![]() V. Ezequiel Méttola1,
V. Ezequiel Méttola1, ![]() Guillermo E. Terán1
Guillermo E. Terán1 ![]() ,
, ![]() Gastón Aguilera1,
Gastón Aguilera1, ![]() Martin M. Montes2,
Martin M. Montes2, ![]() Marina Ibáñez Shimabukuro2,
Marina Ibáñez Shimabukuro2, ![]() Felipe Alonso3 and
Felipe Alonso3 and ![]() J. Marcos Mirande1
J. Marcos Mirande1
PDF: EN XML: EN | Supplementary: S1 S2 S3 S4 S5 | Cite this article
Associate Editor: ![]() Hermán López-Fernández
Hermán López-Fernández
Editor-in-chief: ![]() Carla Pavanelli
Carla Pavanelli
Abstract
The genus Characidium comprises a diverse group of small Neotropical freshwater fishes. Among the 89 valid species, eight occur in Argentina. Characidium borellii, originally described based on material from the río Salí basin, show morphological similarities to specimens from nearby basins, often identified as Characidium cf. zebra. We redescribe C. borellii, based on detailed morphological and molecular analyses of specimens collected from the Salí, Bermejo, and Juramento-Salado basins. Characidium borellii can be distinguished from its congeners by an isthmus completely covered with scales, 14 scale rows around the caudal peduncle, a single row of teeth on the dentary, 13 or 14 pectoral-fin rays, a complete lateral line with 36–38 scales, fins mostly hyaline, regular transversal bars on the body, and the presence of hooks on the pelvic and pectoral fins. The distribution of C. borellii is here extended to include the Bermejo and Juramento-Salado basins. Additional data are provided regarding its phylogenetic position and sexual dimorphism.
Keywords: Biogeography, Characidiinae, Freshwater fish, Phylogeny, Taxonomy.
Introduction
The genus Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) is a group of Neotropical freshwater fishes commonly referred to as “tritolos” and “mariposas” in Argentina and Uruguay (Serra et al., 2014; Almiron et al., 2015; Loureiro et al., 2023). It currently includes 89 valid species (Fricke et al., 2025) and is geographically restricted to tropical South America and southern Panama. The genus was defined by Buckup (1993a,b) as a monophyletic group based on a single synapomorphy: a black spot near the base of the middle caudal-fin rays, although this character has been lost in some species within the genus.
The río de La Plata basin, one of major hydrographic systems in South, extends across Argentina, Bolivia, Brazil, Paraguay, and Uruguay. It includes three major basins: the río Paraná (including the Iguacu and Salado rivers), the Paraguay (with the río Bermejo and Pilcomayo as western tributaries) and the río Uruguay (Pasquini, Depetris, 2007). Currently, 24 species of Characidium have been recorded in the La Plata basin, including: C. borellii (Boulenger, 1895); C. chicoi Graça, Ota & Domingues, 2019; C. dumonti Stabile, Reis, Oliveria & Graça, 2025; C. etzeli Zarske & Géry, 2001; C. fasciatum Reinhardt, 1867; C. gomesi Travassos, 1956; C. heirmostigmata Graça & Pavanelli, 2008; C. itarare Stabile, Reis, Frota, Graça & Oliveira, 2024; C. lagosantense Travassos, 1947; C. laterale (Boulenger, 1895); C. nupelia Graça, Pavanelli & Buckup, 2008; C. occidentale Buckup & Reis, 1997; C. oiticicai Travassos, 1967; C. onca Melo, Brito Ribeiro & Lima, 2021; C. pterostictum Gomes, 1947; C. rachovii (Regan, 1913); C. schubarti Travassos, 1955; C. serrano Buckup & Reis, 1997; C. stigmosum Melo & Buckup, 2002; C. tenue (Cope, 1894); C. travassosi Melo, Buckup & Oyakawa, 2016; C. vestigipinne Buckup & Hahn, 2000; C. xanthopterum Silveira, Langeani, Graça, Pavanelli & Buckup, 2008; C. cf. zebra Eigenmann, 1909 (Buckup, Reis, 1997; Casciotta et al.,2015; Dagosta et al., 2024; Fricke et al., 2025). Among these, eight species have been recorded for Argentina: C. borellii, C. heirmostigmata, C. occidentale, C. pterostictum, C. rachovii, C. serrano, C. tenue,and C. cf. zebra (Mirande, Koerber, 2020).
Among these species, C. borellii appears to be endemic to the río Salí-Dulce, an endorheic basin in northwestern Argentina (Butí, Cancino, 2005; Mirande, Aguilera, 2009). Initially described as part of the genus Nanognathus by Boulenger (1895), the species was later reassigned to Characidium by Géry et al.(2001) and Zarske, Géry (2001), who also provided a supplementary description and clarified its type-locality as San Pablo, Tucumán, Argentina.
The morphology of C. borellii closely resembles that of C. zebra (Boulenger, 1895; Eigenmann, 1909; Buckup, 1992; Buckup, Reis, 1997; Zarske, Géry, 2001) a species described by Eigenmann (1909) from Maripicru Creek, a tributary of the río Branco basin in the Guiana Shield. Due to the limited number of diagnostic characters, many specimens distributed across South America basins have been identified as C. cf. zebra, forming a species complex (Buckup, 1992). This taxonomic ambiguity also occurs in northwestern Argentina, where specimens from basins adjacent to the río Salí basin, such as the Bermejo and Juramento-Salado basins have traditionally been identified as C. cf. zebra (Liotta, 2005; Cancino, Aguilera, 2016; Terán et al., 2016; Alonso et al., 2025).
In this study, we perform an integrative analysis of Characidium populations from northwestern Argentina. We present a revised redescription of C. borellii that allows for its unequivocal distinction from congeneric species. Additionally, we assess the taxonomic status of specimens from adjacent basins based on morphological and meristic data and discuss their phylogenetic placement using molecular and morphological evidence.
Material and methods
Morphological analyses. Morphometric measurements were obtained as straight-line distances using a caliper to the nearest 0.1 mm. Morphological and meristic data followed Buckup (1993a), with modifications from Melo, Oyakawa (2015). Additional variables measured included: 1) number of scales between the anus and the anal-fin origin, 2) number of scales between the anus and the caudal fin, 3) number of scales between the adipose fin and the caudal fin, 4) number of scales between the pectoral and pelvic fins, 5) lengths of the dorsal, anal, pectoral and pelvic fins, measured to the distal tips of longest branched ray, 6) peduncle width measured at the vertical through the lowest caudal-peduncle depth, 7) head height and width measured at the posterior region of the orbit. A summary of these measurements is presented in Tab. 1, with additional data provided in Tab. S1. Selected specimens were cleared and stained (cs) following the protocol of Taylor, Van Dyke (1985). Specimens are deposited in the ichthyological collection of Fundación Miguel Lillo (CI-FML) and Instituto de Bio y Geociencias del Noroeste Argentino (IBIGEO-I).
TABLE 1 | Morphometric summary table of Characidium borellii. Morphometric data from holotype of C. borellii were obtained from Géry et al. (2001).
Variables |
| Holotype | N | Min-Max | Mean | SD | K |
Total length | TL (mm) |
| 83 | 32.4–84.5 | 61.3 | 11.1 | – |
Standard length | SL (mm) | 67.3 | 84 | 25.5–70.0 | 50.1 | 9.2 | – |
Head length | HL (mm) | 14.2 | 84 | 6.9–17.4 | 11.5 | 2.1 | – |
Percentages of standard length | |||||||
Head length (%) | HL% | 21.1 | 84 | 19.7–26.9 | 23.1 | 1.4 | 0.9 |
Pre-pectoral distance | PPD | 20.1 | 84 | 17.3–27.9 | 22.3 | 1.6 | 0.9 |
Pectoral-fin height | PFH |
| 84 | 15.3–23.1 | 19.9 | 1.5 | 0.8 |
Pre-dorsal distance | PDD | 45.5 | 83 | 40.5–49.3 | 45.9 | 1.5 | 1.1 |
Dorsal-fin height | DFH | 17.7 | 84 | 13.8–23.0 | 18.4 | 1.6 | 0.8 |
Dorsal-fin base | DFB |
| 84 | 11.4–15.4 | 14.1 | 0.8 | 1.0 |
Pre-pelvic distance | PPVD | 52.1 | 76 | 46.7–54.7 | 51.6 | 1.6 | 1.1 |
Pelvic-fin height | PVFH |
| 84 | 9.1–16.2 | 12.6 | 1.2 | 0.8 |
Pre-anal distance | PAD |
| 76 | 71.2–80.4 | 76.5 | 1.5 | 1.0 |
Anal-apex distance | AAD |
| 76 | 88.9–97.5 | 94.0 | 1.7 | 1.0 |
Anus to anal fin distance | AAFD |
| 84 | 3.8–9.9 | 6.5 | 1.2 | 0.9 |
Anal-fin height | AFH | 12.9 | 70 | 12.0–17.1 | 14.3 | 1.1 | 0.8 |
Anal-fin base | AFB |
| 84 | 6.5–10.4 | 8.0 | 0.7 | 1.2 |
Adipose-fin height | ADFH |
| 84 | 3.3–6.8 | 5.6 | 0.6 | 0.9 |
Peduncle length | PL | 16.6 | 83 | 15.5–19.2 | 17.4 | 1.0 | 1.0 |
Body depth at dorsal-fin origin | BDDFO | 24.2 | 76 | 17.4–25.2 | 21.5 | 1.7 | 1.3 |
Body depth at anal-fin origin | BDAFO |
| 76 | 12.9–19.9 | 15.9 | 1.4 | 1.2 |
Body depth at caudal penducle | BDCP | 11.9 | 82 | 9.0–12.8 | 11.6 | 0.6 | 1.1 |
Body width | BW |
| 84 | 9.3–13.2 | 11.2 | 0.9 | 1.2 |
Percetages of head length | |||||||
Snout length | SNL | 25.4 | 84 | 20.7–29.6 | 24.2 | 1.8 | 1.1 |
Snout-maxillary tip | SMT |
| 83 | 20.3–26.6 | 23.5 | 1.2 | 0.9 |
Anterior-naris orbit | ANO |
| 84 | 7.2–12.0 | 9.7 | 0.9 | 0.9 |
Posterior-naris orbit | PNO |
| 84 | 2.9–6.3 | 4.4 | 0.7 | 1.0 |
Cheek | CH |
| 83 | 5.9–14.5 | 9.9 | 1.6 | 1.3 |
Orbital diameter | OD | 19.9 | 84 | 19.7–30.9 | 25.0 | 2.4 | 0.5 |
Interorbital distance | IOD | 18.3 | 84 | 11.3–20.8 | 15.8 | 1.7 | 1.0 |
Diagnostic characters were compared with the original description of Characidium species and related taxonomic works (Buckup, 1992; Taphorn, 1990; Buckup, Reis, 1997; Taphorn et al., 2006; Netto-Ferreira et al., 2013; Leitão, Buckup, 2014; Melo, Espíndola, 2016; Agudelo-Zamora et al., 2020; Flausino et al., 2020; Armbruster et al., 2021). Sex identification was based on the presence of secondary sexual characteristics (Teixeira, Melo, 2021) and on gonadal examination.
