![]() Maura Palacios1,
Maura Palacios1, ![]() Alfonso A. González-Díaz2,
Alfonso A. González-Díaz2, ![]() Lenin Arias Rodriguez3,
Lenin Arias Rodriguez3, ![]() Mariana Mateos1,
Mariana Mateos1, ![]() Rocío Rodiles-Hernández2,
Rocío Rodiles-Hernández2, ![]() Michael Tobler4 and
Michael Tobler4 and ![]() Gary Voelker1
Gary Voelker1 ![]()
PDF: EN XML: EN | Supplementary: S1 S2 S3 S4 | Cite this article
Abstract
Mexico is a megadiverse region with a complex geological history, but it remains unclear to what extent the distribution of freshwater fish has been influenced by geographic barriers. This study examines the population level genetic divergence and phylogenetic relationships of species in the shortfin group of the subgenus Mollienesia (genus Poecilia), a group of live-bearing fishes that are widely distributed across Mexico, with sampling at a small geographic scale. Samples from over 50 locations were analyzed for six species by using phylogenetic and haplotype network approaches to assess genetic diversity across geographic ranges and to refine the distributions of species in this group. The results indicate that Mexican species have diversified following multiple, independent invasions from Middle America. Two species found north of the Trans-Mexican Volcanic Belt (TMVB) and one transversal species exhibited weak phylogenetic structure, likely due to the lack of physiographic barriers, recent colonization, and high dispersal rates among regions. In contrast, three species found south of the TMVB exhibited strong phylogenetic structure, reflecting a longer presence in the area and multiple physiographic barriers that isolated populations. This study identified mechanisms driving divergence and speciation, expanded the known range of several species, and resolved taxonomic uncertainties of populations.
Keywords: Diversification, Fishes, Phylogenetics, Phylogeography, Trans-Mexican Volcanic Belt.
México es una región megadiversa con una historia geológica compleja, pero se desconoce el nivel de influencia de las barreras geográficas sobre las distribuciones de los peces dulceacuícolas. Este estudio examina las relaciones filogenéticas, a escala geográfica pequeña, de las especies del grupo de aletas cortas del subgénero Mollienesia (género Poecilia), un grupo de peces vivíparos ampliamente distribuidos en México. Se analizaron muestras de seis especies en más de 50 localidades, utilizando métodos filogenéticos y de redes de haplotipos, para evaluar la diversidad genética y precisar las distribuciones de especies en este grupo. Los resultados indican que las especies mexicanas se han diversificado a partir de múltiples invasiones independientes desde Mesoamérica. Se detectó estructura filogenética débil en dos especies distribuidas al norte del Eje Neovolcánico y una especie que atraviesa el Eje Neovolcánico, posiblemente debido a la ausencia de barreras fisiográficas, colonización reciente y altas tasas de dispersión entre regiones. En contraste, se detectaron niveles altos de estructura filogenética en tres especies distribuidas del Eje Neovolcánico, lo que refleja una presencia más prolongada en el área y la existencia de múltiples barreras fisiográficas que aislaron a las poblaciones. Este estudio identificó mecanismos que promueven la divergencia y la especiación, expandió el rango conocido de varias especies y resolvió incertidumbres taxonómicas de algunas poblaciones.
Palabras clave: Cinturón Volcánico Transmexicano, Diversificación, Filogenética, Filogeografía, Peces.
Introduction
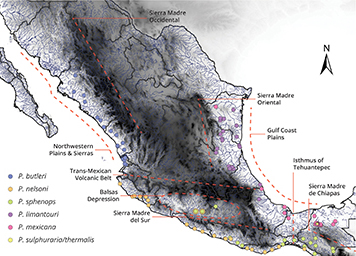
Mexico’s complex geological history has led to diverse ecosystems, making the region rich in flora and fauna. The historical geological activity is reflected in a landscape dominated by mountain chains that give rise to isolated hydrological units and regions with starkly different ecological conditions (Domínguez-Domínguez et al., 2006). In the north of the country, the mountain ranges of the Sierra Madre Oriental and the Sierra Madre Occidental extend along the eastern and western coast, respectively, and in the south, they join the Trans-Mexican Volcanic Belt (TMVB; Fig. 1), which transects the country at a latitude of about 20 ºN and marks the southern edge of the North American plate (Ferrari et al., 1999). In the southern region of Mexico, the Sierra Madre del Sur and the Sierra Madre de Chiapas are separated by the Isthmus of Tehuantepec (Fig. 1). These mountain ranges produce a wide range of geographical landforms, including high mountains with permanent snow cover, highland plateaus, deep valleys, and coastal plains (Contreras-Balderas et al., 2008). Mexico is also unique in encompassing organisms from both the Neartic (North America south to the Mexican plateau) and Neotropical (Mexican central coasts to South America) biogeographic zones (Morrone, Márquez, 2001). The combination of the geological diversity and the interface of different biogeographic zones make Mexico a megadiverse region in terms of ecological habitats, species diversity, and levels of endemism (Conabio, 1998; Metcalfe et al., 2000; Hufnagel, Mics, 2021).
FIGURE 1| Sampling localities of species in the subgenus Mollienesia in Mexico and the main physiographic barriers throughout the country. The main physiographic barriers in Mexico from north to south are Northwestern Plains and Sierras, Sierra Madre Occidental, Sierra Madre Oriental, Gulf Coast Plain, Trans Mexican Volcanic Belt, Balsas Depression, Sierra Madre del Sur, Isthmus of Tehuantepec, and Sierra Madre de Chiapas.
Mexico’s high levels of biodiversity are also reflected in its freshwater ichthyofauna, which is comprised of 50 families, 155 genera, and 536 species (Miller et al., 2005; Mejía et al., 2012; Lyons et al., 2020), with 271 species being endemic to the country (Contreras-Balderas et al., 2008) and new species continually being described (Langerhans et al., 2012; De la Maza-Benignos et al., 2015; Ornelas-García et al., 2015; Domínguez-Domínguez et al., 2016; Matamoros et al., 2018; Conway et al., 2019). Previous phylogeographic studies have shown that some Mexican fishes have diversified, and their distribution patterns are in accordance with ancient geological events rather than modern hydrographic patterns (Mateos et al., 2002; Strecker et al., 2004; Huidobro et al., 2006; Agorreta et al., 2013; Zúñiga-Vega et al., 2014). However, population-level studies are rarely conducted, and there is a poor understanding of the microevolutionary dynamics that shape fish distributions and population structures in Mexico.
The most species-rich fish group in Mexico is the family Poeciliidae (Contreras-MacBeath et al., 2014), which includes small cyprinodontiform fishes inhabiting fresh, brackish, and coastal waters (Reis et al., 2003; Bragança et al., 2018). This group is characterized by a set of unique traits: adult males possess a modified anal fin, with rays 3, 4, and 5 forming a gonopodium for sperm transfer, and reproduction occurs via internal fertilization and matrotrophic or lecithotrophic embryonic development (Rosen, Bailey, 1963; Parenti, 1981; Greven, 2011). Recent studies have shed light on the systematics, phylogeography, and biodiversity of this family in Mexico and other areas of the Neotropics (Hrbek et al., 2007; Meredith et al., 2011; Agorreta et al., 2013; Reznick et al., 2017). Among the most diverse groups of poeciliids in Mexico is the subgenus Mollienesia LeSueur, 1821 (genus Poecilia Bloch & Schneider, 1801; see Tab. 1), whichis represented by 15 species (Palacios et al., 2016). Mollienesia species are found in a variety of habitats, including streams and rivers, coastal marshes, volcanic lakes, cave systems, and hydrogen-sulfide-rich springs (Miller, 1975; Miller et al., 2005; Tobler, Plath, 2011; Palacios et al., 2016). Despite the wide distribution of these species in Mexico, the levels of intra-specific diversity and the distributional ranges of several species in this group remain poorly described (Bussing, 1976; Miller, 1983; Miller et al., 2005). Recent molecular studies of Mollienesia across its distribution in North, Central, and northern South America have revealed three species complexes (Poecilia latipinna LeSueur, 1821, P. sphenops Valenciennes, 1846, and P. mexicana Steindachner, 1863; Breden et al., 1999), a number of putatively undescribed species (Alda et al., 2013; Bagley et al., 2015; Ho et al., 2016; Palacios et al., 2016), and a complex history of colonization from nuclear Central America into North and South America, and back (Ho et al., 2016). In Mexico, some species are widespread and occur on both the Atlantic and Pacific slopes (e.g., P. sphenops and P. mexicana; Alda et al., 2013; Bagley et al., 2015; Ho et al., 2016; Palacios et al., 2016), while others are endemic to a few locations (P. catemaconis Miller, 1975, P. chica Miller, 1975, P. sulphuraria Álvarez, 1948, and P. thermalis Miller, 1975; Palacios et al., 2013, 2016). At a finer scale, previous studies have shown genetic differentiation among closely related lineages along environmental gradients (in P. mexicana, P. sulphuraria, and P. thermalis; Palacios et al., 2013) as well as diversification across physiographic barriers (in P. butleri Jordan, 1889, and P. nelsoni, Meek, 1904; Zúñiga-Vega et al., 2014). The variation of distribution patterns among species makes this subgenus a promising group to investigate phylogeographic patterns and microevolutionary processes. This study specifically applied phylogenetic and haplotype network analyses of seven species in the subgenus Mollienesia (P. butleri, P. limantouri Jordan & Snyder, 1899, P. mexicana, P. nelsoni, P. sphenops, P. sulphuraria,and P. thermalis) from 66 locations across Mexico to (1) determine the number of colonization events of Mexico from nuclear Central America, (2) assess patterns of phylogeography and haplotype diversity in different species, and (3) contribute to a better understanding of the distribution of species in this group.
