![]() Angela Maria Zanata1
Angela Maria Zanata1 ![]() ,
, ![]() Leonardo Oliveira-Silva1 and
Leonardo Oliveira-Silva1 and ![]() Willian M. Ohara2,3
Willian M. Ohara2,3
PDF: EN XML: EN | Cite this article
Abstract
A new miniature species of Characidium is described from the midde rio Guaporé, rio Madeira basin, Rondônia, Brazil. The new species can be readily distinguished from all congeners by the sexual dichromatism, with females having narrow dark bars on body absent in males, and by the presence, in both sexes, of a black midventral dashed line extending from area between contralateral pectoral fins to at least the anal-fin origin. It can also be diagnosed from congeners by having strongly tricuspid teeth, with well developed and similar sized cusps in the premaxilla and outer series of dentary, and short lateral line with 6–8 perforated scales. Morphological novelties to Characidium observed in the new species include a rounded process on the first pleural rib near the vertebra, three or four haemal spines of the first caudal vertebrae distinctly elongate, neural and haemal spines of the antepenultimate vertebra not reaching origins of dorsal and ventral procurrent rays nor the origin of the caudal-fin rays.
Keywords: Amazon fishes, First rib, Haemal spine, Sexual dimorphism, Tricuspid teeth
Uma nova espécie miniatura de Characidium é descrita do médio rio Guaporé, bacia do rio Madeira, Rondônia, Brasil. A nova espécie pode ser facilmente distinguida de todas as congêneres pelo dicromatismo sexual, com barras escuras estreitas no corpo das fêmeas ausentes nos machos, e por ter uma linha preta mediana ventral tracejada em ambos os sexos, que se estende da área entre as bases das nadadeiras peitorais até pelo menos a origem da nadadeira anal. A espécie também pode ser diagnosticada das congêneres por ter dentes fortemente tricúspides, com cúspides bem desenvolvidas e de tamanho semelhante no pré-maxilar e série externa do dentário, e linha lateral curta com 6–8 escamas perfuradas. Novidades morfológicas para Characidium observadas na nova espécie incluem presença de um processo arredondado na primeira costela pleural próximo à vértebra, três ou quatro espinhos hemais das primeiras vértebras caudais distintamente alongados, espinhos neurais e hemais da antepenúltima vértebra não atingindo as origens dos raios procorrentes dorsal e ventral, nem a origem dos raios da nadadeira caudal.
Palavras-chave: Dentes tricuspidados, Dimorfismo sexual, Espinho hemal, Peixes amazônicos, Primeira costela.
Introduction
Characidium Reinhardt, 1867 includes 84 species of South American darters, distributed from eastern Panama to Argentina (Fricke et al., 2022; Oliveira-Silva et al., 2022). Along with continuous rising of the taxonomic knowledge of the genus, the last decades witnessed novelties such as new dimorphic features(e.g., Melo, Oyakawa, 2015; Teixeira, Melo, 2021), morphological specializations, or new structures (e.g., Zanata et al., 2020; Melo et al., 2021), events of miniaturization and discussions of reductive characters (e.g., Netto-Ferreira et al., 2013; Zanata et al., 2015; Graça et al., 2019), cytogenetic studies and genetic divergences used to highlight the occurrence of hidden biodiversity (Pansonato-Alves et al., 2014; Sales et al., 2018; Serrano et al., 2019), and new behavior (e.g., Flausino Junior et al., 2020). However, studies dealing with interspecific phylogenetic relationships within Characidium or testing the monophyly of the genus as it is composed nowadays are not yet available.
A new species of Characidium from upper rio Guaporé sub basin, rio Madeira basin, with a series of putative apomorphic morphological features not reported previously to the genus, including sexually dimorphic structures, is described herein. The new species is the sixth of the genus described from the Mamoré-Guaporé rivers, in addition to C. barbosai Flausino Junior, Lima, Machado & Melo, 2020, C. heinianum Zarske & Géry, 2001, C. nambiquara Zanata & Ohara, 2020, C. schindleri Zarske & Géry, 2001, and C. summum Zanata & Ohara, 2015 (Fricke et al., 2022). Characidium bolivianum Pearson, 1924 also was described from rio Madeira basin (Pearson, 1924), but from the rio Beni sub basin. The new species is morphologically very distinctive from all congeners and apparently not closely related to those from the rio Madeira basin.
Material and methods
After capture, specimens were prior anesthetized with eugenol solution (1 ml/L), then fixed in 10% formalin, and finally preserved in 70% ethanol. The field studies did not involve endangered species. Counts and measurements were taken according to Buckup (1993a), Melo, Oyakawa (2015), and Zanata et al. (2020). New measurements include pectoral-fin length measured from origin of first unbranched pectoral-fin ray to the distal tip of longest fin ray, and pelvic-fin length, measured from the origin of the first unbranched fin ray to the distal tip of the longest fin ray. Measurements were taken with a digital caliper to the nearest 0.1 mm and expressed as percents of standard length (SL), except subunits of head, which are given as percentages of head length (HL). Measurements of the swimbladder were taken in dissected specimens; total lengh of the structure is expressed as percent of standard length (SL), and length of each chamber is expressed as percent of swimbladder length. In the list of paratypes, an asterisk indicates lots which measurements were included in Tab. 1. Meristic data are given in the description, an asterisk indicates counts of the holotype, and the frequency of each count is given in parentheses. Counts on vertebrae, ectopterygoid teeth, branchiostegal rays, procurrent caudal-fin rays, caudal-fin rays, epurals, and other osteological observations were made only in cleared and stained (c&s) paratypes, prepared according to the method of Taylor, Van Dyke (1985). Dentary teeth were counted in c&s specimens and in the largest alcohol preserved specimens. The pattern of circuli and radii were observed on scales situated between dorsal-fin base and lateral line, after being stained in alizarin. The pseudotympanum morphology was examined after the removal of the overlying skin, adipose tissue, and lateral-line nerve of alcohol-preserved specimens. The sex of some specimens was confirmed by dissection, following Vazzoler (1996). Institutional abbreviations follow Fricke, Eschmeyer (2021). Comparisons with C. longum Taphorn, Montaña & Buckup,2006, C. pellucidum Eigenmann, 1909 and C. pteroides Eigenmann, 1909 where based mainly on the information available in their original descriptions and photos of the holotypes (Eigenmann, 1909; Taphorn et al., 2006), further complemented by examination of specimens listed in the ‘Comparative material examined’. Other comparisons with species not cited in the ‘Comparative material examined’ were also based on the literature (e.g., Zarske, 1997; Zarske, Géry, 2001; Peixoto, Wosiacki, 2013; Melo et al., 2016).
