![]() Laís de C. T. Chaves1,5
Laís de C. T. Chaves1,5 ![]() ,
, ![]() João L. L. Feitosa2,5,
João L. L. Feitosa2,5, ![]() Túlio F. Xavier2,
Túlio F. Xavier2, ![]() Beatrice P. Ferreira3 and
Beatrice P. Ferreira3 and ![]() Carlos E. L. Ferreira4
Carlos E. L. Ferreira4
PDF: EN XML: EN | Cite this article
Abstract
Damselfishes are known keystone species of reef environments, however large-scale distribution patterns are poorly studied in the southwestern Atlantic. We evaluated main drivers of distribution of three conspicuous damselfishes, along tropical and subtropical coastal systems, in Brazil. Abundances were assessed against wave exposure, depth (within 1–7 m in tropical and 1–11 m in subtropical reefs) and benthic cover. Despite differences between systems, exposure and depth consistently explained damselfishes distribution. Stegastes fuscus, the larger damselfish species of the genus in the southwestern Atlantic, was dominant in both systems, inhabiting preferably shallow and sheltered reefs. Conversely, Stegastes variabilis occupied shallow habitats with higher exposure. Stegastes pictus was absent from tropical reefs sampled, inhabiting depths >7 m, in subtropical reefs. Species were weakly associated with benthic features, which poorly predicted changes in abundances. Regardless, S. fuscus showed association with articulated calcareous algae, and S. variabilis juveniles associated with erect macroalgae. Despite occurring in very distinctive reef systems, Brazilian damsels habitat requirements are consistent in both tropical and subtropical reefs. While highly persistent species, long term monitoring will inform us how they respond to pervasive global changes and human impacts along Brazilian reefs.
Keywords: Habitat partitioning, Pomacentridae, Reef fish, Stegastes, Territorial herbivores.
Os peixes donzela são espécies-chave em ambientes recifais, contudo seus padrões de distribuição em grande escala são pouco conhecidos no sudoeste do Atlântico. Avaliamos os principais descritores de distribuição para três espécies comuns em sistemas recifais tropicais e costões rochosos subtropicais costeiros no Brasil, em resposta a diferentes regimes de exposição a ondas, profundidade e cobertura bentônica. Apesar das grandes diferenças entre estes sistemas, a exposição das ondas e a profundidade consistentemente explicaram sua distribuição. Stegastes fuscus é numericamente dominante, sendo a maior espécie do gênero no Atlântico sudoeste, dominando recifes rasos e menos hidrodinâmicos, enquanto S. variabilis foi mais abundante em habitats com maior hidrodinamismo. Stegastes pictus foi ausente nos recifes rasos tropicais amostrados e, nos recifes rochosos subtropicais, foi mais abundante em profundidades acima de 7 m. Características do substrato influenciaram pouco a densidade, porem S. fuscus apresentou associação a algas calcárias articuladas, enquanto juvenis de S. variabilis se associaram a macroalgas eretas. Peixes donzelas são generalistas e persistentes, ocorrendo em ambientes distintos, e mesmo assim, regulados de forma especifica e consistente ao longo da costa. O monitoramento a longo prazo nos informará como elas responderão às constantes mudanças globais e impactos antrópicos nos recifes brasileiros.
Palavras-chave: Herbívoros territoriais, Partição de habitat, Peixes recifais, Pomacentridae, Stegastes.
Introduction
Damselfishes play a major functional role in shallow tropical to temperate reef communities (Ceccarelli, 2001). Numerically dominant in different reef zones and habitats (Ceccarelli et al., 2001; Ceccarelli, 2007; Eurich et al., 2018), they present high trophic versatility, from herbivores to invertivores, but many species are considered omnivores (Feitosa et al., 2012; Frédérich, Parmentier, 2016; Eurich et al., 2019). These relatively small fishes take part in multiple energy pathways within reef food webs (Cowan et al., 2016; Pratchett et al., 2016), while exhibiting high site fidelity (Gardiner, Jones, 2005). Multiple species are known to hold territories and by doing so, they enhance algal productivity and overall diversity of benthic algae and associated cryptofauna within large reef habitats (Ferreira et al., 1998; Cleveland, Montgomery, 2003; Hamilton, Dill, 2003; Irving, Witman, 2009; Casey et al., 2014).
Territories are generally presented with abundant turf algae forming a complex, nominally the epilithic algae matrix (EAM), a substrate rich in algae and associated invertebrates and microbiota (e.g., meiofauna, bacteria, detritus) (Ferreira et al., 1998; Ceccarelli, 2007; Casey et al., 2014; Hata, Ceccarelli, 2016). Not surprisingly, these species are among the most aggressive reef fishes (Fontoura et al., 2020), fiercely defending their territories, while affecting the foraging behavior patterns of other species (Francini-Filho et al., 2010). At the same time, the EAM is a major substrate used as primary food source for a diverse set of other reef fishes and invertebrates (Wilson, Bellwood, 1997; Wilson et al., 2003). The degree of habitat modification exerted is, however, highly variable. The level of aggressive behavior displayed, feeding pressure, territory size and weeding intensity may change within species, life phase and at different spatial scales (Hata et al., 2002, 2010; Hata, Kato, 2002, 2004; Ceccarelli, 2007; Feitosa et al., 2012).
As for other species with high site fidelity, habitat selection during recruitment of damselfishes is considered one of the most important driving forces for adult distribution (Munday et al., 1997; Gutiérrez, 1998). However, post-settlement processes (i.e., predation, competition, and migration) are likewise critical, affecting patterns of their distribution at different spatial and temporal scales (Jones, 1991; Carr, Hixon, 1995; Feitosa, Ferreira, 2015). The use of space in territorial reef fish can, therefore, be determined by synergistic factors, such as the availability of preferred microhabitats (Holbrook et al., 2000), abundance of predators (Hixon, Beets, 1993; Almany, 2004), intra- and interspecific competition (Sweatman, 1985; Tolimieri, 1998; Bay et al., 2001) and swimming limitations in high-energy environments (Denny, 2005).
Although damselfishes show clearly structured distribution patterns according to habitat characteristics, spatial distribution patterns of reef fishes can be highly variable on the scale of tens to thousands of meters (Williams, 1982; Meekan et al., 1995; Holbrook et al., 2000; Emslie et al., 2012). At local scales, the relationships of fish composition and abundance were correlated with depth gradients (Green, 1996; Richardson, 1999; Medeiros et al., 2010), exposure (Russ, 1984; Williams, 1991; Gust, 2002; Depczynski, Bellwood, 2005; Floeter et al., 2007) and live coral cover (Bouchon-Navaro, Bouchon, 1989). Large-scale studies have examined the major limiting factors for species distribution, such as temperature (Floeter et al., 2005) and habitat connectivity (Mora, Robertson, 2005). However, for species with extensive distribution ranges, such as several species of damselfishes, the influence of major drivers to their local abundance and distribution has been comparatively poorly investigated (but see Emslie et al., 2012, 2019).
The Brazilian coast extends over 8,000 km of the southwestern Atlantic plus oceanic islands, while reef fishes are distributed along a gradient from tropical to subtropical reefs (Pinheiro et al., 2018). Coral reefs dominate from north to central tropical coast, while subtropical rocky reefs are the main features southwards (Ferreira et al., 2004; Pinheiro et al., 2018). Although considered relatively poor in diversity when compared to the Caribbean and Pacific (Cooper et al., 2009; Tang et al., 2021), Stegastes species are one of the most conspicuous and abundant component of reef fish communities in Brazil (Ferreira et al., 2004; Floeter et al., 2005; Morais et al., 2017; Araújo et al., 2020). Three territorial damselfish species from the genus Stegastes occur along nearshore reefs (Ferreira et al., 2004), while other three endemic species inhabit the oceanic islands (Pinheiro et al., 2018).
Such wide latitudinal distribution and high abundance of damselfishes offer an opportunity to examine driving factors shaping habitat use and relationships between sympatric species along their geographical range in the southwestern Atlantic, both within and among regions of the Brazilian coast. So far, many macro-ecological studies have considered the Brazilian coast an ideal gradient model for reef fish (Ferreira et al., 2004; Floeter et al., 2005; Barneche et al., 2009; Liedke et al., 2016; Morais et al., 2017; Longo et al., 2019) and benthic communities (Aued et al., 2018), but although large patterns of damselfish demography along the coast had been demonstrated, no study has been done to compare abundance patterns at a reef zonation scale. It is critical to understand the dynamics of damselfish assemblages, since they act as habitat modifiers (Ferreira et al., 1998). They occur in high numbers, virtually on the entire Brazilian coast (Ferreira et al., 2004), thus being an important functional group in reef ecosystems.
