![]() Mateus S. Chaves1,
Mateus S. Chaves1, ![]() Renildo R. de Oliveira2,
Renildo R. de Oliveira2, ![]() Alany P. Gonçalves1,3,
Alany P. Gonçalves1,3, ![]() Leandro M. Sousa4 and
Leandro M. Sousa4 and ![]() Lúcia H. Rapp Py-Daniel2
Lúcia H. Rapp Py-Daniel2 ![]()
PDF: EN XML: EN | Cite this article
Abstract
A new species of Scobinancistrus from the Xingu River, Brazil, is described. It can be distinguished from its congeners by color pattern and a combination of non-exclusive characters: overall body covered by large yellow spaced blotches over a dark background (vs. small round and densely packed spots over light or dark background in S. pariolispos and S. aureatus); lack of orange to yellow/orange distal band on dorsal and caudal fins (vs. presence in S. aureatus), dorsal fin not reaching adipose-fin supporting plate when adpressed (vs. reaching the adipose-fin plate in S. pariolispos and S. aureatus). The new species is only known from a portion of the middle Xingu River, ranging from the Volta Grande do Xingu, an area under a strong anthropic impact due to the construction of the Belo Monte dam, to near the Iriri River confluence with the Xingu River. Aspects concerning the species’ threats and its conservation status are discussed.
Keywords: Ancistrini, Brazilian shield, L082, Rapids, Rheophilic species.
Uma nova espécie de Scobinancistrus do rio Xingu, Brasil, é descrita. Ela pode ser distinguida de suas congêneres pelo padrão de cores e uma combinação de caracteres não exclusivos: corpo geral coberto por grandes manchas amarelas espaçadas sobre um fundo escuro (vs. pequenos pontos redondos e densamente compactados sobre fundo claro ou escuro em S. pariolispos e S. aureatus); falta de banda distal laranja a amarela nas nadadeiras dorsal e caudal (vs. presença em S. aureatus), nadadeira dorsal não alcançando a placa da nadadeira adiposa quando comprimida (vs. atingindo a placa da nadadeira adiposa em S. pariolispos e S. aureatus). A nova espécie só é conhecida de uma porção do médio rio Xingu, que vai da Volta Grande do Xingu, área de forte impacto antrópico devido à construção da barragem de Belo Monte, até próximo à confluência do rio Iriri com o rio Xingu. Aspectos relativos às ameaças à espécie e seu estado de conservação são discutidos.
Palavras-chave: Ancistrini, Corredeiras, Escudo Brasileiro, L082, Peixes reofílicos.
Introduction
Endemic and widely distributed throughout the Neotropical region, Loricariidae is the most prominent family of Siluriformes and the fifth largest among vertebrates, with currently 114 genera and more than 1,000 valid species, and still several to be described (Ota et al., 2015; Reis et al., 2016; Fricke et al., 2023). Currently, Loricariidae is subdivided into six subfamilies, and Hypostominae is the most diverse and species-rich (Armbruster, 2004; Reis et al., 2006; Lujan et al., 2015). Among Hypostominae, the tribe Ancistrini is the most diverse, with approximately 265 valid species (Ferraris, 2007; Fisch-Muller et al., 2012).
The Ancistrini Scobinancistrus was proposed by Isbrücker, Nijssen (1989) and diagnosed by the presence of up to five teeth on the dentary and premaxillary; dentary teeth twice larger than premaxillary; teeth strong, robust, bicuspid and spatulated, with a vertical division between cusps; internal cusp significantly larger than external cusp (Isbrücker, Nijssen, 1989; Burgess, 1994). Until now, the genus had two valid species: S. pariolispos Isbrücker & Nijssen, 1989, type species of the genus distributed on Tapajós, Xingu, and Tocantins/Araguaia rivers, and S. aureatus Burgess, 1994, with distribution restricted to the Xingu River basin (Reis et al., 2003).
Besides its worldwide attractiveness for novelties and economic importance, the ornamental aquarium trade is a potent source of potential taxonomic novelties (Tencatt, Britto, 2016; Tencatt, Ohara, 2016). The species of Scobinancistrus are very popular in the aquarium trade due to its beautiful color pattern, with bright yellow, almost golden spots over a dark background (Gonçalves et al., 2009; Camargo et al., 2012). Such as other loricariid genera from the Xingu River, Scobinancistrus species are exported to the US, Asia, and Europe, reaching an incredibly high final price (Rapp Py-Daniel et al., 2011; Ramos et al., 2015; Araújo et al., 2017). Many of these exported loricariids are unknown to science or show a remarkable variation in color pattern, making their identification difficult or misleading. In the absence of formal names, aquarists developed a morphotype-coded identification system for loricariids known as L-Numbers, published initially in the DATZ Magazine (Dignall, 2014).
The Xingu River is one of the largest tributaries of the Amazonas River, originating in Central Brazil and resulting from the confluence of several smaller rivers such as Culuene, Ferro, Jatobá, and Suiá Miçu (Soares, 1953). The Xingu River is characterized by its clear and high-energy waters (Goulding et al., 2003; Junk et al., 2011). Since 2013, the middle Xingu River has been receiving a strong anthropic impact with the construction of the Belo Monte hydroelectric dam (Fearnside, 2015). The construction and the activation of the power station required the construction of a reservoir and the deviation of 80% of the water that used to flow to the Volta Grande, an area characterized by a complex set of strong river deflections and cataracts (Sabaj Peréz, 2015; Sawakuchi et al., 2015).
Recent expeditions in the Xingu River to the areas impacted by the Belo Monte dam provided several specimens of an undescribed species of Scobinancistrus, known since the early 80s and popularly named “tubarão” (shark) by locals, an allusion to its round and depressed body. This species is known under the DATZ code as L082. In the present paper, we confirm that this is a new taxon as part of a significant revision of the genus prepared by the first author (MSC). Herein we describe the new taxon, comment on its distribution, and discuss its conservation status.
Material and methods
Morphological measurements were taken as point-to-point linear distances with a digital caliper to the nearest 0.1 mm following Boeseman (1968), with the addition of cleithrum length, cleithrum process width (at the insertion of the cleithrum), eye-nare length, lower lip width, lower lip length, length of barbel, adipose-caudal fin distance (measured from the posterior tip of the adipose membrane to the anterior portion of the first procurrent ray), internare distance; interbranchial distance; distance from pectoral fin insertion to branchial opening. Standard length (SL) is expressed in millimeters, and all other measurements are expressed as percentages of standard length or head length (HL). Measurements were recorded exclusively in specimens above 60.0 mm SL; descriptive statistics (mean, mode) included measurements of the holotype. Specimens were preserved in 75% alcohol (alc.). Additional osteological observations were made from skeletons prepared from formalin-fixed specimens according to Bemis et al. (2004) and are quoted as “skel.” in the list of examined material. Vertebral counts include Weberian (five) and ural complexes (one), following Lundberg, Baskin (1969). The nomenclature of the dermal plates on the lateral series follows Schaefer (1997) and Oyakawa et al. (2005); osteological nomenclature follows Arratia, Huaquin (1995). Institutional acronyms: ANSP, Academy of Natural Sciences of Philadelphia, Philadelphia; INPA, Instituto Nacional de Pesquisas da Amazônia, Manaus; LIA, Laboratório de Ictiologia de Altamira, Altamira; and MZUSP, Museu de Zoologia da Universidade de São Paulo, São Paulo.