Statistical analyses. Occurrence localities for Characidium borellii were grouped into three major geographic basins: Salí, Bermejo, and Juramento-Salado. Specimens from the arheic río Urueña and río Horcones basins are included in the Juramento-Salado group, as both connected with the río Salado during rainy seasons (Tenchini, 1984). In total, 84 specimens were analyzed (36 specimens from the Salí basin, 33 from the Bermejo basin and the 15 from the Juramento-Salado basin), using 36 morphometric variables, including the standard length (SL) and head length (HL). Allometric coefficient (K) were calculated for each measurement using an ordinary least square (OLS) regression line of base 10 log transformed data (Huxley, 1993; Klingenberg, 1996; Kilmer, Rodrigez, 2017), with SL as the reference. Measurements were transformed into percentages of SL and HL to mitigate size effects and missing values were imputed using mean value substitution.To compare populations, a Principal Component Analysis (PCA) was conducted using the PAST software package (Hammer et al., 2001). Significant components were determined using the broken-stick model (Frontier, 1976) and scree plot analysis (Cattell, 1966). Eigenvalues were calculated from the covariance matrix of the morphometric variables. Sexual dimorphism features and intraspecific variation were assessed using both original and transformed measurements. Statistical differences between populations and groups were evaluated using a PERMANOVA test (based on the Euclidean similarity index). Differences among major variables were tested using the Kruskal-Wallis test, followed by a Mann-Whitney pairwise test with Bonferroni correction. Statistical significance was set at p < 0.05.
DNA extraction. Total genomic DNA was extracted from muscle tissue samples, preserved in cold 96% ethanol. Extractions were performed using the PURO-Genomic DNA kit (PB-L Productos Bio-Lógicos® S.A.), following the manufacturer’s protocol. The mitochondrial Cytochrome Oxidase Subunit I (COI) and the Cytochrome b (cytB) genes were amplified using Polymerase Chain Reaction (PCR) on an Eppendorf Mastercycler thermal cycler (Hamburg, Germany) in a 50-µl reaction mixture consisting of 25 µl of PB-L master mix (PB-L Productos Bio-Lógicos, Quilmes, Argentina), 0.4 μM of each forward and reverse primer and 4 μl of the template DNA. The partial COI gene was amplified with the primers Fish F1 (5′-TCA ACC AAC CAC AAA GAC ATT GGC AC-3′) and Fish R1 (5′- AGA CTT CTG GGT GGC CAA AGA ATC A-3′) (Ward et al., 2005), according to Souza-Shibatta et al. (2018). The fragment of cytB was amplified with the forward primer CB3-H (GGC AAA TAG GAA RTA TCA TTC) and reverse primer Gludg-L (TGA CTT GAA RAA CCA YCG TTG) (Palumbi et al., 2002) following thermocycling conditions as described by Loureiro et al. (2018). Sequencing was performed bidirectionally using an ABI 3730XL sequencer (Macrogen Inc. Korea), to ensure sequence accuracy.
Molecular analyses. Molecular data included sequences of the Cytochrome Oxidase Subunit I (COI) and Cytochrome b (cytB) markers. New sequences (Tab. S2A), were compared alongside sequences mainly from Oliveira-Silva et al. (2024) and Serra et al. (2025), and other sequences available in GenBank (Tab. S2B). Sequences were aligned using MUSCLE (Edgar, 2004), via MEGA (Kumar et al., 2018), which was also used to calculate genetic distances for the COI marker under the Kimura 2-parameter (K2) substitution model, chosen for its standard use in DNA barcoding (Díaz et al., 2016). Phylogenetic analyses were conducted using two approaches: (1) Parsimony Analysis: conducted in TNT (v. 1.6 Goloboff et al., 2008) under extended implied weighting exploring a broad range of concavity (K) values (3–27) (Goloboff, 2014); clade support was estimated using symmetric resampling (10,000 replicates), with GC values (“Group present/Contradicted”) reported (Goloboff et al., 2003); separate analyses were run for COI alone and the combined COI + cytB dataset. (2) Maximum Likelihood (ML) analysis: performed using IQ-TREE (Trifinopoulos et al., 2016). Edge confidence intervals were calculated using fast bootstrapping with 10,000 repetitions (Hoang et al., 2018). The TIM2+F+I+G4 model was identified as the best-fit for COI (Kalyaanamoorthy et al., 2017), was used for tree inference. For both methods, Hoplias malabaricus (Bloch, 1794) was used as the root. In cases of incongruent species delimitation (e.g., Characidium bimaculatum Fowler, 1941; C. bahiense Almeida, 1971; C. fasciatum, and C. gomesi), only sequences corresponding to type-locality ecoregions or basins were included.
Collection’s acronyms:CI-FML, Colección Ictiológica Fundación Miguel Lillo, San Miguel de Tucumán; FMNH, Field Museum of Natural History, Chicago; IBIGEO, Instituto de Bio y Geociencias del NOA, Salta; IAvH-P, Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Colección de Peces, Villa de Leyva; MG-ZV-P, Museo Provincial de Ciencias Naturales “Dr. Ángel Gallardo”, Rosario; MZUSP, Museu de Zoologia, Universidade de São Paulo, São Paulo; and MZUT, Dipartimento di Biologia Animale e dell’Uomo, Museo Zoologico, Università di Torino, Torino.
Results
Characidium borellii (Boulenger, 1895)
(Figs. 1–5; Tab. 1)
Nanognathus borellii Boulenger, 1895:3 (original description; type-locality: San Pablo, Tucuman Province, Argentina).
Characidium borellii (Boulenger, 1895). —Buckup, 1993b:101 (generic location to Characidium). —Géry et al., 2001:27 (correction on type-locality). —Zarske, Gery, 2001:238 (comparative material). —Mirande, 2009:579 (phylogeny). —Mirande, 2010:466 (phylogeny). —Mirande, Koerber, 2015:27 (listed). —Alonso et al., 2015:274 (anatomical study) —Saravia et al., 2023:333 (listed)..
Characidium cf. zebra (non Eigenmann, 1909). —Butí, Cancino, 2005:26 (listed). —Mirande, Aguilera, 2009:178 (listed). —Cancino, Aguilera, 2016:43 (listed). —Terán et al., 2016:199 (mention). —Alonso et al., 2025:18 (mention). —Cortés Hernández et al., 2025:10 (listed).
Characidium fasciatum (non Reinhardt, 1867). —Ringuelet et al., 1967:188 (listed). —Butí, Miquelarena, 1995:26 (listed). —Liotta, 2005:88 (listed).
Diagnosis. Characidium borellii can be distinguished from all congeners, except C. bahiense, C. barbosai Flausino, Lima, Machado & Melo, 2020; C. bimaculatum; C. brevirostre Pellegrin, 1909; C. cacah Zanata, Ribeiro, Araújo-Porto, Pessali & Oliveira-Silva, 2020; C. caucanum Eigenmann, 1912; C. chancoense Agudelo-Zamora, Ortega-Lara & Taphorn, 2020; C. chicoi; C. chupa Schultz, 1944; C. clistenesi Melo & Espíndola, 2016; C. dumonti; C. deludens Zanata & Camelier, 2015; C. etheostoma Cope, 1872; C. etzeli; C. fleurdelis Zanata, Oliveira-Silva & Ohara, 2023; C. geryi (Zarske, 1997); C. heiniamum Zarske & Géry, 2001; C. heirmostigmata; C. interruptum Pellegrin, 1909; C. itarare; C. lagosantense; C. laterale; C. litorale Leitão & Buckup, 2014; C. longum Taphorn, Montaña & Buckup, 2006; C. marshi Breder, 1925; C. mirim Netto-Ferreira, Birindellii & Buckup, 2013; C. nana Mendonça & Netto-Ferreira, 2015; C. nupelia; C. occidentale; C. onca; C. orientale Buckup & Reis, 1997; C. papachibe Peixoto & Wosiacki, 2013; C. pellucidum Eigenmann, 1909; C. phoxocephalum Eigenmann, 1909; C. pteroides Eigenmann, 1909; C. pumarini Teixeira & Melo, 2021; C. rachovii; C. sanctjohanni Dahl, 1960; C. samurai Zanata & Camelier, 2014; C. satoi Melo & Oyakawa, 2015; C. schindleri Zarske & Géry, 2001; C. serrano; C. steindachneri Cope, 1878; C. sterbai (Zarske, 1997); C. stigmosum; C. tapuia Zanata, Ramos & Oliveira-Silva, 2018; C. tenue; C. varii Zanata, Oliveira & Oliveira-Silva, 2024; C. vestigipinne; C. xanthopterum; C. xavante Graça, Pavanellii & Buckup, 2008; and C. zebra by having an isthmus covered with scales (vs. unscaled). It differs from C. bahiense, C. barbosai, C. brevirostre, C. cacah, C. caucanum, C. chancoense, C. chicoi, C. clistenesi, C. dule, C. etzeli, C. fleurdelis, C. heiniamum, C. heirmostigmata, C. itarare, C. lagosantense, C. laterale, C. litorale, C. longum, C. marshi, C. mirim, C. nana, C. nupelia, C. occidentale, C. orientale, C. papachibe, C. pellucidum, C. phoxocephalum, C. pteroides, C. rachovii, C. schinderi, C. serrano, C. tapuia, C. tenue, C. vestigipinne, and C. xavante by having 14 scales around the caudal peduncle (vs. 12 or fewer scales); from C. chupa, C. onca, C. pumarini, C. samurai and C. zebra by having one row of dentary teeth (vs. two rows, inner teeth present); from C. etheostoma, C. geryi, C. sterbai and C. sanctjohanni by having 13 or 14 pectoral-fin rays (vs. 11 or fewer rays); from C. interruptum by having a complete lateral line (vs. incomplete); from C. varii by having fins mostly hyaline (vs. dark stripes present on fins); from C. stigmosum by lacking dots on body (vs. dots present); and from C. satoi and C. deludens by having regular transversal bars (vs. irregular). Additionally, Characidium borellii can be distinguished from congeners, except C. bahiense, C. barbosai, C. bimaculatum, C. clistenesi, C. deludens, C. lagosantense, C. laterale, C. litorale, C. mirim, C. nupelia, C. satoi, C. stigmosum, C. xanthoptertum, and C. xavante, by possessing bony processes (hooks) on pelvic fins. It is further distinguished from C. bahiense, C. bimaculatum, C. clistenesi, C. lagosantense, C. laterale, C. mirim, C. nupelia, C. stigmosum,and C. xavante by having a parietal branch of the supraorbital canal (vs. absent); from C. litorale by lacking secondary bars (vs. thin dark secondary bars present);from C. satoi and C. xanthopterum by having conspicuous transversal bars (vs. absent in breeding males of C. satoi and adults of C. xanthopterum); from C. barbosai by having a preorbital stripe (vs. absent); and from C. deludens by having hooks on pectoral fins of males (vs. hooks absent).
