João D. Ferraz1,2![]()
![]() , Armando C. R. Casimiro1,2
, Armando C. R. Casimiro1,2![]() , Diego A. Z. Garcia2
, Diego A. Z. Garcia2 ![]() , Alan D. Pereira2
, Alan D. Pereira2![]() , Lucas R. Jarduli2,3
, Lucas R. Jarduli2,3![]() , Fernanda S. de Almeida4
, Fernanda S. de Almeida4![]() and Mário L. Orsi1,2
and Mário L. Orsi1,2![]()
PDF: EN XML: EN | Cite this article
Abstract
We evaluated the fish composition and ecological attributes of the ichthyofauna collected in a limnological zone of the Taquaruçu Reservoir, lower Paranapanema River. Information about the fish community was updated when compared to the previous study (2006). Non-metric multidimensional scaling (NMDS) showed differences in species composition between periods and community weighted means (CWMs) exhibited changes in functional composition over time. Four functional indices were used in the principal coordinate analysis (PcoA) to measure changes in the functional space of species, whereas functional β-diversity inspected differences in the traits composition between the periods. 1,203 individuals were sampled of 43 species, being 16 non-native and 14 new records. Compared to 2006, 27 species were absent, most of them native to Loricariidae and Anostomidae, while Curimatidae and Pimelodidae decreased in abundance. Functional indexes showed a reduction in functional diversity, whereas new species records exhibited functional redundancy. It might have occurred a simplification of the fish community over time, excluding the migratory and specialists species such as the herbivores and detritivores. Accordingly, we concluded that the ichthyofauna of the Taquaruçu Reservoir might have been undergoing a process towards biotic homogenization.
Keywords: Biological invasion, Damming, Homogenization, Long Term Changes, South America.
Avaliamos a composição de peixes e atributos ecológicos da ictiofauna coletada em uma zona limnológica do reservatório de Taquaruçu, baixo rio Paranapanema. As informações sobre a comunidade de peixes foram atualizadas quando comparadas ao estudo anterior (2006). O escalonamento multidimensional não métrico (NMDS) mostrou diferenças na composição de espécies entre os períodos, e as médias ponderadas da comunidade (CWMs) exibiram mudanças na composição funcional ao longo do tempo. Quatro índices funcionais foram utilizados na análise de coordenadas principais (PcoA) para medir as mudanças no espaço funcional das espécies, enquanto a β-diversidade funcional inspecionou diferenças na composição de traços entre os períodos. Foram amostrados 1,203 indivíduos de 43 espécies, sendo 16 não-nativas e 14 novos registros. Em comparação com 2006, 27 espécies estavam ausentes, a maioria nativa de Loricariidae e Anostomidae, enquanto Curimatidae e Pimelodidae diminuíram em abundância. Os índices funcionais mostraram redução na diversidade funcional, enquanto novos registros de espécies exibiram redundância funcional. Pode ter ocorrido uma simplificação da comunidade de peixes ao longo do tempo, excluindo as espécies migradoras e especialistas como os herbívoros e detritívoros. Dessa forma, concluímos que a ictiofauna do reservatório de Taquaruçu pode estar sofrendo um processo de homogeneização biótica.
Palavras-chave: América do Sul, Barramento, Homogeneização, Invasão biológica, Mudanças de longo prazo.
Introduction
Studies based on ichthyofauna surveys are relevant to unveil biodiversity patterns (Langeani et al., 2007) and the effects of anthropogenic impacts on communities (Orsi, Britton, 2014; Loures, Pompeu, 2019; Ganassin et al., 2021). Nevertheless, studies based only on the taxonomic composition of communities may not cover all aspects of biodiversity (Mouchet et al., 2010). Currently, the functional facet of communities has been used as a complementary measure of biodiversity (Villéger et al., 2017). The use of functional traits (i.e., any measurable biological characteristic that might influence the environmental fitness of species) has become key information to disentangle the aspects of functional ecology (Villéger et al., 2017; Pimiento et al., 2020). Thus, the investigation of species organized in a multidimensional functional space can be a useful approach (Villéger et al., 2008), resulting in a reliable source to evaluate biodiversity patterns and mechanisms (Mouillot et al., 2013). This approach also provides a powerful tool when it comes to change and loss of biodiversity due to human activities (Toussaint et al., 2018; McLean et al., 2019), as well as the dynamics of native and non-native species (Olden et al., 2006; Pool et al., 2010). Hence, functional diversity indexes can be used to compare biodiversity patterns across spatial and temporal scales (Mouchet et al., 2010; Pimiento et al., 2020).
The upper Paraná River basin is known to be exploited by hydropower (Agostinho et al., 2007), regardless of its importance for hydrological and ichthyofaunistic aspects (Langeani et al., 2007). The biotic-abiotic interactions in reservoirs are classified in distinct temporal phases (Agostinho et al., 1999), and these interactions may act as an environmental filter on the composition and structure of fish communities (Orsi, Britton, 2014; Smith et al., 2018). Reservoirs might select fish individuals with biological characteristics suitable for such environments (Oliveira et al., 2018), where some species may be non-natives (Olden et al., 2006; Pool et al., 2010). Thus, a taxonomic and/or functional simplification of the ichthyofauna may occur, as non-adapted native species, such as dourado Salminus brasiliensis (Cuvier, 1816) and pintado Pseudoplatystoma corruscans (Spix & Agassiz, 1829) decline and/or disappear over time (Arantes et al., 2019). Therefore, reservoirs should become faunistically similar due to a biotic homogenization process (Daga et al., 2020; Magalhães et al., 2020). In this sense, studies on the structure and composition of ichthyofauna in reservoirs across time are essential to understand the impact of human activity on fish communities (Loures, Pompeu, 2019; Ganassin et al., 2021).
The Paranapanema River, one of the major tributaries of the upper Paraná River (Agostinho et al., 2007), is heavily fragmented by dams into a cascade of 11 reservoirs (Duke Energy, 2008), and it is as well highly impacted by invasive species (Garcia et al., 2018; Pelicice et al., 2018; Jarduli et al., 2020). Biological invasions in this basin are mainly influenced by the flooding of a natural barrier by the Itaipu Reservoir (e.g., Sete Quedas Falls) (Júlio Junior et al., 2009). Also, stocking programs (Agostinho et al., 2007), aquaculture escapes (Casimiro et al., 2018), sporting angling and aquarium dumping (Langeani et al., 2007; Jarduli et al., 2020) have all negatively affects this area (Garcia et al., 2018; Pelicice et al., 2018). The Taquaruçu Reservoir is the penultimate one in a series of reservoirs in the lower Paranapanema River (Duke Energy, 2008). This reservoir belongs to the portion of the basin with the largest amount of non-native species, such as the South American silver croaker Plagioscion squamosissimus (Heckel, 1840) and the armored catfish Loricariichthys platymetopon Isbrücker & Nijssen, 1979 (Garcia et al., 2018), and it is known to be under additional anthropogenic activities like deforestation and discharge of human effluents (Vidotto-Magnoni et al., 2015).
The ichthyofauna of the Taquaruçu Reservoir was widely investigated more than ten years ago (data ranging from 1993 to 2000) (Britto, Carvalho, 2006). Based on knowledge about the dynamics in reservoirs and its implications to the ichthyofauna, we aimed here to update the previous information on the ichthyofauna (i.e., Britto, Carvalho, 2006), and to elucidate changes overtime in the taxonomic and functional composition of the fish community. Our hypotheses were coined after the damming period, as follows: (1) The taxonomic and functional composition of the fish community changed between the distinct time-periods analyzed; (2) Generalist species that are functionally similar, mostly non-natives, are favored over specialist native species between the distinct time-periods.
Material and methods
Study area. The Paranapanema River rises in the Atlantic Plateau of the “Serra de Paranapiacaba”, municipality of Capão Bonito, in the São Paulo State, Southeastern Brazil (Sampaio, 1944). It runs through approximately 930 km until it flows into the upper Paraná River (Sampaio, 1944). Extending from the Southeast of SP to the North of the Paraná State, about 330 km from its main channel define the borders between these Brazilian states (Maack, 1981). The lower Paranapanema River is the portion of the basin that begins after Salto Grande Falls (currently flooded by the Salto Grande Reservoir), covering in extension (from upstream to downstream) the reservoirs of Salto Grande, Canoas II, Canoas I, Capivara, Taquaruçu (our study area), and Rosana (Duke Energy, 2008).
