![]() Julia Marx1,2
Julia Marx1,2 ![]() ,
, ![]() Caio R. Pimentel3,
Caio R. Pimentel3, ![]() Luiz A. Rocha4 and
Luiz A. Rocha4 and ![]() Hudson T. Pinheiro5
Hudson T. Pinheiro5
PDF: Download Here | Supplementary: S1 S2 S3 S4 S5 S6 S7 | Cite this article
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Gradientes ambientais e a geografia são fatores-chave na determinação da biodiversidade e na estruturação das comunidades em ecossistemas recifais. Para investigar como a biogeografia e os gradientes de profundidade moldam as comunidades de peixes recifais, analisamos dois arquipélagos oceânicos do Atlântico Sul-Ocidental: Fernando de Noronha, maior e mais próximo do continente, e São Pedro e São Paulo, menor e mais isolado. Utilizando mergulhos em circuito fechado de gases mistos até 120 m de profundidade, realizamos censos visuais de 6.982 indivíduos de 95 espécies, categorizadas por estratos de profundidade: raso, mesofótico superior e mesofótico inferior. Esperávamos que as assembleias de peixes fossem estruturadas de acordo com as previsões da Teoria da Biogeografia de Ilhas e gradientes ambientais: ilhas maiores e mais próximas e recifes rasos sustentando maior riqueza funcional e de espécies, enquanto ilhas menores e isoladas e recifes mais profundos apresentariam maior originalidade e especialização funcional. Nossos resultados revelaram assembleias estruturadas geograficamente e por profundidade, com o estrato mesofótico inferior mais isolado. Enquanto Fernando de Noronha apresentou maior riqueza de espécies geral, São Pedro e São Paulo exibiu, inesperadamente, maior riqueza funcional, especialmente nos estratos raso e mesofótico inferior, além de maior originalidade e especialização funcional. Esses achados destacam os processos evolutivos e ecológicos combinados que moldam as comunidades recifais insulares e reforçam a necessidade de integrar a diversidade funcional na conservação de recifes profundos e isolados.
Palavras-chave: Atributos funcionais, Conservação marinha, Ecossistemas mesofóticos, Endemismo, Teoria da Biogeografia de Ilhas.
Introduction
Depth gradients have emerged as a key factor influencing the assembly of reef fish communities on a global scale (Jankowski et al., 2015; Rocha et al., 2018; Pinheiro et al., 2023), offering valuable insights into how environmental and biogeographic processes shape biodiversity. Mesophotic ecosystems (MEs), which occur between 30 and 150 m, are particularly relevant in this context. These ecosystems exhibit high species turnover, reduced biodiversity, and significant shifts in trophic structure with increasing depth (Pinheiro et al., 2016; Rocha et al., 2018; Pimentel et al., 2022), reflecting the interplay between environmental conditions (e.g., temperature, light availability, turbidity, reef rugosity), and biological interactions (e.g., predation, competition, habitat selection), that govern species distribution. For instance, in tropical reef systems, depth-related decreases in light and temperature have been shown to limit the distribution of herbivorous fish, while complex reef structures provide refuges that reduce predation pressure and allow higher species coexistence (Nemeth, Appeldoorn, 2009; Smallhorn-West et al., 2017; Williams et al., 2019). By exploring the functional connections between shallow and deep reef habitats, including the role of depth-related fish endemism, we can better understand how these ecosystems contribute to assemblage structure and ecosystem processes (Lindfield et al., 2016; Simon et al., 2016).
Understanding depth-related processes requires approaches that extend beyond species counts, focusing on the functional role organisms play within ecosystems. In marine environments, fishes are key regulators of food webs and nutrient cycling, underscoring the value of functional approaches for ecological and conservation studies (Ives, Hughes, 2017; Albouy et al., 2019; Auber et al., 2022). Biodiversity is now widely understood to encompass genetic, phylogenetic, and functional dimensions, rather than being limited to species richness (Cardinale et al., 2012; Mouillot et al., 2013a). Functional diversity, in particular, has emerged as a powerful tool for predicting ecosystem responses to environmental changes, as it directly reflects the traits that mediate species’ interactions with their environment (Villéger et al., 2008; Yeager et al., 2017). As anthropogenic impacts on ecosystems intensify, functional approaches are increasingly used in conservation biology to better preserve ecosystem functionality and resilience (Buisson et al., 2013; Parravicini et al., 2021). Examining the functional traits of fish assemblages across habitats and regions provides critical insights into the ecological consequences of species loss and helps pinpoint areas where conservation efforts should be prioritized (Mouillot et al., 2013a; Parravicini et al., 2014).
This functional perspective is particularly relevant when examining isolated marine ecosystems, such as Fernando de Noronha Archipelago and Saint Peter and Saint Paul Archipelago, which stand out as critical study sites, where recent discoveries of new species have expanded our understanding of biogeography and biodiversity patterns in Brazilian oceanic islands (Souza et al., 2015; Pinheiro et al., 2018a; Pimentel et al., 2020; Mincarone et al., 2022). These locations are about 600 km apart, exhibiting unique characteristics. Fernando de Noronha, the larger archipelago, harbours higher species richness and shares more species with the continental reefs (Pimentel et al., 2020). In contrast, Saint Peter and Saint Paul Archipelago, characterized by its extreme isolation and small insular area, displays higher rates of endemism (Rosa et al., 2016). Both historical and current levels of isolation are acknowledged as key factors influencing the distribution patterns of reef fish species (Floeter et al., 2008; Parravicini et al., 2013, Ferrari et al., 2023) and the composition of assemblages on oceanic islands (Bender et al., 2017; Quimbayo et al., 2019).
These unique features underline the ecological significance of both archipelagos, emphasizing the need for tailored conservation efforts. Reef are the most diverse marine ecosystem and support extremely high biodiversity in addition to providing ecosystem services that benefit humans (Graham, Nash, 2013; Harborne et al., 2017; Hughes et al., 2017; Brandl et al., 2019). However, these ecosystems are rapidly degrading due to multiple anthropogenic impacts, which increases the urgency for effective management (Bellwood et al., 2004; Pinheiro et al., 2023). Importantly, both archipelagos are part of Marine Protected Areas, with zones designated as no-take and sustainable use (Pimentel et al., 2020). However, despite their scientific and economic significance, management predominantly focuses on shallower no-take zones, highlighting the need to extend protection to deeper habitats, particularly those beyond 50 m (Jacquemont et al., 2024). Additionally, studying assemblage structure across depth gradients is crucial to understanding how biodiversity and ecosystem processes vary with depth, providing valuable insights to inform conservation strategies.