Results
Scobinancistrus raonii, new species
urn:lsid:zoobank.org:act:A7052468-F11E-4AE8-BFFD-C2658EA8F86A
(Figs. 1–3; Tab. 1)
L082 Ancistrinae gen. sp. —Schraml, Schäfer, 2004:79–80 (Aquarium fish atlas; citation and brief description).
Ancistrini sp. (L 82). —Seidel, Evers, 2005:936–38 (Wels atlas; citation and brief description).
L082. —Seidel, 2008:198 (citation and brief description; figs. 1–3). —Camargo et al., 2012:113–18 (Book).
Scobinancistrus sp. 2 “Arábia ou Tubarão”. —Gonçalves et al., 2009:239 (Book chapter). —Camargo et al., 2012:183 (Book chapter).
Gênero novo, “Arábia”, L082. —Camargo et al., 2012:167–68 (Book, ornamental fish).
Scobinancistrus sp. L82. —Ramos et al., 2015:96 (Article).
Scobinancistrus n. sp. Xingu L082. —Lujan et al., 2015:282 (Article). —Lujan et al., 2017:330 (Article).
FIGURE 1| Lateral, dorsal and ventral views of Scobinancistrus raonii, INPA 59493, holotype, 99.6 mm SL, Senador José Porfírio, Xingu River, ca. 3.5 km downstream from BR-230. Photo: Leandro M. Sousa.
FIGURE 2| Lateral view of live specimens of Scobinancistrus raonii, Brazil, Pará, Xingu River, Volta Grande do Xingu. Paratypes: INPA 40512, 80.9 mm SL, juvenile (above); LIA 5054, 94.4 mm SL, subadult (middle); ANSP 194989, 120.8 mm SL, adult (below). Photos: Leandro M. Sousa (juvenile and subadult) and Mark H. Sabaj (adult).
Holotype. INPA 59493, 99.6 mm SL, Brazil, Pará, Senador José Porfírio, Xingu River, about 3.5 km downstream from BR-230, 03°06’18.90”S 51°43’28.30”W, 19 Nov 2014, M. H. Sabaj, L. M. Sousa & A. P. Gonçalves.
Paratypes. All from Brazil, Pará, Xingu River. ANSP 193045, 1, 75.3 mm SL, 50 km southeast from Altamira, 03°39’19.4”S 52°23’30.1”W, 11 Oct 2012, M. H. Sabaj, M. Arce & L. M. Sousa. ANSP 194819, 1, 109.3 mm SL, low portion of the main channel going from northwest to southwest, 43 km from Altamira, 03°34’05.0”S 52°20’46.1”W, 11 Sep 2013, M. H. Sabaj, L. M. Sousa, A. Gonçalves, N. K. Lujan, D. B. Fitzgerald, P. Madoka Ito, A. Oliveira & R. Robles. ANSP 194989, 1, 121.5 mm SL, middle of Volta Grande, area of rocks in the main channel of the largest branch of the river between the mouths of the rio Bacajaí and the rio Bacajá, 03°31’55.5”S 51°45’41.3”W, 17 Sep 2013, M. H. Sabaj, L. M. Sousa, A. Gonçalves, N. K. Lujan, D. B. Fitzgerald, P. Madoka Ito, A. Oliveira & R. Robles. ANSP 200828, 1, 69.7 mm SL, Gorgulho da Rita, conglomerate rocks exposed on the main channel arm, 15 km south of Altamira, 03°20’32.1”S 52°11’09.8”W, 25 Sep 2015, M. H. Sabaj, M. Arce, L. M. Sousa, M. Kalacska, J. P. Arroyo & O. Lucanus. INPA 31426, 1, 75.2 mm SL, Landi waterfall, 03°35’01”S 51°49’21”W, 8 Nov 2008, L. Rapp Py-Daniel, R. R. de Oliveira, J. Bessa & H. Anatole. INPA 31459, 1, 55.1 mm SL, Itaobinha, Pedral, 02°53’22”S 51°56’26”W, 4 Nov 2008, L. Rapp Py-Daniel, R. R. de Oliveira, J. Bessa & H. Anatole. INPA 31787, 1, 91.6 mm SL, Merence Island (Pontão), 03°06’18.0”S 51°43’33.6”W, 5 Nov 2008, L. Rapp Py-Daniel, R. R. de Oliveira, J. Bessa & H. Anatole. INPA 31789, 3 (1 skel., 78.4 mm SL, 2 alc.), 74.0–120.2 mm SL, Babaquara, Gorgulho da Rita, 03°22’22.3”S 52°11’51.0”W, 7 Nov 2008, L. Rapp Py-Daniel, R. R. de Oliveira, J. Bessa & H. Anatole. INPA 40117, 2, 60.2–67.8 mm SL, low portion of the main channel, 43 km southwest of Altamira, 03°34’04”S 52°20’45”W, 11 Sep 2013, M. H. Sabaj, L. M. Sousa, A. Gonçalves, N. K. Lujan, D. B. Fitzgerald, P. Madoka Ito, A. Oliveira, R. Robles & fishermen. INPA 40383, 1, 125.1 mm SL, a central area of the Volta Grande, on the right margin of the main arm of the Xingu, above of mouth of the Bacajá River, 03°31’55”S 51°45’41”W, 17 Sep 2013, M. H. Sabaj, L. M. Sousa, A. Gonçalves, N. K. Lujan, D. B. Fitzgerald, P. Madoka Ito, A. Oliveira, R. Robles & fishermen. INPA 40512, 1, 80.9 mm SL, deep channel, 38 km southeast of Vitória do Xingu, 03°05’32”S 51°44’14”W, 21 Sep 2013, M. H. Sabaj, L. M. Sousa, A. Gonçalves, N. K. Lujan, D. B. Fitzgerald, P. Madoka Ito, A. Oliveira & R. Robles. INPA 40766, 1, 102.3 mm SL, downstream Volta Grande, narrow region of the main channel, 03°11’03”S 51°37’02”W, 28 Sep 2013, M. H. Sabaj, L. M. Sousa, A. Gonçalves, N. K. Lujan, D. B. Fitzgerald, P. Madoka Ito, A. Oliveira & R. Robles. INPA 47255, 1, 72.0 mm SL, channel Gorgulho da Rita, ca. 15 km from Altamira, 03°20’32”S 52°11’10”W, 5 Nov 2014, M. H. Sabaj, L. M. Sousa, R. R. de Oliveira, A. Gonçalves, D. Fitzgerald, V. Machado & P. Madoka Ito. INPA 52347, 1, 120.9 mm SL, Santo Antônio, 03°06’0.7”S 51°46’25.3”W, 5 Sep 2012, E. D. Ribeiro & L. Sousa. INPA 58778, 1, 120.4 mm SL, near Paquiçamba Indigenous Land, at the island, 03°23’42.4”S 51°43’35.6”W, 20 Oct 2013, A. Gonçalves & P. Trindade. INPA 58779, 2, 92.9–129.7 mm SL, Pimental cofferdam, 03°26’22.4”S 51°57’27.9”W, Oct 2010, A. Gonçalves & P. Trindade. LIA 5054, 1, 94.4 mm SL, rio Xingu, right area Gorgulho da Rita, ca. 15 km south-southeast of Altamira, 03°20’51.4”S 52°10’56.1”W, 5 Nov 2014, M. H. Sabaj, L. M. Sousa & A. P. Gonçalves. LIA 6912, 1, 88.4 mm SL, Espelho Waterfall, 03°39’05.2”S 52°22’42.6”W, 30 Sep 2017, L. Sousa. MZUSP 107209, 5, 100.9–124.9 mm SL, below Volta Grande do Xingu, Pontão, Belo Monte, 03°06’49.0”S 51°43’23.0”W, 12 Jul 2010, Equipe ECIX.
