![]() Carla Patrícia de Souza1,2,
Carla Patrícia de Souza1,2,![]() Carlos Alberto de Sousa Rodrigues-Filho1,2,
Carlos Alberto de Sousa Rodrigues-Filho1,2,![]() Francisco Antônio Rodrigues Barbosa3 and
Francisco Antônio Rodrigues Barbosa3 and![]() Rafael Pereira Leitão1
Rafael Pereira Leitão1
Abstract
Biological invasions are leading several species to extinction and are projected as a main driver of biodiversity changes in lakes for this century. However, the knowledge of their impacts on the Neotropical ichthyofauna over time remains largely incipient, especially when considering the functional diversity of native communities. Here we aim to identify the effects of non-native species, especially the non-native piscivorous Cichla kelberi and Pygocentrus nattereri, on the functional diversity of the native ichthyofauna of the Carioca Lake, Middle Rio Doce basin, state of Minas Gerais. Using fish occurrence data for eight years from 1983 to 2010 combined with an ecomorphological-trait analysis, we found that while the native species richness dropped to 56%, the functional richness is only 27% of that found before introductions. In other words, more than species, the ichthyofauna suffered an impressive decline in the range of functional traits, which can further have severe impacts on ecological processes within that system. When considering all the components of the current ichthyofauna (native and non-native species), neither taxonomic nor functional richness have changed over time. However, even keeping biodiversity levels, non-native species are not able to fully compensate for the extinct native ones in terms of functions.
Keywords: Biodiversity erosion, Biological invasions, Functional traits, Non-native fish, Rio Doce basin.
Invasões biológicas vêm levando várias espécies à extinção, sendo projetado como o principal causador de mudanças na biodiversidade em lagos neste século. Entretanto, o conhecimento dos impactos sobre a ictiofauna Neotropical ao longo do tempo permanece bastante incipiente, especialmente quando se considera a diversidade funcional de comunidades nativas. Neste estudo, nós procuramos identificar os efeitos das espécies não nativas, especialmente os piscívoros não nativos Cichla kelberi and Pygocentrus nattereri, sobre a diversidade funcional da ictiofauna da Lagoa Carioca, bacia do médio rio Doce, Minas Gerais. Utilizando dados de ocorrência de oito anos entre 1983 e 2010, combinados a uma análise de atributos ecomorfológicos, observamos que a riqueza de espécies decaiu a 56%, enquanto a riqueza funcional é de apenas 27% da encontrada antes das introduções. Ou seja, mais do que espécies, a ictiofauna sofreu um declínio significativo na amplitude de atributos funcionais, o que pode levar a impactos severos em processos ecológicos neste sistema. Ao considerar todos os componentes da ictiofauna atual (espécies nativas e não nativas), as métricas de riqueza não sofreram alterações ao longo do tempo. Entretanto, mesmo mantendo os níveis de biodiversidade, espécies não nativas não são capazes de compensar totalmente as espécies nativas extintas em termos funcionais.
Palavras-chave: Atributos funcionais, Bacia do rio Doce, Erosão da biodiversidade, Invasões biológicas, Peixes não nativos. Brasileiro de Geografia e Estatística.
Introduction
Biological invasions cause strong changes in ecosystems resulting in devastating ecological consequences. Together with resource overexploitation, landscape alterations (e.g., deforestation), pollution and climatic changes, species invasion is considered one of the most important causal factors of biodiversity erosion on Earth (Vitule et al., 2009). Particularly to freshwater systems, invasions have already led several species to extinction (Clavero, García-Berthou, 2005), and are projected as the main driver of biodiversity changes in lakes for this century (Sala et al., 2000). As a result of globalization, the introduction of aquatic species out of their original area of distribution became more frequent and intense, with main vectors related to water released from ships’ ballast, biological control agents, aquarium trade, sport fishing, and food allowance (Millennium Ecosystem Assessment, 2005). After introduced, non-native species can settle down in a native community and extirpate native organisms by a variety of mechanisms frequently related to ecological interactions, such as predation (Kaufman, 1992; Pereira et al., 2015) and competition (Bøhn et al., 2008; Pelicice et al., 2017). Nevertheless, the sequence of events involved behind these general mechanisms is largely unknow and highly unpredictable, hampering effective actions to detect, control and restore natural conditions (Blackburn et al., 2011; Lockwood et al., 2013).
Systematic studies on the impact of invasive species in tropical aquatic ecosystems are still incipient. One of the biggest challenges for understanding the consequences of biological invasions is the lack of long-term historical series that evidence the temporal alterations on the structure of native biological communities (e.g., Olden, 2006; Magalhães et al., 2020). Additionally, despite of the recent conceptual and methodological advances to describe alterations on the structure of communities induced by anthropogenic disturbances, the taxonomic approach remains the rule. This traditional view to quantify biodiversity (i.e., based purely on species richness and/or diversity indices) has been constantly shown as scarcely predictive, given that it neglects the species functional traits and how they mediate ecological interactions (Villéger et al., 2008). Therefore, these community descriptors are frequently considered as a limited quantitative tool in monitoring environmental impacts. Different processes may affect species from different ways, potentially providing delayed or even mistaken signs of perturbation (Mouillot et al., 2013). Therefore, approaches such as the functional diversity of communities in the context of anthropogenic disturbances have been growing in several fields of ecology and for different biological groups (Biswas, Mallik, 2010; Dolbeth et al., 2016; Arnan et al., 2018; Teresa et al., 2021).
The functional diversity of a community can be described as the range of the species ecological traits (Mouillot et al., 2013). Such an approach allows the characterization of species by their functional relations with the environment and with other species, regardless of their taxonomic identities (Cianciaruso et al., 2009). For freshwater fishes, traits related to ecomorphology and life’s history have shown to be particularly good predictors of habitat degradation and biological invasions (Olden, 2006; Leitão et al., 2018). As so, it is possible to better evaluate the causes of species extinctions, and the consequences on the sustainability of ecological processes (Leitão et al., 2016). The functional approach is thus gaining importance, once that it provides a more mechanistic way to assess the human-induced impacts on biodiversity (Magurran, 2004; Ernst et al., 2006; Petchey, Gaston, 2006). Therefore, using this approach in the context of biological invasions seems to be a promising strategy to increase our capacity to detect and predict their impacts on Neotropical aquatic communities.
One of the most emblematic cases of biological invasion in Brazilian freshwater ecosystems has been occurring in the Middle Rio Doce basin. This region holds one of the largest lacustrine systems in South America (Maia-Barbosa et al., 2010), and harbor about a third of all fish species of the basin (Godinho, Vieira, 1998). The introduction of fishes since the 1970’s culminated with the local extinction of several native species, such as the small characin Moenkhausia vittata (Castelnau, 1855) and the carnivore Oligosarcus solitarius Menezes, 1987, currently not found in many lakes (Sunaga, Verani, 1991; Latini et al., 2004; Latini, Petrere, 2004; Oporto, 2013; Fragoso-Moura et al., 2016). The history of introduction and dispersion of fish species across the Rio Doce basin is related mostly to aquaculture and sport fishing (Godinho et al., 1994; Latini et al., 2004; Magalhães et al., 2021). The latter is likely the reason for the establishment of the peacock bass Cichla kelberi Kullander & Ferreira, 2006, native to the Tocantins-Araguaia basin, and the red piranha Pygocentrus nattereri Kner, 1858, native to the Amazon, Paraguay-Paraná, northeastern Brazilian coastal rivers and Essequibo basins (Fricke et al., 2021), impacting the local ichthyofauna given their piscivorous habits and high voracity (Sazima, Machado, 1990; Chellappa et al., 2003).
