![]() Laura Rincón1,
Laura Rincón1, ![]() Felipe Redondo1
Felipe Redondo1 ![]() ,
, ![]() Wendy Kobrinsky1,
Wendy Kobrinsky1, ![]() Matías Pandolfi1 and
Matías Pandolfi1 and ![]() Andrea G. Pozzi1
Andrea G. Pozzi1
PDF: EN XML: EN | Cite this article
Abstract
The cardinal tetra Paracheirodon axelrodi is a species of the family Characidae of great interest as an ornamental fish. Many aspects of the biology of this species are still unknown. The present work presents a complete description of the different components of the digestive tract of P. axelrodi, analyzing its global anatomical arrangement, the adjacent glands (liver and pancreas) and the previously ignored pyloric caeca, using histological and histochemical techniques with light microscopy and scanning electron microscopy. Three levels of analysis were performed: macroscopic anatomy, histology of the gastrointestinal tract and scanning electron microscopy of mucosal surfaces. The intestinal coefficient (Ci) of P. axelrodi calculated here (0.94) is within the expected range for a carnivorous species. The anatomy of the alimentary and the resulting Ci is consistent with carnivorous diet in line with dietary studies in the wild in this species. The digestive tract, including adjacent glands (liver and pancreas), presented a relatively conserved general pattern within that described for other species of the family Characidae, especially at the histological level.
Keywords: Cardinal tetra, Carnivorous diet, Intestinal coefficient, Histology, SEM.
El tetra cardenal Paracheirodon axelrodi es una especie de la familia Characidae de gran interés como pez ornamental. Muchos aspectos de la biología de esta especie son aún desconocidos. En este trabajo se presenta una descripción completa de los diferentes componentes del tracto digestivo de P. axelrodi, analizando su disposición anatómica global, las glándulas adyacentes (hígado y páncreas) y la presencia de ciegos pilóricos, mediante técnicas histológicas e histoquímicas con microscopía óptica y electrónica de barrido. Se realizaron tres niveles de análisis: anatomía macroscópica, histología del tracto gastrointestinal y microscopía electrónica de barrido de las superficies mucosas. El coeficiente intestinal (Ci) de P. axelrodi calculado aquí (0,94) está dentro del rango esperado para una especie carnívora. Los estudios anatómicos del tracto alimentario y el Ci son consistentes con una dieta carnívora en línea con los estudios de la dieta en ejemplares silvestres en esta especie. El tracto digestivo, incluyendo las glándulas adyacentes (hígado y páncreas), presentó un patrón general relativamente conservado dentro de lo descrito para otras especies de la familia Characidae, especialmente a nivel histológico.
Palabras clave: Cardenal tetra, Coeficiente intestinal, Dieta carnívora, Histología, MEB.
Introduction
In recent years, the Digestive tract (DT) of teleost attracts considerable interest because of its anatomical diversity related to dietary habits. Therefore, the gross morphology and histology of DT has long been a topic of research, either from a purely descriptive point of view (Al-Hussaini, 1946; Hale, 1965; Clarke, Witcomb, 1980; Sis et al., 1979; Morrison, Wright, 1999), or in association with the eating habits of the study species (Sinha, 1976; De Silva et al., 1980; Albrecht et al., 2001; Rodrigues et al., 2006; Hanke et al., 2008; Alabssawy et al., 2019). Currently, the existing information on the neotropical ichthyofauna, the most morphologically diverse group of all extant species (Vari, Malabarba, 1998), is still scarce, and even more so, the descriptive information for many South American species. That is why this kind of study could provide crucial information for the reproduction and conservation of species (Santos et al., 2015).
The DT in fish shows a remarkable diversity in terms of morphological and functional characteristics, associated with different feeding habits, taxonomy, as well as body weight, size, and sex (Kapoor et al., 1975; Díaz et al., 2008; Banan Khojasteh et al., 2009). Its functions include digestion, nutrient absorption, hormone secretion and immune protection. The DT comprises distinct portions: the oral cavity, the pharynx, the esophagus, the stomach pyloric caeca, the intestine, and pancreas and liver as accessory glands of DT (Banan Khojasteh, 2012). Al-Hussaini (1946) described that the shape of the teeth shows a correlation with the type of diet. For example, the carnivorous fish Gobio gobio (Linnaeus, 1758) has biserial hooked teeth, the omnivorous fish Rutilus rutilus (Linnaeus, 1758) has uniserial teeth, some of which are hooked, while the herbivorous fish Cyprinus carpio Linnaeus, 1758 has three rows of teeth converging towards each other forming a stud-like ‘molariform’ prominence. Both, the relative overall size of the pharyngeal jaws as well as the muscles associated to them are best developed in herbivorous and least in carnivorous, while the hardness of the horny pad is also in direct relationship to the amount of plant food ingested (Al-Hussaini, 1946; Albrecht et al., 2001; Xiong et al., 2011).
Also, the DT can show a remarkable difference in the pyloric caeca which can vary in position, size, and number. Houssain, Dutta (1998) observed that the greater the caecum size, the better its functional efficiency. In this sense, species with shorter intestines should have either more or larger pyloric caeca (Albrecht et al., 2001). Similarly, the stomach in the teleost presents a variety of different morphologies (Feist, 2009). In some teleost species, the entire stomach appears as a tube of uniform diameter with no marked anatomical differences between the esophagus and stomach. In others, it is described as a round and muscular structure situated at the end of the esophagus. While in some species, the stomach can be divided into a cardiac and a pyloric part and possesses a blind sac of variable size. In other families can be completely absent (Cyprinidae, Labridae, Gobiidae, Scaridae, Cyprinodontidae, some Poeciliidae) (Albrecht et al., 2001; Feist, 2009; Xiong et al., 2011).
The DT structure can vary too according to the nature of the fish diet; it can be quite short in carnivorous species (20% of body length), or much longer in herbivorous species (20 times body length) (Banan Khojasteh, 2012). The intestinal coefficient (Ci) is widely used to classify species into trophic categories, and in fact can help situate species in broad categories (Albrecht et al., 2001; Becker et al., 2010). A general pattern can be extrapolated from several studies on distantly related species (Barbieri et al., 1994) or within the same family (Fryer, Iles, 1972). Accordingly, high Ci values group herbivorous species, whereas intermediate values are associated with omnivores, and low values with carnivores (Ward-Campbell et al., 2005; Becker et al., 2010).
The pancreas and liver are two important organs which take part in digestion. The structure of the liver varies considerably among teleost species, in direct relationship to sex, age, available food (especially regarding glycogen and fat content), temperature, and with endocrine influences strongly connected to the environmentally regulated breeding conditions (Feist, 2009). The liver is usually a compact structure and is in the first third of the abdominal cavity. It can be unilobed, multi lobed, without distinct lobes or with an irregular pattern of cells (Rocha et al., 1994; Ribeiro, Fanta, 2000; Hernandez et al., 2009).
There are two general basic types of fish livers: those that contain pancreatic tissue versus those that do not. Fish livers with exocrine pancreatic tissue are often called “hepatopancreas”. The pancreas is generally diffusely spread within the fat and mesenteries that connect the intestine, stomach, liver, and gallbladder. It can also form a discrete organ like in the Chondrichthyans (Langer et al., 1979; Rombout, Taverne-Thiele, 1982; Ribeiro, Fanta, 2000). The pancreas of teleost has exocrine cells as well as endocrine cells that are organized as Islets of Langerhans or Brockman’s bodies (Ribeiro, Fanta, 2000; Feist, 2009).
