![]() Eduardo Mejia1
Eduardo Mejia1 ![]() ,
, ![]() Gustavo A. Ferraro1 and
Gustavo A. Ferraro1 and ![]() Paulo A. Buckup1
Paulo A. Buckup1
PDF: EN XML: EN | Supplementary: S1 S2 S3 | Cite this article
Abstract
A new species of Rineloricaria is described from the Iguaçu, Magé, Saracuruna, Guapiaçu, Macacu, Macaé, and Paraíba do Sul River basins in southeastern Brazil based on morphological and molecular data. The new species is diagnosed among congeners by the combination of the following characters: absence of plates in most of the ventral surface of pectoral girdle; snout tip with elliptical naked area reaching posteriorly the anteriormost pore of the infraorbital ramus of sensory canal; dorsal-fin spinelet present; supraoccipital and predorsal plates with smooth keels; five series of lateral plates below the dorsal fin; median and mid-ventral series of plates with well-developed keels. The new species is distinguished from congeners that inhabit the rivers draining the Serra do Mar in Rio de Janeiro state by the partial plate coverage of the ventral surface of the pectoral girdle and the abdomen (vs. pectoral girdle and abdomen completely covered by plates in Rineloricaria nigricauda, R. steindachneri,and R. zawadzkii). The new species shares the absence of plates on the abdominal surface with other species from the Paraná, Uruguay, and other Brazilian coastal river basins.
Keywords: DNA barcode, Freshwater fishes, Loricariinae, Suckermouth armored catfishes, Integrative taxonomy.
Uma nova espécie de Rineloricaria é descrita das bacias dos rios Iguaçu, Magé, Saracuruna, Guapiaçu, Macacu, Macaé e Paraíba do Sul, no sudeste do Brasil, com base em caracteres morfológicos e moleculares. A nova espécie é diagnosticada entre as congêneres pela combinação dos seguintes caracteres: ausência de placas na maior parte da superfície ventral da cintura peitoral; extremidade do focinho com área nua elíptica estendida posteriormente, alcançando o poro mais anterior do canal sensorial infraorbital; presença de spinelet na origem da nadadeira dorsal; placas supraoccipitais e pré-dorsais com quilhas pouco proeminentes; cinco séries de placas laterais abaixo da origem da nadadeira dorsal; placas das séries mediana e médio-ventral com quilhas bem desenvolvidas. A nova espécie distingue-se das congêneres que habitam os rios que drenam a Serra do Mar no Estado do Rio de Janeiro pela cobertura parcial de placas da superfície ventral da cintura peitoral e do abdômen (vs. cintura peitoral e abdômen completamente cobertos por placas em Rineloricaria nigricauda, R. steindachneri e R. zawadzkii). A nova espécie compartilha a ausência de placas na superfície abdominal com outras espécies das bacias hidrográficas dos rios Paraná e Uruguai e rios costeiros do Brasil.
Palavras-chave: Cascudos, Códigos de barra de DNA, Loricariinae, Peixes de água doce, Taxonomia integrativa.
Introduction
The genus Rineloricaria Bleeker, 1862, includes 69 valid species of armored catfishes (Fricke et al., 2022) widely distributed in Central and South America from Costa Rica to Argentina, on both sides of the Andes. They inhabit large rivers, streams, and ponds, associated with sandy or rocky bottoms, from high mountain streams to large floodplain rivers (Reis, Cardoso, 2001; Covain, Fisch-Muller, 2007; Fichberg, Chamon, 2008). Rineloricaria is distinguished from other Loricariinae by the following characters: postorbital notch present; surface of lower lip with short round papillae; premaxilla with seven to 15 teeth on each ramus; dentary teeth strong, deeply bicuspidate; dorsal region with dark brown bars or blotches with the first one at the origin of dorsal fin; dorsal fin usually with the first element as a spinelet; abdomen with a polygonal preanal plate, usually bordered by three large trapezoidal plates (Fichberg, Chamon, 2008). In addition, there are features associated with sexual dimorphism: in most species mature males possess numerous hypertrophied odontodes along the sides of the head, on the dorsal surface of pectoral-fin, and predorsal area (Isbrücker, Nijssen, 1992; Fichberg, Chamon, 2008).
In the last two decades, 23 new species of Rineloricaria have been described (Fricke et al., 2022). However, as in other highly diverse and widely distributed genera of Loricariidae, several species remain undescribed. In addition, older descriptions are incomplete and poorly informative (Reis, Cardoso, 2001; Ingenito et al., 2008). Recent studies based on DNA sequences of coastal drainages of southern Brazil and the upper Paraná River suggest that several candidate species need to be evaluated and eventually described (Costa-Silva et al., 2015). Accordingly, our ichthyological surveys in the rivers draining the coastal slope of the Serra do Mar, in the central region of Rio de Janeiro State, revealed an undescribed Rineloricaria species from the Iguaçu, Magé, Saracuruna, Guapiaçu, Macacu, Macaé and Paraíba do Sul River basins. This new species is described herein.
Material and methods
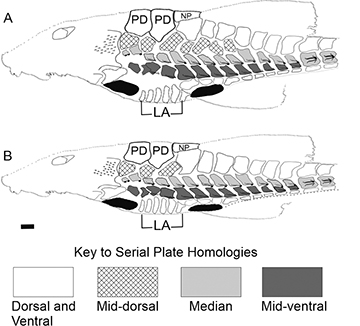
Measurements and counts of bilateral structures were obtained from the left side of the body; otherwise, in cases of damaged structures on the left side, data were obtained from the right side. Measurements followed Isbrücker, Nijssen (1978) and Vera-Alcaraz et al. (2012). All measurements were taken point-to-point to the nearest 0.1 mm, with digital calipers under stereomicroscope when necessary. The body measurements were expressed as proportions of standard length (SL), except for subunits of the head, which were expressed as proportions of head length (HL). Terminology and counts of the series of lateral plates followed Schaefer (1997). Series of plates were identified and counted as illustrated in Fig. 1. The median plate series were identified by the presence of lateral-line canal in each plate (bearing a single odontode ridge) and extend between the compound pterotic bone and the posterior end of the caudal peduncle, including the posterior lateral plates which bear a double odontode ridge. The remaining plate series were identified based on their extent and position relative to adjacent series (Fig. 1). Nomenclature of posterior, median, and anterior abdominal plate complex follows Isbrücker, Nijssen (1979).
FIGURE 1| Schematic illustration of lateral plate series in species of Rineloricaria with (A) extended (described in this study) and (B) short (e.g., R. steindachneri) mid-dorsal series. The black areas indicate the insertion of the pectoral and pelvic fins. LA: lateral abdominal plates; PD: predorsal plates; NP: nuchal plate. Scale bar = 2 mm.
In the description, numbers in parentheses represent the total number of specimens with the associated counts, and asterisks indicate counts of the holotype. Specimens that were directly examined are listed under comparative material. Morphological and meristic characters for species not represented by examined specimens were taken from published species descriptions, and images of type specimens available from the All Catfish Species Inventory Image Base (Morris et al., 2006). To ensure uniform representation, the specimens for the type series were selected from a single location in the Macacu River basin, which was sampled multiple times in different years. Institutional abbreviations follow Sabaj (2020).
