![]() Isadora Cristina Bianchi-Costa1
Isadora Cristina Bianchi-Costa1 ![]() ,
, ![]() Bárbara Angélio Quirino1,
Bárbara Angélio Quirino1, ![]() Ana Lúcia Paz Cardozo1,
Ana Lúcia Paz Cardozo1, ![]() Kátia Yasuko Yofukuji1,
Kátia Yasuko Yofukuji1, ![]() Matheus Henrique Ferreira1 Aleixo and
Matheus Henrique Ferreira1 Aleixo and ![]() Rosemara Fugi1,2
Rosemara Fugi1,2
PDF: EN XML: EN | Supplementary: S1 S2 S3 S4 | Cite this article
Abstract
The hydrological regime leads to fluctuations in the availability of food resources for fish, which may reflect in their diet variation. Our main goal was to analyze the relationship between water-level fluctuations and the diet and the body condition of Trachelyopterus galeatus in the Upper Paraná River floodplain. The fish used in the study were sampled quarterly along nine years, at nine sampling stations. Our results showed that diet of T. galeatus varied in response to oscillations in the hydrometric level, with the consumption of terrestrial invertebrates being positively related to hydrometric level, while the consumption of aquatic invertebrates was negatively related. The trophic niche breadth and feeding activity were not affected by hydrometric level, but the fish body condition was positively affected. The high food plasticity allows T. galeatus to consume the most available resources (e.g., terrestrial invertebrates in high water levels), which probably contributed to the increase in its body condition in higher hydrometric levels. In summary, the hydrological dynamic is fundamental for the diet variation of omnivorous fish, which can take advantage of the resource availability according to the hydrometric level, increasing its body condition when allochthonous resources as terrestrial invertebrates are supposed to be more available.
Keywords: Body condition, Feeding activity, Hydrological cycle, Trachelyopterus galeatus, Trophic ecology.
O regime hidrológico leva a flutuações na disponibilidade de recursos alimentares para os peixes, o que pode refletir na variação de sua dieta. O principal objetivo foi analisar a relação entre as oscilações do nível hidrométrico e a dieta e condição corporal de Trachelyopterus galeatus na planície de inundação do alto rio Paraná. Os peixes foram amostrados trimestralmente durante nove anos, em nove locais. Nossos resultados mostraram que a dieta de T. galeatus variou em resposta às oscilações do nível hidrométrico, com o consumo de invertebrados terrestres sendo positivamente relacionado com o nível hidrométrico, enquanto o consumo de invertebrados aquáticos foi negativamente relacionado. O nicho trófico e a atividade alimentar não foram afetados pelo nível hidrométrico, mas a condição corporal dos peixes foi positivamente afetada. A alta plasticidade alimentar permite que T. galeatus aproveite recursos mais disponíveis (por exemplo, invertebrados terrestres em níveis elevados), o que provavelmente contribui para o aumento da condição corporal em níveis hidrométricos elevados. Em resumo, a dinâmica hidrológica é fundamental para a variação da dieta de peixes onívoros, que podem aproveitar a disponibilidade de recursos de acordo com o nível hidrométrico, aumentando sua condição corporal quando recursos alóctones, como invertebrados terrestres, são supostamente mais disponíveis.
Palavras-chave: Atividade alimentar, Ciclo hidrológico, Condição corporal, Ecologia trófica, Trachelyopetrus galeatus.
Introduction
The natural dynamics of the hydrological cycle in river-floodplain systems are characterized by alternating periods of drought and flood. The hydrological regime is the main factor that influences the functioning of these ecosystems, changing the connections among environments, regulating the quantity and quality of habitats for fish populations (Junk et al., 1989), controlling the growth of these animals (De Graaf, 2003), and changing the limnological characteristics and biological processes (Thomaz et al., 2004). Hydrological cycles with regular drought and flood periods are essential for maintaining a high productivity and diversity of these ecosystems (Junk et al., 1989; Agostinho et al., 2004a). These seasonal changes lead to fluctuations in the availability and quality of food resources for fish (Goulding et al., 1988; Luz-Agostinho et al., 2008; Correa, Winemiller, 2014; Quirino et al., 2017).
In low water periods, a reduction in the abundance of resources is expected (Bonvillain, Fontenot, 2020), which leads to the intensification of interactions, such as competition and predation (Junk et al., 1989; Gomes et al., 2012; Medeiros, Arthington, 2014), making the species diet less diversified and leading to a contraction of the trophic niche (Balcombe et al., 2005; Quirino et al., 2017). In contrast, in high water periods, there is an increase in the availability of food, when a large amount of terrestrial resources, such as fruits, seeds and insects are available to fishes that occupy the flooded area (Goulding, 1988; Junk et al., 1989; Esteves, Galetti, 1995; Pereira et al., 2011; Quirino et al., 2017). Greater ecological opportunity, that is, greater availability of resources that may be exploited, associated with the high waters period (Araújo et al., 2011) contribute greatly to the diet composition, generally expanding the trophic niche breadth some fish species (Delbeek, Williams 1987; Walker et al., 2013; Quirino et al., 2015, 2017; Brambilla et al., 2019). The inclusion of more profitable food resources that are available during the high-water period (Costa-Pereira et al., 2017), may reflect in a better body condition for omnivorous fish, since the condition of a fish is affected, among other factors, by environmental and nutritional variations (Abujanra et al., 2009; Jin et al., 2015; Cardozo et al., 2018). The inclusion of new resources may release individuals from intense intraspecific competition (Araújo et al., 2011), probably favoring their well-being. Especially for floodplain systems, understanding the relationship between water-level fluctuations and the diet variability can help to clarify the mechanisms that allow the persistence and the high abundance of a species in a dynamic environment (Costa-Pereira et al., 2017).
Floodplains under the influence of dams upstream, modifies the natural dynamics of the hydrological regime (Agostinho et al., 2004b; Roberto et al., 2009; Santos et al., 2017), changing the intensity, frequency, duration, and the time when floods occur, besides reducing the seasonally flooded areas (Thomaz et al., 2004; Agostinho et al., 2007; Souza-Filho, 2009). The water flow in the Upper Paraná River floodplain is controlled by the presence of a cascade of reservoirs upstream and this control has become more intense with the construction of the Porto Primavera Reservoir (formed in 1998) (Souza-Filho, 2009). Although the river dynamics are still associated with the hydrological regime, and the oscillation between periods of drought and flood still define the composition of the environments in floodplains (Thomaz et al., 2004; Agostinho et al., 2004b; Santos et al., 2017), the control of the water flow can lead to extreme droughts, which do not represent natural disturbances (Moi et al., 2020).
