![]() Oscar A. Shibatta1
Oscar A. Shibatta1 ![]() ,
, ![]() Marcelo S. Rocha2 and
Marcelo S. Rocha2 and ![]() Renildo R. de Oliveira3
Renildo R. de Oliveira3
PDF: EN XML: EN | Cite this article
Abstract
A new species of Rhyacoglanis is described from the rapids of Jatobal, Pará State, Brazil. This species differs from the congeners by fusing hypurals 3, 4, and 5 (vs. hypural 5 free). It also differs from the other species, except for Rhyacoglanis epiblepsis, by the color pattern with numerous dark spots on the body, short post-cleithral process, rounded pectoral and caudal fins, and incomplete lateral line. This species is known only from the type locality, which is currently flooded by the Tucuruí reservoir.
Keywords: Biodiversity, Bumblebee catfish, Ostariophysi, Systematics, Taxonomy.
Uma nova espécie de Rhyacoglanis é descrita das corredeiras de Jatobal, estado do Pará, Brasil. Essa espécie difere das congêneres pela fusão dos hipurais 3, 4 e 5 (vs. hipural 5 livre). Também difere das demais espécies, exceto de Rhyacoglanis epiblepsis, pelo padrão de colorido com inúmeras pintas escuras no corpo, processo pós-cleitral curto, nadadeiras peitoral e caudal arredondadas, e linha lateral incompleta. Essa espécie é conhecida apenas da localidade tipo, que atualmente se encontra inundada pelo reservatório de Tucuruí.
Palavras-chave: Bagrinho, Biodiversidade, Ostariophysi, Sistemática, Taxonomia.
Introduction
Rhyacoglanis Shibatta & Vari, 2017, consists of five small bumblebee catfish species (Shibatta, Vari, 2017). They are easily recognized by the body’s color pattern that alternates dark brown bands on a yellowish background, grayish head with a light spot on the cheek, a dark stripe along the middle of the upper and lower caudal-fin lobes confluent with dark caudal peduncle blotch, lateral line elongated, and premaxillary tooth plate posterolaterally pointed.
Until the description of Rhyacoglanis by Shibatta, Vari (2017), species of this genus had already been identified as Pseudopimelodus variolosus Miranda Ribeiro, 1914, Pseudopimelodus zungaro zungaro by Mees, 1974, and even as Microglanis sp. (Castro, 1999). However, the hypothesis of monophyly of the genus was presented by Shibatta, Vari (2017), pointing out Rhyacoglanis as Pseudopimelodus sister group.
The genus is widely distributed in South America, occurring in the Orinoco, Amazonas, and Paraná-Paraguay river basins. Three species are known from the Amazon region: Rhyacoglanis pulcher (Boulenger, 1887) from the upper rio Amazonas, R. epiblepsis Shibatta & Vari, 2017 from rio Madeira, and R. seminiger Shibatta & Vari, 2017 from the upper rio Tapajós. The analysis of specimens from the lower rio Tocantins in Pará highlighted a new species from the rapids of Jatobal, a region flooded by the Tucuruí reservoir. This new species is described herein.
Material and methods
Measurements of the body were made point-to-point, on the left side of the specimens, with a digital caliper of 0.01mm accuracy. Proportions of standard length (SL) and head length (HL) were rounded to 0.1 mm precision. A total of 29 morphometric variables of 14 larger and best-preserved specimens (including the holotype) were measured for statistical purposes. Head laterosensory pores were surveyed in five specimens (MZUEL 20873) and identified according to Shibatta (2019). The counts, whenever possible to observe, were made in 10 specimens preserved in ethanol (MZUEL 20873), two cleared and stained (c&s; INPA 7957), and seven x-rays (holotype and INPA 59634). The total number of specimens can vary as results because not all characters were easily visible in consequence of skin cover (in alcohol-preserved specimens) and overlaps of structures in x-rayed specimens. In general, only diaphanized specimens allowed safer counts of all variables than other preparations. Specimens were cleared and stained according to Dingerkus, Uhler (1977); osteological nomenclature followed Shibatta (2019). The method of obtaining and analyzing the data and the comparative material are presented in detail by Shibatta, Vari (2017). The institutional acronyms used are INPA, Instituto Nacional de Pesquisas da Amazônia, Manaus, and MZUEL, Museu de Zoologia da Universidade Estadual de Londrina, Londrina.
