![]() Luiza Bangoim Leal1,
Luiza Bangoim Leal1,![]() David J. Hoeinghaus2,
David J. Hoeinghaus2, ![]() Zacchaeus G. Compson2,
Zacchaeus G. Compson2,![]() Angelo A. Agostinho3,
Angelo A. Agostinho3,![]() Rodrigo Fernandes4 and
Rodrigo Fernandes4 and![]() Fernando M. Pelicice5
Fernando M. Pelicice5
PDF: EN XML: EN | Supplementary: S1 S2 S3 | Cite this article
Abstract
The introduction of non-native predators is a matter of great concern, but their impacts on ecosystem functions remain poorly understood. We investigated how changes in fish diversity following the invasion of Cichla kelberi affected ecosystem functions generated by fish populations. Fish assemblages were sampled in macrophyte patches in a Neotropical impoundment over a 5-year period, before and after the introduction of the predator. We assigned seven ecosystem functions (26 trait-states) to each fish species, and examined how these functions behaved after the invasion. We collected 577 fish belonging to 25 species. Species richness, fish biomass and main species declined significantly over periods. The biomass of ecosystem functions changed significantly over time, and most trait-states declined. Few trait-states were lost, but all functions had at least one trait-state reduced by more than 85%. A null model analysis showed that changes in functions were not driven by species identities, while species richness correlated positively with total biomass and with most functions, suggesting that the loss of taxa and biomass drove observed changes in ecosystem functions. Our study provided evidence that community disassembly associated with the invasion of C. kelberi translated to the decline of several ecosystem functions, affecting energy mobilization and transference.
Keywords: Biological invasion, Energy transference, Fish diversity, Reservoir.
A introdução de predadores não-nativos tem gerado grande preocupação, mas seus impactos sobre a geração de funções ecossistêmicas permanecem pouco investigados. O presente estudo investigou como mudanças na diversidade de peixes, associadas com a invasão de Cichla kelberi, afetaram funções ecossistêmicas geradas por populações de peixes. As assembleias de peixes foram amostradas em bancos de macrófitas em um grande reservatório Neotropical durante um período de cinco anos, antes e depois da introdução do predador. Um conjunto de funções ecossistêmicas (26 trait-states) foi atribuído a cada espécie de peixe, e examinamos como as funções se comportaram depois da invasão. Foram coletados 577 peixes pertencentes a 25 espécies. Registramos declínio significativo da riqueza de espécies, biomassa total e biomassa das principais espécies ao longo dos períodos. A biomassa das funções ecossistêmicas mudou significativamente ao longo do tempo, e a maioria dos trait-states declinou. Poucos trait-states foram perdidos, mas todas as funções tiveram pelo menos um trait-state reduzido em mais de 85%. Uma análise de modelos nulos mostrou que as mudanças nas funções não foram impulsionadas pela identidade das espécies, enquanto que a riqueza de espécies correlacionou positivamente com a biomassa total e com a maioria das funções, sugerindo que a perda de espécies e biomassa impulsionou as mudanças observadas nas funções ecossistêmicas. Nosso estudo apresenta evidências de que a desestruturação da comunidade, associada com a invasão de C. kelberi, se traduziu no declínio de várias funções ecossistêmicas, afetando a mobilização e transferência de energia.
Palavras-chave: Diversidade de peixes, Invasão biológica, Reservatório, Transferência de energia.
Introduction
Ecosystem functions are ecological, biological and physical/chemical processes that maintain ecosystem functioning (Naeem et al., 1999; Hooper et al., 2005; Naeem et al., 2012). They include, for example, energy transfer, pollination, decomposition, primary production, biomass accumulation, population control, nutrient cycling, and climate regulation, among many others. Organisms generate or affect the performance of these functions through a set of functional traits, often defined as effect traits (Lavorel, Garnier, 2002; Cadotte et al., 2011; Raffard et al., 2017), i.e., a set of phenotypic attributes linked to trophic relationships, metabolic processes, behavioral aspects, and interactions with the environment that affect ecosystem functioning. Much recent research effort has been devoted to understanding how biodiversity, and especially biodiversity loss, affects ecosystem functioning. Although results can vary depending on ecological context and spatial and temporal scales, in general, there is compelling evidence that species and functional diversity of local communities strongly affect ecosystem functioning (Cadotte et al., 2011; Cardinale et al., 2012; Hooper et al., 2012; Naeem et al., 2012; Balvanera et al., 2014; Tilman et al., 2014; Daam et al., 2019).
In aquatic and riparian ecosystems, fishes contribute to various ecosystem functions through the assimilation of energy and matter from different sources, the production, accumulation and transfer of biomass, dispersal of seeds, population control, and nutrient cycling, among others (Holmlund, Hammer, 1999; Correa et al., 2007; Hoeinghaus et al., 2009; Flecker et al., 2010; Pendleton et al., 2014). However, fish diversity is being eroded in freshwater ecosystems worldwide, with anthropogenic impacts and introduction of non-native fishes acting as major drivers of biodiversity loss. In general, invasive fishes affect ecological interactions, habitat structure and biogeochemical cycles, causing the loss of taxonomic and functional diversity in natural communities (Cucherousset, Olden, 2011). In the Neotropics, the continuous invasion of multiple species has impaired the structure and functioning of freshwater ecosystems (Vitule et al., 2009), induced biotic homogenization (Toussaint et al., 2016; Bezerra et al., 2019) and threatened fish diversity (Pelicice et al., 2017). The introduction of predatory fish is a matter of great concern, as they reduce or extirpate local populations, permanently changing the structure of fish assemblages (Eby et al., 2006; Sharpe et al., 2017; Marshall, 2018). However, the understanding of how human activities impair ecosystem functions generated by fish populations remains incipient (Holmlund, Hammer, 1999; Nash et al., 2013), including the impacts caused by non-native predators. Invasive predators are expected to induce strong effects on ecosystem functions due to strong demographic effects on prey populations.
The introduction of Cichla species, also known as peacock bass in English or tucunaré in Portuguese, provides a well-known system for assessing invasive predator effects on ecosystem functioning. Native to the Amazon basin (Kullander, Ferreira, 2006), these predators are appreciated by sport fishing and aquaculture activities (Britton, Orsi, 2012), which led to the introduction of several species to different freshwater ecosystems around the world (Ortega et al., 2015; Sastraprawira et al., 2020). Cichla are voracious predators that consume small fish and invertebrates (Bacheler et al., 2004; Fugi et al., 2008; Mendonça et al., 2018), with strong potential to disturb the invaded community, particularly in reservoirs (Zaret, Paine, 1973; Latini, Petrere Jr., 2004; Pelicice, Agostinho, 2009; Sharpe et al., 2017; Carvalho et al., 2021; Franco et al., 2021). However, their effects on ecosystem functioning remain poorly known, with the classic work by Zaret, Paine (1973) the only study reporting consequences on ecosystem functions and services. This limitation is not restricted to the invasion of Cichla, since studies on biological invasions have devoted more attention to changes in taxonomic diversity (Olden et al., 2018), possibly due to the difficulty of measuring ecosystem functions in the field.
In this context, the present study investigated how the invasion of Cichla kelberi Kullander & Ferreira, 2006 affected ecosystem functions generated by fish assemblages associated with aquatic macrophytes in a tropical reservoir (Rosana Reservoir, Paranapanema River, Brazil). Previous studies have shown drastic changes in taxonomic diversity after the introduction (Pelicice, Agostinho, 2009), and provided evidence that the invader caused these changes (Pelicice et al., 2015). No study, however, has investigated how the loss of biodiversity affected ecosystem functioning, and this study examined temporal changes in the composition and performance of ecosystem functions before and after the introduction of the predator. Adapting methodology established by functional ecology, i.e., assignment of functional traits (e.g., Violle et al., 2007; Mouillot et al., 2012), we assigned a set of ecosystem functions to each species. Specifically, we classified species into seven functions (26 trait-states) related to the transfer of energy, matter and genetic information in the system. The study investigated the hypothesis that changes in fish diversity after the invasion of C. kelberi affected the generation of ecosystem functions, causing the erosion of functions due to strong demographic effects (i.e., population decline) and the loss of some prey species. Furthermore, the approach developed for this study serves as a model for initial investigations of effects of changing biodiversity on ecosystem functioning in poorly studied regions where traditional functional trait approaches are not suitable due to data limitations.
Material and methods
Study area. Rosana Reservoir is the last of a series of reservoirs that regulates the discharge of the Paranapanema River, one of the main tributaries of the Upper Paraná River. The dam was closed in 1986 for hydroelectric production and created a shallow reservoir with 276 km2 of surface area. Aquatic macrophytes, especially Egeria densa and E. najas, readily colonized shallow areas (Pelicice et al., 2005). Currently, 65 fish species are recorded in the reservoir, of which 23 are non-native (Pelicice et al., 2018).