The hydroelectric power plant “Escola Politécnica” (Taquaruçu Dam) was built in 1989 and started to operate in 1991. The Taquaruçu Reservoir is characterized by a run-of-river regime, a length of 80 km, a surface area of 105.5 km2, and a maximum depth of 18 m (Britto, Carvalho, 2006). This reservoir has few tributaries, and among them, the most important ones are located on the left bank, following: the Capim, Centenário, and Tenente rivers; and on the right bank: the Anhumas River. Nonetheless, a large part of those tributaries is undergoing several impacts due to anthropogenic activities, such as riparian forest deforestation, land use (agriculture and pasture), urbanization, and discharge of domestic and industrial effluents (Vidotto-Magnoni et al., 2015).
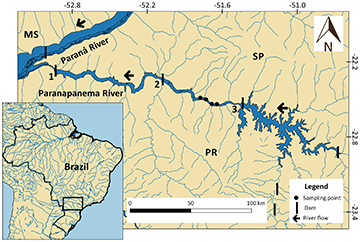
Samplings sites. Samplings were performed from September 2018 to September 2019 at four sites in the reservoir transitional zone: site 1 (22°39’15.0”S 51°40’42.0”W); site 2 (22°39’37.0”S 51°37’53.8”W); site 3 (22°41’24.7”S 51°34’50.5”W); site 4 (22°41’10.8”S 51°32’20.9”W) (Fig. 1). The transitional zone was determined according to Ward et al. (1999) to survey species that inhabit lacustrine and fluvial zones. Following the approach used by Britto, Carvalho (2006), our samplings were standardized and carried out quarterly, where fish were caught using gill-nets (30 to 120 mm between opposed knots), which were set for 24 h and checked every 12 h.
Fish were anesthetized and euthanized by overexposure to 1g/ml clove oil, fixed with 10% formalin, and further transferred to 70% alcohol. The fishes identification were carried out using specific literature (Ota et al., 2018), and with the help of a specialist (Dr. Fernando C. Jerep, Museu de Zoologia da Universidade Estadual de Londrina, MZUEL). Further, a collection of voucher specimens was deposited at MZUEL. As for the previous data (Britto, Carvalho, 2006), all species captured in the reservoir transitional zone and their abundances were selected. Subsequently, species were organized according to Fricke et al. (2021).
Functional traits. Ten functional traits were selected to cover the largest spectrum possible of the fish community functional space. Hence, traits selected here were associated with habitat occupation, feeding habits, life-history, and reproductive strategy (Villéger et al., 2017).
For habitat occupation, we used the following range of adult maximum body size: small size (≤ 200 mm), medium size (≥ 200 – 400 ≤ mm), and large size (≥ 400 mm) (Ota et al., 2018). Regarding the water column position, the following classification was utilized: benthonic, demersal, and pelagic (Graça, Pavanelli, 2007). Classification in trophic guilds was used as feeding habit traits (i.e., detritivore, herbivore, insectivore, invertivore, omnivore, and piscivore) (Hahn et al., 2002; Vidotto-Magnoni, 2009). As traits associated with life-history, our classification followed: periodic, equilibrium, opportunistic, or intermediate (Winemiller, 1995). Finally, for traits related to reproductive strategies, we utilized: migratory behavior (non-migratory, short-distance migratory, long-distance migratory); fertilization (internal or external); parental care (parental care or non-parental care) (Agostinho et al., 2003); spawning (single or multiple); oocyte diameter (mm), and fecundity (number of oocytes matured/gonad mature) (Vazzoler, 1996; Orsi,2010; Froese, Pauly, 2020).
FIGURE 1 | Location of the samplings points in the Taquaruçu Reservoir, lower Paranapanema River (transitional zone, 2020). Hydroelectric power plants: 1– Rosana; 2– Taquaruçu; 3– Capivara. MS = Mato Grosso do Sul State; PR = Paraná State; SP = São Paulo State.
Functional indexes. The distribution of species and their abundance in the functional space characterize functional diversity (Mouillot et al., 2013). Here, we evaluated functional diversity based on four indexes: richness, evenness, divergence, and dispersion. Functional richness (FRic) was defined as the position occupied by species in the functional space (Villéger et al., 2008), while functional evenness (FEve) was based on the uniformity of their abundance in this same space (Mouillot et al., 2013). Functional divergence (FDiv) was calculated as the proportional abundance that was concentrated in species occupying the extremes of the functional space, in the same way as functional dispersion (FDis) was calculated by weighting the average distance of abundance for the extreme traits in this same space (Mouillot et al., 2013).
Statistical analyses. Non-metric multidimensional scaling (NMDS) ordination was employed to elucidate the differences in species composition between the time-periods (i.e., 2006 and 2020). Based on a matrix of distances (e.g., Jaccard method), we verified the degree of similarity between the periods following the species composition data. Thus, in our functional space, we plotted our species composition based on the first two dimensions of the NMDS (final stress level = 0.181). NMDS was run using the function “metaMDS” in the Vegan package (version 2.4–1; Oksanen et al., 2019). Also, to test for significant differences between the groupings of the NMDS, we utilized permutational analysis of variance (PERMANOVA) (Anderson, 2008) using 999 permutations and the function “adonis” in the Vegan package (Oksanen et al., 2019).
We used the community‐weighted mean (CWM) to characterize the functional structure of fish community calculated by each time-period. The CWMs were calculated as the abundance-weighted mean of trait values in the community, which shows an advantage to face the absolute increase or decrease in trait composition (McLean et al., 2019). As a result, the proportions of categorical and continuous trait values can be determined (McLean et al., 2019). For species with no trait data available, we have completed the dataset using the closest phylogenetic equivalent at the genus level (Penone et al., 2014). We assessed changes in the functional structure of the fish community by estimating and comparing the four indexes described above (i.e., richness, evenness, divergence, and dispersion) (Mouillot et al., 2013). When plotted in a two-dimensional functional space, this approach enables the visualization of potential changes in different components of the community’s functional structure, where the axis represents the qualitative traits extracted from a principal coordinates analysis (PCoA) (Mouillot et al., 2013). To verify significant differences in the functional diversity indices between the periods was used a one-tailed test (p < 0.05) proposed by Swenson (2014). We also used functional dissimilarity indexes to assess the changes in the functional β-diversity based on its functional turnover and functional nestedness-resultant components. Hence, in our study, functional β-diversity measured the functional trait composition differences between two distinct periods in time (i.e., 2006 and 2020 datasets) (Mouillot et al., 2013; Villéger et al., 2013).
The multidimensional functional spaces were computed based on a dissimilarity matrix using a principal coordinates analysis (PCoA, a generalization of the principal component analysis (PCA)), which produces a Euclidean space encompassing the two first PCoA axis minus one (Maire et al., 2015). This distance matrix was computed using Gower’s distance (Gower, 1971), allowing thus quantitative and qualitative variables to be mixed while giving them equal weights. Also, we compared the mean squared deviation of the functional spaces. Following the approach devised by Maire et al. (2015), a PCoA was run using this functional distance matrix, and the best functional dendrogram was obtained based on the procedure implemented by Mouchet et al. (2008): using the multidimensional functional spaces from two to seven dimensions, and corresponding to the axis obtained after running the PCoA. Finally, we retained the species coordinates on the first three PCoA axis as the values of the traits (10 traits) describing our fish functional strategies (Villéger et al., 2011; Mouillot et al., 2013). We performed analyses on the functional structure of community using a set of functions: “quality_funct_space”, “plot_funct_space”, “multidimFD”, “multidimFbetaD” (Mouillot et al., 2013; Villéger et al., 2013). All these functions are available from (http://villeger.sebastien.free.fr/Rscripts.html), and analyses were carried out using the R Programming software version 3.5.3 (R Development Core Team, 2020).
Results
We had 1,203 individuals captured belonging to five orders, 17 families, and 43 species. Out of the 43 species, 16 were non-natives to the upper Paraná River basin (Tab. 1). The families showing the greatest richness were: Pimelodidae (seven species), followed by Anostomidae, Cichlidae, and Serrasalmidae (five species each); whereas the most abundant families were Characidae (n = 338), Loricariidae (n = 195), and Sciaenidae (n = 136). Out of the fish composition data obtained by Britto, Carvalho (2006), 27 species were not captured, being most of them native species of Loricariidae (e.g., nine species) and Anostomidae (e.g., six species). Also, 14 species in our results (i.e., mostly non-native species from different families) were new records to the reservoir transitional zone (Tab. 1). The families with a decrease in abundance were Loricariidae, Anostomidae, Curimatidae, and Pimelodidae; whereas Characidae, Cichlidae, and Parodontidae have displayed an increase in abundance (Tab. 1).