Results
Characidium fleurdelis, new species
urn:lsid:zoobank.org:act:8D2995F6-A0DD-4FFA-8977-46327C995DA4
(Figs. 1–5; Tab. 1)
Holotype. MZUSP 126956, 21.6 mm SL, Brazil, Rondônia, Corumbiara town, Vitória da União district, stream tributary of rio Oimerê, rio Corumbiara, rio Guaporé sub basin, rio Madeira basin, 12°59’56.32”S 61°08’51.09”W, 197 m above sea level, 23 Jan 2022, W. Ohara.
FIGURE 1| Characidium fleurdelis: A. Holotype, MZUSP126956, female, 21.6 mm standard length, lateral view; B. Paratype, UFBA 9234, female, 22.9 mm SL, lateral, dorsal and ventral views; C. Paratype, UFBA 9234, female, 23.8 mm SL, lateral view; D. Paratype, UFBA 9234, male, 20.6 mm SL, lateral view; E. Paratype, UFBA 9233, juvenile, 17.4 mm SL, lateral view. All from rio Guaporé sub basin, rio Madeira basin, Rondônia, Brazil.
TABLE 1 | Morphometric data of holotype and paratypes of Characidium fleurdelis (n = 30), range includes the holotype. SD = standard deviation.
Holotype | Range | Mean | SD | |
Total length
(mm) | 28.2 | 25.7–30.3 | – | – |
Standard length
(mm) | 21.6 | 19.8–24.1 | – | – |
Percents of standard length | ||||
Depth at dorsal-fin origin | 23.6 | 21.1–26.9 | 23.4 | 1.6 |
Depth at anal-fin origin | 13.4 | 13.3–16.9 | 15.1 | 1.1 |
Caudal-peduncle
depth | 9.3 | 9.0–12.6 | 10.4 | 1.1 |
Caudal-peduncle
length | 17.6 | 16.4–19.5 | 18.0 | 0.8 |
Pectoral-fin length | 25.5 | 23.1–27.9 | 24.9 | 1.1 |
Pelvic-fin length | 26.4 | 23.2–28.8 | 26.1 | 1.3 |
Dorsal-fin adpressed | 25.9 | 24.5–30.9 | 27.4 | 2.0 |
Dorsal to adipose fin | 10.2 | 7.7–12.0 | 9.8 | 1.5 |
Snout to dorsal-fin origin | 44.4 | 41.1-45.9 | 44.5 | 1.1 |
Snout to pectoral-fin origin | 27.8 | 25.3–29.8 | 27.9 | 1.2 |
Snout to pelvic-fin origin | 51.9 | 49.5–54.4 | 51.7 | 1.1 |
Snout to anal-fin origin | 77.3 | 73.7–77.6 | 75.5 | 1.3 |
Anal-apex distance | 95.8 | 89.4–95.9 | 93.7 | 1.9 |
Anus to anal-fin origin | 11.1 | 9.6–13.5 | 11.5 | 1.1 |
Body width | 10.6 | 10.3–15.4 | 12.4 | 1.3 |
Head length | 27.8 | 25.1–29.9 | 27.5 | 1.1 |
Percents of head length | ||||
Horizontal eye
diameter | 28.3 | 27.6–31.1 | 29.4 | 1.1 |
Snout length | 18.3 | 18.2–21.8 | 19.8 | 1.0 |
Snout to maxillary tip | 21.7 | 20.0–24.1 | 21.8 | 1.2 |
Anterior naris
to orbit | 8.3 | 6.9–10.3 | 9.1 | 0.9 |
Posterior naris
to orbit | 3.3 | 3.2–5.5 | 3.5 | 0.4 |
Cheek depth | 5.0 | 4.8–7.3 | 5.7 | 0.9 |
Least interorbital width | 23.3 | 22.2–25.5 | 24.5 | 1.0 |
Paratypes. All from Brazil, Rondônia, Corumbiara town, Vitória da União district, stream tributary of rio Oimerê, tributary of rio Corumbiara, rio Guaporé sub basin, rio Madeira basin, 12°59’56.32”S 61°08’51.09”W. UFBA 9231*, 3, 20.3–21.7 mmSL, 1 c&s, 20.3 mm SL, 6 May 2021, W. M. Ohara & W. C. S. Tomicha. INPA 59849, 5, 16.3–24.4 mm SL; UFBA 9233, 21, 14.5–22.2 mmSL; MZUSP 126957, 12,15.1–21.3 mm SL, 19 Aug 2021, W. Ohara & V. O. Alves.UFBA 9234*, 22, 20.2–24.1 mmSL, 4 c&s, 20.2–23.5 mm SL; MZUSP 126958,12, 18.4–23.3 mm SL; INPA 59848, 24, 19.3–24.1 mm SL; collected with holotype.
Non-types. All from Brazil, Rondônia, Corumbiara town, Vitória da União district, stream tributaries of rio Corumbiara, rio Guaporé sub basin, rio Madeira basin. INPA 59850, 1, 21.5 mm SL, stream tributary of rio Oimerê, 12°59’56.32”S 61°08’51.09”W, 5 May 2021, W. M. Ohara & W. C. S. Tomicha.INPA 59851, 2, 15.9–19.6 mm SL, stream tributary of rio Oimerê, 13°00’47.92”S 61°08’28.04”W, 6 May 2021, W. M. Ohara & W. C. S. Tomicha. INPA 59852, 10, 13.7–17.0 mm SL, rio Bocão, 13° 5’14.16”S 61°10’7.50”W, 6 May 2021, W. M. Ohara & W. C. S. Tomicha. INPA 59853, 1, 15.5 mm SL, stream tributary of rio Bocão, 13°03’43.00”S 61°03’47.00”W, 6 May 2021, W. M. Ohara & W. C. S. Tomicha.