From tropical to subtropical reefs along the southwestern Atlantic, different habitat features and demographic drivers are expected to influence the spatial distribution and abundance of site-attached fish species. We analyzed patterns of abundance and distribution of three conspicuous territorial damselfishes comparatively in shallow tropical (within depths of 1–7 m) and subtropical reefs (within 1–11 m) of the Brazilian coast. Specifically, we pose these questions: (i) what are the major determinants for Stegastes spp. distribution within different shallow reef habitats? (ii) Are there particular requirements for different life stages (i.e., juveniles vs. adults)? (iii) Are these factors consistent across spatial scales studied (e.g., systems, habitats)?
Material and methods
Study species. The dusky damselfish, Stegastes fuscus (Cuvier, 1830), the cocoa damselfish, Stegastes variabilis (Castelnau, 1855) and the yellowtip damselfish, Stegastes pictus (Castelnau, 1855) are endemic to and widely distributed in the Brazilian Province (Ferreira et al., 2004; Pinheiro et al., 2018; Araújo et al., 2020). The dusky damselfish is one of the most abundant fish on shallow Brazilian coastal reef systems (Ferreira et al., 1998; Menegatti et al., 2003; Osório et al., 2006). Its distribution ranges from Ceará (03°34’5 S 38°24’W), to Santa Catarina (27°36’S 48°23’W). The cocoa damselfish is less conspicuous, but also occurring all along the coast, from Parcel Manoel Luis (00°52’S 44°15’W) to Santa Catarina. The yellowtip damselfish occurs all along the coast and some of the oceanic islands (Pinheiro et al., 2018). Stegastes fuscus and S. variabilis are territorial farming herbivorous (Ferreira et al., 1998; Feitosa et al., 2012), whereas S. pictus is an invertivorous species, feeding either on benthos or on the water column when plankton is abundant (Floeter et al., 2007; authors’ pers. obs.).
Study area and sampling design. Sites studied comprised the tropical fringing reefs of Tamandaré in the northeastern coast of Brazil and the subtropical rocky reefs of Arraial do Cabo in the southeastern coast. These two locations are International Long-term Ecological Program Sites in Brazil: the ILTER Site 18 (PELD-TAMS) and the ILTER Site 22 (RECA) (Muelbert et al., 2019).
The tropical fringing reefs of Tamandaré in the State of Pernambuco (08º44’S 35º05’W) are located in the upper limit of the Marine Protected Area APA Costa dos Corais, which extends 135 km along the northeastern coast of Brazil. Coral reefs are composed of three main reef lines parallel to the coast (Maida, Ferreira, 1997). Stegastes territories are widespread along shallow water reef lines, occupying reef surfaces (up to 2 m deep), where benthic cover in their surroundings is mainly composed of thick articulated calcareous algae (Jania spp. and Halimeda opuntia), and other macroalgae (Feitosa et al., 2012). The zoanthid Palythoa caribaeorum and sparse colonies of the fire-coral Millepora alcicornis are also important components of benthic coverage. The area has a tropical climate with an established regime of rainy (May to September) and dry (October to May) seasons that reach minimum and maximum water temperatures of 26 ºC and 30 ºC, respectively (Maida, Ferreira, 1997).
The subtropical rocky reefs of Arraial do Cabo in the state of Rio de Janeiro (22°59’S 42°00’W) was established as a Marine Extractive Reserve (RESEX Mar Arraial do Cabo) since 1997, a type of sustainable use conservation unit. The rocky shores formed by granite boulders reach 20–30 m of extension from the surface to the interface with the sand flats, with maximum depths of 15 m during high tides. Stegastes territories have dense algal mats 1–3 cm high that are usually dominated by the articulated calcareous algae, Jania spp. and Amphiroa sp. Within territories, these algae alternate with colonies of the fire coral M. alcicornis and the zoanthid P. caribaeorum (Ferreira et al., 1998). The mean water temperature in the study sites is approximately 22 ºC year round, reaching an averaged maximum of 25 ºC (Ferreira et al., 2001) and a minimum of 18 ºC, indicating that this region is under the influence of coastal upwelling during the summer/spring periods (Valentin, 1984). The reefs studied, however, are sporadically affected by cold waters and typically only in the deeper portions (Ferreira et al., 1998).
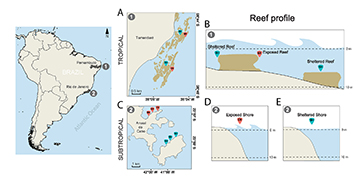
The distribution of the damselfishes S. fuscus, S. variabilis and S. pictus within systems were examined between December 2009 and February 2010, the summer season at both latitudes. Given the inherent structural differences of distinct reef formations, the sampling was adjusted to examine effects of exposure to wave surge in each habitat. In coral reefs of Tamandaré exposure was assessed in the two reef lines further away from the shoreline and at different sections. In the second line, the back reef was characterized as sheltered and the fore reef as exposed. In the third line, both back and fore reefs of deeper reefs (up to 7 m) were characterized as sheltered, due to lower wave surge effects observed. The selected reefs were large enough to separate second and third reef lines by at least 700 meters, and several small sites were sampled in two portions of the reef complex, 2 km apart from each other, allowing distance enough for site independence among samples (Figs. 1A,B). To assess exposure in Arraial do Cabo, two continuous rocky reefs were selected, subject to distinct intensities of wave surge, i.e., exposed and sheltered rocky shores, where three sites at least 700 meters apart from each other were determined (Figs. 1C–E). Also, each site comprised an area wide enough to ensure spatial independence among samples, observing the territorial nature of damselfishes studied (which holds territories of 1–2 m2, Ferreira et al., 1998; Osório et al., 2006; Medeiros et al., 2010). The sites chosen within each reef system presented the same orientation to the coast, so that levels of exposure to wave surge were comparable. Sampling was done at a continuous range of depth from 1 to 11 m, which represent depths where the majority of damselfish can be found.
FIGURE 1 | Map of studied tropical and subtropical reef systems in the Brazilian coast (South America) with respective reef profiles. 1. Fringing coral reefs (tropical system); 2. Rocky reefs (subtropical system). A. Overview of sites sampled in Tamandaré tropical reefs; B. Reef profiles indicating the positioning of sheltered and exposed tropical reefs; C. Overview of sites sampled in Arraial do Cabo subtropical reefs; D. Reef profile at exposed reefs; E. Reef profile at sheltered reefs. EX = Exposed sites; SH = Sheltered sites.
Fish surveys. Replicated 10 x 2 m belt transects were laid with an overall total of 180 transects (90 for each system). Samples were collected along a measuring tape laid on the reef surface, which was always placed at least 20 m away from another transect and sampled on the same day for each site and depth to avoid pseudo-replication. The visual census started after a 3 min period to allow fish to acclimate to the diver’s presence. To avoid edge effects, if an individual was not within a given transect, but at least 50% of its territory was inside the transect boundaries, the fish was still included in the counts. Fish were counted and grouped into life stages (juveniles and adults). While individual fish length was visually estimated, the conspicuous coloration of juveniles was the decisive factor in assigning fish into each life stage. It is important to note that length was slightly different for the studied reef systems (i.e., subtropical fish attain larger size ranges within stages; LCTC and collaborators, work in progress), as well as the distinct coloration patterns of adult S. variabilis between the two systems (Souza et al., 2011) (Fig. 2).
FIGURE 2 | Color patterns of damselfishes studied in their juvenile and adult life phases. A. Stegastes fuscus juvenile; B. S. fuscus adult; C. Stegastes variabilis juvenile; D. S. variabilis adult (in subtropical reefs); E. S. variabilis adult (in tropical reefs); F. Stegastes pictus juvenile; G. S. pictus adult. Image credits to A Bertoncini (A, C, D, F and G) and JLL Feitosa (B and E).
Benthic communities and reef complexity. In both reef systems, the benthic cover was estimated using 40 x 40 cm photoquadrats, taken at each 2 m of the transect used for fish censuses, distributed along the transect from 0 to 10 m (6 samples per transect; totaling 1080 photos). The images were later processed in CPCe 3.5 Software (Kohler, Gill, 2006), where 30 points were overlaid on each image, and organisms underneath were identified and classified in functional groups. The major functional groups defined were: massive corals (Siderastrea stellata, Montastraea cavernosa, Mussismilia hartii, Mussismilia hispida, Porites astreoides), branching corals (Millepora spp.), zoanthids (P. caribaeorum, Zoanthus spp.), erect macroalgae (mainly from the genera Sargassum, Caulerpa, Codium, Gracilaria, Dictyota, Dictyopteris), crustose calcareous algae, articulated calcareous algae (Jania spp., Amphiroa sp., H. opuntia), filamentous algae (Gelidium sp., Ceramium sp., Bryopsis sp.), urchins (Echinometra lucunter, Lytechinus variegatus), non-biotic substrate (sand, bare rock, rubble) and other organisms (sponges, ascidians, barnacles). Reef structural complexity was characterized by two metrics: number of holes per size class and rugosity. Quantity and diameters of holes were estimated on photoquadrats, measured in CPCe, and later categorized in size classes by maximum diameter (< 5 cm, 5–10 cm, and > 10 cm). Rugosity was measured in situ, using a modified chain-link method from Luckhurst, Luckhurst (1978), where a chain was laid along the entire transect line. The rugosity index was obtained from the relationship of the chain length divided by the length of the transect line (i.e., 10 m), which was used as the linear distance.