Non-types. Brazil, Pará, Xingu River.INPA 54819, 11, 71.7–166.2 mm SL, Altamira, uncertain location.
Diagnosis. Scobinancistrus raonii differs from its congeners by presenting a deeper caudal peduncle, 11.7–13.5% in SL (vs. 9.8–11.7% in S. aureatus and 9.9–11.4% in S. pariolispos); by having large yellow irregularly shaped and widely spaced spots over entire body, spots larger than pupil diameter (vs. spots smaller than pupil diameter and densely packed); by presenting last dorsal-fin ray not reaching adipose-fin plate when adpressed in specimens larger than 80 mm SL (vs. last dorsal-fin ray reaching adipose-fin plate when adpressed in young and adults); by lacking contact between hyomandibular and quadrate posteroventrally (vs. contact present). Additionally, S. raonii differs from S. aureatus by lacking a yellow large distal band on the dorsal and caudal fins (vs. present).
Description. Morphometric and meristic data in Tab. 1. Thirty-eight specimens examined and measured. Small to medium-sized Scobinancistrus, largest specimen measured reached 166.2 mm SL (INPA 54819). Body bulky, ellipsoid and rounded in cross section; evenly deep post-cranially. Snout triangular to hexagonal in dorsal view; head and snout elongate, slightly depressed. Lateral view with subtle elevation from snout tip to orbit; slight elevation from this point to dorsal-fin origin, declining from this point to dorsal procurrent caudal-fin ray; complete dorsal profile almost straight or slightly convex. Body wider in cleithrum, deeper at dorsal-fin origin. Ventral surface flat, straight from snout to caudal fin.
TABLE 1 | Morphometric data and counts of Scobinancistrus raonii. H = holotype, SD = standard deviation, N = number of measured specimens.
N | H | Range | Mean | SD | |
Standard length (mm) | 38 | 99.6 | 67.8–166.2 | – | – |
Percentage of standard length | |||||
Predorsal length | 38 | 36.3 | 34.2–40.3 | 37.3 | 1.2 |
Head length | 38 | 31.1 | 28.4–33.2 | 30.9 | 1.2 |
Cleithral width | 38 | 26.3 | 25.3–28.0 | 26.5 | 0.6 |
Cleithral process
width | 33 | 24.8 | 23.6–26.1 | 24.1 | 0.6 |
Cleithral length | 34 | 8.7 | 6.8–10.2 | 8.9 | 0.6 |
Pectoral-pelvic origin
length | 38 | 24.7 | 17.6–26.1 | 22.9 | 1.5 |
Pectoral-spine length | 38 | 30.5 | 24.9–30.6 | 27.8 | 1.7 |
Pelvic-anal origin
length | 38 | 23.7 | 21.2–25.4 | 23.3 | 1.0 |
Pelvic-spine length | 38 | 25.5 | 21.5–27.5 | 24.4 | 1.4 |
Postanal length | 38 | 30.8 | 28.8–33.7 | 31.1 | 1.2 |
Anal-fin spine
length | 38 | 12.3 | 8.5–15.8 | 13.2 | 1.4 |
Dorsal spine length | 38 | 26.9 | 20.5–29.0 | 25.0 | 2.1 |
Dorsal-fin base length | 38 | 27.4 | 26.4–31.6 | 28.4 | 1.1 |
Dorsal-adipose distance | 38 | 18.2 | 13.6–18.2 | 15.5 | 1.1 |
Caudal peduncle depth | 38 | 12.9 | 11.7–13.5 | 12.6 | 0.4 |
Adipose-spine length | 38 | 9.7 | 6.6–11.7 | 10.1 | 1.3 |
Adipose-caudal length | 38 | 7.8 | 5.2–23.3 | 9.6 | 4.8 |
Body depth at dorsal-fin origin | 38 | 18.1 | 13.5–20.2 | 16.9 | 1.8 |
Body width at dorsal-fin origin | 38 | 23.9 | 20.9–26.5 | 23.4 | 1.3 |
Body width at anal-fin origin | 38 | 18.8 | 15.1–19.3 | 17.2 | 0.9 |
Posdorsal length | 38 | 33.8 | 22.9–37.1 | 32.6 | 2.5 |
Anus-anal fin length | 38 | 5.5 | 4.6–7.9 | 6.2 | 0.7 |
Percentage of head length | |||||
Orbital diameter | 38 | 18.5 | 15.9–21.0 | 18.0 | 1.0 |
Snout length | 38 | 61.5 | 57.3–66.1 | 61.8 | 1.8 |
Internares width | 38 | 12.0 | 9.9–13.9 | 11.6 | 1.0 |
Interorbital width | 38 | 32.1 | 28.8–35.3 | 31.3 | 1.6 |
Head depth | 38 | 52.2 | 45.7–58.2 | 50.2 | 2.6 |
Dentary tooth
row length | 38 | 13.6 | 9.3–18.3 | 14.0 | 2.1 |
Premaxillary tooth row length | 38 | 14.6 | 7.7–16.5 | 13.3 | 2.0 |
Head width | 38 | 71.1 | 64.8–80.9 | 71.3 | 3.5 |
Eye-nare length | 38 | 16.5 | 12.1–18.4 | 15.6 | 1.6 |
Interbranchial distance | 38 | 47.5 | 41.7–53.3 | 47.3 | 2.6 |
Pectoral spine origin to gill-opening
distance | 38 | 24.6 | 18.9–28.2 | 23.7 | 2.5 |
Lower lip
width | 38 | 39.7 | 34.8–45.2 | 39.7 | 2.8 |
Lower lip
length | 38 | 17.2 | 8.6–19.8 | 14.9 | 2.2 |
Length barbel | 34 | 10.1 | 5.0–13.4 | 10.5 | 2.0 |
Orbit not elevated, eye dorsolaterally positioned. In lateral view, eye inserted on vertical through branchial opening. Interorbital area flattened. Inconspicuous odontodes on cheek plates on young individuals; in larger specimens, cheek odontodes weakly developed, not surpassing branchial opening. Opercle eversible, not exposed; supraopercle area covered by several small plates, some fused. Parieto-supraoccipital almost flat, indistinct from rest of skull bones. Parieto-supraoccipital process small but conspicuous, pointed posteriorly, not elevated, located between first pair of predorsal plates.
