![]() Victor A. Tagliacollo1,
Victor A. Tagliacollo1, ![]() Fernando Cesar Paiva Dagosta2,
Fernando Cesar Paiva Dagosta2,![]() Mário de Pinna1,
Mário de Pinna1,![]() Roberto E. Reis3 and
Roberto E. Reis3 and![]() James S. Albert4
James S. Albert4
PDF: EN XML: EN | Cite this article
Abstract
The IUCN Red List (RL) provides high-quality conservation assessments for individual species, yet the rate and scale of environmental deterioration globally challenges the conservation community to develop expedited methods for risk assessment. Here we compare threat assessments for 3,001 species of Neotropical freshwater fishes (NFF) in the IUCN–RL using readily accessible data types as proxies for extinction risk: geographic range, elevation, and species publication date. Furthermore, using geographic and taxonomic data alone, we generated preliminary conservation assessments for 2,334 NFF species currently awaiting IUCN assessment, identifying an additional 671 NFF species as potentially threatened. This number of potentially threatened species represents an increase of 59% over the number of species currently assigned to threat categories by the IUCN–RL. These results substantially expand the number of threatened NFF species from 422 currently on the IUCN RL to 1,093 species as threatened or potentially threatened, representing about 18% of all NFF species. Extinction risk is greater in species with smaller geographic ranges, which inhabit upland rivers, and which were described more recently. We propose the Central and Southern Andes, and Eastern Guiana Shield as priorities in the upcoming IUCN RL assessment of NFF species conservation risk.
Keywords: Biodiversity, Conservation status, Extent of Occurrence, IUCN Red List.
A Lista Vermelha (IUCN) fornece avaliações precisas sobre status de conservação das espécies, porém a taxa e escala de deterioração ambiental desafia a comunidade conservacionista a desenvolver métodos rápidos para avaliações de riscos de extinção. Aqui, comparamos as avaliações da IUCN para 3.001 espécies de peixes dulcícolas neotropicais com dados facilmente acessíveis de risco de extinção: extensão de ocorrências, altitude e data de publicação das espécies. Além disso, usando apenas dados geográficos e taxonômicos, geramos avaliações preliminares de conservação para 2.334 espécies de peixes neotropicais aguardando avaliação da IUCN, e identificamos 671 espécies adicionais como potencialmente ameaçadas. Este número de espécies potencialmente ameaçadas representa um aumento de 59% em relação aquelas classificadas nas categorias de ameaça pela IUCN. Estes resultados expandem o número de espécies ameaçadas segundo a IUCN de 422 para 1.093 espécies ameaçadas ou potencialmente ameaçadas, representando cerca de 18% das espécies de peixes neotropicais. O risco de extinção é maior para espécies com distribuições geográficas restritas, que habitam rios de terras altas e que foram descritas mais recentemente. Sugerimos a região Central e Meridional do Andes e o Escudo das Guianas Orientais como prioridades para as próximas avaliações da IUCN sobre a conservação das espécies de peixes dulcícolas neotropicais.
Palavras-chave: Biodiversidade, Conservação, Extensão de Ocorrência, Lista Vermelha (IUCN).
Introduction
Continental freshwaters are home to vertebrate species density higher than most other ecosystems on Earth. Almost half of the world’s fish species (about 14,750 of 35,700) are restricted to continental freshwater environments, including rivers, streams, springs, lakes, ponds, swamps, and wetlands, in a habitat volume that comprises less than 0.001% of the Earth’s total water supply. Freshwaters are a renewable, yet a finite natural resource, being essential to maintain hydro-climatic regimes and almost all economic human activities (Dudgeon et al., 2006; Hoekstra, Mekonnen, 2012). Freshwater ecosystems are increasingly threatened worldwide, especially by the expansion of energy production (Finer, Jenkins, 2012), mining (Ferreira et al., 2014), aquaculture (Valladão et al., 2018), agriculture (Rosa et al., 2020) and urban landscapes (McKinney, 2006). Besides, freshwater environments are being depleted more quickly than their terrestrial counterparts (Albert et al., 2020a), and biodiversity losses are proportionately greater in freshwater than in terrestrial ecosystems (Turak et al., 2017). Fishes and amphibians are among the most threatened of all vertebrates, and their species richness and abundances are declining rapidly at regional and continental scales (Reid et al., 2019).
Freshwater ecosystems are unequally distributed across the Earth’s surface, with c. 30% of the global river discharge flowing through Neotropical river basins. Neotropical freshwaters comprise continental waters of South America, Middle America, and the Caribbean islands (Berra, 2007; Nelson et al., 2016). This vast realm encompasses a variety of aquatic environments, with distinct faunas adapted to torrential mountain rapids and cascades, upland rainforest rivers and streams, karstic and phreatic caverns, lowland floodplains, oxbow lakes, deep (to 100 m) river channels, and coastal rivers and estuaries (Albert et al., 2011a; Crampton, 2011). Those environments are distributed throughout the northern, central, and southern portions of the Andean cordilleras, Central America and the Greater Antilles, the upland Guiana and Brazilian shields, and the lowland Amazon, Orinoco and La Plata River basins. Each of those regions displays a heterogeneous combination of aquatic ecosystems, and a unique composition of fish species and local communities (e.g., Albert et al., 2020a).
Neotropical freshwater fishes (NFF) constitute the most species-rich vertebrate fauna on Earth (Fig. 1). With over 6,200 valid species (Albert et al., 2020b), the NFF species represent c. 30% of the world’s continental fishes, or about 10% of all living vertebrates (Nelson et al., 2016). Dozens of new species are described each year (Fricke et al., 2020), such that total NFF species richness has been projected to exceed 9,000 species (Reis et al., 2016). As with most ichthyofauna, the remarkable NFF species diversity is unevenly distributed among higher taxa, with approximately 70% of the species concentrated within the two most species-rich of the 40 NFF taxonomic orders, and 95% in just the top five orders: Siluriformes (catfishes), Characiformes (tetras, piranhas and allies), Cyprinodontiformes (killifishes, rivulids, and allies), Cichliformes (cichlids), and Gymnotiformes (Neotropical electric fishes). Species richness is also unevenly distributed within taxonomic orders, with 60% of the species in the top five of the 97 NFF taxonomic families (van der Sleen, Albert, 2017; Dagosta, de Pinna, 2019). The phylogenetic diversity represented by these diverse NFF taxa is accompanied by an enormous diversity of functional traits, ecophysiological specializations for feeding and habitat utilization, reproductive modes, and life history strategies (Albert, Reis, 2011; Crampton, 2011; Toussaint et al., 2016).
The mega-diverse Neotropical ichthyofauna also has an uneven distribution of geographic range sizes in which most species have small ranges, and a few species have exceptionally widespread geographic ranges across multiple ecoregions (Albert et al., 2011b). In the Amazon, the largest hydrographic basin in the world, most fish species are not exclusive to a single drainage (Dagosta, de Pinna, 2019), corroborating the hypothesis that the NFF’s biogeographical history is complex and that basins are historically composite (Dagosta, de Pinna, 2017). Range-restricted NFF species are usually limited to rugged upland regions with high topographic relief located towards the continental periphery (e.g., Andes, shields), while the more geographically widespread species occur mostly on low-relief (i.e., flat) lowland river basins at the continental core (Albert et al., 2011b; Dagosta, de Pinna, 2019). Consequently, upland drainages generally exhibit relatively lower local species richness (i.e., lower alpha diversity) but a relatively greater change in species composition across neighboring watersheds (i.e., higher geographic beta diversity) as compared with lowlands (Albert et al., 2017). Even though lowland rivers at the continental core are centers of species richness, upland rivers and coastal drainages at the continental periphery have higher species endemism (Albert et al., 2018).
Neotropical waterways, as with other tropical freshwater systems worldwide, are experiencing accelerating conservation threats because of myriad anthropogenic activities (Dagosta et al., 2020). Worldwide, an average of 30% of freshwater fishes are classified under threat categories by the International Union for Conservation of Nature (IUCN) Red List (RL) assessments (IUCN, 2020). Extinction risk in much of the Neotropics is generally lower than in other continents, where about 10% of all continental Brazilian fishes are currently assigned to threatened categories (Reis et al., 2016; ICMBio, 2018). Despite commendable efforts, approximately half of all NFF currently await IUCN assessment, an expensive, laborious and time-consuming process, and many Latin American countries do not have reliable conservation information on potentially threatened species.
The urgency to prioritize species conservation has encouraged the development of alternative tools for performing preliminary conservation assessments based on geographic and taxonomic information alone (Bachman et al., 2011; Dauby et al., 2017; Zizka et al., 2020). Recently, efforts have been made in combining IUCN RL assessments with species’ traits to assign preliminary extinction risks to not-evaluated or data‐deficient species (Bland et al., 2015; Pelletier et al., 2018; Gonzalez-del-Pliego et al., 2019; Lughadha et al., 2019). Although body size and functional traits are widely known to be correlated with extinction risk in fish faunas (Poff et al., 2012; Kalinkat et al., 2017), the relationships among those variables have only recently been studied in NFFs (Castro, Polaz, 2020; Tagliacollo et al., 2020). Estimates based on recent rates of species discovery suggest that almost 3,000 species of NFF species have yet to be formally described by taxonomists (Reis et al., 2016), and hundreds of fish species with small adult body size and restricted geographic ranges are already known to be threatened with extinction (Castro, Polaz, 2020; Tagliacollo et al., 2020).
Here we investigate the use of geographic and taxonomic data as a proxy to rapidly assess potential conservation threats for species of the NFF species. Specifically, we explore whether species geographic ranges, topographic elevation data, and publication date are associated with extinction risk using a dataset of 3,001 NFF species with threat categories assigned by the IUCN–RL. Furthermore, we use geographic coordinates data to generate a preliminary extinction risk assessment for 2,334 NFF currently awaiting formal IUCN assessment. We identify data types correlated with extinction risk, illustrate geographic distributions of NFF threatened species, predict distributions of potentially threatened species, and suggest possible priority areas for upcoming conservation assessments. Given the runaway rate of habitat destruction and the disproportionately limited resources available for such studies, we provide an important tool for urgently-needed conservation of neotropical freshwater fishes.