Prolonged drought periods can promote the maximization of competitive exclusion by increasing local extinctions (Thomaz et al., 2007). For example, Moi et al. (2020) observed that periods of extreme drought in the Paraná River, which become more frequent after the damming of the river, negatively impacted the benthic community and, consequently, the ecosystem, since macroinvertebrates are one of the main food resources for several organisms, especially fish. Likewise, the elimination of the annual flood pulse in the Barataria basin (Mississippi River floodplain) reduced the abundance of freshwater crayfish, the main prey for carnivorous fish, and potentially altered all trophic dynamics of this system (Bonvillain, Fontenot, 2020).
Based on the premises that flood pulses change the composition, amount, and quality of food resources, our main goal was to analyze the relationship between water-level fluctuations and Trachelyopterus galeatus Linnaeus, 1766 (Siluriformes: Auchenipteridae) diet and body condition variations. It is one of most abundant species in the Upper Paraná River floodplain (Julio-Júnior et al., 2009; Tonella et al., 2018; Long Term Ecological Research (LTER) (unpublished data), a medium size omnivorous species that has a broad trophic niche breadth (Tonella et al., 2018; Garcia et al., 2018, 2020). Omnivory can be considered a beneficial trait for many species, once the broad feeding spectrum allows the species to exploit a high diversity of food items, which can generate a relaxation in competitive interactions (Ricciardi, Rasmussen, 1998; Ribeiro et al., 2007; Nurkse et al., 2016). Changes in diet associated with the water level, increasing, or reducing the trophic niche, can be considered evolutive answers to the environmental conditions associated with water level oscillations in river-floodplain ecosystems. Thus, we hypothesize that T. galeatus changes its feeding habit regarding diet composition and the trophic niche breadth according to the water level once this species is able to take advantage of the different resources available in periods of increased water level. We expect that (i) the consumption of allochthonous items for T. galeatus is positively affected by increasing hydrometric levels; (ii) the consumption of autochthonous items for T. galeatus is negatively affected by increased hydrometric level; (iii) the trophic niche breadth is positively influenced by increased hydrometric level; and (iv) an increase in the feeding activity and body condition with the increase of the hydrometric level.
Material and methods
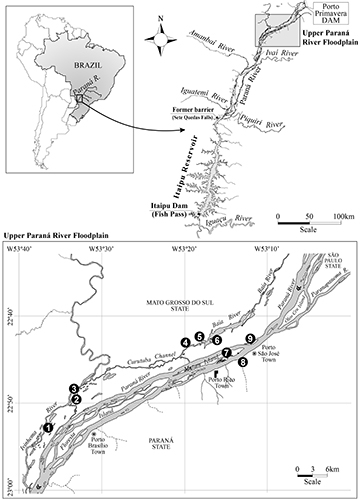
Study area. The Upper Paraná River floodplain is located between the Porto Primavera dam (upstream) and the beginning of the Itaipu reservoir (Fig. 1). This is the last undammed stretch of the Upper Paraná River in the Brazilian territory, with 230 km in length. It presents a great diversity of habitats, including the alluvial plain with numerous secondary channels, connected and isolated lakes, and the main channels of the Paraná, Baía, and Ivinhema rivers (Agostinho et al., 2007). Three protected areas are located in this region: Várzeas do Rio Ivinhema State Park, Environmental Protection Area of the Paraná River Islands and Floodplains, and Ilha Grande National Park. In this study, nine environments were sampled: the main channel of the Paraná, Baía, and Ivinhema rivers (three sampling stations) and six lakes (Fig. 1).
FIGURE 1| Location of the Upper Paraná River floodplain (downstream of the Porto Primavera dam and upstream of the Itaipu reservoir) and the nine sampling stations on the Paraná, Baía, and Ivinhema rivers.
Sampling. The fish used in this study were sampled quarterly (March, June, September and December) along eight years (2005, 2008, 2009, 2010, 2013, 2016, 2018, 2019), and in March and September of 2017, at nine sampling stations, being three rivers (Paraná, Baía and Ivinhema) and six lakes (Fig. 1). Fish were sampled with gillnets with different mesh sizes (3 to 16 cm between opposite nodes), which were exposed for 24 h, and fish were removed at 8, 16 and 22 h. Voucher specimens are deposited at the Coleção Ictiológica of the Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), of the Universidade Estadual de Maringá (NUP 11104).
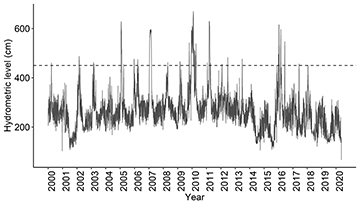
Hydrometric level. Data from the hydrometric levels of the Paraná River were obtained from the National Water Agency (Hidroweb; www.ana.gov.br), measured at the fluviometric station (64575003) located in the municipality of Porto São José, Paraná State, Brazil. The hydrological level is measured daily (Fig. 2), however, in view that fish sampling occurs quarterly, we summarize the river level in a quarterly mean
FIGURE 2| Daily variation in the level of the Paraná River between 2000 and 2020 (provided by the Agência Nacional de Águas e Saneamento Básico – ANA). The horizontal line represents the 4.5 m level when the Paraná River overflows.
The hydrological cycle of the floodplain has been altered due to upstream reservoirs, which was intensified after the construction of the Porto Primavera reservoir in 1998 (Souza-Filho, 2009). However, the floodplain still has a flood pulse, with the flood period usually occurring from November/December to April/May, with maximum hydrometric levels prevailing between January and March, and the drought between June and October with minimum values between July and September (Agostinho et al., 2004c).
Laboratory procedures. Fish were measured (Standard Length = SL in mm), weighted (g) and their stomachs were visually assessed for the degree of stomach repletion using the following numerical scale: DR0 = empty stomach; DR1 = up to 25%; DR2 = 25% to 75%; DR3 ≥ 75% (Pelicice, Agostinho, 2006; Kovalenko et al., 2009). Stomachs with DR2 and DR3 were analyzed and, the food contents were identified at the lowest possible taxonomic level (McCafferty, 1983; Elmoor-Loureiro, 1997) with a stereoscopic microscope. After identification, food items were quantified using the volumetric method (Hellawell, Abel, 1971; Hyslop, 1980), in two ways: (i) by displacement of the water column, using graduated beakers; and (ii) using a gridded dish, in which the volume of the items was obtained in 1 mm³, and later transformed into mL (Bastos et al., 2013). The second method was used in cases where the items were very small in size, which makes it impossible to measure the volume in graduated cylinders. For some analysis items were grouped into large ones, which are: i) Allochthonous items: terrestrial invertebrates and plants; ii) Autochthonous items: aquatic invertebrates, detritus, and fish.
Data analysis. To verify whether consumption of allochthonous items for T. galeatus is positively affected by increased hydrometric level (prediction i), we performed a generalized mixed model (GLMM), with the Gaussian distribution, using the quarterly mean of hydrometric level as the explanatory variable (fixed effect) and the volume of allochthonous items from each sampling as the response variable. Furthermore, we performed another two models using the volume of each group of allochthonous items separately (terrestrial invertebrates and terrestrial plants) as the response variables.