Data analyses. Species densities were compared between systems using the non-parametric Mann-Whitney tests, performed at each life stage (see Tab. S1). To characterize the diversity of microhabitats observed within both reef systems, variables associated with benthic cover and reef complexity were analyzed through a Principal Components Analysis, performed after standardization of variables per sample. These variables were then reduced into groups (PC scores), and later included in the predictive models for distribution of damselfishes, as described below. This analysis was performed in Primer-e 6 software (Clarke, Warwick, 2001).
To determine the most important factors describing distribution in damselfishes, a generalized linear mixed model (GLMM) was performed for each species and life phase, considered separately for each reef system (Tabs. S2). As S. pictus was only detected in one system and did not have enough individuals to enable analysis by separate life stages, data on juvenile and adult densities were pooled and nine models were fitted. We explored the data for each species and determined that a negative binomial distribution was the most adequate for most of the damselfish species, except for S. fuscus adults, which in both reef systems showed a better fit with Gaussian distribution. Five factors were used for fitting the model: exposure (considered as a categorical predictor, with exposed and sheltered sites as levels), depth (continuous variable), and the PC Axes (1, 2 and 3), obtained in the aforementioned PCA analysis, which represented proxies for microhabitat features. Such an approach allowed condensing 14 microhabitat parameters analyzed into three, avoiding oversaturated models, while considering the effects of the major differences in microhabitat. Prior to model fitting, these variables were tested for collinearity using a logistic regression model with all variables. We also examined the variance inflation factor (VIF) for each variable, considering VIF > 2 as a threshold to determine collinearity, following Graham (2003), and all variables were independent. GLMMs were computed using the ‘glmmadmb’ function of the ‘glmmADMB’ package available in R (Skaug et al., 2016). This analysis accounts for zero-inflation, an attribute observed for damselfish data. Spatial variation between samples under the same exposure regime (within exposed or within sheltered sites) was considered as a random effect variable. The backward stepwise removal of non-significant terms from the full model, based on log-likelihood ratio tests was applied for model selection (Zuur et al., 2009), also considering models with lowest value of Akaike information criterion (AIC) (Burnham, Anderson, 2004) (see Tabs. S3). All data in the manuscript can be made available upon request to the corresponding author.
Results
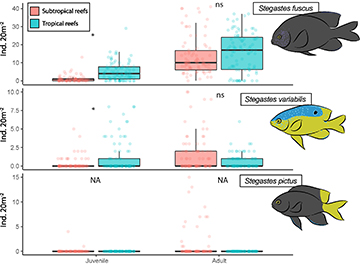
More than 3500 damselfish were counted during this study: ~1500 individuals were observed in subtropical reefs and ~2000 in tropical coral reefs. Stegastes fuscus was the most abundant species in both systems, corresponding to ~88% of total damselfish abundance (73% adults, 15% juveniles), followed by S. variabilis, which accounted for 9% of the fish counted (5% adults, 4% juveniles). Slightly less than a hundred individuals of Stegastes pictus were observed (~3% of the total damselfish abundance), all of them recorded in subtropical reefs. No Stegastes pictus were counted in most censuses, but at some sheltered subtropical sites they reached densities of up to 15 individuals/20 m², indicating a specificity of their distribution to some sites only (Fig. 3). A few settlers were occasionally observed outside of our sampling area in tropical reefs (n = 2), indicating that this species has no established populations on the studied shallow reefs of Tamandaré.
FIGURE 3 | Densities of the three damselfish (adults and juveniles) species between tropical and subtropical reefs of the Brazilian coast. Box-plots show the median (line) and quartiles distributions. *Significant differences in Mann-Whitney tests; ns = non-significant differences; NA = no tests were applicable.
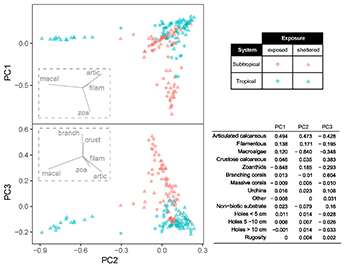
In both tropical and subtropical systems, benthic cover was a major feature for sample segregation and evidence of habitat selection by damselfish at the local scale. The first three PCA axes explained together 84.5% of variation (PC 1 = 38.7%, PC 2 = 27.8% and PC 3 = 18.0%). The main factors responsible for variation between samples in PC 1 were zoanthids (negatively) and articulated calcareous algae; in PC 2 were macroalgae (negatively) and articulated calcareous algae and; in PC 3 branching corals, articulated calcareous algae (negatively) and crustose calcareous algae were the most distinctive features (Fig. 4). Despite the fact that mean rugosity in tropical reefs had higher values than in subtropical reefs (mean ± standard deviation: 1.62 ± 0.2 vs. 1.56 ± 0.2), and higher frequency of holes of different sizes (5.26 ± 2.8 vs. 1.58 ± 1.9), complexity predictors had a minor contribution for explaining the variation among samples. In general, most of the samples were distributed around articulated calcareous algae dominance in both tropical and subtropical reefs. Samples dominated by macroalgae were almost exclusively observed in the tropical system, whereas higher coverage of zoanthids and branching corals/crustose calcareous algae were more frequent in subtropical reefs (Fig. 4).
FIGURE 4 | Principal Component Analysis of benthic cover and habitat predictors. For viewing purposes, vectors for variables with low scores (< 0.100 on axis displayed on each plot) were omitted from graphs. Variable contribution to each PC axis is presented on the table. Macroalgae (macal), articulated calcareous algae (artic), filamentous algae (filam) zoanthids (zoa), branching corals (branch), crustose calcareous algae (crust).
The selected predictors of damselfishes abundances are summarized in Figure 5. Adults of S. fuscus in both systems were highly associated with habitats dominated by articulated calcareous algae (positive relationship with PC2), the dominant species within turfs, but negatively associated with erect macroalgae-dominated habitats. Stegastes fuscus also showed some association with zoanthid-dominated habitats (according to the negative relationship with PC1, in the subtropical system only), which occur at the same depths as turf algae. Similarly, adults of S. variabilis had a negative relationship with PC1, indicating a weak association with articulated calcareous algae and/or a preference for zoanthid-dominated habitats. Conversely, juveniles were associated with habitats dominated by macroalgae, and were the only group with a negative relationship with PC2. Although PC3 contributed to almost 20% of sample variation, none of the species/life phase showed any relationship with the variables pertaining to this axis, so branching corals and crustose algae seem to have little contribution to damselfish distribution in both systems. These benthic groups, although often nearby, are rarely within territorial boundaries of studied damselfishes.
When compared to wave exposure and depth, benthic cover had a lesser explanation power and contributed to finer-scale local changes in damselfish distribution (Fig. 5). Wave exposure was the variable that most contributed to explain damselfish abundances in both systems, composing the models for all species but S. fuscus juveniles in the subtropical region. This life phase of S. fuscus had a distribution that was less consistently predicted by the variables considered herein. Depth was also a significant variable in determining damselfish distribution, and it was selected among five of the nine models fitted (Fig. 5). Although not a good predictor for S. fuscus abundance, in subtropical reefs, S. variabilis — regardless of the life phase — inhabited shallower areas, whereas S. pictus was almost exclusively found in deeper waters (mainly > 7 m). In the tropical system, the juveniles of S. fuscus and adult S. variabilis were exclusively observed in shallower habitats. Stegastes fuscus was more abundant on sheltered sites and this pattern was particularly evident for the subtropical system, where a two to three-fold greater numbers were observed. Adult S. variabilis, although observed in much lower densities, had comparatively higher densities in the subtropical reefs and in more exposed sites (Fig. 5).
FIGURE 5 | Predictors of damselfish densities on subtropical and tropical reefs. GLM coefficients are standardized for the selected factors. PC1–3 are axes extracted from Principal Component Analysis (Fig. 4), representing benthic cover and reef complexity characteristics. Values for non-significant predictors are presented in gray. Error bars denote standard error of coefficients.
