![]() Fábio Flores-Lopes1,2,
Fábio Flores-Lopes1,2, ![]() Rodrigo Quevedo3,
Rodrigo Quevedo3, ![]() Clarice Bernhardt Fialho,
Clarice Bernhardt Fialho, ![]() André Luiz Netto-Ferreira2 and
André Luiz Netto-Ferreira2 and ![]() Luiz R. Malabarba2
Luiz R. Malabarba2 ![]()
PDF: EN XML: EN | Cite this article
Abstract
Males of Rachoviscus crassiceps have two small longitudinal black marks on pectoral-fin rays associated to hard and thick tissues that develop in the dorsal surface of the first and second branched pectoral-fin rays, covered dorsally with pheromone secretion club cells. Besides that, the interradial membrane between the second and third and sometimes that between the third and fourth branched pectoral fin rays are thicker than between other contiguous rays, and covered ventrally with hypertrophied mucous cells. Courtship signaling is essential for mate recognition and is frequently multimodal. Courtship signals are apparently essential for mate recognition in inseminating characiform species of the families Characidae and Stevardiidae, that show more conspicuous or more developed courtship signals than in external fertilization species. We propose the pectoral fin organ in the inseminating acestrorhamphid R. crassiceps of the subfamily Thayeriinae constitutes a putative multicomponent courtship signal, including chemical (pheromones) and visual signals (black mark on pectoral fin).
Keywords: Club cells, Inseminating, Mucous cells, Pheromone, Thayeriinae.
Os machos de Rachoviscus crassiceps apresentam duas pequenas manchas pretas longitudinais nos raios da nadadeira peitoral associadas a tecidos endurecidos e espessos que se desenvolvem na superfície dorsal do primeiro e segundo raios ramificados da nadadeira peitoral, cobertos dorsalmente por células de secreção de feromônios (club cells). Além disso, a membrana interradial entre o segundo e o terceiro e, às vezes, entre o terceiro e o quarto raios ramificados da nadadeira peitoral é mais espessa do que entre outros raios contíguos e é coberta ventralmente por glândulas de muco hipertrofiadas. A sinalização na corte é essencial para o reconhecimento do parceiro e é frequentemente multimodal. Os sinais de corte são aparentemente essenciais para o reconhecimento do parceiro em espécies inseminadoras de caraciformes das famílias Characidae e Stevardiidae, que apresentam sinais de corte mais conspícuos ou mais desenvolvidos do que em espécies de fertilização externa. Propomos que o órgão da nadadeira peitoral de R. crassiceps, espécie inseminadora de Acestrorhamphidae, subfamília Thayeriinae, constitui um suposto sinal multimodal de corte, incluindo sinais químicos (feromônios) e visuais (mancha preta na nadadeira peitoral).
Palavras-chave: Célula de muco, Club cells, Feromônio, Inseminador, Thayeriinae.
Introduction
The genus Rachoviscus Myers, 1926 includes two species, R. crassiceps Myers, 1926 from coastal river systems from Rio de Janeiro to Santa Catarina, Brazil, and R. graciliceps Weitzman & Cruz, 1981 from blackwater streams in the coastal region from the State of Bahia, Brazil. The systematic history of the genus is complex. It was first proposed as a member of the subfamily Cheirodontinae (Myers, 1926), and was later referred to Paragoniatinae (Géry, 1977) and alternatively to Aphyocharacinae (Mirande, 2009, 2010). Weitzman, Cruz (1981) reasoned that there was too little information available to provide hypotheses of relationships with any specific genera of Tetragonopterinae, but pointed out that the Rachoviscus species were Tetragonopterinae rather than Cheirodontinae. In the description of Nematocharax venustus, Weitzman et al. (1986) discussed its possible relationships with Rachoviscus and Hollandichthys Eigenmann, 1909, two genera endemic to the Atlantic Forest that share with Nematocharax Weitzman, Menezes & Britski, 1986 a “well-toothed jaw”.
After such a controversial period in the classification of Rachoviscus, the discovery that the species of the genus are inseminating (males transfer sperm to female ovaries without internal fertilization), the description of sexual characters hypothetically associated to this reproductive strategy, and the analysis of DNA sequences have provided consistent and congruent data for understanding the relationships of the genus. Partially cystic spermiogenesis, with the nucleus elongating forward and opposing the flagellum, and spermatozoa having long, coiled mitochondria in their midpiece, that would allow movement inside the ovaries, are unique primary sexual characters that support a close relationship between Hollandichthys and Rachoviscus (Quágio-Grassiotto et al., 2012). The presence of a body cavity between the pelvic and anal fins internally housing the anus and urogenital openings in males and females, possibly associated to sperm transfer, is also a supposed synapomorphy grouping the species of the two genera (Bertaco, Malabarba, 2013). Multilocus molecular phylogenies further supported Hollandichthys and Rachoviscus as sister taxa and included in a large clade then classified as Stethaprioninae (Javonillo et al., 2010; Thomaz et al., 2010; Oliveira et al., 2011; Tagliacollo et al., 2012; Ferreira et al., 2024).
