![]() Reginaldo Medeiros1
Reginaldo Medeiros1 ![]() ,
, ![]() Cicero D. Oliveira2,
Cicero D. Oliveira2, ![]() Daniele Souto1,
Daniele Souto1, ![]() Jordana Rangely1 and
Jordana Rangely1 and ![]() Nídia N. Fabré1
Nídia N. Fabré1
PDF: EN XML: EN | Cite this article
Abstract
Morphoanatomical or physiological changes coupled with changes in body size are known as allometric relationships. The objective of this study was to identify the points of growth changes in Centropomus based on otolith morphometry and morphogeometry. For this purpose, 455 individuals of C. undecimalis and 176 of C. parallelus were collected from artisanal fishermen of the coast of the state of Alagoas, Brazil. The sagittal otoliths were measured for length, height, perimeter, area and weighed. The potential and polyphasic models were fitted between total fish length and otolith length. The morphotypes otoliths wen describe by form Fourier descriptors and shape indices. The polyphasic model detected three growth phases. The first stanza for C. undecimalis was at 46.8 cm and the second at 75.9 cm. For C. parallelus, it was at 18.8 cm and at 41.2 cm. Each stanza has a specific otoliths morphotype in both species. The otoliths of C. undecimalis and C. parallelus exhibited ontogenetic allometric changes in their growth pattern with two stanzas changing points. The stanzas corresponded to specific lengths reached by individuals over their life cycles, such as their size at maturity and length at sexual reversion.
Keywords: Energy allocation, Hermaphroditism, Morphogeometry, Polyphasic growth, Reproduction.
Mudanças morfoanatômicas ou fisiológicas em conjunto com mudanças no tamanho do corpo, são conhecidas como relações alométricas. O objetivo do estudo foi identificar os pontos de mudanças de crescimento de Centropomus com base na morfometria e morfogeometria dos otólitos. Foram coletados 455 indivíduos de C. undecimalis e 176 de C. parallelus, adquiridos com pescadores artesanais do estado de Alagoas, Brasil. Os otólitos sagitta foram medidos quanto ao comprimento, altura, perímetro, área e pesados. Os modelos potencial e polifásico foram ajustados entre o comprimento do peixe e o comprimento do otólito. Os morfotipos de otólitos são descritos por descritores de Fourier e índices de forma. O modelo polifásico detectou três fases de crescimento. A primeira stanza para C. undecimalis foi em 46,8 cm e a segunda em 75,9 cm. Já C. parallelus,a primeira foi em 18,8 cm e segunda em 41,2 cm. Cada stanza possui um morfotipo específico de otólitos em ambas as espécies. Os otólitos de C. undecimalis e C. parallelus exibiram mudanças alométricas ontogenéticas em seu padrão de crescimento com dois pontos de mudança de stanzas. As stanzas corresponderam a comprimentos específicos alcançados por indivíduos ao longo de seus ciclos de vida, como seu tamanho na maturidade e comprimento na reversão sexual.
Palavras-chave: Alocação energética, Crescimento polifásico, Hermafroditismo, Morfogeometria, Reprodução.
Introduction
The life cycle of living beings is closely related to their maximum size and omset at sexual maturity. Morphoanatomical or physiological changes coupled with changes in body size are known as allometric relationships, and can reflect the compensatory effect necessary to maintain efficiency in bodily functions (Begon et al., 2006). Allometry is a common phenomenon in a variety of taxa, from bacteria (Mitchell, 2002) and invertebrates (Shingleton et al., 2007; Castro et al., 2020) to large mammals, such as whales (Ortega-Ortiz et al., 2018). In fish species, allometry has been extensively described (Gisbert, 1999; Su et al., 2020; Taylor et al., 2020). The beginning of the reproductive life is one of the main causes of changes in growth patterns over the life cycle of fishes, since it implies reallocation of body energy towards reproduction in detriment of growth (Quince et al., 2008; Boukal et al., 2014; Minte-Vera et al., 2016). Several models can be used to evaluate the morphological changes in allometric growth patterns of animals, with the model proposed by Huxley (1924), being one of the most used, because it allows a suitable quantitative description of the growth in a non-linear basis. This model is based on the potential relationship between two morphological variables (y = axb), assuming that body dimensions increase from an allometric coefficient (b of the equation) (Huxley, 1924). This model is usually applied to the relationship between body mass and size of individuals as an indicator of seasonal or interannual allometric changes (Fabré, Saint-Paul, 1998; Froese, 2006; Sousa et al., 2015), as well as of allometry in fish otoliths (Bervian et al., 2006).
Otoliths are calcified structures located in the inner ear of fish, being an important component of their mechanoreceptor system (Popper, Coombs, 1982; Cousseau, 2010). These structures allow the understanding of several aspects of fish’s life history (Campana, Neilson, 1985; Volpedo, Vaz-dos-Santos, 2015; Assis et al., 2020) because otolith growth shows a strict relationship to somatic growth (Francis, 1990). In addition, otoliths are conservative structures that have species-specific morphological characteristics, with the phylogenetic history of a particular taxon being usually reflected in its otolith shape (Nolf, 1985; Monteiro et al., 2005; Tuset et al., 2008, 2016). Therefore, it is not surprising that otolith morphology and morphometry have been used as powerful tools to detect physiological changes caused by many key factors of fish’s life cycle, such as reproduction or sexual maturity (Bervian et al., 2006; Capoccioni et al., 2011; Carvalho et al., 2015; Maciel et al., 2019). Otolith allometry has been widely applied in growth studies of both freshwater and marine species (Pérez, Fabré, 2009, 2013; De Queiroz et al., 2018), whereas otolith shape analysis has been employed in distinguishing fish stocks, populations and species (Capoccioni et al., 2011; Santos et al., 2017; Assis et al., 2020; Song et al., 2020), with Elliptic Fourier Analysis (FEA) being the most commonly used method in shape studies (Santos et al., 2017). Nevertheless, up to the present, the use of otolith allometry and its shape study as a response variable in fish reproductive cycle has been little addressed in hermaphroditic fish (Walker, McCormick, 2004; Munday et al., 2009; Walker, McCormick, 2009a,b; Mejri et al., 2018; Barr et al., 2019), especially for protandrous species.
Hermaphroditism is a polyphyletic condition that has been documented for approximately 2% of teleost species (Avise, Mank, 2009). It can occur sequentially and is characterized by a sex transition, either from male to female (protandry) or from female to male (protogyny) (Warner, 1975). According to the sex allocation theory, sex change is favored to increase the population’s reproductive fitness when the fertility of one particular sex, as a function of size and age, increases more rapidly compared to the other (Charnov, 1982). However, the energetic cost of sex change is as high as that involved in sexual maturation, which means that it can imply in decreased growth rates (Higgins et al., 2015; Matthias et al., 2016; Neves et al., 2017). For many years, sequential hermaphroditism was interpreted as an automatic sex change that occurred when individuals reached a certain body size or age (Allsop, West, 2003; Avise, Mank, 2009). However, some evidence points out that specific conditions in the population structure can trigger the sex change of sequential hermaphrodites (Munday et al., 2006; Walker, McCormick, 2009a), as observed by Shapiro (1987) in reef fish species.