To delineate the new species using molecular data, DNA was extracted from muscle tissue of selected specimens preserved in the field using anhydrous ethanol. Genomic DNA was extracted using the salting out method (Miller et al., 1988). DNA concentration and quality were verified using a NanoDrop ND-2000 spectrophotometer. Partial sequences of the mitochondrial cytochrome oxidase subunit I (COI) gene were amplified with Polymerase Chain Reaction (PCR) using primers FishF1 (5’-TCA ACC AAC CAC AAA GAC ATT GGC AC-3’) and FishR1 (5’-TAG ACT TCT GGG TGG CCA AAG AAT CA-3’) (Ward et al., 2005), and FishF6 (5’-TGT AAA ACG ACG GCC AGT ACY AAY CAC AAA GAY ATT GGC A-3’) and FishR7 (5’-CAG GAA ACA GCT ATG ACC TAR ACT TCT GGR TGD CCR AAG AAY CA-3’) which included M13 sequencing primer sequences appended (Jennings et al., 2019). The PCR protocol for both primers was as follows: denaturation at 94°C/30 s, 35 cycles of 94°C/45 s, 50°C/30 s and 72°C/45 s, and a final step of 72°C/10 min. Quality of PCR products was checked by electrophoresis in agarose gel 2%. Amplified products were purified using Exo-SAP (Handy et al., 2011), and each PCR product was bidirectionally sequenced with the Sanger method, on an automated sequencer ABI3730xl (Applied Biosystems) at the Fundação Oswaldo Cruz (FIOCRUZ). The forward and reverse sequencing chromatographs were manually edited and trimmed in Geneious v. 7.1.3.0 (https://www.geneious.com; Kearse et al., 2012) using reference sequences of Rineloricaria. A Barcode Index Numbers (BIN) (Ratnasingham, Hebert, 2013) was generated for the final sequences using the algorithm available in Barcode of Life Data Systems (BOLD Systems, http://boldsystems.org/, Ratnasingham, Hebert, 2007).
To establish the phylogenetic position of the new species, consensus sequences were aligned with 223 sequences of cytochrome oxidase (COI) from Costa-Silva et al. (2015) (Tab. S1) using the Muscle algorithm (Edgar, 2004) in MEGA X (Kumar et al., 2018). The best-fit evolutionary model (GTR+G+I) was selected using the Akaike information criterion (AIC) and was then used to generate a Maximum Likelihood (ML) tree (Fig. S2). Additionally, the genetic divergence among samples was estimated under the Kimura 2-parameter model (K2P) (Tab. S3) with MEGA X (Kumar et al., 2018) in order to evaluate the genetic variation among populations of the new species from different basins following DNA barcoding standards for fish (Pereira et al., 2013). New nucleotide sequences and associated data, including geospatial coordinates, trace files, and primer details are available in the BOLD Systems V.4 database under their respective Sample ID and Process ID codes (Tab. 1).
TABLE 1 | BOLD Process ID and GeneBank Accession codes for samples of Rineloricaria nudipectoris used to obtain molecular data in this study. Sample ID refers to the DNA Extract Collection of the Laboratório de Pesquisa em Biodiversidade Molecular, Museu Nacional (MNLM) which are also used to record specimen data in BoldSystems (http://www.boldsystems.org/). Voucher code refers to the whole-organism catalog numbers, and Specimen/Tissue code refers to tissue catalog number in the Fish Tissue Collection.
Locality | Voucher | BOLD | BOLD | Genbank Accession |
Paraíba do Sul | MNRJ 51642 | MNLM9287 | MNRJ1308-22 | OP270252 |
Paraíba do Sul | MNRJ 51642 | MNLM9288 | MNRJ1309-22 | OP270253 |
Macaé | MNRJ 50750 | MNLM8195 | MNRJ1026-18 | MH272130 |
Macaé | MNRJ 50807 | MNLM8219 | MNRJ1027-18 | MH272135 |
Macacu | MNRJ 43536 | MNLM6416 | MNRJ787-16 | MH272131 |
Macacu | MNRJ 43536 | MNLM6417 | MNRJ788-16 | MH272133 |
Macacu | MNRJ 43536 | MNLM6418 | MNRJ789-16 | MH272128 |
Macacu | MNRJ 43536 | MNLM6419 | MNRJ790-16 | MH272132 |
Guapiaçu | MNRJ 42451 | MNLM7884 | MNRJ1025-18 | MH272134 |
Iguaçu | MNRJ 43426 | MNLM6404 | MNRJ783-16 | MH272129.1 |
Results
Rineloricaria nudipectoris, new species
urn:lsid:zoobank.org:act:BEFD2515-D72B-425D-AAF0-E1AFF2AA4A9E
(Figs. 2–3, 4A–B; Tab. 2)
Holotype. MNRJ 49345, 88.8 mm SL, male, Brazil, Rio de Janeiro, Cachoeiras de Macacu, rio Macacu behind the Riviera III restaurant at RJ-116 highway, 22°29’01”S 42°39’42”W, 13 Nov 2014, P. A. Buckup, D. F. M. Moraes Jr. & G. L. Vergara.
Paratypes. All from Brazil, Rio de Janeiro State, rio Macacu basin, collected in the same locality as the holotype: MCP 54888, 5, 85.1–96.3 mm SL, 4 Sep 1996, G. W. Nunan, K. M. Grosser, W. Koch & D. F. Moraes. MNRJ 14844, 174, 31.2–113.6 mm SL (27, 83.7–113.6 mm SL), 4 Sep 1996, G. W. Nunan, K. M. Grosser, W. Koch & D. F. Moraes. MNRJ 37103, 12, 36.9–59.8 mm SL, 27 Apr 2010, P. A. Buckup, F. L. K. Salgado, J. R. Gomes, C. Cramer, A. P. C. Rodrigues & A. R. L. Ferreira. MNRJ 43536, 14, 59.5–100.5 mm SL (6, 76.3–98.3 mm SL), collected with the holotype. MNRJ 48493, 9, 60.4–98.6 mm SL (5, 72.6–103.9 mm SL), 27 Apr 2010, P. A. Buckup, F. L. K. Salgado, J. R. Gomes & C. Cramer. MZUSP 126999, 5, 81.5–95.9 mm SL, 4 Sep 1996, G. W. Nunan, K. M. Grosser, W. Koch & D. F. Moraes.
FIGURE 2| Rineloricaria nudipectoris, holotype, MNRJ 49345, male, 88.8 mm SL, Brazil, Cachoeiras de Macacu, rio Macacu; dorsal, lateral, and ventral views.