Hollandichthys and Rachoviscus were again recovered as sister taxa in a phylogenomics study of the Characidae (Melo et al., 2024), and then classified in a separate family and subfamily (Acestrorhamphidae: Thayeriinae) from other inseminating characiforms. The hypothesis of their close relationships as a crown clade among the externally fertilizing species of Thayeriinae supports the presence of insemination in Hollandichthys and Rachoviscus as independently derived from those found in the Stevardiidae and in the characid subfamily Cheirodontinae.
Courtship signals are apparently essential for mate recognition in several inseminating species of the characid subfamily Cheirodontinae and of the Stevardiidae that show more conspicuous or more developed courtship signals than in external fertilized species (Nelson, 1964a,b; Burns et al., 1995; Malabarba, 1998; Burns, Weitzman, 2005; Weitzman et al., 2005; Menezes, Weitzman, 2009; Ferreira et al., 2011; Amcoff, Kolm, 2014, 2015; Fukakusa, 2020; Malabarba et al., 2020). So far, none of these putative courtship signals have been described in the inseminating species of the Hollandichthys + Rachoviscus clade. Our goal in this study is to investigate the presence of courtship signals in Rachoviscus crassiceps.
Material and methods
External morphology examinations and histological preparations were performed on dead and preserved specimens available in a scientific fish collection (MCP – Museu de Ciências e Tecnologia, Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre, Brazil). Examined specimens were fixed with 10% formaldehyde, as usual in specimens available in fish collections. After this process, the animals were transferred and preserved in 70% alcohol. The entire pectoral fin was removed, dehydrated with ethanol, embedded in paraffin, sectioned at 0.5 µm and stained with routine histological technique of hematoxylin-eosin.
Institutional abbreviations: MHNCI, Museu de História Natural Capão da Imbuia, Curitiba, Brazil; MHNG, Museum d’Histoire Naturelle, Département d’Herpétologie et Ichthyologie, Genève, Switzerland; Museu Nacional, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil; MZUSP, Museu de Zoologia, Universidade de São Paulo, São Paulo, Brazil.
Results
Males of Rachoviscus crassiceps and R. graciliceps are easily distinguished from females by the presence of bony hooks along the entire length and ventral surface of the second and third pelvic-fin rays. Males of both species also present bony hooks on the second to the 15th–20th branched anal-fin rays, distributed from the middle length to the distal portions of each ray.
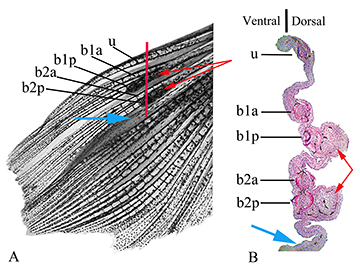
Besides the presence of hooks, males of R. crassiceps can be recognized by the presence of two small longitudinal black marks on pectoral-fin rays. One black mark located on the proximal portion of the posterior ramification of the first branched pectoral-fin ray, and the second on the proximal portion of the posterior ramification of the second branched pectoral-fin ray (Figs. 1, 2).
FIGURE 1| Male of Rachoviscus crassiceps showing the black marks in the pectoral fin (arrow). In this view, the two black lines on the first and second branched pectoral-fin rays are juxtaposed and not discernible as separate (see Fig. 2). Specimen not preserved. Photo by Vinícius Abilhoa.
FIGURE 2| Left pectoral fin of a male of Rachoviscus crassiceps (MCP 37987, 38.2 mm SL). A. Dorsal view of the left pectoral fin before sectioning. B. Histological section of the same fin; the red line in “A”shows approximate location of illustrated sectioning. b1a = anterior branch of the first branched pectoral-fin ray; b1p = posterior branch of the first branched pectoral-fin ray; b2a = anterior branch of the second branched pectoral-fin ray; b2p = posterior branch of the second branched pectoral-fin ray; u = unbranched pectoral-fin ray; red arrows = thick tissues on the dorsal surface of the most proximal portion of the posterior branch of the first branched pectoral-fin ray and on the dorsal surface of the most proximal portion of the posterior branch of the second branched pectoral-fin ray; blue arrows = interradial membrane between the second and third branched pectoral-fin rays.