Discussion
This study investigated the distribution patterns of territorial damselfish assemblages, as well as the associated benthic communities comparatively in tropical and subtropical reef systems approximately 2,200 km apart along the Brazilian coast. These systems are very distinct due to their origin (non-biogenic and biogenic), morphology (rocky reefs and fringing reefs), and latitude (subtropical and tropical), and yet, patterns of distribution of damselfishes inhabiting both systems show similar responses to environmental drivers. Exposure to wave surge and depth were the most important predictors influencing patterns of abundance of these species within both systems. Reef complexity, although important to explain general patterns of richness and abundance in reef fish assemblages (McCormick, 1994; Chabanet et al., 1997; Friedlander, Parrish, 1998; Medeiros et al., 2011), did not play a major role influencing the distribution of damselfishes at the scales of reefs analyzed. Benthic cover accounted only for fine-scale changes in local abundance of different life phases for each species.
Stegastes fuscus was by far the most abundant territorial damselfish in all habitats analyzed regardless of the environmental conditions present. Previous studies along the Brazilian coast corroborate such dominance (Ferreira et al., 2004; Floeter et al., 2005, Longo et al., 2014). Stegastes variabilis and S. pictus occurred at much lower densities than S. fuscus. Stegastes pictus was not detected in shallow tropical habitats studied, where reefs closer to the coastline occur continuously for more than 2 km (Figs. 1A,B). Stegastes pictus is known to occur more frequently in deeper reefs (18–25 m), far from the shore (Floeter et al., 2007; Soares et al., 2018). In contrast, the subtropical rocky shores, with relatively short and steep/vertical relief from shallow to deeper rocky-sandy interface habitats (ca. 10 m deep and average 25 m in length), provided substrate for a well established population of S. pictus (Ferreira et al., 2001).
Exposure to wave surge was a reliable explanatory variable in our models, as previously reported to influence abundance patterns of pomacentrids in both the southeastern (Floeter et al., 2007) and northeastern Brazilian coast (Medeiros et al., 2010). In subtropical reefs, sheltered habitats sustained higher densities of adult S. fuscus, in comparison to adult S. variabilis densities, which is more abundant in exposed sites. Such an effect was detected in the tropical system, but at a much lesser extent, as the exposure to wave surge is attenuated by the barrier reef system. Swimming performance limitations are known to influence the majority of the ecological activities of coral reef fishes including settlement, foraging and reproduction from relatively small to large spatial scales (Kawamata, 1998; Fulton et al., 2001; Fisher, Bellwood, 2003; Blake, 2004; Denny, 2005; Fisher, 2005; Fulton, Bellwood, 2004, 2005). Many studies on reef fish ecomorphology had shown overall distribution patterns explained as a result of adaptations to distinct swimming and feeding modes (Fulton et al., 2001; Bellwood et al., 2002; Johansen, Jones, 2011). For instance, S. fuscus and S. variabilis are both herbivorous species, and very similar in morphology (Araújo et al., 2003; Medeiros et al., 2010; Feitosa et al., 2012). By contrast, S. pictus is smaller than its herbivorous counterparts, with a more pronounced forked caudal fin, which allows it to explore both benthic substrate and the water column, feeding upon both benthic and planktonic invertebrates (Floeter et al., 2007). This morphological adaptation and feeding plasticity also allow this small damselfish to inhabit a diversity of deeper and offshore habitats where water flow and currents provide rich sources of plankton (Pinheiro et al., 2018).
In addition to exposure, depth was also a good predictor of abundance of these damselfishes. As depth increases, environmental conditions, such as light intensity and water motion decline, affecting algal photosynthetic rate and nutrient uptake (Hay, 1981). Following this trend, S. fuscus and S. variabilis, thrive in shallow habitats where primary production peaks and diversity and biomass of preferred filamentous algae is augmented (Ferreira et al., 1998). Their territorial and feeding behaviors increase the likelihood of competition between them. However, S. fuscus is relatively large and more aggressive (Medeiros et al., 2010), hence its dominance.
Benthic communities were variable overall, but seemed to be more stable in sheltered sites, which may be associated with S. fuscus preference for these habitats. It is worth noting that dominant taxa in benthic communities often changed in distribution at the same scales as the damselfish studied here (i.e., latitude, wave exposure and depth), as had been previously described elsewhere (Connolly et al., 2003; Adjeroud et al., 2007; Bongaerts et al., 2013) and more recently, in the Brazilian coast (Aued et al., 2018). The major difference in benthic communities when comparing the more exposed reefs of both systems was the large contribution of macroalgae in the tropics and zoanthids and branching corals (milleporids) in the subtropics. It is generally accepted that the environmental gradients created by the interaction of exposure and depth are linked to both physical (temperature, light, currents) and biological (habitat and food availability) features, thus likely influencing the settlement and survival of damselfishes and benthic organisms (Meekan et al., 2003; Bergenius et al., 2005; McCormick, Hoey, 2006; Sponaugle et al., 2006).
More often than not, the heterogeneity created by such environmental gradients allow multiple species to coexist (Fulton, Bellwood, 2005; Brokovich et al., 2008; Jankowski et al., 2015). In a more diverse community of cohabiting damselfishes, species may develop a high degree of substratum specificity (Waldner, Robertson, 1980; Robertson 1984; Precht et al., 2010; Chaves et al., 2012). For instance, live coral is a major driver of damselfish distribution in many reef systems (Munday et al., 2008; Wilson et al., 2008; Chaves et al., 2012; McCormick, 2012; Garcia-Herrera et al., 2017; MacDonald et al., 2018). In the systems studied herein, the less-diverse damselfish assemblages may lead to lower substrate specificity by species. This is largely supported by our results, where exposure and depth were the most prevalent predictors of damselfish abundance in both tropical and subtropical reef systems.
Additional evidence of this lower substrate specificity is the lack of association of younger individuals to the proxies used for reef structural complexity (i.e., frequency/size of holes and rugosity). It is safe to assume that early life stages are usually under higher mortality rates, along with a high degree of intraspecific competition with adults; a plausible explanation for low numbers of early stage individuals encountered. Damselfishes are known to spawn year-round (e.g., S. fuscus; Souza et al., 2007) and a high degree of self-recruitment is observed for this group (Jones et al., 1999; Christie et al., 2010; Berumen et al., 2012). Additionally, age studies have demonstrated a moderate lifespan for S. fuscus (15–17 years), with accumulation of several adult cohorts on the same reef (Schwamborn, Ferreira, 2002; LCTC and collaborators, work in progress). Not surprisingly, juveniles of all species (but S. variabilis in the tropics) somewhat shared similar habitat relationships with adults of their species. Nonetheless, high quality refuges, as a means of predator avoidance are still relevant for younger and/or smaller individuals (Hixon, Beets, 1993; Carr, Hixon, 1995; Figueira et al., 2008; Komyakova et al., 2013; Quadros et al., 2019).
As much as our results show congruence with previously reported drivers of damselfish distribution, Brazilian reefs hold particularities associated with a low diversity system. Size actually matters, as S. fuscus largely dominate damselfish assemblages in varied habitats, while other species attain higher densities in peripheral, less-preferred sites. This scenario clearly indicated how important interspecific competition shapes damselfish assemblages along the Brazilian coast, where the large species, S. fuscus dominates shallow and more productive habitats to establish territories. This pattern is prevalent all along the Brazilian coast from tropical to subtropical reefs (Ferreira et al., 2004; Pinheiro et al., 2018). Parcel de Manuel Luiz, a mid-shore reef in the north coast of Brazil, is the only reef system known where S. variabilis is the dominant damselfish (Cordeiro et al., 2021).
Turfs are the dominant component in tropical and subtropical reefs in the Brazilian Province (Aued et al., 2018). The synergistic effects of warming oceans, increasing coral bleaching, eutrophication and overfishing (Bellwood et al., 2004; Pratchett et al., 2011; Hughes et al., 2017; Hughes et al., 2018) are leading coral reefs towards phase-shifts, where turf algae prevail. In most reef systems of the Brazilian coast, turf communities are largely dominated by articulated calcareous algae (e.g., Jania spp., Amphiroa spp.) (Ferreira et al., 1998). These algae form a thick turf matrix, composed by the epilithic algal community. This matrix is very productive and provide primordial substrate to epiphytes (i.e., red filamentous and diatoms), which are comparatively more nutritious than algal communities in the surroundings, and comprise the bulk of S. fuscus and S. variabilis diets (Ferreira et al., 1998; Feitosa et al., 2012). A scenario of homogenization of benthic features can be detrimental to the functional diversity of reef communities, but in effect, may benefit damselfishes, such as S. fuscus. However, in the Mediterranean and Australia, “bad turfs” with high percentage of innutritious sediment had been indicated as an undesirable habitat condition for herbivorous fishes (Airoldi, 1998; Tebbett et al., 2018), even for these territorial fish exhibiting high tolerance to variable environmental conditions (Feitosa et al., 2012; Eurich et al., 2019).
