![]() Thais L. Quintão1,
Thais L. Quintão1, ![]() João Luiz Gasparini3,
João Luiz Gasparini3, ![]() Jean-Christophe Joyeux2,
Jean-Christophe Joyeux2, ![]() Luiz A. Rocha4 and
Luiz A. Rocha4 and ![]() Hudson T. Pinheiro4,5
Hudson T. Pinheiro4,5 ![]()
PDF: EN XML: EN | Supplementary: S1 | Cite this article
Abstract
The genus Acyrtus (Gobiesocidae) is represented by four valid species distributed in the western Atlantic, and a recently described fifth species from the eastern Pacific. Here, we describe a new species endemic to Trindade Island, Brazil, and provide the first phylogenetic inference for the genus including all representatives. The new species can be distinguished from all its congeners by meristic and morphometric characters, as well as genetic differences. It presents low genetic diversity and, contrarily to other Trindade Island endemic fishes, shows no evidence of recent population growth. Our phylogeny reveals cryptic species and the paraphyletic nature of Acyrtus, which included Arcos nudus (western Atlantic) in a clade that separated from Arcos erythrops (tropical eastern Pacific) around 20 Mya. The three species found in the Brazilian Province, including one that remains undescribed, form a monophyletic clade which colonized the western South Atlantic around 2.6 Mya. Our study suggests that Arcos nudus should be placed in Acyrtus, and that the relationships among the closely-related Gobiesocidae genera Acyrtus (mostly from the Atlantic Ocean) and Arcos (from the Pacific Ocean) need further investigation.
Keywords: Brazilian Province, Endemism, Evolution, Phylogeny, Reef fish.
O gênero Acyrtus (Gobiesocidae) é representado por quatro espécies válidas encontradas no Atlântico ocidental e uma recentemente descrita do Pacífico oriental. Aqui descrevemos uma nova espécie endêmica da Ilha da Trindade, Brasil, e apresentamos a primeira inferência filogenética para o gênero incluindo todos os representantes. A nova espécie pode ser distinguida de suas congêneres por caracteres merísticos e morfométricos, bem como por diferenças genéticas. A espécie apresenta baixa diversidade genética, entretanto, diferentemente de outras espécies endêmicas da Ilha da Trindade, não mostra evidência de expansão populacional recente. A filogenia obtida revelou a existência de espécies crípticas e a natureza parafilética de Acyrtus, o qual inclui Arcos nudus (do Atlântico ocidental), e que é separado de Arcos erythrops (do Pacifico tropical oriental) por cerca de 20 milhões de anos. As três espécies encontradas no Brasil, incluindo uma ainda não descrita, formam um clado monofilético que colonizou o Atlântico Sul ocidental há cerca de 2,6 milhões de anos. Nosso estudo sugere que Arcos nudus deva ser alocado no gênero Acyrtus, e que as relações entre os gêneros Acyrtus (em maioria do Oceano Atlântico) e Arcos (do Oceano Pacífico) precisam ser estudadas em mais detalhes.
Palavras-chave: Endemismo, Evolução, Filogenia, Peixes recifais, Província Brasileira.
Introduction
The cryptobenthic fish family Gobiesocidae (sensu Brandl et al., 2018), commonly known as clingfishes, comprises 190 recognized species (Fricke et al., 2022) distributed in freshwater, brackish, and coastal areas of the world’s tropical and subtropical regions (Briggs, 1955; Conway et al., 2017). Although occupying different environments and microhabitats (Gonçalves et al., 2002; Henriques et al., 2002), including a wide bathymetric range (Sparks, Gruber, 2012; Fricke et al., 2017), many species are morphologically similar and there is recognized uncertainty in their classification (Conway et al., 2020).
In this context, the use of molecular approaches combined with traditional taxonomy has provided a better understanding of evolutionary relationships (e.g., Henriques et al., 2002; Fricke et al., 2017; Wagner et al., 2019) and identification of cryptic lineages (e.g., Henriques et al., 2002;Craig, Randall, 2008; Conway et al., 2014; Wagner et al., 2019; Torres-Hernández et al., 2020). Though recent molecular studies have provided important progress in the knowledge of this family (Conway et al., 2017, 2020; Fricke et al., 2017), the evolutionary history of most species and the full extent of Gobiesocidae diversity remain unknown. Phylogenetic work including the New World Gobiesocinae suggested a recent diversification, the presence of several cryptic species and paraphyletic genera (Conway et al., 2014, 2017, 2020; Tavera et al.,2021), all of which we will explore in this study within the genus Acyrtus Schultz, 1944.
Acyrtus is represented by five valid species, Acyrtus artius Briggs, 1955, Acyrtus lanthanum Conway, Baldwin & White, 2014, Acyrtus pauciradiatus Sampaio, Anchieta, Nunes & Mendes, 2004 and Acyrtus rubiginosus (Poey, 1868) are restricted to the western Atlantic, while Acyrtus arturo Tavera, Rojas-Vélez & Londoño-Cruz, 2021, was recently described from the eastern Pacific. These are small-bodied fishes (maximum size of less than 30 mm) with cryptobenthic habits, some presenting large secretory cells similar to those present in the venom glands of other teleost fishes (Conway et al., 2014). In the Atlantic, three species are found in the Caribbean and the fourth (A. pauciradiatus) is endemic to Fernando de Noronha Archipelago and Rocas Atoll, in Brazil. However, two other undescribed species have been found in restricted locations of the southwestern Atlantic, one on the Brazilian continental shelf and the other in the oceanic Trindade Island (Pinheiro et al., 2017).
Morphologically, Acyrtus is closely related to Rimicola Jordan & Evermann, 1896, a genus restricted to the eastern Pacific. However, a recent molecular phylogenetic study exploring Gobiesocidae has shown a close relationship between Acyrtus and Arcos Schultz, 1944 and, under low bootstrap values, has placed Arcos nudus (Linnaeus, 1758), from the western Atlantic, within the Acyrtus clade (Conway et al., 2014). This result agrees with the distribution of Arcos and Acyrtus genera since Arcos is mainly restricted to the eastern Pacific and Acyrtus to the western Atlantic. However, the recent description of A. arturo (Tavera et al., 2021) from Malpelo Island opens discussion about the evolutionary history of Acyrtus and Arcos. In addition, both genera share morphological characteristics that obscure their evolutionary proximity (e.g., Tavera et al., 2021). Species of both genera have previously been described as Gobiesox (Schultz, 1944; Conway et al., 2017), indicating that morphological similarities often lead to misidentifications (Conway et al., 2017). In this study, we provide the first phylogenetic inference for the genus Acyrtus containing all known species and including the undescribed species from Trindade Island as new, and an undescribed species from Brazilian coast. In addition, we analyzed the evolutionary history of the genus, and the phylogeographic and demographic history of the Brazilian species complex.
Material and methods
Morphological analysis. The eight type specimens were collected with hand nets during a field expedition to Trindade Island in June 2009. Specimens were fixed in formalin 10% for 24 h, and then transferred to alcohol 70%. Counts were performed with a stereo microscope (Leica S9i, Amplification 6.1–55x) and X-rays obtained with a radiography system Faxitron LX60. Morphological characters were measured to the nearest 0.01 mm using an ocular micrometer mounted on a dissecting scope. Measurements and counts followed the methods presented in Briggs (1955), with the addition of predorsal and preanal lengths, which are the shortest distances between the tip of the upper lip and the dorsal and anal-fin origin, respectively (see Conway et al., 2014). Vertebral counts are presented as precaudal + caudal. The anterior-most vertebra with a haemal spine was counted as the first caudal vertebra, the urostylar complex the last. Following Smith-Vaniz (1971), the principal caudal ray counts included only those rays that articulate with the hypural plate, and are provided as upper+lower counts. Procurrent caudal rays are also provided as upper+lower counts. Measurements are expressed as a percentage of either standard length (SL) or head length (HL). Type specimens were deposited in the fish collections of Universidade Federal do Espírito Santo, Vitória (CIUFES), California Academy of Sciences, San Francisco (CAS-ICH), Museu Nacional do Rio de Janeiro, Rio de Janeiro (MNRJ), Museu de Zoologia da Universidade de São Paulo, São Paulo (MZUSP), Instituto de Biodiversidade e Sustentabilidade – NUPEM, Universidade Federal do Rio de Janeiro, Macaé (NPM), and Museu de Zoologia da Universidade Estadual de Campinas “Prof. Dr. Adão José Cardoso” (ZUEC). In the Description section, counts, measurements, and proportions of the holotype are followed by the range for paratypes inside parentheses. Teeth type and counts were obtained from a dissected specimen.