Material and methods
Spatial database. Estimating species ranges for conservation assessment requires compiling, organizing, and proofing a comprehensive database with thousands of geographic coordinates. We compiled a database of NFF species based on geographic information of preserved specimens from the taxonomic literature and museum collections, the latter accessible through metadata repositories (e.g., GBIF, FishBase, SpeciesLink). After combining specimen occurrences from multiple sources, we applied an automated cleaning pipeline (Robertson et al., 2016) to remove duplicates and geographic records with apparent geo-referenced mistakes. This procedure excluded incongruent occurrences, such as those placed in the ocean or outside the Neotropics, those without precise locality or country names, coordinates along the whole degree latitude or longitude (i.e., latitude or longitude exactly zero) or collected on coarse-scale grid lines without decimal precision. We validated the NFF species distributions by plotting maps of individual species and comparing them with those published in the primary taxonomic literature. For many taxa, in particular those without range maps in the specialized literature, we solicited expert opinions by taxonomists who provided additional data points to improve species distributions and suggestions to exclude unreliable occurrences. In the absence of published maps or expert opinions, we kept only the coordinates of the holotype and paratypes.
FIGURE 1 | Sample of the phenotypic diversity of Neotropical freshwater fishes.Upper left to lower right: Lycengraulis grossidens (Spix & Agassiz, 1829); Hyphessobrycon hexastichos Bertaco & Carvalho, 2005; Geophagus neambi Lucinda, Lucena & Assis, 2010; Crenicichla lepidota Heckel, 1840; Trachelyopterus galeatus (Linnaeus, 1766); Anablepsoides xinguensis (Costa, 2010); Abramites hypselonotus (Günther 1868); Pituna xinguensis Costa & Nielsen, 2007; Gymnotus cuia Craig, Malabarba, Crampton & Albert, 2018; Apteronotus caudimaculosus de Santana, 2003; Colomesus tocantinensis Amaral, Brito, Silva & Carvalho, 2013; Corydoras britskii (Nijssen & Isbrücker, 1983). Species not shown in scale.
IUCN threat categories. We compiled a list of 3,001 NFF species and their respective extinction risks based on assessments using the IUCN criteria and categories (IUCN, 2019). This list contains information about species extinction risks from the IUCN Red List database (IUCN, 2020), Brazilian Ministry of Environment Red List of endangered species (ICMBio, 2018), and Colombian Red List Book (Mojica et al., 2012). Those extinction risk assessments list recognized species as Least Concern (LC), Near Threatened (NT), Vulnerable (VU), Endangered (EN), Critically Endangered (CR), and Data Deficient (DD). We changed the threat classification scheme by combining the categories Least Concern (LC) and Near Threatened (NT) into a single category considered “non-threatened” to match the classification scheme exported by the R package ConR (Dauby et al., 2017).
Variables. We investigated the utility of three variables as proxies for extinction risk categorizations recognized by the IUCN–RL. We compiled a list of NFF species from William Eschmeyer’s Catalog of Fishes (Fricke et al., 2020), including all 6,226 valid NFF species names, publication dates, and synonyms. We replaced all junior synonyms in the spatial database with valid names. We sub-sampled this list of publication dates to include 5,335 valid species having geographic coordinates in the spatial database, including 3,001 NFF species with IUCN–RL extinction risk assessments, and additionally 2,334 awaiting IUCN assessment. We discretized the species publication dates for the dataset of 3,001 NFF species into 15 intervals, each with approximately equal frequency values, including around 200 data points per interval.
We measured the Extent of Occurrence (EOO) as a proxy for species geographic distribution. The intent of the EOO estimates is to measure the degree of extinction risks from threatening factors across the taxon’s geographical distribution (IUCN, 2019). We measured species EOO using coordinates plotted on geographic maps projected in the WGS84 coordinate reference system. We calculated the EOO for each species in the database as the area in square kilometers of the minimum convex polygon encompassing all localities of the species. To avoid overestimating geographic ranges, we removed projected regions extending beyond the Neotropical boundaries into the oceans. For species with less than three geographic coordinates, we calculated the Area of Occupancy (AOO) on a 2 x 2 km grid line (i.e., 4 km2 per coordinate). We discretized the species EOOs for the dataset of 3,001 NFF species into 15 range-size intervals, each including approximately equal frequency values, including around 200 data points per interval.
We assessed differences in elevation of geographic coordinates as a proxy for species topographic elevation ranges. For each specimen of every species in the spatial database, we gathered elevation estimates using NASA’s Shuttle Radar Topography Mission (SRTM) dataset in a Digital Elevation Model (DEM) (Farr et al., 2007). We gathered these elevation estimates on geographic maps projected in the WGS84 coordinate reference system. As before, we discretized species altitude ranges into 15 elevation intervals, each including approximately equal frequency values, including around 5,930 data points per interval.
Preliminary assessments. We used the R package ConR (Dauby et al., 2017) to provide a preliminary conservation assessment for 2,334 NFF species for which extinction risks have not been assigned by the IUCN–RL. We used the ConR package to assign threat categories to the unclassified species assuming only criterion B, specifically sub-criterion B1 (i.e., EOO). This sub-criterion is one of the necessary conditions used by the IUCN–RL for classifying species into threat categories. It must also apply other conditions when assigning extinction risks properly; e.g., number of locations or population fragmentation, continuing decline of species distributions or habitat quality, population extreme fluctuations through time (IUCN, 2019). However, these conditions are too complex for automated conservation assignments, because they depend on detailed knowledge of species ecology and behavior, habitat utilization, and existing actual threats to part or all of the species’ geographic range. Preliminary conservation assessments by ConR classified species into Non-threatened (Least Concern or Near Threatened), and three threat categories: Vulnerable (VU), Endangered (EN), and Critically Endangered (CR). Species with fewer than three geographic coordinates received the status of Data Deficient (DD).
Results
Species occurrences. After verification and validation steps we obtained a database of 125,685 unique geographic coordinates for 5,335 species or 83% of the entire Neotropical ichthyofauna. Of those, 4,154 species were represented by three or more coordinates, with a median of 11 occurrences per species with interquartile range (i.e., Q3–Q1) of 26. By taxonomic order, the database included 60,374 (48.0%) occurrences for 1,758 characiform species, 32,407 (25.8%) occurrences for 1,964 siluriform species, 12,262 (9.7%) occurrences for 465 cichliform species, 6,495 (5.2%) occurrences for 596 cyprinodontiform species, 6,811 (5.4%) occurrences for 258 gymnotiform species, and 7,336 (5.8%) for 294 species in the other taxonomic orders. The geographic coordinates were heterogeneously distributed throughout most of the Neotropical region, with geographic gaps in the Argentinean pampas, a region with a few rivers and comparatively lower species density.
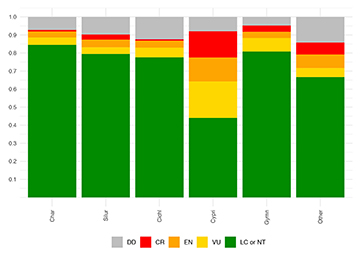
IUCN–RL: conservation assessments. We evaluated the extinction risks for 3,001 NFF species with existing IUCN assessments, including 1,068 Characiformes, 1,106 Siluriformes, 302 Cyprinodontiformes, 259 Cichliformes, 146 Gymnotiformes, and 120 species in other taxonomic orders. We identified that about 14% (or 422 out 3,001 species) of NFF species are under extinction risk, including 176 as VU, 147 as EN, and 99 as CR. Partitioned by taxonomic order, NFF species are classified by the IUCN-RL in threat categories in the following proportions: (i) Characiformes: 8.2% (88 out 1068 species), including 43 as VU, 36 as EN, and nine as CR; (ii) Siluriformes: 10.8% (119 out 1106 species), including 41 as VU, 47 as EN, and 31 as CR; (iii) Cyprinodontiformes: 48.0% (or 145 out 302 species), including 61 as VU, 40 as EN, and 44 as CR; (iv) Cichliformes: 10.0% (26 out 259 species), including 14 as VU, 10 as EN, and two as CR; (v) Gymnotiformes: 14.9% (21 out 146 species), including 11 as VU, five as EN, and five as CR, and (vi) species in other clades: 19.2% (23 out 120 species); including six as VU, nine as EN, and eight as CR (Tab. 1; Fig. 2).
TABLE 1 | Summary of the IUCN–RL data for 3,001 Neotropical freshwater fish (NFF) species. Threatened = number of threatened NFF species.
| Order | NFF | Threatened | % |
| Characiformes | 1068 | 88 | 8.2 |
| Siluriformes | 1106 | 119 | 10.8 |
| Cyprinodontiformes | 302 | 145 | 48.0 |
| Cichliformes | 259 | 26 | 10.0 |
| Gymnotiformes | 146 | 21 | 14.4 |
| Other orders | 120 | 23 | 19.2 |
| TOTAL | 3,001 | 422 | 14.1 |
FIGURE 2 | Extinction risks in 3,001 Neotropical freshwater fishes (NFF). On average, 14% (422 of 3,001) NFF species are classified by the IUCN Red List (RL) as Vulnerable (VU), Endangered (EN) or Critically Endangered (CR). Extinction risks are relatively similar among the orders Characiformes (8.2%), Siluriformes (10.8%), Cichliformes (10.0%), and Gymnotiformes (14.9%). An exception is the clade Cyprinodontiformes, where about 48% of species are classified as either VU, EN, or CR. LC or NT: Least Concern or Near Threatened; DD: Data Deficient.
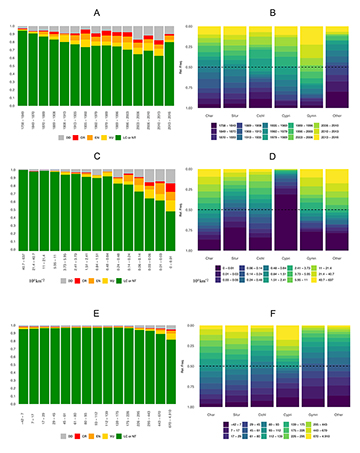
Taxonomic and spatial variables. We obtained information on publication dates, EOOs, and elevation for 3,001 NFF species with IUCN–RL extinction risks. This dataset comprises NFF species published from 1758 to 2016. The Fig. 3 shows general trends of relationships among these three variables and extinction risks in NFF species.