Diagnosis. Characidium fleurdelis differs from congeners, except C. longum, C. pellucidum, and C. pteroides by having teeth in the premaxilla and outer series of dentary strongly tricuspid, with three well developed and similar sized cusps (Fig. 2) (vs. teeth uni- or if tricuspid with median cusp distinctly larger; e.g., Agudelo-Zamora et al., 2020, Fig. 4; Melo et al., 2021; Figs. 3A,B). The new species differs from the aforementioned congeners by the presence of a dark (occasionally interrupted) narrow midlateral stripe on the body (vs. absence of dark midlateral stripe), the presence of a black midventral dashed line extending from area between contralateral pectoral fins to at least the anal-fin origin (Fig. 1B) (vs. absence of interrupted black line throughout ventral midline of body in C. longum and C. pteroides, and midventral black line restricted to the area posterior to pelvic fins in C. pellucidum), and the presence of sexual dichromatism, with females having narrow dark bars on laterals of body and males without it (Figs. 1, 5) (vs. sexual dichromatism absent). Characidium fleurdelis also differs from the three aforementioned species and remaining congeners, except C. bahiense Almeida, 1971, C. cacah Zanata, Ribeiro, Araújo-Porto, Pessali & Oliveira-Silva, 2020, C. chicoi Graça, Ota & Domingues, 2019, C. interruptum Pellegrin,1909, C. laterale (Boulenger, 1895), C. mirim Netto-Ferreira, Birindelli Buckup,2013, C. nana Mendonça & Netto-Ferreira, 2015, C. nupelia Graça, Pavanelli & Buckup, 2008, C. rachovii Regan, 1913, C. stigmosum Melo & Buckup, 2002, and C. xavante Graça, Pavanelli Buckup, 2008, by having a short lateral line with 6–8 perforated scales (vs. lateral line with more than 14 perforated scales). The new species can be distinguished from C. cacah, C. chicoi, C. mirim, C. nana, C. nupelia, C. stigmosum, and C. xavante by the presence of adipose fin (vs. absence), from C. bahiense and C. laterale by having two dentary teeth rows (vs. one), from C. rachovii by the absence of roundish black marks on the pelvic, dorsal, and anal fins (vs. presence)and from C. interruptum by having 12 circumpeduncular scales (vs. 14). Other features useful to diagnose the new species from most congeners includes: first pleural rib with a rounded process posteriorly directed near the vertebra (Fig. 3A) (vs. absence of such a process or, if present, triangular or L shaped); neural and haemal spines of the antepenultimate vertebra not reaching origins of dorsal and ventral procurrent rays, respectively, nor the origin of the caudal-fin rays (Fig. 3B) (vs. neural and haemal spines of the antepenultimate vertebra reaching origins of procurrent rays or the origin of the caudal-fin rays); and three to four haemal spines of caudal vertebrae anterior to the anal fin flattened and distinctly elongate (Figs. 3C, D) (vs. haemal spines anterior to the first anal-fin proximal radial with tapered tip, comparatively shorter than the neural spine of the same vertebra and similar in length or rarely longer than the haemal spines situated above the anal fin). See “Discussion” for additional comments.
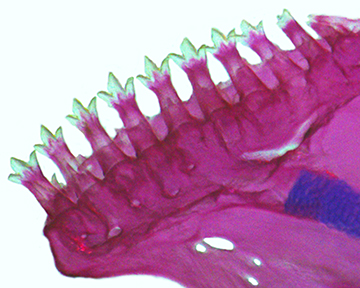
FIGURE 2| Dentary of Characidium fleurdelis, UFBA 9234, paratype, male, 21.0 mm SL, outer view.
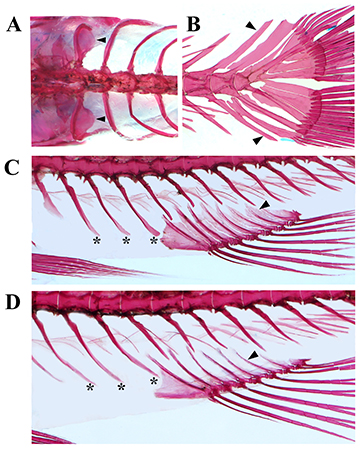
FIGURE 3| Osteological characteristics in cleared and stained specimens of Characidium fleurdelis, UFBA 9234, paratypes: A. Weberian apparatus and first ribs, black arrows indicate dorsal rounded processes on first ribs, female, 23.5 mm SL, dorsal view; B. Caudal-fin bony elements, black arrows indicate neural and haemal spines of the antepenultimate vertebra, posteriormost intermuscular bones removed, female, 23.5 mm SL, lateral view; C. Anal-fin radials and first haemal spines in male, 21.0 mm SL, asterisks indicate elongate and flattened haemal spines, black arrow indicates one radial with plate-like bony expansions, lateral view; D. Anal-fin radials and first haemal spines in female, asterisks indicate elongate haemal spines, black arrow indicates one radial without plate-like bony expansion, 23.5 mm SL, lateral view.
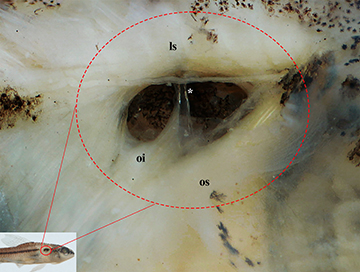
FIGURE 4| Pseudotympanum of Characidium fleurdelis UFBA 9234, paratype, 20.6 mm SL, right side in lateral view. Overlying skin and adipose tissue removed. ls, lateralis superficialis; oi, obliquus inferioris; os, obliquus superioris. Asterisk indicates the rib of fifth vertebrae.
Description. Morphometric data of holotype and paratypes in Tab. 1. Largest specimen reaching 24.4 mm SL. Body elongate. Highest body depth at vertical through dorsal-fin origin. Anterior portion of head convex in lateral view; dorsal profile convex from snout to posterior tip of supraoccipital, straight or slightly convex from supraoccipital to origin of dorsal-fin base, convex along dorsal-fin base, slightly concave or straight from that point to origin of anteriormost dorsal procurrent caudal-fin rays. Ventral profile of body nearly straight or slightly convex from dentary symphysis to isthmus, straight or slightly concave from that point to pelvic-fin origin in males, non-reproductive females, and immature specimens (Fig. 1), convex in mature females (Fig. 5). Profile straight or concave from pelvic-fin insertion to anal-fin origin, straight or slightly concave along anal-fin base, and straight or slightly concave from end of anal-fin base to anterior most ventral procurrent caudal-fin ray.
FIGURE 5| Characidium fleurdelis, paratypes, INPA 59848, lateral view: A. Freshly preserved specimens, top male, 22.2 mm SL; bottom mature female with ovocytes seen by transparency on belly as a yellowish area, 24.1 mm SL; B. Live specimens, top male, 20.6 mm SL; bottom mature female, 24.0 mm SL. All from rio Guaporé sub basin, rio Madeira basin, Rondônia, Brazil.
Snout triangular-shaped in lateral view. Mouth subterminal, aligned or slightly superior than ventral margin of orbit. Distal tip of maxilla barely reaching anterior margin of orbit. Orbit approximately round, distinctly larger than snout length. Cheek narrow, its depth approximately one fifth of orbit diameter. Nares separated, without distinctly raised margins; posterior naris distinctly closer to orbit than anterior naris. Supraorbital broad, roughly triangular-shaped, outer border somewhat concave and inner border with angle. Nasal bones restricted to ossified canal. Parietal fontanel limited anteriorly by frontals and laterally by parietals. Parietal branch of supraorbital canal absent.