Genetic data. Cytochrome Oxidase I (COI) sequences of Acyrtus (A. artius, A. arturo, A. lanthanum, A. rubiginosus, Acyrtus sp.n., and Acyrtus sp.), Arcos (A. erythrops (Jordan & Gilbert, 1882)and A. nudus) and Gobiesox Lacepède, 1800 (G. maeandricus (Girard, 1858), G. punctulatus (Poey, 1876), and G. rhessodon Smith, 1881) were obtained from previous studies (Conway et al., 2014; Conway et al., 2017; Pinheiro et al., 2017; Tavera et al., 2021) and applied to phylogenetic and phylogeographic analyses. The DNA sequences from the Brazilian species A. pauciradiatus were extracted and amplified following protocols detailed in Weigt et al. (2012) for COI gene. Species of the families Gobiesocidae, Pseudochromidae, and Grammatidae were added as outgroups for the molecular-clock calibration. All sequences were aligned using ClustalW algorithm implemented in MEGA 7 (Kumar et al., 2016). Accession numbers of the sequences used are shown in Tab. S1.
Phylogenetic analysis and molecular clock calibration. Interspecific and intraspecific genetic divergences were calculated in MEGA 7 using Tamura-Nei model. Phylogenetic relationships among Acyrtus and Arcos were reconstructed by Bayesian Inference using Mr. Bayes 3.2.6 (Huelsenbeck, Ronquist, 1997). The analysis was performed for two parallel runs of 10 million generations, with four chains each and sampling trees every 1,000 generations. The burn-in value and the effective sample size (ESS) were assessed using Tracer 1.5 (Rambaut et al., 2018). All parameters exceed 200 in ESS values. The consensus tree was obtained from the maximum credibility clades with TreeAnnotator 1.7.5 (Drummond et al., 2012). The appropriate substitution model used was determined using PartitionFinder (Lanfear et al., 2017). We used SYM+I+G model for the first codon position, F81+I for the second position, and GTR+I+G for the third position. We used species of Gobiesox as outgroups based on Conway et al. (2017).
We estimated the divergence times among Acyrtus species using the Relaxed Clock Log Normal model and the Birth/Death prior implemented in BEAUTi & The BEAST 2.5.0 software (Bouckaert et al., 2014). Species of Gobiesocidae, Pseudochromidae, and Grammatidae family were incorporated in the alignment to implement a secondary calibration derived from the results of Near et al. (2013), which were also used by Conway et al. (2017) in the Gobiesocidae family phylogeny. We choose the nodes A (80.3 Ma), B (75.8 Ma), F (42.9 Ma), and G (23.1 Ma) from Conway et al. (2017) to represent the divergence of the last common ancestors between (A) Pseudochromidae + Grammatidae + Gobiesocidae, (B) Grammatidae + Gobiesocidae, (F) all Gobiesocidae species, and (G) Acyrtus + Arcos + Gobiesox. We constrained all node calibrations based on the topology obtained by Conway et al. (2017), which represents the most complete phylogeny obtained for the subfamily Gobiesocinae. The analysis was run in the Cipres Portal (http://www.phylo.org/) using 100 million generations and sampling every 3,000 generations. The effective sample size (ESS) and appropriate burn-in values were visualized in Tracer 1.5 (Rambaut et al., 2018). All parameters exceed 200 in ESS values. We used TreeAnnotator 1.4.3 (Drummond et al., 2012) to obtain the maximum clade credibility tree that was edited in FigTree (Rambaut, 2014) and Inkscape (Free Software Foundation, Boston, USA).
Phylogeographic analysis and demography. We calculated the genetic differentiation between Brazilian species (A. pauciradiatus, Acyrtus sp.n., and Acyrtus sp.) using the FST index (Wright, 1965) implemented in Arlequin 3.5.2.2 (Excoffier, Lischer, 2010). The COI haplotype network was constructed with the median-joining algorithm using PopArt (Bandelt et al., 1999). We performed demographic analysis only for Acyrtus sp.n. Haplotype and nucleotide diversity were estimated in DnaSP6 (Rozas et al., 2017) and neutrality tests were implemented in Arlequin 3.5.2.2. We used Tajima’s D and Fu’s F values to estimate possible events of population expansion. Finally, historical population dynamics were also evaluated using Bayesian Coalescent Skyline method implemented in Beast. We fixed the clock rate using the values obtained by the molecular clock analysis previously cited. The run comprised 10 million generations and samplings every 1,000 generations. We used Tracer 1.5 to check the parameters and to obtain the coalescent analysis output.
Results
Acyrtus simon Gasparini & Pinheiro, new species
urn:lsid:zoobank.org:act:242B5ECF-E791-41DD-92E3-0BC8BA496910
(Fig. 1; Tab. 1)
Arcos sp. —Gasparini, Floeter, 2001:1646 [Trindade Island]. —Pereira-Filho et al., 2011:204 [Trindade Island]. —Simon et al., 2013:2123 [Trindade Island].
Acyrtus sp. —Macieira et al., 2015:390 [Trindade Island]. —Pinheiro et al., 2015:5 [Trindade Island]. —Pinheiro et al., 2017:83 [Trindade Island]. —Pinheiro et al., 2018:86 (Supplementary material) [Trindade Island]. —Guabiroba et al., 2020:701 [Martin Vaz Archipelago].
Acyrtus sp. nov. —Gasparini, 2017:78 [Trindade Island].
Holotype. CIUFES 2915, 26.41 mm SL, Brazil, Trindade Island, Calheta, 28 Jun 2009, H. T. Pinheiro & J. L. Gasparini (Fig. 1).
Paratypes. CAS-ICH 247280, 1, 22.21 mm SL; CIUFES 2914, 1, 26.00 mm SL; CIUFES 4448, 1 (dissected specimen), 32.23 mm SL; MNRJ 52794, 1, 24.71 mm SL; MZUSP 125855, 2, 21.81–25.44 mm SL; NPM 6839, 1, 25.79 mm SL; ZUEC 17336, 1, 30.60 mm SL, same data as for the holotype.
FIGURE 1| Acyrtus simon, holotype CIUFES 2915, 26.41 mm SL. A. Specimen alive, photo taken by J. L. Gasparini in June 2009; B–D. Specimen preserved, photo taken by R. M. Macieira on 31 October 2020. E. X-ray taken by M. M. Mincarone on 11 May 2022.
Diagnosis. Acyrtus simon differs from A. artius by having a deeper head depth (19–21% vs. 14–18% SL), a larger pelvic disc (36–39% vs. 29–36% SL), larger eye diameter (32–40% vs. 24–31% HL), and number of pectoral-fin rays (21–23 vs. 24–27) (Tabs. 1–2). Acyrtus simon can be distinguished from A. lanthanum by a deeper head depth (19–21% vs. 15–18% SL), larger eye diameter (32–40% vs. 24–31% HL), and number of pectoral-fin rays (21–23 vs. 24–25) (Tabs. 1–2). The new species differs from A. rubiginosus by a deeper head depth (19–21% vs. 13–16% SL), a larger pelvic disc (36–39% vs. 27–31% SL), wider pelvic disc (30–36% vs. 22–30% SL), larger eye diameter (32–40% vs. 23–28% HL), and number of pectoral-fin rays (21–23 vs. 24–27) (Tabs. 1–2). It differs from A. pauciradiatus by a larger pelvic disc (36–39% vs. 27–34% SL), shallower caudal peduncle (8–10% vs. 10–11% SL), longer caudal peduncle (10–17% vs. 5–8% SL), shorter anus–disc distance (6–12% vs. 12–13% SL), longer anus–anal fin distance (11–15% vs. 6–9% SL), longer snout length (20–33% vs. 9–15% HL), and narrower interorbital space (18–29% vs. 40–45% HL) (Tabs. 1–2). Acyrtus simon also differs from Acyrtus arturo by having shorter anus–disk distance (6–12 vs. 13–18% SL), longer anus–anal fin distance (11–15 vs. 4–9% SL), and longer snout length (20–33 vs. 10–15% HL) (Tabs. 1–2). It also differs from Arcos nudus [= Acyrtus nudus] by havinga different number of pectoral-fin rays (21–23 vs. 23–25) (Tab. 1).