The forecast scenarios of sea level rise and increased sedimentation on coral reefs (Morgan et al., 2020), as well as changes in oceanographic conditions such as prevalence of strong winds and more hydrodynamic environments (Saunders et al., 2014), may also directly affect the distribution of these species, and consequently, influence their role under adverse conditions. Only by continuously monitoring and conducting experiments, we will understand how these fishes will persist in shallow, ever-changing reef systems of the Brazilian coast and reefs worldwide.
Acknowledgments
Many thanks to the staff of Centro de Pesquisa e Extensão Pesqueira do Nordeste (CEPENE) in Tamandaré and ICMBio (RESEX Mar AC) for providing facilities and local support. Special thanks to Andreza Pacheco, Felipe Ribeiro, Renata Mazzei, Diego Medeiros, Simone Marques and Pedro Pereira for field support. I also thank Allan Souza, Francisco Barros, Paulo Santos, Caroline Feitosa, Liz McGinty for revision and contributions in earlier drafts of this manuscript. Photo credits in Fig. 2 (A, C, D, F and G) to Áthila Bertoncini through the projects Meros do Brasil and Ilhas do Rio. The financial support for this work was provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) as a PhD scholarship and PADI Project Aware awarded to LCTC during the period of study. CELF and BPF are continuously supported by a CNPq research grant.
References
Adjeroud M, Penin L, Carroll A. Spatio-temporal heterogeneity in coral recruitment around Moorea, French Polynesia: Implications for population maintenance. J Exp Mar Biol Ecol. 2007; 341(2):204–18. https://doi.org/10.1016/j.jembe.2006.10.048
Airoldi L. Roles of disturbance, sediment stress, and substratum retention on spatial dominance in algal turf. Ecology. 1998; 79(8):2759–70. https://doi.org/10.1890/0012-9658(1998)079[2759:RODSSA]2.0.CO;2
Almany GR. Differential effects of habitat complexity, predators and competitors on abundance of juvenile and adult coral reef fishes. Oecologia. 2004; 141:105–13. https://doi.org/10.1007/s00442-004-1617-0
Araújo ME, Mattos FMG, Melo FPL, Chaves LCT, Feitosa CV, Lippi DL et al. Diversity patterns of reef fish along the Brazilian tropical coast. Mar Environ Res. 2020; 160:105038. https://doi.org/10.1016/j.marenvres.2020.105038
Araújo ME, Paiva ACG, César FB, Silva JCC. A sutil diagnose morfológica entre as espécies simpátricas de Stegastes fuscus e S. variabilis (Actinopterygii: Pomacentridae). Arq Cienc Mar. 2003; 36:37–43. Available from: http://www.repositorio.ufc.br/bitstream/riufc/54221/1/2003_art_meara%c3%bajo.pdf
Aued AW, Smith F, Quimbayo JP, Cândido DV, Longo GO, Ferreira CEL et al. Large-scale patterns of benthic marine communities in the Brazilian Province. PLoS ONE. 2018; 13(6): e0198452. https://doi.org/10.1371/journal.pone.0198452
Barneche DR, Floeter SR, Ceccarelli DM, Frensel DMB, Dinslaken DF, Mário HFS, Ferreira CEL. Feeding macroecology of territorial damselfishes (Perciformes: Pomacentridae). Mar Biol. 2009; 156:289–99. https://doi.org/10.1007/s00227-008-1083-z
Bay LK, Jones GP, McCormick MI. Habitat selection and aggression as determinants of spatial segregation among damselfish on a coral reef. Coral Reefs. 2001; 20:289–98. https://doi.org/10.1007/s003380100173
Bellwood DR, Hughes TP, Folke C, Nyström M. Confronting the coral reef crisis. Nature. 2004; 429(6994):827–33. https://doi.org/10.1038/nature02691
Bellwood DR, Wainwright PC, Fulton CJ, Hoey A. Assembly rules and functional groups at global biogeographical scales. Funct Ecol. 2002; 16(5):557–62. https://doi.org/10.1046/j.1365-2435.2002.00655.x
Bergenius MAJ, McCormick MI, Meekan MG, Robertson DR. Environmental influences on larval duration, growth and magnitude of settlement of a coral reef fish. Mar Biol. 2005; 147:291–300. https://doi.org/10.1007/s00227-005-1575-z
Berumen ML, Almany GR, Planes S, Jones GP, Saenz-Agudelo P, Thorrold SR. Persistence of self-recruitment and patterns of larval connectivity in a marine protected area network. Ecol Evol. 2012; 2(2):444–52. https://doi.org/10.1002/ece3.208
Blake RW. Fish functional design and swimming performance. J Fish Biol. 2004; 65(5):1193–222. https://doi.org/10.1111/j.0022-1112.2004.00568.x
Bongaerts P, Frade PR, Ogier JJ, Hay KB, van Bleijswijk J, Englebert N et al. Sharing the slope: Depth partitioning of agariciid corals and associated Symbiodinium across shallow and mesophotic habitats (2-60 m) on a Caribbean reef. BMC Evol Biol. 2013; 13(1):205. https://doi.org/10.1186/1471-2148-13-205
Bouchon-Navaro Y, Bouchon C. Correlations between chaetodontid fishes and coral communities of the Gulf of Aqaba (Red Sea). In: Motta PJ, editor. The butterflyfishes: success on the coral reef. Dordrecht: Springer; 1989. p.47–60. (Developments in environmental biology of fishes; vol 9). https://doi.org/10.1007/978-94-009-2325-6_3
Brokovich E, Einbinder S, Shashar N, Kiflawi M, Kark S. Descending to the twilight-zone: changes in coral reef fish assemblages along a depth gradient down to 65 m. Mar Ecol Prog Ser. 2008; 371:253–62. https://doi.org/10.3354/meps07591
Burnham KP, Anderson DR. Multimodel inference: understanding AIC and BIC in model selection. Sociol Methods Res. 2004; 33(2):261–304. https://doi.org/10.1177%2F0049124104268644
Carr MH, Hixon MA. Predation effects on early post-settlement survivorship of coral- reef fishes. Mar Ecol Prog Ser. 1995; 124:31–42. https://doi.org/10.3354/meps124031
Casey JM, Ainsworth TD, Choat JH, Connolly SR. Farming behaviour of reef fishes increases the prevalence of coral disease associated microbes and black band disease. Proc R Soc B. 2014; 281:20141032. https://doi.org/10.1098/rspb.2014.1032
Ceccarelli DM, Jones GP, McCook LJ. Territorial damselfishes as determinants of the structure of benthic communities on coral reefs. Oceanogr Mar Biol. 2001; 39:355–89.
Ceccarelli DM. Modification of benthic communities by territorial damselfish: A multi-species comparison. Coral Reefs. 2007; 26:853–66. https://doi.org/10.1007/s00338-007-0275-1
Chabanet P, Ralambondrainy H, Amanieu M, Faure G, Galzin R. Relationships between coral reef substrata and fish. Coral Reefs. 1997; 16:93–102. https://doi.org/10.1007/s003380050063
Chaves LCT, Ormond CGA, McGinty ES, Ferreira BP. Space partitioning among damselfishes in the Caribbean coast of Panama: the role of habitat preferences. Neotrop Ichthyol. 2012; 10(3):633–42. http://dx.doi.org/10.1590/S1679-62252012000300017
Christie MR, Johnson DW, Stallings CD, Hixon MA. Self-recruitment and sweepstakes reproduction amid extensive gene flow in a coral-reef fish. Mol Ecol. 2010; 19(5):1042–57. https://doi.org/10.1111/j.1365-294X.2010.04524.x
Clarke K, Warwick R. Change in marine communities: an approach to statistical analysis and interpretation. Plymouth: PRIMER -E Ltd; 2001.