The neotropical fish studied in this work is the cardinal tetra Paracheirodon axelrodi (Schultz, 1956), a small member of the Characidae family that lives in the Orinoco, and Rio Negro basins. The Characidae family, with more than 1245 described species, is the most diverse among Neotropical fish and the fourth most diverse in the world (Fricke et al., 2022). The great morphological and behavioral diversity observed in this family is related to the variety of environments that they inhabit (Mirande, 2010). Paracheirodon axelrodi is a non-migratory freshwater fish, which generally lives in shoals in the middle water layers with temperatures between 23°C and 27°C (Anjos, Anjos, 2006; Brito, Bazzoli, 2009). The cardinal tetra exhibits high market value and growing global importance for ornamental aquaculture. It is important both ecologically and economically, being the most abundant species (21%) in the middle basin of the Río Negro (Evers et al., 2019).
Gómez-Ramírez et al. (2011) made a preliminary description of the digestive tract of P. axelrodi, reporting the absence of pyloric caeca for this species. This work aims to expand the description presented by Gómez-Ramírez et al. (2011), whence, the main objective is to describe the different components of the digestive tract of P. axelrodi, analyzing their global anatomical arrangement, and the adjacent glands (liver and pancreas). This paper contributes to the knowledge of the digestive system in P. axelrodi, adding the relation of the anatomical and histological structures of the DT with dietary studies in the wild indicate that it feeds on mesofauna that adhere to submerged leaf litter, roots and aquatic plants (Walker, 2004).
Material and methods
Animals. Sixteen adults of P. axelrodi (eight males and eight females) were obtained from commercial aquariums in Buenos Aires that import specimens from Manaus, Brazil. Animals were housed in 50 liters aquaria mimicking their natural conditions: 26-28 °C, pH of 5.0-6.0, constant photoperiod (14L: 10D). Fish were fed twice daily with commercial food for tropical fish (Tetracolor granules®), containing 47.5% crude protein (min), ether extract 6.5% (min), 2% crude fiber (max), calcium 2.5% (max), 1.5% phosphorus (min), 14% ash (max), vitamin C 100 mg/kg. All fishes were acclimatized for at least one week before analysis.
Animals with a standard length (SL) of 2.07 (mean) ± 0.12 (standard deviation) cm, a total length (TL) of 2.57 ± 0.13 cm and a weight of 0.26 ± 0.05 g, were used. Three levels of analysis were performed: gross anatomy (n = 6, 3 males and 3 females), histology of the digestive tract (n = 10, 5 males and 5 females) and scanning electron microscopy (SEM) (n = 4, 2 males and 2 females) of mucosal surfaces. All animals were anesthetized with 0.1 g/l of benzocaine (Scherck, Moyle, 1990), and euthanized by decapitation. The sections of the body were fixed in Bouin solution for 24 h at 4ºC in the dark and subsequently transferred to ethanol 70%. Voucher specimens are deposited at the Ichthyological collection of the Museo Argentino de Ciencias Naturales “Bernardino Rivadavia” (MACN-Ict 12710), Buenos Aires, Argentina.
Gross anatomy. Animals (n = 6) were dissected under a Leica EZ4D stereomicroscope after euthanization. Dissections consisted of an opening along the midline of the ventral surface of the abdominal cavity. Afterwards, body walls were removed, and the esophagus was sectioned in its anterior portion to free the digestive tract, liver, pancreas, and gas bladder. Pictures were taken using a Nikon D3200 digital camera equipped with a macro lens attached to the stereomicroscope.
For each specimen, the total length (TL; cm), standard length (SL; cm), body weight (W; g) and intestine length (IL; cm) were recorded. Subsequently, the intestinal coefficient (Ci) was calculated using the equation: Ci = IL/SL. The enhanced image-analysis software Fiji was used to measure the IL (cm). This Software is freely available and can be downloaded via the link http://Fiji.sc/Downloads). Results are presented as mean ± standard deviation (M ± SD).
Histology. Full bodies (n = 10) were dehydrated through an ascending ethanol series, clarified in xylene, and embedded in Paraplast® (Sigma). Embedded bodies were sectioned at 7 μm thickness (transversely and longitudinally oriented) and mounted on gelatin-coated glass slides. Then, sections were deparaffinized in xylene, rehydrated through a descending series of ethanol concentration, and stained with Masson Trichromic (MT), Periodic Acid-Schiff (PAS) and Alcian Blue pH 2.5 (AB). The histochemical analysis was performed in a qualitative way, based on the staining intensity. Finally, sections were mounted in DPX (Sigma), examined using a Zeiss Primo Star microscope, and digitally photographed with a Canon PowerShot A640 camera.
Scanning Electron Microscopy (SEM). For SEM, portions of the stomach and intestine were cut longitudinally to expose its mucous surface. The tissues were fixed in Bouin solution for 24 h at 4ºC, dehydrated in a series of increasing concentrations of ethanol (70, 90, 96, and 100%, for 30 min in each bath). Then, samples were critical point dried in CO2 using an EMS 850, Electron Microscopy Science. The tissues were coated with gold using a Quorum Technologies SC 7620, examined with an electron microscope, Philips XL 30, and pictures were obtained with an Olympus PM10AD photomicrographic system.
Results
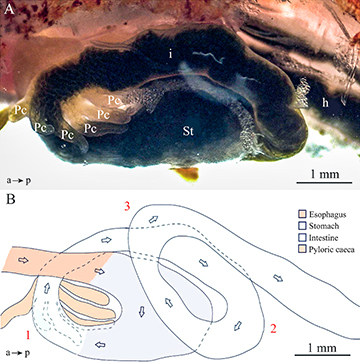
Gross anatomy. The DT of Paracheirodon axelrodi consists of the oropharyngeal cavity, esophagus, stomach, pyloric caeca, intestine, and the associated glands: exocrine pancreas, and liver. Head and snout are relatively short, the mouth showing a terminal position and narrow opening, with a single row of five premaxillary teeth and single maxillary tooth, each with five cusps as previously observed by Weitzman, Fink (1983). The digestive tract (DT) was made up of four macroscopically different parts: esophagus, stomach, pyloric caeca, and intestine (Fig. 1A). The esophagus was a short rectilinear tube with a thick and elastic wall, which extended from the posterior end of the buccopharyngeal cavity to the anterior cardiac region of the stomach. The stomach was a big muscular u-shaped siphon (Fig. 1B).
FIGURE 1| Paracheirodon axelrodi, anatomical arrangement of the digestive tract. A. Left lateral view of the digestive tract under stereoscope showing pyloric caeca (Pc), stomach (St), intestine (i), hindgut (h). B. Schematic representation of the left lateral view of the digestive tract showing the curvatures of the intestine. The most anterior portion of the intestine presents the first dorso-lateral curve (1), from which the six pyloric caeca are observed. At the second curve (2), the intestine was followed by a posterior extension that lay over the stomach. From this point posteriorly, it developed in an antero-dorsal direction, on the left side of the stomach. The intestine is followed by a third curve (3) in a postero-ventral direction. a → p, antero-posterior axes.