Results
Rhyacoglanis rapppydanielae, new species
urn:lsid:zoobank.org:act:53858744-FB46-4BE9-B939-B5DC79E3E551
(Figs. 1–6; Tab. 1)
FIGURE 1 | Rhyacoglanis rapppydanielae, holotype, INPA 8060, 37.5 mm SL, rio Tocantins, state of Pará, Brazil.
Holotype. INPA 8060, 37.5 mm SL, Brazil, Pará, Tucuruí, rio Tocantins, small rapids downstream Jatobal, about 04º28’26.5”S 49º27’18.24”W, 8 Jul 1982, Equipe Ictiologia INPA.
Paratypes. INPA 59634, 183, 23.5–37.4 mm SL, same data as the holotype. INPA 7957, 21 (2 c&s), 16.1–36.0 mm SL, Brazil, Pará, Tucuruí, rio Tocantins, Jatobal rapids, about 04º28’26.5”S 49º27’18.24”W, 8 Jul 1982, Equipe Ictiologia INPA. MZUEL 20873, 30, 15.0–39.0 mm SL, same data as the holotype.
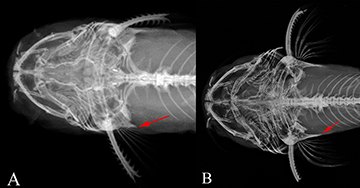
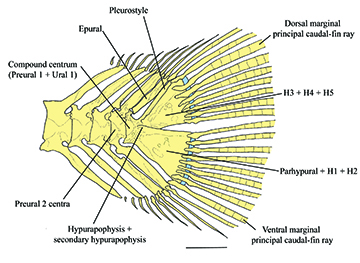
Diagnosis. Rhyacoglanis rapppydanielae is diagnosed from congeners by presenting the hypural bones 3, 4, and 5 fused (vs. hypural 5 free; Fig. 5). The new species also differs from its congeners, except for Rhyacoglanis epiblepsis, by the short post-cleithral process, with only a tiny visible tip after the opercular membrane (vs. elongated, more than half length visible after opercular membrane; Fig. 2), body scattered with numerous dark brown spots of diameter approximately equal to that of the eye (vs. few dark brown spots or absent), rounded pectoral and caudal fins (vs. pointed fins), and incomplete long lateral line (vs. complete). Rhyacoglanis rapppydanielae is distinguished from R. epiblepsis by the more tapered shape of the caudal peduncle’s dark brown band and the thin caudal fin’s dark brown band.
FIGURE 2 | Posterior cleithral process (red arrows) of A. Rhyacoglanis rapppydanielae, holotype, INPA 8060, 37.5 mm SL, and B. Rhyacoglanis seminiger, holotype, LIRP 12466, 74.2 mm SL.
Description. Body proportions are presented in Tab. 1. Body depressed from snout tip to dorsal-fin origin; progressively compressed from that point to caudal-fin base. Head dorsal profile slightly convex. Predorsal region between head and dorsal fin more convex than head. Dorsal-fin base somewhat straight. Dorsal-fin base to adipose-fin origin profile slightly convex. Head ventral profile convex. Abdomen ventral profile convex from pectoral girdle to anus. Caudal peduncle dorsal profile concave. Caudal peduncle ventral profile slightly straight from anus to anal-fin origin, concave posteriorly.