In this context of sequential hermaphroditic fishes, two species of the genus Centropomus, C. undecimalis (Bloch, 1792) and C. parallelus (Poey, 1860), stand out in Brazil because of their both wide distribution along the coast and high commercial value. These two species are protandrous hermaphrodites, being commonly found throughout the tropical western South Atlantic ( Rivas, 1986; Taylor et al., 2000; De Figueiredo-Filho et al., 2021). Both species inhabit different habitats throughout their life cycle, being characterized as estuarine dependent fish (they breeding and growing in estuarine areas) that migrate to the sea to grow and feed (Blewett et al., 2009; Daros et al., 2016; Da Silva et al., 2018). They play important ecological roles such as top-down control in tropical coastal ecosystems (Lira et al., 2018). In addition, these two species have high commercial value in artisanal and sport fisheries (Alvarez-Lajonchère, Tsuzuki, 2008; Dantas, Barletta, 2016; Garrone-Neto et al., 2018). In Brazil, for example, an average of 3,541 tons of snooks are annually caught (considering catches between 2009 and 2018), making it the second highest catching country for this group, behind Mexico only (FAO, 2019). Despite this high catch, there is no evidence to date of population decline for these species. However the rising trend in fishing effort is worrying (Mendonça et al., 2019a,b).
Motivated by the biological peculiarities of these species, we hypothesized that the inherent sequential sex change is expressed by changes in otolith allometric patterns and shape. This is based on the assumption that otoliths are able to record changes due to shifts in growth patterns over specific stages of fish’s life cycle. Therefore, the objective of this study was to detect changes in the growth pattern of C. undecimalis and C. parallelus throughout their life cycle, based on the morphometry and shape of sagittal otoliths, as well as to identify what factors are related to these changes.
Material and methods
Study area and material collection. The coast of the state of Alagoas is 230 km long (08°54’51.3” to 10°30’24.2”S and 35°09’07.1” to 36°23’15.1”W), being characterized by a narrow continental shelf (50 km) with different coastal ecosystems throughout its extension (e.g., estuaries, coastal ridges, coral reefs and sandstone reefs) (Dorigo Correia, Sovierzoski, 2008; Oliveira, Kjerfve, 1993). These area has a semi-humid tropical climate, with rainy periods from March to August and dry periods between September and February. There Centropomus are targeted by small-scale coastal fisheries, conducted primarily with gillnets at maximum depths of 30 meters (Rangely et al., 2010), with young Centropomus individuals being very abundant in mangrove areas (Teixeira, 1997; Da Silva et al., 2018).
The sampling of individuals were conducted throughout the coast between July 2017 and January 2020, focusing on the main landing point of the state, the Port of Jaraguá (Rangely et al., 2010). Individuals were acquired directly from artisanal fishermen right after landing, being captured by different types of fishing gear (e.g., gillnet, trawlnet, longline, and hand-line) to collect fish from all size-classes. Specimens were taken to the laboratory to identification following Menezes, Figueiredo (1980).
Data analysis. The total length (TL, cm) of each fish was measured and the sagittal otoliths were removed. Fish lengths were organized by classes following the Sturges rule (Sturges, 1926), thus, C. undecimalis was grouped into size classes of 5 cm and C. parallelus data was grouped into 3 cm classes. The right otolith of each fish, when available, was weighed (OW, g) on a high precision scale (0.0001 g), then photographed by a Leica S8 APO stereo microscope with camera. Larger otoliths were photographed by a Canon Pc2264 camera in macro mode with a standardized height. Length (OL, mm) and height (OH, mm) were measured from otoliths images (Fig. 1), as well as perimeter (OP, mm) and area (AO, mm2). All measurements were recorded using ImageJ software version 1.48 (Schneider et al., 2012).
Initially, we fitted the potential Huxley model (y = axb); where a is the angular coefficient and b is the allometric coefficient) and tested for heteroscedasticity of these relationships by the Breusch-Pagan test (Breusch, Pagan, 1979). This test found heteroscedasticity for both species (P < 0.05). To correct for this, we calculated the residuals proportional to OL, given by the equation , where PR is the proportional residual, OLo is the observed otolith length, and OLp is the predicted otolith length (Barradas et al., 2016). Proportional residuals were applied because the variance of the error should be constant (homoscedastic) when conditionally analyzing the explanatory variable. We subsequently recalculated the values of a and b from Huxley’s potential relationship applying the nonlinear least-squares method, based on the proportional residuals, using Microsoft Excel Solver (Fylstra et al., 1998).
FIGURE 1 | Otoliths of Centropomidae. A. Centropomus undecimalis. B. Centropomus parallelus. OL = Otolith length; OH = Otolith height; a = anterior; p = posterior; v = ventral; d = dorsal.
With this new value of a, we used the polyphasic approach which considers the variable a, as invariant, and the value of b as variant according to size class (Bervian et al., 2006). This approach assumes a growth pattern composed of different phases, separated by a stanza change point (SCP), which indicates the TL at the moment when the growth pattern changes. Therefore, TL (in the size class of 5 and 3 cm for C. undecimalis and C. parallelus, respectively) was plotted against the mean OL values. Considering the fixed value of a (Huxley model), we calculated the allometric coefficient of each size class (bsc), from the equation: OL = aTLbsc. Then, a third-degree polynomial model was fitted between the bsc and the average TL classes. This model was derived (bsc’), giving rise to a second-degree equation. The obtained parabola was used to calculate the TL values (bsc’ = 0), at which growth changes occur.
To validate the growth stanzas, we analyzed whether the otolith contour followed the same changing pattern. For that, we analyzed the harmonics of the Elliptic Fourier Descriptors (EFD) of 120 otoliths from C. undecimalis (40 per allometric group) and 90 otoliths from C. parallelus (30 per allometric group), using the Shape software (Iwata, Ukai, 2002; Assis et al., 2020). This difference in sample size between species was due to the low number of C. parallelus in the study. From the otolith harmonics, a linear discriminant analysis (LDA) was performed to test the power of shape discrimination by growth stanzas, followed by a PERMANOVA test. R statistical software (R Development Core Team, 2013) and its Vegan package (Oksanen et al., 2020) were used to run LDA and PERMANOVA.
Finally, after establishing the morphotypes for both species, a characterization of otoliths’ morphotypes was performed for each group. We described the morphotypes by calculating shape indices related to the aspect ratio, circularity, roundness, rectangularity, and ellipticity of otoliths (Tuset et al., 2003; Volpedo, Echeverría, 2003; Bani et al., 2013) (Tab. 1). Circularity and roundness compare the otolith shape to a perfect circle; whereas rectangularity describes the variation in length and width in relation to the total area; ellipticity indicates whether the changes in the axes are proportional; and the aspect ratio estimates the irregularity of the surface area (Tuset et al., 2003; Volpedo, Echeverría, 2003; Bani et al., 2013). The Kruskal-Wallis test was used to compare these indices between morphotypes, considering a significance level of 0.05. When significant differences between the various classifications were detected, the Mann-Whitney U-test was subsequently applied for pairwise comparison between species (Zar, 2014).
TABLE 1 | Equations used to estimate the shape indices of the otoliths. OL = otolith length, OH = otolith height, P = perimeter, and A = area.
shape
index | Equation |
Aspect ratio | OL / OH |
Circularity | P2 / A |
Roundness | 4A / π x OL2 |
Rectangularity | A / OL x OH |
Ellipticity | OL – OH / OL + OH |
Results
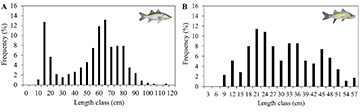
We collected 455 specimens of C. undecimalis, with total length ranging from 9 to 111 cm, with a bimodal distribution (peaks at 15 and 65 cm) (Fig. 2A). The number of C. parallelus was smaller, with only 176 individuals ranging in total length from 8 to 57 cm (Fig. 2B).
FIGURE 2 | Frequency distribution of Centropomus undecimalis (A) and Centropomus parallelus (B) collected in the state of Alagoas, Brazil.