The two black lines on the first and second branched pectoral-fin rays are respectively associated to thick and somewhat hard tissues that develop in the dorsal surface of the most proximal portion of the posterior branch of the first branched pectoral-fin ray and in the dorsal surface of the most proximal portion of the posterior branch of the second branched pectoral-fin ray (Fig. 2A). The interradial membrane between the second and third branched pectoral fin rays, and sometimes that between the third and fourth branched pectoral fin rays, also differs from the membranous tissue between other pectoral fin rays, being thicker than usual between other contiguous rays (Fig. 2B).
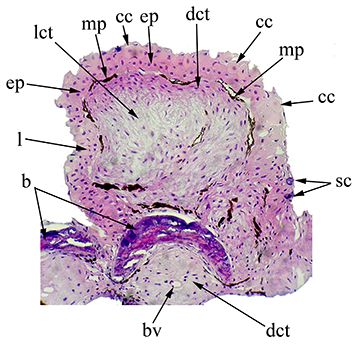
Under histological examination, the structures located dorsally to the fin rays are characterized by a projection of loose and dense conjunctive tissue, covered with melanophores and associated stratified epithelium. The integument in this region is formed by cubical cells, with a high density of club cells around them. The lamina propria of this structure is formed by connective tissue, mainly fibroblasts and matrix (collagen, elastic fiber and reticular fiber), blood cells (lymphocytes) and few blood vessels (Fig. 3).
FIGURE 3| Transversal section of the posterior branch of the second branched pectoral-fin ray of Rachoviscus crassiceps (MCP 37987, 38.2 mm SL) showing the thick tissue located dorsally. Magnification: 400X. b = bone; bv = blood vessel; cc = club cells; dct = dense connective tissue; ep = epithelial tissue; l = lymphocyte; lct = loose connective tissue; mp = melanin pigments; sc = secretory cells.
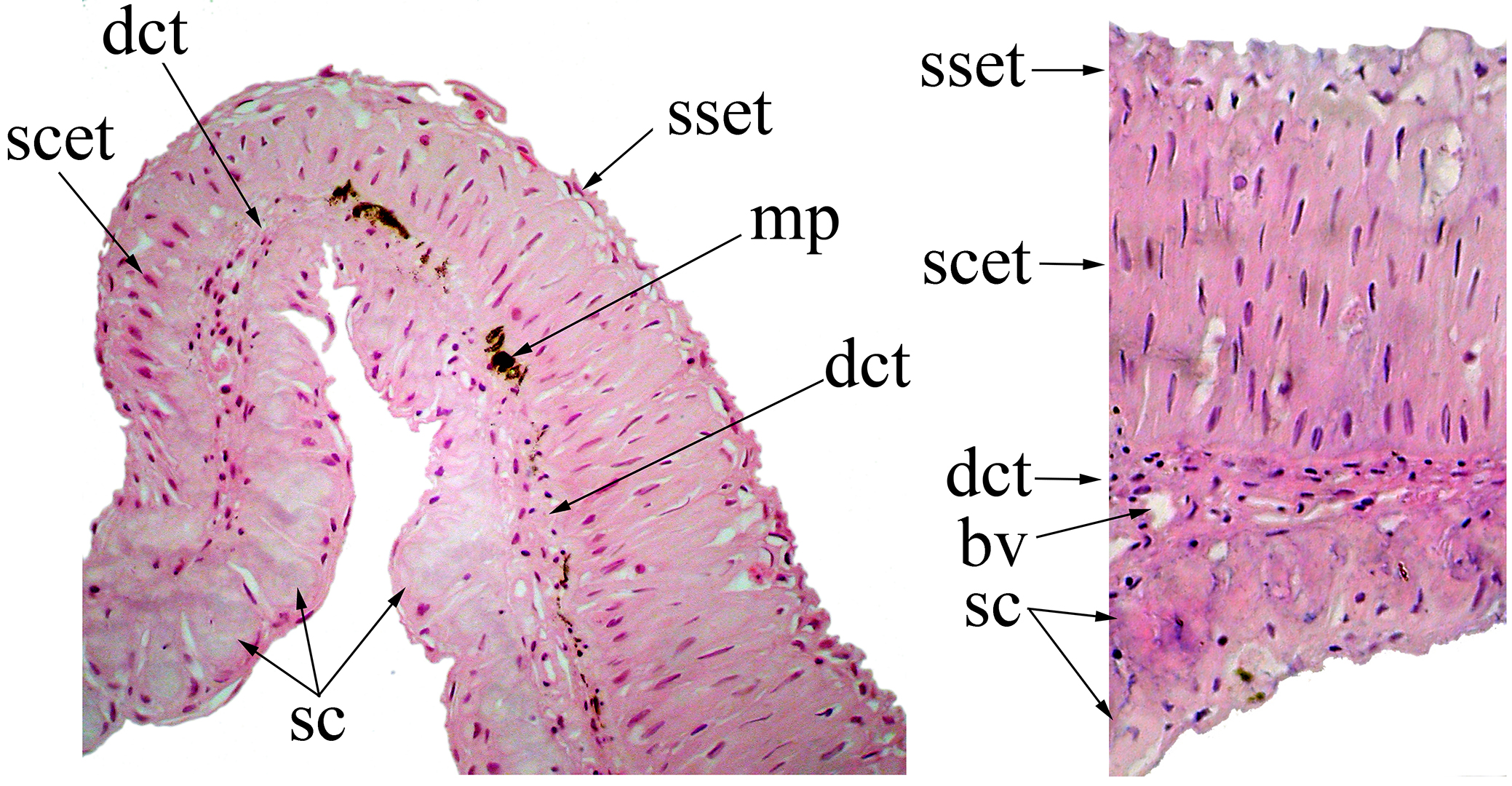
Under histological examination, it is also possible to observe a modified inter-radial membrane between the second and third branched pectoral-fin rays, and sometimes also extending onto the inter-radial membrane between the third and fourth branched pectoral-fin rays (Fig. 4). The membranous tissue is covered dorsally by stratified epithelial tissue, with columnar and squamous cells distally; the ventral surface is constituted of secretory tissue. Just below the basal membrane, the presence of dense connective tissue is observed, with several blood vessels. In this tissue, close to the epithelial tissue, it is possible to observe variably distributed melanophores.
FIGURE 4| Section of the interradial membrane located between the second and third branched pectoral-fin rays of Rachoviscus crassiceps (MCP 37987, 38.2 mm SL) showing the stratified epithelial tissue on the dorsal surface and the secretory cells on the ventral surface of the interradial membrane. bv = blood vessel; dct = dense connective tissue; mp = melanin pigments; sc = secretory cells; scet = stratified columnar epithelial tissue; sset = stratified squamous epithelial tissue.