Recently, Fragoso-Moura et al. (2016) conducted a thorough compilation of a rare historical data of the ichthyofauna from Carioca Lake, a well-studied system located at the Rio Doce State Park (PERD acronym in Portuguese, hereafter) and detected a broad dominance of C. kelberi and P. nattereri, besides the presence of other introduced fishes in the community, e.g., the oscar Astronotus cf. crassipinnis (Heckel, 1840), the cascarudo Hoplosternum littorale (Hancock, 1828),and the African catfish Clarias gariepinus (Burchell, 1822). The authors detected an impressive decline in the number of native species during the last four decades. Still unknown, however, is how this community has been changed in its functional structure. We intend here to fill such a gap from this alternative perspective and point out some mechanistic ways to explain this biodiversity erosion. For this, we compared the levels of fish taxonomic and functional diversity over time, from 1983 to 2010, in Carioca Lake. Given that the functional approach is supposedly more sensible to anthropogenic disturbances (Mouillot et al., 2013), we hypothesize that the decline in functional richness is more pronounced than that of species richness after the introduction of non-native fish species, especially C. kelberi and P. nattereri. We expect to get a better comprehension of the processes behind biological invasions and thus helping to implement conservation and management actions for the ichthyofauna in the region.
Material and methods
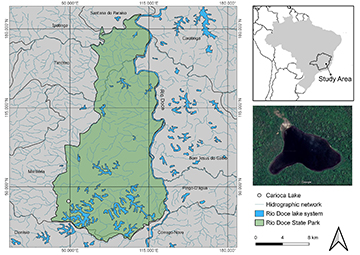
Study area. The Middle Rio Doce basin, state of Minas Gerais, comprises one of the largest lacustrine systems of the Brazilian territory (Tundisi, de Meis, 1985), with more than 250 natural lakes of different sizes, dynamics, biodiversity, and levels of ecological integrity (Maillard et al., 2012). Studies have shown that this system originated around ten thousand years ago as a result of blockages of the mouth of ancient Rio Doce tributaries, creating natural dams (Godinho, 1996). Their ichthyofaunas can thus be treated as closed communities and dispersion rates of species between them can be considered very low over time (Latini et al., 2004). The relief of the region is sinuous with elevation varying from 195 to 525 m and depressions occupied by water bodies (Gilhuis, 1986). The weather is hot and wet, with annual rainfall between 1000 and 1250 mm and defined dry (April to September) and rainy (October to March) seasons. The region comprises the Rio Doce State Park (PERD; Fig. 1), which represents the biggest remnant portion of the Atlantic Forest in Minas Gerais (~36,000 ha), surrounded by Eucalyptus plantations, pasture and diverse agriculture (Barbosa, Moreno, 2002). In 1998, PERD was included in the Long-Term Ecological Research Programme (PELD Rio Doce – Site MLRD), and in 2009 the region was recognized internationally as a major area for conservation of biodiversity (i.e., Ramsar site), as well as for ecological, economic, cultural, scientific, and recreational services (Mikhailova, Barbosa, 2002).
More than 40 lakes are found within the limits of PERD which are well preserved, at least in terms of adjacent forest and water quality (Maillard et al., 2012). Amongst them, Carioca Lake was chosen in this study because its fish community has been repeatedly sampled over the last decades and the introduction history of the non-native predators Cichla kelberi and Pygocentrus nattereri is known. Located at the southern portion of PERD, Carioca Lake has 0.12 km², is considered mesotrophic, with water temperature varying between 24.3 ºC to 33.4 ºC, maximum depth of 11.8 m, and surrounded by dense vegetation (Bezerra-Neto et al., 2010).
FIGURE 1 | The lacustrine system of the Middle Rio Doce basin, state of Minas Gerais, Brazil. The green polygon delimits the area of the Rio Doce State Park (PERD) and the white circle indicates the location of Carioca Lake.
Database. We used occurrence data of native fishes from Carioca Lake obtained from the literature that spans nearly three decades (from 1983 to 2010) (Sunaga, Verani, 1991; Godinho et al., 1994; Latini et al., 2004; Vasconcellos et al., 2005; Pinto-Coelho et al., 2008; Fragoso-Moura et al., 2016), already compiled in Fragoso-Moura et al. (2016). Fish samples were conducted with gillnets of different mesh sizes, seine, dragging and casting nets (specific details given in each published study). Sampling effort was different among studies, with new records of some native species occurring only in the most recent ones. However, given that Carioca Lake is a closed system and the last studies applied greater sampling effort, we assumed that native species listed only in recent records were already present in the fish assemblage since the earliest event of the temporal series. We also limited the analysis to presence-absence, rather than abundance data, to minimize the different sampling effort between years. Finally, we excluded species identified only at the family level in Fragoso-Moura et al. (2016). When needed, we updated the nomenclature of species, based on Fricke et al. (2021).
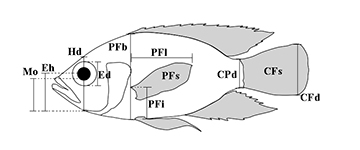
Functional diversity. We first conducted an ecomorphological analysis to functionally characterize all species. Eleven morphometric measures (Fig. 2) were taken from a digital picture (obtained from the authors’ image banks or from the original description of the species) of one adult individual from each species, and processed in ImageJ software (Abràmoff et al., 2004). These measures were then combined in eight ecomorphological traits representing three key functions: foraging, locomotion, and preferential habitat use (Tab. 1; values for each species in Tab. S1). All traits were standardized (mean zero and one standard deviation) to make it possible to compare different measurement units and to avoid trivial correlation with body size (Villéger et al., 2010). We used ecomorphological traits first because they represent an important dimension of the species niche (Winemiller et al., 2015), and are the only set of characteristics available for all species recorded in Carioca Lake over the time series; i.e., the lack of information on fish diet, life history and other biological traits are still a recurrent limitation for functional studies of the Neotropical freshwater ichthyofauna (Leitão et al., 2016; Teresa et al., 2021). Additionally, although some categorical traits could have been used based on literature (e.g., feeding guild), many species characteristics may change over time, even as a result of non-native introductions. For example, there is evidence that after the arrival of P. nattereri (introduced) in PERD lakes, the native Hoplias malabaricus (Bloch, 1794) has changed from a piscivorous to an invertivorous diet (Pompeu, Godinho, 2001). Using such categorical trait would thus weakly express the functional roles of some species and could represent circularity on the relations investigated here.