Dentary teeth in two rows; outer row with 7(1), 8(1), 9(1), or 10(2) teeth; majority of teeth tricuspid, pointed cusps well developed and similar in size (Fig. 2), and posteriormost teeth smallest and conical; inner row with several small conical teeth inserted on edge of replacement tooth trench. Premaxilla with single row of 8(3) or 9(2) teeth tricuspid with cusps well developed and similar in size; teeth decreasing in size from symphysis, posteriormost tooth unicuspid in one c&s specimen. Maxillary edentulous. Ectopterygoid with one row of 7(2) or 8(3) teeth, minute and conical. Endopterygoid teeth absent. Branchiostegal rays 4(5), 3 connected to anterior ceratohyal, 1 connected to posterior ceratohyal.
Scales cycloid; circuli on exposed portion of scales absent; up to seven divergent radii present on exposed portion of scales. Lateral line reduced, with 6(17), 7*(12), or 8(1) perforated scales; total scales along lateral line series 32(1), 33*(12), 34(12), or 35(2); horizontal scale rows 8*(11) or 9(19). Scales along middorsal line between supraoccipital and origin of dorsal fin 8(16) or 9*(14). Scale rows around caudal peduncle 12*(30). Three (4), 4*(22), or 5(4) scales between anus aperture and anal-fin insertion. Isthmus and belly completely covered with scales. Pseudotympanum present, limited dorsally by lateralis superficialis, anteriorly and posteriorly by obliquus inferioris, and ventrally by obliquus superioris. Humeral hiatus divided into anterior and posterior chambers by pleural rib of fifth vertebra (Fig. 4). Swimbladder with two well-developed chambers, both with anterior and posterior extremities rounded, without pointed tip in the second chamber. Total length of swimbladder 20.1–24.8% of SL in four specimens of 20.5–22.9 mm SL; anterior chamber of females 41.7–50.0% and posterior chamber 50.0%–58.3% of swimbladder length; anterior chamber of males 46.1–46.6% and posterior chamber 53.8%–57.1% of swimbladder length.
Fins broad with fragile rays. Dorsal-fin rays ii,9*(30); distal margin of fin nearly straight or somewhat concave. Adipose fin well-developed, somewhat aligned vertically with end of anal-fin base. Pectoral fin with 7–10 total rays; iii,3,i(1), iii,4,i(1), iii,5,i*(17), iii,6(5), or iii,6,i(6); last unbranched and first branched fin rays longest, first branched ray distinctly elongate, posterior tip of fin usually reaching beyond pelvic-fin origin. Postcleithrum 1 absent. Pelvic-fin rays i,6,ii(4) or i,7,i*(26); second branched fin ray elongated, posterior tip of fin reaching anal-fin ray origin (Figs. 1, 5). Anal-fin rays ii,6,i*(18) or ii,7(12) visible in alcohol (2 of the c&s specimens with com iii rays); posterior margin of fin straight or slightly concave, without distinctly elongate rays. First anal-fin radial inserts behind 21st(2) or 22th(3) vertebra, behind the fourth (1) or fifth (4) caudal vertebra; fin elements on last pterygiophore 1(5). Anal-fin proximal radials of males with distinctly developed plate-like bony expansions anteriorly and posteriorly directed (Fig. 3 C), meeting each other at midline between adjacent radials; radials of females without distinctly developed bony expansions (Fig. 3 D). Caudal-fin rays i,9,8,i*(30). Dorsal procurrent caudal-fin rays 6(5); ventral procurrent caudal-fin rays 5(4) or 6(1). Total vertebrae 33(5); precaudal vertebrae 16(3) or 17(2); caudal vertebrae 16(1) or 17(4). First pleural rib with two distinctly developed processes (5), one dorsally positioned, close to vertebra, posteriorly directed and with rounded posterior margin (Fig. 3A), and a second process more ventrally positioned, approximately at midlenght of rib, thumb-like or somewhat triangular-shaped, directed towards swimbladder (see Buckup 1993b, fig. 11). Haemal spines of first three or four caudal vertebrae distinctly elongate almost reaching ventralmost portion of body (Fig. 3 C, D; distal portion of haemal spines flattened laterally with rounded tip in males (Fig. 3 C) and pointed in females (Fig. 3D). Supraneural bones 2(1), 3(1), or 4(3). Epural bones 2(4) or 3(1). Uroneural bone 1(5).
Color in alcohol. Female(Figs. 1, 5A). Ground color of head and body pale yellow. Dorsal surface of head darker than lateral, with a distinct dark patch of pigmentation, usually forming a heart-shaped large blotch behind eyes; melanophores distributed in smaller inconspicuous blotches between eyes or anterior to it. A dark stripe from anterior margin of snout to anterior margin of eye and a more diffuse posterior to eye, with few and large scattered melanophores, somewhat aligned with stripe of snout and continuous with midlateral stripe of body. Ventral half of head without melanophores, except by a few sparsely distributed in parts of opercle. Ventral surface of head pale, without melanophores. Humeral region without humeral blotch apart from longitudinal stripe. A dark line or narrow stripe extending from rear of opercle to end of caudal peduncle; stripe somewhat homogeneous in form and continuous along its extension or with irregular borders; stripe slightly enlarged at end of caudal peduncle; specimens up to 18.0 mm SL with stripe usually interrupted or dashed, separated by less pigmented or unpigmented short stretches. Laterals of body with 7(1), 8(1), 10 (5), 11(5), 12(2), 13(3), 14(3), 15*(3), 16(1), 19(1) narrow and vertically elongated dark bars (Figs. 1, 5); some main bars with shorter bars between them (Fig. 1E, 5); position and form of bars also variable, reaching ventral half of body in some specimens (Figs. 1A, E, 5B) or centered along midlateral line in others (Fig. 1B); some specimens with area of bars close to dorsum roughly connected with each other resulting in a somewhat continuous dark area (Fig. 1B). Dorsum with up to 11 blotches, continuous or not with lateral bars; Basicaudal spot well marked. Ventral surface of body pale yellow with few, small and scattered chromatophores, except by a black dashed midventral line extending from area between pectoral fins to caudal peduncle (Fig. 1B). All fin-rays with tinny melanophores along its borders, forming or not inconspicuous dark lines bordering rays. Proximal portion of caudal fin usually with inconspicuous narrow black blotch on its ventral half, resembling a less elongation of the basicaudal spot. Adipose fin hyaline or with tinny sparse melanophores.
Male (Figs. 1D, 5A). Overall head, background and fins colorations similar to described above. A very conspicuous dark narrow stripe extending from rear of opercle to end of caudal peduncle; specimens up to 18.0 mm SL with stripe usually interrupted or dashed, separated by less pigmented or unpigmented short stretches. Body conspicuously pigmented with melanophores equally distributed, including on ventral half of body, mainly on area posterior do pelvic fins. Flanks without dark bars, with melanophores concentrated on border of one or two dorsalmost longitudinal series of scales, resulting in an inconspicuous reticulate pattern. Black dashed midventral line conspicuous from area between pectoral fins to anal-fin origin, but absent or less evident on caudal peduncle.
