![]() Renata Rúbia Ota1
Renata Rúbia Ota1 ![]() ,
, ![]() Gabriel de Carvalho Deprá1,
Gabriel de Carvalho Deprá1, ![]() Sven Kullander2,
Sven Kullander2, ![]() Weferson Júnio da Graça1,3,4 and
Weferson Júnio da Graça1,3,4 and ![]() Carla Simone Pavanelli 1,3
Carla Simone Pavanelli 1,3
PDF: EN XML: EN | Supplementary: S1 | Cite this article
Abstract
A new species of Satanoperca is described from the Rio Araguaia, Rio Tocantins basin, Brazil, and non-native records are available in the upper Rio Paraná basin. It differs from congeneric species by color pattern characters, such as head and flank marks. It is included in the Satanoperca jurupari species group, characterized by the absence of black rounded blotches on the flank, and low meristic values. A description of the ontogeny of melanophore marks of the S. jurupari species group revealed two different types of arrangement on the flank and numerous melanophore marks on the head. A discussion on morphologically diverse assemblages in the S. jurupari species group is also provided.
Keywords: Freshwater, Neotropical region, Non-native species, Pigmentation, Taxonomy.
Uma nova espécie de Satanoperca é descrita do rio Araguaia, bacia do rio Tocantins, Brasil, e registros não-nativos estão disponíveis para a bacia do alto rio Paraná. Pode ser diagnosticada de suas congêneres por caracteres do padrão de colorido como marcas na cabeça e flanco. É incluída no grupo Satanoperca jurupari pela ausência de máculas pretas arredondadas no flanco e menores valores de contagens. Uma descrição da ontogenia das marcas melanofóricas do grupo S. jurupari revelou dois tipos diferentes de arranjos no flanco e várias marcas melanofóricas na cabeça. Uma discussão sobre assembleias morfologicamente diversas no grupo S. jurupari também é fornecida.
Palavras-chave: Água doce, Espécies não-nativas, Pigmentação, Região Neotropical, Taxonomia.
Introduction
Satanoperca Günther, 1862 is a widely distributed genus in cis-Andean South America, comprising nine species distributed in two species groups sensu Willis et al. (2012). The S. daemon species-group includes S. acuticeps (Heckel, 1840), S. daemon (Heckel, 1840) and S. lilith Kullander & Ferreira, 1988 (considered as separated S. acuticeps and S. daemon species-groups by Kullander, Ferreira, 1988), and is characterized by the presence of black rounded blotches on the flank, and high meristic values. On the other hand, the S. jurupari species-group includes S. curupira Ota, Kullander, Deprá, da Graça & Pavanelli, 2018, S. jurupari (Heckel, 1840), S. leucosticta (Müller & Troschel, 1849), S. mapiritensis (Fernandéz-Yépez, 1950), S. pappaterra (Heckel, 1840), and S. rhynchitis Kullander, 2012 and is characterized by the absence of black rounded blotches on the flank, and low meristic values. Molecular phylogenetic analyses supported the monophyly of Satanoperca (e.g., López-Fernández et al., 2005, 2010; Ilves et al., 2017) and contained species groups (e.g., Willis et al., 2012).
Morphologically, among South American cichlids, Satanoperca is distinguished by a unique body shape with long snout and small mouth, and a unique combination of characters: absence of scales on dorsal- and anal-fin inter-radial membranes, ribs present on caudal vertebrae, post-abdominal extensions of swim bladder, presence of tooth plates on the ceratobranchial 4; scales on anterior half of cheek, a black blotch on the base of caudal-fin upper lobe, one or two rows of dentary teeth, three infraorbitals, equal number of abdominal and caudal vertebrae, or just one abdominal more, gill rakers attached to skin covering gill filaments, and supracleithrum frequently serrated (Kullander, 1986).
Studies on the Satanoperca jurupari species group revealed diverse assemblages, indicating the existence of new species (Kullander, Ferreira, 1988; Kullander, Nijssen, 1989; Kullander, 2012; Willis et al., 2012; Ota, 2013; Ota et al., 2018a). Despite meristic and morphometric data being conservative, the color pattern has been useful to diagnose species, especially head and flank marks (Ota et al., 2018a). Here we describe a new species in the S. jurupari species group, with meristic and morphometric data similar to other species in that group, but with a distinct color pattern. We also introduce a detailed analysis of juvenile color pattern and ontogenetic shifts in color pattern as a method of species discrimination in Satanoperca.
Material and methods
Measurements and counts follow Kullander (1980; 1983; 1986; 1990), and Kullander, Nijssen (1989), modified by Deprá et al. (2014). An explanation of the measurements taken herein is provided in S1. Regarding meristic data (which were taken under a stereomicroscope), the numbering of the caudal-fin rays (i.e., D1, D2, V1, V2 etc.; D standing for dorsal and V standing for ventral) follows Kullander (1986), while the numbering of scale rows (i.e., E1, E2, H1, H2; E standing for epaxial and H standing for hypaxial) follows Kullander (1990). The series of scales between the dorsal-fin base and the upper lateral line were counted from two different points: from the scale reaching the base of the first spine, and from the scale reaching the base of the last soft ray. If the only scale in one of those series to reach the respective spine or ray is a very small one (half the size of the others, or less), it is counted as a half scale. The number of scales on the posterior branch of the lateral line is counted as the number of scales until the distal extremity of hypurals (larger scales). The posterior end of the E1 scale row is understood to lie at the distal end of the hypurals. However, because part of the posteriormost E1 scale occasionally surpasses the border of the hypurals, making it difficult to determine whether it is in fact an E1 scale or a caudal-fin scale, we suggest a different criterion for recognizing that scale, which is not contradictory with the one just mentioned, but easier. The posteriormost scale in the E1 series is usually the one dorsal (and slightly posterior) to the posteriormost lateral-line scale on the hypural region. Scales beyond that one are smaller and cannot be unambiguously assigned to any scale series.
Counts of holotype are indicated by an asterisk. Each count is followed by its frequency in parentheses (when different from the total number of specimens). Osteological notes were based on specimens cleared and stained (c&s) using the protocol of Taylor, Van Dyke (1985). Vertebrae counts follow Deprá et al. (2021a), with modifications. Vertebrae classified as type A/B are transitional between types A and B, i.e., the bony ring formed between the contralateral parapophyses is incomplete. Those classified as type B/D are similar to type B, but with a complete haemal arch and spine in addition to the parapophyses. The lower pharyngeal tooth plate was analyzed as described by Barel et al. (1976).
The basic coloration elements in the Satanoperca jurupari species group are, except in a few details, the same as described by Říčan et al. (2005) for several species of Neotropical cichlid species. Important acronyms are the following: dorsal melanophore patches (DMPs), along the dorsal midline of the flank; and lateral melanophore patches (LMPs; derived from the septal and abdominal melanophore lines), along the lateral midline of the flank.
Satanoperca jurupari was analyzed from two points of view: S. jurupari species group (sensu Kullander, Ferreira, 1988; Ota et al., 2018a), including different valid species (S. curupira, S. jurupari, S. leucosticta, S. mapiritensis, S. pappaterra, and S. rhynchitis); and S. jurupari sensu lato, including different populations of possible undescribed species (see Discussion). Specimens were deposited in the following institutions: Instituto Nacional de Pesquisas da Amazônia, Manaus (INPA); Museu de Ciências e Tecnologia, Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre (MCP); Museu Paraense “Emilio Goeldi”, Belém (MPEG); Museu de Zoologia da Universidade de São Paulo, São Paulo (MZUSP), and Coleção Ictiológica do Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura, Universidade Estadual de Maringá, Maringá (NUP).
The non-type material list includes specimens without full data, damaged, or non-native (see Discussion). INPA TOC refers to an original information provided in the label and means “Tocantins”. Only works with systematic approach or listing the occurrence of the species were considered in the list of references. Localities from which coordinates were not originally georeferenced were determined using Google Earth and are approximate. The new species is part of the project: “Sistemática, taxonomia e biogeografia de ciclídeos neotropicais” (# 305200/2018–6 CNPq and # 4937/2020 UEM) registered in SisGen n° A954837.
Results
Satanoperca setepele, new species
urn:lsid:zoobank.org:act:128E9A30-4451-4AA5-B960-B92192C6EF4A
(Figs. 1–8; Tab. 1)
Geophagus jurupari. —Cetesb, 1977:117 [Brazil, Itaipu area; listed]. —CTSA, 1981:44 [Brazil, Itaipu Binacional; key to species; brief characterization; photograph]. —Santos et al., 1984:48 [Brazil, lower Rio Tocantins; photograph of fresh specimen; brief description].
Geophagus brasiliensis. —Godoy, 1986:16 [Brazil, UHE Ilha Grande; in part; lower photograph].
Satanoperca pappaterra. —Agostinho et al., 1997:187 [Brazil, upper Rio Paraná floodplain; listed; considered an introduced species]. —Duke Energy, 2003:100 [Brazil, Rio Paranapanema; brief description]. —Kullander, 2003:642 [in part; specimens from Rio Paraná basin; distribution]. —Agostinho et al., 2004:229 [Brazil, upper Rio Paraná floodplain; listed; photograph]. —Pavanelli et al., 2007:62 [Brazil, Corumbá Reservoir, upper Rio Paraná; listed]. —Graça, Pavanelli, 2007:214 [Brazil, upper Rio Paraná floodplain; photograph of fresh specimen; brief description]. —Langeani et al., 2007:190 [Brazil, upper Rio Paraná basin; listed]. —Oyakawa, Menezes, 2011:29 [Brazil, State of São Paulo; listed in the upper Rio Paraná]. —Froehlich et al., 2017:10 [Brazil, in part; specimens from upper Rio Paraná basin; listed]. —Jarduli et al., 2020:12 [Brazil, Rio Paranapanema basin; listed].
Satanoperca jurupari (sensu lato). —Stawikowski, Werner, 2004:408 [Brazil, in part; specimens from Rio Tocantins basin; photograph of fresh specimen].
Satanoperca jurupari. —Lucinda et al., 2007:82 [Brazil, Lajeado Reservoir; listed]. —Soares et al., 2009:27 [Brazil, Peixe Angical, Rio Tocantins; listed]. —Bartolette et al., 2017:12 [Brazil, Lajeado Reservoir; listed]. —Guimarães et al., 2020a:45 [Brazil, in part; specimens from Rio Tocantins basin; listed]. —Guimarães et al., 2021:49 [Brazil, in part; specimens from Rio Tocantins basin; listed].
Satanoperca sp. aff. jurupari. —Bartolette et al., 2012:66 [Brazil, UHE Serra da Mesa; listed].
Satanoperca sp. “Araguaia-Paraná”. —Willis et al., 2012:803 [Brazil, species-level phylogenetic analysis].
Satanoperca sp. —Ota et al., 2018b:e170094[9] [Brazil, upper Rio Paraná floodplain; new identification; photograph of fresh specimen; brief description]. —Azevedo-Santos et al., 2018 [Brazil, Ribeirão Frutal; photograph of fresh specimen; listed]. —Reia et al., 2020:715 [Brazil, Ribeirão Sucuri; photograph of fresh specimen; brief description]. —Reis et al., 2020:473 (Tab. 1), 478 [Brazil, Paraná State; listed to the upper Rio Paraná basin].
Holotype. NUP 13671, 151.0 mm SL, Brazil, state of Goiás, municipality of Nova Crixás, lago Japonês, tributary to the Rio Araguaia, 13°25’54”S 50°37’54”W, 2 Nov 2011, Nupélia staff.
