![]() Sara Elizabeth Domínguez-Cisneros1,
Sara Elizabeth Domínguez-Cisneros1, ![]() Ernesto Velázquez-Velázquez1
Ernesto Velázquez-Velázquez1 ![]() ,
, ![]() Omar Domínguez-Domínguez2 and
Omar Domínguez-Domínguez2 and ![]() Rosa Gabriela Beltrán-López3
Rosa Gabriela Beltrán-López3
PDF: EN XML: EN | Supplementary: S1 S2 S3 | Cite this article
Abstract
Se describe una nueva especie de Profundulus de varias localidades de la cuenca del río Grijalva y ríos del Istmo de Tehuantepec, en el sur de México, utilizando una combinación de caracteres morfológicos y moleculares. Genéticamente, la nueva especie se separa de otras especies de Profundulus analizadas por un mínimo de 3.86% de distancia no corregida p, basadas en el gen mitocondrial nd2. La nueva especie se distingue de sus congéneres por la combinación de los siguientes caracteres: presencia de un conjunto de grandes puntos negros en las escamas de los costados del cuerpo, los puntos negros con un diámetro igual o mayor al 50% del diámetro de la pupila, un mayor número de escamas en la línea lateral y un mayor número de escamas predorsales. Con base en el análisis filogenético, la nueva especie se recupera como un taxón cercanamente relacionado a P. chimalapensis, con una probabilidad posterior de 0.96, y este clado fue recuperado como grupo hermano de P. oaxacae con una probabilidad posterior de 1.0 y un soporte bootstrap de 100. Esta especie representa el duodécimo miembro del género Profundulus.
Palabras clave: Biodiversidad, Filogenia, Peces de agua dulce, Profundúlidos, Taxonomía.
Introduction
Profundulidae is one of the few freshwater fish families endemic to Mesoamerica; it constitutes a lineage with a limited geographical distribution that extends from southern Mexico to Central America (Morcillo et al., 2016; Domínguez-Cisneros et al., 2023). Profundulid killifishes are represented by two genera (Tlaloc Álvarez & Carranza, 1951 and Profundulus Hubbs, 1924) and 15 valid species (Calixto-Rojas et al., 2023; Domínguez-Cisneros et al., 2023). Members of the Profundulidae are small (maximum standard length around 120 mm) and are often the only fish species found in the upper parts of river basins they inhabit (Calixto-Rojas et al., 2021).
The genus Profundulus currently comprises eleven species. On the Pacific slope, these species are found from the Río Malinaltepec in the Papagayo River basin, in Guerrero state, southeastern Mexico, southward to the Río Lempa in El Salvador and Honduras; on the Atlantic slope, are found from the upper reaches of the Río Papaloapan of Oaxaca, Mexico, to the Chamelecón and Ulúa rivers in northwestern Honduras (Morcillo et al., 2016; Domínguez-Cisneros et al., 2023).
Our knowledge of the Profundulidae is growing rapidly, and the number of valid species has more than doubled since Miller’s comprehensive revision of Profundulus in 1955 (Domínguez-Cisneros et al., 2023). Recent molecular studies have demonstrated that Mexican populations of P. punctatus (Günther, 1866) (sensu Miller, 1955) include several highly divergent lineages (Doadrio et al., 1999; Morcillo et al., 2016; Calixto-Rojas et al., 2023) that were later described as new species (P. mixtlanensis Ornelas-García, Martinez-Ramírez & Doadrio, 2015, P. parentiae Matamoros, Domínguez-Cisneros, Velázquez-Velázquez & McMahan, 2018, P. chimalapensis Del Moral-Flores, López-Segovia & Hernández-Arellano, 2020, P. adani Domínguez-Cisneros, Velázquez-Velázquez, McMahan & Matamoros, 2021, P. emilioi Calixto-Rojas, Lira-Noriega, Rubio-Godoy, Pérez-Ponce de León & Pinacho-Pinacho, 2023,and P. rei Calixto-Rojas, Lira-Noriega, Rubio-Godoy, Pérez-Ponce de León & Pinacho-Pinacho, 2023). Furthermore, P. balsanus Ahl, 1935 was redescribed and recognized as a valid species (Jamangapé et al., 2016). One of these distinct lineages was found in the headwaters of the Grijalva basin by Morcillo et al. (2016) and Domínguez-Cisneros et al. (2023), being well differentiated from P. punctatus. Miller (1955) also detected that the populations from Piedra Parada, Chiapas (a tributary of the Grijalva River) differed morphologically from most populations of P. punctatus.
In this study, we evaluated the morphological and molecular distinctiveness of P. aff. punctatus from the headwaters of the Grijalva basin and rivers of the Isthmus of Tehuantepec in southern of Mexico. We determined that its taxonomic status did not correspond to any other described species and therefore is formally describe as a new species of Profundulus.
Material and methods
The description of the new species was based on specimens and nd2 sequences obtained from fin clips (ethanol-preserved tissues) collected and preserved over the last 20 years. The specimens are deposited in the ichthyological collection of the Museum of Zoology, Universidad de Ciencias y Artes de Chiapas, Tuxtla Gutierrez, Chiapas, Mexico (MZ-UNICACH). Institutional abbreviations follow Sabaj (2020). Measurements and counts follow Miller (1955) and Domínguez-Cisneros et al. (2023). Measurements are presented as percentages of standard length (SL), except for those related to head morphology, which are expressed as percentages of head length.
Seventeen morphometric measurements (in mm) and nine meristic variables were recorded. The morphometric variables were: 1, standard length; 2, head length; 3, predorsal length; 4, prepelvic length; 5, anal origin to caudal base; 6, body, greatest depth; 7, body, greatest width; 8, caudal peduncle, length; 9, caudal peduncle, least depth; 10, dorsal fin, basal length; 11, anal fin, basal length; 12, head depth; 13, head width; 14, interorbital, least bony width; 15, orbit length; 16, snout length; 17, upper jaw length. The meristic variables were: 1, dorsal fin rays; 2, anal fin rays, 3, caudal fin rays; 4, pectoral fin rays; 5, pelvic fin rays; 6, scales in lateral series; 7, predorsal scales; 8, scale count around the body; 9, scale count around peduncle.
The osteological characters were obtained from cleared and double stained specimens (c&s, bone alizarin and cartilage counter-stained with alcian blue), according to the technique described by Taylor (1967), with some modifications proposed by Taylor, Van Dyke (1985). The identification of bone elements was based on the bone nomenclature proposed by Gosline (1961), Parenti (1981), Costa (1998), and Domínguez-Cisneros et al. (2023). The terminology of anal fin rays and the count of vertebrae follow Arratia (2008) and Schultze, Arratia (2013).
We extracted total genomic DNA using the conventional proteinase K/phenol/chloroform method (Hillis et al., 1996). The polymerase chain reaction (PCR) was used to amplify the mitochondrial NADH dehydrogenase subunit 2 (mt-nd2: 1047 bp), using the primers MET and TRP (Schelly et al., 2006); mitochondrial ATP synthase 8 and 6 (mt-atp8&6: 850 bp), using the primers L8331 and H9236 (Perdices et al., 2002); and the first and second introns of the nuclear S7 ribosomal protein gene (nuc-S7: 937), with the primers S7RPEX1F and S7R0EX3R (Chow, Hazama, 1998). The PCR conditions were the same as those followed by Beltrán-López et al. (2021). The PCR products were visualized on 1.5% agarose gel and submitted to MACROGEN Korea for sequencing. The sequences were alignment manually with MEGAX (Kumar et al., 2018). We obtained the evolutionary substitution model using the program PartitionFinder2 (Lanfear et al., 2016) and Akaike’s Information Criterion (AIC).