Cleveland A, Montgomery WL. Gut characteristics and assimilation efficiencies in two species of herbivorous damselfishes (Pomacentridae: Stegastes dorsopunicans and S. planifrons). Mar Biol. 2003; 142:35–44. https://doi.org/10.1007/s00227-002-0916-4
Connolly SR, Bellwood DR, Hughes TP. Indo-pacific biodiversity of coral reefs: deviations from a mid-domain model. Ecology. 2003; 84(8):2178–90. https://doi.org/10.1890/02-0254
Cooper WJ, Smith LL, Westneat MW. Exploring the radiation of a diverse reef fish family: phylogenetics of the damselfishes (Pomacentridae), with new classifications based on molecular analyses of all genera. Mol Phylogenet Evol. 2009; 52(1):1–16. https://doi.org/10.1016/j.ympev.2008.12.010
Cordeiro CAMM, Quimbayo JP, Nunes JACC, Nunes LT, Sissini MN, Sampaio CLS et al. Conservation status of the southernmost reef of the Amazon Reef System: the Parcel de Manuel Luís. Coral Reefs. 2021; 40:165–85. https://doi.org/10.1007/s00338-020-02026-1
Cowan ZL, Dworjanyn SA, Caballes CF, Pratchett MS. Predation on crown-of-thorns starfish larvae by damselfishes. Coral Reefs. 2016; 35(4):1253–62. https://doi.org/10.1007/s00338-016-1491-3
Denny CM. Distribution and abundance of labrids in northeastern New Zealand: the relationship between depth, exposure and pectoral fin aspect ratio. Environ Biol Fishes. 2005; 72:33–43. https://doi.org/10.1007/s10641-004-4178-5
Depczynski M, Bellwood DR. Wave energy and spatial variability in community structure of small cryptic coral reef fishes. Mar Ecol Prog Ser. 2005; 303:283–93. https://doi.org/10.3354/meps303283
Emslie MJ, Logan M, Ceccarelli DM, Cheal AJ, Hoey AS, Miller I et al. Regional-scale variation in the distribution and abundance of farming damselfishes on Australia’s Great Barrier Reef. Mar Biol. 2012; 159:1293–304. https://doi.org/10.1007/s00227-012-1910-0
Emslie MJ, Logan M, Cheal AJ. The distribution of planktivorous damselfishes (Pomacentridae) on the great barrier reef and the relative influences of habitat and predation. Diversity. 2019; 11(3):33. https://doi.org/10.3390/d11030033
Eurich JG, Matley JK, Baker R, McCormick MI, Jones GP. Stable isotope analysis reveals trophic diversity and partitioning in territorial damselfishes on a low-latitude coral reef. Mar Biol. 2019; 166:17. https://doi.org/10.1007/s00227-018-3463-3
Eurich JG, Shomaker SM, McCormick MI, Jones GP. Experimental evaluation of the effect of a territorial damselfish on foraging behaviour of roving herbivores on coral reefs. J Exp Mar Biol Ecol. 2018; 506:155–62. https://doi.org/10.1016/j.jembe.2018.06.009
Feitosa JLL, Concentino AM, Teixeira SF, Ferreira BP. Food resource use by two territorial damselfish (Pomacentridae: Stegastes) on Southwestern Atlantic algal-dominated reefs. J Sea Res. 2012; 70:42–49. https://doi.org/10.1016/j.seares.2012.03.006
Feitosa JLL, Ferreira BP. Distribution and feeding patterns of juvenile parrotfish on algal-dominated coral reefs. Mar Ecol. 2015; 36(3):462–74. https://doi.org/10.1111/maec.12154
Ferreira CEL, Floeter SR, Gasparini JL, Ferreira BP, Joyeux JC. Trophic structure patterns of Brazilian reef fishes: a latitudinal comparison. J Biogeogr. 2004; 31(7):1093–106. https://doi.org/10.1111/j.1365-2699.2004.01044.x
Ferreira CEL, Gonçalves JEA, Coutinho R, Peret AC. Herbivory by the dusky damselfish Stegastes fuscus (Cuvier, 1830) in a tropical rocky shore: Effects on the benthic community. J Exp Mar Biol Ecol. 1998; 229(2):241–64. https://doi.org/10.1016/S0022-0981(98)00056-2
Ferreira CEL, Goncçalves JEA, Coutinho R. Community structure of fishes and habitat complexity on a tropical rocky shore. Environ Biol Fishes. 2001; 61(4):353–69. https://doi.org/10.1023/A:1011609617330
Figueira WF, Lyman SJ, Crowder LB, Rilov G. Small-scale demographic variability of the bicolor damselfish, Stegastes partitus, in the Florida Keys USA. Environ Biol Fishes. 2008; 81:297–311. https://doi.org/10.1007/s10641-007-9201-1
Fisher R. Swimming speeds of larval coral reef fishes: impacts on self-recruitment and dispersal. Mar Ecol Prog Ser. 2005; 285:223–32. https://doi.org/10.3354/meps285223
Fisher R, Bellwood DR. Undisturbed swimming behaviour and nocturnal activity of coral reef fish larvae. Mar Ecol Prog Ser. 2003; 263:177–88. https://doi.org/10.3354/meps263177
Floeter SR, Behrens MD, Ferreira CEL, Paddack MJ, Horn MH. Geographical gradients of marine herbivorous fishes: Patterns and processes. Mar Biol. 2005; 147:1435–47. https://doi.org/10.1007/s00227-005-0027-0
Floeter SR, Krohling W, Gasparini JL, Ferreira CEL, Zalmon IR. Reef fish community structure on coastal islands of southeastern Brazil: the influence of exposure and benthic cover. Environ Biol Fishes. 2007; 78(2):147–60. https://doi.org/10.1007/s10641-006-9084-6
Fontoura L, Cantor M, Longo GO, Bender MG, Bonaldo RM, Floeter SR. The macroecology of reef fish agonistic behaviour. Ecography. 2020; 43(9):1278–90. https://doi.org/10.1111/ecog.05079
Francini Filho RB, Moura RL. Predation on the toxic zoanthid Palythoa caribaeorum by reef fishes in the Abrolhos Bank, eastern Brazil. Braz J Oceanogr. 2010; 58(1):77–79. https://doi.org/10.1590/S1679-87592010000100008
Frédérich B, Parmentier E. Biology of damselfishes. Liège: CRC Press; 2016.
Friedlander AM, Parrish JD. Habitat characteristics affecting fish assemblages on a Hawaiian coral reef. J Exp Mar Biol Ecol. 1998; 224(1):1–30. https://doi.org/10.1016/S0022-0981(97)00164-0
Fulton CJ, Bellwood DR, Wainwright PC. The relationship between swimming ability and habitat use in wrasses (Labridae). Mar Biol. 2001; 139:25–33. https://doi.org/10.1007/s002270100565
Fulton CJ, Bellwood DR. Wave exposure, swimming performance, and the structure of tropical and temperate reef fish assemblages. Mar Biol. 2004; 144:429–37. https://doi.org/10.1007/s00227-003-1216-3
Fulton CJ, Bellwood DR. Wave-induced water motion and the functional implications for coral reef fish assemblages. Limnol Oceanogr. 2005; 50(1):255–64. https://doi.org/10.4319/lo.2005.50.1.0255
Garcia-Herrera N, Ferse SCA, Kunzmann A, Genin A. Mutualistic damselfish induce higher photosynthetic rates in their host coral. J Exp Biol. 2017; 220(10):1803–11. https://doi.org/10.1242/jeb.152462
Gardiner NM, Jones GP. Habitat specialisation and overlap in a guild of coral reef cardinalfishes (Apogonidae). Mar Ecol Prog Ser. 2005; 305:163–75. https://doi.org/10.3354/meps305163
Graham MH. Confronting multicollinearity in ecological multiple regression. Ecology. 2003; 84(11):2809–15. https://doi.org/10.1890/02-3114
Green AL. Spatial, temporal and ontogenetic patterns of habitat use by coral reef fishes (Family Labridae). Mar Ecol Prog Ser. 1996; 133:1–11. https://doi.org/10.3354/meps133001
Gust N. Scarid biomass on the northern Great Barrier Reef: the influence of exposure, depth and substrata. Environ Biol Fishes. 2002; 64:353–66. https://doi.org/10.1023/A:1016112312009
Gutiérrez L. Habitat selection by recruits establishes local patterns of adult distribution in two species of damselfishes: Stegastes dorsopunicans and S. planifrons. Oecologia. 1998; 115:268–77. https://doi.org/10.1007/s004420050516
Hamilton IM, Dill LM. The use of territorial gardening versus kleptoparasitism by a subtropical reef fish (Kyphosus cornelii) is influenced by territory defendability. Behav Ecol. 2003; 14(4):561–68. https://doi.org/10.1093/beheco/arg023
Hata H, Ceccarelli DM. Farming behaviour of territorial damselfishes. In: Frédérich B, Parmentier E, editors. Biology of damselfishes. Liège: CRC Press; 2016. p.140–70.
