![]() Johanna Maritza Cano Velarde1
Johanna Maritza Cano Velarde1 ![]() ,
, ![]() Natalia Marins Bastos2,
Natalia Marins Bastos2, ![]() Laícia Carneiro-Leite3,
Laícia Carneiro-Leite3, ![]() Laís Pedroso Borges3,
Laís Pedroso Borges3, ![]() Edson Guilherme Vieira4,
Edson Guilherme Vieira4, ![]() Rosicleire Veríssimo-Silveira4 and
Rosicleire Veríssimo-Silveira4 and ![]() Alexandre Ninhaus-Silveira4
Alexandre Ninhaus-Silveira4
PDF: EN XML: EN | Cite this article
Abstract
This study aimed to develop a protocol for the cryopreservation of Pseudoplatystoma corruscans semen. For this, mature males were hormonally induced with a single dose of carp pituitary extract (5 mg/kg body weight). Semen was collected and evaluated. Two cryoprotectants were tested to compose the diluents: dimethyl acetamide (DMA) and dimethyl sulfoxide (Me2SO), in two concentrations (8% and 10%), + 5.0% glucose + 10% egg yolk. The semen was diluted in a 1: 4 ratio (semen: extender), packed in 0.5 mL straws and frozen in a dry shipper container in liquid nitrogen vapors. After thawing, sperm kinetics, sperm morphology and DNA integrity of cryopreserved sperm were evaluated. Pseudoplatystoma corruscans males produced semen with sperm motility > 80%. After thawing, all treatments provided semen with total sperm motility > 40%, with no significant difference (P < 0.05) between them, as well as between the other sperm kinetic parameters evaluated. The treatments with DMA provided a smaller fragmentation of the DNA of the gametes. Sperm malformations were identified in both fresh and cryopreserved semen, with a slight increase in these malformations being identified in sperm from thawed P. corruscans semen samples.
Keywords: Freezing, Sperm quality, Pimelodidae, DNA integrity, Germplasm.
Este estudo teve como objetivo desenvolver um protocolo para a criopreservação do sêmen de Pseudoplatystoma corruscans. Para tal, machos maduros foram induzidos hormonalmente com uma dose única de extrato de hipófise de carpa (5 mg/kg de peso vivo). O sêmen foi coletado e avaliado. Sendo testados para compor os diluentes, dois crioprotetores: dimetil acetamida (DMA) e dimetil sulfóxido (Me2SO), em duas concentrações (8% e 10%), + 5,0% glicose + 10% gema de ovo. O sêmen foi diluído na proporção 1: 4 (sêmen: extensor), embalado em palhetas de 0,5 mL e congelado em container dryshipper em vapores de nitrogênio líquido. Após o descongelamento, foram avaliados os aspectos cinéticos espermáticos, a morfologia espermática e a integridade do DNA dos espermatozoides criopreservados. Os machos de P. corruscans produziram sêmen com motilidade espermática > 80%. Todos os tratamentos proporcionaram após o descongelamento sêmen com motilidade espermática total > 40%, sem diferença significativa (P < 0,05) entre eles, como também entre os demais parâmetros cinéticos espermáticos avaliados. Os tratamentos com DMA proporcionaram uma menor fragmentação do DNA dos gametas. Malformações espermáticas foram identificadas, tanto no sêmen fresco, como no criopreservado, sendo identificado um aumento discreto dessas malformações nos espermatozoides das amostras de sêmen descongeladas de P. corruscans.
Palavras-chave: PTCongelamento, Germoplasma, Integridade do DNA, Pimelodidae, Qualidade do sêmen.
Introduction
Pseudoplatystoma corruscans (Spix & Agassiz, 1829), commonly known as pintado or surubim, belongs to the order Siluriformes, family Pimelodidae, and is native to the São Francisco and Paraná basins of Brazil. This species is a migratory piscivore that is widely used for the production of hybrids, and commercially used for its high-quality meat and in the sports fishing industry (Porto-Foresti et al., 2013; Barzotto et al., 2017). Overfishing and environmental degradation have caused this species to be considered endangered (MMA, 2022).
Pseudoplatystoma corruscans is a species of catfish that requires large reproductive migrations, and its seasonal reproductive cycle is linked to rainfall levels. Similar to many captive-bred catfish, it has some reproductive dysfunction, leading to a semen reduction or low seminal quality in most males. Therefore, there is a need to use hormonal induction of spermiation to increase the volume and quality of semen (Viveiros, 2011; Barzotto et al., 2017). Thus, the development of technologies aimed at optimizing their production in captivity and conserving the species is necessary. The development of protocols for the cryopreservation of germ cells and embryos will allow for the formation of a genetic bank for the species as well as assist producers in eliminating the reproductive asynchrony between males and females, abolishing the need for a large breeding stock, preserving the genetic material of the superior specimens, and facilitating research for genetic improvement (Ninhaus-Silveira et al., 2006; Bashiyo-Silva et al., 2015). However, the development of effective cryopreservation protocols necessitates the use and analysis of high-quality sperm, which is determined by such aspects as sperm motility, sperm concentration, membrane and DNA integrity, and sperm morphology (Kowalski, Cejko, 2019). Knowledge of the seminal characteristics of fish species is essential for evaluating semen quality indices, since the parameters involved directly influence the process of artificial fertilization, as well the establishment of seminal cryopreservation processes and their use in fertilization of oocytes for viable offspring (Beirão et al., 2019).
