![]() Janaína dos Santos Pedron1,
Janaína dos Santos Pedron1, ![]() Jurandir Joaquim Bernardes Junior1
Jurandir Joaquim Bernardes Junior1 ![]() ,
, ![]() Josiane Ribolli1,
Josiane Ribolli1, ![]() Jennyffer Souza2,
Jennyffer Souza2, ![]() Aline Guimarães Pereira2,
Aline Guimarães Pereira2, ![]() Hugo Tolentino2,
Hugo Tolentino2, ![]() Romuel Barros2,
Romuel Barros2, ![]() Carine de Freitas Milarch3,
Carine de Freitas Milarch3, ![]() Rafael José Gonçalves Dias4,
Rafael José Gonçalves Dias4, ![]() Renata Maria Guereschi1,
Renata Maria Guereschi1, ![]() Alexandra Latini2 and
Alexandra Latini2 and ![]() Alex Pires de Oliveira Nuñer1
Alex Pires de Oliveira Nuñer1
PDF: EN XML: EN | Cite this article
Abstract
This study investigated injuries sustained by Pimelodus maculatus caused by transient operating conditions in a Brazilian hydroelectric power plant. Fish (n = 43) that inhabited the tailrace and appeared on the water surface after changes in the operating conditions were evaluated (IF). Additionally, fish (n = 10) captured using longline in the vicinity of the tailrace were used for comparisons (NF). Blood samples were evaluated for cortisol, glucose, and lactate concentrations. Tissue samples (gill, liver, brain, and muscle) were analyzed for oxidative stress biomarkers (TBARS, protein carbonyl content, and catalase activity) and histology (gill, liver, and spleen). The most prominent findings in IF were barotraumas with intra-abdominal hemorrhage and swim bladder rupture. Cortisol and glucose concentrations were lower for IF when compared to NF. IF also suffered from oxidative stress, as indicated by increased TBARS in the liver and brain, as well as increased protein carbonyl content in the gills and liver. The main histological alterations induced by the adverse conditions were circulatory disorders, such as telangiectasia and gill aneurysm, hepatic congested veins, and hemorrhage in the liver and spleen. The abrupt drop of pressure resulting from changes in the operating conditions of the turbines caused barotraumas and oxidative stress, which were the leading causes of fish mortality
Keywords: Barotrauma, Blood parameters, Hemorrhage, Oxidative stress, Uruguay River.
Este estudo investigou os efeitos induzidos por condições transitórias de operação em uma usina hidrelétrica brasileira em Pimelodus maculatus. Peixes (n = 43) no canal de fuga que apareceram na superfície da água após mudanças nas condições operacionais foram avaliados. Adicionalmente, peixes (n = 10) capturados a jusante da barragem com espinhel foram utilizados para comparações (NF). Amostras de sangue foram avaliadas para as concentrações de cortisol, glicose e lactato. Amostras de tecidos (brânquia, fígado, cérebro e músculo) foram analisadas para biomarcadores de estresse oxidativo (TBARS, conteúdo de proteína carbonil e atividade da catalase) e histologia (brânquia, fígado e baço). Os achados mais proeminentes dos peixes afetados (IF) foram barotraumas com hemorragia intra-abdominal e ruptura da bexiga natatória. As concentrações de cortisol e glicose foram menores para IF quando comparadas aos NF. IF também sofreram estresse oxidativo representado pelo aumento de TBARS no fígado e cérebro, e aumento do conteúdo das proteínas carboniladas nas brânquias e fígado. As principais alterações histológicas induzidas pelas condições adversas foram distúrbios circulatórios, como telangiectasia e aneurisma nas brânquias, congestão das veias hepáticas, e hemorragia no fígado e baço. A queda abrupta da pressão resultante de mudanças nas condições operacionais das turbinas causou barotrauma e estresse oxidativo, que foram as principais causas da mortalidade dos peixes.
Palavras-chave: Barotrauma, Estresse oxidativo, Hemorragia, Parâmetros sanguíneos, Rio Uruguai.
Introduction
Fish passage through turbines from upstream to downstream is considered one of the major causes of fish mortality at hydroelectric power plants (HPPs) in the Northern Hemisphere (Cada, 2001; Brown et al., 2014). In Brazilian dams, however, fish mortality related to HPP operation is mainly associated with fish movements from downstream to upstream where fish can enter the tailrace and draft tube during transient operating conditions of the turbine, such as shutdown, startup, and synchronous condenser states (Silva et al., 2018). In all cases, the leading causes of fish mortality and injuries are strike (mechanical shock against building structures, as well as runner blades), shear stress (friction between two water bodies at different velocities), cavitation (bubble formation that causes shock waves at high pressure), and abrupt pressure changes under the runner (Beirão et al., 2016; Carvalho, Marques, 2018).
Rapid decompression in the turbines causes the fish to suffer internal and external tissue injuries, known as barotrauma. Barotrauma includes various injuries such as hemorrhage, embolism, stomach eversion, exophthalmia, swim bladder, and other organ rupture (Algera et al., 2020).
When fish undergo external adverse conditions, such as passage through turbines or rapid decompression, physiological and biochemical responses are triggered, along with compensatory or adaptive mechanisms, in an attempt to overcome the stressor. For instance, the stress hormone cortisol triggers physiologic responses like increasing metabolic parameters, such as glucose and lactate, to recover fish homeostasis (Barton, Iwama, 1991; Trushenski et al., 2010). Also, fish can activate their antioxidant defense systems by producing catalase enzymes, to overcome oxidative cell damage, such as lipid and protein peroxidation, measured as TBARS (thiobarbituric acid reactive substances), and protein carbonylation, respectively (Velisek et al., 2011). Through histology, it is also possible to verify fish health because tissue changes express stress-induced conditions (Copper et al., 2018). Thus, evaluating blood parameters, oxidative tissue stress, and histology are tools for monitoring and understanding the health of downstream fish captured after abrupt decompression exposure at HPPs, as well as turbine-passed fish.
In Brazil, catfish comprise one of the groups most frequently found downstream during mortality episodes owing to transient operating conditions of HPPs (Silva et al., 2018). Pimelodus maculatus (Lacepède, 1803) is a yellow-mandi catfish widely distributed in several South American hydrographic basins, including the Uruguay River (Zaniboni Filho, Schulz, 2003), where Machadinho HPP is located. This is the fish species most frequently identified with lethal injuries after switching the turbine operating regime at this HPP, as in other dams in Brazil (Andrade et al., 2012). Although several studies reported fish mortality at HPPs, the physiological and biochemical responses are unclear because they commonly focus on macroscopic lesions. Therefore, this study aimed to investigate the injuries of P. maculatus exposed to abrupt decompression inside the draft tube at Machadinho HPP by evaluating morphological, physiological, and biochemical parameters.