Non-types. All from Brazil, Rio de Janeiro State. Rio Macaé basin: MNRJ 28472, 1, 115.2 mm SL, Casimiro de Abreu, rio Macaé, about 2 km downstream of Galico farmstead, 22°25’58”S 42°11’15”W, 5 Nov 2003, M. F. G. Brito. MNRJ 28474, 1, 94.2 mm SL, Nova Friburgo, Lumiar, rio Macaé, at the mouth of rio Santiago, 22°21’33”S 42°21’22”W, 6 Dec 2002, M. F. G. Brito. MNRJ 28476, 2, 100.4–112.4 mm SL, Nova Friburgo, Lumiar, rio Macaé, at Poço Feio, 22°21’05”S 42°19’56”W, 6 Dec 2002, M. F. G. Brito. MNRJ 50750, 15, 48.9–115.8 mm SL, Nova Friburgo, rio Macaé, in front of Campinho Doce campsite, downstream of Lumiar, 22°21’27”S 42°18’54”W, 27 Jun 2017, P. A. Buckup, D. F. Moraes Jr., V. Brito, I. C. A. S. Santos & G. A. Ferraro. MNRJ 50807, 1, 82.3 mm SL, Casimiro de Abreu, rio Macaé near Casimiro de Abreu, Sana highway, 22°25’19”S 42°12’27”W, 28 Jun 2017, P. A. Buckup, D. F. Moraes Jr., V. Brito, I. C. A. S. Santos & G. A. Ferraro. MNRJ 51075, 7, 70.5–118.7 mm SL, Nova Friburgo, rio Macaé, in front of Campinho Doce campsite, downstream of Lumiar, 22°21’27”S 42°18’54”W, 27 Jun 2017, P. A. Buckup, V. De Brito, E. Malanski, R. M. Dias, V. Brito & I. C. A. S. Santos. Rio Macacu basin (upstream of confluence with rio Guapiaçu): MNRJ 13741, 12, 73.4–91.2 mm SL, Cachoeiras de Macacu, rio Branco, left bank tributary of rio Bengala, 22°33’55”S 42°37’43”W, 21 Mar 1995, P. A. Buckup & D. F. Moraes Jr. MNRJ 23829, 6, 42.6–87.7 mm SL, Cachoeiras de Macacu, rio Macacu downstream of Cachoeiras de Macacu, near the cemetery on RJ-122 highway, 22°28’43”S 42°39’23”W, 9 Apr 1996, D. F. Moraes, W. Koch, K. Grosser & M. R. Fernandes. MNRJ 23830, 4, 63.3–90.6 mm SL, Cachoeiras de Macacu, rio Soarinho at bairro de Granada, locality of Papucaia, 22°37’00”S 42°43’00”W, 21 Jul 2001, M. R. S. Melo & I. Silveira. MNRJ 23832, 2, 98.6–107.8 mm SL, Cachoeiras de Macacu, rio Macacu, 22°36’07”S 42°45’03”W, 21 Jul 2001, M. R. S. Melo. MNRJ 47199, 9, 1 cs, 72.3–91.2 mm SL, Cachoeiras de Macacu, Rio Branco, access to Jororo road east of Japuíba, 22°33’37”S 42°40’34”W, 28 Apr 2016, P. A. Buckup, M. R. Britto, C. R. Moreira, D. F. Moraes, S. A. Santos & G. S. Araujo. MNRJ 52324, 10, 48.7–72.2 mm SL, Cachoeiras de Macacu, rio Macacu, tributary west of Riviera III restaurant, 22°29’13”S 42°39’46”W, 30 Aug 2019, P. A. Buckup, I. Souto-Santos, G. A. Ferraro, K. C. Ferreira & T. F. de Barros. Rio Guapiaçu drainage: MNRJ 17797, 5, 53.9–93.5 mm SL, Guapimirim, rio Iconha at the bridge at Rancho Dom Pedrito, 22°31’00”S 42°58’04”W, 9 Nov 1998, F. A. G. Melo, A. R. Cardoso & T. A. Jorge. MNRJ 18024, 1, 78.7 mm SL, Guapimirim, rio Paraiso near the entrance of fazenda Iguaçu, 22°31’09”S 42°53’50”W, 10 Dec 1998, P. A. Buckup, F. A. G. Melo, M. R. S. Melo, L. M. Porto & M. C. Amorim. MNRJ 19621, 7, 68.1–94.6 mm SL, Cachoeiras de Macacu,rio Guapiaçu 5 km from RJ-122 highway at fazenda Santo Estevão FEEMA, 22°31’00”S 42°49’00”W, 28 Aug 1999, F. A. G. Melo & R. Rezende. MNRJ 20279, 5, 42.1–87.4 mm SL, Guapimirim, rio Iconha, dirt road marginal to rio Iconha, between Guapi-Mirim and RJ-122, 22°32’01”S 42°55’41”W, 25 Apr 2000, M. R. S. Melo, A. T. Aranda, F. A. G. Melo & G. Souza. MNRJ 23831, 17, 50.9–65.7 mm SL, Cachoeiras de Macacu, first bridge of rio Rabelo River crossing the road from Friburgo to Parada Modelo, 22°33’26.18”S 42°47’25.34”W, 29 May 1983, Ictiologia and Malacologia labs (MNRJ). MNRJ 26664, 2, 55.6–91.8 mm SL, Cachoeiras de Macacu, córrego do Gato at Serra do Mar farm, right margin of Guapiaçu, Guapi-Açu Private Ecological Reserve, 22°26’08”S 42°45’32”W, 15 Apr 2004, P. A. Buckup, R. Campos-da-Paz & M. Ghazzi. MNRJ 27749, 1, 75.2 mm SL, Cachoeiras de Macacu, córrego do Gato at Serra do Mar farm, tributary of the right bank of Guapiaçu, 22°26’08”S 42°45’32”W, 11 Apr 2003, L. M. Sarmento-Soares, J. H. C. Gomes & J. Soares Filho. MNRJ 27781, 4, 76.3–86.1 SL, Cachoeiras de Macacu, pool at rio Guapiaçu, next to Guapiaçu vilage, 22°27’13”S 42°46’20”W, 13 Apr 2003, P. A. Buckup & M. R. Britto. MNRJ 27805, 4, 89.3–103.9 mm SL, Cachoeiras de Macacu, córrego do Gato upstream of the bridge, 22°26’01”S 42°45’38”W, 1 May 2004, L. M. Sarmento-Soares, A. T. Aranda, R. Pinheiro, P. Lopes & R. Guedes. MNRJ 27826, 3, 73.2–89.6 mm SL, Cachoeiras de Macacu, rio Passa Vaca, tributary of Mariquita river, about 2 km upstream of Guapiaçu River, 22°25’54”S 42°44’59”W, 2 May 2004, A. T. Aranda, R. Guedes & R. Pinheiro. MNRJ 42451, 1, 76.8 mm SL, Guapimirim, rio Paraiso, access bridge to number 6660 of the Adutora Paraíso road, 22°30’14”S 42°53’35”W, 14 Jun 2012, P. A. Buckup, M. R. Britto, C. C. D. Quijada, G. A. Ferraro & J. C. Pascoli. MNRJ 48350, 1, 97.3 mm SL, Cachoeiras de Macacu, rio Rabelo, at road from Funchal to Guapiaçu, 22°31’01”S 42°46’43”W, 27 Apr 2010, P. A. Buckup, F. K. Salgado, J. R. Gomes, C. Cramer, A. P. C. Rodrigues & A. R. L. Ferreira. MNRJ 48495, 5, 61.1–83.6 mm SL, Cachoeiras de Macacu, rio Guapiaçu, at the access bridge to the Guapi-Açu Private Ecological Reserve, 22°27’26”S 42°46’01”W, 15 Jul 2016, E. B. Neuhaus & R. M. Dias. MNRJ 50652, 2, 76.1–99.7 mm SL, Cachoeiras de Macacu, rio Guapiaçu, on the bridge before the access to Guapiaçu Ecological Reserve, 22°27’25”S 42°46’01”W, 11 Jun 2017, E. B. Neuhaus & R. M. Dias. MNRJ 51247, 13, 34.9–100.5 mm SL, Cachoeiras de Macacu, rio Guapiaçu, downstream and upstream of the access bridge to the headquarters of the Guapi-Açu Ecological Reserve, 22°27’28”S 42°46’05”W, 14 Dec 2017, P. A. Buckup, V. de Brito, E. Malanski, R. M. Dias & E. Neuhaus. MNRJ 51263, 2, 95.5–105.1 mm SL, Cachoeiras de Macacu, rio do Gato, upstream of the bridge from the road to the researchers house at the Guapi-Açu Ecological Reserve, 22°26’08”S 42°45’33”W, 14 Dec 2017, P. A. Buckup, V. de Brito, E. Malanski, R. M. Dias & E. Neuhaus. MNRJ 51802, 2, 80.2–94.5 mm SL, Cachoeiras de Macacu, córrego do Gato, right bank tributary of rio Guapiaçu, 22°26’08”S 42°45’33”W, 