According to Eilts (2005), the resistance of cells to cryopreservation varies by individual, and this variability may be linked to the genetic makeup of each animal, as well as this sensitivity is species specific. Some characteristics deteriorate the heterogeneous permeability of the sperm cell membrane, which can cause changes in the movement of intracellular water during freezing and thawing processes. Ultimately, this can cause the formation of intracellular ice and consequently affect cell survival.
To protect sperm during the refrigeration and seminal freezing processes, diluent media are used to preserve sperm motility and fertility, provide energy substrates, and prevent the harmful effects of changes in pH and osmolarity to improve semen quality. Several diluting media have been studied with adapted formulations in an attempt to mitigate damage to sperm cells (Ahn et al., 2018). Thus, the present study aimed to develop a cryopreservation protocol that is appropriate for the semen of P. corruscans, while maintaining the sperm characteristics.
Material and methods
Obtaining gametes. Mature adult males of Pseudoplatystoma corruscans (Fig. 1.) were selected from the broodstock of the Hydrobiology Station of the Jupiá Hydroelectric Power Plant, Castilho, SP (20°52’20”S 51°29’15”W), formed by animals captured in the Paraná River, with more than three years of age. The experiments were carried out in the months of November and December in the reproductive period of the species. The fish were hormonally induced with carp pituitary extract (Hoeffers agricultural products) at a single dose of 5 mg/kg body weight via intramuscular injection. After 12 h, the fish were anesthetized with a solution of menthol (10 mL/L of water; peppermint oil, Sigma Aldrich, EUA), and semen was collected in 10 mL test tubes that were previously sterilized and dried. Contamination with blood, feces, and urine was carefully avoided.
FIGURE 1| Adult male specimen of Pseudoplatystoma corruscans, belonging to the Hydrobiology Station of the Jupiá Hydroelectric Plant.
There is no voucher referring to an ichthyological record in a scientific collection of the specimens used in the development of this study, because all were adults belonging to the breeding stock, and there were no juveniles at the station that could be sent for ichthyological analysis. As well as, soon after the end of this experiment, all were donated to other institutions and/or released in their natural environment, the Paraná River.
Evaluation of seminal characteristics. Semen color was estimated considering the translucent and white patterns, and volume was measured using a graduated 10 mL test tube. To measure the sperm concentration (spermatozoa/mL), the semen was diluted in a formaldehyde-saline solution at a proportion of 1:1000 and counted in a Neubauer hematimetric chamber (Bright-Line; New Optics) (Silveira et al., 1990). The quality of fresh semen was evaluated through quantification of subjective motility (Ninhaus-Silveira et al., 2006) and the duration of sperm motility under optical microscopy (400x), from the moment of sperm activation until the complete stoppage of sperm movement. Fourteen males were used for the seminal characteristic evaluations, with three replicates per sample. The CASA system was not used to analyze the sperm motility of fresh semen because it was not available at the hydrobiology station where the fish were located.
Sperm cryopreservation. For the semen cryopreservation experiments, only semen with greater than 80% mobile cells (n = 6 males) were used, along with four extenders composed of 5% glucose, 10% egg yolk, and 8 or 10% of a cryoprotective substance [dimethyl acetamide (DMA) or dimethyl sulfoxide (Me2SO)] composing four treatments, the solutions were augmented with distilled water to 100 mL (Tab. 1).
TABLE 1 | Extenders (Treatments) used in Pseudoplatystoma corruscans sperm freezing.
Treatments | Composition |
T1 | Egg
yolk 10% + Glucose 5% + DMA 8% |
T2 | Egg
yolk 10% + Glucose 5% + DMA 10% |
T3 | Egg
yolk 10% + Glucose 5% + Me2SO 8% |
T4 | Egg
yolk 10% + Glucose 5% + Me2SO 10% |
The semen was diluted in a 1:4 ratio (semen:extender) in the four extenders and transferred into 0.5 mL straws at room temperature (28 °C). After a 15 min equilibrium period, the straws were placed individually inside a dry shipper cryogenic tank (MVE SC 4/2 V). The samples were frozen in liquid nitrogen vapors at a cooling rate of 7.3 °C/min (Thermocouple OMEGA® RDXL4SD). Subsequently, the samples were transferred to a cryogenic “liquid” type container (ABS, Pecplan; model ABS20, MVE). After 60 days, the samples were thawed in a water bath at 37 °C for 10 s (Ninhaus-Silveira et al., 2006).