To construct the phylogenetic hypotheses, we included ten valid species of the genus Profundulus, samples of the new species, and two mt-nd2 sequences identified as P. chimalapensis in Arroyave et al. (2024), collected from the Sabinal River, the same river system as our holotype. We used Bayesian Inference (BI) and Maximum Likelihood (ML) with the three concatenated genes, and also, with each separate genes using Tlaloc portillorum (Matamoros & Schaefer, 2010) as the outgroup. The ML tree was calculated with RAxML on XSEDE (Stamatakis, 2014) using the GTR+GAMMA substitution model. The nodes were supported using 1,000 bootstrap replicates. For BI analysis the simulation was run for 20,000,000 generations with four Markov chains sampling every 500 samples, using MrBayes on XSEDE (Ronquist et al., 2012). Values of Effective Sample Size (ESS) were reviewed to verify that values were > 200. Ten percent of the resulting trees were discarded as burn-in. All of the analyses described above were assessed in the portal CIPRES (Miller et al., 2010). To evaluate the degree of divergence the genetic distances (uncorrected p-distances) were calculated between species in MEGA X (Kumar et al., 2018).
Results
Profundulus hectori,new species
urn:lsid:zoobank.org:act:8319F726-1682-483C-B5F5-FF4A518F8FC1
(Figs. 1, 2A–B; Tabs. 1–2)
Holotype. MZ-UNICACH 7812, male, 51.15 mm SL, Mexico, Chiapas, Sabinal River, tributary of Grijalva River, 1 km from Monterrey Institute of Technology and Higher Education, Tuxtla campus, in the municipality of Tuxtla Gutiérrez, Chiapas, 16°45’36.25”N 93°12’18.62”W, altitude 611 m, 13 Apr 2002, E. Velázquez-Velázquez.
Paratypes. All from Mexico. MZ-UNICACH 006, 16 (1 c&s), 38.89–51.15 mm SL, same data as holotype. MZ-UNICACH 3100, 9, 35.83–57.07 mm SL, Chiapas, Copainalá stream, in the municipality of Copainalá, 17°3’29.43”N 93°11’50.97”W, 23 Apr 2009, M. Anzueto-Calvo & E. Velázquez-Velázquez. MZ-UNICACH 4332, 4, 38.42–53.24 mm SL, Chiapas, Pozas del Chorreadero, in the municipality of Chiapa de Corzo, 16°45’19.04”N 92°58’17.55”W, 23 Oct 2010, M. Anzueto-Calvo & L. Velázquez Valencia. MZ-UNICACH 5085, 18, 38.42–70.79 mm SL, Chiapas, Nandalumí stream, in the municipality of Chiapa de Corzo, 16°43’22.25”N 92°59’3.01”W, 29 Nov 2016, M. Anzueto-Calvo & A. Basulto. MZ-UNICACH 5752, 4, 40.58–43.53 mm SL, Chiapas, stream of the Rancho San José, Colonia Cuauhtémoc, in the municipality of Villaflores, 16°20’45.07”N 93°12’43.68”W, 26 Dec 2014, M. Anzueto-Calvo & E. Velázquez-Velázquez. MZ-UNICACH 6586, 17 (1 c&s), 37.60–58.40 mm SL, Chiapas, stream of the Tierra y Libertad, in the municipality of Jiquipilas, 16°29’16.07”N 94°5’55.21”W, 15 Apr 2015, A. Gómez & E. Velázquez-Velázquez. MZ-UNICACH 7569, 15 (6 c&s), 40.77–68.25 mm SL, Chiapas, El Arenal stream, in the municipality of Cintalapa, 16°26’3.82”N 94°4’54.28”W, 12 Oct 2021, E. Velázquez-Velázquez & S. Domínguez-Cisneros. MZ-UNICACH 7570, 4, 38.19–65.05 mm SL, Chiapas, Waterfall of the Novillero River, in the municipality of Cintalapa, 16°25’51.21”N 94°5’59.08”W, 20 Oct 2021, E. Velázquez-Velázquez & S. Domínguez-Cisneros. MZ-UNICACH 7620, 4 (1 c&s), 40.57–59.38 mm SL, Chiapas, Sabinal River, Pozas de Berriozabal, in the municipality of Tuxtla Gutierrez, 16°45’59.57”N 93°13’48.65”W, 16 Dec 2022, E. Velázquez-Velázquez & M. Anzueto-Calvo. MZ-UNICACH 7622, 10 (2 c&s), 38.53–56.50 mm SL, Oaxaca, upper course of the Negro River, Colonia Rudolfo Figueroa, in the municipality of San Miguel Chimalapa, 16°33’10.46”N 94°10’54.90”W, 27 Jan 2020, E. Velázquez-Velázquez & M. Anzueto-Calvo.
FIGURE 1| Profundulus hectori, MZ-UNICACH 7812, holotype, male, 51.15 mm SL, Sabinal River, tributary of Grijalva River, in the municipality of Tuxtla Gutiérrez, Chiapas, Mexico, 16°45’36.25”N 93°12’18.62”W.
Diagnosis. Profundulus hectoriis a memberof genus Profundulus by the presence of a small mesethmoid, crescent-shaped, that does not extend beyond the margin of the vomer, the autopterotic fossa is large and humeral spot present. The following combination of characters distinguishes P. hectori from congeners: presence of a set of large black spots on the scales on the sides of the body (Figs. 2A–B) (absent in P. guatemalensis, P. kreiseri, P. adani, and P. mixtlanensis). The set of black spots are equal to or greater than 50% of the diameter of the pupil in P. hectori, whereas in P. balsanus, P. parentiae, and P. oaxacae (Meek, 1902), the set of black spots are small (less than 50% of the diameter of the pupil). It is distinguished from P. chimalapensis by a larger number of scales on the lateral line (30–33) (vs. 29–32) and a larger number of predorsal scales (18–24) (vs. 18–21) (Tab. 2). Profundulus hectori can be further differentiated from P. punctatus by the origin of the dorsal fin positioned at a vertical line slightly anterior to the origin of the anal fin (vs. origin of the dorsal fin positioned at a vertical line posterior to the origin of the anal fin) (Figs. 3A–B). The neural and hemal spines of the preural centra 2 in P. hectori has no serrations along its edges (vs. serrations along its outer edge in P. punctatus) (Figs. 4A–B). The lacrimal bone has a lateral extension of round or blunt shape in P. hectori (vs. the lacrimal bone with a lateral extension of triangular shape in P. puntatus) (Figs. 5A–D).
TABLE 1 | Morphometric data for holotype and paratypes of Profundulus hectori (N = 101), and non-types of P. punctatus (N = 111), P. oaxacae (N = 28) and P. chimalapensis (N = 38). In the paratypes and other species columns, the first two numbers represent the range of measurements, followed by the mean in parentheses.