30 Aug 2019, P. A. Buckup, I. C. A. Souto-Santos, F. A. Ferraro & M. D. V. Leão. Rio Magé basin: MNRJ 48823, 1, 111.4 mm SL, Magé, rio Santo Aleixo on the Magé-Piabetá highway bridge, 22°34’42”S 43°03’50”W, 19 May 2007, M. R. Britto, P. A. Buckup & L. Villa-Verde. Rio Saracuruna basin: MNRJ 31606, 7, 57.1–81.1 mm SL, Magé, rio Caiobá-Mirim, in Inhomirim, in front of Raiz da Serra cemetery, near the railroad, 22°34’40”S 43°11’05”W, 18 Sep 2007, L. Villa-Verde, J. R. Gomes & D. Muller. Rio Iguaçu basin: MNRJ 23849, 3, 66.1–86.1 mm SL, Nova Iguaçu, rio Ana Felícia, tributary of the left bank of rio Tinguá, at RJ-111, 22°36’48”S 43°24’39”W, 23 Apr 2002, P. A. Buckup, A. T. Aranda, F. P. Silva, L. M. S. Porto, C. A. Brandão & D. F. Almeida. MNRJ 23850, 28, 45.8–96.9 mm SL, Nova Iguaçu, rio Tinguá, tributary of rio Iguaçu, at BR-111 (Zumbi dos Palmares), 22°36’00”S 43°24’55”W, 23 Apr 2002, P. A. Buckup, A. T. Aranda, F. P. Silva, L. M. S. Porto, C. A. Brandão & D. F. Almeida. MNRJ 43371, 10, 40.5–64.4 mm SL, Nova Iguaçu, rio Boa Esperança at Prainha locality, tributary of rio Iguaçu, 22°34’41”S 43°23’34”W, 13 Aug 2014, M. R. Britto, D. F. M. Moraes Jr., V. S. Meza-Vargas, K. Ferreira, F. Cordeiro & R. Dias. MNRJ 43426, 31, 44.6–101.1 mm SL, Nova Iguaçu, rio Boa Esperança at Tinguá near to ONG Onda Verde, tributary of rio Iguaçu, 22°35’33”S 43°24’52”W, 15 Aug 2014, M. R. Britto, D. F. M. Moraes Jr., V. S. Meza-Vargas & R. M. Dias. MNRJ 43924, 6, 75.2–94.9 mm SL, Nova Iguaçu, rio Ana Felícia, tributary of rio Tinguá, 22°36’44”S 43°25’05”W, 6 Apr 2015, M. Britto, D. F. Moraes Junior, R. Dias, G. Araujo, D. Luiz & R. Delamare. Rio Paraíba do Sul basin (Piabanha drainage): MNRJ 14501, 16, 62.3–99.2 mm SL, Teresópolis, arroio Santa Rita, tributary of the left bank of rio Paquequer near BR-116 bridge, 22°17’42”S 42°56’04”W, 11 Mar 1993, P. A. Buckup, D. F. Moraes Jr., A. Suppa & A. Amaral. MNRJ 51642, 9, 60.5–112.7 mm SL, Teresópolis, Rio Paquequer, at Caledônia Alpina neighborhood, 22°22’03”S 42°57’45”W, 13 Sep 2018, S. A. Santos, E. B. Neuhaus, M. R. Britto & I. C. A. Souto-Santos.
TABLE 2 | Morphometric data of holotype and paratypes of Rineloricaria nudipectoris. Range includes the holotype. N = number of specimens measured; SD = standard deviation.
Holotype | N | Range | Mean | SD | |
Standard length (mm) | 88.79 | 39 | 72.7–113.6 | – | – |
Percent of
standard length | |||||
Head length | 22.8 | 39 | 19.9–23.8 | 21.7 | 0.9 |
Predorsal length | 34.8 | 39 | 30.1–35.3 | 32.9 | 1.2 |
Postdorsal length | 66.0 | 39 | 64.4–70.3 | 67.3 | 1.4 |
Prepectoral length | 17.2 | 39 | 15.8–19.5 | 16.8 | 0.9 |
Postpectoral length | 83.7 | 39 | 82.6–87.4 | 85.2 | 1.0 |
Prepelvic length | 34.3 | 39 | 29.6–36.7 | 32.7 | 1.7 |
Postpelvic length | 66.5 | 39 | 64.9–72.2 | 68.7 | 1.6 |
Preanal length | 48.4 | 39 | 43.7–50.2 | 46.4 | 1.7 |
Postanal length | 51.6 | 39 | 50.2–57.1 | 53.4 | 1.8 |
Unbranched dorsal-fin
ray | 20.8 | 38 | 17.9–23.2 | 20.6 | 1.2 |
Unbranched pectoral-fin ray | 16.9 | 39 | 14.6–19.0 | 17.1 | 1.0 |
Unbranched pelvic-fin
ray | 16.2 | 39 | 14.0–17.2 | 15.4 | 0.7 |
Unbranched anal-fin
ray | 16.3 | 38 | 14.6–17.8 | 16.0 | 0.8 |
Thoracic length | 17.7 | 39 | 14.4–19.7 | 17.1 | 1.2 |
Abdominal length | 16.1 | 39 | 14.8–17.6 | 16.3 | 0.7 |
Cleithral width | 20.0 | 39 | 15.7–20.7 | 18.0 | 1.2 |
Depth at
dorsal-fin origin | 12.0 | 39 | 8.3–12.0 | 10.5 | 0.8 |
Width at
anal-fin origin | 12.4 | 39 | 9.8–13.2 | 11.9 | 0.6 |
Caudal peduncle depth | 2.0 | 39 | 1.6–2.3 | 1.9 | 0.1 |
Caudal peduncle width | 3.5 | 39 | 2.8–3.5 | 3.1 | 0.2 |
Percent of head length | |||||
Snout length
| 47.3 | 39 | 45.1–49.1 | 47.5 | 1.0 |
Eye diameter | 15.7 | 39 | 14.2–18.2 | 16.1 | 0.8 |
Maximum orbital diameter | 22.0 | 39 | 19.0–25.0 | 22.0 | 1.2 |
Interorbital width | 25.0 | 39 | 23.6–27.6 | 25.5 | 1.1 |
Internarial width | 6.3 | 39 | 4.9–8.9 | 6.8 | 0.9 |
Head depth | 45.7 | 39 | 39.8–46.8 | 42.7 | 2.0 |
Head width | 80.3 | 39 | 70.7–80.7 | 75.8 | 2.5 |
Free maxillary
barbel | 7.7 | 39 | 4.4–15.0 | 9.6 | 2.2 |
Ventrorostral length | 6.5 | 39 | 4.5–8.5 | 6.5 | 0.9 |
Lower lip
length | 9.6 | 39 | 2.0–14.8 | 11.4 | 2.2 |
Diagnosis. Rineloricaria nudipectoris is distinguished from most of its congeners, except for R. aequalicuspis Reis & Cardoso, 2001, R. anhaguapitan Ghazzi, 2008, R. baliola Rodriguez & Reis 2008, R. capitonia Ghazzi, 2008, R. latirostris (Boulenger, 1900),
R. maacki Ingenito, Ghazzi, Duboc & Abilhoa, 2008, R. malabarbai Rodriguez & Reis, 2008, R. maquinensis Reis & Cardoso, 2001, R. microlepidogaster (Regan, 1904), R. misionera Rodriguez & Miquelarena, 2005, R. reisi Ghazzi, 2008, R. setepovos Ghazzi, 2008, and
R. tropeira Ghazzi, 2008, by the absence of plates in most of the skin of the ventral surface of the pectoral girdle (vs. pectoral girdle completely covered by plates). Rineloricaria nudipectoris is distinguished from R. setepovos by the presence of plates in the abdominal region (Fig. 3) (vs. abdominal region completely naked). It differs from the remaining naked-breast species, except for R. anhaguapitan, R. baliola, and R. capitonia, by its snout having an anterior area of exposed skin without odontodes extended laterally to the most anterior pore of the infraorbital ramus of the sensory canal (vs. naked area of snout not reaching the most anterior pore of infraorbital ramus of sensory canal) (Fig. 4). It is distinguished from R. anhaguapitan and R. capitonia by the upper unbranched caudal-fin ray extending beyond the margin of the fin as a short filament (vs. absence of filamentous extension) and by the leading-edge pectoral-fin ray reaching pelvic-fin origin (vs. leading-edge pectoral-fin ray reaching the first third of the pelvic fin). It differs from
R. baliola by the spotted color pattern in the dorsal, pectoral, and pelvic-fin rays (vs. color pattern composed of a dark, wide band covering most of the distal half of the fins).