Sperm motility. After thawing, motility analysis was performed (n = 6, with 6 replicates/treatment) using computer assisted sperm analyses (CASA, Medea Lab, Germany), with a sperm class analyzer (SCA™ 2010, Microptics, SL version 5.1, Barcelona, Spain) at the Laboratório de Zootecnia da Universidade Federal de Lavras (UFLA). Thawed semen (100 μL) was diluted in 400 μL of a 5% glucose solution, activated using a 1% sodium bicarbonate solution, and pipetted into a Makler® chamber (Sefi-Medical Instruments Ltd., Haifa, Israel) for motility analyses [total motility (TM, %), curvilinear velocity (VCL, μm/s), straight line velocity (VSL, μm/s), and average path velocity (VAP, μm/s)]. For the calibration of the CASA system for analysis of sperm motility, it was considered: for image capture 100 frames per second, minimum sperm velocity of 10 µm/s and an interval of 10 s between activation and the beginning of the measurement.
Morphological analysis. For ultrastructural analysis, fresh (n = 6) and thawed semen (n = 6/treatment) samples were prefixed in 2% glutaraldehyde in a 0.1 M phosphate buffer at pH 7.3. Subsequently, the material was centrifuged for 3 min at 1000 rpm and the liquid was removed. The pellet was resuspended in distilled water, washed for 10 min, and re-centrifuged (3 min/1000 rpm) three times. The material was then transferred to a 13 mm diameter coverslip covered with 1% poly-L-lysine in water and post-fixed in 0.5% osmium tetroxide in distilled water for 30 min. The samples were then dehydrated in increasing concentrations of ethanol (7.5% to 100%), dried in Balzers CPD-20 critical point equipment, covered with a 10 nm thick gold film (Metallizer MED-010), and analyzed and photomicrographed using a scanning electron microscope (SEM Quanta 200, FEI).
DNA Integrity. To determine the integrity of cryopreserved sperm DNA, the comet assay method was used, based on the protocol proposed by Singh et al. (1988) and Klaude et al. (1996), histological glass slides were prepared with a thin layer of normal melting point agarose (NMP). The thawed semen samples were diluted in Ringer solution (96724 Ringer Solution 25% strength tablets, Sigma Aldrich) to replace extracellular fluids, since this solution is isotonic with multiple electrolytes. The diluted semen (10 µL) was then mixed with 120 µL of low melting point agarose (LMP), placed on slides previously prepared with normal agarose (low melting), covered with a coverslip, and stored in a refrigerator for approximately 30 min to solidify. Subsequently, the slides were immersed in a pH 10 lysis solution (2.5 M NaCl, 100 mM EDTA, 10 mM Tris, and 35 mM lauryl) for 1 h at 4 °C.
The slides were then placed in an electrophoresis tank filled with a pH 13 solution (200 mM EDTA, 10 M NaOH) and maintained for 30 min at 4 °C. Electrophoresis was performed for 20 min at 25 V/300 mA, after which the slides were covered with a pH 7.5 neutralization solution (Tris 0.4 M) for 5 min with three cycles. Finally, they were dried at room temperature (28 °C), fixed by immersion in cold absolute ethanol for 5 min, and dried again at the same temperature.
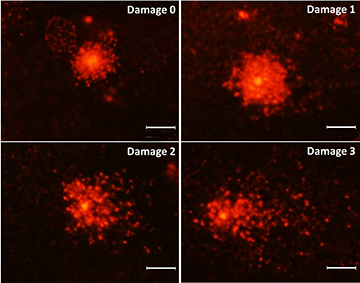
To visualize the comet, each slide was stained by adding 100 µL of GelRed solution, covered with a coverslip, and analyzed with a 40x objective lens with the aid of a fluorescence microscope (Olympus, BX-FLA, Japan). Five samples per treatment were analyzed with three replicates (n = 5; four treatments; three replicates). To determine the damage index, three random fields were chosen per sample/treatment, and 100 cells per field were analyzed and classified according to the intensity and size of the tail (Fig. 2): class 0, no apparent damage; and 1 to 3, increasing DNA damage depending on the length and intensity of tail staining (Kobayashi et al., 1995).
FIGURE 2| Fluorescence photomicrograph of nuclei with different classes of damage assessed by the Comet Assay analysis: 0, no apparent damage; 1, little damage; 2, average damage; 3, major damage. Scale bars = 95 µm.
The damage index (total ID) was measured using the formula below, where “n” represents the number of cells in each damage class:
Total ID = 0 (n Class 0) + 1 (n Class 1) + 2 (n Class 2) + 3 (n Class 3).
Each slide received a score assigned to its total ID, which was subjected to analysis of variance (ANOVA) with the means compared using Tukey’s test (P < 0.05).
