![]() Tatiane Pires de Sousa1
Tatiane Pires de Sousa1 ![]() ,
, ![]() Andréa Bialetzki2 and
Andréa Bialetzki2 and ![]() Lucia Aparecida de Fatima Mateus3
Lucia Aparecida de Fatima Mateus3
PDF: EN XML: EN | Supplementary: S1 S2 S3 | Cite this article
Abstract
The conservation of fish assemblages depends on the longitudinal and lateral connectivity between riverine habitats, in particular during the breeding season and the initial development. This study investigated the composition and spatio-temporal structure in the ichthyoplankton of the hydrographic basin of the Paraguay River in western Brazil to identify the local spawning grounds and the importance of the longitudinal connectivity of economically valuable migratory species. Data were collected at 10 sites between two breeding seasons (2017/2018 and 2018/2019). Were collected 8,635 larvae, representing 55 taxa in 25 families, including the migratory species Brycon hilarii, Hemisorubim platyrhynchos, Piaractus mesopotamicus, Prochilodus lineatus, Pseudoplatystoma spp., Salminus brasiliensis, Sorubim lima, and Zungaro jahu, which are important fishery resources, with the highest larval densities being recorded between November and January. The Sepotuba, Paraguay, Jauru, and Cabaçal rivers were the areas of greatest connectance, and should thus be considered critical for the conservation of the longitudinal connectivity of this fluvial system, indicating that the migratory species spawn upriver. More efficient fisheries management mechanisms are needed, respecting the spawning period of migratory species, maintaining quality and longitudinal connectivity between habitats, and characteristics necessary for successful larval recruitment.
Keywords: Breeding migrations, Connectivity, Conservation and dispersal, Ichthyoplankton, Spawning grounds.
A conservação das assembleias de peixes depende da conectividade longitudinal e lateral entre habitats ribeirinhos, em especial durante o período reprodutivo e de desenvolvimento inicial. Objetivou-se avaliar a composição e a estrutura espaço-temporal do ictioplâncton da bacia hidrografia do rio Paraguai no oeste do Brasil para identificar as áreas de desova e a importância da conectividade longitudinal para a reprodução das espécies migradoras de interesse comercial. Os dados foram coletados em 10 pontos entre dois períodos reprodutivos (2017/2018 e 2018/2019). Capturaram-se 8635 larvas, pertencentes a 25 famílias e 55 táxons, incluindo espécies migradoras de interesse comercial, Brycon hilarii, Hemisorubim platyrhynchos, Piaractus mesopotamicus, Prochilodus lineatus, Pseudoplatystoma spp., Salminus brasiliensis, Sorubim lima e Zungaro jahu, que são importantes recursos pesqueiros,com maiores densidades larvais entre novembro e janeiro. Os locais com maiores interações e conectância foram os rios Sepotuba, Paraguai, Jauru e Cabaçal e, portanto, devem ser considerados áreas críticas para a conservação da conectividade longitudinal no sistema fluvial, indicando que as espécies migradoras têm desovado a montante dos locais amostrados. São necessários mecanismos de gestão pesqueira mais eficientes, respeitando o período de desova das espécies migradoras, mantendo a qualidade e conectividade longitudinal entre habitats, características necessárias ao sucesso do recrutamento larval.
Palavras-chave: Áreas de desova, Conectividade, Conservação e dispersão, Ictioplâncton, Migrações reprodutivas.
Introduction
Rivers are complex aquatic ecosystems that typically encompass a considerable heterogeneity of lotic and lentic habitats, as well as backwaters and floodplains, which may vary in their distribution and connectivity (Nannini et al., 2012). The fish exploit these fluvial biotopes during different stages of their life cycle, in particular during spawning and initial development (Kallasvuo et al., 2017), which guarantee larval recruitment. However, the rivers of the Neotropical region are coming under increasing pressure from hydroelectric dams, which have impacted many hydrographic basins on a large scale (Suzuki et al., 2016). Hydroelectric power plants impose different barriers on the dispersal of many fish, in particular, long-distance migrants and rheophilic species. The impoundment of the river impedes movements upriver from the dam, while the reservoir affects primarily the movements downriver from the dam, through the reduction of lotic environments and river flow changes (Pelicice et al., 2015).
During the reproduction season, long-distance migrants search for spawning habitats, which are typically found in upriver locations (Makrakis et al., 2012). This results in longitudinal movements along with the river system toward its headwaters, where the fish spawn in the main channel of the river and the eggs drift passively downriver, with the larvae hatching downstream (Lopes et al., 2014), and subsequently disperse further until they reach an area appropriate for their early development (Baumgartner et al., 2004). The success of the larval recruitment will depend on the drift of the lotic waters as far as swampy riparian habitats, where the larvae encounter feeding resources and refuges from predators (Suzuki et al., 2009). The spawning period of the long-distance migrants coincides with the flood pulse and relatively high temperatures (Zaniboni-Filho et al., 2017), when new habitats become available and floodplain lakes are connected to the main channel of the river through the inundation of marginal environments (Junk et al., 1989). In this scenario, the recruitment of the juveniles into the adult population will depend on the preservation of both the spawning grounds and the nurseries in which the larvae grow and develop (Bower et al., 2015; Teixeira et al., 2019), as well as the connectivity between these different habitats within the fluvial system (Branco et al., 2016).
The Brazilian upper Paraguay River basin in encompasses two distinct sectors — the plateau and the Pantanal floodplain. The plateau rivers fed the Pantanal wetlands’ flood pulse, forming a complex aquatic ecosystem. Modern-day complication is the increasing impoundment of the tributaries of the Paraguay River for the implantation of hydroelectric power plants, which not only affects the hydrological regime of the Pantanal but also interrupts the connectivity of the fluvial systems, which reduces the access of fish to the nursery habitats and feeding grounds on the floodplains (Winemiller et al., 2016; Alho, Reis, 2017). This reinforces the need to identify the key areas of larval recruitment of migratory fish, given that these species require long stretches of well-preserved riverine habitat in which to complete their life cycle. It is also important to identify the importance of the connectivity of the habitats used by the fish species during the initial phases of their life cycle, in particular in areas subject to impoundment for hydroelectric projects (Agostinho et al., 2002). The temporal variation in using of the different habitats is an additional facet of the life cycle of the species, which helps to define the spawning period and provides important guidelines for the establishment of protective measures for the breeding stocks.
In this context, the spatio-temporal analysis of ichthyoplankton distribution provides important insights into the location of the spawning grounds and nursery areas of fish species (Baumgartner et al., 2004), and in particular, the dispersal process, which is fundamental to the reproductive success of these species (Barthem et al., 2014). Furthermore, this study uses the abundance of fish larvae to evaluate the reproductive patterns of different species in the Brazilian upper Paraguay River basin. Specifically, the study (i) analyzes the temporal and spatial distribution of the fish larvae, (ii) determines the taxonomic composition of the fish larvae, with emphasis on long-distance migrants and species with a high commercial value, (iii) detects the spawning grounds of the species, in particular the most valuable fishery resources, and the dispersal of their offspring, and (iv) demonstrates the importance of longitudinal connectivity for the reproduction of commercially valuable migratory species. We predicted that the fish larvae disperse passively within the fluvial system according to the migratory patterns of the spawning stock, and tested two hypotheses: (i) that there will be a higher density of fish larvae at the sampling sites further downstream within the Paraguay River basin (i.e., on the Cabaçal [site CAB2], Jauru, Sepotuba [site SEP3], and Paraguay rivers), and (ii) that the migratory species will depend on the longitudinal connectivity of the fluvial system to ensure their spawning migrations, with the larvae subsequently dispersing to the marginal areas of the floodplain.