Paratypes. All from Brazil, Rio Araguaia. INPA 59667, 4, 72.1–122.0 mm SL, state of Goiás, municipality of Nova Crixás, unnamed lagoon, tributary to Rio Araguaia, 13°21’53”S 50°37’46”W, 2 Nov 2011, Nupélia staff. MCP 54759, 5, 75.3–125.3 mm SL, state of Goiás, municipality of Nova Crixás, Rio Crixás-açu, tributary to Rio Araguaia, 13°21’30”S 50°36’38”W, 2 Nov 2011. MZUSP 125895, 3, 97.1–118.2 mm SL, state of Goiás, municipality of São Miguel do Araguaia, Lago Luiz Alves 2, tributary to Rio Araguaia, 13°13’22”S 50°34’40”W, 5 Nov 2011, Nupélia staff. NUP 13003, 7, 69.7–107.5 mm SL, state of Mato Grosso, municipality of Cocalinho, Lago Montaria, tributary to Rio Araguaia, 13°24’11”S 50°43’10”W, 3 Nov 2011, Nupélia staff. NUP 13585, 1 c&s, 104.6 mm SL, same data as NUP 13003. NUP 23219, 7 larvae, same data as NUP 13003. NUP 23124, 2, 94.8–144.8 mm SL, same data of holotype.
FIGURE 1 | Satanoperca setepele, NUP 13671, 151.0 mm SL, holotype, Brazil, state of Goiás, municipality of Nova Crixás, lago Japonês, tributary to Rio Araguaia, 13°25’54”S 50°37’54”W, 2 Nov 2011, Nupélia staff.
Non-types. All from Brazil, state of Mato Grosso, Rio Araguaia basin: MZUSP 103210, 5, 138.9–146.9 mm SL, municipality of Cocalinho, Ribeirão Água Preta, tributary to Rio Cristalino, 14°08’57”S 51°32’21”W; NUP 12755, 7 (3, 86.0–117.4 mm SL), municipality of Cocalinho, Lago Goiaba, tributary to Rio Araguaia, 12°50’54”S 52°32’02”W, 5 Dec 2011, Nupélia staff; NUP 12994, 3 (2, 91.4–112.6 mm SL), Lago Montaria, tributary to Rio Araguaia, municipality of Cocalinho, 13°24’11”S 50°43’10”W, 3 Nov 2011, Nupélia staff; NUP 13017, 1, 81.8 mm SL, municipality of Cocalinho, Lago Varal, tributary to Rio Araguaia, 13°00’52”S 50°36’08”W, 3 Nov 2011, Nupélia staff; NUP 13046, 5, 89.7–116.7 mm SL, municipality of Cocalinho, Lago Piranha, tributary to Rio Araguaia, 13°02’57”S 50°37’32”W, 3 Nov 2011, Nupélia staff; NUP 13058, 2, 81.9–120.5 mm SL, municipality of Cocalinho, Lago Comprido 1, tributary to Rio Araguaia, 12°52’06”S 50°35’44”W, 5 Nov 2011, Nupélia staff; NUP 13060, 19, 72.3–95.5 mm SL, municipality of Cocalinho, Lago Comprido 2, tributary to Rio Araguaia, 12°51’5”S 50°34’30”W, 5 Nov 2011, Nupélia staff. State of Goiás, Rio Araguaia basin: NUP 13049, 6, 69.4–88.4 mm SL, municipality of São Miguel do Araguaia, Lago Brito, tributary to Rio Araguaia, 13°10’40”S 50°34’46”W, 14 Nov 2011, Nupélia staff. State of Goiás, Rio Tocantins basin: INPA 14599, 2, 146.7–166.1 mm SL, Municipality of Niquelândia, Reservatório Serra da Mesa, tributary to Rio Tocantins, 14°7’38.4”S 48°17’10.7”W, 1 Oct 1997, E. P. Caramaschi. State of Pará, Rio Tocantins basin: INPA TOC 1105, 2, 120.0–162.2 mm SL, Municipality of Itupiranga, Lago Moroiteiro, tributary to Rio Tocantins, 05°12’42”S 49°22’27”W, 4 Jul 1982; MPEG 17101, 1, 139.5 mm SL, Municipality of Marabá, Rio Itacaiúnas, 05°24’48.1”S 49°10’22.7”W, 27 Jul 2009, F. R. Silva; MPEG 20490, 1, 98.4 mm SL, Municipality of Marabá, Rio Castanha, 05°45’24.3”S 49°41’18.4”W, 21 Jun 2006, A. Almeira; MZUSP 105587, 2 (1, 130.6 mm SL), Municipality of Marabá, Rio Itacaiúnas, upstream of confluence with Rio Tapirapé, 05°41’18”S 50°17’51”W, 12 Aug 2008, I. Fichberg & Muriel-Cunha. State of Tocantins, Rio Araguaia basin: INPA 20063, 1, 109.0 mm SL, Municipality of Caseara, Lago das Ariranhas, tributary to Rio Araguaia, 09°17’47”S 49°56’37”W, 20 May 2000, INPA Ichthyology staff; INPA 21265, 2, 109.4–138.0 mm SL, Municipality of Caseara, praia do Coco, Parque Estadual do Cantão, 09°14’49”S 50°00’11”W, 17 Nov 2000, Projeto Cantão staff. State of Goiás, upper Rio Paraná basin: NUP 235, 4, 127.4–168.7 mm SL, municipality of Itumbiara, Reservatório de Itumbiara, 18°25’08”S 49°12’54”W, 15 Jan 2009, G. Santos; NUP 1134, 22 (7, 115.4–150.5 mm SL), municipality of Caldas Novas, Reservatório de Corumbá, tributary to Rio Paranaíba, 17°43’37”S 48°32’54”W, 9 Apr 1999, Nupélia staff; NUP 1336, 13, 88.4–136.7 mm SL, municipality of Caldas Novas, Rio Corumbá, 17°29’01”S 48°22’12”W, 1 Jun 1996, Nupélia staff; NUP 8829, 1, 137.6 mm SL, border between Alexânia and Santo Antônio do Descoberto, Reservatório de Corumbá IV, 16°16’S 48°20’W, 10 Jun 2006, R. L. Silva et al.; NUP 8830, 3, 136.2–141.9 mm SL, municipality of Luziânia, Reservatório de Corumbá IV, 16°19’20”S 48°10’57”W, 25 Mar 2009, R. L. Silva; NUP 8831, 2, 114.0–128.8 mm SL, municipality of Santo Antônio do Descoberto, Reservatório de Corumbá IV, 16°14’49”S 48°46’42”W, 18 Apr 2009, R. L. Silva; NUP 8895, 1, 170.6 mm SL, border between Caçu and São Simão, Rio Claro, tributary to Rio Paranaíba, 19°07’36”S 50°38’36”W, 16 Out 2008, R. L. Silva; NUP 8896, 2, 37.3–55.2 mm SL, border between Caçu and São Simão, Rio Claro, tributary to Rio Paranaíba, 19°06’17”S 50°39’22”W, 4 Jun 2009, R. L. Silva et al.; NUP 8897, 1, 127.2 mm SL, municipality of Caçu, Rio Claro, tributary to Rio Paranaíba, 19°02’31”S 50°40’08”W, 20 Jul 2009, R. L. Silva et al.; NUP 8898, 1, 149.0 mm SL, border between Caçu and São Simão, Rio Claro, tributary to Rio Paranaíba, 19°07’36”S 50°38’36”W; NUP 11399, 2, 105.3–120.3 mm SL, municipality of Porteirão, Rio dos Bois, tributary to Rio Paranaíba, 17°49’23”S 50°15’11”W, 24–25 Jan 2008, Nupélia staff. State of Mato Grosso do Sul, upper Rio Paraná basin: MZUSP 88409, 3, 61.5–107.1 mm SL, municipality of Três Lagoas, unnamed stream on MS-320 before córrego Taboca, 20°45’03”S 51°40’42”W; NUP 12618, 2, 178.9–199.0 mm SL, municipality of Taquarussu, lagoa do Gavião, tributary to Rio Paraná, 22°40’48”S 53°13’53”W, 18 Sep 2009, Nupélia staff; NUP 12619, 2, 130.4–138.4 mm SL, municipality of Taquarussu, Lagoa das Garças, tributary to Rio Paraná, 22°43’27”S 53°13’05”W, 20 Sep 2011, Nupélia staff; NUP 12780, 1, 143.8 mm SL, same data of NUP 12619; NUP 12815, 1, 150.2 mm SL, municipality of Taquarussu, Lagoa das Garças, tributary to Rio Paraná, 22°43’27”S 53°13’05”W, Nupélia staff; NUP 13285, 1, 190.3 mm SL, municipality of Taquarussu, Lagoa do Gavião, tributary to Rio Paraná, 22°40’48”S 53°13’53”W, 11 Jun 2011, Nupélia staff; NUP 13286, 1, 156.3 mm SL, municipality of Taquarussu, Canal Baía, 22°41’27”S 53°13’29”W, 12 Jun 2011, Nupélia staff; NUP 13287, 2, 91.9–92.1 mm SL, municipality of Taquarussu, Lagoa Fechada, tributary to Rio Paraná, 22°42’38”S 53°16’33”W, 13 Jul 2011, Nupélia staff; NUP 13291, 1, 96.8 mm SL, municipality of Taquarussu, Lagoa Peroba, tributary to Rio Paraná, 22°54’30”S 53°38’24”W, 16 Sep 2011, Nupélia staff; NUP 13292, 1, 107.6 mm SL, municipality of Taquarussu, Lagoa Pousada das Garças, tributary to Rio Paraná, 22°42’01”S 53°15’23”W, 17 Sep 2011, Nupélia staff; NUP 13424, 6 (5, 30.3–58.2 mm SL), municipality of Taquarussu, Lagoa fechada, tributary to Rio Paraná, 22°42’38”S 53°16’33”W, 27 Feb 2012, Nupélia staff. State of Minas Gerais, upper Rio Paraná basin: NUP 234, 2, 114.3–150.9 mm SL, municipality of Furnas, Reservatório de Furnas, tributary to Rio Turvo, 20°42’22”S 45°55’03”W, 15 Jan 1999, G. Santos. State of Paraná, upper Rio Paraná basin: MZUSP 21653, 1, 127.0 mm de SL, municipality of Guaíra, Rio Paraná, above Sete Quedas, 24°04’S 54°15’W, 1977 to 1980, Cetesb staff; NUP 422, 22, 55.8–90.4 mm SL, municipality of Porto Rico, lagoas (ilhas), Rio Paraná, 22°46’19”S 53°16’01”W, Mar 1991, S. Veríssimo et al.; NUP 1727, 5, 64.0–95.4 mm SL, Riacho Caracu, tributary to Rio Paraná, 22°45’S 53°16’W, 1990, Nupélia staff; NUP 1841, 3, 85.6–120.2 mm SL, municipality of Porto Rico, Rio Paraná, 1983, Nupélia staff; NUP 1842, 36, 46.0–94.2 mm SL, municipality of Guaíra, Reservatório de Itaipu, 24°03’S 54°15’W to 25°31’S 54°38’W, 1983, Nupélia staff; NUP 2122, 38, 50.0–101.6 mm SL, municipality of Porto Rico, Lagoa Genipapo (ilha Porto Rico), 22°45’S 53°16’W, 2 Jul 1998, Nupélia staff; NUP 2364, 41, 40.2–87.9 mm SL, municipality of Santa Helena, Reservatório de Itaipu, 24°51’S 54°21’W, 1986, Nupélia staff; NUP 2365, 14, 50.2–75.7 mm SL, municipality of Santa Helena, Reservatório de Itaipu, 25°00’S 54°15’W, 12 Feb 1988, Nupélia staff; NUP 3181, 4, 70.1–96.5 mm SL, municipality of Porto Rico, Riacho Caracu, tributary to Rio Paraná, 28 Apr 2004, students of Biogeography and Systematics PEA; NUP 3309, 11, 117.8–139.7 mm SL, Riacho Água Nanci, tributary to Rio Paraná, 22°47’S 53°17’W, 7 Apr 2004, Nupélia staff; NUP 4406, 3, 136.3–145.4 mm SL, municipality of Pato Bragado, Riacho Arroio Fundo (foz), tributary to Rio São Francisco Verdadeiro, 24°38’16”S 54°11’11”W, 1 Nov 2005, Nupélia staff; NUP 4644, 7, 139.1–144.7 mm SL, municipality of Foz do Iguaçu, Reservatório de Itaipu, 25°32’49”S 54°35’17”W, 13 Nov 2005, Nupélia staff; NUP 4661, 2, 120.0–128.2 mm SL, border between Pato Bragado and Entre Rios do Oeste, Rio São Francisco Verdadeiro (lentic), tributary to Rio Paraná, 24°38’16”S 54°11’11”W, Nov 2005, Nupélia staff; NUP 4901, 2, 136.0–145.2 mm SL, municipality of Foz do Iguaçu, Lago