TABLE 1 | Ichthyofauna and functional traits of the Taquaruçu Reservoir, lower Paranapanema River (the transitional zone between 2006 and 2020). * = non-native species to the upper Paraná River basin; # = species only recorded in 2006; + = species only recorded in 2020; n = abundance; SI = size (mm); WP = water column position; TG = trophic guild; LH = life-history; MB = migratory behavior; FE = fertilization; PC = parental care; SP = spawning; OD = oocyte diameter (mm); FC = fecundity (number of oocytes matured/gonad mature). SM = small; MD = medium; LA = large; BE = benthonic; DE = demersal; PE = pelagic; DET = detritivore; HER = herbivore; INS = insectivore; INV = invertivore; OMN = omnivore; PIS = piscivore; E = equilibrium; O = opportunistic; P = periodic; NM = non-migratory; SDM = short-distance migratory; LDM = long-distance migratory; EF = external fertilization; IF = internal fertilization; PC = parental care; NPC = non-parental care; MU = Multiple; SI = single. P&DII 1092 = field number, MZUEL voucher uncataloged.
| Family/Species | n | Functional traits | Vouchers | ||||||||||
| 2006 | 2020 | SI | WP | TG | LH | MB | FE | PC | SP | OD | FC | ||
| Erythrinidae | |||||||||||||
| Hoplias malabaricus (Bloch, 1794) | 35 | 20 | LA | DE | PIS | E | NM | EF | PC | MU | 1.3085 | 43972.96 | MZUEL 20741 |
| Hoplias sp.+ | 0 | 20 | LA | DE | PIS | E | NM | EF | PC | MU | 1.3085 | 43972.96 | MZUEL 20761 |
| Parodontidae | |||||||||||||
| Apareiodon affinis (Steindachner, 1879) | 13 | 44 | SM | DE | DET | P | SDM | EF | NPC | SI | 0.8900 | 4345.82 | MZUEL 20770 |
| Cynodontidae | |||||||||||||
| Rhaphiodon vulpinus Spix & Agassiz, 1829# | 5 | 0 | LA | PE | PIS | P | LDM | EF | NPC | SI | 0.9800 | 215841 | – |
| Serrasalmidae | |||||||||||||
| Metynnis lippincottianus (Cope, 1870)* | 4 | 33 | SM | PE | HER | P | SDM | EF | NPC | MU | 1.0783 | 11990.53 | MZUEL 20737 |
| Myloplus levis (Eigenmann & McAtee, 1907)*+ | 0 | 1 | ME | PE | HER | P | SDM | EF | NPC | SI | 2.3260 | 7000 | MZUEL 20783 |
| Myloplus tiete (Eigenmann & Norris, 1900)# | 26 | 0 | ME | PE | HER | P | SDM | EF | NPC | SI | 2.3260 | 7000 | – |
| Serrasalmus maculatus Kner, 1858 | 41 | 21 | ME | PE | PIS | E | NM | EF | PC | MU | 1.4078 | 34177.77 | MZUEL 20743 |
| Serrasalmus marginatus Valenciennes, 1837* | 67 | 40 | ME | PE | PIS | EP | NM | EF | PC | MU | 1.2339 | 68082.19 | MZUEL 20744 |
| Anostomidae | |||||||||||||
| Leporellus vittatus (Valenciennes, 1850)# | 10 | 0 | SM | BE | INS | P | SDM | EF | NPC | SI | 1.0300 | 12745.39 | – |
| Leporinus amblyrhynchus Garavello & Britski, 1987# | 2 | 0 | SM | DE | INS | P | SDM | EF | NPC | MU | 1.0300 | 12745.39 | – |
| Leporinus friderici (Bloch, 1794) | 42 | 1 | LA | DE | OMN | P | SDM | EF | NPC | SI | 0.6870 | 244593.6 | MZUEL 20788 |
| Leporinus lacustris Amaral Campos, 1945# | 1 | 0 | SM | BE | HER | P | SDM | EF | NPC | MU | 1.1717 | 62038.11 | – |
| Leporinus octofasciatus Steindachner, 1915 | 6 | 1 | ME | DE | OMN | P | SDM | EF | NPC | MU | 1.0400 | 13308.99 | MZUEL 20792 |
| Leporinus paranensis Garavello & Britski, 1987# | 3 | 0 | LA | DE | OMN | P | SDM | EF | NPC | SI | 0.8900 | 130527.35 | – |
| Megaleporinus obtusidens (Valenciennes, 1837) | 2 | 5 | LA | DE | INS | P | LDM | EF | NPC | SI | 0.6751 | 846826.4 | MZUEL 20745 |
| Schizodon altoparanae Garavello & Britski, 1990# | 3 | 0 | ME | DE | HER | P | SDM | EF | NPC | MU | 0.7545 | 541605.3 | – |
| Schizodon borellii (Boulenger, 1900)*# | 10 | 0 | LA | DE | HER | P | LDM | EF | NPC | MU | 0.8807 | 541605.3 | – |
| Schizodon intermedius Garavello & Britski, 1990 | 50 | 11 | SM | DE | HER | P | SDM | EF | NPC | SI | 1.0200 | 632157.6 | MZUEL 20797 |
| Schizodon nasutus Kner, 1858 | 83 | 32 | LA | DE | HER | P | SDM | EF | NPC | MU | 1.2657 | 265214.6 | MZUEL 20758 |
| Curimatidae | |||||||||||||
| Cyphocharax modestus (Fernández-Yépez, 1948) | 16 | 1 | SM | DE | DET | P | SDM | EF | NPC | MU | 0.6800 | 15948.4 | MZUEL 20793 |
| Cyphocharax naegelii (Steindachner, 1881)# | 8 | 0 | SM | DE | DET | P | SDM | EF | NPC | SI | 0.5600 | 4787.25 | – |
| Prochilodus lineatus (Valenciennes, 1837)# | 5 | 0 | LA | DE | DET | P | LDM | EF | NPC | SI | 0.9800 | 1280722 | – |
| Steindachnerina brevipinna (Eigenmann & Eigenmann, 1889)*+ | 0 | 17 | SM | DE | DET | P | SDM | EF | NPC | SI | 0.4874 | 56353.49 | MZUEL 20776 |
| Steindachnerina insculpta (Fernández-Yépez, 1948) | 253 | 7 | SM | DE | DET | P | SDM | EF | NPC | SI | 0.4637 | 33503.14 | MZUEL 20746 |
| Triportheidae | |||||||||||||
| Triportheus nematurus (Kner, 1858)*+ | 0 | 1 | SM | PE | INS | P | SDM | EF | NPC | MU | 0.9700 | 10953.4 | MZUEL 20756 |
| Acestrorhynchidae | |||||||||||||
| Acestrorhynchus lacustris (Lütken, 1875) | 90 | 33 | ME | PE | PIS | P | NM | EF | NPC | MU | 0.7248 | 25993.55 | MZUEL 20777 |
| Characidae | |||||||||||||
| Astyanax lacustris (Lütken ,1875) | 84 | 99 | SM | PE | INS | P | SDM | EF | PC | MU | 0.6800 | 7442.77 | MZUEL 20753 |
| Moenkhausia intermedia Eigenmann, 1908 | 23 | 100 | SM | PE | INS | OP | NM | EF | NPC | SI | 0.7100 | 18949.21 | MZUEL 20790 |
| Roeboides descalvadensis Fowler, 1932* | 12 | 139 | SM | PE | INS | P | NM | EF | NPC | MU | 0.9200 | 280 | MZUEL 20772 |
| Sternopygidae | |||||||||||||
| Eigenmannia virescens (Valenciennes, 1836)# | 7 | 0 | ME | BE | INS | E | NM | EF | PC | MU | 1.9000 | 531.96 | – |
| Gymnotidae | |||||||||||||
| Gymnotus inaequilabiatus (Valenciennes, 1839)*+ | 0 | 13 | LA | BE | INS | E | NM | EF | PC | MU | 1.7491 | 10360.15 | MZUEL 20748 |