Predorsal area short with subtle elevation, with three pairs of plates: first pair of moderately-sized plates divided by parieto-supraoccipital process; second pair almost indistinct; third pair with large, rectangular spaced plates immediately anterior to nuchal plate. Nuchal plate small, anterior to dorsal spinelet.
Mouth and lips moderate in size; oral disc almost round, slightly rectangular. Lips densely covered by small round papilla (papilla larger medially); small papilla surrounding mandibles. Lower lip short, not reaching scapular girdle. Maxillary barbel short, thick at insertion, thin distally, not reaching beyond lower lip border.
Buccal teeth long, strong, and bicuspid. Teeth spatulate with asymmetric dental cusp, with vertical divide between cups (Fig. 3). Mesial cusp much larger than lateral cusp, in young and adults. Dentary cups set apart, almost parallel to each other; premaxillary cups small, closely attached; up to 4 teeth in premaxilla and dentary. Several oral papillae set immediately posterior to premaxilla and dentary organized as a block.
FIGURE 3| Photo in ventral view of the mouth of Scobinancistrus raonii,INPA 40512, paratype, 80.9 mm SL. Photo: Leandro M. Sousa.
Body smooth, almost without keels; light carena along dorsal-fin base until adipose fin. First mid-lateral plates swiftly bent, forming delicate keel. Ventral surface of caudal peduncle flattened, slightly round. Rest of body without keels or carena. Dorsally, head and trunk completely covered by large plates, except by naked area surrounding dorsal-fin base. Ventral surface utterly devoid of plates in juveniles (up to 70 mm SL) until urogenital opening. Larger specimens (70 to 110 mm SL) with small plates only on lateral border of abdomen, also dispersed on scapular girdle area. Specimens larger than 110 mm SL with small plates covering scapular girdle completely, between pelvics towards urogenital papilla, forming elongate and narrow abdominal plated area; remaining abdomen naked, unplated. Caudal peduncle deep, short, compressed, bulky and wholly plated. Twenty-seven median plates. Posterior plates strongly marked with lines of more developed odontodes on males. Five rows of plates on caudal peduncle. Odontodes short, poorly developed.
Dorsal-fin origin slightly anterior to vertical through pelvic-fin origin. Dorsal fin II,7; locking mechanism present, not functional. Dorsal-fin height moderate; last dorsal-fin ray not reaching adipose-fin supporting plate when adpressed. Dorsal fin without posterior hypertrophied membrane; five to eight plates between last dorsal-fin ray and adipose fin; inter-dorsal area flattened. Adipose fin small with posterior membrane weakly developed. Two pairs of dorsal plates between adipose fin and first dorsal procurrent caudal-fin ray. Caudal fin i,14,i, bilobed, slightly emarginate, with unbranched dorsal caudal-fin ray shorter than unbranched ventral caudal-fin ray. Pectoral fin I,6; pectoral spine short to moderate in size, reaching but not surpassing pelvic-fin base when adpressed. Pectoral spine robust, strong, covered by odontodes; odontodes larger and flexible distally. Pelvic fin i,5, reaching posteriorly vertical through adipose-fin base, not beyond. Anal fin short, i,4. All rays covered by minute odontodes on free surface. Vertebrae 31(1) to 32(1). Six pairs of ribs (2).
Color in alcohol. Similar to living specimens, with dark background, pale, whitish blotches. Ventral surface light to gray with opaque blotches; in some specimens, ventral surface clearer without any blotch, especially among juveniles.
Color in life. Whole body with dark background, green olive to black, presence of conspicuous yellow spaced spots or blotches (Fig. 2). Blotches can be round, sometimes merged to each other, forming large irregular patterns. Blotches all over head, body, and fins larger and more spaced in juveniles than in adults. During growth, blotches become more rounded and numerous. Ventral surface light brown with pale spaced blotches; sometimes inconspicuous.
Sex dimorphism. Mature males with hypertrophied odontodes on opercle surpassing posterior border of orbit, on distal portion of pectoral spine, and along caudal peduncle. Head of males relatively wider than in females. Mature females wider than males in abdominal area.
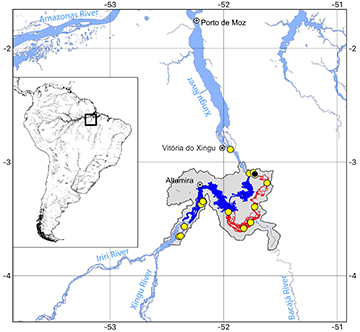
Geographical distribution. Scobinancistrus raonii is endemic to the Xingu River basin, restricted to the main channel, immediately downstream of its confluence with the Iriri River up to the Itaobinha region, upstream from Vitória do Xingu (Fig. 4).
FIGURE 4| Partial map of the Xingu River, showing the distribution of Scobinancistrus raonii (yellow circles). Black circle indicates type locality. Shaded area indicates the Impact Zone of the Belo Monte Dam Complex. Dark blue stretch of the river represents the dammed area. Red stretch indicates the reduced flow impacted area.
