched rays slightly longer than ossified portion of pectoral-fin spine, remaining rays with similar size or shorter than spine; some specimens with first and second branched rays with similar size; posterior margin of pectoral-fin spine with 18 to 25 strongly reduced to poorly-developed serrations along almost its entire length, absent close to origin of spine; typically, most serrations antrorse, with some serrations perpendicularly directed (in relation to main axis of spine) and/or bifid or trifid; serrations fused at base variably present; some specimens with most serrations smoothly antrorse, nearly perpendicular in relation to main axis of spine; small odontodes on anterior, dorsal and ventral surfaces of spine (Figs. 7B, C). Anteroventral portion of cleithrum and anterolateral portion of scapulocoracoid exposed; posterolateral portion of scapulocoracoid moderately developed, exposed, with anterior portion slightly to moderately expanded anteriorly, not in contact with anteroventral portion of cleithrum; exposed areas bearing small odontodes. Opening of axillary gland sensu Kiehl et al. (2006) located just posterior to pectoral-fin spine base.
FIGURE 7| Lateral view of (A) the dorsal-fin spine and dorsal view of (B) the left pectoral-fin spine of a c&s paratype (CPUFMT 4889, 44.7 mm SL), and of (C) the left pectoral-fin spine of a non-type specimen (MNRJ 55728, 41.1 mm SL) of Osteogaster oharai, showing their serration patterns. Area where the illustrated bones are located in fish’s body marked in red in the miniature photo of an uncatalogued specimen in life (lateral view). Scale bars = 1 mm.
Pelvic fin oblong, typically located just below second ventrolateral body plate, and at vertical through first or second dorsal-fin branched ray. Pelvic-fin rays i,5* (36); second branched ray typically as longest fin element, with rays decreasing in size towards both anterior and posterior margins of fin; unbranched ray as shorter fin element, or similar in size to last branched ray. Anterior internal process of basipterygium well developed and moderately laterally expanded, with obliquely placed dorsal lamina, smoothly converging mesially towards anterior edge of process; ventral laminar expansion obliquely placed, converging mesially towards anterior edge of process, clearly less developed than dorsal lamina; anterior external process laminar, somewhat sickle-shaped, roughly rounded or quadrangular, or falciform (see Britto, 2003:141, fig. 22C), well developed and slightly to moderately expanded posteriorly; dorsal ischiac process well developed, with anterior laminar expansion irregular to roughly triangular, trapezoid or rounded, slightly to moderately expanded anteriorly, and posterior laminar expansion roughly triangular or rounded, moderately expanded posteriorly; ventral ischiac process clearly smaller than dorsal process, roughly triangular, bent anteriorly (Fig. 8); specimen MNRJ 55728, 41.1 mm SL, with anterior internal process of basipterygium conspicuously narrow, anterior external process irregular, and right dorsal ischiac process just smoothly expanded anteriorly, apparently due to malformations (Fig. 8D). Adipose fin roughly triangular, separated from base of last dorsal-fin ray by six to eight dorsolateral body plates. Anal fin subtriangular, located just posterior to 11th, 12th or 13th ventrolateral body plates, and at vertical through adipose-fin spine base or region of preadipose platelets. Anal-fin rays ii,5,i (3), ii,6* (31); third ray typically as longest fin element, with rays decreasing in size towards both anterior and posterior margins of fin; fourth ray variably similar in size to third ray; first or last ray as shorter fin element, both variably similar in size. Caudal fin bilobed, with dorsal and ventral lobes similar in size or dorsal lobe slightly larger than ventral lobe; some specimens undergoing caudal regeneration with dorsal lobe smaller than ventral lobe. Caudal-fin rays i,11,i (1), i,12,i* (35), with generally five dorsal and ventral procurrent rays increasing in size posteriorly; small cartilage between upper principal and procurrent caudal-fin rays not observed (Fig. 9).
FIGURE 8| Pelvic girdle in c&s specimens of Osteogaster oharai, showing its morphological variations in dorsal view, depicting (A) a paratype (CPUFMT 4889, 44.7 mm SL), and three non-type specimens, (B) MNRJ 55728, 35.6 mm SL, (C) MNRJ 55728, 40.9 mm SL, and (D) MNRJ 55728 41.1 mm SL. Abbreviations: bp: basipterygium, pae: anterior external process, pai: anterior internal process, pi: dorsal ischiac process. Pelvic girdle position shown in red in the miniature photo of the preserved holotype (MNRJ 55884, 43.4 mm SL) in ventral view. Scale bars = 1 mm.
FIGURE 9| General morphology of caudal skeleton in a c&s paratype of Osteogaster oharai (CPUFMT 4889, 44.7 mm SL). Abbreviations: ccc: compound caudal centrum, cfr: caudal-fin principal rays, dpcr: dorsal procurrent rays, epu: epural, has: haemal spine, hyp 1–5: hypurals 1 to 5, nes: neural spine, par: parhypural, pu 2–4: preural centra 2 to 4, un: uroneural, vpcr: ventral procurrent rays. Area where the illustrated bones are located in fish’s body marked in red in the miniature photo of an uncatalogued specimen in life (lateral view). Scale bar = 1 mm.
Two to four laterosensory canals on trunk; first ossicle tubular, second ossicle laminar, both bearing small odontodes; third and fourth, when present, encased in third and fourth dorsolateral body plates, respectively. Body plates with minute odontodes scattered over exposed area, with conspicuous line of odontodes confined to posterior margins. Dorsolateral body plates 23 (12), 24* (24); ventrolateral body plates 20 (6), 21* (26), 22 (4). Dorsolateral body plates along dorsal-fin base 6 (24), 7* (12); dorsolateral body plates between adipose- and caudal-fin 6 (2), 7* (23), 8 (10), 9 (1). Preadipose platelets 2 (10), 3 (20), 4 (4), 5* (2). Ventral surface of trunk between posteroventral margin of cleithrum and pelvic-fin origin typically laterally delimited only by first ventrolateral body plate, variably by first and second ventrolateral body plates; ventral portion of first ventrolateral body plate ranging from slightly to moderately expanded anteriorly. Small platelets covering base of caudal-fin rays; small platelets disposed dorsally and ventrally between junctions of lateral plates on posterior portion of caudal peduncle. Anterior margin of orbit, above region of junction between frontal and lateral ethmoid, variably with small-sized platelets bearing odontodes. Ventral surface of trunk devoid of platelets, or covered by sparse and conspicuously small platelets in some specimens; platelets irregular in shape and bearing odontodes.
Vertebral count 21 (2), 22 (6); ribs 6 (5), 7 (3); first pair conspicuously large, middle portion closely connected to first ventrolateral body plate; tip close but not directly connected to anterior external process of basipterygium. Parapophysis of complex vertebra ranging from moderately to well developed.