Coloration in life. Ground color pale brown in males and pale yellow in females, with silvery belly (Fig. 5B). Dark pattern of stripe and bars similar to the condition observed in preserved specimens. Males usually darker than females. Ventral half of head pale and opercle silvery. Pigmentation on fins similar to preserved specimens but with pale brown dorsal-, anal-, and caudal-fin rays; pectoral and pelvic fins pale.
Sexual dimorphism. Males can be readily distinguished from females by the absence of dark bars or blotches on laterals and dorsum of body, possessing instead a more homogeneously dark coloration on body (Figs. 1, 5). Males and females of C. fleurdelis possess elongated haemal spines of first caudal vertebrae situated anterior to the anal fin, with the distal portion of the elongated spines distinctly flattened in males (3) and pointed in females (2) (Figs. 3C, D; see Discussion). Males of C. fleurdelis also have proximal radials of the anal fin with distinctly developed plate-like bony expansions (3), anteriorly and posteriorly directed, meeting each other at midline between adjacent radials (Fig. 3C; see Discussion). No similar expansion was observed in females (2; Fig. 3D).
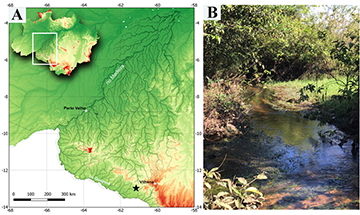
Geographical distribution. Characidium fleurdelis is known to occur in tributaries of the rio Corumbiara, rio Guaporé sub basin, rio Madeira basin, Corumbiara, Rondônia State, Brazil (Fig. 6A).
FIGURE 6| A. Map with part of the rio Madeira basin, Brazil, showing the type locality (black star) of Characidium fleurdelis;symbol may represent more than one locality or lot. B. Sampling locality of Characidium fleurdelis, Rondônia, Corumbiara town, Vitória da União district, stream tributary of rio Oimerê, tributary of rio Corumbiara, rio Guaporé sub basin, rio Madeira basin.
Ecological notes. Characidium fleurdelis was observed and captured only in backwater areas, near the substrate, predominantly composed by sand and mud and covered by leaves, in small clear water and slow current streams, approximately 1–2 m wide, 0.4–1.3 m deep, and bordered by aquatic vegetation and grass (Fig. 6B). The type locality is surrounded by large cultivated farmlands (e.g.,soy, cotton and corn). Other species sampled syntopically near the bottom includes Apistogramma linkei Koslowski, 1985, A. staecki Koslowski, 1985, Brachyrhamdia marthae Sands & Black, 1985, Corydoras caudimaculatus Rössel, 1961, Characidium aff. zebra Eigenmann, 1909 and Mikrogeophagus altispinosus (Haseman, 1911). A dissected mature female of 24.1 mm SL had a total of 500 mature ovocytes.
Etymology. Named in allusion to the similarity of the three pointed flower-like teeth of the species with a stylized flower called fleur-de-lis. Géry (1960) was the first author, apparently, to point out the resemblance of such form of teeth with a fleur-de-lis in his description of Microcharacidium eleotrioides (Géry, 1960). Lily or iris, the flower upon which the emblem fleur-de-lis is historically related, was the flower of Hera, the Greek moon goddess, being the symbol of purity. Also,the fleur-de-lis is often related with the sacred lotus symbol for “plant” meaning “Tree of Life”. A noun in apposition.
Conservation status. The species is known from four localities, two in streams of the rio Oimerê, and two in the rio Bocão, both from the middle rio Corumbiara, tributary of rio Guaporé, rio Madeira basin. The area is strongly impacted by deforestation and severely converted in large cultivated farmlands of soybean, cotton, and corn, suffering effects of erosion, herbicides, pesticides, fertilizers, and silting. The Extension of Occurrence (EOO) is 6.300 km2. Therefore, we recommend that C. fleurdelis be classified as Near Threatened (NT) approaching of Vulnerable (VU) by criterion B1(biii), according to the guidelines published by the International Union for Conservation of Nature Standards and Petitions Committee (IUCN, 2022).
Discussion
Morphological novelties present in Characidium fleurdelis. 1. Posteriorly directed rounded process near the base of the first rib (Fig. 3A). According to Buckup (1993b: 239) in Characidiinae the ribs associated with the fifth vertebral centrum have a conspicuous medial process located below the anterior chamber of the swimbladder, that serves as a point of attachment for the fibrous sheath of thick connective tissue that encapsulates the ventral surface of the swim bladder. Characidium fleurdelis do have the process reported by Buckup (1993b:240, fig. 11), but also possesses a posteriorly directed rounded process, more dorsally positioned than the other process and located near the base of the first rib. Such process is apparently described for the first time to a member of Crenuchidae. Within examined Characidium, similar condition, with two processes as described in C. fleurdelis, was observed in C. cf. pteroides and C. cf. longum. In fact, the c&s specimen of C. cf. longum examined possesses a dorsalmost process also in the second rib. Agudelo-Zamora et al. (2020: figs. 8a, c) described a dorsal upper process triangular or L shaped in C. tatama Agudelo-Zamora, Tavera, Murillo & Ortega-Lara, 2020and C. dule Agudelo-Zamora, Tavera, Murillo & Ortega-Lara, 2020, respectively, but with distinct format and medially directed instead of posteriorly directed as present in C. fleurdelis (Fig. 3A). A posteriorly directed triangular projection near base of first rib was recorded by Zanata, Vari (2005) in the members of the family Alestidae (with the exception of Arnoldichthys Myers, 1926, Chalceus Cuvier, 1818and Lepidarchus Roberts, 1966) and in Astyanax Baird & Girard, 1854, Cheirodon Girard, 1855, Serrasalmus Lacepède, 1803, Piaractus Eigenmann, 1903, Tetragonopterus Cuvier, 1916,and Xenocharax Günther, 1867. The process, according to the authors, serves as the area of attachment on the rib for ligaments extending to the base of the immediately posterior rib and its parapophysis. The rounded process described herein in C. fleurdelis is apparently different from that described by Zanata, Vari (2005) and its function and occurence among members of Crenuchidae needs to be better evaluated in the future.
