![]() Leonardo Schlogel Bueno1
Leonardo Schlogel Bueno1 ![]() ,
, ![]() Matheus Oliveira Freitas1,
Matheus Oliveira Freitas1, ![]() Áthila Andrade Bertoncini1,2,
Áthila Andrade Bertoncini1,2, ![]() Christopher Carl Koenig3,
Christopher Carl Koenig3, ![]() Johnatas Adelir-Alves1,
Johnatas Adelir-Alves1, ![]() Jonas Rodrigues Leite1,
Jonas Rodrigues Leite1, ![]() Felipe Alexandre de Lisboa Miranda Daros4 and
Felipe Alexandre de Lisboa Miranda Daros4 and ![]() Maurício Hostim-Silva1,5
Maurício Hostim-Silva1,5
PDF: EN XML: EN | Cite this article
Abstract
O mero (Epinephelus itajara) forma agregações reprodutivas, cada uma com cerca de 100 indivíduos. No entanto, nenhum estudo foi realizado comparando essas agregações em ambientes naturais e artificiais. Neste artigo, determinamos padrões de abundância de agregação por meio de observações que fizemos entre 2007 e 2014 em três recifes naturais e quatro recifes artificiais no sul do Brasil, comparando avistamentos por unidade de esforço (APUE) entre esses recifes. A sua abundância difere significativamente entre recifes artificiais e naturais. Nenhuma agregação foi observada em recifes naturais, porém agregações de até 54 meros foram observadas em recifes artificiais. Esta mudança no padrão de abundância pode ser devida à pressão da pesca e/ou atração por recifes artificiais. Discutimos os potenciais efeitos ecológicos da diminuição da abundância nos recifes naturais e do aumento da abundância nos recifes artificiais.
Palavras-chave: Atlântico Sul, Epinephelidae, Espécies ameaçadas, Peixes recifais, Recifes artificiais.
Introduction
Atlantic goliath grouper (AGG), Epinephelus itajara (Lichtenstein, 1822), is one of the largest Epinephelidae in the Atlantic Ocean, and are threatened throughout their range (Craig et al., 2009; Ferreira et al., 2011; Barreiros, Coleman, 2023) and classified as Vulnerable (Bertoncini et al., 2018). In Brazil, the species is considered Critically Endangered due to population decline caused by overfishing and habitat loss (ICMBio, 2018). Atlantic goliath grouper grows to 2.5 m and 300 kg (Bullock et al., 1992), feeding mostly on crabs, other crustaceans and slow-moving fishes (Koenig, Coleman, 2009; Freitas et al., 2015), and they play an important role shaping reef communities and modifying the environment where they occur (Koenig et al., 2011).
Epinephelus itajara spawning aggregations are relatively small, about 100 individuals (Koenig et al., 2011; Bueno et al., 2016) when compared with other species of Epinephelidae. In Brazil their spawning aggregations seem to be even smaller, hitting a maximum of ~50 individuals (Bueno et al., 2016). These aggregations occur on shallow (e.g., < 50 m) natural and artificial reefs (Ferreira et al., 2011) that are spatially and temporally consistent, presenting strong interannual fidelity to spawning sites (Koenig et al., 2011; Bueno et al., 2016).
Reef fish, like Atlantic goliath grouper, migrate to form annual spawning aggregations during specific periods of the year (Domeier, Colin, 1997; Sadovy, Eklund, 1999), showing variable consistency of site use (Mitcheson, Colin, 2011) and plausibly representing a learned reproductive strategy (Helfman, Schultz, 1983). That said, aggregations can be eliminated by fishing pressure (Aguilar-Perera, 2006) or shift from natural to artificial reefs (this study).
Some impacts on the marine environment have been attributed to artificial reefs (Brickhill et al., 2005). Impacting their surrounding natural environments as they modify a pre-existent ecosystem that may affect adjacent natural reefs, soft bottoms and pelagic ecosystems (physical, chemical or ecological) (Brickhill et al., 2005).
Artificial reefs may provide cues beyond the evolutionary experience of fishes and elicit responses that are not necessarily adaptive, such as shifting aggregation sites to areas that lack currents that would transport developing larvae to essential juvenile habitat. One of these impacts is the attraction of large reef fish by these structures as studied and discussed by many authors (Bohnsack, 1989; Bohnsack et al., 1997; Lindberg, 1997; Mann et al., 2009; Hackradt et al., 2011). There is clear evidence of the ecological impacts that artificial reefs have on natural reef communities, especially on reef fish (Osenberg et al., 2002; Paxton et al., 2020a; Medeiros et al., 2021).
Many human-made structures intended for other purposes attract fish, including piers and docks, oil and gas platforms, power generation constructions and anti-trawling devices. Others are intentionally deployed to aggregate fishes, including scuttled boats and ships, scrap metal, rocks and concrete structures, thus creating better opportunities for anglers. All of these structures apparently attract AGGs and their aggregations (Sadovy, Eklund, 1999; Bohnsack et al., 1999; Seaman, 2002; Koenig, Coleman, 2009; Koenig et al., 2011; Bueno et al., 2016). The use of artificial reefs along the Brazilian coast has increased in recent years, encouraging research on this subject (Simon et al., 2011). However, the behavioral and environmental effects of enhancement need to be further studied, especially for AGG (Simon et al., 2011; Bueno et al., 2016).
The aim of this study is to evaluate the relationship between the disappearance of known AGG aggregations from natural rocky reefs and the installation of artificial reefs in southern Brazil and discusses how this shift in site affects the rocky reef environments, the distribution and success of this species.
Material and methods
Study area. The study areas were located in southern Brazil coast (states of Paraná and Santa Catarina), between 25o and 27o S latitude in the Western Atlantic. The region has distinct seasonality with summer (late December through late March) being the warmest period. During this time, moderate east and northeast winds predominate, bringing warm (up to 28oC) clear waters from the east. The weather is more variable in the fall (late March through late June), and spring (late September through late December) with an increase in large eastern and southeastern swells and coincident decrease in underwater visibility. Winter (late June through late September) is characterized by cold fronts that bring large swells from the south and southeast, decreasing water temperature (18°C) and increasing turbidity in coastal waters (Bueno et al., 2016).