Coloration in alcohol. Overall color pattern variations in Figs. 1 and 10. Ground color of body pale- to brownish yellow or beige, with top of head dark brown. Dorsal and lateral surface of head, and lateral surface of cleithrum covered by dark brown or black chromatophores, not forming blotches; posterior margin of cleithrum typically with conspicuous concentration of dark brown or black chromatophores, forming thin dark line, more evident on dorsal half of cleithrum. Border of pores of laterosensory canals typically with conspicuous concentration of dark brown or black chromatophores. Flank with oblique dorsolateral longitudinal dark brown or black stripe, typically starting at first dorsolateral body plate and gradually descending towards midventral portion of caudal-fin base, variably extending diffuse on ventral lobe of caudal fin; stripe slightly less inclined downwards, ending at middorsal portion of caudal-fin base in some specimens; stripe variably less evident on predorsal portion of flank, fragmented and/or not reaching caudal-fin base; stripe typically interrupted on dorsolateral body plate just ventral to dorsal-fin spine base; remaining portions of dorso- and ventrolateral body plates covered by dark brown or black chromatophores, not forming blotches. Ventrolateral body plates with conspicuous concentration of dark brown or black chromatophores on their anterior or middle portion, forming diffuse, transversally elongated patches, typically more evident on anterior half of flank; ventral portion of ventrolateral plates of body, especially on region around pelvic fin, typically devoid of or with sparse chromatophores. Posterior margin of body plates typically with conspicuous concentration of dark brown or black chromatophores, forming thin dark lines; such pattern variably absent or less evident in some body plates. Fins covered by dark brown or black chromatophores, not forming blotches; dorsal, pectoral, pelvic and caudal fins with chromatophores typically more concentrated on rays; dorsal-fin membranes variably with concentrations of dark brown or black chromatophores, especially close to dorsal-fin spine and/or on proximal portion of fin; ventral caudal-fin lobe slightly darker than dorsal lobe, especially on its proximal portion in some specimens.
FIGURE 10| Variations on snout shape and color pattern in paratypes of Osteogaster oharai, showing left and right side of specimen UFRO-ICT 21931, 42.5 mm SL, and left side of specimen CPUFMT 5015, 35.4 mm SL.
Coloration in life. Similar to color pattern of preserved specimens, but with lighter ground color of body, which is typically greyish orange; specimens in larval stage with typical dark blotched pattern, and juveniles with large, ill-defined roughly elliptical dark patch on anterodorsal portion of trunk (Fig. 11). Some specimens alternatively with greyish yellow ground color of body, which tends to be brighter on pectoral and pelvic fins (Fig. 11E). Body covered by greenish yellow iridescent coloration; some specimens with yellow to green bright patches on opercle (Figs. 11B, C, D, F).
FIGURE 11| Variation in general body shape and color pattern in life of uncatalogued and unmeasured aquarium specimens of Osteogaster oharai, showing (A) specimens in larval stage, (B) a juvenile specimen, and (C–F) adult specimens. Except for specimen (F), which is from the rio Juruena basin, remaining specimens are from the rio Aripuanã basin. Photos (A–C) by Ingo Seidel, (D) by Hans Evers, (E) by Willian Ohara, and (F) by LFCT.
Sexual dimorphism. As well-documented in Corydoradinae (see Nijssen, Isbrücker, 1980a; Britto, 2003; Spadella et al., 2017), male specimens of Osteogaster oharai present a genital papilla, which is somewhat tubular or lanceolate in shape.
Geographical distribution. Osteogaster oharai is currently known from its type-locality, the rio Aripuanã basin, rio Madeira drainage, and from the rio Juruena basin, rio Tapajós drainage, both in Mato Grosso State, Brazil (Fig. 12).
FIGURE 12| Map showing the known geographic records of Osteogaster oharai (yellow star: type-locality, a stream tributary of the rio Aripuanã; purple circles: additional records in the rio Aripuanã basin; red triangles: records in the rio Juruena basin) in Mato Grosso State, Brazil.
Ecological notes. In the rio Aripuanã basin, the new species was captured in small streams tributaries of the rio Aripuanã upstream the Dardanelos-Andorinhas waterfalls complex (Silva et al., 2019:610, figs. 2A, B). The average width was estimated in 2.18 m ± 0.80, depth in 17.0 cm ± 7.6, water current in 3.83 m/s ± 7.4, dissolved oxygen in 1.18 mg/l ± 0.99, and temperature in 25.1 °C ± 2.0 (Silva et al., 2019:611, tab. 2). Upstream the Dardanelos-Andorinhas waterfalls complex, the only additional species of Corydoradinae found is a putatively undescribed species of Hoplisoma, referred as Corydoras sp. 2 in Silva et al. (2019:617, fig. 4). In the rio Juruena basin, most specimens were captured in small and shallow isolated pools of intermittent streams, directly flowing into the rio Juruena (Fig. 13A), also occurring in larger/deeper streams, as the igarapé do Boi Preto (Fig. 13B). In these sites (rio Juruena basin), the streams present dense marginal vegetation, muddy or dark tea-colored water, and substrate composed by sand and/or clay, leaf litter, submerged logs and branches, and large rocks (Fig. 13). In the rio Juruena basin, no additional Corydoradinae species was found in syntopy.
FIGURE 13| Collecting sites of Osteogaster oharai, showing (A) a small/shallow pool of an intermittent stream, and (B) a larger/deeper stream, the igarapé do Boi Preto, where the specimens UFRO-ICT 22449 were captured, both tributaries of the rio Juruena basin, Mato Grosso, Brazil.
Etymology. Osteogaster oharai is named in honor of Dr. Willian Massaharu Ohara, dear friend and distinctive ichthyologist, not only for his extensive contributions to knowledge of South American freshwater fishes, but for having played a fundamental role in the professional history of LFCT, having generously donated dozens of species of Corydoradinae collected by him over the last decade, some of which were described in collaboration. A noun in a genitive case.
Conservation status. Currently, the new species is known from its type-locality, the rio Aripuanã basin, with an additional record in the rio Juruena basin, both Mato Grosso State, Brazil. Additionally, the species seems to occur in high abundance considering the available material, especially in the rio Juruena basin. It is also important to emphasize that the species is relatively frequent in the aquarium trade. Therefore, considering the currently available data and according to the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN Standards and Petitions Subcommittee, 2024), Osteogaster oharai can be classified as Least Concern (LC).