The presence of six pyloric caeca marked the beginning of the intestine, this being the longest portion of the DT. The most anterior portion of the intestine presented the first curve (1) dorso-lateral (Fig. 1B), to the right side of the stomach. In this section, there were six pyloric caeca (Fig. 1A), which are blind-bottomed evaginations located perpendicular to the intestinal tube, with an interior lumen. In the second curve (2), the intestine was followed by a posterior extension that lay over the stomach. From this point posteriorly, it developed in an antero-dorsal direction, on the left side of the stomach (Fig. 1B). Afterwards, the intestine was followed by a third curve (3) in a postero-ventral direction (Fig. 1B). From the third curve, the intestine projected posteriorly beyond the second curve. It was not possible to distinguish macroscopically where the intestine ends and where the anus begins (Figs. 1A,B).
Intestinal folds were observed under a stereoscopic microscope by transparency of the wall of the digestive tract. The liver was in the anterior third of the abdominal cavity and had an irregular shape. Regarding the pancreas, it was not possible to observe it macroscopically since it is diffuse within the liver in this species. The digestive tract of P. axelrodi was 2.07 ± 0.12 cm in length and had an Ci of 0.94 ± 0.01.
Histology and Scanning Electron Microscopy (SEM)
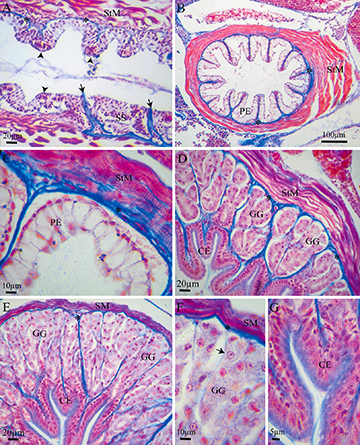
Mouth and Pharynx. The mouth and pharynx presented a mucosa with a stratified squamous epithelium and the lamina propria was formed of loose connective tissue. The muscular layer was composed of striated muscle fibers. In the oral cavity, the fibers were arranged in at least three layers with longitudinal and transversal orientations. In the pharynx, two fiber layers were observed, an inner circular layer and an outer longitudinal one. Pharyngeal teeth were observed too, as well as taste buds (Fig. 2A). The alimentary canal showed a four-layered general structure: mucosa, submucosa, muscularis, and serosa. The muscularis mucosa was not detected.
FIGURE 2| Histological sections of the digestive tract of Paracheirodon axelrodi, stained with Masson Trichrome.A. Longitudinal section of pharynx, showing a mucosa with a stratified squamous epithelium (SS) and underneath this, a layer of loose connective tissue and a thin inner circular layer and an outer longitudinal layer of striated muscle (StM). Pharyngeal teeth (arrows) and taste buds (arrowheads) were observed. B. Cross section of the esophageal mucosa with multiple foldings, composed by a pseudostratified epithelium (PE) and abundant mucous cells. A layer of loose connective tissue was observed, surrounded by a muscular tunica with an inner longitudinal and an external circular layer of StM. C. Detail of B showing pseudostratified epithelium (PE) with abundant mucous cells, a dense septum of connective tissue and external circular layer of StM. D. Transverse section of the first portion of the stomach. where a simple columnar epithelium (CE) formed by a layer of secreting mucus cells, short gastric glands (GG) and a circular layer of StM are observed. E. Transverse section of the fundic stomach which presents a simple columnar epithelium (CE) and long ramified gastric glands (GG) surrounded by a loose connective tissue. The muscular layer at this point contained two sublayers of smooth muscle (SM), a circular inner layer and an outer longitudinal layer. F. Detail of E showing GG composed by oxintopeptics cells (black arrow). G. Detail of E showing CE as a layer of secreting mucus cells. Asterisk, loose connective tissue; CE, simple columnar epithelium; GG, gastric glands; PE, pseudostratified epithelium; SM, smooth muscle; StM, striated muscle.
Esophagus.The esophagus presented a mucosa with many folds, composed by a pseudostratified epithelium with abundant mucous cells (Figs. 2B,C). The epithelial mucous cells had PAS-positive cytoplasm, suggesting the production of neutral mucins. The lamina propria was formed of loose connective tissue, surrounded by a muscular tunica, with an inner longitudinal and an external circular layer of striated muscle fibers (Fig. 2C). The striated muscle fibers continued into the first portion of the stomach.
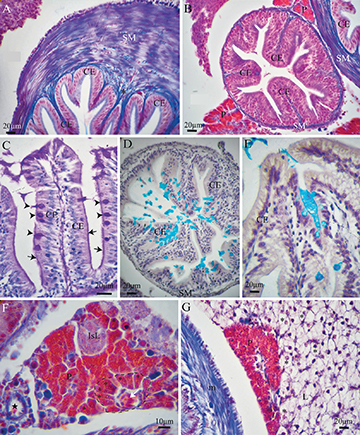
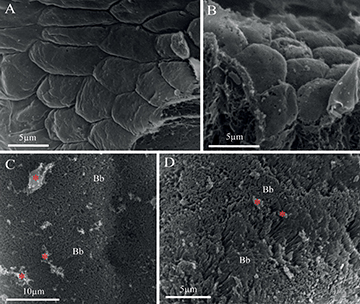
Stomach.The mucosa in the stomach was composed of a simple columnar epithelium (Fig. 3A), which was PAS-positive only in the apical portion. Under SEM, the epithelial cells showed a polyhedral surface (Figs. 4A,B). The lamina propria was formed of a thin layer of dense connective tissue. In the most anterior portion, the stomach presented short tubular-acinous glands (Fig. 2D). In the medial portion, the fundic stomach showed ramified tubular glands surrounded by a loose connective tissue and a dense septum of connective tissue (Figs. 2E,F,G). The gastric glands consisted of oxintopeptic cells (Fig. 2F). In the most caudal portion of the stomach, the pyloric stomach, was characterized by a great development of the smooth muscle and lack of acinous glands (Fig. 3A). The muscular layer at this point contained two sublayers of smooth muscle, except for the most anterior portion which had striated muscle. The orientation of the inner layer of smooth muscle was circular, while the orientation of the outer layer was longitudinal. The serosa layer consisted of dense connective tissue and blood vessels, and, externally, presented a layer of mesothelial squamous cells.
FIGURE 3| Histological sections of the digestive tract of Paracheirodon axelrodi, stained with Masson Trichrome (MT) (A,B,F,G), Periodic Acid-Schiff (PAS) (C) and Alcian Blue (AB) (D,E). A. Cross section of the pyloric portion of the stomach, showing the CE mucosa and a layer of loose connective tissue (asterisk) under the epithelium. Note the absence of GG and a great development of the inner circular layer of SM. B. Cross section of a pyloric cecum, showing mucosal CE cells arranged in a brush border fashion and with numerous folds, and a thin layer of SM. Part of the pyloric stomach can be seen in the upper right corner. A portion of the pancreas (P) can be seen in the upper and lower corners. C. Cross section of intestine showing the PAS positive goblet cells (GCs) (arrowheads) with cytoplasm filled with glycoconjugates. The PAS reaction for neutral glycoconjugates produces an intense purple stain in the mucous granules of GCs and emphasizes the brush border of the enterocytes (arrow). D-E. Pyloric cecum (D) and intestine (E) showing AB positive GCs, with a similar distribution pattern in the epithelium. Thanks to the AB technique for acid mucosubstances, GCs show a strong positive blue staining. F. Pancreas section. The exocrine pancreas is formed by acini (segmented line), which are made up of a group of pyramidal-shaped secretory cells, called acinar cells. Towards the central part of the acinus, a small afferent vein is observed (white arrow). A prominent feature of acinar cells is the presence in their apical regions of aggregated bright eosinophilic zymogen granules (asterisk) that face the narrow lumen. The endocrine pancreas is formed by the Islets of Langerhans (IsL) composed of small cells with a pale-stained granular cytoplasm, in contrast to the larger acinar cells of the exocrine gland, which stain more strongly. On the left is a pancreatic duct (black star) with simple cuboidal epithelium. G. Liver (L) and pancreas (P) section. Many fish livers contain diffuse exocrine pancreatic tissue and are therefore called hepatopancreas. The liver showed polyhedral hepatocytes (asteriks) with a granular and poorly stained cytoplasm, the nuclei are spherical and centrally located presenting well delineated nucleoli. On the left is the smooth muscle layer (SM) of a portion of the intestine. CE, simple columnar epithelium; GCs, goblet cells; GG, gastric glands; SM, smooth muscle.