TABLE 1 | Proportional measurements and counts of type specimens of Acyrtus simon. H = Holotype.
CIUFES 2915 (H) | CAS 247280 | CIUFES 2914 | MNRJ 52794 | MZUSP 125855 | MZUSP 125855 | NUPEM 6839 | ZUEC 17336 | |
Total length (in mm) | 32.19 | 26.94 | 30.91 | 30.26 | 32.94 | 26.50 | 31.68 | 36.38 |
Standard length (SL, in mm) | 26.41 | 22.21 | 26.00 | 24.71 | 25.44 | 21.81 | 25.79 | 30.60 |
Measurements in % of SL | ||||||||
Head length | 33.7 | 36.5 | 34.9 | 35.0 | 47.3 | 40.9 | 36.5 | 34.2 |
Head width | 40.7 | 35.1 | 39.4 | 39.7 | 32.9 | 36.3 | 39.1 | 40.3 |
Head depth | 20.1 | 18.8 | 20.5 | 20.5 | 19.2 | 19.2 | 21.1 | 19.9 |
Pelvic disc length | 37.2 | 37.9 | 38.1 | 38.9 | 39.2 | 36.2 | 37.1 | 37.8 |
Pelvic disc width | 34.6 | 30.3 | 34.6 | 35.6 | 33.6 | 34.3 | 32.8 | 35.6 |
Pre-dorsal distance | 68.7 | 64.1 | 68.5 | 67.7 | 65.4 | 65.5 | 67.7 | 65.7 |
Pre-anal distance | 71.2 | 71.2 | 74.6 | 72.1 | 70.2 | 71.9 | 71.8 | 71.6 |
dorsal length | 17.3 | 20.7 | 14.3 | 16.5 | 20.9 | 16.4 | 18.4 | 15.7 |
Pectoral length | 20.6 | 16.3 | 19.0 | 15.4 | 16.2 | 18.4 | 16.9 | 15.9 |
Caudal Peduncle length | 12.9 | 09.5 | 15.0 | 14.6 | 16.9 | 11.0 | 12.0 | 10.8 |
Caudal peduncle depth | 09.0 | 09.6 | 08.1 | 09.8 | 08.2 | 09.6 | 09.4 | 08.6 |
Anus-Disk distance | 07.1 | 09.5 | 09.1 | 08.7 | 05.8 | 12.4 | 07.3 | 11.2 |
Anus-anal fin distance | 11.4 | 11.5 | 12.9 | 13.4 | 12.1 | 12.7 | 14.5 | 11.5 |
Head length (HL, in mm) | 8.91 | 8.12 | 9.09 | 8.65 | 12.05 | 8.94 | 9.43 | 10.47 |
Measurements in % of HL | ||||||||
Snout length | 32.5 | 28.4 | 26.8 | 25.8 | 21.9 | 20.0 | 30.2 | 27.6 |
Eye diameter | 40.1 | 38.0 | 36.5 | 35.7 | 32.9 | 32.2 | 39.6 | 39.9 |
Interorbital space | 24.2 | 26.1 | 18.2 | 18.3 | 20.4 | 19.2 | 29.2 | 24.0 |
Counts | ||||||||
Dorsal-fin rays | 8 | 9 | 9 | 8 | 8 | 9 | 9 | 8 |
Anal-fin rays | 6 | 7 | 8 | 7 | 6 | 8 | 8 | 8 |
Principal caudal-fin rays | 5+5 | 5+5 | 5+5 | 5+5 | 5+5 | 5+5 | 5+5 | 5+5 |
Procurrent caudal-fin rays | 6+5 | 7+6 | 7+5 | 6+5 | 6+5 | 5+5 | 7+7 | 6+5 |
Pectoral-fin rays | 21 | 22 | 23 | 22 | 21 | 23 | 25 | 23 |
Pelvic-fin rays | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
Vertebrae | 12+17 | 11+18 | 12+17 | 12+17 | 12+17 | 12+17 | 12+17 | 13+16 |
Description. Meristic and proportional measurements of the holotype and seven paratypes given in Tab. 1. Dorsal-fin rays 8 (8–9). Anal-fin rays 6 (6–8). Principal caudal-fin rays 5+5. Procurrent caudal-fin rays 6 (5–7) + 5 (5–7). Pectoral-fin rays 21 (21–23; one with 25). Pelvic-fin rays I,4 (I, 4). Vertebrae 12 (11–13) + 17 (16–18). Body moderately depressed anteriorly, depth 4.9 (4.7–5.3) in SL. Head depressed, head width 2.4 (2.4–3.0) and head length 2.9 (2.1–2.9) in SL. Snout steep with a rounded outline, 3.1 (3.3–4.9) in head length. Posterior nostril surrounded by low fleshy rim and situated in front of anterior edge of eye; anterior nostril tubular, with a bifurcated cirri extending from posterior margin. Mouth terminal, upper lip broad, much wider in front of snout than on the sides. Upper jaw with 2+2 incisiform teeth anteriorly, followed by a single row of 10 small coniform teeth. Lower jaw with 2+2 incisiform teeth anteriorly, followed by single row of 6 coniform teeth. Diameter of eye 2.5 (2.5–3.1) and interorbital space 4.1 (3.4–5.4) in HL. Adhesive disc length 2.7 (2.5–2.7) and width 2.9 (2.8–3.3) in SL; 8 (7–9) transverse rows of papillae across width of disc region A; 10 (9–12) transverse rows of papillae across width of disc region B; 5–5 (3–5) longitudinal rows of papillae across width of disc region C. Pectoral length 4.8 (5.2–6.1) in SL. Pre-dorsal distance 1.4 (1.4–1.5). Dorsal length 5.7 (4.7–6.9). Caudal peduncle length 7.7 (5.9–10.4) and depth 11.0 (10.1–12.3) in SL.
TABLE 2 | Measurement comparisons among Acyrtus species. Data for A. simon and A. pauciradiatus were obtained in the present study. Data for A. artius, A. lanthanum, and A. rubiginosus were taken from Conway et al. (2014), and A. arturo from Tavera et al. (2021).
Acyrtus simon | A. pauciradiatus | A. artius | A. lanthanum | A. rubiginosus | A. arturo | |
Proportion in standard length | ||||||
Head length | 34–47 | 40–44 | 44–47 | 39–43 | 32–39 | 41–43 |
Head depth | 19–21 | 18–2 | 14–18 | 15–18 | 13–16 | 19–27 |
Pelvic disc length | 36–39 | 27–34 | 29–36 | 30–39 | 27–31 | 34–39 |
Pelvic disc width | 30–36 | 32–36 | 29–34 | 31–38 | 22–30 | 31–32 |
Pre-dorsal distance | 64–69 | 67–71 | 65–71 | 64–73 | 61–71 | 62–72 |
Pre-anal distance | 70–75 | 69–76 | 67–78 | 71–77 | 66–80 | 70–76 |
Caudal Peduncle length | 10–17 | 05–08 | 09–15 | 08–11 | 09–13 | 03–11 |
Caudal peduncle depth | 08–10 | 10–11 | 07–10 | 09–14 | 07–09 | 07–09 |
Anus-Disk distance | 06–12 | 12–13 | 08–15 | 04–08 | 12–14 | 13–18 |
Anus-anal fin distance | 11–15 | 06–09 | 08–13 | 10–14 | 12–14 | 04–09 |
Proportion in head length | ||||||
Snout length | 20–33 | 09–15 | 20–27 | 22–30 | 26–32 | 10–15 |
Diameter eye | 32–40 | 24–33 | 24–31 | 24–31 | 23–28 | 26–33 |
Interorbital | 18–29 | 40–45 | 14–21 | 15–21 | 18–25 | 18–25 |
Color in alcohol. Body overall pale, with orange blotches and bands on the dorsal side and on the head; fins hyaline; pupils hyaline with black margin; orange blotches on the iris (Fig. 1).
