![]() Gastón Aguilera1
Gastón Aguilera1 ![]() ,
, ![]() Guillermo E. Terán1,
Guillermo E. Terán1, ![]() Miguel Ángel Cortés Hernández1,2,
Miguel Ángel Cortés Hernández1,2, ![]() Axel M. Katz3,
Axel M. Katz3, ![]() Valter M. Azevedo-Santos4,5,6,
Valter M. Azevedo-Santos4,5,6, ![]() Mauricio Benitez7 and
Mauricio Benitez7 and ![]() Marcos Mirande1
Marcos Mirande1
PDF: Download Here | Supplementary: S1 S2 | Cite this article
Associate Editor: ![]() Carlos DoNascimiento
Carlos DoNascimiento
Editor-in-chief: ![]() José Birindelli
José Birindelli
Abstract
Phenacorhamdia comprende 15 especies distribuidas en las principales cuencas hidrográficas de América del Sur, siendo P. tenebrosa la única especie registrada en Argentina. Tras el examen de ejemplares de Phenacorhamdia provenientes de la cuenca alta del río Bermejo, cuenca del río Paraná, en el noroeste de Argentina, concluimos que pertenecen a una nueva especie, la cual se describe en el presente trabajo. La nueva especie se distingue de los demás miembros del género por una combinación de caracteres no exclusivos que incluyen: barbilla maxilar sobrepasando la punta distal de la aleta pectoral adosada; la coloración general del cuerpo de tono marrón claro; la presencia de dientes cónicos; 41–42 vértebras; iii–iv,7–9 radios de la aleta anal, entre otras características. El análisis morfológico realizado muestra una clara distinción de la nueva especie de sus congéneres, y la gran distancia genética entre P. bermejo y P. tenebrosa basada en el gen COI, proveen soporte adicional para el reconocimiento de la nueva especie aquí descrita
Palabras clave: Análisis morfológico,Bagres, Delimitación de especies, Divergencia genética, Heptapterinae.
Introduction
Heptapteridae is one of the most species-rich families within Siluriformes and is widely distributed throughout the Neotropical region, from Mexico to southern Argentina (Bockmann, Guazelli, 2003; Bockmann, Slobodian, 2018; Thomas, Sabaj, 2020). According to the last phylogenetic hypothesis (i.e., Faustino-Fuster et al., 2021; Silva et al., 2021; Slobodian, Bockmann, 2025) the family comprises two major clades, the subfamilies Rhamdiinae and Heptapterinae supported, respectively, by five synapomorphies, and 14 discrete and two continuous characters proposed by de Pinna (1993), Lundberg et al. (1991), Bockmann (1998), and Bockmann, Miquelarena (2008); see Slobodian, Bockmann (2025) for a complete description of synapomorphies and characters. Heptapterinae includes the tribes Brachyglaniini and Heptapterini, diagnosed by seven and 14 synapomorphies, respectively (Slobodian, Bockmann, 2025). Within Heptapterini, Phenacorhamdia Dahl, 1961 was recovered as monophyletic (although the type species, P. macarenensis, has never been sampled), and as the sister group of Magdalenichthys DoNascimiento, Conde-Saldaña, Albornoz-Garzón, Villa-Navarro in DoNascimiento et al. (2025), with Pariolius Cope, 1872 as sister to Phenacorhamdia and Magdalenichthys; those results partially align with the phylogenetic hypothesis proposed by Solabodian, Bockmann (2025), who recovered a clade formed by Phenacorhamdia as sister to ‘Imparfinis’ microps Eigenmann & Fisher, 1916 and an undescribed, incertae sedis species, that clade being sister to Pariolius.
Phenacorhamdia is a genus composed of small catfishes distinguished from other Heptapteridae by a prognathous mouth, eyes without free orbital margin, the second pore of the supraorbital canal (s2+i2) located far posterior to the anterior nostril, the epioccipital process prominent, and the neural and haemal spines of the posterior vertebrae inclined 30º or less (Britski, 1993; Bockmann, 1998:367). Additionally, members of the genus can be recognized by a combination of non-unique characters that includes a moderate to very elongate body; small eyes, located dorsally; maxillary barbel usually reaching to the last third of the pectoral fin or slightly surpassing its posterior border; the first pectoral-fin ray usually longer than the second one, mostly flexible (except its basal third) and prolonged as a short filament; distal margin of pectoral, dorsal, pelvic, and anal fins convex; pectoral fin usually with 6–9 branched rays; first dorsal-fin ray (spinelet) absent; origin of the pelvic fin at a vertical through the middle of dorsal-fin base or slightly anterior; adipose fin rectangular and moderately long, not confluent with the caudal fin; anal fin short to moderately long, bearing 12–18 rays; caudal fin deeply forked with the ventral lobe slightly to markedly longer than the dorsal lobe, usually with seven branched rays on the dorsal lobe and eight branched rays on the ventral lobe; lateral line continuous to the base of the caudal-fin rays; head and body with uniform brown coloration; anterior and posterior fontanels open; mandibular adductor muscles restricted to the facial region; posterior supraoccipital process short; and usually 7–8 branchiostegal rays (Bockmann, 1998; Bockmann, Slobodian, 2018). Phenacorhamdia shares with its closest relatives, Magdalenichthys and Pariolius, the anal-fin origin usually situated anteriorly to a vertical through the adipose-fin origin (although in P. macarenensis they are located on the same vertical); the anteriorly positioned pelvic-fin origin, which commonly is situated anteriorly to a vertical through the dorsal-fin origin; a usually prognathous mouth (isognathous in some species of the clade); and a usually uniformly pigmented body, with no conspicuous dark bars on the dorsum (although lightcolored marks may be present).
Phenacorhamdia currently is composed of 15 valid species, distributed in various watersheds in South America (Castro, Wozniacki, 2022; Silva et al., 2022). From the Orinoco River basin, P. anisura (Mees, 1987), P. macarenensis Dahl, 1961, P. provenzanoi DoNascimiento & Milani, 2008 and P. taphorni DoNascimiento & Milani, 2008 are recorded. In the Amazon River basin, P. boliviana (Pearson, 1924) is found in the Beni River; P. nigrolineata Zarske, 1998 occurs in the Branco, Ucayali, Solimões, Purus, Madeira, and Tapajós Rivers; and P. somnians (Mees, 1974) in the das Mortes River. In turn, P. cuspidata Silva, Castro, Ohara & Oliveira, 2024 is restricted to the upper Tapajós River basin; and P. suia Silva, Ochoa & Castro, 2022 to the Xingu River basin. Regarding the river basins in northern South America draining into the Atlantic Ocean, P. cabocla Rocha, Ramos & Ramos, 2018 is found in the upper Parnaíba River basin and P. tenuis (Mees, 1986) in the Marowijne River basin. Four species are recognized in the Paraná River basin: P. hoehnei (Miranda Ribeiro, 1914), in the Taquari River, a tributary of the Paraguay River; P. roxoi Silva, 2020, in the upper Paranapanema River basin; P. unifasciata Britski, 1993, endemic to the São Bartolomeu River; and P. tenebrosa (Schubart, 1964), originally described from the upper Paraná River basin, but also recorded in the lower reaches of the basin in Argentina (Giorgis et al., 2012).
After the examination of specimens from the upper Bermejo River, Paraná River basin, we conclude that they belong to a new species which is described herein, corresponding to the sixteenth species of the genus. In addition, we provide a brief discussion on its phylogenetic relationships within Phenacorhamdia, based on the cytochrome oxidase 1 (COI) and cytochrome b (Cytb) genes.
Material and methods
The specimens were collected by electrofishing and euthanized by immersion in tricaine methanesulfonate (MS222), and fixed following standard methods. In some fresh individuals, a small tissue sample was taken and immediately preserved in absolute ethanol for genetic analysis. The tissue aliquots were deposited in the Ichthyological Tissue Collection of the Fundación Miguel Lillo (CIT-FML). Institution abbreviations follow Sabaj (2025).