Examined material. Rachoviscus crassiceps: 37 specimens of 14.3–42.5 mm SL. All from Brazil. Santa Catarina: MCP 37897 (8, 23.0–37.5mm SL; 2 c&s), Ilha de São Francisco do Sul, 26°17’10”S 48°35’17”W; MHNCI 11083 (8, 28.0–42.5 mm SL), Araquari. Paraná: MCP 35554 (3, 24.4–34.7 mm SL), small blackwater stream in area urbanized in the Guaratuba city, 25°52’58”S 48°34’29”W; MCP 37986 (10, 18.1–22.3 mm SL), stream in Brejatuba, Guaratuba Beach; MHNG 2188.72 (1, 15.1 mm SL), Guaratuba Beach, five km of Matinhos; MZUSP 18564 (5, 19.3–23.4 mm SL), Brejatuba Beach, 10 km south of Guaratuba; MZUSP 35723 (2, 14.3–15.2 mm SL), Guaratuba Beach, five km north of Matinhos. Rachoviscus graciliceps: 19 specimens (14.4–36.3 mm SL). All from Brazil, Bahia: MHNG 2514.34 (3 of 4, 14.4–17.5 mm SL), Prado, small blackwater stream tributary of the Rio Jucurucu; MNRJ 10600, paratype, (1, 37.2 mm SL), Prado, small blackwater stream about 1 km north of Prado; MNRJ 19477 (2, 32.0–32.9 mm SL), Prado, small blackwater stream about 500 m from the Atlantic Ocean; MZUSP 90590 (10 of 20, 3 c&s, 25.5–36.3 mm SL) Canavieiras, stream east on the road to Santa Luzia.
Discussion
Courtship signaling is essential for mate recognition and is frequently multimodal. Animals can use chemical signals (pheromones), vocalizations, color patterns, and movements during courtship displays (Mendelson, Shaw, 2012; Higham, Hebets, 2013). Courtship signals are apparently essential for mate recognition in inseminating characiforms species of the families Stevardiidae and Characidae (subfamily Cheirodontinae), that show more conspicuous or more developed courtship signals than in external fertilization species.
Courtship pheromones are chemical signals that play a significant role in reproduction in many taxa as a means of, for example, attracting mates and deterring potential rivals (Wyatt, 2003). The use of chemical signals (pheromones) has been widely hypothesized among stevardiids- and cheirodontine-inseminating species (Nelson, 1964a) based on the presence of hypertrophied glandular tissues associated to modified scales and fin rays of the anal (Weitzman et al., 2005) or caudal fins (Atkins, Fink, 1979; Malabarba, 1998; Menezes, Weitzman, 2009; Ferreira et al., 2011; Fukakusa, 2020), and/or gill filaments on the first gill-arch of male specimens (Burns, Weitzman, 1996; Bushmann et al., 2002; Weitzman et al., 2005; Oliveira et al., 2012; Shrestha et al., 2013).