Coloration in life. Based on color photographs of live specimens (Figs. 1–2): body with variable red and white bands covered by small red spots; white bands might present red blotches; bands wider anteriorly and narrowing towards the caudal fin; pupil rounded and black, with thin white margin; white and red stripes and bands radiating outward from pupil across iris; iris also with thin white margin; first one-third of pectoral fin red, the posterior part hyaline; dorsal fin over red band red, and over white band hyaline with small red spots; caudal fin with variable white and red bands.
Geographical distribution and habitat. Acyrtus simon is only known from Trindade Island, situated 1,160 km from the Brazilian coast. There are unconfirmed records for its presence in the Martin Vaz Archipelago (Guabiroba et al., 2020), which lies 40 km from Trindade. The species lives from intertidal habitat down to reefs 15 m depth (Fig. 2A). Acyrtus simon is commonly found taking shelter in cavities used by Diadema antillarum during the day (Fig. 2B), often sharing the protection from predators offered by spines with the Vitória-Trindade Chain (VTC) endemic Hypleurochilus brasil Pinheiro, Gasparini & Rangel, 2013, Apogon americanus Castelnau, 1855 and a number of others hosts.
FIGURE 2| Acyrtus simon in the natural environment at Trindade Island. A. Photos taken during a night dive by J. L. Gasparini; and B. During the day by J-C. Joyeux.
Etymology. The specific name honors Thiony Emanuel Simon, our ichthyologist friend, who dedicated his career to study reef fishes, especially the fish biodiversity of the Vitória-Trindade Chain. To be treated as a noun in apposition.
Popular name. Thiony’s clingfish; Peixe-ventosa de Thiony.
TABLE 3 | Interspecific and intraspecific (blue boxes) divergence among species of Acyrtus, Arcos, and Gobiesox (percentage values – %).Acy sim: Acyrtus simon; Acy sp: Acyrtus sp; Acy pau: Acyrtus pauciradiatus; Acy aff arti: Acyrtus aff. artius; Acy arti: Acyrtus artius; Acy lan: Acyrtus lanthanum; Acy rub 1: Acyrtus rubiginosus lineage 1; Acy rub 2: Acyrtus rubiginosus lineage 2; Acy artu: Acyrtus arturo; Arc nud: Arcos nudus; Arc ery: Arcos erythrops; Gob pun: Gobiesox punctulatus; Gob rhe: Gobiesox rhessodon; Gob mae: Gobiesox maeandricus.
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | |
1. Acy sim | 0.11 | ||||||||||||
2. Acy sp | 1.25 | 0.23 | |||||||||||
3. Acy pau | 1.85 | 1.93 | 0.09 | ||||||||||
4. Acy aff arti | 5.71 | 6.60 | 6.10 | – | |||||||||
5. Acy arti | 6.60 | 7.61 | 7.85 | 7.08 | 0.45 | ||||||||
6. Acy lan | 7.62 | 10.31 | 10.46 | 11.16 | 10.09 | 0.40 | |||||||
7. Acy artu | 19.36 | 19.63 | 19.55 | 19.30 | 17.59 | 20.51 | 0.00 | ||||||
8. Acy rub 1 | 19.42 | 18.50 | 18.84 | 17.92 | 18.08 | 20.03 | 20.63 | 0.96 | |||||
9. Acy rub 2 | 20 | 19.68 | 20.08 | 19.80 | 19.06 | 19.46 | 20.91 | 5.81 | 0.12 | ||||
10. Arc nud | 19.94 | 20.57 | 19.64 | 21.38 | 20.52 | 19.38 | 21.33 | 20.89 | 19.18 | – | |||
11. Arc ery | 31.80 | 31.63 | 31.12 | 30.60 | 30.82 | 30.46 | 31.47 | 32.00 | 33.79 | 31.33 | – | ||
12. Gob pun | 26.28 | 27.41 | 26.07 | 27.22 | 25.77 | 25.27 | 25.33 | 28.31 | 29.57 | 29.24 | 36.53 | – | |
13. Gob mae | 26.97 | 26.53 | 27.68 | 28.53 | 30.53 | 27.69 | 27.95 | 26.98 | 27.19 | 27.88 | 36.10 | 27.55 | – |
14. Gob rhe | 28.35 | 28.62 | 29.89 | 29.22 | 28.93 | 28.41 | 28.64 | 28.42 | 27.86 | 27.56 | 37.69 | 29.57 | 18.55 |
Conservation status. Acyrtus simon is endemic of Trindade Island. Endemic fishes from this island have not been assessed by the International Union for Conservation Nature (IUCN, 2021), with the exception of Scartella poiti Rangel, Gasparini & Guimarães, 2004; it is considered Vulnerable (VU) due the possibility of habitat degradation associated with its limited distributional range. The latest Brazilian Red List (Portaria MMA Nº 148, de 7 de junho de 2022) considers several Trindade Island endemic species as VU (i.e., Halichoeres rubrovirens Rocha, Pinheiro & Gasparini, 2010, Malacoctenus brunoi Guimarães, Nunan & Gasparini, 2010, Stegastes trindadensis Gasparini, Moura & Sazima, 1999, and Sparisoma rocha Pinheiro, Gasparini & Sazima, 2010, but not S. poiti nor Elacatinus pridisi Guimarães, Gasparini & Rocha, 2004), possibly for the same reasons, and in addition to the risks of fishing and ornamental trade. Therefore, Acyrtus simon is recommended to be categorized as VU according to the IUCN categories and criteria (IUCN Standards and Petitions Subcommittee, 2019).
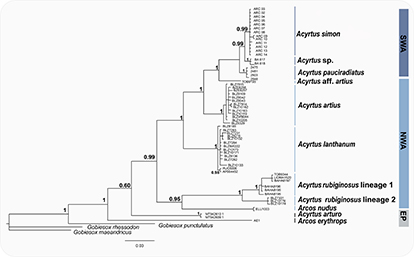
FIGURE 3| Bayesian phylogeny of Acyrtus based on the COI gene. Bayesian posterior probability values are shown and the biogeographic province of lineages presented in the right. SWA: southwestern Atlantic; NWA: northwestern Atlantic; EP: eastern Pacific.
Phylogenetic analyses of Acyrtus. COI sequences of 573 bp were obtained for 73 individuals of 20 species. Our analyses suggest the northwestern Atlantic as the center of the diversification of Acyrtus, and show that the genus is composed by at least nine species, forming a non-monophyletic group of three major clades (Fig. 3). One clade is composed by Caribbean and Brazilian species, another clade by the Caribbean A. rubiginosus lineage 1, A. rubiginosus lineage 2 and Arcos nudus, and a third solely composed by A. arturo, from the tropical eastern Pacific. Although three species (A. artius, A. lanthanum and A. rubiginosus) present a high distributional overlap along most of the Caribbean, our analysis revealed two cryptic species supported by high values of posterior probability, Acyrtus aff. artius from Tobago Island, and Acyrtus aff. rubiginosus from Belize (Fig. 3). Three species are endemic to the Brazilian Province and form a monophyletic group, which has Acyrtus aff. artius as the closest related species (Fig. 3). Brazilian species present small distributions: Acyrtus simon is restricted to Trindade Island, A. pauciradiatus, is restricted to Fernando de Noronha Archipelago and Rocas Atoll, and the undescribed species (Acyrtus sp., Fig. 3) is only known from the Salvador region, on the northeastern Brazilian coast. In general, Caribbean species present higher interspecific divergence than Brazilian species (Tab. 3).