Principal, Canal da Piracema, 25°26’03”S 54°34’51”W, 3 Nov 2006, Nupélia staff; NUP 5665, 2, 136.77–140.7 mm SL, municipality of Foz do Iguaçu, Canal da Piracema, 25°26’03”S 54°34’51”W, 19 Feb 2008, Nupélia staff; NUP 6305, 3, 145.0–166.5 mm SL, municipality of Diamante do Norte, Reservatório Rosana (Rio Paranapanema), tributary to Rio Paraná, 22°36’01”S 52°52’20”W, Apr 2008, Nupélia staff; NUP 6343, 1, 92.4 mm SL, municipality of Porto Rico, Lagoa do Osmar, tributary to Rio Paraná, 22°46’27”S 53°19’56”W, 1 Feb 2005, Nupélia staff; NUP 6367, 2, 50.8–100.1 mm SL, municipality of Foz do Iguaçu, Canal da Piracema, 25°26’03”S 54°34’51”W, 30 Jul 2008, Nupélia staff; NUP 7333, 5, 49.8–84.8 mm SL, municipality of Foz do Iguaçu, Canal da Piracema, 25°26’03”S 54°34’51”W, 16 Nov 2005, Getech staff; NUP 7353, 8, 48.3–78.5 mm SL, municipality of Foz do Iguaçu, Canal da Piracema, 25°26’50”S 54°36’14”W, 31 Jul 2008, Getech staff; NUP 11127, 2, 23.1–38.4 mm SL, municipality of Porto Rico, Ressaco do Pau Véio (ilha Porto Rico), tributary to Rio Paraná, 22°44’50”S 53°15’11”W, 23 Nov 2010, Nupélia staff; NUP 11581, 2, 111.4–137.5 mm SL, municipality of Porto Rico, Rio Paraná, 22°41’S 53°16’W, Dec 2003, Nupélia staff; NUP 12349, 3, 101.9–130.8 mm SL, municipality of Porto Rico, Ressaco do Pau Véio (ilha Porto Rico), tributary to Rio Paraná, 22°44’50”S 53°15’11”W, 17 Sep 2009, Nupélia staff; NUP 12500, 1, 147.6 mm SL, municipality of Taquarussu, Lagoa do Finado Raimundo, tributary to Rio Paraná, 22°47’58”S 53°32’29”W, Nupélia staff; NUP 12620, 1, 121.8 mm SL, same data of NUP 12349; NUP 12882, 2, 133.6–142.9 mm SL, municipality of Porto Rico, Ressaco do Bilé (ilha Mutum), 22°45’14”S 53°17’09”W, 21 Sep 2011, Nupélia staff; NUP 13289, 1, 120.0 mm SL, municipality of Porto Rico, Rio Paraná, 22°45’39”S 53°15’07”W, 14 Jul 2011, Nupélia staff; NUP 13254, 1, c&s, 65.3 mm SL, municipality of Porto Rico, Riacho Caracu, 22°45’55”S 53°15’28”W, 28 apr 2004, C.S. Pavanelli; NUP 13404, 6, 125.6–136.3 mm SL, municipality of Porto Rico, Ressaco do Pau Véio (ilha Porto Rico), tributary to Rio Paraná, 22°44’50”S 53°15’11”W, Nupélia staff; NUP 13586, 1 c&s, 93.5 mm SL, same data as NUP 422; NUP 14390, 4, 85.4–159.1 mm SL, municipality of Pato Bragado, Rio São Francisco Verdadeiro (Ajuricaba), tributary to Rio Paraná, 24°55’37”S 54°03’39”W, 22 Aug 2005, Nupélia staff; NUP 14391, 16, 40.2–85.2 mm SL, municipality of Porto Rico, Rio Paraná, 22°45’40”S 53°15’07”W, Apr 2004, Nupélia staff. NUP 23225, 1, c&s, 82.0 mm SL, municipality of Porto Rico, Rio Paraná, Nupélia staff. State of São Paulo, upper Rio Paraná basin: MZUSP 79355, 1, 165.7 mm SL, municipality of Promissão, Rio dos Dourados, near to mouth, tributary to Rio Tietê, tributary to Reservatório de Promissão, 21°24’12”S 49°42’13”W, Jan 2000, W. S. Smith; MZUSP 83386, 5, 95.0–132.5 mm SL, municipality of Bariri, Rio Tietê, downstream UHE Bariri, 22°08’50”S 48°45’06”W, 3–7 Nov 2011, A. Akama.
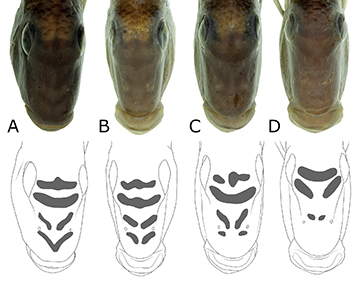
Diagnosis. Satanoperca setepele differs from S. acuticeps, S. daemon, and S. lilith by the absence of black rounded blotches on the flank (vs. presence: one in S. lilith; two in S. daemon; three in S. acuticeps). It is distinguished from S. leucosticta and S. mapiritensis by the absence of small light blotches on the head (vs. presence). It differs from S. curupira by the presence of two oblique stripes on the lachrymal, one at the superior and another at the inferior margin (vs. 3–7 dark-brown oblique stripes across the lachrymal), dark brown markings absent of cheek and opercular series (vs. irregular pattern of dark-brown stripes present on the cheek and opercular series), by the longitudinal band present and conspicuous in preserved specimens (vs. lateral band absent or inconspicuous in preserved specimens), and by presence of 15–19, mode 18, gill rakers on ceratobranchial 1 (vs. 14–16, mode 15). It differs from S. rhynchitis by the presence of 27–29 scales on E1 series (vs. 26). It differs from S. pappaterra by having the dorsal melanophore patches continuous with flank bars, as conspicuous as, and indistinguishable from them (vs. dorsal melanophore patches continuous with flank bars, but much more conspicuous, and clearly distinguishable from them), and longitudinal band as conspicuous as flank bars along its entire length (vs. longitudinal band much more conspicuous at meeting with flank bars and lateral melanophore patches). From S. jurupari, by the following combination of characters: head always without markings (spots, blotches or stripes) on cheek and opercular series, in both living and preserved specimens (vs. frequently light spots, in living specimens; or dark-brown stripes, in preserved specimens), longitudinal band and flank bars very conspicuous, in preserved specimens (vs. both less conspicuous), longitudinal band as conspicuous as flank bars, in preserved specimens (vs. longitudinal band less conspicuous than flank bars), frequently six flank bars, more visible in juvenile specimens (vs. frequently seven flank bars), and supra-cleithrum smooth, without serrations (vs. frequently with serrations) (Fig. 2). Additionally, S. setepele can be distinguished from the remaining species in the S. jurupari group, except S. pappaterra and S. rhynchitis, by the interorbital and nasal stripes not fragmented into spots and fading with growth (vs. fragmented into spots with growth) (Fig. 3), by the presence, in adults, of double scale series on the caudal-fin inter-radial membranes (vs. all caudal-fin inter-radial scale series single), and a secondary scale series on the caudal-fin inter-radial membranes (i.e., between the branches of a ray; vs. secondary series absent).
FIGURE 2 | Supracleithrum with well-developed serrations in. A. Satanoperca acuticeps, MZUSP 8215, 70.0 mm SL; and smooth, without serrations in B. S. setepele, NUP 23125, 97.1 mm SL.
FIGURE 3 | Ontogenetic development of the interorbital and nasal stripes in Satanoperca setepele. A. NUP 8306, 59.2 mm SL. B. MCP 54759, 76.5 mm SL. C. NUP 13003, 102.7 mm SL. D. NUP 13038, 123.5 mm SL.
Description. Morphometric data summarized in Tab. 1. Body laterally compressed; greatest depth at vertical through pelvic-fin origin. In lateral view, dorsal contour more arched than ventral. Dorsal profile of head ascending straight to slightly convex from tip of snout to nape, slightly elevated above orbit. Dorsal profile of trunk moderately convex from nape to end of dorsal fin; slightly concave along caudal peduncle. Ventral profile almost straight from tip of snout to pelvic-fin origin; straight or slightly convex between pelvic- and anal-fin origin; ascending straight or slightly convex along anal fin; slightly concave along caudal peduncle.
Snout long and pointed in lateral view, its tip below median axis of body. Mouth terminal, far below level of orbits; lips of “American type”, upper lip with continuous fold, lower lip interrupted symphysially. Tip of maxilla reaching about vertical between nostril and orbit. Nostril dorsolaterally situated, below horizontal through lower margin of orbit, halfway between tip of snout and orbit. Orbit large, dorsally situated on posterior half of head; pupil at same level of upper lateral line. Interorbital distance narrower than mouth. Posterior margin of precopercle and suprachleitrum smooth, without serrations.
E1 scales 27(37), 28*(171) or 29(20); scales between upper lateral line and dorsal-fin 3½–4 anteriorly, 1½–2½ posteriorly. Scales in transverse row 8+1+3½. Scales on lateral line 17/9(2), 17/10(1), 17/11(2), 18/8(2), 18/9(11), 18/10(19), 18/11(17), 19/8(4), 19/9(16), 19/10(64), 19/11(37), 19/12(3), 20/9(8), 20/10*(23), 20/11(11), 21/10(2), 21/11(1) or 21/12(1); two scales of lower lateral line continuing onto caudal fin. Cheek scales ctenoid posterodorsally, cycloid anteroventrally, arranged in 4(2), 5(164), 6*(63) or 7(2) rows. Opercle scales cycloid, with different sizes and stochastically arranged. Subopercle covered with 1(47) or 2*(168) rows of cycloid scales. Interopercle with up to three scales embedded in skin. Scales absent on preopercle. Circumpeduncular scale rows 16*(235), including lateral line scales. Predorsal region with cycloid scales, approximately size of flank scales. Flank scales ctenoid. Prepelvic scales cycloid, gradually decreasing in size towards gular region. Abdominal scales ctenoid, slightly smaller than flank scales. Pectoral, pelvic, dorsal, and anal fins naked. Caudal-fin base covered with stochastically distributed transition scales, intermediate in size between peduncular and inter-radial scales; caudal fin with inter-radial cycloid scales from base of rays to half of its length; series of scales with pores and canals between rays D3–D4 (1–6 scales) and V4–V5 (6–14 scales continuing from lower branch of lateral line).