Hypotheses that these tissues produce a reproduction-related pheromone are mostly based on the empirical observation that hypertrophied glandular cells are only detected in sexually active alpha males examined during the process of courtship behavior (Atkins, Fink, 1979), or by the observation that secondary sexually dimorphic organs that houses hypertrophied glandular tissues are found only or are more developed in mature males (Fukakusa, 2020), and that sometimes are associated to putative pheromone pump mechanisms (Weitzman, Fink, 1985). The hypothesis that these organs secret pheromones was tested by Fukakusa (2020) who demonstrated that the extract of the caudal organ of males of the stevardiid Mimagoniates inequalis (Eigenmann, 1911) includes chemicals that attract females and increases the aggressiveness of males in controlled experiments. The species of Rachoviscus do not have hypertrophied glandular tissues in the caudal or anal fins and neither in the gill filaments. Instead, it is observed an augmented number of club cells and other secretory cells in the epithelial tissue of an organ in the pectoral fin of males of one of its species, R. crassiceps. Based on the above observations made in other inseminating characids, we also hypothesize that the pectoral organ found in R. crassiceps males serves to produce pheromones related to reproduction.
The pectoral-fin organ of R. crassiceps males has one portion located on the dorsal surface of the fin rays covered externally with secretory club cells. The club cells are observed in the skin of Ostariophysan fish (Abd-El Hafez et al., 2013). The cytoplasm of club cells is rather poor in organelles and rich in non-vesicle secretion. Their function is related to the production, storage, and release of an alarm pheromone (Schreckstoff). The disruption of the cytoplasmic membrane of the club cells due to skin lesions results in exposition and release of its content into the water, leading to an alarm reaction in phylogenetically close species (Mokhtar, Abd-El Hafez, 2021). Club cells, ordinarily found deep in the skin, are histologically indistinguishable from the surface club cells found in the anal fins of male Attonitus Vari & Ortega, 2000, Bryconadenos Weitzman, Menezes, Evers & Burns, 2005, or other characids (Weitzman et al., 2005). Ferreira et al. (2011) found that a wide variety of characids have club cells concentrated at the surface of the epidermis of parts of the anterior region of the anal fin of sexually mature males, often associated with the bony hooks present on the anal-fin rays. The large number of epithelial surface club cells found in the pectoral-fin organ of R. crassiceps may have a pheromone function associated with the reproduction, similar to that of surface club cells of other male characiforms.
The pectoral-fin organ of male specimens of R. crassiceps has another portion located in the ventral surface of the interradial membrane covered with hypertrophied mucous cells. In the Stevardiid species Corynopoma riisei Gill, 1858 (Atkins, Fink, 1979) and Hysteronotus megalostomus Eigenmann, 1911, histological sections through the caudal gland region revealed the presence of hypertrophied mucous cells that have been hypothesized as related to sexual pheromone production. Yet, similar hypertrophied mucous cells had not been discovered in the glandular tissue of the caudal fin of other stevardiid genera (Weitzman et al., 2005; Menezes, Weitzman, 2009). The presence of hypertrophied mucous cells in the pectoral-fin organ of males only in R. crassiceps is also suggestive of a possible pheromone reproduction function of that structure.
Courtship signals can also be represented by movements during courtship displays, and these have long been known in several inseminating stevardiids (Nelson, 1964a; Fukakusa, 2011). Visual signals also play important roles in species recognition, inter- and intraspecific competition and mate choice (Andersson, 1994), as observed in males of species of the four genera of the stevardiid subfamily Stevardiinae (Nelson, 1964a,b; Burns et al., 1995; Melo et al., 2024). These visual signs vary from elongate lateral body scales with distal black marks in Pterobrycon Eigenmann, 1913, a paddle-shaped ornament extended posteriorly from the operculum in Corynopoma Gill, 1858 (Nelson, 1964a,b), and the anterior pectoral-fin rays expanded and pigmented distally in at least one species of both genera Gephyrocharax Eigenmann, 1912 and Pseudocorynopoma Perugia, 1891 (Burns, Weitzman, 2005; Malabarba et al., 2020). In Pterobrycon and Corynopoma, these ornaments have been proposed to function as a prey mimic to attract females and as signaling devices during court-ship (Amcoff, Kolm, 2014, 2015; Nelson, 1964a,b). Males of Lophiobrycon weitzmani Castro, Ribeiro, Benine & Melo, 2003 of the stevardiid subfamily Glandulocaudinae have the middle portions of the unbranched and first branched pectoral-fin rays with globular expansions formed by the lepidotrichia and hypertrophied soft tissue (Castro et al., 2003). Even though this structure is located in a partially similar position to that observed in R. crassiceps, and its histological nature is unknown for comparison, the illustration provided by Castro et al. (2003: fig. 5) and the fact that L. weitzmani and R. crassiceps are not closely related supports a hypothesis of non-homology between the organs found in the two species. It is remarkable, however, the independent evolution of similar signaling devices in the pectoral fin of inseminating species of the subfamilies Glandulocaudinae and Stevardiinae of the Stevardiidae and in R. crassiceps of the subfamily Thayeriinae of the Acestrorhamphidae.