In this study, we investigated how depth and biogeographic factors (e.g., isolation, habitat availability and species composition), shape the functional structure of reef fish assemblages in two oceanic islands of the Southwestern Atlantic Ocean. Following the Theory of Island Biogeography (MacArthur, Wilson, 1967), we expect Fernando de Noronha, being larger and closer to the continent, to sustain higher species and functional richness due to greater habitat diversity and connectivity to the continental coast (Pinheiro et al., 2018b). In contrast, the smaller and more isolated São Pedro and São Paulo Archipelago is predicted to harbours lower species richness but higher functional originality and specialization, reflecting stronger ecological filtering and higher endemism (Floeter et al., 2008; Ferrari et al., 2023). Additionally, within both archipelagos, we predict that species and functional richness will decline with increasing depth, consistent with environmental constraints and turnover processes along depth gradients (Rocha et al., 2018; Lesser et al., 2019; Pinheiro et al., 2023).
Material and methods
Study site. We sampled two oceanic islands in the Brazilian Province: Saint Peter and Saint Paul’s Archipelago (SPSPA), situated about 1,010 km from the northeastern Brazilian coast (00º55’N 29º21’W), and Fernando de Noronha Archipelago (FNA), located 345 km off the northeastern Brazilian coast (03º50’S 32º25’W) (Fig. 1). Both locations are part of a mosaic of marine protected areas, containing both sustainable-use and no-take zones. Whereas SPSPA is inhabited by only four people (researchers and military), FNA is relatively close to the mainland and is the most accessible oceanic island in Brazil, with over 3,000 residents and receiving over 100,000 tourists every year (Cristiano et al., 2020).
FIGURE 1| Location of the Saint Peter and Saint Paul Archipelago and Fernando de Noronha Archipelago, Brazil (South-western Atlantic).
The shallow (≤ 30 m depth) marine zone of SPSPA consists of rocky shores covered by macroalgae, coralline algae, turf, and bryozoans (Magalhães et al., 2015). The mesophotic component of the SPSPA reef system is composed mainly by vertical rocky walls and reef habitat dominated by sponges and black corals (Rosa et al., 2016). Saint Peter and Saint Paul’s Archipelago is directly influenced by the surface South Equatorial Current, which flows westwards, and the Equatorial Undercurrent, flowing eastwards at depths between 40 and 150 m (Rosa et al., 2016). The presence of strong up and downwelling currents increases the amount of nutrients in the water and favours the occurrence of many pelagic species, forming an important fishing ground (Pinheiro et al., 2020). Additionally, SPSPA is the world’s smallest remote tropical archipelago, formed by a group of small rocky islands (Vaske Jr. et al., 2005).
Fernando de Noronha is the largest Brazilian oceanic archipelago and consists of 21 islands and islets. The main island has two distinct sides: the leeward side, locally known as the “inside sea” facing north-west; and the windward side, locally known as the “outside sea” facing southeast, which receives constant winds and is strongly influenced by the low nutrient waters of the South Equatorial current (Martins et al., 2021). FNA reef ecosystems comprise a mosaic of habitats, where the shallow reefs (≤ 30 m) are composed of volcanic rocks, mainly covered by turf algae and brown macroalgae, with a low coral cover (Aued et al., 2018; Pimentel et al., 2020). The deeper reefs are mainly covered by sponges, scleractinian corals and the insular edge platform is followed by a steep wall, characterizing the lower mesophotic reefs (Pimentel et al., 2020).
Data sampling. We assessed the reef fish assemblages by underwater visual censuses (UVC). A total of 87 belt transects (20 x 2 m) were conducted across different depths by using closed-circuit mixed gas rebreather dives in shallow and mesophotic ecosystems. Different sampling strategies were adopted in each island due to differences in island sizes and shelf width, which also affected the technical diving logistics (Tab. S1). Due to a larger shelf, five diving sites ranging from 4 to 112 m depth were sampled around FNA in 2019, while just one site covering a depth gradient spanning 9–107 m was sampled at SPSPA in 2017. The samples were classified into three strata: shallow, 0–30 m, and upper (31–60 m) and lower mesophotic (61–120 m deep).
Data analysis. To assess whether sampling effort was comparable across depths, we first constructed rarefaction curves (Fig. S2) using the “iNEXT” package (Hsieh et al., 2016). The analyses were conducted using total abundance data at the transect scale. Data was Hellinger-transformed and dissimilarity matrices between pairs of transects were computed using Bray-Curtis distances. To evaluate patterns in assemblage structure, we first performed a Principal Coordinates Analysis (PCoA). We then tested the effects of depth and location using a Permutational Multivariate Analysis of Variance (PERMANOVA), with dissimilarities calculated using ‘vegdist’ function and PERMANOVA performed with ‘adonis2’. Also, we measured species’ contribution to dissimilarities between the strata trough a Similarity Percentages (SIMPER) analysis, using the ‘simper’ function. All multivariate analyses were performed using the “vegan” package (Oksanen et al., 2017).
We used afunctional diversity approach to assess differences in the structure of fish assemblages between the two studied archipelagos and their responses to environmental gradients (Mouillot et al., 2013). Six traits were selected to describe the morphological, behavioural, and biological functions of the reef fish species surveyed (Mouillot et al., 2014): size class (0–7 cm, 7.1–15 cm, 15.1–30 cm, 30.1–50 cm, 50.1–80 cm, > 80 cm), home range (sedentary, mobile and very mobile), diel activity (day, night and both), schooling (solitary, pair, small, medium and large school), water position (high, low and bottom) and diet (herbivores, omnivores, carnivores, planktivorous, mobile invertebrates feeders and sessile invertebrates feeders), (see Tab. S3). The functional traits have been compiled from available literature (Quimbayo et al., 2021) and FishBase (Froese, Pauly, 2024).
First, we calculated pairwise species trait distances using the Gower distance metric using ‘funct.dist’ function in “mFD” package (Magneville et al., 2022), which is appropriate for datasets containing mixed trait types (Gower, 1971). The resulting distance matrix was then used to build the functional trait space for each assemblage, across locations (FNA and SPSPA) and depth strata, as well as for the global species pool, defined as all species recorded in both locations.The ‘quality.fspaces’ function was used to assess the quality of functional space, leading to the decision to calculate trait space across two axes (PC4 and PC5, Tab. S4). We used Principal Coordinate Analysis (PCoA) to visualize the functional trait space occupied by fish assemblages, with polygons delineating the functional distances among species within each transect. At the transect level, we computed four functional α–diversity indices using the ‘alpha.fd.multidim’ function: functional richness (FRic), based on species presence, and functional specialization, divergence and originality, all weighted by species abundance. These indices capture different aspects of the ecological niche (spatial, temporal, trophic dimensions), and were used to evaluate how the functional structure of assemblages’ changes with depth. Functional richness, measured by the diversity of the species biological traits presented in the studied assemblages, represents the niche space occupied by the species within a community (Villéger et al., 2008), and it was used to test whether functional space contracts with decreasing species richness at greater depths and geographical isolation. Additionally, functional divergence refers to the degree of ecological differentiation between assemblages, with higher values indicating lower overlap in ecological/functional traits, which imply greater difference in resource use and the ecological niches among species (Mason et al., 2005). Furthermore, functional originality quantifies the distinctiveness of a species’ traits within the community, reflecting how rare or unique a species trait is. A species tends to be less original as it shares more traits with others, which in turn increases redundancy (Pavoine et al., 2017). Thus, assemblages with higher originality values are characterized by lower redundancy, since the functions provided by these species are less likely to be replaced by others (Kondratyeva et al., 2019). Functional specialization indicates how traits can restrict the niche use of a species (Ferry-Graham et al., 2002), i.e., species with highly specialized traits are limited to narrower sets of resources or habitats, whereas generalist species can exploit a broader range. Together, these functional metrics allow us to explore the effects of depth and regional endemism on the assemblages. Finally, to assess the significance of differences in functional indices (richness, specialization, divergence and originality), across depth zones and between locations, we performed a Kruskal-Wallis test (Kruskal, Wallis, 1952). All analyses were performed in R v. 4.3.2 (R Development Core Team, 2023).