Representative individuals werecleared and stained (c&s) following Taylor, Van Dyke (1985). Measurements were taken following DoNascimiento, Milani (2008). Counts include the number of fin rays, branchiostegal rays, ribs, and vertebrae, as well as the number and position of support elements for the dorsal and anal fins, and number of procurrent rays of the caudal fin. Total vertebrae count includes the anterior five, associated with the Weberian complex (Lundberg, McDade, 1986) counted as separate, the free vertebrae, and the compound caudal centrum (PU1+U1) counted as one element (Lundberg, Baskin, 1969). An asterisk indicates counts from the holotype. Morphological data of Phenacorhamdia species, including fin rays, vertebral and rib counts and body measurements were obtained from original species descriptions when available, and from DoNascimento, Milani (2008) for Phenacorhamdia anisura, and from Azevedo-Santos et al. (2025) for P. tenebrosa. Additionally, X-ray images were used for assess osteological data of Phenacorhamdia boliviana and P. somnians, available at the California Academy of Sciences, San Francisco (CAS) Ichthyology Primary Types Imagebase (https://researcharchive.calacademy.org/research/ichthyology/types/Index.asp), and at the Natural History Museum, London (NHMUK) online database (https://data.nhm.ac.uk/dataset/56e711e6-c847-4f99-915a-6894bb5c5dea/resource/05ff2255-c38a-40c9-b657-4ccb55ab2feb/record/2536084), respectively.
Morphological analysis. An exploratory PCA was conducted in R Stats package v. 4.4.2 (R Development Core Team, 2024), using the original measurements without transformations or size corrections, excluding standard length from the analyses. The morphological data matrix analyzed comprised eight species, including the new species herein described and P. boliviana (3 specimens), P. cabocla (5), P. hoehnei (4), P. roxoi (1), P. sommians (2), P. unifasciata (7)and P. tenebrosa (3), and 24 morphological measurements. Missing data were imputed using the missRanger package v. 2.6.0 (Mayer, 2024). All these analyses were performed in R Studio (R Development Core Team, 2024).
DNA extraction. Total genomic DNA was extracted from ethanol-preserved muscle tissue, using the Qiagen DNeasy kit. PCR amplifications were carried out in 30 µL reactions using 0.2 µL Taq (Genbiotech). Mitochondrial gene Cytochrome oxidase subunit I (COI) was amplified by Polymerase Chain Reaction (PCR) using a cocktail of primers VF2_t1, FishF2_t1, and FishR2_t1, Fr1d_t1 (Ivanova et al., 2007); and following conditions implemented by Ward et al. (2005). Cytochrome B (Cytb) was amplified using the primers GLUDGL (Palumbi, 1996) and H16460 (Perdices et al., 2002), following the PCR protocol proposed by Ríos et al. (2017). PCR-amplified products were visualized on 1% agarose gel and cleaned using AccuPrep PCR Purification Kit. Products were sequenced by the Korean company Macrogen (http://www.macrogen.com) and all samples were sequenced in both directions to check for potential errors. Chromatograms obtained from the automated sequencer were processed and edited using GENEIOUS PRO v. 4.8.5 (www.geneious.com). Genbank accession numbers of the sequences used in the phylogenetic analyses are provided in Tab. S1. The sequences generated for the new species herein described have been deposited in GenBank under the accessionnumbers PX978871 (COI) and PX989894 (CYTB).
Phylogenetic analysis. Phylogenetic analyses were performed based on a concatenated dataset including 55 sequences of the cytochrome oxidase subunit 1 gene (COX1) and 22 sequences of the cytochrome b gene (CYTB) to have some estimation of the relationships of the new species within the family. The dataset is composed of 1,494 sites (577 parsimony informative) and 55 terminal specimens obtained from Genbank (Tab. S1). Analyses were rooted on Pimelodus maculatus Lacepède, 1803. These analyses, however, should be taken as provisional, given that no data from most species of the genus are available and its main aim is to assess the generic assignment of the new species. Analyses were performed both under parsimony in TNT (Goloboff, Morales, 2023) and maximum likelihood in RAxML (Stamatakis et al., 2014). Parsimony analyses were done under equal weighting and implied weighting (Goloboff, 1993). We explored a broad range of the concavity constant K, which determines how strong the analysis downweights the characters according to their homoplasy (Goloboff, 1993). Results herein presented and calculations of support correspond to K = 10 that, considering the size of the dataset, produces a mild weight against homoplasy. Searches were done from Wagner trees, whose branches were swapped with complete cycles of TBR (i.e., connecting each terminal taxon and internal clade on every position of the tree, using all the possible rootings in the case of prunned branches). After that, trees were refined with sectorial searches and tree fusing (Goloboff, 1999). Clade supports were calculated through symmetric resampling and expressed as values of GC (Goloboff et al., 2003).
Maximum likelihood analysis was carried out with defaults parameters of RAxML (Stamatakis, 2014) under the GTR+Gamma model. Analyses started from 10 random plus 10 parsimony trees that were swapped by 1000 bootstrap cycles in which fast SPR rounds were performed (i.e., computing the likelihoods according to the branch lengths of the initial tree). As default in RaxML, during the SPR cycles, the distance at which each prunned taxon or clade (its SPR radius) is connected was set to “auto” (Togkousidis et al., 2023). Support estimation was done under Bootstrap, using convergence as stopping criterion (Pattengale et al., 2010).
Results
Phenacorhamdia bermejo, new species
urn:lsid:zoobank.org:act:D3A5ABE5-2E2F-431C-9321-03887FC06B62
(Figs. 1–2; Tab. 1)
Holotype. CI-FML 8284, 42.6 mm SL, Argentina, Ledesma department of Jujuy Province, San Francisco River at Yuto, 23°40’02.0”S 64°28’44.6”W, 345 m asl, 10 Oct 2024, G. Aguilera, G. Terán, M. A. Cortés Hernández & A. López-Méndez.
Paratypes. Argentina. CI-FML 8289, 2 c&s, 41.3–47.7 mm SL, collected with the holotype. CI-FML 7747, 1, 49.2 mm SL, confluence between San Francisco and San Lorenzo rivers, Ledesma department of Jujuy Province, 23°50’27.9”S 64°37’38.1”W, 16 Aug 2019, G. Aguilera, G. Terán & M. Casalinuovo. CI-FML 7790, 1, 45.0 mm SL, San Francisco River under bridge of Route N. 1, Ledesma department of Jujuy Province, 23°42’2.4”S 64°32’08.1”W, 350 m a.s.l., 15 Aug 2019, G. Aguilera, G. Terán & M. Casalinuovo. CI-FML 8285, 9, 1 c&s, 40.5–51.0 mm SL, confluence between San Francisco and San Lorenzo rivers, Ledesma department of Jujuy Province, 23°50’27.9”S 64°37’38.1”W, 365 m a.s.l., 20 Dec 2016, G. Aguilera, G. Terán & D. Delgado. CI-FML 8286, 2, 38.0–38.7 mm SL, confluence between San Francisco and San Lorenzo rivers, Ledesma department of Jujuy Province, 23°50’27.9”S 64°37’38.1”W, 365 m a.s.l., 10 Dec 2019, G. Aguilera, G. Terán & M. Casalinuovo. CI-FML 8287, 1, 51.1 mm SL, Bermejo River, Orán department of Salta Province, 23°11’30.4”S 64°11’02.1”W, 300 m a.s.l., Sep 2016, M. Casalinuovo. CI-FML 8288, 4, 32.4–45.6 mm SL, San Lorenzo River, Ledesma department of Jujuy Province, 23°49’46.4”S 64°39’09.3”W, 370 m a.s.l., Dec 2022, G. Aguilera, G. Terán, A. López-Méndez & H. Bugeau. MACN-Ict 13513, 2, 41.4–43.6 mm SL, confluence between Sauzalito and San Francisco rivers, Ledesma department of Jujuy Province, 23°42’29.7”S 64°31’51.1”W, 350 m a.s.l., 7 Dec 2019, M. Casalinuovo.