Statistical analysis. The data were transformed into a square root (2√) to ensure a normal distribution as the data were nonparametric. Analysis of variance (ANOVA) and multiple mean comparison tests were performed using Tukey’s and Fisher’s tests at 5% probability. Before performing the statistical analysis of the results, Brown and Forsythe tests were conducted to determine the homogeneity of variance, as well as the Kolmogorov–Smirnov test for the normality of the data. The results showed that the percentages were not subjected to transformations to be normalized. Data were analyzed using the R Studio statistical program (RStudio Team, 2015).
Results
Seminal characteristics of fresh semen. The fresh semen was white, presented little viscosity, and had an average seminal volume of 6.8 ± 2.4 mL. The average sperm concentration was 96.7 ± 28.9 ×109 sperm/mL, subjective total motility was 92.3 ± 9.7%, and duration of motility was 31 ± 3.0 s. Pseudoplatystoma corruscans sperm had a head length of 2 μm and a width of 2.32 μm. The middle section was 1.5 μm long, and the flagellum was 32.6 μm in length from the base of the flagellum to the end of the terminal portion. Sperm were also identified with abnormalities such as a curled tail, a shortened middle section, and the absence of flagellum (Fig. 3).
FIGURE 3| Morphological characteristics of the Pseudoplatystoma corruscans sperm. A and B, normal spermatozoa; C and D, sperm with malformations. Legends: h1, width; h2, length; mp, intermediate part; #, sperm without the flagellum; c, curled tail; rmp, reduced middle piece. Scale bars; A and C = 20 µm; B and D = 5 µm.
Sperm motility. The thawed semen showed sperm motility in all treatments tested, and the highest percentage of mobile sperm was observed in the 10% Me2SO treatment (45.57 ± 9.6%) (Fig. 4A). There were no significant differences between the treatments for the analyzed velocity parameters VCL, VSL, and VAP. For total motility, the use of 10% Me2SO resulted in higher means of VSL and VAP (45.02 ± 16.3 μm/s and 61.41 ± 8.8 μm/s, respectively). Regarding curvilinear velocity (VCL), the highest mean was occurred with in the treatment with 10% DMA (89.25 ± 11.2 μm/s) (Figs. 4B–D).
FIGURE 4| Analysis of the sperm movement of thawed semen from Pseudoplatystoma corruscans. A. Total motility (MT), B. Curvilinear velocity (VCL, µm/s), C. Straight line velocity (VSL, µm/s), D. Average path velocity (VAP, µm/s). Same letters indicate no significant differences (P < 0.05).
Morphological analysis. Scanning microscopy analysis revealed possible changes in the structure of thawed P. corruscans sperm. Spermatozoa were observed to have increased head sizes, wrinkling and rupture of the plasma membranes, possible leakage of cytoplasm in the head region, damage to the middle sections and flagella, exposure of the structural fibers, and presence of cytoplasmic gout (Figs. 5A, C, D). Sperm with wound flagella were also observed (Fig. 5B).
FIGURE 5| Morphological analysis of thawed sperm from Pseudoplatystoma corruscans. A. Sperm with changes in the head, rupture of the membrane and damage to the intermediate part. B. Curled flagellum. C. Spermatozoon with damage to the head, intermediate piece and flagellum. D. Sperm head with changes in the head, damage to the membrane and damage to the intermediate part. Legends: ● = membrane rupture, * = possible cytoplasmic material, # = flagellar fibers, ♦ = cytoplasmic gout. Scale bars; A, B, and D = 2 µm; C = 5 µm.
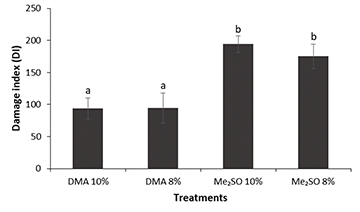
DNA integrity. DNA integrity analysis showed that there was no significant difference between treatments T1 and T2 in relation to the DMA concentration used (8% or 10%), and the same result was observed for treatments T3 and T4 in the Me2SO concentrations used (8% or 10%). However, a significant difference was observed in the use of different cryoprotectants (DMA and Me2SO), with DMA returning the lowest levels of DNA damage, thus demonstrating better protection of the genetic integrity of P. corruscans sperm (Fig. 6).
FIGURE 6| Index of damage caused to DNA by the cryopreservation process of semen from Pseudoplatystoma corruscans. Different letters indicate significant differences (P < 0.05), by the Turkey test
Discussion
For the seminal parameters observed for P. corruscans, the sperm concentration showed the greatest variation among the analyzed males, and this fact was also reported by Ferreira et al. (2001), Caneppele et al. (2015) and Sant’Anna et al. (2019) with the siluriform species, Rhamdia quelen, Steindachneridion melanodermatum, and Steindachneridion parahybae, respectively. In addition to variations in sperm concentration between species, changes in this parameter are often considered to be associated with age, time of year, animal conditions, collection methodology, and/or successive semen collection, among other factors.