TABLE 4 | Divergence times (Mya) among Acyrtus species and Arcos nudus, and the 95% highest posterior density.
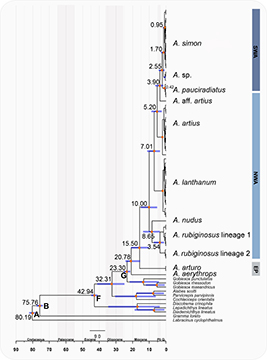
FIGURE 4| Bayesian estimates of divergence time based on the mitochondrial COI gene. Posterior probability values reached 1–0.99 for all the main nodes (represented by the orange circle). The horizontal purple bars indicate 95% credibility intervals of node age estimation. The calibration nodes represent the divergence of the last common ancestors between (A) Pseudochromidae + Grammatidae + Gobiesocidae, (B) Grammatidae + Gobiesocidae, (F) all Gobiesocidae species, and (G) Acyrtus + Arcos + Gobiesox (see Conway et al., 2017).
The estimated date for the most recent common ancestor of the Atlantic species and A. arturo (eastern Atlantic) is at least 15 Mya, and the origin of Arcos nudus and the Acyrtus rubiginosus clade was among the oldest diversification events of the genus within the Atlantic (Fig. 4; Tab. 4). Most of the diversification in Acyrtus is recent, occurring during the Pliocene and Pleistocene. The Brazilian clade is the youngest, diversifying around 2.55 Mya, and the divergence time between Acyrtus simon and Acyrtus sp. is around 1.7 Mya (Fig. 4). Divergences were higher among Caribbean species than among Brazilian species.
TABLE 5 | FST index among Acyrtus in the Southwestern Atlantic. All values were significant (p<0.05).
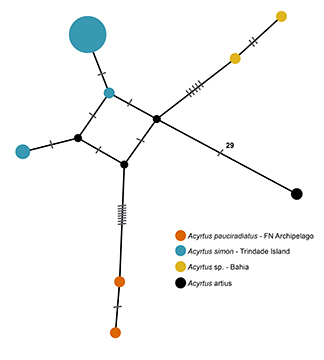
FIGURE 5| Haplotype network of Acyrtus artius and the representatives of Acyrtus in the southwestern Atlantic. FN = Fernando de Noronha.
Evolutionary history of Acyrtus simon. The close relationship between Acyrtus simon, from Trindade Island, and Acyrtus sp. from the Brazilian coast was also revealed in the haplotype network (Fig. 5) and through smaller FST values than Acyrtus pauciradiatus (Tab. 5). Acyrtus simon, represented by only three haplotypes (Fig. 5), presented low haplotype and nucleotide diversity (Hd = 0.362; π = 0.001). Neutrality tests presented negative values (Tab. 6), though the Skyline plot did not present evidence of recent population growth in Trindade Island (Fig. 6) due to the low number of haplotypes.
TABLE 6 | Diversity and neutrality indexes calculated for Acyrtus simon, Trindade Island.h = haplotypes number; Hd = haplotype diversity; SD Hd = standard deviation of Hd; π = nucleotide diversity; SD π = standard deviation of π.
FIGURE 6| Bayesian skyline plot showing the effective population size fluctuation of Acyrtus simon through time (Mya) (black line: median estimation; purple: confidence interval).
Discussion
Our study presents the first phylogeny of the genus Acyrtus including all known representatives, revealing: 1) insights on the evolutionary history of the genus; 2) the current paraphyletic status of the genus; 3) the Brazilian Province species complex as a monophyletic group; 4) and the presence of undescribed and cryptic species. Although our phylogenetic inference is based on a single DNA marker, our conclusions are based on high statistical support.
The absence of reliable fossil data for the family Gobiesocidae (but see Schwarzhans et al., 2017) constitutes as one of the main barriers to the understanding of its evolutionary history. The secondary calibration from the divergence times obtained by Near et al. (2013), and used by Conway et al. (2017), was used here to study the genus Acyrtus. Near et al. (2013) estimated the origin of the family Gobiesocidae in the Eocene, around 42.9 Mya, which is consistent with the emergence of most reef fish families, between 66 and 34 Mya (Bellwood, 2015). Around 39 Mya (Conway et al., 2017), shortly after the origin of the family Gobiesocidae, its New World lineage (subfamily Gobiesocinae) was formed during a period marked by great diversification in the Tethys Sea (Renema et al., 2008). It is possible that earlier Gobiesocidae lineages originated in the Tethys Sea and then colonized both the Atlantic/eastern Pacific and the Indian-western Pacific oceans (Floeter et al., 2008). However, earlier Gobiesocidae lineages could also have had their origins in any of the oceans, using the Tethys as a passage. For instance, Gobiosomatini (Gobiidae) (Thacker, 2015), also endemic of the new world, originated in a period similar to Gobiesocinae, likely via dispersal from the western Indian Ocean through the Tethys passage (Thacker, 2015). While little can be concluded about the center of origin of Gobiesocidae, it seems that the Tethys Sea was important for dispersal and early diversification in the family.
After the rise of the Gobiesocinae, Acyrtus lineagesand Arcos erythrops diverged much earlier than the closure of the Isthmus of Panama, around ~21 Mya. Therefore, this event did not influence the divergence between Gobiesocinae genera as commonly seen in other groups (Lessios, 2008). Tavera et al. (2021) found a similar divergence time for Arcos erythrops, although the placement of this species did not evidence the monophyly of Arcos and Acyrtus. Despite this, our results support the topology obtained by Conway et al. (2020) based on seven different genes. Nevertheless, further investigation of the Arcos phylogeny with broader taxon sampling is needed.
Our results also show the emergence of the western Atlantic Acyrtus at least 15 Mya (the age of the last common ancestor of Acyrtus + Arcos nudus) in a period of origin and diversification of most reef fish genera (Bellwood et al., 2015). Moreover, the origin of Acyrtus coincides with the emergence of the Amazon barrier, which could have prevented an earlier dispersal from the Caribbean to the Brazilian Province. It could explain the latter diversification of Acyrtus in the history of this genus (~3.9 Mya). In this case, the glacioeustatic sea-level changes of the Pleistocene/Pliocene could have contributed to the connectivity between regions, and to the crossing of the biogeographic barrier, as suggested for many other reef fishes (Rocha, 2003).
The diversification of the Brazilian complex of species started during the Pleistocene and these three species present restricted distributions: two are endemic to oceanic islands, and a third is only known from a narrow geographic range of the Brazilian northeastern coast. There are two hypotheses for speciation and colonization routes. The first involves the colonization of the Fernando de Noronha archipelago, with posterior colonization of the Brazilian coast, which recently colonized Trindade Island. This hypothesis is corroborated by the fact that a few Caribbean species are also found in Fernando de Noronha but not along the Brazilian coast (Rocha, 2003). This archipelago could be a gateway for Caribbean species to colonize Brazilian waters. A second hypothesis would involve the colonization of the Brazilian coast first, with a subsequent and earlier colonization of Fernando de Noronha, and a more recent colonization of Trindade also from the coast. Some reef fish lineages were more widely distributed along the southwestern Atlantic in the past, persisting as relicts in restricted locations (Rocha et al., 2010; Pinheiro et al., 2017). The exposure of seamounts during low sea-levels could have favored the colonization of the Vitória-Trindade Chain (Macieira et al., 2015; Pinheiro et al., 2017).