2. Neural and haemal spines of the antepenultimate vertebra far from the procurrent and caudal-fin rays (Fig. 3B). The common condition observed among species of Characidium is to have the haemal spine of pleural centrum of the antepenultimate vertebra (HPU 3) reaching the origins of the ventral procurrent rays or the origin of the ventralmost caudal-fin rays (Melo, Oyakawa, 2015: fig. 4; Melo et al., 2016: figs. 3b-d; Melo et al., 2021: fig. 4; see also Comparative material examined) and usually the neural spine of ural centrum of the same vertebra (NPU 3) reaching the origins of dorsal and procurrent rays and the origin of the dorsalmost caudal-fin rays. Conversely, the neural and haemal spines of the antepenultimate vertebra of C. fleurdelis do not reach the bases of dorsal and ventral procurrent rays, respectively, nor the origin of the caudal-fin rays. Similar condition to that of C. fleurdelis was observed solely in a few specimens of C. travassosi Melo, Buckup & Oyakawa, 2016 (see Melo et al., 2016: fig 3a).
3. Elongation and flattening of the haemal spines of three or four first caudal vertebrae (Figs. 3C, D). The common condition observed in species of Characidium is to have haemal spines anterior to the first anal-fin proximal radial with tapered tip, comparatively shorter than the neural spine of the same vertebra and similar in length to the haemal spines situated above the anal fin (e.g., Melo et al., 2021, fig. 4b), or rarely longer (e.g., Melo, Oyakawa, 2015, fig. 4), but not reaching close to the ventral border of the body. Conversely, in C. fleurdelis three or four haemal spines of the initial caudal vertebrae situated anterior to the anal-fin origin are distinctly longer than the haemal spines situated above the anal fin and reaching close to the ventral margin of body (Figs. 3C, D), and longer than the neural spines of the same vertebrae. Such elongation of haemal spines is similar in males and females of the species, but in males the distal tips are rounded instead of pointed as observed in females. Illustrations of Characidium provided in the literature to these bones and the c&s congeners examined herein to do not have the condition observed in C. fleurdelis, with the exception of C. cf. pteroides (see Comparative material examined). Male and female of C. cf. pteroides possess haemal spines anterior to the anal fin longer than the neural spines of the same vertebra, and only the male possesses its distal portion flattened. However, in both specimens of C. cf. pteroides the haemal spines are not as elongated as in C. fleurdelis. Furthermore, the common condition observed in Characidium is to have only one or two haemal spines positioned anterior to the anal fin radials (e.g., Melo et al., 2021, fig. 4b), instead of three or four as observe in C. fleurdelis.
4. Proximal radials of the anal fin with plate-like bony expansions (Fig. 3C). The proximal radials of the anal fin are usually rod-like in the species of Characidium, with poorly developed flattened plate-like anterior and/or posterior expansions, being almost absent or restricted to proximal half of the bone element (e.g., Melo et al., 2021: fig. 4b). Females of C. fleurdelis have such condition (Fig. 3D). Conversely, in males of the species the proximal radials bear distinctly developed plate-like bony expansions, anteriorly and posteriorly directed. The expansions of adjacent rays approximate each other at midline between them. The dimorphic condition described above was observed also in C. cf. pteroides. A c&s immature male of C. cf. longum examined to the feature possesses laterally expanded proximal radials, but not as developed as in the two species cited.
5. Pelvic-fin elongated (Fig. 1). Having the pectoral fin longer than the pelvic fin apparently is the common condition in species of Characidium, a feature observable either in illustrations (e.g., Lujan et al., 2013; Leitão, Buckup, 2014; Malanski et al., 2019; Melo et al., 2021) or data of the longest axis of the fins length (e.g., Zanata et al., 2020; Oliveira-Silva et al., 2022). In contrast, the pelvic fin in C. fleurdelis possesses similar length or is slightly longer than the pectoral fin (pectoral length = 23.1–27.9% of SL, mean = 24.9 vs. pelvic = 23.2–28.8% of SL, mean = 26.1). Such elongation of the pelvic fin was not observed in other congeners examined to the feature.
Miniaturization and shared features. Characidium fleurdelis can be considered a miniature South American fish once it fits in the criteria suggested by Weitzman, Vari (1988): a species that becomes sexually mature under 20.0 mm SL and may sometimes reach larger sizes; or a species for which maturity data are unknown but that are not reported to exceed 25 to 26 mm SL in the wild. Examined specimens of C. fleurdelis reach up to 24.4 mm SL, with females highly mature at 21.0 mm SL (males were not extensively dissected to define smallest mature). Along with small size of maturation, the new species also has some apparent paedomorphic features, such as absence of parietal branch of the supraorbital canal, short lateral line, and low number of pectoral-fin rays.
Characidium fleurdelis shares the absence of the parietal branch of the supraorbital canal, used by Buckup (1993c) to diagnose the Clade C4 group of species which, according to the author, includes C. bahiense, C. interruptum and C. lanei Travassos, 1967. Netto-Ferreira et al. (2013) expanded the Clade C4, supported also by the presence of more than 12 bars on body and the presence of a single row of dentary teeth. The new species does not share the presence of a single row of dentary teeth, and possess variable presence of bars. Males of C. fleurdelis do not have bars while in females up to 20 irregular narrow, somewhat inconspicuous bars occurs.
Furthermore, the new species shares an incomplete lateral line and the absence of the parietal branch of the supraorbital canal with C. cacah, C. chicoi, C. nana, C. nupelia, C. stigmosum,and C. xavante, and shares thepresence of only four branchiostegal rays with C. bahiense, C. cacah, C. nana, C. nupelia, C. stigmosum, C. summum, and C. xavante. On the other hand, C. fleurdelis shares presence of teeth strongly tricuspid,with three well developed and similar sized cusps, with C. longum, C. pellucidum, and C. pteroides. Buckup (1993c) included C. pellucidum and C. pteroides in the Clade C7. According to the author, the synapomorphies supporting his clades C5 to C7 corresponds to a gradient in pigmentation pattern ranging from relatively wide vertical dark bands (e.g., C. zebra) to a highly modified pattern represented by dark inconspicuous vertical lines on the body. The Clade C7, according to the author, is diagnosed by the fragmentation of the vertical lines, which do not connect with the dorsal roots and are modified into posteriorly-directed semicircles. In C. fleurdelis the vertical bars or lines present in females are variably connected to the dorsal blotches, with specimens possessing both forms, therefore not fitting completely on the condition cited to the C7 clade. As stated in the previous section, C. fleurdelis shares with C. cf. pteroides the presence of two processes in the first rib(also shared with C. cf. longum), the elongation and flattening of the haemal spines of the first caudal vertebrae,and proximal radials of anal fin with plate-like bony expansions.