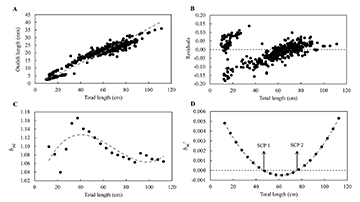
The relationship between TL and OL, using the proportional residuals, in C. undecimalis individuals showed positive allometry, b > 1 (OL = 0.235 x TL1.09, r² = 0.948, P = 0.001) (Fig. 3A), with the residual analysis showing three growth phases (Fig. 3B). The third-degree polynomial model resulted in the following equation bsc = 7E-7 x TL3 – 1.4E-4 x TL2 + 8E-3 x TL +0,99 (r² = 0.529) (Fig. 3C). The model fitted for the allometry coefficient derivative (bsc’) as a function of the TL(bsc’ = 2E-6 x TL2 – 3E-4 x TL2 + 8E-3), detected two growth stanzas, one at 46.8 cm and another at 75.9 cm (Fig. 3D).
FIGURE 3 | Growth changes in Centropomus undecimalis based on a polyphasic growth model. A. Potential regression for fish total length as a function of otolith radius; B. Residual distribution; C. Exponential coefficient (bsc) for length class; D. Derivative of the third-degree polynomial function, and indication of the change points of the stanza (SCP 1 and SCP 2).
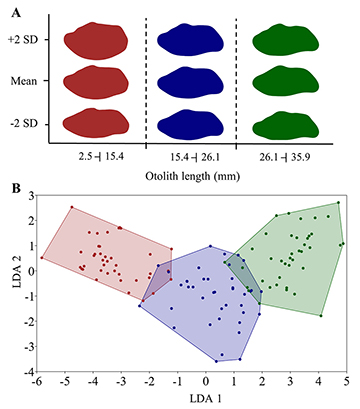
Reconstruction of the otolith shape of C. undecimalis indicated three different morphotypes. Morphotype 1, with OL smaller than 15.4 mm, morphotype 2 with OL between 15.4 and 26.1 mm, and the third comprising otoliths with lengths greater than 26.1 mm (Fig. 4A). The LDA showed high jack-knifed classification success (91.7%) (Fig. 4B), with significant differences among the three morphotypes (P = 0.0001).
FIGURE 4 | Shape of otoliths of Centropomus undecimalis by morphotypes. A. Otolith mean outline and standard deviation (SD). B. Linear discriminant (LD) analysis of harmonics of elliptic Fourier descriptors of otoliths. red = Morphotype 1; blue = Morphotype 2; green = Morphotype 3.
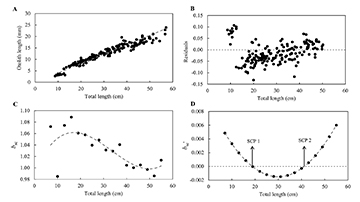
For C. parallelus, the relationship between TL and OL, using the proportional residuals, showed an isometric growth, b = 1(OL = 0.389 x TL1.01, r² = 0.931, P = 0.001) (Fig. 5A), with residual analysis pointing to three growth phases (Fig. 5B). The third-degree polynomial model resulted in (bsc = 2E-6 x TL2 – 3E-4 x TL2 + 8E-3) (r² = 0.559) (Fig. 5C). The model fitted for the allometry coefficient derivative (bsc’ = 2E-6 x TL2 – 3E-4 x TL + 8E-3), also detected two growth stanzas, first at 18.8 cm and the second at 41.2 cm (Fig. 5D).
FIGURE 5 | Growth changes in Centropomus parallelus based on a polyphasic growth model. A. Potential regression for fish total length as a function of otolith radius; B. Residual distribution; C. Exponential coefficient (bsc) for length class; D. Derivative of the third-degree polynomial function, and indication of the change points of the stanza (SCP 1 and SCP 2).
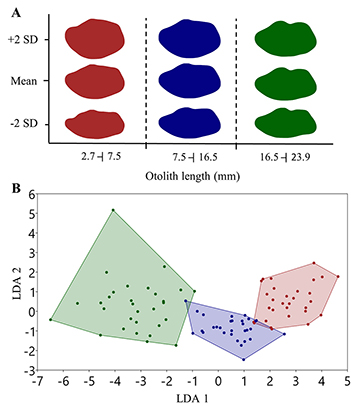
The otolith contours also indicated three morphotypes: first composed of the otoliths with OL smaller than 7.5 mm; second between 7.5 and 16.5 mm; and the last one OL larger than 16.5 mm (Fig. 6A). The LDA showed a jack-knifed classification success of 92.2% (Fig. 6B), with a significant difference between morphotypes (P = 0.0001).
FIGURE 6 | Shape of otoliths of Centropomus parallelus by morphotypes. A. Otolith mean outline and standard deviation (SD). B. Linear discriminant (LD) analysis of harmonics of elliptic Fourier descriptors of otoliths. red = Morphotype 1; blue = Morphotype 2; green = Morphotype 3.
Therefore, both species exhibited three morphotypes. In C. undecimalis, the ratios between OL/OH and OL/OW grow differently among the morphotypes. Furthermore, all shape indices showed a significant difference. Morphotype 1 presented a less elongated aspect and a higher circularity value than the other two morphotypes. Morphotype 2, on its turn, is elongated and presents higher rectangularity and roundness, and Morphotype 3 presented higher aspect ratio and ellipticity values (Tab. 2). The otoliths of C. parallelus showed a similar trend to its congener C. undecimalis, with different relative growth between the biometric proportions of their morphotypes. Two shape indices showed some similarities among the groups (roundness and circularity), but the other indices were significantly different among morphotypes. Morphotype 1 was less elongated and had a higher circularity value, whereas Morphotype 2 presented the greatest roundness, and Morphotype 3 was the most elongated and the most rectangular (Tab. 2).
TABLE 2 | Morphometric relations and shape index of the otoliths of Centropomus undecimalis and C. parallelus per morphotype. Means with the different lower-case letter superscripted are significantly different (P < 0.05). OW = Otolith weigh.