The first record of C. kelberi (initially identified as C. monoculus) in the basin occurred in 2001 in Capivara Reservoir, which is located approximately 150 km upstream of Rosana Reservoir (Orsi, Britton, 2014). The first record in Rosana Reservoir occurred in 2005 (Pelicice, Agostinho, 2009), but according to local fishermen, the species had been captured in the reservoir since 2004. Dispersal from Capivara Reservoir, or even new clandestine fish releases, are possible causes for the introduction of C. kelberi in Rosana Reservoir. Currently, the species has invaded almost all of the reservoirs of the Paranapanema River (Pelicice et al., 2018). Previous studies analyzed the structure of fish assemblages in Rosana Reservoir and their response to the invasion of C. kelberi (Pelicice, Agostinho, 2009; Ferrareze, Nogueira, 2015; Pelicice et al., 2015). These studies reported significant changes in abundance, richness and composition, mainly of small fishes associated with aquatic macrophytes. Pelicice et al. (2015), in particular, provided evidence that the loss of fish diversity in macrophyte patches was associated with the invasion of C. kelberi.
Fish sampling. Fish assemblages were monitored in an arm of the transition zone of Rosana Reservoir (22º34’07”S 52º33’34”W), upstream from Euclides da Cunha Paulista district, São Paulo State. The arm occupies approximately 40 ha, with shores massively colonized by E. densa. For more information about the study area and the fish fauna, see Pelicice et al. (2005). Sampling was carried out over six periods between 2003 and 2007 during warm months (> 30 ºC). The first sampling period occurred in the summer of 2003, before C. kelberi invaded the reservoir (hereafter referred to as ‘Pre’). Five other sampling periods started one year after the introduction of C. kelberi, and occurred during the summer/autumn and spring seasons between 2005 and 2007 (hereafter referred to as ‘Post-1’ through ‘Post-5’). In total, we collected 123 samples with approximately 20 samples per period (see Tab. S1).
Fish were collected in mixed patches of E. densa and E. najas, distributed along both shores of the study area. Sampling methodology consisted of a 1 m2 throw trap, with collections occurring during daylight hours (07:00–18:00). The trap was comprised by a rectangular aluminum frame (1.0 x 1.0 x 1.5 m) with 0.5 cm mesh covering all sides except for the superior and inferior ends. A boat was silently positioned above Egeria patches and used as a platform for trap deployment. Immediately following boat positioning, the trap was quickly thrown into the water and pressed to the bottom. Macrophytes were then removed from the trap with a metal fork and were washed and weighed in the field (fresh mass, g). Next, fish inside the trap were collected with a sieve (0.90 x 1.5 m) and an aluminum dip-net (49 x 49 cm), both with 0.5 cm mesh. The sieve was handled until three procedures captured no fish. The dip-net was hauled inside the trap area until 10 successive hauls resulted in no additional capture. All collected individuals were preserved in 10% formalin and taken to the laboratory, where they were identified, counted and weighted (g). Species identification was based on Graça, Pavanelli (2007) and Ota et al. (2018). Voucher specimens were deposited in the Coleção Ictiológica do Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia), Universidade Estadual de Maringá, Maringá (see Tab. S2).
Categorization of ecosystem functions. Linking biodiversity to ecosystem functioning is often aided by functional approaches that quantify phenotypic traits of species related to various aspects of performance that mediate ecosystem processes (Naeem et al., 1999; Lavorel, Garnier, 2002; Violle et al., 2007; Raffard et al., 2017). A significant limiting factor for functional traits-based approaches is the quantification or availability of relevant traits, especially for specious communities in comparatively poorly studied regions such as the Neotropics (Vitule et al., 2017). For this reason, we developed a new method for initial investigations of effects of changing biodiversity on ecosystem functioning that is particularly suitable for regions where traditional functional trait approaches are inhibited due to the lack of and/or infeasibility of generating traditional functional traits data. The method consists of directly assigning ecosystem functions performed by each species (trait-states), with the magnitude of performance of each function in a community determined by relative biomass of each species and summed across all species contributing to a particular function. Trait-state assignments are fixed, meaning that spatial, temporal and intra-specific variations were not considered (e.g., Isbell et al., 2011; Raffard et al., 2017). We used biomass as a measure of functional intensity (i.e., performance), with the assumption that greater biomass implies greater functioning. Although the relationship between biomass and ecosystem functioning is variable (e.g., terHost, Munguia, 2008), some studies show positive associations (Barnes et al., 2016; Vargas-Larreta et al., 2021).
Based on previous works, particularly Holmlund, Hammer (1999), Groot et al. (2002), Eby et al. (2006), Flecker et al. (2010), and Bauer, Hoye (2014), we defined seven ecosystem functions described by 26 trait-states related to the transfer of energy, matter and genetic information in the system (Tab. 1). Trait-states were assigned to each species based on information available in the literature. The Energy Source ecosystem function was quantified based on species’ diet, primarily following data published by Casatti et al. (2003) and Pelicice, Agostinho (2006). We considered the proportion of resources consumed grouped into classes: algae, plant, invertebrate, vertebrate, organic material/detritus. Other functions were assigned in terms of presence (1) or absence (0), and indicate whether the species directly contributes to the ecosystem function or not (see Tab. S2).
TABLE 1 | Ecosystem functions generated by fish populations considered in the present study. We defined a number of trait-states to describe each function, which were then assigned to each fish species.
| Ecosystem function | Trait-states | Description |
| Energy source | Algae Vegetal Invertebrates Vertebrates Organic matter/detritus | Function related to the components of the system (sources) from which fishes mobilize energy, matter and nutrients. |
| Habitat | Terrestrial Bottom Column Littoral | Function related to the environment where fishes mobilize energy. |
| Regional flow (migration) | Prey Herbivory Predator Genetic exchange | Function related to the transference of energy, matter or genetic information between distant habitats, performed by migratory animals. These animals may feed during migration, or become prey, establishing energy links between habitats. The function refers to the delivery (prey) and mobilization (herbivory, predation) of energy between habitats. |
| Regional flow (local) | Aquatic prey Predator Terrestrial prey | Function related to the transference of energy and matter between distant habitats, performed by local animals. Animals residing in a given location may interact with passengers (as prey or predators), establishing energy links between habitats. In this case, the function refers to the delivery (prey) or mobilization (predation) of energy between the resident and the passenger. |
| Plant Disperser | Propagule disperser | Function related to the dispersal of seeds, fruits or propagules. |
| Ecosystem Engineering | Bioturbation/Cycling Predation Herbivory | Function related to the control of physical-chemical conditions of the environment via trophic, behavioral and metabolic relationships, which affect the availability of nutrients (bottom-up control). The function is also related to the control of lower trophic levels by high consumption rates (top-down control). |
| Services | Food Sport fishing Bait Ornamental Aquaculture | Ecosystem services (provisioning and cultural) generated by fish species. |
Data analysis. We used generalized linear models (GLM) to test whether species richness, total biomass, biomass of dominant species (the nine most abundant species, including C. kelberi) and the biomass of each trait-state differed among periods (Pre against Post-1, 2, 3, 4 and 5). We selected Poisson and Gamma models with a log link function to account for overdispersion in discrete (e.g., species richness) and continuous data (all other variables), respectively. When models were significant, we performed a post-hoc linear contrast between Pre and each Post-period. In order to assess the magnitude of variation, we calculated the “loss” or “gain” in biomass for each trait-state calculated as the percent difference (decline or increase) between mean biomass values in the first (Pre) and last (Post-5) periods. Trait-states registered in Pre but absent in Post-5 were considered as lost, while trait-states absent in Pre but registered in Post-5 were considered as gained.
Ecological null modeling was used to test whether the observed ecosystem functions were different than expected based on chance given the number of species and relative biomass in the assemblages. This analysis distinguishes whether the pattern of change in estimated trait-states is due to species richness and biomass alone or whether the identities of species plays an important role in determining ecosystem functioning. This analysis controls for the ‘sampling effect’ (i.e., higher ecosystem functioning with greater diversity due to the likelihood of including a species that strongly contributes to ecosystem functioning; Huston, 1997) and species identities, which can have significant impacts on ecosystem function regardless of biomass (e.g., rare species that contribute strongly to ecosystem functioning; Pendleton et al., 2014). Species richness and the biomass distribution were held constant for each sample and null assemblages were generated by randomly assigning species identities (i.e., suite of trait values) for each species in an assemblage. We ran 1,000 permutations for each sample in each time-period, and trait-states were calculated for each random assemblage based on the species identity (i.e., trait values) assigned to each biomass in the sample. For each trait-state, the mean of observed values was compared with the null distribution, and p-values were calculated as the probability that the null was as or more extreme than the observed values (i.e., direct null hypothesis test; Veech, 2012).