There is a wide variation in the reported efficacy of permeable cryoprotective agents for Siluriformes sperm. Dimethyl sulfoxide (Me2SO), methanol (MeOH), and dimethyl acetamide (DMA) are most commonly used to protect catfish sperm, as well as ethylene glycol (EG) and propylene glycol (PG), among others (Viveiros, 2011). Ogier de Baulny et al. (1999) reported that DMA was more efficient as a cryoprotectant substance than glycerol, DMSO, and propylene glycol in preserving membrane integrity, mitochondrial activity, ATP level, and sperm motility of European catfish, Silurus glanis, after cryopreservation. The use of methyl glycol (MG), as opposed to MeOH and Me2SO, provided greater sperm motility after thawing for Steindachneridion scriptum (Pereira et al., 2019), whereas, MeOH and Me2SO resulted in increased motility, sperm vigor, and motility duration after thawing in Leiarius marmoratus (Borges et al., 2020). Herrera-Cruz et al. (2019) tested Me2SO, DMA, and EG in the cryopreservation of Pseudoplatystoma magdaleniatum sperm and did not observe significant differences with the use of these cryoprotectants; however, Rhamdia quelen presented higher values of sperm motility after thawing when EG was used as a sperm cryoprotectant (Neumann et al., 2019). Carolsfeld et al. (2003) reported no sperm motility after thawing the semen of P. corruscans when using Me2SO as a cryoprotectant, which differs from our results, which showed significant levels of motility with this compound. This variation is common and usually related to factors such as sperm sensitivity and the specificity of the cryopreservation protocols used, and not the cryoprotective substance itself (Pereira et al., 2019).
The VCL, VSL, and VAP velocities of the P. corruscans spermatozoa observed after thawing in the present study were relatively high and did not present significant changes between any of the cryopreservation protocols tested (P < 0.05), indicating that the freezing and thawing protocols were effective in preserving sperm cells. The total motility (TM) of sperm is the most used parameter to determine the quality and effectiveness of the cryopreservation process. However, the spermatozoa velocities VCL, VSL and VAP together with the TM are highly correlated with the fertilization capacity and hatching, because the faster spermatozoa can reach the oocyte micropyle and carry out fertilization (Viveiros et al., 2010; Gallego et al., 2013).
Gametes with morphological changes were observed in both the fresh and cryopreserved semen samples. However, there was an increase in the amount of structural damage observed in thawed sperm, which is common in studies on cryopreserved semen quality. The changes observed in fresh semen may be related to stress, reproductive diseases, feed management, and animal genetics (Solis-Murgas et al., 2011). This fact was verified by Navarro et al. (2019), who found morphological changes in 92% of the sperm analyzed and a reduction in normal sperm between the collection months of December (47%) and February (8%). According to those authors, this can likely be attributed to stress during breeder handling, as well as the collection process and changes in climatic parameters between months.
The increase in morphological changes after freezing is due to osmotic stress, dehydration, low temperature exposure, ice crystal formation, and cryoprotectant action. These changes in sperm cells by freezing/thawing processes can cause cell death or damage to cell structures (membrane cells, intermediate section, and flagellum). The relationship between the presence of morphological abnormalities in the spermatozoa and the ability to fertilize is direct, mainly linked to defects in the flagellum and in the intermediate piece where the mitochondria are located, or any other alteration that affects the motility capacity of the gametes that reduces the fertilizing capacity of the sperm (Ninhaus-Silveira et al., 2006; Galo et al., 2019; Medina-Robles et al., 2019; Borges et al., 2020).
DNA integrity is one of the important parameters in assessing the quality of thawed semen. It is known that during cryopreservation the formation of ice crystals, the production of reactive oxygen species (ROS), and cryoprotective substances can cause DNA damage. Even having damaged DNA, the sperm may fertilize the oocyte, however embryo development may be impaired, which leads to reduced fertilization rates (Pérez-Cerezales et al., 2011; Gallego et al., 2013 ).
Unlike the other parameters analyzed to evaluate the quality of the freeze/thaw process, the comet assay analysis showed a significant positive effect of DMA use as an internal cryoprotectant regardless of its concentration, as it resulted in less DNA fragmentation in P. corruscans. The results suggested that amides offered more protection to the DNA than other cryoprotective agents, such as glycerol, MG, and Me2SO due to their low molecular weight and viscosity, which facilitates their transition through the cell membrane, thus minimizing the cell damage caused by osmotic stress (Varela-Junior et al., 2012; Figueroa et al., 2016; Perry et al., 2019). The use of Me2SO has been effective in protecting sperm DNA of several species, including Mugil cephalus (Balamurugan et al., 2019), Steindachneridion scriptum (Pereira et al., 2019), and Salmo salar (Figueroa et al., 2016).
Based on the results obtained and, on the considerations raised, the semen of P. corruscans can be frozen using the proposed cryogenic protocols with the extenders used, taking into account that the extenders that had DMA as cryoprotectant substance provided better protection of the gamete DNA.