FIGURE 3| Variation of abdominal plate coverage in the anterior region of abdomen in paratypes of Rineloricaria nudipectoris: A. MNRJ 14844, female, 88.8 mm SL. B. MNRJ 14844, female, 88.9 mm SL. C. MNRJ 14844, female, 98.2 mm SL
Description. Morphometric data of type specimens in Tab. 2. Head and body strongly depressed. Dorsal profile of head ascending from tip of snout until posterior limit of supraoccipital. Dorsal profile of body almost straight to slightly convex between highest point at supraoccipital and dorsal-fin origin, slightly concave along dorsal-fin base, straight from this point to caudal-fin origin. Body widest at pelvic-fin origin, progressively narrowing until end of caudal peduncle.
FIGURE 4| Lateral view of head showing extend of naked skin area (dashed line) of the snout relative to anterior pore (indicated by arrow) on sensory line system in (A) Rineloricaria nudipectoris,holotype, MNRJ 49345, male, 88.8 mm SL, (B) paratype,MNRJ 43536, female, 97.9 mm SL, and (C) R. zawadzkii,MNRJ 38254, female, 98.7 mm SL.
Head length larger than cleithral width. Supraoccipital and predorsal plates with low ridges. In dorsal view, head triangular in females and immature males, laterally rounded with hypertrophied odontodes in males (Figs. 2–3; details in Sexual Dimorphism below). Snout tip with elliptical naked area extended laterally to most anterior pore of infraorbital ramus of sensory canal (Fig. 4). Posterior margin of orbit slightly raised; eye large (14.4–18.2% of HL), rounded to slightly oval horizontally, with large, deep postorbital notch. Iris operculum present, large in preserved specimens. Oral disk covered by randomly distributed, rounded-tip papillae. Margins of both lips with short fringes. Maxillary barbel slightly larger than length of lower lip. Teeth strongly bicuspid, long; mesial cusp greater than lateral, dentary teeth larger than premaxillary teeth. Premaxilla teeth 6(2), 7(2), 8(17), 9*(12), 10(6); dentary teeth 8(8), 9*(25), 10(6).
Two predorsal plates between parieto-supraoccipital and nuchal plates with two low, inconspicuous ridges. Five (dorsal, mid-dorsal, median, mid-ventral, ventral) lateral-plate series below dorsal fin. Dorsal series starting at dorsal-fin base with 21(2), 22*(33), 23(4) plates. Mid-dorsal series with 6(1), 7*(18), 8(18), 9(2) plates; with weak lateral ridge. Median series with 27(2), 28*(32), 29(5) plates. Lateral line complete. Mid-ventral plate series with 15*(5), 16(18), 17(14), 18(2) plates. Median and mid-ventral series with well-developed keels formed by hypertrophied odontodes; these keels meeting in last 10th(2), 11th(12), 12th*(20), 13th(5) plate of median series. Ventral series with 22*(5), 23(28), 24(6) plates. Lateral abdominal series with 6(1), 7(25), 8*(13) plates between pectoral-fin and pelvic-fin origin; number of plates on opposite sides of body sometimes different. Adjacent lateral and medial abdominal plates separated by slender area of skin. Posterior abdominal plate complex with large well-developed preanal plate anteriorly bordered by three polygonal plates, bordered by five or, sometimes, more plates. Middle abdominal plate complex, between lateral abdominal plates, with few minute polygonal plates bordered by naked area, or with plates irregularly arranged in three to five rows, anterior plates smaller, less numerous. Ventral surface of pectoral girdle covered by skin without plates, except for two individuals in lot MNRJ 14844 with few small plates of the anterior plate complex (Fig. 3).
Unbranched dorsal-fin ray (dorsal spine) length (17.9–23.2% SL) similar to head length (19.9–23.8% SL). Dorsal-fin origin slightly posterior to pelvic-fin origin; dorsal-fin rays i,7*(39); dorsal-fin spinelet present. Dorsal-fin posterior margin truncate. Tip of dorsal fin when adpressed surpassing anal-fin origin; reaching eighth or ninth plate posterior to its origin. Pectoral-fin rays i,6*(39); posterior pectoral-fin margin slightly convex; first three branched rays slightly larger than unbranched pectoral-fin ray, when adpressed not reaching origin of pelvic fin, or just passing pelvic-fin origin in small individuals (<50 mm). Pelvic-fin rays i,5*(39); posterior pelvic-fin margin reaching to or slightly beyond anal-fin origin; posterior pelvic-fin margin truncate in juveniles, slightly convex in large individual. Anal-fin rays i,5*(39), posterior anal-fin margin rounded; second or third branched ray longest. Caudal fin truncate with i,10,i*(39) rays. Upper unbranched ray longer than lower one, extended as a short filament; profile of expanded caudal fin concave.
Color in alcohol. Background coloration of dorsal surface of head and trunk light brown. Pores of sensory system on head and lateral medial plates notably dark. Six dark brown transverse bars on dorsal surface of body; first at dorsal-fin origin, second at level of tip of reclined dorsal fin, following bars located on caudal peduncle. Chromatophores concentrated on posterior edge of supraoccipital bone forming dark conspicuous area, sometimes forming transverse bar. All fins covered by dark dots aligned across fin rays. Caudal fin with two broad vertical dark bars: one basally, one at distal margin. Ventral surface pale yellow (Fig. 2).
Sexual dimorphism. Head triangular in females and immature males without hypertrophied odontodes; head margin rounded in mature males, with abundant hypertrophied odontodes. Dorsal region of pectoral-fin rays covered almost entirely with thin, long odontodes with curved tips in males (Fig. 2). Unbranched pectoral-fin ray almost straight in females and juvenile males; thick, strongly curved in mature males, with short, densely arranged odontodes.
Etymology. The specific name “nudipectoris” is a compound Latin word formed by the neutral noun pectus, meaning breast, and the adjective nudus, meaning bare, in the genitive declension. Rineloricaria nudipectoris, thus, means Rineloricaria of the naked breast, in reference to the absence of plates in the ventral portion of the pectoral girdle and anterior medial portion of the abdomen.