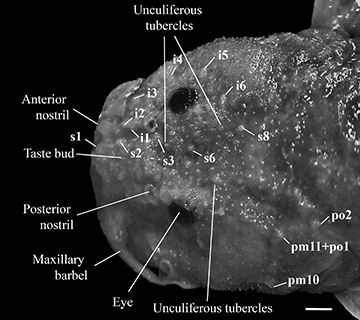
Head depressed, slightly longer than wide. Head anterior margin convex in dorsal view. Head dorsally covered by epidermal papillae (unculiferous tubercles larger than taste buds) (Fig. 4). Opercular membrane well developed; margin reaching pectoral-fin base. Mouth terminal, broad, more than one-half of HL. Upper jaw slightly longer than lower jaw. Lips thick, well developed primarily on lateral surface of jaws. Premaxillary tooth plate with posteriorly pointed projection. Anterior nostril immediately posterior of vertical through rictus. Eye small, superior, covered by skin, slightly anterior head half-length. Maxillary barbel short, not reaching the upper edge of operculum; base enlarged. Tip of adpressed maxillary barbel falling short of opercular margin. Adpressed inner mental barbel extending beyond base of outer mental barbel but falling short of barbel tip. Tip of adpressed outer mental barbel reaching margin of opercular membrane. Head laterosensory system pores conspicuous, six infraorbital (i1-i6), five supraorbital (S1-S3, S6, and S8), ten premaxillaries (pm1-pm10), one premaxillary-postorbital compound pore (pm11+po1), one postorbital (po2).
Dorsal fin trapezoidal; distal margin rounded; length of longest ray shorter than fin base. Dorsal-fin origin anterior to one-third of body length. Tip of adpressed dorsal fin falling short of midpoint between bases of dorsal and adipose fins. First dorsal-fin ray (spinelet) small, rigid, and forming dorsal-fin locking mechanism. Second dorsal-fin ray (spine) with anterior margin smooth and posterior margin bearing retrorse serrations. Dorsal-fin rays II,6* (14). Adipose fin long; base longer than that of other fins; posterior extremity free and angled. Pectoral fin slightly triangular with distal margin rounded. Tip of adpressed pectoral fin falling short of pelvic-fin origin but reaches vertical through second branched ray of dorsal fin. First pectoral-fin ray strong, rigid, and forming spine with retrorse serrations along almost anterior and posterior margins (Figs. 2–3). Pectoral-fin rays I,6* (14). Pelvic fin almost triangular, with distal margin rounded. Pelvic-fin origin at the vertical through penultimate dorsal-fin ray base. Tip of adpressed pelvic fin falling short of anal-fin origin or vertical through adipose-fin origin. Pelvic fin rays i+5*(16). Anal fin rounded distally; base length shorter than adipose-fin base length. Anal-fin rays iii,5*(3), iii+6(2), iv,5(1). Caudal fin slightly forked; rounded lobes; lower lobe longer than upper lobe. Principal caudal-fin rays i,6,8,i* (14). Dorsal procurrent caudal-fin rays 8(1), 10(2), 11(2), 12*(4); ventral procurrent caudal-fin rays 9(3), 10(2), 11*(3), 12(1). Parhypural, hypural 1, and hypural 2 fused; hypurals 3, 4, and 5 fused (Fig. 5).
Posterior cleithral process underdeveloped; pointed tip visible after opercular membrane (Fig. 2A). Axillary pore present. Lateral line long, incomplete, falling short of caudal-fin base, extending to vertical through posterior third of adipose fin. Total vertebrae 31*(5), 32(3), 33(1). Ribs 7(3), 8*(4), 9(2). Gill rakers on first branchial arch, 0,1,3(1), 0,1,4(3), 1,1,2(1), 1,1,3*(2), 1,1,4(2), 1,1,5(1).
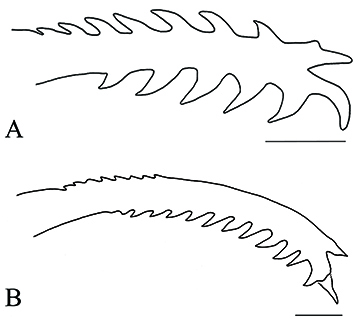
FIGURE 3 | Pectoral-fin spine: A. Left, ventral view of Rhyacoglanis rapppydanielae, paratype, INPA 7957, 33.3 mm SL, rio Tocantins, state of Pará, Brazil, scale bar = 1 mm; and B. Right, dorsal view of Rhyacoglanis seminiger, holotype, LIRP 12466, 74.2 mm SL, scale bar = 2.0 mm (after Shibatta, Vari, 2017).