Species | Class | N | OW | Aspect ratio | Roundness | Circularity | Rectangularity | Ellipticity |
Centropomus undecimalis | All | 455 | 0.423 ± 0.329 | 2.031 ± 0.154 | 0.425 ± 0.082 | 23.605 ± 4.573 | 0.680 ± 0.138 | 0.339 ± 0.033 |
Morphotype 1 | 146 | 0.085 ± 0.098 | 1.904 ± 0.115a | 0.377 ± 0.124a | 26.859 ± 6.501a | 0.567 ± 0.198a | 0.310 ± 0.027a | |
Morphotype 2 | 238 | 0.489 ± 0.134 | 2.075 ± 0.120b | 0.454 ± 0.030b | 21.870 ± 1.661b | 0.737 ± 0.026b | 0.349 ± 0.025b | |
Morphotype 3 | 71 | 0.917 ± 0.329 | 2.146 ± 0.156c | 0.430 ± 0.038c | 22.728 ± 2.372c | 0.721 ± 0.027c | 0.363 ± 0.031c | |
Centropomus parallelus | All | 176 | 0.157 ± 0.121 | 1.814 ± 0.156 | 0.510 ± 0.075 | 19.999 ± 5.037 | 0.724 ± 0.102 | 0.287 ± 0.037 |
Morphotype 1 | 33 | 0.035 ± 0.023 | 1.727 ± 0.152a | 0.445 ± 0.129a | 24.804 ± 9.736ab | 0.602 ± 0.181a | 0.264 ± 0.040a | |
Morphotype 2 | 103 | 0.135 ± 0.071 | 1.797 ± 0.111b | 0.529 ± 0.036b | 18.241 ± 1.464a | 0.743 ± 0.025b | 0.284 ± 0.028b | |
Morphotype 3 | 40 | 0.318 ± 0.121 | 1.919 ± 0.193c | 0.517 ± 0.065b | 20.689 ± 2.045b | 0.772 ± 0.059c | 0.312 ± 0.040c |
Discussion
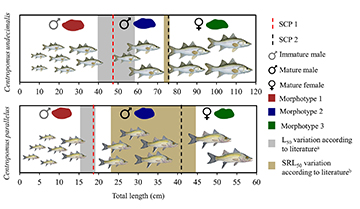
The otoliths of C. undecimalis and C. parallelus exhibited clear ontogenetic changes in their growth pattern with two stanzas changing points (SCPs) (P < 0.001 for both species). There stanzas corresponded to specific lengths reached by individuals over their life cycles, such as their size at maturity (L50) and length at sexual reversion (SRL50) (Fig. 7). In both species, SCPs (SCP 1 and SCP 2) allowed us to identify three growth phases, which were expressed by significant morphogeometric variations in otoliths’ shape (P < 0.001 for both species) (Fig. 7), creating a specific morphotype for each phase (Morphotype 1, Morphotype 2 and Morphotype 3). Interspecific differences were observed in the ontogenetic development of the otoliths, in which for both species Morphotype 1 was less elongated and rectangular, typical of more sedentary fish conversely as the structure grows it becomes more rectangular and elongated, which are features of more active fish (Volpedo et al., 2008; Tuset et al., 2015). There were high shape differences by morphotypes (classification = 90%), with the greatest similarities occurring especially in otoliths close in size to the transition lengths between morphotypes. However, the limitations of these results for Morphotype 1, can be attributed to intra-population variations, which could not be tested due to uncertainty of the type of environment where they were caught. Comparing the timing of the changes in growth pattern, it was observed that the first stanza occurs earlier in C. parallelus (31.4% of the maximum length) than in C. undecimalis (39.1%). In contrast, the second stanza occurs earlier in C. undecimalis (63.3%), with C. parallelus only changing its growth pattern when it reaches 68.6% of its maximum length (Fig. 7). Allometric changes in the otoliths of Centropomus are observed in the first phase as a greater energetic investment in increasing fish size to the detriment of otolith size. The second allometry is produced by an inverse process, where otoliths grow more than fish size proportionally. Therefore, the morphogeometry of otoliths of Centropomus may be an appropriate indicator of their ontogenetic phases.
FIGURE 7 | Ontogenetic phases for Centropomus undecimalis and Centropomus parallelus, and indications of growth stanzas. L50 = maturation length; SRL50 = sexual reversion length; literaturea for C. undecimalis = Gassman et al. (2017) and Cruz-Botto et al. (2018); literaturea for C. parallelus = Cunha Chaves, Nogueira (2018), Assis et al. (2019) and Costa e Silva et al. (2021); literatureb for C. undecimalis = Gassman et al. (2017) and Young et al. (2020); literatureb for C. parallelus = Cerqueira (2005), Gómez-Ortiz et al. (2011) and Assis et al. (2019).
The allometry found in our study is well known in fish (Bervian et al., 2006; Carvalho et al., 2015; Barradas et al., 2016; Maciel et al., 2019). In gonochoric fish, this pattern is commonly associated with the onset of sexual maturity as observed by Carvalho et al. (2015), for Anchoa tricolor; Barradas et al. (2016) for Corydoras paleatus; and Maciel et al. (2019) for Genidens genidens. These authors also made use of the polyphasic method and found two growth stanzas, the first being very similar to the maturation size (L50) of these species. This occurs because sexual maturation is a key-point in the life history of fish, where changes in energy allocation typically occurs. Before sexual maturation, it is exclusively directed to body growth, whereas after first maturation, the energy allocation changes to support the development of reproductive structures (Quince et al., 2008; Wilson et al., 2018).
For sequential hermaphroditic fishes, this polyphasic growth methodology to break down when growth changes occur had not yet been applied. However, it did not show any difference for the gonochoric fishes, as the growth stanzas found were also very similar with estimated reproductive parameters for C. undecimalis and C. parallelus (Tab. 3). In both species, the first stanza was within the estimated L50 size, and the second stanza was close to the sexual reversal of males into active female size (SRL50). Therefore, the reproductive factor was also the main possible cause, similar to that found for gonochoric fishes (Bervian et al., 2006; Carvalho et al., 2015; Barradas et al., 2016; Maciel et al., 2019). Sequential hermaphroditic species also require high energetic demands due to the amount of complex physiological processes involved in gametogenesis and subsequent sex change (Higgins et al., 2015; Matthias et al., 2016).
TABLE 3 | Reproductive parameters of Centropomus undecimalis and C. parallelus. L50 = length at first sexual maturation; SRL50 = length at sexual reversal of males into active female size; NI = Not informed.
Species | L50
(cm) | SRL50
(cm) | Geographical
coordinates | References |
C. undecimalis | 40–50 | NI | 10°58’55”N 74°18’29.1”W | Cruz-Botto et al. (2018) |
C. undecimalis | 59 | 73.6 | 10°11’30” – 10°20’20”N 65°41’10” – 65°57’20”W | Gassman et al. (2017) |
C. undecimalis | NI | 73.1 | 27°50’00” – 26°50’00”N 80°00’00” – 80°30’00”W | Young et al.
(2020) |
C. parallelus | 19 | NI | 25º52’00”S 48º39’00”W | Chaves, Nogueira
(2018) |
C. parallelus | 15 | 23 | 15°43’00”S 38°55’00”W | Assis et al.
(2019) |
C. parallelus | 18 | NI | 25°45’00”S 48°20’00”W | Costa e Silva et al. (2021) |
C. parallelus | NI | 45 | 27°35’02”S 48°26’35”W | Cerqueira (2005) |
C. parallelus | NI | 32 | 21°30’00” – 22°48”00”N 97°36’00” – 98°00’00”W | Gómez-Ortiz et al.
(2011) |
The SRL50 shows high variation, especially for C. parallelus (Tab. 3). This may be related to fishing (Hunter et al., 2015), since overfishing can directly affect size at maturity, longevity, and fecundity (Hutchings, 2002; Hunter et al., 2015; Da Silva et al., 2019). In addition, sex allocation theory points out that sex change occurs when reproductive potential in the first sex is no longer as efficient (Charnov, 1982). However, Charnov, Skúladóttir (2000) proposed a dimensionless model, which presents the idea that sex change occurs when individuals reach a certain size/age (low variability). In contrast to this approach, in some species sex changes take place as a response to known social or demographic conditions of local populations, such as local sexual proportion (Shapiro, Lubbock, 1980; Shapiro, 1987). Since empirical evidences associate body size and otolith morphometry in both species, our study supports the model proposed by Charnov, Skúladóttir (2000). By applying the same conceptual approach, Allsop, West (2003) predicted sex change at 80% of maximum body size as an invariant relation modeled for over 50 species. If it is assumed that the second stanza coincides with the sex change in both examined species, this phenomenon should occur at 63% of maximum body size for C. undecimalis and 68% of maximum body size for C. parallelus. These changes, which could be considered early according to the empirical model proposed by Allsop, West (2003), may be related to higher growth rates, which are typical of tropical regions (Houde, 1989). Therefore, for both species, the first stanza may be related to sexual maturation and the second stanza may be related to sexual reversion.