Even though we observed low genetic diversity in our results for A. simon, there was no sign of population bottlenecks or expansion in the neutrality test, differently from other VTC endemics (Pinheiro et al., 2017). Differently from species with wider distribution (Pinheiro et al., 2017), the restriction of Acyrtus to shallow reefs and its absence on the VTC seamounts may have constrained its connectivity between the coast and the islands and limited the input of new haplotypes. Recent population expansions are seen in many species along the western Atlantic (da Silva et al., 2015; Liedke et al., 2020) that seem to be related to a ~90% increase in reef area caused by the rise in sea-level following the last glacial maximum (Ludt, Rocha, 2015).
Both Conway et al. (2014) and our study show three undescribed Caribbean Acyrtus species, one of them closely related to Acyrtus artius and the two other to A. rubiginosus. The existence of many hidden lineages in the same genus may be attributed to the species small size, cryptic behavior, and a low morphological divergence. The latter, in particular, is commonly related to recent speciation and/or stabilizing selection on the ancestral phenotype (Milá et al., 2017). A similar pattern is found for Gouania (Gobiesocidae) in the Mediterranean Sea, where recent diversification and cryptic lineages were recognized by Wagner et al. (2019). These results evidence that future molecular studies for Gobiesocidae should lead to the discovery of many cryptic species. The most likely cause for the high diversification in this family (even within provinces) is related to their weak dispersal potential, small size, sedentary habit, and demersal eggs (Pires, Gibran, 2011). These life-history characteristics are among the most important drivers of speciation in the Brazilian Province (Pinheiro et al., 2018; Mazzei et al., 2021; Simon et al., 2021), and other taxonomic groups that share similar traits (e.g., Labrisomidae and Gobiidae) also show strong genetic structure and cryptic speciation within the Great Caribbean (Baldwin et al., 2011; Victor, 2014). Additionally, the distribution of the closely-related Brazilian Acyrtus species in different environments (mainland coast and oceanic islands) and localities with distinct levels of isolation suggests that ecological and peripatric speciation processes might be important drivers of diversification in Gobiesocidae (Rocha et al., 2005; Pinheiro et al., 2017; Simon et al., 2021).
Our phylogenetic inference discloses the paraphyletic nature of Acyrtus, which includes Arcos nudus in its clade. The first Acyrtus species (A. rubiginosus) was described in 1868, originally assigned to the genus Sicyases, a valid genus described based on Sicyases sanguineus Müller & Troschel, 1843. The genera Acyrtus and Arcos were described in the same article (Schultz, 1944), based on Acyrtus rubiginosus and Arcos erythrops, respectively. Arcos nudus was originally described by Linnaeus (1758) as Cyclopterus, a valid and now monotypic genus described based on Cyclopterus lumpus Linnaeus, 1758 (Cyclopteridae or lumpfishes). It was later reassigned as Gobiesox nudus by Briggs (1955), and afterwards as Arcos nudus by Fernholm, Wheeler (1983). All other Arcos species are from the tropical eastern Pacific and were originally described as Gobiesox, a valid genus described based on Gobiesox cephalus Lacépède, 1800.
Our phylogenetic analyses indicate that Arcos nudus should be reassigned to Acyrtus. This species shares several distinguishing morphological characters with other Acyrtus species, including large secretory cells present inside the groove present in the subopercular spine (Conway et al., 2014). Therefore, according to our data, the genus Arcos seems to be exclusive to the eastern Pacific, while most Acyrtus are found in the western Atlantic. Additional studies including more eastern Pacific Gobiesocidae species are necessary to better assess the status of Acyrtus arturo, from Malpelo Island (Tavera et al., 2021), and Arcos nudus, from the Atlantic, which could belong to other clades. Alternatively, a more complete phylogenetic analysis including all Arcos species could also show clades uniting Acyrtus and Arcos erythrops, suggesting the unification of Acyrtus and Arcos in a single genus. Finally, our results highlight that a broad sampling in Gobiesocidae family will bring important insights about evolutionary patterns of cryptobenthic fishes.
Comparative material examined. All from Brazil. Acyrtops beryllinus: CIUFES 2930, 3, 12.36–16.52 mm SL. CIUFES 130851, 1, 25.12 mm SL; Acyrtus pauciradiatus: CIUFES 2475, 1, 11.08 mm SL. CIUFES 2481, 2, 9.36–11.24 mm SL. CIUFES 2503, 1, 19.02 mm SL. CIUFES 2548, 1, 10.03 mm SL. Gobiesox barbatulus: CIUFES 307, 6, 20.26–47.64 mm SL. CIUFES 1368, 14, 40.77–57.50 mm SL. CIUFES 2923, 3, 34.30–49.72 mm SL. CIUFES 2883, 6, 13.14–30.47 mm SL. Tomicodon australis: CIUFES 130269, 2, 19.78–25.31 mm SL. CIUFES 130294, 2, 14.11–19.12 mm SL. CIUFES 130712, 1, 15.42 mm SL. CIUFES 130853, 13, 20.36–31.21 mm SL.
Acknowledgments
We thank the Brazilian Navy for support during scientific expeditions to Trindade Island. We also thank Raphael M. Macieira, C. L. S. Sampaio, R. G. Santos for support in the field and with samples, Fabio Di Dario, Kevin W. Conway and Sergio R. Floeter for providing valuable literature. TLQ is supported by a FAPES doctoral fellowship (Fundação de Amparo à Pesquisa e Inovação do Espírito Santo), JLG by a CAPES doctoral fellowship (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and HTP by a FAPESP JP fellowship (Fundação de Amparo à Pesquisa do Estado de São Paulo; 2019/24215–2; 2021/07039–6). LAR and HTP are funded by the Hope for Reefs initiative of the California Academy of Sciences. We thank Michael M. Mincarone, Raphael M. Macieira, and an anonymous reviewer for their insightful and constructive comments on the various versions of our manuscript.
References
Baldwin CC, Castillo CI, Weigt LA, Victor BC. Seven new species within western Atlantic Starksia atlantica, S. lepicoelia, and S. sluiteri (Teleostei, Labrisomidae), with comments on congruence of DNA barcodes and species. Zookeys. 2011; 79:21–72. https://doi.org/10.3897/zookeys.79.1045
Bandelt H-J, Forster P, Rohl A. Median-Joining networks for inferring intraspecific phylogenies. Mol Biol Evol.1999; 16(1):37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Bellwood DR, Goatley CH, Cowman PF, Bellwood O. The evolution of fishes on coral reefs: fossils, phylogenies and functions. In: Mora C, editor. Ecology of Fishes on Coral Reefs. Cambridge: Cambridge University Press; 2015; p.55–63.
Bouckaert R, Heled J, Kühnert D, Vaughan T, Wu C-H, Xie D et al. BEAST 2: A software platform for bayesian evolutionary analysis. PLoS Comput Biol. 2014; 10(4):e1003537. https://doi.org/10.1371/journal.pcbi.1003537
Brandl SJ, Goatley CHR, Bellwood DR, Tornabene L. The hidden half: ecology and evolution of cryptobenthic fishes on coral reefs. Biol Rev Camb Philos Soc. 2018; 93(4):1846–73. https://doi.org/10.1111/brv.12423
Briggs J. A monograph of the clingfishes (Order Xenopterygii). Stanford Ichthyology Bulletin 6. Standford, California: Natural History Museum of Standford University. 1955.
Conway KW, Baldwin C, White MD. Cryptic diversity and venom glands in Western Atlantic clingfishes of the genus Acyrtus (Teleostei: Gobiesocidae). PLoS ONE. 2014;9(5):e97664. https://doi.org/10.1371/journal.pone.0097664
Conway KW, Kim D, Rüber L, Pérez HSE, Hastings PA. Molecular systematics of the New World clingfish genus Gobiesox (Teleostei: Gobiesocidae) and the origin of a freshwater clade. Mol Phylogenet Evol. 2017; 112:138–47. https://doi.org/10.1016/j.ympev.2017.04.024
Conway KW, King CD, Summer AP, Kim D, Hasting PA, Moore GI et al. Molecular phylogenetics of the clingfishes (Teleostei: Gobiesocidae) – Implications for classification. Copeia. 2020; 108(4):886–906. https://doi.org/10.1643/CI2020054
Craig MT, Randall JE. Two new species of the Indo-Pacific clingfish genus Discotrema (Gobiesocidae). Copeia. 2008; 2008(1):68–74. https://doi.org/10.1643/CI-07-025
Drummond AJ, Suchard MA, Xie D, Rambaut A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol. 2012;29(8):1969–73. https://doi.org/10.1093/molbev/mss075
Excoffier L, Lischer HEL. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour. 2010;10(3):564–67. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Fernholm BO, Wheeler A. Linnaean fish specimens in the Swedish Museum of Natural History, Stockholm. J Linn Soc London Zool. 1983; 78:199–286.