FIGURE 4 | Epidermal structures of the dorsal region of the head of Rhyacoglanis rapppydanielae, paratype, MZUEL 20873, 38.3 mm SL, rio Tocantins, state of Pará, Brazil. Scale bar = 1 mm. Abbreviations: i1 to 6 = pores one to six of the infraorbital canal of laterosensory system; s1 to 8 = pores one to eight of the supraorbital canal; pm10 and 11 = pores 10 and 11 of preoperculomandibular canal; po1 and 2 = pores one and two of postotic canal.
Coloration in alcohol. Ground color yellowish brown. Head with small dark brown spots dorsally, on operculum, and ventral to orbit. Region over adductor mandibulae muscles yellowish-brown in many specimens. Dorsal region between opercula with slightly dark brown band. Lateral and dorsolateral surface of trunk covered by small dark brown spots. Trunk with three well-defined dark brown bands. Subdorsal dark brown band nearly triangular, larger width approximately equal to dorsal-fin base length or narrower; ventral tip, usually exceeding the lower limit of abdomen region but not joining counterpart band. Subadipose dark brown band wide, slightly rectangular, extending dorso-ventrally. Caudal peduncle dark brown band narrow; anterior and posterior margin irregular shaped, usually biconcave. Dorsal fin with C-shaped dark brown band from base to middle, distal margin and across ventral one-third of posterior five rays hyaline. Pectoral fin hyaline, small dark brown spots in the middle of rays. Pelvic fin hyaline. Adipose fin dark brown with anterior and posterior region yellowish-brown spot. Anal fin hyaline, base with dark brown band. Caudal fin hyaline; with thin open and lying down W-shaped dark brown band in middle of rays. Caudal-fin dark brown band continuous like in holotype (Fig. 1) or discontinuous.
FIGURE 5 | Caudal-fin skeleton of Rhyacoglanis rapppydanielae, paratype, INPA 7957, 33.3 mm SL, rio Tocantins, state of Pará, Brazil. H1–5 = hypurals 1 to 5. Scale bar = 1 mm.
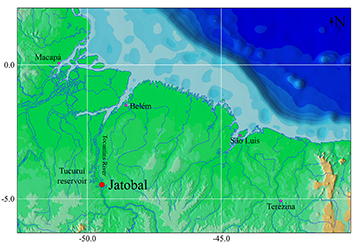
Geographical distribution. Rhyacoglanis rapppydanielae is known only from the type locality, Jatobal rapids, lower rio Tocantins basin, Pará, Brazil (Fig. 6). Unfortunately, collectors did not determine the exact geographic localization. However, the coordinates 04º28’26.5”S 49º27’18.24”W were recovered with Google Maps to Jatobal, Goianésia do Pará municipality, state of Pará.
Etymology. The specific name rapppydanielae is a homage to the Brazilian ichthyologist Lucia H. Rapp Py-Daniel for her outstanding contribution to Neotropical fishes’ systematics. She is the founder and current curator of the fish collection at Instituto Nacional de Pesquisas da Amazônia (INPA), whose scientific importance is recognized by the national and international communities.
FIGURE 6 | Map of Jatobal localization (red dot) in the rio Tocantins, type locality of Rhyacoglanis rapppydanielae, currently flooded by Tucuruí reservoir, state of Pará, Brazil. Pink dots = state capitals.