We also evaluated the decay of assemblage similarity over time, considering the composition of species and trait-states, and the relationship between taxonomic diversity (species richness) and the diversity of ecosystem functioning. Composition similarity (Bray-Curtis distance, biomass-based) was compared between each Post period and the Pre period. Spearman’s non-parametric correlation was used to assess relationships between species richness and (i) total biomass, (ii) functional richness, quantified as the number of ecosystem functions performed by an assemblage, and (iii) functional intensity, quantified as biomass. For these analyses, samples were combined within periods and trait-states were combined within their respective ecosystem function in order to represent the overall composition of species and ecosystem functions in each period.
Analyses were performed using Statistica 7.1 (Statsoft, 2005), Past 2.17 (Hammer et al., 2001) and R 2.13.1 (R Development Core Team, 2011). GLMs were performed using the glm function from the stat package, and post-hoc comparisons were conducted using the emmeans package (Lenth, 2018). The inference of statistical significance followed Dushoff et al. (2019).
Results
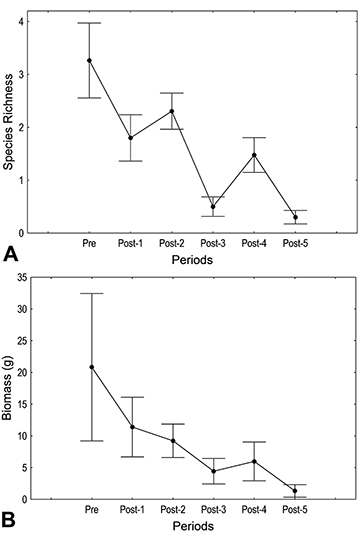
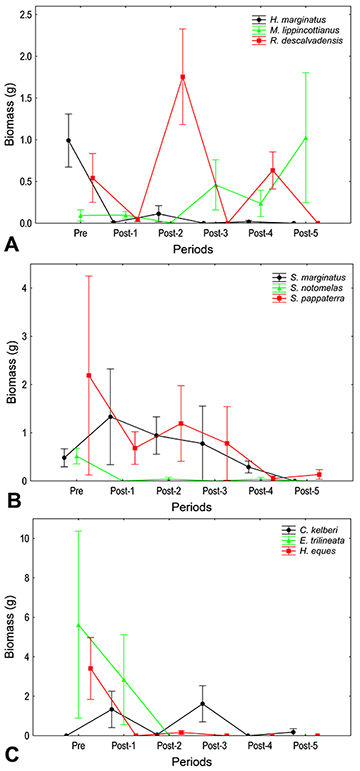
Assemblage structure and populations. In total across all sampling events, we collected 577 individuals belonging to 25 species (see Tab. S2). In the period that preceded the introduction of C. kelberi (Pre), we collected 256 individuals representing 17 species, with a total biomass of 395.68 g. After the introduction (Post-1 to 5), we captured an average of 321 individuals of 21 species, with an average biomass of 681.28 g. There was a progressive and statistically clear reduction in species richness (Fig. 1A) and total biomass (Fig. 1B) over the periods (Tab. 2). Considering the most abundant species, the majority showed statistically clear changes in biomass (Tab. 2). Some showed oscillatory trends, such as Roeboides descalvadensis, Metynnis lippincottianus, and C. kelberi, but the majority declined progressively (Fig. 2). Some species were extirpated, such as Hemigrammus marginatus, R. descalvadensis, Serrasalmus marginatus, Serrapinnus notomelas, Eigenmannia trilineata, and Hyphessobrycon eques (Fig. 2).
FIGURE 1 | Species richness (A) and total biomass (B) in fish assemblages associated with macrophyte beds in Rosana Reservoir, before (Pre) and after (Post 1 to 5) the introduction of Cichla kelberi. Mean ± standard error.
TABLE 2 | GLM results contrasting Pre and Post periods, considering the structure of fish assemblages (species richness, total biomass), main species and ecosystem functions (trait-states). A post-hoc linear test contrasted the Pre period with each Post-period (p values).
| Post-1 | Post-2 | Post-3 | Post-4 | Post-5 | DF | Deviance | p | |
| Assemblage structure | ||||||||
| Species richness | 0.052 | 0.427 | <0.001 | 0.004 | <0.001 | 5 | 84.77 | <0.001 |
| Total biomass | 0.986 | 0.998 | 0.344 | 0.807 | 0.003 | 5 | 34.72 | 0.008 |
| Main species | ||||||||
| Hemigrammus marginatus | <0.001 | 0.014 | <0.001 | <0.001 | <0.001 | 5 | 76.04 | <0.001 |
| Metynnis lippincottianus | 0.998 | 0.671 | 0.946 | 0.997 | 0.762 | 5 | 44.38 | 0.133 |
| Roeboides descalvadensis | 0.007 | 0.361 | <0.001 | 0.9839 | <0.001 | 5 | 167.65 | <0.001 |
| Serrasalmus marginatus | 1.000 | 0.993 | 0.922 | 0.993 | 0.001 | 5 | 76.04 | 0.002 |
| Serrapinnus notomelas | <0.001 | 0.002 | <0.001 | 0.0026 | <0.001 | 5 | 146.68 | <0.001 |
| Satanoperca pappaterra | 1.000 | 1.000 | 0.968 | 0.239 | 0.741 | 5 | 45.41 | 0.243 |
| Cichla kelberi | 0.009 | 0.818 | 0.002 | 1.000 | 0.428 | 5 | 141.87 | <0.001 |
| Eigenmannia trilineata | 0.992 | <0.001 | <0.001 | <0.001 | <0.001 | 5 | 248.47 | <0.001 |
| Hyphessobrycon eques | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 5 | 276.11 | <0.001 |
| Ecosystem functions Energy source | ||||||||
| Algae | 0.993 | 0.695 | 0.096 | 0.161 | 0.021 | 5 | 35.72 | 0.009 |
| Plants | 1.000 | 0.995 | 0.323 | 0.037 | 0.009 | 5 | 39.57 | 0.001 |
| Invertebrates | 0.859 | 0.992 | 0.221 | 0.848 | 0.003 | 5 | 38.32 | 0.009 |
| Vertebrates | 1.000 | 1.000 | 0.999 | 0.998 | 0.044 | 5 | 22.29 | 0.061 |
| Organic matter and detritus | 0.994 | 0.984 | 1.000 | 0.911 | 0.187 | 5 | 33.69 | 0.050 |
| Habitat | ||||||||
| Terrestrial | 0.990 | 0.992 | 0.161 | 0.965 | <0.001 | 5 | 119.22 | 0.001 |
| Bottom | 1.000 | 0.973 | 0.921 | 0.973 | 0.232 | 5 | 21.46 | 0.254 |
| Water column | 0.991 | 1.000 | 1.000 | 0.858 | 0.893 | 5 | 10.83 | 0.785 |
| Littoral | 0.986 | 0.998 | 0.358 | 0.711 | 0.003 | 5 | 35.06 | 0.009 |
| Regional flow (migration) | ||||||||
| Prey | 0.943 | 0.999 | 1.000 | 0.997 | 1.000 | 5 | 18.23 | 0.782 |
| Herbivory | 0.699 | 0.318 | 1.000 | 1.000 | 0.984 | 5 | 68.28 | 0.036 |
| Predator | 0.097 | 0.446 | 0.097 | 0.090 | 0.097 | 5 | 140.65 | <0.001 |
| Genetic exchange | 0.943 | 0.999 | 1.000 | 0.997 | 1.000 | 5 | 18.23 | 0.782 |
| Regional flow (local) | ||||||||
| Aquatic prey | 0.874 | 0.969 | 0.046 | 0.460 | 0.012 | 5 | 44.86 | 0.009 |
| Predator | 0.982 | 0.727 | 0.998 | 0.991 | 0.037 | 5 | 56.33 | 0.006 |
| Terrestrial prey | 0.872 | 1.000 | 0.010 | 0.314 | 0.001 | 5 | 88.93 | <0.001 |
| Plant Disperser | ||||||||
| Propagule disperser | 0.774 | 0.987 | 0.053 | 0.163 | 0.013 | 5 | 43.41 | 0.004 |
| Ecosystem Engineering | ||||||||
| Bioturbation/Cycling | 1.000 | 1.000 | 0.884 | 0.122 | 0.526 | 5 | 56.86 | 0.089 |
| Predator | 0.022 | 0.867 | 0.005 | 0.935 | 0.528 | 5 | 117.66 | 0.002 |
| Herbivory | 1.000 | 0.678 | 0.947 | 0.997 | 0.768 | 5 | 44.74 | 0.136 |
| Services | ||||||||
| Food | 0.942 | 0.974 | 1.000 | 0.842 | 0.752 | 5 | 22.42 | 0.110 |
| Sport fishing | 0.022 | 0.867 | 0.005 | 0.935 | 0.528 | 5 | 117.66 | 0.002 |
| Bait | 0.999 | 0.809 | 0.000 | 0.799 | 0.000 | 5 | 174.84 | <0.001 |
| Ornamental | 0.871 | 0.991 | 0.311 | 0.659 | 0.007 | 5 | 33.13 | 0.020 |
| Aquaculture | 0.873 | 0.765 | 0.607 | 1.000 | 0.979 | 5 | 28.39 | 0.427 |
FIGURE 2 | Biomass of the most abundant fish species associated with macrophyte beds in Rosana Reservoir, before (Pre) and after (Post 1 to 5) the introduction of Cichla kelberi. Mean ± standard error. A. Hemigrammus marginatus, Metynnis lippincottianus, Roeboides descalvadensis; B. Serrasalmus marginatus, Serrapinnus notomelas, Satanoperca pappaterra; C. Cichla kelberi, Eigenmannia trilineata, and Hyphessobrycon eques.
