![]() Lais Reia1
Lais Reia1 ![]() ,
, ![]() Gabriel de Souza Costa e Silva1,2,
Gabriel de Souza Costa e Silva1,2, ![]() Claudio Oliveira1 and
Claudio Oliveira1 and ![]() Ricardo Cardoso Benine3
Ricardo Cardoso Benine3
PDF: EN XML: EN | Supplementary: S1 S2 | Cite this article
Abstract
A new species of Moenkhausia from the rio Culuene, rio Xingu basin, in Mato Grosso State is described here through genetic and morphological data. This new species differs from all congeners by the following combination of characters: the presence of reddish color on the posterior portion of the body, as well as on the base of anal, adipose, and pelvic fins in live specimens; a conspicuous midlateral dark stripe, which extends from posterior margin of opercle to the middle of caudal-fin rays, becoming wider and more conspicuous from the vertical through the dorsal-fin origin; absence of humeral blotch; absence of blotches on caudal-fin lobes; by having the concentration of dark pigments on the anterior margin of the caudal-fin rays; and presence of 2–3 maxillary teeth. A brief discussion about its putative relationships with other congeners is presented.
Keywords: Amazon basin, Mitochondrial DNA, Moenkhausia lopesi, Taxonomy, Tetra.
Uma nova espécie de Moenkhausia do rio Culuene, bacia do rio Xingu é descrita aqui através da utilização de dados morfológicos e genéticos. Esta nova espécie difere de todas as suas congêneres por possuir a seguinte combinação de caracteres: a presença da porção posterior do corpo e a base das nadadeiras anal, adiposa e pélvicas avermelhadas em espécimes vivos; uma faixa meio-lateral escura que se estende da margem posterior do opérculo até os raios medianos da nadadeira caudal, tornando-se larga a partir da origem da nadadeira dorsal; ausência de uma mancha na região umeral; ausência de mancha nos lobos da nadadeira caudal; por ter uma concentração de pigmentos escuros na margem anterior dos raios da nadadeira caudal; e presença de 2–3 dentes no maxilar. Uma breve discussão sobre suas prováveis relações com as demais congêneres é apresentada.
Palavras-chave: Bacia Amazônica, DNA mitocondrial, Moenkhausia lopesi, Taxonomia, Piaba.
Introduction
Moenkhausia was proposed byEigenmann (1903) and defined by himself (Eigenmann, 1917) by the following character combination: a complete pored lateral line, premaxillary teeth in two rows, with at least five teeth in the inner row, and a caudal fin partially covered by scales. Nevertheless, phylogenetic studies established the homoplastic condition of those characters and the non-monophyly of Moenkhausia (Mirande, 2010, 2019; Mariguela et al., 2013). Currently, Moenkhausia comprises100 valid species widely distributed across the main South American rivers (Fricke et al., 2023), with its greater diversity in the cis-Andean region, being considered the third most species-rich genus in the Amazonian basin (Marinho et al., 2016; Dagosta, de Pinna, 2019).
A recent expedition to the rio Culuene, upper rio Xingu basin, revealed the existence of a new species of Moenkhausia, which is described here using the integrative approach. In addition to the description, we provide a discussion on its possible phylogenetic relationships.
Material and methods
Morphological analysis.Counts and measurements follow Fink, Weitzman (1974), and Menezes, Weitzman (1990) with the exception of the counts of the horizontal scales series below the lateral line, which was counted at the row below the lateral line to the pelvic-fin origin and addition of the head depth, which was measured at the vertical through the middle of the orbit. Measurements were taken point to point using a digital caliper (0.1 mm) on the left side of specimens whenever possible and, as well as the counts, performed under a stereomicroscope. Specimens were cleared and counterstained (c&s) following the procedure of Taylor, Van Dyke (1985). The number of vertebrae, supraneurals, procurrent, and caudal-fin rays were taken from two c&s specimens and posterior small dentary teeth from four specimens, which were dissected, clear, and stained only jaws. The four vertebrae of the Weberian apparatus were counted as four separate elements, and the fused PU1+U1 as a single element. The frequency of each count is provided in parentheses after the respective range, with the count of the holotype marked by an asterisk. The sex was determined based on pelvic-fin elongation and confirmed by direct examination of gonads, under a stereomicroscope, which follows the methodology of Vazzoler (1996). The covariance analysis was realized with eight males and eight females using the package “car” (Fox, Weisberg, 2019) in the software RStudio v. 2023.06.1 (RStudio Team, 2020). Data of Moenkhausia iris Marinho & Dagosta, 2023 and M. rubra Pastana & Dagosta, 2014 were taken in the original description. Institutional abbreviations follow Sabaj (2020).
Molecular data and analysis.DNA extraction followed Ivanova et al. (2006) and partial sequences of the mitochondrial gene cytochrome c oxidase subunit I (COI) were amplified by polymerase chain reaction (PCR), with primers FishF1/R1and FishF2/R1 described by Ward et al. (2005) and FishF6/R7 described by Jennings et al. (2019). Reactions were carried out in a 12.5 μL reaction volume containing 1.25 μL of 10× PCR buffer, 0.40 μL MgCl2 (50 mM), 0.30 μL dNTPs (2 mM), 0.25 μL of each primer (5 μM), 0.20 μL of PHT Taq DNA polymerase (Phoneutria), and 2 μL DNA template (200 ng), and 7.85 μL of ddH2O. The PCR consisted of denaturation (5 min at 95°C) followed by 30 cycles of denaturation (1 min at 95°C), primer hybridization (45 sec at 52°C), nucleotide extension (1 min at 68°C), and a final extension (10 min at 68°C). All PCR products were checked using 1% agarose gel and purified with ExoSap-IT (USB Corporation) following the manufacturer’s instructions. The purified PCR products were sequenced using the Big DyeTM Terminator v. 3.1 Cycle Sequencing Ready Reaction Kit (Applied Biosystems, Austins, USA), and purified through ethanol precipitation. Amplified fragments were then loaded into an ABI 3500 Genetic Analyzer (Applied Biosystems), in the Instituto de Biotecnologia (IBTEC), Instituto de Biociências, Universidade Estadual Paulista Júlio de Mesquita Filho, Botucatu, Brazil.