Acknowledgments
The authors would like to thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the financial support and the scholarship granted and to Prof. Dr. Ana Viveiros from the Universidade Federal de Lavras, MG for the seminal analyzes at CASA and the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP process 2012/12522–9), for financial support.
References
Ahn JY, Park JY, Lim HK. Effects of different diluents, cryoprotective agents, and freezing rates on sperm cryopreservation in Epinephelus akaara. Cryobiology. 2018; 83:60–64. https://doi.org/10.1016/j.cryobiol.2018.06.003
Balamurugan R, Prapaporn W, Munuswamy N. Sperm activation and effects of cryopreservation on motility, ultrastructureand DNA integrity in Grey mullet, Mugil cephalus. Aquac Rep. 2019; 14:100204. https://doi.org/10.1016/j.aqrep.2019.100204
Barzotto E, Oliveira M, Mateus L. Reproductive biology of Pseudoplatystoma corruscans (Spix and Agassiz, 1829) and Pseudoplatystoma reticulatum (Eigenmann and Eigenmann, 1889), two species of fisheries importance in the Cuiabá River basin, Brazil. J Appl Ichthyol.2017; 33(1):29–36. https://doi.org/10.1111/jai.13162
Bashiyo-Silva C, Costa RS, Ribeiro DC, Senhorini JA, Veríssimo-Silveira R, Ninhaus-Silveira A. Hormonal induction of Brycon cephalus (Characiformes, Characidae) to spermiation using D-ala6, pro9net-mGnRH + metoclopramide. Zygote. 2015; 24(3):319–25. https://doi.org/10.1017/S0967199415000210
Beirão J, Boulais M, Gallego V, O’Brien JK, Peixoto S, Robeck TR, Cabrita E. Sperm handling in aquatic animals for artificial reproduction. Theriogenology. 2019; 133:161–78. https://doi.org/10.1016/j.theriogenology.2019.05.004
Borges AM, Araújo KO, Pivato I, Navarro RD. Ultraestrutura e criopreservação de sêmen de jundiá amazônico (Leiarius marmoratus) em cativeiro. Arq Bras Med Vet Zootec. 2020; 72(1):253–62. https://doi.org/10.1590/1678-4162-10709
Caneppele D, Sanches EA, Romagosa E. Sperm production of Steidachneridion parahybae (Steindachner, 1877) and the effect of hormonal induction throughout one reproductive cycle. J Appl Ichthyol. 2015; 31(1):54–61. http://dx.doi.org/10.1111/jai.12732
Carolsfeld J, Godinho HP, Zaniboni-Filho E, Harvey BJ. Cryopreservation of sperm in Brazilian migratory fish conservation. J Fish Biol. 2003; 63(2):472–89. https://doi.org/10.1046/j.1095-8649.2003.00170.x
Eilts BE. Theoretical aspects of canine cryopreserved semen evaluation. Theriogenology 2005; 64(3):685–91. https://doi.org/10.1016/j.theriogenology.2005.05.017
Ferreira AA, Nuñer APO, Luz RK, Tataje DAR, Esquivel JR, Restrepo JB. Avaliação qualitativa e quantitativa do sêmen do jundiá, Rhamdia quelen. B Inst Pesca. 2001; 27:57–60.
Figueroa E, Valdebenito I, Merino O, Ubilla A, Risopatrón J, Farias JG. Cryopreservation of Atlantic Salmon Salmo salar sperm: effects on sperm physiology. J Fish Biol. 2016; 89(3):1537–50. https://doi.org/10.1111/jfb.13052
Gallego V, Pérez L, Asturiano JF, Yoshida M. Relationship between spermatozoa motility parameters, sperm/egg ratio, and fertilization and hatching rates in pufferfish (Takifugu niphobles). Aquaculture. 2013; 416–417:238–43. https://doi.org/10.1016/j.aquaculture.2013.08.035
Galo JM, Streit-Junior DP, Oliveira CA, Povh JP, Fornari DC, Digmayer M, Ribeiro RP. Quality of fresh and cryopreserved semen and their influence on the rates of fertilization, hatching and quality of the larvae of Piaractus mesopotamicus. Braz J Biol. 2019; 79(3):438–45. https://doi.org/10.1590/1519-6984.182391
Herrera-Cruz EE, Aristizabal-Regino JL, Yepes-Blandón JA, Estrada-Posada AL, Espinosa-Araújo JA, Atencio-García VJ. Criopreservación de semen de bagre rayado Pseudoplatystoma magdaleniatum con tres diferentes crioprotectores. Rev Colomb Biotecnol. 2019; 21(2):55–62. http://dx.doi.org/10.15446/rev.colomb.biote.v21n2.77847
Klaude M, Eriksson S, Nygren J, Ahnström G. The comet assay: mechanisms and technical considerations. Mutation. 1996; 363(2):89–96. https://doi.org/10.1016/0921-8777(95)00063-1