In addition, C. fleurdelis has a low number of total pectoral-fin rays (7–10), compared to most of its congeners, which have 13 rays (Melo et al., 2016). According to the authors, a low count of total pectoral-fin rays occurs also in C. bahiense Almeida (9–11 rays), C. bimaculatum Fowler, 1941 (12), C. etzeli Zarske & Géry, 2001 (11–12), C. geryi (Zarske, 1997) (8–9), C. heinianum Zarske & Géry, 2001 (10), C. helmeri Zanata, Sarmento-Soares & Martins-Pinheiro, 2015, (8–12), C. mirim (9–11), C. nupelia (11–13), C. sterbai (Zarske, 1997) (9–10), C. travassosi (10–12) and C. xavante, with 11–13 pectoral-fin rays. Characidium fleurdelis differs from these species by a series of features, including body coloration, number of pored lateral-line scales, and presence of strongly tricuspid teeth with similar sized cusps (see Diagnosis). Zarske, Géry (2001) considered C. heinianum closely related to C. pteroides and C. pellucidum, speciesthat possess morphological similarities with C. fleurdelis. In addition to the features cited above that can distinguish C. fleurdelis from C. heinianum, the former promptly differs from the latter by having a short lateral line with 6–8 perforated scales (vs. completely pored lateral line), and by having haemal spine of the antepenultimate vertebra far from the procurrent and caudal-fin rays(vs. haemal spine of the antepenultimate vertebra reaching the origins of the ventralmost caudal-fin rays, see Zarke, Géry 2001:232, fig. 3). Characidium papachibe Peixoto & Wosiacki, 2013 was also considered by the authors as slightly similar to the psammophile species C. heinianum, C. longum, C. pellucidum,and C. pteroides due to its reticulated color pattern. However, C. papachibe can be distinguished from those species, and from C. fleurdelis, by the presence of anteriorly oblique, midlateral bars centered on the lateral line or just ventral thereof and not reaching either the dorsum or the ventrum of the body. Furthermore, C. fleurdelis can be distinguished from C. papachibe by having incompletely pored lateral line (vs. completely pored), 12 series of scales around caudal peduncle (vs. 10), and tricuspid teeth with similarly sized cusps (vs. teeth with medial cusp larger).
The uniqueness of C. fleurdelis is unquestionable based on the morphological traits reported for the first time within the genus, but the evolution of such traits and the relationships of the species should be evaluated in future comprehensive phylogenetic studies of Characidium.
Comparative material examined. Comparative material was obtained from the list of species provided by Zanataet al. (2018) and Oliveira-Silva et al. (2022), with addition ofCharacidium cf. longum,MZUSP 122131, 10, 28.8–32.5 SL, 1 male c&s 32.5 mm SL, rio Manicoré, rio Madeira basin, Amazonas, Brazil; Characidium pteroides, LBP 6934, 36, 20.1–27.1 mm SL, rio Negro basin, São Gabriel da Cachoeira, Amazonas, Brazil; Characidium cf. pteroides,MZUSP 87646, 15, 17.9–36.3 SL, 2 c&s, 28.7–35.9 mm SL, male and female respectively, rio Preto da Eva, rio Amazonas basin, Amazonas, Brazil.
Acknowledgments
For the loan or permission to examine material we thank Osvaldo T. Oyakawa (MZUSP) and Lúcia Rapp Py-Daniel (INPA). We thank Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) for providing the collecting permits, Universidade Federal de Rondônia for support and logistic, Weder Tomicha and Valmir Alves (UNIR) for assistance during fieldwork. LOS was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brasil (CAPES, Finance Code 001).
References
Agudelo-Zamora HD, Tavera J, Murillo YD, Ortega-Lara A. The unknown diversity of the genus Characidium (Characiformes: Crenuchidae) in the Chocó biogeographic region, Colombian Andes: two new species supported by morphological and molecular data. J Fish Biol. 2020; 97(6):1662–75. https://doi.org/10.1111/jfb.14527
Buckup PA. Review of the characidiin fishes (Teleostei: Characiformes), with description of four new genera and ten new species. Ichthyol Explor Freshw. 1993a; 4(2):97–154.
Buckup PA. The monophyly of the Characidiinae, a Neotropical groups of characiform fishes (Teleostei: Ostariophysi). Zool J Linn Soc. 1993b; 108(3):225–45. https://doi.org/10.1111/j.1096-3642.1993.tb00297.x
Buckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993c; 9(3):305–41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
Eigenmann CH. Reports on the expedition to British Guiana of the Indiana University and the Carnegie Museum, 1908. Report no. 1. Some new genera and species of fishes from British Guiana. Ann Carnegie Mus. 1909; 6(1):4–54.
Flausino Junior N, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique midwater behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350–60. https://doi.org/10.11646/zootaxa.4816.3.5
Fricke R, Eschmeyer WN. Eschmeyer’s catalog of fishes: guide to fish collections. [Internet]. San Francisco: California Academy of Science; 2021. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/collections.asp
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2022. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Géry J. Jobertina eleodrioides [sic] n. sp. (Characidiinae) from French Guiana with considerations about the genus and redescription of the type-species. Contributions to the study of the Characoid fishes (Ostariophysi) III. Opusc. zool. Herausgegeben von der Zoologischen Staatssammlung in München 1960; 47:1–10.
Graça WJ, Ota RR, Domingues WM. A new species of miniature Characidium (Characiformes: Crenuchidae) from the upper rio Paraguay basin, Mato Grosso State, Brazil. J Fish Biol. 2019; 95(6):1480–85. https://doi.org/10.1111/jfb.14169
International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for using the IUCN Red List categories and criteria. Version 14 [Internet]. Gland; 2022. Available from: https://www.iucnredlist.org/resources/redlistguidelines
Leitão RP, Buckup PA. A new species of Characidium (Characiformes: Crenuchidae) from coastal basins of Serra do Mar, southeastern Brazil. Copeia. 2014; 2014(1):14–22. https://doi.org/10.1643/CI-12-137
Lujan NK, Agudelo-Zamora H, Taphorn DC, Booth PN, López-Fernández, H. Description of a new, narrowly endemic South American darter (Characiformes: Crenuchidae) from the central Guiana Shield highlands of Guyana. Copeia. 2013; 2013(3):454–63. https://doi.org/10.1643/CI-12-079
Malanski E, Sarmento-Soares LM, Silva-Malanski ACG, Lopes MM, Ingenito LFDS, Buckup PA. A new species of Characidium (Characiformes: Crenuchidae) from coastal basins in the Atlantic Rainforest of eastern Brazil, with phylogenetic and phylogeographic insights into the Characidium alipioi species group. Neotrop Ichthyol. 2019; 17(2):e180121. https://doi.org/10.1590/1982-0224-20180121
Melo MRS, Oyakawa OT. A new species of Characidium Reinhardt (Characiformes, Crenuchidae) with a distinctively dimorphic male. Copeia. 2015; 103(2):281– 89. https://doi.org/10.1643/CI-14-073
Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná state, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. http://dx.doi.org/10.1590/1982-0224-20160014
Melo MRS, Bouquerel BB, Masumoto FT, França RS, Netto-Ferreira AL. A new species of Characidium (Characiformes: Crenuchidae) from the Chapada dos Veadeiros, Goiás, Brazil. Neotrop Ichthyol. 2021; 19(2):e200152. https://doi.org/10.1590/1982-0224-2020-0152
Netto-Ferreira AL, Birindelli JLO, Buckup PA. A new miniature species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from the headwaters of the rio Araguaia, Brazil. Zootaxa. 2013; 3664(3):361–68. http://dx.doi.org/10.11646/zootaxa.3664.3.6
Oliveira-Silva L, Santos SAD, Lopes MM, Zanata AM. A new species of Characidium (Characiformes: Crenuchidae) from the rio Doce basin, Brazil. Neotrop Ichthyol. 2022; 20(1):e210125 https://doi.org/10.1590/1982-0224-2021-0125
Pansonato-Alves JC, Serrano EA, Utsunomia R, Camacho JP, Costa Silva GJ, Vicari MR et al. Single origin of sex chromosomes and multiple origins of B chromosomes in fish genus Characidium. PLoS ONE. 2014; 9(9):e107169. https://doi.org/10.1371/journal.pone.0107169
Pearson NE. The fishes of the eastern slope of the Andes. I. The fishes of the rio Beni basin, Bolivia, collected by the Mulford expedition. Ind Univ Stud. 1924; 11(64):1–83.