We identified about 33.5% (or 1,007 of 3,001 species) NFF species described in a time-interval of 20 years, between 1996 to 2016. Approximately 18.7% (or 188 of 1,007 species) of these recently described NFF species are classified by the IUCN–RL as either VU, EN or CR (Tab. 2). Partitioned by taxonomic order, we verified that NFF species described in this 20-year interval are classified by the IUCN–RL in threat categories in the following proportions: (i) Characiformes: 11.6% (34 of 294 species), including 16 as VU, 12 as EN, and six as CR; (ii) Siluriformes: 15.1% (59 of 390 species), including 19 as VU, 26 as EN, and 14 as CR; (iii) Cyprinodontiformes: 44.1% (67 of 152 species), including 34 as VU, 13 as EN, and 20 as CR; (iv) Cichliformes: 11.3% (seven of 62 species), including five as VU, and two as EN; (v) Gymnotiformes: 21.8% (19 of 87 species), including 10 as VU, four as EN and five as CR; and (vi) species in other clades: 9.1% (two of 22 species), including two as EN (Tab. 2).
TABLE 2 | Summary of the IUCN–RL data for 1,007 NFF species described in a 20–year interval, from 1996 to 2016.
| Publ. dates: 1996–2016 | |||
| Order | NFF | Threatened | % |
| Characiformes | 294 | 34 | 11.6 |
| Siluriformes | 390 | 59 | 15.1 |
| Cyprinodontiformes | 152 | 67 | 44.1 |
| Cichliformes | 62 | 7 | 11.3 |
| Gymnotiformes | 87 | 19 | 21.8 |
| Other orders | 22 | 2 | 9.1 |
| TOTAL | 1,007 | 188 | 18.7 |
FIGURE 3 | Association among three variables and extinction risks in 3,001 Neotropical freshwater fishes (NFF). A. IUCN-Red List (RL) threat categories by species description dates. B. Species description dates by taxonomic orders. C. IUCN-RL categories by geographic ranges. D. Geographic ranges by taxonomic orders. E. IUCN-RL categories by elevation ranges. F. Elevational ranges by taxonomic orders. Threatened status are usually higher for recently described NFF species, those inhabiting narrow geographic ranges, and those confined to upland river drainages. CR: Critically Endangered; EN: Endangered; VU: Vulnerable; DD: Data Deficient.
We estimate 44.5% (1,337 of 3,001 species) of NFF species have an EOO under 20,000 km2. Approximately 27.9% (373 of 1,337 species) of these NFF species with EOOs under 20,000 km2 are classified by the IUCN–RL as either VU, EN or CR (Tab. 3). Partitioned by taxonomic order, NFF species with EOO estimates of less than 20,000 km2 are classified by the IUCN–RL in threat categories in the following proportions: (i) Characiformes: 21.8% (70 of 321 species), including 33 as VU, 29 as EN and eight as CR; (ii) Siluriformes: 18.6% (106 of 570 species), including 34 as VU, 44 as EN and 28 as CR; (iii) Cyprinodontiformes: 52.7% (138 of 262 species), including 55 as VU, 39 as EN and 44 as CR; (iv) Cichliformes: 22.1% (21 of 95 species), including 13 as VU, seven as EN and one as CR; (v) Gymnotiformes: 37.2% (19 of 51 species), including 10 as VU, four as EN and five as CR; and (vi) species in other clades: 50% (19 of 38 species), including three as VU, nine as EN and seven as CR (Tab. 3).
TABLE 3 | Summary of the IUCN–RL data for 1,337 NFF species with geographic range estimates less than 20,000 km2. EOO = Extent of Occurrence.
| Geographic ranges: EOO < 20,000 km2 | |||
| Order | NFF | Threatened | % |
| Characiformes | 321 | 70 | 21.8 |
| Siluriformes | 570 | 106 | 18.6 |
| Cyprinodontiformes | 262 | 138 | 52.7 |
| Cichliformes | 95 | 21 | 22.1 |
| Gymnotiformes | 51 | 19 | 37.2 |
| Other orders | 38 | 19 | 50.0 |
| TOTAL | 1,337 | 373 | 27.8 |
We obtained elevation data from 88,977 geographic coordinates, ranging from -42 to 4,910 meters elevation. Dividing this elevation range by interquartile, we showed that species in upland river systems above 253.00 meters are more often classified by the IUCN–RL in threat categories (Tab. 4). Approximately 23.6% (217 of 918 species) of the NFF species with median altitudes ranging between 253.0–4911.0 m (Q4) are classified by the IUCN–RL as either VU, EN or CR (Tab. 4). Partitioned by taxonomic order, NFF species with median altitudes ranging between 253.0 – 4911.0 meters (Q4) are classified by the IUCN–RL in threat categories in the following proportions: (i) Characiformes: 14.9% (47 of 316 species), including 24 as VU, 17 as EN and 6 as CR; (ii) Siluriformes: 17.8% (73 of 409 species), including 27 as VU, 26 as EN and 20 as CR; (iii) Cyprinodontiformes: 60% (73 of 121 species), including 36 as VU, 24 as EN and 13 as CR; (iv) Cichliformes: 17.5% (seven of 40 species), including 40 as VU and three as CR; (v) Gymnotiformes: 41.2% (six of 17 species), including one as VU, four as EN and one as CR; and (vi) species in other clades: 73.3% (11 of 15 species), including two as VU, four as EN and five as CR (Tab. 4).
TABLE 4 | Summary of the IUCN–RL data for 3,001 NFF species, with estimates of median elevation within interquartile ranges. Thr = Threatened species.
| Q1 | Q2 | Q3 | Q4 | |||||||||
| -42.0 : 42.0 m | 42 : 105.0 m | 105.0 : 253.0 m | 253.0 : 4910.0 m | |||||||||
| Order | NFF | Thr | % | NFF | Thr | % | NFF | Thr | % | NFF | Thr | % |
| Characiformes | 112 | 11 | 9.8 | 358 | 10 | 2.8 | 282 | 20 | 7.1 | 316 | 47 | 14.9 |
| Siluriformes | 179 | 15 | 8.4 | 277 | 17 | 6.1 | 241 | 14 | 5.8 | 409 | 73 | 17.8 |
| Cyprinodontiformes | 89 | 43 | 48.3 | 40 | 14 | 35.0 | 52 | 15 | 28.8 | 121 | 73 | 60.3 |
| Cichliformes | 60 | 5 | 8.3 | 94 | 10 | 10.6 | 65 | 4 | 6.2 | 40 | 7 | 17.5 |
| Gymnotiformes | 57 | 6 | 10.5 | 50 | 6 | 12.0 | 22 | 3 | 13.6 | 17 | 6 | 35.3 |
| Other orders | 46 | 5 | 10.9 | 47 | 5 | 10.6 | 12 | 2 | 16.7 | 15 | 11 | 73.3 |
| TOTAL | 543 | 85 | 15.7 | 866 | 62 | 7.2 | 674 | 58 | 8.6 | 918 | 217 | 23.6 |
ConR: preliminary conservation assessments. The R package ConR generated preliminary conservation assessments for 2,334 NFF species awaiting IUCN assessments, including 858 Siluriformes, 690 Characiformes, 294 Cyprinodontiformes, 206 Cichliformes, 112 Gymnotiformes, and 174 species in other orders. We verified in this preliminary survey that about 28.7% (671 of 2,334) species were considered under potential extinction risk, including 301 as VU, 346 as EN, and 24 as CR (Tab. 5). Partitioned by taxonomic order, we verified that NFF species are assigned by the ConR package in threat categories in the following proportions: (i) Characiformes 30.4% (210 of 690 species), including 97 as VU, 106 as EN, and seven as CR; (ii) Siluriformes: 29.2% (251 of 858 species), including 109 as VU, 130 as EN, and 12 as CR; (iii) Cyprinodontiformes 30.9% (91 of 294 spp.), including 40 spp. as VU, 49 spp. as EN, and two spp. as CR; (iv) Cichliformes: 26.2% (54 of 206 spp.), including 25 spp. as VU, 27 spp. as EN, and two spp. as CR; (v) Gymnotiformes 32.1% (36 of 112 spp.), including 14 spp. as VU, 21 spp. as EN, and one as CR; and (vi) species in other clades: 16.6% (29 of 174 spp.), including 16 spp. as VU, and 13 spp. as EN (Tab. 5).
TABLE 5 | Summary of the ConR preliminary conservation assignments for 2,334 NFF species.
| Order | NFF | Threatened | % |
| Characiformes | 690 | 210 | 30.4 |
| Siluriformes | 858 | 251 | 29.3 |
| Cyprinodontiformes | 294 | 91 | 31.0 |
| Cichliformes | 206 | 54 | 26.2 |
| Gymnotiformes | 112 | 36 | 32.1 |
| Other orders | 174 | 29 | 16.7 |
| TOTAL | 2,334 | 671 | 28.7 |
Discussion
Threat status in NFF species. The Neotropical ichthyofauna is among the most diverse on Earth, with published estimates of more than 9,000 species or about 14% of all vertebrate species. This diversity of NFF species is dominated by three orders of ostariophysan fishes (Characiformes, Siluriformes, and Gymnotiformes) and two orders of acanthomorph fishes (i.e., Cichliformes, Cyprinodontiformes). This study suggests that ca. 14%, or about one species in seven, of all NFF species are under some level of extinction risk (Fig. 2). The estimated 14% of threatened NFF species is lower than corresponding figures for the freshwater ichthyofauna in other continents; e.g., about 37% in Europe (Freyhof, Brooks, 2011), 27% in North America (IUCN, 2020), and 22% in Africa (Snoeks et al., 2011). However, absolute numbers are alarming with 422 species at extinction risk (Tab. 1) and it will certainly increase because nearly half of all NFF species are still awaiting IUCN assessment. The extinction risk is similar among ostariophysans and cichliform species, with an average of 11.0% of species assigned to threat categories, but cyprinodontiform killifishes are at much greater risk, with nearly 48% of species in threat categories (Tab. 1). Cyprinodontiformes include a high proportion of small-bodied species living in seasonal pools and other ephemeral aquatic habitats, with narrow geographic distributions within hydrologically isolated upland and coastal river basins (Fig. 3D), often in areas of the Brazilian shield with high human impact (Costa, 2019). These features place killifishes among the most vulnerable vertebrates in the Neotropical region (Costa, 2016; Costa, 2019).