Seven reef complexes were selected as sampling sites (Fig. 1), three natural reefs, including Arquipélago das Graças (AG), Tamboretes (TA) and Laje dos Lobos outcrops (LL) and four artificial reefs, including Balsa Sul (BS), Balsa Norte (BN), Recifes Artificiais Marinhos (RAM), and Monobóia (MB). The longest distance between sites is from BN (state of Paraná) to LL (state of Santa Catarina) ~91 km, the closest sites are MB and AG ~11 km distance, both in the state of Santa Catarina.
FIGURE 1| Study areas in southern Brazil in the states of Paraná and Santa Catarina, between 25o and 27o S latitude in the Western Atlantic. Four artificial reefs Balsa Norte (BN), Recifes Artificiais Marinhos (RAM), Balsa Sul (BS), and Monobóia (MB). Three natural reefs: Arquipélago das Graças (AG), Tamboretes (TA), and Laje dos Lobos outcrops (LL).
The natural reef sites (AG, TA and LL), are rocky reefs, close to coastal islands with a maximum depth of 19 m and presenting similar structural complexity and variable relief. These reefs are located in the northern region of the state of Santa Catarina.
The sites BS, BN and RAM are artificial reefs installed to encourage fishing tourism and discourage trawl fisheries. BS and BN are ~5 m high, ~75 m long, and ~11 m wide. These iron barges were intentionally sunk offshore on 21 January 2001, 27 m deep, over sandy bottom ~32 km from Pontal do Paraná, state of Paraná (Bueno et al., 2016).
The study site RAM is part of a large artificial reef program that includes ~ 40 reef areas ~12 km offshore formed by concrete blocks and reef balls ~1.5 m high (Fig. 2). For the present study two of those artificial reef areas were selected (sites: Parque dos Meros and RAM-G4). These two reefs are about 1.8 km apart and were considered a single site for abundance analyses (RAM), because of the great proximity and the registered easy movement of the AGG between these reefs. Both were sunk on 9 June 2000 at a depth of 18 m and in 2013 were included in the Currais Island National Marine Park, a marine protected area (Daros et al., 2012).
FIGURE 2| Atlantic goliath grouper Epinephelus itajara on artificial reefs in southern Brazil. A. Using the bottom structure under the Monobóia (MB); B. Seeking shelter in a quadrilateral-shape artificial reef on Recifes Artificiais Marinhos (RAM). Photos by Leonardo S. Bueno.
The MB site is an oil ship single point mooring buoy, installed 8 km offshore in 1977 at depth of ~24 m, composed by a vertical (bottom-surface) structure with many underwater structures such as pipelines, concrete, large anchors and chains, with approximately 300 m radius, off São Francisco do Sul, state of Santa Catarina (Bueno et al., 2016). These artificial sites offer large sheltering areas to AGG (Fig. 2).
Dive surveys. Between October 2007 and March 2014, we conducted dive surveys on AGG at all seven study sites during each season, with higher frequency during austral summer, the known reproductive period (Bullock et al., 1992; Koenig, Coleman, 2009; Gerhardinger et al., 2009; Bueno et al., 2016; Koenig et al., 2017). Roving Diver Technique (RDT) (Jones, Thompson, 1978) were performed in which two scientific divers performed 30 min surveys whenever visibility was greater than three meters. AGG with a total length greater than 100 cm are considered adults and suitable for reproduction (Bullock et al., 1992; Koenig et al., 2007; Bueno et al., 2016). Thus, to infer the reproductive period, all observed AGG had their total lengths estimated (Harvey et al., 2002) in two size classes (< 99 cm and > 100 cm). Natural reef locations chosen for RDT samples were obtained from local spearfishers taken on both aggregation and occurrence described sites (Gerhardinger et al., 2009) and artificial reefs were selected among well-known sites. On large artificial reefs RDT surveys were conducted over as much of the structure as practicable, but on shipwrecks (BS and BN) it was possible to survey all structures.
Data analysis. Diving surveys (RDT) were standardized as sightings-per-unit-effort (SPUE) (Equation: SPUE = N * (T/t); see Bueno et al., 2016). The unit of effort was 30 min, the approximate time of a survey. Where: SPUE = sightings per unit effort, N = number of AGG observed, T = duration in minutes of each survey and t = 30 min to standardize efforts.
To compare abundances between the seven sample areas and within areas (four artificial and three natural reefs) the non-parametric Kruskal-Wallis test was used. When significant differences (p < 0.05) were observed, the Mann-Whitney posteriori test was used (Sokal, Rohlf, 1995). Data were log (x+1) transformed prior to a hierarchical cluster analysis which was used to represent the average similarity (Bray-Curtis) among sampled sites. The consistency of visually defined clusters was tested through the analysis of similarity (ANOSIM).
Results
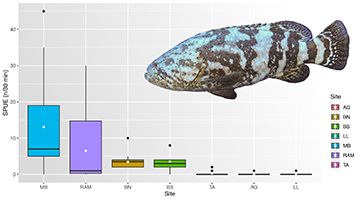
Three hundred and sixteen RDT surveys (160 on artificial reefs and 156 on natural reefs) distributed over the seven sites were performed, with approximately 192 total diving hours. The maximum SPUE was observed at MB with 54 AGGs, followed by RAM (32), BN (30), BS (10), TA (2), LL (2) and AG (1). The AGGs observed during RTD were considered adults, with estimated total length above 100 cm. Combined, natural reefs showed 11% frequency of occurrence of AGG, against 88% at artificial reefs with SPUE mean higher than 4 (Tab. 1).
TABLE 1 | Sightings-per-unit-effort SPUE for Atlantic goliath grouper Epinephelus itajara on artificial reefs: Monobóia (MB), Recifes Artificiais Marinhos (RAM), Balsa Norte (BN), and Balsa Sul (BS), and natural reefs: Tamboretes (TA), Arquipélago das Graças (AG), and Laje dos Lobos (LL), in southern Brazil. Samples (spring/summer/fall/winter in brackets, respectively) number of dive surveys; Max, maximum abundance of Atlantic Goliath Grouper; Mean, mean abundance; SD, standard deviation; frequencyofoccurrence(%); unit effort = 30 min survey.