Although some features of the reproductive biology of the species may impact the overall shape of otoliths, which could explain why three otoliths morphotypes were found for both studied species, there are many other factors that may also be related to changes in otoliths shape. For instance, habitat type can also be an important factor as changes in habitat use can directly impact otolith shape (Tuset et al., 2003, 2018; Vignon, 2012; Zischke et al., 2016). In this context, it might be expected that C. undecimalis and C. parallelus would present different morphotypes as found in this study, due to their migratory behaviour between marine, estuarine and continental waters throughout their life history (Daros et al., 2016; Bot Neto et al., 2020; Purtlebaugh et al., 2020; Stevens et al., 2021). However, since we acquired specimens from fishermen at the time of landing, we cannot state whether habitat type, feeding, depth, or temperature influenced the morphotypes due to the uncertainties of where the fish were captured. Therefore, we can observe that the allometry present in the otoliths of C. undecimalis and C. parallelus represents complex processes associated with evolutionary and demographic issues that need to be better studied by evaluating growth rates and the effect of habitat changes characteristic of these estuarine dependent species.
Finally, we can conclude that C. undecimalis and C. parallelus show polyphasic growth, with observed changes in growth resulting from reproductive processes such as maturation and sexual reversion. Furthermore, we detected that the otolith shape also followed the pattern of allometric relationships, indicating three different morphotypes for each species. This study reinforces the importance of life-history studies of C. undecimalis and C. parallelus.
Acknowledgments
The present study is part of the Projeto Ecológico de Longa Duração da Costa dos Corais – Alagoas, funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, process number 441657/2016–8), the Alagoas Research Foundation (process number 60030.1564/2016), and the Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES) (process number 23038.000452/2017–16). The authors would like to express their gratitude to CNPq for providing a grant to N. Fabré, PhD (process number 306624/2014–1); to all the fishermen who helped in the field work; and to all members of the Laboratory of Ecology, Fish, and Fishery and the Laboratory of Conservation and Management of Renewable Resources at the Universidade Federal de Alagoas.
References
Allsop DJ, West SA. Constant relative age and size at sex change for sequentially hermaphroditic fish. J Evol Biol. 2003; 16(5):921–29. https://doi.org/10.1046/j.1420-9101.2003.00590.x
Alvarez-Lajonchère L, Tsuzuki MY. A review of methods for Centropomus spp. (snooks) aquaculture and recommendations for the establishment of their culture in Latin America. Aquac Res. 2008; 39(7):684–700. https://doi.org/10.1111/j.1365-2109.2008.01921.x
Assis DAS, Nobre DM, Freitas MC, Moraes LE, Santos ACA. Reproductive biology of the protandric hermaphrodite fat snook Centropomus parallelus Poey 1860 in a tropical estuary, northeastern Brazil. Stud Neotrop Fauna Environ. 2019; 54(3):225–35. https://doi.org/10.1080/01650521.2019.1663594
Assis IO, Silva VEL, Souto-Vieira D, Lozano AP, Volpedo AV, Fabré NN. Ecomorphological patterns in otoliths of tropical fishes: Assessing trophic groups and depth strata preference by shape. Environ Biol Fishes. 2020; 103(4):349–61. https://doi.org/10.1007/s10641-020-00961-0
Avise JC, Mank JE. Evolutionary perspectives on hermaphroditism in fishes. Sex Dev. 2009; 3(2–3):152–63. https://doi.org/10.1159/000223079
Bani A, Poursaeid S, Tuset VM. Comparative morphology of the sagittal otolith in three species of south Caspian gobies. J Fish Biol. 2013; 82(4):1321–32. https://doi.org/10.1111/jfb.12073
Barr EE, Cabello MG, Gómez MP, Boa AG. Study of the age of Centropomus robalito by otoliths analysis of sagitta, asteriscus and lapillus in Mexican Central Pacific. J Aquac Fisheries. 2019; 3:13. https://doi.org/10.24966/AAF-5523/100013
Barradas JRS, Lermen IS, Larré GG, Martins TP, Fontoura NF. Polyphasic growth in fish: A case study with Corydoras paleatus (Siluriformes, Callichthyidae). Iheringia Ser Zool. 2016; 106:e2016017. https://doi.org/10.1590/1678-4766e2016017
Begon M, Townsend CR, Harper JL. Ecology: From individuals to ecosystems. 4th ed. Blackwell Publishing; 2006.
Bervian G, Fontoura NF, Haimovici M. Statistical model of variable allometric growth: Otolith growth in Micropogonias furnieri (Actinopterygii, Sciaenidae). J Fish Biol. 2006; 68(1):196–208. https://doi.org/10.1111/j.0022-1112.2006.00890.x
Blewett DA, Stevens PW, Champeau TR, Taylor RG. Use of rivers by common snook Centropomus undecimalis in Southwest Florida: A first step in addressing the overwintering paradigm. Fla Sci. 2009; 72(4):310–24. Available from: https://www.jstor.org/stable/24321646
Bot Neto RL, Carvalho BM, Schwarz Júnior R, Spach HL. Ontogenetic variation in the sagitta otolith of Centropomus undecimalis (Actinopterygii: Perciformes: Centropomidae) in a tropical estuary. Acta Ichthyol Piscat. 2020; 50(4):433–43. https://doi.org/10.3750/AIEP/03014
Boukal DS, Dieckmann U, Enberg K, Heino M, Jørgensen C. Life-history implications of the allometric scaling of growth. J Theor Biol. 2014; 359:199–207. https://doi.org/10.1016/j.jtbi.2014.05.022
Breusch TS, Pagan AR. A simple test for heteroscedasticity and random coefficient variation. Econometrica. 1979; 47(5):1287–94. https://doi.org/10.2307/1911963
Campana SE, Neilson JD. Microstructure of fish otoliths. Can J Fish Aquat Sci. 1985; 42(5):1014–32. https://doi.org/10.1139/f85-127
Capoccioni F, Costa C, Aguzzi J, Menesatti P, Lombarte A, Ciccotti E. Ontogenetic and environmental effects on otolith shape variability in three Mediterranean European eel (Anguilla anguilla, L.) local stocks. J Exp Mar Biol Ecol. 2011; 397(1):1–07. https://doi.org/10.1016/j.jembe.2010.11.011
Carvalho BM, Vaz-dos-Santos AM, Spach HL, Volpedo AV. Ontogenetic development of the sagittal otolith of the anchovy, Anchoa tricolor, in a subtropical estuary. Sci Mar. 2015; 79(4):409–18. https://doi.org/10.3989/scimar.04218.31A
Castro RB, Azevedo-Farias AK, Silva VEL. Macrobrachium amazonicum (Crustacea: Decapoda: Palaemonidae) shows a polyphasic growth pattern in the Lower São Francisco River, Northeastern Brazil. Panam J Aquat Sci. 2020; 16(1):52–56.
Cerqueira VR. Cultivo do robalo-peva, Centropomus parallelus. In: Baldisserotto B, Gomes LC, editors. Espécies nativas para psicultura no Brasil. Santa Maria: Editora UFSM; 2005. p.403–31.
Charnov EL, Skúladóttir U. Dimensionless invariants for the optimal size (age) of sex change. Evol Ecol Res. 2000; 2:1067–71.
Charnov EL. The theory of sex allocation. Monographs in population biology. New Jersey: Princeton University Press; 1982. https://doi.org/10.2307/j.ctvx8b6km
Chaves PTC, Nogueira AB. Biologia reprodutiva do robalo-peva, Centropomus parallelus (Teleostei), na Baía de Guaratuba (Brasil). Acta Biol Parana. 2018; 47(3–4):69–84. https://doi.org/10.5380/abpr.v47i0.62590
Costa e Silva GH, Freitas MO, Abilhoa V. Reproductive biology of the fat snook Centropomus parallelus Poey, 1860 (Teleostei, Centropomidae) and implications for its management in the southern Atlantic Ocean. J Fish Biol. 2021; 99(2):669–72. https://doi.org/10.1111/jfb.14718
Cousseau MB. Ictiología: Aspectos fundamentales. La vida de los peces sudamericanos. Mar Del Plata: EUDEM; 2010.