To verify whether consumption of autochthonous items for T. galeatus is negatively affected by increased hydrometric level (prediction ii), we performed a GLMM, with the Gaussian distribution, using the quarterly mean hydrometric level as the explanatory variable (fixed effect) and the volume of autochthonous items from each sampling as the response variable. Furthermore, we performed another three models using the volume of each group of aquatic items separately (aquatic invertebrates, detritus and fish) as the response variables.
Variations in the trophic niche breadth were assessed using the Permutation Analysis of Multivariate Dispersion (PERMIDISP, Anderson, 2006), performed using the vegan package (Oksanen et al., 2019). PERMDISP assesses the dispersion of the diet in the multivariate space, that is, the mean distance of individuals in relation to the centroid of each population. Thus, if the population has a greater mean distance from the centroid, the greater the inter-individual variability and consequently the greater the trophic niche breadth (Correa, Winemiller, 2014). To verify the influence of the hydrometric level in trophic niche breadth (prediction iii) we performed a GLMM, with the Gaussian distribution, using the hydrometric level as the explanatory variable and the mean of centroid distance (CD) from each sampling as the response variable.
The body condition was assessed through the mean of Fulton’s Condition Factor for each sampling, calculated according to the equation: K = 100*(W/L³) where W is the body weight in grams and L is the standard length in cm. Factor 100 is used to bring K close to unity (Froese, 2006). The body condition analyze is a useful tool for comparing the condition, fatness, or well-being of a fish (Tesch, 1968). We also assessed the feeding activity of T. galeatus by the mean degree of stomach repletion (mDR), expressed by: mDR = (N0*0) + (N1*1) + (N2*2) + (N3*3)/N, where N0, N1, N2 and N3 correspond to the number of individuals with stomach fullness of 0, 1, 2 and 3, respectively, and N is the total number of analyzed individuals in each sampling (Carniatto et al., 2012, 2019). The body condition of a species can be influenced by feeding activity (Pereira et al., 2016; Cardozo et al., 2018). We performed a generalized mixed model (GLMM) using the Gaussian distribution in order to assess the relationship between the hydrometric level (fixed effect) and the K and mDR data of each sampling (response variable) (prediction iv).
Statistical analyses and graphs were performed with R 4.0 software (R Development Core Team, 2020), using the “lme” function from the nlme package (Pinheiro et al., 2021) for analysis and the package ggplot2 for graphs (Wickham, 2016). Because mixed models allow for the inclusion of a random term (Zuur et al.,2009), for all GLMMs, we included the sampled years as the random effect because close years may be very similar and therefore have an influence in our analysis. In order to achieve homoscedasticity of the data, for the models of allochthonous resources (terrestrial invertebrates, terrestrial plants and detritus), the response variables were log x+1 transformed. The model assumption was visually tested for residual normality and homoscedasticity. Cook’s distances were used to graphically analyze influential observations (Zuur et al., 2009). In addition, the Bonferroni test was used to detect outliers (Cook, Weisberg, 1982), performed using the car package (Fox et al.,2022) and did not show significant results (Tab. S1). Model quality was assessed using pseudo marginal R² (the proportion of variation explained by fixed effects) and pseudo conditional R² (the proportion of variation explained by fixed and random effects) (Nakagawa, Schielzeth, 2013). All assumptions were checked and values of P < 0.05 were considered statistically significant.
Results
The stomachs of 295 individuals of Trachelyopterus galeatus,whose body length ranged from 6.1 cm to 18.3 cm, were analyzed. The T. galeatus diet was composedof 27 items from allochthonous and autochthonous origins, including higher plants, aquatic and terrestrial invertebrates, fish and detritus (Tab. S2). Among the resources of allochthonous origin, the invertebrates stood out, comprising a richness of 17 prey resources, among which adult Coleoptera, Orthoptera and Formicidae composed the species diet in almost all samplings (Tab. S2). Among the terrestrial plants, leaves, fruit and seeds occurred in the diet of T. galeatus. On the other hand, autochthonous items were composed of invertebrates, detritus and fish. Odonata nymph, Ephemeroptera larvae and Diptera pupae were the predominant aquatic invertebrates in the diet, and fish items were consumed at most samples (Tab. S2).
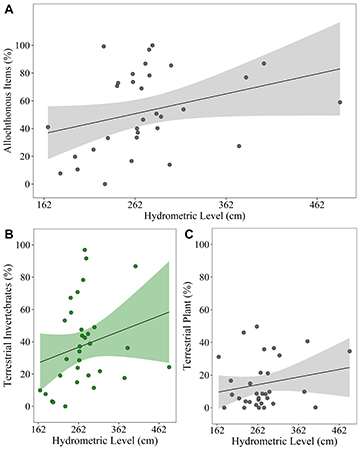
Considering the origin of food items consumed by T. galeatus as a function of the hydrometric level (prediction i), the percentage of consumption of allochthonous items showed a tendency to increase as the hydrometric level increases, but the model showed only a marginally significant result (pseudo-R²m = 0.1 – pseudo-R²c = 0.1 – p = 0.077) (Tab. 1; Fig. 3A; Tab. S3). On the other hand, when analyzing the consumption of each group of allochthonous items separately, the consumption of terrestrial invertebrates was positively affected by hydrometric level (pseudo-R²m = 0.17 – pseudo-R²c = 0.17 – p = 0.021) (Tab. 1; Fig. 3B; Tab. S4), while the percentage of terrestrial plant consumption was not affected by the hydrometric level (pseudo-R²m = 0.02 – pseudo-R²c = 0.02 – p = 0.389) (Tab. 1; Fig. 3C; Tab. S4).
FIGURE 3| A. Relationship between the hydrometric level and the consumption of all allochthonous items; B. The consumption of terrestrial invertebrates; C. And the consumption of terrestrial plants for Trachelyopterus galeatus. The green graphs indicate a significant positive influence of the variable as a function of hydrometric level, while grayscale graphs indicate a non-significance.
TABLE 1 | Results of generalized linear mixed model analysis between the consumption of allochthonous items by Trachelyopterus galeatus in function of the hydrometric level of the floodplain. P-values < 0.05 are in bold. SE = Standard Error.