| Rhamphichthyidae | |||||||||||||
| Rhamphichthys hahni (Meinken, 1937)*+ | 0 | 1 | LA | BE | INS | E | NM | EF | NPC | MU | 3.0000 | 1085 | MZUEL 20759 |
| Rhamphichthys sp.# | 2 | 0 | LA | BE | INS | E | NM | EF | NPC | MU | 3.0000 | 1085 | – |
| Loricariidae | |||||||||||||
| Hypostomus ancistroides (Ihering, 1911)# | 11 | 0 | ME | BE | DET | E | NM | EF | PC | MU | 3.2900 | 420.45 | MZUEL 20778 |
| Hypostomus margaritifer (Regan, 1908)# | 1 | 0 | ME | BE | DET | E | NM | EF | PC | MU | 3.3325 | 1326.387 | – |
| Hypostomus regani (Ihering, 1905)# | 9 | 0 | ME | BE | DET | E | NM | EF | PC | MU | 3.3325 | 1326.387 | – |
| Hypostomus sp. | 2 | 13 | ME | BE | DET | E | NM | EF | PC | MU | 3.2900 | 420.45 | MZUEL 20749 |
| Hypostomus sp2.# | 5 | 0 | ME | BE | DET | E | NM | EF | PC | MU | 3.2900 | 420.45 | – |
| Hypostomus sp4.# | 15 | 0 | ME | BE | DET | E | NM | EF | PC | MU | 3.2900 | 420.45 | – |
| Hypostomus sp6.# | 34 | 0 | ME | BE | DET | E | NM | EF | PC | MU | 3.2900 | 420.45 | – |
| Loricaria sp.# | 19 | 0 | ME | BE | DET | E | NM | EF | PC | SI | 4.2800 | 429.2 | – |
| Loricariichthys platymetopon Isbrücker & Nijssen, 1979* | 175 | 170 | ME | BE | DET | E | NM | EF | PC | MU | 2.3200 | 1451 | MZUEL 20738 |
| Megalancistrus parananus (Peters, 1881)# | 1 | 0 | LA | BE | DET | E | NM | EF | PC | SI | 4.2900 | 13022 | – |
| Proloricaria prolixa (Isbrücker & Nijssen, 1978) | 13 | 12 | ME | BE | DET | E | NM | EF | PC | SI | 4.2800 | 429.2 | MZUEL 20728 |
| Rhinelepis aspera Spix & Agassiz, 1829# | 50 | 0 | LA | BE | DET | P | LDM | EF | NPC | SI | 1.3200 | 181200 | – |
| Auchenipteridae | |||||||||||||
| Auchenipterus nuchalis (Spix & Agassiz, 1829)*# | 53 | 0 | ME | DE | INS | P | NM | IF | NPC | MU | 1.3200 | 2466 | – |
| Auchenipterus osteomystax (Miranda Ribeiro, 1918)*+ | 0 | 40 | ME | DE | INS | P | NM | IF | PNC | MU | 1.3200 | 2466 | MZUEL 20739 |
| Tatia neivai (Ihering, 1930)+ | 0 | 1 | SM | DE | INV | P | SDM | IF | PC | SI | 1.6700 | 297.24 | MZUEL 20794 |
| Trachelyopterus galeatus (Linnaeus, 1766)* | 96 | 38 | ME | DE | OMN | P | NM | IF | NPC | MU | 1.9000 | 750 | MZUEL 20755 |
| Doradidae | |||||||||||||
| Ossancora eigenmanni (Boulenger, 1895)*+ | 0 | 5 | SM | BE | 0 | P | NM | EF | NPC | MU | 0.9000 | 22400.1 | MZUEL 20779 |
| Pterodoras granulosus (Valenciennes, 1821)*# | 8 | 0 | LA | DE | PIS | P | LDM | EF | NPC | MU | 1.0600 | 296000 | – |
| Trachydoras paraguayensis (Eigenmann & Ward, 1907)* | 43 | 9 | P1 | BE | INV | P | NM | EF | NPC | MU | 0.9000 | 22400.1 | MZUEL 20767 |
| Pimelodidae | |||||||||||||
| Iheringichthys labrosus (Lütken, 1874) | 81 | 16 | ME | DE | INV | P | SDM | EF | NPC | MU | 1.1200 | 10954.81 | MZUEL 20747 |
| Pimelodella gracilis (Valenciennes, 1835)+ | 0 | 4 | ME | DE | INS | P | SDM | EF | NPC | MU | 0.6400 | 10955 | MZUEL 20789 |
| Pimelodella sp.# | 6 | 0 | ME | DE | INS | P | SDM | EF | NPC | MU | 0.6400 | 10955 | – |
| Pimelodus maculatus Lacepède, 1803 | 87 | 34 | LA | DE | OMN | P | LDM | EF | NPC | SI | 0.5860 | 76184.2 | MZUEL 20768 |
| Pimelodus ornatus Kner, 1858* | 5 | 1 | LA | DE | OMN | P | SDM | EF | NPC | SI | 0.3952 | 54677.58 | MZUEL 20765 |
| Pimelodus sp.+ | 0 | 1 | LA | DE | OMN | P | LDM | EF | NPC | SI | 0.5860 | 76184.2 | P&DII 1092 |
| Pinirampus pirinampu (Spix & Agassiz, 1829) | 13 | 1 | LA | DE | PIS | P | LDM | EF | NPC | SI | 1.1800 | 189858.2 | MZUEL 20740 |
| Sorubim lima (Bloch & Schneider, 1801)*+ | 0 | 1 | LA | DE | PIS | P | LDM | EF | NPC | SI | 1.5400 | 1200000 | MZUEL 20769 |
| Zungaro jahu (Ihering, 1898)# | 4 | 0 | LA | DE | PIS | P | LDM | EF | NPC | SI | 1.6000 | 862405 | – |
| Cichlidae | |||||||||||||
| Cichla cf. kelberi Kullander & Ferreira, 2006*+ | 0 | 10 | LA | DE | PIS | E | NM | EF | PC | MU | 1.6087 | 15694.12 | MZUEL 20760 |
| Crenicichla britskii Kullander, 1982 | 19 | 10 | SM | DE | INS | E | NM | EF | PC | SI | 2.0500 | 955.46 | MZUEL 20752 |
| Crenicichla jaguarensis Haseman, 1911# | 0 | 1 | SM | DE | INS | E | NM | EF | PC | SI | 2.0500 | 955.46 | MZUEL 20784 |
| Crenicichla niederleinii (Holmberg, 1891)# | 11 | 0 | SM | DE | INS | E | NM | EF | PC | SI | 2.9200 | 562.91 | – |
| Crenicichla sp. | 17 | 57 | SM | DE | INS | E | NM | EF | PC | SI | 2.0500 | 955.46 | MZUEL 20764 |
| Geophagus iporangensis Haseman, 1911+ | 0 | 3 | SM | DE | INS | E | NM | EF | PC | MU | 1.7800 | 1539 | MZUEL 20785 |
| Sciaenidae | |||||||||||||
| Plagioscion squamosissimus (Heckel, 1840)* | 131 | 136 | LA | PE | PIS | P | SDM | EF | NPC | MU | 0.5300 | 108314.1 | MZUEL 20742 |
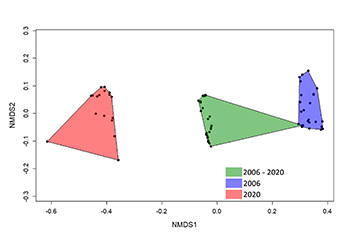
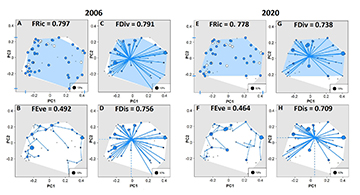
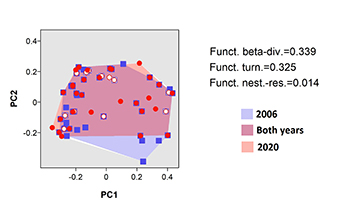
NMDS evidenced a clustering of three groups (i.e., species occurring in both years, species captured exclusively in 2006, and species captured exclusively in 2020), indicating divergences in the fish community over time (Fig. 2). The PERMANOVA confirmed that the species composition in 2006 was significantly distinct from that in 2020 (R2 = 0.22, P = 0.01). The CWMs demonstrated a change in six out of ten traits between the datasets of 2006 and 2020, following: size, water column position, trophic guild, migratory behavior, oocyte diameter, and fecundity (Tab. 2). All the functional indexes calculated showed a decreased between 2006 and 2020 datasets (Fig. 3). Changes in functional β-diversity between 2006 and 2020 datasets were observed as well, where the turnover component was almost the total value of total functional β-diversity (Fig. 4).