Conservation status. The rapids where the species was collected are flooded by the 200 km long Tucuruí hydroelectric power plant reservoir, which covered a vast territory and flooded several Tocantins’ tributaries. Rhyacoglanis rapppydanielae (previously identified as Pseudopimelodus sp.) was collected two years before the plant’s inauguration, which took place on November 22, 1984 (Santana et al., 2014), and the species was not collected again. The collection of a high number of specimens (n = 214) contained in one sample (originally INPA 8060) was possible using timbó to stum fishes. This substance is extracted from plants by indigenous and was allowed in experimental fisheries at the time but is no longer used. The species was not found in samples from the upper rio Tocantins. Thus, recent collection expeditions nearby the Tucuruí reservoir are necessary to verify the species’ occurrence in the remaining tributaries. Description of a species known only from a place where a large dam was built also happened to Tatia britskii (Sarmento-Soares & Birindelli, 2015) collected in 1965 in the region of Ilha Solteira reservoir, upper rio Paraná (Sarmento-Soares, Birindelli, 2015). Therefore, following the guidelines given in the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN Standards and Petitions Committee, 2019), we consider that Rhyacoglanis rapppydanielae should be categorized as Data Deficient (DD).
Comparative material examined. Same as listed in Shibatta, Vari (2017), with addition of Rhyacoglanis sp: Roraima: INPA 8062, 1, 32.4 mm SL, rio Uraricoera, ilha de Maracá, furo Maracá, 11 Mar 1988, M. Jégu.
TABLE 1 | Body proportions of Rhyacoglanis rapppydanielae (N = 14; INPA 8060, holotype; MZUEL 20873, 10 paratypes, and INPA 7957, 3 paratypes) from rio Tocantins, state of Pará, Brazil. Statistics includes the holotype. Min = Minimum; Max = Maximum; SD = Standard deviation.
Holotype | Min–Max | Mean±SD | |
Standard length (mm) | 37.5 | 25.7–38.3 | 31.2±3.7 |
Percent of standard length | |||
Head length | 29.6 | 27.3–31.0 | 28.9±1.1 |
Pectoral-girdle width | 27.3 | 26.3–28.5 | 27.1±0.7 |
Predorsal distance | 37.9 | 36.7–39.1 | 37.9±0.9 |
Dorsal-fin base length | 16.5 | 13.1–16.9 | 15.4±1.3 |
Adipose-fin base length | 20.3 | 16.8–21.2 | 19.3±1.3 |
Prepelvic length | 49.8 | 48.5–53.1 | 51.0±1.2 |
Pelvic fin to anal fin distance | 23.2 | 22.8–28.0 | 25.2±1.6 |
Anal-fin base length | 11.6 | 9.1–11.9 | 10.2±0.8 |
Body depth | 20.5 | 16.0–24.5 | 19.7±2.3 |
Caudal peduncle length | 13.9 | 13.3–19.5 | 15.4±1.7 |
Caudal peduncle depth | 11.2 | 9.3–11.2 | 10.3±0.5 |
Pectoral-fin spine length | 16.1 | 14.1–20.1 | 17.3±1.7 |
Dorsal-fin spine length | 11.2 | 10.4–14.0 | 12.3±1.2 |
Pelvic-fin length | 18.5 | 16.9–20.1 | 18.2±1.0 |
Post-cleithral process length | 9.4 | 8.0–10.1 | 8.9±0.6 |
Dorsal-fin origin to pelvic fin | 25.1 | 22.4–28.2 | 25.0±1.7 |
Distance between pelvic fins | 15.0 | 12.6–15.0 | 14.1±0.8 |
Distance between pelvic fin to the anus | 13.6 | 10.2–14.3 | 12.3±1.2 |
Distance between anus to anal fin | 16.0 | 11.9–16.5 | 14.3±1.3 |
Percent of head length | |||
Eye diameter | 10.0 | 10.0–13.9 | 12.1±1.3 |
Interorbital distance | 29.2 | 29.2–36.4 | 32.2±2.2 |
Snout length | 37.7 | 31.1–41.6 | 36.4±2.7 |
Mouth width | 70.2 | 51.6–70.2 | 60.4±5.0 |
Head depth | 45.0 | 30.5–47.1 | 36.6±4.6 |
Maxillary barbel length | 92.2 | 55.6–99.4 | 78.1±12.9 |
Distance between anterior and posterior nostrils | 18.3 | 15.3–22.7 | 19.5±2.2 |
Distance between posterior nostril to eye | 9.3 | 4.1–11.2 | 8.6±1.9 |
Distance between posterior nostrils | 17.5 | 15.7–23.8 | 19.7±2.1 |
Discussion
To date, Rhyacoglanis rapppydanielae is the smallest described species of the genus. However, despite the small body size (25.7–38.3 mm SL), the fused hypural bones 3, 4, and 5 may indicate that the specimens are already adults because some Pseudopimelodidae juveniles, like Lophiosilurus alexandri Steindachner, 1876, present these bones still free when young but fused in adults (OAS, pers. obs.). Furthermore, Shibatta, Vari (2017) evidenced that Rhyacoglanis and Lophiosilurus Steindachner, 1876 evolved in different evolutionary lineages, which signals us that this fusion of hypural bones is a homoplasy among R. rapppydanielae and L. alexandri.