Cruz-Botto S, Roca-Lanao B, Gaitán-Ibarra S, Chaparro-Muñoz N, Villamizar N. Natural vs laboratory conditions on the reproductive biology of common snook Centropomus undecimalis (Bloch, 1792). Aquaculture. 2018; 482:9–16. https://doi.org/10.1016/j.aquaculture.2017.09.013
Dantas DV, Barletta M. Habitat use by Centropomus undecimalis in a rocky area of estuarine beach in north-east Brazil. J Fish Biol. 2016; 89(1):793–803. https://doi.org/10.1111/jfb.13001
Daros FA, Spach HL, Correia AT. Habitat residency and movement patterns of Centropomus parallelus juveniles in a subtropical estuarine complex. J Fish Biol. 2016; 88(5):1796–810. https://doi.org/10.1111/jfb.12944
Dorigo Correia M, Sovierzoski HH. Gestão e desenvolvimento sustentável da zona costeira do Estado de Alagoas, Brasil. Revista de Gestão Costeira Integrada. 2008; 8(2):25–45.
Fabré NN, Saint-Paul U. Annulus formation on scales and seasonal growth of the Central Amazonian anostomid Schizodon fasciatus. J Fish Biol. 1998; 53(1):1–11. https://doi.org/10.1111/j.1095-8649.1998.tb00103.x
Food and Agriculture Organization of the United Nations (FAO). FishStatJ – Software for fishery and aquaculture statistical time series. Rome: Fisheries and Aquaculture Division (NFI) – FAO; 2019. Available from: http://www.fao.org/fishery/statistics/software/fishstatj/en
De Figueiredo-Filho JM, Marceniuk AP, Feijó A, Siccha-Ramirez R, Ribeiro GS, Oliveira C et al. Taxonomy of Centropomus Lacépède, 1802 (Perciformes: Centropomidae), with focus on the Atlantic species of the genus. Zootaxa. 2021; 4942(3):301–38. https://doi.org/10.11646/zootaxa.4942.3.1
Francis RICC. Back-calculation of fish length: A critical review. J Fish Biol. 1990; 36(6):883–902. https://doi.org/10.1111/j.1095-8649.1990.tb05636.x
Froese R. Cube law, condition factor and weight-length relationships: History, meta-analysis and recommendations. J Appl Ichthyol. 2006; 22(4):241–53. https://doi.org/10.1111/j.1439-0426.2006.00805.x
Fylstra D, Lasdon L, Watson J, Waren A. Design and use of the Microsoft Excel Solver. Interfaces (Providence). 1998; 28(5):29–55. https://doi.org/10.1287/inte.28.5.29
Garrone-Neto D, Sanches EA, Daros FALM, Imanobu CMR, Moro PS. Using the same fish with different rules: A science-based approach for improving management of recreational fisheries in a biodiversity hotspot of the Western South Atlantic. Fish Manag Ecol. 2018; 25(4):253–60. https://doi.org/10.1111/fme.12288
Gassman J, López Rojas H, Padrón D. Reproducción de los róbalos Centropomus undecimalis y C. ensiferus (Perciformes: Centropomidae) en una laguna costera tropical. Rev Biol Trop. 2017; 65(1):181–94. https://doi.org/10.15517/rbt.v65i1.23614
Gisbert E. Early development and allometric growth patterns in Siberian sturgeon and their ecological significance. J Fish Biol. 1999; 54(4):852–62. https://doi.org/10.1111/j.1095-8649.1999.tb02037.x
Gómez-Ortiz MG, López-Navarrete H, Arteaga-Peña R, Balderas-Telles J, Acosta-Barbosa G. Aspectos biológicos-pesqueros de Robalo blanco Centropomus undecimalis y Chucumite Centropomus parallelus en el Sur de Tamaulipas y Norte de Veracruz. CRIP Tampico: INAPESCA; 2011.
Higgins RM, Diogo H, Isidro EJ. Modelling growth in fish with complex life histories. Rev Fish Biol Fish. 2015; 25(3):449–62. https://doi.org/10.1007/s11160-015-9388-8
Houde ED. Comparative growth, mortality, and energetics of marine fish larvae: Temperature and implied latitudinal effects. Fish Bull. 1989; 87(3):471–95.
Hunter A, Speirs DC, Heath MR. Fishery-induced changes to age and length dependent maturation schedules of three demersal fish species in the Firth of Clyde. Fish Res. 2015; 170:14–23. https://doi.org/10.1016/j.fishres.2015.05.004
Hutchings JA. Life histories of fish. In: Hart PJB, Reynolds JD, editors. Handbook of fish biology and fisheries: Fish biology, Volume 1. Oxford: Blackwell Publishing; 2002. p.149–74. https://doi.org/10.1002/9780470693803.ch7
Huxley JS. Constant differential growth-ratios and their significance. Nature. 1924; 114(2877):895–96. https://doi.org/10.1038/114895a0
Iwata H, Ukai Y. SHAPE: A computer program package for quantitative evaluation of biological shapes based on elliptic Fourier descriptors. J Hered. 2002; 93(5):384–85. https://doi.org/10.1093/jhered/93.5.384
Lira A, Angelini R, Le Loc’h F, Ménard F, Lacerda C, Frédou T et al. Trophic flow structure of a neotropical estuary in northeastern Brazil and the comparison of ecosystem model indicators of estuaries. J Mar Syst. 2018; 182:31–45. https://doi.org/10.1016/j.jmarsys.2018.02.007
Maciel TR, Vaz-dos-Santos AM, Barradas JRS, Vianna M. Sexual dimorphism in the catfish Genidens genidens (Siluriformes: Ariidae) based on otolith morphometry and relative growth. Neotrop Ichthyol. 2019; 17(1):e180101. https://doi.org/10.1590/1982-0224-20180101
Matthias BG, Ahrens RNM, Allen MS, Lombardi-Carlson LA, Fitzhugh GR. Comparison of growth models for sequential hermaphrodites by considering multi-phasic growth. Fish Res. 2016; 179:67–75. https://doi.org/10.1016/j.fishres.2016.02.006
Mejri M, Trojette M, Allaya H, Ben Faleh A, Jmil I, Chalh A et al. Use of otolith shape to differentiate two lagoon populations of Pagellus erythrinus (Actinopterygii: Perciformes: Sparidae) in Tunisian waters. Acta Ichthyol Piscat. 2018; 48(2):153–61. https://doi.org/10.3750/AIEP/02376
Mendonça JT, Chao L, Albieri RJ, Giarrizzo T, Fadré NN, da Silva FMS et al. Centropomus parallelus. The IUCN Red List of Threatened Species. 2019a; e.T190327A82662519 [Internet]. IUCN: 2019a. https://dx.doi.org/10.2305/IUCN.UK.2019-2.RLTS.T190327A82662519.en
Mendonça JT, Chao L, Albieri RJ, Giarrizzo T, Silva FMS, Castro MG et al. Centropomus undecimalis. The IUCN Red List of Threatened Species. 2019b; e.T191835A82665184 [Internet]. IUCN: 2019b. https://dx.doi.org/10.2305/IUCN.UK.2019-2.RLTS.T191835A82665184.en
Menezes NA, Figueiredo JL. Manual de peixes marinhos do Sudeste do Brasil. IV. Teleostei (3). São Paulo: Museu de Zoologia, Universidade de São Paulo; 1980.