Data analysis. We conducted all analytical procedures considering two different scenarios: “all species”, which included both native and non-native species; and “only native species”. From the eight ecomorphological traits, we built a Euclidean distance matrix considering the entire time series (1983–2010) and then performed a Principal Component Analysis (PCA) to represent the multidimensional functional space for each scenario. After accounting for the balance between the number of dimensions and the quality of the functional space (according to Maire et al., 2015), we kept the first three PCA axes to synthesize the eight ecomorphological traits. From this three-dimensional functional space, we quantified Functional Richness (FRic); i.e., the volume of the convex hull polygon occupied by the species found in each sampled year (1983, 1985, 1987, 1992, 2003, 2005, 2008, and 2010). FRic quantitatively describes the range of trait combinations by the focal set of species (Villéger et al., 2008).
FIGURE 2 | Morphometric measures taken from digital pictures: CPd – caudal-peduncle minimal depth, CFd – caudal-fin maximum depth, CFs – caudal-fin surface, PFi – distance from pectoral-fin insertion to the bottom of the body, PFb – body depth at the level of the pectoral-fin insertion, PFl – pectoral-fin length, PFs – pectoral-fin surface, Hd – head depth along the vertical axis of the eye, Ed – eye diameter, Eh – distance from the center of the eye to the bottom of the head, Mo – distance from the tip of the upper jaw to the bottom of the head along the head depth axis. Adapted from Leitão et al. (2016).
TABLE 1 | List of the eight ecomorphological traits used to functionally characterize fish species from the Carioca Lake, Middle Rio Doce basin, state of Minas Gerais, southeastern Brazil.
| Morphological trait | Calculation | Ecological meaning | References |
| Oral-gape position (Ops) | Mo/Hd | Feeding method in the water column | Adapted from Sibbing, Nagelkerke (2000) |
| Eye size (Edst) | Ed/Hd | Prey detection | Adapted from Boyle, Horn (2006) |
| Eye position (Eps) | Eh/Hd | Vertical position in the water column | Gatz (1979) |
| Pectoral-fin position (PFps) | PFi/PFb | Use of pectoral fin for maneuverability | Dumay et al. (2004) |
| Pectoral-fin aspect ratio (PFar) | PFl2/PFs | Use of pectoral fin for propulsion | Adapted from Fulton et al. (2001) |
| Caudal-peduncle throttling (CPt) | CFd/CPd | Use of caudal fin for propulsion and/or direction | Webb (1984) |
| Caudal-fin aspect ratio (CFar) | CFd2/CFs | Caudal fin use for propulsion and/or direction | Webb (1984) |
| Fins surface ratio (Frt) | 2 x PFs/CFs | Main type of propulsion between caudal and pectoral fins | Villéger et al. (2010) |
To investigate the changes in fish diversity over the years for each scenario, we conducted an analysis of covariance (ANCOVA), which considers the differences between taxonomic and functional approaches, as well as the interaction between both factors (year × diversity approach). All analyses were carried out in the software R (R version 4.0.0; R Core Team, 2020). FRic was computed using the multidimFD (http://villeger.sebastien.free.fr/Rscripts.html), and ANCOVA was conducted using the lm (base package) and the Anova function (type III test; car package).
Results
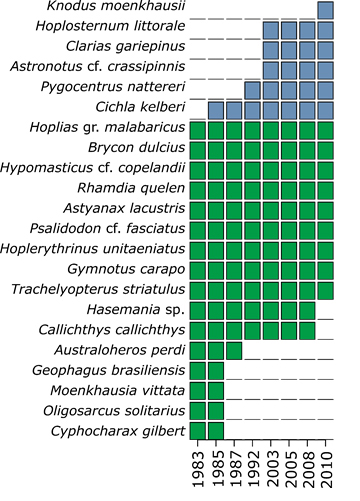
Fish species composition of Carioca Lake changed over the sampling years, with the replacement of seven native (the saguiru Cyphocharax gilbert (Quoy & Gaimard, 1824), the lambari-bocarra Oligosarcus solitarius, the lambari-chatinha Moenkhausia vittata, the cará Geophagus brasiliensis (Quoy & Gaimard, 1824), the cará-verde Australoheros perdi Ottoni, Lezama, Triques, Fragoso-Moura, Lucas & Barbosa, 2011, the tamboatá Callichthys callichthys (Linnaeus, 1758) and the lambari Hasemania sp.) by six non-native species (Cichla kelberi, Pygocentrus nattereri, Astronotus cf. crassipinnis, Clarias gariepinus, Hoplosternum littorale,and Knodus moenkhausii (Eigenmann & Kennedy, 1903)) between 1983 and 2010 (Fig. 3). An updated list of the 22 species registered in Fragoso-Moura et al. (2016) is available in Tab. S2.
FIGURE 3 | Compositional change of the ichthyofauna from Carioca Lake, Middle Rio Doce basin state of Minas Gerais, southeastern Brazil. Green and blue squares indicate, respectively, the presence of native and introduced species in each year.
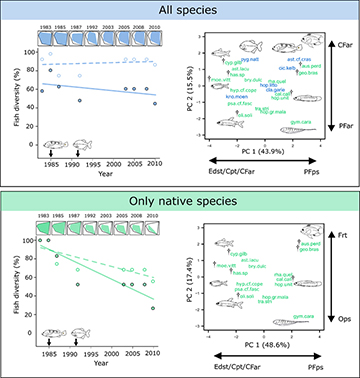
We did not detect changes in taxonomic or functional richness (FRic) over time when considering all species, native and non-native, of the fish assemblage (years: F1,12 = 2.13, p = 0.17; diversity approach: F1,12 = 0.06, p = 0.81; interaction: F1,12 = 1.33, p = 0.27; Fig. 4). On the other hand, when considering only the native ichthyofauna, both diversity indicators decreased (years: F1,12 = 20.12, p < 0.001; diversity approach: F1,12 = 0.72, p = 0.41; interaction: F1,12 = 4.49, p = 0.06; Fig. 4), firstly after the introduction of C. kelberi (decay of 16.5% in FRic) and especially with the introduction of P. nattereri, in 1992 (decay of 47.3% in FRic). Overall, while the number of native species decreased to 56.3% (9 species in 2010 out of 16 species in 1983), FRic dropped down to 27.2% in relation to the pre-introductions condition (Fig. 4).
FIGURE 4 | Temporal changes in species richness (dashed lines) and functional richness (FRic; continous lines) of the ichthyofauna from Carioca Lake, Middle Rio Doce basin, state of Minas Gerais, considering two scenarios: “all species”, including native and non-native; and “only native species” (left figures). Plotted values expressed as a proportion to the maximum richness. The arrows represent the first records of the introduced piscivorous Cichla kelberi (in 1985) and Pygocentrus nattereri (in 1992) in the system. The upper plots represent the functional space (only two dimensions for simplify visualization), with polygons indicating the proportion filled by the set of species (FRic) in each year. Right figures illustrate the functional space showing the position of each species. Green and blue colors indicate, respectively, the native and introduced species, and crosses indicate native species extirpated from the lake. Codes at the ends of the arrows are the most important ecomorphological traits for each axis of the PCA (for functional trait and species codes, see Tab. 1 and Tab. S2).