Floeter SR, Rocha LA, Robertson DR, Joyeux J-C, Smith-Vaniz WF, Wirtz P et al. Atlantic reef fish biogeography and evolution. J Biogeogr. 2008; 35(1):22–47. https://doi.org/10.1111/j.1365-2699.2007.01790.x
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2022. Available from: http://researcharchive.calacademy.org/research/ ichthyology/catalog.
Fricke R, Chen J-N, Chen W-J. New case of lateral asymmetry in fishes: A new subfamily, genus and species of deep water clingfishes from Papua New Guinea, western Pacific Ocean. C R Biol. 2017; 340(1):47–62. https://doi.org/10.1016/j.crvi.2016.11.002
Gasparini JL, Floeter SR. The shore fishes of Trindade Island, southwestern Atlantic. J Nat Hist. 2001; 35(11):1639–56. https://doi.org/10.1080/002229301317092379
Gasparini JL. Peixes dos recifes e das ilhas de Vitória e adjacências. Espírito Santo, Brasil. Fauna – Editora, Estudos & Projetos LTDA. Gráfica GSA. Vitória. 2017.
Gonçalves EJ, Barbosa M, Cabral HN, Henriques M. Ontogenetic shifts in patterns of microhabitat utilization in the small-headed clingfish, Apletodon dentatus (Gobiesocidae). Environ Biol Fishes. 2002; 63:333–39. https://doi.org/10.1023/A:1014302319622
Guabiroba HC, Pimentel CR, Macieira RM, Cardozo-Ferreira GC, Teixeira JB, Gasparini JL et al. New records of fishes for the Vitória-Trindade Chain, southwestern Atlantic. Check List. 2020; 16(3):699–705. https://doi.org/10.15560/16.3.699
Henriques M, Lourenço R, Almada F, Calado G, Gonçalves D, Guillemaud T et al. A revision of the status of Lepadogaster lepadogaster (Teleostei: Gobiesocidae): sympatric subspecies or a long misunderstood blend of species? Biol J Linn Soc Lond. 2002; 76(3):327–38. https://doi.org/10.1046/j.1095-8312.2002.00067.x
Huelsenbeck JP, Ronquist F. Bayesian analysis of molecular evolution using MrBayes. In: Nielsen R, editor. Statistical Methods in Molecular Evolution. Statistic for Biology and Health. New York: Springer.1997; p.183–226.
International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for using the IUCN Red List categories and criteria. Version 14 [Internet]. Gland; 2019. Available from: https://www.iucnredlist.org/resources/redlistguidelines
International Union for Conservation of Nature (IUCN). The IUCN Red List of Threatened Species. Version 2021-3. Available from: https://www.iucnredlist.org. 2021.
Kumar S, Stecher G, Tamura K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol. 2016; 33(7):1870–74. https://doi.org/10.1093/molbev/msw054
Lanfear R, Frandsen PB, Wright AM, Senfeld T, Calcott B. Partitionfinder 2: New methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol Biol Evol. 2017; 34(3):772–73. https://doi.org/10.1093/molbev/msw260
Lessios HA. The great American schism: divergence of marine organisms after the rise of the central American Isthmus. Annu Rev Ecol Evol Syt. 2008; 39:63–91. https://doi.org/10.1146/annurev.ecolsys.38.091206.095815
Liedke AMR, Pinheiro HT, Floeter SR, Bernardi G. Phylogeography of the banded butterflyfish, Chaetodon striatus, indicates high connectivity between biogeographic provinces and ecosystems in the western Atlantic. Neotrop Ichthyol. 2020; 18(1):e190054. https://doi.org/10.1590/1982-0224-2019-0054
Ludt WB, Rocha LA. Shifting seas: the impacts of Pleistocene sea-level fluctuations on the evolution of tropical marine taxa. J Biogeogr. 2015; 42(1):25–38. https://doi.org/10.1111/jbi.12416
Macieira RM, Simon T, Pimentel CR, Joyeux J-C. Isolation and speciation of tidepool fishes as a consequence of Quaternary sea-level fluctuations. Environ Biol Fishes. 2015; 98:385–93. https://doi.org/10.1007/s10641-014-0269-0
Mazzei EF, Pinheiro HT, Simon T, Moura RL, Macieira RM, Pimentel CR et al. Mechanisms of dispersal and establishment drive a stepping stone community assembly on seamounts and oceanic islands. Mar Biol. 2021; 168(109). https://doi.org/10.1007/s00227-021-03919-7
Milá B, Van Tassel JL, Calderón JA, Ruber L, Zardoya R. Cryptic lineage divergence in marine environments: genetic differentiation at multiple spatial and temporal scales in the widespread intertidal goby Gobiosoma bosc. Ecol Evol. 2017; 7(14):5514–23. https://doi.org/10.1002/ece3.3161
Near TJ, Dornburg A, Eytan RI, Keck BP, Smith WL, Kuhn KL et al. Phylogeny and tempo of diversification in the super radiation of spiny-rayed fishes. Proc Natl Acad Sci USA. 2013; 110(31):12738–43. https://doi.org/10.1073/pnas.1304661110
Pereira-Filho GH, Amado-Filho GM, Guimarães SMPB, Moura RL, Sumida PYG, Abrantes DP et al. Reef fish and benthic assemblage of the Trindade and Martin Vaz Island group, southwestern Atlantic. Braz J Oceanogr. 2011; 59(3):2001–12. Available from: https://bityli.com/hxchez
Pinheiro HT, Gasparini JL, Rangel CA. A new species of the genus Hypleurochilus (Teleostei: Blennidae) from Trindade Island and Martin Vaz Archipelago, Brazil. Zootaxa. 2013; 3709(1):95–100. https://doi.org/10.11646/zootaxa.3709.1.5
Pinheiro HT, Mazzei E, Moura RL, Amado-Filho GM, Carvalho-Filho A, Braga AC et al. Fish biodiversity of the Vitória-Trindade seamount chain, southwestern Atlantic: an update database. PLoS ONE. 2015; 10(3):e0118180. https://doi.org/10.1371/journal.pone.0118180
Pinheiro HT, Bernardi G, Simon T, Joyeux J-C, Macieira RM, Gasparini JL et al. Island biogeography of marine organisms. Nature. 2017;549:82–85. https://doi.org/10.1038/nature23680
Pinheiro HT, Rocha LA, Macieira RM, Caravalho-Filho A, Anderson AB, Bender MG et al. South-Western Atlantic reef fishes: zoogeographical patterns and ecological drivers reveal a secondary biodiversity centre in the Atlantic Ocean. Divers Distrib. 2018; 24(7):951–65. https://doi.org/10.1111/ddi.12729
Pires THS, Gibran FZ. Interdital life: field observations on the clingfish Gobiesox barbatulus in southeastern Brazil. Neotrop Ichthyol. 2011; 9(1):233–40. https://doi.org/10.1590/S1679-62252011005000001
Rambaut A. Figtree, a graphical viewer of phylogenetic trees [Internet]. 2014. Available from: http://tree.bio.ed.ac.uk/software/figtree.