Predictor variables of extinction risks. To evaluate species’ extinction risks, the IUCN-RL uses a standardized protocol with a widely-accepted set of criteria (IUCN, 2019). This assessment is a laborious and time-consuming process based on experts’ opinions and data that are not readily available for many taxonomic groups. The urgency to prioritize species conservation of threatened ecosystems has motivated the use of alternatives for speeding conservation assessments, by identifying geographic regions and species traits linked to higher extinction risks (Bland et al., 2015; Gonzalez-del-Pliego et al., 2019). Our findings support the longstanding view that geographic range size is a predictor of extinction risk (Purvis et al., 2000; Poff et al., 2012), and also more recently hypothesis that threatened status is, to some degree, associated with elevation gradients (Reis et al., 2016) and species’ publication date (Tagliacollo et al., 2020) (Fig. 3).
Biodiversity patterns seen here for NFF resembles those of most biotas on Earth, in being characterized by a highly-skewed frequency distribution of organisms among species, in which most individuals are members of just a few highly abundant species (>80% measured as numbers of individuals). Some argue that conservation efforts should prioritize ecologically dominant species that are purported to perform most of the current ecosystem functions (e.g., Gaston, Fuller, 2008; Winfree et al., 2015). This is a shortsighted strategy from both ecological and evolutionary perspectives. Some less abundant species (e.g., top predators, ecosystem engineers) contribute disproportionately to the functional structure of species assemblages (Leitão et al., 2016; Jousset et al., 2017). Other species are spatially structured so that they are only less abundant locally, despite being geographically widespread (Violle et al., 2017). In general, less abundant species often buffer ecosystem resilience to disturbance by providing stabilizing functional redundancy (Lyons et al., 2005; Mouillot et al., 2013; Dee et al., 2019). Although the functional diversity of NFFs is still poorly documented, preliminary studies show it is greater than other freshwater faunas (Su et al., 2019).
This study estimates that 44.5% (1,337 of 3,001 species) of NFF species have an EOO smaller than 20,000 km2, which is an area equivalent to a square land parcel just 213 km on each side (Tab. 1; Fig. 3D). Like all species, freshwater fishes have particular habitat requirements (e.g., large river channels, floodplain lakes, rainforest streams, etc.) with specialized ecological and physiological traits, and therefore most NFF species occupy only a fraction of the theoretical maximum amount of aquatic habitat, with many species occupying a tiny spatial footprint of less than 100 km2 of aquatic habitat, equivalent to a parcel less than 10 km on a side.
The effect of geographic range size on extinction risk varies both by region and among taxa. Fish diversity is often correlated with forest cover at a regional scale, although this pattern is more heterogeneous at more local scales (Lo et al., 2020). Both paleontological and macroecological studies have shown that geographic range is often the most important predictor of long-term evolutionary survival (Harnik et al., 2012; Toledo et al., 2014; Foote et al., 2016; Longrich et al., 2016). These conclusions suggest that current reductions in geographic range size will lead to pronounced increases in long-term extinction risk even if local populations are relatively large and stable at present. Such a pattern is even more critical for groups whose distributions are already naturally restricted, as seen in Cyprinodontiformes (Tab. 3; Fig. 3D).
The diverse NFF resembles that of other continents in exhibiting pronounced elevation biodiversity gradients, with greatest species richness in the lowlands below about 250 meters (Oberdorff et al., 2011; Costa et al., 2018; Albert et al., 2020b). This elevation-diversity gradient is part of a larger core-periphery biodiversity pattern observed in South American freshwater fishes, with higher species richness and lower percent endemism in lowland basins of the continental core (i.e., Amazon-Orinoco lowlands) and lower species richness and higher percent endemism in upland basins of the continental periphery (e.g., Shields and Andes) (Albert et al., 2011b; Dagosta, de Pinna, 2019; Oberdorff et al., 2019). Similar core-periphery patterns of species richness and percent endemism are also observed in many groups of South American plants (Ramírez-Barahona et al., 2011; Antonelli et al., 2018), frogs (Vasconcelos et al., 2019), and snakes (Azevedo et al., 2020).
Our results suggest that extinction risks for NFF species are higher in upland rivers at the continental periphery (Fig. 3E; Fig. 4). About 23.6% of species at an elevation above 253.0 meters are recognized as threatened species by the IUCN-RL (Tab. 4). As noted above, many cyprinodontiform species are at higher extinction risk in uplands areas at the continental periphery, including regional species flocks in the Altiplano (e.g., Orestias; Guerrero-Jiménez et al., 2017), Central America (e.g., Goodeidae; Foster, Piller, 2018), and Mata Atlântica (e.g., Nematolebias; Costa et al., 2014). Although phenotypic specialization is often thought to promote adaptive diversification (Petren et al., 2005; Seehausen, 2006; Pinto et al., 2008), the specializations of many NFF species to specific habitat types in upland rivers (e.g., waterfalls, torrential hill streams) potentially constrain their elevational and geographic distributions. Therefore the same traits that may contribute to higher local species richness may be disadvantageous for survival in environments affected by humans (Ceretta et al., 2020). Although the aggregated effects of ecological and physiological constraints on elevation distributions in NFF species are poorly understood, it is likely that restricted geographic ranges, habitat availability, and habitat connectivity all strongly contribute to the threatened status of NFF species in upland rivers (Lanés et al., 2014; Silva et al., 2015).
We found that the dates of NFF species’ publications are associated with extinction risks, in which more recently described species are assigned to higher threat categories by the IUCN–RL (Fig. 3). This association is most apparent in Cyprinodontiformes, with about 44% of species described in a 20–year interval from 1996 to 2016 (Tab. 2; Fig. 3). As in other taxonomic groups (e.g., birds, mammals), NFF species with widespread geographic distributions and/or high local abundances are often described earlier in the discovery process, and these species therefore also suffer lower extinction risk. In contrast, range-restricted species, often located in remote areas or unusual habitats of South America, are only recently being collected and described by ichthyologists (e.g., Akawaio penak Maldonado-Ocampo, López-Fernández, Taphorn, Bernard, Crampton & Lovejoy, 2014, and Tarumania walkerae de Pinna, Zuanon, Rapp Py-Daniel & Petry, 2017), and we generally have much less knowledge about their actual geographic and ecological ranges.
Using recent rates of species discovery and description, studies have forecast that about ca. 3,000 NFF species remain to be described (Reis et al., 2016, and reference therein). Results from this study indicate that most of these “yet-to-be-described” NFF species will be range-restricted, potentially threatened, and possibly data deficient on arrival. This is obviously a large and alarming number, which potentially exceeds the total number of (breeding) Neotropical birds (ca. 2,250 species; Rahbek et al., 2007), or the total number of obligate freshwater fish species in North America and Europe combined (ca. 1,460 species). The highly endemic spatial structure of Neotropical fishes means this fauna is vulnerable to mass extinction due to anthropogenic activities (Pelicice et al., 2017; Bezerra et al., 2019; Grasel et al., 2019).
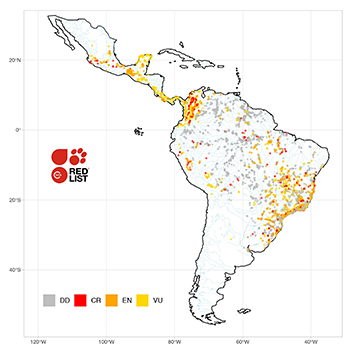
FIGURE 4 | Collection points for 442 threatened Neotropical freshwater fishes (NFF). Threatened NFF species classified by the IUCN Red List (RL) are often distributed in the upland rivers of the Brazilian Shield and the Colombian Andes, and coastal Atlantic and Caribbean drainages. CR: Critically Endangered; EN: Endangered; VU: Vulnerable; DD: Data Deficient. Data for 4,412 localities with geographic coordinates.
Distribution range sizes of threatened NFFs species. Threatened NFF species are often distributed in upland rivers at the continental periphery (Fig. 4). In general, such species include those described decades ago with range-restricted distributions in the Brazilian Shield and the Colombian Andes, and coastal Atlantic and Caribbean drainages (Fig. 5). Higher concentration of threatened species in Colombia and Brazil, and a few species in other Latin American countries, is explained by the efforts of those two countries in generating national lists of threatened species in accordance with IUCN protocols (Mojica et al., 2012; ICMBio, 2018). NFF species in those aquatic environments are under higher extinction risks due to increasing threats caused by, among other things, expansion of agriculture (Rosa et al., 2020), implementation of hydropower plants (Finer, Jenkins, 2012), and urbanization (McKinney, 2006).
The amount of land surface area converted to agricultural activities varies substantially by region, but across the Neotropics natural vegetation cover is being removed to support human demands for commodities like soy, corn, palm oil, among others (Pütz et al., 2014). The conversion of natural lands to agriculture fields has been increasing in recent years with tropical forests, savannas and temperate forest being the most affected areas (Poorter et al., 2016). Agriculture activities can have a significant impact on freshwater ecosystems due to the flowing of fertilizers, herbicides and pesticides into rivers affecting the ecology of aquatic organisms and imposing threat on fish populations already under extinction risk (Albert et al., 2020a).