Results
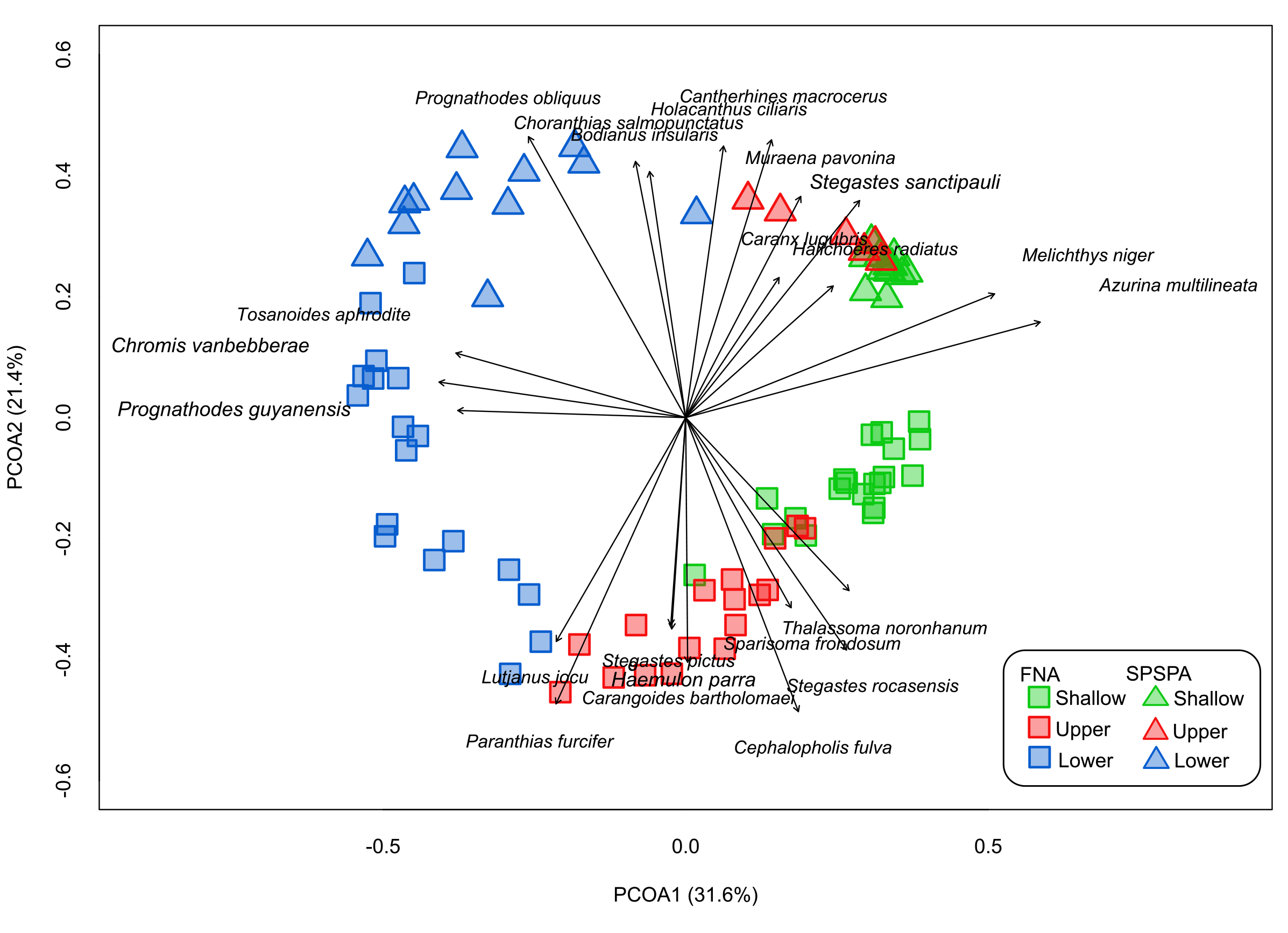
Taxonomic structure of the fish assemblages. We conducted a total of 85 transects across depths ranging from 4 to 111 m. In FNA, 4,600 individuals representing 63 species were recorded across 54 transects, whereas in SPSPA, 2,376 individuals belonging to 31 species were recorded across 31 transects (Tab. S1). The reef fish assemblages were both geographically and depth-structured (Fig. 2), with the lower mesophotic zone more isolated, and a small overlap between upper and shallow strata. The PERMANOVA (Tab. S5) results showed a significant effect of depth (R² = 0.34, p = 0.001), location (R² = 0.18, p = 0.001) and their interaction (R² = 0.08, p = 0.001), in the structure of the reef fish assemblages. A total of 49 species occurred exclusively in FNA, while 17 were only recorded in SPSPA; 17 species were limited to shallow reefs, while 41 were unique to MEs (Tab. S6). Although a marked separation was identified, some degree of convergence can be observed between islands and along the depth gradient, such as the dominance of planktivorous species in all strata. In shallow waters, Azurina multilineata and Melichthys niger were found in both islands, while Tosanoides aphrodite and Chromis vanbebberae predominate in their deeper waters (Fig. 2). Moreover, Paranthias furcifer was the most important contributor to the dissimilarities between the lower mesophotic and upper mesophotic zones of FNA (Tab. S7), accounting for 14% of the dissimilarity. Holocentrus adscensionis and Thalassoma noronhanum contributed most to the similarity between shallow and upper mesophotic zones of this same island (7% and 5%, respectively). In SPSPA assemblages, A. multilineata was the largest contributor to the dissimilarity in shallow and lower mesophotic zones (41% of dissimilarity). In upper and lower mesophotic zones, accounted for 33% of the dissimilarity, followed by M. niger (10%) and Choranthias salmopunctatus (8%).
FIGURE 2| Principal Coordinate Analysis (PCoA) performed on species abundance by transect. Each symbol represents one transect. Axis 1 represents the depth while axis 2 represents the locations. The plotted species are those with r2 greater than or equal to 10%.
Functional structure of the fish assemblages. Fish assemblages exhibited significant differences in species and functional richness across islands and depth strata (Kruskal-Wallis test: p < 0.001). Higher values of species richness were recorded in FNA (Fig. 3), compared to SPSPA, except in the lower mesophotic zone, where SPSPA showed a slightly higher richness (mean: 7.1) than FNA (mean: 6.5). In contrast, functional richness showed an inverse result. SPSPA presented higher functional richness values in both shallow (Fric: 0.32) and lower mesophotic zone (Fric: 0.26), compared to FNA (Fric shallow: 0.22; Fric lower: 0.23). For both locations, the upper mesophotic exhibited the highest functional richness (FNA Fric: 0.36; SPSPA Fric: 0.34).