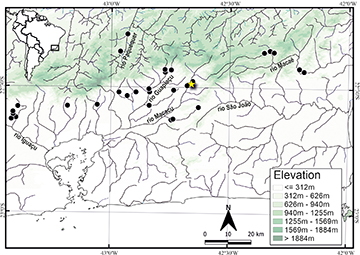
Geographical distribution. Rineloricaria nudipectoris is known from the Macaé, Macacu, Guapiaçu, Magé, Saracuruna, Iguaçu, and Paraíba do Sul river basins in southeastern Brazil (Fig. 5). Rineloricaria nudipectoris was recorded in sympatry with R. zawadzkii (Costa-Silva et al., 2022)and is associated with running clear waters and sandy or rocky substrates with marginal vegetation.
FIGURE 5| Geographic distribution of Rineloricaria nudipectoris. Star represents the type-locality; black circles may represent more than one collection event in the same locality.
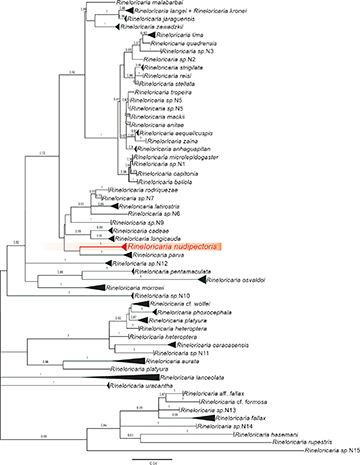
Molecular delimitation. DNA barcodes based on the amplified 655-nucleotide partial sequence of COI mitochondrial gene were generated for a total of 10 specimens of Rineloricaria nudipectoris (Tab. 1), encompassing specimens from the Iguaçu (1 ex.), Guapiaçu (1 ex.), Macacu (4 ex.), Macaé (2 ex.), and Paraíba do Sul (2 ex.) river basins. All samples share the same haplotype, except for a specimen from the Iguaçu River, which differs from specimens of the other drainages by three nucleotides. Rineloricaria nudipectoris received the Barcode Index Number BOLD:ADA2055. Based on the maximum likelihood tree (Fig. 6), the COI haplotypes of R. nudipectoris are most similar to those of Rineloricaria parva (Boulenger, 1895) (LBP_5136, LBP_9160, LBP_8471, LBP_9837, LBP_12964, LBP_12964, LBP_19025), which are included in BIN BOLD:AAZ4949 (Fig. S2). The overall mean distance among species is 12%. The lowest genetic distance between R. nudipectoris and any other known species is 6.2% from R. parva, followed by 7% from R. cadeae (Hensel, 1868), and 7.7% from R. longicauda Reis, 1983 (Tab. S3).
FIGURE 6| Maximum Likelihood tree based on the sequences of the cytochrome c oxidase subunit I gene under the GTR+G+I model. The numbers at each branch indicate the bootstrap values (100 pseudoreplicates), except for those below 0.5 which were suppressed. Branches are collapsed based on Costa-silva et al. (2015).
Conservation status. Rineloricaria nudipectoris is known from the Macaé, Macacu, Guapiaçu, Magé, Saracuruna, Iguaçu and Paraíba do Sul river basins. Its Extent of Occurrence (EOO) is approximately 2,047 km2, based on the minimum convex method, but the population is not severely fragmented, extreme fluctuations in area or number of individuals or subpopulations have not been observed, and there is no evidence of continuing decline in area, extent or quality of habitat, number of locations or number of mature individuals. The species is easily collected downstream of farmland areas and small towns. It occurs in protected areas, such as the Tinguá Biological Reserve and the Guapiaçu Natural Patrimony Reserves. Considering its presence in main coastal river drainages, its abundance in areas of moderate land use, and its presence in protected areas, R. nudipectoris is classified in the Least Concern (LC) category according to the criteria of the International Union for Conservation of Nature (IUCN) criteria (IUCN Standards and Petitions Subcommittee, 2022).
Discussion
Rineloricaria nudipectoris is assigned to the genus Rineloricaria based on the possession of the following features proposed by Fichberg, Chamon (2008): postorbital notch present; surface of lower lip with short round papillae; premaxilla with 6 to 10 teeth on each ramus; dentary teeth strong, deeply bicuspidate; six dark brown transversal bars on dorsal surface of body (first at dorsal-fin origin, second at level of posterior end of the reclined dorsal-fin rays, following bars located along caudal peduncle); abdomen with polygonal preanal plate anteriorly bordered by three trapezoidal plates, in turn bordered by five additional plates (Fig. 3). Although these characters are present in other Loricariinae, the phylogenetic analysis of our molecular data corroborates the hypothesis that R. nudipectoris is a member of a monophyletic group of species of Rineloricaria
(Fig. S2).Its close relationship with other species deeply inserted within the group confirms its generic assignment.
According to our DNA sequence results, specimens of Rineloricaria nudipectoris from the various drainages are remarkably similar. DNA barcodes of specimens from all drainages are genetically identical, except for the sample from the Iguaçu River which exhibits three nucleotide mutations. This level of genetic divergence (Tab. S3) is less than 1% and considerably smaller than the 2% threshold that has been suggested to identify separate species of fish lineages (Ward, et al, 2009; Pereira et al., 2013; Gomes et al., 2015; Shimabukuro-Dias et al., 2016; Ribolli et al., 2017). Although some molecular species delimitation algorithms are not based on genetic distance (Nogueira et al., 2021), these results corroborate the hypothesis that our samples represent a single species, which can be diagnosed by both morphological and molecular characters.
Within the framework of the comprehensive analysis of Rineloricaria presented by Costa-Silva et al. (2015), R. nudipectoris is most closely related to members of BIN BOLD:AAZ4949, which is assigned to samples of R. parva from the Paraná and Paraguay drainages in the La Plata River system. This relationship is unexpected because these species occur in geographically distant areas and exhibit considerable morphological differences. Rineloricaria nudipectoris can be distinguished from R. parva by having five lateral plate series below the dorsal fin vs. four series. In addition, R. parva has the caudal fin with both unbranched rays extended as long filaments and pectoral girdle completely covered by plates (Vera-Alcaraz et al., 2012: fig. 11), vs. upper unbranched caudal fin ray prolonged as a short filament and the skin covering the pectoral girdle naked or almost naked in R. nudipectoris (Figs. 2–3). The genetic similarity between these two species may be due to the inability of the COI marker to retain information from old phylogenetic relationships. Further testing of the hypothesis of close relationship between R. nudipectoris and R. parva requires additional molecular data, possibly including nuclear genes. Such a comprehensive study, however, is beyond the scope of the present study.