Hydroelectric dams impose other threats to NFF species, by transforming rivers into reservoirs leading to the extirpation of rheophilic species and collapse of migratory fish populations (Winemiller et al., 2016; Hrbek et al., 2018). Besides changes in environmental landscapes, hydropower plants modify the hydrological regime downstream, disturbing seasonal reproductive, feeding cycles, and migratory routes (Helfman, 2007). Small capacity dams have proliferated across the South America continent blocking headwater streams in upland regions (Grill et al., 2019). Brazil alone has built close to 500 small dams and many other projects are pending licensing by the national agency of energy production (ANEEL, 2015). As a consequence, the connectivity of fish populations in headwater river systems have been fragmented, preventing faunal movements and the expansion of geographic ranges.
Worldwide about 1.7 billion people live in cities that draw water from freshwater ecoregions of high biodiversity value (Abell et al., 2019). Some of the largest Brazilian metropolitan areas lie at the headwaters of large tropical river basins. For example, the São Paulo greater metropolitan area (ca. 23.4 million people) strides the headwaters of the Tietê River, Brasília (ca. 4.3 million people) the headwaters of the Tocantins, São Francisco and Paraná Rivers, Belo Horizonte (ca. 2.7 million people) at headwaters of the das Velhas River (largest tributary of the São Francisco River), Curitiba (ca. 1.9 million people) at headwaters of the Iguaçu River, and Cuiabá (ca. 600,000 people) at headwaters of the Cuiabá River (major tributary of the Paraguay River). Outside of Brazil, there are examples of the same pattern in Colombia: metropolitan areas of Medellín (ca. 3.7 million people) and Cali (ca. 2.3 million people) at the headwaters of the Cauca-Magdalena River, and Barranquilla (ca. 2 million people) at its mouth. These large cities significantly reduce both the quality and quantity of water flowing downstream, from the combined effects of water withdrawals that lower the regional water table, and water pollution from urban and agricultural runoff, as well as industrial and residential sewage effluents. Finally, human footprint on aquatic resources from these and other cities throughout Latin America is expected to rise dramatically in coming years, with projected increases both in absolute numbers of people and per capita consumption of water, energy, food and other resources (Hoekstra, Mekonnen, 2012; Mekonnen, Gerbens-Leenes, 2020).
Additional impacts are projected in Atlantic coastal drainages, as more seashore and coastal estuaries are converted to urban and agricultural landscapes, affecting the quantity and quality of freshwater habitats in these areas with high local fish endemism (Camelier, Zanata, 2014; Bertaco et al., 2016; Hughes et al., 2020; Silva et al., 2020). Urbanization has distinct patterns in the Neotropics, with remote regions in, e.g., the Western Amazon, Guianas and Altiplano having the lowest fraction of land converted to urban or agricultural purposes, at least to date. However, the southeast Atlantic, Maracaibo-Caribbean and Eastern Atlantic regions have suffered the highest degree of urbanization (Gwynne, 2017), pressuring biotas with the highest concentration of threatened species in the Neotropics.
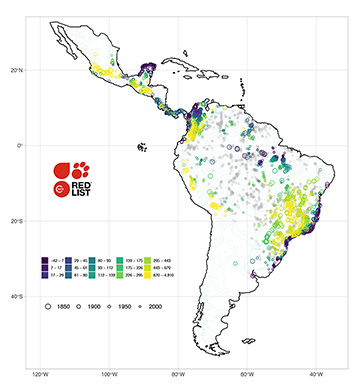
FIGURE 5 | Collection points for 442 threatened Neotropical freshwater fishes (NFF) colored by elevation and sized by species’ description year. Threatened NFF species are often those described decades ago, with range-restricted distributions in the upland rivers of the Brazilian Shield and the Colombian Andes, and coastal Atlantic and Caribbean drainages. CR: Critically Endangered; EN: Endangered; VU: Vulnerable; DD: Data Deficient (gray). Data for 4,412 localities with geographic coordinates.
Distribution range sizes of potentially threatened NFF species. This study generated preliminary conservation assessments using EOO for 2,334 NFF species currently awaiting IUCN assessment, identifying an additional 671 NFF species as potentially threatened. This number represents 29% of NFF species currently lacking IUCN assessment. With 422 species currently on the IUCN RL, and the addition of 671 species suggested by the ConR package, the total number of threatened or potentially threatened Neotropical freshwater fish species has risen to 1,093 species, or about 18% of all species in the fauna.
Geographic range is widely regarded as an important predictor of extinction risk in freshwater taxa (Olden et al., 2010; Collen et al., 2014). The ConR package integrates information on species geographic data to calculate EOOs and compute IUCN threatened categories without explicit locations (sensu IUCN, 2019) that may affect species’ spatial ranges. A location is defined by the IUCN guidelines as a geographically or ecologically distinct area in which a single threatening event can rapidly affect all individuals of the taxon. Thus, the ConR provides a preliminary solution to estimate species conservation threat status without detailed information (e.g., locations) required to achieve complete IUCN–RL status. We applied ConR to generate a data-driven baseline to identify potentially threatened species and their geographic ranges in the Neotropical region. We stress the ConR package is not at all intended to replace the IUCN Red Listing assessments; rather, it aims to assist and facilitate this process by red-flagging potentially threatened species and geographic areas. ConR estimates of threat status must be determined carefully, and the flagged species must be understood as potentially threatened which may be endangered in the presence of environmental threats.
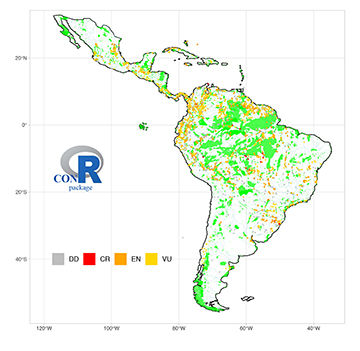
ConR reveals that potentially threatened NFF species are often restricted to single river basins located outside protected areas at high altitudes of the northern, central and southern Andes, and Eastern Guiana Shield (Fig. 6). These highland regions in the Neotropics are relatively species-poor, comprising less than 30.6% of the Neotropical ichthyofauna inhabiting river drainages above 253.0 meters (Tab. 4). The low diversity contrasts with the high endemism in both the Andean Cordillera (Schaefer, 2011) and Eastern Guiana Shield (Lujan, Armbruster, 2011), which encompasses an ichthyofauna comprised of relictual lineages with specialized phenotypes. Potentially threatened species along the Andean Cordilleras includes emblematic species in the Chilean ichthyofauna (e.g., the catfishes Diplomystes and Nematogenys (Guichenot, 1848)), climbing catfishes (Astroblepus) in Peru, Ecuador and Colombia, and Altiplano killifishes (Orestias) in Lake Titicaca, to mention a few. The isolated Brazilian coastal drainages and the Guiana Shield also harbor a disproportionate number of low-diversity fish clades (i.e., with few species), but which represent long phylogenetic branches (i.e., relatively early-branching) within all the major taxonomic orders; e.g., Conorhynchus conirostris (Valenciennes, 1840), Delturus, Lithogenes, Trichogenes and Wertheimeria maculata Steindachner, 1877(Siluriformes), Hollandichthys, Lignobrycon myersi (Miranda Ribeiro 1956), Mimagoniates, Nematocharax,and Spintherobolus (Characiformes), Akawaio penak and Japigny kirschbaum Meunier, Jégu & Keith, 2011 (Gymnotiformes), Guianacara and Mazarunia (Cichliformes), and Nematolebias (Cyprinodontiformes). Peripheral basins in the Guiana Shield have been proposed as museums of diversity where lineages have resisted extinction for many millions of years (Albert et al., 2011b). The unique ichthyofaunas of the South American uplands are centers of taxonomic and phylogenetic diversity (Faith, 1992; Magurran, 2013), and important biodiversity measures should be considered by policy makers when developing conservation actions (Strecker et al., 2011; Li et al., 2020).
The upland reaches of large Amazonian tributaries draining the central Brazilian Shield are also important centers of threatened freshwater fish species; e.g., the upper Aripuanã, Juruena, Teles Pires, and Iriri Rivers. That region concentrates species highlighted by the ConR package as potentially Critically Endangered (CR) including highly specialized rheophilic lineages; e.g., Gymnotus Lamontianus (Gymnotiformes), Baryancistrus longipinnis (Kindle 1895), Scobinancistrus (Siluriformes), Rhinopetitia, Leporinus tristriatus Birindelli & Britski, 2013, Sartor, Ossubtus, Utiaritichthys (Characiformes), Retroculus, Teleocichla (Cichliformes). That portion of the northern Brazilian Shield is a high conservation priority for freshwater fishes, as a region of high endemism, few protected areas and numerous planned or existing hydropower dams (Winemiller et al., 2016; Dagosta et al., 2020). This region is also the focus of intense deforestation along the expanding agricultural frontier of the Brazilian uplands (Spera et al., 2016). The analysis also highlights the occurrence of some potentially threatened species in drainages near the South American Atlantic margin, with emphasis on the Iguaçu River (Daga et al., 2016) and Uruguay River (Bertaco et al., 2016). The latter two basins belong to the most threatened Brazilian biome, the Mata Atlântica, and are located close to large urban centers.
FIGURE 6 | Collection points for 671 potentially threatened Neotropical Freshwater Fishes (NFF). Potentially threatened NFF species predicted by the ConR package using EOO estimates are usually distributed outside protected areas (e.g., national parks, indigenous lands: green) and more often located in the upland rivers of the northern, central and southern Andes, and Eastern Guiana Shield. CR: Critically Endangered; EN: Endangered; VU: Vulnerable; LC or NT: Least Concern or Near Threatened; DD: Data Deficient. Data for 4,412 localities with geographic coordinates. Protected areas (green) from: https://www.protectedplanet.net.