Pyloric caeca and intestine.In the first portion of the intestine, after the stomach, six pyloric caeca were observed which were histologically like the rest of the intestine. The pyloric caeca were characterized by a mucosa that consisted of a simple columnar epithelium with many deep folds and a thin layer of connective tissue under the epithelium, which in turn was surrounded by a thin layer of smooth muscle (Fig. 3B). The intestine was characterized by a simple columnar epithelium too, with a brush border and mucus secreting goblet cells. The brush border and the secreted mucus were also observed under SEM (Figs. 4C,D). Goblet cells had a cytoplasm filled with glycoconjugates that were stained strongly with PAS and AB pH 2.5 (Figs. 3C,D,F). These cells were interspersed between epithelial cells, both in the intestine and in the caeca (Tab. 1). The serosa layer was composed of a thin layer of connective tissue and a cubical mesothelium.
FIGURE 4| Scanning Electron Microscopy (SEM) microphotographs of the digestive tract of Paracheirodon axelrodi. A-B. SEM view of mucosal surface showing a polyhedral surface of the epithelial cells in two different sections of the stomach. C. SEM view of mucosal surface of the intestine showing the brush border (Bb) present in the apical portion of the epithelium and abundant mucous secretion (red asterisk) from the GCs. D. Detail with higher magnification of C, in which the Bb is better defined.
TABLE 1 | Qualitative analysis of epithelial mucous cells based on staining intensity to Periodic Acid-Schiff (PAS) and Alcian Blue (AB) pH = 2,5. (–) not stained, (+) poorly stained, (++) strongly stained.
Mouth | Esophagus | Stomach | Caecum | Anterior
intestine | Posterior
intestine | |
PAS | + | ++ | – | + | + | ++ |
AB | + | – | – | + | + | ++ |
Liver and pancreas.The hepatocytes were polyhedral, with a granular and poorly stained cytoplasm. Their nuclei were spherical and centrally located presenting well delineated nucleoli. The liver was highly irrigated, as numerous arteries and veins were seen in the tissue. The liver was covered by a thin capsule of simple squamous epithelium and a thin adjacent layer of connective tissue (Fig. 3G).
Histologically, it was possible to distinguish an exocrine and an endocrine portion in P. axelrodi pancreas (Figs. 3B,F,G). The exocrine portion had two forms, (1) disseminated in the mesentery around the intestine and (2) intrahepatically around branches of the portal vein, forming what is known as hepatopancreas. Acini are the enzyme-secreting units of the exocrine pancreas. In P. axelrodi,each acinus consists of an ovoid cluster of pyramid-shaped secretory cells (acinar cells) surrounding a common luminal space. The acinar cells had a central spherical nucleus with one nucleolus, a basophilic basal portion, and an eosinophilic apical portion with zymogen granules. Occasionally, it was possible to observe next to the acini, pancreatic ducts with cuboidal epithelium. The endocrine pancreas consisted of areas scattered through the exocrine part. This endocrine tissue forms the so-called Islets of Langerhans (Fig. 3F) which were composed of small cells with a pale stained granular cytoplasm, in contrast to the larger protein-secreting cells of the exocrine gland, which stained more strongly.
The results of the histochemical analysis, performed in a qualitative way, are shown in Tab. 1 based on staining intensity. The caeca and intestine showed a similar pattern of distribution of the mucous cells that were AB and PAS positive. Towards the last portion of the intestine, the number of AB and PAS positive mucous secreting cells increased in number and staining intensity (Figs. 3C,D,F).
Discussion
Depending on the diet, the DT of the different teleost species shows a wide variation between species, as it plays an important role in the digestion and absorption of nutrient ingredients (Murray et al., 1996). For this reason, studies of the morphology and histology of DT are considered an effective tool when it comes to understand the mechanisms of ingestion, digestion, and absorption in fish (Xiong et al., 2011). The anatomy and histology of the digestive tract have been described for some species of the Characidae family (Rodrigues, Menin, 2008; Alonso et al., 2015). More specifically for P. axelrodi, only a preliminary description of its digestive tract has been published (Gómez-Ramírez et al., 2011). In general terms, the DT of P. axelrodi presents a relatively conserved general pattern with respect to that described in other species of the Characidae family, especially at the anatomical and histological level. However, when comparing P. axelrodi to other Characids, this species shows certain variations that can be highlighted, especially about the characteristics observed in mouth and teeth, esophageal mucosa and number and disposition of intestinal caeca.
The mouth mucosa in P. axelrodi presents a stratified squamous epithelium, a submucosa with loose connective tissue, and a muscular layer composed of striated muscle fibers, same as observed by Gómez-Ramírez et al. (2011). As described by Weitzman, Fink (1983), the mouth of P. axelrodi has a single row of five premaxillary teeth and one maxillary tooth, each of the with five cusps. This arrangement of the teeth is similar to what has been described in Phycocharax rasbora Ohara, Mirande & Lima, 2017, another species of the Characidae family (Ohara et al., 2017). However, a greater variation in tooth placement has been observed in related species such as Paracheirodon innesi (Myers, 1936). For example, wild-caught specimens of P. innesi have a slightly irregular series of six or seven tricuspid teeth with one or more teeth offset anteriorly to other teeth of the row (Géry, 1960; Weitzman, Fink, 1983). In fish, the oral and oropharyngeal cavities play important roles in selecting, seizing, and directing food to the esophagus (Al-Hussaini, 1946; Rodrigues et al., 2006; Wilson, Castro, 2010). Anatomical and histological studies enable us to relate those cavities to the eating habits (Al-Hussaini, 1946) and the food processing mechanisms (Rodrigues et al., 2006; Hanke et al., 2008). Considering these characteristics reported in terms of the mouth and teeth, it could be suggested that P. axelrodi is a species that swallows whole but small prey (Walker, 2004; Weitzman, Fink, 1983).
In the DT, the esophagus, stomach, intestine, and pyloric caeca are tubular organs that enable food conveyance, storage, digestion, and absorption (Seixas-Filho et al., 2000; Albrecht et al., 2001; Rodrigues, Menin, 2008; Wilson, Castro, 2010). All components of the DT present a wall consisting of four layers: mucosa, submucosa, muscularis and serosa (Albrecht et al., 2001; Wilson, Castro, 2010; Xiong et al., 2011). Histological studies have shown that the structure of the four layer varies among species, mostly in the epithelium, and that the differences are related to the feeding habits (Albrecht et al., 2001), type of food ingested (Germano et al., 2014), absorption and production of mucosubstances (Albrecht et al., 2001; Santos et al., 2007).