FIGURE 1| Lateral, dorsal, and ventral views of holotype of Phenacorhamdia bermejo, CI-FML 8284, 42.6 mm SL
Diagnosis. Phenacorhamdia bermejo is distinguishedfrom the other congeners, except P. anisura, P. hoehnei, P. macarenensis, P. nigrolineata, P. tenebrosa,by having fewer number of total vertebrae (41–42 vs. 39 in P. taphorni, 43 in P. boliviana, 43–45 in P. suia,44 in P. cabocla,48–52 in P. cuspidata,47–48 in P. provenzanoi, 45–46 in P. roxoi,45 in P. somnians, 46 in P. unifasciata, and 53–55 in P. tenuis). Additionally, it can be distinguished by having a longer maxillary barbel (29.8–38.0% of SL vs. 26.2–28.3 in P. anisura,12.5–23.3 in P. cabocla, 10.5–18.7 in P. cuspidata, 19.3–23.0 in P. hoehnei,21.0–27.8 in P. provenzanoi, 15.9–22.1 in P. roxoi, 19.6–21.1 in P. somnians, 16.5–21.7 in P. suia, 22.6–27.4 in P. tenebrosa and 14.6–18.8 in P. unifasciata); longer outer mentonian barbel (16.1–19.9% of SL vs. 6.2–13.4 in P. cuspidata, 11.0–15.0 in P. roxoi, 11.8–14.2 in P. suia, and 9.6–15.1 in P. unifasciata); longer inner mentonian barbel (12.8–16.2% of SL vs. 5.8–10.4 in P. cuspidata, 9.9–12.4 in P. roxoi, 8.7–14.7 in P. suia, and 6.9–11.2 in P. unifasciata); conical teeth in the jaws (vs. multicuspidate teeth in P. cuspidata, P. suia,and P. taphorni); 11–12 anal-fin rays (vs. 10 in P. somnians, 16 in P. tenuis, and 16–18 in P. provenzanoi); 15 branched caudal-fin rays (vs. 14 in P. unifasciata and 16–17 in P. tenuis); and longer head (20.9–24.5% of SL vs. 15.1–19.0 in P. provenzanoi and 16.5–20.7 in P. suia). Phenacorhamdia bermejo is distinguishedfrom P. anisura, P. hoehnei, P. nigrolineata, P. provenzanoi,and P. unifasciata by having longer upper caudal-fin lobe (24.2–28.6% of SL vs. 21.4–23.7 in P. anisura, 20.4–24.0 in P. hoehnei, 17.6–23.1 in P. nigrolineata, and 14.0–18.2 in P. provenzanoi, and 15.0–23.4 in P. unifasciata). It is distinguished from P. hoehnei, P. macarenensis and P. nigrolineata by having more branched pectoral-fin rays (7–8 vs. 5–6 in P. hoehnei, 6 in P. macarenensis and P. nigrolineata). From P. hoehnei, P. taphorni, and P. tenebrosa by having longer lower caudal-fin lobe (29.0–33.8% of SL vs. 24.0–28.0 in P. hoehnei, 21.5–28.6 in P. taphorni, and 21.7–24.2 in P. tenebrosa). From P. nigrolineata and P. tenebrosa by havinglonger caudal peduncle (20.1–23.4% of SL vs. 12.3–18.6% in P. nigrolineata and 17.5–17.9% in P. tenebrosa). From P. hoehnei and P. nigrolineata by the mouth gape (30.8–36.6% of HL vs. 14.5–20.1 in P. hoehnei and 37.2–53.7 in P. nigrolineata). From P. nigrolineata and P. provenzanoi by the shorter anal-fin base (11.4–14.8% of SL vs. 15.1–22.1 in P. nigrolineata and 16.2–19.4 in P. provenzanoi). From P. nigrolineata and P. roxoi by having deeper body (11.5–17.0% of SL vs. 6.8–10.6 in P. nigrolineata and 8.2–10.2 in P. roxoi). From P. anisura and P. provenzanoi by having deeper caudal peduncle (7.0–8.6% of SL vs. 5.7–6.9 in P. anisura and 4.3–5.1 in P. provenzanoi).
The new species is also distinguished from P. boliviana by the insertion of dorsal-fin pterygiophore behind vertebra 11 (vs. 14); from P. cabocla by having fewer upper caudal-fin lobe branched rays (7, one with 6, vs. 8–9), and longer snout length (36.0–45.7% of HL vs. 30.0–35.9); from P. suia by the shorter distance between dorsal fin to adipose fin (17.9–21.4% of SL vs. 25.5–34.9); from P. macarenensis by having more branched anal-fin rays (7–9 vs. 6); from P. tenebrosa by the insertion of dorsal-fin first basal radial between neural spine of 11th−12th vertebrae (vs. 9th−10th), and last basal radial between neural spine of 16th−17th vertebrae (vs.13th−14th or 14th−15th). Phenacorhamdia bermejo is additionally distinguished from P. provenzanoi and P. tenuis by the greater number of gill rakers on first branchial arch (7 vs. 4–5 in P. provenzanoi and 2 in P. tenuis). It is also distinguished from the remaining species of the genus, except P. macarenensis, P. nigrolineata, P. provenzanoi, P. roxoi, P. taphorni, P. tenuis and P. unifasciata,by the maxillary barbel surpassing the distal tip of the adpressed pectoral fin and reaching up to the dorsal-fin base in some specimens (vs. reaching up to the distal tip of the adpressed pectoral fin in P. anisura, P. boliviana and P. tenebrosa; to the pectoral-fin base in P. cabocla, P. hoehnei, and P. suia; to the end of the opercle in P. somnians; and not reaching to pectoral-fin origin in P. cuspidate, Fig. S2).
Description. Morphometric data are summarized in Tab. 1. Body elongated, elliptical in cross section through dorsal-fin origin. Greater body depth at dorsal-fin origin; body becoming compressed towards caudal peduncle. Dorsal profile of body, from snout tip to dorsal-fin origin, convex (except for concave area between anterior margin of posterior nostril to posterior margin of eye). Straight or slightly convex from end of dorsal fin to adipose-fin origin, straight and descending along adipose-fin base, and straight from that point to caudal-fin origin. Ventral profile of body, from snout tip to pelvic-fin origin, convex; thence almost straight to anal-fin end; and concave or almost straight from that point to caudal-fin origin.
TABLE 1 | Morphometric measurement of Phenacorhamdia bermejo. N = 21 specimens;SD = Standard deviation.
| Holotype | Range | Mean | SD |
Standard length (mm) | 42.6 | 32.4–52.3 | – | – |
Percents of standard length | ||||
Body depth | 13.8 | 11.5–17.0 | 13.6 | 1.4 |
Cleithral width | 14.4 | 13.0–16.7 | 14.9 | 0.9 |
Predorsal length | 41.6 | 37.9–42.0 | 40.4 | 1.2 |
Dorsal-fin base | 10.8 | 9.5–12.6 | 11.1 | 0.7 |
Dorsal fin to adipose fin | 20.3 | 17.9–21.4 | 19.5 | 0.9 |
Preadipose length | 72.4 | 67.3–72.4 | 70.1 | 1.3 |
Adipose-fin length | 18.1 | 16.7–20.3 | 18.5 | 1.1 |
Prepelvic length | 39.7 | 38.0–43.9 | 40.4 | 1.2 |
Preanal length | 66.4 | 63.6–69.6 | 65.8 | 1.4 |
Anal-fin base | 12.5 | 11.4–14.8 | 12.8 | 0.9 |
Caudal-peduncle length | 21.8 | 20.1–23.4 | 21.5 | 1.0 |
Caudal-peduncle depth | 7.0 | 7.0–8.6 | 7.6 | 0.4 |
Upper caudal lobe length | 26.2 | 24.2–28.6 | 26.5 | 1.3 |
Lower caudal lobe length | 29.7 | 29.0–33.8 | 31.3 | 1.5 |
Head length | 22.5 | 20.9–24.5 | 22.5 | 1.0 |
Maxillary-barbel length | 35.0 | 29.8–38.0 | 34.2 | 2.4 |
Outer mental-barbel length | 18.2 | 16.1–19.9 | 18.0 | 1.0 |
Inner mental-barbel length | 14.9 | 12.8–16.2 | 14.5 | 1.2 |
Percents of head length | ||||
Head width | 58.9 | 56.4–76.5 | 66.0 | 4.7 |
Head depth | 44.8 | 42.9–55.9 | 49.4 | 4.0 |
Mouth gape | 30.8 | 30.8–36.6 | 33.5 | 1.7 |
Snout length | 40.3 | 36.0–45.7 | 39.5 | 2.3 |
Fleshy interorbital | 28.2 | 22.2–29.2 | 25.6 | 2.1 |
Eye diameter | 8.6 | 8.6–11.3 | 9.8 | 0.8 |
Head depressed, deepest and widest at occipital region. Snout short, anterior margin rounded in dorsal view. Mouth terminal or slightly prognathous; lower jaw slightly longer than upper jaw. Premaxillary and dentary teeth conical, arranged in rectangular patch with four to eight irregular rows. Anterior nares tubular, smaller than posterior nares, situated closer to snout tip than to posterior nares. Posterior nares ovoid, closer to eye than to anterior nares, with fleshy anterior flap covering nostril when retracted. Eyes small, dorsally positioned and covered with skin, located closer to snout tip than to posterior opercular margin. Maxillary barbel long, surpassing distal tip of pectoral fin when adpressed, reaching pelvic-fin origin or dorsal-fin origin in some specimens. Mental barbels (inner and outer) aligned in straight line; outer barbel longer, reaching pectoral-fin origin; inner barbel extending just anterior to vertical through opercular margin. Branchiostegal membranes free, anteriorly united to isthmus, bearing seven rays (3 c&s). Gill rakers seven, along anterior margin of first ceratobranchial.