TABLE 2 | Correlation matrix between ecomorphological traits and each of the three axes of the PCAs representing the functional space of the two scenarios (“all species” and “only native”) of the fish assemblage from Carioca Lake, Middle Rio Doce basin. The most significant traits for each dimension are represented in bold.
| Morphological traits | “All species” | “Only native” | ||||
| PC1 | PC2 | PC3 | PC1 | PC2 | PC3 | |
| Oral-gape position (Ops) | -0.38 | -0.33 | 0.73 | -0.57 | -0.54 | 0.50 |
| Eye size (Edst) | -0.80 | 0.01 | 0.17 | -0.87 | 0.07 | -0.28 |
| Eye position (Eps) | 0.50 | 0.07 | 0.42 | 0.34 | -0.49 | -0.71 |
| Pectoral-fin position (PFps) | 0.83 | 0.26 | 0.28 | 0.84 | 0.08 | -0.20 |
| Pectoral-fin aspect ratio (PFar) | -0.50 | -0.46 | -0.49 | -0.42 | -0.29 | -0.31 |
| Caudal-peduncle throttling (CPt) | -0.77 | 0.54 | 0.07 | -0.86 | 0.31 | -0.20 |
| Caudal-fin aspect ratio (CFar) | -0.78 | 0.60 | 0.04 | -0.84 | 0.40 | -0.19 |
| Fins surface ratio (Frt) | 0.60 | 0.46 | -0.32 | 0.58 | 0.71 | 0.01 |
| Variance explained (%) | 43.9 | 15.5 | 14.6 | 48.6 | 17.4 | 13.4 |
The morphological traits more negatively associated with PC1 were eye size (Edst), caudal-peduncle throttling (Cpt) and caudal-fin aspect ratio (CFar) (Fig. 4; Tab. 2). Species with high values for these traits are visually oriented and predominantly use the water column due to their swimming capacity (Tab. 1). The loss of the species with these characteristics was the main responsible for the first FRic decay in 1987 (Fig. 4). Pectoral-fin position (PFps) was positively correlated with PC1, while fins surface ratio (Frt) and oral-gape position (Ops) were, respectively, the most positively and negatively correlated morphological traits with PC2 (Fig. 4; Tab. 2). These characteristics are found in species with good maneuverability in complex habitats and association with the bottom for feeding (Tab. 1), whose loss in the assemblage were responsible for the great FRic decay in 1992 (Fig. 4).
Discussion
Fish species introductions in the Middle Rio Doce basin have led to one of the most emblematic cases of biological invasion in Brazilian freshwater ecosystems, imperiling the aquatic biota even in protected areas. After compiling published studies and building a precious temporal dataset of the ichthyofauna from Carioca Lake, Fragoso-Moura et al. (2016) registered a significant decay in the number of native species. Over the last decades, many native fishes, including the endemic (and with the type locality inside PERD) Australoheros perdi and Oligosarcus solitarius, were no more captured soon after the detection of the non-native piscivorous C. kelberi and P. nattereri, which indicates that they became rare or extinct in the lake. By considering the functional diversity of this fish assemblage, we have now shown that the impacts of the biological invasion were even more severe than the loss of species itself. Relative to the condition found before the introductions, the current functional richness of the native ichthyofauna is more than 70% lower, a loss two times higher than its taxonomic counterpart. In other words, more than species, there is a drastic reduction in the range of functional traits related to forms of locomotion and use of habitat and food resources.
The erosion of functional richness of the native ichthyofauna occurred in sequential steps, a first one soon after the introduction of C. kelberi in 1985, with a decrease of ~16% in FRic, and a second just with the first record of P. nattereri in 1992, with an additional fall of 30%. This degradation profile is related to the position of the species in the ecomorphological space. Of the seven native species not registered in the system after the introductions, four are located at the edge of the space, which means the extirpation of highly specialized functions. Particularly the strongest second fall can be explained by the sequential loss of the cichlids Geophagus brasiliensis and A. perdi, which hold unique sets of ecomorphological traits within the local assemblages. In the following years after that, new introductions were recorded and, after relative stability in FRic levels, the native species Hasemania sp. and C. callichthys were not recorded in the last sampling event (2010). Particularly, the absence of C. callichthys, also a morphologically specialized species, caused a significant final decay in FRic of the native ichthyofauna. Among several factors, the impact of invasive species in natural environments is dependent upon local biotic characteristics, and systems with few native species tend to present higher vulnerability to invasion (Moyle, Light, 1996). The strong domination of the two predators at initial phases of invasion may have created a destabilized environment more conducive to the establishment of other introduced species. This can be seen as an empirical example of the invasive meltdown hypothesis, a process by which non-native species facilitate the invasion of each other through positive interspecific interactions (Simberloff, 2006).
The set of lost morphological traits is likely sensitive to predation. The first species lost have large eyes and high values of caudal-peduncle throttling and caudal-fin aspect ratio, characteristics of diurnal species (Boyle, Horn, 2006) and good swimming ability in open waters (Webb, 1984), feeding along the vertical column (e.g., O. solitarius and M. vittata) or closer to the lake bottom (e.g., C. gilbert). Although not positioned close to these species in the ecomorphological space, Cichla kelberi also has a diurnal habit and preys both in open waters or close to shelters (Chellappa et al., 2003; Pelicice et al., 2015). Species with more developed and upper-inserted pectoral fins (G. brasiliensis and later A. perdi) were also lost. These characteristics favor maneuverability (Villéger et al., 2010) and allows the occupation of structured habitats, such as areas dominated by aquatic macrophytes and wood debris. This habitat type is frequently occupied by P. nattereri, which uses it as spawning sites, development of juveniles (Pauly, 1994), and as suitable shelters to ambush their preys (Sazima, Machado, 1990). Therefore, the sequence of species extirpation in Carioca Lake seems to follow a predictable relation between prey characteristics associated with the use of habitats and the different predation strategies applied by the two introduced piscivores.
Besides predation, the native ichthyofauna of Carioca Lake has probably been also affected by competitive interactions with introduced species. Studying the diet of Hoplias gr. malabaricus, Pompeu, Godinho (2001) observed a clear difference when comparing lakes (including Carioca Lake) with and without C. kelberi and P. nattereri, with high proportion of shrimps rather than fish in the diet when occurring in sympatry with the introduced piscivores. The authors suggested that this feeding plasticity may be allowing the maintenance of Hoplias gr. malabaricus in the system.On the other hand, O. solitarius, a species endemic of Rio Doce’s lakes, disappeared from Carioca Lake just after the introduction of C. kelberi. It is one of the smallest piscivores of the region (Godinho, 1996), and perhaps it was disfavored by a combination of predation and competition with the invasive peacock bass. Finally, given their similar ecological demands, it is plausible that Hasemania sp. and C. callichthys have been negatively affected by competition with, respectively, the small characin Knodus moenkhausii and the catfish Hoplosternum littorale, more recently introduced in the system. Although feasible, these are yet tentative interpretations to explain the absence of such native species in Carioca Lake, given that testing competitive hypotheses in the field is operationally difficult (Jackson et al., 2001). However, we believe this represents a vast avenue for further investigation on the role of interspecific interactions in the context of freshwater biological invasions.