TABLE 1 | List of species from the Poecilia sphenops and P. mexicana species complexes present in Mexico. The table provides information about the slope on which a species occurs, a broad description of the distribution, the endemism status in Mexico, and phylogeographic breaks uncovered in this study. Asterisks indicate the inclusion of a species in phylogenetic analyses; crosses indicate focal species used for phylogeographic analyses based on Cyt b.
Species | Slope | Distribution | Endemic | Phylogeographic Break Cyt b |
P. sphenops complex | ||||
Poecilia catemaconis Miller, 1975* | Atlantic | Laguna Catemaco and surrounding
rivers, Veracruz | X | Sierra de Tuxtlas |
Poecilia chica Miller, 1975* | Pacific | Three small rivers in Jalisco | X | Jalisco block |
Poecilia maylandi Meyer, 1983 | Atlantic | Rio Balsas, Guerrero | X | |
Poecilia sphenops Valenciennes, 1846*? | Atlantic,
Pacific | Tamaulipas, Veracruz,
Guerrero, Oaxaca, Chiapas, and
Guatemala | ||
P. mexicana complex | ||||
Poecilia butleri Jordan, 1889*? | Pacific | Rio Fuerte,
Sonora, Sinaloa, and Nayarit | X | Trans Mexican Volcanic Belt |
Poecilia mexicana Steindachner,
1863*? | Atlantic | Nuevo Leon to Panama | ||
Poecilia limantouri Jordan, 1899*? | Atlantic | Rio Grande to
N. Veracruz | X | |
P. nelsoni (Meek, 1904)
*? | Pacific | Colima to S.
Guatemala | X | Trans Mexican Volcanic Belt, Balsas Depression |
Poecilia orri Fowler, 1943* | Atlantic | Yucatan, Quintana Roo to
Honduras | ||
Poecilia sulphuraria (Álvarez, 1948)*? | Atlantic | Sulfide springs in Tabasco and Chiapas | X | |
Poecilia thermalis Steindachner, 1863*? | Atlantic | Sulfide springs in
Chiapas | X |
Material and methods
Specimen acquisition. We sampled specimens of the subgenus Mollienesia throughout the subgenus’s distribution in Mexico (Fig. 1; see Tab. S1 for locality data). Fish were captured using electrofishing, seines, cast and dip nets. Immediately after capture, some sampled fish were euthanized with buffered MS222; others were sampled and released (per permit restrictions). Regardless of sampling method, fin clips (right pectoral fin) were preserved in 95% ethanol for molecular analyses, and specimens were fixed in a 10% formaldehyde solution following protocols approved by the Texas A&M University and Oklahoma State University Committees on Use and Care of Animals (IACUC 2011-118 & ACUP AS10-15). Ethanol preserved tissues, DNA extractions, and formalin fixed specimens are housed in the Biodiversity, Research and Teaching Collections of the Department of Wildlife & Fisheries Sciences at Texas A&M University, College Station, Texas, USA, and the El Colegio de la Frontera Sur, San Cristóbal, Chiapas, Mexico. Voucher numbers for new specimens are provided in Tab. S1, where possible; many of our samples were fin clips (per permit restrictions) and some were used for life history dissections, and as such were not suitable as voucher specimens.
Samples collected for this study were supplemented with sequence data obtained from GenBank to examine the relationship of Mollienesia species in Mexico to other members of the genus from North and Central America. We included sequence data for multiple genes (see below) for 18 out of 26 recognized species in the subgenus Mollienesia (Mateos, 2005; Alda et al., 2013; Ho et al., 2016; Palacios et al., 2016): P. kykesis, P. latipinna, P. latipunctata Meek, 1904, and P. velifera (Regan, 1914) (members of the sailfin molly clade); P. catemaconis, P. chica, P. marcellinoi, and P. sphenops (members of the P. sphenops species complex of the shortfin molly clade), as well as P. butleri, P. gillii (Kner, 1863), P. hondurensis Poeser, 2011, P. mexicana, P. nelsoni, P. orri Fowler, 1943, P. petenensis Günther, 1866 (sometimes referred to as P. gracilis), P. salvatoris Regan, 1907, P. sulphuraria, and P. thermalis (members of the P. mexicana species complex of the shortfin molly clade). In addition, we supplemented the cytochrome b dataset generated for our focal species with additional sequences. This expanded the distributional coverage for P. butleri and P. nelsoni (Zúñiga-Vega et al., 2014), spanning their entirerangesalong the Pacific coast of Mexico. The distribution coverage was also extended north of our sampling for P. limantouri (Stöck et al., 2010) and south of our sampling in P. mexicana (Tobler et al., 2011).
Molecular analyses. Total genomic DNA was extracted from ethanol-preserved fin clips with the DNeasy Blood & Tissue Kit (Qiagen, Inc., Valencia, CA) following the manufacturer’s protocol. The samples were amplified for several presumably neutral genes commonly used for phylogenetic reconstruction in fishes, including poeciliids (Hrbek et al., 2007; Meredith et al., 2010, 2011). Focal genes included the mitochondrial cytochrome b gene (cyt b, 1,140 base pairs) with LA and HA primers (Schmidt et al., 1998), the mitochondrial gene NADH subunit 2 (ND2, 1,047 bp) with ND2B-L (Broughton, Gold 2000) and ASN (Kocher et al., 1995) primers, and a nuclear gene, exon 3 of recombination activating gene-1 (RAG1, 1,561 bp) with the primers L2492 RAG1 and H4054 RAG1 (Hrbek et al., 2007). PCR protocols followed Palacios et al. (2016), and PCR products were purified with Exosap-IT enzyme reaction (GE Healthcare Bio-Sciences Corp., Piscataway, NT), directly sequenced with a dye-labeled terminator kit (Big Dye Terminator version 3.1, Applied Biosystems, Foster City, CA), and run on an ABI automated sequencer (Applied Biosystems, Foster City, CA). Sequence electrophenograms were edited with Sequencher version 4.8 (Gene Codes) and aligned with MAFFT v. 6.0 (Katoh, Toh, 2008).
Alignment and phylogenetic analyses. The data were partitioned into three datasets: (1) a concatenated dataset (cyt b, ND2, and RAG1,3748 base pairs), (2) the nuclear gene alone (RAG1 1561 bp), and (3) cyt b alone(1140 bp). A strictly mitochondrial dataset (cyt b and ND2) was not analyzed because samples missing sequences for ND2 reduced the size of the dataset. All datasets were analyzed separately using FaBox online (Villesen, 2007) to determine the total number of haplotypes. The cyt b dataset was reduced from 953 sequences to 475 (139 haplotypes for the 6 species of interest). We used Partition Finder (Lanfear et al., 2012)to determine thebest partition scheme and the most likely model of DNA substitution among 24 candidate models on a fixed BioNJ-JC tree based on the Bayesian information criterion (BIC; Tab. S2), separately for each gene. Phylogenetic analyses for the concatenated and the cyt b datasets were conducted with a maximum likelihood (ML) approach as implemented in RAxML GUI v. 1.0 (Stamatakis, 2006, 2008), with 500 Rapid Bootstrap searches followed by an ML search. The complex general time reversible (GTR) + Γ(gamma distribution for rate variation among sites) model was chosen for each partition. The bootstrap trees were summarized with a Sumtrees script that used a 50% percent majority rule consensus parameter in DendroPy v. 3.10.1 (Sukumaran, Holder, 2010), and the final tree was rooted and visualized in FigTree v. 1.4.2 (http://tree.bio.ed.ac.uk/software/figtree/). Bayesian analyses were conducted in MrBayes v. 3.2.1 (Ronquist, Huelsenbeck, 2003; Ronquist et al., 2012), implementing two runs with four chains under default parameters for 50 million generations, sampling every 10 generations. A 25% burn-in was applied and stable posterior probability values examined in Tracer v. 1.5 (Rambaut et al., 2018). Pairwise genetic distances were calculated under the Kimura-2 parameter model in MEGA v. 7 (Kumar et al., 2016) with pairwise deletion for missing data.