Parameter estimate | SE | t-value | P-value | ||
Total | Intercept | 13.19 | 21.79 | 0.55 | 0.55 |
Hydrometric level | 0.14 | 0.08 | 1.85 | 0.07 | |
Terrestrial invertebrates | Intercept | -7.21 | 4.25 | -1.69 | 0.10 |
log (level+1) | 1.88 | 0.76 | 2.48 | 0.02 | |
Terrestrial plant | Intercept | 1.34 | 0.96 | 1.39 | 0.18 |
log (level+1) | 0.002 | 0.003 | 0.88 | 0.39 | |
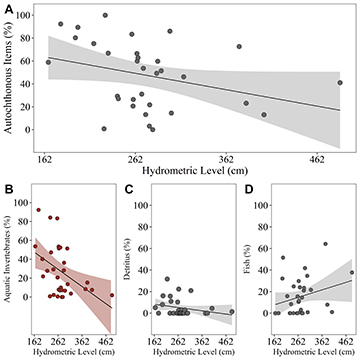
Regarding the consumption of autochthonous items, the percentage of consumption showed a negative tendency as a function of the hydrometric level, but the model only showed a marginally significant result (pseudo-R²c = 0.10 – pseudo-R²m = 0.10 – p = 0.077) (Tab. 2; Fig. 4A; Tab. S3). As for the volumetric percentage of aquatic invertebrates in the diet of T. galeatus, unlike the consumption of terrestrial invertebrates, it was negatively affected by the hydrometric level (pseudo-R²c = 0.17 – pseudo R²m = 0.17 – p = 0.012) (Tab. 2; Fig. 4B; Tab. S4), while for detritus consumption there was no significance of the model (pseudo-R²c = 0.05 – pseudo-R²m = 0.05 p = 0.238) (Tab. 2; Fig. 4C; Tab. S4). As well, the percentage of consumption of fish species for T. galeatus, was not affected by the hydrometric level (pseudo-R²c = 0.06 – pseudo-R²m = 0.06 – p = 0.354) (Tab. 2; Fig. 4D; Tab. S4).
FIGURE 4| A. Relationship between the hydrometric level and the consumption of all autochthonous items; B. The consumption of aquatic invertebrates; C. The consumption of detritus; D. And the consumption of fish for Trachelyopterus galeatus. Red graphs indicate a significant negative influence of the variable as a function of hydrometric level, while grayscale graphs indicate a non-significance.
TABLE 2 | Results generalized linear mixed model analysis between the consumption of autochthonous items by Trachelyopterus galeatus in the function of the hydrometric level of the floodplain. P-values < 0.05 are in bold. SE = Standard Error.
| Parameter estimate | SE | t-value | P-value | |
Total | Intercept | 86.81 | 21.79 | 3.98 | <0.001 |
Hydrometric level | -0.14 | 0.08 | -1.85 | 0.07 | |
Aquatic invertebrates | Intercept | 77.14 | 18.84 | 4.09 | 0.005 |
Hydrometric level | -0.18 | 0.07 | -2.73 | 0.01 | |
Detritus | Intercept | 2.08 | 0.94 | 2.21 | 0.03 |
Hydrometric level | -0.004 | 0.003 | -1.21 | 0.238 | |
Fish | Intercept | -4.49 | 20.81 | -0.22 | 0.83 |
Hydrometric level | 0.07 | 0.08 | 0.95 | 0.35 | |
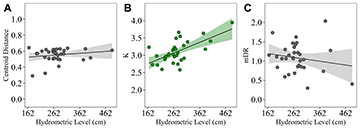
The values of the trophic niche breadth, evaluated through the PERMDISP, ranged from 0.3 to 0.6, remaining above 0.5 in 87.5% of samplings. The results did not show any tendency, furthermore, the linear model did not show any significant effect of the hydrometric level on trophic niche breadth (adjusted-R² = 0.04 – p = 0.371) (Fig. 5A; Tab. 3), rejecting our third prediction. The GLMM model for the body condition (Fig. 5B) showed a significant and positive relationship for K and the hydrometric level (p < 0.01; Tab. 3), however, the data varied greatly between the levels of the random effects (years sampled – pseudo-R²m = 0.28 – pseudo-R²c = 0.52). We did not find any evidence that the feeding activity of fish (mDR; Fig. 5C) increased with the hydrometric level, rather it presented a slight decline. The model did not show any significant effect of the hydrometric level on feeding activity and the data varied greatly with the increase of the hydrometric level (Tab. 3). This model also varied greatly between the levels of the random effects (years sampled – pseudo-R²m = 0.02; pseudo-R²c = 0.08).
FIGURE 5| A. Relationship between the hydrometric level and the trophic niche breadth; B. Body condition; C. And the feeding activity of Trachelyopterus galeatus. The green graphs indicate a significant positive influence of the variable as a function of hydrometric level, while grayscale graphs indicate a non-significance.
TABLE 3 | Results of generalized linear mixed model analysis for Centroid Distance (CD), K and mDR values of Trachelyopterus galeatus with hydrometric level. SE = Standard Error.
| Parameter estimative | SE | t-value | P-value | |
CD | Intercept | 1.999 | 0.223 | 8.959 | <0.0001 |
Hydrometric Level | -0.00069 | <0.0001 | -0.894 | 0.371 | |
K | Intercept | 2.394 | 0.197 | 12.123 | <0.0001 |
Hydrometric Level | 0.002 | 0.0006 | 3.692 | 0.0013 | |
mDR | Intercept | 1.326 | 0.292 | 4.533 | 0.0002 |
Hydrometric Level | -0.00088 | 0.001 | -0.853 | 0.4024 |
Discussion
Our results show that the diet of Trachelyopterus galeatus varied in response to oscillations in the hydrometric level, with the consumption of different food resources or different proportions of them. The consumption of allochthonous resources tends to increase with the increase of the hydrometric level, while the consumption of autochthonous resources tends to decrease. The terrestrial invertebrates were positively influenced by the hydrometric level, while the aquatic invertebrates were negatively influenced by the hydrometric level, both showing significant relationships. However, variations of the hydrometric level showed no influence on the trophic niche breadth, which hardly varied in most of the samplings. As expected, fish body condition was positively affected by the increase in the hydrometric level, while the feeding activity was not influenced by such increase. These results indicate that different hydrological conditions could influence the availability of resources in the environment, and it is likely that fish with trophic opportunism change their diet as a consequence.
The diet of T. galeatus was composed of many food items (27), including fish, plants (leaves, seeds and fruits), detritus, and invertebrates (mainly insects). A broad diet allows the exploitation of different resources according to availability, providing a competitive advantage (Courant et al., 2017). Trachelyopterus galeatus has an omnivorous habit (Tonella et al., 2018; Garcia et al., 2020), and our result showed that it presents different diet patterns according to river level fluctuations, changing it according to the availability of allochthonous and autochthonous resources provided by the hydrological dynamics (Prejs, Prejs, 1987; Winemiller, Kelso-Winemiller, 2003; Quirino et al., 2017). Some omnivorous species can change their trophic position according to variations in the hydrometric level (e.g., Quirino et al.,2015; McMeans et al., 2019). In our study, the consumption of these basal resources (e.g., fruits and seeds) did not show a significant relationship with the hydrometric level, but there was a trend to replace autochthonous resources with allochthonous ones. Terrestrial invertebrates may represent a high quality food resource because of their high energy density compared to aquatic ones (Sullivan et al., 2014) and may also justify the high consumption of terrestrial invertebrates by T. galeatus, once they were higher in relation to the consumption of plants.