Remarks. As described for the new species (see “Coloration in alcohol” section), some congeners, especially O. zygata, and other species of Corydoradinae, such as Brochis arcuatus (Elwin, 1938), B. bethanae (Bentley, Grant & Tencatt, 2021), Corydoras narcissus Nijssen & Isbrücker, 1980, Hoplisoma granti (Tencatt, Lima & Britto, 2019), and H. urucu (Britto, Wosiacki & Montag, 2009), commonly present a small, vertical “pigmentation gap” (variably more or less intense depending on specimen) just ventral to dorsal-fin spine base, which can be extended further ventrally towards first ventrolateral body plate as a bright whitish vertically elongated patch, the latter typically more evident in living specimens (Figs. 1, 10, 11B–F, 14; see Bentley et al., 2021). In some specimens, only the pigmentation gap or the bright whitish vertically elongated patch are present, while in others both are simultaneously present. Interestingly, such feature seems to be typically present (or at least more evident) in the Corydoradinae displaying dorsolateral stripe. This pigmentation gap and the vertically elongated whitish patch are placed just above the transverse process of the second pterygiophore (likely formed by the fusion of pterygiophores 2 and 3 according to Reis (1998)) and the strong ligament connecting this process to the first rib, respectively. Interestingly, there is a precise alignment between these structures and such distinct coloration (i.e., reduced dark brown or black pigmentation and bright whitish vertically elongated patch) (Bentley et al., 2021), but a possible relationship between them requires further investigation.
Identification key to species of Osteogaster
1a. Posterior process of the parieto-supraoccipital not in contact with the nuchal plate……………….. 2 (variable in O. oharai)
1b. Posterior process of the parieto-supraoccipital in contact with thenuchal plate……………….. 4 (variable in O. oharai)
2a. Dark patch on flanks restricted to anterodorsal portion of flank (clearly overing less than 50% of its surface), somewhat rounded to roughly diamond shaped……………….. O. maclurei
2b. Dark patch on flanks extended towards caudal-fin base, oblong, covering at least about 50% of its surface……………….. 3
3a. Flank almost entirely dark brown or black……………….. O. hephaestus
3b. Flank with clear background and a dark brown or black stripe……………….. 5
4a. Longitudinal dark stripe almost entirely covering dorsal and middle portion of flank, running in parallel to longitudinal axis of the body or slightly inclined upwards anteriorly, with anterior portion slightly wider, smoothly becoming narrower posteriorly……………….. O. melanotaenia
4b. Oblique dorsolateral longitudinal dark stripe on flanks, starting at first dorsolateral body plate and gradually descending towards middle portion of caudal-fin base, variably extending diffuse on ventral lobe of caudal fin……………….. O. oharai
5a. Posterolateral portion of scapulocoracoid moderately-developed, not expanded mesially……………….. 6
5b. Posterolateral portion of scapulocoracoid strongly well-developed, conspicuously expanded mesially, contacting its counterpart and almost entirely covering ventral surface of trunk……………….. 7
6a. Flank with large, conspicuous roughly rounded to longitudinally elliptical dark brown or black blotch on its anterior portion, variably extending towards caudal-fin base, forming a somewhat mace-shaped longitudinal patch running parallel to longitudinal axis of the body or slightly inclined upwards anteriorly, with wider portion on anterior half of trunk, gradually becoming narrower posteriorly……………….. O. aenea
6b. Flank always with oblique dorsolateral longitudinal dark stripe (see step 4b)……………….. O. oharai
7a. Dorsolateral stripe indistinct from the dark patch almost entirely covering flanks, only visible on predorsal region of body……………….. O. eques
7b. Dorsolateral dark stripe as the only dark patch on flanks, conspicuous……………….. 8
8a. Snout typically rounded; ventral and anterior laminar expansions of infraorbital 1 strongly well developed, almost entirely covering snout’s lateral surface……………….. O. rabauti
8b. Snout typically pointed; ventral and anterior laminar expansions of infraorbital 1 strongly well developed, but covering up to about 50% of snout’s lateral surface……………….. O. zygata
Discussion
The rio Aripuanã basin is known by its relatively high degree of endemism, with about 15 species considered as endemic, 11 of which are restricted to its upper stretch, upstream of the Dardanelos-Andorinhas waterfalls complex (see Kullander, 1995; Benine, 2002; Deprá et al., 2014; Ferreira et al., 2024). In the rio Juruena basin, the number of endemic species is even higher, totaling 24 species (Dagosta, de Pinna, 2019). Even though each river system presents a considerably high degree of endemism, species or group of species shared between the rivers Aripuanã, especially its upper portion, and Juruena basins is well documented (see Varella et al., 2012; Dagosta, de Pinna, 2019; Casatti et al., 2020). In a broader analysis, the interfluvial region between the Madeira and Tapajós rivers can be considered an important area of endemism in the Amazon basin (see Tencatt, Ohara, 2016a; Dagosta, de Pinna, 2019). Considering that the new species was only recorded upstream the Dardanelos-Andorinhas waterfalls complex, the description of O. oharai reinforces the close relation between the upper rio Aripuanã and rio Juruena basins.
Due to its peculiar color pattern, Osteogaster is surely one of the most well-defined groups within Corydoradinae (Dias et al., 2024). The species of this genus can be promptly distinguished from the other Corydoradinae by typically having ground color of body in shades of orange in living specimens, with only a single, large dark patch on flanks, with all fins unblotched (vs. ground color of body in shades of yellow, typically with small dark spots at least in some part of the body and/or more than one large, dark patch on body in most species of remaining genera). Although the diagnosis provided by Dias et al. (2024) remains mostly accurate, the description of O. oharai resulted in small modifications in it. The authors have used, among other features, the typical ground color of body in shades of orange plus the presence of a single dark patch on flanks and unblotched fins to distinguish the representatives.
Even though this color pattern is found in all Osteogaster species, including O. oharai, some specimens alternatively display a bright greyish yellow coloration in life (Fig. 11E). This small modification in the variation range brings an important implication, as a color pattern composed by background color of body in shades of yellow combined with a single dark area on flanks and unblotched fins is similar to that observed in some females or juveniles of some Brochis (e.g., B. pantanalensis) and Gastrodermus (e.g., G. latus), or even some adult Brochis, like B. splendens. Albeit similar, we still consider them as distinct, non-homologous, color patterns, since in these groups there is no formation of a large, well-defined dark patch on flanks, conspicuously standing out from the background color of body, as in Osteogaster, but rather a dusky area on flanks. In any case, although the color pattern of Osteogaster is quite conservative, and potentially synapomorphic, we suggest that the recognition of this and other genera within Corydoradinae be performed through the analysis of combined features for an unambiguous identification (see Dias et al., 2024).