Ecosystem functioning. We recorded statistically clear changes in the intensity of ecosystem functioning over time (Tab. 2). With the exception of the functions Regional Flow (migration) and Ecosystem Engineering, most trait-states lost biomass over time (Fig. 3A-G). For the ecosystem functions Energy Source, Habitat, Regional Flow (local), Plant Disperser and Services, most or all trait-states declined (Tab. 2).
FIGURE 3 | Ecosystem functions generated by fish populations associated with macrophyte beds in Rosana Reservoir, before (Pre) and after (Post 1 to 5) the introduction of Cichla kelberi. Mean ± standard error. Ecosystem functions: A. Energy Source; B. Habitat; C. Regional Flow (migration); D. Regional Flow (local); E. Plant Disperser; F. Engineering; G. Services.
No ecosystem function was totally lost, but all functions had at least one trait-state reduced by more than 85% of the original intensity (Tab. 3). All trait-states of the Energy Source function declined by more than 90%. The Regional Flow (migration) function lost the Predator trait-state, but Prey and Genetic Exchange increased by more than 200%, while Herbivory increased by more than 400%. The Ecosystem Engineering function declined by more than 94% in the Bioturbation/Cycling trait-state, but Herbivory experienced a 900% increase. The Services function lost the Bait trait-state, but Aquaculture increased by more than 500% (Tab. 3).
Results from null modeling indicate that in almost all cases (trait-states in different periods) the observed intensity of ecosystem functioning in a time period was not different from expected based on the randomly generated distribution (see Fig. S3). In general, this indicates that the observed changes in the intensity of ecosystem functions were driven by the loss of taxa and biomass, not changes in species identities per se.
TABLE 3 | Increasing (positive values) and decreasing (negative values) trends in the intensity of ecosystem functions (trait-states) generated by fish populations, calculated from the variation in fish biomass between the first (Pre) and last (Post-5) periods. “+” = function added, absent in Pre but present in Post; “-” = function lost, present in Pre but absent in Post-5.
| Ecosystem functions | % |
| Energy source | |
| Algae | -91.5 |
| Plants | -94.2 |
| Invertebrates | -94.3 |
| Vertebrates | -90.9 |
| Organic matter and detritus | -90.7 |
| Habitat | |
| Terrestrial | – |
| Benthic | -91 |
| Water column | 0.6 |
| Littoral | -93.6 |
| Regional Flow (migration) | |
| Prey | 209 |
| Herbivory | 409.8 |
| Predator | – |
| Genetic exchange | 226.2 |
| Regional Flow (local) | |
| Aquatic prey | -94.4 |
| Predator | -85.1 |
| Terrestrial prey | -98.3 |
| Plant Disperser | |
| Propagule disperser | -91.9 |
| Ecosystem Engineering | |
| Bioturbation/Cycling | -94.1 |
| Predator | + |
| Herbivory | 936.5 |
| Services | |
| Food | -56.6 |
| Sport fishing | + |
| Bait | – |
| Ornamental | -93.3 |
| Aquaculture | 504.3 |
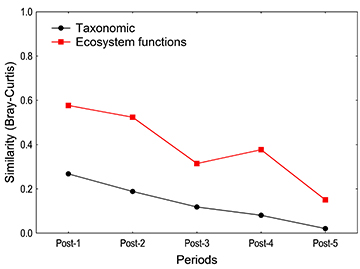
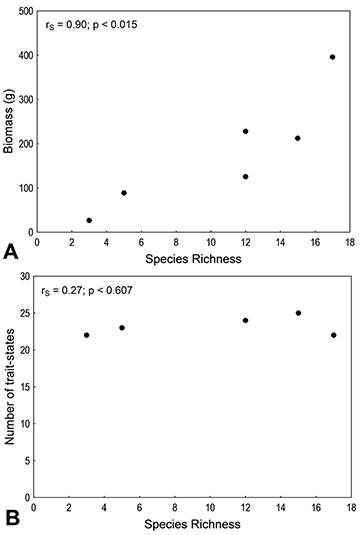
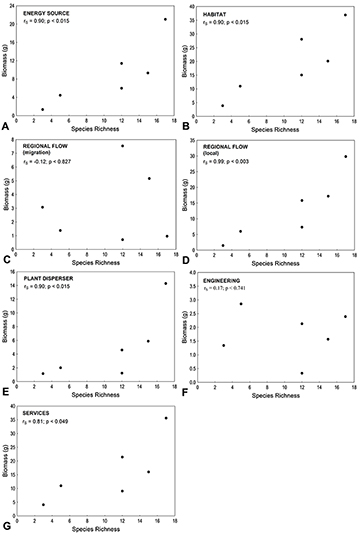
Taxonomic and functional similarity (based on biomass) progressively declined over time (Fig. 4). Taxonomic similarity showed lower values, and approached zero in the last period. Species richness correlated positively with total biomass (Fig. 5A), but not with the richness of ecosystem functions (Fig. 5B). Species richness was also positively correlated with the intensity (biomass) of the Energy Source, Habitat, Regional Flow (Local), Plant Disperser, and Services functions (Fig. 6A-G).
FIGURE 4 | Temporal decay of taxonomic and functional similarity (Bray Curtis, biomass-based) during the study period, calculated as the composition similarity of the Pre period against each Post period.
FIGURE 5 | Correlations between species richness and (A) total biomass and (B) number of ecosystem functions performed by fish populations. The significance of correlations was tested through Spearman’s non-parametric correlation.
FIGURE 6 | Correlations between species richness and the intensity of ecosystem functions performed by fish populations. The significance of correlations was tested through Spearman’s non-parametric correlation. Ecosystem functions: A. Energy Source; B. Habitat; C. Regional Flow (migration); D. Regional Flow (local); E. Plant Disperser; F. Engineering; G. Services.
Discussion
Non-native fishes have the potential to cause significant disturbances to the invaded ecosystem, especially in modified environments (Rahel, 2007; Johnson et al., 2008). Studies have reported a myriad of consequences that emerge from fish invasions (e.g., Leprieur et al., 2008; Olden et al., 2018; Toussaint et al., 2018; Bezerra et al., 2019), but the effects on ecosystem functions remain poorly investigated. This topic has high theoretical and practical relevance (Hooper et al., 2012; Naeem et al., 2012; Balvanera et al., 2014), because biodiversity generates essential ecosystem functions and non-native species have the potential to erode biodiversity. In this sense, our work investigated how changes in fish diversity following the invasion of Cichla kelberi affected ecosystem functions potentially generated by fish species associated with macrophyte beds in Rosana Reservoir. Considering a set of ecosystem functions (7 functions based on 26 trait-states), we were able to demonstrate that ecosystem functions were significantly altered after the introduction, supporting the hypothesis that species losses and especially strong demographic effects (i.e., population declines) translated to the erosion of ecosystem functioning. Our findings also indicate that the loss of ecosystem functioning was not associated with the loss of specific taxa, but with a severe process of community disassembly where the invasion coincided with a sharp decline in species richness and fish abundance (Pelicice, Agostinho, 2009). Thus, our results demonstrate that the invasion of C. kelberi was followed by the loss of fish diversity and the functional simplification of the ecosystem, affecting aspects related to the transfer of energy, matter and genetic information.