Hata H, Kato M. Monoculture and mixed-species algal farms on a coral reef are maintained through intensive and extensive management by damselfishes. J Exp Mar Biol Ecol. 2004; 313(2):285–96. https://doi.org/10.1016/j.jembe.2004.08.009
Hata H, Kato M. Weeding by the herbivorous damselfish Stegastes nigricans in nearly monocultural algae farms. Mar Ecol Prog Ser. 2002; 237:227–31. https://doi.org/10.3354/meps237227
Hata H, Nishihira M, Kamura S. Effects of habitat-conditioning by the damselfish Stegastes nigricans (Lacepède) on the community structure of benthic algae. J Exp Mar Biol Ecol. 2002; 280(1–2):95–116. https://doi.org/10.1016/S0022-0981(02)00395-7
Hata H, Watanabe K, Kato M. Geographic variation in the damselfish-red alga cultivation mutualism in the Indo-West Pacific. BMC Evol Biol. 10(2010):185. https://doi.org/10.1186/1471-2148-10-185
Hay ME. The functional morphology of turf-forming seaweeds: persistence in stressful marine habitats. Ecology. 1981; 62(3):739–50. https://doi.org/10.2307/1937742
Hixon MA, Beets JP. Predation, prey refuges, and the structure of coral-reef fish assemblages. Ecol Monogr. 1993; 63(1):77–101. https://doi.org/10.2307/2937124
Holbrook SJ, Forrester GE, Schmitt RJ. Spatial patterns in abundance of a damselfish reflect availability of suitable habitat. Oecologia. 2000; 122:109–20. https://doi.org/10.1007/PL00008826
Hughes TP, Anderson KD, Connolly SR, Heron SF, Kerry JT, Lough JM et al. Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science. 2018; 359(6371):80–83. https://doi.org/10.1126/science.aan8048
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, et al. Global warming and recurrent mass bleaching of corals. Nature. 543(2017):373–77. https://doi.org/10.1038/nature21707
Irving AD, Witman JD. Positive effects of damselfish override negative effects of urchins to prevent an algal habitat switch. J Ecol. 2009; 97(2):337–47. https://doi.org/10.1111/j.1365-2745.2008.01467.x
Jankowski MW, Gardiner NR, Jones GP. Depth and reef profile: effects on the distribution and abundance of coral reef fishes. Environ Biol Fishes. 2015; 98:1373–86. https://doi.org/10.1007/s10641-014-0365-1
Johansen JL, Jones GP. Increasing ocean temperature reduces the metabolic performance and swimming ability of coral reef damselfishes. Glob Chang Biol. 2011; 17(9):2971–79. https://doi.org/10.1111/j.1365-2486.2011.02436.x
Jones GP. Post recruitment processes in the ecology of coral reef fish populations: A multifactorial perspective. In: Sale PF, editor. The ecology of fishes on coral reefs. Auckland: Academic Press, inc.; 1991. p.294–328
Jones GP, Millcich MJ, Emslie MJ, Lunow C. Self-recruitment in a coral fish population. Nature. 402(1999):802–04. https://doi.org/10.1038/45538
Kawamata S. Effect of wave-induced oscillatory flow on grazing by a subtidal sea urchin Strongylocentrotus nudus (A. Agassiz). J Exp Mar Biol Ecol. 1998; 224(1):31–48. https://doi.org/10.1016/S0022-0981(97)00165-2
Kohler KE, Gill SM. Coral point count with excel extensions (CPCe): A visual basic program for the determination of coral and substrate coverage using random point count methodology. Comput Geosci. 2006; 32(9):1259–69. https://doi.org/10.1016/j.cageo.2005.11.009
Komyakova V, Munday PL, Jones GP. Relative importance of coral cover, habitat complexity and diversity in determining the structure of reef fish communities. PLoS ONE. 2013; 8(12):e83178. https://doi.org/10.1371/journal.pone.0083178
Liedke AMR, Barneche DR, Ferreira CEL, Segal B, Nunes LT, Burigo AP et al. Abundance, diet, foraging and nutritional condition of the banded butterflyfish (Chaetodon striatus) along the western Atlantic. Mar Biol. 163(2016):06. https://doi.org/10.1007/s00227-015-2788-4
Longo GO, Ferreira CEL, Floeter SR. Herbivory drives large-scale spatial variation in reef fish trophic interactions. Ecol Evol. 2014; 4(23):4553–66. https://doi.org/10.1002/ece3.1310
Longo GO, Hay ME, Ferreira CEL, Floeter SR. Trophic interactions across 61 degrees of latitude in the Western Atlantic. Glob Ecol Biogeogr. 2019; 28(2):107–17. https://doi.org/10.1111/geb.12806
Luckhurst BE, Luckhurst K. Analysis of the influence of substrate variables on coral reef fish communities. Mar Biol. 49(1978):317–23. https://doi.org/10.1007/BF00455026
MacDonald C, Tauati MI, Jones GP. Depth patterns in microhabitat versatility and selectivity in coral reef damselfishes. Mar Biol. 165(2018):138. https://doi.org/10.1007/s00227-018-3396-x
Maida M, Ferreira BP. Coral reefs of Brazil: overview and field guide. Proceedings of the 8th International Coral Reef Symposium. 1997; 1:263–74.
McCormick MI. Comparison of field methods for measuring surface tomography and their associations with a tropical reef fish assemblage. Mar Ecol Prog Ser. 1994; 112:87–96. https://doi.org/10.3354/meps112087
McCormick MI. Lethal effects of habitat degradation on fishes through changing competitive advantage. Proc R Soc B. 2012; 279:3899–904. https://doi.org/10.1098/rspb.2012.0854
McCormick MI, Hoey AS. Biological and physical correlates of settlement and survival for a coral reef fish, Pomacentrus amboinensis (Pomacentridae).10th International Coral Reef Symposium. 2006; 425–30.
Medeiros PR, Rosa RS, Francini-Filho RB. Dynamics of fish assemblages on a continuous rocky reef and adjacent unconsolidated habitats at Fernando de Noronha archipelago, tropical western Atlantic. Neotrop Ichthyol. 2011; 9(4):869–79. https://doi.org/10.1590/S1679-62252011005000048
Medeiros PR, Souza AT, Ilarri MI. Habitat use and behavioural ecology of the juveniles of two sympatric damselfishes (Actinopterygii: Pomacentridae) in the south-western Atlantic Ocean. J Fish Biol. 2010; 77(7):1599–615. https://doi.org/10.1111/j.1095-8649.2010.02795.x
Meekan MG, Carleton JH, McKinnon AD, Flynn K, Furnas M. What determines the growth of tropical reef fish larvae in the plankton: food or temperature? Mar Ecol Prog Ser. 2003; 256:193–204. https://doi.org/10.3354/meps256193
Meekan MG, Steven ADL, Fortin MJ. Spatial patterns in the distribution of damselfishes on a fringing coral reef. Coral Reefs. 1995; 14:151–61. https://doi.org/10.1007/BF00367233
Menegatti JV, Vescovi DL. Interações agonísticas e forrageamento do peixe-donzela, Stegastes fuscus (Peciformes: Pomacentridae). Natureza online. 2003; 1:45–50. Available from: http://www.naturezaonline.com.br/natureza/conteudo/pdf/Revista_Online_Menegatti.pdf
Mora C, Robertson DR. Factors shaping the range-size frequency distribution of the endemic fish fauna of the Tropical Eastern Pacific. J Biogeogr. 2005; 32(2):277–86. https://doi.org/10.1111/j.1365-2699.2004.01155.x
Morais RA, Ferreira CEL, Floeter SR. Spatial patterns of fish standing biomass across Brazilian reefs. J Fish Biol. 2017; 91(6):1642–67. https://doi.org/10.1111/jfb.13482
Morgan KM, Perry CT, Arthur R, Williams HTP, Smithers SG. Projections of coral cover and habitat change on turbid reefs under future sea-level rise. Proc R Soc B. 2020; 287:20200541. https://doi.org/10.1098/rspb.2020.0541
Muelbert JH, Nidzieko NJ, Acosta ATR, Beaulieu SE, Bernardino AF, Boikova E et al. Ilter – the international long-term ecological research network as a platform for global coastal and ocean observation. Front Mar Sci. 2019; 6:1–14. https://doi.org/10.3389/fmars.2019.00527
Munday PL, Jones GP, Caley MJ. Habitat specialisation and the distribution and abundance of coral-dwelling gobies. Mar Ecol Prog Ser. 1997; 152:227–39. https://doi.org/10.3354/meps152227
Munday PL, Jones GP, Pratchett MS, Williams AJ. Climate change and the future for coral reef fishes. Fish Fish. 2008; 9(3):261–85. https://doi.org/10.1111/j.1467-2979.2008.00281.x
Munday PL. Habitat loss, resource specialization, and extinction on coral reefs. Glob Chang Biol. 2004; 10(10):1642–47. https://doi.org/10.1111/j.1365-2486.2004.00839.x
Osório R, Rosa IL, Cabral H. Territorial defence by the Brazilian damsel Stegastes fuscus (Teleostei: Pomacentridae). J Fish Biol. 2006; 69(1):233–42. https://doi.org/10.1111/j.1095-8649.2006.01095.x
Pinheiro HT, Rocha LA, Macieira RM, Carvalho-Filho A, Anderson AB, Bender MG, et al. South-western Atlantic reef fishes: Zoogeographical patterns and ecological drivers reveal a secondary biodiversity centre in the Atlantic Ocean. Divers Distrib. 2018; 24(7):951–65. https://doi.org/10.1111/ddi.12729
Pratchett MS, Bay LK, Gehrke PC, Koehn JD, Osborne K, Pressey RL et al. Contribution of climate change to degradation and loss of critical fish habitats in Australian marine and freshwater environments. Mar Freshw Res. 2011; 62(9):1062–81. https://doi.org/10.1071/MF10303
Pratchett MS, Hoey AS, Wilson SK, Hobbs JPA, Allen GR. Habitat-use and specialisation among coral reef damselfishes. In: Frédérich B, Parmentier E, editors. Biology of damselfishes. Liège: CRC Press; 2016. p.84–121.
