![]() Rayane G. Aguiar1,
Rayane G. Aguiar1, ![]() Erick C. Guimarães1,2,3
Erick C. Guimarães1,2,3 ![]() ,
, ![]() Pâmella S. de Brito1,3,
Pâmella S. de Brito1,3, ![]() Jadson P. Santos3,
Jadson P. Santos3, ![]() Axel M. Katz4,
Axel M. Katz4, ![]() Luiz Jorge B. da S. Dias5,
Luiz Jorge B. da S. Dias5, ![]() Luis Fernando Carvalho-Costa1 and
Luis Fernando Carvalho-Costa1 and ![]() Felipe P. Ottoni6
Felipe P. Ottoni6
PDF: EN XML: EN | Cite this article
Abstract
A new species of Knodus from the Mearim and Munim River basins, Northeastern Brazil, is herein described based on integrative taxonomy, by using different molecular based species delimitation methods and independent approaches. The new species possesses the combination of character states that usually diagnoses the genus. The new species possesses a similar colour pattern to K. victoriae, which is also morphologically similar to it. The species described herein differs from K. victoriae by possessing more total vertebrae, more branched anal-fin rays, and fewer circumpeduncular scales. We also provide a detailed discussion of the morphological diagnostic features exhibited by Knodus species from adjacent river basins.
Keywords: Cryptic species, Integrative Taxonomy, Stevardiinae.
Uma nova espécie de Knodus das bacias dos rios Mearim e Munim, Nordeste do Brasil, é descrita com base em taxonomia integrativa, utilizando diferentes métodos moleculares de delimitação de espécies e abordagens independentes. A nova espécie possui a combinação de estados de caráter que geralmente é utilizada para diagnosticar o gênero. A nova espécie possui um padrão de coloração semelhante a K. victoriae, que também é morfologicamente semelhante a ela. A espécie aqui descrita difere de K. victoriae por possuir mais vértebras totais, mais raios ramificados na nadadeira anal e menos escamas circumpedunculares. Nós também fornecemos uma discussão detalhada das características morfológicas diagnósticas exibidas por espécies de Knodus de bacias hidrográficas adjacentes.
Palavras-chave: Espécie críptica, Stevardiinae, Taxonomia Integrativa.
Introduction
Knodus Eigenmann, 1911 is one of the most species-rich characid genera within the subfamily Stevardiinae (Thomaz et al.,2015; Mirande, 2019; Ferreira et al.,2021), including 35 valid species (Menezes, Marinho, 2019; Sousa et al.,2020; Fricke et al., 2022). It is distributed among the Amazon, Tocantins-Araguaia, Orinoco, Paraná-Paraguay, Parnaíba, São Francisco, and Jequitinhonha river basins (Fricke et al.,2022), with its diversity peaking in the Amazon River basin (García-Melo et al.,2019; Fricke et al.,2022).
The genus Knodus is diagnosed only by the possession of two rows of pre-maxillary teeth – the inner row with four teeth – and a scaled caudal-fin (Eigenmann, 1918; Géry, 1972, 1977). This combination traditionally used to diagnose the genus has been questioned by some authors (e.g., Schultz, 1944; Taphorn, 1992; Román-Valencia, 2000; Román-Valencia et al., 2008) who consider Knodus a synonym of the closely related genus Bryconamericus Eigenmann, 1907.
Recent phylogenetic hypotheses based on molecular and morphological data have improved our knowledge on Knodus diversity and its intrageneric relationships (e.g., Thomaz et al.,2015; Mirande, 2019; García-Melo et al.,2019). However, although some species have been described within Knodus in recent years, the alpha taxonomy of most species and related genera is still somewhat confusing (García-Melo et al.,2019; Sousa et al., 2020).
Recent sampling efforts conducted in tributaries of the Mearim and Munin river basins (Northeast Brazil) revealed the existence of a new species morphologically similar to but still distinct from Knodus victoriae (Steindachner, 1907), whose type locality is in the upper Parnaiba River basin (Steindachner, 1907). Thus, we describe a new cryptic speciesof Knodussensu Bickford et al.(2007), from the Mearim and Munim river basins, based on an integrative taxonomic approach.
Material and methods
Taxon sampling, specimen collection, and preservation. Specimens were captured with a manual trail-net (2 m long × 1.8 m high; mesh size, 2 mm) and euthanized in a buffered solution of ethyl-3-amino-benzoat-methanesulfonate (MS–222) at a concentration of 250 mg/L until complete cessation of opercular movements, according to animal welfare laws and guidelines (Close et al.,1996; Leary et al.,2013). Specimens selected for morphological analysis were fixed in formalin for 10 days, after which they were preserved in 70% ethanol. Molecular data were obtained from specimens fixed and preserved in absolute ethanol. Specimens for morphological analysis are included in the lists of type and comparative material. Specimens for molecular approaches are listed in Tab. 1. Type material are deposited in the following ichthyological collections: Laboratório de Biologia e Genética de Peixes, Universidade Estadual Paulista Júlio de Mesquita Filho, Botucatu (LBP); Instituto de Biologia, Universidade Federal do Rio de Janeiro, Rio de Janeiro (UFRJ); Coleção Ictiologica do Centro de Ciências Agrárias e Ambientais da Universidade Federal do Maranhão, Chapadinha (CICCAA); and Coleção Ictiológica da Universidade Estadual do Maranhão, São Luís (CIUEMA).
Morphological analysis. Measurements and counts were made according to Fink, Weitzman (1974), with exception of the scale rows below the lateral line, which were counted to the insertion of the pelvic fin. Vertical scale rows between the dorsal-fin origin and lateral line do not include the scale of the median predorsal series situated just anterior to the first dorsal fin ray. Counts of supraneurals, vertebrae, procurrent caudal fin rays, unbranched dorsal and anal fin rays, branchiostegal rays, gill-rakers, premaxillary, maxillary, and dentary teeth were taken only from cleared and stained paratypes (C&S), prepared according to Taylor, Van Dyke (1985). The four modified vertebrae which constitute the Weberian apparatus were not included in the vertebral counts and the fused PU1 + U1 was considered a single element. Osteological nomenclature follows Weitzman (1962). Comparisons with other species of Knodus were based on examined material, as well as information from literature (Zarske, Géry, 2006; Ferreira, Lima, 2006; Zarske, 2007, 2008; Ferreira, Carvajal, 2007; Román-Valencia et al., 2008; Menezes et al.,2009, 2020; Ferreira, Netto-Ferreira, 2010; Esguícero, Castro, 2014;Menezes, Marinho, 2019; Sousa et al.,2020; Deprá et al.,2021).
DNA extraction, amplification, and sequencing. DNA was extracted from fin clips using a saline buffer extraction protocol (Aljanabi, Martinez, 1997). Fragments of the mitochondrial gene cytochrome c oxidase subunit 1 (COI) were amplified with the universal primers designed by Ward et al.(2005) (FISHF1 5´-TCAACCAACCACAAAGACATTGGCAC-3´ and FISHR1 5´-TAGACTTCTGGGTGGCCAAAGAATCA-3´), primers from Melo et al. (2011) (COI L6252-Asn 5’-AAGGCG GGGAAAGCCCCGGCA G-3’ and H7271-COXI 5’-TCC TATGTAGCCGAATGGTTC TTT T-3’) and primers developed in the present study (KNODUS-TOF 5´-GGGCGATGACCAAATCTA-3´ and KNODUS-TOR 5´-AGGGTCGAAGAATGAGGTAT-3’). Polymerase chain reactions (PCR) for samples from Knodus sp. “Mearim”, Knodus cf. savannensis, Knodus sp. “Maracaçumé”, and Knodus cf. victoriae comprised a total volume of 15 µL containing 1x polymerase buffer, 1.5 mM MgCl2, 200 µM dNTP, 0.2 µM of each primer, 1U Taq polymerase (Invitrogen), 100 ηg DNA template, and ultrapure water. The PCR cycles were as follows: 2 min at 94ºC, followed by 35 cycles of 94ºC for 30s, 54ºC for 30s and 72ºC for 1 min and 10 min at 72ºC. Polymerase chain reactions (PCR) for samples from the Knodus sp. “Munim”, and Knodus sp. “Itapecuru” comprised a total volume of 15 µl containing 1x Polymerase buffer, 400 µM dNTP, 0,4 uM of each primer, 1U Taq Polymerase (Invitrogen), 100 ηg DNA template, and ultrapure water. The PCR cycles were as follows: 5 min at 94ºC, followed by 35 cycles of 94ºC for 45s, 48°-52ºC for 45s, and 72ºC for 1 min, and 10 min at 72ºC. Amplicons were purified using ExoSAP-IT PCR Product Clean-up (Thermo Fisher Scientific) and Gel Purification Kit (GE Healthcare Systems), and sequenced using forward and reverse primers and the BigDye Terminator 3.1 Cycle Sequencing kit in a ABI 3730 DNA Analyzer (Thermo Fisher Scientific).
Data partitioning, evolutionary models, and alignment. The dataset included the COI sequences (401 base pairs, bp) of individuals from 10 Knodus species, including the species here described, specimens that we were not able to identify at the species level, as well as two Bryconamericus species. Many sequences were newly generated for this project, while others originated from the Barcode of Life Database (BOLD) and the National Center for Biotechnology Information (NCBI) (Tab. 1). Sequences were aligned using ClustalW (Chenna et al., 2003) and translated into amino acid residues using the program MEGA 7 (Kumar et al.,2016) to test if the sequences came from NUMTs (nuclear mitochondrial DNA sequences), in which case premature stop codons or indels would be expected. The best-fit evolutionary model (GTR+I+G) was selected using the Akaike Information Criterion (AIC) and the Corrected Akaike Information Criterion (AICc) by jModelTest 2.1.7 (Darriba et al.,2012), and used in all analyses, except for ABGD (Automatic Barcode Gap Discovery) which is a model-free approach based only on genetic distances.
Phylogenetic analysis. A Bayesian inference phylogenetic (BI) tree was estimated in MrBayes 3.2 (Ronquist et al., 2012) to reconstruct the evolutionary relationships among terminals using the General Time Reversible (GTR+I+G) evolutionary model. The BI analysis was conducted with the following parameters: two independent Markov chain Monte Carlo (MCMC) runs of two chains each for 10 million generations, with a tree sampling frequency at every 1,000 generations. The convergence of the MCMC chains and the proper burn-in value were assessed by evaluating the stationary phase of the chains using Tracer v. 1.6 (Rambaut et al., 2014). The final consensus tree and its posterior probabilities were generated with the remaining tree samples after removing the first 25% samples (burn-in). We used as outgroups sequences of Bryconamericus exodon Eigenmann, 1907 and B. iheringii (Boulenger, 1887). The remaining haplotypes were used as ingroups.