Dorsal fin with i,6(21)* rays; its origin anterior to midbody. First basal radial between neural spines of vertebrae 11–12; last between vertebrae 16–17(3 c&s). Long supraneural, anterior to first dorsal-fin pterygiophore, spanning two vertebrae in length. Adipose fin moderate in length (16.7–20.3% of SL), not confluent with caudal fin and far from reaching first caudal-fin ray. Pectoral fin with i,7(18)* or i,8(3) rays; unbranched ray prolonged into short filament, extending beyond distal fin margin. Pelvic fin with i,5 rays and rounded margin; insertion anterior to or at vertical through dorsal-fin origin. Anal fin with iii(8) or iv(12)* unbranched rays and 7(1), 8(17)* or 9(2) branched rays, in combinations: iv,7(1), iii,8(8), iv,8(9)*, iv,9(2). Origin posterior to midbody; first basal radial between hemal spine of vertebrae 22–23, and last distal radial between vertebrae 28–29.
Caudal fin deeply forked, with i,7+8,i (20)* principal rays (one specimen with i,6 rays on dorsal lobe). Lobes elongate and pointed; ventral lobe longer (29.0–33.8% SL) than dorsal (24.2−28.6% SL). Procurrent rays 9–10 in dorsal lobe, and 8–9 on ventral lobe. Caudal skeleton of ventral lobe with hypurals 1+2 fused into single plate; parahypural closely adjacent but separated from it. Dorsal lobe with hypural 3+4 fused into single plate; hypural 5 ossified and free; uroneural ossified, fused to complex centrum (preural centrum 1+ural centrum 1). Precaudal vertebrae 15(2) and 16(1), caudal vertebrae 25(1), 26(1), 27(1), 41(2) or 42(1) total vertebrae. First hemal spine on 16th (2) or 17th (1) vertebra. Bifid hemal spine on vertebra 24th (3) to 29th (2) or 30th (1). Pleural ribs 8(1) or 9(2).
Supraorbital sensory canal with 4 pores: s1, opening medially adjacent to anterior nares, s2 fused to infraorbital pore i2 (s2+i2) and opening approximately half length between anterior and posterior nares, s3 on internal posterior margin of posterior nare, s4 and s5 absent, supraorbital canals connected medially by epiphysial membranous canal, above anterior fontanel, without superficial pore (s6), and s8 at the posterior surface of frontal. Infraorbital canal with six pores: i1, located at vertical through posterior margin of anterior nares, near the groove of maxillary barbel, i2 fused to s2, i3 located half distance between anterior and posterior nares, into maxillary groove, i4 located just anterior to vertical through anterior eye margin, i5 behind eye in lateral margin of maxillary groove, and i6 placed behind eye. Preoperculomandibular canal with 11 pores, pm 1 displaced just from midventral line at dentary, pm2 and pm3 on posterior margin of ventral lip, pm4 anterior to outer mentonian barbel insertion, pm5 at outer mentonian barbel level, pm6 at vertical through anterior margin of posterior nares, pm7 at vertical through half distance between posterior nares and anterior eye margin, pm8 at vertical through eye, pm9 and pm10 separated by a short stretch from principal preoperculomandibular canal, and pm11 in preopercle at eye level.
Coloration in alcohol. Coloration after fixation: body background light beige dorsally and laterally, almost white ventrally. The pectoral, pelvic, anal, and dorsal fins remain hyaline, while the pigmentation pattern in the adipose and caudal fins persists, although it becomes less pronounced. In fixed specimens, the small indentation of the lateral line system appears as a thin, grayish line.
Coloration in life. Coloration in live specimens (Fig. 2): the overall pigmentation consists of uniform, minute mottling, producing a homogeneous reddish-brown hue across the body. The head is slightly darker than the rest of the body, while pigmentation on the caudal peduncle is concentrated midlaterally, gradually fading towards the dorsal and ventral regions. The pectoral, pelvic, and anal fins are hyaline, with the dorsal fin similarly transparent but displaying chromatophores concentrated along the rays, which becoming less conspicuous towards the distal tips. The adipose fin exhibits chromatophores concentrated at the base, extending to about middle depth of the fin, with a gradual fading towards the distal margin. The caudal fin shows denser pigmentation near the base, which progressively fades towards the distal margin. Head, in ventral view, with chromatophores restricted to the preoperculomandibular laterosensory canal. Clear absence of pigmentation from the origin of the pectoral fin to the anal-fin origin, giving it a silvery appearance.
FIGURE 2| Live specimen of Phenacorhamdia bermejo,CI-FML 8289, 47.7 mm SL, in dorsal, lateral, and ventral views. Photo by Alejandro López-Méndez.
Geographical distribution. Phenacorhamdia bermejo inhabits the upper Bermejo River basin, where it was found at four localities, three on the San Francisco River basin in Jujuy Province and one at the Bermejo River in Salta (Fig. 3).
FIGURE 3| Distributional map of Phenacorhamdia bermejo. Red diamond indicates the type locality, and green dots indicate additional localities.
Conservation status. Phenacorhamdia bermejo is exposed to some environmental pressures within its range, including agricultural runoff, deforestation, and industrial pollution affecting freshwater ecosystems. However, no major threats have been identified that would currently place the species at risk across its distribution. The species occur in relatively well-preserved habitats and does not meet the criteria for any threat category. Following the IUCN Red List categories and criteria (IUCN, 2024), P. bermejo is best assessed as Least Concern (LC).
Etymology. The specific epithet “bermejo” is a Spanish word meaning reddish in English and refers to both the coloration of live specimens which present a reddish-brown hue across the body, and to the name of the river basin where the specimens of Phenacorhamdia bermejo were collected. A noun in apposition.
