Jaqueline de Bem1 ![]()
![]() , Josiane Ribolli
, Josiane Ribolli![]() , Cristhiana Röpke2
, Cristhiana Röpke2![]() , Kirk O. Winemiller3
, Kirk O. Winemiller3![]() and Evoy Zaniboni-Filho1
and Evoy Zaniboni-Filho1![]()
PDF: EN XML: EN | Cite this article
Abstract
Dams reduce the longitudinal connectivity of rivers and thereby disrupt fish migration and the spatial distribution of species, impacts that remain poorly studied for some Neotropical rivers from mega-diverse basins. We investigated the spatial distribution of fish species with different trophic and movement/reproductive/size characteristics to assess how functional groups have responded to a cascade of dams on the Uruguai River in southern Brazil. Fish abundance, biomass, and species composition were evaluated at eight locations along the longitudinal gradient. The fish assemblage in the upper stretch was mainly characterized by small and medium-sized species at higher trophic levels, whereas the sites located furthest downstream displayed more medium and large-sized species, including many carnivorous species. Species with high fecundity, seasonal migrants, and catfishes with internal fertilization were common in the river´s middle and lower reaches. Detritivorous species dominated areas distant from the dams. Overall, functional diversity of local fish assemblages was greater in lower reaches. The cascade of dams has impacted the distribution of functional groups of local fish assemblages of Uruguai River. The alteration of functional groups in upper reaches of the river has potential consequences for ecosystem processes and services, such as nutrient cycling and fisheries.
Keywords: Ichthyofauna, Reproductive tactics, Reservoirs, River fragmentation, Trophic groups.
As barragens reduzem a conectividade longitudinal dos rios e interrompem a migração e a distribuição espacial das espécies, configurando impactos pouco estudados para alguns rios Neotropicais megadiversos. Investigamos a distribuição espacial de espécies de peixes com diferentes características tróficas e reprodutivas para avaliar como grupos funcionais responderam a uma cascata de reservatórios no rio Uruguai, sul do Brasil. Abundância de peixes, biomassa e composição de espécies foram avaliadas em oito locais ao longo do gradiente longitudinal. A assembleia de peixes no trecho superior foi caracterizada principalmente por espécies de pequeno e médio porte em níveis tróficos mais elevados, enquanto que os ambientes localizados mais a jusante apresentaram mais espécies de médio e grande porte, incluindo muitas espécies carnívoras. Espécies com alta fecundidade, migradores sazonais e bagres com fertilização interna foram comuns no curso médio e inferior. Espécies detritívoras dominaram áreas distantes das barragens. A diversidade funcional das assembleias de peixes locais foi maior nas partes inferiores. A cascata de barragens impactou a distribuição dos grupos funcionais das assembleias de peixes locais no rio Uruguai. A alteração da diversidade funcional no curso superior do rio tem consequências potenciais para a dinâmica e serviços do ecossistema, como ciclagem de nutrientes e pesca.
Palavras-chave: Fragmentação de rios, Grupos tróficos, Ictiofauna, Reservatórios, Táticas reprodutivas.
Introduction
Hydropower dams impact river ecosystems by reducing river longitudinal connectivity (Agostinho et al., 2016; Vitule et al., 2017; Barbarossa et al., 2020), hydrology and sediment dynamics (Araújo et al., 2013; Forsberg et al., 2017), and water physicochemistry (Zaniboni-Filho, Schulz, 2003). Dams also impact fish populations by altering in-stream and riparian habitats and blocking migration corridors (Mueller et al., 2011; Granzotti et al., 2018). River fragmentation impacts are both local and regional (Rosenberg et al., 1997), with cumulative effects that can affect regions located hundreds of kilometers downstream and upstream of the dam (Grill et al., 2015). When dams and their associated reservoirs are built in series, i.e., a cascade of dams, impacts to aquatic biota and ecosystem dynamics may be compounded. Hydropower dams constitute one of the main threats to freshwater biodiversity (Vörösmarty et al., 2010), and impacts are particularly devastating for the mega-diverse fish faunas in the tropics (Winemiller et al., 2016; Vitule et al., 2017). Migratory species are especially vulnerable because hydroelectric projects obstruct corridors essential for seasonal movements for reproduction and exploitation of habitats critical for feeding or providing refuge from predation (Carolsfeld et al., 2003; Hoeinghaus et al., 2009; Pelicice et al., 2015). Even for non-migratory fishes, dams cause population fragmentation and reductions in the sizes of local stocks and gene flow among them (Allendorf et al., 2012). As a consequence, river fragmentation compromises the adaptive capacity and the long-term persistence of fish stocks (Allendorf et al., 2012).