Minte-Vera CV, Maunder MN, Casselman JM, Campana SE. Growth functions that incorporate the cost of reproduction. Fish Res. 2016; 180:31–44. https://doi.org/10.1016/j.fishres.2015.10.023
Mitchell JG. The energetics and scaling of search strategies in bacteria. Am Nat. 2002; 160(6):727–40. https://doi.org/10.1086/343874
Monteiro LR, Di Beneditto APM, Guillermo LH, Rivera LA. Allometric changes and shape differentiation of sagitta otoliths in sciaenid fishes. Fish Res. 2005; 74(1–3):288–99. https://doi.org/10.1016/j.fishres.2005.03.002
Munday PL, Buston PM, Warner RR. Diversity and flexibility of sex-change strategies in animals. Trends Ecol Evol. 2006; 21(2):89–95. https://doi.org/10.1016/j.tree.2005.10.020
Munday PL, Ryen CA, McCormick MI, Walker SPW. Growth acceleration, behaviour and otolith check marks associated with sex change in the wrasse Halichoeres miniatus. Coral Reefs. 2009; 28(3):623. https://doi.org/10.1007/s00338-009-0499-3
Neves A, Vieira AR, Sequeira V, Paiva RB, Gordo LS. Modelling the growth of a protogynous sparid species, Spondyliosoma cantharus (Teleostei: Sparidae). Hydrobiologia. 2017; 797(1):265–75. https://doi.org/10.1007/s10750-017-3188-1
Nolf D. Otolith piscium. In: Schultze H-P, editor. Handbook of Paleoichthyology: Volume 10. Stuttgart, New York: Gustav Fisher Verlag; 1985.
Oliveira AM, Kjerfve B. Environmental responses of a tropical coastal lagoon system to hydrological variability: Mundaú-Manguaba, Brazil. Estuar Coast Shelf Sci. 1993; 37(6):575–91. https://doi.org/10.1006/ecss.1993.1074
Ortega-Ortiz CD, Gómez-Muñoz VM, Gendron D. Allometry and morphometry of blue whales photographed in the Gulf of California: Insights into subspecies taxonomy in the Eastern North Pacific. Endanger Species Res. 2018; 37:183–94. https://doi.org/10.3354/esr00910
Oksanen J, Guillaume Blanchet F, Friendly M, Kindt R, Legendre P, McGlinn D et al. Vegan: Community Ecology Package. Version 2.5-7 [Internet]. CRAN; 2020. Available from: https://cran.r-project.org/web/packages/vegan/index.html
Pérez A, Fabré NN. Seasonal growth and life history of the catfish Calophysus macropterus (Lichtenstein, 1819) (Siluriformes: Pimelodidae ) from the Amazon floodplain. J Appl Ichthyol. 2009; 25(3):343–49. https://doi.org/10.1111/j.1439-0426.2008.01104.x
Pérez A, Fabré NN. Spatial population structure of the Neotropical tiger catfish Pseudoplatystoma metaense: Skull and otolith shape variation. J Fish Biol. 2013; 82(5):1453–68. https://doi.org/10.1111/jfb.12046
Popper AN, Coombs S. The morphology and evolution of the ear in actinopterygian fishes. Am Zool. 1982; 22(2):311–28. https://doi.org/10.1093/icb/22.2.311
Purtlebaugh CH, Martin CW, Allen MS. Poleward expansion of common snook Centropomus undecimalis in the northeastern Gulf of Mexico and future research needs. PLoS ONE. 2020; 15(6):e0234083. https://doi.org/10.1371/journal.pone.0234083
De Queiroz JDGR, Salvador NLA, Sousa MF, Da Silva VEL, Fabré NN, Batista VS. Life-history traits of Chloroscombrus chrysurus (Actinopterygii: Perciformes: Carangidae) in tropical waters of the Atlantic Ocean. Acta Ichthyol Piscat. 2018; 48(1):1–08. https://doi.org/10.3750/AIEP/02315
Quince C, Abrams PA, Shuter BJ, Lester NP. Biphasic growth in fish I: Theoretical foundations. J Theor Biol. 2008; 254(2):197–206. https://doi.org/10.1016/j.jtbi.2008.05.029
R Development Core Team. R: A language and environment for statistical computing. Vienna, Austria: The R Foundation for Statistical Computing; 2013. Available from: http://www.R-project.org/
Rangely J, Fabré NN, Tiburtino C, Batista VS. Estratégias de pesca artesanal no litoral marinho alagoano (Brasil). Bol Inst Pesca. 2010; 36(4):263–75.
Rivas LR. Systematic review of the perciform fishes of the genus Centropomus. Copeia. 1986; 1986(3):579–611. https://doi.org/10.2307/1444940
Santos RS, Azevedo MCC, Albuquerque CQ, Araújo FG. Different sagitta otolith morphotypes for the whitemouth croaker Micropogonias furnieri in the Southwestern Atlantic coast. Fish Res. 2017; 195:222–29. https://doi.org/10.1016/j.fishres.2017.07.027
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012; 9(7):671–75. https://doi.org/10.1038/nmeth.2089
Shapiro DY. Differentiation and evolution of sex change in fishes. Bioscience. 1987; 37(7):490–97. https://doi.org/10.2307/1310421
Shapiro DY, Lubbock R. Group sex ratio and sex reversal. J Theor Biol. 1980; 83(3):411–26. https://doi.org/10.1016/0022-5193(80)90048-X
Shingleton AW, Frankino WA, Flatt T, Nijhout HF, Emlen DJ. Size and shape: The developmental regulation of static allometry in insects. Bioessays. 2007; 29(6):536–48. https://doi.org/10.1002/bies.20584
Da Silva VEL, Teixeira EC, Batista VS, Fabré NN. Spatial distribution of juvenile fish species in nursery grounds of a tropical coastal area of the south-western Atlantic. Acta Ichthyol Piscat. 2018; 48(1):9–18. https://doi.org/10.3750/AIEP/02299
Da Silva VEL, Vieira DS, Teixeira EC, Ferreira ACL, Assis IO, Rangely J et al. Maturity, fecundity, and reproductive cycle of Conodon nobilis (Actinopterygii: Perciformes: Haemulidae) in tropical waters of the Atlantic Ocean. Acta Ichthyol Piscat. 2019; 49(3):235–42. https://doi.org/10.3750/aiep/02597
Song J, Dou S, Cao L, Liu J. Sulcus and otolith outline analyses: Complementary tools for stock discrimination in white croaker Pennahia argentata in northern Chinese coastal waters. J Oceanol Limnol. 2020; 38(5):1559–71. https://doi.org/10.1007/s00343-020-0023-8
Sousa MF, Fabré NN, Batista VS. Seasonal growth of Mugil liza Valenciennes, 1836 in a tropical estuarine system. J Appl Ichthyol. 2015; 31(4):627–32. https://doi.org/10.1111/jai.12704
Stevens PW, Kimball ME, Elmo GM, Williams KL, Ritch JL, Crane DP. Investigation into the occurrence of juvenile common snook Centropomus undecimalis, a subtropical estuarine sport fish, in saltmarshes beyond their established range. Estuaries Coast. 2021; 44(5):1477–83. https://doi.org/10.1007/s12237-020-00884-0
Sturges HA. The choice of a class interval. J Am Stat Assoc. 1926; 21(153):65–66. https://doi.org/10.1080/01621459.1926.10502161
Su L, Luo S, Qiu N, Xu C, Hou M, Xiong X et al. Comparative allometric growth of rare minnow (Gobiocypris rarus) in two culture environments. Hydrobiologia. 2020; 847(9):2083–95. https://doi.org/10.1007/s10750-020-04232-y
Taylor MD, Fowler AM, Suthers IM. Insights into fish auditory structure–function relationships from morphological and behavioural ontogeny in a maturing sciaenid. Mar Biol. 2020; 167(2):21. https://doi.org/10.1007/s00227-019-3619-9
Taylor RG, Whittington JA, Grier HJ, Crabtree RE. Age, growth, maturation, and protandric sex reversal in common snook, Centropomus undecimalis, from the east and west coasts of South Florida. Fish Bull. 2000; 98(3):612–24.