Sites/Reef type | Samples | Max | Mean | SD (±) | % |
Artificial reefs | |||||
MB | 52 [19/27/02/04] | 54 | 12.55 | 0.88 | 96 |
RAM | 62 [05/34/19/04] | 32 | 7.73 | 10.18 | 74 |
BN | 30 [01/18/08/03] | 30 | 4.73 | 5.07 | 100 |
BS | 16 [02/06/06/02] | 10 | 4.09 | 3.21 | 63 |
Sub-total | 160 [27/85/37/13] | 54 | 8.37 | 9.67 | 88 |
Natural reefs | |||||
TA | 65 [14/28/19/04] | 2 | 0.26 | 0.60 | 22 |
AG | 40 [09/16/13/02] | 1 | 0.02 | 0.14 | 3 |
LL | 51 [14/07/26/04] | 2 | 0.06 | 0.32 | 4 |
Sub-total | 156 [37/51/58/10] | 2 | 0.14 | 0.45 | 11 |
Total | 316 [64/136/95/23] | 54 | 4.54 | 8.25 | 64 |
The AGG abundance on artificial reefs was significantly (p < 0.05) higher than on natural reefs (Tab. 2). Maximum values of SPUE showed large differences, the artificial reefs being more abundant than the natural reefs group (Fig. 3). Except for MB and RAM in the artificial reefs group, no statistically significant differences (p < 0.05) were observed among the reef sites of each group. However, the groups were significantly different (p < 0.05) from one another (Tab. 2).
TABLE 2 | Multiple comparison using data on Atlantic goliath grouper Epinephelus itajara abundance presenting p-value for the studied sites: Monobóia (MB); Recifes Artificiais Marinhos (RAM); Balsa Norte (BN); Balsa Sul (BS); Tamboretes (TA); Arquipélago das Graças (AG); Laje dos Lobos (LL). Sites are distributed according to reef types artificial or natural. Kruskal-Wallis test: H (6, N = 316) = 206.2508; p = 0.000 (*Significant values).
Groups | Sites | MB | RAM | BN | BS | TA | AG | LL |
Artificial | MB |
| 0.0091* | 1.0000 | 1.0000 | < 0.001* | < 0.001* | < 0.001* |
RAM | 0.0091* |
| 1.0000 | 1.0000 | < 0.001* | < 0.001* | < 0.001* | |
BN | 1.0000 | 1.0000 |
| 1.0000 | < 0.001* | < 0.001* | < 0.001* | |
BS | 1.0000 | 1.0000 | 1.0000 |
| 0.0023* | 0.0003* | 0.0002* | |
Natural | TA | < 0.001* | < 0.001* | < 0.001* | 0.0023* |
| 1.0000 | 1.0000 |
AG | < 0.001* | < 0.001* | < 0.001* | 0.0003* | 1.0000 |
| 1.0000 | |
LL | < 0.001* | < 0.001* | < 0.001* | 0.0002* | 1.0000 | 1.0000 |
|
FIGURE 3| Boxplots of Atlantic goliath grouper Epinephelus itajara abundances by sampled sites: Artificial reefs (Monobóia (MB), N = 52; Recifes Artificiais Marinhos (RAM), N = 62; Balsa Norte (BN), N = 30; Balsa Sul (BS), N = 16) and natural rock reefs (Tamboretes (TA), N = 65; Arquipélago das Graças (AG), N = 40; Laje dos Lobos (LL), N = 51) in southern Brazil. RDT data pooled from 2007 to 2014. Black dots = outliers, horizontal line in box = median, white box = mean values.
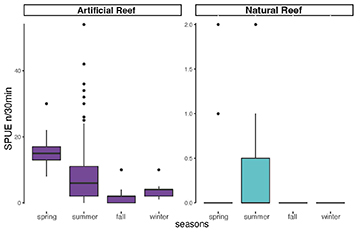
Comparing the abundances on natural and artificial reefs by seasons, AGG were more abundant in artificial reefs in all seasons. The artificial reefs had a higher abundance during spring and summer relative to fall and winter, whereas natural reef abundance remained low during all seasons (Fig. 4).
FIGURE 4| Boxplots of Atlantic goliath grouper Epinephelus itajara abundances by season in all sampled sites in southern Brazil divided into artificial reef group in purple (Monobóia, RAM, Balsa Norte, and Balsa Sul) (Spring N = 27; Summer N = 85; Fall N = 35; Winter N = 13) and natural reef group in teal (Tamboretes, Arquipélago das Graças, and Laje dos Lobos) (Spring N = 37; Summer N = 51; Fall N = 58; Winter N = 10), pooled years 2007 to 2014. Black dots = outliers, horizontal line in box = median.
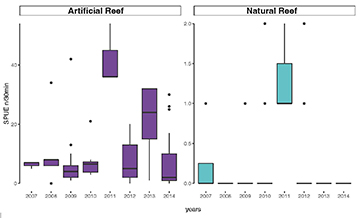
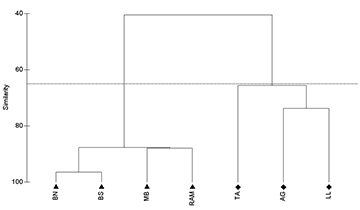
Comparing the abundance between natural and artificial reefs among years, there was low variance in the natural reefs abundance, never being higher than two AGG. On the artificial reefs some variation in abundance was observed, with the higher values being observed in 2011 and 2013. At artificial reef sites the mean abundance was always higher than 7 AGG for all years (Fig. 5). Cluster analysis resulted in the formation of two groups (Fig. 6), the artificial group and the natural group with the artificial reef group significantly different from the natural reefs (ANOSIM, R = 1, p = 0.029).
FIGURE 5| Boxplots of Atlantic goliath grouper Epinephelus itajara abundances in southern Brazil from 2007–2014 in all sampled sites divided into artificial reefs in purple (Monobóia, Recifes Artificiais Marinhos, Balsa Norte, and Balsa Sul) and natural reefs in teal (Tamboretes, Arquipélago das Graças, and Laje dos Lobos). Black dots = outliers, horizontal line in box = median.