A haplotype network was generated with a statistical parsimony analysis of the cyt b gene dataset for the six focal species in TCS v. 1.2.1 (Clement et al., 2000), as implemented in the Popart program (Leigh, Bryant, 2015). This approach calculated the number of significant substitutions connecting haplotypes in a network by applying the algorithm developed by Templeton et al. (1992). A connection limit was set to the default of 95% to generate the haplotype networks between closely related sequences.
Results
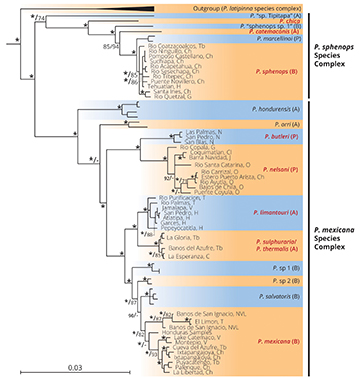
Phylogenetic relationships of Mexican mollies to other species in the subgenus Mollienesia. To determine the number of colonization events from nuclear Central America into Mexico, we generated a phylogenetic tree with 18 of the 26 recognized species in the subgenus Mollienesia, and overall, there was high concordance between maximum-likelihood and Bayesian analyses.The phylogeny of the concatenated dataset showed moderate to high support for the two clades representing the P. sphenops and P. mexicana species complexes (Fig. 2). Most of the phylogenetic signal came from the mitochondrial genes. Analysis of RAG1 alone resulted in poorly supported phylogenetic relationships due to the low levels of sequence variation in our samples (1–2 base pairs across the entire gene; results not shown). The genetic distances across the two complexes ranged from 6.9–10.5%, while they ranged from 1.0–8.2% within the P. sphenops species complex and 0.9–6.0% within the P. mexicana complex (Tab. S3).
FIGURE 2| Bayesian tree from the MrBayes partitioned analysis of Poecilia spp. for two mitochondrial genes (Cyt b and ND2, 2187 base pairs) and one nuclear (RAG1, 1561 base pairs) rooted with other poeciliid outgroups. Species names in black pertain to species outside of Mexico and species names in red are species found within Mexico. Species labels represent slope: (A) = Atlantic, (P) = Pacific, and (B) = Bi-coastal. Nodal support shown (left to right; respectively): Bayesian Posterior Probabilities followed by RAxML bootstrap support values. Asterisks denote nodal support of 95% or above for the two methods, and a single asterisk at a node indicates support values of 95% or above for both methods. Nodes with no values present either had low values or were of little interest for this study. The capital letters at the end of each sample represents the state of origin in Mexico, from North to South: T = Tamaulipas, NVL = Nuevo León, V = Veracruz, H = Hidalgo, M = Michoacan, G = Guerrero, O = Oaxaca, Tb = Tabasco, C = Chiapas.
The P. sphenops species complex was strongly (Bayesian, > 95%) or moderately (maximum-likelihood, 74%) supported and included three species from Mexico (P. sphenops and the two endemic species P. chica and P. catemaconis), which were interspersed among three species from Middle America (P. marcellinoi and two putatively undescribed species, P. “sp. Tipitapa” and P. “sphenops sp1”; Bagley et al., 2015) (Fig. 2). Within P. sphenops, our phylogeny did not provide support for the relationships among the sampled locations that cover both the Atlantic and Pacific versants of Mexico.
The P. mexicana species complex was strongly supported (>95% with both methods) and included five Mexican and five Middle American species (Fig. 2). Poecilia hondurensis and P. orri from Middle America represented the most basal divergences in the complex (Fig. 2). The next divergence in the complex was strongly supported and included two species from Mexico’s Pacific slope, P. butleri and P. nelsoni. Next, our results recovered a strongly supported P. limantouri cladeon the Atlantic slope of Mexico (which lacks resolution among different sampling sites) as sister to the sulfide spring endemics P. sulphuraria and P. thermalis (Fig. 2). The P. limantouri/P. sulphuraria–P. thermalis clade is sister to a clade comprised of two putatively undescribed lineages from Middle America (P. “sp 1” and P. “sp 2”; Palacios et al., 2016), P. salvatoris (also from Middle America), and the strongly/moderately supported P. mexicana clade. Poecilia mexicana is split into three clades. Two of these clades are of samples from Mexico, with one strongly supported clade comprised of the most northern sampling locations, from the Trans-Mexican Volcanic Belt north to the state of Nuevo León. The other clade includes the remaining samples, which come from south of the Trans-Mexican Volcanic Belt and span both sides of the Isthmus of Tehuantepec (Fig. 2). The two Mexican clades are not sisters, as a lineage consisting of samples from Honduras is more closely related to the southern clade.
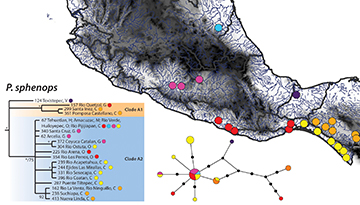
Phylogeography of Poecilia sphenops. To assess patterns of phylogeography and haplotype diversity within different species, we generated haplotype networks and observed potential patterns of differentiation across known geographic barriers. The widespread species P. sphenops is found across the Atlantic and Pacific versants in Mexico, and genetic distances within this species ranged from 0 to 1.0% (Tab. S4). Our analyses revealed three divergent groups. The first of these (Texistepec) is represented by a single haplotype from the Rio Coatzacoalcos of the southern Atlantic coast (state of Veracruz), just west of the Isthmus of Tehuantepec (Fig. 3). The second group (clade A1) was composed of one Pacific slope haplotype from the Guerrero coast west of the Isthmus of Tehuantepec, and two haplotypes from the Atlantic versant in Chiapas west of the Isthmus of Tehuantepec (Fig. 3). The main P. sphenops clade (clade A2 in Fig. 3) included one widely distributed haplotype, found along the Atlantic coast north of the Trans-Mexican Volcanic Belt, in the Balsas depression, and along the Pacific coast east and west of the Isthmus of Tehuantepec. Clade A2 also exhibited three strongly supported sub-clades, the first consisting of one haplotype from the Balsas depression and one from a Pacific location at the Isthmus of Tehuantepec (Rio Ostuta on the Oaxacan coast). The second sub-clade consisted of four Pacific haplotypes from the Chiapas coast east of the Isthmus of Tehuantepec, and the third sub-clade was comprised of three haplotypes from the Atlantic versant east of the Isthmus of Tehuantepec in Chiapas (Fig. 3). Additionally, we found five other P. sphenops haplotypes with unresolved relationships; these samples were geographically dispersed across the Balsas depression and the Pacific versant on both sides of the Isthmus of Tehuantepec (Fig. 3).
FIGURE 3| Cytochrome b (1,140 bp) mitochondrial gene Bayesian phylogeny, parsimony haplotype network, and sampling distribution of Poecilia sphenops (aqua-Atlantic North of the Trans Mexican Volcanic Belt, pink- Balsas River Drainage, purple- Atlantic North of the Isthmus of Tehuantepec, red- Pacific North of the Isthmus of Tehuantepec, orange- Atlantic South of the Isthmus of Tehuantepec, and yellow- Pacific South of the Trans Mexican Volcanic Belt) across geographic barriers along the coasts of Mexico. The phylogeny has Bayesian posterior values followed by bootstrap values with asterisks representing support of 95% or above. The Parsimony network values correspond to the haplotype values and are colored according by geographic locations separated by barriers; circle sizes correspond to the number of individuals with that haplotype (larger circles reflect more individuals), and black circles indicate unsampled haplotypes. The capital letters at the end of each sample represents the state of origin in Mexico, from North to South: V = Veracruz, H = Hidalgo, M = Michoacan, G = Guerrero, O = Oaxaca, C = Chiapas.
The haplotype network of P. sphenops indicated that haplotypes were separated between 1–10 mutational steps. The most common haplotype included samples from the Pacific coast, including both sides of the Isthmus of Tehuantepec and the Balsas depression, and one sample from Atlantic versant north of the Trans-Mexican Volcanic Belt. The most divergent haplotypes in P. sphenops were found along the Pacific coast west of the Isthmus of Tehuantepec and in rivers of the Atlantic versant, both west (Rio Coatzocoalcos) and east (Chiapas highlands) of the Isthmus (Fig. 3). The remaining haplotypes, with two exceptions, were all found on the Pacific versant, including the Balsas depression. Overall, the lack of clear phylogeographic patterns and haplotype sharing across major biogeographic boundaries indicate low levels of differentiation within P. sphenops.
Phylogeography of Poecilia butleri and P. nelsoni. The sister taxa P. butleri and P. nelsoni are found on either side of the Trans-Mexican Volcanic Belt. The genetic distances within both P. butleri and P. nelsoni ranged from 0–2.0%, and the two species differed by 4.6% (Tab. S4).