The functional characteristics of fish can be useful for assessing the impacts of dams on river ecology (Hoeinghaus et al., 2009; Mouillot et al., 2013; Pendleton et al., 2014; Santos et al., 2017, 2020; Zhang et al., 2020). Traits, such as morphology associated with modes of swimming and use of habitat, exploitation of various types of food resources and how food is obtained, defense tactics, reproductive strategies, and other aspects of performance that affect fitness, can be used to group species into functional groups or niches (Violle et al., 2007; Winemiller et al., 2015). These attributes can be used to assess how fish assemblages respond to human impacts and predicting the fish community’s responses to anthropic impacts (Angermeier, Winston, 1998; Arantes et al., 2019; Dias et al., 2020).
Impacts from dams differentially affect species and locations along the fluvial gradient. Rheophilic species, feeding specialists and species that depended on seasonal access to floodplain habitats generally are excluded from impounded areas (Agostinho et al., 2008). Ecological generalists and species adapted for lentic conditions may prosper in modified habitats (Arantes et al., 2019). Evaluation of assemblage functional groups facilitates comparative study of natural communities as well as impact assessment for dams and other human disturbances (Hoeinghaus et al., 2007; Cella-Ribeiro et al., 2017; Pelicice et al., 2018). These impacts can be more intense in cascade reservoirs system, in which changes in physical and biological characteristics have a substantial influence on trait compositions of fish communities (Santos et al., 2017, 2020; Arantes et al., 2019). Here, we assess the distribution of fish functional groups across a dam/reservoir cascade system in the Uruguai River within the subtropical region of South America.
Functional groups of aquatic organisms are expected to be longitudinally distributed in relation to fairly predictable riverscape characteristics, according to the assumptions of the River Continuum Concept (RCC; Vannote et al., 1980). For example, greater proportions of insectivorous fishes should occur in the upper river reaches where the riparian vegetation canopy covers the channel and provides allochthonous inputs in the form of terrestrial invertebrates and leaf litter that supports aquatic insects. In lower river reaches, where the channel is broader and sunlight and nutrients fuel autochthonous primary production, omnivorous and detritivorous fishes should comprise greater proportions of fish assemblages. However, this theory applies well to unobstructed rivers. Impoundments disrupt longitudinal connectivity and therefore should alter many predictions of the RCC (Vannote et al., 1980). The Serial Discontinuity Concept (SDC; Ward, Stanford, 1995) and Cascading Reservoir Continuum Concept (CRCC; Barbosa et al., 1999) predict how reservoir cascades alter physical and biodiversity patterns and processes described by the RCC.
We make several predictions about how the cascade of dams and reservoirs in the Uruguai River should affect the functional composition of local fish assemblages. Areas unaffected by dams with relatively natural flow regimes and habitats with fast-flowing water should retain the full complement of native fishes, including species that are rheophilic, trophic specialists and migratory species (Schork, Zaniboni-Filho, 2018). In areas directly impacted by dams, fish assemblages should be dominated by small and medium-sized fishes with opportunistic life-history strategies (Schork, Zaniboni-Filho, 2017). We further predict that the river’s altered longitudinal gradient (with dams blocking fish migration, lentic conditions in reservoirs, modified flow regimes in stretches downstream from dams, and transition zones in between (Thornton et al., 1990; Monaghan et al., 2019; Dias et al., 2020) has not only affected functional composition of local fish assemblages, but also functional diversity, numerical abundance, and biomass.
Material and methods
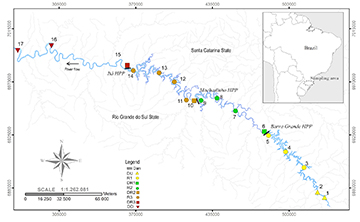
Sampling. The study was conducted along an approximately 600 km reach of the Upper Uruguai River where three hydroelectric dams have been constructed: Barra Grande (Upper stretch – completed in 2005), Machadinho (Middle stretch – completed in 2002), and Itá (Lower stretch – completed in 2000). Barra Grande has an area of 94 km2 and 90.6 days of the water residence time, while these values are 79 km2 and 54 days for Machadinho, and 141 km2 and 55 days for Itá. All of these reservoirs are formed by dams that are more than 100 m height. The spatial distribution of fish functional groups was investigated in eight locations, listed here from upstream to downstream: distant environment upstream from the uppermost dam [Distant Upstream – DU]; Barra Grande Reservoir [R1]; immediately downstream from Barra Grande Dam [Downstream Barra Grande DR1]; Machadinho Reservoir [R2]; immediately downstream from Machadinho Dam [Downstream Machadinho – DR2]; Itá Reservoir [R3]; immediately downstream from Itá Dam [Downstream Itá – DR3]; and distant downstream from the last dam [Distant Downstream – DD] (Fig. 1; Tab. 1). The environments Downstream R1, R2 and R3 were located approximately 250 m downstream from the dams, and the DD is a comparatively long and unimpeded stretch where several free-flowing tributaries join the river.