Dorsal-fin rays XIV.9(1), XIV.10(1), XIV.11(2) XV.9(9), XV.10*(130), XV.11(11), XVI.9(21) or XVI.10(14); dorsal spines increasing in size up to 6th, first spine about one-fourth length of last. Dorsal fin with distal slightly rounded blotches in soft rays, in some specimens rays 3–5 forming filament reaching up to distal portion of caudal fin; lappets pointed, with posterior margin free, slightly surpassing tip of spines. Anal-fin rays III.6(3), III.7*(187) or III.8(7); middle rays longest, pointed, in some specimens forming filament reaching up to half of caudal-fin length. Caudal fin straight or slightly rounded, with 16 principal rays. Total pectoral-fin rays 13(56), 14(64), 15*(12) or 16(6). Pectoral fin rounded to pointed, third or fourth ray longest, reaching up to third anal-fin spine. Pelvic-fin rays I,5; first ray longest, with filamentous extension, reaching up to half of anal fin.
Gill rakers externally on first epibranchial lobe 9(2), 10(12), 11(62), 12*(115) or 13(35); 0–1 on angle; 15(10), 16(74), 17(91), 18*(41) or 19(10) on ceratobranchial 1.
Lower pharyngeal tooth plate elongated. Anterior teeth slender, with unique retrorse cusp. Medio posterior teeth more robust, with large posterior cusp antrorse; 8–9 teeth in median row, 11–12 teeth in posterior row, 27–29 in lateral margin (Fig. 4).
Teeth unicuspid and retrorse, of uniform size. Symphysis of both jaws lacking teeth. Hemiseries of upper/lower jaw teeth15–35/12–26; one or two inner rows anteriorly in lower jaw.
Vertebrae 15+13 (1 c&s; penultimate caudal one representing fusion of two vertebrae), 15+14 (3 c&s). All specimens with type-A’ third vertebra. Penultimate abdominal vertebra type A (1) or A/B (3). Last one type A (1) or B (3). First caudal vertebra type B/D (2) or D (2).
TABLE 1 | Morphometric data of Satanoperca setepele. Standard length in millimeters and proportional measurements as percentage of SL. N = total number of specimens examined; SD = Description of taking measurements is shown in Tab. S1.
| Holotype | Paratypes | Non–types – Araguaia–Tocantins | Non–types – upper rio
Paraná | |||||||||
| | N | Range | Mean | SD | N | Range | Mean | SD | N | Range | Mean | SD |
Standard length (mm) | 151.0 | 22 | 69.7–144.8 | 100.7 | – | 57 | 56.5–166.1 | 101.5 | – | 120 | 35.1–173.6 | 110.3 | – |
Head depth | 38.3 | 22 | 34.2–40.7 | 37.3 | 1.9 | 57 | 24.4–40.8 | 36.3 | 3.0 | 120 | 30.6–40.5 | 35.6 | 1.8 |
Body depth | 44.1 | 22 | 40.8–47.3 | 42.7 | 1.4 | 57 | 37.8–47.3 | 42.3 | 2.3 | 120 | 36.3–48.8 | 42.4 | 2.1 |
Caudal peduncle depth | 15.1 | 22 | 13.8–16.7 | 15.4 | 0.6 | 57 | 13.7–17.0 | 15.6 | 0.7 | 120 | 13.0–17.3 | 15.3 | 0.7 |
Head length | 38.6 | 22 | 36.4–41.6 | 39.0 | 1.1 | 57 | 34.2–40.6 | 37.4 | 1.6 | 120 | 30.1–40.2 | 37.4 | 1.3 |
Pectoral-fin length | 37.5 | 22 | 36.1–42.3 | 38.7 | 1.6 | 57 | 33.4–41.4 | 37.4 | 1.7 | 109 | 29.0–40.9 | 35.8 | 2.3 |
Pelvic-fin length | 37.9 | 22 | 33.6–46.7 | 39.9 | 3.9 | 56 | 30.2–52.0 | 39.5 | 4.3 | 107 | 27.2–55.8 | 41.3 | 6.0 |
Caudal peduncle length | 14.6 | 22 | 10.7–19.8 | 14.6 | 1.8 | 57 | 10.7–19.8 | 14.3 | 1.4 | 120 | 10.6–16.6 | 13.8 | 1.3 |
Last anal-fin spine length | 15.0 | 21 | 12.7–19.8 | 16.8 | 1.8 | 56 | 12.2–19.1 | 16.3 | 1.7 | 119 | 11.9–17.7 | 15.1 | 1.3 |
Last dorsal-fin spine length | 19.3 | 18 | 18.3–21.1 | 20.0 | 0.9 | 54 | 16.3–20.9 | 19.1 | 1.1 | 118 | 11.8–21.5 | 18.1 | 1.4 |
Predorsal distance | 45.6 | 22 | 41.9–47.3 | 45.1 | 1.6 | 57 | 38.9–47.2 | 44.6 | 1.5 | 120 | 18.1–46.9 | 43.2 | 2.7 |
Prepectoral distance | 39.9 | 22 | 38.4–44.3 | 40.3 | 1.6 | 57 | 34.4–43.9 | 39.1 | 1.6 | 120 | 35.8–44.2 | 38.7 | 1.4 |
Prepelvic distance | 43.8 | 22 | 41.0–46.3 | 43.4 | 1.3 | 57 | 33.2–46.3 | 42.6 | 1.9 | 120 | 40.0–48.7 | 43.2 | 1.4 |
Head width | 20.0 | 22 | 16.5–21.4 | 19.0 | 1.1 | 57 | 16.1–21.4 | 18.3 | 1.2 | 120 | 15.0–21.3 | 17.9 | 1.3 |
Cheek depth | 14.8 | 22 | 12.4–15.3 | 13.8 | 0.8 | 57 | 9.7–15.8 | 13.7 | 1.2 | 120 | 8.6–33.5 | 14.0 | 2.2 |
Lachrymal depth | 16.9 | 22 | 13.3–17.8 | 15.5 | 1.2 | 57 | 11.3–17.8 | 14.9 | 1.5 | 120 | 9.9–17.8 | 15.2 | 1.4 |
Postorbital head length | 10.2 | 22 | 7.7–11.4 | 9.5 | 0.9 | 57 | 7.7–11.4 | 9.7 | 0.8 | 120 | 6.7–12.4 | 9.3 | 1.1 |
Upper jaw length | 13.2 | 21 | 11.6–13.7 | 12.9 | 0.6 | 54 | 10.4–13.9 | 12.5 | 0.8 | 117 | 7.5–13.9 | 12.2 | 0.7 |
Lower jaw length | 7.4 | 22 | 7.0–9.4 | 8.2 | 0.8 | 54 | 5.3–9.4 | 7.6 | 1.0 | 117 | 6.2–10.6 | 8.2 | 1.0 |
Snout length | 21.0 | 22 | 16.9–23.4 | 20.0 | 1.8 | 57 | 14.6–23.8 | 18.7 | 2.1 | 120 | 13.8–21.7 | 19.0 | 1.4 |
Orbital diameter | 10.2 | 22 | 9.8–12.6 | 11.2 | 0.9 | 57 | 8.7–13.7 | 11.0 | 1.1 | 120 | 8.1–14.6 | 9.9 | 1.1 |
Interorbital distance | 12.2 | 22 | 10.0–12.3 | 11.3 | 0.6 | 57 | 8.6–12.3 | 10.5 | 1.0 | 120 | 8.4–12.1 | 10.3 | 0.7 |
FIGURE 4 | Lower pharyngeal tooth plate of Satanoperca setepele (NUP 23125, 97.1 mm SL) in occlusal view, anterior portion upwards.
Coloration in alcohol. Background pale beige to yellowish-brown; ventral region yellowish-white. Head mainly brownish; ventral region yellowish-white. No apparent sexual dichromatism. Dorsal region of head with nasal and infraorbital stripes faded. Two dark-brown oblique stripes running from anterior to posterior margin of lachrymal, one at superior (pre-lachrymal stripe) and another at inferior margin (post-lachrymal stripe). Dark-brown spots or stripes absent on cheek and opercular series. Seven dorsal melanophore patches, continuous with flank bars, as conspicuous as, and indistinguishable from them; DMP1, at distal half of caudal peduncle; DMP2, at base of last dorsal-fin rays and origin of caudal peduncle; DMP3, slightly posterior to base of first dorsal-fin soft rays; DMP4, at base of last dorsal-fin spines; DMP5, at base of middle dorsal-fin spines; DMP6, halfway to base between first and middle dorsal-fin spines; DMP7, on nape and base of first dorsal-fin spines. Six lateral melanophore patches along E1 scale series; LMP1a, at distal half of caudal peduncle; LMP2–3, at vertical through DMP2; LMP4, halfway at vertical through DMP 3 and 4; LMP5, halfway at vertical through DMP 4 and 5; LMP6, slightly posterior at vertical through DMP 6, between pelvic- and anal-fin origins; LMP7, slightly posterior to vertical through pelvic-fin origin. Six flank bars, as LMPs; FBs 4–6 usually connecting LMP to more than one DMP, Y-shaped. Lateral band dark-brown, mainly concentrated along E1 series, as conspicuous as flank bars. Pale beige to brownish-gray fins. Dorsal fin darker on distal margin; small white blotches on soft portion. Anal fin with small white blotches or irregular stripes on last three soft rays. Caudal fin lighter on distal margin; small white blotches on inter-radial scaleless portion; one black blotch, usually ocellated, at base of D3–D7 rays.
FIGURE 5 | Paratypes of Satanoperca setepele. A. NUP 13003, 102.7 mm SL. B. INPA 59667, 118.7 mm SL. C. MCP 54759, 125.3 mm SL.
Coloration in life. Based on Fig. 6, and Stawikowski, Werner (2004:408). Background greenish; ventral region yellowish-white. Head dark-brown on superior region, including upper lip; lachrymal and cheek greenish; preopercle, interopercle, and subopercle pale beige to yellowish; opercle dark-grey or green; ventral region yellowish-white. Eyes brownish, with light yellowish-golden ring around pupil. Pattern of blotches, stripes, bars, and band similar to color in alcohol. Fins orange to reddish. Pelvic fin with greenish blue iridescent filament. Black blotch at base of D3–D7 caudal-fin rays not ocellated.
FIGURE 6 | Fresh uncataloged specimens of Satanoperca setepele. A. Lajeado Reservoir. B. Rio Paranaíba. C. Upper Rio Paraná floodplain.
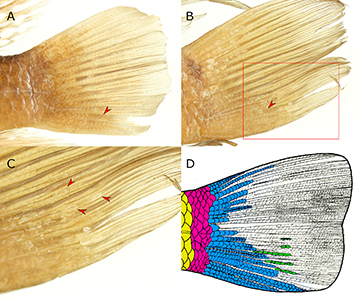
Ontogeny of meristic and morphometric data. Allomery present in squamation of cheek and caudal fin. Scale rows on cheek, 5 rows in specimens between 70.0–130.0 mm SL, 6 rows in specimens between 85.0–155.0 mm SL (R2 = 0.3103). One scale row per interradial membrane in specimens up to 90.0 mm SL; two in larger specimens (double rows, when present, on both lobes or only on ventral one; Fig. 7). Maximum number of scales per row, two at 15.0 mm SL; about five at 40.0 mm SL; 10 at 70.0 mm SL; 15 or more in specimens larger than 100.0 mm SL (either only on ventral lobe or on both lobes). Secondary inter-radial scale series present in specimens larger than 100.0 mm SL. Strong positive allometry in cheek depth (R2 = 0.5113) and lachrymal depth (R2 = 0.7691; reaching a plateau at about 130.0 mm SL). Moderate positive allometry in head depth (R2 = 0.2704; plateau at about 120.0 mm SL); pelvic-fin length (R2 = 0.3363; plateau at about 140.0 mm SL); last dorsal-fin spine length (R2 = 0.1518; plateau at about 110.0 mm SL); snout length (R2 = 0.2525; plateau at about 140.0 mm SL); and interorbital length (R2 = 0.1674). Strong negative allometry in orbital diameter (R2 = 0.6400). Moderate negative allometry in head length (R2 = 0.1701); last anal-fin spine length (R2 = 0.1746); pre-pectoral distance (R2 = 0.1531); and postorbital head length (R2 = 0.2824).