As expected, there was a trend to increase the consumption of allochthonous items with the increase in the hydrometric level (prediction i), which was particularly significant for the consumption of invertebrates. Several studies suggest an increase in the consumption of allochthonous resources, such as invertebrates, in high water periods compared to low water periods (Quirino et al., 2017; Heng et al., 2018; Castello et al., 2019). The high consumption of terrestrial items by T. galeatus is related to the flooding of terrestrial environments as the hydrometric level increases. In these conditions, fish occupy newly flooded areas, where they find allochthonous resources in greater proportions and for a longer time, which is an advantage. In fact, there was a trend to replace aquatic invertebrates with terrestrial ones in the diet, once these resources have a higher energy density when compared to autochthonous resources (Junk, 1983; Francis, Schindler, 2009; Sullivan et al., 2014).
As expected, the diet of T. galeatus in low water levels was mainly composed of detritus, fish and aquatic invertebrates, being the latter significantly consumed in lower hydrological levels. Although autochthonous resources are abundant throughout the year, they are most accessible during periods of low water (Abujanra et al., 2009) – when the area, water volume, depth and connectivity between aquatic environments are reduced (Gomes et al., 2012; Medeiros et al., 2014) – which leads to a concentration of aquatic organisms, facilitating predation (Wantzen et al., 2002). In order to persist during extreme levels of drought, T. galeatus was able to consume energy-dense food items, such as aquatic invertebrates and fish, which are more cost-effective prey than plants or detritus (Dorenbosch, Bakker, 2011). Trachelyopterus galeatus also consumed detritus, mainly at lower hydrological levels. Although very abundant, this food item usually has a low energy value (Barrera-Oro, 2002; Pyrzanowski et al., 2019). Nevertheless, the habit of consuming detritus and fish in different hydrometric levels may help to explain the high abundance of T. galeatus in the studied environment. Even though this fish is not a native species of this floodplain (Ota et al., 2018), it is able to exploit resources that are rarely limited (Moyle, Light, 1996), even in periods of scarcity of more energy-efficient resources.
The trophic niche breadth did not vary significantly regarding changes in the hydrometric level. In trophic ecology studies, there is no consistent pattern of trophic niche breadth for fish (Correa, Winemiller, 2014). While some studies show an expansion of the trophic niche of invertivorous fish in the flood season, when there is an increase in food abundance (Correa et al., 2009; Quirino et al., 2017), a contraction of the niche breadth was also observed in this period (Walker et al., 2013). However, species response to the abundance or limitation of resources in different hydrological seasons depends on the taxa and environments (Correa, Winemiller, 2014). In this way, the opportunistic diet of T. galeatus can explain its persistence throughout the entire hydrological cycle, regardless of water level fluctuation, because it can prey on the most profitable prey types (terrestrial or aquatic). This leads to improvements in their diet based on the abundance and availability of resources, causing low variation in trophic niche breadth across the hydrometric level.
As expected, the condition factor was associated significantly with the hydrometric level. This increase with the increase in the hydrometric level may be associated with access to new food sources due to the high connectivity between the aquatic and terrestrial environments, in contrast to low hydrometric levels, when food resources are restricted (López-Rodríguez et al., 2019). Despite the significant increase in body condition in periods of high hydrometric levels, our results showed a constancy in the degree of stomach repletion. Luz-Agostinho et al. (2008) also found that a piscivorous species, Hoplias aff. malabaricus, showed feeding activity regardless of the flood regime while body condition improved during periods of high water. The authors pointed out that the different patterns observed for feeding activity and body condition may be associated with different energy costs for searching for prey. Generally, a temporal influence on fish feeding activity is expected (Gelós et al., 2010), but specifically in relation to the dry and flood periods, there is also no consensus, as it has already been observed that periods of high water promote a more intense feeding or a null effect, depending on the fish feeding habit (Luz-Agostinho et al., 2008; Abujanra et al., 2009). In our study, T. galeatus did not show significant changes in its feeding activity, in this case, keeping it almost constant at different hydrometric levels. This demonstrates that this species maintains its efficient foraging skills regardless of the water level. This is also another favorable characteristic for this species, as it guarantees adaptation to different environmental conditions (Garcia et al., 2020) and, probably, the stability of the population.
In summary, our results showed that variations in hydrological levels affect fish diet and even their body condition, as they likely change food availability, favoring species capable of taking advantage of such different resources. Although other factors can influence fish feeding, e.g., degree of connectivity between environments, vegetation, and others aspects in the landscape scale, the sampling sites are located in an environmental protection area and are very similar considering land cover, in addition to connecting in high water periods (Thomaz et al., 2007). Our results reinforce the fundamental role of hydrological dynamics in diet composition, generating important implications for omnivorous fish feed behavior in the environment. A species with high food plasticity is likely to become a successful invader (Nurkse et al., 2016; Courant et al., 2017; Tonella et al., 2018), which is one of the reasons why T. galeatus is currently the third most captured species in this floodplain (Tonella et al., 2018). Hence, the high food plasticity, and trophic opportunism allows T. galeatus to take advantage of the most available resources, consuming predominantly aquatic invertebrates with a decrease in the hydrometric level; and terrestrial invertebrates with an increase in the hydrometric level which probably contributed to the increase in its body condition in this situation, while it kept similar trophic niche breadth and feeding activity regardless of hydrological condition.
Acknowledgments
The authors thank Sidinei M. Thomaz who greatly improved this manuscript; Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupelia), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for granting scholarship to ICBC, BAQ, ALPC, KYY and MHFA, and for financial support and infrastructure for the development of this study.
References
Abujanra F, Agostinho AA, Hahn NS. Effects of the flood regime on the body condition of fish of different trophic guilds in the Upper Paraná River flooding. Brazil. Braz J Biol. 2009; 69(2):469–79. https://doi.org/10.1590/S1519-69842009000300003
Agostinho AA, Thomaz SM, Gomes LC. Threats for biodiversity in the floodplain of the Upper Paraná River: effects of hydrological regulation by dams. Ecohydrol Hydrobiol. 2004a; 4(3):255–68.
Agostinho AA, Gomes LC, Thomaz SM, Hahn NS. The Upper Paraná river and its floodplain: main characteristics and perspectives for management and conservation. In: Thomaz SM, Agostinho AA, Hahn NS, editors. The upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004b. p.381–93.