FIGURE 6| Atlantic goliath grouper Epinephelus itajara abundance on southern Brazil study sites displayed in a similarity cluster diagram. Balsa Norte (BN); Balsa Sul (BS); Monobóia (MB); Recifes Artificiais Marinhos (RAM); Tamboretes (TA); Arquipélago das Graças (AG); Laje dos Lobos (LL). Triangles () represent artificial reefs and diamonds (♦) represent natural reefs. Horizontal line defined at 67 % of similarity.
Discussion
This study showed that Atlantic goliath grouper no longer occurred on previously known spawning aggregation sites in southern Brazil, apparently preferring regional artificial reefs over the natural reefs. The apparent preference for artificial reefs may be the result of increased habitat relief and complexity and therefore refuge availability where structurally comparable natural habitats are rare (Fig. 2) (Coleman et al., 2010; Collins et al., 2015). Their greater abundance on artificial reefs was observed during spring and summer, coinciding with the reproductive period of this species in the studied region (Felix-Hackardt, Hackradt, 2008; Bueno et al., 2016). Similar patterns were observed by other researchers operating in other parts of their range (Bullock et al., 1992; Colin, 1994; Eklund, Schull, 2001; Sala et al., 2003; Koenig, Coleman, 2009; Collins et al., 2015), whereas their low abundance on natural reefs could result from direct heavy fishing pressure, which would diminish their abundance and possibly reduced their food resources. Reports from fishers in the same region indicating the same (Gerhardinger et al., 2006, 2009).
Considering the great number of artificial reefs deployed near the study sites, it is likely that AGG temporarily uses natural reefs while migrating from their resident reefs to staging areas (Nemeth, 2012). The flux of significant fish biomass from feeding grounds to aggregation sites to reproduce provides an important and largely overlooked ecological component of connectivity within marine ecosystems that alters habitat use, interspecific interactions, and energy transfer resulting from feeding and defecation (Nemeth, 2012). Although little information exists on predator-prey dynamics at aggregation sites, it is known that the AGG feeds at aggregation sites and probably along migratory pathways (Koenig, Coleman, 2009).
According to the local fishers, the natural reefs around AG, TA and LL were historically known as AGG spawning aggregation sites, with more than 60 AGG being seen on one site in a single day. LL was the site with the highest abundance of AGG (> 60) followed by TA (~15), and AG (~5) (Gerhardinger et al., 2009). Our surveys on these sites (LL, TA and AG) showed much lower abundance (highest SPUE; TA, 2; LL, 2 and AG, 1) and low frequency of occurrence (TA, 22%; LL, 4% and AG, 3%) even during the reproductive period (spring and summer). Such abundances suggest there is no longer a formation of spawning aggregations.
Experienced spearfishers have reported never observing more than 2 AGGs on any natural reef studied (LSB, pers. obs.). Also, no AGG were sighted on natural reefs close to site RAM in a reef fish study conducted from October 2008 to August 2009 (Daros et al., 2012). Similar results are found, while performing a checklist of the fishes of LL and nearby natural reef sites in 2006 (Adelir-Alves, Pinheiro, 2011). Studies with reef fish in the Currais Archipelago, Itacolumis Islands, Graças Archipelago and in the Tamboretes Archipelago, conducted between 2007 and 2010, also had similar results (Adelir-Alves et al., 2018; Daros et al., 2018). This suggests that the use of natural environments by AGG decreased concomitant with the increased deployment of artificial reefs at the end of 1999 and beginning of 2000.
The observed patterns of AGG aggregations, shifting from natural to artificial reefs suggests a significant behavioral change for these fishes in southern Brazil. So far, empirical research focusing on how climate change influences fish reproduction is limited but studies on Caribbean fishes have suggested a series of impacts on fish aggregations, from reduced habitat availability including suitable ocean spawning habitats (Gokturk et al., 2022) and increased fishing vulnerability, to range shifts correlated with temperature sensitivity (Fodrie et al., 2010; Maharaj et al., 2018). Such phenology appraisal (Gokturk et al., 2022) should be considered in future studies of AGG, given that the changes we have observed (aggregation shifts from natural to artificial reefs) may have ecological impacts on natural areas, precipitating the loss of an important reef predator and its significant seasonal energy contribution through spawning events. Having a large population of spawning adults can significantly modify food webs as their massive egg output can attract a wide variety of pelagic predators attempting to take advantage of this temporary and predictable food source (Nemeth, 2012).
Atlantic goliath groupers have a positive effect on the biodiversity and abundance of associated reef fishes based in large part on their excavating behavior (Koenig et al., 2011), similar to red grouper Epinephelus morio, which results in a positive influence on species richness at a local scale (Coleman et al., 2010). The excavations increase habitat complexity on reefs buried by sediment suspended by storms thereby increasing habitat complexity making additional structure available for other species (Coleman, Koenig, 2010). Reproductive aggregation events of reef fish, especially large species such as AGG, bring several ecological benefits to the marine ecosystem (e.g., nutrients), attracting several species (Mitcheson, Colin, 2011). Artificial reefs affect the distribution of ichthyofauna, attracting reef fishes that originally would stay on natural reefs (Osenberg et al., 2002). Also, large predatory fishes apparently abandon natural reefs in favor of artificial reefs which can alter ecological processes such as competition and predation (Simon et al., 2011) and perhaps reproductive success. Such changes alter the ecological stability of the ecosystem, consequently lowering resilience and resistance to invasions by exotics species relative to unaltered ecosystems (Stallings, 2009).
Crustaceans and slow-moving fishes constitute the primary food of the AGG (Koenig, Coleman, 2009; Freitas et al., 2015), and the low abundance of these animals combined with the high rugosity of natural reefs hampers the access to prey. The attraction of snappers and groupers to artificial reefs is related to a high prey concentration and lower energy cost for encountering food on artificial reefs (Simon et al., 2011).