Kobayashi H, Sugiyama C, Morikawa Y, Hayashi M, Sofuni T. A comparison between manual microscopic analysis and computerized image analysis in the single cell gel electrophoresis assay. MMS Commun. 1995; 3:103–15. https://www.scienceopen.com/document?vid=3c9a53e6-a18e-49ae-8655-11b6acd50b63
Kowalski RK, Cejko BI. Sperm quality in fish: Determinants and affecting factors. Theriogenology. 2019; 135:94–108. https://doi.org/10.1016/j.theriogenology.2019.06.009
Medina-Robles VM, Guaje-Ramírez DN, Marin-Cossio LC, Sandoval-Vargas LY, Cruz-Cassalas PE. Crioconservación seminal de Colossoma macropomum como estrategia de producción y conservación en la Orinoquia Colombiana. Orinoquia. 2019; 23(1):15–24. http://dx.doi.org/10.22579/20112629.537
Ministério do Meio Ambiente (MMA). Portaria Nº 148, de 7 de junho de 2022. 2022. Available from: https://www.in.gov.br/en/web/dou/-/portaria-mma-n-148-de-7-de-junho-de-2022-406272733
Navarro RD, Lemos JV, Ribeiro MT. Quality of semen in the reproductive cycle of Cachara (Pseudoplatystoma fasciatum) raised in captivity. Acta Sci Biol Sci. 2019; 41:1–06. https://doi.org/10.4025/actascibiolsci.v41i1.46517
Neumann G, Sanches PV, Bombardelli RA. Effects on fertility of motile sperm to egg ratio with use of cryopreserved Rhamdia quelen semen at different post-activation times. Anim Reprod Sci. 2019; 201:84–92. https://doi.org/10.1016/j.anireprosci.2019.01.001
Ninhaus-Silveira A, Foresti F, Veríssimo-Silveira R, Senhorini JA. Seminal analysis, cryogenic preservation, and fertility in matrinxã fish, Brycon cephalus (Günther, 1869). Braz Arch Biol Techn. 2006; 49(4):651–59. https://doi.org/10.1590/S1516-89132006000500015
Ogier de Baulny B, Labbé C, Maisse G. Membrane integrity, mitochondrial activity, ATP content, and motility of the European catfish (Silurus glanis) testicular spermatozoa after freezing with different cryoprotectants. Cryobiology. 1999; 39(2):177–84. https://doi.org/10.1006/cryo.1999.2200
Pereira JR, Pereira FA, Perry CT, Pires DM, Muelbert JRE, Garcia JRE, Corcini CD, Varela-Junior AS. Dimethylsulfoxide, methanol and methylglycol in the seminal cryopreservation of Suruvi, Steindachneridion scriptum. Anim Reprod Sci. 2019; 200:7–13. https://doi.org/10.1016/j.anireprosci.2018.09.024
Pérez-Cerezales S, Gutiérrez-Adán A, Martínez-Páramo S, Beirão J, Herráez MP. Altered gene transcription and telomere length in trout embryo and larvae obtained with DNA cryodamaged sperm. Theriogenology. 2011; 76(7):1234–45. https://doi.org/10.1016/j.theriogenology.2011.05.028
Perry CT, Corcini CD, Anciuti AN, Otte MV, Soares SL, Garcia JRE, Muelbet JRE, Varela-Junior AS. Amides as cryoprotectants for the freezing of Brycon orbignyanus sperm. Aquaculture. 2019; 508:90–97. https://doi.org/10.1016/j.aquaculture.2019.03.015
Porto-Foresti F, Hashimoto DT, Prado FD, Senhorini JA, Foresti F. Genetic markers for the identification of hybrids among catfish species of the family Pimelodidae. J Appl Ichthyol. 2013; 29(3):643–47. https://doi.org/10.1111/jai.12092
RStudio Team. RStudio: Integrated Development for R. RStudio, Inc., Boston, MA. 2015. Available from: http://www.rstudio.com/
Sant’Anna JFM, Moraes-Neto A, Castro JP, Sczepanski TS, Pohv JA, Lopera-Barrero N, Artoni RF. Quantitative and qualitative variables of semen from surubim do Iguaçu, Steindachneridion melanodermatum Garavello, 2005 (Siluriformes: Pimelodidae). Braz K Biol. 2019; 79(1):1–05. https://doi.org/10.1590/1519-6984.09016
Silveira WF, Kavamoto ET, Cestarolli MA, Godinho HM, Ramos SM, Ninhaus-Silveira A. Spermatogenic evaluation, cryogenic preservation and fertility of the “pacu”, Piaractus mesopotamicus [Holmberg, 1887], sperm, from induced spawning. Bol Inst Pesca. 1990; 17:1–13.
Singh NP, Mccoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low-levels of DNA damage in individual cells. Exp Cell Res. 1988; 175(1):184–91. https://doi.org 10.1016/0014-4827(88)90265-0
Solis-Murgas LD, Felizardo VO, Ferreira MR, Andrade ES, Veras GC. Importância da avaliação dos parâmetros reprodutivos em peixes nativos. Rev Bras Reprod Anim. 2011; 35(2):186–91.