This study covered processes related to the origin, assimilation and flow of matter/energy between components and compartments of the ecosystem. The Energy Source function referred to the connection between fish and the energy source (e.g., algae, invertebrates), whereas the Habitat function referred to the environment where the energy is obtained (e.g., water column, benthos). Both functions were drastically reduced after the invasion of C. kelberi, which must affect the transfer of matter and energy through the system. The Energy Source function was the most affected, with a decline of more than 90% for each trait-state. It is important to note that fish species in macrophyte beds are small-sized and feed predominantly on invertebrates in shallow littoral environments (Casatti et al., 2003; Pelicice, Agostinho, 2006). This link is important, because invertebrates assimilate energy from sources that may be inaccessible for fishes (e.g., macrophyte, debris, periphyton, algae; Covich et al., 1999; Benetti et al., 2014). The loss of this link likely affects the structure of food webs in littoral habitats, with negative effects on higher trophic levels, such as larger fish species and terrestrial predators. Concerning the Habitat function, the Littoral state declined by more than 90%, whereas the Terrestrial state was completely lost. These trait-states were shared by many small-sized species, specifically Astyanax lacustris, Crenicichla britskii, Cichlasoma paranaense, Eigenmannia trilineata, Gymnotus carapo, Hemigrammus marginatus, and Oligossarcus pintoi. The decline of the littoral link has high potential to affect food webs in the reservoir, since littoral areas play a key role for biodiversity in Neotropical reservoirs (Agostinho et al., 2016). The complete loss of the terrestrial link, in turn, affects the connection between aquatic and terrestrial environments, impairing the flow of matter and energy between environments (e.g., consumption of terrestrial invertebrates, predation of fish by birds).
Our study also investigated functions related to the transference of matter/energy between distant sites. The Regional Flow (migration) function considered fish that could migrate to other locations, whereas Regional Flow (local) considered local fish species that interact with migratory species. Studies indicate that migrants have strong effects on ecosystem functioning, as they link distant environments through energy flows (Flecker et al., 2010; Bauer, Hoye, 2014; Uno, Power, 2015). However, Regional Flow (via migration) was not affected by the invasion, probably because fish species associated with macrophyte beds are small and sedentary (Casatti et al., 2003; Pelicice et al., 2005), indicating that this function was not generated by the pre-invasion fauna. Regional Flow (via local), on the other hand, was significantly affected, since small fish serve as prey for young and adult migratory species (Bozza, Hahn, 2010). Each trait-state reduced by more than 85%, implying a reduction in the supply of prey for aquatic and terrestrial migrants. Although we have not recorded migratory fish species in the macrophyte beds, there are eight migratory fish species found in Rosana Reservoir (Pelicice et al., 2018). Therefore, the loss of local prey may negatively affect populations of migratory fishes, if they visit macrophyte beds during their migratory movements or if they use these habitats as nursery grounds. Furthermore, migratory prey not found in macrophyte beds may subsidize C. kelberi populations (e.g., Hoeinghaus et al., 2006) and thus contribute to increased effects of C. kelberi on ecosystem functions performed by fish assemblages in macrophyte beds.
The dispersal of seeds, fruits and animal propagules is essential for the maintenance of communities, and many fish species perform this function in Neotropical environments (Correa et al., 2007). Many fish species that occurred in macrophyte beds in Rosana Reservoir consume small seeds, and may have a potential role as seed dispersers. Following invasion by C. kelberi, the Plant Disperser function declined by more than 90%. The decline of this function, therefore, points to potential negative effects on the dispersal and recruitment of aquatic and terrestrial plants (Silveira, Weiss, 2014; Costa-Pereira, Galetti, 2015). The invasion also affected the Ecosystem Engineering function, specifically functions related to population and biomass control (top-down processes), and bioturbation and nutrient cycling (bottom-up processes). The Bioturbation/Cycling trait-state reduced by 94% after the invasion, primarily associated with the decline and extirpation of the benthic consumer Satanoperca pappaterra from macrophyte beds (Hahn, Cunha, 2005; Cassemiro et al., 2008). The loss of this function can decrease the mobilization of nutrients stored in the sediment, with potential negative effects on primary and secondary production (Biles et al., 2002; Adámek, Marsálek, 2013). The Predation trait-state did not differ between periods, but it appeared in the system after invasion as it is a function performed almost exclusively by C. kelberi. As previous noted, C. kelberi is a voracious predator with potential to induce strong top-down control. This function oscillated over time and followed variations in the abundance of the invader (Pelicice et al., 2015). The Herbivory trait-state experienced a substantial increase (> 900%), associated with the increase in biomass of M. lippincottianus, which is a non-native herbivore that spread in macrophyte beds after the invasion of C. kelberi.
The study also recorded changes in potential ecosystem services (Services function) generated by fish populations. The main trait-states affected were those related to the provision of ornamental and bait fish resources, because fishes found in macrophyte beds are small-sized. However, the provision of other services showed increasing trends after the invasion. The Aquaculture trait-state increased by more than 500%, and was related to increased biomasses of four species: A. lacustris, C. kelberi, M. lippincottianus and O. niloticus (the last three are non-native). The Sport Fishing trait-state emerged only after the introduction of C. kelberi, and represents the provisioning of a new service (sport fishing) much appreciated by some sectors of society (e.g., tourism, sport-fishing industry, anglers). However, it should be noted that this trait-state was represented by low biomass throughout the study period, in contrast to the effects of the invasive piscivore on the loss of fishing resources with potential to support consumption, ornamental and bait services. Britton, Orsi (2012) contrasted economic benefits and environmental costs associated with the use of non-native species in sport fishing and aquaculture activities, and concluded that some species provide economic services for some sectors, but they usually do that at the expense of environmental, economic and social sustainability in the long-term. Another aspect is that populations of non-native Cichla tend to oscillate and support unproductive fisheries in reservoirs, since these predators reduce prey populations to very small sizes, which constrain the population growth of the invader (Fugi et al., 2008). These tradeoffs must be considered in the assessment of socioeconomic costs and benefits derived from the introduction of Cichla in non-Amazonian reservoirs. Illegal introductions continue to occur and different species are now widespread in many basins (Pelicice et al., 2018; Sastraprawira et al., 2020); recently, legislation amendments have been proposed to protect non-native fish stocks and foster fishing activities (Magalhães et al., 2018).
Several results from prior studies of the relationship between biodiversity and ecosystem functioning indicate that changes in taxonomic diversity are associated with changes in the performance of ecosystem functions (Cadotte et al., 2011; Balvanera et al., 2014; Daam et al., 2019). Modern ecology has devoted substantial attention to understand how biodiversity affects ecosystem functioning, principally in light of the strong effects of human activities on the structure of most natural communities (Cardinale et al., 2012; Tilman et al., 2014). In our study, population declines and loss of species following invasion of C. kelberi triggered a profound process of community disassembly in littoral macrophyte beds (Pelicice, Agostinho, 2009). In fact, results from the null model analysis indicated that the decline of ecosystem functions was not associated with the loss of particular species, but rather is likely a consequence of wholesale changes in taxonomic diversity, biomass and decay of community similarity due to the voracious nature of C. kelberi. Although we observed the erosion of ecosystem functions (i.e., complete loss or significant reductions in the intensity of many trait-states), species richness was not correlated with the richness of ecosystem functions. In fact, none of the seven functions were totally lost, and even in situations of low species richness (i.e., three species), most functions remained in the area – indicating functional redundancy among fish species in these assemblages. Although the assessment of functional redundancy is highly dependent on the number of traits (Petchey, Gaston, 2006), our study considered a reasonable number of trait-states (n = 26), suggesting that many species share functions (often multiple functions). Redundancy is a common phenomenon in communities, as species overlap in various niche dimensions (Hooper et al., 2005). When many species perform the same function, they may replace each other in cases of species loss, maintaining the provisioning of ecosystem functioning (Chapin III et al., 2000). However, although no ecosystem function was lost, results indicated that most trait-states changed significantly (i.e., increased, declined or disappeared) after the invasion, pointing to a strong functional simplification of the studied system. Moreover, the erosion of taxonomic diversity implies a reduction in functional resilience because the loss of species reduces the redundancy in functions, making the system vulnerable to disorganization and novel disturbances (Tilman, 1997; Chapin III et al., 2000).
The loss of ecosystem functions threatens the maintenance of ecosystems, as functions are responsible for the mobilization, assimilation, and transference of energy (Mouillot et al., 2012; Naeem et al., 2012). Therefore, studies on biological invasions should pay more attention to the effects invaders have on ecosystem functions generated by the resident biota. This is difficult, especially where functional traits data are not available or are particularly challenging to quantify (e.g., Vitule et al., 2017). In this study, we used a novel method, derived from the traditional approach of functional ecology (e.g., Mouillot et al., 2012; Toussaint et al., 2016), to indirectly assess the presence and performance of ecosystem functions by assigning trait-states directly to species. Admittedly, indirect assessments have natural limitations, as the function and performance of species are not measured in realistic scenarios. However, indirect assessments, such as the one we present here, can provide an initial understanding of potential ecosystem consequences of biodiversity loss or for development of hypotheses for traditional traits-based studies. Such studies can be particularly useful to initiate biodiversity-ecosystem functioning research in poorly studied and species-rich regions like the Neotropics. Studies using indirect assessments of ecosystem functioning should be followed by direct assessments of ecosystem functioning, measuring empirically how non-native species affect the generation of these functions in invaded ecosystems (e.g., Tilman et al., 1997; Balvanera et al., 2014; Allen et al., 2016).