Material and methods
Study area and sampling. In Brazil, there are two different sectors of the upper Paraguay River basin, the plateau and the Pantanal floodplain, with distinct sets of natural and hydrological resources (Alho, 2008). The plateau is located to the north and east of the upper Paraguay River basin, and covers an area of 214,802 km2, about 59% of the basin in Brazil (Galdino et al., 2006). This area encompasses the headwaters of the principal rivers that flow toward the Pantanal floodplain. The main tributaries of the upper Paraguay River basin include the Paraguay, Jauru, Sepotuba, Cuiabá, and São Lourenço rivers. This region has a seasonal humid tropical climate, with two well-defined seasons, rainy and dry, and mean annual precipitation of 1,400 mm, most that falls between November and March (Fantin-Cruz et al., 2011).
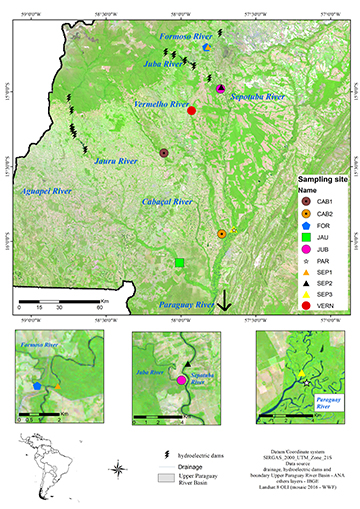
The study area is located on the Paraguay River and its right-margin tributaries on the plateau, a region with several future hydroelectric projects still at the planning stage (Silva et al., 2007; Souza et al., 2017; Calheiros et al., 2018). A total of 10 sampling sites were established within this area (Fig. 1) on the Cabaçal (sites CAB1 and CAB2), Formoso (FOR), Jauru (JAU), Juba (JUB), Sepotuba (sites SEP1, SEP2, and SEP3), Vermelho (VERN), and Paraguay (PAR) rivers, all in the state of Mato Grosso, in western Brazil. Collections considered each water body distinct from each other due to the motionless characteristics of the ichthyoplankton, with one site in the tributary and another in the main river, upstream of the tributary mouth. However, the overlap of the FOR/SEP1, JUB/SEP2, and SEP3/PAR sites corresponds to the proximity of the collected water bodies (see Fig. 1). On the two rivers with impoundments — the Jauru and Juba — the sampling sites were located in non-flooded stretches downriver from the dams because of the loss of connectivity due to the projects. Sites CAB2, JAU, SEP3, and PAR were considered representing downriver areas in the river system studied here (Tab. S1).
FIGURE 1| Study area in the Brazilian upper Paraguay River basin in the Brazilian state of Mato Grosso, sampled during the 2017/18 and 2018/2019 breeding seasons. CAB1 = Cabaçal River site 1; CAB2 = Cabaçal River site 2; FOR = Formoso River; JAU = Jauru River; JUB = Juba River; PAR = Paraguay River; SEP1 = Sepotuba River site 1; SEP2 = Sepotuba River site 2; SEP3 = Sepotuba River site 3; VERN = Vermelho River. The overlap of the FOR/SEP1, JUB/SEP2 and SEP3/PAR sites corresponds to the proximity of the collected water bodies.
To investigate the spatial-temporal variation, monthly samplings were carried out in each of the 10 sites, in two consecutive breeding seasons, November–March 2017/2018 (RP1) and October–March 2018/2019 (RP2), respecting the reproductive cycle of the regional ichthyofauna (Ziober et al., 2012). Ichthyoplankton was collected using conical–cylindrical plankton nets with a mesh size of 0.5 mm and 0.38 m3 diameter, with a flowmeter attached to measure the volume of filtered water. To encompass the spatial variation inside each site, the surface layer of the water column (right and left margins, and center channel) and at the bottom were sampled using nets submerged for 10 and 3 min, respectively. For bottom samples, we use adapted conical-cylindrical plankton nets coupled to a sled-shaped iron structure for sites over 1.5 m deep. The sampling was performed at 6 h intervals (00, 06, 12 and 18h) for to include the nictemeral variation. These samples were grouped to represent the spatio-temporal variation inside the sites. Thus, for each month sampled, 10 replicates were obtained. Ichthyoplankton was fixed in 4% formalin, and buffered with calcium carbonate (CaCO3) to conservation of the biological material. In the laboratory, the larvae were separated and quantified using a stereomicroscope. The abundance of the larvae was standardized to a volume of 10 m3 of filtered water (Tab. S2), following Tanaka (1973), as modified by Nakatani et al. (2001). The larvae were identified using the regressive developmental sequence technique, which involves the morphological comparison of the smaller individuals with more developed forms, using traits such as body shape, the presence of barbels, the sequence of the formation of the fins, the relative position of the anal opening, and the number of vertebrae/myomeres or fin rays (Ahlstrom, Moser, 1976; Nakatani et al., 2001), which was supported by literature (Oldani 1979 a,b; Nascimento, Araújo-Lima, 2000; Nakatani et al., 2001; Oliveira et al., 2012; Andrade et al., 2014, 2016; Taguti et al., 2015; Garcia et al., 2016). Larvae were identified to the lowest possible taxonomic level (larvae identified to at least genus included some long-distance migrant species) (Suzuki et al., 2004). Once identified, the larvae were assigned to one of the four development stages (yolk-sac, preflexion, flexion, and postflexion) following Ahlstrom et al. (1976), as modified by Nakatani et al. (2001), considering the flexion of the notochord and its supporting elements.
Data analysis. Spatio-temporal distribution. We used a longitudinal design, where the same sampling units (10 sites) were sampled over time (months). The analyzes were conducted considering this design to adjust the degrees of freedom since samples in the same sample unit over time are not independent. After testing the normality and homoscedasticity of the data, a nonparametric test with inter-sample dependence (nested month by sampling site) was used. The differences in larval density (i.e., individuals/10 m3) among sites and between periods were verified using the Friedman (repeated-measures analysis of variance – ANOVA) nonparametric test (MacFarland, Yates, 2016), to avoid temporal pseudoreplication, with Dunn’s post hoc test for pairwise comparisons (Dunn, 1964).
Taxonomic composition. We evaluated the differences in the taxonomic composition of the samples using a PERMANOVA based on a Bray-Curtis matrix, with 9999 permutations, considering all the larvae together, independent of the level of their taxonomic identification. A permutation test of the homogeneity of multivariate dispersal was applied to test the significance of the differences in the taxonomic composition of the samples at a spatio-temporal scale (Anderson et al., 2006). The dispersal of the data on the composition of the larval assemblage was used to calculate the homogeneity of the multivariate dispersal (PERMDISP), considering the dissimilarity of a sample concerning its distance from the centroid of the entire group of samples. The values were then configured spatially using a Principal Coordinates Analysis (PCoA). The spatial autocorrelation in the taxonomic composition of the larvae with the distance between the sampling sites was verified using a partial Mantel test. Euclidean distance, with the geographical coordinates transformed into UTM, was used as a matrix of geographic distance, while the Bray-Curtis distance was applied for the taxonomic composition.
Spawning. An indicator species analysis (IndVal; Dufrene, Legendre, 1997) was used to identify associations at spatial (Cáceres, 2022) and temporal scales with 999 permutations. The larval development stages of the commercially valuable migratory species were related to the spatial scale, which permitted the inference of the possible location of the spawning grounds and the dispersal of the fry, while the temporal scale was evaluated using only the larvae that were identified to species.
Longitudinal connectivity. We used a bipartite network approach to evaluate the importance of longitudinal connectivity for economically-valuable migratory species in the upper Paraguay basin (Mateus et al., 2004; Alho, Reis, 2017). This type of approach is typically used for the analysis of pollinator or predator-prey networks (Dormann et al., 2008, 2009), but may also be applied to other types of interaction. Here, we used metrics for quantitative matrices, considering the density of the migratory species identified at each sampling site. We then compiled a bipartite plot, which shows the relationship between the density of species and the sampling sites, to visualize the interaction network.