Teixeira RL. Distribution and feeding habits of the young common snook, Centropomus undecimalis (Pisces: Centropomidae), in the shallow waters of a tropical brazilian estuary. Bol Mus Biol Mello Leitão. 1997; 6:35–46.
Tuset VM, Farré M, Otero-Ferrer JL, Vilar A, Morales-Nin B, Lombarte A. Testing otolith morphology for measuring marine fish biodiversity. Mar Freshw Res. 2016; 67(7):1037–48. https://doi.org/10.1071/MF15052
Tuset VM, Imondi R, Aguado G, Otero-Ferrer JL, Santschi L, Lombarte A et al. Otolith patterns of rockfishes from the northeastern pacific. J Morphol. 2015; 276(4):458–69. https://doi.org/10.1002/jmor.20353
Tuset VM, Lombarte A, Assis CA. Otolith atlas for the western Mediterranean, north and central eastern Atlantic. Sci Mar. 2008; 72(S1):7–198. https://doi.org/10.3989/scimar.2008.72s17
Tuset VM, Lombarte A, González JA, Pertusa JF, Lorente MAJ. Comparative morphology of the sagittal otolith in Serranus spp. J Fish Biol. 2003; 63(6):1491–504. https://doi.org/10.1111/j.1095-8649.2003.00262.x
Tuset VM, Olivar MP, Otero-Ferrer JL, López-Pérez C, Hulley PA, Lombarte A. Morpho-functional diversity in Diaphus spp. (Pisces: Myctophidae) from the central Atlantic Ocean: Ecological and evolutionary implications. Deep Sea Res 1 Oceanogr Res Pap. 2018; 138:46–59. https://doi.org/10.1016/j.dsr.2018.07.005
Vignon M. Ontogenetic trajectories of otolith shape during shift in habitat use: Interaction between otolith growth and environment. J Exp Mar Bio Ecol. 2012; 420–421:26–32. https://doi.org/10.1016/j.jembe.2012.03.021
Volpedo A, Echeverría DD. Ecomorphological patterns of the sagitta in fish on the continental shelf off Argentine. Fish Res. 2003; 60(2–3):551–60. https://doi.org/10.1016/S0165-7836(02)00170-4
Volpedo AV, Tombari AD, Echeverría DD. Eco-morphological patterns of the sagitta of Antarctic fish. Polar Biol. 2008; 31(5):635–40. https://doi.org/10.1007/s00300-007-0400-1
Volpedo AV, Vaz-dos-Santos AM, editors. Métodos de estudios con otolitos: principios y aplicaciones. Ciudad Autónoma de Buenos Aires: CAFP-BA-PIESCI; 2015.
Walker SPW, McCormick MI. Otolith-check formation and accelerated growth associated with sex change in an annual protogynous tropical fish. Mar Ecol Prog Ser. 2004; 266:201–12. https://doi.org/10.3354/meps266201
Walker SPW, McCormick MI. Fish ears are sensitive to sex change. Biol Lett. 2009a; 5(1):73–76. https://doi.org/10.1098/rsbl.2008.0555
Walker SPW, McCormick MI. Sexual selection explains sex-specific growth plasticity and positive allometry for sexual size dimorphism in a reef fish. Proc R Soc Lond B Biol Sci. 2009b; 276(1671):3335–43. https://doi.org/10.1098/rspb.2009.0767
Warner RR. The adaptive significance of sequential hermaphroditism in animals. Am Nat. 1975; 109(965):61–82. https://doi.org/10.1086/282974
Wilson KL, Honsey AE, Moe B, Venturelli P. Growing the biphasic framework: Techniques and recommendations for fitting emerging growth models. Methods Ecol Evol. 2018; 9(4):822–33. https://doi.org/10.1111/2041-210X.12931
Young JM, Yeiser BG, Whittington JA, Dutka-Gianelli J. Maturation of female common snook Centropomus undecimalis: Implications for managing protandrous fishes. J Fish Biol. 2020; 97(5):1317–31. https://doi.org/10.1111/jfb.14475
Zar JH. Biostatistical analysis. 5th ed. Essex: Pearson; 2014.
Zischke MT, Litherland L, Tilyard BR, Stratford NJ, Jones EL, Wang Y-G. Otolith morphology of four mackerel species (Scomberomorus spp.) in Australia: Species differentiation and prediction for fisheries monitoring and assessment. Fish Res. 2016; 176:39–47. https://doi.org/10.1016/j.fishres.2015.12.003
Authors
![]() Reginaldo Medeiros1
Reginaldo Medeiros1 ![]() ,
, ![]() Cicero D. Oliveira2,
Cicero D. Oliveira2, ![]() Daniele Souto1,
Daniele Souto1, ![]() Jordana Rangely1 and
Jordana Rangely1 and ![]() Nídia N. Fabré1
Nídia N. Fabré1
[1] Laboratório de Ecologia, Peixes e Pesca, Instituto de Ciências Biológicas e da Saúde, Universidade Federal de Alagoas, Av. Lourival Melo Mota, Tabuleiro do Martins, 57072-900 Maceió, AL, Brazil. (RM) reginaldo.medeiros@outlook.com.br (corresponding author), (DS) danielesoutovieira@gmail.com, (JR) jordanarangely@gmail.com, (NNF) nidia.fabre59@gmail.com.
[2] Laboratório de Conservação e Manejo de Recursos Renováveis, Instituto de Ciências Biológicas e da Saúde, Universidade Federal de Alagoas, Av. Lourival Melo Mota, Tabuleiro do Martins, 57072-900 Maceió, AL, Brazil. (CDO) linsdiogoc@gmail.com.
Authors Contribution 
Reginaldo Medeiros: Conceptualization, Data Curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Writing-original draft, Writing-review and editing.
Cicero Diogo Oliveira: Conceptualization, Data Curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Daniele Souto: Conceptualization, Data Curation, Formal analysis, Investigation, Methodology, Writing-original draft, Writing-review and editing.
Jordana Rangely: Conceptualization, Formal analysis, Investigation, Methodology, Project administration, Writing-original draft, Writing-review and editing.
Nídia Noeme Fabré: Conceptualization, Data Curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing-original draft, Writing-review and editing.
Ethical Statement
The research was carried out in accordance with the principles of the Basel Declaration and following the recommendations of Brazilian Animal Protection Law. Sampling procedures were approved by the Sistema de Autorização e Informação em Biodiversidade (SISBIO committee) under the license #56293–1.
Competing Interests
The authors declare no competing interests.
How to cite this article
Medeiros R, Oliveira CD, Souto D, Rangely J, Fabré NN. Growth stanza in fish life history using otoliths shape: the protandric Centropomus case (Carangaria: Centropomidae). Neotrop Ichthyol. 2021; 19(4):e200145. https://doi.org/10.1590/1982-0224-2020-0145
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted September 18, 2021 by Ana Petry
Accepted September 18, 2021 by Ana Petry
![]() Submitted December 18, 2020
Submitted December 18, 2020
![]() Epub November 30, 2021
Epub November 30, 2021