Species delimitation and molecular diagnoses. We implemented five distinct and independent single locus species delimitation methods based on molecular data, each of which rely on different operational criteria for species delimitation. DBC, DNA barcoding (hereafter Traditional DNA barcoding) was initially proposed by Hebert et al. (2003a,b). Since then, it has improved and gained supporters due to its practicality and efficiency (e.g., Hajibabaei et al., 2007; Coissac et al., 2016; DeSalle, Goldstein, 2019). The premise of the method consists of standardized sequencing of a specific gene for each species, followed by the organization of the sequences in virtual reference libraries. Once a species is added to the library, any individual of that species, at any ontogenetic stage, or even fragments, can be identified by simple comparison, simply by sequencing the standard gene (Hebert et al., 2003a,b). For fish and other animals, the gene used is the mitochondrial protein cytochrome c oxidase I (COI) and its effectiveness has been frequently demonstrated (e.g., Costa-Silva et al., 2018; García-Melo et al., 2019). The methodology suggests that the maximum genetic distance between individuals of the same species, based on the COI sequences, can be defined for each taxonomic group. Any difference higher than this cut-off value would represent discontinuity between species (Hebert et al., 2003a,b). The other approaches were CBB, a character-based DNA barcoding method (DeSalle et al., 2005) adapted by Ottoni et al. (2019) and Guimarães et al. (2020b); GMYC, the General Mixed Yule Coalescent method, single-threshold version (Fujisawa, Barraclough, 2013); bPTP, the Bayesian implementation of the Poisson tree processes (Zhang et al.,2013); and ABGD, Automatic Barcode Gap Discovery (Puillandre et al., 2012).
Traditional DNA barcoding (DBC). We used the Kimura-2-parameter model (K2P) (Kimura, 1980) to estimate the pairwise genetic distances between species in MEGA 7 software (Kumar et al., 2016). We considered a cutoff of 2% as sufficient to discriminate species, since this threshold is commonly inferred by species delimitations among freshwater Neotropical fish species based on COI (Jacobina et al., 2018).
Molecular diagnosis (CBB). The molecular diagnosis approach delimited the new species by the presence of a unique combination of nucleotides at particular sites. In addition, the new species was diagnosed by nucleotide substitutions following Costa et al. (2014), Ottoni et al. (2019), and Guimarães et al. (2020b). Nucleotide substitutions among lineages were optimized on the Bayesian inference topology using PAUP version 4 (Swofford, 2002). Each nucleotide substitution is represented by its relative numeric position determined through sequence alignment with the complete mitochondrial COI gene of Psalidodon paranae (Eigenmann, 1914)(KX609386.1:5503-7062), followed by the specific nucleotide substitution in parentheses. Unique nucleotide substitutions in our analysis are marked with asterisk.
General Mixed Yule Coalescent (GMYC). The GMYC is a single locus coalescent-based species delimitation approach that relies on branch lengths to establish a threshold between speciation and coalescent processes (Fujisawa, Barraclough, 2013). Here we applied the single-threshold version of the method, which usually outperforms the multiple-threshold version (Fujisawa, Barraclough, 2013). A new reduced dataset was created for this analysis using DAMBE5 (Xia, 2013), including only unique haplotypes following the requirements of this method.
The ultrametric phylogenetic tree needed for input was inferred in BEAST version 1.8.4 (Drummond et al., 2012), with the following parameters: an uncorrelated relaxed clock with lognormal distribution, a Yule Process as tree prior with 10 million generations and sampling frequency of 1,000. We used as outgroups sequences of Bryconamericus exodon and B. iheringii. The remaining haplotypes were used as ingroups. The GMYC analysis was performed on the Exelixis Lab’s server https://species.h-its.org/gmyc.
Bayesian implementation of the Poisson tree processes (bPTP). The bPTP is another single locus coalescent-based species delimitation method that differs from other similar approaches, such as GMYC, by not requiring an ultrametric tree and thus not relying on branch lengths to delimit species (Zhang et al., 2013). The method assumes that more molecular variability (number of nucleotide substitutions) is expected between haplotypes from different species than within a species (Zhang et al., 2013), establishing a threshold between speciation and coalescent processes. The reduced dataset for performing the bPTP was the same at that used in GMYC, following the requirements of this method. The input phylogenetic tree was estimated in software Mrbayes 3.2 (Ronquist et al., 2012) to reconstruct the evolutionary relationships among terminals using the General Time Reversible (GTR+I+G) evolutionary model. The BI analysis was conducted with the following parameters: two independent Markov chain Monte Carlo (MCMC) runs of two chains each for 10 million generations, with a tree sampling frequency at every 1,000 generations. The convergence of the MCMC chains and the proper burn-in value were assessed by evaluating the stationary phase of the chains using Tracer version 1.6 (Rambaut et al., 2014). The final consensus tree and its posterior probabilities were generated with the remaining tree samples after removing the first 25% samples (burn-in). The remaining haplotypes were used as ingroups. The bPTP analysis was performed on the Exelixis Lab’s web server http://species.h-its.org/ptp/, following the default parameters except for designation of a 20% burn-in. Bryconamericus exodon was chosen as the outgroup.
Automatic Barcode Gap Discovery (ABGD). The ABGD is a barcode species delimitation method that aims to establish a minimum gap that probably corresponds to the threshold between interspecific and intraspecific processes (Puillandre et al., 2012). The major advantage of ABGD when compared to the other barcode species delimitation methods is that the inference of the limit between interspecific and intraspecific processes (gap detection) is recursively applied to previously obtained groups to get finer partitions until there is no further partitioning, allowing a more refined search. Basically, the ABGD analysis indicates the number of groups (species) estimated relative to a large spectrum of p values (prior intraspecific values). A value of 0.1 indicates maximum intraspecific variability with all sequences belonging to a single species, whereas a 0.001 value indicates a small intraspecific variability with each distinct haplotype representing a different species. The reduced dataset for performing the ABGD was the same as that used in GMYC, following the requirements of this method. We ran ABGD on the ABGD server website https://bioinfo.mnhn.fr/abi/public/abgd/abgdweb.html with default parameters, except for the X value (we used an X value of 1.0 as recommended by the server for our analysis). After running the ABGD, additional molecular, morphological, or ecological characters are needed to infer the correct number of species if the analysis will follow an integrative taxonomy paradigm.
Results
Knodus guajajara Aguiar, Brito, Ottoni & Guimarães, new species
urn:lsid:zoobank.org:act:4EEDDC6E-4088-4B60-828D-AF79EF84DC26
(Figs. 1–3; Tabs. 2–3)
Knodus victoriae [non Knodus victoriae (Steindachner, 1907)]. ―Guimarães et al.(2020a:90).
―Oliveira et al. (2020:6–7).
FIGURE 1| Knodus guajajara, holotype, CICCAA 4883, 31.4 mm SL, Alto Alegre do Pindaré municipality, Igarapé Arapapá, Pindaré River drainage, Mearim River basin.
Holotype. CICCAA 4883, 31.4 mm SL, Alto Alegre do Pindaré municipality, Igarapé Arapapá, Pindaré River drainage, Mearim River basin, 03°42’26”S 46°00’25”W, Nov 2016. E. C. Guimarães & P. S. Brito.
FIGURE 2| Knodus guajajara, CICCAA 4861, paratype, 31.9 mm SL, jaw suspensorium. A. Premaxillary. B. Maxilla. C. Dentary. Scale bar = 1 mm.
TABLE 1 | Specimens and DNA sequence information included in the study. Sequences made available by this study follow with asteristic.
Nº | Species | Depositories of vouchers | Genbank ID | ||