For this study, we generated three sequences of the new species and 22 sequences of the other three valid species of Moenkhausia. We also used three sequences obtained from Genbank, including the root Psellogrammus kennedyi (Eigenmann, 1903). For more details about sequences and Genbank numbers, see Tab. 1. The sequences were assembled using the software Geneious 7.1.4 (Kearse et al., 2012) and aligned with Muscle (Edgar, 2004) under default parameters. To evaluate the occurrence of substitution saturation in our molecular data, we estimated the index of substitution saturation (Iss) using the method described by Xia et al. (2003) and Xia, Lemey (2009) with the software DAMBE 7.2.1 (Xia, 2018).
TABLE 1 | Collection, vouchers, distributions, and Genbank number of species analyzed.
Species | Collection | Voucher | Locality (river/basin/city/state/country) | Geographic coordinates | Genbank |
Moenkhausia aurantia | LBP 18999 | 75410 | Roncador/Tocantins basin/São João da Aliança/GO/Brazil | 14°43’51.3”S 47°32’34.0”W | OR922605 |
Moenkhausia sp. n. | LBP 16063 | 66490 | Culuene/Xingu basin/Primavera do Leste/MT/Brazil | 14°38’21.2”S 53°55’35.3”W | OR922604 |
Moenkhausia sp. n. | LBP 30660 | 105627 | Culuene/Xingu basin/Primavera do Leste/MT/Brazil | 14°38’21.2”S 53°55’35.3”W | OR922602 |
|
| 105628 | Culuene/Xingu basin/Primavera do Leste/MT/Brazil | 14°38’21.2”S 53°55’35.3”W | OR922603 |
Moenkhausia bonita | CI-FML 7395 | A-4 | Bermejo basin/Orán/Salta/Argentina | 23°07’00.0”S 64°30’00.0”W | MK928340 |
Moenkhausia bonita | LBP 3783 | 22244 | Negro/Paraguai basin/Aquidauana/MS/Brazil | 19°34’17.3”S 56°14’44.8’’W | OR922616 |
|
| 22246 | Negro/Paraguai basin/Aquidauana/MS/Brazil | 19°34’17.3”S 56°14’44.8’’W | OR922619 |
Moenkhausia bonita | LBP 3740 | 22186 | Negro/Paraguai basin/Aquidauana/MS/Brazil | 19°34’54.6”S 56°15’16.5’’W | OR922609 |
Moenkhausia bonita | LBP 8153 | 38041 | Cacequi/Uruguai basin/Casequi/RS/Brazil | 29°53’51.1”S 54°51’05.0”W | OR922607 |
Moenkhausia bonita | LBP 8546 | 43281 | Sepotuba/Paraguay basin/Tangará da Serra/MT/Brazil | 14°20’32.6” 57°31’22.5’’W | OR922618 |
|
| 43283 | Sepotuba/Paraguay basin/Tangará da Serra/MT/Brazil | 14°20’32.6” 57°31’22.5’’W | OR922617 |
|
| 43284 | Sepotuba/Paraguay basin/Tangará da Serra/MT/Brazil | 14°20’32.6” 57°31’22.5’’W | OR922608 |
Moenkhausia bonita | LBP 9699 | 45818 | São Bento/upper Paraná basin/Nova Andradina/MS/Brazil | 22°08’56.9”S 53°25’34.8”W | OR922612 |
|
| 45819 | São Bento/upper Paraná basin/Nova Andradina/MS/Brazil | 22°08’56.9”S 53°25’34.8”W | OR922615 |
Moenkhausia bonita | LBP 13171 | 55083 | Uruguay basin/Uruguaiana/RS/Brazil | 29°30’42.8”S 56°43’09.9”W | OR922610 |
|
| 55084 | Uruguay basin/Uruguaiana/RS/Brazil | 29°30’42.8”S 56°43’09.9”W | OR922611 |
Moenkhausia bonita | LBP 13171 | 55085 | Uruguay basin/Uruguaiana/RS/Brazil | 29°30’42.8”S 56°43’09.9”W | OR922614 |
|
| 55086 | Uruguay basin/Uruguaiana/RS/Brazil | 29°30’42.8”S 56°43’09.9”W | OR922613 |
Moenkhausia bonita | LBP 12158 | 51913 | Paraguay basin/Cáceres/MT/Brazil | 16°03’48.7”S 57°42’27.0”W | OR922620 |
|
| 51914 | Paraguay basin/Cáceres/MT/Brazil | 16°03’48.7”S 57°42’27.0”W | OR922621 |
Moenkhausia aff. lopesi | LBP 8550 | 43297 | Sepotuba/Paraguai basin/Tangará da Serra/MT/Brazil | 14°20’32.6”S 57°31’22.5’’W | OR922606 |
Moenkhausia aff. lopesi | LBP 30595 | 105766 | Mutum/Juruena basin/Padronal/MT/Brazil | 13°05’08.7”S 59°53’32.0”W | OR922601 |
Moenkhausia lopesi | LBP 33989 | 114736 | Correntes/Piquiri basin/Paraguai/basin/Sonora/MS/Brazil | 17°36’21.0”S 54°11’18.0”W | OR922622 |
|
| 114737 | Correntes/Piquiri basin/Paraguai/basin/Sonora/MS/Brazil | 17°36’21.0”S 54°11’18.0”W | OR922623 |
Moenkhausia cf. lopesi | LBP 10237 | 47790 | Manso/Paraguay basin/Campo Verde/MT/Brazil | 15°30’17.2”S 55°21’53.9”W | OR922624 |
|
| 47791 | Manso/Paraguay basin/Campo Verde/MT/Brazil | 15°30’17.2”S 55°21’53.9”W | OR922625 |
Hemigrammus marginatus | 930249 | DCC01369 | São Francisco basin/MG/Brazil | 19°46’26.4”S 45°27’46.8”W | HM906017 |
Psellogrammus kennedyi | LBP 5220 | 26408 | upper rio Paraná basin/Porto Rico/MS/Brazil | 22°47’29”S 53°20’58”W | JN989170 |
The best-fit model of nucleotide evolution was selected according to Akaike Information Criterion with corrections for small sample sizes (AICc). The overall mean genetic distances (among all specimens), as well as interspecific (among species group) and intraspecific distances (among specimens of each species group), were estimated with 1.000 pseudoreplicates and without root. These previous analyses were estimated using MEGA v. 11 (Tamura et al., 2021). Maximum likelihood (ML) analysis was performed in RaxML PTHREADS-SSE3 v. 8 (Stamatakis, 2014) using the GTRGAMMA model in the Gymnotus server at LBP-UNESP. The best tree was accessed through ten random searches with 1,000 bootstrap pseudoreplicates using the autoMRE function with bootstopping criteria (Pattengale et al., 2010), which ran 800 pseudoreplicates. The resulting ML tree was used as an input tree for the Poisson Tree Process model (PTP) analysis (Zhang et al., 2013), which was performed on the PTP web server (https://species.h-its.org), with the option “remove outgroup” and the others parameters in default. The analysis of Assemble Species by Automatic Partitioning (ASAP) (ASAP; Puillandre et al., 2020) is available in the ASAP webserver (https://bioinfo.mnhn.fr/abi/public/asap/asapweb.html) with model Jukes-Cantor (JC69).