The esophageal mucosa of P. axelrodi is similar to that of other teleost and other species of the Characidae family, which is characterized by having a pseudostratified epithelium with many folds and abundant mucosal cells (Rodrigues, Menin, 2008; Alonso et al., 2015). This tissue conformation of the mucosa of the esophagus avoids damage caused by ingesting live prey or food with a hard consistency (Olaya et al., 2007). However, this is different from that observed by Gómez-Ramírez et al. (2011), where it is described that the mucosa of the esophagus of P. axelrodi is composed of stratified epithelium. The goblet cells essentially have two functions: first, to secrete mucopolysaccharides for lubrication, and second, to produce enzymatic cofactors or hydrolytic enzymes (Albrecht et al., 2001; Olaya et al., 2007; Santos et al., 2007; Feist, 2009; Xiong et al., 2011). The lamina propria is formed of loose connective tissue, surrounded by a muscular tunica, with an inner longitudinal and an external circular layer of striated muscle fibers, like that of other teleosts and other species of the Characidae family, as Astyanax rutilus (Jenyns, 1842) and Salminus brasiliensis (Cuvier, 1817) (Alonso et al., 2015; Rodrigues, Menin, 2008). It has been described that the conformation of the muscular tunica into two orientations allows the stretching of the organ, thus giving a greater capacity for distension (Olaya et al., 2007). By having striated muscle fibers, it allows a controlled movement which is important to avoid excessive water intake and regurgitation of unwanted food (Muñoz et al., 2006; Olaya et al., 2007; Wilson, Castro, 2010).
The stomach of teleosts presents a variety of different morphologies. In some species, the entire intestine is a tube of uniform diameter with no marked anatomical differences between the esophagus and stomach, while in other species, the stomach is seen as a round, muscular structure at the end of the esophagus (Feist, 2009). Mohsin (1962) stated that the stomach can be considered as the DT organ that shows the most diet-related adaptations. For example, in some species of carnivorous Characiformes different adaptations of the stomach are observed, mainly regarding the size of the cardiac and cecal regions. This is the case of Hoplias malabaricus (Bloch, 1794), in which the cardiac region is the largest, of Acestrorhynchus britskii Menezes, 1969, and Acestrorhynchus lacustris (Lütken, 1875), where the cecal region is the largest compared to the other gastric regions (Menin, Mimura, 1993). In the case of the stomach of P. axelrodi, it displays a u-shape, with defined cardiac, fundic and pyloric regions, similar to other species of the Characidae family with a carnivore diet, such as S. brasiliensis (Rodrigues, Menin, 2008), Aphyocharax anisitsi Eigenmann & Kennedy, 1903, Cheirodon interruptus (Jenyns, 1842), among others (Escalante, 1987; Cazorla et al., 2003; Alonso et al., 2015).
The cardiac region of the stomach in P. axelrodi presents a simple epithelium with tubular-acinous short glands and a circular layer of striated muscle. The fundic region of the stomach presents long ramified tubular glands surrounded by a loose connective tissue and a muscular layer that exhibit smooth muscle in two different orientations. The gastric glands consisted of oxintopeptic cells, secreting H+, Cl– and pepsinogen (Feist, 2009). In the most caudal portion of the stomach, the pyloric region of the stomach, there is a great development of the smooth muscle and lack of tubular-acinous glands. According to what has been described, the orientation in two directions of the muscular layer of the stomach facilitates that the peristaltic movements are carried out in several directions, and that the enzymatic action is more efficient during the digestion process, which ensures the breakdown of food before reaching the intestine (Olaya et al., 2007; Gómez-Ramírez et al., 2011).
The pyloric caeca in P. axelrodi are in the first portion of the intestine that continues after the stomach and present six folds, mainly in the longitudinal direction, in contrast with those in the rest of the intestine, that are mainly transverse. It has been widely described that the presence of these folds probably facilitates its filling and emptying in a bidirectional way and their main function is nutrient absorption. These are filled and emptied together with the first portion of the intestine, increasing the whole absorptive surface (Feist, 2009; Canan et al., 2012; Alonso et al., 2015). On the other hand, feeding habits in fish generally correlate with intestine length, for example, carnivorous species tend to have shorter intestines than herbivorous fish (Feist, 2009). The intestinal coefficient (Ci) is widely used to classify species into trophic categories. A Ci value below one indicates a carnivorous diet, between one and three indicates an omnivorous diet, and above three an herbivorous or detritivorous diet (Ward-Campbell et al., 2005). In this sense, the Ci: 0.94 obtained for P. axelrodi is within the expected range for a carnivorous species. Similarly to observed in P. axelrodi, Alonso et al. (2015) describe that A. anisitsi, A. rutilus and Characidium borellii (Boulenger, 1895),other three species of the Characidae family, present a low number of intestinal caeca and reduced intestine. For example, A. anisitsi feeds mainly on aquatic insects during the rainy season and terrestrial insects in the dry season (Corrêa et al., 2009) while in the Upper Paraná River floodplain in Brazil, this species fed mainly on microcrustaceans (Cladocera and Copepoda). In the case of C. borellii, although there is no information on diet, other Characidium species show a carnivorous diet too (Aranha et al., 2000; Bastos et al., 2013). Even though A. rutilus has an omnivorous diet for example, the main items ingested by this species are terrestrial insects and microcrustaceans and the development of intestinal caeca and intestine is also moderate (Alonso et al., 2015).
Evidence on ecology and feeding habits suggests that P. axelrodi, depending on changes in the water level, utilizes different habitats and capitalizes omnivorously on a diverse selection of food resources, including algae (Geisler, Annibal, 1987; Marshall et al., 2008). However, in another study in which examining stomach contents of P. axelrodi, authors suggest that the cardinal is principally a predator of small invertebrate prey, feeding mainly on microcrustaceans and chironomid larvae (Walker, 2004). So, considering the characteristics observed in terms of the mouth and teeth, the presence and reduced number of pyloric caeca, the Ci value below one, and the dietary studies in the wild in this species previously reported by Walker (2004), together this evidence supports P. axelrodi as a primarily carnivorous species.
On the other hand, it should be considered that the feeding habits of P. axelrodi may be highly influenced by the adaptive challenges it faces in its habitat and by the fact that it evolved as a miniature species. For example, Geisler, Annibal (1984) have compared the lengths of fish held in aquarium versus wild individuals of P. axelrodi, noting that individuals of this species rarely live more than a year in the wild, and that poor nutrition in its natural habitat further limits the adult size of this species (Weitzman, Vari, 1988). Furthermore, Alonso et al. (2015) suggest that herbivorous species have a larger part of their body dedicated to feeding and consequently it can be expected from this that miniature species such as P. axelrodi are more likely to evolve in “carnivorous clades”.
In teleost fish, there is no separation between the small and large intestine; there is just an undifferentiated tube. The intestinal portions that contain more complex mucosa are generally involved in absorptive processes. The intestinal epithelium of the simple or pseudostratified columnar type, is made up of cells possessing a well-marked striated border (microvilli) and goblet (mucus-secreting) cells. The former is both absorptive and secretory in function (Feist, 2009; Canan et al., 2012). The intestine of P. axelrodi is characterized by a simple columnar epithelium with a brush border and mucus secreting goblet cells with PAS positive glycoconjugates. Gómez-Ramírez et al. (2011) observed an increase in goblet cells towards the posterior part of the intestine of P. axelrodi, which is a mechanism of protection of the epithelium and lubrication to allow the passage of feces (Muñoz et al., 2006).