FIGURE 7 | Caudal-fin squamation in Satanoperca setepele in two ontogenetic stages. A. NUP 7333, 89.5 mm SL (arrowhead points to double inter-radial scale series). B. NUP 4644, 141.8 mm SL (arrowhead points to double inter-radial scale series; red rectangle shows area detailed in C. C. Details of the caudal fin of NUP 4644, 141.8 mm SL, highlighting secondary inter-radial scale series. D. Schematic representation of the same specimen, showing the regionalization of the caudal-peduncle and caudal-fin squamation. Yellow, caudal-peduncle scales (from dorsal to ventral E4 to H4 rows); pink, irregularly arranged, transitional scales on the caudal-fin base; blue, primary inter-radial scale series (rows can be single, double, or a mixture of both); green, secondary inter-radial scale series (i.e., those that develop between two branches of a same ray).
Reproductive biology. Based on the specimens from the upper Rio Paraná floodplain. Some features are attributed to sexual dimorphism, such as body shape (males with deeper bodies, while females are smaller), and presence of long filaments in dorsal and pelvic fins in males. Nonetheless, during sexing analysis, it was not possible to corroborate this assumption, since not all specimens with deeper bodies and long filaments were males. Satanoperca setepele is considered a protogynous hermaphrodite species (with males presenting oocytes, which do not develop); however, this hermaphroditism is unfunctional, as no sex reversion was observed (Harumi I. Suzuki, 2013, pers. comm.). It has external fertilization, displays parental care (larvophile mouthbrooders; pers. observation), and reproduces from October to January, with average monthly frequency of breeding specimens 0–10% in December, and 10–25% in January. The average frequency of breeding specimens is 0–10% in lentic and semi-lentic environments, and 10–25% in lotic environments. The L50 (median length of first gonadal maturation) is 86.0 mm in females, and 75.0 mm in males; and the L100 (length at which all specimens can breed) is 116.0 mm (Vazzoler et al., 1997; Suzuki et al., 2004).
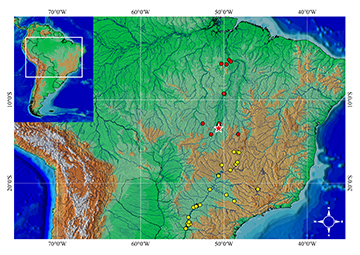
Geographical distribution. Satanoperca setepele is known from the Rio Tocantins basin, including its major tributary, the Rio Araguaia, from Rio Cristalino, Cocalinho (in the Rio Araguaia) and Reservatório Serra da Mesa, Niquelândia (in the Rio Tocantins), to the mouth of the Rio Itacaíunas, Marabá. Non-native records are available from the upper Rio Paraná basin (Fig. 8).
FIGURE 8 | Map of distribution of Satanoperca setepele (red star, type locality; red circles, native localities; yellow circles, non-native localities).
Ecological notes and habitat. Stomachs of Satanoperca setepele from the upper Rio Paraná floodplain mainly contained debris (30–60% of frequency), and aquatic and terrestrial insect larvae, mollusks, and higher plants with less frequency (0–30%) (Hahn et al., 1997; 2004). Localities include rivers (rare occurrence), permanent and temporary lagoons (moderate occurrence), and natural canals (Agostinho et al., 1997); and S. setepele is considered sedentary or with restricted displacements (Suzuki et al., 2004).
Etymology. The specific name setepele (literal translation “sete-pele”, in Portuguese; seven skins, in English) refers to a Brazilian designation for demon, representing its shapeshifter capacity. The name is given in allusion to the folklore around Satanoperca [Greek Σατάν (satan, demon); πέρκα (pérkē, Perca)]. A noun in apposition.
Conservation status. Satanoperca setepele is widely distributed in the Rio Tocantins basin. Therefore, its extinction risk is considered low, based on the currently available data on its geographic distribution. As no specific threats were detected, S. setepele can be categorized as Least Concern (LC), according to the IUCN categories and criteria (IUCN, 2019).
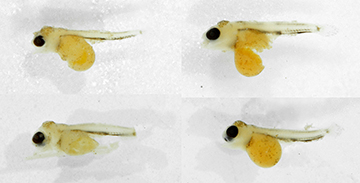
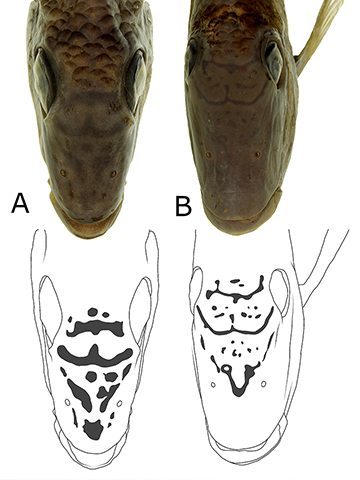
Ontogeny of melanophore marks in the Satanoperca jurupari species group. Larvae of Satanoperca curupira about 4.9 mm SL and S. setepele about 5.0 mm SL are predominantly colorless, except for a melanophore patch on the top of the cranium, another on the caudal-fin base, and four longitudinal lines, which become segmented with growth (Figs. 9–10). In the terminology of Říčan et al. (2005), those lines represent (1) the dorsal melanophore line; (2) the septal melanophore line; (3) the abdominal melanophore line; and (4) the ventral melanophore line. As shown in Fig. 9, the dorsal melanophore line gives place to four melanophore patches, called A, B, C and D, in accordance with Říčan et al. (2005). The three remaining longitudinal lines also give place to a series of melanophore patches, but not necessarily passing through a stage in which four of these patches are present, as in the case of the dorsal line. In S. curupira (Fig. 9) and S. setepele (Fig. 10), it is possible to observe that the dorsal and ventral melanophore lines are more superficial, and the ventral one continues anteriorly dorsal to the abdominal cavity, ending at the same level as the dorsal margin of the eye, dorsal to the pectoral fin. The septal and abdominal lines are deeply imbedded in the body.
At 7.7 mm SL (Fig. 11A), the lateral melanophore patches (LMPs; derived from the septal and abdominal melanophore lines) and dorsal melanophore patches (DMPs) are already visible. The species in the Satanoperca jurupari group analyzed herein present the seven DMPs commonly found in cichlids, but also a distinct DMP8, at least in some specimens (e.g., Figs. 11C–D). As can be seen by comparing Figs. 9 and 11, the DMP2 derives from D; DMPs 3 and 4, from C; DMPs 5 and 6, from B; and DMPs 7 and 8, from A. The spot at the caudal-fin base of the larvae develops into LMP1p and LMP1a.
The seven LMPs, common to many cichlids (see Říčan et al. (2005), are usually present in Satanoperca curupira (Fig. 11) and S. jurupari (Fig. 12), whereas S. pappaterra and S. setepele commonly have only six, with LMPs 2 and 3 fused into an LMP2–3 (Figs. 13 and 14, respectively). In 7.7 mm SL specimens (Fig. 11A), at least four marks are discernible in the ventral midline of the flank, between the anal-fin origin and the end of the caudal peduncle, which are called ventral melanophore patches (VMPs) 1–4 and derive from the ventral melanophore line. Apparently, the anterior portion of this line provides the melanophores that will constitute the visceral pigmentation, instead of developing into VMPs 5–7, as expected by their position in the flank (Fig. 11A). At the same length, there are concentrations of melanophores around the mouth; ventrally and posteriorly to the eye; on and dorsal to the opercle; and on the extra-scapulars. Additionally, the melanophores at the caudal-fin base are concentrated in a conspicuous LMP1p, which is approximately K-shaped (Fig. 11A).
FIGURE 9 | Larvae of Satanoperca curupira (NUP 22961, about 4.9 mm SL). Numbering of melanophore marks derived from the dorsal melanophore line as in Říčan et al. (2005; figs. 1A–B).
FIGURE 10 | Larvae of Satanoperca setepele, NUP 23219, paratypes, lago Montaria, tributary to Rio Araguaia, Brazil.
FIGURE 11 | Ontogenetic variations in juvenile of Satanoperca curupira. A. NUP 22962, 7.7 mm SL. B. NUP 19516, 12.4 mm SL. C. NUP 19516, 13.0 mm SL. D. NUP 19516, 18.2 mm SL. DMP = dorsal melanophore patches; LMP = lateral melanophore patches; VPM = ventral melanophore patches.
FIGURE 12 | Juvenile of Satanoperca jurupari, Igapó at Santa Clara farm, Rio Amazonas basin, NUP 17656, 18.2 mm SL. LMP = lateral melanophore patches.
FIGURE 13 | Ontogenetic and polymorphic variations in juvenile of Satanoperca pappaterra (NUP 23126). A. 11.5 mm SL. B. 12.1 mm SL. C. 12.3 mm SL. D. 13.1 mm SL. E. 13.7 mm SL. F. 14.4 mm SL. G. 14.5 mm SL. H. 15.7 mm SL. I. 17.2 mm SL. J. 18.9 mm SL. K. 19.1 mm SL. L. 23.2 mm SL. Notice the variation in the intensity of the melanophore marks and in the size at which the connection between LMPs, DMPs and VMPs becomes clear.
FIGURE 14 | Ontogenetic and polymorphic variations in juvenile of Satanoperca setepele NUP 8181. A. 13.3 mm SL. B. 14.1 mm SL. C. 16.7 mm SL. D. 17.0 mm SL. E. 18.0 mm SL.
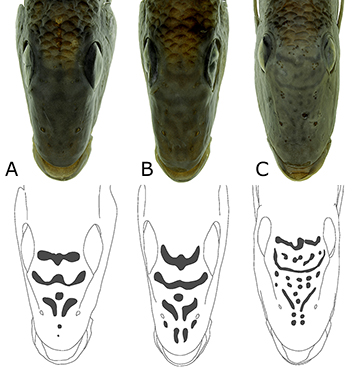
Between 10.0–30.0 mm SL (Figs. 11B–D, 3, 4), the LMPs, DMPs and VMPs gradually become indistinguishable as individual marks, as the migration of melanophores among them causes the formation of the flank bars (FBs). FB1 is formed from the connection of LMP1a with DMP1 and VMP1. LMP2 is connected solely with DMP2 and VMP2 (forming an FB2–3, if fused to LMP3, and FB2, if not fused). However, the migration of melanophores does not necessarily occur between an LMP and the corresponding DMP. In species with an LMP3 distinct from LMP2, the former connects with DMP2 and DMP3, forming a Y-shaped FB3. The same is true of the more anterior bars, which are frequently Y-shaped, being formed by the connection of an LMP with its corresponding DMP and the DMP immediately posterior (Figs. 15–16). However, there are cases in which the more anterior LMPs do connect solely to their corresponding DMPs, or solely to the DMP immediately posterior, forming an unbranched FB (Fig. 17). Additionally, there are some cases in which DMP 4, 5 or 6 can remain completely or almost so disconnected from FBs, which is more common in S. pappaterra than in S. curupira or S. jurupari (Fig. 17C).
In specimens 10.0–30.0 mm SL, some of the melanophore concentrations on the head begin to differentiate into discrete marks. Part of the melanophores on the top of the head form a conspicuous supraorbital stripe; those in the interorbital region and on the dorsal surface of the snout begin to form the interorbital and nasal stripes, respectively; those posterior to the eye begin a postorbital stripe; those ventral to the eye form an infraorbital stripe, occasionally extend onto the ventral arm of the preopercle; and some melanophores situated around the mouth develop into a pre-lachrymal mark. The LMP1p becomes more elongated vertically, usually longer, and darker dorsally than ventrally.