The results of one-tailed test showed a significant difference in each functional diversity index between the two time-periods. FRic (t = 80.253; P = 0.007), FDiv (t = 28.903; P = 0.02), FEve (t = 33.508; P = 0.01) and FDis (t = 31.17; P = 0.02) showed decreasing trends along of the two time-periods (2006 to 2020) indicating the loss of several functional traits and change in the abundance of species with restricted to specific groups functional traits.
TABLE 2 | Functional composition by community-weighted means (CWMs), and changes in the ichthyofauna of the Taquaruçu Reservoir, lower Paranapanema River (the transitional zone between 2006 and 2020).
| Year | Size (mm) | Water column position | Trophic guild | Life-History | Migratory behavior | Fertili- zation | Parental care | Spawning | Oocyte diameter (mm) | Fecundity (n.oocytes) |
| 2006 | Medium | Demersal | Detritivore | Periodic | Short-Distance migratory | External | Non parental care | Multiple | 2.0128 | 17882.09 |
| 2020 | Small | Pelagic | Piscivore | Periodic | Non-Migratory | External | Non parental care | Multiple | 1.4783 | 27474.19 |
FIGURE 2 | Non-metric multidimensional scaling (NMDS) of the ichthyofauna composition of the Taquaruçu Reservoir, lower Paranapanema River (the transitional zone between 2006 and 2020).
FIGURE 3 | Two-dimensional functional space (PCoA) of the ichthyofauna of the Taquaruçu Reservoir, lower Paranapanema River (transitional zone between 2006 and 2020). A, B, C, D = 2006. E, F, G, H = 2020. FRic = Functional richness; FEve = Functional evenness; FDiv = Functional divergence; FDis = Functional dispersion.
FIGURE 4 | Two-dimensional functional space (PCoA) of the functional β-diversity of the ichthyofauna of the Taquaruçu Reservoir, lower Paranapanema River (the transitional zone between 2006 and 2020).
Discussion
We observed the absence of several native species that were registered in 2006, especially from the Loricariidae and Anostomidae families, and the reduction in the abundance of Curimatidae and Pimelodidae families. On the other hand, 14 species were new records for this current study. However, most of them are non-native to the lower Paranapanema River. Statistical analysis provided indications that there was a significant taxonomic discrepancy between the time-periods. In this sense, the CWMs of traits demonstrated that six out of ten traits considered here had changed over time, a fact corroborated by the decrease in all functional indexes between the time-periods. Finally, the β-functional diversity showed that the new species were functionally redundant.
Changes in the functional composition of the fish community might be related to rearrangements in the ichthyofauna between the time-periods of 2006 and 2020, which have acted predominantly in the loss of richness in Loricariidae and Anostomidae, and it has also decreased the abundance in Curimatidae and Pimelodidae. Most of the species pertaining to the Anostomidae and Pimelodidae in the upper Paraná River display medium to large body size (Graça, Pavanelli, 2007; Ota et al., 2018), and migratory behavior (Agostinho et al., 2003). As shown in previous studies (e.g., Agostinho et al., 2007; Hoeinghaus et al., 2009), large migratory fish in the upper Paraná River have been harmed, after the damming, by the blocking of movements and the disconnection of critical habitats to life cycles. Besides, the dam’s water level control neutralizes seasonal floods, which restrains the connection with the marginal lagoons utilized by early life stages to hide, develop, feed, and grow, thus leading to population depletion (Agostinho et al., 2004; Arantes et al., 2019). In this sense, several studies have pointed out a graduate occupation of the reservoirs by opportunistic species of smaller body size (Alvim, Peret, 2004; Lima et al., 2016; Arantes et al., 2019).
Changes in body size and position in the water column due to anthropogenic impacts have been registered in marine ecosystems (e.g., continental shelves of the Atlantic Ocean), where smaller and pelagic fish with short life cycles are favored due to reply faster to environmental changes (McLean et al., 2019). In the Paraná River, damming has caused changes in the composition of ichthyofauna, varying from pelagic to benthopelagic habitat usage (Oliveira et al., 2018), whereas we have found a different pattern (from demersal to pelagic usage of habitat). One could find an explanation for this change in the fish feeding habits, based on the fact that much of Anostomidae species feed on plant material and Pimelodidae species show predator/omnivore feeding, both favored by their sub-terminal mouths (Graça, Pavanelli, 2007), and demersal behavior in the bottom (Vidotto-Magnoni, 2009). Since the previous study was performed few years after the dam has closed (Britto, Carvalho, 2006), species belonging to Anostomidae may have been favored by the trophic upsurge period in the early years of the reservoir, where the increase in transparency and carriage of organic matter from terrestrial floods facilitated primary production (Agostinho et al., 1999). However, these individuals were probably overcome by smaller and pelagic fish (except in the case of the large pelagic invasive P. squamosissimus) due to changes in the availability of food as the reservoir aged (Cunha-Santino et al., 2013; Arantes et al., 2019), as well as specialized feeding habits that precluded the use of different resources (Luz-Agostinho et al.,2006). On the other hand, for the Pimelodidae individuals, this may have occurred mildly due to their wide feeding capacity (Vidotto-Magnoni, 2009).
Back to the study done by Britto, Carvalho (2006), the short-distance migratory behavior of fish was dominantly represented by Anostomidae and Pimelodidae. Species of both groups can carry out short-distance migration that might allow them to accomplish their life cycles in reservoirs (Agostinho et al., 2003; Orsi, 2010). However, the short-distance migrator species have been replaced by non-migrators ones, which may be related to the damming (Agostinho et al., 2004; Cunha-Santino et al., 2013). This scenario was also seen in the Colorado River, North America, where the decline by damming of native migrant species provided unoccupied niches to sedentary non-native species occupation (besides other environmental factors) (Olden et al., 2006; Pool et al., 2010). Our results partially corroborate with this trend since we could demonstrate an increase in native and non-native sedentary species belonging to the families: Characidae (e.g., native tetra Moenkhausia intermedia Eigenmann, 1908; and the non-native saicanga Roeboides descalvadensis Fowler, 1932), Parodontidae [e.g., native jackknife Apareiodon affinis (Steindachner, 1879)], and Cichlidae (e.g., native jacundá Crenicichla sp.; and the non-native peacock bass Cichla cf. kelberi Kullander & Ferreira, 2006).
The reduction of the oocyte diameter and the increased fecundity are believed to be guided by environmental forces selecting periodic life-history species (Winemiller, 1995). Therefore, our results denoted a decline of migratory species of this kind. In this way, changes in trait composition might be associated with the loss of Loricariidae species. A previous study in the Paraná River (Suzuki et al., 2000) have shown that species of Loricariidae have large oocytes (> 4.0 mm), while in the Paranapanema River, it was found that they had the largest oocyte diameters among the entire ichthyofauna (> 3.0 mm) (Orsi, 2010). For both studies, this group presented lower fecundity and possibly, an equilibrium trait concerning Life-History (Winemiller, 1995). However, in our study, we did not find changes in the composition of life-history traits. In this sense, it should be noted that there are intermediate characteristics between periodic and equilibrium extremes of life-history, where the opportunistic species fit, which produce oocytes varying in size (e.g., medium to small oocytes) and medium to large offsprings (Winemiller, 1995). Therefore, we could explain the changes in trait composition here without having changes in life-history because the decrease in oocyte diameter and lower fecundity (Loricariidae), together with the increase in little oocytes and larger offsprings (probably influenced by the non-native opportunistic species) were captured in abundance weighted means (CWMs) (McLean et al., 2019).