Acknowledgments
We thank the Programa de Pós-Graduação em Biodiversidade, Ecologia e Conservação (previous Ecologia de Ecótonos) from the Universidade Federal do Tocantins (UFT), and the Nupélia (UEM) for providing infrastructure and support. Coordenacão de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) provided a scholarship for LBL and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) provided research grants for AAA (Process number 307538/2016-8) and FMP (Process number 312256/2020-5).
References
Adámek Z, Marsálek M. Bioturbation of sediments by benthic macroinvertebrates and fish and its implication for pond ecosystems: A review. Aquac Int. 2013; 21:1–17. http://dx.doi.org/10.1007/s10499-012-9527-3
Agostinho AA, Gomes LC, Santos NCL, Ortega JCG, Pelicice FM. Fish assemblages in Neotropical reservoirs: Colonization patterns, impacts and management. Fish Res. 2016; 173(1):26–36. http://dx.doi.org/10.1016/j.fishres.2015.04.006
Allen DC, Cardinale BJ, Wynn-Thompson T. Plant biodiversity effects in reducing fluvial erosion are limited to low species richness. Ecology. 2016; 97(1):17–24. http://dx.doi.org/10.1890/15-0800.1
Bacheler MN, Neal JW, Noble RL. Diet overlap between native bigmouth sleepers (Gobiomorus dormitor) and Introduced predatory fishes in a Puerto Rico reservoir. Ecol Freshw Fish. 2004; 13(2):111–18. http://dx.doi.org/10.1111/j.1600-0633.2004.00040.x
Balvanera P, Siddique I, Dee L, Paquette A, Isbell F, Gonzalez A, Byrnes J, O’Connor MI, Hungate BA, Griffin JN. Linking biodiversity and ecosystem services: current uncertainties and the necessary next steps. BioScience. 2014; 64(1):49–57. http://dx.doi.org/10.1093/biosci/bit003
Barnes AD, Weigelt P, Jochum M, Ott D, Hodapp D, Haneda NF, Brose U. Species richness and biomass explain spatial turnover in ecosystem functioning across tropical and temperate ecosystems. Phil Trans R Soc B. 2016; 371(1694):20150279. http://dx.doi.org/10.1098/rstb.2015.0279
Bauer S, Hoye BJ. Migratory animals couple biodiversity and ecosystem functioning worldwide. Science. 2014; 344(6179):1242552. http://dx.doi.org/10.1126/science.1242552
Benetti CJ, Pérez-Bilbao A, Garrido J. The determination of food sources for invertebrates in four ponds in NW Spain using stable isotope analysis. Limnetica. 2014; 33(1):89–106. http://dx.doi.org/10.23818/limn.33.08
Bezerra LAV, Ribeiro VM, Freitas MO, Kaufman L, Padial AA, Vitule JRS. Benthification, biotic homogenization behind the trophic downgrading in altered ecosystems. Ecosphere. 2019; 10(6):e02757. http://dx.doi.org/10.1002/ecs2.2757
Biles CL, Paterson DM, Ford RB, Solan M, Raffaelli DG. Bioturbation, ecosystem functioning and community structure. Hydrol Earth Syst Sc. 2002; 6(6):999–1005. http://dx.doi.org/10.5194/hess-6-999-2002
Bozza AN, Hahn NS. Uso de recursos alimentares por peixes imaturos e adultos de espécies piscívoras em uma planície de inundação neotropical. Biota Neotrop. 2010; 10(3):217–26. http://dx.doi.org/10.1590/S1676-06032010000300025
Britton JR, Orsi ML. Non-native fish in aquaculture and sport fishing in Brazil: economic benefits versus risks to fish diversity in the upper River Paraná Basin. Rev Fish Biol Fisher. 2012; 22:555–65. http://dx.doi.org/10.1007/s11160-012-9254-x
Cadotte MW, Carscadden K, Mirotchnik N. Beyond species: functional diversity and the maintenance of ecological processes and services. J Appl Ecol. 2011; 48(5):1079–87. http://dx.doi.org/10.1111/j.1365-2664.2011.02048.x
Cardinale BJ, Duffy JE, Gonzalez A, Hooper DU, Perrings C, Venail P, Narwani A, Mace GM, Tilman D, Wardle DA, Kinzig AP, Daily GC, Loreau M, Grace JB, Larigauderie A, Srivastava DS, Naeem S. Biodiversity loss and its impact on humanity. Nature. 2012; 486:59–67. http://dx.doi.org/10.1038/nature11148
Carvalho TL, Ferreira EA, Pelicice FM, Fernandes R. Comparative functional responses predict the predatory impact of the highly invasive fish Cichla kelberi. Hydrobiologia. 2021; 848:2203–11. http://dx.doi.org/10.1007/s10750-020-04440-6
Casatti L, Mendes HF, Ferrerira KM. Aquatic macrophytes as feeding site for small fishes in the Rosana reservoir, Paranapanema river, southeastern Brazil. Braz J Biol. 2003; 63(2):213–22. http://dx.doi.org/10.1590/S1519-69842003000200006
Cassemiro FAS, Rangel TFLVB, Pelicice FM, Hahn NS. Allometric and ontogenetic patterns related to feeding of a Neotropical fish, Satanoperca pappaterra (Perciformes, Cichlidae). Ecol Freshw Fish. 2008; 17(1):155–64. http://dx.doi.org/10.1111/j.1600-0633.2007.00270.x
Chapin III FS, Zavaleta ES, Eviner V, Naylor RL, Vitousek PM, Reynolds HL, Hooper DU, Lavorel S, Sala OE, Hobbie SE, Mack MC, Diaz S. Consequences of changing biodiversity. Nature. 2000; 405:234–42. https://doi.org/10.1038/35012241
Correa SB, Winemiller KO, López-Hernández H, Galetti M. Evolutionary perspectives on seed consumption and dispersal by fishes. Bioscience. 2007; 57(9):748–56. http://dx.doi.org/10.1641/B570907
Costa-Pereira R, Galetti M. Frugivore downsizing and the collapse of seed dispersal by fish. Biol Conserv. 2015; 191:809–11. http://dx.doi.org/10.1016/j.biocon.2015.07.011
Covich AP, Palmer MA, Crowl TA. The role of benthic invertebrate species in freshwater ecosystems. Bioscience. 1999; 49(2):119–27.
Cucherousset J, Olden JD. Ecological impacts of non-native freshwater fishes. Fisheries. 2011; 36(5):215–30. http://dx.doi.org/10.1080/03632415.2011.574578
Daam AA, Teixeira H, Lillebo AI, Nogueira AJA. Establishing causal links between aquatic biodiversity and ecosystem functioning: Status and research needs. Sci Total Environ. 2019; 656:1145–56. http://dx.doi.org/10.1016/j.scitotenv.2018.11.413
Dushoff J, Kain MP, Bolker BM. I can see clearly now: Reinterpreting statistical significance. Methods Ecol Evol. 2019; 10(6):756–59. http://dx.doi.org/10.1111/2041-210X.13159
Eby LA, Roach WJ, Crowder LB, Stanford JA. Effects of stocking-up freshwater food webs. Trends Ecol Evol. 2006; 21(10):576–84. http://dx.doi.org/10.1016/j.tree.2006.06.016
Ferrareze M, Nogueira MG. Impact assessment of the introduction of Cichla kelberi in a large Neotropical reservoir and its lateral lagoons (Upper Paraná River Basin, Brazil). Braz J Biol. 2015; 75(4):1018–26. http://dx.doi.org/10.1590/1519-6984.05814
Flecker AS, McIntyre PB, Moore JW, Anderson JT, Taylor BW, Hall RO. Migratory fishes as material and process subsidies in riverine ecosystems. In: Gido KB, Jackson DA, editors. Community ecology of stream fishes: concepts, approaches, and techniques. Bethesda: American Fisheries Society, Symposium; 2010. p.559–92.
Franco ACS, García-Berthou E, Santos LN. Ecological impacts of an invasive top predator fish across South America. Sci Total Environ. 2021; 761:143296. https://doi.org/10.1016/j.scitotenv.2020.143296
Fugi R, Luz-Agostinho KDG, Agostinho AA. Trophic interaction between an introduced (peacock bass) and a native (dogfish) piscivorous fish in a Neotropical impounded river. Hydrobiologia. 2008; 607:143. http://dx.doi.org/10.1007/s10750-008-9384-2
Graça WJ, Pavanelli CS. Peixes de planície de inundação do alto rio Paraná e áreas adjacentes. Maringá: Eduem; 2007.