FIGURE 3| Boxplot of median and quartiles of species and functional richness. Species Richness (Sprichn): total number of species present in each transect. Functional richness (Fric) indicates the proportion of functional space filled by species of each depth strata. The numbers above the bars indicate the average value of the indices at each strata.
Functional specialization (Fig. 4) increased with depth in both archipelagos, with SPSPA showing lower overall values (SPSPA: Fspe: shallow: 0.32; upper: 0.33; lower: 0.34; and FNA: Fspe: shallow: 0.31; upper: 0.34; lower: 0.48). Functional originality showed a different pattern: SPSPA had consistently higher originality values across all depth strata (e.g., Fori: shallow: 0.16; upper: 0.19; lower: 0.14), whereas FNA showed lower values across the gradient (Fori: shallow: 0.14; upper: 0.12; lower: 0.04). No significant differences (Kruskal-Wallis test: p > 0.001), were observed for functional divergence between locations or depth strata.
FIGURE 4| Boxplot of median and quartiles of multidimensional functional indices by island and depth strata. Functional specialization (Fesp) corresponds to the average distance weighted by abundance to the average position of the species of the global set. Functional originality (Fori) is measured by the average weighted distance to the closest species of the global set of species. Functional divergence (Fdiv) means the proportion of abundance supported by the species with the most extreme functional traits. The numbers above the bars indicate the average value of the indices at each strata.
Discussion
As expected, the reef fish assemblages investigated were structured by island and depth, influenced by the evolutionary history of each location and environmental characteristics, which change along the depth gradient. Many species typically found in mesophotic zones prefer cooler temperatures, while others are associated with shallower and warmer waters. Thus, overlap between the shallow and upper mesophotic zones could be attributed to species that display ontogenetic shifts, such as Stegastes, whose juveniles can recruit in deeper reefs, exploiting a broader range of habitats (Ferreira et al., 1998; Gajdzik et al., 2016; Rosa et al., 2016). Additionally, species with broader bathymetric distributions, such as Lutjanus jocu and Bodianus insularis, can thrive across multiple depth strata, thus contributing to the observed similarities between shallow and deeper zones (Rosa et al., 2016; Pimentel et al., 2022). Conversely, distinct patterns emerge in the lower mesophotic, likely driven by species shifts frequently found along the depth gradient, in response to environmental changes (Rocha et al., 2018; Pinheiro et al., 2023; Gragnolati et al., 2024). We found that the lower mesophotic of both islands shared species like Chromis vanbebberae and Tosanoides aphrodite. Nevertheless, we observed that FNA presented widespread species, such as Paranthias furcifer and Prognathodes guyanensis, showing a positive relationship with greater depths, whereas SPSPA showed local endemics, like Prognathodes obliquus and Choranthias salmopunctatus. Therefore, these deeper ecosystems support distinct ecological assemblages with low connectivity with shallower areas, where SPSPA is more isolated, while FNA receives colonizers from both the isolated island (SPSPA) and the continental shelf.
The comparison between the two archipelagos revealed clear distinctions in their fish assemblages, with FNA exhibiting a higher taxonomic richness than SPSPA. These patterns align with previous expectations regarding differences in island size and isolation, which are known to influence species diversity in coral reef ecosystems (Bellwood, Hughes, 2001; Parravicini et al., 2013; Ferrari et al., 2023). Larger and more complex environments typically support a broader range of species, offering more habitats and resources for ecological interactions (Mora et al., 2003; Hachich et al., 2015; Velasquez et al., 2018). In contrast, smaller and more isolated islands often show lower biodiversity due to limitations in available resources and space (Hachich et al., 2015; Velasquez et al., 2018). These differences in species richness could also be attributed to the geological age of each archipelago, as described in the General Dynamic Theory of Oceanic Island Biogeography (Whittaker et al., 2008; Pinheiro et al., 2018a). Under this framework, island age influences habitat availability, with younger islands providing more newly formed substrates and older islands experiencing gradual habitat loss through erosion and subsidence. Therefore, FNA, younger, larger, and closer to the continental shelf likely provides a wider range of habitats and more opportunities for species colonization (Hachich et al., 2015). In contrast, SPSPA, an older, smaller, and highly isolated island, may have reduced and more spatially restricted habitats due to long-term geological degradation and subsidence, supporting fewer species (Luiz et al., 2012).
Interestingly, contrary to our initial expectations, functional richness in SPSPA exceeded that of FNA. In such contexts, this pattern may emerge from the need of different species to efficiently utilize limited resources and minimize competition, particularly in resource-poor and isolated environments (Mason et al., 2005; Mendes et al., 2019). Additionally, we observed greater species richness at upper mesophotic depths for both islands, suggesting that specific depth zones may facilitate higher diversity and functional richness than initially anticipated. Although different from widescale patterns (Pinheiro et al., 2023), a hump-shaped pattern of richness distribution along the depth gradient has already been described for corals, suggesting the existence of local dynamics such as disturbance frequency and/or competitive exclusion (Roberts et al., 2019). This finding highlights the complexity of ecological interactions in these systems and emphasizes that the diversity of functional groups is not necessarily linearly related to species richness (Halpern, Floeter, 2008). In fact, our results contrast with those of Ferrari et al. (2023), who analysed the fish composition (presence/absence data) of 72 oceanic islands around the world, without incorporating depth-related variation, and reported a positive correlation between species and functional richness. The lack of this positive relationship could be related to differences in the evolutionary history and assembly rules, as previously suggested by Pinheiro et al. (2023), when comparing species vs functional richness patterns of Pacific and Atlantic MEs. Here, this apparent decoupling may reflect the presence of fewer, yet more functionally distinct species, potentially resulted from environmental filtering and the strong selective pressures typical of small, isolated, and resource-limited systems (Hachich et al., 2015). Additionally, differences in sampling scale and consequently species pools may also contribute to the discrepancy in the relationship between taxonomic and functional richness.
The occupation of the available functional space indicates the degree of complementarity among the species (Mouillot et al., 2014). A uniform occupation of functional space by a rich assemblage is often associated with ecological resilience (Mouillot et al., 2014), since the presence of multiple species playing the same functions can confer greater stability to ecosystems under environmental disturbances (Mouillot et al., 2013). However, evolving in geographic isolation and under intense limitation of available area, the remote SPSPA assemblage reached high levels of functional originality, especially in mesophotic zones. The predominance of assemblages with high levels of functional originality in a low-diversity environment may indicates little functional plasticity and redundancy, which potentially makes the assemblage more vulnerable to environmental changes (Robinson et al., 2022; Huang et al., 2024).