Character | Holotype | P. hectori Paratypes | P. punctatus | P. oaxacae | P. chimalapensis |
Standard length (mm) | 51.15 | 34.0–70.79 (45.35) | 36.36–82.17 (53.47) | 35.0–63.2 (45.5) | 35.9–80.3 (45.6) |
Percent of standard length | |||||
Head length | 31.06 | 27.03–32.93 (29.69) | 26.10–32.18 (28.55) | 26.3–30.5 (28.7) | 27.4–33.4 (30.8) |
Predorsal length | 67.17 | 62.19–74.68 (67.09) | 63.87–72.48 (68.64) | 65.5–72.2 (69.0) | 66.3–77.4 (69.4) |
Prepelvic length | 54.60 | 48.07–57.62 (53.02) | 46.95–55.57 (51.92) | 47.9–57.3 (52.2) | 50.2–56.4 (53.9) |
Anal origin to caudal base | 33.35 | 28.68–35.78 (32.40) | 27.09–37.42 (33.57) | 25.2–32.6 (29.1) | 27.1–35.6 (31.2) |
Body, greatest depth | 29.59 | 20.99–35.17 (28.07) | 24.12–33.61 (28.50) | 23.5–30.8 (26.6) | 24.3–30.8 (27.5) |
Body, greatest width | 17.98 | 15.53–23.45 (18.87) | 14.30–25.40 (17.92) | 14.4–18.6 (16.5) | 15.4–19.4 (17.5) |
Caudal peduncle, length | 17.65 | 13.66–21.41 (17.55) | 15.48–21.80 (18.73) | 11.1–19.6 (14.8) | 14.2–19.9 (17.0) |
Caudal peduncle, least depth | 15.65 | 13.63–17.28 (15.44) | 14.00–18.28 (15.67) | 11.9–16.6 (14.4) | 13.9–17.4 (15.3) |
Dorsal fin, basal length | 16.28 | 11.10–18.42 (15.30) | 12.50–17.26 (14.74) | 12.8–17.7 (15.2) | 12.5–16.5 (14.7) |
Anal fin, basal length | 15.83 | 12.77–19.09 (16.63) | 11.59–20.07 (15.71) | 11.7–18.5 (14.7) | 12.6–18.9 (15.2) |
Percent of head length | |||||
Head depth | 66.33 | 58.98–94.86 (74.58) | 56.48–84.75 (70.33) | 66.4–101.5 (81.0) | 59.9–70.5 (65.8) |
Head width | 65.07 | 58.77–75.93 (68.23) | 59.71–80.56 (68.78) | 60.1–72.3 (65.0) | 61.0–71.7 (65.6) |
Interorbital, least bony width | 42.41 | 39.95–53.15 (45.18) | 42.16–53.15 (48.50) | 41.5–48.3 (44.3) | 45.9–52.7 (48.7) |
Orbit length | 23.72 | 20.54–28.91 (25.09) | 19.02–29.87 (25.62) | 21.2–26.3 (23.8) | 22.0–32.6 (28.0) |
Snout length | 32.59 | 25.63–37.38 (31.70) | 28.99–37.10 (32.82) | 24.0–32.2 (28.8) | 28.7–37.9 (33.1) |
Upper jaw length | 27.24 | 11.37–31.73 (24.04) | 17.87–30.58 (26.59) | 21.8–36.1 (26.9) | 23.5–32.0 (26.4) |
TABLE 2 | Meristic data of holotype and paratypes of Profundulus hectori (N = 106), and non-types of P. punctatus (N = 127), P. oaxacae (N = 35) and P. chimalapensis (N = 38). In the paratypes and other species columns, the first two numbers represent the range of measurements, followed by the mode in parentheses. *The vertebral counts were obtained from cleared and double stained specimens: Profundulus hectori (N = 11), P. punctatus (N = 10), P. oaxacae (N = 5) and P. chimalapensis (N = 7).
Character | Holotype | P. hectori Paratypes | P. punctatus | P. oaxacae | P. chimalapensis |
Dorsal rays | 11 | 10–14 (12) | 10–14 (11) | 11–13 (12) | 11–14 (13) |
Anal rays | 12 | 12–16 (14) | 12–16 (14) | 13–15 (14) | 14–16 (15) |
Pectoral rays | 17 | 15–19 (17) | 14–19 (17) | 15–18 (16) | 15–18 (17) |
Caudal rays | 20 | 18–23 (21) | 19–24 (21) | 17–22 (20) | 18–23 (20) |
Pelvic rays | 6 | 5–7 (6) | 5–7 (6) | 6 (6) | 6 (6) |
Lateral scales | 32 | 30–33 (32) | 29–34 (33) | 29–31 (30) | 29–32 (31) |
Predorsal scales | 21 | 18–24 (20) | 18–24 (20) | 22–29 (25) | 18–21 (19) |
Scales around body | 28 | 24–29 (26) | 23–30 (26) | 23–31 (27) | 25–30 |
Scales around peduncle | 20 | 17–22 (20) | 17–25 (20) | 16–22 (19) | 17–21 (20) |
Vertebrae | – | 32–33 (33)* | 32–34 (33)* | 31–33* | 33* |
Gill rakers | 15 | 14–19 (16) | 14–19 (15) | 15–18 (16) | 16–19 (18) |
FIGURE 2| General morphology and live coloration patterns in Profundulus hectori, A. Male, B. Female, Sabinal River, tributary of Grijalva River, Tuxtla Gutierrez, Chiapas. Collected in December 16, 2022 by E. Velázquez-Velázquez and M. Anzueto-Calvo. Profudulus punctatus, C. Male, D. Female, Arroyo tributario del Río Zanatenco, Tonalá Chiapas. Collected on March 21, 2023 by Ernesto Velázquez and Manuel Anzueto.
FIGURE 3| Origin of the dorsal fin positioned slightly anterior to the origin of the anal fin in a vertical line (dotted line): A. Profundulus punctatus, MZ-UNICACH 6326, male, 62.53 mm SL, B. Profundulus hectori, MZ-UNICACH 7812, holotype, male, 51.15 mm SL.
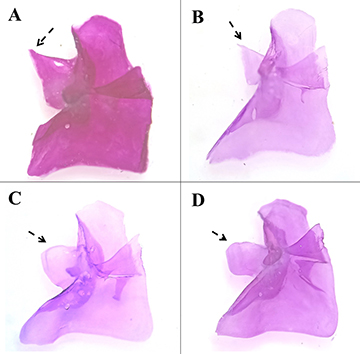
FIGURE 4| Lateral view of the caudal skeleton of A. Profundulus punctatus, MZ-UNICACH 7169, male, 49.24 mm SL, Guatemala, Quetzaltenango, Batza River, B. P. punctatus, MZ-UNICACH 6355, female, 62.71 mm SL, Mexico, Chiapas, Zapata River and C. P. hectori, paratype, MZ-UNICACH 7620, male, 51.15 mm SL, Mexico, Chiapas, Sabinal River, D. P. hectori, MZ-UNICACH 006, paratype, male, 51.15 mm SL, Mexico, Chiapas, Nandalumí stream. Arrows point to spinal neural 2 with serrations. Abbreviations: e, epural; h2-5, hemal spine of preural centra 2-5; Hp, hypural plate; p, parhypural; n2-5, neural spine of preural centra 2-5; r, radial cartilage.