Results
Moenkhausia aurora, new species
urn:lsid:zoobank.org:act:003754E6-0E2E-4F64-A8FD-4E723C3BA46D
(Figs. 1–3; Tab. 2)
Holotype. LBP 34895, 37.9 mm SL, Brazil, Mato Grosso State, municipality of Primavera do Leste, rio Culuene, upper rio Xingu basin, 14°38’21.2”S 53°55’35.3”W, 23 Aug 2021, L. Reia, G. S. C. Silva, C. S. Souza & E. V. Ywamoto.
Paratypes. All from Brazil. LBP 16063, 26, 26.5–36.8 mm SL, same locality of the holotype, 5 Aug 2012, C. Oliveira, M. Taylor, G. J. C. Silva & J. H. M. Martinez. LBP 30660, 36, 23.3–36.7 mm SL, 2 c&s, 31.9–32.8 mm SL, same data of holotype. ANSP 208909, 3, 31.0–34.0 mm SL; MNRJ 54695, 3, 30.8–33.7 mm SL; MZUEL 23348, 3, 30.2–34.7 mm SL, MZUSP 118284, 194, 17.6–43.0 mm SL, Mato Grosso State, municipality of Primavera do Leste, stream tributary of rio Culuene, rio Xingu basin, 14°43’04.4”S 54°04’38.2”W, 17 Nov 2014, F. C. P. Dagosta, W. M. Ohara & V. Giovannetti.
Diagnosis. Moenkhausia aurora is distinguished from all congeners, except M. rubra, and M. iris by the presence of reddish body color in life specimens. The new species can be readily distinguished from M. rubra by having the base of pelvic and anal fins reddish in live specimens (vs. pelvic and anal fins hyaline), and by the absence of dark pigmentation on anteriormost rays of the anal fin (vs. presence). Moenkhausia aurora differs from M. iris by lower number of scales between lateral line and pelvic-fin origin (4 vs. 5), and by a lower number of unbranched anal-fin rays (iii vs. iv-v). Moenkhausia aurora is also distinguished from all congeners, except M. bonita Benine, Castro & Sabino, 2004, M. celibela Marinho & Langeani, 2010, M. dichroura (Kner, 1858), M. intermedia Eigenmann, 1908, and M. lopesi Britski & Silimon 2001, by the absence of a humeral blotch (variable in M. dichroura and M. intermedia see Lima et al., 2020 vs. presence). The new species differs from M. lopesi by having, in life specimens, a reddish color on the posterior portion of the body, as well as on the base of the anal, adipose, and pelvic fins (Fig. 3A) (vs. yellowish, Fig. 3B), and by the lower number of maxillary teeth (2–3 vs. 3–7). Moenkhausia aurora can be distinguished from M. bonita, M. celibela, M. dichroura, and M. intermedia by the absence of dark pigmentation on caudal-fin lobes (vs. two black blotches on caudal fin, one on each lobe in M. bonita, M. dichroura, and M. intermedia, and one blotch on dorsal lobe in M. celibela). Moenkhausia aurora can be distinguished from M. dichroura and M. intermedia by a lower number of gill rakers on the first arch (9–14+1+ 6–8 vs. 18–22 +1+ 10–11). Additionally, M. aurora differs from M. lopesi, M. bonita, M. celibela, M. dichroura, and M. intermedia by having a concentration of dark pigments on the anterior margin of the caudal-fin rays (Figs. 1, 3A) (vs. scarcely pigmented).
FIGURE 1| Moenkhausia aurora, Brazil, Mato Grosso State, municipality of Primavera do Leste, rio Culuene. A. LBP 34895, 37.5 mm SL, holotype, male; B. LBP 30660, 34 mm SL, paratype, male; C. LBP 30660, 31.4 mm SL, paratype, male.
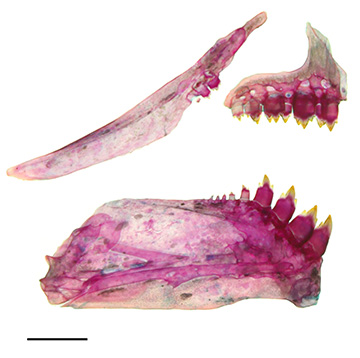
FIGURE 2| Medial view of the left side of maxilla, premaxilla, and dentary. Moenkhausia aurora, paratype, LBP 16660, 34.8 mm SL, Brazil, Mato Grosso, Primavera do Leste, rio Culuene. Scale bar = 1 mm.
FIGURE 3| Color in life of Moenkhausia aurora (A), paratype, LBP 30660, Brazil, Mato Grosso State, municipality of Primavera do Leste, rio Culuene. Specimen not measured. Photo by Eric V. Ywamoto; and Moenkhausia lopesi (B) from Mato Grosso State, municipality of Sonora, rio Piquiri drainage, rio Paraguay basin. Specimen not measured. Photo by Heriberto G. Junior.