Material and methods
Study area and fish sampling. The Machadinho HPP is located on the upper Uruguay River (27°31’31”S and 51°47’07”W). It borders Santa Catarina and Rio Grande do Sul states in southern Brazil. It has three Francis turbines, each with a 380 MW output, totaling 1,140 MW of installed capacity. From September 2019 to June 2021, the tailrace was inspected 11 times during changes in the operating regimes (Tab. 1). The inspections consisted of observing fish that appeared on the water surface for approximately 20 min. When sighted floating with erratic swimming, P. maculatus individuals were sampled (n = 43) for immediate analysis. The species was also captured in the immediate vicinity of the tailrace (n = 10) using longline gear for comparative analyses. A voucher specimen was deposited in the Ichthyological Collection of the Universidade Estadual de Londrina, Brazil (MZUEL 9599).
TABLE 1 | Transient operating conditions of turbines at the Machadinho hydroelectric power plant (HPP) where catfish sustained injuries evaluated in this study.
Transient operating condition | Description |
Startup | The turbine runner goes from rest to
nominal speed in seven min (slow startup scheme), the unit is synchronized to
the grid, and the load is increased (load acceptance). |
Synchronous condenser to generating mode | At the synchronous condenser mode, the
turbine is operated in air conditions rotating at the synchronous speed
driven by the generator acting as a motor. Depending on the energy demand,
the turbine goes from synchronous condenser mode to generating mode. The
scheme adopted by the Machadinho HPP to return to
generating mode involves a sequence of shutdown and slow startup. |
Shutdown | The turbine runner comes to stop after
working as synchronous condenser, by opening the deaeration
valve to expel air from the runner to the atmosphere and break the runner by
friction with water under the runner. |
For convenience, the term “Injured Fish (IF)” refers to fish collected on the water surface during tailrace inspections, whereas “Naïve Fish (NF)” refers to those captured using longline in the proximity of the tailrace.
Sampling and analyses. Fish were measured, weighed, and then anesthetized using 50 mg L-1 eugenol. Blood was sampled from the caudal vein for glucose (Accu Cheek Active, Roche, Germany), lactate (Accutrend Plus, Roche, Germany), and cortisol analyses. A 3.0 mL syringe with anticoagulant (EDTA, 10%) was used, and the collected blood was stored in 2 mL sterile microtubes. Cortisol determination used plasma from centrifuged blood (2,500 x g, 15 min, 4 ºC), maintained frozen in liquid nitrogen (-180 ºC) until analyses, and measured in duplicate using ELISA kits (Arbor Assays Cortisol Immunoassay, Arbor Assays, USA). After blood sampling, fish were euthanized with an overdose of the anesthetic (100 mg L-1 eugenol) and submitted to necropsy. Necropsy consisted of external and internal macroscopic analysis.
Gill, liver, brain, and muscle samples were collected and stored in liquid nitrogen (-180 ºC) until required for biochemical analyses. Levels of the biomarkers thiobarbituric acid reactive substances (TBARS) and protein carbonyl, as well as catalase activity, were measured using the methods described by Reznick, Packer (1994), Buege, Aust (1978), and Nelson, Kiesow (1972), respectively with some modifications as detailed in Thiel et al. (2020). For IF, biochemical and hematological analysis were carried out with 30 randomly sampled fish.
Gill, liver, and spleen tissues sampled for histology were fixed in 10% buffered formalin solution for 24 h, washed in 70% alcohol, and stored in 70% alcohol until analysis. Next, tissues were dehydrated in a series of alcohol solutions (70, 80, 90, and 100%) and embedded in paraffin following a routine technique. Subsequently, 5 μm thick cross-sections were taken using a Leica RM2255 rotary microtome (Leica Microsystems, Germany). Then, each tissue was stained with hematoxylin and eosin using the automatic Leica AutoStainer XL (Leica Microsystems, Germany). Images were captured under a light microscope (Leica DM3000 LED, Leica Microsystems, Germany) with a connected camera and analyzed with Leica Application Suite 3.4.0 (Leica Microsystems, Switzerland).
Statistical analyses. Comparisons among groups were made by one-way ANOVA followed by Tukey-Kramer test after checking for homoscedasticity (Levene’s test) and normality (Shapiro-Wilk test). Outliers were detected and removed from the analysis using the Tukey outlier detection test (k = 2.1). The Kruskal-Wallis test was performed when assumptions were violated. The frequencies of lesions were compared between groups using Fisher’s test. All analyses were performed using Statistica Software 7.0 (Stat Soft, USA) at a 0.05 significance level.
Results
Pimelodus maculatus sampled during inspections of operating regime shifts showed 26 types of injuries, and the percentage of occurrence varied with the operation performed (Tab. 2). Cortisol (F1,39 = 31.4; P < 0.05) and glucose (F1,32 = 15.1; P < 0.05) levels were lower in IF, while no changes were observed in lactate concentrations (F1,33 = 2.2; P > 0.05) (Tab. 3).
TABLE 2 | Types of injuries and their frequency of occurrence (%) as determined following necropsy of Pimelodus maculatus collected in the tailrace at the transient operating regimes of machine shutdown (n = 16), startup (n = 22), and synchronous condenser (SC) to generating mode (n = 5).
Injury | Shutdown | Start-up | SC
to generating mode |
Branchial arch rupture | – | 4.5 | – |
Swim bladder rupture | 93.8 | 90.0 | 80.0 |
Intestine rupture | – | 4.5 | – |
Spleen ruptured | – | – | 20.0 |
Intra-abdominal hemorrhage | 68.8 | 72.7 | 100.0 |
Branchial hemorrhage | 12.5 | 27.3 | 20.0 |
Eye hemorrhage | 6.3 | – | – |
Subcutaneous hemorrhage | 12.5 | 13.6 | – |
Abdominal distension | 68.8 | 54.5 | 100.0 |
Over-inflated swim bladder | 6.3 | 4.5 | 20.0 |
Subcutaneous emphysema | 6.3 | 4.5 | – |
Grazes/Laceration | 18.8 | 9.1 | – |
Stomach eversion | 12.5 | 18.2 | 20.0 |
Exophthalmia | 18.8 | 9.1 | 20.0 |
Viscera extrusion through the urogenital
opening | – | 4.5 | 60.0 |
Eye loss | 6.3 | – | – |
Venous embolism | 6.3 | 18.2 | – |
Embolism in kidney | – | 13.6 | – |
Hemorrhagic fins | 12.5 | 13.6 | – |
Hemorrhagic kidney | – | 27.3 | – |
Hemorrhagic liver | 12.5 | 22.7 | – |
Hemorrhagic spleen | 43.8 | 22.7 | – |
Opercular fracture | 6.3 | 4.5 | – |
Spine fracture | – | 4.5 | – |
Fins fracture | 6.3 | 4.5 | – |
Head fracture | 12.5 | 18.2 | – |
TABLE 3 | Hematological responses of Pimelodus maculatus captured using longline in the vicinity of the tailrace (NF) or collected in the tailrace during transient operating conditions (IF) of the Machadinho hydroelectric power plant (HPP). Data are reported as mean ± standard deviation. Different letters indicate significant differences (P < 0.05) among groups for each biochemical parameter.