Some native species still resist in this competitive and high predation-pressure environment. Taking a deeper look at the ecomorphological space, we can argue that this resistance can be in part associated with characteristics related to the use of habitats not dominated by the introduced piscivores. For instance, a great part of the ecomorphological space not suppressed over time is composed of species with small eyes positioned in the upper portion of the head and subterminal mouth. These traits generally favor benthic habits (Gatz, 1979), which make more difficult the predation by both C. kelberi and P. nattereri. Other traits not directly evaluated here may also be important in the context of antipredatory mechanisms. This is the case of rigid fin spines (e.g., found in Trachelyopterus striatulus (Steindachner, 1877)), large body sizes (e.g., Hoplias gr. malabaricus, Hypomasticus cf. copelandii (Steindachner, 1875), Hoplerythrinus unitaeniatus (Spix & Agassiz, 1829), Rhamdia quelen (Quoy & Gaimard, 1824),and Brycon dulcis Lima & Vieira, 2017), or cryptic coloration (e.g., Gymnotus carapo Linnaeus, 1758) that helps in the camouflage of visually oriented predators (Keenleyside, 1979). Another set of traits that may also explain the maintenance of some native species is related to behavioral and life-history aspects, such as nocturnal habits (e.g., R. quelen, T. striatulus, G. carapo), which minimize the chance of being captured by C. kelberi and P. nattereri (Nico, 1990; Sazima, Machado, 1990; Santos et al., 2011), and parental care (e.g., H. gr. malabaricus and H. unitaeniatus), which aids preventing predation of eggs, larvae and juveniles (Ota et al., 2018).
Taking into account the whole ichthyofauna, considering both native and non-native species, there was no significant changes in functional richness over time. However, this does not mean that the new elements of the ichthyofauna fully compensate for the loss of the native ones. By visualizing the functional space (Fig. 4), we can note that the positions of some non-native fish, particularly P. nattereri, C. kelberi and Astronotus cf. crassipinnis, are at the extreme portions, contributing to the assemblage FRic. However, these areas were not all previously filled by native fishes. Therefore, the maintenance of FRic levels over time, even with the native species losses, indicates more a change in the functional structure than a maintenance of functions; i.e., the functional roles promoted by the non-native are somewhat different from the ones promoted by the extirpated natives. Such a change can lead to significant impacts to community or even to ecosystem properties. For instance, although positioned relatively close to C. kelberi and A. cf. crassipinnis in the morpho-functional space, the natives G. brasiliensis and A. perdi are omnivorous-benthophagous feeders, having thus quite different roles to the trophic structure of the ichthyofauna compared to these non-native piscivores. The extirpated native detritivore C. gilbert stirs the sediment when feeding at the lake bottom (see Sazima, Caramaschi, 1989) for a detailed description of this behavior in related curimatids). Studies on sediment-feeding behavior have already revealed significant effects of fish on ecosystem functioning by modulating carbon flux, primary production and respiration in aquatic ecosystems (e.g., Flecker, 1996; Taylor et al., 2006). Another example of potential loss of a unique and specialized functional role is the extirpation of O. solitarius. Although also being a piscivore, O. solitarius is morphologically very different from C. kelberi and P. nattereri (opposing extremes in the functional space, Fig. 4), possessing higher caudal peduncle throttling, bigger eyes and longer pectoral fins positioned at the lower portion of body. These characteristics favor a high visual accuracy to detect preys in the water column and a long-distance swimming ability (Gatz, 1979; Winemiller, 1991), likely enabling the consumption of different resources and transportation of matter and energy across broader areas in the lake.
This study provided empirical evidence that the functional richness was more sensitive than species richness in response to non-native fish invasions in Carioca Lake. However, we are aware that there are still important gaps to be filled. Firstly, we used only ecomorphological traits that, although widely used and recognized as a suitable proxy to infer some functional aspects, do not inform about several other niche dimensions (Teresa et al., 2021). For example, life-history aspects, such as fecundity and spawning site, may clarify issues related to the resilience of native populations against the pressures promoted by introduced species. Accurate diet information would contribute to our interpretations about the degree in which non-native species compensate the absence of extirpated native for the structure and dynamics of local food webs. Another important limitation is the differences in sampling efforts among the studies used in the temporal series, which led us i) to assume that native species recorded only in the last studies were already present before, and ii) to disregard species abundance data. Precisely knowing the occurrence and abundance for each species in each sampling event would allow a more realistic picture of the biodiversity changes following introductions. This clearly illustrates the need for long-term ecological research programs (LTERs), which unfortunately is receiving much less attention and financial support in recent times in Brazil. Despite the limitations, we consider that this research represents an important first step for understanding the changes in Carioca Lake’s ichthyofauna functional structure.
With the purpose of local and regional biodiversity conservation it is expected that management actions, for instance, population reduction by allowing sport fishing with selective removal of non-native fish (Britton et al., 2011; Coggins et al., 2011), will be taken in PERD and its surroundings for an urgent control against new introductions, considering the impacts that have occurred and those predicted in the medium and long term. Studies on behavior, feeding ecology, reproduction, as well as the impacts of other non-native species in the region are essential to create subsidies that enable the conservation of fish species that still resist. Since most of the introductions were intentional, environmental education activities that raise awareness among the local population and visitors are of great importance for conservation efforts to become effective and to avoid future introductions (Lima et al., 2010).
Acknowledgments
We are grateful to the two anonymous reviewers and the Associate Editor, Prof. Emili García-Berthou, for the critical reading and suggestions, to Diego Castro and Gilberto N. Salvador for important suggestions in earlier versions of the manuscript, to Erica Caramaschi, Carlos B. M. Alves, Dr. Tulio F. Teixeira, Tiago C. Pessali and Jansen Zuanon for helping with discussion on species biology and distribution, and to Anna Quaresma for helping with the translation. We also thanks Agência Nacional de Energia Elétrica and Companhia Energética de Minas Gerais (P&D ANEEL / CEMIG GT599 – PROECOS Project) and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG APQ–01611–17, APQ–00401–19) for financially support Laboratório ECOPeixes-UFMG, and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq 441481/2016–7) and FAPEMIG (APQ-04812–17) for support PELD Rio Doce – Site MLRD.