Collection | Tissue code | Voucher | |||
1 | Bryconamericus
exodon | LBP | 26191 | 5118 | MH002960 |
2 | Bryconamericus
iheringii | LBP | 60645 | 14473 | MH002991 |
3 | Knodus
alpha | STRI | 9564 | 531 | MH003217 |
4 | Knodus
alpha | LBP | 9561 | 663 | MH003216 |
5 | Knodus
borki | LBP | 53820 | 12472 | MH003218 |
6 | Knodus
borki | LBP | – | 53821 | KT248111 |
7 | Knodus
caquetae | MPUJ | 637 | 11073A | MH003221 |
8 | Knodus
caquetae | MPUJ | 639 | 11073B | MH003222 |
9 | Knodus
heteresthes | LBP | 57371 | 13870 | MH003232 |
10 | Knodus
heteresthes | LBP | 37317 | 7959 | MH003233 |
11 | Knodus
megalops | LBP | 54223 | 12566B | MH003234 |
12 | Knodus meridae | LBP | 15818 | 7569 | MH003235 |
13 | Knodus
tiquiensis | LBP | – | 33217 | KT248096 |
14 | Knodus
tiquiensis | LBP | – | 33218 | KT248097 |
15 | Knodus
tiquiensis | LBP | – | 33216 | KT248098 |
16 | Knodus
victoriae | LBP | – | 27366 | KT248128 |
17 | Knodus
victoriae | LBP | 27370 | KT248129 | |
18 | Knodus
victoriae | LBP | 27342 | 5607 | KT248130 |
19 | Knodus
victoriae | LBP | – | 27254 | KT248131 |
20 | Knodus
victoriae | LBP | – | 27368 | KT248132 |
21 | Knodus
victoriae | LBP | – | 27295 | KT248133 |
22 | Knodus
victoriae | LBP | 27369 | KT248134 | |
23 | Knodus
victoriae | LBP | – | 27253 | KT248135 |
24 | Knodus
victoriae | LBP | – | 27255 | KT248136 |
25 | Knodus
victoriae | LBP | – | 27367 | KT248137 |
26 | Knodus aff. victoriae (Balsas)* | CICCAA | 4817.1 | 4817 | MW556675 |
27 | Knodus aff. victoriae (Balsas)* | CICCAA | 4817.2 | 4817 | MW556676 |
28 | Knodus aff. victoriae (Balsas)* | CICCAA | 4817.3 | 4817 | MW556677 |
29 | Knodus aff. victoriae (Balsas)* | CICCAA | 4818.1 | 4818 | MW556678 |
30 | Knodus aff. victoriae (Balsas)* | CICCAA | 4818.2 | 4818 | MW556679 |
31 | Knodus aff. victoriae (Balsas)* | CICCAA | 4818.3 | 4818 | MW556680 |
32 | Knodus aff. victoriae (Itapecuru)* | CICCAA | 2064.1 | 2064 | MW556681 |
33 | Knodus aff. victoriae (Itapecuru)* | CICCAA | 2064.2 | 2064 | MW556682 |
34 | Knodus cf. savannensis | LBP | – | 66360 | KT248199 |
35 | Knodus cf. savannensis | LBP | – | 66339 | KT248200 |
36 | Knodus cf. savannensis | LBP | – | 66340 | KT248201 |
37 | Knodus cf. savannensis | LBP | – | 66356 | KT248202 |
38 | Knodus cf. savannensis | LBP | – | 66362 | KT248203 |
39 | Knodus cf. savannensis | LBP | – | 66363 | KT248204 |
40 | Knodus cf. savannensis | LBP | – | 66363 | KT248218 |
41 | Knodus cf. savannensis | LBP | – | 62500 | KT248219 |
42 | Knodus cf. savannensis (Carolina-MA,
Tocantins River basin)* | CICCAA | 3609.1 | 3609 | MW556695 |
43 | Knodus sp. “Guamá” | LBP | 43070 | 9141 | KT248223 |
44 | Knodus sp. “Guamá” | LBP | – | 43718 | KT248225 |
45 | Knodus sp. “Guamá” | LBP | – | 43067 | KT248226 |
46 | Knodus sp. “Guamá” | LBP | – | 43068 | KT248227 |
47 | Knodus sp. “Tapajós” | LBP | 57048 | 13750 | KT248123 |
48 | Knodus sp. “Tapajós” | LBP | – | 57049 | KT248124 |
49 | Knodus sp. “Xingu” | LHGP | – | 66314 | KT248184 |
50 | Knodus sp. “Xingu” | LHGP | – | 66315 | KT248185 |
51 | Knodus sp. (Marabá)* | CICCAA | 2087.1 | 2087 | MW556692 |
52 | Knodus sp. (Marabá)* | CICCAA | 2087.2 | 2087 | MW556693 |
53 | Knodus sp. (Marabá)* | CICCAA | 2087.3 | 2087 | MW556694 |
54 | Knodus sp. (Maracaçumé)* | CICCAA | 2390.1 | 2390 | MW556683 |
55 | Knodus sp. (Maracaçumé)* | CICCAA | 2390.2 | 2390 | MW556684 |
56 | Knodus sp. (Maracaçumé)* | CICCAA | 2390.5 | 2390 | MW556685 |
57 | Knodus
guajajara* | CICCAA | 2052.2 | 2052 | MW556688 |
58 | Knodus
guajajara* | CICCAA | 2052.3 | 2052 | MW556689 |
59 | Knodus
guajajara* | CICCAA | 2052.4 | 2052 | MW556690 |
60 | Knodus
guajajara* | CICCAA | 2052.5 | 2052 | MW556691 |
61 | Knodus
guajajara* | CICCAA | 2391.1 | 2391 | MW556686 |
62 | Knodus
guajajara* | CICCAA | 2391.2 | 2391 | MW556687 |
Paratypes. All from Brazil, Maranhão State: CICCAA 1535, 1, 29.2 mm SL, collected with holotype. CICCAA 1585, 3, 28.0–34.7 mm SL, Alto Alegre do Pindaré municipality, igarapé Arapapá, Pindaré River drainage, Mearim River basin, 03°42’26”S 46°00’25”W, Nov 2015, E. C. Guimarães & P. S. Brito. CICCAA 4860, 2 C&S, 29.9–31.6 mm SL, Alto Alegre do Pindaré municipality, igarapé Arapapá, Pindaré River drainage, Mearim River basin, 03°42’26”S 46°00’25”W, Nov 2015, E. C. Guimarães & P. S. Brito. LBP 31041, 16, 24.5–31.4 mm SL, Alto Alegre do Pindaré municipality, Igarapé Igarapá, Pindaré River drainage, Mearim River basin, 03°45’51”S 46°08’15”W, Nov 2015, E. C. Guimarães & P. S. Brito. CICCAA 4858, 5 C&S, 22.9–27.0 mm SL, Alto Alegre do Pindaré municipality, Igarapé Igarapá, Pindaré River drainage, Mearim River basin, 03°45’51”S 46° 08’15”W, Nov 2015, E. C. Guimarães & P. S. Brito. CICCAA 1517, 2, 21.8–24.4 mm SL, Alto Alegre do Pindaré municipality, igarapé Caititu, Pindaré River drainage, Mearim River basin, 03°42’30”S 46°01’19”W, Jul 2017, E. C. Guimarães & P. S. Brito. CICCAA 4859, 2 C&S, 23.5–27.6 mm SL, Alto Alegre do Pindaré municipality, igarapé Caititu, Pindaré River drainage, Mearim River basin, 03°42’30”S 46°01’19”W, Jul 2017, E. C. Guimarães & P. S. Brito. CICCAA 1536, 2, 28.8–29.8 mm SL, Buriticupu municipality, Buritizinho River, Pindaré River drainage, Mearim River basin, 04°11’53”S 46°28’41”W, Nov 2016, E. C. Guimarães & P. S. Brito. CICCAA 4861, 1 C&S, 31.9 mm SL, Buriticupu municipality, Buritizinho River, Pindaré River drainage, Mearim River basin, 04°11’53”S 46°28’41”W, Nov 2016, E. C. Guimarães & P. S. Brito. CICCAA 1227, 1, 29.6 mm SL, Buriticupu municipality, Buritizinho River, Pindaré River drainage, Mearim River basin, 04°19’45”S 46°29’46”W, 27 Jan 2017, E. C. Guimarães & P. S. Brito. UFRJ 7023, 10, 22.8–29.3 mm SL, Alto Alegre do Pindaré municipality, igarapé Jenipapo, Pindaré River drainage, Mearim River basin, 03°51’20”S 46°11’09”W, Jul 2017, E. C. Guimarães & P. S. Brito. CICCAA 4862, 5 C&S, 23.9–29.3 mm SL, Brazil, Maranhão State, Alto Alegre do Pindaré municipality, igarapé Jenipapo, Pindaré River drainage, Mearim River basin, 03°51’20”S 46°11’09”W, Jul 2017, E. C. Guimarães & P. S. Brito. CICCAA 1321, 39, 19.4–29.2 mm SL, Miranda do Norte municipality, Mearim River basin, 04°19’45”S 46°29’46”W, Nov 2016, E. C. Guimarães & P. S. Brito. CICCAA 4863, 11 C&S, 19.7–24.4 mm SL, Miranda do Norte municipality, Mearim River basin, 04°19’45”S 46°29’46”W, Nov 2016, E. C. Guimarães & P. S. Brito. CIUEMA 1021, 8, 21.2–26.9 mm SL, Alto Alegre do Pindaré municipality, Igarapé Timbira, Pindaré River drainage, Mearim River basin, 03°32’57”S 44°39’38”W, Nov 2015, E. C. Guimarães & P. S. Brito. CICCAA 3423, 42, 19.5–30.21 mm SL, CICCAA 3424, 57, 22.35–26.9 mm SL,, Chapadinha municipality, Riacho da Raiz, RESEX Chapada Limpa, Munim River basin, 03°53’45”S 43°29’21”W, 11 Aug 2019, J. Reis, L. Oliveira, F. Ottoni, R. Fernandes & A. Silva. Chapadinha municipality, Bandeira River, Povoado Mata do Jeroca, RESEX Chapada Limpa, Munim River basin, 03°59’40”S 43°29’24”W, 11 Aug 2019, J. Reis, L. Oliveira, F. Ottoni, R. Fernandes & A. Silva. CICCAA 3425, 22, 20.22–26.71 mm SL, CICCAA 3426, 6, 28.87–32.6 mm SL, Chapadinha municipality, stream at Bairro Aldeia, Munim River basin, 03°44’53”S 43°21’32”W, 28 Jan 2019, F. Carvalho, H. Silva, L. Oliveira & I. Gôuvea. CICCAA 3467, 2, 21.22–21.43 mm SL, Anapurus municipality, stream on the road at Povoado de Paços, Munim River basin, 03°33’44”S 43°03’52”W, Sep 2019, D. Campos, J. Reis & F. Ottoni. CICCAA 4808, 3, 25.19–34.2 mm SL, CICCAA 4822, 1, 36.89 mm SL, Chapadinha municipality, stream at balneário Repouso do Guerreiro, Bairro Independência, Munim River basin, 03°44’57”S 43°20’26”W, Nov 2019, B. Furtado, M. Paiva, A. Bezerra, M. Coelho & I. Gouvêa. CICCAA 4490, 5, 24.73–29.11 mm SL, Chapadinha municipality, Riacho Fundo, Munim River basin, 03°42’20”S 43°31’46”W, 23 Sep 2018, L. Sousa, L. Oliveira & I. Gouvêa, CICCAA 4491, 3 C&S 23.26–28.19 mm SL, Chapadinha municipality, Riacho Fundo, Munim River basin, 03°42’20”S 43°31’46”W, 23 Sep 2018, L. Sousa, L. Oliveira & I. Gôuvea.
FIGURE 3| Knodus guajajara, CICCAA 4861, paratype, 31.9 mm SL, jaw suspensorium. A. Premaxillary. B. Maxilla. C. Dentary. Scale bar = 1 mm.
TABLE 2 | Morphometric data (N = 110) of the holotype and paratypes of Knodus guajajara from the Mearim River basin. SD = Standard deviation.