To evaluate whether the interruption of longitudinal connectivity, by impoundment, for example, impedes the access of fish to spawning grounds upriver from a sampling site, we simulated the elimination of connections beginning with the sites with a greater frequency of connections and estimated the coefficient of extinction, ‘a’ (Memmott et al., 2004). We assumed that the proportion of larvae (y) reflects an exponential function of the proportion of extinction (disconnected) sites, whose slope is estimated by coefficient ‘a’. The discontinuity of the sites affects the greater the value of ‘a’ (Dormann et al., 2009). We repeated the procedure, this time beginning with the elimination of the sites with the lowest density of connections, as well as the random discontinuity of the sites.
We also evaluated the robustness of the system to the loss of connectivity. Burgos et al. (2007) developed a quantitative measure of robustness, based on a parameter, R, which is defined as the area under the extinction curve. Intuitively, R = 1 corresponds to a curve that declines gently to the scenario in which almost all the sites are eliminated (unconnected). This is consistent with a very robust system in which, for example, most of the species persist, even after a large proportion of the sites have been disconnected. By contrast, R = 0 represents a curve that declines abruptly as soon as any one connection is lost. This is consistent with a fragile system in which, for example, most of the species lose all their interactions (that is, the potential to reach their spawning grounds) and become extinct, even when only a small proportion of the sites have become disconnected (Burgos et al., 2007; Dormann et al., 2009).
The estimates of ‘a’ and ‘R’ were compared with the null models, which were generated based on 1000 simulations, using a randomization algorithm that maintains constant the sum of the lines and columns of the matrix (Oksanen et al., 2019). All the analyses were run in the R environment (R Development Core Team, 2020), using the indicspecies (Cáceres, 2022), FD, lmPerm (Wheeler, Torchiano, 2016), vegan (Oksanen et al., 2019), and bipartite (Dormann et al., 2020) packages, considering a significance level of p < 0.05.
Results
Spatio-temporal distribution. These samples resulted in 8,635 larvae. The Friedman test indicates significant effects of spatial scale (X2(9) = 27.644; p = 0.001) and the sample month (X2(5) = 27.248; p < 001) on the larval density. Dunn’s post hoc test (Tab. S1) showed higher significant larval densities (p < 0.05) in downstream areas, between CAB2, JAU, SEP3, PAR, and VERN and all the other sampling sites. Larval densities recorded in November, December, January, and February were all significantly different (p < 0.05) from October and March. The reproductive peak occurred between November and February in both reproductive periods with the highest larval densities, indicating the onset of spawning between October and late March (months with the lowest larval density, Fig. S3).
Taxonomic composition. The individuals recorded in this study belonged to five taxonomic orders and 25 families, with the highest densities being recorded for the orders Characiformes, Siluriformes. Anostomidae, Characidae, and Pimelodidae were the families with the highest larval densities. The larvae were distributed in 55 lower taxa (Tab. 1), including individuals representing the commercially valuable migratory species Brycon hilarii (Valenciennes, 1850), Hemisorubim platyrhynchos (Valenciennes, 1840), Piaractus mesopotamicus (Holmberg, 1887), Prochilodus lineatus (Valenciennes, 1837), Pseudoplatystoma spp., Salminus brasiliensis (Cuvier, 1816), Sorubim lima (Bloch, Schneider, 1801), and Zungaro jahu (Ihering, 1898). The highest larval densities were recorded for P. lineatus, Rhaphiodon vulpinus Spix & Agassiz, 1829, and H. platyrhynchos, whereas the larvae of Z. jahu were collected only on the Sepotuba River (SEP1, SEP2, and SEP3).
TABLE 1 | Mean density (per 10 m3) of the fish larvae taxa sampled in the upper Paraguay River basin in the Brazilian state of Mato Grosso during the breeding seasons of 2017/2018 and 2018/2019. CAB1 = Cabaçal River site 1; CAB2 = Cabaçal River site 2; FOR = Formoso River; JAU = Jauru River; JUB = Juba River; PAR = Paraguay River; SEP1 = Sepotuba River site 1; SEP2 = Sepotuba River site 2; SEP3 = Sepotuba River site 3; VERN = Vermelho River; aLarvae identified to order; bLarvae identified to family; cLarvae identified to subfamily; *Commercially valuable migratory species.
Taxonomic composition | Sampling
sites | |||||||||
CAB1 | CAB2 | FOR | JAU | JUB | PAR | SEP1 | SEP2 | SEP3 | VERN | |
Beloniformes | | | | | | | | | | |
Belonidae | ||||||||||
Pseudotylosurus angusticeps (Günther,
1866) | | | | 0.04 | | | | |||
Characiformesa | | 43.15 | 0.15 | 6.82 | | 15.18 | 0.04 | | 9.34 | 0.05 |
Acestrorhynchidae | ||||||||||
Acestrorhynchus pantaneiro Menezes, 1992 | 0.04 | |||||||||
Anostomidaeb | 95.83 | 0.07 | 104.59 | 0.45 | 33.35 | 0.17 | 0.34 | 214.20 | 7.24 | |
Leporinus spp. | 0.32 | 0.52 | ||||||||
Bryconidaeb | 0.23 | |||||||||
Brycon hilarii* (Valenciennes,
1850) | 12.61 | 14.37 | ||||||||
Salminus brasiliensis* (Cuvier, 1816) | 0.74 | 1.68 | 0.04 | 0.04 | ||||||
Characidaeb | 98.14 | 12.95 | 3.36 | 0.18 | 7.89 | 0.03 | ||||
Aphyocharax spp. | 0.09 | |||||||||
Bryconamericus exodon Eigenmann, 1907 | 0.89 | 0.05 | ||||||||
Bryconamericus spp. | 0.11 | 10.47 | 0.12 | 0.02 | ||||||
Hemigrammus spp. | 0.29 | |||||||||
Hemigrammus tridens Eigenmann, 1907 | 0.07 | |||||||||
Hyphessobrycon eques (Steindachner,
1882) | 0.08 | |||||||||
Moenkhausia spp. | 0.10 | |||||||||
Piabarchus stramineus (Eigenmann,
1908) | 0.07 | |||||||||
Poptella paraguayensis (Eigenmann,
1907) | 0.03 | 0.02 | ||||||||
Psellogrammus kennedyi (Eigenmann,
1903) | 0.06 | |||||||||
Serrapinnus calliurus (Boulenger,
1900) | 0.01 | |||||||||
Tetragonopterus argenteus Cuvier, 1816 | 0.07 | |||||||||
Xenurobrycon macropus Myers & Miranda Ribeiro, 1945 | 3.01 | |||||||||
Characinaec | 0.13 | 0.02 | 0.02 | |||||||
Crenuchidae | ||||||||||
Characidium spp. | 0.05 | 0.02 | ||||||||
Curimatidaeb | 0.08 | 0.41 | 0.29 | 1.54 | 0.09 | |||||
Cyphocharax gillii (Eigenmann & Kennedy,
1903) | 0.05 | 0.04 | 0.15 | |||||||
Cynodontidae | ||||||||||
Rhaphiodon vulpinus Spix & Agassiz,
1829 | 1.26 | 64.71 | 0.06 | 2.97 | 0.93 | |||||
Erythrinidaeb | 0.15 | |||||||||