Ecological notes. Scobinancistrus raonii inhabits strong, high-energy waters along the main river channel, inhabiting a special microhabitat composed of conglomerate rock formed by gravel, sand, and iron oxide (called “mocororô” by local people). These conglomerates are usually flat rocky plates with spaces and caves carved by the current and can be stacked on the river bottom, giving the tridimensional complexity of this region (Fig. 5). We hypothesize that the yellow blotches along the fish’s body camouflage the fish among the incrusted gravel on the conglomerate. Scobinancistrus raonii is usually found at shallow depths, from 0.5 m to a maximum depth of 11 m. Scobinancistrus raonii is very likely a selective carnivore, such as its congeners, as it feeds on items with its well-developed spatulate teeth (Fig. 3). Analysis of food contents of the other species of Scobinancistrus showed the presence of insects, algae, microcrustaceans and dominance of Porifera (spicula) (Zuanon, 1999; Taís de Jesus, 2020, pers. comm).
FIGURE 5| Scobinancistrus raonii hiding underneath its preferred microhabitat: conglomerate plates of gravel and iron oxide. Photo: Leandro M. Sousa.
Etymology. The new species is named in honor of the Cacique Raoni Metuktire of the Kayapó people. Raoni is one of the most active indigenous leaders in the struggle to preserve the Amazon rainforest and indigenous peoples, being a solid reference for the fight for the conservation of the Xingu River basin for more than 40 years. A patronym.
Conservation status. Scobinancistrus raonii occurs in a limited distribution area, on a stretch of approximately 300 km of the middle Xingu River, Pará State. In an attempt to estimate area of occupancy (AOO – following IUCN, 2022 guidelines) loss, we used the method of 2 x 2 km grid cells to calculate the area between the most distant points in the Xingu River where Scobinancistrus raonii has been registered. The original area before Belo Monte and Pimental dam constructions comprised 1,028 km2. With the water flow reduction and exposition of rocks of the extensive running waters of the Volta Grande do Xingu (VGX) (ca. 460 km2) due to the deviation of the main channel of the Xingu River, we can estimate a loss of 45% of the original area. Plus, part of the Xingu River running waters below Altamira city was transformed into a large and deep reservoir by the Pimental dam (220 km2), causing an additional reduction of 21% of the original area of occupancy of S. raonii. As S. raonii was shown to have restricted habitat requirements (running waters, turbulence, rocky conglomerates), the main impact is the reduction of flow in the reservoir area, which leads to siltation and homogenization of the river bottom (Fig. 6). We are estimating a total loss of AOO of more than 60% of the original area. Approximately more than 95% of the area of occurrence of this species is included in the direct impact area of the Belo Monte Hydroelectric Power Plant, which reduced the natural range of the species due to the environmental degradation of the area. Thus, based in the decline of quality of habitat, area of occupancy (reduced to less than 500 km2), points of threat (two dams in the area of occurrence, Pimental and Belo Monte, could be considered as locations) and consequently reduction on the population, S. raonii could qualify for the Endangered (EN) category under the IUCN (2022) standards, criterion B2abii,iii, raising many concerns about conservancy of these high-energy water dependent loricariids.
FIGURE 6| Underwater photography at the exact same locality in Gorgulho da Rita. Above, October 2015, showing a rapids’ habitat with high tridimensional complexity occupied by several species of fish (on the picture: Teleocichla preta, Leporinus tigrinus, Hypomasticus julii). Below, October 2017, same spot after Belo Monte Dam implementation showing that the absence of current promoted siltation and the disappearance of rheophilic species from the area. Photos: Leandro M. Sousa
Discussion
Scobinancistrus raonii has the diagnostic characteristics to be included in the genus Scobinancistrus, such as 1) the presence of up to five teeth on the dentary and premaxillary; 2) dentary teeth two times larger than premaxillary; 3) teeth strong, robust, bicuspid and spatulated, with a vertical division between cusps and, 4) mesial cusp significantly larger than lateral one. In addition, it differs from Parancistrus Bleeker, 1862, Baryancistrus Rapp Py-Daniel, 1989, and Spectracanthicus Nijssen & Isbrücker, 1987 by having the dorsal fin separated from the adipose fin, without any extended membrane after the last dorsal fin ray; it differs from Hypancistrus Isbrücker & Nijssen, 1991 by having upper and lower jaws with more than four teeth; and it differs from the remaining ancistrins (sensu Armbruster, 2004) by the reduced number of mandibular teeth. Besides, the diagnostic features cited above reinforce the distinction of S. raonii from the other ancistrins.
The color pattern of S. raonii is also quite distinct from its congeners. Scobinancistrus aureatus and S. pariolispos show round, small, densely organized yellow spots all over the head, body, and fins, whereas S. raonii has round or irregularly shaped, large, and spaced yellow blotches along the head, body, and fins. Also, S. aureatus has a bright golden distal band on dorsal and caudal fins and a clearer and spotless ventral surface, which makes this species particularly distinctive from S. raonii and S. pariolispos. Additionally, S. raonii, besides being distinct from S. pariolispos by size and color pattern on the dorsal surface, does not show the abdominal vermiculation pattern found in populations of S. pariolispos.
Scobinancistrus raonii apparently does not reach more than 170 mm SL (largest specimen with 166.2 mm SL), whereas S. pariolispos and S. aureatus can easily reach above 200 mm SL (largest specimen of S. aureatus examined measured 245.5 mm SL and largest S. pariolispos, 290.0 mm SL). Overall body proportions and counts are very similar between S. pariolispos and S. aureatus and distinct from S. raonii. Scobinancistrus raonii has a more depressed head and body, smaller orbit diameter, deeper caudal peduncle, and shorter dorsal fin (last dorsal-fin ray not reaching adipose-fin plate when adpressed, in specimens above 80 mm SL).
Scobinancistrus raonii is apparently confined to the main channel of Xingu River, probably restricted to the fast-running waters, without any records from tributaries or upper reaches of the Xingu. So far, the furthest upstream record of S. raonii is close to the confluence of the Xingu/Iriri rivers, not occurring upstream of the confluence. Downstream, S. raonii can be found below Cachoeira de Belo Monte, in a small village called Santo Antônio. From this point towards the mouth, the Xingu River becomes a deeper river with lower-speed waters, limiting the presence of S. raonii. This geomorphologic discontinuity is almost certainly responsible for the turnover of species composition along the fluvial gradient in the Xingu (Fitzgerald et al., 2018). In addition to that, S. raonii seems to be dependent on the conglomerate rock, as the fish is only found in cracks of rocks composed of incrustation of gravel and iron dioxide (Sabaj Pérez, 2015; Sawakuchi et al., 2015; Freire et al., 2022). Bedrocks or conglomerates are naturally stacked in layers forming a myriad of crevices and caves, ideal for shelter or foraging of different species of invertebrates and fishes (Zuanon, 1999). Thus, all species of Scobinancistrus are found in the main channel, amongst rocks with moderate to strong currents. During high-waters, they concentrate in marginal areas with reduced deposit of sediments (Zuanon, 1999; Silva et al., 2016).