Description. Data summarized in Tab. 2. Small-sized species, largest specimen examined 43.0 mm SL. Body compressed laterally, moderately elongated, greatest body depth at dorsal-fin origin. Dorsal profile of head convex from its tip to vertical through posterior nostril; straight to slightly convex from that point to tip of supraoccipital spine. Dorsal profile of body convex from tip of supraoccipital spine to dorsal-fin origin; dorsal-fin base slightly convex to straight and posteroventrally inclined; slightly convex to straight from last dorsal-fin rays to adipose-fin insertion; adipose-fin base slightly inclined posteroventrally; slightly concave from terminus of adipose-fin base to anteriormost dorsal procurrent caudal-fin rays. Ventral profile of head slightly convex to straight from chin to isthmus. Ventral profile of body slightly convex to anal-fin origin; anal-fin base straight and inclined posterodorsally; slightly concave from terminus of anal fin base to anteriormost ventral procurrent caudal-fin ray.
TABLE 2 | Morphometric data for holotype and paratypes of Moenkhausia aurora. SD = Standard deviation.
| Holotype | N | Range | Mean | SD |
Standart length | 37.9 | 44 | 28.1-43.0 | 34.2 | – |
Porcentages of standart length | |||||
Greatest depth | 36.8 | 44 | 29.9-36.8 | 34.0 | 1.7 |
Snout to dorsal-fin origin | 52.0 | 44 | 49.8-54.2 | 51.6 | 1.0 |
Snout to pelvic-fin origin | 47.2 | 44 | 41.4-51.0 | 47.5 | 1.7 |
Snout to pectoral-fin origin | 29.4 | 44 | 26.7-30.1 | 28.4 | 0.8 |
Snout to anal-fin origin | 63.6 | 44 | 60.1-67.2 | 62.9 | 1.8 |
Caudal peduncle depth | 11.8 | 44 | 10.4-12.3 | 11.4 | 0.5 |
Caudal peduncle length | 11.4 | 44 | 9.7-13.9 | 11.4 | 0.9 |
Pectoral-fin length | 25.3 | 44 | 21.1-26.3 | 23.8 | 1.2 |
Pelvic-fin length | 21.0 | 44 | 15.5-21.6 | 18.3 | 1.5 |
Dorsal-fin length | 32.4 | 43 | 25.5-33.5 | 29.5 | 1.9 |
Dorsal-fin base | 14.5 | 44 | 12.4-15.7 | 13.9 | 0.7 |
Anal-fin length | 20.8 | 43 | 16.9-22.8 | 19.5 | 1.4 |
Anal-fin base | 30.8 | 44 | 26.9-34.0 | 30.3 | 1.8 |
Eye to dorsal-fin origin | 36.6 | 44 | 34.0-38.9 | 36.7 | 1.2 |
Dorsal-fin origin to caudal-fin origin | 53.7 | 44 | 50.6-55.3 | 52.7 | 1.2 |
Head depth | 19.9 | 43 | 18.4-22.3 | 20.7 | 0.9 |
Head length | 27.8 | 44 | 26.0-29.5 | 27.6 | 0.8 |
Porcentages of head length | |||||
Orbital diameter | 40.0 | 44 | 36.9-47.4 | 42.2 | 2.5 |
Interorbital distance | 32.6 | 44 | 30.2-34.4 | 32.0 | 1.2 |
Snout length | 27.2 | 44 | 20.0-31.6 | 26.7 | 3.3 |
Upper jaw length | 48.4 | 44 | 46.8-53.3 | 50.3 | 1.7 |
Eyes large. Mouth terminal. Premaxillary teeth in two rows. Outer row with 3(6), 4*(26) or 5(10) tricuspid teeth; inner row with 5(40) or 6*(2) pentacuspid teeth, the last tooth varying from tricuspid to pentacuspid. Maxillary with 2*(27) or 3(14) pentacuspid to tricuspid teeth. Tip of maxilla through vertical reaching posterior half of second infraorbital. Dentary with 4*(42) pentacuspid teeth, followed by one small tricuspid tooth and a row varying between 5–13(4 c&s) small conic teeth. Central cuspid in all teeth more developed than lateral cusps (Fig. 2). First gill arch with 6(3), 7*(20), 8(5) gill rakers on upper limb, 1(28) gill raker on intermediate cartilage and 9(1), 11*(6), 12(20), 14(1) gill rakers on lower limb.
Scales cycloid. Lateral line complete, slightly curved with 34(12), 35*(22), or 36(8) pored scales. One specimen with interrupted lateral line. Longitudinal scale rows above lateral line 5*(43) or 6(1). Longitudinal scale rows below lateral line 4*(44). Circumpeduncular scale rows 13(10), 14*(30) or 15(1). Single row of scales overlaying basal portion of anterior anal-fin rays. Small scales covering proximal one-third of caudal-fin lobes.
Dorsal-fin rays ii,9*(43). Pectoral-fin rays i,10(23), 11*(9), 12(2), or i,10,i(8) with their tips surpassing pelvic-fin origin. Pelvic-fin rays i,6(2) or 7*(40), with their tips surpassing anal-fin rays only in males. Anal fin slightly falcate, last unbranched, and four first branched rays longest. Anal-fin rays iii,18(1), 19(9), 20*(24), 21(7) or 22(1). Caudal fin forked, lobes of similar size. Caudal-fin rays i,9,8,i. Dorsal procurrent caudal-fin rays 10(2). Ventral procurrent caudal-fins rays 8(2). Total vertebrae 33(2). Supraneurals 4(2).
Coloration in alcohol. Overall body coloration yellowish. Dorsal portions of head and body darkly pigmented. Lips and maxilla densely pigmented; infraorbitals, opercle, and preopercle light beige, with scattered melanophores. Dorsolateral portion of the body with scattered dark chromatophores concentrated on distal margin of scales, exhibiting a reticulated pattern on three first horizontal rows. Conspicuous midlateral dark stripe, extending from posterior margin of opercle to middle caudal-fin rays. Dark stripe narrow anteriorly, becoming wide from vertical line just anterior dorsal-fin origin region, with median portion darker than anterior and posterior ones. Melanophores scattered between midlateral dark stripe and second row of scales below lateral line horizontally. A thin black stripe on the base of anal fin. Pectoral, pelvic, and anal fins hyaline, with melanophores scattered. Anterior portion of caudal-fin rays with concentrated melanophores on lepidotrichia margins, more evident on medial rays, forming a dark stripe, and in the upper and lower unbranched rays. Posterior portion of caudal-fin rays entirely hyaline, without melanophores.