Precht WF, Aronson RB, Moody RM, Kaufman L. Changing patterns of microhabitat utilization by the threespot damselfish, Stegastes planifrons, on caribbean reefs. PLoS ONE. 2010; 5(5):e10835. https://doi.org/10.1371/journal.pone.0010835
Quadros ALS, Barros F, Blumstein DT, Meira VH, Nunes JACC. Structural complexity but not territory sizes influence flight initiation distance in a damselfish. Mar Biol. 2019; 166:65. https://doi.org/10.1007/s00227-019-3508-2
Richardson DL. Correlates of environmental variables with patterns in the distribution and abundance of two anemonefishes (Pomacentridae: Amphiprion) on an Eastern Australian sub-tropical reef system. Environ Biol Fishes. 55(1999):255–63. https://doi.org/10.1023/A:1007596330476
Russ G. Distribution and abundance of herbivorous grazing fishes in the central Great Barrier Reef. I. Levels of variability across the entire continental shelf. Mar Ecol Prog Ser. 1984; 20:23–34. https://doi.org/10.3354/meps020023
Saunders MI, Leon JX, Callaghan DP, Roelfsema CM, Hamylton S, Brown CJ et al. Interdependency of tropical marine ecosystems in response to climate change. Nat Clim Change. 2014; 4:724–29. https://doi.org/10.1038/nclimate2274
Schwamborn SHL, Ferreira BP. Age structure and growth of the dusky damselfish. Environ Biol Fishes. 2002; 16:79–88. https://doi.org/10.1023/A:1013851532298
Skaug H, Fournier D, Nielsen A, Magnusson A, Bolker B. glmmADMB: generalized linear mixed models using AD Model Builder [Computer software manual – Internet], R package, version 0.8. 0.2016 Available from: http://glmmadmb.r-forge.r-project.org/
Soares MO, Davis M, Paiva CC, Carneiro PBM. Mesophotic ecosystems: coral and fish assemblages in a tropical marginal reef (northeastern Brazil). Mar Biodivers. 2018; 48:1631–36. https://doi.org/10.1007/s12526-016-0615-x
Souza AT, Ilarri MI, Medeiros PR, Sampaio CLS, Floeter SR. Unusual colour patterns of territorial damselfish (Pomacentridae: Stegastes) in the south-western Atlantic. Mar Biodivers Rec. 2011; 4:E101. https://doi.org/10.1017/S1755267211001035
Souza LLG, Chellappa S, Gurgel HCB. Biologia reprodutiva do peixe-donzela, Stegastes fuscus Cuvier, em arrecifes rochosos no nordeste do Brasil. Rev Bras Zool. 2007; 24(2):419–25. https://doi.org/10.1590/s0101-81752007000200020
Sponaugle S, Grorud-Colvert K, Pinkard D. Temperature-mediated variation in early life history traits and recruitment success of the coral reef fish Thalassoma bifasciatum in the Florida Keys. Mar Ecol Prog Ser. 2006; 308:1–15. https://doi.org/10.3354/meps308001
Sweatman HPA. The influence of adults of some coral reef fishes on larval recruitment. Ecol Monogr. 1985; 55(1):469–85. https://doi.org/10.2307/2937132
Tang KL, Stiassny ML, Mayden RL, DeSalle R. Systematics of Damselfishes. Ichthyol Herpetol. 2021; 109(1):258–318. https://doi.org/10.1643/i2020105
Tebbett SB, Bellwood DR, Purcell SW. Sediment addition drives declines in algal turf yield to herbivorous coral reef fishes: implications for reefs and reef fisheries. Coral Reefs. 2018; 37:929–37. https://doi.org/10.1007/s00338-018-1718-6
Tolimieri N. Contrasting effects of microhabitat use on large-scale adult abundance in two families of Caribbean reef fishes. Mar Ecol Prog Ser. 1998; 167:227–39. https://doi.org/10.3354/meps167227
Valentin JL. Analyse des paramètres hydrobiologiques dans la remontée de Cabo Frio (Brésil). Mar Biol. 82(1984):259–76. https://doi.org/10.1007/BF00392407
Waldner R, Robertson D. Patterns of habitat partitioning by eight species of territorial Caribbean damselfishes (Pisces: Pomacentridae). Bull Mar Sci. 1980; 30:171–86.
Williams DMCB. Patterns and processes in the distribution of coral reef fishes. In: Sale PF, editor. The ecology of fishes on coral reefs. Auckland: Academic Press, Inc.; 1991. p.437–74.
Williams DMCB. Patterns in the distribution of fish communities across the Central Great Barrier Reef. Coral Reefs. 1982; 1:35–43. https://doi.org/10.1007/BF00286538
Wilson S, Bellwood DR. Cryptic dietary components of territorial damselfishes (Pomacentridae, Labroidei). Mar Ecol Prog Ser. 1997; 153:299–310. https://doi.org/10.3354/meps153299
Wilson SK, Bellwood DR, Choat JH, Furnas MJ. Detritus in the epilithic algal matrix and its use by coral reef fishes. Oceanogr Mar Biol. 2003; 41:279–309.
Wilson SK, Burgess SC, Cheal AJ, Emslie M, Fisher R, Miller I et al. Habitat utilization by coral reef fish: implications for specialists vs. generalists in a changing environment. J Anim Ecol. 2008; 77(2):220–28. https://doi.org/10.1111/j.1365-2656.2007.01341.x
Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM. Mixed effects models and extensions in ecology with R. New York: Springer Science & Business Media; 2009.
Authors
![]() Laís de C. T. Chaves1,5
Laís de C. T. Chaves1,5 ![]() ,
, ![]() João L. L. Feitosa2,5,
João L. L. Feitosa2,5, ![]() Túlio F. Xavier2,
Túlio F. Xavier2, ![]() Beatrice P. Ferreira3 and
Beatrice P. Ferreira3 and ![]() Carlos E. L. Ferreira4
Carlos E. L. Ferreira4
[1] Marine Planning Program, Council of the Haida Nation, #1 Reservoir Road, V0T 1S1 Skidegate, Haida Gwaii, BC, Canada. (LCTC) lctchaves@gmail.com (corresponding author).
[2] Departamento de Zoologia, Universidade Federal de Pernambuco, Av. Professor Moraes Rego s/n, Cidade Universitária, 50670-420 Recife, PE, Brazil. (JLLF) feitosajll@gmail.com, (TFX) tuliofreire982@gmail.com.
[3] Departamento de Oceanografia. Universidade Federal de Pernambuco, Av. Arquitetura s/n, Cidade Universitária, 50670-901 Recife, PE, Brazil. (BPF) beatrice@ufpe.br.
[4] Departamento de Biologia Marinha, Universidade Federal Fluminense, Campus Valonguinho, 24001-970 Niterói, RJ, Brazil. (CELF) carlosferreira@id.uff.br.
[5] Tropical Conservation Consortium, 10413 Southwest 40th Avenue, 97219 Portland, OR, USA.
Authors Contribution 
Laís de Carvalho Teixeira Chaves: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Writing-original draft, Writing-review and editing.
João Lucas Leão Feitosa: Formal analysis, Methodology, Visualization, Writing-review and editing.
Túlio Freire Xavier: Writing-review and editing.
Beatrice Padovani Ferreira: Conceptualization, Funding acquisition, Methodology, Supervision, Writing-original draft, Writing-review and editing.
Carlos E. L. Ferreira: Conceptualization, Methodology, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The authors declare no competing interests.
How to cite this article
Chaves LCT, Feitosa JLL, Xavier TF, Ferreira BP, Ferreira CEL. Drivers of damselfishes distribution patterns in the southwestern Atlantic: tropical and subtropical reefs compared. Neotrop Ichthyol. 2021; 19(4):e210010. https://doi.org/10.1590/1982-0224-2021-0010
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted June 23, 2021 by Fernando Gibran
Accepted June 23, 2021 by Fernando Gibran
![]() Submitted January 11, 2021
Submitted January 11, 2021
![]() Epub December 10, 2021
Epub December 10, 2021