Hoplias spp. | 0.04 | 0.87 | 0.09 | 3.17 | 0.02 | 0.12 | 0.02 | 0.33 | 0.70 | |
Hemiodontidae | ||||||||||
Hemiodus spp. | 0.15 | 0.13 | 1.21 | 12.58 | 7.59 | |||||
Prochilodontidae | ||||||||||
Prochilodus lineatus* (Valenciennes, 1837) | 11.26 | 85.62 | 35.52 | 7.11 | ||||||
Serrasalmidaeb | 1.97 | 1.96 | 3.66 | 1.99 | 0.02 | |||||
Myloplus levis (Eigenmann
& McAtee, 1907) | 0.10 | |||||||||
Piaractus mesopotamicus* (Holmberg, 1887) | 0.40 |
| 2.07 | 0.28 | 0.02 | 13.72 | ||||
Serrasalmus spp. | 0.42 | |||||||||
Triportheidae | ||||||||||
Engraulisoma taeniatum Castro, 1981 | 0.13 | 0.03 | ||||||||
Triportheus angulatus (Spix & Agassiz, 1829) | 0.03 | |||||||||
Triportheus spp. | 0.09 | 0.19 | ||||||||
Gymnotiformesa | | | | | | 0.10 | | | 0.30 | |
Gymnotidae | ||||||||||
Gymnotus spp. | 4.76 | 0.11 | 0.31 | 0.02 | 0.04 | |||||
Hypopomidae | ||||||||||
Brachyhypopomus spp. | 0.12 | 0.05 | 0.03 | 0.05 | ||||||
Rhamphichthyidae | ||||||||||
Rhamphichthys hahni (Meinken,
1937) | 0.02 | |||||||||
Sternopygidaeb | 0.13 | 0.09 | ||||||||
Eigenmannia spp. | 0.07 | 0.61 | 0.56 | 0.57 | 0.01 | 0.96 | 0.05 | |||
Perciformes | | | | | | | | | | |
Cichlidae | ||||||||||
Satanoperca pappaterra (Heckel, 1840) | 0.04 | |||||||||
Siluriformesa | 0.12 | 0.71 | 0.08 | 0.14 | 0.14 | 0.58 | 0.56 | 0.25 | 0.94 | 0.49 |
Auchenipteridae | ||||||||||
Ageneiosus inermis (Linnaeus,
1766) | 0.04 | |||||||||
Ageneiosus spp. | 0.10 | |||||||||
Auchenipterus osteomystax (Miranda Ribeiro, 1918) | 0.37 | 0.49 | 0.31 | |||||||
Auchenipterus spp. | 0.12 | 0.07 | 0.18 | |||||||
Tatia neivai (Ihering,
1930) | 0.02 | 0.09 | 0.13 | |||||||
Trachelyopterus galeatus (Linnaeus,
1766) | 0.04 | |||||||||
Trachelyopterus spp. | 0.18 | 0.38 | 0.34 | 2.18 | 0.01 | |||||
Callichthyidaeb | 0.03 | 0.02 | 0.02 | |||||||
Callichthys callichthys (Linnaeus,
1758) | 0.01 | |||||||||
Hoplosternum littorale (Hancock, 1828) | 0.09 | 0.09 | ||||||||
Doradidaeb | 0.05 | 0.28 | 0.13 | 0.00 | 1.36 | |||||
Pterodoras granulosus (Valenciennes,
1821) | 0.02 | |||||||||
Heptapteridaeb | 0.12 | 0.09 | 0.01 | 0.28 | 0.18 | 0.03 | ||||
Rhamdia quelen (Quoy & Gaimard, 1824) | 1.23 | 0.04 | 0.16 | 0.16 | ||||||
Loricariidaeb | 0.02 | 0.10 | 0.09 | 0.05 | 0.06 | |||||
Loricaria spp. | 0.08 | |||||||||
Otocinclus vittatus Regan, 1904 | 0.08 | |||||||||
Loricariinaec | 0.07 | |||||||||
Pimelodidaeb | 0.01 | 13.17 | 0.05 | 1.46 | 30.17 | 0.04 | 0.70 | 9.85 | 20.45 | |
Hemisorubim platyrhynchos* (Valenciennes, 1840) | 0.15 | 0.02 | 6.13 | 12.88 | 34.37 | |||||
Pimelodus spp. | 15.19 | 11.98 | 3.01 | |||||||
Pseudoplatystoma spp.* | 0.05 | 0.13 | 1.17 | 1.66 | 0.03 | |||||
Sorubim lima* (Bloch & Schneider 1801) | 0.05 | 0.18 | ||||||||
Zungaro jahu* (Ihering, 1898) | 0.08 | 0.34 | 0.43 | |||||||
Trichomycteridaeb | 0.03 | |||||||||
Ituglanis spp. | | | | | 0.03 | | | | | |
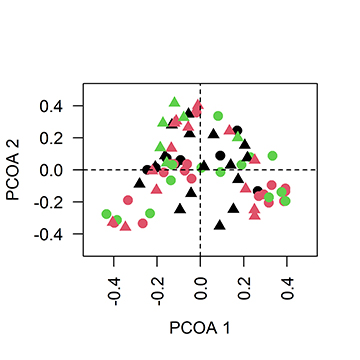
The results of the PERMANOVA revealed significant differences in the total taxonomic composition among the sample sites (F9;26 = 1.62; p < 0.001) and at a temporal scale (F5;70 = 1.40; p = 0.005). However, no significant variation was found in the heterogeneity of the species composition at either spatial or temporal scales (Figs. 2–3). The partial Mantel test detected a significant correlation between the matrix of the taxonomic composition of the larvae and the geographical distance between sampling sites (r = 0.23; p < 0.001), as well as for the composition of the commercially valuable migratory species (r = 0.19; p = 0.003), which indicates that sites closer together have a more similar composition of larval taxa.
FIGURE 2| Results of the Principal Coordinates Analysis (PCoA) based on the Bray-Curtis dissimilarity matrix of the taxonomic composition of the fish larvae in relation to the sites sampled in the upper Paraguay River basin in Mato Grosso State, western Brazil. CAB1 = Cabaçal River site 1 (closed black circle); CAB2 = Cabaçal River site 2 (closed red circle); FOR = Formoso River (black triangle); JAU = Jauru River (red asterisk); JUB = Juba River (blue triangle); PAR = Paraguay River (black cross); SEP1 = Sepotuba River site 1 (open black circle); SEP2 = Sepotuba River site 2 (open red circle); SEP3 = Sepotuba River site 3 (open green circle); VERN = Vermelho River (red square).
FIGURE 3| Results of the Principal Coordinates Analysis (PCoA) based on the Bray-Curtis dissimilarity matrix of the taxonomic composition of the fish larvae in relation to the months sampled in the upper Paraguay River basin in Mato Grosso State, western Brazil. OCT = October (black circle), NOV = November (red circle), DEC = December (green circle), JAN = January (black triangle), FEB = February (red triangle), MAR = March (green triangle).
Spawning. The commercially valuable migratory species presented significant patterns of the association at both spatial and temporal scales (Tab. 2; p < 0.05). The flexion larval stage of B. hilarii was associated with the Paraguay River. Brycon hilarii larvae in yolk-sac stage and Pseudoplatystoma spp. in flexion stage were both associated with site SEP3. The preflexion stage of P. lineatus were associated with the sites JAU/PAR and the yolk-sac stage of P. mesopotamicus, with sites JAU/SEP3, while the yolk-sac stage of Z. jahu were associated with sites SEP1/SEP2. The preflexion larvae of H. platyrhynchos were associated with sites SEP3/VERN. On a temporal scale, P. mesopotamicus was associated with November and December. Considering only the variation in the larval development stages (p < 0.05), the pre and postflexion larvae were associated with sites CAB2/JAU/PAR/SEP3 (COMB1), whereas the larvae in flexion were associated with sites CAB2/JAU/PAR/SEP3/VERN (COMB2) in the Tab. 2.
TABLE 2 | Significant indicator species values (IndVal) recorded at the sampling sites in the upper Paraguay River basin in two breeding seasons (2017–2018 and 2018–2019) in Mato Grosso State, western Brazil. CAB1 = Cabaçal River site 1; CAB2 = Cabaçal River site 2; FOR = Formoso River; JAU = Jauru River; JUB = Juba River; PAR = Paraguay River; SEP1 = Sepotuba River site 1; SEP2 = Sepotuba River site 2; SEP3 = Sepotuba River site 3; VERN = Vermelho River; COMB1 = association among sites CAB2/JAU/PAR/SEP3; COMB2 = association among sites CAB2/JAU/PAR/SEP3/VERN; LS = larval stage; stat = statistic; p = significance level; NA = not analyzed; YS = yolk-sac; PF = preflexion; FL = flexion; FP = postflexion.