Groot RS, Wilson MA, Boumans RMJ. A typology for the classification, description and valuation of ecosystem functions, goods and services. Ecol Economics. 2002; 41(3):393–408. https://doi.org/10.1016/S0921-8009(02)00089-7
Hahn NS, Cunha F. Feeding and trophic ecomorphology of Satanoperca pappaterra (Pisces, Cichlidae) in the Manso Reservoir, Mato Grosso State, Brazil. Braz Arch Biol Technol. 2005; 48(6):1007–12. http://dx.doi.org/10.1590/S1516-89132005000800017
Hammer Q, Harper DAT, Ryan PD. Past: Paleontologia Statistics software package for education and data analysis. Paleontol Electron. 2001; 4(1):1–09.
Hoeinghaus DJ, Agostinho AA, Gomes LC, Pelicice FM, Okada EK, Latini JD, Kashiwaki EAL, Winemiller KO. Effects of river impoundment on ecosystem services of large tropical rivers: embodied energy and market value of artisanal fisheries. Conserv Biol. 2009; 23(5):1222–31. http://dx.doi.org/10.1111/j.1523-1739.2009.01248.x
Hoeinghaus DJ, Winemiller KO, Layman CA, Arrington DA, Jepsen DB. Effects of seasonality and migratory prey on body condition of Cichla species in a tropical floodplain river. Ecol Freshw Fish. 2006; 15(4):398–407. https://doi.org/10.1111/j.1600-0633.2006.00152.x
Holmlund CM, Hammer M. Ecosystem services generated by fish populations. Ecol Economics. 1999; 29:253–68.
Hooper DU, Adair EC, Cardinale BJ, Byrnes JEK, Hungate BA, Matulich KL, Gonzalez A, Duffy JE, Gamfeldt L, O’Connor MI. A global synthesis reveals biodiversity loss as a major driver of ecosystem change. Nature. 2012; 486:105–08. http://dx.doi.org/10.1038/nature11118
Hooper DU, Chapin III FS, Ewel JJ, Hector A, Inchausti P, Lavorel S, Lawton JH, Lodge DM, Loreau M, Naeem S, Schmid B, Setala H, Symstad AJ, Vandermeer J, Wardle DA. Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monog. 2005; 75(1):3–35. https://doi.org/10.1890/04-0922
Huston MA. Hidden treatments in ecological experiments: re-evaluating the ecosystem function of biodiversity. Oecologia. 1997; 110:449–60. https://doi.org/10.1007/s004420050180
Isbell F, Calcagno V, Hector A, Connolly J, Harpole WS, Reich PB, Lorenzen MS, Schmid B, Tilman D, Ruijvens J, Weigelt A, Wilsey BJ, Zavaleta ES, Loreau M. High plant diversity is needed to maintain ecosystem services. Nature. 2011; 477:199–202. http://dx.doi.org/10.1038/nature10282
Johnson PTJ, Olden JD, Vander Zanden MJ. Dam invaders: impoundments facilitate biological invasions into freshwaters. Front Ecol Environ. 2008; 6(7):357–63. http://dx.doi.org/10.1890/070156
Kullander SO, Ferreira EJG. A review of the South American cichlid genus Cichla, with descriptions of nine new species (Teleostei: Cichlidae). Ichthyol Explor Fres. 2006; 17(4):289–398.
Latini AO, Petrere Jr M. Reduction of a native fish fauna by alien species: an example from Brazilian freshwater tropical lakes. Fisheries Manag Ecol. 2004; 11(2):71–79. http://dx.doi.org/10.1046/j.1365-2400.2003.00372.x
Lavorel S, Garnier E. Predicting changes in community composition and ecosystem functioning from plant traits: revisiting the Holy Grail. Func Ecol. 2002; 16(5):545–56. http://dx.doi.org/10.1046/j.1365-2435.2002.00664.x
Lenth R. emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.1.3 [Internet]. R Foundation for Statistical Computing; 2018. Available from: https://CRAN.R-project.org/package=emmeans
Leprieur F, Beauchard O, Blanchet S, Oberdorff T, Brosse S. Fish invasions in the world’s river systems: when natural processes are blurred by human activities. PLOS Biol. 2008; 6(12):e322. http://dx.doi.org/10.1371/journal.pbio.0060028
Magalhães ALB, Pelicice FMP, Lima-Junior DP. Riscos ambientais e socioeconômicos do Projeto de Lei que visa a proteção de espécies invasoras (tucunaré azul e tucunaré amarelo) no Estado do Paraná. Bol Soc Bras Ictiol. 2018; Nota Técnica:1–08.
Marshall BE. Guilty as charged: Nile perch was the cause of the haplochromine decline in Lake Victoria. Can J Fish Aquat Sci. 2018; 75(9):1542–59. http://dx.doi.org/10.1139/cjfas-2017-0056
Mendonça HS, Santos ACA, Martins MM, Araújo FG. Size-related and seasonal changes in the diet of the non-native Cichla kelberi Kullander & Ferreira, 2006 in a lowland reservoir in the southeastern Brazil. Biota Neotrop. 2018; 18(3):e20170493. http://dx.doi.org/10.1590/10.1590/1676-0611-BN-2017-0493
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR. A functional approach reveals community responses to disturbances. Trends Ecol Evol. 2012; 28(3):167–77. http://dx.doi.org/10.1016/j.tree.2012.10.004
Naeem S, Chapin III FS, Costanza R, Ehrlich PR, Golley FB, Hooper DU, Lawton JH, O’neill RV, Mooney HA, Sala OE, Symstad AJ, Tilman D. Biodiversity and ecosystem functioning: Maintaining natural life support processes. Issues Ecology. 1999; 4:1–11. Available from: https://www.esa.org/wp-content/uploads/2013/03/issue4.pdf
Naeem S, Duffy E, Zavaleta E. The functions of biological diversity in an age of extinction. Science. 2012; 336:1401–06. http://dx.doi.org/10.1126/science.1215855
Nash KL, Graham NAJ, Bellwood DR. Fish foraging patterns, vulnerability to fishing, and implications for the management of ecosystem function across scales. Ecol Appl. 2013; 23(7):1632–44. http://dx.doi.org/10.1890/12-2031.1
Olden JD, Comte L, Giam X. The Homogocene: a research prospectus for the study of biotic homogenization. NeoBiota. 2018; 37:23–36. http://dx.doi.org/10.3897/neobiota.37.22552
Orsi ML, Britton JR. Long-term changes in the fish assemblage of a Neotropical hydroelectric reservoir. J Fish Biol. 2014; 84(6):1964–70. http://dx.doi.org/10.1111/jfb.12392
Ortega JCG, Julio Jr HF, Gomes LC, Agostinho AA. Fish farming as the main driver of fish introductions in Neotropical reservoirs. Hydrobiologia. 2015; 746(1):147–58. http://dx.doi.org/10.1007/s10750-014-2025-z
Ota RR, Deprá GC, Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: revised, annotated and updated. Neotrop Ichthyol. 2018; 16(2):e170094. https://dx.doi.org/10.1590/1982-0224-20170094
Pelicice FM, Agostinho AA. Feeding ecology of fishes associated with Egeria spp. patches in a tropical reservoir, Brazil. Ecol Freshw Fish. 2006; 15(1):10–19. http://dx.doi.org/10.1111/j.1600-0633.2005.00121.x
Pelicice FM, Agostinho AA. Fish fauna destruction after the introduction of a non – native predator (Cichla kelberi) in a Neotropical reservoir. Biol Invasions. 2009; 11:1789–801. http://dx.doi.org/10.1007/s10530-008-9358-3
Pelicice FM, Agostinho AA, Thomaz SM. Fish assemblages associated with Egeria in a tropical reservoir: investigating the effects of plant biomass and diel period. Acta Oecol. 2005; 27(3):9–16. http://dx.doi.org/10.1016/j.actao.2004.08.004
Pelicice FM, Azevedo-Santos VM, Esguícero LH, Agostinho AA, Arcifa MS. Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotrop Ichthyol. 2018; 16(2):e170150. http://dx.doi.org/10.1590/1982-0224-20170150
Pelicice FM, Azevedo-Santos VM, Vitule JRS, Orsi ML, Lima Junior DP, Magalhães ALB, Pompeu PS, Petrere Jr M, Agostinho AA. Neotropical freshwater fishes imperiled by unsustainable policies. Fish Fish. 2017; 18(6):1119–33. http://dx.doi.org/10.1111/faf.12228
Pelicice FM, Latini JD, Agostinho AA. Fish fauna disassembly after introduction of a voracious predator: main drivers and the role of the invader’s demography. Hydrobiologia. 2015; 746(1):271–83. http://dx.doi.org/10.1007/s10750-014-1911-8