Rineloricaria nudipectoris is known from rivers draining the coastal slope of the Serra do Mar in the central region of Rio de Janeiro. Eight other species have been described from coastal rivers draining the Serra do Mar between this region and the southern coast of Santa Catarina: Rineloricaria anitae Ghazzi, 2008, R. cubataonis (Steindachner,1907), R. jaraguensis (Steindachner, 1909), R. kronei (Miranda Ribeiro, 1911), R. langei Ingenito, Ghazzi, Duboc & Abilhoa, 2008, R. nigricauda (Regan, 1904), R. steindachneri (Regan, 1904), and R. zawadzkii Costa-Silva, Silva & Oliveira, 2022. Three of these species occur geographically close to R. nudipectoris: R. nigricauda (described from Porto Real, Paraíba do Sul River basin), R. steindachneri (from the lower Paraíba do Sul River), and R. zawadzkii (from Paraíba do Sul River basin and coastal rivers in Rio de Janeiro State). The new species is morphologically distinguishable from those, as well as from most other congeners, by the lack of dermal plates covering the ventral region of the pectoral girdle (Figs. 3A–C) (vs. pectoral girdle extensively covered by plates). Incomplete abdominal cover, however, is associated to species from Paraná basin (R. latirostris, R. maacki), Uruguay basin (R. anhaguapitan, R. capitonia, R. misionera,
R. reisi, R. setepovos,and R. tropeira) and coastal drainages from southeastern to southern Brazil (R. aequalicuspis, R. baliola, R. malabarbai, R. maquinensis, R. microlepidogaster) (Ingenito et al., 2008), and the phylogenetic significance of this character should be further investigated.
The description of R. nudipectoris contributes further to the knowledge of the fish fauna of the coastal drainages of southeastern Brazil, where the new species is exclusively found. The presence of undescribed species of fish in one of the largest metropolitan areas in Brazil, which has been occupied for centuries, illustrates the incompleteness of knowledge about fish diversity in the tropical and subtropical areas of the Neotropical Region, and highlights the need for rapidly describing this unknown diversity facing a prospect of extinction due to growing human occupation of river drainages. It also highlights the role of modern DNA sequencing technology in integrative taxonomic studies, which has accelerated the rate of description of new species of fish during the last two decades.
Comparative material examined. Brazil. Rineloricaria aequalicuspis, Rio Grande do Sul: MNRJ 41101, 44, 52.1–107.8 mm SL. Santa Catarina: MNRJ 41212, 20, 50.2–134.9 mm SL. Rineloricaria anhaguapitan, Rio Grande do Sul: MNRJ 41810, 4, 44.8–61.9 mm SL. MNRJ 41813, 5, 41.2–102.4 mm SL. Rineloricaria beni, Rondônia: MNRJ 27508, 7, 37.1–57 mm SL. Rineloricaria cacerensis, Mato Grosso: MNRJ 645, 1, 62.52 mm SL, lectotype designated by Miranda Ribeiro, 1953. Rineloricaria cadeae, Rio Grande do Sul: MNRJ 14196, 7, 15.3–50.4 mm SL. Rineloricaria capitonia, Santa Catarina: MNRJ 52490, 2, 72.7–73.1mm SL. Rineloricaria cubataonis, Santa Catarina: MNRJ 40946, 54, 50.9–104.1 mm SL. Rineloricaria fallax, Roraima: MNRJ 31555, 8, 59.6–124.6 mm SL. Rineloricaria formosa, Roraima: MNRJ 31556,1, 90.4 mm SL. Rineloricaria hasemani, Rondônia: MNRJ 39265, 58.2 mm SL. Rineloricaria jaraguensis, Santa Catarina: MNRJ 40922, 33, 46.7–85.3 mm SL. Rineloricaria kronei: São Paulo, MNRJ 40756, 7, 59.2–118.4 mm SL. Rineloricaria lanceolata, Rondônia: MNRJ 14972, 35, 43.6–101.6 mm SL. MNRJ 27524, 4, 60.3–109.2 mm SL. Tocantins: MNRJ 49848, 2, 63.9–97.5 mm SL. Rineloricaria langei, Paraná: MNRJ 31157, 5, 1 cs, 81.1–106.2 mm SL, paratypes. Rineloricaria latirostris, São Paulo: MNRJ 29853, 1, 75.12 mm SL. MNRJ 32758, 1, 121.2 mm SL. Rineloricaria maacki, Paraná: MNRJ 31158, 1, 108.4 mm SL, holotype. Rineloricaria malabarbai, Rio Grande do Sul: MNRJ 41069, 9, 43.9–125.3 mm SL. Rineloricaria microlepidogaster: Rio Grande do Sul: MNRJ 22232, 1, 169.9 mm SL. MNRJ 41068, 10, 52.6–132.7 mm SL. Rineloricaria nigricauda, Rio de Janeiro: BMNH 1891.6.16.32, 55.4 mm SL, lectotype designated by Isbrücker, 1979. MNRJ 14017, 20, 53.2–105.3 mm SL. MNRJ 13955, 5, 134.4–142.9 mm SL. Mato Grosso: MNRJ 44569, 7, 52.9–86.1 mm SL. Rineloricaria pentamaculata, Paraná: MNRJ 41049, 20, 57.8–93.9 mm SL. Rineloricaria phoxocephala, Amazonas: MNRJ 46873, 116.5 mm SL. Rineloricaria steindachneri, Rio de Janeiro: MNRJ 14005, 1, 100.1 mm SL. MNRJ 42476, 1, 83.5 mm SL. Rineloricaria strigilata, Santa Catarina: MNRJ 41031, 11, 70.2–104.3 mm SL. Rineloricaria tropeira, Santa Catarina: MNRJ 19037, 2, 74.6–80.1 mm SL. MNRJ 21284, 2, 101.1–113 mm SL, paratypes. Rineloricaria zawadzkii, Rio de Janeiro: MNRJ 38254, 12, 11.8–137.8 mm SL. MNRJ 40325, 2, 64.2–94.4 mm SL. MNRJ 42544, 4, 74.8–110.6 mm SL. Paraguay.Rineloricaria parva, Departamento Central: MNRJ 24316, 6 (3, 70.9–77.6 mm SL).
Acknowledgments
We thank Emanuel Neuhaus, Manuela Dopazo, Igor Souto-Santos (Museu Nacional – UFRJ) for all the help during field and laboratory activities. Several students and postdocs in our laboratory have contributed to studies of Rineloriaria over the past 25 years leading to the description of R. nudipectoris: André S. T. Pereira, Amanda T. dos Santos, Leonardo F. S. Ingenito, Miriam S. Ghazzi, Thais M. Estruc, Tarik Q. B. Carvalho, and Ana C. G. Silva-Malanski. EM is grateful to Guilherme Muricy (UFRJ), for comments and suggestions during manuscript drafting. This work received support from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, PROEX), process 88887.613913/2021-00. PAB research received funding from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPQ proc. 423526/2018–9, 312801/2017–3), CAPES (proc. 88882.156885/2016–01), and Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ proc. 200.063/2019).