Concluding remarks. Continental freshwaters support diverse but fragile ecosystems that are widely imperiled by human activities. The conservation status of many freshwater fishes will benefit by prioritizing geographic areas with the largest number of coexisting and threatened species (Jézéquel et al., 2020; Leal et al., 2020). Hundreds of fishes with smaller adult body sizes and restricted geographic ranges are known to be threatened with extinction (Castro, Polaz, 2020). This study verifies that often threatened NFF species were recently described, are range-restricted, and are confined to certain upland portions of the Brazilian Shield and Northern Andes and to lowland portions of the Brazilian and Caribbean coastal drainages. Based on the ConR preliminary assignment estimates for NFF species, we estimate that about 29% of the species awaiting extinction risk assessments are potentially threatened. These potentially threatened NFF species are more commonly located outside formally protected areas (Azevedo‐Santos et al., 2019; Dagosta et al., 2020) in the Central and Southern Andes and Eastern Guiana Shield. Our results expand the number and geographic distribution of threatened NFF species from 422 species currently on the IUCN RL to 1,093 threatened or potentially threatened, representing about 18% of all described NFF species. Therefore, conservation efforts directed towards Neotropical aquatic biodiversity must prioritize habitats in uplands and coastal lowlands, particularly in the Andean Cordilleras, South American shields, and Brazilian and Caribbean coastal drainages. Rivers of the Central and Southern Andes, and Eastern Guiana Shield should be prioritized in the upcoming IUCN RL assessments for NFF species conservation efforts.
Acknowledgments
We are grateful to Leandro Sousa (UFPA), Douglas Bastos (INPA), and José Birindelli (MZUEL) for sharing the photos of Pituna xinguensis, Anablepsoides xinguensis, and Colomesus tocantinensis, respectively, and to Cristhian Conde for comments on characiform species of Southeastern Brazil. We wish to express our appreciation for William Eschmeyer’s Catalogue of Fishes, without which this study would not have been possible. VAT is supported by Fundação de Auxílio à Pesquisa do Estado de São Paulo (FAPESP process #2018/20806–3); JSA by United States National Science Foundation DEB 0614334, 0741450, and 1354511; FCPD by FAPESP (#2016/19075–9) and by CNPq (#405643/2018–7); RER and MDP by CNPq (#306455/2014–5 and #310688/2019–1, respectively).
References
Abell R, Vigerstol K, Higgins J, Kang S, Karres N, Lehner B et al. Freshwater biodiversity conservation through source water protection: quantifying the potential and addressing the challenges. Aquat Conserv. 2019; 29(7):1022–38. https://doi.org/10.1002/aqc.3091
Agência Nacional de Energia Elétrica (ANEEL). Sistema de informações georeferenciadas do setor elétrico – SIGEL [Internet]. 2015. Available from: https://sigel.aneel.gov.br
Albert JS, Carvalho TP, Petry P, Holder MA, Maxime EL, Espino J et al. Aquatic biodiversity in the Amazon: habitat specialization and geographic isolation promote species richness. Animals. 2011a; 1(2):205–41. https://doi.org/10.3390/ani1020205
Albert JS, Craig J, Tagliacollo VA, Petry P. Upland and lowland fishes: a test of the river capture hypothesis. In: Hoorn C, Perrigo A, Antonelli A, editors. Mountains, climate and biodiversity. Cambridge: Wiley Press; 2018. p.273–94.
Albert JS, Destouni G, Duke-Sylvester SM, Magurran AE, Oberdorff T, Reis RE et al. Scientists’ warning to humanity on the freshwater biodiversity crisis. Ambio. 2020a; 50:85–94. https://doi.org/10.1007/s13280-020-01318-8
Albert JS, Petry P, Reis R. Major biogeographic and phylogenetic patterns. In: Albert JS, Reis R, editors. Historical biogeography of Neotropical freshwater fishes. Berkley, Los Angeles, London: University of California Press; 2011b. p.21–57.
Albert JS, Reis R, editors. Historical biogeography of neotropical freshwater fishes. Berkley, Los Angeles, London: University of California Press; 2011.
Albert JS, Schoolmaster DR Jr, Tagliacollo VA, Duke-Sylvester SM. Barrier displacement on a neutral landscape: toward a theory of continental biogeography. Syst Biol. 2017; 66(2):167–82. https://doi.org/10.1093/sysbio/syw080
Albert JS, Tagliacollo VA, Dagosta F. Diversification of Neotropical freshwater fishes. Annu Rev Ecol Evol Syst. 2020b; 51(1):27–53. https://doi.org/10.1146/annurev-ecolsys-011620-031032
Antonelli A, Zizka A, Carvalho FA, Scharn R, Bacon CD, Silvestro D et al. Amazonia is the primary source of Neotropical biodiversity. Proc Natl Acad Sci USA. 2018; 115(23):6034–39. https://doi.org/10.1073/pnas.1713819115
Azevedo JAR, Guedes TB, Nogueira CC, Passos P, Sawaya RJ, Prudente ALC et al. Museums and cradles of diversity are geographically coincident for narrowly distributed Neotropical snakes. Ecography. 2020; 43(2):328–39. https://doi.org/10.1111/ecog.04815
Azevedo-Santos VM, Frederico RG, Fagundes CK, Pompeu PS, Pelicice FM, Padial AA et al. Protected areas: a focus on Brazilian freshwater biodiversity. Divers Distrib. 2019; 25(3):442–48. https://doi.org/10.1111/ddi.12871
Bachman S, Moat J, Hill A, de la Torre J, Scott B. Supporting Red List threat assessments with GeoCAT: geospatial conservation assessment tool. ZooKeys. 2011; 150:117–26. https://doi.org/10.3897/zookeys.150.2109
Berra TM. Freshwater fish distribution. Chicago: Chicago Press; 2007.
Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America—a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401–40. https://doi.org/10.11646/zootaxa.4138.3.1
Bezerra LAV, Freitas MO, Daga VS, Occhi TVT, Faria L, Costa APL et al. A network meta-analysis of threats to South American fish biodiversity. Fish Fish. 2019; 20(4):620–39. https://doi.org/10.1111/faf.12365
Bland LM, Collen B, Orme CDL, Bielby J. Predicting the conservation status of data-deficient species. Conserv Biol. 2015; 29(1):250–59. https://doi.org/10.1111/cobi.12372
Camelier P, Zanata AM. Biogeography of freshwater fishes from the Northeastern Mata Atlântica freshwater ecoregion: distribution, endemism, and area relationships. Neotrop Ichthyol. 2014; 12(4):683–98. https://doi.org/10.1590/1982-0224-20130228
Castro RMC, Polaz CNM. Small-sized fish: the largest and most threatened portion of the megadiverse neotropical freshwater fish fauna. Biota Neotrop. 2020; 20(1):e20180683. https://doi.org/10.1590/1676-0611-BN-2018-0683
Ceretta BF, Fogliarini CO, Giglio VJ, Maxwell MF, Waechter LS, Bender MG. Testing the accuracy of biological attributes in predicting extinction risk. Perspect Ecol Conserv. 2020;18(1):12–18. https://doi.org/10.1016/j.pecon.2020.01.003
Collen B, Whitton F, Dyer EE, Baillie JEM, Cumberlidge N, Darwall WRT et al. Global patterns of freshwater species diversity, threat and endemism. Glob Ecol Biogeogr. 2014; 23(1):40–51. https://doi.org/10.1111/geb.12096
Costa ID, Petry AC, Mazzoni R. Responses of fish assemblages to subtle elevations in headwater streams in southwestern Amazonia. Hydrobiologia. 2018; 809:175–84. https://doi.org/10.1007/s10750-017-3463-1
Costa WJEM. Inferring Evolution of habitat usage and body size in endangered, seasonal Cynopoeciline killifishes from the South American Atlantic Forest through an integrative approach (Cyprinodontiformes: Rivulidae). PLoS ONE. 2016; 11(7): e0159315. https://doi.org/10.1371/journal.pone.0159315
Costa WJEM. Description of a new species of cynopoeciline killifish (Cyprinodontiformes, Aplocheilidae), possibly extinct, from the Atlantic Forest of south-eastern Brazil. ZooKeys. 2019; 867:73–85. https://doi.org/10.3897/zookeys.867.34034
Costa WJEM, Amorim PF, Aranha GN. Species limits and DNA barcodes in Nematolebias, a genus of seasonal killifishes threatened with extinction from the Atlantic Forest of south-eastern Brazil, with description of a new species (Teleostei: Rivulidae). Ichthyol Explor Freshw. 2014; 24(3):225–36.
Crampton W. An ecological perspective on diversity and distributions. In: Albert JS, Reis R, editors. Historical biogeography of Neotropical freshwater fishes. Berkley, Los Angeles, London: University of California Press; 2011. p.165–89.