During the early years of the damming, piscivores species are favored by the increase of preys (Lima et al., 2016), whereas the rise of the detritivores species occurs later due to high sedimentation during the process of the reservoir aging (Cunha-Santino et al., 2013). However, we have observed an opposite pattern. In the reservoirs of the Iguaçu (Delariva et al., 2013), and Tietê rivers (Smith et al., 2018), detritivores species became more abundant when food resources were largely available. Hence, the higher representation of detritivores of Loricariidae and Curimatidae [e.g., the saguiru Steindachnerina insculpta (Fernández-Yépez, 1948)] in the study carried out by Britto, Carvalho (2006) may be associated again with the early stage of the reservoir, which contributed to the concentration of organic matter in the bottom (Agostinho et al., 2007). Thus, as found here to Anostomidae, the decreasing of richness in Loricariidae and abundance in Curimatidae, most likely, occurred due to the trophic dynamics in the reservoir (Cunha-Santino et al., 2013), and fish inefficiency in exploring other food resources since detritivores are also specialists (Luz-Agostinho et al., 2006; Smith et al., 2018). Nevertheless, the detritivores may have also been harmed over time by the low carriage of organic material in the reservoir sediment due to the deforestation in the surrounding areas (Vidotto-Magnoni et al., 2015), or the dam’s water level control (Alvim, Peret, 2004). Besides, benthonic detritivore species (Loricariidae) may have been suffered from low concentrations of oxygen in the bottom of the reservoir (Arantes et al., 2019), while the detritivores species, in general, may have been experienced the effects of the run-of-river regime over time, which might have had minor sedimentation due to short-time water retention and small flooded perimeter (Nogueira et al., 2012). Likewise, the increase of piscivores species was most likely modulated by the progressive niche occupation left by rheophilic predators (e.g., Pimelodidae) (Olden et al., 2006; Pool et al., 2010). Thus, sedentary species such as Cichla cf. kelberi, which were introduced by human-mediated activity (e.g., sporting angling) (Langeani et al., 2007; Jarduli et al., 2020), probably increased their occupancy.
The functional turnover showed that changes in trait composition were mostly driven by non-native species, a pattern that has been observed in distinct reservoirs of the upper Paraná River (e.g., Orsi, Britton, 2014; Loures, Pompeu, 2019; Daga et al., 2020), and others Brazilian watersheds (Arantes et al., 2019; Magalhães et al., 2020; Ganassin et al., 2021). Previously, a global freshwater review found that, in general, non-native species increase the functional richness of fish communities, in some cases beyond 100% (Toussaint et al., 2018). However, the functional turnover did not increase or re-establish ecological functions since there was a decrease in all functional indexes analyzed. Consequently, taxonomic turnover was mediated by trait-environment relationships, where species presented functional redundancy (Villéger et al., 2010; Pimiento et al., 2020). The FRic and FEve indexes decreased over time, meaning that the functional space has been reduced (Villéger et al., 2008; Mouchet et al., 2010), and the uniformity of traits was modified (Mouillot et al., 2013). This finding was supported by the decrease in FDiv index over time, which have indicated that dominant species became functionally closer while the extremes decreased (Mouillot et al., 2013), meaning that species having extreme traits (e.g., largest oocyte diameters) in the functional space became less represented. Hence, the FDis index decrease over time demonstrated that species had been found closer to the center of functional space, where similar characteristics should remain (Villéger et al., 2008). This functional scenario might be justified by the type of environment studied since large watersheds that historically had richer fauna with many ecological functions should become less susceptible to gain traits when non-natives species become established (Toussaint et al., 2018).
The decrease in the FRic index, even with the introduction of non-native species, was verified in previous studies such as in the estuarine fish community in Mexico (Villéger et al., 2010) and in the Paraná River (Oliveira et al., 2018), once the species were functionally redundant. Additionally, a decline of specialized species was observed, that is, species with extreme traits such as herbivory (Villéger et al., 2010), and migratory behavior (Oliveira et al., 2018) have diminished. Thus, the taxonomic and trait composition of fishes under environmental impacts might substantially change across time, yet the fish communities seem to converge to the same characteristics, showing a functional simplification effect that should drive biotic homogenization (McLean et al.,2019; Magalhães et al., 2020). Thus, our results here might enhance and show that the loss of a significant portion of the taxonomic and functional composition in the Taquaruçu Reservoir should probably drive the fish community towards biotic homogenization, an environmental panorama that is most likely subjected to the damming (Loures, Pompeu, 2019; Daga et al., 2020; Ganassin et al., 2021), and biological invasions (Magalhães et al., 2020).
The implications of the environmental scenario found here is quite worrying since fish participate in the nutrient cycle, are considered ecosystem engineers, and control food chains (Villeger et al., 2017; Arantes et al., 2019). Particularly, the Anostomidae, Curimatidae, and Loricariidae families play an important role in food chains since they promote the flow of nutrients and matter from lower to upper food chain levels (Alvim, Peret, 2004; Luz-Agostinho et al., 2006). Thus, herbivores and detritivores species are responsible for mediating most of the flow of matter and energy in large watersheds from South America (Bowen, 1984; Arantes et al., 2019). On the other hand, predators as Pimelodidae species have a relevant position in food chains by promoting the moderation of food chain levels below (Agostinho et al., 2007). In addition, fish are known to perform several ecosystem services (Villeger et al., 2017; Arantes et al., 2019), as the importance of families such as Anostomidae, Curimatidae, and Pimelodidae in fisheries resources (Hoeinghaus et al., 2009), and Loricariidae in fishkeeping (Graça, Pavanelli, 2007).
In conclusion, we identified a substantial loss or decreased over time in fish species (i.e., 2006 to 2020), that have reduced their functional space in the Taquaruçu Reservoir. This scenario might compromise the ecological relationships and ecosystem services in this reservoir. Our results emphasized that reservoirs play an important “filtering” role in structuring fish communities, which should result in a selection of suitable traits driving assemblages to a simplification of characteristics (i.e., traits) and, most likely, to biotic homogenization influenced by non-native species. Here, our results reinforced the impacts of damming in fish communities, especially on migratory and specialist species. Hence, we showed that after thirty years have passed the damming, we could find relevant changes in the fish communities, demonstrating that there is a pressing need for continuous monitoring of the reservoirs. We encourage the practice of this approach for successful biological and ecological assessments and management of Brazilian freshwater ecosystems, as well as increasing knowledge on the dynamics in reservoirs, recognizing thus local and regional environmental issues.
Acknowledgments
This study was funded by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil – finance code 001 to first and second authors. The third and fifth authors would like to thank the CTG Brasil for support in their postdoctoral research at the Universidade Estadual de Londrina (UEL), and the fourth author would also like to thank CTG Brasil for granting financial assistance for this work. Also, we would like to thank Prof. Dr. Fernando C. Jerep (MZUEL) for identifying the species and Aparecido de Souza and Edson S. da Silva for helping with the fieldwork. We also thank the anonymous reviewers and Associate Editor for their valuable comments.
References
Agostinho AA, Miranda LE, Bini LM, Gomes LC, Thomaz SM, Suzuki HI. Patterns of colonization in neotropical reservoirs, and prognoses on aging. In: Tundisi JG, Straskraba M, editors. Theoretical reservoir ecology and its applications. Leiden: Backhuys Publishers; 1999. p.227–65. Available from: http://repositorio.uem.br:8080/jspui/handle/1/5322
Agostinho AA, Gomes LC, Suzuki HI, Júlio Jr HF. Migratory fishes of the upper Paraná River basin, Brazil. In: Carolsfeld J, Harvey B, Ross C, Baer A, editors. Migratory fishes of South America: Biology, fisheries and conservation status. Ottawa: IRDC, The World Bank & World Fisheries Trust; 2003. p.19–98.
Agostinho AA, Gomes LC, Veríssimo S, Okada EK. Flood regime, dam regulation and fish in the upper Paraná River: Effects on assemblage attributes, reproduction and recruitment. Rev Fish Biol Fish. 2004; 14(1):11–19. https://doi.org/10.1007/s11160-004-3551-y
Agostinho AA, Gomes LC, Pelicice FM. Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil. Maringá: EDUEM; 2007.