Coloration in life. Dorsal portion of head, lips and maxilla emerald green; first, second, and third infraorbitals silver; fifth and sixth infraorbitals gold. Gular region reddish. Laterodorsal portion of trunk emerald green, becoming reddish from dorsal-fin origin to caudal peduncle. Lateroventral portion of trunk from isthmus to pelvic-fin origin silver to gold became reddish from that point to caudal peduncle. Upper portion of eyes golden, ventral portion silver. Inconspicuous midlateral silver stripe becomes black from vertical line just last anal-fin rays to middle caudal-fin rays. A concentration of black pigments on the anterior margin of the caudal-fin rays. Pectoral fin yellow to red. Pelvic, dorsal, adipose, anal, and caudal fins red on base, orange in central, and white on their tips (Fig. 3A).
Sexual dimorphism. Pelvic-fin length in adult males is proportionally longer than in adult females (19.4–21.6 vs. 15.5–17.5% of SL), with their tips surpassing anal-fin rays. This result was corroborated by covariance analysis (Fig. 4), which presents a strong relation between pelvic-fin length and standard length influencing the sex with p-value >0.05. Bony hooks were not found on fin rays.
FIGURE 4| The relationship between standard length and pelvic-fin length in Moenkhausia aurora.
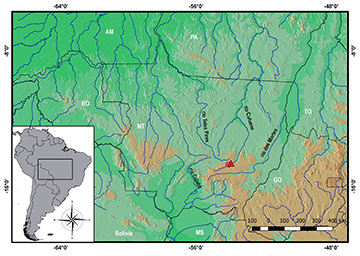
Geographical distribution. Moenkhausia aurora is known from the upper rio Culuene drainage, rio Xingu basin, municipality of Primavera do Leste, Mato Grosso State, Brazil (Fig. 5).
FIGURE 5| Map of central South America indicating the type locality (triangle) of Moenkhausia aurora.
Ecological notes. The type-locality of M. aurora is a tributary of the rio Culuene with about 10 m width, 1.5 m deep, and 470 m above sea level (Fig. 6). The stretch sampled presents riparian vegetation composed of trees and shrubs, fast and transparent water with the substrate formed by sand and submerged aquatic macrophytes. Moenkhausia aurora was collected syntopically with Hyphessobrycon loweae Costa & Géry, 1994, Hemigrammus sp., Rhinotocinclus acuen (Silva, Roxo & Oliveira, 2014), Knodus sp., and Leporinus multimaculatus Birindelli, Teixeira & Britski, 2016.
FIGURE 6| The type-locality of Moenkhausia aurora, rio Culuene, municipality of Primavera do Leste, Mato Grosso State, Brazil. Photo by Camila Souza.
Etymology. The specific epithet aurora comes from Latin, which means dawn or sunrise. In allusion to the red, orange, and gold colors present in specimens in life. A noun in apposition.
Conservation status. Moenkhausia aurora is known from two localities of the upper rio Culuene, rio Xingu basin, and its conservation status is uncertain based on the currently available geographic distribution. However, no imminent threats to the species were detected in the area of occurrence; we suggest that M. aurora should be classified as Least Concern (LC) according to the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN Standards and Petitions Subcommittee, 2022).
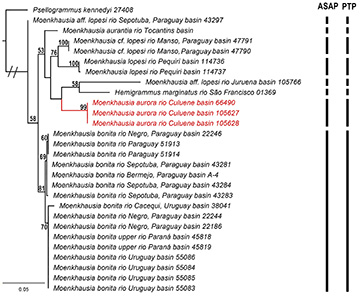
Genetics. The final matrix comprehended 600 pb with 155 variable sites. The composition of frequency of nucleotides obtained was A = 25.2%, C = 25.1%, G = 18.3%, and T = 31.4%. The values of Iss. were lower than Iss.c, indicating an absence of saturation in our matrix. The best evolution nucleotide model was GTR+G (General Time Reversible + Gamma) with an AICc value = 3888.273. However, the genetic distances analysis did not include this model, so we used the third best model TN93+G (Tamura-Nei + Gamma) model, with an AICc value = 3901.128. Genetic analyses supported Moenkhausia aurora as a distinct lineage. Overall, the mean genetic distance was 5%±0.01 without the outgroup. The genetic distance interspecific ranged from 2.2%±0.6 between M. bonita, and M. aff. lopesi rio Sepotuba basin to 10.2%±1.6 between M. aff. lopesi rio Juruena basin and M. lopesi rio Piquiri basin (Tab. 3). Moenkhausia aurora has 5.9%±0.010 of genetic distance from M. lopesi, the most similar species in terms of morphology. The Asap (ASAP score = 2.00, Fig. S1) and PTP methods discriminate the same eight lineages for the data and supported the identity of M. aurora (Figs. 7 and S2).
TABLE 3 | Genetic distances and S.D. of species analyzed in this study based on the TN93+G model. Intraspecific distances are marked in bold.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
1. M. aff. lopesi rio Juruena | – |
|
|
|
|
|
|
|
2. H. marginatus | 0.085±0.013 | – |
|
|
|
|
|
|
3. M. aurora | 0.092±0.014 | 0.07±0.012 | 0±0 |
|
|
|
|
|
4. M. aurantia | 0.086±0.014 | 0.071±0.012 | 0.058±0.011 | – |
|
|
|
|
5. M. aff. lopesi rio Sepotuba | 0.099±0.015 | 0.072±0.012 | 0.056±0.010 | 0.026±0.007 | – |
|
|
|
6. M. bonita | 0.094±0.015 | 0.073±0.012 | 0.060±0.010 | 0.030±0.007 | 0.022±0.006 | 0.004±0.001 |
|
|
7. M. cf. lopesi rio Manso | 0.099±0.015 | 0.069±0.012 | 0.054±0.010 | 0.037±0.008 | 0.041±0.009 | 0.038±0.008 | 0±0 |
|
8. M. lopesi rio Piquiri | 0.102±0.016 | 0.080±0.013 | 0.059±0.010 | 0.044±0.009 | 0.053±0.010 | 0.045±0.009 | 0.034±0.008 | 0.001±0.001 |
FIGURE 7| Maximum likelihood tree of species of the group Moenkhausia lopesi based on the COI gene (600pb). Bars represent the number of species obtained by the ASAP and PTP analyses. Bootstrap values above 50% are represented by numbers near the nodes.