Description. Morphometric data summarized in Tab. 1, full dataset by basin in Tab. S1. Largest specimen 70.0 mm SL; smallest 25.5 mm SL. Body elongated. Dorsal profile moderately convex to dorsal-fin origin, slightly convex to adipose fin, straight to caudal fin. Ventral profile slightly convex from snout to pelvic fins. Body deepest at dorsal fin origin. Caudal peduncle depth smaller than body depth at anal-fin origin (Fig. 1A). Head length equal to or slightly greater than pre-pectoral distance. Snout rounded in lateral view, tip below of ventral eye margin level. Mouth subterminal. Maxillary not or barely reaching anterior orbit margin. Orbit oval, laterodorsally positioned; orbital diameter equal to or slightly longer than snout length. Two nasal openings, well separated; anterior with full dermal flap, posterior with flap on anterior margin only.
FIGURE 1| Specimen of Characidium borellii, male, CI FML 8183, 53.6 mm SL, from río Romano, Tucumán, Argentina, in río Salí basin. A. Lateral view. B. Dorsal view. C. Ventral view. Scale bar = 1 cm.
Cycloid scales lacking circuli. Isthmus completely scaled, except for one (Fig. 1C). Longitudinal scales: 35(2), 36(16), 37(34), 38(28), 39(2); all perforated by lateral line. Transversal scales: 4/5(56), 5/5(18). Pre-dorsal series: 7(1), 9(10), 10(35), 11(27), 12(11). Pre-adipose series: 6(1), 8(8), 9(26), 10(40), 11(8) (Fig. 1B). Pre-anal series: 4(2), 5(60), 6(22). Scales between adipose and caudal fin: 4(2), 5(10), 6(50), 7(17), 8(1), 9(1). Scales between anus and anal-fin origin: 2(18), 3(56), 4(10). Scales between anal and caudal-fin origin: 5(9), 6(45), 7(26), 8(1). Scales between pectoral and pelvic fins: 11(18), 12(39), 13(4). Scales around caudal peduncle: 14(84) (Fig. 1A).
Pectoral fin well-developed; surpassing vertical through dorsal-fin origin; rays: iii,9,i(38); iii,10,i(45); iii,11,i(1). Pelvic fin well-developed, barely reaching anal-fin origin in males; rays: i,6,i(6); i,7,i(74); i,8,i(3). Dorsal fin truncated, narrow base; rays: ii,8(3); ii,9(79); ii,10(2). Anal fin with narrow base; rays: ii,6(2); ii,7(70); ii,8(12). Small unbranched anterior rays in dorsal and anal fins, visible in cs specimens. Adipose fin well-developed. Caudal fin with lower lobe slightly longer; rays: i,8,7,i(2); i,8,8,i(2); i,9,8,i(80).
Pseudotympanum present as muscular hiatus between obliquus superioris and obliquus inferioris, aligned with swimbladder, posterior to humeral spot. Oval shaped opening between ribs of fifth and sixth vertebrae (Fig. 3). Swimbladder reduced, with two chambers; anterior chamber wider than posterior, both of equal length.
FIGURE 2| Specimens of Characidium borellii from río Salí (A–F), río Juramento (G) and río Bermejo (H) basins. A. Male from río Los Sosa, Tucumán, Argentina (IBIGEO-I 524, 50.9 mm SL). B. Female specimen from río Sucio, Tucumán, Argentina (CI FML 8003, 58.1 mm SL). C. Male from río Lules (CI FML 8181, 45.8 mm SL). D. Female from río Lules, Tucumán, Argentina (CI FML 8181, 44.5 mm SL). E. Hyperpigmented male from río Las Tunas, Catamarca, Argentina (CI FML 8005, 63.5 mm SL). F. Gravid female from río Las Tunas, Catamarca, Argentina (CI FML 8005, 60.5 mm SL). G. Female specimen from río Guanaco, Salta, Argentina (CI FML 8014, 57.4 SL, mm SL). H. Female specimen from río Popayan, National Park “El Rey”, Salta, Argentina (CI FML 8012, 46.9 mm SL). Scale bar = 1 cm.
FIGURE 3| Pseudotympanum of Characidium borellii, male, CL FML 8013, 60.0 mm SL, from National Park “El Rey”, Salta, Argentina. Overlying skin, adipose tissue and lateral line nerve removed. os, obliquus superioris; oi, obliquus inferioris; ls, lateralis superficialis. Numbers correspond to vertebrae associated with pleural ribs.
Premaxillary with 6(2 cs) or 7(7 cs) tricuspid teeth, decreasing in size posteriorly. Dentary with a one row of 6(2 cs) or 7(7 cs) tricuspid teeth, decreasing posteriorly. Replacement teeth present on dentary and premaxillary (Figs. 4A, B). Maxillary lacking teeth. Ectopterygoid elongated, bearing 7(4 cs), 8(2 cs) and 10(2 cs) minute conical teeth. Mesopterygoid and endopterygoid lacking teeth. Branchiostegal rays: 5(6 cs). Gill rakers: hypobranchial 1(3 cs); ceratobranchial 6(6 cs); epibranchial 6(1 cs), 7(1 cs), 8(1 cs).
FIGURE 4| A. Right upper jaw in medial view of Characidium borellii (CI FML 3865). B. Right dentary in medial view of C. borellii (CI FML 3865). C. Pelvic girdle and fins in ventral view of a male C. borellii (CI FML 3865) with a detail of bony processes. Abbreviations: BP, bony processes; Den, dentary; dent, dentary teeth; dentr, replacement dentary teeth; IsP: ischiatic process; MEC, Meckel cartilage; MX, maxillary; Pb, pelvic bone; PMX, premaxillary; pmxt, premaxillary teeth. Scale bars = 1 mm.
Parietal branch of supraorbital canal present, reaching parietal. Cranial fontanel present bordered anteriorly by the frontals. Infraorbitals 1–6 present, infraorbitals 4–6 reduced to a canal. Pelvic bone distally rounded; ischiatic process irregular (Fig. 4C). Total vertebrae: 35(2 cs), 36(3 cs). Four anterior vertebrae fused in Weberian apparatus, 32 free vertebrae. Precaudal vertebrae: 15(5 cs). Caudal vertebrae: 16(1 cs), 17(4 cs). Hypural bones: 6(5 cs). Epural bones: 2(2 cs), 3(2 cs). Pair of uroneurals: 1(1 cs). Upper procurrent rays: 8(2 cs), 9(4 cs). Lower procurrent rays: 7(5 cs), 8(1 cs).
Coloration in alcohol. Body and head yellowish, darker dorsally, ventrally whitish. Dark pre-orbital stripe present, oblique, from snout tip to anterior eye margin, stripe poorly defined. Dark post-orbital stripe present with poorly defined edges. Area around nares lacking melanophores, Opercle mostly dark, with quadrangular blotch in some specimens. Dark longitudinal stripe along mid-body, from opercle to caudal fin, edges diffuse, narrower than one scale depth anteriorly, widening posteriorly. Dark transversal bars across body: 7(3), 8(8), 9(38), 10(18), 11(6), or 12(2), wider than scale, regular in shape, surpassing midlateral stripe. Dark stripe from dorsal fin origin to head along mid-dorsal scales. Thin dark stripe between dorsal an adipose fins. Humeral blotch present, slightly vertically elongated. Basicaudal spot present, more conspicuous in small specimens. Peduncular blotch present, circular or oval; more diffuse in larger specimens. Fins mostly hyaline. Few dark-brown chromatophores between rays in the middle dorsal fin. Dark melanophores on posterior margin of first three to five rays of pectoral and pelvic fins.
Coloration in life. Dorsal body silver or light brown, ventral region yellowish or whitish. Dark or silver longitudinal stripe, diffuse anteriorly, from snout to caudal-fin base. Dark transversal bars (9–11) from anterior to posterior region. Fins mostly hyaline, paired fins yellowish, with dark-brown pigmentation on posterior margin of first pectoral and pelvic fin rays (Fig. 5). Small, dark, rounded spots near operculum and caudal-fin base. Peduncular blotch visible or diffuse.
FIGURE 5| Life specimens of Characidium borellii, CI FML 8183, from río Salí basin, Tucumán, Argentina. A. Female, 54.9 mm SL. B. Male, 57.3 mm SL.