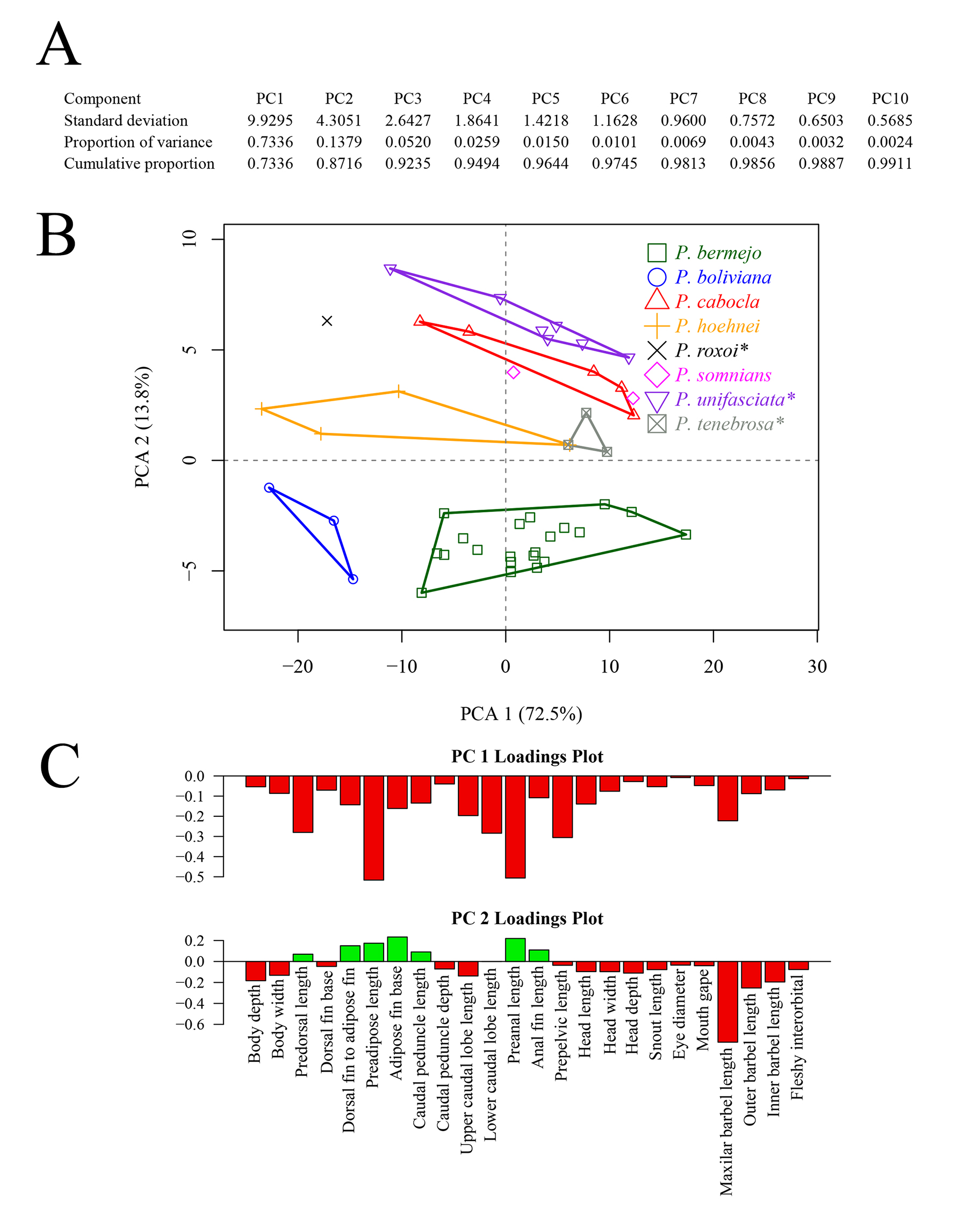
Morphological analysis. The PCA revealed clear morphological differentiation among the examined species of Phenacorhamdia. The first two principal components accounted for 86.3% of the total variation (PC1: 72.5%; PC2: 13.8%). Along PC1, P. bermejo was distinctly separated from all other species, indicating that morphological proportions such as preadipose length, preanal length, prepelvic length, caudal fin lower lobe length, predorsal length and maxillary barbel length allow clear distinction from other congeners (Fig. 4). In contrast, P. cabocla, P. somnians, P. hoehnei, and P. tenebrosa partially overlapped, reflecting shared morphometric characteristics while still maintaining species-level distinctions. Overall, the PCA supports the morphological distinctiveness of the new species, P. bermejo, within the genus.
FIGURE 4| Results of the principal component analysis. A. Importance of each component; B. Biplot of first two principal components, with the proportion of explained variance shown in parenthesis; C. Loading of each variable on the principal components.
Phylogenetic analysis. Both Parsimony and Maximum Likelihood analyses recovered the new species nested with other species of Phenacorhamdia, which formed a well-supported monophyletic group (ML: Freq = 93%; P: GC = 90), supporting the inclusion of the new species in this genus (Fig. 5). In both analyses, Phenacorhamdia was recovered as the sister group to the recently described genus Magdalenichthys. Together, these genera form a clade sister to Mastiglanis, and this larger clade is, in turn, sister to Pariolius. Within Phenacorhamdia twoclades were recovered, one with P. boliviana and P. somnians, and the other one including P. bermejo as sister to the clade including P. unifasciata, P. roxoi and P. tenebrosa. The mean genetic distances based on the COI marker indicated a divergence of 8.8% between P. bermejo and P. tenebrosa, both occurring in the Paraná River basin (Tab. 2). Moreover, P. bermejo exhibited genetic divergences exceeding 6% from P. unifasciata and 13% from P. boliviana, reinforcing the genetic distinctiveness of the new species.
FIGURE 5| Phylogenetic analyses performed under parsimony (A) and maximum likelihood (B).
TABLE 2 | Genetic distances interspecific and intraspecific of Phenacorhamdia under K2 substitution model. Values are represented as percentages.
| 1 | 2 | 3 | 4 | Intraspecific distance |
1. Phenacorhamdia bermejo |
|
|
|
| – |
2. Phenacorhamdia unifasciata | 6.2 |
|
|
| 0 |
3. Phenacorhamdia tenebrosa | 8.8 | 6.5 |
|
| 1.4 |
4. Phenacorhamdia boliviana | 13.5 | 14.2 | 14.9 |
| – |
Discussion
Silva et al. (2021), Faustino-Fuster et al. (2021), Slobodian, Bockmann (2025), and DoNascimiento et al. (2025) corroborated the monophyly of Heptapteridae, and recovered two major clades within it, the subfamilies Rhamdiinae and Heptapterinae, as in the present analyses. The Rhamdiinae includes Brachyrhamdia Myers, 1927, Goeldiella Eigenmann & Norris, 1900, Pimelodella Eigenmann & Eigenmann, 1888, Rhamdella Eigenmann & Eigenmann, 1888 and Rhamdia Bleeker, 1858, and a non-circumscribed genus that includes the species of the group P. leptosoma sensu Slobodian, Pastana (2018) (see Slobodian, Bockmann, 2025). For its part, Heptapterinae is divided in two tribes (Brachyglaniini and Heptapterini), the latter composed of Acentronichthys Eigenmann & Eigenmann, 1916, Cetopsorhamdia Eigenmann & Fisher, 1916, Chasmocranus Eigenmann, 1912, Heptapterus Bleeker, 1858, Horiomyzon Miranda Ribeiro, 1918, Imparfinis Eigenmann & Norris, 1900, Magdalenichthys, Mastiglanis Bockmann, 1994, Nannoglanis Boulenger, 1887, Nemuroglanis Eigenmann & Eigenmann, 1889, Pariolius Cope, 1872, Phenacorhamdia Dahl, 1961, Rhamdioglanis Ihering, 1907, Rhamdiopsis Haseman, 1911, Taunayia Miranda Ribeiro, 1918, and a few clades treated as undescribed genera (see Bockmann, Slobodian 2018; Slobodian, Bockmann, 2025).
The phylogenetic hypothesis proposed in this study does not seek to evaluate the position of Phenacorhamdia within Heptapterinae, as it is based on two mitochondrial genes (COI and CytB), it should be regarded as exploratory and of limited robustness and explanatory power. Nevertheless, the topologies under Parsimony and ML analyses confirm the close relationships obtained by DoNascimiento et al. (2025) between the new genus they described Magdalenichthys and Phenacorhamdia. DoNascimiento et al. (2025) recovered Pariolius in a close relation to that clade, and Mastiglanis as sister to the remaining species within the tribe Hetapterini, unlike the present analyses in which Pariolius and Mastiglanis are successive sister taxa to the clade including Phenacorhamdia and Magdalenichthys. For their part, Slobodian, Bockmann (2025) recovered Pariolius as closely related to a clade comprising Phenacorhamdia as the sister group to ‘Imparfinis’ microps plus a new undescribed genus. Although the tree topologies obtained in the present analyses differs from those recovered in previous studies, Pariolius is consistently recovered in closely relationship to Phenacorhamdia in all cases. Resolution of phylogenetic relationships within Heptapterinae remains limited, which is likely attributable, at least in part, to incomplete taxon sampling in molecular analyses (e.g., Horiomyzon retropinnatus Stewart, 1986, Nannoglanis fasciatus Boulenger, 1887, Rhamdiopsis moreirai Haseman, 1911, ‘Imparfinis’ microps, etc.).