Artificial reefs may not represent the solution or the ideal tool to increase fishing production (Bohnsack et al., 1999; Osenberg et al., 2002; Paxton et al., 2020b). Rather, they may simply affect distribution by artificial attracting fish from natural reefs and concentrating them upon artificial ones, making them more available to fishing, as we often see fishers at our artificial study sites. The availability of artificial reefs appears to modify AGG habitat choice (and therefore, distribution) either by providing additional resources that allow to support greater numbers of individuals overall or by simply aggregating individuals into greater densities than would be expected under natural conditions (Koenig, Coleman, 2009; Coleman, Koenig, 2010; Collins et al., 2015). If true, then resources currently spent on artificial reef programs may be misdirected, if not harmful, to natural reef systems (Osenberg et al., 2002).
Artificial reefs with good planning and effective protection, a remote goal in Brazil, may improve reproductive success in some species such as the AGG, but still remove this fish from natural reefs. If the nursery and growth (bottleneck) from the population occur in non-reef habitats, artificial reefs are unlikely to increase abundance or biomass of the species (Bohnsack, 1989), even if they spawn at these reefs. However, most of the studied artificial reefs do not have any laws or regulations to protect these species against fishing, and in those that have, enforcement is very poor. Unplanned installation of artificial reefs without enforcement or management can result in negative impacts on species that are attracted or that are spawning in the nearby areas. Groupers (Epinephelidae) and snappers (Lutjanidae) are two main fish families targeted by fishers in Brazil and therefore more affected by this problem.
In 2020, the Brazilian Energy Research Company (a governmental institution) reported the potential to generate 700 GW along the Brazilian coast, with offshore wind farms installed in areas up to 50m depth (IBAMA, 2022). To date, in spite of the many uncertainties regarding impacts (in land and offshore), the Brazilian government intends to carry on the licensing process, starting in 2023, of 70 projects along the coast (IBAMA, 2022), totaling 12,059 offshore wind turbines. As we are about to face such a huge deployment of artificial structures in the near future, and knowing a myriad of the impacts that will come along (Vasconcellos, Couto, 2021; Galparsoro et al., 2022; Lloret et al., 2022), offshore wind farms turbine structures will pose a new challenge in protecting and monitoring reef fish aggregations along these artificial reefs.
To better understand the dynamics and processes in the marine environment affected by artificial reefs, long-term monitoring and benthic habitat mapping are necessary (Simon et al., 2011). We do recommend the addition of new techniques such as passive-acoustic telemetry to monitor movement patterns, bioacoustics monitoring to identify specific behaviors and the expansion of the spearfisher collaboration network through participatory research and citizen science programs.
These findings provide evidence that artificial reefs support large fish predators, such as the Atlantic goliath grouper, highlighting the need to strengthen the protection of this endangered reef fish during the reproductive aggregations that occur mainly on artificial reefs in southern Brazil.
Acknowledgments
We want to thank Dr. Thiony Simon for his meaningful ideas for this work. We will always be grateful for his valuable contributions to Brazilian science evolution. We are grateful to Thiago Felipe de Souza, Rafael L. Velo and Leonardo F. Machado for their active participation in this research. We also thank all the team of Projeto Meros do Brasil, and all the team of Fish Ecology Lab (Florida State University Coastal & Marine Laboratory).
LSB benefited from a scholarship from FAPES (Fundação de Amparo à Pesquisa do Espírito Santo) and a Sandwich scholarship from CAPES Foundation, Ministry of Education of Brazil from the PDSE program. AAB benefited from a Postdoctoral scholarship from CAPES/PNPD (23102.004667/2014–42) and CNPq/PDS (160133/2018–1). Thanks to André Cattani and Diogo Augusto Moreira. We would like to acknowledge the support from COMAR Institute, Lancha Furacão and Submarine Serviços for providing important technical help for dive operations. The Cebola family in Paraná, for their invaluable support in the field. We thank Projeto Meros do Brasil (sponsored by Petrobras) and Fundação Grupo O Boticário de Proteção à Natureza (Grant: A0018–2016). Last but not least, we posthumously acknowledge Christopher C. Koenig for his profound contributions, unwavering devotion, and deep love for understanding and conserving the ocean. All of us at Projeto Meros do Brasil are deeply grateful for the time spent together and the exchange of ideas. His commitment, strength, and passion for goliath groupers and nature will forever inspire us, as will his charisma and enthusiasm for shaping better researchers and individuals.