Based on the data exposed we propose the pectoral fin organ in R. crassiceps constitutes a putative multicomponent courtship signal, including chemical (pheromones) and visual signals (black mark on pectoral fin possibly associated with courtship movements) of the pectoral fin. Experimental data are required to test this hypothesis.
Acknowledgments
We thank Carlos A. S. Lucena (MCP) for the loan of specimens. LRM and ALNF are partially funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq grant number 308026/2021–7, LRM; grant number 313834/2021–0, ALNF) and Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS grant number 72550.751.48979, ALNF).
References
Abd-El Hafez EA, Mokhtar DM, Abu-Elhamed AS, Hassan AHS. Comparative histomorphological studies on oesophagus of catfish and grass carp. J Histol. 2013:858674. https://doi.org/10.1155/2013/858674
Andersson M. Sexual selection. Princeton, NJ: Princeton University Press. 1994.
Amcoff M, Kolm N. A test of sensory exploitation in the swordtail characin (Corynopoma riisei) based on colour matching between female prey and a male ornament. Environ Biol Fishes. 2014; 97(3):247–54. https://doi.org/10.1007/s10641-013-0147-1
Amcoff M, Kolm N. Multiple male sexual signals and female responsiveness in the swordtail characin, Corynopoma riisei. Environ Biol Fishes. 2015; 98(7):1731–40. https://doi.org/10.1007/s10641-015-0388-2
Atkins DL, Fink WL. Morphology and histochemistry of the caudal gland of Corynopoma riisei Gill. J Fish Biol. 1979; 14(5):465–69. https://doi.org/10.1111/j.1095-8649.1979.tb03543.x
Bertaco VA, Malabarba LR. A new species of the characid genus Hollandichthys Eigenmann from coastal rivers of southern Brazil (Teleostei: Characiformes) with a discussion on the diagnosis of the genus. Neotrop Ichthyol. 2013; 11(4):767–78. https://doi.org/10.1590/S1679-62252013000400004
Burns JR, Weitzman SH, Grier HJ, Menezes NA. Internal fertilization, testis and sperm morphology in glandulocaudine fishes (Teleostei: Characidae: Glandulocaudinae). J Morphol. 1995; 224(2):131–45. https://doi.org/10.1002/jmor.1052240203
Burns JR, Weitzman SH. Novel gill-derived gland in the male swordtail characin, Corynopoma riisei (Teleostei: Characidae: Glandulocaudinae). Copeia. 1996; 1996(3):627–33. https://doi.org/10.2307/1447526
Burns JR, Weitzman SH. Insemination in ostariophysan fishes. In: Grier HJ, Uribe MC, editors. Viviparous fishes. Florida: New Life Publications. 2005; p.107–34.
Bushmann PJ, Burns JR, Weitzman SH. Gill-derived glands in glandulocaudine fishes (Teleostei: Characidae: Glandulocaudinae). J Morphol. 2002; 253(2):187–95. https://doi.org/10.1002/jmor.1120
Castro RMC, Ribeiro AC, Benine RC, Melo ALA. Lophiobrycon weitzmani, a new genus and species of glandulocaudine fish (Characiformes: Characidae) from the rio Grande drainage, upper rio Paraná system, south-eastern Brazil. Neotrop Ichthyol. 2003; 1(1):11–19. https://doi.org/10.1590/S1679-62252003000100002
Ferreira KM, Menezes NA, Quagio-Grassiotto I. A new genus and two new species of Stevardiinae (Characiformes: Characidae) with a hypothesis on their relationships based on morphological and histological data. Neotrop Ichthyol. 2011; 9(2):281–98. https://doi.org/10.1590/S1679-62252011000200005
Ferreira KM, Ribeiro AC, Lima FC, Silva HPD, Ferreira DC, Mirande JM. A new species of Inpaichthys from the rio Canamã, rio Aripuanã basin, Mato Grosso State, Brazil, with a redefinition of the genus (Characidae: Stethaprioninae). Neotrop Ichthyol. 2024; 22(1):e230113. https://doi.org/10.1590/1982-0224-2023-0113
Fukakusa CK. Comportamentos reprodutivos em Mimagoniates inequalis (Eigenmann, 1911) (Characidae: Stevardiinae: Glandulocaudini). Curso de graduação Ciências Biológicas, UFRGS. 2011. Available from: http://hdl.handle.net/10183/35346
Fukakusa CK. Behavioral evidence of chemical communication by male caudal fin organs of a glandulocaudine fish (Teleostei: Characidae). Ichthyol Explor Freshw. 2020; 1127:1–11. http://doi.org/10.23788/IEF-1127
Géry J. Characoids of the world. Neptune City, T. F. H. Publications; 1977.