Agostinho AA, Gomes LC, Veríssimo S, Okada EK. Flood regime, dam regulation and fish in the Upper Paraná River: effects on assemblage attributes, reproduction and recruitment. Rev Fish Biol Fish. 2004c; 14:11–19. https://doi.org/10.1007/s11160-004-3551-y
Agostinho AA, Pelicice FM, Petry AC, Gomes LC, Júlio Jr. HF. Fish diversity in the upper Paraná River basin: habitats, fisheries management and conservation. Aquat Ecosyst Health Manag. 2007; 10(2):174–86. https://doi.org/10.1080/14634980701341719
Anderson MJ. Distance-based tests for homogeneity of multivariate dispersions. Biometrics. 2006; 62(1):245–53. https://doi.org/10.1111/j.1541-0420.2005.00440.x
Araújo MS, Bolnick DI, Layman CA. The ecological causes of individual specialisation. Ecol Lett. 2011; 14:948–58. https://doi.org/10.1111/j.1461-0248.2011.01662.x
Balcombe SR, Bunn SE, Mckenzie-Smith FJ, Davies PM. Variability of fish diets between dry and flood periods in an arid zone floodplain river. J Fish Biol. 2005; 67(6):1552–67. https://doi.org/10.1111/j.1095-8649.2005.00858.x
Barrera-Oro E. The role of fish in the Antarctic marine food web: differences between inshore and offshore waters in the southern Scotia Arc and west Antarctic Peninsula. Antarct Sci. 2002; 14(4):293–309. https://doi.org/10.1017/S0954102002000111
Bastos RF, Miranda SF, Garcia AM. Diet and feeding strategy of Characidium rachovii (Characiformes, Crenuchidae) in coastal plain streams of southern Brazil. Iheringia Sér Zool. 2013; 103(4):335–41. https://doi.org/10.1590/S0073-47212013000400001
Bolnick DI, Ingram T, Snowberg LK, Stutz WE, Lau OL, Paull JS. Ecological release from interspecific competition leads to decoupled changes in population and individual niche widths. Proc R Soc B. 2010; 277(1689):1789–97. https://doi.org/10.1098/rspb.2010.0018
Bonvillain CP, Fontenot QC. The annual flood pulse mediates crayfish as a major diet constituent of carnivorous fishes in South Louisiana. Freshw Crayfish. 2020; 25(1):69–75. https://doi.org/10.5869/fc.2020.v25-1.069
Brambilla EM, Uieda VS, Nogueira MG. Does seasonality and pool connectivity influence fish trophic ecology in knickzone habitats? Stud Neotrop Fauna Environ. 2019; 54(1):22–30. https://doi.org/10.1080/01650521.2018.1518118
Cardozo ALP, Farias EGG, Rodrigues-Filho JL, Moteiro IB, Scandolo TM, Dantas DV. Feeding ecology and ingestion of plastic fragments by Priacanthus arenatus: What’s the fisheries contribution to the problem? Mar Pollut Bull. 2018; 130:19–27. https://doi.org/10.1016/j.marpolbul.2018.03.010
Carniatto N, Fugi R, Cantanhêde G, Gubiani EA, Hahn NS. Effects of flooding regime and diel cycle on diet of a small sized fish associated to macrophytes. Acta Limnol Bras. 2012; 24(4):363–72. https://doi.org/10.1590/S2179-975X2013005000007
Carniatto N, Fugi R, Quirino BA, Cunha ER, Thomaz SM. An invasive and a native macrophyte species provide similar feeding habitat for fish. Ecol Freshw Fish. 2020, 29(1):112–20. https://doi.org/10.1111/eff.12499
Castello L, Bayley PB, Fabré NN, Batista VS. Flooding effects on abundance of an exploited, long-lived fish population in river-floodplains of the Amazon. Rev Fish Biol Fish. 2019; 29(2):487–500. https://doi.org/10.1007/s11160-019-09559-x
Correa CE, Hahn S, Delariva RL. Extreme trophic segregation between sympatric fish species: the case of small sized body Aphyocharax in the Brazilian Pantanal. Hydrobiologia. 2009; 635:57–65. https://doi.org/10.1007/s10750-009-9861-2
Correa SB, Winemiller KO. Niche partitioning among frugivorous fishes in response to fluctuating resources in the Amazonian floodplain forest. Ecology. 2014; 95(1):210–24. https://doi.org/10.1890/13-0393.1
Costa-Pereira R, Tavares LE, Camargo PB, Araújo M. Seasonal population and individual niche dynamics in a tetra fish in the Pantanal wetlands. Biotropica. 2017; 49(4):531–38. https://doi.org/10.1111/btp.12434
Courant J, Vogt S, Marques R, Measey J, Secondi J, Rebelo R et al. Are invasive populations characterized by a broader diet than native populations? PeerJ. 2017; 5:e3250. https://doi.org/10.7717/peerj.3250
Delbeek JC, Williams DD. Food resource partitioning between sympatric populations of brackishwater sticklebacks. J Anim Ecol. 1987; 56(3):949–67. https://doi.org/10.2307/4959
Dorenbosch ME, Bakker ES. Herbivory in omnivorous fishes: effect of plant secondary metabolites and prey stoichiometry. Freshwater Biol. 2011; 56(9):1783–97. https://doi.org/10.1111/j.1365-2427.2011.02618.x
Elmoor-Loureiro LMA. Manual de identificação de cladóceros límnicos do Brasil. Brasília: Universa; 1997.
Esteves KZ, Galetti PM. Food partitioning among some characids of a small Brazilian floodplain lake from the Paraná River basin. Environ Biol Fishes. 1995; 42(4):375–89. https://doi.org/10.1007/BF00001468
Fox J, Weisberg S, Price B, Adler D, Bates D, Baud-Bovy G et al. Car: Companion to Applied Regression. Package R package version 3.1-0; 2022. Available from: https://CRAN.R-project.org/package=car
Francis TB, Schindler DE. Shoreline urbanization reduces terrestrial insect subsidies to fishes in North American lakes. Oikos. 2009; 118(12):1872–82. https://doi.org/10.1111/j.1600-0706.2009.17723.x
Froese R. Cube law, condition factor and weight–length relationships: history, meta–analysis and recommendations. J Appl Ichthyol. 2006; 22(4):241–53. https://doi.org/10.1111/j.1439–0426.2006.00805.x
Garcia DAZ, Tonella LH, Alves GHZ, Vidotto-Magnoni AP, Benedito E, Britton JR et al. Seasonal and habitat variations in diet of the invasive driftwood catfish Trachelyopterus galeatus in a Neotropical river basin, Brazil. J Appl Ichthyol. 2020; 36(3):326–35. https://doi.org/10.1111/jai.14035
Garcia DAZ, Vidotto-Magnoni AP, Orsi ML. Diet and feeding ecology of non–native fishes in lentic and lotic freshwater habitats. Aquat Invasions. 2018; 13(4):565–73. https://doi.org/10.3391/ai.2018.13.4.13
Gelós M, Teixeira-Mello F, Goyenola G, Iglesias C, Fosalba C, Rodriguez FG et al. Seasonal and diel changes in fish activity and potential cascading effects in subtropical shallow lakes with different water transparency. Hydrobiologia. 2010; 646:173–85. https://doi.org/10.1007/s10750-010-0170-6
Gomes LC, Bulla CK, Agostinho AA, Vasconcelos LP, Miranda LE. Fish assemblage dynamics in a Neotropical floodplain relative to aquatic macrophytes and the homogenizing effect of a flood pulse. Hydrobiologia. 2012; 685:97–107. https://doi.org/10.1007/s10750-011-0870-6
Goulding M, Carvalho LM, Ferreira EG. Rio Negro: rich life in poor water: Amazonian diversity and foodchain ecology as seen through fish communities. SPB Academic Publishing: The Hague; 1988.