References
Adelir-Alves J, Bueno LS, Freitas MO, Spach HL. Unusual habitat for threadfin (Teleostei: Polynemidae) in the South-western Atlantic Ocean. PANAMJAS. 2018; 13(1):131–34. Available from: https://panamjas.org/pdf_artigos/PANAMJAS_13(2)_131-134.pdf
Adelir-Alves J, Pinheiro PC. Peixes recifais das ilhas costeiras do Balneário Barra do Sul, Santa Catarina, Brasil. CEPSUL. 2011; 2(1):10–21. https://doi.org/10.37002/revistacepsul.vol2.30210-21
Aguilar-Perera A. Disappearance of a Nassau grouper spawning aggregation off the southern Mexican Caribbean coast. Mar Ecol Prog Ser. 2006; 327:289–96. https://doi.org/10.3354/meps327289
Barreiros JP, Coleman FC. West African goliath grouper: where are they between Senegal and Angola? Fishes. 2023; 8(6):318. https://doi.org/10.3390/fishes8060318
Bertoncini AA, Aguilar-Perera A, Barreiros J, Craig MT, Ferreira B, Koenig C. Epinephelus itajara (errata version published in 2019) [Internet]. The IUCN Red List of Threatened Species. 2018; e.T195409A145206345. Available from: https://dx.doi.org/10.2305/IUCN.UK.2018-2.RLTS.T195409A145206345.en
Bohnsack JA. Are high densities of fishes at artificial reefs the result of habitat limitation or behavioral preference? Bull Mar Sci. 1989; 44(2):631-45. Available from: https://www.ingentaconnect.com/content/umrsmas/bullmar/1989/00000044/00000002/art00009
Bohnsack JA, Ecklund AM, Szmant AM. Artificial reef research: is there more than the attraction-production issue? Fisheries. 1997; 22:14–23. Available from: https://www.researchgate.net/publication/279561041_Artificial_reef_research_Is_there_more_than_the_attraction-production_issue
Bohnsack JA, McClellan DB, Harper DE, Davenport GS, Konoval GJ, Eklund AM et al. Baseline data for evaluating reef fish populations in the Florida Keys 1979–1998. NOAA Tech, NMFS-SEFSC. 1999; 427:61. Available from: https://www.researchgate.net/publication/265683324_Baseline_Data_For_Evaluating_Reef_Fish_Populations_in_the_Florida_Keys_1979-1998_Miami_FL
Brickhill MJ, Lee SY, Connolly RM. Fishes associated with artificial reefs: attributing changes to attraction or production using novel approaches. J Fish Biol. 2005; 67:53–71. https://doi.org/10.1111/j.0022-1112.2005.00915.x
Bueno LS, Bertoncini AA, Koenig CC, Coleman FC, Freitas MO, Leite JR et al. Evidence for spawning aggregations of the endangered Atlantic goliath grouper Epinephelus itajara in southern Brazil. J Fish Biol. 2016; 89(1):876–89. https://doi.org/10.1111/jfb.13028
Bullock LH, Murphy MD, Godcharles MF, Mitchell ME. Age, growth, and reproduction of jewfish Epinephelus itajara in eastern Gulf of México. Fish Bull. 1992; (90):243–49. Available from: https://spo.nmfs.noaa.gov/content/age-growth-and-reproduction-jewfish-epinephelus-itajara-eastern-gulf-mexico
Coleman FC, Koenig CC. The effects of fishing, climate change, and other anthropogenic disturbances on red grouper and other reef fishes in the Gulf of Mexico. Integr Comp Biol. 2010; 50(2):201–12. https://doi.org/10.1093/icb/icq072
Coleman FC, Koenig CC, Scanlon K, Heppell S, Miller MW. Benthic habitat modification through excavation by red grouper Epinephelus morio (Valenciennes) in the northeastern Gulf of Mexico. TOFISHSJ. 2010; 3(1):1–15. https://doi.org/10.2174/1874401X01003010001
Colin PL. Preliminary investigations of reproductive activity of the Jewfish, Epinephelus itajara (Pisces: Serranidae). In: Goodwin MH, Waugh GT, editors. Proceedings of the 43rd Gulf and Caribbean Fisheries Institute. Miami: Florida; 1994. p.138–47. Available from: https://sedarweb.org/documents/s23rd23-preliminary-investigations-of-reproductive-activity-of-the-jewfish-epinephelus-itajara-pisces-serranidae/
Collins AB, Barbieri LR, McBride RS, McCoy ED, Motta PJ. Reef relief and volume are predictors of Atlantic goliath grouper presence and abundance in the eastern Gulf of Mexico. Bull Mar Sci. 2015; 91(4):399–418. http://dx.doi.org/10.5343/bms.2015.1001
Craig MT, Graham RT, Torres RA, Hyde JR, Freitas MO, Ferreira BP et al. How many species of goliath grouper are there? Cryptic genetic divergence in a threatened marine fish and the resurrection of a geopolitical species. Endanger Species Res. 2009; 7(3):167–74. https://doi.org/10.3354/esr00117
Daros FA, Bueno LS, Soeth M, Bertoncini AA, Hostim-Silva M, Spach HL. Rocky reef fish assemblage structure in coastal islands of southern Brazil. Lat Am J Aquat Res. 2018; 46(1):197–211. http://dx.doi.org/10.3856/vol46-issue1-fulltext-19
Daros FA, Bueno LS, Vilar CC, Passos AC, Spach HL. Checklist of rocky reef fishes from the Currais Archipelago and Itacolomis Island, Paraná state, Brazil. Check List. 2012; 8(3):349–54. https://doi.org/10.15560/8.3.349
Domeier ML, Colin PL. Tropical reef fish spawning aggregations: defined and re-viewed. Bull Mar Sci. 1997; (60):698–726. Available from: https://www.researchgate.net/publication/233709283_Tropical_Reef_Fish_Spawning_Aggregations_Defined_and_Reviewed#:~:text=A%20spawning%20aggregation%20was%20broadly,greater%20number%20and%20take%20up
Eklund AM, Schull J. A stepwise approach to investigating the movement patterns and habitat utilization of goliath grouper, Epinephelus itajara, using conventional tagging, acoustic telemetry and satellite tracking. In: Sibert JR, Nielsen JL, editors. Electronic tagging and tracking in marine fisheries. Springer Netherlands; 2001. p.189–216. https://doi.org/10.1007/978-94-017-1402-0_9
Felix-Hackardt FC, Hackradt CW. Populational study and monitoring of the goliath grouper, Epinephelus itajara (Lichtenstein, 1822), in the coast of Paraná, Brazil. Natureza & Conservação. 2008; 6(2):139–54. Available from: https://www.researchgate.net/publication/229066955_Populational_study_and_monitoring_of_the_goliath_grouper_Epinephelus_itajara_Lichtenstein_1822_in_the_coast_of_Parana_Brazil
Ferreira BP, Hostim-Silva M, Bertoncini AA, Coleman FC, Koenig CC. Atlantic goliath grouper – Epinephelus itajara. In: Sadovy MY, Colin PL, editors. Reef fish spawning aggregations: biology, research and management. Corvallis, USA: Fisheries & Wildlife Department, Oregon State University, Fish & Fisheries Series; 2011. p.417–22.