Specimens over 30.0 mm SL are much like adults, mainly because the FBs are already formed, so that the LMPs, DMPs and VMPs are merged into one another (Figs. 15–19). Additionally, the following head marks are fully differentiated from the melanophores scattered on the head: supraorbital, postorbital, infraorbital, post-lachrymal, pre-lachrymal, nasal, interorbital, extra-scapular and supra-opercular. The postorbital stripe lies between the posterior margin of the eye and the dorsal extremity of the preopercle. In Satanoperca species analyzed herein, it has the aspect of a spot of similar depth and length (Figs. 11–19). However, it is topologically equivalent to the anterior portion of the postorbital stripe of Kullander, Ferreira (2006; Fig. 1), the posterior portion being equivalent to our midopercular mark, or to our supra-opercular mark, or both. The post-lachrymal stripe runs along the posteroventral margin of the lachrymal, while the pre-lachrymal stripe runs along the anterodorsal margin of the bone (Figs. 11–19). They are shown in Ota et al. (2018a; Fig. 3) as “oblique dark stripes on lachrymal”. The nasal stripes develop between the contralateral nasal bones, being one dorsal to the nostril, and one ventral to it (Figs. 3; 20–22), although they become fragmented with growth in S. curupira and S. jurupari among species analyzed herein (Figs. 20–21). The interorbital stripes develop between the anterodorsal margins of the orbits, the posterior one across the clc lateralis canal (sensu Kullander, 1983; Fig. 10), and the anterior one running between the contralateral lateral ethmoids, through the anterior portion of the frontal bone, immediately anterior to the attachment of the epaxialis muscle (Figs. 3; 20–22; like the nasal stripes, interorbital stripes may become fragmented with growth, as in Figs. 3; 20–22). The extrascapular mark (Figs. 11–19) lies mostly on the proximal extra-scapular, being about as deep as wide (apparently a topological equivalent of the posteroventral portion of the occipital bar of Kullander, Ferreira, 2006, Fig. 1a). Finally, the midopercular mark appears to be present in the species analyzed herein, but only as a poorly defined concentration of melanophores, rather than a discrete mark (Figs. 1–8).
Regarding melanophore marks, the main differences between specimens between 30.0–50.0 mm SL (Figs. 15, 18A–B, 19A) and those above that size (Figs. 17, 18C–D, 19B–C) are that, in the former, a suborbital stripe is still present; the nasal and interorbital stripes are still conspicuous and entire; and the caudal spot (which derives from LMP1p) has not reached its final shape. The suborbital stripe becomes clear somewhere between 4.9 and 7.7 mm SL (at least in Satanoperca curupira), first as a set of few, isolated melanophores, and later as a dense mass of such cells. However, above 30.0 mm SL the infraorbital stripe starts to fade out, so that specimens over 50.0 mm SL have only a faint trace of it immediately ventral to the eye, or no trace at all (Figs. 15–19).
FIGURE 15 | Juvenile of Satanoperca pappaterra, NUP 23127, 32.0 mm SL. Notice the Y-shaped flank bars and the persistent infraorbital stripe.
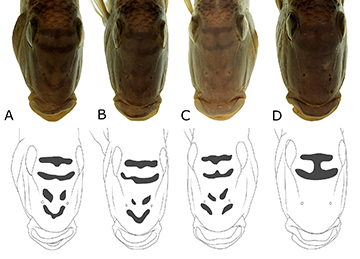
FIGURE 16 | Ontogenetic and polymorphic variations in juvenile of Satanoperca setepele. A. NUP 11127, 23.5 mm SL. B. NUP 17051, 27.8 mm SL. C. NUP 8093, 29.6 mm SL. D. NUP 12825, 36.6 mm SL. E. NUP 11127, 39.9 mm SL. F. Same specimen as in A, interorbital and nasal stripes in their initial stages. G. Same specimen as in C, idem.
FIGURE 17 | Ontogenetic and polymorphic variations in adults of Satanoperca pappaterra, Rio Quilombo, Rio Paraguai basin, Brazil, NUP 2185. A. 80.5 mm SL. B. 96.4 mm SL. C. 116.3 mm SL. D. 126.4 mm SL. Notice how the connection between LMPs and DMPs varies among the specimens, forming either branched or unbranched flank bars. Occasionally, isolated DMPs are present.
Above 50.0 mm SL, the nasal and infraorbital stripes can either become fragmented (Satanoperca curupira and S. jurupari; Figs. 20–21) or faded (S. pappaterra; Fig. 22). In the first case, it appears that some melanophores migrate, establishing at the middle of the distance between those stripes, where they become arranged into spots, while the original stripes become narrower (Figs. 20–21). In the second case, no substantial fragmentation happens, although the nasal stripes disappear almost completely, and the infraorbital ones fade out to some degree (Fig. 22). The LMP1p, which in specimens greater than 10.0 mm SL starts to elongate vertically (Figs. 11–14), and to become darker dorsally, in specimens over 30.0 mm SL is usually completely confined to the base of the dorsal caudal-fin lobe (Figs. 15–19). However, in specimens between30.0–50.0 mm SL it is still bar-shaped (Figs. 15, 18A–B, 19A). Progressively, as the individual surpasses 50.0 mm SL, the ventral portion of the LMP1p disappears (Figs. 17, 18C–D, 19B–C). It is unclear if the melanophores of this ventral portion eventually migrate to the very dorsal portion of the dorsal caudal-fin lobe base or become dispersed on the caudal fin, but the dorsal portion of LMP1p persists as the caudal spot, which is usually roundish and slightly ocellate in adults of the species examined herein (e.g., Figs. 17D, 18D, 19C).
FIGURE 18 | Ontogenetic variations in subadults and adults of Satanoperca curupira. NUP 19490, A. 31.7 mm SL, B. 36.3 mm SL, and C. 60.8 mm SL. NUP 20688, D. 134.4 mm SL, rio Pardo, rio Madeira basin.
FIGURE 19 | Ontogenetic variations in Satanoperca jurupari. A. NUP 20762, 43.0 mm SL. B. NUP 20762, 56.8 mm SL. C. NUP 19402, 138.0 mm
FIGURE 20 | Ontogenetic development of the interorbital and nasal stripes in Satanoperca curupira. A. NUP 19490, 58.7 mm SL. B. NUP 20688, 134.4 mm SL.
FIGURE 21 | Ontogenetic development of the interorbital and nasal stripes in Satanoperca jurupari. A. NUP 20762, 43.0 mm SL. B. NUP 20762, 56.8 mm SL. C. NUP 19402, 138.0 mm SL.
FIGURE 22 | Ontogenetic development of the interorbital and nasal stripes in Satanoperca pappaterra, Rio Quilombo, Rio Paraguai basin, NUP 2185. A. 80.5 mm SL. B. 96.4 mm SL. C. 116.3 mm SL. D. 126.4 mm SL.
Discussion
Satanoperca setepele is herein included in the S. jurupari species-group (sensu Kullander, Ferreira, 1988) by the absence of black rounded blotches on flank and lower meristics, now comprised by S. curupira, S. jurupari, S. leucosticta, S. mapiritensis, S. pappaterra, S. rhynchitis, and S. setepele. Willis et al. (2012), in a species-level phylogenetic analysis of Satanoperca, found two lineages corresponding to possible new species, one identified as “S. sp. Araguaia-Paraná” (= Satanoperca setepele), and another identified as “S. sp. Xingu”. Ota (2013) taxonomically reviewed the genus and found that the specimens from the Rio Xingu present the color pattern of flank bars and longitudinal band similar to Satanoperca setepele, in preserved specimens. However, in living specimens the color pattern is variable from flank bars inconspicuous (almost nonexistent) and longitudinal band conspicuous, to flank bars as conspicuous as longitudinal band. Furthermore, serrations were present in some specimens from the Rio Xingu, whereas serrations in the supracleithrum were absent from S. setepele. Considering the variation in color pattern, and not all specimens from Rio Xingu present serrations on the supracleitrhum, it is not possible to state them as diagnostic features from S. setepele, probably corresponding to still an undescribed species. It is noteworthy that these specimens are from the region of Altamira, Pará, but in the region of Belo Monte, Pará, we identified specimens of S. jurupari. As no consistent diagnostic characters were found, we prefer not to formally describe this species until more data are available.
Another issue involves the specimens from the lower Rio Tocantins basin. Before the filling of the Tucuruí Reservoir, the region was divided into three sections according to geological and hydrological features: Northern, from the mouth of the Rio Tocantins to the Municipality of Tucuruí; Central, corresponding to the Tucuruí Reservoir, between the Municipalities of Tucuruí and Itupiranga; and Southern, region of the Municipality of Itupiranga, characterized by a small floodplain, with isolated lagoons (de Mérona et al., 2010). Satanoperca setepele was recorded by Santos et al. (1984, p. 48, as S. jurupari), between 1980 and 1983, before the filling of the reservoir in 1984. However, 20 years later, Santos et al. (2004), only recorded S. acuticeps (p. 127; identified by us as S. jurupari sensu lato, with lower and elongated body) and S. jurupari (p. 128; also identified by us as S. jurupari sensu lato, with deeper body and overall color brownish-gray). Therefore, it is possible that S. setepele occurred throughout the region of the Tucuruí Reservoir, however after the filling, its distribution became restricted to the Southern section, region of Marabá, Pará, where the Rio Itacaíunas empties, and it does not seem to be found in syntopy with S. jurupari sensu lato. Weidner (2000, p. 225) provided photographs of specimens kept in aquarium, but as it was not specified from which region of the Rio Tocantins they were from, we prefer not to identify them as S. setepele.
In the Rio Mearim and Capim-Guamá basins, Rio Amazonas basin, the preserved specimens can present the color pattern of head without markings (spots, blotches, or stripes), and flank bars and longitudinal band similar to Satanoperca setepele. In the Rio Mearim basin, despite some specimens analyzed by us present the features described above, the photograph of a fresh specimen provided by Soares (2013:103, as S. jurupari) presents an overall color pattern dark-brown and does not resemble S. setepele, even considering those specimens living in environments with high concentrations of organic matter (i.e., Rio Baía, upper Rio Paraná basin). Moreover, Stawikowski, Werner (2004), Abreu et al. (2019, S2) and Guimarães et al. (2020b) also recorded specimens from the Rio Mearim basin (as S. jurupari); however, as no photographs were provided, it was not possible to verify their color pattern. On the other hand, the specimens from the Rio Capim-Guamá basin usually present seven flank bars, less conspicuous than longitudinal band (vs. usually six flank bars, as conspicuous as longitudinal band, in S. setepele). Therefore, these populations are herein considered to be best interpreted as a population of S. jurupari sensu lato.