FIGURE 1 | Sampling locations in the Upper Uruguai River, Brazil. The symbols indicate survey sites: circles represent sites within the reservoirs (R1 = site 3 to 5; R2 = site 7 to 9; R3 = site 11 to 14); squares indicate sites located just below dams [Downstream R1 (DR1) = 6, Downstream R2 (DR2) = 10, Downstream R3 (DR3) = 15], and triangles indicate sites that are most distant from dams [Distant Upstream (DU) = 1 and 2; Distant Downstream (DD) = 16 and 17].
TABLE 1 | Characteristics of survey sites within eight locations along the Upper Uruguai River, Brazil. DU (distant location upstream of the most upstream dam), DD (distant and downstream of the most downstream dam), R1 (Barra Grande Reservoir), R2 (Machadinho Reservoir), R3 (Itá Reservoir), DR1 (immediately downstream from Barra Grande Dam), DR2 (immediately downstream from Machadinho Dam), DR3 (immediately downstream from Itá Dam).
| Locations | Sample Sites | UTM Coordinate (X) | UTM Coordinate (Y) | Distance downstream from the first sample site (km) | Distance between river banks (km) |
| DU | 1 | 529400 | 6873288 | 0 | 0.15 |
| DU | 2 | 523534 | 6880973 | 11.49 | 0.22 |
| R1 | 3 | 512252 | 6898513 | 49.46 | 0.39 |
| R1 | 4 | 496769 | 6912114 | 79.06 | 0.58 |
| R1 | 5 | 481753 | 6926735 | 105.55 | 0.74 |
| DR1 | 6 | 480779 | 6927650 | 106.88 | 0.11 |
| R2 | 7 | 454048 | 6946672 | 156.22 | 0.34 |
| R2 | 8 | 438514 | 6956148 | 189.55 | 0.51 |
| R2 | 9 | 421924 | 6955590 | 224.34 | 0.96 |
| DR2 | 10 | 422047 | 6954598 | 225.76 | 0.33 |
| R3 | 11 | 410968 | 6957484 | 250.04 | 0.40 |
| R3 | 12 | 402272 | 6971232 | 288.40 | 0.54 |
| R3 | 13 | 390945 | 6978846 | 311.01 | 0.67 |
| R3 | 14 | 367311 | 6981165 | 367.48 | 0.77 |
| DR3 | 15 | 361955 | 6981341 | 382.30 | 0.20 |
| DD | 16 | 299465 | 7002554 | 514.81 | 0.28 |
| DD | 17 | 264480 | 7001424 | 583.48 | 0.56 |
Fish assemblage data from surveys conducted at 17 sites were grouped into eight study locations (Distant Upstream – DU; Reservoirs – R1, R2, R3; Downstream Reservoirs – DR1, DR2 and DR3; and Distant Downstream – DD). Those sites are grouped considering similarities in geographical location and abiotic characteristics. Field work was done during January–February, April–May, July–August and October–November from 2006 to 2010, totaling 290 survey events successfully concluded. At each site during each survey seven different nets set were used, totaling 2,145 net samples. During each survey, fishes were caught along a stretch of shoreline with a set of four gill nets and three trammel nets with mesh sizes ranging from 1.5 to 5.0 cm between adjacent knots, nets height ranging from 1.6 to 1.8 m and the length ranging from 10 to 40 m. Nets were placed in the evening and removed in the following morning (set for approximately 12 h). All captured fish were counted and identified at the species level, weighed (in grams) and measured (total length in cm). Collecting license was provided by Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis – IBAMA (52/2007 and 145/2009). The nomenclature of the species list was based on Fricke et al. (2021) and in consultation with taxonomic experts. Vouchers of all species were deposited in the fish collection of Museu de Zoologia da Universidade Estadual de Londrina, Paraná, Brazil (MZUEL) (S1).
Abundance of each fish species was estimated from of numerical Catch Per Unit Effort (CPUE) (sum of individuals per 100 m2 net area/12 h – CPUEN) and biomass (sum of grams per 100 m2 net area/12 h – CPUEB), for each sampling unit (for each sampling site and period). Then, fish species were grouped according to functional groups, and CPUEN and CPUEB.