Moreover, Tencatt et al. (2025) provided an extensive discussion on the mesethmoid anatomy in relation to snout morphology, and its implications to the systematics of the group, proposing standardizations regarding this bone and the snout shape. In the light of the findings of Tencatt et al. (2025), the use of the mesethmoid size and snout shape alone does not allow an unequivocal recognition of the genera of Corydoradinae due to the multiple cases of convergence within the subfamily. Following Tencatt et al. (2025), the size of mesethmoid differs only Osteogaster from Corydoras, presenting overlap with the remaining genera of Corydoradinae. Therefore, we provide an updated diagnosis for Osteogaster, as follows: (I) mesethmoid moderate in size (vs. long in Corydoras), (II) posterior margin of pectoral-fin spine with all or nearly all serrations directed towards the tip of the spine or perpendicularly directed (vs. mostly directed towards the origin of the spine in Brochis, Corydoras, Gastrodermus, except G. hastatus and G. pygmaeus, and Scleromystax), (III) posterior laminar expansion of infraorbital 2 contacting pterotic–extrascapular (vs. laminar expansion typically not in contact with pterotic-extrascapular in Aspidoras and Hoplisoma; not contacting in Scleromystax), (IV) anterodorsal edge of infraorbital 2 expanded anteriorly (vs. not expanded anteriorly in Aspidoras, Hoplisoma, and Scleromystax), and (V) ground color of body yellowish orange to reddish orange, with only a single, large dark patch on flanks; all fins devoid of dark spots (vs. ground color of body pale yellow, brownish yellow, or greyish yellow, typically with small dark spots at least in some part of the body and/or more than one large, dark patch on body in most species of remaining genera).
As previously mentioned, Osteogaster species presents a single, large to extremely large solid dark patch on flanks. Typically, this patch presents a rounded to slightly longitudinally elliptical or roughly diamond-like shape, as in O. maclurei and some specimens of Osteogaster aff. aenea, a conspicuously elongated oblong shape, as in some specimens of O. aenea and Osteogaster aff. aenea, and in O. melanotaenia. In other representatives, i.e., O. eques and O. hephaestus, the dark patch is also conspicuously elongated, but clearly wider, almost entirely covering flanks. Alternatively, some species present a well-defined dorsolateral longitudinal dark stripe on flanks, such as the new species (Figs. 1, 10, 11C–D), O. rabauti (Figs. 15A, C, E) and O. zygata (Figs. 15B, D, F), and, although not well-defined/distinct (especially in living specimens), O. eques (Figs. 15G–L). Except for O. oharai, the species sharing this dorsolateral dark stripe also shares two additional features, namely: (I) posterolateral portion of scapulocoracoid strongly well developed, conspicuously expanded mesially, contacting its counterpart and almost entirely covering ventral surface of trunk (Fig. 15E, F, K, L), and (II) dorsolateral stripe extending anteriorly on head, converging mesially and meeting its counterpart on parieto-supraoccipital, forming a V-shaped pattern in dorsal view, with anteriormost portion variably reaching the frontals (Figs. 15A, B, G, H). Interestingly, an exclusive and peculiar tricolored color pattern on early development is also shared by two of those species, O. rabauti and O. eques, with the fry presenting reddish/yellowish orange anterior third of body, black middle third of body and greyish white posterior third of body. Although apparently forming a distinct morphological group within Osteogaster, this was not corroborated by previous molecular analyses (Alexandrou et al., 2011; Dias et al., 2024).
FIGURE 14| Specimens of Corydoradinae displaying a “pigmentation gap” just ventral to dorsal-fin spine base and/or a bright whitish vertically elongated patch just posterior to cleithrum (indicated by red arrows). (A) Brochis arcuata, (B) B. bethanae, (C) Corydoras narcissus, and (D) Hoplisoma granti. Photos (A and D) by Hans Evers, (B and C) by Steven Grant.
FIGURE 15| General morphology and color pattern of other striped species of Osteogaster, showing (A, C and E, in dorsal, lateral and ventral views, respectively) O. rabauti (MNRJ 3626, 46.0 mm SL, the holotype of Corydoras myersi), (B, D and F, in dorsal, lateral and ventral views, respectively) O. zygata (CAS 24756, unmeasured, the holotype of Corydoras zygatus), (G, I and K, in dorsal, lateral and ventral views, respectively) O. eques (MCZ 8204, unmeasured, paralectotype of Corydoras eques), and (H, J and L, in dorsal, lateral and ventral views, respectively) an unpreserved aquarium specimen of O. eques photographed alive. Photos (A, C and E) by Victor de Brito, (B, D and F) ©California Academy of Sciences, (G, I and K) ©President and Fellows of Harvard College, and (H, J and L) by Willian Ohara.
Backgrounded by previous phylogenetic hypothesis (Reis, 1998; Dias et al., 2024), the different morphological patterns found in Osteogaster provide interesting insights on the systematics of Callichthyidae. As widely known, convergent color patterns are extremely common within Corydoradinae (Nijssen, Isbrücker, 1980b,c; Britto, 2003; Britto et al., 2009; Alexandrou et al., 2011; Tencatt et al., 2019, 2021, 2022a, 2023b, 2024a,b; Tencatt, Pavanelli, 2015; Tencatt, Ohara, 2016a,b; Lima, Sazima, 2017). However, the independent occurrence of other morphological traits is also relatively common in Callichthyidae, especially among the Corydoradinae. The most notorious example refers to the absence of contact between the posterior process of the parieto-supraoccipital and the nuchal plate, which is found independently in all genera within the family, occurring in all Callichhtyinae (Reis, 1998), Aspidoras (typical condition of the genus, but variable in A. belenos; Tencatt et al., 2022b) and Scleromystax (typical condition of the genus; Britto, 2003; Britto et al., 2016), and spread out in Brochis (B. difluviatilis; Britto, Castro, 2002), Gastrodermus (G. hastatus, G. pygmaeus and typically in G. undulatus; Bono et al., 2019; apparently, also in G. latus, LFCT, pers. obs.), Hoplisoma (H. gladysae, H. micracanthus and H. petracinii; Calviño, Alonso, 2009), and Osteogaster (O. maclurei, O. melanotaenia and variably in O. oharai;see Description section; Tencatt et al., 2023a).
Although less common, another feature that illustrates the convergence events in the group refers to the presence of posterolateral portion of scapulocoracoid ranging from well- to strongly well-developed, expanded mesially and variably contacting its counterpart. This feature can be observed in most Callichthyinae (except for Callichthys; with variable degrees of mesial expansion in remaining genera; Reis, 1998), the species of the Brochis splendens group sensu Tencatt et al. (2025) (i.e., B. britskii, B. multiradiata and B. splendens), and Osteogaster (O. eques, O. rabauti and O. zygata). Several other traits absent in Osteogaster also illustrates this, as the presence of posterior margin of pectoral-fin spine mostly with antrorse, well-developed conical serrations (shared by the Callichthyinae, Corydoras, Gastrodermus, and Scleromystax), and a small, roughly rounded parieto-supraoccipital fontanel (shared by Aspidoras, Gastrodermus and Scleromystax). Moreover, features related to mesethmoid and snout morphology are frequently convergent (Tencatt et al., 2025).