Daga V, Debona T, Abilhoa V, Gubiani É, Vitule J. Non-native fish invasions of a Neotropical ecoregion with high endemism: a review of the Iguaçu River. Aquat Invasions. 2016; 11(2):209–23. http://dx.doi.org/10.3391/ai.2016.11.2.10
Dagosta FCP, de Pinna M. Biogeography of Amazonian fishes: deconstructing river basins as biogeographic units. Neotrop Ichthyol. 2017; 15(3):e170034. https://doi.org/10.1590/1982-0224-20170034
Dagosta FCP, de Pinna M. The fishes of the Amazon: distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; (431):1–163. Available from: http://digitallibrary.amnh.org/handle/2246/6940
Dagosta FCP, de Pinna M, Peres CA, Tagliacollo VA. Existing protected areas provide a poor safety-net for threatened Amazonian fish species. Aquat Conserv. 2020; 31(5):1167–89. https://doi.org/10.1002/aqc.3461
Dauby G, Stévart T, Droissart V, Cosiaux A, Deblauwe V, Simo-Droissart M et al. ConR: an R package to assist large-scale multispecies preliminary conservation assessments using distribution data. Ecol Evol. 2017; 7(24):11292–303. https://doi.org/10.1002/ece3.3704
Dee LE, Cowles J, Isbell F, Pau S, Gaines SD, Reich PB. When do ecosystem services depend on rare species? Trends Ecol Evol. 2019; 34(8):746–58. https://doi.org/10.1016/j.tree.2019.03.010
Dudgeon D, Arthington AH, Gessner MO, Kawabata Z-I, Knowler DJ, Lévêque C et al. Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev Cambridge Philos Soc. 2006; 81(2):163–82. https://doi.org/10.1017/s1464793105006950
Faith DP. Conservation evaluation and phylogenetic diversity. Biol Conserv. 1992; 61(1):1–10. https://doi.org/10.1016/0006-3207(92)91201-3
Farr TG, Rosen PA, Caro E, Crippen R, Duren R, Hensley S et al. The shuttle radar topography mission. Rev Geophys. 2007; 45(2):1–33. https://doi.org/10.1029/2005RG000183
Ferreira J, Aragão LEOC, Barlow J, Barreto P, Berenguer E, Bustamante M et al. Brazil’s environmental leadership at risk. Science. 2014; 346(6210):706–07. https://doi.org/10.1126/science.1260194
Finer M, Jenkins CN. Proliferation of hydroelectric dams in the Andean Amazon and implications for Andes-Amazon connectivity. PLoS ONE. 2012; 7(4):e35126. https://doi.org/10.1371/journal.pone.0035126
Foote M, Ritterbush KA, Miller AI. Geographic ranges of genera and their constituent species: structure, evolutionary dynamics, and extinction resistance. Paleobiology. 2016; 42(2):269–88. https://doi.org/10.1017/pab.2015.40
Foster KL, Piller KR. Disentangling the drivers of diversification in an imperiled group of freshwater fishes (Cyprinodontiformes: Goodeidae). BMC Evol Biol. 2018; 18:116. https://doi.org/10.1186/s12862-018-1220-3
Freyhof J, Brooks E. European red list of freshwater fishes. Luxembourg: Publications Office of the European Union; 2011. Available from: https://ec.europa.eu/environment/nature/conservation/species/redlist/downloads/European_freshwater_fishes.pdf
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2020. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Gaston KJ, Fuller RA. Commonness, population depletion and conservation biology. Trends Ecol Evol. 2008; 23(1):14–19. https://doi.org/10.1016/j.tree.2007.11.001
Gonzalez-del-Pliego P, Freckleton RP, Edwards DP, Koo MS, Scheffers BR, Pyron RA et al. Phylogenetic and trait-based prediction of extinction risk for data-deficient amphibians. Curr Biol. 2019; 29(9):1557–63. https://doi.org/10.1016/j.cub.2019.04.005
Grasel D, Fearnside PM, Vitule JRS, Bozelli RL, Mormul RP, Rodrigues RR et al. Brazilian wetlands on the brink. Biodivers Conserv. 2019; 28:255–57. https://doi.org/10.1007/s10531-018-1666-z
Grill G, Lehner B, Thieme M, Geenen B, Tickner D, Antonelli F et al. Mapping the world’s free-flowing rivers. Nature. 2019; 569(7755):215–21. https://doi.org/10.1038/s41586-019-1111-9
Guerrero-Jiménez CJ, Peña F, Morales P, Méndez M, Sallaberry M, Vila I et al. Pattern of genetic differentiation of an incipient speciation process: the case of the high Andean killifish Orestias. PLoS ONE. 2017; 12(2):e0170380. https://doi.org/10.1371/journal.pone.0170380
Gwynne RN. Industrialization and urbanization in Latin America. London: Routledge; 2017.
Harnik PG, Simpson C, Payne JL. Long-term differences in extinction risk among the seven forms of rarity. Proc R Soc B Biol Sci. 2012; 279:4969–76. https://doi.org/10.1098/rspb.2012.1902
Helfman G. Fish conservation: a guide to understanding and restoring global aquatic biodiversity and fishery resources. Washington: Island Press; 2007.
Hoekstra AY, Mekonnen MM. The water footprint of humanity. Proc Natl Acad Sci USA. 2012; 109(9):3232–37. https://doi.org/10.1073/pnas.1109936109
Hrbek T, Meliciano NV, Zuanon J, Farias IP. Remarkable geographic structuring of rheophilic fishes of the lower Araguaia River. Front Genet. 2018; 9:1–12. https://doi.org/10.3389/fgene.2018.00295
Hughes LC, Cardoso YP, Sommer JA, Cifuentes R, Cuello M, Somoza GM et al. Biogeography, habitat transitions and hybridization in a radiation of South American silverside fishes revealed by mitochondrial and genomic RAD data. Mol Ecol. 2020; 29(4):738–51. https://doi.org/10.1111/mec.15350
Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio). Livro vermelho da fauna brasileira ameaçada de extinção: volume VI – Peixes. In: Instituto Chico Mendes de Conservação da Biodiversidade, organizers. Livro vermelho da fauna brasileira ameaçada de extinção. Brasília: ICMBio/MMA; 2018.
International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for using the IUCN Red List categories and criteria. Version 14 [Internet]. Gland; 2019. Available from: https://www.iucnredlist.org/resources/redlistguidelines
International Union for Conservation of Nature (IUCN). The IUCN Red List of Threatened Species Version 2020-3. Gland; 2020. Available from: https://www.iucnredlist.org
Jézéquel C, Tedesco PA, Darwall W, Dias MS, Frederico RG, Hidalgo M et al. Freshwater fish diversity hotspots for conservation priorities in the Amazon Basin. Conserv Biol. 2020; 34(4):956–65. https://doi.org/10.1111/cobi.13466
Jousset A, Bienhold C, Chatzinotas A, Gallien L, Gobet A, Kurm V et al. Where less may be more: how the rare biosphere pulls ecosystems strings. ISME J. 2017; 11:853–62. https://doi.org/10.1038/ismej.2016.174
Kalinkat G, Jähnig SC, Jeschke JM. Exceptional body size–extinction risk relations shed new light on the freshwater biodiversity crisis. Proc Natl Acad Sci USA. 2017; 114(48):E10263–64. https://doi.org/10.1073/pnas.1717087114
Lanés LEK, Gonçalves ÂC, Volcan MV, Lanés LEK, Gonçalves ÂC, Volcan MV. Discovery of endangered annual killifish Austrolebias cheradophilus (Aplocheiloidei: Rivulidae) in Brazil, with comments on habitat, population structure and conservation status. Neotrop Ichthyol. 2014; 12(1):117–24. https://doi.org/10.1590/S1679-62252014000100012
Leal CG, Lennox GD, Ferraz SFB, Ferreira J, Gardner TA, Thomson JR et al. Integrated terrestrial-freshwater planning doubles conservation of tropical aquatic species. Science. 2020; 370(6512):117–21. https://doi.org/10.1126/science.aba7580
Leitão RP, Zuanon J, Villéger S, Williams SE, Baraloto C, Fortunel C et al. Rare species contribute disproportionately to the functional structure of species assemblages. Proc R Soc B Biol Sci. 2016; 283:20160084. https://doi.org/10.1098/rspb.2016.0084
Li D, Olden JD, Lockwood JL, Record S, McKinney ML, Baiser B. Changes in taxonomic and phylogenetic diversity in the Anthropocene. Proc R Soc B Biol Sci. 2020; 287:20200777. https://doi.org/10.1098/rspb.2020.0777
Lo M, Reed J, Castello L, Steel EA, Frimpong EA, Ickowitz A. The influence of forests on freshwater fish in the tropics: a systematic review. BioScience. 2020; 70(5):404–14. https://doi.org/10.1093/biosci/biaa021
Longrich NR, Scriberas J, Wills MA. Severe extinction and rapid recovery of mammals across the Cretaceous–Palaeogene boundary, and the effects of rarity on patterns of extinction and recovery. J Evol Biol. 2016; 29(8):1495–512. https://doi.org/10.1111/jeb.12882
Lughadha EN, Walker BE, Canteiro C, Chadburn H, Davis AP, Hargreaves S et al. The use and misuse of herbarium specimens in evaluating plant extinction risks. Philos Trans R Soc BBiol Sci. 2019; 374:20170402. https://doi.org/10.1098/rstb.2017.0402
Lujan NK, Armbruster JW. The Guiana Shield. In: Albert JS, Reis R, editors. Historical biogeography of Neotropical freshwater fishes. Berkley, Los Angeles, London: University of California Press; 2011. p.211–24.
Lyons KG, Brigham CA, Traut BH, Schwartz MW. Rare species and ecosystem functioning. Conserv Biol. 2005; 19(4):1019–24. Available from: https://www.jstor.org/stable/3591286
Magurran AE. Measuring biological diversity. Malden: John Wiley & Sons; 2013.
McKinney ML. Urbanization as a major cause of biotic homogenization. Biol Conserv. 2006; 127(3):247–60. https://doi.org/10.1016/j.biocon.2005.09.005
Mekonnen MM, Gerbens-Leenes W. The water footprint of global food production. Water. 2020; 12(10):2696. https://doi.org/10.3390/w12102696
Mojica JIE, Usma Oviedo JUE, Alvarez León RE, Lasso CA. Libro rojo de peces dulceacuícolas de Colombia. 2012.
Mouillot D, Bellwood DR, Baraloto C, Chave J, Galzin R, Harmelin-Vivien M et al. Rare species support vulnerable functions in high-diversity rcosystems. PLoS Biol. 2013; 11(5):e1001569. https://doi.org/10.1371/journal.pbio.1001569
Nelson JS, Grande TC, Wilson MVH. Fishes of the World. Hoboken, New Jersey: John Wiley & Sons; 2016.
Oberdorff T, Dias MS, Jézéquel C, Albert JS, Arantes CC, Bigorne R et al. Unexpected fish diversity gradients in the Amazon basin. Sci Adv. 2019; 5:eaav8681. https://doi.org/10.1126/sciadv.aav8681
Oberdorff T, Tedesco PA, Hugueny B, Leprieur F, Beauchard O, Brosse S et al. Global and regional patterns in riverine fish species richness: a review. Int J Ecol. 2011; 2011:967631. https://doi.org/10.1155/2011/967631
Olden JD, Kennard MJ, Leprieur F, Tedesco PA, Winemiller KO, García-Berthou E. Conservation biogeography of freshwater fishes: recent progress and future challenges. Divers Distrib. 2010; 16(3):496–513. https://doi.org/10.1111/j.1472-4642.2010.00655.x
Pelicice FM, Azevedo-Santos VM, Vitule JRS, Orsi ML, Junior DPL, Magalhães ALB et al. Neotropical freshwater fishes imperiled by unsustainable policies. Fish Fish. 2017; 18(6):1119–33. https://doi.org/10.1111/faf.12228
Pelletier TA, Carstens BC, Tank DC, Sullivan J, Espindola A. Predicting plant conservation priorities on a global scale. Proc Natl Acad Sci USA. 2018; 115(51):13027–32. https://doi.org/10.1073/pnas.1804098115
Petren K, Grant PR, Grant BR, Keller LF. Comparative landscape genetics and the adaptive radiation of Darwin’s finches: the role of peripheral isolation. Mol Ecol. 2005; 14(10):2943–57. https://doi.org/10.1111/j.1365-294x.2005.02632.x
Pinto G, Mahler DL, Harmon LJ, Losos JB. Testing the island effect in adaptive radiation: rates and patterns of morphological diversification in Caribbean and mainland Anolis lizards. Proc R Soc B Biol Sci. 2008; 275(1652):2749–57. https://doi.org/10.1098/rspb.2008.0686
Poff NL, Olden JD, Strayer DL. Climate change and freshwater fauna extinction risk. In: Hannah L, editor. Saving a Million species: extinction risk from climate change. Washington, DC: Island Press/Center for Resource Economics; 2012. p.309–36.