FIGURE 5| Ventral view of left lacrimal: A. Profundulus punctatus, MZ-UNICACH 7169, male, 49.24 mm SL, Guatemala, Quetzaltenango, Batza River, B. P. punctatus, MZ-UNICACH 6355, female, 62.71 mm SL, Mexico, Chiapas: Zapata River and C. P. hectori, paratype, MZ-UNICACH 7620, male, 51.15 mm SL, Mexico, Chiapas: Sabinal River, D. P. hectori, paratype, MZ-UNICACH 006, male, 51.15 mm SL, Mexico, Chiapas, Nandalumí stream. Arrow points to lacrimal extension.
Description. Morphometric data appear in Tab. 1. Body slender, sub-cylindrical; head compressed, large (27.03–32.93%); the interorbital broad (39.95–53.15%) and typically concave or nearly flat. Ascending premaxillary process short and broad and bluntly rounded at the tip. Mouth subterminal, lower jaw broad, heavy and protruding so that upper jaw is included. Dorsal fin rounded in both males and females with its basal length large (11.10–22.70%). Anal fins of males rounded, slightly elongated in females (Figs. 2A–B). Caudal peduncle relatively short (13.66–21.41%). All bones in the caudal fin skeleton are directly or indirectly associated with the last compound centrum of the vertebral column; median hypural plate divided into subequal parts by an open groove (Figs. 4C–D). Meristic traits are as follows (Tab. 2).
Coloration in alcohol. In both sexes, head and flank pale brown; the body is dark gray; the humeral spot is very evident; fins hyaline in females, hyaline with pale brown pigmentation in males.
Coloration in life. Live coloration description is largely based on two live specimens (Figs. 2A–B). The form and coloration of the nuptial adult male are shown in Fig. 2A. The male is brown-orange on nearly the whole body. A golden yellow blotch covers the operculum and reaches the base of the pectoral fin. A dark band on the sides of the body, with large irregular dark-brown spots on the scales, runs from just behind the pectoral fin to the base of the caudal fin. The base of dorsal and anal fin yellowish, with greater intensity on the edges. The female is brown-orange on nearly the whole body, with diffuse dark band on the anterior half of the side of the body. A golden yellow blotch covers the operculum and reaches the base of the pectoral fin. All fins yellowish. Most of the body dark, often with irregular brown-dark spots on the scales on the sides of the body to the caudal fin (Fig. 2B).
Sexual dimorphism. The largest specimen recorded was a male (83.9 mm SL), while the maximum size recorded in a female was 77.9 mm SL. In males and females the dorsal and anal fins have a similar morphology (Figs. 2A–B); the males have contact organs or spinules on the surface of the scales and the middle rays of the anal and dorsal fins; this is the most notable, though not striking, difference between males and females.
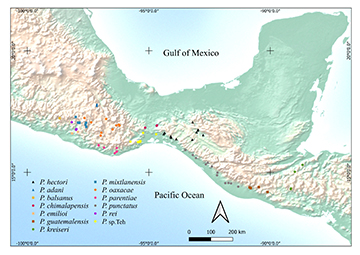
Geographical distribution. On the Atlantic slope: Springs and streams tributaries of the Grijalva Rivers in Chiapas, Mexico (Fig. 6): Sabinal River (Figs. 7A–B), Cintalapa River (Fig. 7C), Negro River, the Venta River, Bombaná River, Chiapilla River, Nandalumí streams, Natura and vos (springs) and Los Amates River, all tributaries of the Grijalva River basin. On the Pacific slope: Novillero River (Fig. 7D) and the Arenal River.
FIGURE 6| Map showing the geographic distribution of Profundulus hectori (black triangles). Also displayed are the geographic distributions of Profundulus species in southern Mexico and Central America.
FIGURE 7 | Habitats of Profundulus hectori in the upper basin of the Grijalva and Novillero Rivers. A. and B. Type-locality, Sabinal River, tributary of Grijalva River, Tuxtla Gutierrez, Chiapas (March 6, 2023). C. Arroyo Rizo de Oro, tributary of Cintalapa River, Chiapas (January 29, 2020). D. Waterfall of the Novillero River, Cintalapa, Chiapas (November 17, 2022).
Ecological notes. The type-locality is near the city of Tuxtla Gutierrez, capital of the state of Chiapas, along the Sabinal River, a tributary of the Grijalva River, at an altitude of 658 m. The streams are surrounded by riparian vegetation with presence of Sabino trees (Taxodium huegelii) (Figs. 7A–B). The substrate was composed of pebbles, sand and Sabino roots; the marginal vegetation was composed predominantly of scrubland and herbaceous banks, where the specimens were found. The new species was found in sympatry at the type-locality with Tlaloc labialis (Günther, 1866), Poeciliopsis pleurospilus (Günther, 1866), Poecilia sphenops Valenciennes, 1846, Chiapaheros grammodes (Taylor & Miller, 1980), Astyanax aeneus (Günther, 1860), and the non-native fish Poecilia reticulata Peters, 1859, which is currently considered a highly invasive species. This species is commonly known as Escamudo del Grijalva in Spanish-spaeking countries, or as Grijalva killifish in english-speaking countries.
Etymology. In recognition of his many contributions to the study and conservation of freshwater fishes in Mexico, this species is named after our friend and colleague, the Mexican ichthyologist Hector Espinosa Perez, recently deceased. A noun in a genitive case.
Conservation status. Profundulus hectori is known as a native and endemic freshwater species of Chiapas, Mexico. Its total population size and population characteristics are unknown. However, ichthyological surveys suggest that this species is common at some localities where it occurs. A number of potential threats to aquatic habitat exist in Chiapas, including deforestation associated with land-use changes, pollution from agriculture, exotic species, and urban and hydropower development. While the direct impacts of these threats on population status are unknown, they are producing continuous inferred declines in habitat quality and, potentially, availability. Therefore, the new species can be listed as Least Concern (LC), according to the IUCN criteria (IUCN, 2024).
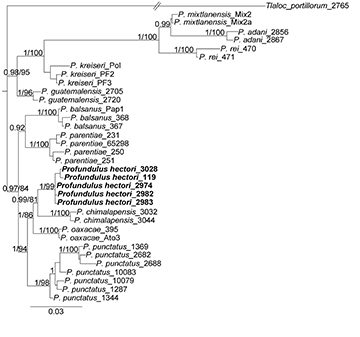
Molecular analysis. Phylogenetic analyses (BI and ML) based on the three concatenated genes suggest that the new species, P. hectori, forms a clade with P. chimalapensis as its sister species. Together, P. hectori and P. chimalapensis were recovered as sister species to P. oaxacae, with strong support in both analyses. This clade of three species is, in turn, the sister clade to P. punctatus, also with high support (Fig. 8). Although the phylogenetic relationships among P. hectori, P. chimalapensis, P. oaxacae, and P. punctatus varied depending on the gene used, P. hectori was consistently recovered as a monophyletic group with high support and as part of the same clade with the same sister species. The results of those genes are presented as supplementary material (Figs. S1, S2 and S3). The specimens identified as P. chimalapensis in Arroyave et al. (2024) were nested within the clade of the new species, P. hectori (Fig. S1). The mean genetic p-distances based on mt-nd2 between P. hectori and congeners ranged from 3.86% (with P. oaxacae), 4.16% (with P. chimalapensis), 4.44% (with P. punctatus) to 15.55% (with P. adani) (Tab. 3).