Taxonomic composition | LS | Sampling sites | |||||||||||||||
PAR | SEP3 | JAU/PAR | JAU/SEP3 | SEP1/SEP2 | SEP3/VERN | COMB1 | COMB2 | ||||||||||
stat | p | stat | p | stat | p | stat | p | stat | p | stat | p | stat | p | stat | p | ||
Characiformes | | | | | | | | | | | | | | | | | |
Bryconidae | |||||||||||||||||
Brycon hilarii | YS | 0.29 | <0.01 | ||||||||||||||
Brycon hilarii | FL | 0.31 | <0.01 | ||||||||||||||
Serrasalmidae | |||||||||||||||||
Piaractus mesopotamicus | YS | 0.25 | 0.03 | ||||||||||||||
Prochilodontidae | |||||||||||||||||
Prochilodus lineatus | PF | 0.36 | <0.01 | ||||||||||||||
Siluriformes | | | | | | | | | | | | | | | | | |
Pimelodidae | |||||||||||||||||
Hemisorubim platyrhynchos | PF | 0.32 | <0.01 | ||||||||||||||
Pseudoplatystoma spp. | FL | 0.20 | 0.03 | ||||||||||||||
Zungaro jahu | YS | 0.23 | <0.01 | ||||||||||||||
NA | PF | | | | | | | | | | | | | 0.87 | <0.01 | | |
FP | 0.78 | <0.01 | |||||||||||||||
FL | | | | | | | | | | | | | | | 0.82 | <0.01 | |
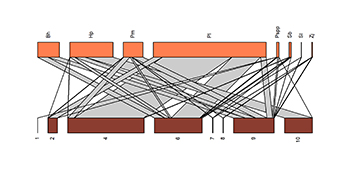
Longitudinal connectivity. The migratory–local larval density network (Fig. 4) is affected by the loss in connectivity among the sites, with low values of the extinction coefficient ‘a’, not only for the scenario in which the sites with the highest frequency of interactions are disconnected (a = 0.72; a̅null = 3.35; sd = 0.24; p < 0.0001) but also for the scenarios in which the disconnection begins with the sites of lower density (a = 0.718; a̅null = 3.36; sd = 0.23; p < 0.001) or is completely random (a = 0.55; a̅null = 3.44; sd = 0.21; p < 0.001). In all cases, the estimate of the value of ‘a’ is much lower than that expected by chance (p < 0.0001), which shows that losing of connectivity is likely to have a major impact on the density of migratory species larvae. Besides to the sensitivity of these species to extinction, the network indicated low levels of robustness when the loss of connectivity begins with the sites with the highest frequency of interaction (R = 0.49), which reflects the vulnerability of the system to the loss of connectivity in even a small proportion of the sites. By contrast, when the disconnection begins with the sites with a low frequency of interaction, the system is more robust (R = 0.78). This measure of robustness (R) was not different from that expected by chance, however, whether losing connectivity started with the sites with greater densities of connections (Rnull = 0.46; sd = 0.027; p = 0.85) or with those of lower density (Rnull = 0.74; sd = 0.010; p = 0.99). When the simulation of the loss of connectivity was random, the robustness was intermediate and, once again, not different from that expected by chance (R = 0.67; Rnull = 0.63; sd = 0.012; p = 0.99).
FIGURE 4| Bipartite plot showing the relationship between the larval densities of the commercially valuable migratory fish species (in orange) and the sampling sites (in brown). The width of the bars is proportional to the number of interactions, that is, the abundance of larvae. The bands (gray) and lines (black) reflect the species-sites connections, with the broader bands representing greater larval densities, while the lines represent the interactions of the species with the sampling sites. Bh = Brycon hilarii; Hp = Hemisorubim platyrhynchos; Pm = Piaractus mesopotamicus;Pl = Prochilodus lineatus; Pspp = Pseudoplatystoma spp.; Sb = Salminus brasiliensis; Sl = Sorubim lima; Zj = Zungaro jahu; Sites: 1 = Cabaçal River site 1; 2 = Cabaçal River site 2; FOR = Formoso River; 4 = Jauru River; 6 = Paraguay River; 7 = Sepotuba River site 1; 8 = Sepotuba River site 2; 9 = Sepotuba River site 3; 10 = Vermelho River.
Discussion
Our findings show the existence of significant spatio-temporal variation in the density of larvae over the course of the breeding season, with a fundamental association with geographic patterns, both for the assemblage and for the migratory species alone. The high larval densities at the downstream sites sampled within the study area (CAB2, JAU, PAR, and SEP3) highlight the importance of these rivers for larval recruitment and, ultimately, the preservation of the local fish fauna. Furthermore, the high density of the larvae collected in the study suggests that many taxa use spawning sites within this area between November and February, with the larvae subsequently dispersing to the habitats further downriver. Sampling took place during the period when most species of regional ichthyofauna are breeding (Resende, 2003). Despite considering the theoretical references, the non-inclusion of annual collection can be interpreted as a limitation of this work.
The presence of the initial embryonic development stages (yolk-sac and preflexion) indicates that the species are spawning near the sampling sites and confirms the drift of the larvae to nursery areas on the floodplain. It is also important to note that the species composition varied among the sites, with migratory species, including important fishery resources, being indicators of specific sampling sites, in particular, Paraguay and Sepotuba rivers (SEP3). It is worth mentioning that the sampling took place only in the rainy season and it is not possible to estimate the species that reproduce in the dry season, which does not guarantee a complete understanding of the reproduction of the ichthyofauna. Then, the interaction network that encompasses both species and sampling sites highlighted the fragility of the system and the importance of preserving the longitudinal connectivity of the fluvial system to guarantee the reproduction of commercially valuable migratory species.
The seasonal variation in the density of larvae in the study area reveals that the breeding season begins in October (the austral spring), and peaks between November and February, with a minor peak in March, representing the end of the breeding season. The groups of fish that make up the ichthyofauna of the Pantanal have several reproductive strategies, with the commercially valuable migratory species of the order Characiformes spawning at the onset of the breeding season, and those of the order Siluriformes spawning later in the season (Vazzoler, 1996). Even considering the absence of annual sampling or at other times of the year, the sampling in this work reflected the reproductive period for most species in the Paraguay River basin, as in other works in the literature. Clearly, each group of fish presents distinct patterns of breeding and larval development, which allow the whole assemblage to reproduce successfully at the end of the breeding season (Silva et al., 2019). The decline in larval density observed in March may be related to the last phase of the breeding season in the region (Tondato et al., 2010; Reynalte-Tataje et al., 2011; Ziober et al., 2012). These temporal data are thus essential for establishing protective measures that ensure the survival of spawning stocks, particularly through the implementation of adequate closed seasons by the regional fishery management authorities to protect these stocks.
The abundance of larvae of migratory species in downstream sites and taxonomic similarities with geographic space are evidence of longitudinal connectivity. The rivers constitute a dendritic network of channels connected by the downstream flow and distinct ecological processes, which may influence the dynamics of both the populations and community through the movement of the different species through the network (Grant et al., 2007). The reproductive success of the fish depends on the preservation of the connectivity of the different bodies of water to ensure the dispersal of individuals within the fluvial system (Branco et al., 2016; Humphries et al., 2020). The migratory fish species require adequate conservation measures that include the maintenance of uninterrupted stretches of river (Pelicice, Agostinho, 2008) and the longitudinal connectivity of spawning grounds and the lateral connectivity of areas of larval recruitment, as well as other types of habitats (Pelicice et al., 2015). However, our findings showed clearly that losing longitudinal connectivity induces the loss of species, reflecting the vulnerability of the network to extinction (low ‘a’ values). In this context, the sites with the highest level of interaction among the commercially valuable migratory species (SEP3, PAR, and CAB2) and larval abundance (JAU) should act as focal sites for the conservation of longitudinal connectivity in the upper Paraguay basin.