Blood parameter | NF | IF |
Cortisol (ng mL-1) | 39.9 ± 16.9a (n = 10) | 13.2 ± 11.4b (n = 30) |
Glucose (mg dL-1) | 86.1 ± 55.7a (n = 10) | 21.6 ± 11.5b (n = 23) |
Lactate (mmol L-1) | 8.2 ± 3.9a (n = 10) | 5.7 ± 4.6a (n = 24) |
IF presented higher levels of TBARS in the liver (F1,39 = 7.7; P < 0.05) and brain (F1,38 = 6.9; P < 0.05), increased protein carbonyl content in the gill (F1,33 = 371.3; P < 0.05) and liver (F1,38=5.4; P < 0.05), and lower protein carbonyl content in the muscle (F1,37 = 8.4; P < 0.05). Catalase activity was reduced in the brain (F1,34 = 20.1; P < 0.05) of IF (Tab. 4).
TABLE 4 | Biochemical responses in different tissues (gill, liver, brain, and muscle) of Pimelodus maculatus captured using longline in the vicinity of the tailrace (NF) or collected in the tailrace during transient operating conditions (IF) of the Machadinho hydroelectric power plant (HPP). Data are reported as mean ± standard deviation. Different letters indicate significant differences (P < 0.05) among groups for each parameter and tissue. TBARS = thiobarbituric acid reactive substances.
Biochemical parameter | Fish group | Tissue | |||
Gill | Liver | Brain | Muscle | ||
TBARS levels | NF | 554.9 ± 36.7a (n = 10) | 147.3 ± 107.8a (n = 10) | 30.4 ± 19.2a (n = 10) | 3.7 ± 2.9a (n = 8) |
IF | 475.1 ± 331.9a (n = 26) | 630.5 ± 543.3b (n = 30) | 46.4 ± 15.7b (n = 30) | 3.9 ± 2.9a (n = 27) | |
Protein carbonyl
content | NF | 137.5 ± 19.8a (n = 9) | 127.6 ± 36.5a (n = 10) | 59.2 ± 48.9a (n = 10) | 32.6 ± 21.7a (n = 10) |
IF | 398.0 ± 38.5b (n = 25) | 168.1 ± 50.2b (n = 29) | 74.6 ± 46.9a (n = 30) | 18.6 ± 8.3b (n = 28) | |
Catalase activity | NF | 100.4 ± 74.3a (n = 10) | 334.2 ± 249.9a (n = 10) | 122.2 ± 54.9a (n = 10) | 31.9 ± 29.5a (n = 10) |
IF | 71.4 ± 59.1a (n = 30) | 488.2 ± 474.3a (n = 30) | 56.6 ± 29.5b (n = 30) | 35.7 ± 23.3a (n = 30) | |
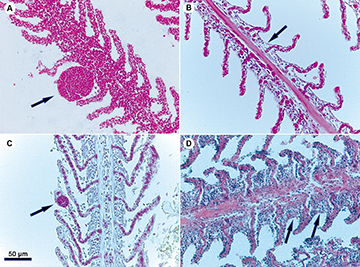
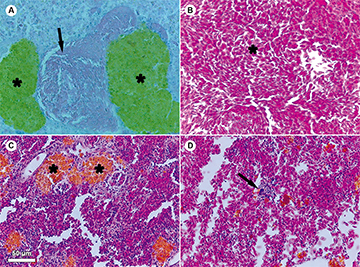
Four types of gill lesions were detected in histological analyses (Fig. 1). Epithelium detachment (Fig. 1B), telangiectasia (Fig. 1C), and interlamellar hyperplasia (Fig. 1D) were higher (P < 0.05) in IF (Tab. 5). Branchial aneurysm (Fig. 1A), telangiectasia (Fig. 1C), and interlamellar hyperplasia (Fig. 1D) were not found in NF. The presence of congested veins and hemorrhage were identified in the liver and spleen of IF, but it was only significant (P < 0.05) in congested veins at the liver (Tab. 6; Figs. 2A–B). Melanomacrophage centers were found in the liver and spleen of all fish and their presence was no different (P > 0.05) between IF and NF (Tab. 6; Figs. 2A, C). Mononuclear inflammatory infiltrates were higher (P < 0.05) in NF spleen (Tab. 6: Fig. 2D). They were not found in the liver of NF or IF.
TABLE 5 | Frequency of occurrence (%) of histological gill lesions found for Pimelodus maculatus captured using longline in the vicinity of the tailrace (NF) or collected in the tailrace during transient operating conditions (IF) of the Machadinho hydroelectric power plant (HPP). Different letters indicate significant differences (P < 0.05) among groups for each lesion.
Fish group | Aneurysm | Epithelium
detachment | Telangiectasia | Interlamellar hyperplasia |
NF | 0.0a | 20.0a | 0.0a | 0.0a |
IF | 28.1a | 71.9b | 46.9b | 87.5b |
TABLE 6 | Frequency of occurrence (%) of histological liver and spleen lesions found in Pimelodus maculatus captured using longline in the vicinity of the tailrace (NF) or collected in the tailrace during transient operating conditions (IF) of the Machadinho hydroelectric power plant (HPP). Different letters indicate significant differences (P < 0.05) among groups for each lesion.
Tissue | Fish group | Melanomacrophage Centers | Mononuclear
inflammatory infiltrate | Congested veins | Hemorrhage |
Liver | NF | 100.0a | 0.0a | 0.0a | 0.0a |
IF | 100.0a | 0.0a | 70.3b | 24.3a | |
Spleen | NF | 100.0a | 100.0a | 0.0a | 0.0a |
IF | 100.0a | 64.5b | 29.0a | 12.9a |
FIGURE 1| Histological gill alterations in Pimelodus maculatus collected during transient operating conditions (magnification: × 400). Arrows indicate the following lesions: A. Aneurysm; B. Epithelium detachment; C. Telangiectasia; and D. Interlamellar hyperplasia.
FIGURE 2| Histological alterations in liver and spleen of Pimelodus maculatus collected during transient operating conditions in the tailrace of Machadinho HPP (magnification: × 400). Arrows and asterisks indicate the following lesions: A. Melanomacrophage centers (asterisk) and congested vein (arrow) in the liver; B. Blood cells indicating hepatic hemorrhage (asterisk); C. Melanomacrophage centers in the spleen (asterisk); D. Mononuclear inflammatory infiltrate in the spleen (arrow).
Discussion
Hydrostatic pressure variation on the turbine runner during transient operating conditions causes fish to decompress and, consequently, suffer from barotrauma (Beirão et al., 2016). This is the same kind of pressure drop experienced by turbine-passed fish during steady state operating conditions, typical of partial load or full load operation at hydropower plants worldwide (Cada et al., 2006; Brown et al., 2014).