FIGURE 8| Bayesian phylogeny with the three concatenated genes showing relationships of Profundulus hectori with respect to other species of Profundulus. Values before diagonal represent the bayesian posterior probabilities, and the values after of diagonal represent the bootstrap support for ML analysis.
TABLE 3 | Below the diagonal: uncorrected genetic distances (p) in percentage between recognized Profundulus species with mitochondrial nd2 gene. Above the diagonal: standard errors.
Species | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 |
1. P. hectori | 0.00 | 0.01 | 0.007 | 0.006 | 0.007 | 0.006 | 0.006 | 0.006 | 0.006 | 0.01 | 0.01 |
2. P. mixtlanensis | 12.53 | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.007 | 0.008 |
3. P. guatemalensis | 5.37 | 11.77 | 0.00 | 0.006 | 0.008 | 0.008 | 0.008 | 0.007 | 0.007 | 0.01 | 0.009 |
4. P. kreiseri | 4.64 | 11.90 | 3.82 | 0.00 | 0.008 | 0.0079 | 0.007 | 0.006 | 0.007 | 0.01 | 0.01 |
5. P. balsanus | 6.07 | 13.45 | 6.57 | 5.68 | 0.00 | 0.0079 | 0.007 | 0.007 | 0.007 | 0.01 | 0.01 |
6. P. punctatus | 4.44 | 12.90 | 6.15 | 5.85 | 6.57 | 0.00 | 0.006 | 0.006 | 0.006 | 0.01 | 0.009 |
7. P. chimalapensis | 4.16 | 13.02 | 6.40 | 5.34 | 6.10 | 4.87 | 0.00 | 0.006 | 0.007 | 0.01 | 0.01 |
8. P. oaxacae | 3.86 | 12.10 | 5.43 | 4.54 | 6.16 | 4.60 | 4.32 | 0.00 | 0.006 | 0.01 | 0.01 |
9. P. parentiae | 5.26 | 13.20 | 5.26 | 4.62 | 5.02 | 5.45 | 5.53 | 4.81 | 0.00 | 0.01 | 0.009 |
10. P. adani | 15.55 | 4.86 | 13.96 | 15.44 | 17.82 | 14.65 | 16.94 | 14.06 | 16.89 | 0.00 | 0.006 |
11. P. rei | 12.69 | 0.86 | 12.32 | 11.44 | 13.71 | 13.36 | 13.31 | 12.45 | 13.40 | 4.07 | 0.00 |
Discussion
Profundulus was first described and diagnosed by Hubbs (1924) based on morphological characters. Since then, the taxonomy of this genus has undergone a long and complex history. Recently, Domínguez-Cisneros et al. (2023) diagnosed and characterized the genera Tlaloc and Profundulus, the sole members of the family Profundulidae. The genus Profundulus is distinguished by six key morphological characters: The mesethmoid is small, crescent-shaped, and does not extend beyond the margins of the vomer; the vomer is greatly broadened anteriorly, lacks lateral processes, and is in contact with the lateral ethmoids; the upper portion of the parasphenoid, which contacts the mesethmoid, does not extend beyond the medial part of the latter; the dorsal margin of the interoperculum has a short extension that does not exceed the edge of the bone; the ventral margin of the lacrimal is slightly concave and the autopterotic fossa is notably large. These diagnostic traits are present in the newly described species, Profundulus hectori.
Phylogenetic analyses of the concatenated genes placed P. hectori in a well-supported clade with P. chimalapensis, P. oaxacae, and P. punctatus. Within this clade, P. hectori is most closely related to P. chimalapensis (Fig. 8). The shortest genetic distance, based on the mt-nd2 gene, was observed between P. hectori and P. oaxacae (3.86%), while the greatest distance was between P. hectori and P. punctatus (4.44%). Although the use of genetic distances for species delimitation is debated, the values observed between P. hectori and its close relatives exceed those documented for well-established species of Cyprinodontiformes based on mitochondrial markers, including nd2 (Doadrio, Domı́nguez, 2004; Morcillo et al., 2016; Matamoros et al., 2018; Domínguez, 2023). These findings align with Morcillo et al. (2016), who proposed that P. punctatus represents a species complex. Our results indicate that P. hectori corresponds to the undescribed Profundulus sp. Tehuantepec (Clade A3) identified by Morcillo et al. (2016). Furthermore, our taxonomic analysis establishes P. hectori as a distinct species, distinguishable from its congeners by its unique coloration patterns, morphological traits, and osteological characteristics.
Recently, Arroyave et al. (2024) documented a range extension for P. chimalapensis, identifying specimens from the Grijalva River Basin and certain Pacific rivers (e.g., the Ostuta River) as P. chimalapensis. Although they claimed to have conducted the first assessment of the phylogenetic position of P. chimalapensis, their study did not include specimens from the species’ type-locality or the Coatzacoalcos River basin, where P. chimalapensis is considered endemic (Del Moral-Flores et al., 2020; Domínguez-Cisneros et al., 2023). Based on our molecular results, we phylogenetically validate P. chimalapensis and, by including specimens from its type-locality, confirm its endemicity to the Coatzacoalcos River basin (Atlantic Slope). In contrast, P. hectori is primarily distributed in the Grijalva River basin and the Novillero and Arenal Rivers on the Pacific Slope. We incorporated two mt-nd2 sequences from Arroyave et al. (2024) for specimens collected from the Sabinal River, the proposed type-locality of P. hectori. These sequences nested within the P. hectori clade. Accordingly, we conclude that the specimens identified as P. chimalapensis by Arroyave et al. (2024) actually belong to P. hectori. Our results demonstrate that P. hectori does not overlap with other closely related species. Specifically, P. chimalapensis is endemic to the Coatzacoalcos River basin; P. punctatus is restricted to Pacific Coast rivers in Chiapas and Guatemala; and P. oaxacae is confined to the upper Verde River basin in Oaxaca, Mexico (Domínguez-Cisneros et al., 2023).