The Volta Grande do Xingu and the upstream area have been profoundly altered by the construction of the Belo Monte dam and by the deviation of the river to fill up a vast reservoir impounded by the Pimental dam. As this large stretch of bedrock of the VGX was exposed, we might expect a large impact on rock-dwellers-fish specialists such as Scobinancistrus raonii, with a substantial reduction in these fishes’ natural habitat (Winemiller et al., 2016). By the end of 2015, the river was dammed, affecting a stretch of nearly 170 km upstream that was transformed into the main reservoir (Pimental reservoir) below Altamira. The type-locality of S. raonii above Belo Monte Powerhouse (see map) is currently submerged. Thus, the occurrence area of the species was divided into two parts: the flooded area for the main reservoir above the Pimental dam and, downstream, the area with drastically reduced flow due to deviation of the water. The chances of survival and adaptation for a species that does not reach depths below 10 m, is highly dependent on oxygenated waters, and shows a sophisticated camouflage mechanism must be investigated. Rheophilic species are more sensitive to hydrologic changes due to their specializations developed for living in fast waters and their dependence on micro-habitats for survival (Lujan, Conway, 2015; Fitzgerald et al., 2018). Besides, the drastic change from a high-energy environment to a reservoir will undoubtedly cause the deposition of sediments on the rocky substrates (a perspective of a mix of sand, silt, and mud). These sediments were usually washed away by the natural flow of the water (Sabaj Pérez, 2015). Scobinancistrus raonii shows a limited range which raises the probability of negative impacts on its survival or adaptation. Ideally, periodic surveys should be conducted in Volta Grande do Xingu to follow up on the high-risk and long-term impacts on populations of S. raonii and other rheophilic fishes in an attempt to evaluate their chances of survival (Castello et al., 2013; Zarfl et al., 2015; Frederico et al., 2016; Azevedo-Santos et al., 2019).
Scobinancistrus species, like many loricariids in Xingu, are highly valued in the ornamental fish international market (Camargo et al., 2004, 2012) due to the attractive bright color pattern over a dark background. Scobinancistrus raonii is already known in the ornamental trade as L082 and is exported to several countries in North America, Europe, and Asia (Ramos et al., 2015; Araújo et al., 2017) and, more recently, to Colombia. However, due to pressure caused by overfishing, Scobinancistrus pariolispos and S. aureatus are classified as Vulnerable (VU) by the ICMBio (2018), the Brazilian agency responsible for the evaluation of the status of conservation of Brazilian species under the IUCN parameters. However, the commercialization of ornamental fish species of Scobinancistrus is currently legal. Thus, it is paramount to know and document the fish species at national and regional levels to increase our knowledge of the ichthyofauna and improve initiatives towards conservation.
Comparative material examined. All from Brazil. Scobinancistrus aureatus: Pará,Xingu River: MZUSP 47690, 205.0 mm SL, holotype. INPA 3453, 1, 196.2 mm SL. INPA 4035, 1, 200.1 mm SL. INPA 31471, 1, 55.6 mm SL. INPA 31791, 1, 91.8 mm SL. INPA 34657, 1, 195.7 mm SL. INPA 40118, 1, 109.4 mm SL. INPA 40153, 1, 113.9 mm SL. INPA 43245, 1, 41.6 mm SL. LIA 1470, 1, 76.2 mm SL. LIA 2432, 1, 39.2 mm SL. LIA 2461, 3, 45.3–51.2 mm SL. Scobinancistrus pariolispos: Pará,Tocantins River: INPA 10910, 117.5 mm SL, holotype. Pará, Araguaia River: INPA 58258, 1, 183.3 mm SL. Pará, Tapajós River: INPA 26485, 6, 53.4–175.3 mm SL. INPA 26491, 1, 56.9 mm SL. INPA 26506, 3, 24.2–48.9 mm SL. INPA 43761, 8, 45.2–78.9 mm SL. Mato Grosso, Tapajós River: MZUSP 116525, 1, 176.1 mm SL. MZUSP 34266, 3, 151.3–180.1 mm SL. Pará, Tocantins River: INPA 20747, 1, 210.6 mm SL. INPA 26673, 1, 84.5 mm SL. INPA 31878, 1, 239.9 mm SL. MZUSP 19282, 2, 145.2–238.1 mm SL. Pará, Xingu River: INPA 31790, 4, 53.5–117.1 mm SL. INPA 40119, 1, 68.5 mm SL. INPA 40173, 2, 51.1–67.3 mm SL. INPA 40405, 1, 58.4 mm SL. INPA 40448, 1, 43.8 mm SL.
Acknowledgments
We thank Rafaela P. Ota for suggestions and contribution to the manuscript. Thanks to Mark Sabaj (ANSP), Aléssio Datovo, Guilherme Dutra, Mario de Pinna and Osvaldo Oyakawa (MZUSP), Cárlison Silva-Oliveira and Frank Ribeiro (UFOPA) for curatorial assistance and/or the loan of specimens. Thanks to Dayane Silva (UFPA), Mark Sabaj (ANSP) and Paulo Trindade (UFPA) for sharing specimens and data on Scobinancistrus collected during their Xingu field studies. Fieldwork supported in part by Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (process 486376/2013–3 to LMS), the federal environmental licensing process associated with the Belo Monte hydroelectric complex (Process # 02001.001848/2006-75 to LMS) and the iXingu Project (NSF DEB–1257813). We also acknowledge the collaboration of the Xingu fishermen (Dani, Edson and others) in all aspects of the fieldwork. Their expertise was fundamental for the accomplishment of this project. This study was part of the master’s thesis of the first author (MSC), who is grateful to Alessandro Bifi, Cárlison Silva-Oliveira and Marcelo Rocha. MSC was supported by a fellowship grant from Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM).
References
Araújo JG, Santos MAS, Rebello KF, Isaac VJ. Cadeia comercial de peixes ornamentais do rio Xingu, Pará, Brasil. Bol Instit Pesca. 2017; 43(2):297–307. https://doi.org/10.20950/1678-2305.2017v43n2p297
Armbruster JW. Phylogenetic relationships of the suckermouth armoured catfishes (Loricariidae) with emphasis on the Hypostominae and the Ancistrinae. J Linn Soc Zool. 2004; 141(1):1–80. https://doi.org/10.1111/j.1096-3642.2004.00109.x
Arratia G, Huaquin L. Morphology of the lateral line system and of the skin of Diplomystid and certain primitive Loricarioid catfishes, and systematic and biological considerations. Bonn: Bonn Zool Monogr. 1995.