Conversely, the lower functional diversity found in the richer FNA suggests that species are more densely packed (patch distribution) within the functional space, indicating a redundancy also found in richer faunas (Mouillot et al., 2014). This pattern may reflect the ecological filtering effect of oceanic islands and specific geographic characteristics of the archipelago, such as its large size and proximity to the continental shelf (Mora et al., 2003). While the oceanic environment constrains the ecological traits and species composition of those able to reach and establish in these areas (Pinheiro et al., 2018a; Mazzei et al., 2021), these geographic characteristics may also favour species packing through higher levels of colonization. High migration levels decrease opportunity for speciation in oceanic islands (Luiz et al., 2012; Pinheiro et al., 2017; Mazzei et al., 2021), probably driving lower originality levels compared to SPSPA.
Variations in habitat availability and usage along the depth gradient can lead to different levels of specialization (Jankowiski et al., 2015). The highest values of functional specialization observed in the lower mesophotic strata of both islands suggest that in deeper zones, the species are more specialized in their ecological functions. This result may be attributed to the more restricted or stable conditions found at greater depths, where the competition for specific resources favours the development of specialized traits (Wilson et al., 2008; Lesser et al., 2009). High species richness on shallow reefs is often linked to environmental characteristics, including physical factors such as high structural complexity, temperature, light availability, and hydrodynamics (Wood, 2001; Pereira-Filho et al., 2011; Francini-Filho et al., 2013). These conditions create diverse habitats that support a variety of species. Additionally, ecological factors such as benthic cover and species interactions, along with biogeographic influences like provinciality and regional enrichment, play a significant role (Pinheiro et al., 2023). These factors may promote shifts in niche breadth, resulting in species to become more specialized in response to local conditions (Lesser et al., 2018).
While FNA’s larger size and higher biodiversity might provide some resilience to their assemblages, the growing impacts of the local population and increasing tourism might counterbalance the benefits of the protection offered by the national park (Mello et al., 2025). Indeed, both archipelagos are susceptible to disturbances like climate change, habitat degradation, and overfishing. SPSPA, with its smaller size and higher levels of endemism, faces even greater risks due to limited habitat availability and lower colonization opportunities. These anthropogenic threats could impact the biodiversity metrics studied mainly through the removal of predator, specialist and sensible species, decreasing both taxonomic and functional richness. Therefore, conservation efforts in oceanic islands like FNA and SPSPA must account for the unique ecological dynamics and the vulnerability of these ecosystems. Expanding protection measures to include deeper zones and incorporating functional diversity into conservation plans is critical (Giglio et al., 2018; Pereira et al., 2020; Jacquemont et al., 2024). By understanding the functional roles of species, particularly in relation to depth gradients, conservation strategies can be more effectively tailored to maintain ecosystem services, protect biodiversity, and ensure the resilience of these ecosystems in the face of ongoing environmental changes.
Acknowledgments
We would like to thank Tyler A. Phelps who, together with HTP and LAR, did the sampling for this work. We thank Guilherme Loyola da Cruz for creating the map used in this article. We are grateful to our friend M. Vilella and the staff at his Pizzaria Namoita for all their care during our stay in Noronha, and to K. V. Bastos (CIUFES) and R. Francini-Filho (CEBIMar-USP) for help with logistics.
References
Albouy C, Archambault P, Appeltans W, Araújo MB, Beauchesne D, Cazelles K et al. The marine fish food web is globally connected. Nat Ecol Evol. 2019; 3(8):1153–61. https://doi.org/10.1038/s41559-019-0950-y
Auber A, Waldock C, Maire A, Goberville E, Albouy C, Algar AC et al. A functional vulnerability framework for biodiversity conservation. Nat Commun. 2022; 13(1):4774. https://doi.org/10.1038/s41467-022-32331-y
Aued AW, Smith F, Quimbayo JP, Cândido DV, Longo GO, Ferreira CEL et al. Large-scale patterns of benthic marine communities in the Brazilian province. PLoS ONE. 2018; 13(6):e0198452. https://doi.org/10.1371/journal.pone.0198452
Bellwood DR, Hughes TP. Regional-scale assembly rules and biodiversity of coral reefs. Science. 2001; 292(5521):1532–35. https://doi.org/10.1126/science.1058635
Bellwood DR, Hughes TP, Folke C, Nyström M. Confronting the coral reef crisis. Nature. 2004; 429, 827–833. https://doi.org/10.1038/nature02691
Bender MG, Leprieur F, Mouillot D, Kulbicki M, Parravicini V, Pie MR et al. Isolation drives taxonomic and functional nestedness in tropical reef fish faunas. Ecography. 2017; 40(3):425–35. https://doi.org/10.1111/ecog.02293
Brandl SJ, Rasher DB, Côté IM, Casey JM, Darling ES, Lefcheck JS et al. Coral reef ecosystem functioning: Eight core processes and the role of biodiversity. Front Ecol Environ. 2019; 17(8):445–54. https://doi.org/10.1002/fee.2088
Buisson L, Grenouillet G, Villéger S, Canal J, Laffaille P. Toward a loss of functional diversity in stream fish assemblages under climate change. Glob Chang Biol. 2013; 19(2):387–400. https://doi.org/10.1111/gcb.12056
Cardinale BJ, Duffy JE, Gonzalez A, Hooper DU, Perrings C, Venail P et al. Biodiversity loss and its impact on humanity. Nature. 2012; 486(7401):59–67. https://doi.org/10.1038/nature11148