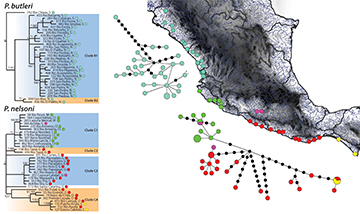
Poecilia butleri was restricted to the Pacific versant north of the Trans-Mexican Volcanic Belt. The phylogenetic structure for this speciesshowed an initial divergence represented by a single haplotype from our most northern sample in Sinaloa (Rio Choix). The next divergence (clade B2 in Fig. 4) included a clade composed of two haplotypes from the southernmost portion of the P. butleri range (San Blas and Rio El Palillo in southern Nayarit). Lastly, the strongly supported clade B1 contained the remaining haplotypes, which formed a five lineage polytomy with no obvious geographic structuring (Fig. 4). Within clade B1, two lineages represented single haplotypes, one from the northern (Sinaloa) and one from the southern portion (Nayarit) of P. butleri’s range. Two other lineages represented moderately to strongly supported sub-clades comprising six and three haplotypes from Sinaloa. The fifth lineage was a large weakly supported clade of individuals collected from both the northern and southern portions of the range. The haplotype network of P. butleri had a maximum of 17 mutational steps between haplotypes, with most haplotypes differing between 1–6 steps (Fig. 4). The most divergent haplotypes were from Rio Choix (192), the most northern sampling locality in Sinaloa, and from El Palillo (456), San Blas (399), and Acaponeta (408), all southern localities in Nayarit.
FIGURE 4| Cytochrome b (1,140 bp) mitochondrial gene Bayesian phylogeny, parsimony haplotype network, and sampling distribution of the Pacific sister taxa Poecilia butleri (blue-North of the Trans Mexican Volcanic Belt) and P. nelsoni (lime green-South of the Trans Mexican Volcanic Belt, pink- Balsas River Drainage, red- North of the Isthmus of Tehuantepec, and yellow- South of the Trans Mexican Volcanic Belt) across geographic barriers along the coast of Mexico. The phylogeny has Bayesian posterior values followed by bootstrap values with asterisks representing support of 95% or above. The Parsimony network values correspond to the haplotype values and are colored according by geographic locations separated by barriers; circle sizes correspond to the number of individuals with that haplotype (larger circles reflect more individuals), and black cirles indicate unsampled haplotypes. The capital letters at the end of each sample represents the state of origin in Mexico, from North to South: S = Sinaloa, N = Nayarit, J = Jalisco, Cl = Colima, M = Michoacan, G = Guerrero, O = Oaxaca, C = Chiapas.
Poecilia nelsoni is distributed on the Pacific versant south of the Trans-Mexican Volcanic belt, spanning both sides of the Isthmus of Tehuantepec. Although all P. nelsoni samples were recovered as monophyletic, the basal node consisted of a polytomy of six lineages (Fig. 4). Two lineages consisting of a single haplotype, a third lineage representing two haplotypes (clade C2), and a fourth lineage representing many haplotypes (clade C3) were mostly collected west of the Isthmus of Tehuantepec in Guerrero, with one individual from the Balsas depression (Fig. 4). Of the two remaining P. nelsoni lineages, one (clade C1) was composed almost entirely of individuals collected just south of the Trans-Mexican Volcanic Belt, including sites in Jalisco, Colima, and Michoacan, and one individual from the Balsas depression. Finally, clade C4 was composed of individuals collected west of the Isthmus of Tehuantepec in Oaxaca and individuals collected south of the Isthmus of Tehuantepec in Chiapas. The haplotype network for P. nelsoni underscored thephylogenetic results, in that multiple divergent haplotype groups were found at the northern extent of P. nelsoni’s range, and along the coast of Guerrero and Oaxaca. In addition, haplotypes were shared across the Isthmus of Tehuantepec, and the Balsas River samples were more closely associated with samples from other regions than with each other.
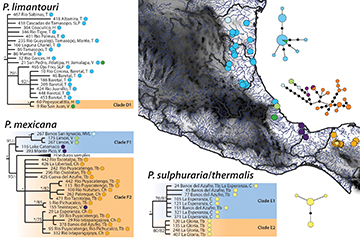
Phylogeography of Poecilia mexicana sensu lato. The closely related species P. limantouri, P. thermalis and P. sulphuraria, and P. mexicana formed, along with several Middle American species, the last major clade in the phylogeny (Figs. 2, 5). Among these species, the hydrogen-sulfide spring endemics P. thermalis and P. sulphuraria were most genetically similar (0–0.5% sequence divergence within each species), whereas P. mexicana (within species range: 0–2.0%) and P. limantouri (within species range: 0–1.7%) were more genetically diverse (Tab. S4). Poecilia limantouri was predominantly found north of the TMVB, but with a small range (represented by two sampling points) just south of this geographic barrier. Most haplotypes of P. limantouri across the distributional range did not exhibit any phylogenetic clustering or lacked support from the maximum likelihood analyses. The single sub-clade with strong support (D1) included two haplotypes: one from the state of Hidalgo and the other from the southern sampling points in the state of Veracruz (Fig. 5). The haplotype network exhibited between 1–6 mutational steps between haplotypes.
FIGURE 5| Cytochrome b (1,140 bp) mitochondrial gene Bayesian phylogeny, parsimony haplotype network, and sampling distribution of Atlantic taxa Poecilia limantouri (Turqouise-North of the Trans Mexican Volcanic Belt, forest green-South of the Trans Mexican Belt), P. sulphuraria/P. thermalis (yellow-South of the Isthmus of Tehuantepec), and P. mexicana (baby blue-North of the Trans Mexican Volcanic Belt, light green-South of the Trans Mexican Volcanic Belt, purple-North of the Isthmus of Tehuantepec, orange-South of the Isthmus of Tehuantepec) across geographic barriers along the Atlantic coast of Mexico. The phylogeny has Bayesian posterior values followed by bootstrap values with asterisks representing support of 95% or above. The Parsimony network values correspond to the haplotype values and are colored according by geographic locations separated by barriers; circle sizes correspond to the number of individuals with that haplotype (larger circles reflect more individuals), and black circles indicate unsampled haplotypes. The capital letters at the end of each sample represents the state of origin in Mexico, from North to South: NVL = Nuevo Leon, T = Tamaulipas, V = Veracruz, H = Hidalgo, Tb = Tabasco, C = Chiapas.
The species P. sulphuraria and P. thermalis from the states of Tabasco and Chiapas were recovered in two sub-clades with moderate support. The first (E1) included individuals of P. sulphuraria from the Baños del Azufre site and of P. thermalis from the La Esperanza spring complex. The second sub-clade (E2) was formed exclusively of haplotypes of P. sulphuraria from the La Gloria spring complex. The haplotype network showed almost no variation in these taxa, and interestingly the Baños del Azufre population of P. sulphuraria clustered more closely with P. thermalis (clade E1) than the La Gloria population of P. sulphuraria (clade E2; Fig. 5).
Finally, phylogenetic analyses recovered a monophyletic P. mexicana, with further phylogenetic structuring of varying support (Fig. 5). One sub-clade (0.91 Posterior Probability) was comprised of individuals from northern Mexico (in Nuevo León), individuals collected just south of TMVB, and two sites just west of the Isthmus of Tehuantepec in the Los Tuxlas region (clade F1 in Fig. 5). Another sub-clade represented individuals collected from the Atlantic versant of Honduras, and this sub-clade was recovered as sister to the last sub-clade (Clade F2), comprising individuals collected from Tabasco and Chiapas, except for one individual collected in Veracruz (Fig. 5). The haplotype network showed a range of variation with 1–15 mutational steps between haplotypes.
Our analyses indicated extensions of the documented ranges for P. mexicana and P. limantouri, which were previously believed to either occur only south and only north of the TMVB, respectively (Miller et al., 2005). Both species’ distributions appear to span the TMVB (Fig. 5), and the northern haplotypes in P. mexicana represent a highly divergent lineage that may represent a yet unrecognized species that also differs in a series of morphological traits from P. mexicana south of the TMVB (M. Tobler, unpublished data). We also found the southern range of Poecilia nelsoni in Mexico to extend into southern Oaxaca and Chiapas.
Discussion
The aim of this study was to provide a better understanding of the patterns of divergence and speciation across the geologically complex Mexican landscape in freshwater fishes of the subgenus Mollienesia. The overall phylogenetic patterns observed among species (Fig. 2) revealed multiple colonization events from lower Central America into Mexico and strong geographic structuring by physiographic barriers and by river basins. The exception was the widely distributed P. sphenops, which was distributed across all geographic barriers discussed here (Figs. 1, 3), thereby overlapping with the distribution of other species in the subgenus Mollienesia. In contrast, P. nelsoni and P. mexicana showed signals of population structure but not in relation to the Isthmus of Tehuantepec (Figs. 4, 5). This phylogeographic structure over small spatial scales, albeit weak at present, is likely the result of limited genetic exchange across smaller geologic barriers and disconnected river basins. Our results indicate that different species in the subgenus Mollienesia-despite their phenotypic similarities-vary in their vagility, and mechanistic studies are needed to understand how morphological, physiological, and behavioral differences among species may contribute to variation in dispersal.