Alvim MCC, Peret AC. Food resources sustaining the fish fauna in a section of the upper São Francisco River in Três Marias, MG, Brazil. Braz J Biol. 2004; 64(2):195–202. https://doi.org/10.1590/S1519-69842004000200003
Anderson MJ. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2008; 26(1):32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x
Arantes CC, Fitzgerald DB, Hoeinghaus DJ, Winemiller KO. Impacts of hydroelectric dams on fishes and fisheries in tropical rivers through the lens of functional traits. Curr Opin Environ Sustain. 2019; 37:28–40. https://doi.org/10.1016/j.cosust.2019.04.009
Bowen SH. Detritivory in Neotropical fish communities. In: Zaret TM, editor. Evolutionary ecology of Neotropical freshwater fishes. Dordrecht: Springer; 1984. p.59–66. https://doi.org/10.1007/978-94-015-7682-6_4
Britto SGC, Carvalho ED. Ecological attributes of fish fauna in the Taquaruçu Reservoir, Paranapanema River (Upper Paraná, Brazil): composition and spatial distribution. Acta Limnol Bras. 2006; 18(4):377–88. Available from: http://www.ablimno.org.br/acta/pdf/acta_limnologica_contents1804E_files/18(4)_04.pdf
Casimiro ACR, Garcia DAZ, Vidotto-Magnoni AP, Britton JR, Agostinho AA, Almeida FS et al. Escapes of non-native fish from flooded aquaculture facilities: The case of Paranapanema River, southern Brazil. Zoologia. 2018; 35:e14638. https://doi.org/10.3897/zoologia.35.e14638
Cunha-Santino MB, Bitar AL, Bianchini Jr I. Chemical constraints on new man-made lakes. Environ Monit Assess. 2013; 185(12):10177–90. https://doi.org/10.1007/s10661-013-3322-0
Daga VS, Olden JD, Gubiani EA, Piana PA, Padial AA, Vitule JRS. Scale-dependent patterns of fish faunal homogenization in Neotropical reservoirs. Hydrobiologia. 2020; 847(18):3759–72. https://doi.org/10.1007/s10750-019-04145-5
Delariva RL, Hahn NS, Kashiwaqui EAL. Diet and trophic structure of the fish fauna in a subtropical ecosystem: Impoundment effects. Neotrop Ichthyol. 2013; 11(4):891–904. https://doi.org/10.1590/S1679-62252013000400017
Duke Energy. Peixes do rio Paranapanema. São Paulo: Horizonte Geográfico; 2008.
Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: species by family/subfamily [Internet]. San Francisco: California; 2021. Available from: https://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Froese R, Pauly D. FishBase [Internet]. Leiden: Netherlands; 2020. Available from: www.fishbase.org
Ganassin MJM, Muñoz-Mas R, Oliveira FJM, Muniz CM, Santos NCL, García-Berthou E et al. Effects of reservoir cascades on diversity, distribution, and abundance of fish assemblages in three Neotropical basins. Sci Total Environ. 2021; 778:146246. https://doi.org/10.1016/j.scitotenv.2021.146246
Garcia DAZ, Britton JR, Vidotto-Magnoni AP, Orsi ML. Introductions of non-native fishes into a heavily modified river: Rates, patterns and management issues in the Paranapanema River (upper Paraná ecoregion, Brazil). Biol Invasions. 2018; 20(5):1229–41. https://doi.org/10.1007/s10530-017-1623-x
Gower JC. A general coefficient of similarity and some of its properties. Biometrics. 1971; 27(4):857–71. https://doi.org/10.2307/2528823
Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes. Maringá: EDUEM; 2007.
Hahn NS, Fugi R, Peretti D, Russo MR, Loureiro-Crippa VE. Estrutura trófica da ictiofauna da planície de inundação do alto rio Paraná. In: Seeliger U, Cordazzo CV, Barbosa FAR, editors. Os sites e o programa brasileiro de pesquisas ecológicas de longa duração. Belo Horizonte: UFMG; 2002. p.123–26.
Hoeinghaus DJ, Agostinho AA, Gomes LC, Pelicice FM, Okada EK, Latini JD et al. Effects of river impoundment on ecosystem services of large tropical rivers: Embodied energy and market value of artisanal fisheries. Conserv Biol. 2009; 23(5):1222–31. https://doi.org/10.1111/j.1523-1739.2009.01248.x
Jarduli LR, Garcia DAZ, Vidotto-Magnoni AP, Casimiro ACR, Vianna NC, Almeida FS et al. Fish fauna from the Paranapanema River basin, Brazil. Biota Neotrop. 2020; 20(1):e20180707. https://doi.org/10.1590/1676-0611-bn-2018-0707
Júlio Junior HF, Dei To’s C, Agostinho AA, Pavanelli CS. A massive invasion of fish species after eliminating a natural barrier in the upper Paraná River basin. Neotrop Ichthyol. 2009; 7(4):709–18. https://doi.org/10.1590/S1679-62252009000400021
Langeani F, Castro RMC, Oyakawa OT, Shibatta OA, Pavanelli CS, Casatti L. Diversidade da ictiofauna do alto rio Paraná: Composição atual e perspectivas futuras. Biota Neotrop. 2007; 7(3):181–97. https://doi.org/10.1590/S1676-06032007000300020
Lima AC, Agostinho CS, Sayanda D, Pelicice FM, Soares AMVM, Monaghan KA. The rise and fall of fish diversity in a neotropical river after impoundment. Hydrobiologia. 2016; 763(1):207–21. https://doi.org/10.1007/s10750-015-2377-z
Loures RC, Pompeu PS. Temporal changes in fish diversity in lotic and lentic environments along a reservoir cascade. Freshw Biol. 2019; 64(10):1806–20. https://doi.org/10.1111/fwb.13372
Luz-Agostinho KDG, Bini LM, Fugi R, Agostinho AA, Júlio Jr HF. Food spectrum and trophic structure of the ichthyofauna of Corumbá reservoir, Paraná river Basin, Brazil. Neotrop Ichthyol. 2006; 4(1):61–68. https://doi.org/10.1590/S1679-62252006000100005
Maack R. Geografia física do Estado do Paraná. 2nd ed. Rio de Janeiro: José Olympio; 1981.
Magalhães ALB, Daga VS, Bezerra LAV, Vitule JRS, Jacobi CM, Silva LGM. All the colors of the world: Biotic homogenization-differentiation dynamics of freshwater fish communities on demand of the Brazilian aquarium trade. Hydrobiologia. 2020; 847(18):3897–915. https://doi.org/10.1007/s10750-020-04307-w
Maire E, Grenouillet G, Brosse S, Villéger S. How many dimensions are needed to accurately assess functional diversity? A pragmatic approach for assessing the quality of functional spaces. Glob Ecol Biogeogr. 2015; 24(6):728–40. https://doi.org/10.1111/geb.12299
McLean M, Mouillot D, Lindegren M, Villéger S, Engelhard G, Murgier J et al. Fish communities diverge in species but converge in traits over three decades of warming. Glob Change Biol. 2019; 25(11):3972–84. https://doi.org/10.1111/gcb.14785
Mouchet M, Guilhaumon F, Villéger S, Mason NWH, Tomasini JA, Mouillot D. Towards a consensus for calculating dendrogram-based functional diversity indices. Oikos. 2008; 117(5):794–800. https://doi.org/10.1111/j.0030-1299.2008.16594.x
Mouchet MA, Villéger S, Mason NWH, Mouillot D. Functional diversity measures: an overview of their redundancy and their ability to discriminate community assembly rules. Funct Ecol. 2010; 24(4):867–76. https://doi.org/10.1111/j.1365-2435.2010.01695.x
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR. A functional approach reveals community responses to disturbances. Trends Ecol Evol. 2013; 28(3):167–77. https://doi.org/10.1016/j.tree.2012.10.004
Nogueira MG, Perbiche-Neves G, Naliato DAO. Limnology of two contrasting hydroelectric reservoirs (storage and run-of-river) in Southeast Brazil. In: Samadi-Boroujeni H, editor. Hydropower: Practice and application. Rijeka: In Tech; 2012. p.167–84.
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D et al. Vegan: Community Ecology Package [Internet]. R Package version 2.5-6; 2019. Available from: https://cran.r-project.org/package=vegan
Olden JD, Poff NL, Bestgen KR. Life-history strategies predict fish invasions and extirpations in the Colorado River basin. Ecol Monogr. 2006; 76(1):25–40. https://doi.org/10.1890/05-0330
Oliveira AG, Baumgartner MT, Gomes LC, Dias RM, Agostinho AA. Long-term effects of flow regulation by dams simplify fish functional diversity. Freshw Biol. 2018; 63(3):293–305. https://doi.org/10.1111/fwb.13064
Orsi ML. Estratégias Reprodutivas de Peixes: Estratégias reprodutivas de peixes da região média-baixa do rio Paranapanema, reservatório de Capivara. São Paulo: Editora Edgard Blucher Ltda.; 2010.