Rhyacoglanis rapppydanielae is morphologically similar to R. epiblepsis of the río Beni. Some shared characters, such as the color pattern, the shape of the pectoral and caudal fins, the post-cleithral process’s length, and the lateral line, may indicate phylogenetic proximity.
The pectoral-fin spine shape of R. rapppydanielae is like R. epiblepsis and R. annulatus Shibatta & Vari, 2017. They share well-developed serrae distributed along almost entire anterior and posterior margins. On the contrary, R. pulcher, R. paranensis Shibatta & Vari, 2017, and R. seminiger have smaller serrae in the pectoral-fin spine’s anterior margin than the posterior ones (Figs. 2–3). In the phylogenetic hypothesis presented by Shibatta, Vari (2017), R. epiblepsis was the sister species of R. annulatus. Therefore, the relative size of anterior and posterior serrae may have some phylogenetic signal, but it is still necessary to test this hypothesis with a more encompassing phylogenetic analysis, including R. rapppydanielae.
The new species has the body size and external morphology roughly like Microglanis robustus Ruiz & Shibatta, 2010, which occurred in syntopy. However, R. rapppydanielae can be distinguished from M. robustus by the posterior extension of the premaxillary tooth plate (vs. lateral not prolonged) and the more elongated lateral line (extending to vertical through posterior third of adipose fin vs. short, reaching only the vertical through origin of adipose fin). Nevertheless, the morphological similarity among syntopic species leads to classifying it as Mullerian mimicry, as both species have pungent spines in the dorsal and pectoral fins as a defensive mechanism.
Comparing the cephalic laterosensory system of R. rapppydanielae with Microglanis robustus (Ruiz, Shibatta, 2010) and Batrochoglanis castaneus Shibatta, 2019 (Shibatta, 2019), it is observed that the arrangement and quantity of pores are similar, which may indicate that this system is conservative in the family. However, an analysis involving more species, including other Pseudopimelodidae genera, is still necessary to corroborate its coverage in the family.
Currently, R. rapppydanielae is known only from the rio Tocantins basin. A specimen examined from the rio Uraricoera, Roraima State, Brazil (INPA 8062) also showed morphometric characters and the rounded shape of the caudal fin lobes like R. rapppydanielae. However, slight differences in the color pattern and the low number of specimens (n = 1) prevent us from identifying it as a known species. Nevertheless, more species related to R. rapppydanielae and R. epiblepsis can occur along rio Amazonas basin.
Acknowledgments
We thank Lucia H. Rapp Py-Daniel for allowing the specimens to be examined. To Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the research productivity grant to OAS (proc. #303685/2018–2).
References
Castro RMC. Evolução da ictiofauna de riachos sul-americanos: padrões gerais e possíveis processos causais. In: Caramaschi EP, Mazzoni R, Peres-Neto PR, editors. Ecologia de peixes de riachos. Oecologia Brasiliensis; 1999; 6(1):139–55. https://doi.org/10.4257/oeco.1999.0601.04
Dingerkus G, Uhler LD. Enzyme clearing alcian blue stained whole vertebrates for demonstration of cartilage. Stain Technol. 1977; 52(4):229–32. https://doi.org/10.3109/10520297709116780
IUCN Standards and Petitions Committee. Guidelines for using the IUCN Red List Categories and Criteria. Version 14. Prepared by the Standards and Petitions Committee. 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
Mees GF. The Auchenipteridae and Pimelodidae of Suriname (Pisces, Nematognathi). Zool Verh. 1974; 132(1):1–256. Available from: https://repository.naturalis.nl/pub/317592/ZV1974132001.pdf
Miranda-Ribeiro A. Pimelodidae, Trachycorystidae, Cetopsidae, Bunocephalidae, Auchenipteridae e Hypophthalmidae. Rio de Janeiro: Comissão de Linhas Telegraphicas Estrategicas de Matto-Grosso ao Amazonas. 1914; 15 (anexo 5):1-13.