The liver parenchyma of P. axelrodi is composed primarily of hepatocytes, the cells are polyhedral in shape, have central nuclei and are variable in cytoplasmic content due to their ability to deposit either lipids or glycogen (Dyková et al., 2022). As previously described in a great variety of teleost fishes, the liver is the largest of the extramural organs (outside of the digestive tract) and fulfills important functions that include nutrient assimilation, bile production, detoxification, and maintenance of the body metabolic homeostasis. The liver also plays a role in the synthesis of plasma proteins, such as albumin, fibrinogen, and complementary factors (Feist, 2009; Dyková et al., 2022).
The pancreas of P. axelrodi has a structure comparable to that described in many other teleost and in the Characidae (Alonso et al., 2015), which is characterized by being diffuse throughout the body cavity and surrounding various organs, especially the digestive tract. As described in teleost, the pancreas is the gland responsible for the storage and secretion of various important digestive enzymes, and generally is present in two morphologically and functionally distinct forms, exocrine and endocrine (Feist, 2009; Mokhtar, 2015; Dyková et al., 2022). The present study revealed that the exocrine portion of the P. axelrodi pancreas has two forms, disseminated in the mesentery around the intestine and intrahepatic around the branches of the portal vein, forming what is known as hepatopancreas. The exocrine pancreas of P. axelrodi consists of clusters of pyramidal cells organized primarily into acini that have a cytoplasm with many large granules of eosinophilic zymogen. It has been described that zymogen granules often contain enzymes responsible for the digestion of proteins, carbohydrates, fats, and nucleotides (Alboughobish, Khaksari-Mahabadi, 2005; Feist, 2009; Mokhtar, 2015). The endocrine pancreas of P. axelrodi is organized into the Islets of Langerhans, as has been described for other teleost species. These islets are of endodermal origin and are formed by clusters of hormone-secreting cells (insulin, glucagon, somatostatin) surrounded by a capillaries network. Because these cell types secrete peptide hormones, they exhibit the usual features of cells engaged in active protein synthesis. The size of islet cells may vary according to food and season (Feist, 2009; Mokhtar, 2015).
In summary, this work presents a complete description of all the components of P. axelrodi DT including its anatomical arrangement, the description of adjacent glands involved in digestion (liver and pancreas), and the presence of the pyloric caeca that had not been previously observed or described in this species. The characteristics observed in the mouth and teeth, the presence of a reduced number of pyloric caeca and the value obtained for the intestinal coefficient (Ci) are compatible with a carnivorous diet, as observed previously in the diet of this species in the wild (Walker, 2004). The DT of P. axelrodi presented a relatively conserved general pattern within that described for other species of the Characidae family, especially at the histological level (Rodrigues, Menin, 2008; Alonso et al., 2015).
Acknowledgments
This study was supported by grants from Consejo Nacional de Ciencias y Tecnológicas (PIP 11220200100047CO) and Universidad de Buenos Aires (UBACyT 20020190100010BA).
References
Alabssawy AN, Khalaf-Allah HMM, Gafar AA. Anatomical and histological adaptations of the digestive tract in relation to food and feeding habits of lizardfish, Synodus variegatus (Lacepède, 1803). Egypt J Aquat Res. 2019; 45(2):159–65. https://doi.org/10.1016/j.ejar.2019.05.006
Alboughobish N, Khaksari Mahabadi M. Histological study of liver and pancreas in Ctenopharyngodon idella. Sc-Res Iran Vet J. 2005; 9(11):25–34.
Albrecht MP, Ferreira MFN, Caramaschi EP. Anatomical features and histology of the digestive tract of two related Neotropical omnivorous fishes (Characiformes; Anostomidae). J Fish Biol. 2001;58(2):419–30. https://doi.org/10.1111/j.1095-8649.2001.tb02261.x
Al-Hussaini AH. On the functional morphology of the alimentary tract of some fish in relation to differences in their feeding habits: anatomy and histology. J Cell Sci. 1946; 3(10):109–39.
Alonso F, Mirande JM, Pandolfi M. Gross anatomy and histology of the alimentary tract of Characidae (Teleostei: Ostariophysi: Characiformes) and potential phylogenetic information. Neotrop Ichthyol. 2015; 13(2):273–86. https://doi.org/10.1590/1982-0224-20140137
Anjos H, Anjos C. Biologia reprodutiva e desenvolvimento embrionario e larval do cardinal tetra, Paracheirodon axelrodi Schultz, 1956 (Characiformes: Characidae), em laboratorio. B Inst Pesca. 2006; 32:151–60.
Aranha JMR, Gomes JHC, Fogaça FNO. Feeding of two sympatric species of Characidium, C. lanei and C. pterostictum (Characidiinae) in a coastal stream of Atlantic Forest (Southern Brazil). Braz Arch Biol Technol. 2000; 43(5):527–31. https://doi.org/10.1590/S1516-89132000000500013
Banan Khojasteh SM. The morphology of the post-gastric alimentary canal in teleost fishes: a brief review. Int J Aquat Sci. 2012; 3(2):71–88.
Banan Khojasteh SM, Sheikhzadeh F, Mohammadnejad D, Azami A. Histological, histochemical, and ultrastructural study of the intestine of rainbow trout (Oncorhynchus mykiss). World Appl Sci J.2009; 6(11):1525–31.
Barbieri G, Peret A, Verani JR. Notas sobre a adaptação do trato digestivo ao regime alimentar em espécies de peixes da região de São Carlos (SP) I: Quociente intestinal. Rev Bras Biol. 1994; 54:63–69.
Bastos RF, Miranda SF, Garcia AM. Dieta e estratégia alimentar de Characidium rachovii (Characiformes, Crenuchidae) em riachos de planície costeira do sul do Brasil. Iheringia Sér. Zool, 2013; 103(4):335–41. https://doi.org/10.1590/S0073-47212013000400001
Becker AG, Gonçalves JF, Garcia LO, Behr ER, Graça DL, Kurtz Filho M, Martins T, Baldisserotto B. Morphometric parameters comparisons of the digestive tract of four teleosts with different feeding habits. Ciência Rural. 2010;40(4):862–66. https://doi.org/10.1590/S0103-84782010005000049
Brito MFG, Bazzoli N. Oogenesis of the cardinal tetra Paracheirodon axelrodi Schultz (1956): A histological and histochemical study. Braz J Morphol Sci. 2009; 26(1):14–18.
Canan B, Nascimento WS, Silva NB, Chellappa S. Morphohistology of the digestive tract of the damsel fish Stegastes fuscus (Osteichthyes: Pomacentridae).Scientific World J. 2012; 787316. https://doi.org/10.1100/2012/787316
Cazorla AL, Durán W, Tejera L. Alimentación de la ictiofauna del río Sauce Grande, provincia de Buenos Aires, Argentina. Biología Acuática. 2003; 20:73–79.