Pendleton RM, Hoeinghaus DJ, Gomes LC, Agostinho AA. Loss of rare fish species from tropical floodplain food webs affects community structure and ecosystem multifunctionality in a mesocosm experiment. PLoS One. 2014; 9(1):e84568. http://dx.doi.org/10.1371/journal.pone.0084568
Petchey OL, Gaston KJ. Functional diversity: back to basics and looking forward. Ecol Lett. 2006; 9(6):741–58. http://dx.doi.org/10.1111/j.1461-0248.2006.00924.x
R Development Core Team. R: A Language and Environment for Statistical Computing [Internet]. R Foundation for Statistical Computing; 2011. Available from: www.r-project.org
Raffard A, Lecerf A, Cote J, Buoro M, Lassus R, Cucherousset J. The functional syndrome: linking individual trait variability to ecosystem functioning. Proc R Soc B. 2017; 284(1868):20171893. http://dx.doi.org/10.1098/rspb.2017.1893
Rahel FJ. Biogeographic barriers, connectivity and homogenization of freshwater faunas: it’s a small world after all. Freshwater Biol. 2007; 52(4):696–710. http://dx.doi.org/10.1111/j.1365-2427.2006.01708.x
Sastraprawira SM, Razak IHA, Shahimi S, Pati S, Edinur HA, John AB, Ahmad A, Kumaran JV, Martin MB, Chong JL, Chowdhury AJK, Nelson BR. A review on introduced Cichla spp. and emerging concerns. Heliyon. 2020; 6(11):e05370. https://doi.org/10.1016/j.heliyon.2020.e05370
Sharpe DMT, De León LF, González R, Torchin ME. Tropical fish community does not recover 45 years after predator introduction. Ecol. 2017; 98(2):412–24. http://dx.doi.org/10.1002/ecy.1648
Silveira RML, Weiss B. Evidence for herbaceous seed dispersal by small-bodied fishes in a Pantanal seasonal wetland. Braz J Biol. 2014; 74(3):588–96. http://dx.doi.org/10.1590/bjb.2014.0089
Statsoft. Statistica (data analysis software system), version 7.1 [Internet]. Tulsa: StatSoft Inc; 2005. Available from: http://www.statsoft.com
Vargas-Larreta B, López-Martínez JO, González EJ, Corral-Rivas JJ, Hernandez FJ. Assessing above-ground biomass-functional diversity relationships in temperate forests in northern Mexico. For Ecosyst. 2021; 8(8):1–14. https://doi.org/10.1186/s40663-021-00282-3
Veech JA. Significance testing in ecological null models. Theor Ecol. 2012; 5:611–16. http://dx.doi.org/10.1007/s12080-012-0159-z
Violle C, Navas ML, Vile D, Kazakou E, Fortunel C, Hummel I, Garnier E. Let the concept of trait be functional! Oikos. 2007; 116(5):882–92. http://dx.doi.org/10.1111/j.2007.0030-1299.15559.x
Vitule JRS, Agostinho AA, Azevedo-Santos VM, Daga VS, Darwall WRT, Fitzgerald DB, Frehse FA, Hoeinghaus DJ, Lima Jr. DP, Magalhães ALB, Orsi ML, Padial AA, Pelicice FM, Petrere Jr. M, Pompeu PS, Winemiller KO. We need better understanding about functional diversity and vulnerability of tropical freshwater fishes. Biodivers Conserv. 2017; 26:757–62. https://doi.org/10.1007/s10531-016-1258-8
Vitule JRS, Freire CA, Simberloff D. Introduction of nonnative freshwater fish can certainly be bad. Fish Fish. 2009; 10(1):98–108. https://doi.org/10.1111/j.1467-2979.2008.00312.x
terHost CP, Munguia P. Measuring ecosystem function: consequences arising from variation in biomass-productivity relationships. Community Ecol. 2008; 9(1):39–44. https://doi.org/10.1556/ComEc.9.2008.1.5
Tilman D, Knops J, Wedin D, Reich P, Ritchie M, Siemann E. The influence of functional diversity and composition on ecosystem processes. Science. 1997; 277(5330):1300–02. http://dx.doi.org/10.1126/science.277.5330.1300
Tilman D, Isbell F, Cowles JM. Biodiversity and ecosystem functioning. Annu Rev Ecol Evol Syst. 2014; 45:471–93. http://dx.doi.org/10.1146/annurev-ecolsys-120213-091917
Toussaint A, Charpin N, Beauchard O, Grenouillet G, Oberdorff T, Tedesco PA, Brosse S, Villéger S. Non-native species led to marked shifts in functional diversity of the world freshwater fish faunas. Ecol Lett. 2018; 21(11):1649–59. http://dx.doi.org/10.1111/ele.13141
Toussaint A, Charpin N, Brosse S, Villéger S. Global functional diversity of freshwater fish is concentrated in the Neotropics while functional vulnerability is widespread. Sci Rep. 2016; 6:22125. http://dx.doi.org/10.1038/srep22125
Uno H, Power ME. Mainstem-tributary linkages by mayfly migration help sustain salmonids in a warming river network. Ecol Lett. 2015; 18(10):1012–20. http://dx.doi.org/10.1111/ele.12483
Zaret TM, Paine RT. Species introduction in a tropical lake. Science. 1973; 182(4111):449–55. http://dx.doi.org/10.1126/science.182.4111.449
Authors
![]() Luiza Bangoim Leal1,
Luiza Bangoim Leal1,![]() David J. Hoeinghaus2,
David J. Hoeinghaus2, ![]() Zacchaeus G. Compson2,
Zacchaeus G. Compson2,![]() Angelo A. Agostinho3,
Angelo A. Agostinho3,![]() Rodrigo Fernandes4 and
Rodrigo Fernandes4 and![]() Fernando M. Pelicice5
Fernando M. Pelicice5
[1] Programa de Pós-Graduação em Ecologia de Ecótonos (current Biodiversidade, Ecologia e Conservação), Universidade Federal do Tocantins. Rua 3, Quadra 17, Jardim dos Ipês, 77500-000 Porto Nacional, TO, Brazil. luizableal@gmail.com (corresponding author).
[2] Department of Biological Sciences, Advanced Environmental Research Institute, University of North Texas, 1155 Union Circle #310559, Denton, TX 76203-5017, U.S.A. (DJH) david.hoeinghaus@unt.edu, (ZGC) zacchaeus.compson@unt.edu.
[3] Programa de Pós-Graduação em Ecologia de Ambientes Aquáticos Continentais, Núcleo de Pesquisa em Limnologia, Ictiologia e Aqüicultura, Departamento de Biologia, Universidade Estadual de Maringá, Av. Colombo, 5790, 87020-900 Maringá, PR, Brazil. agostinhoaa@gmail.com.
[4] Laboratório de Ecologia de Comunidades e Paisagens, Departamento de Biociências, Universidade Federal Rural do Semi-Árido, Av. Presidente Costa e Silva, 572, 59625-900 Mossoró, RN, Brazil. rfernandes@ufersa.edu.br.
[5] Núcleo de Estudos Ambientais, Programa de Pós-Graduação em Biodiversidade, Ecologia e Conservação, Universidade Federal de Tocantins. Rua 3, Quadra 17, Jardim dos Ipês, 77500-000 Porto Nacional, TO, Brazil. fmpelicice@gmail.com.
Authors Contribution 
Luiza Bangoim Leal: Conceptualization, Formal analysis, Investigation, Writing-original draft.
David Joseph Hoeinghaus: Conceptualization, Formal analysis, Supervision, Validation, Writing-review and editing.
Zacchaeus Greg Compson: Formal analysis, Software, Writing-review and editing.
Angelo Antonio Agostinho: Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing-review and editing.
Rodrigo Fernandes: Formal analysis, Software, Writing-review and editing.
Fernando Mayer Pelicice: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
Fish individuals were collected by license number 004/2001 (IBAMA process number 02017.000.586/01-68).
Competing Interests
The authors declare no competing interests.
How to cite this article
Leal LB, Hoeinghaus DJ, Compson ZG, Agostinho AA, Fernandes R, Pelicice FM. Changes in ecosystem functions generated by fish populations after the introduction of a non-native predator (Cichla kelberi) (Perciformes: Cichlidae). Neotrop Ichthyol. 2021; 19(3):e210041. https://doi.org/10.1590/1982-0224-2021-0041
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted June 28, 2021 by Emili Garcia-Berthou
Accepted June 28, 2021 by Emili Garcia-Berthou
![]() Submitted January 29, 2021
Submitted January 29, 2021
![]() Epub Sept 24, 2021
Epub Sept 24, 2021