Despite the sampling points being downstream of the dams, these projects become physical barriers to reproductive migration as well as ecological barriers to ichthyoplankton. These impoundments interrupt the movements of migratory species during the spawning period (Fernandes et al., 2009; Barzotto et al., 2015) and retain the ichthyoplankton in the reservoir, isolating them from the habitats necessary to complete their life cycle (Pelicice, Agostinho, 2008; Pelicice et al., 2015; Winemiller et al., 2016; Brambilla et al., 2020). Additionally, the fluvial system was not robust (‘R’) to the loss of connectivity, even in widely distinct scenarios, with a lesser or greater density of connections or even a random arrangement. The reduced resilience of the network should be considered a key point for the future implantation of new hydroelectric projects because so many species spawn upriver from the sampling sites. Given this, the alteration and fragmentation of habitats may cause the loss of local species, which cannot be substituted by the process of dispersal, resulting in a simultaneous loss of species richness and an increase in the dissimilarity of the communities (Edge et al., 2017).
The associations observed at the spatio-temporal scale highlight the need to preserve the quality, complexity, and longitudinal connectivity of the region’s rivers to ensure adequate larval development and dispersal to the floodplain downstream. Migratory species can be used as indicators to identify the areas with characteristics intrinsically important for the adequate development and recruitment of the larvae, and the different stages of larval development. Given this, the administrators of freshwater systems must consider the migratory behavior of the fish into account, with their use of habitats within areas of overlap, to ensure the conservation of the largest possible number of species (Bower et al., 2015) through the preservation of the longitudinal and lateral connectivity among the different habitats (Branco et al., 2012; Pelicice et al., 2015; Cruz et al., 2020). It will also be essential to integrate scientific and traditional knowledge in the development of public policies that ensure the most effective fishery management strategies, to guarantee the reduction of conflicts of interest and socioeconomic inequalities (Schulz et al., 2019), as well as the reproductive success and the maintenance of the stocks of the region’s ichthyofauna.
The larval densities observed in the sampling sites, therefore, corroborate the hypothesis that the highest densities are observed in the areas further downstream (CAB2, JAU, SEP3, PAR, and VERN) due to passive dispersion of ichthyoplankton. This suggests that larvae can find refuge and growth areas toward the floodplain, favoring the recruitment of ichthyofauna. Although the Jauru River presents hydropower reservoirs upstream of the sampled area, the dam-free stretch offers suitable conditions for fish reproduction. However, losing connectivity in the dendritic network will have a great impact on the density of migratory larvae, corroborating the second hypothesis of this study. However, the simulation of losing connectivity of the fluvial system reflects in reduced population recruitment, interfering with the population dynamics and in the fishery production in the region.
Acknowledgments
We are grateful to the members of the Laboratório de Ecologia e Recursos Pesqueiros (LEMARPE) for all their assistance in the field. The fieldwork was financed by the Agência Nacional de Águas e Saneamento Básico (ANA) and the Fundação Eliseu Alves (FEA). We also thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), for providing TPS with a doctoral scholarship. We are also grateful to the Instituto de Biociências for the research infrastructure available at the Cuiabá campus of UFMT and the team of the Ichthyoplankton Laboratory of the Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (NUPÉLIA) at Universidade Estadual de Maringá for their support for the taxonomic identification of specimens.
References
Agostinho AA, Gomes LC, Fernadez DR, Suzuki HI. Efficiency of fish ladders for Neotropical ichthyofauna. River Res Appl. 2002; 18(3):299–306. https://doi.org/10.1002/rra.674
Ahlstrom EH, Butler JL, Sumida BY. Pelagic stromateoid fishes (Pisces, Perciformes) of the Eastern Pacific: kinds, distributions, and early life histories and observations on five of these from the northwest Atlantic. Bull Mar Sci. 1976; 26(3):285–402.
Ahlstrom EH, Moser HG. Eggs and larvae of fishes and their role in systematic investigations and in fisheries. Rev Trav Inst Peches Marit. 1976; 40(3/4):379–98.
Alho CJR. Biodiversity of the Pantanal: response to seasonal flooding regime and to environmental degradation. Braz J Biol. 2008; 68(4):957–66. http://dx.doi.org/10.1590/s1519-69842008000500005
Alho CJR, Reis RE. Exposure of fishery resources to environmental and socioeconomic threats within the Pantanal wetland of South America. Int J Aquact Fish Sci. 2017; 3(2):22–29. http://dx.doi.org/10.17352/2455-8400.000024
Anderson MJ, Ellingsen KE, McArdle BH. Multivariate dispersion as a measure of beta diversity. Ecol Lett. 2006; 9(6):683–93. https://doi.org/10.1111/j.1461-0248.2006.00926.x
Andrade FF, Lima AF, Assumpção L, Makrakis S, Kasai RID, Makrakis MC. Characterization of the early development of Pseudoplatystoma reticulatum Eigenmann & Eigenmann, 1889 (Siluriformes: Pimelodidae) from the Paraguay River Basin. Neotrop Ichthyol. 2016; 14(2):e150032. http://dx.doi.org/10.1590/1982-0224-20150032
Andrade FF, Makrakis MC, Lima AF, Assumpção L, Makrakis S, Pini SFR. Embryonic, larval, and juvenile development of Hemisorubim platyrhynchos (Siluriformes, Pimelodidae) of the Paraná River basin. Iheringia, Ser Zool. 2014; 104(1):70–80. http://dx.doi.org/10.1590/1678-4766201410417080
Barthem R, Costa MC, Cassemiro F, Leite RG, Silva Jr N. Diversity and abundance of fish larvae drifting in the Madeira River, Amazon Basin: sampling methods comparison. In: Grillo O, editor. Biodiversity – The Dynamic Balance of the Planet. Croatia: IntechOpen; 2014. p.137–58. http://dx.doi.org/10.5772/57404
Barzotto E, Sanches PV, Bialetzki A, Orvati L, Gomes LC. Larvae of migratory fish (Teleostei: Ostariophysi) in the lotic remnant of the Paraná River in Brazil. Rev Bras Zool. 2015; 32(4):270–80. http://dx.doi.org/10.1590/S1984-46702015000400002
Baumgartner G, Nakatani K, Gomes LC, Bialetzki A, Sanches PV. Identification of spawning sites and natural nurseries of fishes in the upper Paraná River, Brazil. Environ Biol Fishes. 2004; 71:115–25. https://doi.org/10.1007/s10641-004-0098-z
Bower SD, Lennox RJ, Cooke SJ. Is there a role for freshwater protected areas in the conservation of migratory fish? Inland Waters. 2015; 5(1):1–06. https://doi.org/10.5268/IW-5.1.779
Brambilla EM, Silva LGM, Baumgartner LJ, Bialetzki A, Nogueira MG. Dispersal of fish eggs and larvae in cascade of small hydropower plants with fish ladders. Hydrobiologia. 2020; 849:339–56. https://doi.org/10.1007/s10750-020-04425-5
Branco P, Boavida I, Santos JM, Pinheiro A, Ferreira MT. Boulders as building blocks: improving habitat and river connectivity for stream fish. Ecohydrology. 2012; 6(4):627–34. https://doi.org/10.1002/eco.1290
Branco P, Santos JM, Amaral S, Romão F, Pinheiro AN, Ferreira MT. Potamodromous fish movements under multiple stressors: connectivity reduction and oxygen depletion. Sci Total Environ. 2016; 572:520–25. http://dx.doi.org/10.1016/j.scitotenv.2016.08.070
Burgos E, Ceva H, Perazzo RPJ, Devoto M, Medan D, Zimmermann M, Delbue AM. Why nestedness in mutualistic networks? J Theor Biol. 2007; 249(1):307–13. http://dx.doi.org/10.1016/j.jtbi.2007.07.030
Cáceres M. Indicator species analysis using ‘indicspecies’. 2022. Available from: https://cran.r-project.org/web/packages/indicspecies/vignettes/IndicatorSpeciesAnalysis.html
Calheiros DF, Castrilon SKI, Bampi AC. Hidrelétricas nos rios formadores do pantanal: ameaças à conservação e às relações socioambientais e econômicas pantaneiras tradicionais. Rev Ibero-Am Cienc Amb. 2018; 9(1):119–39. http://doi.org/10.6008/CBPC2179-6858.2018.001.0009
Cruz DO, Kingsford RT, Suthers IM, Rayner TS, Smith JA, Arthington AH. Connectivity but not recruitment: response of the fish community to a large-scale flood on a heavily regulated floodplain. Ecohydrology. 2020; 13:e2194. https://doi.org/10.1002/eco.2194
Dormann CF, Fruend J, Gruber B. Visualising bipartite networks and calculating some (ecological) indices [Internet]. 2020. Available from: https://cran.r-project.org/web/packages/bipartite/bipartite.pdf
Dormann CF, Frund J, Bluthgen N, Gruber B. Indices, graphs and null models: analyzing bipartite ecological networks. The Open Ecol J. 2009; 2(1):7–24. http://dx.doi.org/10.2174/1874213000902010007
Dormann CF, Gruber B, Frund J. Introducing the bipartite package: analysing ecological networks. R News. 2008; 8(2):8–11.