The taxonomic validity of Profundulus adani has been questioned. Calixto-Rojas et al. (2023) proposed treating P. adani as a synonym of P. mixtlanensis. However, we do not agree with this conclusion. Calixto-Rojas et al. (2023) argued that phylogenetic approaches assume species should form monophyletic groups. In our concatenated analyses, P. adani is recovered as monophyletic (posterior probability = 1), and even in the phylogenetic tree presented by Calixto-Rojas et al. (2023: fig. 2), P. adani appears monophyletic, albeit without a reported support value at the corresponding node. Furthermore, the relationships among P. adani, P. mixtlanensis, and P. rei in their analysis remain unresolved, with the samples of P. mixtlanensis failing to form a monophyletic group. Calixto-Rojas et al. (2023) acknowledged possible sources of phylogenetic discrepancies, such as introgression, ancestral polymorphism, incomplete lineage sorting, and the time required for some loci to reach reciprocal monophyly. They also stated that such discrepancies do not necessarily allow the detection of species boundaries. Interestingly, in the same study, they described P. rei as a new species despite it not being monophyletic in some of their analyses, justifying this by emphasizing the importance of integrating multiple lines of evidence in species delimitation. Taking this into account, Calixto-Rojas et al. (2023) highlighted the need for integrative taxonomy, stating that resolving taxonomically complex groups requires combining different sources of evidence. Despite this, they decided to synonymize P. adani based primarily on concatenated phylogenetic analyses and species delimitation methods, without considering it as a separate group in subsequent morphological and ecological analyses. This approach overlooked additional evidence, including data from Domínguez-Cisneros et al. (2021), contradicting their stated commitment to a total-evidence framework for assessing diversity within Profundulus. The challenges associated with relying solely on molecular data for species delimitation have been widely discussed in the literature. Issues such as researcher biases, a lack of standardized practices, and the variable information content of molecular markers (e.g., gene flow, sampling design, lineage sorting, selective pressures, and others) can influence results (Huang, 2020; Magoga et al., 2021; Smith, Carstens, 2022). As Burriel-Carranza et al. (2023) emphasized, such practices risk producing unnecessary and unwarranted taxonomic changes with long-term consequences for science and conservation. Even Calixto-Rojas et al. (2023) recognized limitations in species delimitation tools for freshwater fishes, noting that individual methods have inherent biases and weaknesses and emphasizing the importance of complementary data sources, an approach they did not fully apply when addressing the taxonomic status of P. adani.
In this study, we included sequences from the type locality of P. rei (as described by Calixto-Rojas et al., 2023) and from P. mixtlanensis at the locality where paratypes were collected in Río Grande, Santiago Juxtlahuaca (Ornelas-García et al., 2015). Our results support the monophyly of P. adani, with relationships among P. mixtlanensis, P. adani, and P. rei well resolved, although we acknowledge that few populations were included for P. mixtlanensis (see Fig. 8). We also found a high p-distance (> 4.86%) in the nd2 mitochondrial gene between P. adani and other Profundulus species (see Domínguez-Cisneros et al., 2023 for comparisons using additional markers), a value higher than those observed between well-recognized species pairs within the genus (see Tab. 3). Furthermore, we consider the morphological differences reported by Domínguez-Cisneros et al. (2021, 2023) as additional evidence for species distinction, including a greater number of predorsal scales, a relatively larger upper jaw, and the absence of a humeral spot in P. adani, distinguishing it from P. mixtlanensis. Considering this molecular, morphological, and phylogenetic evidence, we maintain that P. adani should continue to be recognized as a valid taxon until a more comprehensive phylogenetic and taxonomic study, including extensive sampling across populations of the P. rei, P. adani, and P. mixtlanensis species group, as well as the incorporation of multiple molecular markers is conducted.
The family Profundulidae has undergone numerous taxonomic revisions, largely due to challenges in species delimitation stemming from a lack of external morphological differentiation among certain species within the family. This phenomenon, commonly referred to as cryptic speciation (Morcillo et al., 2016), has complicated classification efforts. Comparative morphology remains a fundamental tool for reconstructing evolutionary history and refining the classification of cyprinodontiform fishes, often providing valuable phylogenetic insights across taxonomic levels (Domínguez-Cisneros et al., 2023). Our integrative taxonomic analysis, incorporating molecular, morphological, and osteological characters, supports the clades previously defined in Profundulus. Accordingly, we advocate for the combined use of morphological and molecular data to reliably differentiate profundulid species.
Comparative material examined. Profundulus punctatus: Mexico. MZ-UNICACH 6326, 25 (4 c&s), 37.9–72.6 mm SL, Río Lagartero, Arriaga, Chiapas, 16°19’19.18”N 93°52’16.95”W, 7 Apr 2015, A. E. Gomez & E. Velázquez. MZ-UNICACH 6632, 21 (3 c&s), 43.3–72.0 mm SL, Río Cintalapa en Escuintla, Chiapas, 15°23’39.71”N 92°34’29.74”W, 9 Apr 2015, A. E. Gomez & E. Velázquez. MZ-UNICACH 6578, 18 (2 c&s), 45.9–56.3 mm SL, Cascada Velo de Novia Tonala, Chiapas, 8 Apr 2015, A. E. Gomez & E. Velázquez. Guatemala. MZ-UNICACH 7169 (3 c&s), 32, 37.9–72.6 mm SL, Río Batza, Colonia Campolla, puente Batza, Quetzaltenango, 14°33’37.33”N 91°51’15.83”W, 9 Apr 2017, A. E. Gomez & E. Velázquez. MZ-UNICACH 7182, 16, 36.3–75.0 mm SL, Rio Nil, puente Nil, El Acintal, Retalhuleo, 14°33’53.56”N 91°43’23.49”W, 9 Apr 2017, A. E. Gomez & E. Velázquez. MZ-UNICACH 7186, 14 (2 c&s), 39.4–82.1 mm SL, Río el Puentecito, Caserio, Retalhuleo, 14°32’55.71”N 91°37’6.57”W, 9 Apr 2017, A. E. Gomez & E. Velázquez. Profundulus chimalapensis: Mexico. MZ-UNICACH 7559, 38 (7 c&s), 35.8–80.3 mm SL, Arroyo Piedra del Rio el Corte (Paso Lagarto), Santa María Chimalapa, Oaxaca, 16°56’3.10”N 94°39’46.21”W, 20 Jan 2022, E. Velázquez & M. J. Anzueto. Profundulus oaxacae: Mexico. MZ-UNICACH 6714, 27 (3 c&s), 35.0–63.1 mm SL, Río Salado. MZ-UNICACH 6715, 22 (2 c&s), 39.2–70.5 mm SL, Arroyo Grande tributario del Río la Hormiga, Mitla, Oaxaca, 27 Apr 2015, M. J. Anzueto. Profundulus balsanus: Mexico. MZ-UNICACH 6712, 22, 34.2–64.6 mm SL, Río Malinaltepec. Profundulus parentiae: Mexico. CNPE-IBUNAM 28804, holotype, 65.9 mm SL, Río Huatulco, Santa Maria Huatulco, Oaxaca, 15°50’24.56”N 96°19’48.75”W, 25 Apr 2015, W. Matamoros, M. J. Anzueto, J. A. Jamangapé & I. Aguilar. CNPE-IBUNAM 22805, 3, Rio Huatulco, Santa Maria Huatulco, Oaxaca, 15°50’24.56”N 96°19’48.75”W, 25 Apr 2015, W. Matamoros, M. J. Anzueto, J. A. Jamangapé & I. Aguilar. FMNH 131755, 2, paratypes, 26.9–35.3 mm SL, Río Huatulco, Santa María Huatulco, Oaxaca, 15°50’24.56”N 96°19’48.75”W, 25 Apr 2015, W. Matamoros, M. J. Anzueto, J. A. Jamangapé & I. Aguilar. MZ-UNICACH 6575, 10, 50.3–68.3 mm SL, Río Huatulco, Toma de Agua, Río Huatulco, Santa María Huatulco, Oaxaca, 15°50’24.56”N 96°19’48.75”W, 25 Apr 2015, W. Matamoros, M. J. Anzueto, J. A. Jamangapé & I. Aguilar. MZ-UNICACH 6576, 10, 40.2–67.7 mm SL, Arroyo de la calle central, San Miguel Figueroa, Pochutla, Oaxaca, 15°47’24.1”N 96°24’3.9”W, 21 Apr 2015, W. Matamoros, M. J. Anzueto, J. A. Jamangapé & I. Aguilar. Profundulus adani: Mexico. CNPE-IBUNAM 23796, holotype, 64.7 mm SL, Arroyo Santa Maria Ixcaltlan, Oaxaca, 17°51’3”N 97°11’57.4”W, 19 Jun 2018, E. Velázquez & M. J. Anzueto. MZ-UNICACH 7382, 44, paratypes, 36.6–68.2 mm SL. Santa Maria Ixcatlan, Oaxaca, 17°47’57.59”N 97°8’10.40”W, 19 Jun 2018, E. Velázquez & M. J. Anzueto. MZ-UNICACH 7343, 11, 52.3–77.9 mm SL. Rio Huatla, San Miguel Huatla, Oaxaca, 17°44’28”N 97°20’15.4”W, 20 Jun 2018, E. Velázquez & M. J. Anzueto. Profundulus mixtlanensis: Mexico. MZ-UNICACH 6716, 26, 39.3–60.7 mm SL, Ranchería la Soledad, Rio San Isidro, Putla, Oaxaca, 17°2’48.92”N 97°51’16.31”W, 29 Apr 2015, M. J. Anzueto & E. Velázquez. Profundulus guatemalensis: Guatemala. MZ-UNICACH 7194, 12, 38.3–83.8 mm SL, Balneario el cantil, Rio Cantil, Escuintla, 14°21’21.18”N 90°48’30.10”W, 10 Apr 2017, W. Matamoros & A. E. González. Profundulus kreiseri: El Salvador. MZ-UNICACH 7205, 26, 37.1–72.6 mm SL, Quebrada los Tecomates, tributario del rio Lempa, Chalatenango, 14°18’50.34”N 89°9’39.91”W, 12 Apr 2017, W. Matamoros & A. E. González.