Fodrie FJ, Heck KL, Powers SP, Graham WM, Robinson KL. Climate-related, decadal-scale assemblage changes of seagrass-associated fishes in the northern Gulf of Mexico. Glob Chang Biol. 2010; 16(1):48–59. https://doi.org/10.1111/j.1365-2486.2009.01889.x
Freitas MO, Abilhoa V, Giglio VJ, Hostim-Silva M, Moura RL, Francini-Filho RB et al. Diet and reproduction of the goliath grouper, Epinephelus itajara (Actinopterygii: Perciformes: Serranidae), in eastern Brazil. Acta Ichthyol Piscat. 2015; 45(1):1–11. https://doi.org/10.3750/AIP2015.45.1.01
Galparsoro I, Menchaca I, Garmendia JM, Borja A, Maldonado AD, Iglesias G et al. Reviewing the ecological impacts of offshore wind farms. NPJ Ocean Sustain. 2022; 1(1):1–08. https://doi.org/10.1038/s44183-022-00003-5
Gerhardinger LC, Medeiros R, Marenzi RC, Bertoncini AA, Hostim-Silva M. Local ecological knowledge on the goliath grouper Epinephelus itajara. Neotrop Ichthyol. 2006; 4(4):441–50. https://doi.org/10.1590/S1679-62252006000400008
Gerhardinger LC, Hostim-Silva M, Medeiros RP, Matarezi J, Bertoncini AA, Freitas MO et al. Fishers’ resource mapping and goliath grouper Epinephelus itajara (Serranidae) conservation in Brazil. Neotrop Ichthyol. 2009; 7(1):93–102. https://doi.org/10.1590/S1679-62252009000100012
Gokturk EN, Bartlett BS, Erisman B, Heyman W, Asch RG. Loss of suitable ocean habitat and phenological shifts among grouper and snapper spawning aggregations in the Greater Caribbean under climate change. Mar Ecol Prog Ser. 2022; 699:91–115. https://doi.org/10.3354/meps14165
Hackradt CW, Félix-Hackradt FC, García-Charton JA. Influence of habitat structure on fish assemblage of an artificial reef in southern Brazil. Mar Environ Res. 2011; 72(5):235–47. https://doi.org/10.1016/j.marenvres.2011.09.006
Harvey E, Fletcher D, Shortis M. Estimation of reef fish length by divers and by stereo-video: a first comparison of the accuracy and precision in the field on living fish under operational conditions. Fish Res. 2002; 57(3):255–65. https://doi.org/10.1016/S0165-7836(01)00356-3
Helfman GS, Schultz ET. Social transmission of behavioural traditions in a coral reef fish. Animal Behaviour. 1983; 32(2):379–84. https://doi.org/10.1016/S0003-3472(84)80272-9
Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis (IBAMA). Mapas de projetos em licenciamento – Complexos Eólicos Offshore [Internet]. Brasília; 2022. Available from: https://www.gov.br/ibama/pt-br/assuntos/laf/consultas/mapas-de-projetos-em-licenciamento-complexos-eolicos-offshore
Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio). Livro Vermelho da Fauna Brasileira Ameaçada de Extinção: Volume VI – Peixes. Brasília: ICMBio/MMA; 2018. Available from: https://www.gov.br/icmbio/pt-br/centrais-de-conteudo/publicacoes/publicacoes-diversas/livro_vermelho_2018_vol1.pdf
Jones RS, Thompson MJ. Comparison of Florida reef fish assemblages using a rapid visual technique. Bull Mar Sci. 1978; 28(1):159–72. Available from: https://www.ingentaconnect.com/content/umrsmas/bullmar/1978/00000028/00000001/art00011?crawler=true
Koenig C, Bueno L, Coleman F, Cusick J, Ellis R, Kingon K et al. Diel, lunar, and seasonal spawning patterns of the Atlantic goliath grouper, Epinephelus itajara, off Florida, United States. Bull Mar Sci. 2017; 93(2):391–406. https://doi.org/10.5343/bms.2016.1013
Koenig CC, Coleman FC. Population density, demographics, and predation effects of adult goliath grouper (Epinephelus itajara). Silver Spring, MD: Final Report to NOAA MARFIN for Project NA05NMF4540045; 2009. Available from: https://www.reef.org/sites/default/files/SEDAR23_RD_19_GoliathFINAL.pdf
Koenig CC, Coleman FC, Eklund AM, Schull J, Ueland J. Mangroves as essential nursery habitat for goliath grouper (Epinephelus itajara). Bull Mar Sci. 2007; 80:567–86. Available from: https://sedarweb.org/documents/s23rd07-mangroves-as-essential-nursery-habitat-for-goliath-grouper-epinephelus-itajara/
Koenig CC, Coleman FC, Kingon K. Pattern of recovery of the goliath grouper Epinaphelus itajara population in the southeastern US. Bull Mar Sci. 2011; 87(4):891–911. https://doi.org/10.5343/bms.2010.1056
Lindberg JW. Can science resolve the attraction-production issue? Fisheries. 1997; (22):10–13.
Lloret J, Turiel A, Solé J, Berdalet E, Sabatés A, Olivares A et al. Unravelling the ecological impacts of large-scale offshore wind farms in the Mediterranean Sea. Sci Total Environ. 2022; 824:153803. http://dx.doi.org/10.1016/j.scitotenv.2022.153803
Maharaj RR, Lam VW, Pauly D, Cheung WWL. Regional variability in the sensitivity of Caribbean reef fish assemblages to ocean warming. Mar Ecol Prog Ser. 2018; 590:201–09. https://doi.org/10.3354/meps12462
Mann DA, Locascio JV, Coleman FC, Koenig CC. Goliath grouper (Epinephelus itajara) sound production and movement patterns on aggregation sites. Endanger Species Res. 2009; 7(3):229–36. https://doi.org/10.3354/esr00109
Medeiros AP, Ferreira BP, Betancur‐R R, Cardoso AP, Matos MR, Santos BA. Centenary shipwrecks reveal the limits of artificial habitats in protecting regional reef fish diversity. J Appl Ecol. 2021; 59(1):286–99. https://doi.org/10.1111/1365-2664.14053
Mitcheson YS, Colin PL, editors. Reef fish spawning aggregations: biology, research and management. Fish & Fisheries Series, vol 35. Dordrecht: Springer; 2011.