Sexual dimorphism. Mature males of Characidium borellii can be distinguished from mature females and juveniles by the presence of bony processes (hooks) on pelvic and pectoral fins, and by pelvic-fin length. Hooks are consistently present on the pelvic fins of males from all basins examined but are rarely found on pectoral fins. When present, these hooks are located on the dorsal and distal half of the first four unbranched rays of the pair fins (Fig. 4C). These bony processes are classified as “spine type” according to classification proposed by Teixeira, Melo (2021), exhibiting a cylindrical cross-section along their entire length. Females typically lack hooks on their pair fins, one female was observed with three small hooks on the pelvic fins. Pelvic-fin length is sexually dimorphic. In males, the pelvic fin reaches or slightly surpasses the anal-fin origin, whereas in females, it does not reach the anal-fin origin (Fig. S4). One hyperpigmented male from río Tunas, characterized by a darker body and well-developed pelvic and pectoral hooks, was examined (Fig. 2E). No differences in counts were observed between males and females. However, males generally have a longer head and pelvic fin, as well as greater body depth at dorsal-fin origin, compared to females.
Historical remarks. Ringuelet et al. (1967) erroneously cited Characidium borellii for San Pablo, Paraguay, and C. fasciatum for Argentina. Géry et al. (2001) later clarified the type-locality of C. borellii as San Pablo, Tucumán, Argentina. Buckup (1992) determined that many specimens were incorrectly identified as C. fasciatum,leading to the species exclusion from checklists of freshwater fishes in Argentina (Mirande, Koerber, 2020). Subsequently, specimens previously identified as C. fasciatum were re-identified as C. borellii from Salí basin or C. cf. zebra in several other basins in Argentina (Butí, Cancino, 2005; Liotta, 2005; Almirón et al., 2015; Cancino, Aguilera, 2016; Casciotta et al., 2016; Saravia et al., 2023).
Statistical analysis. Allometry. The allometric coefficients (K) for Characidium borellii indicated a range of negative and positive allometry, with some variables displaying isometry. Measurements exhibiting the greatest positive allometry were peduncle width (K = 1.3), body depth at the dorsal-fin origin (K = 1.3) and cheek (K = 1.3). Measurements with the greatest negative allometry included pelvic-fin height (K = 0.8), dorsal-fin height (K = 0.8), pelvic-fin length (K = 0.8), anal-fin height (K = 0.8) and orbital diameter (K = 0.5). Orbital diameter exhibited strong negative allometry with a K value of 0.5.
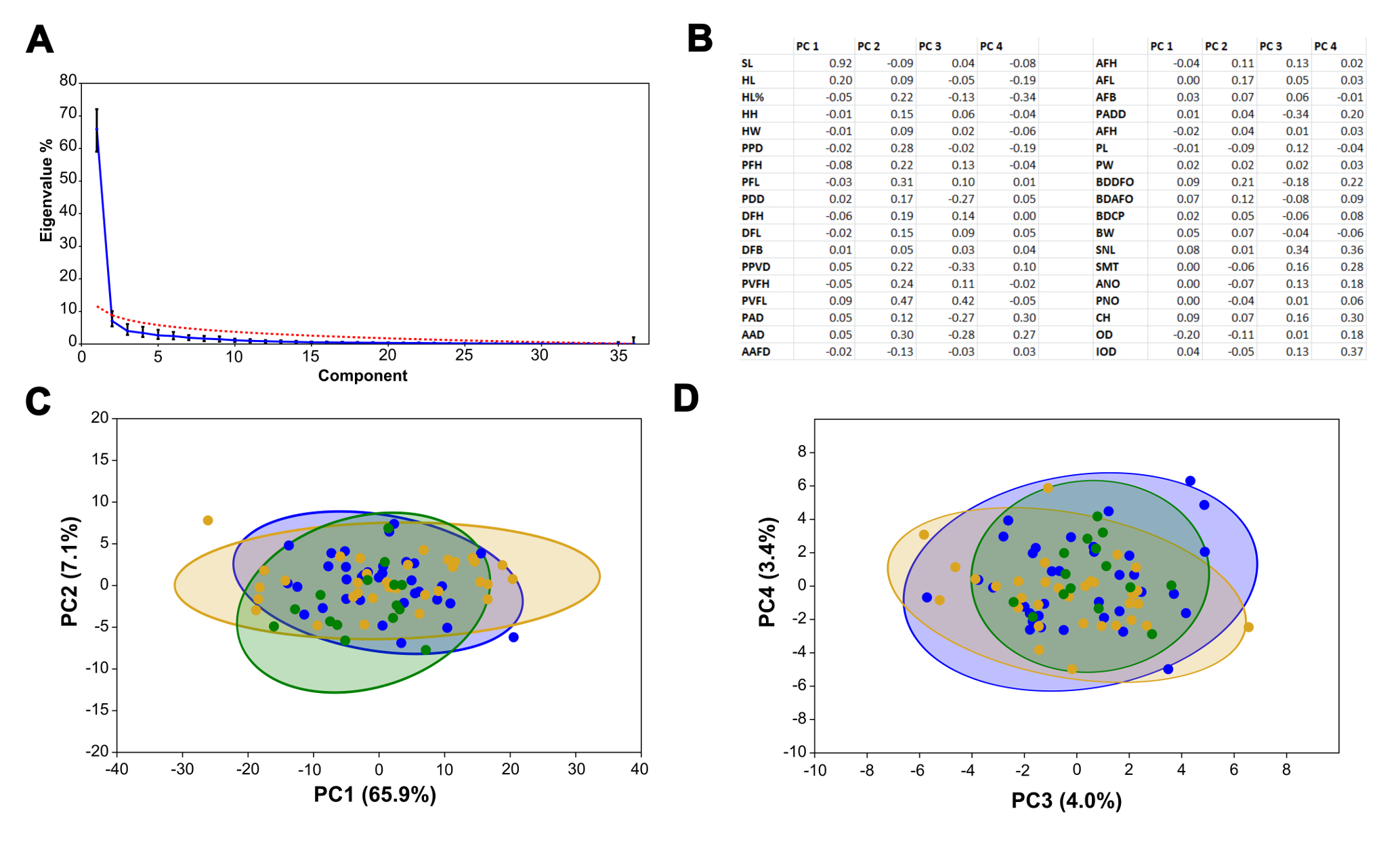
Principal Component Analysis. A consensus of scree plot and broken stick methods, resulted in the selection of four relevant components: PC1 (65.9%), PC2 (7.1%), PC3 (4.0%) and PC4 (3.4%), which together accounted for 80.4% of the variability (Fig. 6A; Tab. S3A). The plot of the first two components, PC1 vs. PC2 (Fig. 6C), explained 73% of the variability, whereas PC2 vs. PC4 (Fig. 7D) explained 7.4%. Both plots suggested no significant differences in overall morphology among specimens from Salí, Bermejo, and Juramento-Salado basins. The loadings of measurements are presented in Fig. 6B and Tab. S3B.
FIGURE 6| Principal component analysis (PCA) of morphometric variables for specimens of Characidium borellii from the Salí (blue), Bermejo (yellow/orange) and Juramento-Salado (green) basins. A. The curves start to flatten out after the five-six components (scree plot method – blue line) and to two-three components (broken stick model – red line). B. Measurement loadings for relevant components C. PC1 vs. PC2, showing no significant differences between the specimens of the different basins. D. PC3 vs. PC4, showing no significant differences between the specimens of the different basins.
FIGURE 7| Distribution map of Characidium borellii in Northwestern Argentina. Blue spots represent specimens from the río Salí basin, green spots from the río Juramento-Salado basin and orange spots from the río Bermejo basin. The red star indicates the type-locality: San Pablo, Tucumán, Argentina.
Intraspecific variability. The PERMANOVA test did not detect statistically significant morphological differences among specimens of Characidium borellii from the Bermejo, Juramento-Salado and Salí basins.
Sexual dimorphism. The PERMANOVA test revealed significant statistical morphological differences among males, females, and juveniles. The SIMPER test indicated that the following variables contributed cumulatively to over 80% of the observed dissimilarity: standard length, orbital diameter, pelvic-fin length, head length, snout length, body depth at dorsal-fin origin, anal apex distance, check, dorsal-fin height, and interorbital distance (Tab. S3C). Kruskal-Wallis and pairwise Mann-Whitney tests identified significant differences in several variables: pelvic-fin length (males > females), orbital diameter (juveniles > males and females), head length (males > females), anal apex distance (juveniles < males and females), check (juvenile < males and females), body depth at the dorsal-fin origin (males > females), and dorsal-fin height (juveniles > males and females).
Geographical distribution. Characidium borellii is distributed across the upper basins of the río Salí-Dulce, Bermejo, and río Juramento-Salado in Northwestern Argentina (NOA). This species has been recorded in the provinces of Tucumán, Catamarca, Salta, Jujuy, and Santiago del Estero (Fig. 7). In the Salí and Juramento-Salado basins, C. borellii occurs sympatrically with C. rachovii.
Molecular analysis. The final COI and cytB alignments were 654 and 747 base pairs (bp), respectively.
Parsimony analysis. Under parsimony, the consensus tree (K reference = 12, length = 2130, fit = 79.82753) of 15 most parsimonious trees (MPTs) using the COI marker (Fig. S5A) recovered C. borellii from the Salí. Bermejo, and Juramento-Salado basins as a highly supported monoplyletic group (GC value = 99). This clade was sister to a group comprising C. bahiense, C. lagosantense, and C. samurai (Fig. 8). Specimens of C. cf. zebra from Chocó region and Maroni basin (near to C. zebra distribution) formed a highly supported monophyletic clade (GC value = 100), more closely related to C. etheostoma, C. nana, C. cf. steindachneri,and C. cf. tapuia than to C. borellii. Sequences of C. rachovii from río Salí basin and C. pterostictum from río Paraná basin clustered with their respective conspecifics, supporting their identification. A single MPT from combined COI and cytB (K reference = 12, length = 2608, fit = 91.14823) revealed a topology consistent with the COI-only analysis (Fig. 9A).
FIGURE 8| COI + cytB markers under extended implied weighting (K reference = 12, length = 2608, fit = 91.14823), GC supporting values generated from 10,000 replicates.
FIGURE 9| Clade I or “Nanognathus”. A. Clade obtained using COI marker under extended weighting, parsimony (K = 12). B. Clade obtained using COI marker under maximum likelihood (ML). C. Clade obtained using COI and cytB under extended weighting, parsimony (K = 12). Black dots, high support (60–100 GC); grey dots, medium support (30–59 GC); white dots, low support (0–29 GC), red dots, high statistical support (≥ 95%); red triangle, sequences of Characidium borellii.