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA. Posterior summarization in bayesian phylogenetics using Tracer 1.7. Syst Biol. 2018; 67(5):901–04. https://doi.org/10.1093/sysbio/syy032
Renema W, Bellwood DR, Braga JC, Bromfield K, Hall R, Johnson KG et al. Hopping hotspots: global shifts in marine biodiversity. Science. 2008; 321(5889):654–57. https://doi.org/10.1126/science.1155674
Rocha LA. Patterns of distribution and processes of speciation in Brazilian reef fishes. J Biogeogr. 2003; 30(8):1161–71. https://doi.org/10.1046/j.1365-2699.2003.00900.x
Rocha LA, Robertson DR, Roman J, Bowen BW. Ecological speciation in tropical reef fishes. Proc R Soc Lond B Biol Sci. 2005; 272(1563):573–79. https://doi.org/10.1098/2004.3005
Rocha LA, Pinheiro HT, Gasparini JL. Description of Halichoeres rubrovirens, a new species of wrasse (Labridae: Perciformes) from the Trindade and Martin Vaz Island group, southeastern Brazil, with a preliminary mtDNA molecular phylogeny of New World Halichoeres. Zootaxa. 2010; 2422:22–30. https://doi.org/10.11646/zootaxa.2422.1.2
Rozas J, Ferrer-Mata A, Sanchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE et al. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol. 2017;34(12):3299–302. https://doi.org/10.1093/molbev/msx248
Schultz LP. A revision of the American clingfishes, family Gobiesocidae, with descriptions of new genera and forms. Proc U S Nat Mus. 1944; 96(3187):47–77. https://doi.org/10.5479/si.00963801.96-3187.47
Schwarzhans W, Carnevale G, Japundžić S, Bradić-Milinović K. Otoliths in situ from Sarmatian (Middle Miocene) fishes of the Paratethys. Part IV: Scorpaenidae, Labridae, and Gobiesocidae. Swiss J Palaeontol. 2017; 136(Suppl. 1):93–108. https://doi.org/10.1007/s13358-017-0124-y
Silva R, Veneza I, Sampaio I, Araripe J, Schneider, Gomes G. High levels of genetic connectivity among populations of yellowtail snapper, Ocyurus chrysurus (Lutjanidae – Perciformes), in the western south Atlantic revealed through multilocus analysis. PLoS ONE. 2015; 10(3):e0122173. https://doi.org/10.1371/journal.pone.0122173
Simon T, Macieira RM, Joyeux J-C. The shore fishes of the Trindade-Martin Vaz insular complex: and update. J Fish Biol. 2013; 82:2113–27. https://doi.org/10.1111/jfb.12126
Simon T, Pinheiro HT, Santos S, Macieira RM, Ferreira YSS, Bernardi G et al. Comparative phylogeography of reef fishes indicates seamounts as stepping stones for dispersal and diversification. Coral Reefs. 2021.
Smith-Vaniz WF. Another new species of the clingfish genus Derilissus from the Western Atlantic (Pisces: Gobiesocidae). Copeia. 1971; 1971(2):291–94. https://doi.org/10.2307/1442829
Sparks JS, Gruber DF. A new mesophotic clingfish (Teleostei: Gobiesocidae) from the Bahamas. Copeia. 2012; 2012(2):251–56. https://doi.org/10.1643/CI-11-124
Tavera J, Rojas-Vélez S, Londoño-Cruz E. A new species of the genus Acyrtus on the eastern Pacific: A cornerstone for the evolution and biogeography of the genus. J Fish Biol. 2021; 99(5):1550–60. https://doi.org/10.1111/jfb.14860
Thacker CE. Biogeography of goby lineages (Gobiiformes: Gobioidei): origin, invasions and extinction throughout the Cenozoic. J Biogeogr. 2015; 42(9):1615–25. https://doi.org/10.1111/jbi.12545
Torres-Hernández E, Betancourt-Resendes I, Díaz-Jaimes P, Angulo A, Espinoza E, Domínguez-Domínguez O. Independent evolutionary lineage of the clingfish Gobiesox adustus (Gobiesocidae) from Isla del Coco, Costa Rica. Rev Biol Trop. 2020; 68(S1):S306–19. https://doi.org/10.15517/rbt.v68iS1.41201
Victor BC. Three new endemic cryptic species revealed by DNA barcoding of the gobies of the Cayman Islands (Teleostei: Gobiidae). J Ocean Sci Found. 2014; 12:25–60. https://doi.org/10.5281/zenodo.1049119
Wagner M, Bračun S, Skofitsch G, Kovačić M, Zogaris S, Iglésias SP et al. Diversification in gravel beaches: A radiation of interstitial clingfish (Gouania, Gobiesocidae) in the Mediterranean Sea. Mol Phylogenet Evol. 2019; 139:106525. https://doi.org/10.1016/j.ympev.2019.106525
Weigt LA, Driskell AC, Baldwin CC, Ormos A. DNA barcoding fishes. In: Kress WJ, Erickson DL, editors. DNA barcodes: methods and protocols, Methods in Molecular Biology. New York: Springer. 2012; p.109–26.
Wright S. The interpretation of population structure by F-statistics with special regard to systems of mating. Evolution. 1965;19(3):395–420. https://doi.org/10.2307/2406450
Authors
![]() Thais L. Quintão1,
Thais L. Quintão1, ![]() João Luiz Gasparini3,
João Luiz Gasparini3, ![]() Jean-Christophe Joyeux2,
Jean-Christophe Joyeux2, ![]() Luiz A. Rocha4 and
Luiz A. Rocha4 and ![]() Hudson T. Pinheiro4,5
Hudson T. Pinheiro4,5 ![]()
[1] Programa de Pós Graduação em Biologia Animal, Dept de Ciências Biológicas, Universidade Federal do Espírito Santo, 29075-910 Vitória, ES, Brazil (TLQ) thaislquintao@hotmail.com.
[2] Departamento de Oceanografia e Ecologia, Universidade Federal do Espírito Santo, 29075-910 Vitória, ES, Brazil. (JCJ) jean.joyeux@ufes.br.
[3] Programa de Pós-Graduação em Ciências Ambientais e Conservação (PPG-CiAC), Instituto de Biodiversidade e Sustentabilidade, Universidade Federal do Rio de Janeiro (NUPEM-UFRJ), 27965-045 Macaé, RJ, Brazil. (JLG) gaspa.vix@gmail.com.
[4] Department of Ichthyology, California Academy of Sciences, 94118 San Francisco, CA, USA. (LAR) Lrocha@calacademy.org.
[5] Centro de Biologia Marinha, Universidade de São Paulo, 11600-000 São Sebastião, SP, Brazil. (HTP) htpinheiro@gmail.com (corresponding author).
Authors’ Contribution 

Thais L. Quintão: Formal analysis, Investigation, Methodology, Visualization, Writing-original draft, Writing-review and editing.
João Luiz Gasparini: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Supervision, Writing-original draft, Writing-review and editing.
Jean-Christophe Joyeux: Data curation, Formal analysis, Investigation, Writing-review and editing.
Luiz A. Rocha: Investigation, Methodology, Validation, Writing-review and editing.
Hudson T. Pinheiro: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Fish collection was authorized by the Brazilian Environmental Agency, Instituto Chico Mendes de Conservação da Biodiversidade (SISBIO 12786–1 and 20880–2), and was in accordance with the ethical principles for animal experimentation, approved by the Ethics Committee for the Use of Animals of the Universidade Federal do Espírito Santo (CEUA-UFES 017–2009).
Competing Interests
The authors declare no competing interests.
How to cite this article
Quintão TL, Gasparini JL, Joyeux J-C, Rocha LA, Pinheiro HT. Recent dispersal and diversification within the clingfish genus Acyrtus (Actinopterygii: Gobiesocidae), with the description of a new western Atlantic species. Neotrop Ichthyol. 2022; 20(3):e210137. https://doi.org/10.1590/1982-0224-2021-0137
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2022 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted May 30, 2022 by Michael Mincarone
Accepted May 30, 2022 by Michael Mincarone
![]() Submitted September 19, 2021
Submitted September 19, 2021
![]() Epub October 03, 2022
Epub October 03, 2022