Acknowledgments
We thank Esteban Pineda for preparation of the map and to Manuel Anzueto and Fabiola Maza for their technical assistance in the field and laboratory. Special thanks to Gloria Solis-Guzman from the Aquatic Biology Laboratory at UMSNH for support with molecular procedures. This article is part of the doctoral thesis of the first author (SEDC), a PhD student in Biodiversity and Conservation of Tropical Ecosystems, from the Universidad de Ciencias y Artes de Chiapas (UNICACH) Mexico. The manuscript improved substantially thanks to the suggestions of two anonymous critics.
References
Arroyave J, Hernández-Ávila SG, Matus-Martínez M, Matamoros W. Range extension of the Mexican-endemic killifish Profundulus chimalapensis (Cyprinodontiformes: Profundulidae), with comments on its phylogenetic placement and possible intergeneric hybridization with Tlaloc Álvarez & Carranza, 1951. Zootaxa. 2024; 5536(3):454–64. https://doi.org/10.11646/zootaxa.5536.3.5
Beltrán-López RG, González-Díaz AA, Soria-Barreto M, Garduño-Sánchez MA, Xochitla-Castrejón C, Rodiles-Hernández R et al. Genetic diversity and structure of one of the most endangered freshwater fish species in Mexico: Tlaloc hildebrandi (Miller, 1950) and recognition of its evolutionarily significant units. PeerJ. 2021; 9:e11952. https://doi.org/10.7717/peerj.11952
Burriel-Carranza B, Estarellas M, Riaño G, Talavera A, Tejero-Cicuéndez H, Els J et al. Species boundaries to the limit: Integrating species delimitation methods is critical to avoid taxonomic inflation in the case of the Hajar banded ground gecko (Trachydactylus hajarensis). Mol Phylogenet Evol. 2023; 186:107834. https://doi.org/10.1016/j.ympev.2023.107834
Calixto-Rojas M, Lira-Noriega A, Rubio-Godoy M, Pérez-Ponce de León G, Pinacho-Pinacho CD. Phylogenetic relationships and ecological niche conservatism in killifish (Profundulidae) in Mesoamerica. J Fish Biol; 2021; 99(2):396–410. https://doi.org/10.1111/jfb.14727
Calixto-Rojas M, Lira-Noriega A, Rubio-Godoy M, Pérez-Ponce de León G, Pinacho-Pinacho CD. Delimitation and species discovery in the Profundulidae fish family: using genetic, environmental and morphologic data to address taxonomic uncertainty. Mol Phylogenet Evol. 2023. 186:107856. https://doi.org/10.1016/j.ympev.2023.107856
Chow S, Hazama K. Universal PCR primers for S7 ribosomal protein gene introns in fish. Mol Ecol. 1998; 7:1247–63.
Costa WJEM. Phylogeny and classification of the Cyprinodontiformes (Euteleostei: Atherinomorpha) a reappraisal. In: Malabarba LR, Reis RE, Vari RP, Lucena ZMS, Lucena CAS, editors. Phylogeny and classification of Neotropical fishes. Porto Alegre: Edipucrs; 1998. p.537–60.
Del Moral-Flores LF, López-Segovia E, Hernández-Arellano T. Profundulus chimalapensis, una nueva especie de pez ciprinodóntido (Cyprinodontiformes: Profundulidae) del Río Coatzacoalcos, México. Rev Biol Trop. 2020; 68(4):1185–97. https://doi.org/10.15517/rbt.v68i4.40340
Doadrio I, Carmona JA, Martínez E, Sostoa A. Genetic variation and taxonomic analysis of the subgenus Profundulus. J Fish Biol. 1999; 55(4):751–66. https://doi.org/10.1111/j.1095-8649.1999.tb00715.x
Doadrio I, Domínguez O. Phylogenetic relationships within the fish family Goodeidae based on cytochrome b sequence data. Mol Phylogenet Evol. 2004; 31(2):416–30. https://doi.org/10.1016/j.ympev.2003.08.022
Domínguez CSE. Revisión taxonómica y filogenía molecular de la familia Profundulidae (Teleostei: Cyprinodontiformes). [PhD Thesis]. Tuxtla Gutiérrez: Universidad de Ciencias y Artes de Chiapas; 2023. Available from: https://hdl.handle.net/20.500.12753/4983
Domínguez-Cisneros SE, Domínguez-Domínguez O, Velázquez-Velázquez E, Pérez-Rodríguez R. Redescription and diagnoses of the genera Profundulus and Tlaloc (Cyprinodontiformes: Profundulidae), Mesoamerican endemic fishes. Neotropical Ichthyol. 2023; 21(1):e220089. https://doi.org/10.1590/1982-0224-2022-0089
Gosline W. Some osteological features of modern lower Teleostean Fishes. Smithsonian Misc Collect.1961; 142(3):1–42.
Hillis DM, Mable BK, Larsen A, Davis SK. Nucleic acids IV: sequencing. In: Hillis DM, Moritz C, Mable BK, editors. Molecular systematics. Sinauer, Sunderland, Massachsetts; 1996. p.321–81.
Huang J-P. Is population subdivision different from speciation? From phylogeography to species delimitation. Ecol Evol. 2020; 10(14):6890–96. https://doi.org/10.1002/ece3.6524
Hubbs CL. Studies of the fishes of the order Cyprinodontes. Misc Pubs Univ Michigan Mus Zool. 1924; 13:1–31.