In this context, even though such features are often useful for recognizing taxa at species or generic (when combined with additional traits) level within Corydoradinae, they are surely not synapomorphic, but rather homoplastic at genus level. A similar situation is also observed in shared reductive features (see Weitzman, Fink, 1983; Weitzman, Fink, 1985; Schaefer et al., 1989). Moreover, the substantial advances on the taxonomy (e.g., Britto, Reis, 2005; Britto et al., 2005, 2007, 2009; Tencatt et al., 2013, 2014a,b, 2016, 2019, 2020, 2021, 2022a,b, 2023a,b, 2024a,b; Tencatt, Pavanelli, 2015; Tencatt, Britto, 2016; Tencatt, Evers, 2016, Tencatt, Ohara, 2016a,b; Espíndola et al., 2018; Bono et al., 2019) and interrelationships (e.g., Alexandrou et al., 2011; Marburger et al., 2018; Dias et al., 2024) of Corydoradinae since the work of Britto (2003) revealed that most of the synapomorphies presented therein are restricted to more terminal levels (i.e., species or group of species levels), occurring independently among its genera. Albeit the genera within Corydoradinae are relatively well-diagnosed by morphological features (see Dias et al., 2024), proper synapomorphies are apparently scarce at generic level.
Among the putative exclusive features within the Corydoradinae at genus level we suggest: (I) base of pectoral-fin branched rays with small laminar expansions on its inner margin, generally more evident on first rays and variably with irregular margins, forming pointed structures, for Aspidoras; (II) posterior margin of dorsal-fin spine with most serrations retrorse and with blade-like aspect for Brochis; (III) branch of the temporal sensory canal at sphenotic, which gives rise to the supraorbital canal, with two pores, (IV) region just above posterodorsal margin with nearly straight to slightly arched dorsally, horizontally elongated eyebrow-like marking, and (V) area at the corner of the mouth, ventral to the maxillary barbel, with a roughly triangular fleshy flap, which can be variably elongated, similar to a barbel, for Corydoras; (VI) orbit positioned on middle portion of lateral surface of head for Gastrodermus; (VII) unblotched/hyaline fins, flanks devoid of small, dark markings, only displaying a single, large and conspicuous dark patch for Osteogaster; (VIII) odontodes inserted in fleshy papillae on preopercular-opercular region of males for Scleromystax.
In the case of Hoplisoma, the genus can only be recognized by the combination of non-synapomorphic features, which are essentially the presence of (i) posterior margin of both dorsal- and pectoral-fin spines with all or nearly all serrations directed towards the tip of the spine or perpendicularly directed (shared with Osteogaster, G. hastatus and G. pygmaeus), (ii) ground color of body in shades of yellow, typically with small dark spots at least in some part of the body and/or more than one large, dark patch on body in most species of remaining genera (shared with G. hastatus and G. pygmaeus), and (iii) anterodorsal edge of infraorbital 2 not expanded anteriorly (shared with Aspidoras and Scleromystax). As discussed above, even though there were notable advances on the taxonomy and interrelationships (molecular-based hypotheses), the only available morphology-based phylogenetic hypothesis (Britto, 2003), published more than 20 years ago, has become outdated due to substantial advances accomplished in the last decade (e.g., Tencatt et al., 2013, 2014a,b, 2016, 2019, 2020, 2021, 2022a,b, 2023a,b, 2024a,b; Tencatt, Pavanelli, 2015; Britto et al., 2016; Tencatt, Britto, 2016; Tencatt, Evers, 2016, Tencatt, Ohara, 2016a,b; Espíndola et al., 2018; Bono et al., 2019), which is reflected in the remarkable difference between such morphological hypothesis and the recent molecular-based topologies (Marburger et al., 2018; Dias et al., 2024). We therefore highlight the need to scrutinize Britto’s (2003) primary-homology hypotheses from a revisionist perspective, even though with specific assessments, as carried out by Tencatt et al. (2025), so that the knowledge gathered over the last two decades of studies on Corydoradinae systematics led by MRB can be used to refine knowledge of the interrelationships within the subfamily.
Comparative material examined. Osteogaster aenea: USNM 1116, lectotype of Hoplosoma aeneum, 38.2 mm SL; USNM 92819, 1, 49.5 mm SL, paralectotype; USNM 205649, 3, 32.7–44.9 mm SL, paralectotypes. Osteogaster eques: MCZ 8204, 4 of 12, 37.6–44.4 mm SL, paratypes of Corydoras eques. Osteogaster hephaestus: MZUSP 119087, holotype of Corydoras hephaestus, 22.6 mm SL. Osteogaster maclurei: MUSM 70671, holotype of Corydoras maclurei, 37.0 mm SL. Osteogaster melanotaenia: BMNH 1909.7.23.41, lectotype of Corydoras melanotaenia, 38.3 mm SL; BMNH 1909.7.23.42, 1, 30.1 mm SL, paralectotype. Osteogaster rabauti: MNRJ 3626, holotype of Corydoras myersi, 46.0 mm SL; USNM 163252, 2, both 37.0 mm SL, paratypes of Corydoras myersi. Osteogaster zygata: MZUSP 30858, 4 of 15, 41.7–47.3 mm SL.