Poorter L, Bongers F, Aide TM, Almeyda Zambrano AM, Balvanera P, Becknell JM et al. Biomass resilience of Neotropical secondary forests. Nature. 2016; 530:211–14. https://doi.org/10.1038/nature16512
Purvis A, Gittleman JL, Cowlishaw G, Mace GM. Predicting extinction risk in declining species. Proc R Soc Lond B Biol Sci. 2000; 267(1456):1947–52. https://doi.org/10.1098/rspb.2000.1234
Pütz S, Groeneveld J, Henle K, Knogge C, Martensen AC, Metz M et al. Long-term carbon loss in fragmented Neotropical forests. Nat Commun. 2014; 5:5037. https://doi.org/10.1038/ncomms6037
Rahbek C, Gotelli NJ, Colwell RK, Entsminger GL, Rangel TFLVB, Graves GR. Predicting continental-scale patterns of bird species richness with spatially explicit models. Proc R Soc B Biol Sci. 2007; 274(1607):165–74. https://doi.org/10.1098/rspb.2006.3700
Ramírez-Barahona S, Luna-Vega I, Tejero-Díez D. Species richness, endemism, and conservation of American tree ferns (Cyatheales). Biodivers Conserv. 2011; 20:59–72. https://doi.org/10.1007/s10531-010-9946-2
Reid AJ, Carlson AK, Creed IF, Eliason EJ, Gell PA, Johnson PTJ et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol Rev. 2019; 94(3):849–73. https://doi.org/10.1111/brv.12480
Reis RE, Albert JS, Dario FD, Mincarone MM, Petry P, Rocha LA. Fish biodiversity and conservation in South America. J Fish Biol. 2016; 89(1):12–47. https://doi.org/10.1111/jfb.13016
Robertson MP, Visser V, Hui C. Biogeo: an R package for assessing and improving data quality of occurrence record datasets. Ecography. 2016; 39(4):394–401. http://dx.doi.org/10.1111/ecog.02118
Rosa L, Chiarelli DD, Rulli MC, Dell’Angelo J, D’Odorico P. Global agricultural economic water scarcity. Sci Adv. 2020; 6(18):eaaz6031. http://dx.doi.org/10.1126/sciadv.aaz6031
Schaefer S. The Andes: riding the tectonic uplift. In: Albert JS, Reis R, editors. Historical biogeography of Neotropical freshwater fishes. Berkley, Los Angeles, London: University of California Press; 2011. p.259–78.
Seehausen O. African cichlid fish: a model system in adaptive radiation research. Proc R Soc B Biol Sci. 2006; 273(1597):1987–98. https://doi.org/10.1098/rspb.2006.3539
Silva AT, Chagas RJ, Santos ACA, Zanata AM, Rodrigues BK, Polaz CNM et al. Freshwater fishes of the Bahia State, Northeastern Brazil. Biota Neotrop. 2020; 20(4):e20200969. https://doi.org/10.1590/1676-0611-BN-2020-0969
Silva JFM, Jerep FC, Bennemann ST. New record and distribution extension of the endangered freshwater fish Cnesterodon hypselurus (Cyprinodontiformes: Poeciliidae) in the upper Paraná River basin, Brazil. Check List. 2015; 11(6):1811. https://doi.org/10.15560/11.6.1811
van der Sleen P, Albert JS. Field Guide to the Fishes of the Amazon, Orinoco, and Guianas. New Jersey: Princeton University Press; 2017.
Snoeks J, Harrison IJ, Stiassny MLJ. The status and distribution of freshwater fishes. In: Darwall W, Smith K, Allen D, Holland R, Harrison I, Brooks E, editors. The diversity of life in African freshwaters: underwater, under threat. An analysis of the status and distribution of freshwater species throughout mainland Africa. Cambridge, Gland: IUCN; 2011. p.42–73. Available from: https://portals.iucn.org/library/sites/library/files/documents/RL-6-001.pdf
Spera SA, Galford GL, Coe MT, Macedo MN, Mustard JF. Land-use change affects water recycling in Brazil’s last agricultural frontier. Glob Chang Biol. 2016; 22(10):3405–13. https://doi.org/10.1111/gcb.13298
Strecker AL, Olden JD, Whittier JB, Paukert CP. Defining conservation priorities for freshwater fishes according to taxonomic, functional, and phylogenetic diversity. Ecol Appl. 2011; 21(8):3002–13. https://doi.org/10.1890/11-0599.1
Su G, Villéger S, Brosse S. Morphological diversity of freshwater fishes differs between realms, but morphologically extreme species are widespread. Glob Ecol Biogeogr. 2019; 28(2):211–21. https://doi.org/10.1111/geb.12843
Tagliacollo VA, Camelier P, Zanata AM, Reis RE. A shocking discovery of threat risks on newly described species of weakly electric fishes. J Fish Biol. 2020; 96(5):1077–86. https://doi.org/10.1111/jfb.14096
Toledo LF, Becker CG, Haddad CFB, Zamudio KR. Rarity as an indicator of endangerment in neotropical frogs. Biol Conserv. 2014; 179:54–62. https://doi.org/10.1016/j.biocon.2014.08.012
Toussaint A, Charpin N, Brosse S, Villéger S. Global functional diversity of freshwater fish is concentrated in the Neotropics while functional vulnerability is widespread. Sci Rep. 2016; 6:22125. https://doi.org/10.1038/srep22125
Turak E, Harrison I, Dudgeon D, Abell R, Bush A, Darwall W et al. Essential biodiversity variables for measuring change in global freshwater biodiversity. Biol Conserv. 2017; 213(Part B):272–79. https://doi.org/10.1016/j.biocon.2016.09.005
Valladão GMR, Gallani SU, Pilarski F. South American fish for continental aquaculture. Rev Aquac. 2018; 10(2):351–69. https://doi.org/10.1111/raq.121640
Vasconcelos TS, Silva FR, Santos TG, Prado, VHM, Provete DB. Biogeographic patterns of South American anurans. Switzerland: Springer; 2019. https://doi.org/10.1007/978-3-030-26296-9
Violle C, Thuiller W, Mouquet N, Munoz F, Kraft NJB, Cadotte MW et al. Functional rarity: the ecology of outliers. Trends Ecol Evol. 2017; 32(5):356–67. https://doi.org/10.1016/j.tree.2017.02.002
Winemiller KO, McIntyre PB, Castello L, Fluet-Chouinard E, Giarrizzo T, Nam S et al. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science. 2016; 351(6269):128–29. https://doi.org/10.1126/science.aac7082
Winfree R, Fox JW, Williams NM, Reilly JR, Cariveau DP. Abundance of common species, not species richness, drives delivery of a real-world ecosystem service. Ecol Lett. 2015; 18(7):626–35. https://doi.org/10.1111/ele.12424
Zizka A, Silvestro D, Vitt P, Knight TM. Automated conservation assessment of the orchid family with deep learning. Conserv Biol. 2020; 35(3):897–908. https://doi.org/10.1111/cobi.13616
Authors
![]() Victor A. Tagliacollo1,
Victor A. Tagliacollo1,![]() Fernando Cesar Paiva Dagosta2,
Fernando Cesar Paiva Dagosta2,![]() Mário de Pinna1,
Mário de Pinna1,![]() Roberto E. Reis3 and
Roberto E. Reis3 and![]() James S. Albert4
James S. Albert4
[1] Museu de Zoologia da Universidade de São Paulo, Av. Nazaré, 481, 04263-000 São Paulo, SP, Brazil. (VAT) vatagliacollo@gmail.com, (MP) pinna@ib.usp.br.
[2] Faculdade de Ciências Biológicas e Ambientais, Universidade Federal da Grande Dourados, Rodovia Dourados/Itahum, km 12, 79804-970 Dourados, MS, Brazil. ferdagosta@gmail.com.
[3] Pontifícia Universidade Católica do Rio Grande do Sul, Av. Ipiranga, 6681, 90619-900 Porto Alegre, RS, Brazil. reis@pucrs.br.
[4] Department of Biology, University of Louisiana at Lafayette, Louisiana, Lafayette, LA, 70503, USA. jalbert@louisiana.edu (corresponding author).
Authors Contribution 
Victor A. Tagliacollo: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Software, Validation, Visualization, Writing-original draft, Writing-review and editing.
Fernando Cesar Paiva Dagosta: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Mário de Pinna: Conceptualization, Formal analysis, Investigation, Resources, Supervision, Writing-review and editing.
Roberto E. Reis: Conceptualization, Supervision, Validation, Writing-original draft, Writing-review and editing.
James S. Albert: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The authors declare no competing interests.
How to cite this article
Tagliacollo VA, Dagosta FCP, de Pinna M, Reis RE, Albert JS. Assessing extinction risk from geographic distribution data in Neotropical freshwater fishes. Neotrop Ichthyol. 2021; 19(3):e210079. https://doi.org/10.1590/1982-0224-2021-0079
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted July 12, 2021 by Carla Pavanelli
Accepted July 12, 2021 by Carla Pavanelli
![]() Submitted January 21, 2021
Submitted January 21, 2021
![]() Epub Sept 24, 2021
Epub Sept 24, 2021