Cristiano SC, Rockett GC, Portz LC, Souza Filho JR. Beach landscape management as a sustainable tourism resource in Fernando de Noronha island (Brazil). Mar Pollut Bull. 2020; 150:110621. https://doi.org/10.1016/j.marpolbul.2019.110621
Ferrari DS, Floeter SR, Leprieur F, Quimbayo JP. A trait-based approach to marine island biogeography. J Biogeogr. 2023; 50(3):528–38. https://doi.org/10.1111/jbi.14549
Ferreira CEL, Gonçalves JEA, Coutinho R, Peret AC. Herbivory by the dusky damselfish Stegastes fuscus (Cuvier, 1830) in a tropical rocky shore: Effects on the benthic community. J Exp Mar Biol Ecol. 1998; 229(2):241–64. https://doi.org/10.1016/S0022-0981(98)00056-2
Ferry-Graham LA, Bolnick DI, Wainwright PC. Using functional morphology to examine the ecology and evolution of specialization. Integr Comp Biol. 2002; 42(2):265–77. https://doi.org/10.1093/icb/42.2.265
Floeter SR, Rocha LA, Robertson DR, Joyeux JC, Smith-Vaniz WF, Wirtz P et al. Atlantic reef fish biogeography and evolution. J Biogeogr. 2008; 35(1):22–47. https://doi.org/10.1111/j.1365-2699.2007.01790.x
Francini-Filho RB, Coni EOC, Meirelles PM, Amado-Filho GM, Thompson FL, Pereira-Filho GH et al. Dynamics of coral reef benthic assemblages of the Abrolhos bank, eastern Brazil: inferences on natural and anthropogenic drivers. PLoS ONE. 2013; 8(1):e54260. https://doi.org/10.1371/journal.pone.0054260
Froese R, Pauly D, editors. FishBase [Internet]. 2024. Available from: https://www.fishbase.org
Gajdzik L, Parmentier E, Sturaro N, Frédérich B. Trophic specializations of damselfishes are tightly associated with reef habitats and social behaviours. Mar Biol. 2016; 163(12):249. https://doi.org/10.1007/s00227-016-3020-x
Giglio VJ, Pinheiro HT, Bender MG, Bonaldo RM, Costa-Lotufo LV, Ferreira CEL et al. Large and remote marine protected areas in the South Atlantic Ocean are flawed and raise concerns: comments on Soares and Lucas. Mar Policy. 2018; 96:13–17. https://doi.org/10.1016/j.marpol.2018.07.017
Gower JC. A general coefficient of similarity and some of its properties. Biometrics. 1971; 27(4):857–71. https://doi.org/10.2307/2528823
Gragnolati M, Rolim FA, Pereira-Filho GH, Athayde ACS, Ciotti ÁM, Motta FS. Vertical structure of reef fish assemblages and light penetration reveal new boundaries of mesophotic ecosystems in the subtropical Southwestern Atlantic. Mar Environ Res. 2024; 198:106527. https://doi.org/10.1016/j.marenvres.2024.106527
Graham NAJ, Nash KL. The importance of structural complexity in coral reef ecosystems. Coral Reefs. 2013; 32(2):315–26. https://doi.org/10.1007/s00338-012-0984-y
Hachich NF, Bonsall MB, Arraut EM, Barneche DR, Lewinsohn TM, Floeter SR. Island biogeography: patterns of marine shallow-water organisms in the Atlantic Ocean. J Biogeogr. 2015; 42(10):1871–82. https://doi.org/10.1111/jbi.12560
Halpern BS, Floeter SR. Functional diversity responses to changing species richness in reef fish communities. Mar Ecol Prog Ser. 2008; 364:147–56. https://doi.org/10.3354/meps07553
Harborne AR, Rogers A, Bozec YM, Mumby PJ. Multiple stressors and the functioning of coral reefs. Annu Rev Mar Sci. 2017; 9(1):445–68. https://doi.org/10.1146/annurev-marine-010816-060551
Hsieh TC, Ma KH, Chao A. iNEXT: An R package for rarefaction and extrapolation of species diversity (Hill numbers). Methods Ecol Evol. 2016; 7(12):1451–56. https://doi.org/10.1111/2041-210X.12613
Huang C, Xu Y, Zang R. Low functional redundancy revealed high vulnerability of the subtropical evergreen broadleaved forests to environmental change. Sci Total Environ. 2024; 935:173307. https://doi.org/10.1016/j.scitotenv.2024.173307
Hughes TP, Barnes ML, Bellwood DR, Cinner JE, Cumming GS, Jackson JBC et al. Coral reefs in the Anthropocene. Nature. 2017; 546(7656):82–90. https://doi.org/10.1038/nature22901
Ives AR, Hughes JB. General relationships between species diversity and stability in competitive systems. Am Nat. 2017; 159(4):388–95. https://doi.org/10.2307/3079248
Jacquemont J, Loiseau C, Tornabene L, Claudet J. 3D ocean assessments reveal that fisheries reach deep but marine protection remains shallow. Nat Comm. 2024; 15(1):4027. https://doi.org/10.1038/s41467-024-47975-1
Jankowski MW, Graham NAJ, Jones GP. Depth gradients in diversity, distribution and habitat specialisation in coral reef fishes: implications for the depth-refuge hypothesis. Mare Ecol Prog Ser. 2015; 540:203–15. https://doi.org/10.3354/meps11523
Kondratyeva A, Grandcolas P, Pavoine S. Reconciling the concepts and measures of diversity, rarity and originality in ecology and evolution. Biol Rev. 2019; 94(4):1317–37. http://doi.org/10.1111/brv.12504
Kruskal WH, Wallis WA. Use of ranks in one-criterion variance analysis. J Am Stat Assoc. 1952; 47(260):583–621. http://dx.doi.org/10.1080/01621459.1952.10483441
Lesser MP, Slattery M, Leichter JJ. Ecology of mesophotic coral reefs. J Exp Mar Biol Ecol. 2009; 375(1–2):1–08. https://doi.org/10.1016/j.jembe.2009.05.009
Lesser MP, Slattery M, Mobley CD. Biodiversity and functional ecology of mesophotic coral reefs. Source: Annu Rev Ecol Evol Syst. 2018; 49(1):49–71. https://doi.org/10.1146/annurev-ecolsys-110617-062423
Lindfield SJ, Harvey ES, Halford AR, McIlwain JL. Mesophotic depths as refuge areas for fishery-targeted species on coral reefs. Coral Reefs. 2016; 35(1):125–37. https://doi.org/10.1007/s00338-015-1386-8
Luiz OJ, Madin JS, Ross Robertson D, Rocha LA, Wirtz P, Floeter SR. Ecological traits influencing range expansion across large oceanic dispersal barriers: insights from tropical Atlantic reef fishes. Proc Biol Sci. 2012; 279(1730):1033–40.
MacArthur RH, Wilson EO. The theory of island biogeography. New Jersey: Princeton University Press; 1967.