Holotype | Paratypes | Mean | SD | |
Standard length | 31.4 | 19.4‒34.7 | 25.3 | ‒ |
Percents of standard length | ||||
Depth at dorsal-fin origin (body depth) | 29.8 | 20.1‒30.2 | 24.7 | 3.3 |
Snout to dorsal-fin origin | 55.3 | 47.8‒57.1 | 52.2 | 5.5 |
Snout to pectoral-fin origin | 27.1 | 21.7‒29.32 | 24.9 | 3.0 |
Snout to pelvic-fin origin | 45.7 | 37.6‒49.3 | 42.2 | 4.9 |
Snout to anal-fin origin | 60.2 | 44.0‒61.6 | 53.9 | 6.1 |
Caudal peduncle depth | 10.5 | 7.6‒11.7 | 9.3 | 1.2 |
Caudal peduncle length | 15.3 | 7.1‒15.2 | 11.1 | 1.9 |
Pectoral-fin length | 20.5 | 14.8‒23.8 | 18.7 | 2.6 |
Pelvic-fin length | 13.5 | 9.8‒15.4 | 12.6 | 1.7 |
Dorsal-fin base length | 9.1 | 8.7‒15.2 | 11.5 | 1.7 |
Dorsal-fin height | 26.9 | 17.7‒27.0 | 21.3 | 2.8 |
Anal-fin base length | 29.4 | 25.5‒36.3 | 30.5 | 3.8 |
Anal-fin lobe length | 13.0 | 9.8‒20.3 | 15.3 | 2.4 |
Eye to dorsal-fin origin | 42.5 | 33.6‒43.8 | 38.9 | 4.5 |
Dorsal-fin origin to caudal-fin base | 52.5 | 39.4‒56.7 | 46.9 | 5.8 |
Percents of head length | ||||
Head length | 23.9 | 21.3‒26.5 | 23.3 | 2.5 |
Horizontal eye diameter | 41.3 | 32.6‒45.6 | 39.5 | 4.8 |
Snout length | 24.9 | 15.8‒29.6 | 21.8 | 3.3 |
Least interorbital width | 35.1 | 23.7‒36.8 | 32.3 | 4.0 |
Upper jaw length | 47.5 | 31.5‒48.8 | 38.3 | 5.3 |
Morphological diagnosis. Knodus guajajara differs from K. borki Zarske, 2008 and K. delta Géry, 1972 by having a complete lateral line (vs. incomplete lateral line) and from K. cupariensis de Sousa, Silva-Oliveira, Canto & Ribeiro, 2020 and K. geryi Lima, Britski & Machado, 2004 by having caudal fin lobes with sparse chromatophores and lacking basal blotches (vs. a dark basal blotch on each caudal fin lobe, Sousa et al., 2020; fig. 1); from Knodus borki, K. diaphanus (Cope, 1878), K. victoriae, K. heteresthes (Eigenmann, 1908), K. deuterodonoides (Eigenmann, 1914), K. longus Zarske & Géry, 2006, K. septentrionalis Géry, 1972, K. figueiredoi Esguícero & Castro, 2014, K. geryi, K. meridae Eigenmann, 1911, K. nuptialis Menezes & Marinho, 2019, K. orteguasae (Fowler, 1943), K. tiquiensis Ferreira & Lima, 2006, K. angustus Menezes, Ferreira & Netto-Ferreira, 2020, K. rufford Deprá, Ota, Vitorino-Júnior & Ferreira, 2021 and K. obolus Deprá, Ota, Vitorino-Júnior & Ferreira, 2021 by having 20–25 branched rays in the anal-fin (mode 23) (vs. 12–19, combined); and from K. tiquiensis by having a single humeral spot (vs. two). Knodus guajajara is distinguished from K. breviceps (Eigenmann, 1908) and K. savannensis Géry, 1961 by having a conspicuous round humeral blotch (vs. inconspicuous and vertically elongate); from K. dorsomaculatus Ferreira & Netto-Ferreira, 2010 by having a hyaline dorsal-fin (vs. dark blotch on the base of the first five branched dorsal fin rays); from K. alpha (Eigenmann, 1914), K. chapadae (Fowler, 1906), K. geryi, K. hypopterus (Fowler, 1943), K. mizquae (Fowler, 1943) and K. shinahota Ferreira & Carvajal, 2007 by having 4 or 5 rows of scales between the lateral line and the dorsal-fin origin (vs. 6 rows of scales); from K. cinarucoensis (Román-Valencia et al., 2008), K. gamma Géry, 1972 and K. longus Zarske & Géry, 2006 by having 12 or 13 scales in the median series between the tip of the supraoccipital spine and the dorsal-fin origin (vs. 10 or 11 rows of scales in K. gamma and 17 to 18 in K. longus); from K. jacunda (Fowler, 1913), K. moenkhausii (Eigenmann & Kennedy, 1903), Knodus cismontanus (Eigenmann, 1914), Knodus caquetae Fowler, 1945, K. tanaothoros (Weitzman, Menezes, Evers & Burns, 2005), K. weitzmani (Menezes, Netto-Ferreira & Ferreira, 2009) and by having 3 to 5 maxillary teeth (vs. absence in K. jacunda,one in K. caquetae, and 2 in K. moenkhausii, K. tanaothoros, K. weitzmani and K. cismontanus); from K. megalops Myers, 1929 by having 3 or 4 tricuspid teeth in the premaxillary outer row (vs. 5); from K. jacunda by having 3 to 5 maxillary teeth (vs. absence); from K. smithi (Fowler, 1913) by having 3 to 5 cusps on the teeth of the inner row of the premaxilla (vs. 7); and from K. figueredoi, K. heteresthes, K. meridae, K. mizquae, K. moenkhausii, K. victoriae, K. pasco Zarske, 2007 by having 12 circumpeduncular scales (vs. 13–14 combined). Furthermore, Knodus guajajara differs from K. victoriae by having more total vertebrae 33–35, mode 34 (vs. 30–33, mode 32).
FIGURE 4| Knodus guajajara, CICCAA 4861, paratype, male, 31.9 mm SL Maranhão, Mearim River basin. A. Hooks on pelvic fin. B. Hooks on anal fin. (Photographed by F. P. Ottoni).
TABLE 3 | Morphometric data (N = 136) for the paratypes of Knodus guajajara from the Munim River basin. SD = Standard deviation.
Range | Mean | SD | |
Standard length | 19.5‒37.7 | 25.3 | ‒ |
Percents of standard length | |||
Depth at dorsal-fin origin (body depth) | 20.4‒37.2 | 24.7 | 2.06 |
Snout to dorsal-fin origin | 47.4‒53.4 | 50.3 | 1.13 |
Snout to pectoral-fin origin | 21.9‒27.1 | 24.4 | 0.79 |
Snout to pelvic-fin origin | 39.4‒45.7 | 42.5 | 1.24 |
Snout to anal-fin origin | 53.1‒59.8 | 56.2 | 1.38 |
Caudal peduncle depth | 8.3‒10.4 | 9.3 | 0.41 |
Caudal peduncle length | 9.2‒12.8 | 10.4 | 0.75 |
Pectoral-fin length | 18.5‒23.4 | 20.9 | 0.74 |
Pelvic-fin length | 12.0‒15.7 | 13.8 | 0.68 |
Dorsal-fin base length | 8.4‒12.6 | 10.1 | 0.76 |
Dorsal-fin height | 20.1‒24.6 | 22.1 | 0.86 |
Anal-fin base length | 22.8‒28.7 | 26.4 | 1.17 |
Anal-fin lobe length | 16.1‒20.2 | 18.1 | 0.82 |
Eye to dorsal-fin origin | 33.1‒39.2 | 36.1 | 1.12 |
Dorsal-fin origin to caudal-fin base | 47.4‒54.3 | 49.5 | 1.14 |
Percents of head length | |||
Head length | 21.5‒25.7 | 23.4 | 0.82 |
Horizontal eye diameter | 33.8‒42.6 | 37.4 | 1.56 |
Snout length | 20.1‒25.9 | 22.5 | 1.34 |
Least interorbital width | 27.1‒35.3 | 31.7 | 1.77 |
Upper jaw length | 34.5‒41.2 | 37.9 | 1.47 |
Description. Morphometric data presented in Tabs. 2–3. Body comparatively small, with largest specimen examined measuring 37.7 mm SL. Greatest body depth at dorsal-fin origin. Dorsal profile of head convex from upper lip to vertical through middle portion of eye; slightly concave from this point to tip of supraoccipital spine; straight to slightly convex from posterior tip of supraoccipital spine to dorsal-fin origin; dorsal-fin base straight; slightly convex to straight from end of dorsal-fin base to adipose fin and concave from latter point to anterior dorsal-procurrent ray. Ventral profile of body convex from lower lip to anal-fin origin; straight, posterodorsally inclined along anal-fin base. Dorsal and ventral profile of caudal peduncle slightly concave.
TABLE 4 | Kimura-2-parameters pairwise genetic distances among species.
Species | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |
1 | Bryconamericus exodon | ||||||||||||||||||
2 | Bryconamericus iheringii | 0.12 | |||||||||||||||||
3 | Knodus heteresthes | 0.15 | 0.16 | ||||||||||||||||
4 | Knodus cf. savanensis (Clade 1) | 0.13 | 0.17 | 0.09 | |||||||||||||||
5 | Knodus cf. savanensis (Clade 2) | 0.13 | 0.16 | 0.08 | 0.06 | ||||||||||||||
6 | Knodus sp. “Tapajós” | 0.13 | 0.18 | 0.08 | 0.10 | 0.09 | |||||||||||||
7 | Knodus sp. “Xingu” | 0.15 | 0.16 | 0.05 | 0.09 | 0.10 | 0.09 | ||||||||||||
8 | Knodus tiquiensis | 0.14 | 0.18 | 0.13 | 0.11 | 0.11 | 0.12 | 0.14 | |||||||||||
9 | Knodus borki | 0.11 | 0.15 | 0.12 | 0.12 | 0.11 | 0.11 | 0.12 | 0.15 | ||||||||||
10 | Knodus megalops | 0.13 | 0.14 | 0.14 | 0.14 | 0.14 | 0.12 | 0.13 | 0.15 | 0.08 | |||||||||
11 | Knodus caquetae | 0.11 | 0.14 | 0.14 | 0.14 | 0.13 | 0.12 | 0.14 | 0.15 | 0.07 | 0.04 | ||||||||
12 | Knodus meridae | 0.16 | 0.16 | 0.12 | 0.11 | 0.10 | 0.12 | 0.13 | 0.15 | 0.11 | 0.13 | 0.13 | |||||||
13 | Knodus alpha | 0.14 | 0.18 | 0.10 | 0.06 | 0.09 | 0.13 | 0.09 | 0.15 | 0.14 | 0.14 | 0.15 | 0.15 | ||||||
14 | Knodus sp. “Marabá” | 0.14 | 0.16 | 0.16 | 0.13 | 0.12 | 0.14 | 0.15 | 0.14 | 0.15 | 0.15 | 0.15 | 0.17 | 0.15 | |||||
15 | Knodus sp. “Maracaçumé” | 0.12 | 0.14 | 0.12 | 0.13 | 0.11 | 0.12 | 0.13 | 0.11 | 0.11 | 0.12 | 0.10 | 0.12 | 0.15 | 0.14 | ||||
16 | Knodus victoriae | 0.15 | 0.18 | 0.07 | 0.08 | 0.08 | 0.10 | 0.08 | 0.13 | 0.11 | 0.15 | 0.14 | 0.12 | 0.10 | 0.16 | 0.12 | |||
17 | Knodus aff. victoriae (Balsas) | 0.13 | 0.19 | 0.09 | 0.09 | 0.09 | 0.10 | 0.10 | 0.13 | 0.12 | 0.13 | 0.15 | 0.10 | 0.10 | 0.14 | 0.14 | 0.05 | ||
18 | Knodus aff. victoriae (Itapecuru) | 0.14 | 0.15 | 0.10 | 0.09 | 0.09 | 0.10 | 0.11 | 0.13 | 0.12 | 0.11 | 0.14 | 0.12 | 0.09 | 0.14 | 0.12 | 0.08 | 0.06 | |
19 | Knodus guajajara | 0.16 | 0.13 | 0.15 | 0.13 | 0.13 | 0.14 | 0.14 | 0.16 | 0.11 | 0.13 | 0.11 | 0.13 | 0.13 | 0.13 | 0.11 | 0.13 | 0.15 | 0.13 |
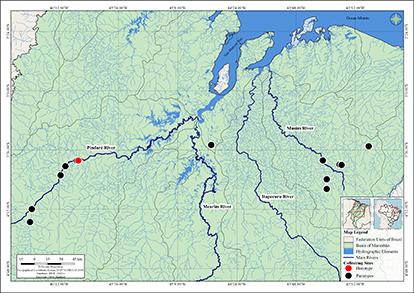
FIGURE 5| Geographical distribution of Knodus guajajara.