References
Abràmoff MD, Magalhães PJ, Ram SJ. Image processing with ImageJ. Biophotonics. 2004; 11(7):36–42. Available from: https://imagej.nih.gov/ij/docs/pdfs/Image_Processing_with_ImageJ.pdf
Arnan X, Arcoverde GB, Pie MR, Ribeiro-Neto JD, Leal IR. Increased anthropogenic disturbance and aridity reduce phylogenetic and functional diversity of ant communities in Caatinga dry forest. Sci Total Environ. 2018; 631–632:429–38. https://doi.org/10.1016/j.scitotenv.2018.03.037
Barbosa FAR, Moreno P. Mata Atlântica e sistema lacustre do Médio Rio Doce. In: Seeliger U, Cordazzo C, Barbosa FAR, editors. Os sites e o programa brasileiro de pesquisas ecológicas de longa duração. Belo Horizonte: Universidade Federal de Minas Gerais; 2002. p.69–81.
Bezerra-Neto JF, Briguenti LS, Pinto-Coelho RM. A new morphometric study of Carioca Lake, Parque Estadual do Rio Doce (PERD), Minas Gerais State, Brazil. Acta Sci Biol Sci. 2010; 32(1):49–54. https://doi.org/10.4025/actascibiolsci.v32i1.4990
Biswas SR, Mallik AU. Disturbance effects on species diversity and functional diversity in riparian and upland plant communities. Ecology. 2010; 91(1):28–35. https://doi.org/10.1890/08-0887.1
Blackburn TM, Pyšek P, Bacher S, Carlton JT, Duncan RP, Jarošík V, Wilson JRU, Richardson DM. A proposed unified framework for biological invasions. Trends Ecol Evol. 2011; 26(7):333–39. https://doi.org/10.1016/j.tree.2011.03.023
Bøhn T, Amundsen PA, Sparrow A. Competitive exclusion after invasion? Biol Invasions. 2008; 10(3):359–68. https://doi.org/10.1007/s10530-007-9135-8
Boyle KS, Horn MH. Comparison of feeding guild structure and ecomorphology of intertidal fish assemblages from central California and central Chile. Mar Ecol Prog Ser. 2006; 319:65–84. https://doi.org/10.3354/meps319065
Britton JR, Gozlan RE, Copp GH. Managing non-native fish in the environment. Fish Fish. 2011; 12(3):256–74. https://doi.org/10.1111/j.1467-2979.2010.00390.x
Chellappa S, Câmara MR, Chellappa NT, Beveridge MCM, Huntingford FA. Reproductive ecology of a Neotropical cichlid fish, Cichla monoculus (Osteichthyes: Cichlidae). Braz J Biol. 2003; 63(1):17–26. http://dx.doi.org/10.1590/S1519-69842003000100004
Cianciaruso MV, Silva IA, Batalha MA. Diversidades filogenética e funcional: novas abordagens para a Ecologia de comunidades. Biota Neotrop. 2009; 9(3):93–103. http://dx.doi.org/10.1590/S1676-06032009000300008
Clavero M, García-Berthou E. Invasive species are a leading cause of animal extinctions. Trends Ecol Evol. 2005; 20(3):110. https://doi.org/10.1016/j.tree.2005.01.003
Coggins LG, Jr., Yard MD, Pine WE. Nonnative fish control in the Colorado River in Grand Canyon, Arizona: an effective program or serendipitous timing? TransAm Fish Soc. 2011; 140(2):456–70. https://doi.org/10.1080/00028487.2011.572009
Dolbeth M, Vendel AL, Pessanha A, Patrício J. Functional diversity of fish communities in two tropical estuaries subjected to anthropogenic disturbance. Mar Pollut Bull. 2016; 112(1–2):244–54. https://doi.org/10.1016/j.marpolbul.2016.08.011
Dumay O, Tari PS, Tomasini JA, Mouillot D. Functional groups of lagoon fish species in Languedoc Roussillon, southern France. J Fish Biol. 2004; 64(4):970–83. https://doi.org/10.1111/j.1095-8649.2004.00365.x
Ernst R, Linsenmair KE, Rödel MO. Diversity erosion beyond the species level: dramatic loss of functional diversity after selective logging in two tropical amphibian communities. Biol Conserv. 2006; 133(2):143–55. https://doi.org/10.1016/j.biocon.2006.05.028
Flecker AS. Ecosystem engineering by a dominant detritivore in a diverse tropical stream. Ecology. 1996; 77(6):1845–54. https://doi.org/10.2307/2265788
Fragoso-Moura EN, Oporto LT, Maia-Barbosa PM, Barbosa FA. Loss of biodiversity in a conservation unit of the Brazilian Atlantic Forest: the effect of introducing non-native fish species. Braz J Biol. 2016; 76(1):18–27. https://doi.org/10.1590/1519-6984.07914
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2021. https://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Fulton C, Bellwood D, Wainwright P. The relationship between swimming ability and habitat use in wrasses (Labridae). Mar Biol. 2001; 139(1):25–33.
Gatz AJ, Jr. Community organization in fishes as indicated by morphological features. Ecology. 1979; 60(4):711–18. https://doi.org/10.2307/1936608
Gilhuis JP. Vegetation survey of the Parque Florestal Estadual do Rio Doce, MG, Brasil. [Master Dissertation]. Viçosa: Universidade Federal de Viçosa; 1986.
Godinho AL. Peixes do Parque Estadual do Rio Doce. Belo Horizonte: Instituto Estadual de Florestas/ Universidade Federal de Minas Gerais; 1996.
Godinho AL, Fonseca MT, Araújo LM. The ecology of predator fish introductions: the case of Rio Doce Valley Lakes. In: Pinto-Coelho RM, Giani A, von Sperling M, editors. Ecology and human impact on lakes and reservoirs in Minas Gerais with special reference to future development and management strategies. Belo Horizonte: SEGRAC; 1994. p.77–83.
Godinho AL, Vieira F. Ictiofauna. In: Costa CMR, Hermann G, Martins CS, editors. Biodiversidade em Minas Gerais: Um atlas para sua conservação. Belo Horizonte: Fundação Biodiversitas; 1998. p.44–46.
Jackson DA, Peres-Neto PR, Olden JD. What controls who is where in freshwater fish communities – the roles of biotic, abiotic, and spatial factors. Can J Fish Aquat Sci. 2001; 58(1):157–70. https://doi.org/10.1139/f00-239
Kaufman L. Catastrophic change in species-rich freshwater ecosystems. BioScience. 1992; 42(11):846–58. https://doi.org/10.2307/1312084
Keenleyside MH. Diversity and adaptation in fish behaviour. Berlin: Springer; 1979.