Acknowledgments
This work is dedicated to Rob McLure, dear friend and renowned aquarist, for the extensive support to LFCT throughout the years, especially on topics related to Aspidoras, Scleromystax and Osteogaster. The Universidade Federal de Mato Grosso (UFMT) provided logistical support. The authors are grateful to Carla Pavanelli (NUP), Carlos Lucena and Margarete Lucena (MCP), Cláudio Oliveira (LBP), Mário de Pinna, Aléssio Datovo, Michel Gianeti, Murilo Pastana and Osvaldo Oyakawa (MZUSP), Kris Murphy, Sandra Raredon and Jeffrey Clayton (USNM), Mark Sabaj and Mariangeles Arce (ANSP), James Maclaine and Oliver Crimmen (BMNH), and Otávio Froehlich (in memoriam) (ZUFMS) for hosting museum visits and loaning of material. To Emanuel Neuhaus (MNRJ), Francisco Severo-Neto and Thomaz Sinani (ZUFMS-PIS), Carlos Lucena and Héctor Vera-Alcaraz (MCP), Cláudio Oliveira, Ricardo Britzke, Fábio Roxo, Bruno Melo and Gabriel Silva (LBP), and Willian Ohara, Vinícius Espíndola and Túlio Teixeira (MZUSP) for generously welcoming LFCT during museum visits. To Fernando Vaz-de-Mello and Jorge Arias from the Laboratório de Scarabaeoidologia (UFMT) for allowing the use and general support of the photomontage equipment Leica M205C (subproject EECBio UFMT/Finep #01.12.0359.00), respectively. This study was partially supported by INCT – Peixes, funded by MCTIC/CNPq (proc. 405706/2022–7) through the sub-project “Peixes de Mato Grosso: preenchendo lacunas de coleta em riachos de cabeceira das bacias do alto Rio Paraguai e Rio Amazonas (incluindo sistema Tocantins-Araguaia)”. To Thiago Izzo (UFMT) and Izaias Fernandes (UNIR) for the donation of most specimens of O. oharai from the rio Tapajós basin analyzed herein. To Steven Grant for kindly reviewing the English language of this manuscript. To the members of the fish keeping groups Corydoras World (United Kingdom), Catfish Study Group (United Kingdom), Internationale Gemeinschaft Barben Salmler Schmerlen Welse e.V. (Germany), Potomac Valley Aquarium Society (United States of America), Ohio Cichlid Association (USA), Greater Pittsburgh Aquarium Society, Inc. (USA), and All Oddball Aquatics (USA) for the invaluable support to LFCT. The Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; proc. #88887.902472/2023–00) provided fellowship to VCG, and the Conselho Nacional de Desenvolvimento Científico e Tecnológico provided financial support to MRB (CNPq; proc. #311294/2021–9).
References
Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
Arratia G, Gayet M. Sensory canals and related bones of Tertiary siluriform crania from Bolivia and North America and comparison with recent forms. J Vertebr Paleontol. 1995; 15:482–505. https://doi.org/10.1080/02724634.1995.10011243
Benine RC. Moenkhausia levidorsa, a new species from rio Aripuanã, Amazon basin, Brazil (Characiformes: Characidae). Ichthyol Explor Freshw. 2002; 13:289–94.
Bentley RF, Grant S, Tencatt LFC. A new arc-striped species of Corydoras Lacépède, 1803 (Teleostei: Callichthyidae) from the Peruvian Amazon. Zootaxa. 2021; 4948(2):184–200. https://doi.org/10.11646/zootaxa.4948.2.2
Bono A, Tencatt LFC, Alonso F, Lehmann A P. Redescription of Corydoras undulatus Regan, 1912 (Siluriformes: Callichthyidae), with comments on the identity of Corydoras latus Pearson, 1924. PLoS ONE. 2019; 14(1):e0211352. https://doi.org/10.1371/journal.pone.0211352
Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153:119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO2
Britto MR, Castro RMC. New corydoradine catfish (Siluriformes: Callichthyidae) from the upper Paraná and São Francisco: the sister group of Brochis and most of Corydoras species. Copeia. 2002; 2002(4):1006–15. https://doi.org/10.1643/0045-8511(2002)002[1006:NCCSCF]2.0.CO;2
Britto MR, Fukakusa CK, Malabarba LR. New species of Scleromystax Günther, 1864 (Siluriformes: Callichthyidae) – extending the meridional distribution of genera endemic to the Atlantic Forest. Neotrop Ichthyol. 2016; 14(3):e150158. https://doi.org/10.1590/1982-0224-20150158
Britto MR, Lima FCT. Corydoras tukano, a new species of Corydoradine catfish from the rio Tiquié, upper rio Negro basin, Brazil (Ostariophysi: Siluriformes: Callichthyidae). Neotrop Ichthyol. 2003; 1(2):83–91. https://doi.org/10.1590/S1679-62252003000200002
Britto MR, Lima FC, Hidalgo MH. Corydoras ortegai, a new species of corydoradine catfish from the lower río Putumayo in Peru (Ostariophysi: Siluriformes: Callichthyidae). Neotrop Ichthyol. 2007; 5(3):293–300. https://doi.org/10.1590/S1679-62252007000300009
Calviño PA, Alonso F. Two new species of the genus Corydoras (Ostariophysi: Siluriformes: Callichthyidae) from northwestern Argentina, and redescription of C. micracanthus Regan, 1912. Rev Mus Argent Cienc Nat. 2009; 11(2):199–214. Available from: https://www.scielo.org.ar/pdf/rmacn/v11n2/v11n2a06.pdf
Casatti L, Brejão GL, Carvalho FR, Silva HP, Pérez-Mayorga MA, Manzotti AR et al. Stream fish from recently deforested basins in the Meridional Amazon, Mato Grosso, Brazil. Biota Neotrop. 2020; 20(1):e20190744. https://doi.org/10.1590/1676-0611-BN-2019-0744
Dagosta FCP, de Pinna MCC. The fishes of the amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 2019(431):1–163. https://doi.org/10.1206/0003-0090.431.1.1
Deprá GC, Kullander SO, Pavanelli CS, Graça WJ. A new colorful species of Geophagus (Teleostei: Cichlidae), endemic to the rio Aripuanã in the Amazon basin of Brazil. Neotrop Ichthyol. 2014; 12(4):737–46. https://doi.org/10.1590/1982-0224-20140038
Dias AC, Tencatt LFC, Roxo FF, Silva GC, Santos SA, Britto MR et al. Phylogenomic analyses in the complex Neotropical subfamily Corydoradinae (Siluriformes: Callichthyidae) with a new classification based on morphological and molecular data. Zool J Linn Soc. 2024; zlae053. https://doi.org/10.1093/zoolinnean/zlae053
Espíndola VC, Tencatt LFC, Pupo FM, Villa-Verde L, Britto MR. From the inside out: a new species of armoured catfish Corydoras with the description of poorlyexplored character sources (Teleostei, Siluriformes, Callichthyidae). J Fish Biol. 2018; 92(5):1463–86. https://doi.org/10.1111/jfb.13602
Ferreira KM, Ribeiro AC, Lima FCT, Silva HP, Ferreira DC, Mirande JM. A new species of Inpaichthys from the rio Canamã, rio Aripuanã basin, Mato Grosso State, Brazil, with a redefinition of the genus (Characidae: Stethaprioninae). Neotrop Ichthyol. 2024; 22(1):e230113. https://doi.org/10.1590/1982-0224-2023-0113
Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: species by family/subfamily [Internet]. San Francisco: California; 2025. Available from: https://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Huysentruyt F, Adriaens D. Descriptive osteology of Corydoras aeneus (Siluriformes: Callichthyidae). Cybium. 2005; 29:261–73. https://doi.org/10.26028/cybium/2005-293-004
International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee.Guidelines for using the IUCN Red List categories and criteria. Version 16 [Internet]. Gland; 2024. Available from: https://www.iucnredlist.org/resources/redlistguidelines
Kiehl E, Rieger C, Greven H. Axillary gland secretions contribute to the stress-induced discharge of a bactericidal substance in Corydoras sterbai (Callichthyidae, Siluriformes). Verh Ges Ichthyol. 2006; 5:111–15.