Pimelodus maculatus collected in the tailrace after changing the operating condition were found with various barotraumas, such as swim bladder rupture, hemorrhage, embolism, stomach eversion, and exophthalmia. Bone fractures as result of mechanical shocks were also observed, as described for other species (Brown et al., 2009; Algera et al., 2020).
The swim bladder controls fish buoyancy in the water column, acting as a gas reservoir, and its retention capacity depends on fish morphology (Stephenson et al., 2010; Pflugrath et al., 2012). Pimelodus maculatus is a benthic and physoclistous species that exchanges gas through the circulatory system. It often maintains negative buoyancy by retaining a little air in the swim bladder (Silva et al., 2018). Therefore, when this fish undergoes rapid decompression, the dissolved gases boil (Henry’s Law) and expand (Boyle’s Law), causing the swim bladder to overinflate and burst (Brown et al., 2009), a condition that affected 90% of the IF evaluated in this study.
Hemorrhage is a frequent finding that results from gas embolism, i.e., bubble formation in blood vessels. It increases blood volume and reduces blood density, causing organ rupture and blood loss, leading to death (Brown et al., 2012, 2014). More than 70% of IF presented widespread bleeding, which extended to the peritoneal cavity, liver, spleen, kidney, gills, eyes, skin, and fins.
Adaptive stress responses, classified as primary and secondary, follow a pattern in most fish, and several of these responses can be analyzed through blood (Wandelaar-Bonga, 1997; George et al., 2013). Among the primary responses, cortisol is the most important corticosteroid hormone produced during stressor events. The hypothalamic-pituitary-interrenal axis controls the release of cortisol that acts mainly on the gills, intestine, and liver, regulating metabolic energy and hydromineral balance (Barton, Iwama, 1991). Secondary responses are triggered by primary responses and are mainly metabolic in origin. The release of glucose from the liver by glycogenolysis, known as hyperglycemia, is a metabolic response mediated by catecholamines, acting as a substrate for energy production. Also, in anaerobic situations, lactate is released as an energy source for cellular metabolism (Olsen et al., 1995; Polakof et al., 2012).
The reference values of blood stress parameters are unknown for fish exposed to pressure drops. However, it is possible to infer those values from fish farming studies. Barcellos et al. (2003), for example, have found increased cortisol (seven times) and glycose (three times) levels in the juvenile silver catfish Rhamdia quelen (Quoy & Gaimard, 1834) after harvesting. In juvenile cobia Rachycentron canadum (Linnaeus, 1766) exposed to air, cortisol, glucose, and lactate have increased six, three, and eight times, respectively (Trushenski et al., 2010).
The cortisol and glucose concentrations in fish under stress rise to establish homeostasis (Iversen et al., 2009). Accordingly, we were expecting higher cortisol and glucose levels in IF compared to those in NF owing to the severity and irreversibility of the trauma herein described. Notably, even fishing is a stressor able to drive both stress indicators away from the baseline in NF. However, systemic response did not occur in IF, possibly because of hemorrhage. Typically, cortisol travels through the bloodstream signaling the onset of a stressful situation (Wandelaar-Bonga, 1997). However, the severe bleeding found in most IF (70%) blocked the activation of physiological responses that would otherwise have increased cortisol and glucose levels. Therefore, in the present study, neither cortisol nor glucose was suitable as a stress marker in the assessment of fish exposed to pressure drop.
To the best of our knowledge, the present study is the first to perform biochemical analyses of oxidative stress parameters on fish affected by HPP operation, a common approach in ecotoxicological studies (Parvez, Raisuddin, 2005; Menezes et al., 2011; Toni et al., 2011). Oxidative stress is the imbalance between the generation of oxidant compounds and the action of antioxidant defense systems. A deficiency in the antioxidant system or insufficient ability to repair oxidative damage can increase reactive oxygen species and cause cell death (Barbosa et al., 2010; Halliwell, Gutteridge, 2015).
Lipid peroxidation, measured as TBARS has been reported as the one of the main factors responsible for the loss of cellular functions under oxidative stress conditions (Almroth et al., 2005; Barata et al., 2005; Menezes et al., 2011). Lipid peroxidation acts as a mechanism of cell injury caused by oxygen free radicals in biological membranes. These reactions produce lipid hydroperoxides which break down the double bond of unsaturated fatty acids and damage the cell membrane (Barata et al., 2005). The formation of lipid hydroperoxides can, in turn, further oxidize proteins, causing an excessive formation of carbonylated proteins, a biomarker for protein oxidation assessment (Lackner, 1998; Almroth et al., 2005).
TBARS levels have been reported to increase in the gills, liver, and muscle of lambari Astyanax jacuhiensis (Cope, 1894) (= A. lacustris (Lütken, 1875)) in the presence of five pesticides in the water (Loro et al., 2015). Also, the increased levels of TBARS in the gill, liver, and kidney previously demonstrated that Prochilodus lineatus (Valenciennes, 1837) is living under stressful field conditions in the Salado River basin (Santa Fé, Argentina) (Cazenave et al., 2009). Similarly,increased TBARS levels in the liver (328%) and brain (52%) of IF in the present study showed that changes in the operating conditions of HPP turbines also caused cellular damage to P. maculatus tissues as a direct result of stress and trauma.
IF also showed increased protein carbonyl levels in the gills (190%) and liver (32%). Similarly, increased protein carbonyl content was reported in the gill (200%), liver (111%), and kidney (190%) of Channa punctata (Bloch, 1793) exposed to the pesticide deltamethrin (Parvez, Raisuddin, 2005), as well as the liver of silver catfish exposed to herbicides (Ferreira et al., 2010; Menezes et al., 2011). Unexpectedly, IF muscle showed a reduction of protein carbonyl content (44%). In this case, exhaustion caused by the attempt to escape from the longline could explain the higher oxidative stress in NF muscle compared to that in IF. Protein carbonylation is induced by reactive oxygen species (ROS), which are produced in low amount during muscle contractility (Dalle-Donne et al., 2003). Under strenuous activity, however, ROS accumulate in the muscle, increasing oxidative stress (Duberstein et al., 2009).
Organisms have developed antioxidant defense systems to minimize oxidative damage to cellular components, as determined through the activity of antioxidant enzymes, such as catalase (Barata et al., 2005; Monteiro et al., 2006). Catalase is an enzyme that belongs to the first line of antioxidant defense and converts hydrogen peroxide into water and oxygen (Halliwell, Gutteridge, 2015). Catalase and other antioxidant enzymes can increase, decrease, or remain unchanged, depending on the intensity and duration of the stressor event, or even vary according to fish species (Ballesteros et al., 2009).
Catalase activity was reported to decreased in the liver of R. quelen infected with Aeromonas hydrofila. This was attributed to the low capacity of this species to neutralize the hydrogen peroxide produced by high TBARS, contributing, in turn, to the oxidative liver damage (Baldissera et al., 2017). A similar trend in these biomarker levels was observed in the brain of IF, where high TBARS levels occurred, along with a decrease in catalase activity, indicating oxidative brain damage.