Defining functional groups. Functional groups were defined with an emphasis on characteristics predicted to affect responses to damming and reservoir formation: trophic niche, body size, and reproductive strategy (Arantes et al., 2019). Information on feeding and reproductive biology of species analyzed in this study were based on the scientific literature (e.g., Vazzoler, 1996; Hahn et al., 1998; Zaniboni-Filho et al., 2004; Reynalte-Tataje, Zaniboni-Filho, 2008; Araújo et al., 2009; Gubiani et al., 2012), including accounts for congeneric species when no information was available, and body size was recorded as the largest specimen captured from the Upper Uruguai River during regular surveys conducted by our research group from 1995 to 2014.
Fourteen functional groups were classified considering trophic and movement/reproductive/size categories. There were five trophic groups, based on diet: i) detritivore (species which fed on detritus, organic layer, periphytic algae and mud), ii) omnivore (defined as species with a generalist diet without predominance of either plant or animal tissue), iii) invertivore (species which fed on invertebrates), iv) carnivore (i.e., invertivore with a tendency for piscivore), and v) piscivore (species which fed mainly on fish). Movement/reproductive/size groups were nine combinations of this three characteristics: movement – considering fish with Sedentary habits or Short Migration (S/SM) or Long Migration (LM); reproductive strategy – with Parental Care (PC), Internal Fertilization (IF) or neither (No Parental Care – NPC); and body size – Small, maximum total length < 20 cm (S); Medium, total length between 20 and 40 cm (M); and Large, total length > 40 cm (L). These characteristics (movement, reproductive strategy and body size) were grouped based on their assumed functional and evolutionary interdependence. Thus, the nine movement/reproductive/size groups were: LNL (LM-NPC-L), SIM (S/SM-IF-M); SIS (S/SM-IF-S), SNL (S/SM-NPC-L), SNM (S/SM-NPC-M), SNS (S/SM-NPC-S), SPL (S/SM-PC-L), SPM (S/SM-PC-M), and SPS (S/SM-PC-S).
Numerical abundance and biomass of each functional group were determined from the sum of CPUEN and CPUEB of species for each sampling unit.
Data analysis. The CPUEN and CPUEB of the fourteen functional groups were log-transformed (log (x+1)) and nonmetric multidimensional scaling (NMDS) using the Bray-Curtis index was applied to evaluate dissimilarity in abundance (CPUEN) and biomass (CPUEB) of the functional groups between the sampled environments. Analysis were performed separately for trophic groups and movement/reproduction/size categories. The differences of functional assemblage structures (based on both trophic and movement/reproductive/size) among the eight locations were tested by Permutational Multivariate Analysis of Variance (PERMANOVA; Anderson, Walsh, 2013; Anderson, 2014), followed by post hoc tests performed with PERMANOVA pairwise comparisons, and Bonferroni method. The multivariate data were analyzed using the Bray-Curtis index generated from the transformed data with 9.999 permutations.
An analysis of homogeneity of multivariate dispersions (PERMDISP, Anderson et al., 2006) using the Bray-Curtis dissimilarity index was applied to evaluate if the eight locations had different degrees of homogeneity of assemblage structures based on different to CPUEN and CPUEB of functional groups. All analyzes were performed in software R (Version 3.2.4; http://cran.r-project.org), with Vegan, Mass, Car and RVAideMemoire packages.
Results
Surveys conducted at the eight locations in Upper Uruguai River yielded 77 fish species from 49 genera and 24 families (S1), distributed into eight study locations (S2). PERMANOVA analyses with adonis function revealed a significant difference between fish assemblage structures in the different habitat categories based on trophic (numerical abundance R = 0.27, p = 0.001; biomass R = 0.23, p = 0.001) and movement/reproductive/size categories (numerical abundance R = 0.32, p = 0.001; biomass R = 0.27, p = 0.001). Results of the PERMANOVA pairwise comparisons for eight environmental presents no differences between DUxR2, DUxDD, DR1xDR2, DUxR3 and DDxR3 for trophic categories (p > 0.05), and DUxR2 for movement/reproductive/size categories (p > 0.05), but with significant differences for the others comparisons.
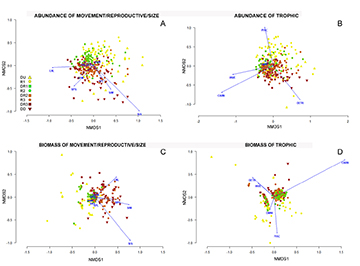
The results of ordination (NMDS; Fig. 2) indicated that piscivorous fishes were predominant in most upstream reservoir (R1), long-distance migratory and carnivorous fishes were most common immediately downstream from reservoirs (DR), and detritivorous fishes and those with internal fertilization were most common in the location Distant and Downstream from the lowest dam in the cascade (DD).