Mouth sub-terminal; jaws isognathous. Posterior terminus of maxilla extending beyond anterior margin of orbit. Premaxillary teeth in two rows; outer row with 3(18) and 4(8) tricuspid teeth, inner row with 4(26) tri-to pentacuspid teeth. Maxilla with 3(11), 4(13), 5(2) tri-to pentacuspid teeth. Dentary with three anteriormost teeth large, tri and pentacuspid teeth, followed by 4(2), 5(2), 6(4), 7(13) and 8(5) smaller, uni, tricuspid or pentacuspid teeth (Fig. 3).
Scales cycloid, moderately large, with 6–8 well-marked radii; circuli only present proximally. Lateral line complete, slightly curved ventrally along its anterior third, with 36(8), 37(61), 38*(137), 39(13), or 40(7) perforated scales. Longitudinal series of scales between dorsal-fin origin and lateral line 4(30) or 5*(202). Longitudinal series of scales between lateral line and pelvic-fin origin 4*(63), or 5(184). Predorsal scales 11(3), 12(106) or 13*(122). Circumpeduncular scales 12*(220). Single series of scales on anal-fin base.
Dorsal fin rays ii,7(45) or 8*(191). Dorsal-fin origin slightly posterior to middle of body, and slightly posterior to vertical through pelvic-fin origin. First unbranched ray approximately half length of second unbranched ray. Pectoral-fin rays i,9(9), 10(142) or 11*(94); tip of pectoral fin reaching or exceeds pelvic fin origin when adpressed. Pelvic-fin rays i,5,i(41) or i,6,i*(189); tip of pelvic fin usually reaching first anal fin rays when adpressed. Anal-fin rays iv, 20(2), 21(8), 22(40), 23*(140), 24(37) or 25(15). Anal-fin origin located at vertical through middle of dorsal fin. Anal-fin rays decreasing gradually in length. Caudal fin forked, lobes equal in size. Principal caudal-fin rays i,9,8,i(219). Dorsal procurrent caudal-fin rays 8(1), 9(1), 10(18), 11(3) or 12(3) and ventral procurrent caudal-fin rays 9(3), 10(2), 11(15), 12(3) or 13(3). Adipose-fin origin approximately at vertical through base of 19th to 21st branched anal-fin rays. Gill rakers on first gill arch 15(2), 16(4) or 17(2): hypobranchial 2(4) or 3 (4), ceratobranchial 7(8), cartilage between epibranchial and ceratobranchial 1(8), epibranchial 5(4) or 6(4). Gill rakers setiform. Branchiostegal rays 4(8), 3(8) on anterior ceratohyal and 1(8) on posterior ceratohyal. Total vertebrae 33(3), 34(18) or 35(8). First dorsal fin pterygiophore inserted between 10th and 12th vertebrae. First anal fin pterygiophore inserted between 16th and 17th vertebrae. Supraneurals 4(2), 5(12) or 6(9).
Color in alcohol. Ground of body and head light brown, with dorsal region of body (especially dorsal profile), dorsal portion of head (just above eyes), and upper snout area (upper jaw and adjacent areas) darker. Ventral region of trunk and head with light brown coloration, especially on scales adjacent to anal-fin base. Dark colored chromatophores concentrated on scales of dorsal region (from both body and head), on opercle, and lateral band of flank (especially on posterior region, near caudal peduncle and caudal-fin base). Conspicuous vertically elongated humeral spot, with some dark scattered chromatophores above and below this blotch. Lateral band formed by chromatophores located above lateral line, extending gradually from humeral blotch to middle rays of caudal fin, where they become thicker and more concentrated. All fins including adipose hyaline, with chromatophores on inter-radial membranes of dorsal fin. Anal and caudal fins with posterior margins dark grey.
Color in life. Yellowish coloration in dorsolateral region and silver coloration in ventrolateral region of body (Fig. 2). Gray lateral band located in middle portion of trunk, with bright pigment extending from operculum to caudal-fin base. Dorsal portion of eye yellowish-orange and lower portion silver; mouth and dorsolateral portion of head yellowish with concentrated chromatophores; operculum with silver coloration. Fins hyaline with chromatophores present and especially concentrated in medial region of dorsal fin, distal region of anal and caudal fins, on first pectoral-fin ray, and just posterior to medial lateral band on caudal fin. Adipose fin with yellowish-orange pigmentation; yellowish-orange pigments also present in middle region of caudal fin.
Sexual dimorphism. Hooks present on pelvic fins and anal fin of sexually mature males. Pelvic fins with 5 to 14 hooks per branched ray; anal fin with 2 to 8 hooks per ray. Hooks concentrated on anterior anal fin rays (Fig. 4).
Geographical distribution. Knodus guajajara occurs in the rivers, streams, and lagoons of the Mearim and Munim river basins, in the State of Maranhão, Northeast Brazil (Fig. 5).
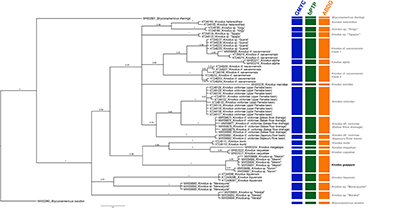
FIGURE 6| Bayesian Inference phylogenetic tree generated by this study. The results of GMYC, bPTP and ABGD are included. Numbers above and below branches are posterior probability values. The gray bars correspond to the congruent results of the different species delimitation methods (Con.).
Etymology. The specific epithet honors the Guajajara indigenous tribe, which is one of the most numerous indigenous peoples in Brazil. They inhabit more than 10 Indigenous Lands on the eastern margin of the Amazon, all located in Maranhão. A noun in apposition the language spoken by them is the Teneteara, from the Tupi-Guarani linguistic family.
Conservation status. Knodus guajajara occurs in two distinct river basins of Maranhão, the Mearim and Munim river basins. It was found in several collecting sites along these basins, and it is always abundant. Therefore, according to the guidelines given in the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN, 2019), we recommend that Knodus guajajara should be categorized as Least Concern (LC).
Molecular species delimitation
Molecular diagnosis. Knodus guajajara is diagnosed molecularly by a combination of 19 nucleotide substitutions, four of them unique* (listed below):
COI 279 (C→A), COI 282 (C→T), COI 306 (C→T), COI 327 (C→T), COI 357 (A→G*), COI 372 (A→G*), COI 378 (A→G), COI 414 (T→C), COI 450 (T→C), COI 459 (C→T), COI 510 (T→C), COI 528 (C→T), COI 537 (C→T), COI 565 (C→T), COI 585 (T→C), COI 589 (C→T), COI 597 (C→T), COI 627 (A→G*), COI 645 (T→A*).
Traditional DNA Barcoding. COI sequences support the existence of a new species of Knodus inhabiting the Mearim and Munim River basins. After trimming, the final alignment yielded 401 base pairs with 132 variable sites and 62 haplotypes. Knodus guajajara is 14% divergent, on average, from the other taxa, with a minimum genetic distance of 11% to K. borki, K. caquetae and Knodus sp. “Maracaçumé”, and a maximum genetic distance of 16% to Bryconamericus exodon Eigenmann,1907 and Knodus tiquiensis. The genetic distance between the new species and K. victoriae is 13% (Tab. 4).
Phylogenetic analysis. The BI phylogenetic analysis delimited 19 lineages: two of them are outgroups (Bryconamericus species) and the remaining correspond to Knodus species (Fig. 6). One of the 17 lineages of Knodus represents the new species described herein (Knodus guajajara)with haplotypes from the Mearim and Munim river basins forming an exclusive clade supported by the highest possible support value (PP = 1) (Fig. 6). The BI phylogenetic analysis also delimited new lineages of Knodus inhabiting the Tocantins River (Knodus sp. “Marabá”) and the Maracaçumé River (Knodus sp. “Maracaçumé”). In addition, two well supported clades were recovered within Knodus cf. savannensis: the first one containing Knodus cf. savannensis (Tocantins River, Carolina, MA), Knodus “Guamá” and Knodus cf. savannensis (KT248218 and KT248219) (posterior probability = 0.96). The second clade of Knodus cf. savannensis corresponds to six haplotypes (KT248199, KT248200, KT248201, KT248202, KT248203, and KT248204) of Knodus cf. savannensis (PP = 1). Two other lineages closely related to K. victoriae were detected: Knodus cf. victoriae (Balsas River drainage) and Knodus cf. victoriae (Itapecuru River basin) (PP = 1).
GMYC, bPTP and ABGD. The results of the three species delimitation methods (GMYC, bPTP, and ABGD) were congruent (Fig. 6), and all three delimited the same 19 lineages. Two of them are outgroups (Bryconamericus species), and 17 are Knodus lineages (species) (Fig. 6). The new species herein described (K. guajajara) was recovered in all three species delimitation methods. Furthermore, some potential undescribed species were detected: Knodus “Marabá”, Knodus “Maracaçumé”, Knodus cf. victoriae (Itapecuru River basin) and Knodus cf. victoriae (Balsas River drainage). Knodusvictoriae (upper Parnaiba River basin) was also recovered, as well as two K. cf. savannensis clades.
Discussion
Knodus guajajara is the first species of the genus described using a combination of morphological and genetic data. The new species has the traditional morphological characteristics that define Knodus, especially the small scales covering the procurrent rays of the caudal fin (Eigenman, 1918; Géry, 1972, 1977; Román-Valencia et al., 2008).
There are some congeners that occur geographically close to the new species described here: Knodus cupariensis, K. dorsomaculatus and K. heteresthes (from the Tapajós River basin); K. nuptialis and K. weitzmani (from the Xingu River basin); K. savannensis (from the lower/middle Tocantins River basin); and K. victoriae (from the upper Parnaiba River basin) (Aguiar et al., 2021; Fricke et al., 2022; and this study). Furthermore, Knodus septentrionalis from Ecuador’s upper Pastaza River basin was previously considered by Géry (1972) as a subspecies of Knodus victoriae, but was elevated to the species category by Chang, Ortega (1995). The new species is morphologically distinguished from these aforementioned species, as well as all the other congeners, by a unique combination of character states (see diagnosis). Of the geographically proximate species to Knodus guajajara, K. savannensis and K. victoriae are the closest ones (lower/middle Tocantins River and Parnaiba River basins, respectively). However, Knodus guajajara is distinguished from K. savannensis in the form of its conspicuous and slightly rounded humeral patch, which is inconspicuous and vertically elongated in K. savannensis. It is morphologically most similar to K. victoriae, having been previously misidentified as that species in the Munim and Mearim river basins (Oliveira et al., 2020; Guimarães et al.,2018a, 2020a). However, some morphological characteristics differ between these two species, such as the presence of fewer circumpeduncular scales, more branched rays in the anal fin and more vertebrae in Knodus guajajara (see diagnosis).