The brain has very low antioxidant defenses compared to other tissues, e.g., the liver (Halliewell, Gutteridge, 2015). In addition, the brain is characterized by high oxygen consumption and lipid-rich content; therefore, it is highly susceptible to oxidative stress. In IF, decreased catalase activity and increased TBARS in the brain indicate the induction of oxidative damage and further indicate that such damage may be related to the severity of the lesions. It is postulated that catalase levels are low in all brain regions compared to the liver. Owing to the intrinsic characteristics of the central nervous system and its metabolism, several antioxidant defense systems are, however, present in the brain, making it more susceptible to damage caused by oxidant species (Halliwell, Gutteridge, 2015; Streck et al., 2013).
Two fish species, Oreochromis niloticus (Linnaeus, 1758) and Clarias gariepinus (Burchell, 1822), reported to be chronically exposed to metal concentrations in water and sediments, showed marked increases in catalase activity and TBARS levels in their liver and gills (Moussa et al., 2022). Since these species have antioxidant defense capacity against different stressors, such findings suggest these organs are good indicators of pollution. In the present study, our analyses showed an increase in TBARS and protein carbonyl content in the liver, but no change in catalase activity. Perhaps the time required for an increase in catalase expression in this tissue was not sufficient to generate antioxidant responses to trauma. Thus, our data provide new information on the use of oxidative markers in IF, mainly catalase activity, TBARS levels and protein carbonyl content in the different organs analyzed.
For P. maculatus, no changes in catalase activity were observed in the liver and gills, even with an increase in TBARS levels and protein carbonyl in the liver. The lack of antioxidant enzyme response contributes to more substantial lipid and protein oxidation and, consequently, more severe oxidative damage (Toni et al., 2011). Toni et al. (2011) also did not observe changes in catalytic activity of in Cyprinus carpio (Linnaeus, 1758) exposed to tebuconazol, even with the increased levels of TBARS and protein carbonyl content.
Histology is a widely used tool to determine fish health and physical condition because tissue changes express conditions induced by stress, pathogens, inadequate nutrition, improper water quality, or injuries suffered (Copper et al., 2018). However, this approach is rarely applied in investigating the possible causes of fish mortality at HPPs (Brown et al., 2009). Our evaluation of IF histology revealed several lesions related to circulatory disorders, such as aneurysm and telangiectasia in the gills, congested veins in the liver, and hemorrhage in the liver and spleen.
Aneurysms and telangiectasia in the gill are related to physical or chemical damage, usually linked to trauma, intense handling, or chemical contaminants (Flores-Lopes, Thomaz, 2011). These circulatory injuries are associated with an increase in blood flow increase owing the dilatation of blood capillaries, starting with blood congesting the extremity of the secondary lamellae (telangiectasia) and then developing into an aneurysm, congesting all lamellae (Strzyżewska-Worotyńska et al., 2017). In most cases, these are irreversible lesions, and when many lamellae are affected, the respiratory capacity of fish is compromised (Roberts, 2002). Among the hemorrhagic findings from the necropsy of the injured fish, both aneurysm and telangiectasia resulted from abrupt hydrostatic pressure variation.
Congested veins are indicative of blockages in blood passage from the hepatic artery and spleen to the central vein, requiring greater effort from the heart to pump blood (Kostić et al., 2017). However, because of abdominal and local bleeding in organs like the liver and spleen, even greater pumping could not supply the circulatory loss in severe hemorrhage.
The liver and spleen of NF and IF presented melanomacrophage centers, which are typically located in the hematopoietic tissues of these organs, as well as kidneys (Wolke, 1992). Therefore, it is implausible that they were associated with the injuries caused by changes in the operating conditions. The melanomacrophage centers are associated with inflammatory processes, metabolizing toxic compounds by phagocytosis, and iron cycling; accordingly, they have been considered suitable water quality biomarkers of environmental pollution (Agius, Roberts, 2003; Sayed, Younes, 2017). Some studies have related the higher incidence of melanomacrophage centers in P. maculatus liver to water contaminants (Brito et al., 2012; Paulino et al., 2014).
Mononuclear inflammatory infiltrates, which include first-line defense cells such as macrophages, lymphocytes, and plasmocytes (Manrique et al., 2017), were observed in the spleen of most fish evaluated, indicating that changes in the operating conditions did not trigger the inflammatory process. Araújo et al. (2019) have also found mononuclear inflammatory infiltrates in the liver of P. maculatus collected in the Funil’s HPP (Rio de Janeiro, Brazil), suggesting that these infiltrates likely result from sublethal concentrations of pollutants.
Morphological changes were also found in IF gills, such as interlamellar hyperplasia and detachment of the lamellar epithelium. Excessive cell proliferation, such as that which occurs in hyperplasia, is an adaptive response that increases the distance between the external environment and the blood, reducing the contact surface area with water. As this process intensifies, the diffusion of respiratory gases reduces causing hypoxia and death (Mallatt, 1985; Sepici-Dinçel et al., 2009). The detachment of the lamellar epithelium can lead to osmoregulatory disorders, also caused by the difficulty in carrying out gas exchange (Pereira et al., 2014).
Pimelodus maculatus inhabiting the tailrace when exposed to the transient operating conditions showed barotrauma as the main cause of mortality, with swim bladder rupture and hemorrhage being the most recurrent traumas. Diffuse hemorrhage was the most critical effect because it caused circulatory disorders that promoted gill lesions, such as aneurysms. Furthermore, liver and spleen lesions, such as congested veins and blood circulation blockage, inhibited the activation of hormonal cascade essential for fish recovery and oxygen transport. In addition, lipid and protein oxidative damage to all analyzed tissues probably prevented the synthesis of antioxidant enzymes that would have otherwise helped in fish recovery, thus causing irreversible cell damage.
Acknowledgments
We thank the LAPAD (Laboratório de Biologia e Cultivo de Peixes de Água Doce) staff (Maurício Machado, Pedro Iaczinski, and Ronaldo Silva) who cooperated in fieldwork. We are also grateful to LABOX (Laboratório de Bioenergética e Estresse Oxidativo) and LAMEB (Laboratório Multiusuário de Estudos em Biologia) for collaborating with the processing and analysis of oxidative stress and histology, respectively. This research was developed within the scope of the Research & Development Program regulated by ANEEL (PD-00403-0046/2019) and supported by Engie Brasil Energia.