Kullander SO. Three new cichlid species from Southern Amazonia: Aequidens gerciliae, A. epae and A. michaeli. Ichthyol Explor Freshw. 1995; 6:149–70.
Lundberg JG. The evolutionary history of North American catfishes, family Ictaluridae. [PhD Thesis]. Michigan: The University of Michigan; 1970.
Lima FCT, Sazima I. Corydoras desana, a new plated catfish from the upper rio Negro, Brazil, with comments on mimicry within Corydoradinae (Ostariophysi: Siluriformes: Callichthyidae). Aqua. 2017; 23(1):19–31.
Marburger S, Alexandrou MA, Taggart JB, Creer S, Carvalho G, Oliveira C et al. Whole genome duplication and transposable element proliferation drive genome expansion in Corydoradinae catfishes. Proc R Soc B Biol Sci. 2018; 285(1872):20172732. https://doi.org/10.1098/rspb.2017.2732
Nijssen H, Isbrücker IJH. Aspidoras virgulatus n. sp., a plated catfish from Espírito Santo, Brazil (Pisces, Siluriformes, Callichthyidae). Bull Zool Mus Univ. 1980a; 7:133‒39.
Nijssen H, Isbrücker IJH. A review of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Bijdr Dierkd. 1980b; 50:190–220. https://doi.org/10.1080/01650528609360697
Nijssen H, Isbrücker IJH. Three new Corydoras species from French Guiana and Brazil (Pisces, Siluriformes, Callichthyidae). Neth J Zool. 1980c; 30(3):494–503. https://doi.org/10.1163/002829680X00113
Reis RE. Revision of the Neotropical catfish genus Hoplosternum (Ostariophysi: Siluriformes: Callichthyidae), with the description of two new genera and three new species. Ichthyol Explor Freshw. 1997; 7:299–326.
Reis RE. Anatomy and phylogenetic analysis of the neotropical callichthyid catfishes (Ostariophysi, Siluriformes). Zool J Linn Soc. 1998; 124:105–68. https://doi.org/10.1006/zjls.1997.012
Reis RE. Family Callichthyidae (Armored catfishes). In: Reis RE, Kullander SO, Ferraris Jr. CJ, editors. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.291–309.
Sabaj MH. Codes for Natural History Collections in Ichthyology and Herpetology. (online supplement). Version 9.7. 2025. Available from: https://asih.org
Schaefer SA. Homology and evolution of the opercular series in the loricarioid catfishes (Pisces: Siluroidei). J Zool. 1988; 214:81–93. https://doi.org/10.1111/j.1469-7998.1988.tb04988.x
Schaefer SA, Aquino AE. Postotic laterosensory canal and pterotic branch homology in catfishes. J Morphol. 2000; 246:212–27. https://doi.org/10.1002/1097-4687(200012)246:3<212::AID-JMOR5>3.0.CO;2-S
Schaefer SA, Weitzman SH, Britski HA. Review of the Neotropical catfish genus Scoloplax (Pisces: Loricarioidea: Scoloplacidae) with comments on reductive characters in phylogenetic analysis. Proc Acad Nat Sci Phila. 1989; 141:181–211.
Silva HP, Zawadzki CH, Lourenço LS, Fernandes IM. Stream fish in the Aripuanã river upstream and downstream of the Dardanelos-Andorinhas waterfall complex, state of Mato Grosso, Brazil. Oecol Austr. 2019; 23(3):606–19. https://doi.org/10.4257/oeco.2019.2303.17
Slobodian V, Pastana MNL. Description of a new Pimelodella (Siluriformes: Heptapteridae) species with a discussion on the upper pectoral girdle homology of Siluriformes. J Fish Biol. 2018; 93(5):901–16. https://doi.org/10.1111/jfb.13795
Spadella MA, Desan SP, Henriques TCBPO, Oliveira C. Variation in male reproductive system characters in Corydoradinae (Loricarioidei: Callichthyidae) reflects the occurrence of different lineages in this subfamily. Neotrop Ichthyol. 2017; 15(1):e160039. https://doi.org/10.1590/1982-0224-20160039
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19. https://doi.org/10.26028/cybium/1985-92-001
Tencatt LFC, Britto MR. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Araguaia basin, Brazil, with comments about Corydoras araguaiaensis Sands, 1990. Neotrop Ichthyol. 2016; 14(1):e150062. https://doi.org/10.1590/1982-0224-20150062
Tencatt LFC, Britto MR, Isbrücker IJH, Pavanelli CS. Taxonomy of the armored catfish genus Aspidoras (Siluriformes: Callichthyidae) revisited, with the description of a new species. Neotrop Ichthyol. 2022b; 20(3):e220040. https://doi.org/10.1590/1982-0224-2022-0040
Tencatt LF, Britto MR, Pavanelli CS. A new long-finned Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the lower rio Paraná basin, Brazil. Neotrop Ichthyol. 2014a; 12(1):71–79. https://doi.org/10.1590/S1679-62252014000100007
Tencatt LFC, Britto MR, Pavanelli CS. A new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the upper rio Paraná basin, Brazil. Neotrop Ichthyol. 2014b; 12(1):89–96. https://doi.org/10.1590/S1679-62252014000100009
Tencatt LFC, Britto MR, Pavanelli CS. Revisionary study of the armored catfish Corydoras paleatus (Jenyns, 1842) (Siluriformes: Callichthyidae) over 180 years after its discovery by Darwin, with description of a new species. Neotrop Ichthyol. 2016; 14(1):e150089. https://doi.org/10.1590/1982-0224-20150089
Tencatt LFC, Couto OLP, Santos AS, Sousa LM. A new long-snouted Corydoras (Siluriformes: Callichthyidae) from the rio Xingu and rio Tapajós basins, Brazilian Amazon. Neotrop Ichthyol. 2024a; 22(1):e230112. https://doi.org/10.1590/1982-0224-2023-0112
Tencatt LFC, Evers HG. A new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the río Madre de Dios basin, Peru. Neotrop Ichthyol. 2016; 14(1):13–26. https://doi.org/10.1590/1982-0224-20150019
Tencatt LFC, Gomes VC, Evers HG. A new species of Corydoras (Siluriformes: Callichthyidae) from the rio Madre de Dios basin, Peruvian Amazon, with comments on Corydoras aeneus identity. Neotrop Ichthyol. 2023a; 21(2):e230023. https://doi.org/10.1590/1982-0224-2023-0023
Tencatt LFC, Grant S, Bentley RF. A new color-changing species of Corydoras (Siluriformes: Callichthyidae) from the rio Jutaí, Brazilian Amazon. Neotrop Ichthyol. 2023b; 21(4):e230105. https://doi.org/10.1590/1982-0224-2023-0105