Magalhães G, Amado-Filho G, Rosa M, Moura R, Brasileiro P, Moraes F et al. Changes in benthic communities along a 0–60 m depth gradient in the remote St. Peter and St. Paul Archipelago (Mid-Atlantic Ridge, Brazil). Bull Mar Sci. 2015; 91:377–96. https://doi.org/10.5343/bms.2014.1044
Magneville C, Loiseau N, Albouy C, Casajus N, Claverie T, Escalas A et al. mFD: an R package to compute and illustrate the multiple facets of functional diversity. Ecography. 2022; 2022(1):1–15. https://doi.org/10.1111/ecog.05904
Martins K, Pelage L, Justino AKS, Frédou FL, Vaske Júnior T, Le Loc’h F et al. Assessing trophic interactions between pelagic predatory fish by gut content and stable isotopes analysis around Fernando de Noronha archipelago (Brazil), Equatorial West Atlantic. J Fish Biol. 2021; 99(5):1576–90. https://doi.org/10.1111/jfb.14863
Mason NWH, Mouillot D, Lee WG, Wilson JB. Functional richness, functional evenness and functional divergence: The primary components of functional diversity. Oikos. 2005; 111(1):112–18. https://doi.org/10.1111/j.0030-1299.2005.13886.x
Mazzei EF, Pinheiro HT, Simon T, Moura RL, Macieira RM, Pimentel CR et al. Mechanisms of dispersal and establishment drive a stepping stone community assembly on seamounts and oceanic islands. Mar Biol. 2021; 168(7):109. https://doi.org/10.1007/s00227-021-03919-7
Mello TJ, Longhini CM, Wanderley BMS, Silva CA, Lehrback BD, Bom FC et al. Pollution affects even oceanic marine protected areas in Southwestern Atlantic. Environ Poll. 2025; 366 :125485. https://doi.org/10.1016/j.envpol.2024.125485
Mendes TC, Quimbayo JP, Bouth HF, Silva LPS, Ferreira CEL. The omnivorous triggerfish Melichthys niger is a functional herbivore on an isolated Atlantic oceanic island. J Fish Biol. 2019; 95(3):812–19. https://doi.org/10.1111/jfb.14075
Mincarone MM, Eduardo LN, Di Dario F, Frédou T, Bertrand A, Lucena-Frédou F. New records of rare deep-sea fishes (Teleostei) collected from off north-eastern Brazil, including seamounts and islands of the Fernando de Noronha Ridge. J Fish Biol. 2022; 101(4):945–59. https://doi.org/10.1111/jfb.15155
Mora C, Chittaro PM, Sale PF, Kritzer JP, Ludsin SA. Patterns and processes in reef fish diversity. Nature. 2003; 421:933–36. https://doi.org/10.1038/nature01393
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR. A functional approach reveals community responses to disturbances. Trends Ecol Evol. 2013a; 28(3):167–77. https://doi.org/10.1016/j.tree.2012.10.004
Mouillot D, Bellwood DR, Baraloto C, Chave J, Galzin R, Harmelin-Vivien M et al. Rare species support vulnerable functions in high-diversity ecosystems. PLoS Biol. 2013b; 11(5):e1001569. https://doi.org/10.1371/journal.pbio.1001569
Mouillot D, Villéger S, Parravicini V, Kulbicki M, Arias-González JE, Bender M et al. Functional over-redundancy and high functional vulnerability in global fish faunas on tropical reefs. PNAS. 2014; 111(38):13757–62. https://doi.org/10.1073/pnas.1317625111
Nemeth M, Appeldoorn R. The distribution of herbivorous coral reef fishes within fore-reef habitats: the role of depth, light and rugosity. Caribb J Sci. 2009; 45(2–3):247–53. https://doi.org/10.18475/cjos.v45i2.a11
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D et al. Vegan: community ecology package. Version 2.4-3 [Internet]. 2017. Available from: https://CRAN.R-project.org/package=vegan
Parravicini V, Bender MG, Villéger S, Leprieur F, Pellissier L, Donati FGA et al. Coral reef fishes reveal strong divergence in the prevalence of traits along the global diversity gradient. Proc Biol Sci. 2021; 288(1961):20211712. https://doi.org/10.1098/rspb.2021.1712
Parravicini V, Kulbicki M, Bellwood DR, Friedlander AM, Arias-Gonzalez JE, Chabanet P et al. Global patterns and predictors of tropical reef fish species richness. Ecography. 2013; 36(12):1254–62. https://doi.org/10.1111/j.1600-0587.2013.00291.x
Parravicini V, Villéger S, McClanahan TR, Arias-González JE, Bellwood DR, Belmaker J et al. Global mismatch between species richness and vulnerability of reef fish assemblages. Ecol Lett. 2014; 17(9):1101–10. https://doi.org/10.1111/ele.12316
Pavoine S, Bonsall MB, Dupaix A, Jacob U, Ricotta C. From phylogenetic to functional originality: Guide through indices and new developments. Ecol Indic. 2017; 82:196–205. https://doi.org/10.1016/j.ecolind.2017.06.056
Pereira PHC, Macedo CHR, Lima GV, Benevides LJ. Effects of depth on reef fish flight initiation distance: Implications of deeper reefs conservation. Environ Biol Fish. 2020; 103(10):1247–56. https://doi.org/10.1007/s10641-020-01017-z
Pereira-Filho GH, Amado-Filho GM, Guimarães SMPB, Moura RL, Sumida PYG, Abrantes DP et al. Reef fish and benthic assemblages of the Trindade and Martin Vaz island group, Southwestern Atlantic. Braz J Oceanogr. 2011; 59(3):201–12. https://doi.org/10.1590/s1679-87592011000300001
Pimentel CR, Rocha LA, Shepherd B, Phelps TAY, Joyeux J-C, Martins AS et al. Mesophotic ecosystems at Fernando de Noronha archipelago, Brazil (South-western Atlantic), reveal unique ichthyofauna and need for conservation. Neotrop Ichthyol. 2020; 18(4):e200050. https://doi.org/10.1590/1982-0224-2020-0050
Pimentel CR, Pinheiro HT, Giarrizzo T, Francini-Filho RB, Reis-Filho JA, Rocha LA et al. Ecological links between pelagic and mesophotic reef fishes in an oceanic archipelago of the Equatorial Atlantic Ocean. Diversity. 2022; 14(4):273. https://doi.org/10.3390/d14040273
Pinheiro HT, Bernardi G, Simon T, Joyeux J-C, Macieira RM, Gasparini JL et al.Island biogeography of marine organisms. Nature. 2017; 549:82–85. https://doi.org/10.1038/nature23680
Pinheiro HT, Goodbody-Gringley G, Jessup ME, Shepherd B, Chequer AD, Rocha LA. Upper and lower mesophotic coral reef fish communities evaluated by underwater visual censuses in two Caribbean locations. Coral Reefs. 2016; 35(1):139–51. https://doi.org/10.1007/s00338-015-1381-0
Pinheiro HT, MacDonald C, Quimbayo JP, Shepherd B, Phelps TA, Loss AC et al. Assembly rules of coral reef fish communities along the depth gradient. Curr Biol. 2023; 33(8):1421–30. https://doi.org/10.1016/j.cub.2023.02.040
Pinheiro HT, Macena B, Francini-Filho R, Ferreira C, Albuquerque F, Alves Bezerra N et al. Fish biodiversity of Saint Peter and Saint Paul’s Archipelago, Mid-Atlantic Ridge, Brazil: new records and a species database. J Fish Biol. 2020; 97(4):1143–53. https://doi.org/10.1111/jfb.14484
Pinheiro HT, Rocha LA, Macieira RM, Carvalho-Filho A, Anderson AB, Bender MG et al. South-western Atlantic reef fishes: zoogeographical patterns and ecological drivers reveal a secondary biodiversity centre in the Atlantic Ocean. Divers Distrib. 2018a; 24(7):951–65. https://doi.org/10.1111/ddi.12729
Pinheiro HT, Rocha C, Rocha LA. Tosanoides aphrodite, a new species from mesophotic coral ecosystems of St. Paul’s Rocks, Mid Atlantic Ridge (Perciformes, Serranidae, Anthiadinae). ZooKeys. 2018b; 786:105–15. https://doi.org/10.3897/zookeys.786.27382