Accurate discussions regarding phylogenetic relationships, as well as the biogeography of the species in the studied area, would be speculative due to the lack of available data, poor taxonomic resolution of some species and knowledge gaps for Knodus species in this region, as well as for the coastal river systems of Maranhão.
Using only morphological characteristics to delimit species in Knodus has caused identification errors in taxonomic studies (García-Melo et al., 2019). For this reason, García-Melo et al. (2019) did not solely consider the presence of scales at the base of the tail fin as a convincing feature to delimit the genus, but rather emphasized the use of DNA sequences to delimit Knodus species more precisely.
In accordance with that recommendation, in addition to morphological evidence, the description of Knodus guajajara is also supported by molecular data (see Fig. 6 and Tab. 4) from five different and independent single locus species delimitation methods. It has a high genetic distance value (13%) (Tab. 4) relative to K. victoriae, the most morphologically similar species, and appears to be phylogenetically distant (Fig. 6). Knodus guajajara is the sister group to a clade comprising K. borki, K. caquetae and K. megalops, possessing 11%, 11% and 13% of genetic distance, respectively, to these species. It is important to emphasize that K. guajajara is more genetically similar to these three species than to several geographically closer congeners (see Tab. 4). Knodus borki is known from near Iquitos, in Peru, K. caquetae occurs in the Amazon and Caquetá River basins of Colombia, and K. megalops is distributed in the Upper Amazon River basin, in Peru and Ecuador (Fricke et al.,2022). All three of these regions are far away from the known geographical distribution of Knodus guajajara. This pattern may be an artifact of the gap of knowledge for Knodus or of the scarcity of genetic data for the genus.
In addition to the new species described here, our results reveal the presence of even more potentially undescribed species for the genus Knodus (Fig. 6) than García-Melo et al.(2019), indicating that a knowledge gap for the group still likely exists and that more undescribed species await discovery. Studies that incorporate both molecular and morphological data, as well as, different operational criteria (integrative taxonomy) have shown success in delimiting and describing species and complexes of cryptic or hidden species (e.g., Mazetti et al.,2012; Guimarães et al.,2018b, 2019, 2020b; Brito et al.,2019, 2021; Ottoni et al.,2019; Faria et al.,2020). The same result is observed for the present study, in which the different species delimitation methods based on molecular data demonstrate congruence with each other. All these methods supported the validity of the new species, the hidden diversity in K. victoriae and the possible presence of new species of Knodus in the Maracaçumé and Tocantins river basins (Fig. 6).
Knodus savannensis is distributed throughout the Tocantins basin and has two possible type-localities according to Lima, Géry (2001): “Brazil, state of Tocantins, municipality of Itacajá, between the Manoel Alves Pequeno river and the Vermelho river, Tocantins river basin approx. 8°19’S, 47°25W”, or “Brazil, Tocantins river basin, Javaés river, Bananal Island, approx. 11°S, 51°W”. A more comprehensive study focusing on K. savannensis is necessary to solve the taxonomy complexity of this species and its type locality.
Our results point to another lineage (Knodus sp. “Marabá”) in the Tocantins River that is apparently not closely related to the Knodus savannensis complex, suggesting two distinct colonization or speciation events in the basin. The occurrence of geographically close species in the genus was recently recorded by Deprá et al. (2021), in which two new species (K. rufford and K. obolus) share the same type locality in the upper Tocantins river basin, thus demonstrating another case of hidden diversity within Knodus.
Still another case of cryptic diversity occurs in the Maracaçumé river basin (Maranhão State). In this river a divergent lineage occurs (Knodus sp. “Maracaçumé”, see Tab. 4), but despite its geographic proximity, it possesses a high genetic distance (11%) to Knodus guajajara, suggesting that is a new species according to the species delimitation methods conducted. The occurrence of new characids has been demonstrated for the Maracaçumé River basin, including cryptic species (e.g., Brito et al.,2019; Guimarães et al.,2020b). The description of new fish species for this basin is expected since according to Abreu et al. (2019, 2020), biogeographic processes favored the isolation of drainages in Maranhão, providing opportunities for the emergence of new species.
We also identified cryptic diversity occurring in Knodus victoriae with the detection of two additional lineages: one from the Balsas River drainage of the upper Parnaiba River basin [Knodus aff. victoriae (Balsas)]; and the other from the upper Itapecuru River basin [Knodus aff. victoriae (Itapecuru)] (Fig 6; Tab. 4), totaling three lineages under the name Knodus victoriae. A noteworthy aspect is that two of these lineages, Knodus aff. victoriae (Balsas) and Knodus victoriae,occur in upper sections of the Parnaiba River basin (Aguiar et al., 2021; this study). It is likely that the presence of these sister lineages in the upper Parnaiba and upper Itapecuru river basins is due to past headwater captures between these river systems.
In addition to contributing to the description of a new species (Knodus guajarara), the results of our present work revealed that a hidden diversity of species of the genus Knodus inhabits the hydrographic basins of Maranhão and adjacent systems. The use of different data sources (morphology, molecular, geographic distribution, etc.) can help to identify genetically distinct groups with high morphological similarity, thereby avoiding identification errors and helping to solving problems of validity and taxonomic placement within Characidae.
Comparative material examined. Knodus borki: Peru. MTDF 31357, 1, 33.99 mm SL, holotype, Umgebung von Iquitos.Knodus longus: Bolivia. MTDF 28853, 1, 46.1 mm SL, holotype, río Warinilla (oder Harinilla) nahe Choiro. Knodus pasco: Peru. MTDF 30634, 1, 55.4 mm SL, holotype, Departamento Pasco, erste Quebrada bei San Antonio de Cacazú (eigentlich nach dem Vorort Baja Cacazú) auf der Straße, nach Puerto Bermúdez.Knodus savannensis: Brazil. USNM 196088, 1, 30.4 mm SL, holotype, Tocantins, Tocantins basin.Knodus savannensis (Clade 1): Brazil. CICCAA 2397, 108, 36.83–48.67 mm SL; CICCAA 2399, 73, 31.46–38.52 mm SL; CICCAA 2400, 27, 38.37–61.08 mm SL, Maranhão, Carolina. Knodus sp. “Marabá”: Brazil. CICCAA 1522, 2, 29.16–33.92 mm SL; CICCAA 1306, 14, 22.92–36.22 mm SL; CICCAA 1503, 3, 34.36–38.24 mm SL, Pará, Marabá. CICCAA 1525, 9, 24.4–37.74 mm SL, Pará, Curinópolis. CICCAA 1436, 20, 17.74–27.51 mm SL, Pará, Paragominas. CICCAA 1409, 6, 30.7–38.9 mm SL; CICCAA 1521, 4, 29.85–37.67 mm SL; CICCAA 1516, 1, 29.62 mm SL; CICCAA 1524, 5, 20.68–20.82 mm SL;CICCAA 1509, 3, 34.86–38.24 mm SL, Maranhão, São Pedro da Água Branca.CICCAA 1295, 27, 20.36–28.27 mm SL; CICCAA 1523, 8, 17.82–22.58 mm SL, Maranhão, Vila Nova dos Martírios. Knodus victoriae: Brazil. NMW 57823, 16 syntypes; NMW 57824, 13 syntypes; NMW 57825, 18 syntypes of Tetragonopterus victoriae, Maranhão, Parnaíba basin; CICCAA 2708, 18; CICCAA 2709, 17; CICCAA 2710, 7 C&S, Maranhão, Alto Parnaíba municipality, Parnaíba river basin; Knodus aff. victoriae: Brazil. CICCAA 2842, 1, 29.5 mm SL, Maranhão, Itapecuru River basin. CICCAA 4809, 9, 15.57–28.47 mm SL; CICCAA 4810, 31, 20.73–43.48 mm SL; CICCAA 4811, 29, 17.18–42.22 mm SL; CICCAA 4812, 34, 17.32–42.18 mm SL; CICCAA 4892, 8 C&S, CICCAA 4893, 2 C&S, Maranhão, Parnaíba basin. Knodus cf. victoriae: Brazil. CICCAA 2395, 6, 25.6–36.08 mm SL; CICCAA 2391, 5 C&S, 27.51–31.4 mm SL, Maranhão, Itapecuru River basin.
Acknowledgments
We thank Axel Zarske, Ingo Schindler and Ronald Fricke for providing useful literature; Vale S.A, Amplo Engenharia and Instituto Maranhense de Estudos Socioeconômicos e Cartográficos (IMESC) for sharing part of the data analyzed in this study; Mark Sabaj Pérez (CAS), James Maclaine (FMNH), Riedel Bettina (NMW), Sandra Raredon (Smithsonian Institution) for providing photographs, x-ray images, and information on the type material of some species; Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES – Finance Code 001) and Fundação de Amparo à Pesquisa e ao Desenvolvimento Científico e Tecnológico do Maranhão (FAPEMA) for providing the scholarship under the process 88882.445737/2019–01 to RGA, BD–05604/19 to ECG, 88887.159561/2017–00 to PSB; Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; grant 307974/2021–9 to FPO) and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ; grant E–26/202.005/2020 to AMK). This study was supported by the project “PROCESSO UNIVERSAL – 00724/17”, from FAPEMA.