Ruiz WBG, Shibatta AO. A new species of Microglanis (Siluriformes, Pseudopimelodidae) from lower Rio Tocantins basin, Pará Brazil, with description of superficial neuromasts and pores of lateral line system. Zootaxa. 2010; 2632(1):53–66. http://dx.doi.org/10.11646/zootaxa.2632.1.3
Santana AC, Bentes ES, Homma AKO, Oliveira, FA, Oliveira, CM. Influência da barragem de Tucuruí no desempenho da pesca artesanal, estado do Pará. Rev Econ Sociol Rural. 2014; 52(2):249–66. http://dx.doi.org/10.1590/S0103-20032014000200003
Sarmento-Soares LM, Birindelli JLO. A new species of the catfish genus Centromochlus (Siluriformes: Auchenipteridae: Centromochlinae) from the upper rio Paraná basin, Brazil Neotrop Ichthyol. 2015; 13(1):77–86. http://dx.doi.org/10.1590/1982-0224-20140042
Shibatta OA. New species of bumblebee catfish of the genus Batrochoglanis Gill, 1858 (Siluriformes: Pseudopimelodidae) from the Aripuanã River basin, Mato Grosso, Brazil. Zootaxa. 2019; 4674(2):243–63. https://doi.org/10.11646/zootaxa.4674.2.6
Shibatta OA, Vari RP. A new genus of Neotropical rheophilic catfishes, with four new species (Teleostei: Siluriformes: Pseudopimelodidae). Neotrop Ichthyol. 2017; 15(2):e160132. http://dx.doi.org/10.1590/1982-0224-2016013
Authors
![]() Oscar A. Shibatta1
Oscar A. Shibatta1 ![]() ,
, ![]() Marcelo S. Rocha2 and
Marcelo S. Rocha2 and ![]() Renildo R. de Oliveira3
Renildo R. de Oliveira3
[1] Museu de Zoologia, Departamento de Biologia Animal e Vegetal, Centro de Ciências Biológicas, Universidade Estadual de Londrina, 86057-970 Londrina, PR, Brazil. shibatta@uel.br (corresponding author)
[2] Universidade do Estado do Amazonas, Av. Djalma Batista 2470, Manaus, AM, Brazil. marcelo.inpa@gmail.com
[3] Coordenação de Biodiversidade, Coleção de Peixes, Instituto Nacional de Pesquisas da Amazônia-INPA, Av. André Araújo 2936, Petrópolis, 69067-375 Manaus, AM, Brazil. deoliveirarr@gmail.com
Authors Contribution 
Oscar Akio Shibatta: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Marcelo Salles Rocha: Data curation, Formal analysis, Investigation, Writing-original draft, Writing-review and editing.
Renildo Ribeiro de Oliveira: Data curation, Investigation, Validation, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The authors declare no competing interests.
How to cite this article
Shibatta OA, Rocha MS, Oliveira RR. New species of Rhyacoglanis (Siluriformes: Pseudopimelodidae) from rio Tocantins basin, northern Brazil. Neotrop Ichthyol. 2021; 19(4):e210083. https://doi.org/10.1590/1982-0224-2021-0083
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted July 18, 2021 by Marcelo Britto
Accepted July 18, 2021 by Marcelo Britto
![]() Submitted May 5, 2021
Submitted May 5, 2021
![]() Epub 01 Dec 2021
Epub 01 Dec 2021