Quimbayo JP, Dias MS, Kulbicki M, Mendes TC, Lamb RW, Johnson AF et al. Determinants of reef fish assemblages in tropical Oceanic islands. Ecography. 2019; 42(1):77–87. https://doi.org/10.1111/ecog.03506
Quimbayo JP, Silva FC, Mendes TC, Ferrari DS, Danielski SL, Bender MG et al. Life-history traits, geographical range, and conservation aspects of reef fishes from the Atlantic and Eastern Pacific. Ecology. 2021; 102(5):e03298. https://doi.org/10.1002/ecy.3298
R Development Core Team. R: A language and environment for statistical computing. Version 4.3.2 [Internet]. Vienna, Austria; 2023. Available from: https://www.R-project.org/
Roberts TE, Keith SA, Rahbek C, Bridge TCL, Caley MJ, Baird AH. Testing biodiversity theory using species richness of reef-building corals across a depth gradient. Biol Lett. 2019; 15(10):20190493. http://dx.doi.org/10.1098/rsbl.2019.0493
Robinson BJO, Barnes DKA, Grange LJ, Morley SA. The extremes of disturbance reduce functional redundancy: Functional trait assessment of the shallow Antarctic benthos. Front Mar Sci. 2022; 8:797112. https://doi.org/10.3389/fmars.2021.797112
Rocha LA, Pinheiro HT, Shepherd B, Papastamatiou YP, Luiz OJ, Pyle RL et al. Mesophotic coral ecosystems are threatened and ecologically distinct from shallow water reefs. Science. 2018; 361(6399):281–84. https://doi.org/10.1126/science.aaq1614
Rosa MR, Alves AC, Medeiros DV, Coni EOC, Ferreira CM, Ferreira BP et al. Mesophotic reef fish assemblages of the remote St. Peter and St. Paul’s archipelago, Mid-Atlantic ridge, Brazil. Coral Reefs. 2016; 35(1):113–23. https://doi.org/10.1007/s00338-015-1368-x
Simon T, Pinheiro HT, Moura RL, Carvalho-Filho A, Rocha LA, Martins AS et al. Mesophotic fishes of the Abrolhos Shelf, the largest reef ecosystem in the South Atlantic. J Fish Biol. 2016; 89(1):990–1001. https://doi.org/10.1111/jfb.12967
Smallhorn-West PF, Bridge TCL, Munday PL, Jones GP. Depth distribution and abundance of a coral-associated reef fish: Roles of recruitment and post-recruitment processes. Coral Reefs. 2017; 36(1):157–66. https://doi.org/10.1007/s00338-016-1509-x
Souza AS, Soares RX, Amorim KDJ, Ribeiro RS, Molina WF, García-Jr J. First record of Diodon eydouxii Brisout de Barneville, 1846 (Osteichthyes: Diodontidae) in Saint Peter and Saint Paul archipelago, Brazil. Pan-Am J Aquat Sci. 2015; 10(2):179–81.
Vaske Jr. T, Lessa RP, Nobrega M, Montealegre-Quijano S, Marcante Santana F, Bezerra Jr. JL. A checklist of fishes from Saint Peter and Saint Paul archipelago, Brazil. J Appl Ichthyol. 2005; 21(1):75–79. https://doi.org/10.1111/j.1439-0426.2004.00600.x
Velasquez E, Bryan SE, Ekins M, Cook AG, Hurrey L, Firn J. Age and area predict patterns of species richness in pumice rafts contingent on oceanic climatic zone encountered. Ecol Evol. 2018; 8(10):5034–46. https://doi.org/10.1002/ece3.3980
Villéger S, Mason NWH, Mouillot D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology. 2008; 89(8):2290–301. https://doi.org/10.1890/07-1206.1
Whittaker RJ, Triantis KA, Ladle RJ. A general dynamic theory of oceanic island biogeography. Journal of Biogeography. 2008; 35, 977–94.
https://doi.org/10.1111/j.1365-2699.2008.01892
Williams J, Jordan A, Harasti D, Davies P, Ingleton T. Taking a deeper look: Quantifying the differences in fish assemblages between shallow and mesophotic temperate rocky reefs. PLoS ONE. 2019; 14(3):e0206778. https://doi.org/10.1371/journal.pone.0206778
Wilson SK, Burgess SC, Cheal AJ, Emslie M, Fisher R, Miller I et al. Habitat utilization by coral reef fish: Implications for specialists vs. generalists in a changing environment. J Anim Ecol. 2008; 77(2):220–28. https://doi.org/10.1111/j.1365-2656.2007.01341.x
Wood R. Biodiversity and the history of reefs. Geol J. 2001; 36(3–4):251–63. https://doi.org/10.1002/gj.898
Yeager LA, Deith MCM, McPherson JM, Williams ID, Baum JK. Scale dependence of environmental controls on the functional diversity of coral reef fish communities. Glob Ecol Biogeogr. 2017; 26(10):1177–89. https://doi.org/10.1111/geb.12628
Authors
![]() Julia Marx1,2
Julia Marx1,2 ![]() ,
, ![]() Caio R. Pimentel3,
Caio R. Pimentel3, ![]() Luiz A. Rocha4 and
Luiz A. Rocha4 and ![]() Hudson T. Pinheiro5
Hudson T. Pinheiro5
[1] Programa de Pós-Graduação em Zoologia, Instituto de Biociências, Universidade de São Paulo, 05508-900, São Paulo, SP, Brazil. (JM) jmarxsouza@gmail.com (corresponding author).
[2] Centro de Biologia Marinha (CEBIMar), Universidade de São Paulo, 11612-109, São Sebastião, SP, Brazil. (HTP) htpinheiro@usp.br.
[3] Departamento de Oceanografia e Ecologia, Universidade Federal de Espírito Santo, Vitória, ES, Brazil. (CRP) caiopimentelr@gmail.com.
[4] California Academy of Sciences, San Francisco, CA 94118, USA. (LAR) lrocha@calacademy.org.
Authors’ Contribution 

Julia Marx: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Caio R. Pimentel: Funding acquisition, Investigation, Writing-review and editing.
Luiz A. Rocha: Data curation, Funding acquisition, Investigation, Resources, Writing-review and editing.
Hudson T. Pinheiro: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
Data availability statement
The data supporting the findings of this study are available from the corresponding author, upon reasonable request.
AI statement
The authors did not use any AI-assisted technologies in the creation of this manuscript or its figures.
Funding
We thank Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; 2019/24215–2) and the California Academy of Sciences through the Hope For Reefs Initiative for funding. JM and HTP received FAPESP fellowships (2022/08684–5 and 2021/07039–6, respectively). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) – finance code 001. CRP also thanks the Fundação Estadual de Amparo à Pesquisa do Estado do Espírito Santo for the PhD scholarship (54/2018) and CAPES for the PDSE fellowship (88881.362262/2019–01). We are grateful to NGO Voz da Natureza for supporting the project, and to Fundação Grupo Boticário de Proteção à Natureza for funding.
Supplementary Material
Supplementary material S1
Supplementary material S2
Supplementary material S3
Supplementary material S4
Supplementary material S5
Supplementary material S6
Supplementary material S7
How to cite this article
Marx J, Pimentel CR, Rocha LA, Pinheiro HT. Functional diversity and ecological patterns of reef fish assemblages across depth gradients in oceanic islands of the Brazilian Province. Neotrop Ichthyol. 2026; 24(1):e250035. https://doi.org/10.1590/1982-0224-2025-0035
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 10, 2025
Accepted December 10, 2025
![]() Submitted March 10, 2025
Submitted March 10, 2025
![]() Epub April 27, 2026
Epub April 27, 2026