References
Abreu JMS, Craig JM, Albert JS, Piorski NM. Historical biogeography of fishes from coastal basins of Maranhão State, northeastern Brazil. Neotrop Ichthyol. 2019; 17(2):e180156. https://doi.org/10.1590/1982-0224-20180156
Abreu JMS, Saraiva ACS, Albert JS, Piorski NM. Paleogeographic influences on freshwater fish distributions in northeastern Brazil. J South Am Earth Sci. 2020; 102:102692. https://doi.org/10.1016/j.jsames.2020.102692
Aguiar RG, Guimarães EC, Carvalho-Costa LF, Ottoni FP. Historical review on the type locality of Knodus victoriae (Steindachner 1907) (Teleostei: Characidae) and Loricaria parnahybae Steindachner 1907 (Teleostei: Loricariidae). Biota Neotrop. 2021; 21(4):e20211226. https://doi.org/10.1590/1676-0611-BN-2021-1226
Aljanabi SM, Martinez I. Universal and rapid salt-extraction of high quality genomic DNA for PCR-based techniques. Nucleic Acids Res. 1997; 25(22):4692–93. https://doi.org/10.1093/nar/25.22.4692
Bickford D, Lohman DJ, Sodhi NS, Ng PK, Meier R, Winker K, Das I. Cryptic species as a window on diversity and conservation. Trends Ecol Evol. 2007; 22(3):148–55. https://doi.org/10.1016/j.tree.2006.11.004
Brito PS, Guimarães EC, Carvalho-Costa LF, Ottoni FP. A new species of Aphyocharax Günther, 1868 (Characiformes, Characidae) from the Maracaçumé river basin, eastern Amazon. Zoosyst Evol. 2019; 95(2):507–16. https://doi.org/10.3897/zse.95.36788
Brito PS, Guimarães EC, Guimarães KLA, Rodrigues LRR, Anjos MR, Katz AM. Cryptic speciation in populations of the genus Aphyocharax (Characiformes: Characidae) from eastern Amazon coastal river drainages and surroundings revealed by single locus species delimitation methods. Neotrop Ichthyol. 2021; 19(4):e210095. https://doi.org/10.1590/1982-0224-2021-0095
Chang F, Ortega H. Additions and corrections to the list of freshwater fishes of Perú. Publ Mus Hist Nat UNMSM (A). 1995; 50:1–11.
Chenna R, Sugawara H, Koike T, Lopez R, Gibson TJ, Higgins DG et al. Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res. 2003; 31(13):3497–500. https://doi.org/10.1093/nar/gkg500
Close B, Banister K, Baumans V, Bernoth EM, Bromage N, Bunyan J et al. Recommendations for euthanasia of experimental animals: Part 1. Lab Anim. 1996; 30(4):293–316. https://doi.org/10.1258/002367796780739871
Coissac E, Hollingsworth PM, Lavergne S, Taberlet P. From barcodes to genomes: Extending the concept of DNA barcoding. Mol Ecol. 2016; 25(7):1423–28. https://doi.org/10.1111/mec.13549
Costa WJEM, Amorim PF, Aranha GN. Species limits and DNA barcodes in Nematolebias, a genus of seasonal killifishes threatened with extinction from the Atlantic Forest of south-eastern Brazil, with description of a new species (Teleostei: Rivulidae). Ichthyol Explor Freshw. 2014; 24(3):225–36.
Costa-Silva GJ, Ashikaga FY, Dias CKS, Pereira LHG, Foresti F, Oliveira C. DNA barcoding techniques used to identify the shared ichthyofauna between the Pantanal floodplain and Upper Paraná River. Mitochondrial DNA A. 2018; 29(7):1063–72. https://doi.org/10.1080/24701394.2017.1404046
Darriba D, Taboada GL, Doallo R, Posada D. jModelTest 2: More models, new heuristics and parallel computing. Nat Methods. 2012; 9(8):772. https://doi.org/10.1038/nmeth.2109
DeSalle R, Egan MG, Siddall M. The unholy trinity: Taxonomy, species delimitation and DNA barcoding. Philos Trans R Soc Lond B Biol Sci. 2005; 360(1462):1905–16. https://doi.org/10.1098/rstb.2005.1722
DeSalle R, Goldstein P. Review and interpretation of trends in DNA barcoding. Front Ecol Evol. 2019; 7:302. https://doi.org/10.3389/fevo.2019.00302
Deprá GC, Ota RR, Vitorino Júnior OB, Ferreira KM. Two new species of Knodus (Characidae: Stevardiinae) from the upper rio Tocantins basin, with evidence of ontogenetic meristic changes. Neotrop Ichthyol. 2021; 19(1):e200106 https://doi.org/10.1590/1982-0224-2020-0106
Drummond AJ, Suchard MA, Xie D, Rambaut A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol. 2012; 29(8):1969–73. https://doi.org/10.1093/molbev/mss075
Esguícero ALH, Castro RCC. Knodus figueiredoi, a new characid from the Rio das Garças, upper Rio Araguaia basin, Brazil, with comments on the taxonomic limits of the genera Knodus and Bryconamericus (Teleostei: Characidae). Ichthyol Explor Freshw. 2014; 25(1):39–48.
Eigenmann CH. The American Characidae. Mem Mus Comp Zool; 1918; 43(2):103–208.
Faria TC, Bastos DA, Zuanon J, Lima FCT. A new Hyphessobrycon (Characiformes: Characidae) of the Hyphessobrycon heterorhabdus species-group from the Central Amazon basin, Brazil. Zootaxa. 2020; 4859(2):zootaxa-4859. https://doi.org/10.11646/zootaxa.4859.2.6
Ferreira KM, Lima FCT. A new species of Knodus (Characiformes: Characidae) from the rio Tiquié, upper rio Negro system, Brazil. Copeia. 2006; 2006(4):630–39. https://doi.org/10.1643/0045-8511(2006)6[630:ANSOKC]2.0.CO;2
Ferreira KM, Carvajal FM. Knodus shinahota (Characiformes: Characidae) a new species from the río Shinahota, río Chapare basin (Mamoré system), Bolivia. Neotrop Ichthyol. 2007; 5(1):31–36. https://doi.org/10.1590/S1679-62252007000100004
Ferreira KM, Netto-Ferreira AL. Knodus dorsomaculatus (Characiformes: Characidae), a new species from Teles Pires River, Tapajós River basin, Brazil. J Fish Biol. 2010; 77(3):468–78. https://doi.org/10.1111/j.1095-8649.2010.02680.x
Ferreira KM, Mirande JM, Quagio-Grassiotto I, Santana JCO, Baicere-Silva CM, Menezes NA. Testing the phylogenetic hypotheses of Stevardiinae Gill, 1858 in light of new phenotypic data (Teleostei: Characidae). J Zool Syst Evol Res. 2021; 59(8):2060–85. https://doi.org/10.1111/jzs.12517
Fink WL, Weitzman SH. The so-called cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Smithson Contrib Zool. 1974; 172:1–46. https://doi.org/10.5479/si.00810282.172
Fricke R, Eschmeyer WN. Eschmeyer’s Catalog of Fishes: Guide to fish collections [Internet]. San Francisco: California Academy of Sciences; 2022. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/collections.asp
Fricke R, Eschmeyer W, Van Der Laan R. Eschmeyer’s Catalog of Fishes: genera, species, references [Internet]. San Francisco: California Academy of Sciences; 2022. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Fujisawa T, Barraclough TG. Delimiting species using single-locus data and the GeneralizedMixed Yule Coalescent approach: A revised method and evaluation on simulated data sets. Syst Biol. 2013; 62(5):707–24. https://doi.org/10.1093/sysbio/syt033
García-Melo JE, Oliveira C, Silva GJC, Ochoa-Orrego LE, Pereira LHG, Maldonado-Ocampo JA. Species delimitation of neotropical Characins (Stevardiinae): Implications for taxonomy of complex groups. PLoS ONE. 2019; 14(6):e0216786. https://doi.org/10.1371/journal.pone.0216786
Géry J. Contribution à l’étude des poissons characoïdes de l’équateur. Avec une révision du genre Pseudochalceus et la description d’une nouveaux genre endémique du Rio Cauca en Colombie. Acta Humboldtiana, Ser Geológica, Palentológica et Biológica. 1972; 2:1–110.
Géry J. Characoids of the world. Neptune City: TFH Publications; 1977.
Guimarães EC, Brito PS, Feitosa LM, Carvalho-Costa LF, Ottoni FP. A new cryptic species of Hyphessobrycon Durbin, 1908 (Characiformes, Characidae) from the Eastern Amazon, revealed by integrative taxonomy. Zoosyst Evol. 2019; 95(2):345–60. https://doi.org/10.3897/zse.95.34069
Guimarães EC, Brito PS, Ferreira BRA, Ottoni FP. A new species of Charax (Ostariophysi, Characiformes, Characidae) from northeastern Brazil. Zoosyst Evol. 2018a; 94(1):83–93. https://doi.org/10.3897/zse.94.22106
Guimarães EC, Brito PS, Feitosa LM, Carvalho-Costa LF, Ottoni FP. A new species of Hyphessobrycon Durbin from northeastern Brazil: Evidence from morphological data and DNA barcoding (Characiformes, Characidae). ZooKeys. 2018b; 765:79–101. https://doi.org/10.3897/zookeys.765.23157
Guimarães EC, Brito PS, Gonçalves CS, Ottoni FP. An inventory of Ichthyofauna of the Pindaré River drainage, Mearim River basin, Northeastern Brazil. Biota Neotrop. 2020a; 20(4):e20201023. https://doi.org/10.1590/1676-0611-BN-2020-1023
Guimarães EC, Brito PS, Bragança PHN, Santos JP, Katz AM, Carvalho Costa LF. Integrative taxonomy reveals two new cryptic species of Hyphessobrycon Durbin, 1908 (Teleostei: Characidae) from the Maracaçumé and middle Tocantins River basins, Eastern Amazon region. Eur J Taxon. 2020b; 723(1):77–107. https://doi.org/10.5852/ejt.2020.723.1145
Hajibabaei M, Singer GAC, Hebert PDN, Hickey DA. DNA barcoding: How it complements taxonomy, molecular phylogenetics and population genetics. Trends Genet. 2007; 23(4):167–72. https://doi.org/10.1016/j.tig.2007.02.001
Hebert PDN, Cywinska A, Ball SL, deWaard JR. Biological identifications through DNA Barcodes. Proc R Soc Lond B Biol Sci. 2003a; 270(1512):313–21. https://doi.org/10.1098/rspb.2002.2218
Hebert PDN, Ratnasingham S, de Waard JR. Barcoding animal life: Cytochrome c oxidase subunit 1 divergences among closely related species. Proc R Soc Lond B Biol Sci. 2003b; 270(Suppl_1):S96–99.
International Union for Conservation of Nature (IUCN). Standards and Petitions Committee. Guidelines for using the IUCN Red List Categories and Criteria. Version 14 [Internet]. Gland; 2019. Available from: https://www.iucnredlist.org/resources/redlistguidelines
Jacobina UP, Lima SMQ, Maia DG, Souza G, Batalha-Filho H, Torres RA. DNA barcode sheds light on systematics and evolution of Neotropical freshwater trahiras. Genetica. 2018; 146(6):505–15. https://doi.org/10.1007/s10709-018-0043-x
Kimura M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol. 1980; 16(2):111–20. https://doi.org/10.1007/BF01731581
Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol. 2016; 33(7):1870–74. https://doi.org/10.1093/molbev/msw054
Lima FCT, Géry J. Correction of the type locality of Hyphessobrycon stegemanni Géry and Knodus savannensis Géry (Teleostei: Characiformes: Characidae). Comun Mus Ciênc Tecnol PUCRS, Sér. Zool. 2001; 14(1):95–97.