Latini AO, Lima-Junior DP, Giacomini HC, Latini RO, Resende DC, Espírito-Santo HMV, Barros DF, Pereira TL. Alien fishes in lakes of the Doce river basin (Brazil): range, new occurrences and conservation of native communities. Lundiana. 2004; 5(2):135–42. Available from: http://hdl.handle.net/11449/67794
Latini AO, Petrere M, Jr. Reduction of a native fish fauna by alien species: an example from Brazilian freshwater tropical lakes. Fish Manag Ecol. 2004; 11(2):71–79. https://doi.org/10.1046/j.1365-2400.2003.00372.x
Leitão RP, Zuanon J, Mouillot D, Leal CG, Hughes RM, Kaufmann PR, Villéger S, Pompeu PS, Kasper D, Paula FR, Ferraz SF, Gardner TA. Disentangling the pathways of land use impacts on the functional structure of fish assemblages in Amazon streams. Ecography. 2018; 41:219–32. https://doi.org/10.1111/ecog.02845
Leitão RP, Zuanon J, Villéger S, Williams SE, Baraloto C, Fortunel C, Mendonça FP, Mouillot D. Rare species contribute disproportionately to the functional structure of species assemblages. Proc R Soc Lond B Biol Sci. 2016; 283(1828):20160084. https://doi.org/10.1098/rspb.2016.0084
Lima FP, Latini AO, Marco Júnior PD. How are the lakes? Environmental perception by fishermen and alien fish dispersal in Brazilian tropical lakes. Interciencia. 2010; 35(2):84–91. Available from: https://repositorio.bc.ufg.br/handle/ri/12092
Lockwood JL, Hoopes MF, Marchetti MP. Invasion ecology. Oxford: Blackwell Publishing; 2013.
Magalhães AL, Daga VS, Bezerra LA, Vitule JR, Jacobi CM, Silva LG. All the colors of the world: biotic homogenization-differentiation dynamics of freshwater fish communities on demand of the Brazilian aquarium trade. Hydrobiologia. 2020; 847:3897–915. https://doi.org/10.1007/s10750-020-04307-w
Magalhães ALB, Azevedo-Santos VM, Pelicice FM. Caught in the act: Youtube™ reveals invisible fish invasion pathways in Brazil. J Appl Ichthyol. 2021; 37:125–28. https://doi.org/10.1111/jai.14159
Magurran AE. Measuring Biological Diversity. Oxford: Blackwell Publishing; 2004.
Maia-Barbosa PM, Barbosa LG, Brito SL, Garcia F, Barros CF, Souza MB, Mello N, Guimarães AS, Barbosa FA. Limnological changes in Dom Helvécio Lake (South-East Brazil): natural and anthropogenic causes. Braz J Biol. 2010; 70(Suppl. 3):795–802. http://dx.doi.org/10.1590/S1519-69842010000400010
Maillard P, Pivari MO, Luis CHP. Remote sensing for mapping and monitoring wetlands and small lakes in Southeast Brazil. In: Chemin Y, editor. Remote sensing of planet Earth. Rijeka: InTech; 2012. p.23–46.
Maire E, Grenouillet G, Brosse S, Villéger S. How many dimensions are needed to accurately assess functional diversity? A pragmatic approach for assessing the quality of functional spaces. Glob Ecol Biogeogr. 2015; 24(6):728–40. https://doi.org/10.1111/geb.12299
Mikhailova I, Barbosa F. Estimativa preliminar do valor de recursos ambientais do Parque Estadual do Rio Doce (MG): uma aplicação dos métodos “Disposição a Pagar”. Belo Horizonte: Anais do V Simpósio Nacional sobre Recuperação de Áreas Degradadas; 2002. p.11–16.
Millennium Ecosystem Assessment. Ecosystems and human well-being: synthesis. Washington, DC: Island Press; 2005. Available from: http://www.millenniumassessment.org/documents/document.356.aspx.pdf
Mouillot D, Graham NA, Villéger S, Mason NW, Bellwood DR. A functional approach reveals community responses to disturbances. Trends Ecol Evol. 2013; 28(3):167–77. https://doi.org/10.1016/j.tree.2012.10.004
Moyle PB, Light T. Biological invasions of fresh water: empirical rules and assembly theory. Biol Conserv. 1996. 78(1–2):149–61. https://doi.org/10.1016/0006-3207(96)00024-9
Nico LG. Feeding chronology of juvenile piranhas, Pygocentrus notatus, in the Venezuelan llanos. Environ Biol Fish. 1990; 29:51–57. https://doi.org/10.1007/BF00000568
Olden JD. Biotic homogenization: a new research agenda for conservation biogeography. J Biogeogr. 2006; 33(12):2027–39. https://doi.org/10.1111/j.1365-2699.2006.01572.x
Oporto LT. Aspectos ecológicos e sociais da invasão de peixes em lagos naturais do médio Rio Doce, MG, Brasil. [PhD Thesis]. Belo Horizonte: Universidade Federal de Minas Gerais; 2013. Available from: http://pos.icb.ufmg.br/pgecologia/teses/T95_Lorena_Oporto.pdf
Ota RR, Deprá GD, Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: revised, annotated and updated. Neotrop Ichthyol. 2018; 16(2):e170094. https://doi.org/10.1590/1982-0224-20170094
Pauly D. Quantitative analysis of published data on the growth, metabolism, food consumption, and related features of the red-bellied piranha, Serrasalmus nattereri (Characidae). In: Balon EK, Bruton MN, Noakes DLG, editors. Women in ichthyology: an anthology in honor of ET, Ro and Genie. Dordrecht: Springer; 1994. p.423–37. https://doi.org/10.1007/978-94-011-0199-8_30
Pelicice FM, Azevedo‐Santos VM, Vitule JR, Orsi ML, Lima Junior DP, Magalhães AL, Pompeu PS, Petrere M, Jr., Agostinho AA. Neotropical freshwater fishes imperilled by unsustainable policies. Fish Fish. 2017; 18(6):1119–33. https://doi.org/10.1111/faf.12228
Pelicice FM, Latini JD, Agostinho AA. Fish fauna disassembly after introduction of a voracious predator: main drivers and the role of the invaders demography. Hydrobiologia. 2015; 746:271–83. https://doi.org/10.1007/s10750-014-1911-8
Pereira LS, Agostinho AA, Gomes LC. Eating the competitor: a mechanism of invasion. Hydrobiologia. 2015; 746:223–31. https://doi.org/10.1007/s10750-014-2031-1
Petchey OL, Gaston KJ. Functional diversity: back to basics and looking forward. Ecol Lett. 2006; 9(6):741–58. https://doi.org/10.1111/j.1461-0248.2006.00924.x
Pinto-Coelho RM, Bezerra-Neto JF, Miranda F, Mota TG, Resck R, Santos AM, Maia-Barbosa PM, Mello NA, Marques MM, Campos MO, Barbosa FA. The inverted trophic cascade in tropical plankton communities: impacts of exotic fish in the Middle Rio Doce lake district, Minas Gerais, Brazil. Braz J Biol. 2008; 68(suppl. 4):1025–37. https://doi.org/10.1590/S1519-69842008000500010
Pompeu PS, Godinho AL. Mudança na dieta da traíra Hoplias malabaricus (Bloch) (Erythrinidae, Characiformes) em lagoas da bacia do rio Doce devido à introdução de peixes piscívoros. Rev Bras Zool. 2001; 18(4):1219–25. https://doi.org/10.1590/S0101-81752001000400016
R Core Team. R: A language and environment for statistical computing [Internet]. 2020; Available from: http://www.R-project.org
Sala OE, Chapin FS, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH. Global biodiversity scenarios for the year 2100. Science. 2000; 287(5459):1770–74. http://dx.doi.org/10.1126/science.287.5459.1770
Santos AFGN, Santos LN, Araújo F. Digestive tract morphology of the Neotropical piscivorous fish Cichla kelberi (Perciformes: Cichlidae) introduced into an oligotrophic Brazilian reservoir. Rev Biol Trop. 2011; 59(3):1245–55. Available from: http://www.scielo.sa.cr/scielo.php?script=sci_arttext&pid=S0034-77442011000300025&lng=en&nrm=iso
Sazima I, Caramaschi EP. Comportamento alimentar de duas espécies de Curimata, sintópicas no Pantanal de Mato Grosso (Osteichthyes, Characiformes). Rev Bras Biol. 1989; 49(2):325–33.