Clarke AJ, Witcomb DM. A study of the histology and morphology of the digestive tract of the common eel (Anguilla anguilla). J Fish Biol. 1980; 16(2):159–70. https://doi.org/10.1111/j.1095-8649.1980.tb03695.x
Corrêa CE, Hahn NS, Delariva RL. Extreme trophic segregation between sympatric fish species: the case of small sized body Aphyocharax in the Brazilian Pantanal. Hydrobiologia. 2009; 635:57–65. https://doi.org/10.1007/s10750-009-9861-2
Díaz AO, García AM, Goldemberg AL. Glycoconjugates in the mucosa of the digestive tract of Cynoscion guatucupa: a histochemical study. Acta Histochem. 2008; 110(1):76–85. https://doi.org/10.1016/j.acthis.2007.08.002
Dyková I, Žák J, Blažek R, Reichard M, Součková K, Slabý O. Histology of major organ systems of Nothobranchius fishes: short-lived model species. J Vert Biol. 2022; 71(21074):21074.1–50. https://doi.org/10.25225/jvb.21074
Escalante AH. Dieta comparativa de Cheirodon interruptus (Osteichthyes, Characidae) en ambientes lénticos y lóticos de la Provincia de Buenos de Aires. Revista Mus La Plata, Sec Zool. 1987; 14:35–45.
Evers H-G, Pinnegar JK, Taylor MI. Where are they all from? – sources and sustainability in the ornamental freshwater fish trade. J Fish Biol. 2019; 94(6):909–16. https://doi.org/10.1111/jfb.13930
Feist SW. Atlas of fish histology – Edited by F. Genten, E. Terwinghe and A. Danguy. J Fish Biol. 2009; 75(3):757–58. https://doi.org/10.1111/j.1095-8649.2009.02318_3.x
Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: genera/species by family/subfamily [Internet]. San Francisco: California Academy of Sciences; 2022. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Fryer G, Iles TD. The cichlid fishes of the Great Lakes of Africa. Their biology and evolution. Edinburgh: Oliver, Boyd eds; 1972.
Geisler R, Annibal SR. Ecology of the cardinal-tetra Paracheirodon axelrodi (Pisces, Characoidea) in the river basin of the Rio Negro/Brazil as well as breeding-related factors. TFH Magazine. 1987; 3512:66–70.
Geisler R, Annibal SR. Okologie des Cardinal-Tetra Paracheirodon axelrodi (Pisces, Characoidea) im Stromgebiet des Rio Negro/Brasilien sowie zuchtrelevante Faktoren. Amazoniana. 1984; 91: 53–86. Available from: https://eurekamag.com/research/021/472/021472949
Germano RM, Stabille SR, Mari RB, Pereira JNB, Faglioni JRS, Miranda Neto MH. Morphological characteristics of the Pterodoras granulosus digestive tube (Valenciennes, 1821) (Osteichthyes, Doradidae). Acta Zool (Stockholm). 2014; 95(2):166–75. https://doi.org/10.1111/azo.12016
Géry J. Contributions to the study of Characoid fishes. 11. The generic position Hyphessobrycon innesi and Cheirodon axelrodi, with a review of the morphological affinities of some Cheirodontinae (Pisces-Cypriniformes). Bulletin of Aquatic Biology. 1960; 2(1):1–18.
Gómez-Ramírez E, Obando MJ, Tovar MO, Caldas ML, Hurtado H. Estudio histológico del tracto digestivo del neón cardenal Paracheirodon axelrodi (Characidae). Int J Morphol. 2011; 29(3):782–86. https://dx.doi.org/10.4067/S0717-95022011000300018
Hale PA. The morphology and histology of the digestive systems of two freshwater teleosts, Poecilia reticulata and Gasterosteus aculeatus. Proc Zool Soc. 1965;146(2):132–49. https://doi.org/10.1111/j.1469-7998.1965.tb05206.x
Hanke BV, Meyer AA, Oliveira E. Análise histológica de estruturas sensoriais de Centropomus parallelus (Poey, 1896) (Centropomidae) relacionadas ao hábito alimentar. RUBS. 2008; 1:16–23.
Hernández E, Figueroa J, Iregui C. Streptococcosis on a red tilapia, Oreochromis sp., farm: a case study. J Fish Dis. 2009; 32(3):247–52. https://doi.org/10.1111/j.1365-2761.2008.00981.x
Houssain AM, Dutta HM. Assessment of structural and functional similarities and differences between caeca of the bluegill. J Fish Biol. 1998; 53(6):1317–23. https://doi.org/10.1111/j.1095-8649.1998.tb00251.x
Kapoor BC, Smith H, Verighina IA. The alimentary canal and digestion in teleosts. Adv Mar Biol. 1975; 13:109–239. https://doi.org/10.1016/S0065-2881(08)60281-3
Langer M, Van Noorden S, Polak JM, Pearse AGE. Peptide hormone-like immunoreactivity in the gastrointestinal tract and endocrine pancreas of eleven teleost species. Cell Tissue Res. 1979; 199(3):493–508. https://doi.org/10.1007/BF00236085
Marshall BG, Forsberg BR, Thomé-Souza MJF. Autotrophic energy sources for Paracheirodon axelrodi (Osteichthyes, Characidae) in the middle Negro River, Central Amazon, Brazil. Hydrobiologia. 2008; 596: 95–103. https://doi.org/10.1007/s10750-007-9060-y
Menin E, Mimura OM. Anatomia comparativa do estômago de três peixes Teleostei de água-doce de hábito alimentar ictiófago. Revista Ceres. 1993; 40 (228):203–22.
Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes): from characters to taxonomy. Neotrop Ichthyol. 2010; 8(3):385–568. https://doi.org/10.1590/S1679-62252010000300001
Mohsin SM. Comparative morphology and histology of the alimentary canals in certain groups of Indian teleosts. Acta Zoologica. 1962; 43(1):79–133.
Mokhtar DM. Histological, histochemical and ultrastructural characterization of the pancreas of the grass carp (Ctenopharyngodon idella). Eur J Anat. 2015; 19(2):145–53.
Morrison CM, Wright Jr. JR. A study of the histology of the digestive tract of the Nile tilapia. J Fish Biol. 1999; 54(3):597–606. https://doi.org/10.1111/J.1095-8649.1999.TB00638.x
Morrison CM. Histology of the atlantic cod Gadus morhua:An atlas, digestive tract and associated organs. National Research Council of Canada: Ottawa, Canada; 1987.
Muñoz A, Caldas ML, Hurtado H. Análisis histomorfológico del sistema digestivo y glándulas anexas en alevinos de cachama blanca, Piaractus brachypomus (Characidae: Piaractus). Rev Fac Cs Basicas.2006; 2:137–64.
Murray HM, Wright GM, Goff GP. A comparative histological and histochemical study of the post-gastric alimentary canal from three species of pleuronectids, the Atlantic halibut, the yellowtail flounder and the winter flounder. J Fish Biol. 1996; 48(2):187–206. https://doi.org/10.1111/j.1095-8649.1996.tb01112.x
National Research Council (NRC). Committee for the update of the guide for the care and use of laboratory animals. Guide for the care and use of laboratory animals (8th ed.). Nat Ac Press; 2011.
Ohara WM, Mirande JM, Lima FCT. Phycocharax rasbora, a new genus and species of Brazilian tetra (Characiformes: Characidae) from Serra do Cachimbo, rio Tapajós basin. PLoS ONE. 2017; 12(2):e0170648. https://doi.org/10.1371/journal.pone.0170648
Olaya CM, Ovalle CH, Gómez E, Rodríguez D, Caldas ML, Hurtado H. Histología y morfometría del sistema digestivo del silúrido bagre tigrito (Pimelodus pictus). Rev Fac Med Vet Zoot. 2007; 54(2):311–23. Available from: https://www.redalyc.org/articulo.oa?id=407639214007
Ribeiro CAO, Fanta E. Microscopic morphology and histochemistry of the digestive system of a tropical freshwater fish Trichomycterus brasiliensis (Lutken) (Siluroidei, Trichomycteridae). Rev Bras Zool.2000; 17(4):953–71. https://doi.org/10.1590/S0101-81752000000400007
Rocha E, Monteiro RA, Pereira CA. The liver of the brown trout, Salmo trutta fario: a light and electron microscope study. J Anat. 1994; 185(2):241–49.