Orsi ML, Britton JR. Long-term changes in the fish assemblage of a Neotropical hydroelectric reservoir. J Fish Biol. 2014; 84(6):1964–70. https://doi.org/10.1111/jfb.12392
Ota RR, Deprá GC, Graça WJ, Pavanelli CS. Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: Revised, annotated and updated. Neotrop Ichthyol. 2018; 16(2):e170094. https://doi.org/10.1590/1982-0224-20170094
Pelicice FM, Azevedo-Santos VM, Esguícero ALH, Agostinho AA, Arcifa MS. Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotrop Ichthyol. 2018; 16(2):e170150. https://doi.org/10.1590/1982-0224-20170150
Penone C, Davidson AD, Shoemaker KT, Di Marco M, Rondinini C, Brooks TM et al. Imputation of missing data in life-history trait datasets: Which approach performs the best? Methods Ecol Evol. 2014; 5(9):961–70. https://doi.org/10.1111/2041-210X.12232
Pimiento C, Leprieur F, Silvestro D, Lefcheck JS, Albouy C, Rasher DB et al. Functional diversity of marine megafauna in the Anthropocene. Sci Adv. 2020; 6(16):eaay7650. https://doi.org/10.1126/sciadv.aay7650
Pool TK, Olden JD, Whittier JB, Paukert CP. Environmental drivers of fish functional diversity and composition in the Lower Colorado River basin. Can J Fish Aquat Sci. 2010; 67(11):1791–807. https://doi.org/10.1139/F10-095
R Development Core Team. A language and environment for statistical computing [Internet]. Vienna: R Foundation for Statistical Computing; 2020. Available from: https://www.R-project.org
Sampaio T. Relatório sobre os estudos efetuados nos rios Itapetininga e Paranapanema. Rev Inst Geogr Geol. 1944; 2(3):30–81.
Smith WS, Pereira CGF, Espindola ELG, Rocha O. Trophic structure of the fish community throughout the reservoirs and tributaries of the middle and lower Tietê River (São Paulo, Brazil). Acta Limnol Bras. 2018; 30:e308. https://doi.org/10.1590/s2179-975X0618
Suzuki HI, Agostinho AA, Winemiller KO. Relationship between oocyte morphology and reproductive strategy in loricariid catfishes of the Parana River, Brazil. J Fish Biol. 2000; 57(3):791–807. https://doi.org/10.1111/j.1095-8649.2000.tb00275.x
Swenson NG. Functional and phylogenetic ecology in R. New York: Springer; 2014. https://doi.org/10.1007/978-1-4614-9542-0
Toussaint A, Charpin N, Beauchard O, Grenouillet G, Oberdorff T, Tedesco PA et al. Non-native species led to marked shifts in functional diversity of the world freshwater fish faunas. Ecol Lett. 2018; 21(11):1649–59. https://doi.org/10.1111/ele.13141
Vazzoler AEAM. Biologia da reprodução de peixes teleósteos: Teoria e prática. Maringá: EDUEM; 1996.
Vidotto-Magnoni AP. Ecologia trófica das assembléias de peixes do reservatório de Chavantes (Médio rio Paranapanema, SP/PR). [PhD Thesis]. Botucatu: Universidade Estadual Paulista “Júlio de Mesquita Filho”; 2009. Available from: https://repositorio.unesp.br/handle/11449/106518
Vidotto-Magnoni AP, Garcia DAZ, Costa ADA, Souza JG, Yabu MHS, Almeida FS et al. Ichthyofauna of streams of the Lower Paranapanema River basin, state of Paraná, Brazil. Check List. 2015; 11(5):1756. https://doi.org/10.15560/11.5.1756
Villéger S, Mason NWH, Mouillot D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology. 2008; 89(8):2290–301. https://doi.org/10.1890/07-1206.1
Villéger S, Miranda JR, Hernández DF, Mouillot D. Contrasting changes in taxonomic vs. functional diversity of tropical fish communities after habitat degradation. Ecol Appl. 2010; 20(6):1512–22. https://doi.org/10.1890/09-1310.1
Villéger S, Novack-Gottshall PM, Mouillot D. The multidimensionality of the niche reveals functional diversity changes in benthic marine biotas across geological time. Ecol Lett. 2011; 14(6):561–68. https://doi.org/10.1111/j.1461-0248.2011.01618.x
Villéger S, Grenouillet G, Brosse S. Decomposing functional β-diversity reveals that low functional β-diversity is driven by low functional turnover in European fish assemblages. Glob Ecol Biogeogr. 2013; 22(6):671–81. https://doi.org/10.1111/geb.12021
Villéger S, Brosse S, Mouchet M, Mouillot D, Vanni MJ. Functional ecology of fish: Current approaches and future challenges. Aquat Sci. 2017; 79(4):783–801. https://doi.org/10.1007/s00027-017-0546-z
Ward JV, Tockner K, Schiemer F. Biodiversity of floodplain river ecosystems: Ecotones and connectivity. Regul Rivers Res Manage. 1999; 15(1–3):125–39. https://doi.org/10.1002/(SICI)1099-1646(199901/06)15:1/3%3C125::AID-RRR523%3E3.0.CO;2-E
Winemiller KO. Aspects structurels et fonctionnels de la biodiversité des peuplements de poissons. Bull Fr PêchePiscic. 1995; 1995(337–338–339):23–45. https://doi.org/10.1051/kmae:1995007
Authors
JoãoJoão D. Ferraz1,2![]()
![]() , Armando C. R. Casimiro1,2
, Armando C. R. Casimiro1,2![]() , Diego A. Z. Garcia2
, Diego A. Z. Garcia2 ![]() , Alan D. Pereira2
, Alan D. Pereira2![]() , Lucas R. Jarduli2,3
, Lucas R. Jarduli2,3![]() , Fernanda S. de Almeida4
, Fernanda S. de Almeida4![]() and Mário L. Orsi1,2
and Mário L. Orsi1,2![]()
[1] Programa de Pós-Graduação em Ciências Biológicas, Universidade Estadual de Londrina, Centro de Ciências Biológicas, Rodovia Celso Garcia Cid, PR-445, km 380, 86057-970 Londrina, PR, Brazil. (JDF) jd_ferraz@hotmail.com (corresponding author), (ACRC) armandocesar82@yahoo.com.br.
[2] Laboratório de Ecologia de Peixes e Invasões Biológicas & Laboratório de Ecologia Aquática e Conservação de Espécies Nativas, Universidade Estadual de Londrina, Rodovia Celso Garcia Cid, PR-445, km 380, 86057-970 Londrina, PR, Brazil. (DAZG) diegoazgarcia@hotmail.com, (ADP) alandeivid@uel.br, (MLO) orsi@uel.br.
[3] Departamento de Oceanografia Biológica, Instituto Oceanográfico, Universidade de São Paulo, Praça Centro Universitário das Faculdades Integradas de Ourinhos, Rodovia BR-153, km 338, 19909-100 Ourinhos, SP, Brazil. (LRJ) lucasjarduli@gmail.com.
[4] Laboratório de Genética e Ecologia Animal, Universidade Estadual de Londrina, Centro de Ciências Biológicas, Rodovia Celso Garcia Cid, PR-445, km 380, 86057-970 Londrina, PR, Brazil. (FSA) fernandasa@uel.br.
Authors Contribution 
João D. Ferraz: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Visualization, Writing-original draft, Writing-review and editing.
Armando C. R. Casimiro: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Visualization, Writing-original draft, Writing-review and editing.
Diego A. Z. Garcia: Conceptualization, Data curation, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Alan D. Pereira: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Lucas R. Jarduli: Conceptualization, Data curation, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing-original draft, Writing-review and editing.
Fernanda S. de Almeida: Conceptualization, Funding acquisition, Project administration, Resources, Supervision, Writing-review and editing.
Mário L. Orsi: Conceptualization, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing-review and editing.
Ethical Statement
Fish were anesthetized and euthanized by overexposure to 1g/ml clove oil, fixed with 10% formalin, and further transferred to 70% alcohol. The Animal Ethics Committee authorized the field sampling (CEUA N° 24310.2017.78; collection license Nº 16578).
Competing Interests
The authors declare no competing interests.
How to cite this article
Ferraz JD, Casimiro ACR, Garcia DAZ, Pereira AD, Jarduli LR, Almeida FS, Orsi ML. Taxonomic loss and functional reduction over time in the ichthyofauna of the Taquaruçu Reservoir, lower Paranapanema River, Southern Brazil. Neotrop Ichthyol. 2021; 19(3):e200143. https://doi.org/10.1590/1982-0224-2020-0143
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted May 12, 2021 by Fernando Carvalho
Accepted May 12, 2021 by Fernando Carvalho
![]() Submitted December 16, 2020
Submitted December 16, 2020
![]() Epub Sept 17, 2021
Epub Sept 17, 2021