Generic allocation. Phenacorhamdia is one of the best-known genera of Heptapteridae, whose phylogenetic relationships for its recognition as a monophyletic group have been addressed through morphological and molecular data (Bockmann, 1998; Silva et al., 2021; Slobodian, Bockmann, 2025), although none of these studies has included the type species of the genus P. macarenensis. Bockmann (1998:367) proposed six synapomorphies to define the genus: (a) posterior process of epioccipital very prominent, (b) prognathous mouth, (c) neural and haemal spines of posterior vertebrae inclined 30 degrees or less relative to the longitudinal axis of the body, (d) first ray of pectoral-fin slightly longer than the second ray, (e) eight branched rays on the lower lobe of the caudal-fin, (f) second pore of the supraorbital canal (s2+i2) located farthest from the anterior nostril. All the above-mentioned character-states were verified in P. bermejo (Figs. 1, 6)justifying its inclusion in the genus from its morphology and corroborating our phylogenetic results. Thus, with the addition of P. bermejo, the genus is currently composed of 16 species widely distributed in the main basins of South America.
FIGURE 6| Caudal vertebrae of Phenacorhamdia bermejo,CI-FML 8289. Abbreviations: epu, epural; hu1+hu2, ventral hypural plate formed by the fused hypurals 1 and 2; hu3+hu4, dorsal hypural plate formed by the fused hypurals 3 and 4; hu5, dorsal hypural plate 5; ph, parhypural; pu1+u1, complex centrum composed of preural centrum 1 and ural centrum 1; ur, uroneural.
The new taxon is the fifth species of the genus described from the Paraná River basin, in addition to P. hoehnei, P. roxoi, P. tenebrosa, and P. unifasciata. Phenacorhamdia bermejo can be readily distinguished from these species by its yellowish body background and the absence of a dark brown lateral stripe. Additional characters also reinforce the differences among P. bermejo and the remaining members of the genus from the Paraná River basin, such as the number of vertebrae and the length of the maxillary barbels. Despite these differences, P. bermejo resembles P. tenebrosa in external morphology and meristics, highlighting the conservative morphology of these species. Nevertheless, the PCA encompassing all species from the Paraná River basinrevealed distinct groupings, thereby indicating a clear morphological differentiation between P. bermejo and the remaining species (see Fig. 4). Additionally, the phylogenetic analyses herein performed, under both Parsimony and Maximum Likelihood, recover Phenacorhamdia as monophyletic, with P. bermejo as the sister group of a clade composed of P. tenebrosa and P. unifasciata, with P. boliviana as the sister group of this clade of three species. In both analyses, P. bermejo was recovered as an independent taxon, also showing a high genetic divergence from P. tenebrosa (over 8%), which is, by certain morphological similarity, the main species with which the new taxon could be confused.
Giorgis et al. (2012) recorded P. tenebrosa in Argentina for the first time from the analysis of specimens collected at Yacyretá dam near to Ituzaingó city in Corrientes and the Pre-Delta National Park in Entre Ríos. In that contribution, they mentioned that the maxillary barbel surpasses the distal border of the pectoral fin, and the body presents a reddish-brown coloration in life (see fig. 1 in Giorgis et al., 2012), conditions that agree with those exhibited by P. bermejo. Although we could not analyze the material from the Paraná River basin, considering the above-mentioned characters, the presence of P. tenebrosa in Argentina is dubious and requires further investigation.
Bockmann (1998) recovered two monophyletic clades in Phenacorhamdia, one composed of species with short snout and long, slender bodies; and the other, of species with long snout and relatively short bodies. The short snout group, which includes species with snout lengths between 31.5–34.3% HL (Castro, Wosiacki, 2023) is supported by the angle of the neural and haemal spines of caudal vertebrae smaller than 20° (vs. between 20° and 30°) as their single known synapomorphy. The long snout group includes P. anisura, P. hoehnei, P. macarenensis, P. provenzanoi, P. roxoi, P. somnians, P. taphorni, P. tenebrosa, and P. unifasciata,which possess snout lengths between 34.3–40.0% HL (Castro, Wosiacki, 2023), and is supported by nine synapomorphies. Phenacorhamdia bermejo has a long snout (36.0–45.7% HL), and a relatively short, slender body, and presents the nine synapomorphies supporting the clade according to Bockmann (1998), including a long extension on the postero-lateral angle of premaxilla (ch. 15:2), the lateral margin of the posterior portion of lateral ethmoid sharp and posteriorly directed (ch. 26:2), presence of a foramen on the posterior portion of the lateral ethmoid (ch. 27:1), a vertical fenestra in the anterior portion of the pterosphenoid (ch. 52:1), optic foramen very reduced (ch. 53:2), anterior portion of the corono-meckelian bone tapered (ch. 61:1), the ascending portion of the Meckel cartilage discontinuous (ch. 64:1), and the ventral margin of the metapterygoid without bony contact to the antero-dorsal process of the quadrate (ch. 74:1), which justify its inclusion in the “long-snouted” group and corroborates the phylogenetic results obtained herein.
Material comparative examined. Bolivia. Phenacorhamdia boliviana. MZUSP 27813, 5, 44.6–65.6 mm SL, Villa Tunari, Chapare, 17°00’34.0”S 65°18’29.4”W. Brazil. Phenacorhamdia cabocla. MZUSP 87325, 2, 38.3–39.5 mm SL, Riberão São Raimundo, Tocantins River basin, Estreito, Maranhão, 06°44’22”S 47°07’26”W. MZUSP 84082, 3, 41.0–55.0 mm SL, Formosa do Rio Preto, Brejão, rio Sapão, tributary of rio Preto, São Francisco River basin, Bahia, 10°55’39”S 45°41’54”W. Phenacorhamdia hoehnei. MZUSP 103778, 8, 22.8–63.5 mm SL, córrego Cabeceira Comprida, Fazenda Campina Grande, dirt road between Coxim and Pedro Gomes, upper Taquari basin, Coxim, Mato Grosso do Sul, 18°14’35”S, 54°33’34”W. MZUSP 90022, 1, Tangará da Serra, rio do Sapo, tributary of rio Sepotuba, bellow the waterfall, Mato Grosso, 14°37’04”S 57°44’44”W. MZUSP 59354, 4, 32.6–60.1 mm SL, rio Salobra, about 18 km between Bonito to Bodoquena, Bodoquena, Mato Grosso do Sul. Phenacorhamdia roxoi. MZUSP 125819, holotype, 63.4 mm SL, rio Pardo, rio Paranapanema, Botucatu, São Paulo, 22.941178°S 48.584397°W. In addition, material listed in Azevedo-Santos et al. (2020), Ferrazi et al. (2024), Tinti et al. (2024). Phenacorhamdia somnians. MZUSP 30809, 2, 38.3–48.9 mm SL, rio Itacaiunas, railway, 10 km east of the N-4, Pará. Phenacorhamdia tenebrosa.EEBP 197, 3 syntypes, 38.5–43.8 mm SL, rio Mogi Guaçu, Cachoeira de Emas, São Paulo. CICCAA 8618, 1 c&s, 40.8 mm SL, Porto Ferreira, rio Mogi Guaçu basin, São Paulo, 21°52’37”S 47°30’32”W. Phenacorhamdia unifasciata. MZUSP 36691, holotype, 60.2 mm SL, Ribeirao Santana (São Bartolomeu), Paranaíba basin, Distrito Federal. MZUSP 36692, 1 paratype, 43.8 mm SL, Ribeirão Mestre D’Armas (São Bartolomeu), Paranaíba basin, Distrito Federal. MZUSP 36693, 1 paratype, 46.6 mm SL, córrego Fumal (São Bartolomeu), Paranaíba basin, Distrito Federal. MZUSP 36694, 1 paratype, 53.0 mm SL, córrego Papua, tributary of rio São Bartolomeu, Paranaíba basin, Distrito Federal. MZUSP 36696, 1 paratype, 41.2 mm SL, córrego Quinze (São Bartolomeu), Paranaíba basin, Distrito Federal. MZUSP 36698, 1 paratype, 43.1 mm SL, rio Pipiripau (São Bartolomeu), Paranaíba basin, Distrito Federal. MZUSP 25051, 1 paratype, córrego Mestre D’Armas, tributary of rio São Bartolomeu, Planaltina, Goiás, 15°38’S 47°39’W. Colombia. Phenacorhamdia anisura.MHNU-I 1817, 1 c&s, 37.7 mm SL, Bocas del Guayuriba, Guayuriba River, Puerto López, Meta, 03°55’10.8”N 73°05’05.6”W. Phenacorhamdia taphorni. MHNU-I 2119, 1 c&s, 34.1 mm SL, bridge over Guayuriba River, 03°57’33.5”N 73°16’20.0”W, route Rincón de Pompeya-La Palmera, Villavicencio, Meta.