Rodrigues SS, Menin E. Anatomia do tubo digestivo de Salminus brasiliensis (Cuvier, 1817) (Pisces, Characidae, Salmininae). Biotemas 2008; 21(2):65–75. http://dx.doi.org/10.5007/2175-7925.2008v21n2p65
Rodrigues SS, Navarro RD, Menin E. Adaptações anatômicas da cavidade bucofaringiana de Leporinus macrocephalus Garavello & Britski, 1988 (Pisces, Characiformes, Anostomidae) em relação ao hábito alimentar. Biotemas. 2006; 19(1):41–50.
Rombout JHWM, Taverne-Thiele JJ. An immunocytochemical and electron-microscopical study of endocrine cells in the gut and pancreas of a stomachless teleost fish, Barbus conchonius (Cyprinidae). Cell Tissue Res.1982; 227:577–93. https://doi.org/10.1007/BF00204788
Santos CM, Duarte S, Souza TGL, Ribeiro TP, Sales A, Araújo FG. Histologia e caracterização histoquímica do tubo gastrintestinal de Pimelodus maculatus Pimelodidae, Siluriformes) no reservatório de Funil, Rio de Janeiro, Brasil. Iheringia Sér Zool. 2007; 97(4):411–17. https://doi.org/10.1590/S0073-47212007000400009
Santos M, Arantes F, Santiago K, Santos J. Morphological characteristics of the digestive tract of Schizodon knerii (Steindachner, 1875), (Characiformes: Anostomidae): An anatomical, histological, and histochemical study. An Acad Bras Ciênc. 2015; 87(2):867–78. https://doi.org/10.1590/0001-3765201520140230
Seixas-Filho JT, Brás JM, Gomide ATM, Oliveira MGA, Donzele JL, Menin E. Anatomia funcional e morfometria dos intestinos e cecos pilóricos do teleostei (Pisces) de água doce Brycon orbignyanus (Valenciennes, 1849). R Bras Zootec. 2000; 29(2):2181–92. https://doi.org/10.1590/S1516-35982000000200001
De Silva SS, Cumaranatunga PRT, Silva CD. Food, feeding ecology and morphological features of four co-occurring cyprinids (Pisces; Cyprinidae). Neth J Zool. 1980; 30:54–73.
Sinha GM. Comparative morphology, anatomy, and histology of the alimentary canal of an Indian freshwater major carp, Labeo calbasu (Hamilton) during the different life-history stages in relation to food and feeding habits. Anat Anz. 1976; 139(4):348–62.
Sis RF, Ives PJ, Jones DM, Lewis DH, Haensly WE. The microscopic anatomy of the oesophagus, stomach, and intestine of the channel catfish, Ictalurus punctatus. J Fish Biol. 1979; 14(2):179–86. https://doi.org/10.1111/j.1095-8649.1979.tb03508.x
Vari RP, Malabarba LR. Neotropical Ichthyology: an overview. In: Malabarba LR, Reis RE, Vari RP, Lucena ZMS, Lucena CAS, editors. Phylogeny and Classification of Neotropical Fishes. Porto Alegre: Edipucrs; 1998. p.7–11.
Walker I. The food spectrum of the cardinal – tetra (Paracheirodon axelrodi, Characidae) in its natural habitat. Acta Amaz. 2004; 34(1):69–73. https://doi.org/10.1590/S0044-59672004000100009
Ward-Campbell BMS, Beamish FWH, Kongchaiya C. Morphological characteristics in relation to diet in five coexisting Thai fish species. J Fish Biol. 2005; 67(5):1266–79. https://doi.org/10.1111/j.1095-8649.2005.00821.x
Weitzman SH, Fink W. Relationships of the neon tetras, a group of South American freshwater fishes (Teleostei, Characidae), with comments on the phylogeny of new world Characiformes. Bull Mus Comp Zool. 1983; 150(6):339–95.
Weitzman SH, Vari RP. Miniaturization in South American freshwater fishes; an overview and discussion. Proc Biol Soc Wash. 1988; 101(2):444–65. Available from: http://hdl.handle.net/10088/901
Wilson JM, Castro LFC. Morphological diversity of the gastrointestinal tract in fishes. In: Grossell M, Farrell AP, Brauner CJ, editors. The multifunctional gut of fish. New York: Academic Press, 2010. p.2–44.
Xiong D, Zhang L, Yu H, Xie C, Kong Y, Zeng Y, Huo B, Liu Z. A study of morphology and histology of the alimentary tract of Glyptosternum maculatum (Sisoridae, Siluriformes). Acta Zool. 2011; 92(2):161–69. https://doi.org/10.1111/j.1463-6395.2010.00458.x
Authors
![]() Laura Rincón1,
Laura Rincón1, ![]() Felipe Redondo1
Felipe Redondo1 ![]() ,
, ![]() Wendy Kobrinsky1,
Wendy Kobrinsky1, ![]() Matías Pandolfi1 and
Matías Pandolfi1 and ![]() Andrea G. Pozzi1
Andrea G. Pozzi1
[1] Laboratorio de Neuroendocrinología y Comportamiento en Peces y Anfibios, Departamento de Biodiversidad y BiologíaExperimental, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires. Instituto de Biodiversidad y BiologíaExperimental y Aplicada-CONICET. Ciudad Universitaria, Pabellón 2, 1428, Buenos Aires, Argentina. (LR) laura.rinconc27@gmail.com (corresponding author), (FR) felipe.redondo@yahoo.com.ar, (WK) wendy.ko@hotmail.com, (MP) pandolfi@bg.fcen.uba.ar, (AGP) pozziag@gmail.com
Authors’ Contribution 

Laura Rincón: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Felipe Redondo: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Writing-original draft.
Wendy Kobrinsky: Conceptualization, Data curation, Investigation, Methodology.
Matías Pandolfi: Conceptualization, Data curation, Funding acquisition, Project administration, Resources, Supervision.
Andrea G. Pozzi: Conceptualization, Data curation, Funding acquisition, Project administration, Resources, Supervision, Writing-review and editing.
Ethical Statement
All procedures described in the following sections were conducted in accordance with international standards (Guide for Care and Use of Laboratory Animals – NRC, 2011) on animal welfare as well as being compliant with local regulations (CICUAL, Comisión Institucional para el Cuidado y Uso de Animales de Laboratorio, Protocol # 75b).
Competing Interests
The author declares no competing interests.
How to cite this article
Rincón L, Redondo F, Kobrinsky W, Pandolfi M, Pozzi AG. Morphological study of the digestive tract of the cardinal tetra, Paracheirodon axelrodi (Characiformes: Characidae). Neotrop Ichthyol. 2023; 21(1):e220092. https://doi.org/10.1590/1982-0224-2022-0092
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted February 3, 2023 by Rosemara Fugi
Accepted February 3, 2023 by Rosemara Fugi
![]() Submitted September 21, 2022
Submitted September 21, 2022
![]() Epub March 13, 2023
Epub March 13, 2023