Phylogenetic patterns of Mexican species. The phylogeny of the subgenus Mollienesia shows an interesting pattern with respect to the Mexican species in the P. sphenops and P. mexicana species complexes. Most Mexican species in the subgenus were directly derived from Central American lineages, demonstrating the complexity of the evolution of species in this group. In the P. sphenops species complex, there were three potential invasions that led to the speciation of Mexican species (P. catemaconis, P. chica, and P. sphenops). The differentiation of P. catemaconis on the Atlantic slope of Mexico from the ancestral population was a result of the geographic isolation as Lake Catemaco became geologically separated from other hydrological units by the Sierra de los Tuxtlas (Palacios et al., 2016). The Pacific P. chica possibly also evolved due to volcanic activity isolating the Jalisco block between the Pliocene and Quaternary (Rosas-Elguera et al., 1996).
In the P. mexicana species complex, Mexican species also showed multiple independent invasions from Central America. The Pacific slope species, P. butleri and P. nelsoni, evolved in Mexico due to the uplift of the Trans Mexican Volcanic Belt, isolating populations north and south of this barrier (Mateos, 2005; Zúñiga-Vega et al., 2014). On the Atlantic versant, the evolution of species was a combination of vicariance (P. limantouri from P. petenensis in Central America; Palacios et al., 2016) and adaptation to local environmental conditions in toxic, hydrogen-sulfide-rich springs (P. sulphuraria and P. thermalis; Palacios et al., 2013). Lastly, the most recent invasion of P. mexicana may have included two independent invasion events from Central America; one represented by lineages occurring in the northern distribution and the other into the southern portion in Mexico.
Within species, strong phylogeographic structuring was observed in two species (P. nelsoni and P. mexicana) over small spatial scales. This is likely due to the fragmentation and disruption of connections between drainages, potentially by the presence of physiographic barriers such as the Balsas Depression, thereby limiting dispersal and isolating populations. This pattern is common in other Neotropical fish species distributed in Mexico, such as the genera Astyanax Baird & Girard, 1854 (Coghill et al., 2014), Profundulus Hubbs, 1924 (Morcillo et al., 2016), and Poeciliopsis Regan, 1913 (Mateos et al., 2002), although other species, like P. sphenops, appear to be able to cross the very same barriers.
Geological barriers influencing diversification in Mexican mollies. The Trans Mexican Volcanic Belt (TMVB) is a physiographic feature that formed west to east between 25–2.5 million years ago (Miller et al., 2005). The TMVB is composed of ridges and volcanoes that decrease eastwardly in altitude toward Veracruz (Miller et al., 2005). This barrier is a phylogeographic break for both terrestrial and aquatic species (Huidobro et al., 2006), and our results suggest that the TMVB was associated with a vicariant speciation event on the Pacific versant, resulting in the evolution of sister species north (P. butleri) and south (P. nelsoni) of this barrier. This pattern has been previously observed in these sister taxa (Mateos, 2005; Zúñiga-Vega et al., 2014) and in other vertebrate species (Devitt, 2006; Blair, Sánchez-Ramírez, 2016; Light et al., 2016). However, the most common recent ancestors of several fish groups distributed across the TMVB originated north of the TMVB (Pérez-Rodríguez et al., 2015), whereas our study suggests an origin south of the TMVB because of the higher genetic variation in the populations of P. nelsoni (observed in most southern populations) and due to the phylogenetic structure pointing toward repeated colonization from Central America. A similar south to north crossing of the TMVB has recently been documented for Poeciliopsis scarli Meyer, Riehl, Dawes & Dibble, 1985 (Conway et al., 2019). Invasion by or isolation of populations of Neotropical freshwater fish to the north of the TMVB on the Pacific Coast has also been documented in fish of the genera Astyanax (Strecker et al., 2012) and Poeciliopsis (Mateos et al., 2002).
On the Atlantic slope, the eastern most point of TMVB is called Punta del Morro (PDM) and serves as a phylogeographic break (Contreras-Balderas et al., 1996). The uplift of this barrier has caused vicariant events evident in species pairs of various families of Neartic and Neotropical freshwater fish (e.g., Lepisosteidae, Clupeidea, Cichlidae, Characidae, and Poeciliidae; Contreras-Balderas et al., 1996; Hulsey et al., 2004; Agorreta et al., 2013). The mountains at PDM serve as a filter for primary and secondary Neartic and Neotropical fishes (Obregon-Barboza et al., 1994), frequently setting distribution limits and influencing phenotypic differences (Hulsey et al., 2010). For P. mexicana, unique haplotypes exist north of the PDM, which may be indicative of such isolation. In contrast, our results for P. sphenops and P. limantouri, which exhibited shared haplotypes on either side of the TMVB, suggested that the PDM is not a strong isolating barrier for these species at this time. However, the presence of P. sphenops outside of the described range (Miller et al., 2005), especially north of the TMVB, may also be a result of human introductions (Espinosa Perez, Ramírez, 2015).
The Balsas River system is the largest hydrological system on the Pacific slope of Mexico and houses many endemic species. The high rate of endemism in the Balsas Depression (BD) is a result of the isolation due to the formation of the Trans Mexican Volcanic Belt and Sierra Madre del Sur (Ferrusquía-Villafranca, 1998; Ferrari et al., 2000). The Balsas River system has been linked to events of dispersals and colonization by Neartic fish from the Mesa Central, a high plateau in central Mexico (Domínguez-Domínguez et al., 2010), a pattern coinciding in fish parasites (Martínez-Aquino et al., 2014), or with isolation of populations due the uplift of the TMVB (Pérez-Rodríguez et al., 2015). In our case, the Balsas River Valley is a potential area of transition for the northern expansion of P. nelsoni and P. sphenops. For P. nelsoni, the southern distribution of our sampling in Chiapas showed higher genetic diversity, potentially in relation to older populations, and for P. sphenops, theBD formed part of the historical documented range (Miller et al., 2005). This idea is supported by the shared populations or sister taxa in surrounding areas of the BD for Mollienesia and other species and coincides with the most recent geological isolation of this area by Plio-Pleistocene volcanism (Marshall, Liebherr, 2000). Future fine scale sampling may reveal additional patterns in the BD, as the Sierra de Taxco divides the Balsas basin into western and eastern units (Cabral-Cano et al., 2000). Interestingly, this ichthyogeographical province (Miller, Smith, 1986; Miller et al., 2005) houses the native endemic species P. maylandi of the subgenus Mollienesia,but sampling effortsin and around the type locality (Meyer, 1983)were unsuccessful, instead finding only P. nelsoni and P. sphenops.
Finally, the Isthmus of Tehuantepec is a low elevation area recognized as a strong faunal barrier that emerged during the Pliocene and Pleistocene (Campbell, 1999; Marshall, Liebherr, 2000; Mulcahy et al., 2006). This potential barrier does not appear to have played a significant role in shaping inter-specific variation in Mollienesia taxa (Fig. 2). However, our results for P. mexicana suggest that the Isthmus may be a moderate barrier, with one sub-clade being distributed almost entirely south of the Isthmus of Tehuantepec (and this sub-clade was sister to Guatemalan samples), and another being distributed entirely north of the Isthmus of Tehuantepec (Fig. 5). The same pattern did not hold for either P. sphenops or P. nelsoni, as both species had shared haplotypes occurring across the Isthmus (Figs. 3, 4). Our results for these species contrast with a recent study of Poeciliopsis pleurospilus and P. gracilis which, based on ddRADseq data, showed that the Isthmus of Tehuantepec was a clear geographic barrier between them (Ward et al., 2022).
In summary, the phylogenetic patterns of Mexican species in the subgenus Mollienesia demonstrate several independent invasions from Central America and subsequent diversification of species associated with isolation, both because of major physiogeographic barriers and ecological selection. The phylogeographic patterns observed within Mexican species either indicated strong genetic structuring across a physiographic barrier (P. nelsoni and P. mexicana) or a lack of genetic structuring (P. sphenops, P. butleri, P. limantouri, and P. sulphuraria/thermalis), indicating that different species may vary significantly in their dispersal abilities. In fact, P. sulphuraria and P. thermalis are so closely related that they may represent the same species, but a formal taxonomic revision is required to determine whether P. sulphuraria should be treated as a synonym to P. thermalis (Palacios etal., 2013). Our analyses also indicated broader distributions than currently described for several species. This study also sheds insight into the taxonomic status of Pacific populations of P. sphenops, which was morphologically hypothesized as distinct based on the scale count around the peduncle (Miller et al., 2005). Since we find lack of genetic differentiation across the species’ distribution, these morphological differences are likely indicative of geographic variation in phenotypes and not species differences.