References
Costa-Silva GJ, Rodriguez MS, Roxo FF, Foresti F, Oliveira C. Using different methods to access the difficult task of delimiting species in a complex Neotropical hyperdiverse group. PLoS ONE. 2015; 10:1–12. https://doi.org/10.1371/journal.pone.0135075
Costa-Silva GJ, Costa e Silva GS, Oliveira C. A new species of spiny Rineloricaria (Siluriformes: Loricariidae) from the Rio Paraíba do Sul basin and coastal rivers from Rio de Janeiro State. Zootaxa. 2022; 5175(2):285–92. https://doi.org/10.11646/zootaxa.5175.2.6
Covain R, Fisch-Muller S. The genera of the Neotropical armored catfish subfamily Loricariinae (Siluriformes: Loricariidae): A practical key and synopsis. Zootaxa. 2007; 1462(1):1–40. https://doi.org/10.11646/zootaxa.1462.1.1
Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004; 32(5):1792–97. http://doi.org/10.1093/nar/gkh340
Fichberg I, Chamon CC. Rineloricaria osvaldoi (Siluriformes: Loricariidae): A new species of armored catfish from rio Vermelho, Araguaia basin, Brazil. Neotrop Ichthyol. 2008; 6(3):347–54. https://doi.org/10.1590/S1679-62252008000300008
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2022. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Gomes LC, Pessali TC, Sales NG, Pompeu PS, Carvalho DC. Integrative taxonomy detects cryptic and overlooked fish species in a Neotropical river basin. Genetica. 2015; 143:581–88. https://doi.org/10.1007/s10709-015-9856-z
Handy SM, Deeds JR, Ivanova NV, Hebert PDN, Hanner RH, Ormos A et al. A single-laboratory validated method for the generation of DNA barcodes for the identification of fish for regulatory compliance. J AOAC Int. 2011; 94(1):201–10. https://doi.org/10.1093/jaoac/94.1.201
Ingenito LFS, Ghazzi MS, Duboc LF, Abilhoa V. Two new species of Rineloricaria (Siluriformes: Loricariidae) from the rio Iguaçu basin, southern Brazil. Neotrop Ichthyol. 2008; 6(3):355–66. https://doi.org/10.1590/S1679-62252008000300009
Isbrücker, IJH, Nijssen H. Two new species and a new genus of neotropical mailed catfishes of the subfamily Loricariinae Swainson, 1838 (Pisces, Siluriformes, Loricariidae). Beaufortia. 1978; 27(339):177–206. Available from: https://repository.naturalis.nl/pub/504901
Isbrücker IJH, Nijssen H. Three new South American mailed catfishes of the genera Rineloricaria and Loricariichthys (Pisces, Siluriformes, Loricariidae). Bijdr Dierkd. 1979; 48:191–211.
Isbrücker IJH, Nijssen H. Sexualdimorphismus bei Harnischwelsen (Loricariidae). DATZ-Sonderheft Harnischwelse. 1992; 19–33. Available from: https://www.datz.de/images/stories/gallerien/Sonderheft_Harnischwelse/sexualdimorphismus%20bei%20harnischwelsen.pdf
International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN Red List categories and criteria, Version 15.1 [Internet], Gland; 2022. Available from: https://www.iucnredlist.org/resources/redlistguidelines
Jennings WB, Ruschi PA, Ferraro G, Quijada CC, Silva-Malanski ACG, Prosdocimi F et al. Barcoding the Neotropical freshwater fish fauna using a new pair of universal COI primers with a discussion of primer dimers and M13 primer tails. Genome. 2019; 62(2):77–83. https://doi.org/10.1139/gen-2018-0145
Kearse M, Moir R, Wilson A, StonesHavas S, Cheung M, Sturrock S et al. Geneious basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics. 2012; 28(12):1647–49. https://doi.org/10.1093/bioinformatics/bts199
Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol Biol Evol. 2018; 35(6):1547–49. https://doi.org/10.1093/molbev/msy096
Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988; 16(3):1215. https://doi.org/10.1093/nar/16.3.1215
Morris PJ, Yager HM, Sabaj Pérez MH. ACSImagebase: A digital archive of catfish images compiled by participants in the All Catfish Species Inventory. 2006. Available from: http://acsi.acnatsci.org/base
Nogueira AF, Oliveira C, Langeani F, Netto-Ferreira AL. Molecular species delimitation of the genera Anodus, Argonectes, Bivibranchia and Micromischodus (Ostariophysi: Characiformes). Neotrop Ichthyol. 2021; 19(4):e210005. https://doi.org/10.1590/1982-0224-2021-0005
Pereira LHG, Hanner R, Foresti F, Oliveira C. Can DNA barcoding accurately discriminate megadiverse Neotropical freshwater fish fauna? BMC Genet. 2013; 14(20). https://doi.org/10.1186/1471-2156-14-20
Ratnasingham S, Hebert PDN. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Mol Ecol Notes. 2007; 7(3):355–64.
Ratnasingham S, Hebert PDN. A DNA-based registry for all animal species: The Barcode Index Number (BIN) System. PLoS ONE. 2013; 8(8):e66213. https://doi.org/10.1371/journal.pone.0066213
Reis RE, Cardoso AR. Two new species of Rineloricaria from southern Santa Catarina and northeastern Rio Grande do Sul, Brazil (Teleostei: Loricariidae). Ichthyol Explor Freshw. 2001; 12(4):319–32.
Ribolli J, Scaranto BM, Shibatta OA, Bombardelli RA, Zaniboni-Filho E. DNA barcoding confirms the occurrence of Rhamdia branneri and Rhamdia voulezi (Siluriformes: Heptapteridae) in the Iguaçu River Basin. Neotrop Ichthyol. 2017; 15(1):e160147. https://doi.org/10.1590/1982-0224-20160147
Sabaj MH. Codes for natural history collections in ichthyology and herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
Schaefer SA. The Neotropical cascudinhos: systematics and biogeography of the Otocinclus catfishes (Siluriformes: Loricariidae). Proc Acad Nat Sci Phila. 1997; 148:1–120. https://www.jstor.org/stable/4065046
Shimabukuro-Dias CK, Costa Silva GJD, Ashikaga FY, Foresti F, Oliveira C. Molecular identification of the fish fauna from the Pantanal flood plain area in Brazil. Mitochondrial DNA A. 2016; 28(4):588–92. https://doi.org/10.3109/24701394.2016.1149826
Vera-Alcaraz HS, Pavanelli CS, Zawadzki CH. Taxonomic revision of the Rineloricaria species (Siluriformes:Loricariidae) from the Paraguay River basin. Neotrop Ichthyol. 2012; 10(2):285–311. https://doi.org/10.1590/S1679-62252012000200006
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PDN. DNA barcoding Australia’s fish species. Phil Trans R Soc B. 2005; 360(1462):1847–57. https://doi.org/10.1098/rstb.2005.1716
Ward RD, Hanner R, Hebert PDN. The campaign to DNA barcode all fishes, FISH-BOL. J Fish Biol. 2009; 74(2):329–56. https://doi.org/10.1111/j.1095-8649.2008.02080.x
Authors
![]() Eduardo Mejia1
Eduardo Mejia1 ![]() ,
, ![]() Gustavo A. Ferraro1 and
Gustavo A. Ferraro1 and ![]() Paulo A. Buckup1
Paulo A. Buckup1
[1] Programa de Pós-Graduação em Ciências Biológicas (Zoologia), and Departamento de Vertebrados, Museu Nacional, Universidade Federal do Rio de Janeiro, Quinta da Boa Vista, São Cristóvão, 20940-040 Rio de Janeiro, RJ, Brazil. (EM) edu.mejia@ufrj.br (corresponding author), (GAF) gustavoandresferraro@yahoo.com.br, (PAB) buckup@acd.ufrj.br.
Authors’ Contribution 

Eduardo Mejia: Data curation, Formal analysis, Investigation, Methodology, Validation, Writing-original draft, Writing-review and editing.
Gustavo A. Ferraro: Data curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Paulo A. Buckup: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The author declares no competing interests.
How to cite this article
Mejia E, Ferraro GA, Buckup PA. A new species of Rineloricaria (Siluriformes: Loricariidae) from coastal drainages of Rio de Janeiro, southeastern Brazil. Neotrop Ichthyol. 2023; 21(1):e220083. https://doi.org/10.1590/1982-0224-2022-0083
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted February 22, 2023 by Marcelo Britto
Accepted February 22, 2023 by Marcelo Britto
![]() Submitted August 23, 2022
Submitted August 23, 2022
![]() Epub April 7, 2023
Epub April 7, 2023