The occurrence of Satanoperca setepele in the upper Rio Paraná is an uncertain question until current days. Fowler (1954), in the catalog of freshwater fishes from Brazil, kept S. pappaterra as a junior synonym of S. jurupari and did not mention its distribution in that basin. Years later, Britski (1972), studying the freshwater fishes from the State of São Paulo, only recorded Geophagus iporangensis Haseman, 1911 [as G. brasiliensis (Quoy & Gaimard, 1824)] among cichlids. The oldest record of S. setepele in the upper Rio Paraná is MZUSP 21653 (1 specimen), above Sete Quedas Falls, during the monitoring of the Itaipu area (Cetesb, 1977; CTSA, 1981); whereas no records of Satanoperca spp. were found below Sete Quedas Falls, in the lower Rio Paraná. Many species from the Rio Araguaia and Rio Tocantins basins are known to have been introduced in the upper Rio Paraná basin, such as Cichla kelberi Kullander & Ferreira, 2006, Cichla piquiti Kullander & Ferreira, 2006 (Kullander, Ferreira, 2006), Geophagus sveni Lucinda, Lucena & Assis, 2010 (Góis et al., 2015), Hyphessobrycon moniliger Moreira, Lima & Costa, 2002 (Ota et al., 2018b), Leporinus tigrinus Borodin,1929 (Santos et al., 2013; Cavaretto et al., 2020), Leporinus unitaeniatus Garavello & Santos, 2009 (Message et al., 2020), and Serrasalmus geryi Jegú & Santos, 1988 (Deprá et al., 2021b). According to Langeani et al. (2007), the occurrence of S. setepele (as S. pappaterra) can be associated with fish-farming, and according to local fishermen (pers. comm.) it is possible that the species was accidentally introduced along with “tilapias” [i.e., Coptodon rendalli (Boulenger, 1897), and Oreochromis niloticus (Linnaeus, 1758)]. Therefore, S. setepele is herein considered a non-native species in the upper Rio Paraná basin, also highlighted by Agostinho et al. (1997), Langeani et al. (2007), Kullander (2012), Ota et al. (2018a), and Reis et al. (2020).
The analysis of the melanophore coloration in species of the Satanoperca jurupari group showed the same basic elements as in Geophagus species (Gabriel C. Deprá, 2021, pers. comm.), with a few divergent details. In the S. jurupari species group, how the melanophore marks on the flank (DMPs, LMPs and VMPs) become interconnected to form FBs is much more simplified than in Geophagus Heckel, 1840. Whereas in Geophagus those connections differ between species in numerous details, in the S. jurupari group there are only two types of arrangement: one in which the LMPs 2 and 3 are fused, and there are only six FBs; and one in which those marks are not fused, and there are seven FBs. However, differently from Geophagus, the species of the S. jurupari group exhibit numerous melanophore marks on the head, revealing hints of a possible serial homology with the marks on the flank. In other words, there may be a relation between all melanophoric marks on the head and the four melanophore lines described by Říčan et al. (2005): dorsal, septal, abdominal, and ventral.
Říčan et al. (2005) partially recognized that relationship by attributing the marks on the nape and on the opercle to his bar 8, and the marks on the top of the skull (supra-orbital and interorbital stripes), along with the infraorbital stripe, to his bar 9. However, that hypothesis of serial homology does not account for the more anterior marks, i.e., the nasal, pre-lachrymal and post-lachrymal marks. Based on topology alone, we interpret the dorsal extremity of the supraorbital stripe as derived from a melanophore patch related to the dorsal melanophore line, which could be referred to as DMP9. Concurrently, the medial extremity of the interorbital stripes would putatively derive from DMP10, whereas the nasal stripes would derive from DMP11. According to those hypotheses, the putative corresponding LMPs 9, 10 and 11 would have to lie either on the eye rim or on the eye proper. Topology also seems to indicate that the pre-lachrymal and post-lachrymal stripes originate from the connection of a dorsal melanophore line-derivative (i.e., DMP 12) with a septal or abdominal melanophore line-derivative lying on the eye or on the eye rim.
Although Říčan et al. (2005) did not name DMPs, LMPs and VMPs, they seem to have considered the ventral extremity of the suborbital stripe as belonging to the ventral melanophore line, i.e., equivalent to VMP9. Concurrently, the midopercular mark would be equivalent to VMP8, and the supra-opercular mark, to LMP8. Although those hypotheses seem plausible, they do not account for the serial homology of the postorbital and extra-scapular marks observed in the species of the Satanoperca jurupari group. In any case, there is no clear evidence that the melanophore lines described by Říčan et al. (2005) extend to the anterior portion of the head. If such an extension can be demonstrated to exist, the hypotheses of homology for the interobital, nasal, pre-lachrymal and post-lachrymal stripes proposed herein can be tested properly, as well as the implicit hypotheses proposed by Říčan et al. (2005) for the infraorbital stripe and for the supra-opercular and midopercular marks.
Comparative material examined. The same as described in Ota (2013), and Ota et al. (2018a).
Acknowledgments
The authors are grateful to Nupélia and Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais (PEA) for logistic support and Proex/CAPES for travel support as part of the Master dissertation of RRO. To Marli Campos (NUP), Lúcia H. Rapp Py-Daniel and Renildo R. de Oliveira (INPA), Carlos and Margarete Lucena, and Roberto Reis (MCP), Wolmar B. Wosiacki (MPEG), Osvaldo T. Oyakawa, Mário de Pinna, Michel Gianetti, and Aléssio Datovo (MZUSP) for museum, technical support and/or loan of material. To Luiz F. C. Tencatt for photographing the holotype. To Carlos Bernardo M. Alves for providing photographs of fresh specimens from the Rio Paranaíba. To Renata Bartolette and Marcelo Brito for providing photographs of fresh specimens from Lajeado and Serra da Mesa Reservoirs. To Alessandra B. da Silva, Harumi I. Suzuki, and Wladimir M. Domingues for providing photographs of fresh specimens from the upper Rio Paraná floodplain. To Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for granting scholarship and providing grants to RRO (Proc. # 130680/2011–7), WJG (Proc. # 401317/2016–1 and 305200/2018–6) and CSP (Proc. # 308777/2019-0). And to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for granting scholarship to GCD (Proc # 946013).
References
Abreu JMS, Craig JM, Albert JS, Piorski NM. Historical biogeography of fishes from coastal basins of Maranhão State, northeastern Brazil. Neotrop Ichthyol. 2019; 17(2):e180156. http://dx.doi.org/10.1590/1982-0224-20180156
Agostinho AA, Bini LM, Gomes LC, Júlio Júnior HF, Pavanelli CS, Agostinho CS. Fish assemblages. In: Thomaz SM, Agostinho AA, Hahn NS, editors. The upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004. p.223–46.
Agostinho AA, Júlio Júnior HF, Gomes LC, Bini LM, Agostinho CS. Composição, abundância e distribuição espaço-temporal da ictiofauna. In: Vazzoler AEAM, Agostinho AA, Hahn NS, editors. A planície de inundação do alto rio Paraná: aspectos físicos, biológicos e socioeconômico. Maringá: Eduem; 1997. p.179–208.
Azevedo-Santos VM, Coelho PN, Deprá GC. Ichthyofauna of the Ribeirão Frutal and tributaries, upper Rio Paraná basin, Minas Gerais, Southeastern Brazil. Biota Neotrop. 2018; 18(3):e20180517. http://dx.doi.org/10.1590/1676-0611-BN-2018-0517
Barel CDN, Van Oijen MJO, Witte F, Witte-Maas ELM. An introduction to the taxonomy and morphology of the haplochromine Cichlidae from Lake Victoria. Part A. Text. Neth J Zool. 1976; 27(4):333–80. http://dx.doi.org/10.1163/002829677X00199
Bartolette R, Souza-Lima R, Figueiredo CAA, Moraes Júnior DF, Caramaschi EP. Composição taxonômica da ictiofauna da área da UHE Serra da Mesa. In: Mazzoni R, Caramaschi EP, Iglesias-Rios R, editors. Usina Hidrelétrica de Serra da Mesa: 15 anos de estudos da ictiofauna do Alto Tocantins. Rio de Janeiro: Furnas Centrais Elétricas S.A; 2012. p.53–90.
Bartolette R, Vieira CS, Santos JFL, Santos CDC, Luduvice JSV, Passos TS, D’avilla T, Nascimento BO, Ernesto D, Argolo FH, Aguiar AJM, Argolo F, Pereira MSA, Santos TF, Brito MFG. The ichthyofauna in the influence area of the Lajeado reservoir, Tocantins state, Brazil. Check List. 2017; 13(3):2156. http://dx.doi.org/10.15560/13.3.2156
Britski HA. Peixes de água doce do estado de São Paulo. In: Comissão Interestadual da bacia Paraná-Uruguai, organizers. Poluição e Piscicultura. São Paulo: Faculdade de Saúde Pública da USP, Instituto de Pesca; 1972. p.79–107.
Cavaretto RC, Souza-Shibatta L, Celestino LF, Silva OS, Shibatta OA, Sofia SH, Makrakis S, Makrakis M. Evidence for the unexpected dispersal of Leporinus tigrinus Borodin, 1929 through hydroelectric dams in the most regulated Neotropical river basin. Aquat Invasions. 2020; 15(3):497–513. http://dx.doi.org/10.3391/ai.2020.15.3.09
Companhia Ambiental do Estado de São Paulo (Cetesb), Itaipu. Histórico de coleções de peixes efetuadas pela Cetesb na área de Itaipu. Cetesb, Itaipu; 1977.
Companhia de Tecnologia de Saneamento Ambiental (CTSA), Itaipu. Itaipu Binacional-Ictiofauna: complementação do inventário ictiofaunístico. Curitiba: Cetesb, 3; 1981.
Deprá GC, Kullander SO, Pavanelli CS, Graça WJ. A new colorful species of Geophagus (Teleostei: Cichlidae), endemic to the rio Aripuanã in the Amazon basin of Brazil. Neotrop Ichthyol. 2014; 12(4):737–46. http://dx.doi.org/10.1590/1982-0224-20140038
Deprá GC, Ota RR, Vitorino Júnior OB, Ferreira KM. Two new species of Knodus (Characidae: Stevardiinae) from the upper rio Tocantins basin, with evidence of ontogenetic meristic changes. Neotrop Ichthyol. 2021a; 19(1):e200106. https://doi.org/10.1590/1982-0224-2020-0106
Deprá GC, Oliveira AG, Silva AB, Frota A, Proença HC, Message HJ, Reis RB, Ota RR. A new potential invader: first record of the pirambeba Serrasalmus geryi (Characiformes: Serrasalmidae) in the upper Paraná River floodplain, Brazil. J Ichthyol. 2021b; 61(2)190–95. https://doi.org/10.1134/S0032945221020041
Duke Energy International Geração Paranapanema. Peixes do rio Paranapanema. São Paulo: Horizonte Geográfico; 2003.
Fowler HW. Os peixes de água doce do Brasil. Arq Zool. 1954; 9:1–400.
Froehlich O, Cavallaro M, Sabino J, Súarez YR, Vilela MJA. Checklist da ictiofauna do Estado de Mato Grosso do Sul, Brasil. Iheringia. 2017; 107(S):e2017151. http://dx.doi.org/10.1590/1678-4766e2017151
Godoy MP. Peixes e pesca do rio Paraná: área do futuro reservatório de Ilha Grande. Florianópolis: Eletrosul; 1986.
Góis KS, Pelicice FM, Gomes LC, Agostinho AA. Invasion of an Amazonian cichlid in the upper Paraná River: facilitation by dams and decline of a phylogenetically related species. Hydrobiologia. 2015; 746:401–13. http://dx.doi.org/10.1007/s10750-014-2061-8
Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes. Maringá: Eduem; 2007.
Guimarães EC, Brito OS, Ottoni, FP. Peixes. In: Dornas, RAP, Rolim, SG, organizers. Fauna de vertebrados do entorno da estrada de ferro Carajá. Belo Horizonte: Rupestre; 2020a. p.32–51.
Guimarães EC, Brito PS, Gonçalves CS, Ottoni FP. An inventory of ichthyofauna of the Pindaré River drainage, Mearim River basin, Northeastern Brazil. Biota Neotrop. 2020b; 20(4):e20201023. http://dx.doi.org/10.1590/1676-0611-BN-2020-1023
Guimarães EC, Brito PS, Santos JP, Oliveira, Ottoni FP. Supplementary material to Guimarães et al. (2020): Peixes. Fauna de vertebrados ao longo da Estrada de Ferro Carajás. Ichthyol Contrib Peces Criollos. 2021; 76:1–54.