Tencatt LF, Lima FC, Britto MR. Deconstructing an octogenarian misconception reveals the true Corydoras arcuatus Elwin 1938 (Siluriformes: Callichthyidae) and a new Corydoras species from the Amazon basin. J Fish Biol. 2019; 95(2):453–71. https://doi.org/10.1111/jfb.13980
Tencatt LFC, Ohara WM. A new long-snouted species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madre de Dios basin. Zootaxa. 2016a; 4144(3):430–42. https://doi.org/10.11646/zootaxa.4144.3.9
Tencatt LFC, Ohara WM. Two new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madeira basin, Brazil. Neotrop Ichthyol. 2016b; 14(1):139–54. https://doi.org/10.1590/1982-0224-20150063
Tencatt LFC, Ohara WM, Carvalho V, Grant S, Britto MR. Two exquisite new species of Hoplisoma (Siluriformes: Callichthyidae) from the rio Tapajós basin, Brazil, with a discussion on the morphology of the mesethmoid within Corydoradinae. Neotrop Ichthyol. 2025; 23(1):e240100. https://doi.org/10.1590/1982-0224-2024-010
Tencatt LFC, Ohara WM, Sousa LM, Britto MR. Science and hobby joining forces for the discovery of three new Corydoras (Siluriformes: Callichthyidae) from the rio Tapajós basin, Brazil, with comments on Corydoras sp. CW111. Neotrop Ichthyol. 2022a; 20(3):e220070. https://doi.org/10.1590/1982-0224-2022-0070
Tencatt LFC, Pavanelli CS. Redescription of Corydoras guapore Knaack, 1961 (Siluriformes: Callichthyidae), a midwater Corydoradinae species from the rio Guaporé basin. Neotrop Ichthyol. 2015; 13(2):287–96. https://doi.org/10.1590/1982-0224-20150018
Tencatt LFC, Ruiz-Tafur M, Chuctaya J. A new masked Corydoras (Siluriformes: Callichthyidae) from the Itaya and Nanay River basins, Peruvian Amazon. Neotrop Ichthyol. 2024b; 22(2):e240016. https://doi.org/10.1590/1982-0224-2024-0016
Tencatt LFC, Santos SA, Britto MR. Taxonomic review of the typical long-snouted species of Corydoras (Siluriformes: Callichthyidae) from the río de La Plata basin. Neotrop Ichthyol. 2020; 18(4):e200088. https://doi.org/10.1590/1982-0224-2020-0088
Tencatt LFC, Santos SA, Evers HG, Britto MR. Corydoras fulleri (Siluriformes: Callichthyidae), a new catfish species from the rio Madeira basin, Peru. J Fish Biol. 2021; 99(2):614–28. https://doi.org/10.1111/jfb.14750
Tencatt LFC, Vera-Alcaraz HS, Britto MR, Pavanelli CS. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio São Francisco basin, Brazil. Neotrop Ichthyol. 2013; 11(2):257–64. https://doi.org/10.1590/S1679-62252013000200003
Varella HR, Kullander SO, Lima FCT. Crenicichla chicha, a new species of pike cichlid (Teleostei: Cichlidae) from the rio Papagaio, upper rio Tapajós basin, Mato Grosso, Brazil. Neotrop Ichthyol. 2012; 10(2):233–34. https://doi.org/10.1590/S1679-62252012000200002
Vera-Alcaraz HS. Relações filogenéticas das espécies da família Callichthyidae (Ostariophysi, Siluriformes). [PhD Thesis]. Porto Alegre: Pontifícia Universidade Católica do Rio Grande do Sul; 2013. Available from: https://tede2.pucrs.br/tede2/handle/tede/263
Weitzman SH, Fink SV. Xenurobryconin phylogeny and putative pheromone pumps in Glandulocaudinae fishes (Teleostei: Characidae). Smithson Contrib Zool. 1985; 421:1–119. https://doi.org/10.5479/si.00810282.421
Weitzman SH, Fink WL. Relationships of the neon tetras, a group of South American freshwater fishes (Teleostei: Characidae), with comments on the phylogeny of New World characiforms. Bull Mus comp Zool. 1983; 150(6):339–95. https://doi.org/10.5962/BHL.PART.28701
Authors
![]() Luiz Fernando Caserta Tencatt1
Luiz Fernando Caserta Tencatt1 ![]() ,
, ![]() Vandergleison de Carvalho2,
Vandergleison de Carvalho2, ![]() Hugmar Pains da Silva3 and
Hugmar Pains da Silva3 and ![]() Marcelo Ribeiro de Britto2
Marcelo Ribeiro de Britto2
[1] Universidade Federal de Mato Grosso, Instituto de Biociências, Departamento de Biologia e Zoologia, Laboratório de Sistemática de Loricarioidea, Avenida Fernando Corrêa da Costa, 2367, Boa Esperança, 78060-900 Cuiabá, MT, Brazil. (LFCT) luiztencatt@hotmail.com (corresponding author).
[2] Universidade Federal do Rio de Janeiro, Museu Nacional, Departamento de Vertebrados, Setor de Ictiologia, Quinta da Boa Vista s/n, São Cristóvão, 20940-040 Rio de Janeiro, RJ, Brazil. (VC) dcarvalhojet@hotmail.com, (MRB) mrbritto@mn.ufrj.br.
[3] Universidade do Estado de Mato Grosso, Campus Universitário Professor Eugênio Carlos Stieler de Tangará da Serra, Faculdade de Ciências Agrárias, Biológicas, Engenharia e Saúde, Avenida Inácio Bittencourt Cardoso, 6967 E, Jardim Aeroporto,78301-532 Tangará da Serra, MT, Brazil. (HPS) painsbio@gmail.com.
Authors’ Contribution 

Luiz Fernando Caserta Tencatt: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Vandergleison de Carvalho: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-original draft, Writing-review and editing.
Hugmar Pains da Silva: Conceptualization, Funding acquisition, Methodology, Resources, Validation, Writing-review and editing.
Marcelo Ribeiro de Britto: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
How to cite this article
Tencatt LFC, Carvalho V, Silva HP, Britto MR. A new striped Osteogaster (Siluriformes: Callichthyidae) from the Madeira and Tapajós river basins, Brazil, with a new generic diagnosis and an identification key to the species of Osteogaster. Neotrop Ichthyol. 2025; 23(2):e240134. https://doi.org/10.1590/1982-0224-2024-0134
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted March 31, 2025
Accepted March 31, 2025
![]() Submitted December 16, 2024
Submitted December 16, 2024
![]() Epub July 11, 2025
Epub July 11, 2025