Discussion
Moenkhausia aurora, the new species described here, is morphologically similar to M. lopesi in terms of general color pattern in alcohol-preserved specimens. However, these two species can be readily distinguished by the coloration of live specimens. Individuals of M. aurora have a reddish coloration (trunk and base of fins) versus yellowish in M. lopesi. (Figs. 3A,B) (see Diagnosis section). Britski, Silimon (2001) described color variations in some freshly collected specimens of M. lopesi, with dorsal fin varying from yellow (Fig. 3B) to orange and caudal fin varying from yellow (Fig. 3B) to orange or red (Bertaco et al., 2011:36, fig. 6b), but they did not report any specimen with dorsal, pelvic, anal or adipose fins red, or even, the trunk reddish, as occurs in M. aurora. The color variation observed in M. lopesi is most likely associated with sexual dimorphism since orange and red (carotenoid-based) reported in fins are two of the sets of pigments more common to expressed sexual dichromatism in Characiformes species (Pastana et al., 2017). In contrast, all collected specimens of M. aurora, including males and females, and small individuals showed a reddish color pattern.
Recent phylogenetic studies recovered a subclade of Moenkhausia composed of M. bonita, Hemigrammus marginatus Ellis, 1911, and Moenkhausia aff. lopesi (this paper, voucher 43297, Tab. 1) (Mariguela et al., 2013; Britzke et al., 2018). Moenkhausia aurora, M. lopesi, and M. aurantia are genetically close to the aforementioned species (Fig. 7) and probably also belong to this clade. Additionally to molecular data, this cluster of species, which include M. bonita, H. marginatus, M. aurora, M. lopesi, and M. aurantia, share a set of characters that can be putative synapomorphies for this group, such as (1) presence of a dark midlateral stripe, which extends from the posterior margin of the opercle (or from the vertical line just anterior to dorsal-fin origin, in M. aurantia) to middle caudal-fin rays, becoming wide from dorsal-fin origin; (2) dorsolateral portion of the body with concentrated chromatophores along distal margin of scales resulting in a reticulated pattern along the first three horizontal rows of scales; (3) absence of humeral blotch or inconspicuous in M. aurantia; (4) a thin black stripe on base of anal fin; (5) proximal portion of fins colorful (yellow, orange or red) and their distal portion usually white in life specimens.
Our molecular delimitation analysis was useful in discriminating eight mitochondrial lineages within this putative monophyletic group: Moenkhausia aurora, M. bonita, Hemigrammus marginatus, M. aurantia, M. lopesi, and more three other species that are probably new to science (revision in progress by the first author) morphologically similar to M. lopesi: Moenkhausia aff. lopesi from rio Juruena, Moenkhausia cf. lopesi from rio Manso, and Moenkhausia aff. lopesi from rio Sepotuba (Fig. 5). Additionally, our molecular results corroborated that M. bonita a species that occurs throughout Paraguay, Uruguay, and upper Paraná river basins (Vanegas-Ríos et al., 2019), is a unique widespread evolutionary lineage.
The overall mean of genetic distance found for this group is relatively low (mean~5%) when compared with values found in other monophyletic and genetically well-studied groups of Characidae, such as Tetragonopterus Cuvier, 1816 (mean~11%) (Melo et al., 2011; Silva et al., 2013), Moenkhausia oligolepis group (mean~19.0%) (Benine et al., 2009; Reia et al., 2021), and Priocharax Weitzman & Vari, 1987 (mean~22%) (Mattox et al., 2020, 2023). In other words, we can find evident morphological diagnostic characters even in lineages within a few DNA changes (e.g., 2.2% between M. bonita and Moenkhausia cf. lopesi). Certainly, intrinsic and/or extrinsic mechanisms are involved in the degree of morphological diversification in different rates for different clades of Characidae, constraining the phenotypic disparity (e.g., Moenkhausia oligolepis group) or promoting fast morphological diversification, as in the case of the group herein studied.
Comparative material examined. Brazil. Moenkhausia aurantia:rio Tocantins basin: LBP 18999, 4, 26.9–45.4. Moenkhausia bonita: rio Uruguay basin: ANSP 168836, 5, 25.2–32.5 mm SL. Moenkhausia celibela: rio Xingu basin: ANSP 194586, 6, 21.4–26.8 mm SL. ANSP 197467, 24, 15.5–31.1 mm SL. Moenkhausia lopesi: MZUSP 64480, 1, 36.5 mm SL, holotype; MZUSP 64481, 4 of 410, 29.6–34.9 mm SL, paratypes. Moenkhausia aff. lopesi: rio Sepotuba basin: LBP 8550, 1, 44.4 mm SL. Moenkhausia aff. lopesi: rio Juruena basin: LBP 30595. Hemigrammus marginatus: rio Itapecuru basin: ANSP 187197, 50, 14.4–31.6 mm SL; ANSP 187198, 49, 16.9–30.1 mm SL. Rio São Francisco basin: ANSP 171945, 29, 25.1–31.4 mm SL. Colombia. Moenkhausia intermedia: rio Amazonas basin: ANSP 135936, 28.5–31.8 mm SL. Guyana. Moenkhausia shideleri: rio Essequibo basin: ANSP 177002, 1, 64.6 mm SL. Paraguay. Moenkhausia bonita: rio Paraguay basin: ANSP 168836, 5, 25.2–32.3 mm SL; ANSP 175110, 5, 22.1–32.3 mm SL. Rio Paraná basin: ANSP 170335, 19, 23.4–31.7 mm SL. Moenkhausia dichroura: rio Paraguay basin: ANSP 169719, 51, 44.8–78.1 mm SL.