Acknowledgments
To Fundación Miguel Lillo and CONICET for permanent support. To Miguel Casalinuovo (in memorium), Baltazar Bugeau (UEL-CONICET), Diego Delgado and Alejandro López-Méndez for help during sampling trips. We thank Felipe Alonso and Ezequiel Mettola for permanent support. We thank Carlos DoNascimiento for providing the COI sequences of Magdalenichthys species. We also thank Gabriel Depra and Ithalo Castro for their valuable comments, which greatly improved this manuscript.
References
Azevedo-Santos VM, Pelicice FM, Henry R. Knowing biodiversity: fishes from the Guareí River basin, a tributary of the Jurumirim reservoir, Paranapanema River, Brazil. Biota Neotrop. 2020; 20(3):e20201031. https://doi.org/10.1590/1676-0611-BN-2020-1031
Azevedo-Santos VM, Souto-Santos ICA, Baskin JN, Deprá GC, Katz AM. On the type material of two Heptapteridae species (Siluriformes) originally deposited in a deactivated collection in Brazil. Zootaxa. 2025; 5696(1):133–41. https://doi.org/10.11646/zootaxa.5696.1.9
Bockmann FA. Análise filogenética da família Heptapteridae (Teleostei, Ostariophysi, Siluriformes) e redefinição de seus gêneros. [PhD Thesis]. São Paulo, Universidade de São Paulo; 1998.
Bockmann FA, Guazzelli GM. Family Heptapteridae (Heptapterids). In: Reis RE, Kullander SO, Ferraris Jr. CJ, editors. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.407–33.
Bockmann FA, Miquelarena AM. Anatomy and phylogenetic relationships of a new catfish species from northeastern Argentina with comments on the phylogenetic relationships of the genus Rhamdella Eigenmann & Eigenmann, 1888 (Siluriformes, Heptapteridae). Zootaxa. 2008; 1780(1):1–54. https://doi.org/10.11646/zootaxa.1780.1.1
Bockmann FA, Slobodian V. Family Heptapteridae. In: Van der Sleen P, Albert J, editors. Field guide to the fishes of the Amazon, Orinoco and Guianas. Princeton and Oxford: Princeton University Press; 2018. p.233–52.
Britski HA. Uma nova espécie de Phenacorhamdia da bacia do alto Paraná (Pisces, Siluriformes). Comun Mus Ciênc Tecnol Pucrs, Sér Zool. 1993; 6:41–50.
Castro IS, Wosiacki WB. Redescription and expansion of the geographic distribution of Phenacorhamdia nigrolineata (Siluriformes: Heptapteridae). Neotrop Ichthyol. 2023; 21(2):e220107. https://doi.org/10.1590/1982-0224-2022-0107
DoNascimiento C, Milani N. The Venezuelan species of Phenacorhamdia (Siluriformes: Heptapteridae), with the description of two new species and a remarkable new tooth morphology for siluriforms. PNAS. 2008; 157(1):163–80. https://doi.org/10.1635/0097-3157(2008)157[163:TVSOPS]2.0.CO;2
DoNascimiento C, Villa-Navarro FA, Albornoz-Garzón JG, Conde-Saldaña CC, Silva GSC, Méndez-López A et al. An unexpectedly diverse new genus of catfishes (Siluriformes, Heptapteridae) endemic to the Magdalena River basin, Colombia. Zool J Linn Soc. 2025; 204(3):zlaf048. https://doi.org/10.1093/zoolinnean/zlaf048
Faustino-Fuster DR, López-Castaño JA, Quiñones JM, Meza-Vargas V. Increasing the species diversity of the monotypic genus Pariolius Cope 1872 (Siluriformes: Heptapteridae) after more than 150 years. Zootaxa. 2024; 5433(3):389–403. https://doi.org/10.11646/zootaxa.5433.3.6
Faustino-Fuster DR, Meza-Vargas V, Lovejoy NR, Lujan NK. Multi-locus phylogeny with dense Guiana Shield sampling supports new suprageneric classification of the Neotropical three-barbeled catfishes (Siluriformes: Heptapteridae). Mol Phylogenet Evol. 2021; 162:107186. https://doi.org/10.1016/j.ympev.2021.107186
Ferrazi R, Ottoni FP, Tinti F, Molina ME, Correia-Silva G, Birindelli JLO et al. Ichthyofauna of streams from the Piraju and Tejupá municipalities, São Paulo state, Brazil. Oecol Aust. 2024; 28(1):61–70. Available from: https://revistas.ufrj.br/index.php/oa/article/view/58773
Giorgis P, Ciotek L, Almirón A, Casciotta J. First record of Phenacorhamdia tenebrosa (Shubart, 1964) (Siluriformes, Heptapteridae) from the Paraná river basin in Argentina. ICP. 2012; 24:1–04. Available from: https://pecescriollos.de/wp-content/uploads/2020/12/ICP-24-Giorgis-et-al-2012-First-record-of-Phenacorhamdia-tenebrosa-in-Argentina.pdf
Goloboff P A. Estimating character weights during tree search. Cladistics. 1993; 9(1):83–91. https://doi.org/10.1111/j.1096-0031.1993.tb00209.x
Goloboff PA. Analyzing large data sets in reasonable times: solutions for composite optima. Cladistics. 1999; 15(4):415–28. https://doi.org/10.1111/j.1096-0031.1999.tb00252.x
Goloboff PA, Farris JS, Källersjö M, Oxelman B, Ramírez MJ, Szumik CA. Improvements to resampling measures of group support. Cladistics. 2003;19(3):324–32. https://doi.org/10.1111/j.1096-0031.2003.tb00376.x
Goloboff PA, Morales ME. TNT version 1.6, with a graphical interface for macOS and Linux, including new routines in parallel. Cladistics. 2023; 39(2):144–53. https://doi.org/10.1111/cla.12524
Ivanova NV, Zemlak TS, Hanner RH, Hebert PD. Universal primer cocktails for fish DNA barcoding. Mol Ecol Notes. 2007; 7(4):544–48. https://doi.org/10.1111/j.1471-8286.2007.01748.x
International Union for Conservation of Nature (IUCN). Standards and petitions committee. Guidelines for using the IUCN Red List categories and criteria. Version 16 [Internet]. Gland; 2024. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Lundberg JG, Baskin JN. The caudal skeleton of the catfishes, order Siluriformes. Am Mus Novit. 1969; 2398:1–49. Available from: https://www.biodiversitylibrary.org/item/328705#page/1/mode/1up
Lundberg JG, Bornbusch AH, Mago-Leccia F. Gladioglanis conquistador n. sp. from Ecuador with diagnoses of the subfamilies Rhamdiinae Bleeker and Pseudopimelodinae n. subf. (Siluriformes: Pimelodidae). Copeia. 1991; 1991(1):190–209. http://dx.doi.org/10.2307/1446263
Lundberg JG, McDade LA. On the South American catfish Brachyrhamdia imitator Myers (Siluriformes, Pimelodidae), with a phylogenetic evidence for a large intrafamilial lineage. Acad Nat Sci Phila. 1986; 463:1–24.