Azevedo-Santos VM, Frederico RG, Fagundes CK, Pompeu PS, Pelicice FM, Padial AA et al. Protected areas: A focus on Brazilian freshwater biodiversity. Diversity Distrib. 2019; 25(3):442–48. https://doi.org/10.1111/ddi.12871
Bemis WE, Hilton EJ, Brown B, Arrindell R, Richmond AM, Little CD et al. Methods for preparing dry, partially articulated skeletons of Osteichthyans, with notes on making ridewood dissections of the cranial skeleton. Copeia. 2004; 2004(3):603–09. http://www.jstor.org/stable/1448479
Boeseman M. The genus Hypostomus Lacépède, 1803, and its Surinam representatives (Siluriformes, Loricariidae). Zool Verh. 1968; 99:1–89.
Burgess WE. Scobinancistrus aureatus, a new species of Loricariid Catfish from the Rio Xingu (Loricariidae: Ancistrinae). TFH Magazine. 1994; 43(1):236–42.
Camargo M, Giarrizzo T, Isaac VJ. Review of the geographic distribution of fish fauna of the Xingu River basin, Brazil. Ecotropica. 2004; 10:123–47. Available from: https://mamiraua.org.br/documentos/682b5dbf3fd22e70ea0f8b16083a0bff.pdf
Camargo M, Gimênes-Junior H, Rapp Py-Daniel L. Acaris ornamentais do Médio Rio Xingu. Belém, FAPESPA/FUNCEFET. 2012.
Castello L, McGrath DG, Hess LL, Coe MT, Lefebvre PA, Petry P et al. The vulnerability of Amazon freshwater ecosystems. Conserv Lett. 2013; 6(4):217–29. https://doi.org/10.1111/conl.12008
Dignall J. List of original L-Numbers: Online reference. 2014. Available from: https://www.planetcatfish.com/shanesworld/shanesworld.php?article_id=514
Fearnside PM. Amazon dams and waterways: Brazil’s Tapajós basin plans. Ambio. 2015; 44(5):426–39. https://doi.org/10.1007/s13280-015-0642-z
Ferraris Jr. CJ. Checklist of catfishes, recent and fossil (Osteichthyes: Siluriformes), and catalogue of siluriform primary types. Zootaxa. 2007; 1418:1–628.
Fisch-Muller S, Montoya-Burgos JI, Le Bail PY, Covain R. Diversity of the Ancistrini (Siluriformes: Loricariidae) from the Guianas: the Panaque group, a molecular appraisal with descriptions of new species. Cybium. 2012; 36(1):163–93.
Fitzgerald DB, Sabaj-Pérez MH, Sousa LM, Gonçalves AP, Rapp Py-Daniel L, Lujan NK et al. Diversity and community structure of rapids-dwelling fishes of the Xingu River: Implications for conservation amid large-scale hydroelectric development. Biol Conserv. 2018; 222:104–12. https://doi.org/10.1016/j.biocon.2018.04.002
Frederico RG, Olden JD, Zuanon J. Climate change sensitivity of threatened, and largely unprotected, Amazonian fishes. Aquat Conserv Mar Freshw Ecosyst. 2016; 26(1):91–102. https://doi.org/10.1002/aqc.2658
Freire MP, Góes AM, Fairchild TR, Gautheron C, Parra M, Pupim FN et al. Quaternary ironstones in the Xingu River, eastern Amazonia (Brazil). Quat Res. 2022:1–14. https://doi.org/10.1017/qua.2022.15
Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: genera/species by family/subfamily [Internet]. San Francisco: California Academy of Sciences; 2023. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Goulding M, Barthem R, Ferreira EJG. The Smithsonian Atlas of the Amazon. Washington: Smithsonian Books; 2003.
Gonçalves AP, Camargo M, Carneiro CC, Camargo AT, Paula GJX, Giarrizzo T. A pesca de peixes ornamentais. In: Camargo M, Ghilardi R, editors. Entre a terra, as águas e os pescadores do médio rio Xingu – uma abordagem ecológica. Belém, Pará; 2009. p.233–64.
Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio). Livro Vermelho da Fauna Brasileira Ameaçada de Extinção: Volume VI – Peixes. Brasília: ICMBio/MMA; 2018. https://www.gov.br/icmbio/pt-br/centrais-de-conteudo/publicacoes/publicacoes-diversas/livro_vermelho_2018_vol6.pdf
Isbrücker IJH, Nijssen H. Diagnose dreier neuer Harnischwelsgattungen mit fünf neuen Arten aus Brasilien (Pisces, Siluriformes, Loricariidae). DATZ. 1989; 42(9):541–47. Available from: https://www.datz.de/images/stories/Erstbeschreibungen/Scobinancistrus%20pariolispos-1989-9-Teil1.pdf
International Union for Conservation of Nature (IUCN). Standards and petitions committee. Guidelines for using the IUCN Red List categories and criteria. Version 15.1 [Internet]. Gland; 2022. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Junk WJ, Piedade MTF, Schöngart J, Cohn-Haft M, Adeney JM, Wittmann F. A classification of major naturally-occurring Amazonian lowland wetlands. Wetlands. 2011; 31:623–40. https://doi.org/10.1007/s13157-011-0190-7
Lujan NK, Conway KW. Life in the fast lane: A review of rheophily in freshwater fishes. In: Riesch R, Tobler M, Plath M, editors. Extremophile fishes: Ecology, evolution, and physiology of Teleosts in extreme environments. Springer; 2015. p.107–36. https://doi.org/10.1007/978-3-319-13362-1_6
Lujan NK, Armbruster JW, Lovejoy NR, López-Fernández H. Multilocus molecular phylogeny of the suckermouth armored catfishes (Siluriformes: Loricariidae) with a focus on subfamily Hypostominae. Mol Phylogenet Evol. 2015; 82:269–88. https://doi.org/10.1016/j.ympev.2014.08.020
Lujan NK, Cramer CA, Covain R, Fisch-Muller S, López-Fernández H. Multilocus molecular phylogeny of the ornamental wood-eating catfishes (Siluriformes, Loricariidae, Panaqolus and Panaque) reveals undescribed diversity and parapatric clades. Mol Phylogenet Evol. 2017; 109:321–36. http://dx.doi.org/10.1016/j.ympev.2016.12.040
Lundberg JG, Baskin JN. The caudal skeleton of the catfishes, order Siluriformes. Am Mus Nov. 1969; 2398:1–49. Available from: http://hdl.handle.net/2246/2608
Ota RR, Message HJ, Graça WJ, Pavanelli CS. Neotropical Siluriformes as a model for insights on determining biodiversity of animal groups. PLoS ONE. 2015; 10(7):1–13. https://doi.org/10.1371/journal.pone.0132913
Oyakawa OT, Akama A, Zanata AM. Review of the genus Hypostomus Lacépède, 1803 from rio Ribeira de Iguape basin, with description of a new species (Pisces, Siluriformes, Loricariidae). Zootaxa. 2005; 92(1):1–27. https://doi.org/10.11646/zootaxa.921.1.1
Ramos FM, Araújo MLG, Prang G, Fujimoto RY. Ornamental fish of economic and biological importance to the Xingu River. Braz J Biol. 2015; 75(3):95–98. http://dx.doi.org/10.1590/1519-6984.02614BM
Rapp Py-Daniel LH, Zuanon JAS, Oliveira RR. Two new ornamental loricariid catfishes of Baryancistrus from rio Xingu drainage (Siluriformes: Hypostominae). Neotrop Ichthyol. 2011; 9(2):241–52. https://doi.org/10.1590/S1679-62252011000200001
Reis RE, Kullander SO, Ferraris CJ, Jr., editors. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003.