Acknowledgments
We are grateful to Francisco S. Neto for providing specimens of M. lopesi from rio Piquiri basin to molecular data. We are also grateful to Michel D. Gianeti, Alessio Datovo, and Mario C. C. de Pinna (MZUSP), Mariangeles Arce H. and Mark H. Sabaj (ANSP), Cristiano Moreira and Durval S. Santos (MNRJ), and José L. O. Birindelli (MZUEL) for curatorial assistance and loan of material. Eric V. Ywamoto and Heriberto G. Junior provided photos in life of Moenkhausia aurora and M. lopesi, respectively. James R. Garcia-Ayala provided the photos of the dentary, premaxilla, and maxilla of M. aurora. We are grateful to Camila S. Souza for providing the photo of the habitat of M. aurora and for financing the expedition to rio Culuene with the support of FAPESP grant# 2017/06551. We are grateful to William M. Ohara, Juan M. Mirande, and Francisco Langeani for their careful review of the article. Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq proc. #306054/2006–0 (CO) and CNPq proc. #312074/2022–0 (RCB) and Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP grant #2023/09902–9 (LR), FAPESP grant #2020/13433–6 (CO), and FAPESP grant #2021/12979–8 and #2022/13025–0 (GSCS).
References
Benine RC, Mariguela TC, Oliveira C. New species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) with comments on the Moenkhausia oligolepis species complex. Neotrop Ichthyol. 2009; 7(2):161–68. https://doi.org/10.1590/S1679-62252009000200005
Bertaco VA, Jerep FC, Carvalho FR. A new characid fish, Moenkhausia aurantia (Ostariophysi: Characiformes: Characidae), from the upper rio Tocantins basin in Central Brazil. Zootaxa. 2011; 2934(1):29–38. https://doi.org/10.11646/zootaxa.2934.1.3
Britski HA, Silimon KZ. Descrição de uma nova espécie de Moenkhausia da bacia do rio Paraguai (Teleostei: Ostariophysi: Characiformes). Comun Mus Ciênc Tecnol PUCRS, Sér Zool. 2001; 14(2):111–20.
Britzke R, Troy WP, Oliveira C, Benine RC. Description of a new species of Moenkhausia (Characiformes: Characidae) from the upper Paraguay basin, Central Brazil, with comments on its phylogenetic relationships. Neotrop Ichthyol. 2018; 16(2):e170086. https://doi.org/10.1590/1982-0224-20170086
Dagosta FCP, de Pinna M. The fishes of the Amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 2019(431):1–63. https://doi.org/10.1206/0003-0090.431.1.1
Edgar RC. Muscle: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics. 2004; 5:113. https://doi.org/10.1186/1471-2105-5-113
Eigenmann CH. New genera of South American fresh-water fishes and new names for some old genera. Smith Misc Collect. 1903; 45:144–48.
Eigenmann CH. The American Characidae – I. Mem Mus Comp Zool. 1917; 43:1–102.
Fink WL, Weitzman SH. The so-called Cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Smithson Contrib Zool. 1974; 172:1– 46. http://dx.doi.org/10.5479/si.00810282.172
Fox J, Weisberg S. An R Companion to applied regression. Sage, Thousand Oaks CA, 3rd edition, 2019. Available from: http://z.umn.edu/carbook
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: Genera, species, references [Internet]. San Francisco: California Academy of Sciences; 2023. Available from: https://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
International Union for Conservation of Nature (IUCN). Standards and Petitions Committee. Guidelines for Using the IUCN Red List Categories and Criteria. Version 15.1 [Internet]. 2022. Available from: https://www.iucnredlist.org/documents/RedListGuidelines.pdf
Ivanova NV, Dewaard JR, Hebert PDN. An inexpensive, automation-friendly protocol for recovering high-quality DNA. Mol Ecol Notes. 2006; 6(4):998–1002. https://doi.org/10.1111/j.1471-8286.2006.01428.x
Jennings WB, Ruschi PA, Ferraro G, Quijada CC, Silva-Malanski ACG, Prosdocimi F et al. Barcoding the Neotropical freshwater fish fauna using a new pair of universal COI primers with a discussion of primer dimers and M13 primer tails. Genome. 2019; 62(2):77– 83. https://doi.org/10.1139/gen-2018-0145
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al. Geneious basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics. 2012; 28(12):1647–49. https://doi.org/10.1093/bioinformatics/bts199
Lima FCT, Faria TC, Ohara WM, Barros SF. Revalidation and redescription of Moenkhausia madeirae Fowler from the rio Madeira basin in Brazil and Bolivia (Characiformes: Characidae). Proc Acad Nat Sci Phila. 2020; 167(1):147–58. https://doi.org/10.1635/053.167.0109
Marinho MMF, Dagosta FCP, Camelier P, Lima FCT. Description of a new species of Moenkhausia (Characiformes, Characidae) from the Upper Rio Tapajós Basin, Brazil. Copeia. 2016; 104(1):243–49. https://doi.org/10.1643/CI-14-216
Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA 16 evidence. J Zool Syst Evol Res. 2013; 51(4):327–32. https://doi.org/10.1111/jzs.12025
Mattox GMT, Britz R, Souza CS, Casas ALS, Lima FCT, Oliveira C. Two new species of miniature tetras of the fish genus Priocharax from the Rio Juruá drainage, Acre, Brazil (Teleostei: Characiformes: Characidae). Can J Zool. 2023; 101(4):248–66. https://doi.org/10.1139/cjz-2022-0136
Mattox GMT, Souza CS, Toledo-Piza M, Britz R, Oliveira C. A new miniature species of Priocharax (Teleostei: Characiformes: Characidae) from the Rio Madeira drainage, Brazil, with comments on the adipose fin in characiforms. Vertebr Zool. 2020; 70(3):417–33. https://doi.org/10.26049/VZ70-3-2020-11
Melo BF, Benine RC, Mariguela TC, Oliveira C. A new species of Tetragonopterus Cuvier, 1816 (Characiformes: Characidae: Tetragonopterinae) from the rio Jari, Amapá, northern Brazil. Neotrop Ichthyol. 2011; 9(1):49–56. https://doi.org/10.1590/S1679-62252011000100002
Menezes NA, Weitzman SH. Two new species of Mimagoniates (Teleostei: Characidae: Glandulocaudinae), their phylogeny and biogeography and a key to the glandulocaudin fishes of Brazil and Paraguay. Proc Biol Soc Wash. 1990; 103(2):380–426.
Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes): from characters to taxonomy. Neotrop Ichthyol. 2010; 8(3):385–68. https://doi.org/10.1590/S1679-62252010000300001
Mirande JM. Morphology, molecules and the phylogeny of Characidae (Teleostei, Characiformes). Cladistics. 2019; 35(3):282–300. https://doi.org/10.1111/cla.12345
Pastana MNL, Dagosta FCP, Esguícero ALH. A new sexually dichromatic miniature Hyphessobrycon (Teleostei: Characiformes: Characidae) from the Rio Formiga, upper Rio Juruena basin, Mato Grosso, Brazil, with a review of sexual dichromatism in Characiformes. J Fish Biol. 2017; 91(5):1301–18. https://doi.org/10.1111/jfb.13449
Pattengale ND, Alipour M, Bininda-Emonds ORP, Moret BME, Stamatakis A. How many bootstrap replicates are necessary? J Comput Biol. 2010; 17(3):337–54. https://doi.org/10.1089/cmb.2009.0179
Reia L, Oliveira C, Benine RC. Moenkhausia andrica (Characiformes: Characidae): a new species from the rio Tapajos basin, Brazil, with minute fin hooklets in females. J Fish Biol. 2021; 99(4):1380–92. https://doi.org/10.1111/jfb.14847
RStudio Team. Integrated Development Environment for R. Posit Software, PBC, Boston, MA. 2020. Available from: http://www.rstudio.com/
Sabaj MH. Codes for natural history collections in ichthyology and herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
Silva GSC, Melo BF, Oliveira C, Benine RC. Morphological and molecular evidence for two new species of Tetragonopterus (Characiformes: Characidae) from central Brazil. J Fish Biol. 2013; 82(5):1613–31. https://doi.org/10.1111/jfb.12094
Stamatakis A. RaxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. J Bioinform. 2014; 30(9):1312–13. https://doi.org/10.1093/bioinformatics/btu033
Tamura K, Stecher G, Kumar S. MEGA11: Molecular Evolutionary Genetics Analysis version 11. Mol Biol Evol. 2021; 38(7):3022–27. https://doi.org/10.1093/molbev/msab120
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Vanegas-Rios JA, Britzke R, Mirande JM. Geographic variation of Moenkhausia bonita (Characiformes: Characidae) in the rio de la Plata basin, with distributional comments on M. intermedia. Neotrop Ichthyol. 2019; 17(1):e170123. https://doi.org/10.1590/1982-0224-20170123
Vazzoler AEAM. Biologia da reprodução de peixes Teleósteos: teoria e prática. Maringá, EDUEM; 1996.
Xia X, Xie Z, Salemi M, Chen L, Wang Y. An index of substitution saturation and its application. Mol Phylogenet Evol. 2003; 26(1):1–07. https://doi.org/10.1016/S1055-7903(02)00326-3
Xia X, Lemey P. Assessing substitution saturation with DAMBE. In: Lemey P, Salemi M, Vandamme AM, editors. The phylogenetic handbook. A practical approach to phylogenetic analysis and hypothesis testing. Cambridge: Cambridge University Press; 2009. p.615–30.
Xia X. DAMBE7: New and improved tools for data analysis in molecular biology and evolution. Mol Biol Evol. 2018; 35(6):1550–52. https://doi.org/10.1093/molbev/msy073
Ward RD, Zemlak TS, Innes BH, Last RP, Hebert PDN. DNA barcoding Australia’s fish species. Phil Trans R Soc B. 2005; 360(1462):1847–57. https://doi.org/10.1098/rstb.2005.1716
Authors
![]() Lais Reia1
Lais Reia1 ![]() ,
, ![]() Gabriel de Souza Costa e Silva1,2,
Gabriel de Souza Costa e Silva1,2, ![]() Claudio Oliveira1 and
Claudio Oliveira1 and ![]() Ricardo Cardoso Benine3
Ricardo Cardoso Benine3
[1] Laboratório de Biologia e Genética de Peixes, Departamento de Biologia Estrutural e Funcional, Instituto de Biociências de Botucatu, Universidade Estadual Paulista, R. Prof. Dr. Antônio C. W. Zanin, s/n, Rubião Jr., 18618-689 Botucatu, SP, Brazil. (LR) laisreia@yahoo.com.br (corresponding author), (GSCS) gabriel_biota@hotmail.com, (CO) claudio.oliveira@unesp.br.
[2] Department of Ichthyology, The Academy of Natural Sciences of Drexel University, 19103-1195, Philadelphia, PA, USA.
[3] Laboratório de Ictiologia, Departamento de Biodiversidade e Bioestatística, Instituto de Biociências de Botucatu, Universidade Estadual Paulista, R. Prof. Dr. Antônio C. W. Zanin, s/n, Rubião Jr., 18618-689 Botucatu, SP, Brazil. (RCB) ricardo.benine@unesp.br.
Authors’ Contribution 

Lais Reia: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Writing-original draft, Writing-review and editing.
Gabriel de Sousa Costa e Silva: Conceptualization, Investigation, Writing-original draft, Writing-review and editing.
Claudio Oliveira: Data curation, Funding acquisition, Methodology, Resources, Writing-review and editing.
Ricardo C. Benine: Data curation, Formal analysis, Investigation, Methodology, Validation, Writing-review and editing.
Ethical Statement
Collecting permits were issued by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio 13843–5). The use of animals in this research was conducted under Brazilian animal welfare laws, and the research was approved by the Ethics Committee on Animal Use of the Instituto de Biociências, UNESP (protocol number 1058/2020).
Competing Interests
The author declares no competing interests.
How to cite this article
Reia L, Silva GSC, Oliveira C, Benine RC. New reddish species of Moenkhausia Eigenmann (Characiformes: Characidae) from the upper rio Xingu basin, Brazil. Neotrop Ichthyol. 2024; 22(1):e230089. https://doi.org/10.1590/1982-0224-2023-0089
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2024 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 2, 2023 by Paulo Lucinda
Accepted December 2, 2023 by Paulo Lucinda
![]() Submitted August 2, 2023
Submitted August 2, 2023
![]() Epub February 12, 2024
Epub February 12, 2024