Acknowledgments
The authors thank C. Passow and Z. Culumber for help in the field. We thank G. Sosa with assistance in file transfer of analyses. Permits issued by the Municipio de Tacotalpa-Tabasco (DFET/23/2011), the Mexican Federal Agencies SEMARNAT (SGPA/DGVS/04315/11 for Poecilia sulphuraria and SGPA/DGVS/02076/12 for P. nelsoni (issued under butleri), CONAPESCA (DGOPA. 09004.041111.3088 for Poecilia sp.). Financial support came from Texas A&M University and the National Council for Science and Technology Exchange Program for Visiting Student Researchers in Mexico (to MP) and the National Science Foundation (IOS–1121832, IOS–1463720, and IOS–1557860 to MT; DEB–0743782 to Luis A. Hurtado and MM).
References
Agorreta A, Domínguez-Domínguez O, Reina RG, Miranda R, Bermingham E, Doadrio I. Phylogenetic relationships and biogeography of Pseudoxiphophorus (Teleostei: Poeciliidae) based on mitochondrial and nuclear genes. Mol Phylogenet Evol. 2013; 66(1):80–90. https://doi.org/10.1016/j.ympev.2012.09.010
Alda F, Reina RG, Doadrio I, Bermingham E. Phylogeny and biogeography of the Poecilia sphenops species complex (Actinopterygii, Poeciliidae) in Central America. Mol Phylogenet Evol. 2013; 66(3):1011–26. https://doi.org/10.1016/j.ympev.2012.12.012
Álvarez del Villar J. Descripción de una nueva especie de Mollienisia capturada en Baños del Azufre, Tabasco (Pisces, Poeciliidae). An Esc Nac Cienc Biol Mex. 1947; 5:275–81.
Bagley JC, Alda F, Breitman MF, Bermingham E, van den Berghe EP, Johnson JB. Assessing species boundaries using multilocus species delimitation in a morphologically conserved group of neotropical freshwater fishes, the Poecilia sphenops species complex (Poeciliidae). PLoS ONE. 2015; 10:e0121139. https://doi.org/10.1371/journal.pone.0121139
Blair C, Sánchez-Ramírez S. Diversity-dependent cladogenesis throughout western Mexico: Evolutionary biogeography of rattlesnakes (Viperidae: Crotalinae: Crotalus and Sistrurus). Mol Phylogenet Evol. 2016(April); 97:145–54. https://doi.org/10.1016/j.ympev.2015.12.020
Bragança PHN, Amorim PF, Costa WJ. Pantanodontidae (Teleostei, Cyprinodontiformes), the sister group to all other cyprinodontoid killifishes as inferred by molecular data. Zoosyst Evol. 2018; 94(1):137–45. https://doi.org/10.3897/zse.94.22173
Breden F, Ptacek MB, Rashed M, Taphorn D, Figueiredo CA. Molecular phylogeny of the live-bearing fish genus Poecilia (Cyprinodontiformes: Poeciliidae). Mol Phylogenet Evol. 1999; 12(2):95–104. https://doi.org/10.1006/mpev.1998.0600
Broughton RE, Gold JR. Phylogenetic relationships in the North American cyprinid genus Cyprinella (Actinopterygii: Cyprinidae) based on sequences of the mitochondrial ND2 and ND4L genes. Copeia. 2000; 2000(1):1–10. https://doi.org/10.1643/0045-8511(2000)2000[0001:PRITNA]2.0.CO;2
Bussing WA. Geographic distribution of the San Juan ichthyofauna of Central America with remarks on its origin and ecology. In: Thorson TB, editor. Investigations of the ichthyofauna of Nicaraguan Lakes – A monumantal work on Nicaraguan Fishes. Lincoln, Nebraska: The school of life sciences, University of Nebraska Lincoln; 1976. p.57–175.
Cabral-Cano E, Lang HR, Harrison CGA. Stratigraphic assessment of the Arcelia–Teloloapan area, southern Mexico: implications for southern Mexico’s post-Neocomian tectonic evolution. J South Amer Earth Sci. 2000(4–5); 13:443–57. https://doi.org/10.1016/S0895-9811(00)00035-3
Campbell JA. Distribution patterns of amphibians in Middle America. In: Duellman WE, editor. Patterns distribution of amphibians: A global perspective. The John Hopkins University Press, Baltimore and London; 1999. p.111–120.
Clement M, Posada D, Crandall KA. TCS: A computer program to estimate gene genealogies. Mol Ecol. 2000; 9(10):1657–59. https://doi.org/10.1046/j.1365-294x.2000.01020.x
Coghill LM, Hulsey CD, Chaves-Campos J, García de Leon FJ, Johnson SG. Next generation phylogeography of cave and surface Astyanax mexicanus. Mol Phylogenet Evol. 2014; 79:368–74. https://doi.org/10.1016/j.ympev.2014.06.029
Conabio. La diversidad biológia de México estudio de País. 1998. CONABIO, México.
Contreras-Balderas S, Ogregon SH, Lozano-Vilano M. Punta del Morro, una interesante barrera de los patrones de distribución de los peces continentales el Norte y Centro de Veracruz, Mexico. Acta Biol Venez. 1996; 16(4):37–42.
Contreras-Balderas S, Ruiz-Campos G, Schmitter-Soto JJ, Díaz-Pardo E, Contreras-McBeath T, Medina-Soto M et al. Freshwater fishes and water status in México: A country-wide appraisal. Aquat Ecosyst Health Manag. 2008; 11(3):246–56. https://doi.org/10.1080/14634980802319986
Contreras-MacBeath T, Rodríguez MB, Sorani V, Goldspink C, Reid MG. Richness and endemism of the freshwater fishes of Mexico. Journal of Threatened Taxa. 2014; 6(2):5421–33. https://doi.org/10.11609/JoTT.o3633.5421-33
Conway KW, Mateos M, Vrijenhoek RC. A new species of the live-bearing fish genus Poeciliopsis from northern Mexico (Cyprinodontiformes, Poeciliidae). ZooKeys. 2019; 883:91–118. https://doi.org/10.3897/zookeys.883.37586
De la Maza-Benignos M, Ornelas-García CP, Lozano-Vilano ML, García-Ramírez E, Doadrio I. Phylogeographic analysis of genus Herichthys (Perciformes: Cichlidae), with descriptions of Nosferatu new genus and H. tepehua n. sp. Hydrobiologia. 2015; 748:201–31. https://doi.org/10.1007/s10750-014-1891-8
Devitt TJ. Phylogeography of the Western Lyresnake (Trimorphodon biscutatus): testing aridland biogeographical hypotheses across the Nearctic-Neotropical transition. Mol Ecol. 2006; 15(14):4387–407. https://doi.org/10.1111/j.1365-294X.2006.03015.x
Domínguez-Domínguez O, Bernal-Zuñiga DM, Piller KR. Two new species of the genus Xenotoca Hubbs and Turner, 1939 (Teleostei, Goodeidae) from central-western Mexico. Zootaxa. 2016; 4189(1). https://doi.org/10.11646/zootaxa.4189.1.3
Dominguez-Domínguez O, Doadrio I, Pérez-Ponce de León G. Historical biogeography of some river basins in central Mexico evidenced by their goodeine freshwater fishes: a preliminary hypothesis using secondary Brooks parsimony analysis. J Biogeogr. 2006; 33(8):1437–47. https://doi.org/10.1111/j.1365-2699.2006.01526.x
Domínguez-Domínguez O, Pedraza-Lara C, Gurrola-Sánchez N, Perea S, Pérez-Rodríguez R, Israde-Alcántara I et al. Historical biogeography of the Goodeinae (Cyprinodontiforms). In: Uribe M, Grier HJ, editors. Viviparous Fishes II. Homestead, Florida: New Life Publications; 2010. p.13–30.
Espinosa Perez H, Ramírez HM. Exotic and invasive fishes in Mexico. CheckList. 2015; 11(3):1–13. https://doi.org/10.15560/11.3.1627
Ferrari L, Conticelli S, Vaggelli G, Petrone CM, Manetti P. Late Miocene volcanism and intra-arc tectonics during the early development of the Trans-Mexican Volcanic Belt. Tectonophysics. 2000; 318:161–85. https://doi.org/10.1016/S0040-1951(99)00310-8
Ferrari L, López-Martínez M, Aguirre-Díaz G, Carrasco-Núñez G. Space-time patterns of Cenozoic arc volcanism in central Mexico: From the Sierra Madre Occidental to the Mexican Volcanic Belt. Geology. 1999; 27(4):303–06. https://doi.org/10.1130/0091-7613(1999)027%3C0303:STPOCA%3E2.3.CO;2
Ferrusquía-Villafranca I. Geología de Mexico: una sinopsis. In: Ramamoorthy TP, Bye R, Lot A, editors. Diversidad biológica de México: orígenes y distribución. Universidad Nacional Autónoma de México: Instituto de Biologia; 1998. p.3–108.