References
Agius C, Roberts RJ. Melano-macrophage centres and their role in fish pathology. J Fish Dis. 2003; 26(9):499–509. https://doi.org/10.1046/j.1365-2761.2003.00485.x
Algera DA, Rytwinski T, Taylor JJ, Bennett JR, Smokorowski KE, Harrison PM et al. What are the relative risks of mortality and injury for fish during downstream passage at hydroelectric dams in temperate regions? A systematic review. Environ Evid. 2020; 9(3). https://doi.org/10.1186/s13750-020-0184-0
Almroth BC, Sturve J, Berglund Å, Förlin L. Oxidative damage in eelpout (Zoarces viviparus), measured as protein carbonyls and TBARS, as biomarkers. Aquat Toxicol. 2005; 73(2):171–80. https://doi.org/10.1016/j.aquatox.2005.03.007
Andrade F, Prado IG, Loures RC, Godinho AL. Evaluation of techniques used to protect tailrace fishes during turbine maneuvers at Três Marias Dam, Brazil. Neotrop Ichthyol. 2012; 10(4):723–30. https://doi.org/10.1590/S1679-62252012000400005
Araújo FG, Gomes ID, Nascimento AA, Santos MAJ, Sales A. Histopathological analysis of liver of the catfish Pimelodus maculatus in a tropical eutrophic reservoir from Southeastern Brazil. Acta Sci Biol Sci. 2019; 41(1):e41039. https://doi.org/10.4025/actascibiolsci.v41i1.41039
Baldissera MD, Souza CF, Júnior GB, Vargas AC, Boligon AA, Campos MMA et al. Melaleuca alternifolia essential oil enhances the non-specific immune system and prevents oxidative damage in Rhamdia quelen experimentally infected by Aeromonas hydrophila: Effects on cholinergic and purinergic systems in liver tissue. Fish Shellfish Immunol. 2017; 61:1–08. https://doi.org/10.1016/j.fsi.2016.12.016
Ballesteros ML, Wunderlin DA, Bistoni MA. Oxidative stress responses in different organs of Jenynsia multidentata exposed to endosulfan. Ecotoxicol Environ Saf. 2009; 72(1):199–205. https://doi.org/10.1016/j.ecoenv.2008.01.008
Barata C, Varo I, Navarro JC, Arun S, Porte C. Antioxidant enzyme activities and lipid peroxidation in the freshwater cladoceran Daphnia magna exposed to redox cycling compounds. Comp Biochem Physiol C Toxicol Pharmacol. 2005; 145(2):175–86. https://doi.org/10.1016/j.cca.2005.01.013
Barbosa KBF, Costa NMB, Alfenas RCG, Paula SO, Minim VPR, Bressan J. Estresse oxidativo: conceito, implicações e fatores modulatórios. Rev Nutr. 2010; 23(4):629–43. https://doi.org/10.1590/S1415-52732010000400013
Barcellos LJG, Kreutz LC, Rodrigues LB, Fioreze I, Quevedo RM, Cericato L et al. Haematological and biochemical characteristics of male jundiá (Rhamdia quelen Quoy & Gaimard Pimelodidae): changes after acute stress. Aquac Res. 2003; 34(15):1465–69. https://doi.org/10.1111/j.1365-2109.2003.00972.x
Barton BA, Iwama GK. Physiological changes in fish from stress in aquaculture with emphasis on the response and effects of corticosteroids. Annu Rev Fish Dis. 1991; 1:3–26. http://dx.doi.org/10.1016/0959-8030(91)90019-g
Beirão BV, Marcian NCB, Dias LS, Falcão RC, Dias EW, Fabrino DL et al. Barotrauma em peixes em usinas hidrelétricas: ferramentas para o estudo. Bol Soc Bras Ictiol. 2016; 115:26–36.
Brito IA, Freire CA, Yamamoto FY, Assis HCS, Souza-Bastos LR, Cestari MM et al. Monitoring water quality in reservoirs for human supply through multi-biomarker evaluation in tropical fish. J Environ Monit. 2012; 14(2):615–25. https://doi.org/10.1039/c2em10461j
Brown RS, Carlson TJ, Gingerich AJ, Stephenson JR, Pflugrath BD, Welch AE et al. Quantifying mortal injury of juvenile chinook salmon exposed to simulated hydro-turbine passage. T Am Fish Soc. 2012; 141(1):147–57. https://doi.org/10.1080/00028487.2011.650274
Brown RS, Carlson TJ, Welch AE, Stephenson JR, Abernethy CS, Ebberts BD et al. Assessment of barotrauma from rapid decompression of depth-acclimated juvenile chinook salmon bearing radiotelemetry transmitters. T Am Fish Soc. 2009; 138(6):1285–301. https://doi.org/10.1577/t08-122.1
Brown RS, Colotelo AH, Pflugrath BD, Boys CA, Baumgartner LJ, Deng ZD et al. Understanding barotrauma in fish passing hydro structures: a global strategy for sustainable development of water resources. Fisheries. 2014; 39(3):108–22. https://doi.org/10.1080/03632415.2014.883570
Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1978; 52:302–10. https://doi.org/10.1016/s0076-6879(78)52032-6
Cada G, Loar J, Garrison L, Fisher Jr. R, Neitzel D. Efforts to reduce mortality to hydroelectric turbine-passed fish: Locating and quantifying damaging shear stresses. Environ Manage. 2006; 37:898–906. https://doi.org/10.1007/s00267-005-0061-1
Carvalho AG, Marques EE. Mitigação de injúrias e mortalidade de peixes em turbinas e vertedouros de hidrelétricas: meta-síntese de pesquisas científicas publicadas em periódicos. Rev Cereus. 2018; 10(4):45–67. https://doi.org/10.18605/2175-7275/cereus.v10n4p45-67
Cazenave J, Bacchetta C, Parma MJ, Scarabotti PA, Wunderlin DA. Multiple biomarkers responses in Prochilodus lineatus allowed assessing changes in the water quality of Salado River basin (Santa Fe, Argentina). Environ Pollut. 2009; 157(11):3025–33. https://doi.org/10.1016/j.envpol.2009.05.055
Copper JE, Budgeon LR, Foutz CA, Rossum DBV, Vanselow DJ, Hubley MJ et al. Comparative analysis of fixation and embedding techniques for optimized histological preparation of zebrafish. Comp Biochem Physiol C Toxicol Pharmacol. 2018; 208:38–46. https://doi.org/10.1016/j.cbpc.2017.11.003
Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R. Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta. 2003; 329(1–2):23–38. https://doi.org/10.1016/S0009-8981(03)00003-2
Duberstein KJ, Johnson SE, McDowell LR, Ott EA. Protein carbonyl assay to measureoxidative stress in muscle of exercising horses supplemented with vitamin E. Comp Exerc Physiol. 2009; 6(1):1–05. https://doi.org/10.1017/S1478061509356121
Ferreira D, Motta AC, Kreutz LC, Toni C, Loro VL, Barcellos LJG. Assessment of oxidative stress in Rhamdia quelen exposed to agrichemicals. Chemosphere. 2010; 79(9):914–21. https://doi.org/10.1016/j.chemosphere.2010.03.024
Flores-Lopes F, Thomaz AT. Histopathologic alterations observed in fish gills as a tool in environmental monitoring. Braz J Biol. 2011; 71(1):179–88. https://doi.org/10.1590/S1519-69842011000100026
George N, Peter VS, Peter MCS. Physiologic implications of inter-hormonal interference in fish: Lessons from the interaction of adrenaline with cortisol and thyroid hormones in climbing perch (Anabas testudineus Bloch). Gen Comp Endocr. 2013; 181:122–29. https://doi.org/10.1016/j.ygcen.2012.11.002
Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. 5th ed. Oxford: Oxford University Press; 2015. https://doi.org/10.1093/acprof:oso/9780198717478.001.0001
Iversen M, Eliassen RA, Finstad B. Potential benefit of clove oil sedation on animal welfare during salmon smolt, Salmo salar L. transport and transfer to sea. Aquac Res. 2009; 40(2):233–41. https://doi.org/10.1111/j.1365-2109.2008.02091.x
Kostić J, Kolarević S, Kračun-Kolarević M, Aborgiba M, Gačić Z, Paunović M et al. The impact of multiple stressors on the biomarkers response in gills and liver of freshwater breams during different seasons. Sci Total Environ. 2017; 601–602:1670–81. https://doi.org/10.1016/j.scitotenv.2017.05.273
Lackner R. “Oxidative stress” in fish by environmental pollutants. In: Braunbeck T, Hinton DE, Streit B, editors. Fish ecotoxicology. Basel: Birkhäuser Basel; 1998. p.203–24.