International Union for Conservation of Nature (IUCN). Standards and petitions committee. Guidelines for using the IUCN Red List categories and criteria. Version16 [Internet]. Gland; 2024. Available from: https://nc.iucnredlist.org/redlist/content/ attachment_files/RedListGuidelines.pdf
Jamangapé JA, Velázquez-Velázquez E, Martínez-Ramirez E, Anzueto-Calvo MJ, Gomez EL, Domínguez-Cisneros SE et al. Validity and redescription of Profundulus balsanus Ahl, 1935 (Cyprinodontiformes: Profundulidae). Zootaxa. 2016; 4173(1):55–65. https://doi.org/10.11646/zootaxa.4173.1.5
Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. 2018; 35(6):1547–49. https://doi.org/10.1093/molbev/msy096
Lanfear R, Frandsen PB, Wright AM, Senfeld T, Calcott B. PartitionFinder 2: new methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol Biol Evol. 2016; 34(3):772–73. https://doi.org/10.1093/molbev/msw260
Magoga G, Fontaneto D, Montagna M. Factors affecting the efficiency of molecular species delimitation in a species‐rich insect family. Mol Ecol Resour. 2021; 21(5):1475–89. https://doi.org/10.1111/1755-0998.13352
Matamoros WA, Domínguez-Cisneros SE, Velázquez-Velázquez E, McMahan CD. Description of a new species of killifish of the genus Profundulus (Atherinomorpha: Profundulidae) from the Mexican State of Oaxaca. Copeia. 2018; 106(2):239–46. https://doi.org/10.1643/i2020156
Morcillo F, Ornelas-García CP, Alcaraz L, Matamoros WA, Doadrio I. Phylogenetic relationships and evolutionary history of the Mesoamerican endemic freshwater fish family Profundulidae (Cyprinodontiformes: Actinopterygii). Mol Phylogenet Evol. 2016; 94:242–51. https://doi.org/10.1016/j.ympev.2015.09.002
Miller RR. A systematic review of the middle American fishes of the genus Profundulus. Misc Pubs Univ Michigan Mus Zool. 1955; 92:1–64. https://deepblue.lib.umich.edu/handle/2027.42/57270
Miller MA, Pfeiffer W, Schwartz T. “Creating the CIPRES science gateway for inference of large phylogenetic trees”. In: Proceedings of the gateway computing environments workshop (GCE). New Orleans, LA: Institute of Electrical and Electronics Engineers; 2010. p.1–08. https://doi.org/10.1109/GCE.2010.5676129
Parenti LR. A phylogenetic and biogeographic analysis of Cyprinodontiform fishes (Teleostei, Atherinomorpha). Bull Am Mus Nat Hist. 1981; 168(4):335–557. http://hdl.handle.net/2246/438
Perdices A, Bermingham E, Montilla A, Doadrio I. Evolutionary history of the genus Rhamdia (Teleostei: Pimelodidae) in Central America. Mol Phylogenet Evol. 2002; 25:172–89. https://doi.org/10.1016/S1055-7903(02)00224-5
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol. 2012; 61(3):539–42. https://doi.org/10.1093/sysbio/sys029
Schultze HP, Arratia G. The caudal skeleton of basal teleosts, its conventions, and some of its major evolutionary novelties in a temporal dimension. In: Arratia G, Schultze P. Wilson MVH, editors. Mesozoic Fishes 5-Global Diversity and Evolution; 2013. p.187–246.
Sabaj MH. Codes for natural history collections in ichthyology and herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
Schelly R, Salzburger W, Koblmüller S, Duftner N, Sturmbauer C. Phylogenetic relationships of the lamprologine cichlid genus Lepidiolamprologus (Teleostei: Perciformes) based on mitochondrial and nuclear sequences, suggesting introgressive hybridization. Mol Phylogenet Evol. 2006; 38:426–38. https://doi.org/10.1016/j.ympev.2005.04.023
Smith ML, Carstens BC. Species delimitation using molecular data. In: Wilkins JS, Zachos FE, Pavlinov IY, editors. Species problems and beyond. Florida, Boca Raton: CRC Press; 2022. p.145–60.
Stamatakis A. RAxML version 8: a tool for phylogenetic analysis andpost-analysis of large phylogenies. Bioinformatics. 2014; 30(9):1312–13. https://doi.org/10.1093/bioinformatics/btu033
Taylor WR. An enzyme method of clearing and staining small vertebrates. Proc U S Natl Mus. 1967; 122(3596):1–17. https://doi.org/10.5479/si.00963801.122-3596.1
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium; 1985; 9(2):107–09. https://doi.org/10.26028/cybium/1985-92-001
Authors
![]() Sara Elizabeth Domínguez-Cisneros1,
Sara Elizabeth Domínguez-Cisneros1, ![]() Ernesto Velázquez-Velázquez1
Ernesto Velázquez-Velázquez1 ![]() ,
, ![]() Omar Domínguez-Domínguez2 and
Omar Domínguez-Domínguez2 and ![]() Rosa Gabriela Beltrán-López3
Rosa Gabriela Beltrán-López3
[1] Colección de Peces, Museo de Zoología, Instituto de Ciencias Biológicas, Universidad de Ciencias y Artes de Chiapas. Libramiento Norte Poniente, 1150, Colonia Lajas Maciel, C.P. 29039, Tuxtla Gutiérrez, Chiapas, Mexico. (SEDC) sara.domínguez@unicach.mx, (EVV) ernesto.velazquez@unicach.mx (corresponding author).
[2] Laboratorio de Biología Acuática, Facultad de Biología, Universidad Michoacana de San Nicolás de Hidalgo, Edificio “R” planta baja, Ciudad Universitaria, Francisco J. Mujica, s/n, Morelia, Michoacán, C.P. 58030, Mexico. (ODD) goodeido@yahoo.com.mx.
[3] Laboratorio de Ictiología, Centro de Investigaciones Biológicas, Universidad Autónoma del Estado de Morelos, Av. Universidad, 1001, Col. Chamilpa, Cuernavaca, Morelos, C.P. 62209, Mexico. (RGBL) rosa.beltran@uaem.mx.
Authors’ Contribution 

Sara Elizabeth Domínguez-Cisneros: Conceptualization, Data curation, Formal analysis, Methodology, Writing-original draft.
Ernesto Velázquez-Velázquez: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Project administration, Supervision, Writing-original draft, Writing-review and editing.
Omar Domínguez-Domínguez: Funding acquisition, Investigation, Methodology, Resources, Supervision, Writing-review and editing.
Rosa Gabriela Beltrán-López: Formal analysis, Methodology, Software, Validation.
Ethical Statement
Fish samples were collected in accordance with Mexican environmental legislation (collecting permit: SPARN/DGVS/13321/23, SGPA/DGVS/03479/09 and SGPA/DGVS/02208/13).
Competing Interests
The author declares no competing interests.
How to cite this article
Domínguez-Cisneros SE, Velázquez-Velázquez E, Domínguez-Domínguez O, Beltrán-López RG. A new species of Profundulus (Cyprinodontiformes: Profundulidae) from southeastern Mexico. Neotrop Ichthyol. 2025; 23(2):e240096. https://doi.org/10.1590/1982-0224-2024-0096
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted April 11, 2025
Accepted April 11, 2025
![]() Submitted September 26, 2024
Submitted September 26, 2024
![]() Epub August 04, 2025
Epub August 04, 2025