Dufrene M, Legendre P. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr. 1997; 67(3):345–66. https://doi.org/10.1890/0012-9615
Dunn OJ. Multiple comparisons using rank sums. Technometrics. 1964; 6(3):241–52. http://dx.doi.org/10.1080/00401706.1964.10490181
Edge CB, Fortin MJ, Jackson DA, Lawrie D, Stanfield L, Shrestha N. Habitat alteration and habitat fragmentation differentially affect beta diversity of stream fish communities. Landsc Ecol. 2017; 32:647–62. https://doi.org/10.1007/s10980-016-0472-9
Fantin-Cruz I, Pedrollo O, Castro NMR, Girard P, Zeilhofer P, Hamilton SK. Historical reconstruction of floodplain inundation in the Pantanal (Brazil) using neural networks. J Hydrol. 2011; 399(1):376–84. https://doi.org/10.1016/j.jhydrol.2011.01.014
Fernandes R, Agostinho AA, Ferreira EA, Pavanelli CS, Suzuki HI, Lima DP, Gomes LC. Effects of the hydrological regime on the ichthyofauna of riverine environments of the Upper Paraná River floodplain. Braz J Biol. 2009; 69(2):669–80. https://doi.org/10.1590/S1519-69842009000300021
Galdino S, Vieira LM, Pellegrin LA. Impactos ambientais e socioeconômicos na Bacia do Rio Taquari – Pantanal. Corumbá: EMBRAPA Pantanal; 2006.
Garcia DAZ, Costa ADA, Leme GLA, Almeida FS, Silva ES, Orsi ML. A bacia do Paranapanema. In: Orsi ML, Almeida FS, Swarça AC, Claro-Garcia A, Vianna NC, Garcia DAZ, Bialetzki A, editores. Ovos, larvas e juvenis dos peixes da bacia do rio Paranapanema: uma avaliação para a conservação. Assis: Triunfal Gráfica and Editora; 2016. p.25–30.
Grant EHC, Lowe WH, Fagan WF. Living in the branches: population dynamics and ecological process in dendritic networks. Ecol Lett. 2007; 10:165–75. http://dx.doi.org/10.1111/j.1461-0248.2006.01007.x
Humphries P, King A, McCasker N, Kopf RK, Stoffels R, Zampatti B, Price A. Riverscape recruitment: a conceptual synthesis of drivers of fish recruitment in rivers. Can J Fish Aquat Sci. 2020; 77(2):213–25. https://doi.org/10.1139/cjfas-2018-0138
Junk WJ, Bayley PB, Sparks RE. The flood pulse concept in river-floodplain systems. In: Dodge DP, editor. Proceedings of the International Large River Symposium. Can Spec Publ Fish Aquat Sci. 1989. p.110–27.
Kallasvuo M, Vanhatalo J, Veneranta L. Modeling the spatial distribution of larval fish abundance provides essential information for management. Can J Fish Aquat Sci. 2017; 74(5):636–49. https://doi.org/10.1139/cjfas-2016-0008
Lopes CA, Garcia V, Reynalte-Tataje DA, Zaniboni-Filho E, Nuñer APO. Temporal distribution of ichthyoplankton in the Forquilha river, upper Uruguay river – Brazil: relationship with environmental factors. Acta Sci Biol Sci. 2014; 36(1):59–65. http://doi.org/10.4025/actascibiolsci.v36i1.17993
MacFarland TW, Yates JM. Friedman Twoway Analysis of Variance (ANOVA) by ranks. In: MacFarland TW, Yates JM, editors. Introduction to Nonparametric Statistic for the Biological Sciences Using R. Cham: Springer; 2016. p.213–47. https://doi.org/10.1007/978-3-319-30634-6_7
Makrakis MC, Miranda LE, Makrakis S, Fontes Júnior HK, Morlis WG, Dias JHP, Garcia JO. Diversity in migratory patterns among Neotropical fishes in a highly regulated river basin. J Fish Biol. 2012; 81:866–81. http://dx.doi.org/10.1111/j.1095-8649.2012.03346.x
Mateus LAF, Penha JMF, Petrere Jr M. Fishing resources in the rio Cuiabá basin, Pantanal do Mato Grosso, Brazil. Neotrop Ichthyol. 2004; 2(4):217–27. https://doi.org/10.1590/s1679-62252004000400004
Memmott J, Waser NM, Price M. Tolerance of pollination networks to species extinctions. Proc R Soc Lond B. 2004; 271(1):2605–11. http://dx.doi.org/10.1098/rspb.2004.2909
Nakatani K, Agostinho AA, Baumgartner G, Bialetzki A, Sanches PV, Makrakis MC, Pavanelli CS. Ovos e larvas de peixes de água doce: desenvolvimento e manual de identificação. Maringá: EDUEM; 2001.
Nannini MA, Goodrich J, Dettmers JM, Soluk DA, Wahl DH. Larval and early juvenile fish dynamics in main channel and backwater lake habitats of the Illinois River ecosystem. Ecol Freshw Fish. 2012; 21(2):499–509. https://doi.org/10.1111/j.1600-0633.2012.00568.x
Nascimento FL, Araújo-Lima CARM. Descrição das larvas das principais espécies utilizadas pela pesca, no Pantanal. Corumbá: EMBRAPA Pantanal; 2000. Available from: https://ainfo.cnptia.embrapa.br/digital/bitstream/item/37433/1/BP19.pdf
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagener H. Vegan: community ecology package [Internet]. 2019. Available from: https://cran.r-project.org/web/packages/vegan/index.html
Oldani N. Identification and morphology of the larvae and juveniles of Thoracocharax stellatus (Kner, 1860) (Pisces, Gasteropelecidae). Rev Asoc Cienc Nat Litoral. 1979(a); 10:49–60.
Oldani N. Identification and morphology of the larvae and juveniles of Triportheus paranensis (Gunther, 1874) (Pisces, Characidae). Rev Asoc Cienc Nat Litoral. 1979(b); 10:61–71.