Greven H. Gonads, genitals, and reproductive biology. In: Evans JP, Pilastro A, Schlupp I, editors. Ecology and evolution of poeciliid fishes. Chicago: The University of Chicago Press. 2011; p.3–17.
Ho ALFC, Pruett CL, Lin J. Phylogeny and biogeography of Poecilia (Cyprinodontiformes: Poeciliinae) across Central and South America based on mitochondrial and nuclear DNA markers. Mol Phylogenet Evol. 2016; 101:32–45. https://doi.org/10.1016/j.ympev.2016.04.032
Hrbek T, Seckinger J, Meyer A. A phylogenetic and biogeographic perspective on the evolution of poeciliid fishes. Mol Phylogenet Evol. 2007; 43(3):986–98. https://doi.org/10.1016/j.ympev.2006.06.009
Hufnagel L, Mics F. Introductory Chapter: Biodiversity of Mexico. In: Hufnagel L, editor. Natural history and ecology of Mexico and Central America. Rijeka: IntechOpen; 2021.
Huidobro L, Morrone JJ, Villalobos JL, Alvarez F. Distributional patterns of freshwater taxa (fishes, crustaceans and plants) from the Mexican Transition Zone. J Biogeogr. 2006; 33(4):731–41. https://doi.org/10.1111/j.1365-2699.2005.01400.x
Hulsey CD, García de León FJ, Sánchez Johnson Y, Hendrickson DA, Near TJ. Temporal diversification of Mesoamerican cichlid fishes across a major biogeographic boundary. Mol Phylogenet Evol. 2004; 31(2):754–64. https://doi.org/10.1016/j.ympev.2003.08.024
Hulsey CD, Mims MC, Parnell NF, Streelman JT. Comparative rates of lower jaw diversification in cichlid adaptive radiations. J Evol Biol. 2010; 23(7):1456–67. https://doi.org/10.1111/j.1420-9101.2010.02004.x
Katoh K, Toh H. Recent developments in the MAFFT multiple sequence alignment program. Brief Bioinform. 2008; 9(4):286–98. https://doi.org/10.1093/bib/bbn013
Kocher TD, Conroy JA, McKaye KR, Stauffer JR, Lockwood SF. Evolution of NADH dehydrogenase subunit 2 in east African cichlid fish. Mol Phylogenet Evol. 1995; 4(4):420–32. https://doi.org/10.1006/mpev.1995.1039
Kumar S, Stecher G, Tamura K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol. 2016; 33(7):1870–74. https://doi.org/10.1093/molbev/msw054
Lanfear R, Calcott B, Ho SYW, Guindon S. Partitionfinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol. 2012; 29(6):1695–701. https://doi.org/10.1093/molbev/mss020
Langerhans RB, Gifford ME, Domínguez-Domínguez O, García-Bedoya D, DeWitt TJ. Gambusia quadruncus (Cyprinodontiformes: Poeciliidae): a new species of mosquitofish from east-central México. J Fish Biol. 2012; 81(5):1514–39. https://doi.org/10.1111/j.1095-8649.2012.03397.x
Leigh JW, Bryant D. Popart : Full-feature software for haplotype network construction. Methods Ecol Evol. 2015; 6(9):1110–16. https://doi.org/10.1111/2041-210X.12410
Light JE, Ostroff MO, Hafner DJ. Phylogeographic assessment of the northern pygmy mouse, Baiomys taylori. J Mammal. 2016; 97(4):1081–94. https://doi.org/10.1093/jmammal/gyw065
Lyons T, Máiz-Tomé L, Tognelli MF, Daniels A, Meredith C, Bullock R et al, editors. The status and distribution of freshwater fishes in Mexico. IUCN Freshwater Biodiversity Unit; 2020. https://portals.iucn.org/library/node/49039
Marshall CJ, Liebherr JK. Cladistic biogeography of the Mexican transition zone. J Biogeogr. 2000; 27(1):203–16. https://doi.org/10.1046/j.1365-2699.2000.00388.x
Martínez-Aquino A, Ceccarelli FS, Eguiarte LE, Vásquez-Dominguez E, Pérez-Ponce de León G. Do the historical biogeography and evolutionary history of the digenean Margotrema spp. across central Mexico mirror those of their freshwater fish hosts (Goodeinae)? PLoS ONE. 2014; 9:e101700. https://doi.org/10.1371/journal.pone.0101700
Matamoros WA, Domínguez-Cisneros SE, Velázquez-Velázquez E, McMahan CD. Description of a new species of killifish of the genus Profundulus (Atherinomorpha: Profundulidae) from the Mexican State of Oaxaca. Copeia. 2018; 106(2):239–46. https://doi.org/10.1643/CI-17-677
Mateos M. Comparative phylogeography of livebearing fishes in the genera Poeciliopsis and Poecilia (Poeciliidae: Cyprinodontiformes) in central Mexico. J Biogeogr. 2005; 32(5):775–80. https://doi.org/10.1111/j.1365-2699.2005.01236.x
Mateos M, Sanjur OI, Vrijenhoek RC. Historical biogeography of the livebearing fish genus Poeciliopsis (Poeciliidae: Cyprinodontiformes). Evolution. 2002; 56(5):972–84. https://doi.org/10.1111/j.0014-3820.2002.tb01409.x
Mejía O, León-Romero Y, Soto-Galera E. DNA barcoding of the ichthyofauna of Pánuco-Tamesí complex: evidence for taxonomic conflicts in some groups. Mitochondrial DNA. 2012; 23(6):471–76. https://doi.org/10.3109/19401736.2012.710207
Meredith RW, Pires MN, Reznick DN, Springer MS. Molecular phylogenetic relationships and the coevolution of placentotrophy and superfetation in Poecilia (Poeciliidae: Cyprinodontiformes). Mol Phylogenet Evol. 2011; 59(1):148–57. https://doi.org/10.1016/j.ympev.2011.01.014
Meredith RW, Pires MN, Reznick DN, Springer MS. Molecular phylogenetic relationships and the evolution of the placenta in Poecilia (Micropoecilia) (Poeciliidae: Cyprinodontiformes). Mol Phylogenet Evol. 2010; 55(2):631–39. https://doi.org/10.1016/j.ympev.2009.11.006
Metcalfe SE, O’Hara SL, Caballero M, Davies SJ. Records of Late Pleistocene–Holocene climatic change in Mexico – a review. Quat Sci Rev. 2000; 19(7):699–721. https://doi.org/10.1016/S0277-3791(99)00022-0
Meyer MK. Une nouvelle espèce de Poecilia du Guerrero, Mexique (Pisces: Poeciliidae). RFAH. 1983; 10(2):55–58.
Miller RR. Checklist and key to the mollies of Mexico (Pisces, Poeciliidae, Poecilia, Subgenus Mollienesia). Copeia. 1983; 1983(3):817–22. https://www.jstor.org/stable/1444354
Miller RR. Five new species of Mexican poeciliid fishes of the genera Poecilia, Gambusia, and Poeciliopsis. Occasional papers of the Museum of Zoology, University of Michigan. 1975; 672:1–44. Available from: https://deepblue.lib.umich.edu/handle/2027.42/57108
Miller RR, Minckley WL, Norris SM, Gach MH. Freshwater fishes of Mexico. University of Chicago Press, Chicago; 2005.
Miller RR, Smith ML. Origin and geography of the fishes of central Mexico. In: Hocutt CH, Wiley EO, editors. The zoogeography of north american freshwater fishes. New York: Wiley; 1986. p.487–519.