Hahn NS, Andrian IF, Fugi R, Almeida VLLA. Ecologia trófica. In: Vazzoler AEAM, Agostinho AA, Hahn NS, editors. A planície de inundação do alto rio Paraná: aspectos físicos, biológicos e socioeconômico. Maringá: Eduem; 1997. p.209–28.
Hahn NS, Fugi R, Andrian IF. Trophic-ecology of the fish assemblages. In: Thomaz SM, Agostinho AA, Hahn NS, editors. The upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004. p.247–69.
Ilves KL, Torti D, López-Fernández H. Exon-based phylogenomics strengthens the phylogeny of Neotropical cichlids and identifies remaining conflicting clades (Cichliformes: Cichlidae: Cichlinae). Mol Phylogenet Evol. 2017; 118(2018):232–43. http://dx.doi.org/10.1016/j.ympev.2017.10.008
International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN Red List categories and criteria. Version 14 [Internet]. Gland; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Jarduli LR, Garcia DAZ, Vidotto-Magnoni AP, Casimiro ACR, Vianna NC, Almeida FS, Jerep FC, Orsi ML. Fish fauna from the Paranapanema River basin, Brazil. Biota Neotrop. 2020; 20(1):e20180707. http://dx.doi.org/10.1590/1676-0611-BN-2018-0707
Kullander SO. A taxonomical study of the genus Apistogramma Regan, with a revision of Brazilian and Peruvian species (Teleostei: Percoidei: Cichlidae). Bonn Zool Monogr. 1980; 14:1–152.
Kullander SO. Taxonomic studies on the percoid freshwater fish family cichlid in South America. Review of the South America Cichlidae. Stockholm: University of Stockholm, Naturhistoriska Riksmuseet; 1983.
Kullander SO. Cichlid fishes of the Amazon River drainage of Peru. Stockholm: Swedish Museum of Natural History, Naturhistoriska Riksmuseet; 1986.
Kullander SO. Mazarunia mazarunii (Teleostei: Cichlidae), a new genus and species from Guyana, South America. Ichthyol Explor Freshw. 1990; 1(1):3–14.
Kullander SO. Family Cichlidae (Cichlids). In: Reis RE, Kullander SO, Ferraris CJ, Jr., organizers. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.605–54.
Kullander SO. A taxonomic review of Satanoperca (Teleostei: Cichlidae) from French Guiana, South America, with a description of a new species. Cybium. 2012; 36(1):247–62. http://dx.doi.org/10.26028/cybium/2012-361-014
Kullander SO, Ferreira EJG. A new Satanoperca species (Teleostei, Cichlidae) from the Amazon River basin in Brazil. Cybium. 1988; 12(4):343–55.
Kullander SO, Ferreira EJG. A review of the South American cichlid genus Cichla, with descriptions of nine new species (Teleostei: Cichlidae). Ichthyol Explor Freshw. 2006; 17(4):289–398.
Kullander SO, Nijssen H. The cichlids of Surinam. Teleostei: Labroidei. Leiden: E. J. Brill; 1989.
Langeani F, Castro RMC, Oyakawa OT, Shibatta OA, Pavanelli CS, Casatti L. Diversidade da ictiofauna do alto rio Paraná: composição atual e perspectivas futuras. Biota Neotrop. 2007; 7(3):181–97. Available from: https://www.biotaneotropica.org.br/v7n3/pt/abstract?article+bn03407032007
López-Fernández H, Honeycutt RL, Stiassny MLJ, Winemiller KO. Morphology, molecules, and character congruence in the phylogeny of South American geophagine cichlids (Perciformes, Labroidei). Zool Scr. 2005: 34(6):627–51. https://doi.org/10.1111/j.1463-6409.2005.00209.x
López-Fernández H, Winemiller KO, Honeycutt RL. Multilocus phylogeny and rapid radiations in Neotropical cichlid fishes (Perciformes: Cichlidae: Cichlinae). Mol Phylogenet Evol. 2010; 55(3):1070–86. https://doi.org/10.1016/j.ympev.2010.02.020
Lucinda PHF, Freitas IS, Soares AB, Marques EE, Agostinho CS, Oliveira RF. Fish, Lajeado Reservoir, rio Tocantins drainage, State of Tocantins, Brazil. Check List. 2007; 3(2):70–83. http://dx.doi.org/10.15560/3.2.70
de Mérona B, Juras AA, Santos GM, Cintra IHA. Os peixes e a pesca no baixo Rio Tocantins: vinte anos depois da UHE Tucuruí. Brasília: Eletronorte; 2010.
Message HJ, Deprá GC, Silva AB, Proença HC, Penido IS, Oliveira RC, Ota RR. Leporinus unitaenatus Garavello, Santos, 2009. Bol Soc Bras Ictiologia. 2020; 133:62–65. Available from: https://www.sbi.bio.br/images/sbi/boletim-docs/2020/setembro_133.pdf
Ota RR. Revisão taxonômica de Satanoperca Günther, 1862 (Perciformes, Cichlidae), com a descrição de três espécies novas. [Master Dissertation]. Maringá: Universidade Estadual de Maringá; 2013.
Ota RR, Kullander SO, Deprá GC, Graça WJ, Pavanelli CS. Satanoperca curupira, a new cichlid species from the rio Madeira basin in Brazil (Teleostei: Cichlidae). Zootaxa. 2018a; 4379(1):103–12. http://dx.doi.org/10.11646/zootaxa.4379.1.6
Ota RR, Deprá GC, Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: revised, annotated and updated. Neotrop Ichthyol. 2018b; 16(2):e170094. http://dx.doi.org/10.1590/1982-0224-20170094
Oyakawa OT, Menezes NA. Checklist of freshwater fishes from São Paulo State, Brazil. Biota Neotrop. 2011; 11(S1a):19–31. Available from: http://www.biotaneotropica.org.br/v11n1a/en/abstract?inventory+bn0021101a2011
Pavanelli CS, Graça WJ, Zawadzki CH, Britski HA, Vidotti AP, Avelino GS, Veríssimo S. Fishes from the Corumbá Reservoir, Paranaíba River drainage, Upper Paraná River basin, State of Goiás, Brazil. Check List. 2007; 3(1):58–64. https://doi.org/10.15560/3.1.58
Reia L, Costa e Silva GS, Garcia-Ayala JR, Vicensotto MPF, Benine RC. Ichthyofauna of the ribeirão Sucuri, a tributary of the rio Tietê, upper rio Paraná basin, southeastern Brazil. Check List. 2020; 16(3):711–28. http://dx.doi.org/10.15560/16.3.711
Reis R, Frota A, Deprá GC, Ota RR, Graça WJ. Freshwater fishes from Paraná State, Brazil: an annotated list, with comments on biogeographic patterns, threats, and future perspectives. Zootaxa. 2020; 4868(4):451–94. http://dx.doi.org/10.11646/zootaxa.4868.4.1
Říčan O, Musilová Z, Muška M, Novák J. Development of coloration patterns in Neotropical cichlids (Teleostei: Cichlidae: Cichlasomatinae). Folia Zool. 2005; 54(monog1):1–46.
Santos CJ, Tencatt LFC, Ota RR, Graça WJ. Second record of Leporinus tigrinus Borodin, 1929 (Characiformes: Anostomidae) in the upper Paraná River basin, Brasil. Check List. 2013; 9(6):1543–44. http://dx.doi.org/10.15560/9.6.1543
Santos GM, Jegú M, Mérona B. Catálogo de peixes comerciais do baixo rio Tocantins; projeto Tucuruí. Manaus: Eletronorte, CNPq, INPA; 1984.
Santos GM, Mérona B, Juras AA, Jégu M. Peixes do baixo rio Tocantins: 20 anos depois da Usina Hidrelétrica de Tucuruí. Brasília: Eletronorte; 2004.
Soares EC. Peixes do Mearim. Plural. 2013; (10):1–130. Available from: https://livrozilla.com/doc/305084/peixes-do-mearim
Soares AB, Pelicice FM, Lucinda PHF, Akama A. Diversidade de peixes na área de influência da barragem de Peixe Angical, antes e após a formação do reservatório. In: Agostinho CS, Pelicice FM, Marques EE, editors. Reservatório de Peixe Angical: bases ecológicas para o manejo da ictiofauna. São Carlos: RiMa; 2009. p.15–27.
Stawikowski R, Werner U. Die Buntbarsche Amerikas. Band 3: Erdfresser, Hecht– und Kammbuntbarsche. Stuttgart: Ulmer; 2004.
Suzuki HI, Vazzoler AEAM, Marques EE, Lizama MAP, Inada P. Reproductive ecology of the fish assemblages. In: Thomaz SM, Agostinho AA, Hahn NS, editors. The upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004. p.271–91.
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19. Available from: https://sfi-cybium.fr/en/node/2423
Vazzoler AEAM, Suzuki HI, Marques EE, Lizama MAP. Primeira maturação gonadal, períodos e áreas de reprodução. In: Vazzoler AEAM, Agostinho AA, Hahn NS, editors. A planície de inundação do alto rio Paraná: aspectos físicos, biológicos e socioeconômicos. Maringá: Eduem; 1997. p.249–66.
Weidner T. Südamerikanische Erdfresser. El Paso: Cichlid Press: 2000.
Willis SC, López-Fernández H, Montaña CG, Farias IP, Ortí G. Species-level phylogeny of ‘Satan’s perches’ based on discordant gene trees (Teleostei: Cichlidae: Satanoperca Günther 1862). Mol Phylogenet Evol. 2012; 63(3):798–808. http://dx.doi.org/10.1016/j.ympev.2012.02.018
Authors
![]() Renata Rúbia Ota1
Renata Rúbia Ota1 ![]() ,
, ![]() Gabriel de Carvalho Deprá1,
Gabriel de Carvalho Deprá1, ![]() Sven Kullander2,
Sven Kullander2, ![]() Weferson Júnio da Graça1,3,4 and
Weferson Júnio da Graça1,3,4 and ![]() Carla Simone Pavanelli 1,3
Carla Simone Pavanelli 1,3
[1] Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil. (RRO) renatarubia.ota@gmail.com (corresponding author), (GCD)
gabrieldepra@gmail.com.
[2] Department of Zoology, Swedish Museum of Natural History. Stockholm, Sweden. (SK) sven.kullander@svenkullander.se.
[3] Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), Centro de Ciências Biológicas (CCB), Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil. (CSP) carlasp@nupelia.uem.br.
[4] Departamento de Biologia, Centro de Ciências Biológicas, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil. (WJG) weferson@nupelia.uem.br.
Authors Contribution 
Renata Rúbia Ota: Conceptualization, Data curation, Formal analysis, Investigation, Writing-original draft, Writing-review and editing.
Gabriel de Carvalho Deprá: Data curation, Formal analysis, Investigation, Writing-original draft, Writing-review and editing.
Sven Kullander: Conceptualization, Data curation, Formal analysis, Investigation, Writing-review and editing.
Weferson Júnio da Graça: Investigation, Supervision, Writing-review and editing.
Carla Simone Pavanelli: Investigation, Supervision, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The authors declare no competing interests.
How to cite this article
Ota RR, Deprá GC, Kullander S, Graça WJ, Pavanelli CS. A new species of Satanoperca (Teleostei: Cichlidae) from the Rio Tocantins basin, Brazil. Neotrop Ichthyol. 2021; 19(4):e210116. https://doi.org/10.1590/1982-0224-2021-0116
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted October 26, 2021 by George Mattox
Accepted October 26, 2021 by George Mattox
![]() Submitted July 19, 2021
Submitted July 19, 2021
![]() Epub December 17, 2021
Epub December 17, 2021