Reis RE, Pereira EHL, Armbruster JW. Delturinae, a new loricariid catfish subfamily (Teleostei, Siluriformes), with revisions of Delturus and Hemipsilichthys. J Linn Soc Zool. 2006; 147(2):277–99. https://doi.org/10.1111/j.1096-3642.2006.00229.x
Reis RE, Albert JS, Di Dario F, Mincarone, Petry P, Rocha LA. Fish biodiversity and conservation in South America. J Fish Biol. 2016; 89(1):12–47. https://doi.org/10.1111/jfb.13016
Sabaj Pérez M. Where the Xingu bends and will soon break. Am Sci. 2015; 103(6):395–403.
Sawakuchi AO, Hartmann GA, Sawakuchi HO, Pupim FN, Bertassoli Jr. DJ, ParraM et al. The Volta Grande do Xingu: reconstruction of past environments and forecasting of future scenarios of a unique Amazonian fluvial landscape. Sci Drill. 2015; 20:21–32. https://doi.org/10.5194/sd-20-21-2015
Schraml E, Schäfer F. Aqualog – Loricariidae: All L-Numbers, New 2nd edition. Hollywood Import & Export, Inc; 2004.
Seidel I, Evers H-G. Wels atlas, band 2. Mergus Verlag GmbH; 2005.
Seidel I. Back to Nature – Handbuch für L-Welse. Dahne; 2008.
Silva GSC, Roxo FF, Lujan NK, Tagliacollo VA, Zawadzki CH, Oliveira C. Transcontinental dispersal, ecological opportunity and origins of an adaptive radiation in the Neotropical catfish genus Hypostomus (Siluriformes: Loricariidae). Mol Ecol. 2016; 25(7):1511–29. https://doi.org/10.1111/mec.13583
Schaefer SA. The Neotropical cascudinhos: Systematics and biogeography of the Otocinclus catfishes (Siluriformes: Loricariidae). Proc Acad Nat Sci. 1997; 148:1–120.
Soares LC. Limites meridionais e orientais da área de ocorrência da floresta Amazônica em território brasileiro. Rev Bras Geogr. 1953; 15(1):3–95.
Tencatt LFC, Britto MR. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Araguaia basin, Brazil, with comments about Corydoras araguaiaensis Sands, 1990. Neotrop Ichthyol. 2016; 14(1):e150062. https://doi.org/10.1590/1982-0224-20150062
Tencatt LFC, Ohara WM. Two new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madeira basin, Brazil. Neotrop Ichthyol. 2016; 14(1):e150063. https://doi.org/10.1590/1982-0224-20150063
Winemiller KO, McIntyre PB, Castello L, Flute-Chouinard E, Giarrizzo T, Nam S et al. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science. 2016; 351(6269):128–29. https://doi:10.1126/science.aac7082
Zarfl C, Lumsdon AE, Berlekamp J, Tydecks L, Tockner K. A global boom in hydropower dam construction. Aquat Sci. 2015; 77:161–70. https://doi.org/10.1007/s00027-014-0377-0
Zuanon JAS. História natural da ictiofauna de corredeiras do rio Xingu, na região de Altamira, Pará. [PhD Thesis]. Campinas: Universidade Estadual de Campinas; 1999.
Authors
![]() Mateus S. Chaves1,
Mateus S. Chaves1, ![]() Renildo R. de Oliveira2,
Renildo R. de Oliveira2, ![]() Alany P. Gonçalves1,3,
Alany P. Gonçalves1,3, ![]() Leandro M. Sousa4 and
Leandro M. Sousa4 and ![]() Lúcia H. Rapp Py-Daniel2
Lúcia H. Rapp Py-Daniel2 ![]()
[1] Programa de Pós-Graduação em Biologia de Água Doce e Pesca Interior, Instituto Nacional de Pesquisas da Amazônia, Avenida André Araújo, 2936, Petrópolis, 69067-375 Manaus, AM, Brazil. (MSC) mateussantana58@gmail.com.
[2] Coleção de Peixes, Programa de Coleções Científicas e Biológicas – PCCB, Coordenação de Biodiversidade, Instituto Nacional de Pesquisas da Amazônia, Av. André Araújo, 2936, Petrópolis, 69067-375 Manaus, AM, Brazil. (RRO) deoliveirarr@gmail.com, (LHRPD) luciarpd60@gmail.com (corresponding author).
[3] Laboratório de Ecologia e Biologia de Peixes, Instituto de Desenvolvimento Sustentável Mamirauá, Estrada do Bexiga, 2584, Fonte Boa, 69553-225 Tefé, AM, Brazil. (APG) alany.xingu@gmail.com.
[4] Laboratório de Ictiologia de Altamira, Universidade Federal do Pará, Rua Coronel José Porfírio, 2515, 68372-040 Altamira, PA, Brazil. (LMS) leandro.m.sousa@gmail.com.
Authors’ Contribution 

Mateus S. Chaves: Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Renildo R. Oliveira: Conceptualization, Methodology, Writing-review and editing.
Alany P. Gonçalves: Investigation, Methodology, Project administration, Writing-review and editing.
Leandro M. Sousa: Funding acquisition, Investigation, Methodology, Project administration, Writing-review and editing.
Lúcia H. Rapp Py-Daniel: Conceptualization, Data curation, Formal analysis, Funding acquisition, Methodology, Project administration, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
How to cite this article
Chaves MS, Oliveira RR, Gonçalves AP, Sousa LM, Rapp Py-Daniel LH. A new species of armored catfish of the genus Scobinancistrus (Loricariidae: Hypostominae) from the Xingu River basin, Brazil. Neotrop Ichthyol. 2023; 21(3):e230038. https://doi.org/10.1590/1982-0224-2023-0038
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted August 24, 2023 by Marcelo Britto
Accepted August 24, 2023 by Marcelo Britto
![]() Submitted April 24, 2023
Submitted April 24, 2023
![]() Epub October 30, 2023
Epub October 30, 2023