Morcillo F, Ornelas-García CP, Alcaraz L, Matamoros WA, Doadrio I. Phylogenetic relationships and evolutionary history of the Mesoamerican endemic freshwater fish family Profundulidae (Cyprinodontiformes: Actinopterygii). Mol Phylogenet Evol. 2016; 94:242–51. https://doi.org/10.1016/j.ympev.2015.09.002
Morrone JJ, Márquez J. Halffter’s Mexican Transition Zone, beetle generalized tracks, and geographical homology. J Biogeogr. 2001; 28(5):635–50. https://doi.org/10.1046/j.1365-2699.2001.00571.x
Mulcahy DG, Morrill BH, Mendelson JR. Historical biogeography of lowland species of toads (Bufo) across the Trans-Mexican Neovolcanic Belt and the Isthmus of Tehuantepec. J Biogeogr. 2006; 33(11):1889–904. https://doi.org/10.1111/j.1365-2699.2006.01546.x
Obregon-Barboza H, Contreras-Balderas S, Lozano-Vilano MD. The fishes of Northern and Central Veracruz, Mexico. Hydrobiologia. 1994; 286:79–95. https://doi.org/10.1007/BF00008499
Ornelas-García CP, Martínez-Ramírez E, Doadrio I. A new species of killifish of the family Profundulidae from the highlandsof the Mixteca region, Mexico. Rev Mex Biodivers. 2015; 86(4):926–33. https://doi.org/10.1016/j.rmb.2015.10.001
Palacios M, Arias-Rodriguez L, Plath M, Eifert C, Lerp H, Lamboj A et al. The rediscovery of a long described species reveals additional complexity in speciation patterns of poeciliid fishes in sulfide springs. PLoS ONE. 2013; 8:e71069. https://doi.org/10.1371/journal.pone.0071069
Palacios M, Voelker G, Arias Rodríguez L, Mateos M, Tobler M. Phylogenetic analyses of the subgenus Mollienesia (Poecilia, Poeciliidae, Teleostei) reveal taxonomic inconsistencies, cryptic biodiversity, and spatio-temporal aspects of diversification in Middle America. Mol Phylogenet Evol. 2016; 103:230–44. https://doi.org/10.1016/j.ympev.2016.07.025
Parenti LR. A phylogenetic and biogeographic analysis of cyprinodontiform fishes (Teleostei, Atherinomorpha). Bull Am Mus Nat Hist. 1981; 168:335–557. http://hdl.handle.net/2246/438
Pérez-Rodríguez R, Domínguez-Domínguez O, Doadrio I, Cuevas-García E, Pérez-Ponce de León G. Comparative historical biogeography of three groups of Nearctic freshwater fishes across central Mexico. J Fish Biol. 2015; 86(3):993–1015. https://doi.org/10.1111/jfb.12611
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA. Posterior Summarization in Bayesian Phylogenetics Using Tracer 1.7. Syst Biol. 2018; 67(5):901–04. https://doi.org/10.1093/sysbio/syy032
Reis RE, Kullander SO, Ferraris CJ, editors. The check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003.
Reznick DN, Furness AI, Meredith RW, Springer MS. The origin and biogeographic diversification of fishes in the family Poeciliidae. PLoS ONE. 2017; 12:e0172546. https://doi.org/10.1371/journal.pone.0172546
Ronquist F, Huelsenbeck JP. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics. 2003; 19(12):1572–74. https://doi.org/10.1093/bioinformatics/btg180
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol. 2012; 61(3):539–42. https://doi.org/10.1093/sysbio/sys029
Rosas-Elguera J, Ferrari L, Garduño-Monroy VH, Urrutia-Fucugauchi J. Continental boundaries of the Jalisco block and their influence in the Pliocene-Quaternary kinematics of western Mexico. Geology. 1996; 24(10):921–24. https://doi.org/10.1130/0091-7613(1996)024%3C0921:CBOTJB%3E2.3.CO;2
Rosen DE, Bailey RM. The poeciliid fishes (Cyprinodontiformes), their structure, zoogeography and systematics. Bull Amer Mus Nat Hist. 1963; 126:1–176. https://digitallibrary.amnh.org/handle/2246/1123
Schmidt TR, Bielawski JP, Gold JR. Molecular phylogenetics and evolution of the cytochrome b gene in the cyprinid genus Lythrurus (Actinopterygii: Cypriniformes). Copeia. 1998; 1998(1):14–22. https://www.jstor.org/stable/pdf/1447697.pdf
Stamatakis A. RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics. 2006; 22(21):2688–90. https://doi.org/10.1093/bioinformatics/btl446
Stamatakis A. A rapid bootstrap algorithm for the RAxML web servers. Syst Biol. 2008; 57(5):758–71. https://doi.org/10.1080/10635150802429642
Stöck M, Lampert KP, Möller D, Schlupp I, Schartl M. Monophyletic origin of multiple clonal lineages in an asexual fish (Poecilia formosa). Mol Ecol. 2010; 19(23):5204–15. https://doi.org/10.1111/j.1365-294X.2010.04869.x
Strecker U, Faundez VH, Wilkens H. Phylogeography of surface and cave Astyanax (Teleostei) from Central and North America based on cytochrome b sequence data. Mol Phylogenet Evol. 2004; 33(2):469–81. https://doi.org/10.1016/j.ympev.2004.07.001
Strecker U, Hausdorf B, Wilkens H. Parallel speciation in Astyanax cave fish (Teleostei) in Northern Mexico. Mol Phylogenet Evol. 2012; 62(1):62–70. https://doi.org/10.1016/j.ympev.2011.09.005
Sukumaran J, Holder MT. DendroPy: a Python library for phylogenetic computing. Bioinformatics. 2010; 26(12):1569–71. https://doi.org/10.1093/bioinformatics/btq228
Templeton AR, Crandall KA, Sing CF. A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping and DNA sequence data. III. Cladogram estimation. Genetics. 1992; 132(2):619–33. https://doi.org/10.1093/genetics/132.2.619
Tobler M, Palacios M, Chapman LJ, Mitrofanov I, Bierbach D, Plath M et al. Evolution in extreme environments: replicated phenotypic differentiation in livebearing fish inhabiting sulfidic springs. Evolution. 2011; 65(8):2213–28. https://doi.org/10.1111/j.1558-5646.2011.01298.x
Tobler M, Plath M. Living in extreme habitats. In: Evans J, Pilastro A, Schlupp I, editors. Ecology and evolution of poeciliid fishes. Chicago: University of Chicago Press; 2011. p.120–27.
Villesen P. FaBox: an online toolbox for fasta sequences. Mol Ecol Notes. 2007; 7(6):965–68. https://doi.org/10.1111/j.1471-8286.2007.01821.x
Ward SJ, McMahan CD, Khakurel B, Wright AM, Piller KR. Genomic data support the taxonomic validity of Middle American livebearers Poeciliopsis gracilis and Poeciliopsis pleurospilus (Cyprinodontiformes: Poeciliidae). PLoS ONE 17(1):e0262687. https://doi.org/10.1371/journal.pone.0262687
Zúñiga-Vega JJ, Ingley SJ, Unmack PJ, Johnson JB. Do freshwater ecoregions and continental shelf width predict patterns of historical gene flow in the freshwater fish Poecilia butleri? Biol J Linn Soc Lond. 2014; 112(3):399–416. https://doi.org/10.1111/bij.12289
Authors
![]() Maura Palacios1,
Maura Palacios1, ![]() Alfonso A. González-Díaz2,
Alfonso A. González-Díaz2, ![]() Lenin Arias Rodriguez3,
Lenin Arias Rodriguez3, ![]() Mariana Mateos1,
Mariana Mateos1, ![]() Rocío Rodiles-Hernández2,
Rocío Rodiles-Hernández2, ![]() Michael Tobler4 and
Michael Tobler4 and ![]() Gary Voelker1
Gary Voelker1 ![]()
[1] Department of Ecology and Conservation Biology, Texas A&M University, 2258 TAMU, College Station, TX 77843, USA. (MP) mauraeva@gmail.com, (MM) mariana.mateos@ag.tamu.edu, (GV) gvoelker@tamu.edu (corresponding author).
[2] Departamento de Conservación de la Biodiversidad, El Colegio de la Frontera Sur (ECOSUR), Carretera Panamericana y Periférico Sur s/n, Barrio María Auxiliadora, San Cristóbal de Las Casas, Chiapas CP 29290, Mexico. (AAGD) agonzalez@ecosur.mex, (RRH) rrodiles@ecosur.mex.
[3] División Académica de Ciencias Biológicas, Universidad Juárez Autónoma de Tabasco (UJAT), C.P. 86150 Villahermosa, Tabasco, Mexico. (LAR) leninariasrodriguez@hotmail.com.
[4] Division of Biology, Kansas State University, 116 Ackert Hall, Manhattan, KS 66506, USA. (MT) michi.tobler@gmail.com.
Authors’ Contribution 

Maura Palacios: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft.
Alfonso A. González-Díaz: Methodology, Writing-review and editing.
Lenin Arias Rodriguez: Investigation, Methodology, Writing-review and editing.
Mariana Mateos: Formal analysis, Funding acquisition, Investigation, Writing-review and editing.
Rocío Rodiles-Hernández: Methodology, Writing-review and editing.
Michael Tobler: Conceptualization, Investigation, Methodology, Writing-original draft.
Gary Voelker: Conceptualization, Formal analysis, Investigation, Writing-original draft.
Ethical Statement
Protocols approved by the Texas A&M University and Oklahoma State University Committees on Use and Care of Animals (IACUC 2011–118 and ACUP AS10–15).
Competing Interests
The author declares no competing interests.
How to cite this article
Palacios M, González-Díaz AA, Rodriguez LA, Mateos M, Rodiles-Hernández R, Tobler M, Voelker G. Population level genetic divergence and phylogenetic placement of Mexican shortfin mollies (Mollienesia: Poecilia: Poeciliidae). Neotrop Ichthyol. 2023; 21(2):e220101. https://doi.org/10.1590/1982-0224-2022-0101
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted April 2, 2023 by Paulo Lucinda
Accepted April 2, 2023 by Paulo Lucinda
![]() Submitted December 5, 2022
Submitted December 5, 2022
![]() Epub May 1, 2023
Epub May 1, 2023