Maximum Likelihood analysis. The phylogenetic tree (TIM2+F+I+G4 model, Log-likelihood = -9310.2156, BIC = 19891.1597) exhibited a topology similar to parsimony (Fig. S5B). Specimens of Characidium borellii from the Salí, Bermejo, and Juramento-Salado basins formed a monophyletic clade with high statistical support (bootstrap value ≥ 95%) (Fig. 9B). Characidium borellii was found to be more closely related to C. samurai, C. lagosantense,and C. bahiense than to taxa in the C. zebra complex. Seven of the nine main clades mentioned by Serra et al. (2025) were recovered across all analyses: “Characidium cf. crandellii”, “Characidium”, “Characidium alipioi” “Characidium caucanum”, “Poecilosomatops”, “Chorimycterus”, and “Characidium bimaculatum”. The “Chorimycterus” and “Characidium pellucidum” cladeswere not recovered by parsimony under low K values (K = 3). Topologies among analyses were not entirely congruent, and most major clades showed low support. Characidium borellii was placed within the “C. bimaculatum” clade, which is hereby renamed the “Nanognathus” clade to reflect the historical placement of C. borellii as the first and only species originally described within the genus Nanognathus (Fig. 9A).
Genetic distances. The between-group mean distances using the COI marker showed over 1.6% divergence between C. borellii and other species of Characidium, exceeding the maximum intraspecific distance (Tab. 2). Species within “Nanognathus” clade, such as C. bahiense, C. bimaculatum, C. cf. zebra (from Paraná and São Francisco), C. deludens, C. lagosantense, C. samurai, and C. varii, exhibited interspecific distances ranging from 5.0% to 8.5% compared to C. borellii. Greater divergences with Characidium borellii were observed for morphologically distinct species, such as: C. fasciatum (23.1%, a species with unscaled isthmus)and C. rachovii (16.0%, a sympatric species of C. borellii). Sequences of C. cf. zebra species showed genetic distances ranging from 5.6% to 19.2%.Theintraspecific genetic distance within C. borellii was less than 0.6%. Characidium rachovii and C. pterostictum showed intraspecific distances of 0.3% and 1.6%, respectively.
TABLE 2 | Interspecific and intraspecific genetic distances of Characidium species under K2 substitution model.
| Species | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | Intraespecific |
1 | C. borellii | – |
|
|
|
|
|
|
|
|
|
|
|
|
|
| 0.6 |
2 | C. deludens | 5.0 | – |
|
|
|
|
|
|
|
|
|
|
|
|
| 0.0 |
3 | C. bimaculatum | 5.4 | 1.2 | – |
|
|
|
|
|
|
|
|
|
|
|
| 0.1 |
4 | C. cf. zebra Paraná | 5.6 | 1.3 | 1.6 | – |
|
|
|
|
|
|
|
|
|
|
| 1.1 |
5 | C. cf. zebra Sao Francisco | 5.7 | 1.4 | 1.5 | 1.8 | – |
|
|
|
|
|
|
|
|
|
| 0.1 |
6 | C. varii | 6.3 | 4.5 | 4.4 | 4.7 | 4.8 | – |
|
|
|
|
|
|
|
|
| 0.0 |
7 | C. bahiense | 6.6 | 6.2 | 6.6 | 6.5 | 6.3 | 6.9 | – |
|
|
|
|
|
|
|
| 0.2 |
8 | C. lagosantense | 8.0 | 5.6 | 6.0 | 5.9 | 5.9 | 7.5 | 3.5 | – |
|
|
|
|
|
|
| 0.0 |
9 | C. samurai | 8.5 | 6.5 | 7.0 | 6.7 | 6.4 | 8.3 | 6.7 | 6.5 | – |
|
|
|
|
|
| 0.0 |
10 | C. cf. zebra Tocantins | 12.8 | 13.0 | 14.2 | 14.0 | 14.2 | 13.6 | 14.6 | 14.7 | 15.3 | – |
|
|
|
|
| 0.0 |
11 | C. rachovii | 16.0 | 14.1 | 14.5 | 14.2 | 13.6 | 14.4 | 12.8 | 13.3 | 14.1 | 12.6 | – |
|
|
|
| 0.3 |
12 | C. cf. zebra Choco | 17.7 | 15.8 | 16.5 | 16.9 | 16.1 | 15.9 | 18.4 | 17.3 | 15.5 | 17.9 | 19.7 | – |
|
|
| 0.0 |
13 | C. cf. zebra Maroni | 19.2 | 17.1 | 17.9 | 18.4 | 18.1 | 17.8 | 18.8 | 19.0 | 17.8 | 18.7 | 19.2 | 5.5 | – |
|
| 0.1 |
14 | C. pterostictum | 20.3 | 20.0 | 21.2 | 20.0 | 20.3 | 17.5 | 19.1 | 18.3 | 19.6 | 20.4 | 19.8 | 15.9 | 15.6 | – |
| 1.6 |
15 | C. fasciatum | 23.1 | 21.8 | 22.7 | 21.4 | 21.9 | 20.4 | 20.4 | 18.8 | 19.2 | 19.5 | 20.0 | 19.2 | 20.3 | 15.1 | – | 0.0 |
16 | P. weitzmani | 40.6 | 37.6 | 37.7 | 37.6 | 37.7 | 36.7 | 34.5 | 33.7 | 34.1 | 31.6 | 34.7 | 35.9 | 36.6 | 31.2 | 32.0 | n/c |
Material examined. All from Argentina, river basins:Salí:MZUT P 2563, 1, 67.3 mm SL, holotype, (photographs in Zarske, Géry, 2001). CI FML 3865, 103, 42.9–53.6 mm SL. CI FML 7822, 1, 43.3 mm SL. CI FML 7862, 2, 51.0–52.6 mm SL. CI FML 7835, 4, 33.75–45.62 mm SL. CI FML 7924, 6, 41.9–54.6 mm SL. CI FML 8003, 3, 58.1–70.0 mm SL. CI FML 8004, 1, 57.0 mm SL. CI FML 8005, 4, 54.9–63.5 mm SL. CI FML 8179, 3, 37.6–55.8 mm SL. CI FML 8180, 4, 37.5–49.3 mm SL. CI FML 8181, 8, 36.4–45.6 mm SL. CI FML 8182, 3, 47.2–53.6 mm SL. CI FML 8183, 3, 54.9–57.3 mm SL. IBIGEO-I 524, 4, 35.8–50.9 mm SL. Bermejo: CI FML 8006, 1, 63.2 mm SL. CI FML 8007, 22, 69.1–36.2 mm SL. CI FML 8008, 1, 52.7 mm SL. CI FML 8009, 8, 66.9–46.9 mm SL. CI FML 8010, 3, 25.9–41.7 mm SL. CI FML 8011, 45, 33.1–55.7 mm SL. CI FML 8012, 20, 32.7–54.0 mm SL. CI FML 8013, 33, 43.8–60.3 mm SL. CI FML 8015, 1, 46.7 mm SL. CI FML 8184, 1, 47.5 mm SL. CI FML 8185, 1, 49.7 mm SL. CI FML 8186, 1, 32.9 mm SL. CI FML 8192, 20, 45.1–62.5 mm SL. CI FML 8193, 2, 53.4–58.8 mm SL. IBIGEO-I 520, 10, 44.5–51.3 mm SL. IBIGEO-I 521, 12, 27.3–46.9 mm SL. IBIGEO-I 522, 4, 48.1–54.6 mm SL. IBIGEO-I 523, 5, 38.7–63.2 mm SL. Juramento: CI FML 632, 1, 50.3 mm SL. CI FML 6177, 2, 43.2–44.5 mm SL. CI FML 8014, 2, 57.4–54.2 mm. CI FML 8015, 2, 44.4–48.4 SL. Horcones: CI FML 8016, 1, 35.2 mm SL. Urueña:CI FML 8017, 7, 38.1–53.1 mm SL.
Discussion
Historically, limited information has been available regarding the morphology, phylogenetic relationships and distribution of C. borellii, with only a brief redescription by Géry et al. (2001). Several characters described by these authors differ from the findings of this study, including iii,9,i or iii,10,i pectoral-fin rays (vs. iii,9 rays), tricuspid teeth in the dentary and premaxillary (vs. conical teeth), and confirmation of a single row of teeth in the dentary. Additional features with phylogenetic potential, such as hooks on the pelvic and pectoral fins of males (Mirande, 2010; Teixeira, Melo, 2021); swimbladder morphology (Agudelo-Zamora et al., 2020) and pseudotympanum anatomy (Zanata, Camelier, 2014), are also provided.
Secondary sexual characteristics including bony process on the pelvic, also observed by Mirande (2010), and pectoral fins, and longer pelvic fin in males, are recorded in C. borellii. These dimorphic characters are also found in several other species of Characidium (Teixeira, Melo, 2021): bony processes on paired fins in C. bahiense, C. barbosai, C. bimaculatum, C. clistenesi, C. deludens (only on pelvic-fin rays), C. lagosantense, C. laterale, C. litorale, C. mirim, C. nupelia, C. satoi, C. stigmosum, C. xanthopterum, C. xavante; and longer pelvic fins in males of C. deludens, C. kamakan Zanata & Camelier, 2015, C. rachovii, C. orientale, and C. vestigipinne. The combination of having both characters appears to be unique in C. borellii.
The genus Characidium is diagnosed by the presence of a black basicaudal spot (Buckup, 1993b), a character also present in C. borellii, despite some reversions within the genus. Buckup (1993b) recovered three major clades within Characidium: C1, C4, and C5. Clade C1 (C2+C3) comprises species (e.g., C. fasciatum and C. gomesi) characterized by an unscaled isthmus, reduced or absent poscleithrum 1, and reduction or absence of the cranial fontanel. Clade C4 includes species (e.g., C. rachovii, C. occidentale, and C. laterale) defined by three synapomorphies (Netto-Ferreira et al., 2013; Mendonça, Netto-Ferreira, 2015): absence of the parietal branch of the supraorbital canal, more than 12 transversal bars, and absence of inner row of dentary teeth. Clade C5 includes species with thin transversal bars. Characidium borellii only shares the absence of inner teeth with species in C4 and does not clearly conform to any of these morphological clades.