De Graaf G. The flood pulse and growth of floodplain fish in Bangladesh. Fish Manag Ecol. 2003; 10(4):241–47. https://doi.org/10.1046/j.1365-2400.2003.00341.x
Hellawell JM, Abel RA. A rapid volumetric method for the analysis of the food of fishes. J Fish Biol. 1971; 3(1):29–37. https://doi.org/10.1111/j.1095-8649.1971.tb05903.x
Heng K, Chevalier M, Lek S, Laffaille P. Seasonal variations in diet composition, diet breadth and dietary overlap between three commercially important fish species within a flood-pulse system: the Tonle Sap Lake (Cambodia). PLoS ONE. 2018; 13(6):1–16. https://doi.org/10.1371/J.pone.0198848
Hyslop EJ. Stomach contents analysis – a review of methods and their application. J Fish Biol. 1980; 17(4):411–29. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Jin S, Yan X, Zhang H, Fan W. Weight-length relationships and Fulton’s condition factors of skipjack tuna (Katsuwonus pelanis) in the western and central Pacific Ocean. PeerJ. 2015; 3:e758. http://doi.org/10.7717/peerj.758
Júlio-Júnior HF, Tós CD, Agostinho AA, Pavanelli CS. A massive invasion of fish species after eliminating a natural barrier in the upper Rio Paraná basin. Neotrop Ichthyol. 2009; 7(4):709–18. https://doi.org/10.1590/S1679-62252009000400021
Junk WJ, Bayley PB, Sparks RE. The flood pulse in river flood plain systems. Can Spec Publ Fish Aquat Sci. 1989; 106(1):110–27.
Junk WJ. Ecology of swamps in the middle Amazon. In: Gore AJP, editor. Ecosystems of the world. Mires, Swamp, Bog, Fen and Moor. Amsterdam: Elsevier Publishers Company; 1983. p.269–94.
Kovalenko K, Dibble ED, Fugi R. Fish feeding in changing habitats: effects of invasive macrophyte control and habitat complexity. Ecol Freshw Fish. 2009; 18(2):305–13. https://doi.org/10.1111/j.1600-0633.2008.00348.x
López-Rodríguez A, Silva I, Ávila–Simas S, Stebniki S, Bastian R, Massaro VM et al. Diets and trophic structure of fish assemblages in a large and unexplored subtropical river: the Uruguay River. Water. 2019; 11(7):1374. https://doi.org/10.3390/w11071374
Luz-Agostinho KDG, Agostinho AA, Gomes LC, Júlio-Júnior HF. Influence of flood pulse on diet composition and trophic relationships among fish in the upper Parana River floodplain. Hydrobiologia. 2008; 607:187–98. https://doi.org/10.1007/s10750-008-9390-4
McCafferty WP. Aquatic entomology: the fishermen’s and ecologists’ illustrated guide to insects and their relatives. Boston: Jones & Bartlett Learning Publishers; 1983.
McMeans BC, Kadoya T, Pool TK, Holtgrieve GW, Lek S, Kong H et al. Consumer trophic positions respond variably to seasonally fluctuating environments. Ecology. 2019; 100(2):e02570. https://doi.org/10.1002/ecy.2570
Medeiros ESF, Arthington AH. Fish diet composition in floodplain lagoons of an Australian dryland river in relation to an extended dry period following flooding. Environ Biol Fish. 2014; 97(7):797–812. https://doi.org/10.1007/s10641-013-0180-0
Medeiros TN, Rocha AA, Santos NC, Severi W. Influence of the hydrological level on the diet of Leporinus reinhardtii (Characiformes, Anostomidae) in a semi-arid Brazilian reservoir. Iheringia, Sér Zool. 2014; 104:290–98. https://doi.org/10.1590/1678–476620141043290298
Moi DA, Ernandes-Silva J, Baumgartner MT, Mormul RP. The effects of river-level oscillations on the macroinvertebrate community in a river-floodplain system. Limnology. 2020; 21(2):219–32. https://doi.org/10.1007/s10201-019-00605-y
Moyle PB, Light T. Biological invasions of freshwater: empirical rules and assembly theory. Biol Conserv. 1996; 78(1–2):149–61. https://doi.org/10.1016/0006-3207(96)00024-9
Nakagawa S, Schielzeth H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol. 2013; 4(2):133–42. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Nurkse K, Kotta J, Orav-Kotta H, Ojaveer H. A successful non–native predator, round goby, in the Baltic Sea: generalist feeding strategy, diverse diet and high prey consumption. Hydrobiologia. 2016; 777:271–81. https://doi.org/10.1007/s10750-016-2795-6
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, Mcglinn D et al. Vegan: Community Ecol. Package R package version 24-2; 2020. Available from: https://CRANR-projectorg/package=vegan
Ota RR, Deprá GC, da Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: revised, annotated and updated. Neotrop Ichthyol. 2018; 16(2):e170094. https://doi.org/10.1590/1982-0224-20170094
Pelicice FM, Agostinho AA. Feeding ecology of fishes associated with Egeria spp. patches in a tropical reservoir, Brazil. Ecol Freshw Fish. 2006; 15(1):10–19. https://doi.org/10.1111/j.1600-0633.2005.00121.x
Pereira JO, Silva MT, Vieira LJS, Fugi R. Effects of flood regime on the diet of Triportheus curtus (Garman, 1890) in an Amazonian floodplain. Neotrop Ichthyol. 2011; 9(3):623–28. http://doi.org/10.1590/S1679-62252011005000029
Pereira LS, Agostinho AA, Delariva RL. Effects of river damming in Neotropical piscivorous and omnivorous fish: feeding, body condition and abundances. Neotrop Ichthyol. 2016; 14(1):e150044. https://doi.org/10.1590/1982-0224-20150044
Pinheiro J, Bates D, DebRoy S, Sarkar D. R Core Team. nlme: Modelos de efeitos mistos lineares e não lineares. R versão do pacote 3.1-152; 2021. Available from: https://cran.r-project.org/web/packages/nlme/
Prejs A, Prejs K. Feeding of tropical freshwater fishes: seasonality in resource availability and resource use. Oecologia. 1987; 71:397–404.