Mayer M. missRanger: fast imputation of missing values. R package version 2.6.0. 2024. Available from: https://mayer79.github.io/missRanger/
Palumbi SR. Nucleic acids II: the polymerase chain reaction. In: Hillis DM, Moritz C, Mable BK, editors. Molecular Systematics (2nd ed). Sinauer Associates, Inc. Sunderland, MA; 1996. p.205–47.
Pattengale ND, Alipour M, Bininda-Emonds ORP, Moret BME, Stamatakis A. How many bootstrap replicates are necessary? J Comput Biol. 2010; 17(3):337–54. https://doi.org/10.1089/cmb.2009.0179
Perdices A, Bermingham E, Montilla A, Doadrio I. Evolutionary history of the genus Rhamdia (Teleostei: Pimelodidae) in Central America. Mol Phylogenet Evol. 2002; 25(1):172–89. https://doi.org/10.1016/S1055-7903(02)00224-5
de Pinna MCC. Higher-level phylogeny of Siluriformes (Teleostei, Ostariophysi), with a new classification of the order. [PhD Dissertation]. City University of New York: New York; 1993.
Ríos N, Bouza C, Gutiérrez V, García G. Species complex delimitation and patterns of population structure at different geographic scales in Neotropical silver catfish (Rhamdia: Heptapteridae). Environ Biol Fishes. 2017; 100:1047–67. https://doi.org/10.1007/s10641-017-0622-1
R Development Core Team. R: a language and environment for statistical computing, version 4.2.2. Vienna, Austria: R Foundation for Statistical Computing; 2024. Available from: https://www.r-project.org/
Sabaj MH. Codes for Natural History Collections in Ichthyology and Herpetology (online supplement). Version 9.7. American Society of Ichthyologists and Herpetologists, Washington, DC. 2025. Available from: https://www.asih.org/resources/standard-symbolic-codes
Stamatakis A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014; 30(9):1312–13. https://doi.org/10.1093/bioinformatics/btu033
Silva GSC, Ochoa LE, Castro IS. New species of Phenacorhamdia (Siluriformes: Heptapteridae) from the Xingu River basin. Neotrop Ichthyol. 2022; 20(2):e210143. https://doi.org/10.1590/1982-0224-2021-0143
Silva GSC, Roxo FF, Melo BF, Ochoa LE, Bockmann FA, Sabaj MH et al. Evolutionary history of Heptapteridae catfishes using ultraconserved elements (Teleostei, Siluriformes). Zool Scr. 2021; 50(5):543–54. http://dx.doi.org/10.1111/zsc.12493
Slobodian V, Bockmann FA. Phylogeny and classification of Heptapteridae (Pimelodoidea). In: Arratia G, Reis RE, editors. Catfishes, a highly diversified group, vol. 2. Evolution and phylogeny. Boca Raton: CRC Press; 2025. http://dx.doi.org/10.1201/9781003374312-16
Taylor WR, Van Dyke GC. Revised procedures for stained and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Tinti F, Ferrazi R, Correia-Silva G, Ottoni FP, Molina ME, Vieira LO et al. Ichthyofauna from tributaries of the Paranapanema River basin, Cerqueira César municipality, southeastern Brazil. Braz J Biol. 2024; 84:e287937. https://doi.org/10.1590/1519-6984.287937
Togkousidis A, Kozlov OM, Haag J, Höhler D, Stamatakis A. Adaptive RAxML-NG: accelerating phylogenetic inference under maximum likelihood using dataset difficulty. Mol Biol Evol. 2023; 40(10):msad227. https://doi.org/10.1093/molbev/msad227
Thomas MR, Sabaj MH. Heptapteridae: seven-finned catfishes. In: Warren ML, Burr BM, editors. Freshwater fishes of North America: Characidae to Poeciliidae. Maryland: The Johns Hopkins University Press; 2020. p.123–48.
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PD. DNA barcoding Australia’s fish species. Philos Trans R Soc Lond B Biol Sci. 2005; 360(1462):1847–57. https://doi.org/10.1098/rstb.2005.1716
Authors
![]() Gastón Aguilera1
Gastón Aguilera1 ![]() ,
, ![]() Guillermo E. Terán1,
Guillermo E. Terán1, ![]() Miguel Ángel Cortés Hernández1,2,
Miguel Ángel Cortés Hernández1,2, ![]() Axel M. Katz3,
Axel M. Katz3, ![]() Valter M. Azevedo-Santos4,5,6,
Valter M. Azevedo-Santos4,5,6, ![]() Mauricio Benitez7 and
Mauricio Benitez7 and ![]() Marcos Mirande1
Marcos Mirande1
[1] Unidad Ejecutora Lillo (CONICET) – Fundación Miguel Lillo, Miguel Lillo 251, San Miguel de Tucumán (CP 4000), Tucumán, Argentina. (GA) gaguilera@lillo.org.ar (corresponding author), (GET) guilloteran@gmail.com, (MACH) macortes.hernandez95@gmail.com, (MM) mcmirande@gmail.com.
[2] Fondo para la Investigación Científica y Tecnológica (FONCYT), Argentina
[3] Departamento de Zoologia, Instituto de Biologia, Universidade Federal do Rio de Janeiro, Av. Carlos Chagas Filho, 373, 21941 902, Rio de Janeiro, RJ, Brazil. (AMK) axelmk@gmail.com.
[4] Centro Universitário Eduvale de Avaré, Av. Pref. Misael Eufrásio Leal, 347, 18705-050, Avaré, SP, Brazil. (VMAS) valter.ecologia@gmail.com.
[5] Grupo de Ecologia Aquática, Belém, PA, Brazil.
[6] Programa de Pós-Graduação em Biodiversidade, Ecologia e Conservação, Universidade Federal do Tocantins, 77500-000, Porto Nacional, TO, Brazil.
[7] Instituto de Biología Subtropical, UNaM-CONICET, Laboratorio de Genética Evolutiva, Félix de Azara, 1552, N3300LQH- Posadas, Argentina. (MB) mauriciofbenitez@gmail.com.
Authors’ Contribution 

Gastón Aguilera: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Guillermo E. Terán: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Visualization, Writing-original draft.
Miguel Ángel Cortés Hernández: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-review and editing.
Axel M. Katz: Conceptualization, Data curation, Investigation, Writing-original draft.
Valter M. Azevedo-Santos: Conceptualization, Data curation, Formal analysis, Investigation, Writing-review and editing.
Mauricio Benitez: Conceptualization, Investigation, Methodology, Writing-review and editing.
Marcos Mirande: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing-review and editing.
Ethical Statement
The study complied with the animal welfare laws, guidelines, and policies of the Comité Nacional de Ética en la Ciencia y Tecnología, Argentina. Collection permits were granted by the Ministerio de Ambiente of Jujuy (permits’ numbers: 1103-306 M/2016, Res. N° 137/2016-MA).
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article and in the supplementary material of this article.
AI statement
The authors declare that ChatGPT (OpenAI) was used to assist with grammar correction, language polishing, and reference formatting. It was not used to generate or analyze data in this manuscript.
Funding
This work was partially funded by FONCyT (PICT 2020–02141 and PICT 2019–01004), CONICET (PIP–11220200101214), and Fundación Miguel Lillo (Z–0085).
Supplementary Material
Supplementary material S1
Supplementary material S2
How to cite this article
Aguilera G, Terán GE, Cortéz Hernández MA, Katz AM, Azevedo-Santos VM, Benitez M, Mirande M. Hidden diversity in the Paraná River basin: a new species of Phenacorhamdia (Siluriformes: Heptapteridae) from the Bermejo River, Argentina. Neotrop Ichthyol. 2026; 24(1):e250189. https://doi.org/10.1590/1982-0224-2025-0189
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 28, 2025
Accepted December 28, 2025
![]() Submitted July 29, 2025
Submitted July 29, 2025
![]() Epub April 21, 2026
Epub April 21, 2026