Most recent phylogenetic analyses using mitochondrial markers (COI) recognize different lineages or clades (Oliveira-Silva et al., 2024; Serra et al., 2025) but with distinct relationships among them. According to these clades, we recovered C. borellii in the “Nanognathus” clade (= “C. bimaculatum” clade from Serra et al., 2025 or CI clade from Oliveira-Silva et al., 2024), grouped with C. bahiense, C. bimaculatum, C. cf. zebra (from Paraná and São Francisco basins), C. deludens, C. lagosantense, C. samurai, and C. varii. Genetic distances are consistent with the close phylogenetic relationships among these species. Combining the morphology data, C. borellii appears most closely related to C. bimaculatum and C. deludens, sharing several features: the presence of hooks on the pelvic fins, a muscular hiatus of the pseudotympanum formed by a single aperture, absence of an inner row of dentary teeth (in C. deludens), and the presence of parietal branch of supraorbital canal (in C. deludens). A combined phylogenetic analysis integrating both morphological and molecular data with a broader range of species is needed to confirm these relationships.
Specimens previously identified as Characidium cf. zebra or C. fasciatum from Sali, Bermejo and Juramento-Salado basins (Liotta, 2005; Mirande, Aguilera, 2009; Cancino, Aguilera, 2016; Terán et al., 2016; Alonso et al., 2025) were misidentified and correspond to C. borellii. The molecular and morphological data showed no significant differences among these populations, with high morphological variation attributed to sexual dimorphism or allometry. Therefore, Characidium borellii represents a valid species within the genus Characidium, with a distribution restricted to Northwestern Argentina.
Comparative material examined. Comparative material is listed in Serra et al. (2025), additional comparative material: Characidium pterostictum: Argentina: CI FML 8189, 11, 26.6–54.9 mm SL. CI FML 8190, 31, 33.5–63.1. CI FML 8130, 6, 34.5–52.5. Characidium rachovii: CI FML 5372, 1, 28.2 mm SL.CI FML 8018, 8, 36.4–25.4 mm SL. CI FML 8191, 5, 18.2–21.5 mm SL. MG-ZV-P 2870 (ex-INALI 832), 8, 32.7–34.9 mm SL. Characidium serrano: CI FML 8105, 7, 32.7–52.5 mm SL. CI-FML 8106, 24, 31.1–47.9 mm SL. Characidium tenue: CI FML 8187, 1, 29.9 mm SL. CL FML 8188, 1, 29.1 mm SL. Characidium aff. zebra: CI FML 1739, 14, 13.6–34.7 mm SL. CI FML 5175, 47, 22.9–29 mm SL.CI FML 7680, 5, 26.7–37.2 mm SL. Brazil: Characidium alipioi: MZUSP 109873, 1, 78.6 mm SL. MZUSP 79427, 25, 70.0-86.2 mm SL. Characidium bimaculatum: MZUSP 68959, 3 (1cs), 20.2–25.3 mm SL. Characidium deludens: MZUSP 115009, 1, 48.15 mm SL, holotype. MZUSP 115010, 9, 27.01–40.95 mm SL, paratypes. Characidium fasciatum: MZUSP 37067, 2, 42.4–48.8 mm SL. MZUSP 85981, 13, 21.4–35.6 mm SL. MZUSP 85995, 32 (1cs), 22.6–26.3 mm SL. Characidium gomesi:MZUSP 110032, 3, 31.1 mm SL. MZUSP 110447, 6, 32.9–59.3 mm SL. MZUSP 88349, 6, 31.2–38.7 mm SL. Characidium lagosantense: MZUSP 73708, 2, 19.3–23.0 mm SL. Characidium lanei:MZUSP 114844, 11, 28.9–46.3 mm SL. MZUSP 120519, 1, 41.8 mm SL. Characidium cf. lanei: CI FML 2133, 2, 50.2–67.0 mm SL. Characidium lauroi: MZUSP 110359, 5, 30.8–45.0 mm SL. MZUSP 124033, 12, 37.4–49.1 mm SL. Characidium litorale:MZUSP 129926, 2, 18.8–25.0 mm SL. MZUSP 129946, 1, 34.3 mm SL. Characidium nana: MZUSP 125919, 10, 15.8–20.7 mm SL. Characidium nupelia:MZUSP 87743, 1, 28.96 mm SL, holotype. MZUSP 87742, 20, 21.82–28.95 mm SL, paratypes. Characidium oiticicai:MZUSP 110245, 14, 23.2–55.7 mm SL. MZUSP 87716, 3, 34.0–44.8 mm SL. Characidium onca: MZUSP 125807, 1, 39.7 mm SL, holotype. MZUSP 125803, 6 (2 cs), 26.8–30.5 mm SL, paratypes. Characidium schubarti:MZUSP 71023, 3, 34.2–42.7 mm SL. Characidium stigmosum:MZUSP 40804, 1, 27.9–35.5 mm SL, paratype. Characidium travassosi: MZUSP 85940, 1, 33.7 mm SL, holotype. MZUSP 85938, 6 (1 cs), 32.6–33.3 mm SL, paratypes. Characidium xanthopterum:MZUSP 41454, 20, 21.6–31.3 mm SL. MZUSP 117743, 5, 30.8–39.3 mm SL. MZUSP 117946, 1, 33.6 mm SL. Characidium boavistae: Colombia: IAvH-P-11801, 19, 38.1–49.8 mm SL. IAvH-P-21619, 3, 29.8-38.5 mm SL. IAvH-P-23640, 8, 33.9–44.1 mm SL. IAvH-P-29935, 1, 40.9 mm SL. IAvH-P-9760, 16, 37.5–49.4 mm SL. IAvH-P-9767, 26, 31.0–46.4 mm SL. Characidium caucanum: IAvH-P-1520, 1, 43.8 mm SL. Characidium crandellii: IAvH-P-20573, 3, 38.7–41.7 mm SL. Characidium dule:IAvH-P-13334, 42, 28.2–42.1 mm SL. Characidium phoxocephalum:IAvH-P-8432, 1, 53.9 mm SL. IAvH-P-8434, 1, 89.7 mm SL. Melanocharacidium dispilomma: IAvH-P-12885, 27, 21.2–31.6 mm SL. IAvH-P-26305, 6, 27.5–33.8 mm SL. Characidium zebra: Guyana: FMNH 53547, 52.5 mm SL, holotype (photographs). FMNH 52766, 4, 30–34 mm SL, paratypes (photographs and radiograph).
Acknowledgments
Thanks are due to the Secretaría de Ambiente y Desarrollo Sustentable of Salta province, particularly Yanina Bonduri and Sebastián Musalem, for granting the necessary collecting permits. We would like to express our gratitude to IBIGEO and Fundación Miguel Lillo for their ongoing support. To Killifish Foundation members and to Stefan Koerber, Fernando Lobo, Baltazar Bugeau, Roberto Sanchez, Julio Cruz and Virginia Martínez for their permanent support. To Alejandro Méndez-López for the photographs of the specimens. To Ana S. Méttola for help with the image edition. To Jon Fong and Phil Willink for providing photographs of type-specimens of Characidium zebra. TNT was provided free by the Willi Hennig Society. This work benefited from valuable suggestions and comments of Marcelo R. S. Melo.
References
Agudelo-Zamora HD, Tavera J, Murillo YD, Ortega-Lara A. The unknown diversity of the genus Characidium (Characiformes: Crenuchidae) in the Chocó biogeographic region, Colombian Andes: two new species supported by morphological and molecular data. J Fish Biol. 2020; 97(6):1662–75. https://doi.org/10.1111/jfb.14527
Almeida VG. Descricão de uma nova espécie do gênero Characidium (Pisces, Characidae). Pap Avulsos Zool.1971; 25(14):111–19. https://doi.org/10.11606/0031-1049.1971.25.p111-119
Almirón A, Casciotta J, Ciotek L, Giorgis P. Guía de los peces del Parque Nacional Pre-delta. Ciudad Autónoma de Buenos Aires: Administración de Parques Nacionales; 2015.
Alonso F, Mirande JM, Pandolfi M. Gross anatomy and histology of the alimentary system of Characidae (Teleostei: Ostariophysi: Characiformes) and potential phylogenetic information. Neotrop Ichthyol. 2015; 13(2):273–86. https://doi.org/10.1590/1982-0224-20140107
Alonso F, Terán GE, Aguilera G, Montes MM, Serra Alanís WS, Calviño P et al. Integrative phylogeny of Corydoradinae (Siluriformes: Callichthyidae) with an emphasis on northwestern La Plata species, including descriptions of a new genus and species. Zool Anz. 2025; 314:10–23. https://doi.org/10.1016/j.jcz.2024.11.006
Armbruster JW, Lujan NK, Bloom DD. Redescription of the Guiana Shield darter species Characidium crandellii and C. declivirostre (Crenuchidae) with descriptions of two new species. Ichthyol Herpetol. 2021; 109(1):102–22. https://doi.org/10.1643/i2019299
Boulenger GA. Viaggio del dottor Alfredo Borelli nella Repubblica Argentina e nel Paraguay: XII – Poissons. Bollett Mus Zool Anat Comp R Univ Torino. 1895; 10(196):1–03. Available from: https://biostor.org/reference/206977
Breder CM. New loricariate, characin, and poeciliid fishes from the rio Chucunaque, Panama.Am Mus Novit. 1925; 180:1–09. Available from: http://hdl.handle.net/2246/4358
Buckup PA. Redescription of Characidium fasciatum, type species of the Characidiinae (Teleostei, Characiformes). Copeia. 1992; 1992(4):1066–73. https://doi.org/10.2307/1446639
Buckup PA. Phylogenetic interrelationships and reductive evolution in Neotropical characidiin fishes (Characiformes, Ostariophysi). Cladistics. 1993a; 9(3):305–41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
Buckup PA. Review of the characidiin fishes (Teleostei: Characiformes), with description of four new genera and ten new species. Ichthyol Explor Freshw. 1993b; 4(2):97–154
Buckup PA, Hahn L. Characidium vestigipinne: a new species of Characidiinae (Teleostei, Characiformes) from Southern Brazil. Copeia. 2000; 2000(1):150–55. https://doi.org/10.1643/0045-8511(2000)2000[0150:CVANSO]2.0.CO;2
Buckup PA, Reis RE. Characidiin genus Characidium (Teleostei, Characiformes) in southern Brazil, with description of three new species. Copeia. 1997; 1997(3):531–48. https://doi.org/10.2307/1447557
Butí C, Cancino F. Ictiofauna de la cuenca endorreica del río Salí-Dulce, Argentina. Acta Zool Lilloana. 2005; 49(1–2):9–33.