Loro VL, Murussi C, Menezes C, Leitemperger J, Severo E, Guerra L et al. Spatial and temporal biomarkers responses of Astyanax jacuhiensis (Cope, 1894) (Characiformes: Characidae) from the middle rio Uruguai, Brazil. Neotrop Ichthyol. 2015; 13(3):569–78. https://doi.org/10.1590/1982-0224-20140146
Mallatt J. Fish gill structural changes induced by toxicants and other irritants: A statistical review. Can J Fish Aquat Sci. 1985; 42(4):630–48. https://doi.org/10.1139/f85-083
Manrique WG, Figueiredo MAP, Belo MAA, Martins ML, Moraes FR. Chronic granulomatous inflammation in teleost fish Piaractus mesopotamicus: histopathology model study. Rev Mvz Cordoba. 2017; 22(1):5738–46. https://doi.org/10.21897/rmvz.933
Menezes CC, Loro VL, Fonseca MB, Cattaneo R, Pretto A, Miron DS et al. Oxidative parameters of Rhamdia quelen in response to commercial herbicide containing clomazone and recovery pattern. Pestic Biochem Physiol. 2011; 100(2):145–50. https://doi.org/10.1016/j.pestbp.2011.03.002
Monteiro DA, Almeida JA, Rantin FT, Kalinin AL. Oxidative stress biomarkers in the freshwater characid fish, Brycon cephalus, exposed to organophosphorus insecticide Folisuper 600 (methyl parathion). Comp Biochem Physiol C Toxicol Pharmacol. 2006; 143(2):141–49. https://doi.org/10.1016/j.cbpc.2006.01.004
Moussa MA, Mohamed HRH, Abdel-Khalek AA. The antioxidant defense capacities and histological alterations in the livers and gills of two fish species, Oreochromis niloticus and Clarias gariepinus, as indicative signs of the Batts drain pollution. Environ Sci Pollut R. 2022; 29(47):71731–41. https://doi.org/10.1007/s11356-022-20804-y
Nelson DP, Kiesow LA. Enthalpy of decomposition of hydrogen peroxide by catalase at 25ºC (with molar extinction coefficients of H2O2 solution in the UV). Anal Biochem. 1972; 49(2):474–78. https://doi.org/10.1016/0003-2697(72)90451-4
Olsen YA, Einarsdottir IE, Nilssen KJ. Metomidate anaesthesia in Atlantic salmon, Salmo salar, prevents plasma cortisol increase during stress. Aquaculture. 1995; 134(1–2):155–68. https://doi.org/10.1016/0044-8486(95)00008-P
Parvez S, Raisuddin S. Protein carbonyls: novel biomarkers of exposure to oxidative stress-inducing pesticides in freshwater fish Channa punctata (Bloch). Environ Toxicol Phar. 2005; 20(1):112–17. https://doi.org/10.1016/j.etap.2004.11.002
Paulino MG, Benze TP, Sadauskas-Henrique H, Sakuragui MM, Fernandes JBK, Fernandes MN. The impact of organochlorines and metals on wild fish living in a tropical hydroelectric reservoir: bioaccumulation and histopathological biomarkers. Sci Total Environ. 2014; 497–498:293–306. https://doi.org/10.1016/j.scitotenv.2014.07.122
Pereira DP, Santos DMS, Carvalho Neta AV, Cruz CF, Carvalho Neta RNF. Morphological changes in fish gills of Oreochromis niloticus (Pisces, Cichlidae) as biomarkers of aquatic pollution in the lagoon of Jansen, São Luís, State of Maranhão (Brazil). Biosci J. 2014; 30(4):1213–21.
Pflugrath BD, Brown RS, Carlson TJ. Maximum neutral buoyancy depth of juvenile chinook salmon: Implications for survival during hydroturbine passage. T Am Fish Soc. 2012; 141(2):520–25. https://doi.org/10.1080/00028487.2012.670187
Polakof S, Panserat S, Soengas JL, Moon TW. Glucose metabolism in fish: a review. J Comp Physiol B. 2012; 182:1015–45. https://doi.org/10.1007/s00360-012-0658-7
Reznick AZ, Packer L. Oxidative damage to proteins: Spectrophotometric method for carbonyl assay. Meth Enzymol. 1994; 233:357–63. https://doi.org/10.1016/s0076-6879(94)33041-7
Roberts RJ. Fish pathology. London: Saunders; 2002.
Sayed AH, Younes HaM. Melanomacrophage centers in Clarias gariepinus as an immunological biomarker for toxicity of silver nanoparticles. J Microsc Ultrastruct. 2017; 5(2):97–104.