Leary S, Underwood W, Anthony R, Cartner S, Corey D, Grandin T et al. AVMA guidelines for the euthanasia of animals: 2013 edition. 2013; Schauumburg: American Veterinary Medical Association. Available from: https://www.purdue.edu/research/oevprp/regulatory-affairs/animal-research/docs/2013_AVMA_Guidelines.pdf
Mazeti CM, Maniglia TC, Prioli SMAP, Prioli AJ. Molecular variants in populations of Bryconamericus aff. iheringii (Characiformes, Characidae) in the upper Paraná river basin. Acta Sci Biol Sci. 2012; 35(2):241–48. https://doi.org/10.4025/actascibiolsci.v35i2.11451
Melo BF, Benine RC, Mariguela TC, Oliveira C. A new species of Tetragonopterus Cuvier, 1816 (Characiformes: Characidae: Tetragonopterinae) from the rio Jari, Amapá, northern Brazil. Neotrop Ichthyol. 2011; 9(1):49–56. https://doi.org/10.1590/S1679-62252011000100002
Menezes NA, Netto-Ferreira AL, Ferreira KM. A new species of Bryconadenos (Characiformes: Characidae) from the rio Curuá, rio Xingu drainage, Brazil. Neotrop Ichthyol. 2009; 7(2):147–52. https://doi.org/10.1590/S1679-62252009000200003
Menezes NA, Marinho MMF. A new species of Knodus Eigenmann (Characiformes: Characidae: Stevardiinae) with comments on nuptial tubercles and gill gland in characiform fishes. PLoS ONE. 2019; 14(7):e0217915. https://doi.org/10.1371/journal.pone.0217915
Menezes NA, Ferreira KM, Netto-Ferreira AL. A new species of Knodus Eigenmann (Characiformes: Characidae: Stevardiinae) from the rio Aripuanã, rio Madeira basin, Brazil. Neotrop Ichthyol. 2020; 18(2):e190139. https://doi.org/10.1590/1982-0224-2019-0139
Mirande JM. Morphology, molecules and the phylogeny of Characidae (Teleostei, Characiformes). Cladistics. 2019; 35(3):282–300. https://doi.org/10.1111/cla.12345
Oliveira ES, Guimarães EC, Brito PS, Vieira LO, Oliveira RF, Campos DS et al. Ichthyofauna of the Mata de Itamacaoca, an urban protected area from the upper Munim River basin, Northern Brazilian Cerrado. Biota Neotrop. 2020; 20(4):e20201116. https://doi.org/10.1590/1676-0611-BN-2020-1116
Ottoni FP, Mattos JLO, Katz AM, Bragança PHN. Phylogeny and species delimitation based on molecular approaches on the species of the Australoheros autrani group (Teleostei, Cichlidae), with biogeographic comments. Zoosyst Evol. 2019; 95(1):49–64. https://doi.org/10.3897/zse.95.31658
Puillandre N, Lambert A, Brouillet S, Achaz G. ABGD, Automatic Barcode Gap Discovery for primary species delimitation. Mol Ecol. 2012; 21(8):1864–77. https://doi.org/10.1111/j.1365-294X.2011.05239.x
Rambaut A, Suchard M, Xie D, Drummond A. Tracer v1.6. 2014. Available from: http://beast.bio.ed.ac.uk/Tracer
Román-Valencia C. Tres nuevas especies de Bryconamericus (Ostariophysi: Characidae) de Colombia y diagnóstico del género. Rev Biol Trop. 2000; 48(2–3):449–64.
Román-Valencia C, Taphorn B. DC, Ruiz-C RI. Two new Bryconamericus: B. cinarucoense n. sp. and B. singularis n. sp. (Characiformes, Characidae) from the Cinaruco River, Orinoco Basin, with keys to all Venezuelan Species. Anim Biodivers Conserv. 2008; 31(1):15–27.
Ronquist F, Teslenko M, Van der Mark P, Ayres DL, Darling A, Höhna S et al. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol. 2012; 61(3):539–42. https://doi.org/10.1093/sysbio/sys029
Schultz LP. The fishes of the family Characinidae from Venezuela, with description of seventeen new forms. Proc U S Natl Mus. 1944; 95(3181):235–367. https://doi.org/10.5479/si.00963801.95-3181.235
Sousa DJA, Silva-Oliveira C, Canto ALC, Ribeiro FRV. A new species of Knodus (Characiformes: Characidae) from the rio Cupari drainage, lower rio Tapajós basin, Brazil. Zootaxa. 2020; 4747(3):575–84. https://doi.org/10.11646/zootaxa.4747.3.10
Swofford DL. PAUP. Phylogenetic Analysis Using Parsimony (*and Other Methods), 4.0 edn, Sinauer, Sunderland, MA, 2002.
Taphorn DC. The characiform fishes of the Apure River drainage, Venezuela: UNELLEZ; 1992.
Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and others vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
Thomaz AT, Arcila D, Ortí G, Malabarba LR. Molecular phylogeny of the subfamily Stevardiinae Gill, 1858 (Characiformes: Characidae): Classification and the evolution of reproductive traits. BMC Evol Biol. 2015; 15:146. https://doi.org/10.1186/s12862-015-0403-4
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PDN. DNA barcoding Australia’s fish species. Philos Trans R Soc Lond B Biol Sci. 2005; 360(1462):1847–57. https://doi.org/10.1098/rstb.2005.1716
Weitzman SH. The osteology of Brycon meeki, a generalized characid fish, with an osteological definition of the family. Stanford Ichthyological Bulletin. 1962; 8(1):3–77.
Xia X. DAMBE5: A comprehensive software package for data analysis in molecular biology and evolution. Mol Biol Evol. 2013; 30(7):1720–28. https://doi.org/10.1093/molbev/mst064
Zarske A. Knodus pasco sp. n. – ein neuer Salmler (Teleostei: Characiformes: Characidae) aus Peru. Vertebr Zool. 2007; 57(1):15–21.
Zarske A. Knodus borki sp. n. – ein neuer Salmler aus Peru mit einer erganzenden Beschreibung von Boehlkea fredcochui Géry, 1966 (Teleostei: Characiformes: Characidae). Vertebr Zool. 2008; 58(2):159–71.
Zarske A, Géry J. Knodus longus sp. n. – ein neuer Salmler (Teleostei: Characiformes: Characidae) aus den bolivianischen Andean, Einzugsgebiet des río Beni. Zool Abh. 2006; 55:51–57.
Zhang J, Kapli P, Pavlidis P, Stamatakis A. A general species delimitation method with applications to phylogenetic placements. Bioinformatics. 2013; 29(22):2869–76. https://doi.org/10.1093/bioinformatics/btt499
Authors
![]() Rayane G. Aguiar1,
Rayane G. Aguiar1, ![]() Erick C. Guimarães1,2,3
Erick C. Guimarães1,2,3 ![]() ,
, ![]() Pâmella S. de Brito1,3,
Pâmella S. de Brito1,3, ![]() Jadson P. Santos3,
Jadson P. Santos3, ![]() Axel M. Katz4,
Axel M. Katz4, ![]() Luiz Jorge B. da S. Dias5,
Luiz Jorge B. da S. Dias5, ![]() Luis Fernando Carvalho-Costa1 and
Luis Fernando Carvalho-Costa1 and ![]() Felipe P. Ottoni6
Felipe P. Ottoni6
[1] Universidade Federal do Maranhão, Departamento de Biologia, Laboratório de Ecologia Molecular, Av. dos Portugueses, 1966, Bacanga, Cidade Universitária Dom Delgado, 65080-805. São Luís, MA, Brazil. (RGA) rayaneaguiar1706@gmail.com, (LFCC) luis.costa@ufma.br, (PSB) pamellabrito@hotmail.com.
[2] Universidade Federal do Oeste do Pará, Programa de Pós-Graduação Sociedade Natureza e Desenvolvimento, Instituto de Ciências da Educação, Av. Mendonça Furtado, 2946, Fátima, 68040-470. Santarém, PA, Brazil. erick.ictio@yahoo.com.br (corresponding author).
[3] Universidade Estadual do Maranhão, Laboratório de Ictiofauna e Piscicultura Integrada, Centro de Ciências Agrárias, CidadeUniversitária Paulo VI, 65055-310. São Luís, MA, Brazil. (JPS) jadsonsantos@professor.uema.br.
[4] Universidade Federal do Rio de Janeiro, Laboratório de Sistemática e Evolução de Peixes Teleósteos, Departamento de Zoologia,Instituto de Biologia, Cidade Universitária, 21941-599. Rio de Janeiro, RJ, Brazil. axelmk@gmail.com.
[5] Universidade Estadual do Maranhão, Centro de Educação, Ciências Exatas e Naturais. Cidade Universitária Paulo VI, Tirirical65055970. São Luís, MA, Brazil. luizjorgedias@hotmail.com.
[6] Universidade Federal do Maranhão, Centro de Ciências Agrárias e Ambientais, Laboratório de Sistemática e Ecologia de Organismos Aquáticos, Campus de Chapadinha, BR-222, km 4, S/N, Boa Vista, 65500-000. Chapadinha, MA, Brazil. fpottoni@gmail.com.
Authors’ Contribution 

Rayane Gonçalves Aguiar: Investigation, Methodology, Writing-original draft.
Erick Cristofore Guimarães: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and Editing.
Pâmella Silva de Brito: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and Editing.
Jadson Pinheiro dos Santos: Project administration, Visualization.
Axel Makay Katz: Software, Visualization, Writing-review and Editing.
Luiz Jorge Bezerra da Silva Dias: Project administration, Visualization.
Luis Fernando C. Costa: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing-review and Editing.
Felipe Polivanov Ottoni: Conceptualization, Formal analysis, Visualization, Writing-original draft, Writing-review and Editing.
Ethical Statement
All material was collected with permits 51540–3/ from SISBIO (Sistema de Autorização e Informação em Biodiversidade) and 02001.007241/ 2004–37 IBAMA (Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis).
Competing Interests
The authors declare no competing interests.
How to cite this article
Aguiar RG, Guimarães EC, Brito PS, Santos JP, Katz AM, Dias LJBS, Carvalho-Costa LF, Ottoni FP. A new species of Knodus (Characiformes: Characidae), with deep genetic divergence, from the Mearim and Munim river basins, Northeastern Brazil, and evidence for hidden diversity in adjacent river basins. Neotrop Ichthyol. 2022; 20(2):e210173. https://doi.org/10.1590/1982-0224-2021-0173
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2022 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted April 8, 2022 by Brian Sidlauskas
Accepted April 8, 2022 by Brian Sidlauskas
![]() Submitted January 26, 2022
Submitted January 26, 2022
![]() Epub June 03, 2022
Epub June 03, 2022