Pyrzanowski K, Zięba G, Dukowska M, Smith C, Przybylski M. The role of detritivory as a feeding tactic in a harsh environment – a case study of weatherfish (Misgurnus fossilis). Sci Rep. 2019; 9(1):1–09. https://doi.org/10.1038/s41598-019-44911-y
Quirino BA, Carniatto N, Gaiotto JV, Fugi R. Seasonal variation in the use of food resources by small fishes inhabiting the littoral zone in a Neotropical floodplain lake. Aquat Ecol. 2015; 49(4):431–40. https://doi.org/10.1007/s10452-015-9535-2
Quirino BA, Carniatto N, Guglielmetti R, Fugi R. Changes in diet and niche breadth of a small fish species in response to the flood pulse in a Neotropical floodplain lake. Limnologica. 2017; 62:126–31. https://doi.org/10.1016/j.limno.2016.10.005
R Development Core Team. R: A language and environment for statistical computing Vienna, Austria: R Foundation for Statistical Computing; 2020. Available from: http://wwwR-projectorg/
Ribeiro F, Orjuela R, Magalhães M, Collares-Pereira M. Variability in feeding ecology of a South American cichlid: a reason for successful invasion in Mediterraneantype rivers? Ecol Freshw Fish. 2007; 16(4):559–69. https://doi.org/10.1111/j.1600–0633.2007.00252.x
Ricciardi A, Rasmussen JB. Predicting the identity and impact of future biological. invaders: a priority for aquatic resource management. Can J Fish Aquat Sci. 1998; 55(7):1759–65. https://doi.org/10.1139/f98-066
Roberto MC, Santana N, Thomaz SM. Limnology in the Upper Paraná River floodplain: large–scale spatial and temporal patterns, and the influence of reservoirs. Braz J Biol. 2009; 69(2):717–25. https://doi.org/10.1590/S1519-69842009000300025
Santos NCL, Santana HS, Ortega JCG, Dias RM, Stegmann LF, Silva Araújo IM et al. Environmental filters predict the trait composition of fish communities in reservoir cascades. Hydrobiologia. 2017; 802:245–53. https://doi.org/10.1007/s10750-017-3274-4
Souza-Filho EE. Evaluation of the Upper Paraná River discharge controlled by reservoirs. Braz J Biol. 2009; 69(2):707–16. https://doi.org/10.1590/S1519-69842009000300024
Sullivan ML, Zhang Y, Bonner TH. Carbon and nitrogen ratios of aquatic and terrestrial prey for freshwater fishes. J Freshw Ecol. 2014; 29(2):259–66. https://doi.org/10.1080/02705060.2014.888524
Tesch FW. Age and growth. In: Ricker WE, editor. Methods for assessment of fish production in fresh waters. Oxford: Blackwell Scientific Publications; 1968. p.93–123.
Thomaz SM, Agostinho AA, Hahn, NS. The upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers; 2004.
Thomaz SM, Bini LM, Bozelli RL. Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia. 2007; 579:1–13. https://doi.org/10.1007/s10750-006-0285-y
Tonella LH, Fugi R, Vitorino OB, Suzuki HI, Gomes LC, Agostinho AA. Importance of feeding strategies on the long-term success of fish invasions. Hydrobiologia. 2018; 817:239–52. https://doi.org/10.1007/s10750–017–3404–z
Walker RH, Kluender ER, Inebnit TE, Adams SR. Differences in diet and feeding ecology of similar-sized spotted (Lepisosteus oculatus) and shortnose (Lepisosteus platostomus) gars during flooding of a south-eastern US river. Ecol Freshw Fish. 2013; 22(4):617–25. https://doi.org/10.1111/eff.12066
Wantzen KM, Machado AF, Voss M, Boriss H, Junk WJ. Seasonal isotopic shifts in fish of the Pantanal wetland, Brazil. Aquat Sci. 2002; 64(3):239–51. https://doi.org/10.1007/PL00013196
Wickham H. Data Analysis. In: ggplot2. Use R! Springer, Cham. 2016; p.189–201. https://doi.org/10.1007/978-3-319-24277-4_9
Winemiller KO, Kelso-Winemiller LC. Food habits of tilapiine cichlids of the Upper Zambezi River andflood-plains during the descending phase of the hydrological cycle. J Fish Biol. 2003; 63(1):120–28. https://doi.org/10.1046/j.1095-8649.2003.00134.x
Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM. Mixed effects models and extensions in ecology with R, Statistics for Biology and Health. New York: Springer New York; 2009.
Authors
![]() Isadora Cristina Bianchi-Costa1
Isadora Cristina Bianchi-Costa1 ![]() ,
, ![]() Bárbara Angélio Quirino1,
Bárbara Angélio Quirino1, ![]() Ana Lúcia Paz Cardozo1,
Ana Lúcia Paz Cardozo1, ![]() Kátia Yasuko Yofukuji1,
Kátia Yasuko Yofukuji1, ![]() Matheus Henrique Ferreira1 Aleixo and
Matheus Henrique Ferreira1 Aleixo and ![]() Rosemara Fugi1,2
Rosemara Fugi1,2
[1] Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais, Universidade Estadual de Maringá, Av.Colombo, 5790, 87020-900 Maringá, PR, Brazil. (ICBC) isadorabianchi10@gmail.com (corresponding author), (BAQ) barbara_aq@hotmail.com, (ALPC) analuciapazcardozo@gmail.com, (KYY) kayofukuji96@gmail.com, (MHFA) matheusferreiraaleixo@gmail.com, (RF) rosemarafugi@gmail.com.
[2] Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil.
Authors’ Contribution 

Isadora Cristina Bianchi-Costa: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft.
Bárbara Angélio Quirino: Investigation, Methodology, Writing-review and editing.
Ana Lúcia Paz Cardozo: Investigation, Methodology, Writing-review and editing.
Kátia Yasuko Yofukuji: Investigation, Methodology, Writing-review and editing.
Matheus Henrique Ferreira Aleixo: Investigation, Methodology, Writing-review and editing.
Rosemara Fugi: Conceptualization, Investigation, Methodology, Supervision, Writing-review and editing.
Ethical Statement
Fish were anesthetized with benzocaine and euthanized according to the protocol approved by the Ethics Committee on the Use of Animals at the Universidade Estadual de Maringá (CEUA/UEM nº 1420221018 – ID 001974).
Competing Interests
The author declares no competing interests.
How to cite this article
Bianchi-Costa IC, Quirino BA, Cardozo ALP, Yofukuji KY, Aleixo MHF, Fugi R. Water-level fluctuations lead to changes in the diet of an omnivorous fish in a floodplain. Neotrop Ichthyol. 2023; 21(1):e220064. https://doi.org/10.1590/1982-0224-2022-0064
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted January 23, 2023 by Gerson Araújo
Accepted January 23, 2023 by Gerson Araújo
![]() Submitted July 5, 2022
Submitted July 5, 2022
![]() Epub March 13, 2023
Epub March 13, 2023