Oliveira FG, Bialetzki A, Gomes LC, Santin M, Taguti TL. Desenvolvimento larval de Brycon hilarii (Characiformes, Characidae). Iheringia, Ser Zool. 2012; 102(1):62–70. https://doi.org/10.1590/S0073-47212012000100009
Pelicice FM, Agostinho AA. Fish-passage facilities as ecological traps in large Neotropical rivers. Conserv Biol. 2008; 22(1):180–88. https://doi.org/10.1111/j.1523-1739.2007.00849.x
Pelicice FM, Pompeu PS, Agostinho AA. Large reservoirs as ecological barriers to downstream movements of Neotropical migratory fish. Fish Fish. 2015; 16:697–715. https://doi.org/10.1111/faf.12089
R Development Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria; 2020. Available from: https://www.R-project.org/
Resende EK. Considerações para definição de períodos de defeso de reprodução: o caso do Pantanal [Internet]. EMBRAPA Pantanal; 2003. Available from: https://www.infoteca.cnptia.embrapa.br/infoteca/bitstream/doc/812710/1/ADM039.pdf
Reynalte-Tataje DA, Nakatani K, Fernandes R, Agostinho AA, Bialetzki A. Temporal distribution of icthyoplankton in the Ivinhema River (Mato Grosso do Sul State/Brazil): Influence of environmental variables. Neotrop Ichthyol. 2011; 9(2):427–36. https://doi.org/10.1590/S1679-62252011005000017
Schulz C, Whitney BS, Rossetto OC, Neves DM, Crabb L, Oliveira EC, Lima PLT, Afzal M, Laing AF, Fernandes LCS, Silva CA, Steinke VA, Steinke ET, Saito CH. Physical, ecological and human dimensions of environmental change in Brazil’s Pantanal wetland: synthesis and research agenda. Sci Total Environ. 2019; 687:1011–27. https://doi.org/10.1016/j.scitotenv.2019.06.023
Silva A, Souza CA, Zani H, Freitas DR. Erosion evaluation in Paraguay river right bank downstream of the beach of Julião Cáceres city – MT. Rev Geo Ac. 2007; 1(1):5–19
Silva PS, Miranda LE, Makrakis S, Assumpção L, Dias JHP, Makrakis MC. Tributaries as biodiversity preserves: An ichthyoplankton perspective from the severely impounded Upper Paraná River. Aquatic Conserv: Mar Freshw Ecosyst. 2019; 29(2):258–69. https://doi.org/10.1002/aqc.3037
Souza CA, Leandro GRS, Sousa JB, Cunha SB, Garcia PHM. Sediments contribution of affluents of the right margin of the Paraguay river, superior Pantanal – Mato Grosso – Brazil. Cienc Geo. 2017; 21(1):18–31.
Suzuki FM, Dunham JB, Silva LGM, Alves CBM, Pompeu PS. Factors influencing movements of two migratory fishes within the tailrace of a large Neotropical dam and their implications for hydropower impacts. River Res Appl. 2016; 33(4):514–23. https://doi.org/10.1002/rra.3105
Suzuki HI, Agostinho AA, Bailly D, Gimenes MF, Júlio Jr HF, Gomes LC. Inter-annual variations in the abundance of young-of-the-year of migratory fishes in the upper Paraná River floodplain: relations with hydrographic attributes. Braz J Biol. 2009; 69(2):649–60. https://doi.org/10.1590/s1519-69842009000300019
Suzuki HI, Vazzoler AEAM, Marques EE, Lizama MAP, Inada P. Reproductive ecology of fish assemblages. In Thomaz SM, Agostinho AA, Hahn NS, editores. The upper Paraná River and its floodplain. Netherlands: Backhuys Publishers; 2004. p.271–92.
Taguti TL, Bialetzki A, Severi W, Agostinho AA, Assakawa LF. Early development of two tropical fishes (Perciformes: Sciaenidae) from the Pantanal of Mato Grosso, Brazil. Rev Biol Trop. 2015; 63(4):1105–18. https://doi.org/10.15517/RBT.V63I4.17265
Tanaka S. Stock assessment by means of ichthyoplankton surveys. FAO Fish Tech Paper. 1973; 122:33–51.
Teixeira GE, Bialetzki A, Soares BE, Souza G, Caramaschi EP. Variation in the structure of the ichthyoplankton community in the lower Paraíba do Sul River. Neotrop Ichthyol. 2019; 17(4):e180004. https://doi.org/10.1590/1982-0224-20180004
Tondato KK, Mateus LAF, Ziober SR. Spatial and temporal distribution of fish in marginal lagoons of Pantanal, Mato Grosso State, Brazil. Neotrop Ichthyol. 2010; 8(1):123–34. https://doi.org/10.1590/S1679-62252010005000002
Vazzoler AEAM. Biologia da reprodução de peixes teleósteos: teoria e prática. Maringá: EDUEM; 1996.
Wheeler B, Torchiano M. Permutation tests for linear models. R package version 2.1.0. [Internet]. 2016. Available from: https://github.com/mtorchiano/lmPerm
Winemiller KO, McIntyre PB, Castello L, Fluet-Chouinard E, Giarrizzo T, Nam S, Baird IG, Darwall W, Lujan NK, Harrison I, Stiassny MLJ, Silvano RAM, Fitzgerald DB, Pelicice FM, Agostinho AA, Gomes LC, Albert JS, Baran E, Petrere Jr M, Zarfl C, Mulligan M, Sullivan JP, Arantes CC, Sousa, LM, Koning AA, Hoeinghaus DJ, Sabaj M, Lundberg JG, Armbruster J, Thieme ML, Petry P, Zuanon J, Torrente Vilara G, Snoeks J, Ou C, Rainboth W, Pavanelli CS, Akama A, van Soesbergen A, Sáenz L. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Basin-scale planning in needed to minimize impacts in mega-diverse rivers. Science. 2016; 351(6269):128–29. https://doi.org/10.1126/science.aac7082
Zaniboni-Filho E, Ribolli J, Hermes-Silva S, Nuñer APO. Wide reproductive period of a long-distance migratory fish in a subtropical river, Brazil. Neotrop Ichthyol. 2017; 15(1):e160135. https://doi.org/10.1590/1982-0224-20160135
Ziober SR, Bialetzki A, Mateus LAF. Effect of abiotic variables on fish eggs and larvae distribution in headwater of Cuiabá River, Mato Grosso State, Brazil. Neotrop Ichthyol. 2012, 10(1):123–32. http://dx.doi.org/10.1590/S1679-62252012000100012
Authors
![]() Tatiane Pires de Sousa1
Tatiane Pires de Sousa1 ![]() ,
, ![]() Andréa Bialetzki2 and
Andréa Bialetzki2 and ![]() Lucia Aparecida de Fatima Mateus3
Lucia Aparecida de Fatima Mateus3
[1] Programa de Pós-Graduação em Ecologia e Conservação da Biodiversidade, Instituto de Biociências, Universidade Federal de Mato Grosso, Av. Fernando Corrêa da Costa 2367, 78060-900 Cuiabá, MT, Brazil. (TPS) tatipiressousa@gmail.com (corresponding author).
[2] Laboratório de Ictioplâncton, Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (NUPÉLIA), and Programa dePós-Graduação em Ecologia de Ambientes Aquáticos Continentais, Centro de Ciências Biológicas, Universidade Estadual deMaringá, Av. Colombo 5790, Bloco G-80, 87020-900 Maringá, PR, Brazil. (AB) bialetzki@nupelia.uem.br.
[3] Centro de Biodiversidade, Instituto de Biociências, Universidade Federal de Mato Grosso, Av. Fernando Corrêa da Costa 2367,78060-900 Cuiabá, MT, Brazil. (LAFM) lafmateus@gmail.com.
Authors’ Contribution 

Tatiane Pires de Sousa: Conceptualization, Formal analysis, Investigation, Writing-review and editing.
Andréa Bialetzki: Conceptualization, Investigation, Methodology, Resources, Supervision, Validation, Writing-review and editing.
Lucia Aparecida de Fatima Mateus: Conceptualization, Data curation, Formal analysis, Funding acquisition, Methodology, Project administration, Resources, Supervision, Validation, Writing-review and editing.
Ethical Statement
The collection of biological material was approved by the Instituto Chico Mendes de Conservação da Biodiversidade (license for collection of zoological material number 16313-1, ICMBio).
Competing Interests
The author declares no competing interests.
How to cite this article
Sousa TP, Bialetzki A, Mateus LAF. Dynamics of fish larvae recruitment in the hydrographic basin of the Paraguay River in western Brazil. Neotrop Ichthyol. 2023; 21(1):e220034. https://doi.org/10.1590/1982-0224-2022-0034
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2023 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted February 8, 2023 by Franco Teixeira de Mello
Accepted February 8, 2023 by Franco Teixeira de Mello
![]() Submitted April 17, 2022
Submitted April 17, 2022
![]() Epub April 03, 2023
Epub April 03, 2023