Higham JP, Hebets EA. An introduction to multimodal communication. Behav Ecol Sociobiol. 2013; 67:1381–88. https://doi.org/10.1007/s00265-013-1590-x
Javonillo R, Malabarba LR, Weitzman SH, Burns JR. Relationships among major lineages of characid fishes (Teleostei: Ostariophysi: Characiformes), based on molecular sequence data. Mol Phylogenet Evol. 2010; 54(2):498–511. https://doi.org/10.1016/j.ympev.2009.08.026
Malabarba LR. Monophyly of the Cheirodontinae, characters and major clades (Ostariophysii: Characidae). In: Malabarba LR, Reis RE, Vari RP, Lucena ZMS, Lucena CAS, editors. Phylogeny and classification of Neotropical fishes. Porto Alegre: Edipucrs; 1998. p.193–234.
Malabarba LR, Chuctaya J, Hirschmann A, Oliveira EB, Thomaz AT. Hidden or unnoticed? Multiple lines of evidence support the recognition of a new species of Pseudocorynopoma (Characidae: Corynopomini). J Fish Biol. 2020; 98(1):219–36. https://doi.org/10.1111/jfb.14572
Melo BF, Ota RP, Benine RC, Carvalho FR, Lima FCT, Mattox GMT et al. Phylogenomics of Characidae, a hyper-diverse Neotropical freshwater fish lineage, with a phylogenetic classification including four families (Teleostei: Characiformes). Zool J Linn Soc. 2024; 202(1):zlae101. https://doi.org/10.1093/zoolinnean/zlae101
Mendelson TC, Shaw KL. The (mis) concept of species recognition. Trends Ecol Evol. 2012; 27(8):421–27. https://doi.org/10.1016/j.tree.2012.04.001
Menezes NA, Weitzman SH. Systematics of the Neotropical fish subfamily Glandulocaudinae (Teleostei: Characiformes: Characidae). Neotrop Ichthyol. 2009; 7(3):295–370. https://doi.org/10.1590/S1679-62252009000300002
Mirande JM. Weighted parsimony phylogeny of the family Characidae (Teleostei: Characiformes). Cladistics. 2009; 25(6):574–613. https://doi.org/10.1111/j.1096-0031.2009.00262.x
Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes): from characters to taxonomy. Neotrop Ichthyol. 2010; 8(3):385–568. https://doi.org/10.1590/S1679-62252010000300001
Mokhtar DM, Abd-El Hafez EA. An overview of the structural and functional aspects of immune cells in teleosts. Histol Histopathol. 2021; 36:399–414. https://doi.org/10.14670/HH-18-302
Myers GS. Eine neue characiniden gattung der unterfamilie Cheirodontinae aus Rio de Janeiro, Brasilien. Blätter für Aquarien und Terrarien-Kunde. 1926; 37(24):1–2.
Nelson K. Behavior and morphology in the Glandulocaudine fishes (Ostariophysi, Characidae). Univ Calif Publ Zool. 1964a; 75:59–152.
Nelson K. The evolution of a pattern of sound production associated with courtship in the characid fish, Glandulocauda inequalis. Evolution. 1964b; 18:526–40.
Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G et al. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol. 2011; 11:275. https://doi.org/10.1186/1471-2148-11-275
Oliveira CLC, Malabarba LR, Burns JR. Comparative morphology of gill glands in externally fertilizing and inseminating species of cheirodontine fishes, with implications on the phylogeny of the family Characidae (Actinopterygii: Characiformes). Neotrop Ichthyol. 2012; 10(2):349–60. https://doi.org/10.1590/S1679-62252012005000005
Quágio-Grassiotto I, Malabarba LR, Azevedo MA, Burns JR, Baicere-Silva CM, Quevedo R. Unique derived features in spermiogenesis and sperm morphology supporting a close relationship between the species of Hollandichthys and Rachoviscus (Characiformes: Characidae). Copeia. 2012; 2012(4):609–25. https://doi.org/10.1643/CG-11-171
Shrestha B, Javonillo R, Burns JR, Pirger Z, Vertes A. Comparative local analysis of metabolites, lipids and proteins in intact fish tissues by LAESI mass spectrometry. Analyst. 2013; 138:3444–49. https://doi.org/10.1039/C3AN00631J
Tagliacollo VA, Souza-Lima R, Benine RC, Oliveira C. Molecular phylogeny of Aphyocharacinae (Characiformes, Characidae) with morphological diagnoses for the subfamily and recognized genera. Mol Phylogenet Evol. 2012; 64(2):297–307. https://doi.org/10.1016/j.ympev.2012.04.007
Thomaz AT, Malabarba LR, Bonatto SL. The phylogenetic placement of Hollandichthys Eigenmann 1909 (Teleostei: Characidae) and related genera. Mol Phylogenet Evol. 2010; 57(3):1347–52. https://doi.org/10.1016/j.ympev.2010.10.006
Weitzman SH, Cruz CAG. The South American fish genus Rachoviscus, with a description of a new species (Teleostei: Characidae). Proc Biol Soc Wash. 1981; 93(4):997–1015.
Weitzman SH, Fink SV. Xenurobryconin phylogeny and putative pheromone pumps in glandulocaudine fishes (Teleostei: Characidae). Smithson Contrib Zool. 1985; 421. https://doi.org/10.5479/si.00810282.421
Weitzman SH, Menezes NA, Britski HA. Nematocharax venustus, a new genus and species of fish from the rio Jequitinhonha, Minas Gerais, Brazil (Teleostei: Characidae). Proc Biol Soc Wash. 1986; 99(2):335–46. Available from: https://www.biodiversitylibrary.org/page/34595802#page/358/mode/1up
Weitzman SH, Menezes NA, Evers H-G, Burns JR. Putative relationships among inseminating and externally fertilizing characids, with a description of a new genus and species of Brazilian inseminating fish bearing an anal-fin gland in males (Characiformes: Characidae). Neotrop Ichthyol. 2005; 3(3):329–60. https://doi.org/10.1590/S1679-62252005000300002
Wyatt TD. Pheromones and animal behaviour: communication by smell and taste. Cambridge university press; 2003.
Authors
![]() Fábio Flores-Lopes1,2,
Fábio Flores-Lopes1,2, ![]() Rodrigo Quevedo3,
Rodrigo Quevedo3, ![]() Clarice Bernhardt Fialho,
Clarice Bernhardt Fialho, ![]() André Luiz Netto-Ferreira2 and
André Luiz Netto-Ferreira2 and ![]() Luiz R. Malabarba2
Luiz R. Malabarba2 ![]()
[1] Departamento de Ciências Biológicas, Universidade Estadual de Santa Cruz, Rodovia Ilhéus-Itabuna, km 16, 45662-000 Ilhéus, BA, Brazil. (FFL) In memorian.
[2] Programa de Pós-Graduação em Biologia Animal, IB, Universidade Federal do Rio Grande do Sul, Av. Bento Gonçalves, 9500, 91501-970 Porto Alegre, RS, Brazil. (CBF) clarice.fialho@ufrgs.br, (ALNF) alnferreira@gmail.com, (LRM) malabarb@ufrgs.br (corresponding author).
[3] Programa de Pós-Graduação em Biociências (Zoologia), Pontifícia Universidade Católica do Rio Grande do Sul, Av. Ipiranga, 6681, 90619-900 Porto Alegre, RS, Brazil. (RQ) professorquevedo@gmail.com.
Authors’ Contribution 

Fábio Flores-Lopes: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft.
Rodrigo Quevedo: Formal analysis, Investigation, Writing-original draft.
Clarice Bernhardt Fialho: Writing-review and editing.
André Luiz Netto-Ferreira: Writing-review and editing.
Luiz R. Malabarba: Conceptualization, Investigation, Methodology, Resources, Supervision, Validation, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
How to cite this article
Flores-Lopes F, Quevedo R, Fialho CB, Netto-Ferreira AL, Malabarba LR. Description of new putative multicomponent courtship signal for mate recognition in Rachoviscus crassiceps (Characiformes: Acestrorhamphidae). Neotrop Ichthyol. 2025; 23(1):e240064. https://doi.org/10.1590/1982-0224-2024-0064
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2025 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted January 17, 2025 by Elizete Rizzo
Accepted January 17, 2025 by Elizete Rizzo
![]() Submitted July 8, 2024
Submitted July 8, 2024
![]() Epub March 31, 2025
Epub March 31, 2025