Butí C, Miquelarena AM. Ictiofauna del río Salí superior, departamento Trancas, Tucumán, República Argentina. Acta Zool Lilloana. 1995; 43(1):21–44.
Cancino F, Aguilera G. Ictiofauna. In: Antelo C, Bulacio E, Cancino F, Marigliano N, Peralta M, Ramallo G, Romero F, editors. Biodiversidad y fronteras: Cuenca del río Bermejo (Salta, Argentina). Tucumán: Fundación Miguel Lillo; 2016. p.40–44. Available from: https://www.lillo.org.ar/revis/cnaturaleza/2016-scn-21.pdf
Casciotta JR, Almirón AE, Ciotek L, Giorgis P, Říčan O, Piálek L et al. Visibilizando lo invisible: un relevamiento de la diversidad de peces del Parque Nacional Iguazú, Misiones, Argentina. Hist Nat. 2016; 6(2):5–77.
Casciotta J, Almirón A, Doubnerová K, Piálek L, Říčan O. First records of Characidium heirmostigmata and C. serrano (Characiformes: Crenuchidae) from freshwaters of Argentina. ICP. 2015; 34:1–03.
Cattel RB. The scree test for the number of factors. Multivariate Behav Res. 1966; 1(2):245–76. https://doi.org/10.1207/s15327906mbr0102_10
Cope ED. On the fishes of the Ambyiacu River. Proc Acad Nat Sci Phila.1871; 23(3):250–94.
Cortés Hernández MÁ, Terán G, Méndez López A, Aguilera G. Fishes of the Parque Nacional Calilegua, Jujuy, Argentina. Peces del Parque Nacional Calilegua, Jujuy, Argentina. Hist Nat. 2025; 15(2):5–17.
Dahl G. New fresh-water fishes from Western Colombia. Caldasia. 1960; 8(39):451–84.
Díaz J, Villanova GV, Brancolini F, del Pazo F, Posner VM, Grimberg A et al. First DNA barcode reference library for the identification of South American freshwater fish from the lower Paraná river. PLoS ONE. 2016; 11(7):e0157419. https://doi.org/10.1371/journal.pone.0157419
Edgar RC. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004; 32(5):1792–97. https://doi.org/10.1093/nar/gkh340
Eigenmann CH. Reports on the expedition to British Guiana of the Indiana University and the Carnegie Museum, 1908: Report no 1 – Some new genera and species of fishes from British Guiana. Ann Carnegie Mus. 1909; 6(1):4–54. https://doi.org/10.5962/p.331035
Flausino NJ, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique mid-water behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350–60. https://doi.org/10.11646/zootaxa.4816.3.5
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Sciences; 2025. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Frontier S. Étude de la décroissance des valeurs propres dans une analyse en composantes principales: Comparaison avec le modd́le du bâton brisé. J Exp Mar Biol Ecol. 1976; 25(1):67–75. https://doi.org/10.1016/0022-0981(76)90076-9
Géry J, Zarske A, Paepke H-J. Supplementary descriptions of two overlooked fish taxa, Leporinus macrolepidotus Peters, 1868 and Nanognathus borellii Boulenger, 1895, actually members of the genus Characidium Reinhardt, 1866 (Teleostei: Characiformes: Characidiidae). Zool Abh Staat Tierk Dresden. 2001; 51(3):23–31.
Goloboff PA. Extended implied weighting. Cladistics. 2014; 30(3):260–72. https://doi.org/10.1111/cla.12047
Goloboff PA, Farris JS, Källersjö M, Oxelman B, Ramiréz MJ, Szumik CA. Improvements to resampling measures of group support. Cladistics. 2003; 19(4):324–32. https://doi.org/10.1111/j.1096-0031.2003.tb00376.x
Goloboff PA, Farris JS, Nixon KC. TNT, a free program for phylogenetic analysis. Cladistics. 2008; 24(5):774–86. https://doi.org/10.1111/j.1096-0031.2008.00217.x
Graça WJ, Ota RR, Domingues WM. A new species of miniature Characidium (Characiformes: Crenuchidae) from the upper Rio Paraguay basin, Mato Grosso state, Brazil. J Fish Biol. 2019; 95(6):1480–85. https://doi.org/10.1111/jfb.14169
Graça WJ, Pavanelli CS. Characidium heirmostigmata, a new characidiin fish (Characiformes: Crenuchidae) from the upper rio Paraná basin, Brazil. Neotrop Ichthyol. 2008; 6(1):53–56. https://doi.org/10.1590/S1679-62252008000100006
Hammer Ø, Harper DA, Ryan PD. PAST: Paleontological statistics software package for education and data analysis. Palaeontol Electron. 2001; 4(1):4.
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS. UFBoot2: Improving the ultrafast bootstrap approximation. Mol Biol Evol. 2018; 35(2):518–22. https://doi.org/10.1093/molbev/msx281
Huxley JS. Problems of relative growth.Johns Hopkins University Press; 1993.
Kalyaanamoorthy S, Minh BQ, Wong TKF, von Haeseler A, Jermiin LS. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat Methods. 2017; 14(6):587–89. https://doi.org/10.1038/nmeth.4285
Kilmer JT, Rodríguez RL. Ordinary least squares regression is indicated for studies of allometry. J Evol Biol. 2017; 30(1):4–12. https://doi.org/10.1111/jeb.12986
Klingenberg CP. Multivariate allometry. In: Marcus LF, Corti M, Loy A, Naylor GJP, Slice DE, editors. Advances in morphometrics. Boston: Springer; 1996. p.23–49. https://doi.org/10.1007/978-1-4757-9083-2_3
Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. 2018; 35(6):1547–49. https://doi.org/10.1093/molbev/msy096
Leitão RP, Buckup PA. A new species of Characidium (Characiformes: Crenuchidae) from coastal basins of Serra do Mar, southeastern Brazil. Copeia. 2014; 2014(1):14–22. https://doi.org/10.1643/CI-12-137
Liotta J. Distribución geográfica de los peces de aguas continentales de la República Argentina. Serie Documentos n° 3, ProBiota; 2005. Available from: http://sedici.unlp.edu.ar/bitstream/handle/10915/10949/Documento_completo.pdf?sequence=1&isAllowed=y
Loureiro M, González-Bergonzoni I, Mello F. Peces de agua dulce de Uruguay. Facultad de Ciencias, Udelar; 2023.
Loureiro M, Sá R, Serra SW, Alonso F, Lanés LEK, Volcan MV et al. Review of the family Rivulidae (Cyprinodontiformes, Aplocheiloidei) and a molecular and morphological phylogeny of the annual fish genus Austrolebias Costa, 1998. Neotrop Ichthyol. 2018; 16(3):e180007. https://doi.org/10.1590/1982-0224-20180007
Melo MRS, Buckup PA. Characidium stigmosum (Characiformes: Crenuchidae): a new species of characidiin fish from central Brazil. Ichthyol Herpetol. 2002; 2002(4):988–93. https://doi.org/10.1643/0045-8511(2002)002[0988:CSCCAN]2.0.CO;2
Melo MRS, Espíndola VC. Description of a new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) from the Chapada Diamantina, Bahia, and redescription of Characidium bimaculatum Fowler, 1941. Zootaxa.2016; 4196(4):552–68. https://doi.org/10.11646/zootaxa.4196.4.5
Melo MRS, Oyakawa OT. A new species of Characidium Reinhardt (Characiformes, Crenuchidae) with a distinctively dimorphic male. Copeia, 2015; 103(2):281–89. https://doi.org/10.1643/CI-14-073
Mendonça MB, Netto-Ferreira AL. New species of Characidium (Characiformes: Crenuchidae) from the rio Tapajós and rio Xingu drainages, Pará, Brazil. Zootaxa. 2015; 4021(1):187–94. https://doi.org/10.11646/zootaxa.4021.1.9
Mirande M. Weighted parsimony phylogeny of the family Characidae (Teleostei: Characiformes). Cladistics. 2009; 25(6):574–613. https://doi.org/10.1111/j.1096-0031.2009.00276.x
Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes): From characters to taxonomy. Neotrop Ichthyol. 2010; 8(3):385–568. https://doi.org/10.1590/S1679-62252010000300001
Mirande JM, Aguilera G. Los peces de la selva pedemontana del noroeste argentine. In: Brown AD, Blendinger PG, Lomáscolo T, Bes PG, editors. Selva Pedemontana de las Yungas: historia natural, ecología y manejo de un ecosistema en peligro. Tucumán: Ediciones del Subtrópico; 2009. p.169–211.
Mirande JM, Koerber S. Checklist of the freshwater fishes of Argentina. ICP. 2020; 72:1–181.
Netto-Ferreira AL, Birindelli JLO, Buckup PA. A new miniature species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from the headwaters of the rio Araguaia, Brazil. Zootaxa. 2013; 3664(3):361–68. https://doi.org/10.11646/zootaxa.3664.3.6
Oliveira-Silva L, Batalha-Filho H, Camelier P, Zanata AM. Underestimated diversity in Characidium (Characiformes: Crenuchidae) from Neotropical rivers revealed by an integrative approach. Syst Biodivers. 2024; 22(1):2346510. https://doi.org/10.1080/14772000.2024.2346510
Palumbi SR, Martin A, Romano S, McMillan WO, Stice L, Grabowski G. The simple fool’s guide to PCR. Honolulu: University of Hawaii; 2002. Available from: https://stacks.stanford.edu/file/druid:yh393jm6703/Simple_Fool%27s_Master%20PCR.pdf
Pasquini AI, Depetris PJ. Discharge trends and flow dynamics of South American rivers draining the southern Atlantic seaboard: an overview. J Hydrol. 2007; 333(2–4):385–99. https://doi.org/10.1016/j.jhydrol.2006.09.005
Pellegrin J. Characinidés américains nouveaux de la collection du Muséum d’Histoire naturelle. Bull Mus Natl Hist Nat. 1908; 14(7):342–47.