Peixoto LAW, Wosiacki WB. A new species of Characidium (Characiformes: Crenuchidae) from the lower Amazon. Copeia. 2013; 2013(1):52–57. https://doi.org/10.1643/CI-12-080
Sales NG, Mariani S, Salvador GN, Pessali TC, Carvalho DC. Hidden diversity hampers conservation efforts in a highly impacted Neotropical river system. Front Genet. 2018; 9(271):1–11. https://doi.org/10.3389/fgene.2018.00271
Serrano EA, Melo BF, Freitas-Souza D, Oliveira MLM, Utsunomia R, Oliveira C et al. Species delimitation in Neotropical fishes of the genus Characidium (Teleostei, Characiformes). Zool Scr. 2019; 48(1):69–80. https://doi.org/10.1111/zsc.12318
Taphorn DC, Montaña CG, Buckup PA. Characidium longum (Characiformes: Crenuchidae), a new fish from Venezuela. Zootaxa. 2006; 1247(1):1–12. https://doi.org/10.11646/zootaxa.1247.1.1
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Teixeira TF, Melo MR. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) from the Río Huallaga, central Peruvian Amazon, with a review of secondary sexual characters in the genus. J Fish Biol. 2021; 98(1):178–88. https://doi.org/10.1111/jfb.14568
Vazzoler AEAM. 1996. Biologia da reprodução de peixes teleósteos: teoria e prática. Maringá: Eduem/SBI.
Weitzman SH, Vari RP. Miniaturization in South American freshwater fishes: an overview and discussion. Proc Biol Soc Wash. 1988; 101(2):444–65.
Zanata AM, Vari RP. The family Alestidae (Ostariophysi, Characiformes): a phylogenetic analysis of a trans-Atlantic clade. Zool J Linn Soc. 2005;145(1):1–144. https://doi.org/10.1111/j.1096-3642.2005.00183.x
Zanata AM, Sarmento-Soares ML, Martins-Pinheiro RF. A new species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from coastal rivers in the extreme south of Bahia, Brazil. Zootaxa. 2015; 4040(3):371–83. https://doi.org/10.11646/zootaxa.4040.3.8
Zanata AM, Ohara WM, Oyakawa OT, Dagosta FC. A new rheophilic South American darter (Crenuchidae: Characidium) from the rio Juruena basin, Brazil, with comments on morphological adaptations to life in fast-flowing waters. J Fish Biol. 2020; 97(5):1343–53. https://doi.org/10.1111/jfb.1448
Zanata AM, Ramos TPA, Oliveira-Silva L. A new species of Characidium (Characiformes: Crenuchidae) from the rio Parnaíba basin, northeastern Brazil. Zootaxa. 2018; 4514(1):77–86. https://doi.org/10.11646/zootaxa.4514.1.6
Zarske A. Geryichthys sterbai gen. et spec. nov. and Microcharacidium geryi spec. nov.: Beschreibung einer neuen Gattung und zweier neuer Arten von Bodensalmlern aus dem Einzugsgebiet des Rio Ucayali in Peru (Teleostei: Ostariophysi: Characiformes: Characidiidae). Zool Abh. 1997; 49(2):157–72.
Zarske A, Géry J. Beschreibung von drei neuen Arten der Gattung Characidium Reinhardt, 1866 aus Bolivien und Paraguay (Teleostei: Characiformes: Characidiidae). Zool Abh. 2001; 51(16):229–46.
Authors
![]() Angela Maria Zanata1
Angela Maria Zanata1 ![]() ,
, ![]() Leonardo Oliveira-Silva1 and
Leonardo Oliveira-Silva1 and ![]() Willian M. Ohara2,3
Willian M. Ohara2,3
[1] Programa de Pós-Graduação em Biodiversidade e Evolução, Instituto de Biologia, Universidade Federal da Bahia, Rua Barãode Jeremoabo, 147, Ondina, 40170-115 Salvador, BA, Brazil. (AMZ) zanata.angela@gmail.com (corresponding author), (LOS) leonardoufcg.bio@hotmail.com.
[2] Departamento de Biologia, Universidade Federal do Amazonas, Av. Gen. Rodrigo Octávio, 6200, Coroado, 69080-900 Manaus,AM, Brazil. (WMO) willianmohara@gmail.com.
[3] Programa de Pós-Graduação em Conservação e Uso Recursos Naturais, Departamento de Biologia, Universidade Federal de Rondônia, BR-364, Porto Velho, RO, Brazil.
Authors’ Contribution 

Angela Maria Zanata: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Leonardo Oliveira-Silva: Data curation, Formal analysis, Writing-original draft, Writing-review and editing.
Willian M. Ohara: Conceptualization, Formal analysis, Funding acquisition, Methodology, Project administration, Writing-review and editing.
Ethical Statement
Permission for collecting was granted by ICMBio and by Sistema de Autorização e Informação em Biodiversidade (SISBIO 65628–3).
Competing Interests
The author declares no competing interests.
How to cite this article
Zanata AM, Oliveira-Silva L, Ohara WM. A new sexually dichromatic miniature Characidium (Characiformes: Crenuchidae) from the rio Guaporé, rio Madeira basin, Brazil, with remarkable morphological novelties to the genus. Neotrop Ichthyol. 2023; 21(1):e220059. https://doi.org/10.1590/1982-0224-2022-0059
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted December 27, 2022 by Marcos Mirande
Accepted December 27, 2022 by Marcos Mirande
![]() Submitted June 24, 2022
Submitted June 24, 2022
![]() Epub March 13, 2023
Epub March 13, 2023