Sazima I, Machado FA. Underwater observations of piranhas in western Brazil. In: Bruton MN, editor. Alternative life-history styles of fishes. Dordrecht: Springer; 1990. p.17–31. https://doi.org/10.1007/978-94-009-2065-1_2
Sibbing FA, Nagelkerke LA. Resource partitioning by Lake Tana barbs predicted from fish morphometrics and prey characteristics. Rev Fish Biol Fisher. 2000; 10(4):393–437. https://doi.org/10.1023/A:1012270422092
Simberloff D. Invasional meltdown 6 years later: important phenomenon, unfortunate metaphor, or both? Ecol Lett. 2006; 9(8):912–19. https://doi.org/10.1111/j.1461-0248.2006.00939.x
Sunaga T, Verani JR. The fish communities of the lakes in Rio Doce Valley, Northeast Brazil. Verh Int Ver Theor Angew Limnol. 1991; 24(4):2563–66. https://doi.org/10.1080/03680770.1989.11900015
Taylor BW, Flecker AS, Hall RO, Jr. Loss of a harvested fish species disrupts carbon flow in a diverse tropical river. Science. 2006; 313(5788):833–36. https://doi.org/10.1126/science.1128223
Teresa FB, Rodrigues-Filho CAS, Leitão, RP. Diversidade funcional de comunidades de peixes de riacho. Oecol Aust. 2021; 25(2):416–33. https://doi.org/10.4257/oeco.2021.2502.12
Tundisi JG, de Meis MRM. Geomorphology and limnological processes at the Middle Rio Doce Valley lakes. In: Water Research Institute, Limnological Studies in Central Brazil (1st Report). Chikusa-ku: Nagoya University; 1985. p.11–17.
Vasconcellos MG, Assumpção AM, Soares AS, Lucca JV, Verani JR, Fenerich-Verani N, Rocha O. Análise da diversidade de espécies de peixes de 4 lagoas do Sistema de Lagos do Vale do Médio Rio Doce–MG, em relação à ocorrência de espécies exóticas. In: Rocha O, Espíndola ELG, Fenerich-Verani N, Verani JR, Rietzler AC, editors. Espécies invasoras em águas doces: estudos de caso e propostas de manejo. São Carlos: EDUFSCar; 2005. p.119–29.
Villéger S, Mason NW, Mouillot D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology. 2008; 89(8):2290–301. https://doi.org/10.1890/07-1206.1
Villéger S, Miranda JR, Hernández DF, Mouillot D. Contrasting changes in taxonomic vs. functional diversity of tropical fish communities after habitat degradation. Ecol Appl. 2010; 20(6):1512–22. https://doi.org/10.1890/09-1310.1
Vitule JR, Freire CA, Simberloff D. Introduction of non-native freshwater fish can certainly be bad. Fish Fish. 2009; 10(1):98–108. https://doi.org/10.1111/j.1467-2979.2008.00312.x
Webb PW. Form and function in fish swimming. Sci Am. 1984; 251(1):72–83. Available from: http://www.jstor.org/stable/24969414
Winemiller KO. Ecomorphological diversification in lowland freshwater fish assemblages from five biotic regions. Ecol Monogr. 1991; 61(4):343–65. https://doi.org/10.2307/2937046
Winemiller KO, Fitzgerald DB, Bower LM, Pianka ER. Functional traits, convergent evolution, and periodic tables of niches. Ecol lett. 2015; 18(8):737–51. https://doi.org/10.1111/ele.12462
Authors
![]() Carla Patrícia de Souza1,2,
Carla Patrícia de Souza1,2,![]() Carlos Alberto de Sousa Rodrigues-Filho1,2,
Carlos Alberto de Sousa Rodrigues-Filho1,2,![]() Francisco Antônio Rodrigues Barbosa3 and
Francisco Antônio Rodrigues Barbosa3 and![]() Rafael Pereira Leitão1
Rafael Pereira Leitão1
[1] Laboratório de Ecologia de Peixes (ECO-Peixes), Departamento de Genética, Ecologia e Evolução (DGGE), Instituto de Ciências Biológicas (ICB), Universidade Federal de Minas Gerais (UFMG), Av. Antônio Carlos, 6627, Pampulha, 31270-901 Belo Horizonte, MG, Brazil. (CPS) carlasouza.patricia@yahoo.com.br, (CASRF) carlosfilho918@gmail.com, (RPL) ecorafa@gmail.com (corresponding author).
[2] Programa de Pós-Graduação em Ecologia, Conservação e Manejo da Vida Silvestre (ECMVS), Universidade Federal de Minas Gerais (UFMG) Av. Antônio Carlos, 6627, Pampulha, 31270-901 Belo Horizonte, MG, Brazil.
[3] Laboratório de Limnologia, Ecotoxicologia e Ecologia Aquática (LIMNEA), Departamento de Genética, Ecologia e Evolução (DGGE), Instituto de Ciências Biológicas (ICB), Universidade Federal de Minas Gerais (UFMG), Av. Antônio Carlos, 6627, Pampulha, 31270-901 Belo Horizonte, MG, Brazil. barbosa.ufmg@gmail.com.
Authors Contribution 
Carla Patrícia de Souza: Data curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Carlos Alberto de Sousa Rodrigues-Filho: Data curation, Formal analysis, Investigation, Methodology, Writing-review and editing.
Francisco Antônio Rodrigues Barbosa: Funding acquisition, Investigation, Methodology, Project administration, Validation, Visualization, Writing-review and editing.
Rafael Pereira Leitão: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The authors declare no competing interests.
How to cite this article
Souza CP, Rodrigues-Filho CAS, Barbosa FAR, Leitão RP. Drastic reduction of the functional diversity of native ichthyofauna in a Neotropical lake following invasion by piscivorous fishes. Neotrop Ichthyol. 2021; 19(3):e210033. https://doi.org/10.1590/1982-0224-2021-0033
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted July 16, 2021 by Emilí Garcia-Berthou
Accepted July 16, 2021 by Emilí Garcia-Berthou
![]() Submitted January 25, 2021
Submitted January 25, 2021
![]() Epub Sept 17, 2021
Epub Sept 17, 2021