Peixoto LAW, Wosiacki WB. A new species of Characidium (Characiformes: Crenuchidae) from the lower Amazon. Copeia. 2013; 2013(1):52–57. https://doi.org/10.1643/CI-12-080
Reinhardt JT. Om trende, formeentligt ubeskrevne fisk af Characinernes eller karpelaxenes familie. Overs K Dansk Vidensk Selsk Forth. 1866; 49–68.
Ringuelet RA, Arámburu RH, Alonso de Arámburu AS. Los peces de agua dulce de la República argentina. La Plata: CIC; 1967.
Saravia AN, Méttola VE, Aguilera G, Cano PD, Terán GE. Do we know what we are conserving? Fishes from Parque Nacional Aconquija, Tucumán, northwestern Argentina. Acta Zool Lilloana. 2023; 67(2):327–47.
Schultz LP. The fishes of the family Characinidae from Venezuela, with descriptions of seventeen new forms. Proc US Natl Mus. 1944; 95(3181):235–367.
Serra S, Bessonart J, Mello FT, Duarte A, Malabarba L, Loureiro M. Peces del río Negro. Montevideo: MGAP-DINARA; 2014.
Serra W, Scarabino F, Méttola VE, Montes MM, Terán ET, Moncada M et al. Characidium serrano Buckup & Reis, 1997 (Characiformes, Crenuchidae): new insights into its phylogenetic relationships and distribution. Acta Zool Lilloana. 2025; 69(1):1–28. http://dx.doi.org/10.30550/j.azl/2097
Silveira LGG, Langeani F, Graça WJ, Pavanelli CS, Buckup PA. Characidium xanthopterum (Ostariophysi: Characiformes: Crenuchidae): a new species from the Central Brazilian Plateau. Neotrop Ichthyol. 2008; 6(2):169–74. https://doi.org/10.1590/S1679-62252008000200003
Souza-Shibatta L, Tonini JFR, Abrahão VP, Jarduli LR, Oliveira C, Malabarba LR et al. Reappraisal of the systematics of Microglanis cottoides (Siluriformes, Pseudopimelodidae), a catfish from southern Brazil. PLoS ONE. 2018; 13(7):e0199963. https://doi.org/10.1371/journal.pone.0199963
Stabile BHM, Reis RB, Frota A, Graça WJ, Oliveira AV. Morphology and molecular evidence of a new species of Characidium (Characiformes: Crenuchidae) from the edges of a protected area at rio Itararé, upper rio Paraná, southern Brazil. J Fish Biol. 2024; 105(6):1850–61. https://doi.org/10.1111/jfb.15935
Stabile BHM, Reis RB, Oliveira AV, Graça WJ. A new species of Characidium (Characiformes: Crenuchidae) from the Iguaçu National Park, rio Iguaçu basin, Paraná, Brazil. J Fish Biol. 2025; 107(2):454–65. https://doi.org/10.1111/jfb.70035
Taphorn DC. The characiform fishes of the Apure river drainage, Venezuela. [PhD. Thesis]. University of Florida; 1990.
Taphorn DC, Montaña CG, Buckup PA. Characidium longum (Characiformes: Crenuchidae), a new fish from Venezuela. Zootaxa. 2006; 1247(1):1–12.https://doi.org/10.11646/zootaxa.1247.1.1
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Teixeira TF, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) from the río Huallaga, central Peruvian Amazon, with a review on secondary sexual characters in the genus. J Fish Biol. 2021; 98(1):178–88. https://doi.org/10.1111/jfb.14568
Tenchini AS. Evaluación de la creciente 1983/84 en la cuenca del río Salado en la provincia de Santiago del Estero. Consejo Federal de Inversiones; 1984.
Terán GE, Jarduli L, Alonso F, Mirande JM, Shibatta OA. Microglanis nigrolineatus, a new species from northwestern Argentina (Ostariophysi: Pseudopimelodidae). Ichthyol Explor Freshw. 2016; 27(3):193–202.
Trifinopoulos J, Nguyen L-T, von Haeseler A, Minh BQ. W-IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res. 2016; 44(W1):232–35. https://doi.org/10.1093/nar/gkw256
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PDN. DNA barcoding Australia’s fish species. Philos Trans R Soc Lond B Biol Sci. 2005; 360(1462):1847–57. https://doi.org/10.1098/rstb.2005.1716
Zanata AM, Camelier P. A new species of Characidium (Characiformes: Crenuchidae) from small coastal drainages in northeastern Brazil, with remarks on the pseudotympanum of some species of the genus. Neotrop Ichthyol. 2014; 12(2):333–42. https://doi.org/10.1590/1982-0224-20130168
Zanata AM, Camelier P. Two new species of Characidium Reinhardt (Characiformes: Crenuchidae) from northeastern Brazilian coastal drainages. Neotropl Ichthyol. 2015; 13(3):487–98. https://doi.org/10.1590/1982-0224-20140106
Zanata AM, Oliveira C, Oliveira-Silva L. Integrative taxonomy reveals a new species of Characidium (Characiformes: Crenuchidae) shared by tributaries of upper Tapajós and Xingu river basins, Brazil. J Fish Biol. 2024; 150(6):1929–38. https://doi.org/10.1111/jfb.15938
Zanata AM, Oliveira-Silva L, Ohara WM. A new sexually dichromatic miniature Characidium (Characiformes: Crenuchidae) from the rio Guaporé, rio Madeira basin, Brazil, with remarkable morphological novelties to the genus. Neotrop Ichthyol. 2023; 21(1):e220059. https://doi.org/10.1590/1982-0224-2022-0059
Zanata AM, Ramos TPA, Oliveira-Silva L. A new species of Characidium (Characiformes: Crenuchidae) from the rio Parnaíba basin, northeastern Brazil. Zootaxa. 2018; 4514(1):77–86. https://doi.org/10.11646/zootaxa.4514.1.6
Zanata AM, Ribeiro TC, Araújo-Porto FA, Pessali TC, Oliveira-Silva L. A new species of Characidium (Characiformes: Crenuchidae) from the upper rio São Francisco basin, Minas Gerais, Brazil. Zootaxa. 2020; 4790(3):505–14. https://doi.org/10.11646/zootaxa.4790.3.5
Zarske A. Geryichthys sterbai gen. et spec. nov. und Microcharacidium geryi spec. nov.: Beschreibung einer neuen gattung und zweier neuer arten von bodensalmlern aus dem einzugsgebiet des rio Ucayali in Peru (Teleostei: Ostariophysi: Characiformes: Characidiidae). Zool AbhStaat Tierk Dresden. 1997; 49:157–72.
Zarske A, Géry J. Beschreibung von drei neuen arten der gattung Characidium Reinhardt, 1866 aus Bolivien und Paraguay (Teleostei: Characiformes: Characidiidae). Zool Abh Staat Tierk Dresden. 2001; 51(16):229–46.
Authors
![]() V. Ezequiel Méttola1,
V. Ezequiel Méttola1, ![]() Guillermo E. Terán1
Guillermo E. Terán1 ![]() ,
, ![]() Gastón Aguilera1,
Gastón Aguilera1, ![]() Martin M. Montes2,
Martin M. Montes2, ![]() Marina Ibáñez Shimabukuro2,
Marina Ibáñez Shimabukuro2, ![]() Felipe Alonso3 and
Felipe Alonso3 and ![]() J. Marcos Mirande1
J. Marcos Mirande1
[1] Fundación Miguel Lillo, Unidad Ejecutora Lillo, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Tucumán, Argentina. (VEM) ezequielmettola@gmail.com, (GET) guilloteran@gmail.com (corresponding author), (GA) gaguilera@lillo.org.ar, (JMM) mcmirande@gmail.com.
[2] Centro de Estudios Parasitológicos y Vectores (CEPAVE), Consejo Nacional de Investigaciones Científicas y Técnicas, Universidad Nacional de La Plata, Comision de Investigaciones Cientificas de la Provincia de Buenos Aires (CCT, CONICET-UNLP-CICPBA), La Plata, Buenos Aires, Argentina. (MIS) marinai@cepave.edu.ar, (MMM) martinmiguelmontes@gmail.com.
[3] Instituto de Bio y Geociencias del NOA (IBIGEO), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), and Facultad de Ciencias Naturales, Universidad Nacional Salta (UNSa), Salta Province, Argentina. (FA) felipealonso@gmail.com.
Authors’ Contribution 

V. Ezequiel Méttola: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Guillermo E. Terán: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Gastón Aguilera: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Martin M. Montes: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Marina Ibáñez Shimabukuro: Data curation, Formal analysis, Investigation, Methodology, Validation.
Felipe Alonso: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
J. Marcos Mirande: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
All procedures followed the ethical guidelines and animal welfare regulations established by the Comité Nacional de Ética en la Ciencia y Tecnología, Argentina. The material used in this work comes from specimens deposited in museum collections.
Competing Interests
The author declares no competing interests.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article and in the supplementary material of this article.
Funding
This study was partially funded by the National Scientific and Technical Research Council of Argentina (CONICET) through PIBAA 2872021010 0128CO and a grant from the Fundación Williams “Fondos complementarios para la investigación con impacto en el territorio argentino: Peces de la cuenca del Río Bermejo: biodiversidad, distribución espaciotemporal, efectos de las actividades antrópicas y conservación” to FA, grants from the Agencia Nacional de Promoción de la Investigación, el Desarrollo Tecnológico y la Innovación (ANPCyT) to JMM (PICT–2020–02141) and GA (PICT–2019–1004), and a grant from CONICET to JMM (PIP–2020–0101214).
How to cite this article
Méttola VE, Terán GE, Aguilera G, Montes MM, Shimabukuro MI, Alonso F, Mirande JM. Integrative study of Characidium borellii (Characiformes: Crenuchidae): phylogenetic relationships and geographic distribution. Neotrop Ichthyol. 2025; 23(3):e250022. https://doi.org/10.1590/1982-0224-2025-0022
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted July 19, 2025
Accepted July 19, 2025
![]() Submitted February 7, 2025
Submitted February 7, 2025
![]() Epub November 10, 2025
Epub November 10, 2025