Sepici-Dinçel A, Benli AÇK, Selvi M, Sarıkaya R, Şahin D, Özkul IA et al. Sublethal cyfluthrin toxicity to carp (Cyprinus carpio L.) fingerlings: Biochemical, hematological, histopathological alterations. Ecotoxicol Environ Saf. 2009; 72(5):1433–39. https://doi.org/10.1016/j.ecoenv.2009.01.008
Silva LGM, Beirão BV, Falcão RC, Castro ALF, Dias EW. It’s a catfish! Novel approaches are needed to study the effects of rapid decompression on benthic species. Mar Freshw Res. 2018; 69(12):1922–33. https://doi.org/10.1071/MF18267
Stephenson JR, Gingerich AJ, Brown RS, Pflugrath BD, Deng Z, Carlson TJ et al. Assessing barotrauma in neutrally and negatively buoyant juvenile salmonids exposed to simulated hydro-turbine passage using a mobile aquatic barotrauma laboratory. Fish Res. 2010; 106(3):271–78. https://doi.org/10.1016/j.fishres.2010.08.006
Streck EL, Czapski GA, Silva CG. Neurodegeneration, mitochondrial dysfunction, and oxidative stress. Oxid Med Cell Longev. 2013; 2013:826046. https://doi.org/10.1155/2013/826046
Strzyżewska-Worotyńska E, Szarek J, Babińska I, Gulda D. Gills as morphological biomarkers in extensive and intensive rainbow trout (Oncorhynchus mykiss Walbaum, 1792) production technologies. Environ Monit Assess. 2017; 189:611. https://doi.org/10.1007/s10661-017-6278-7
Thiel NA, Sachett A, Schneider SE, Garbinato C, Decui L, Eichwald T et al. Exposure to the herbicide 2,4-dichlorophenoxyacetic acid impairs mitochondrial function, oxidative status, and behavior in adult zebrafish. Environ Sci Pollut R. 2020; 27:45874–82. https://doi.org/10.1007/s11356-020-10497-6
Toni C, Loro VL, Santi A, Menezes CC, Cattaneo R, Clasen BE et al. Exposure to tebuconazol in rice field and laboratory conditions induces oxidative stress in carp (Cyprinus carpio). Comp Biochem Physiol C Toxicol Pharmacol. 2011; 153(1):128–32. https://doi.org/10.1016/j.cbpc.2010.09.008
Trushenski J, Schwarz M, Takeuchi R, Delbos B, Sampaio LA. Physiological responses of cobia Rachycentron canadum following exposure to low water and air exposure stress challenges. Aquaculture. 2010; 307(1–2):173–77. http://doi.org/10.1016/j.aquaculture.2010.07.015
Velisek J, Stara A, Li Z-H, Silovska S, Turek J. Comparison of the effects of four anaesthetics on blood biochemical profiles and oxidative stress biomarkers in rainbow trout. Aquaculture. 2011; 310(3–4):369–75. https://doi.org/10.1016/j.aquaculture.2010.11.010
Wandelaar-Bonga SE. The stress response in fish. Physiological Reviews. 1997; 67:591–625. https://doi.org/10.1152/physrev.1997.77.3.591
Wolke RE. Piscine macrophage aggregates: A review. Ann Rev Fish Dis. 1992; 2:91–108. https://doi.org/10.1016/0959-8030(92)90058-6Zaniboni Filho E, Schulz UH. Migratory fishes of the Uruguai River. In: Carolsfeld J, Harvey B, Ross C, Baer A, editors. Migratory fishes of South America: Biology, fisheries and conservation status. Canada: IDRC and The World Bank; 2003. p.156–94.
Authors
![]() Janaína dos Santos Pedron1,
Janaína dos Santos Pedron1, ![]() Jurandir Joaquim Bernardes Junior1
Jurandir Joaquim Bernardes Junior1 ![]() ,
, ![]() Josiane Ribolli1,
Josiane Ribolli1, ![]() Jennyffer Souza2,
Jennyffer Souza2, ![]() Aline Guimarães Pereira2,
Aline Guimarães Pereira2, ![]() Hugo Tolentino2,
Hugo Tolentino2, ![]() Romuel Barros2,
Romuel Barros2, ![]() Carine de Freitas Milarch3,
Carine de Freitas Milarch3, ![]() Rafael José Gonçalves Dias4,
Rafael José Gonçalves Dias4, ![]() Renata Maria Guereschi1,
Renata Maria Guereschi1, ![]() Alexandra Latini2 and
Alexandra Latini2 and ![]() Alex Pires de Oliveira Nuñer1
Alex Pires de Oliveira Nuñer1
[1] Laboratório de Biologia e Cultivo de Peixes de Água Doce, Departamento de Aquicultura, Universidade Federal de Santa Catarina, Rodovia Francisco Thomaz dos Santos, 3532, Armação do Pântano do Sul, 88066-260 Florianópolis, SC, Brazil. (JSP) janainapedron@gmail.com, (JJBJ) jurandirjbjr@gmail.com (corresponding author), (JR) josianeribolli@gmail.com, (RMG) rmguereschi@gmail.com, (APON) alex.nuner@ufsc.br.
[2] Laboratório de Bioenergética e Estresse Oxidativo, Departamento de Bioquímica, Universidade Federal de Santa Catarina, Campus Universitário Reitor João David Ferreira Lima, s/nº, Trindade, 88040-900 Florianópolis, SC, Brazil. (JS) jennyffer_souzaa@hotmail.com, (AGP) alinegp77@gmail.com, (HT) hugopharmacist@gmail.com, (RB) roxdim@gmail.com, (AL) alatinilabox@gmail.com.
[3] Faculdade IELUSC, Rua Princesa Isabel, 438, Centro, 89201-270 Joinville, SC, Brazil. (CFM) carinedefs@yahoo.com.br.
[4] Engie Brasil Energia, Rua Paschoal Apóstolo Pítsica, 5064, Agronômica, 88025-255 Florianópolis, SC, Brazil. (RJGD) rafael.dias@engie.com.
Authors’ Contribution 

Janaína dos Santos Pedron: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-original draft, Writing-review and editing.
Jurandir Joaquim Bernardes Junior: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing-original draft, Writing-review and editing.
Josiane Ribolli: Methodology, Validation, Writing-original draft, Writing-review and editing.
Jennyffer Souza: Formal analysis, Investigation, Validation, Writing-review and editing.
Aline Guimarães Pereira: Formal analysis, Investigation, Validation, Writing-review and editing.
Hugo Tolentino: Formal analysis, Investigation, Validation, Writing-review and editing.
Romuel Barros: Formal analysis, Investigation, Validation, Writing-review and editing.
Carine de Freitas Milarch: Formal analysis, Investigation, Validation, Writing-review and editing.
Rafael José Gonçalves Dias: Funding acquisition, Project administration, Resources, Writing-review and editing.
Renata Maria Guereschi: Investigation, Project administration, Resources, Writing-original draft, Writing-review and editing.
Alexandra Latini: Conceptualization, Methodology, Validation, Writing-review and editing.
Alex Pires de Oliveira Nuñer: Funding acquisition, Investigation, Project administration, Supervision, Validation, Writing-review and editing.
Ethical Statement
Collecting permits were issued by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) through license number 70928-1 and subsequent renovations. All procedures complied with the Ethics Committee on the Use of Animals from the Universidade Federal de Santa Catarina (protocol number 7124290719).
Competing Interests
The author declares no competing interests.
How to cite this article
Pedron JS, Bernardes Junior JJ, Ribolli J, Souza J, Pereira AL, Tolentino H, Barros R, Milarch CF, Dias RJG, Guereschi RM, Latini A, Nuñer APO. Fish injuries resulting from transient operating conditions in a Brazilian hydropower plant: morphological, physiological and biochemical evaluation in Pimelodus maculatus (Siluriformes: Pimelodidae). Neotrop Ichthyol. 2023; 21(3):e220104. https://doi.org/10.1590/1982-0224-2022-0104
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted September 4, 2023 by Bernardo Baldisserotto
Accepted September 4, 2023 by Bernardo Baldisserotto
![]() Submitted November 3, 2022
Submitted November 3, 2022
![]() Epub October 20, 2023
Epub October 20, 2023