Varela-Junior AS, Corcini CD, Gheller SMM, Jardim RD, Lucia Jr. T, Streit Jr. DP, Figueiredo MRC. Use of amides as cryoprotectants in extenders for frozen sperm of tambaqui, Colossoma macropomum. Theriogenology. 2012; 78(2):244–51. http://dx.doi.org/10.1016/j.theriogenology.2012.02.029
Viveiros ATM, Nascimento AF, Orfão LH, Isaú ZA. Motility and fertility of the subtropical freshwater fish streaked prochilod (Prochilodus lineatus) sperm cryopreserved in powdered coconut water. Theriogenology. 2010; 74(4):551–56. https://doi.org/10.1016/j.theriogenology.2010.03.018
Viveiros ATM. Current status of sperm cryopreservation in siluriform catfishes. In: Tiersch TR, Green CC, editors. Cryopreservation in aquatic species, 2nd Edition. Louisiana: World Aquaculture Society; 2011. p.387–97.
Authors
![]() Johanna Maritza Cano Velarde1
Johanna Maritza Cano Velarde1 ![]() ,
, ![]() Natalia Marins Bastos2,
Natalia Marins Bastos2, ![]() Laícia Carneiro-Leite3,
Laícia Carneiro-Leite3, ![]() Laís Pedroso Borges3,
Laís Pedroso Borges3, ![]() Edson Guilherme Vieira4,
Edson Guilherme Vieira4, ![]() Rosicleire Veríssimo-Silveira4 and
Rosicleire Veríssimo-Silveira4 and ![]() Alexandre Ninhaus-Silveira4
Alexandre Ninhaus-Silveira4
[1] Pós-Graduação em Biologia Animal, Instituto de Biociências, Letras e Ciências Exatas, Universidade Estadual Paulista “Júlio de Mesquita Filho” (UNESP), Rua Cristóvão Colombo, 2265, Jardim Nazareth, 15054-000 São José do Rio Preto, SP, Brazil. (JMCV) johannavelarde@yahoo.com.br (corresponding author).
[2] Pós-Graduação em Biociência Animal, Faculdade de Zootecnia e Engenharia de Alimentos (FZEA – USP), Rua Duque de Caxias,225, Jardim Elite, 13635-900 Pirassununga, SP, Brazil. (NMB) nataliambastos@usp.br.
[3] Pós-Graduação em Ciências Biológicas: Zoologia, Instituto de Biociências, Universidade Estadual Paulista “Júlio de Mesquita Filho” (UNESP), Av. Rubião Jr., s/n, 18618-970 Botucatu, SP, Brazil. (LCL) laicialeite@hotmail.com; (LPB) lais_borges27@hotmail.com.
[4] Laboratório de Ictiologia Neotropical (L.I.NEO), Departamento de Biologia e Zootecnia, Faculdade de Engenharia, UNESP,Av. Brasil, 56, 15085-000 Ilha Solteira, SP, Brazil. (EGV) edson.g.vieira@unesp.br, (ANS) alexandre.ninhaus@unesp.br, (RVS) rosicleire.verissimo@unesp.br.
Authors’ Contribution 

Johanna Maritza Cano Velarde: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Writing-original draft.
Natalia Marins Bastos: Formal analysis, Methodology.
Laícia Carneiro-Leite: Formal analysis, Writing-original draft.
Laís Pedroso Borges: Data curation, Formal analysis.
Edson Guilherme Vieira: Conceptualization, Writing-review and editing.
Rosicleire Veríssimo-Silveira: Data curation, Formal analysis, Writing-review and editing.
Alexandre Ninhaus-Silveira: Conceptualization, Funding acquisition, Methodology, Project administration, Resources, Supervision, Writing-review and editing.
Ethical Statement
All procedures were performed and approved by the Ethics Committee for the Use of Animals (CEUA) of the Faculdade de Engenharia, UNESP, Ilha Solteira, São Paulo, Brazil (CEUA-FEIS/UNESP 05/2017 process).
Competing Interests
The author declares no competing interests.
How to cite this article
Velarde JMC, Bastos NM, Carneiro-Leite L, Borges LP, Vieira EG, Veríssimo-Silveira R, Ninhaus-Silveira A. Dimethyl acetamide and dimethyl sulfoxide associated at glucose and egg yolk for cryopreservation of Pseudoplatystoma corruscans semen. Neotrop Ichthyol. 2023; 21(1):e220071. https://doi.org/10.1590/1982-0224-2022-0071
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted January 11, 2023 by Bernardo Baldisserotto
Accepted January 11, 2023 by Bernardo Baldisserotto
![]() Submitted November 9, 2022
Submitted November 9, 2022
![]() Epub February 20, 2023
Epub February 20, 2023