Nemeth RS. Ecosystem aspects of species that aggregate to spawn. In: Mitcheson YS, Colin P, editors. Reef fish spawning aggregations: biology, research and management. Fish & Fisheries Series. 2012; 35:21–55. https://doi.org/10.1007/978-94-007-1980-4_2
Osenberg CW, Mary CM, Wilson JA, Lindberg JW. A quantitative framework to evaluate the attraction-production controversy. ICES J Mar Sci. 2002; 59:214–21. https://doi.org/10.1006/jmsc.2002.1222
Paxton AB, Newton EA, Adler AM, Van Hoeck RV, Iversen Jr. ES, Taylor JC et al. Artificial habitats host elevated densities of large reef-associated predators. PLoS ONE. 2020b; 15(9):e0237374. https://doi.org/10.1371/journal.pone.0237374
Paxton AB, Shertzer KW, Bacheler NM, Kellison GT, Riley KL, Taylor JC. Meta-analysis reveals artificial reefs can be effective tools for fish community enhancement but are not one-size-fits-all. Front Mar Sci. 2020a; 7:282. https://doi.org/10.3389/fmars.2020.00282
Sadovy Y, Eklund AM. Synopsis of biological data on the Nassau grouper, Epinephelus striatus (Bloch, 1792), and the jewfish, E. itajara (Lichtenstein, 1822). NOAA Technical Report NMFS. 1999. Available from: https://repository.library.noaa.gov/view/noaa/3090
Sala E, Arbuto-Oropeza O, Paredes G, Thompson G. Spawning aggregations and reproductive behavior of reef fishes in the Gulf of California. Bull Mar Sci. 2003; 72(1):103–201. Available from: https://www.researchgate.net/publication/233708233_Spawning_aggregations_and_reproductive_behavior_of_reef_fishes_in_the_Gulf_of_California
Seaman W. Unifying trends and opportunities in global artificial reef research, including evaluation. ICES J Mar Sci. 2002; 59:14–16. https://doi.org/10.1006/jmsc.2002.1277
Simon T, Pinheiro HT, Joyeux JC. Target fishes on artificial reefs: evidences of impacts over nearby natural environments. Sci Total Environ. 2011; 409:4579–84. https://doi.org/10.1016/j.scitotenv.2011.07.057
Sokal RR, Rohlf FJ. Biometry: the principles and practice of statistics in biological research – 3rd Edition. New York: W.H. Freeman and Co.; 1995.
Stallings CD. Fishery-independent data reveal negative effect of human population density on Caribbean predatory fish communities. PLoS ONE. 2009; 4(5):e5333. https://doi.org/10.1371/journal.pone.0005333
Vasconcellos HAS, Couto LC. Estimation of socioeconomic impacts of wind power projects in Brazil’s northeast region using interregional input-output analysis. Renew Sustain Energy Rev. 2021; 149:111376. https://doi.org/10.1016/j.rser.2021.111376
Authors
![]() Leonardo Schlogel Bueno1
Leonardo Schlogel Bueno1 ![]() ,
, ![]() Matheus Oliveira Freitas1,
Matheus Oliveira Freitas1, ![]() Áthila Andrade Bertoncini1,2,
Áthila Andrade Bertoncini1,2, ![]() Christopher Carl Koenig3,
Christopher Carl Koenig3, ![]() Johnatas Adelir-Alves1,
Johnatas Adelir-Alves1, ![]() Jonas Rodrigues Leite1,
Jonas Rodrigues Leite1, ![]() Felipe Alexandre de Lisboa Miranda Daros4 and
Felipe Alexandre de Lisboa Miranda Daros4 and ![]() Maurício Hostim-Silva1,5
Maurício Hostim-Silva1,5
[1] Instituto Meros do Brasil, Rua Benjamin Constant, 67, Centro, 80060-020 Curitiba, PR, Brazil. (LSB) lecomeros@gmail.com (corresponding author), (MOF) serranidae@gmail.com, (JAA) johnatas_alves@yahoo.com, (JRL) jonasipaq@yahoo.com.br.
[2] Programa de Pós-Graduação em Ecologia, Universidade Federal de Santa Catarina, Rua Eng. Agronômico Andrei Cristian Ferreira, s/n, Trindade, 88040-900 Florianópolis, SC, Brazil. (AAB) athilapeixe@gmail.com.
[3] Florida State University Coastal & Marine Laboratory, 3618, Coastal Highway, 98, St. Theresa, 32358, FL, USA. (CCK) ckoenig@fsu.edu.
[4] Universidade Estadual Paulista, UNESP, Campus de Registro, Av. Nelson Brihi Badur, 430, Vila Tupy, 11900-000 Registro, SP, Brazil. (FALMD) felippe.daros@unesp.br.
[5] Laboratório de Ecologia de Peixes Marinhos, Departamento de Ciências Agrárias e Biológicas, Universidade Federal do Espírito Santo, CEUNES, BR-101, km 60, 29932-540 São Mateus, ES, Brazil. (MHS) mhostim@gmail.com.
Authors’ Contribution 

Leonardo Schlogel Bueno: Conceptualization, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Matheus Oliveira Freitas: Investigation, Methodology, Writing-review and editing.
Áthila Andrade Bertoncini: Investigation, Writing-review and editing.
Christopher Carl Koenig: Conceptualization, Supervision, Writing-review and editing.
Johnatas Adelir-Alves: Investigation, Methodology, Writing-review and editing.
Jonas Rodrigues Leite: Investigation, Project administration, Writing-review and editing.
Felipe Alexandre de Lisboa Miranda Daros: Investigation, Writing-review and editing.
Maurício Hostim-Silva: Project administration, Supervision.
Ethical Statement
This work activity was authorized through licenses issued by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio): SISBIO 15080–2 and SISBIO 31719–1.
Competing Interests
The author declares no competing interests.
How to cite this article
Bueno LS, Freitas MO, Bertoncini AA, Koenig CC, Adelir-Alves J, Leite JR, Daros FALM, Hostim-Silva M. Changes or disappearance of the Atlantic goliath grouper Epinephelus itajara (Perciformes: Epinephelidae) spawning aggregation sites in southern Brazil. Neotrop Ichthyol. 2025; 23(2):e240067. https://doi.org/10.1590/1982-0224-2024-0067
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted May 6, 2025
Accepted May 6, 2025
![]() Submitted July 18, 2024
Submitted July 18, 2024
![]() Epub July 11, 2025
Epub July 11, 2025