Approximately half of the total fish abundance and biomass in the assemblage at the location most Distant Downstream (DD) and the assemblage most Distant Upstream (DU) was composed of detritivorous species (Tab. 2).
The assemblage from the uppermost reservoir of the cascade (R1) had the lowest numerical abundance and biomass of detritivores, invertivores and carnivores. R1 had the highest numerical abundance and biomass of piscivores (41% and 65% of total fish abundance and biomass, respectively). Omnivorous fish were abundant in R1, DR1, and DR3 (44%, 68% and 47%, respectively; Tab. 2). The high abundance of omnivores immediately downstream from Barra Grande Dam (DR1) was due to an extraordinary abundance of a single species (Psalidodon aff. fasciatus).
FIGURE 2 | Ordination plots produced by the non-metric multidimensional scaling analysis (NMDS) using the Bray-Curtis index, considering the abundance (CPUEN) of movement/reproductive/size (A) and trophic categories (B), and biomass (CPUEB) of movement/reproductive/size categories (C) and trophic categories (D), among survey locations. [DU (red triangle), DD (yellow triangle)] = Sampling points away from the dams; [R1 (red circle), R2 (green circle), R3 (orange circle)] = sampling points located in the reservoirs; [DR1 (green square), DR2 (orange square), DR3 (red square)] = locations downstream from dams. Labels are for Detritivores, Invertivores, Carnivores, Omnivores, and Piscivores; S/SM = sedentary/short migration, NPC = no parental care, PC = parental care, IF = internal fertilization, and LM = long migration; and Small, Medium, and Large body sizes.
TABLE 2 | CPUEN and CPUEB of five trophic categories within samples of fish assemblages from eight locations along Upper Uruguai River obtained between 2006–2010. DU (distant upstream from the most upstream dam), DD (distant downstream from the most downstream dam), R1 (Barra Grande Reservoir), R2 (Machadinho Reservoir), R3 (Itá Reservoir), DR1 (immediately downstream from Barra Grande Dam), DR2 (immediately downstream from Machadinho Dam), DR3 (immediately downstream from Itá Dam).
| Trophic category | DU | % | R1 | % | DR1 | % | R2 | % | DR2 | % | R3 | % | DR3 | % | DD | % | |
| Abundance | Piscivorous | 28 | 10 | 85 | 41 | 94 | 8 | 54 | 16 | 104 | 19 | 26 | 10 | 31 | 5 | 11 | 5 |
| Carnivorous | 2 | 1 | 1 | 0 | 10 | 1 | 4 | 1 | 3 | 1 | 10 | 4 | 20 | 3 | 1 | 0 | |
| Omnivorous | 50 | 18 | 90 | 44 | 801 | 68 | 99 | 29 | 229 | 41 | 108 | 39 | 285 | 47 | 57 | 29 | |
| Invertivorous | 79 | 28 | 22 | 11 | 165 | 14 | 129 | 38 | 91 | 16 | 30 | 11 | 85 | 14 | 31 | 16 | |
| Detritivorous | 121 | 43 | 9 | 4 | 114 | 10 | 54 | 16 | 129 | 23 | 101 | 37 | 183 | 30 | 99 | 50 | |
| Biomass | Piscivorous | 5817 | 21 | 13643 | 65 | 17165 | 22 | 6997 | 37 | 21266 | 35 | 3403 | 25 | 7473 | 12 | 1502 | 13 |
| Carnivorous | 419 | 2 | 203 | 1 | 3425 | 4 | 486 | 3 | 840 | 1 | 566 | 4 | 1591 | 3 | 108 | 1 | |
| Omnivorous | 2847 | 10 | 4816 | 23 | 19214 | 25 | 5353 | 28 | 9062 | 15 | 3927 | 29 | 26011 | 42 | 3611 | 30 | |
| Invertivorous | 2735 | 10 | 485 | 2 | 13311 | 17 | 2787 | 15 | 6486 | 11 | 958 | 7 | 2727 | 4 | 1660 | 14 | |
| Detritivorous | 15618 | 57 | 1741 | 8 | 23226 | 30 | 3215 | 17 | 23817 | 39 | 4853 | 35 | 24826 | 40 | 5023 | 42 |
Fish assemblages at DR1 and Machadinho Reservoir (R2) had highest biomass and numerical abundance, respectively, of invertivorous fishes (Tab. 2). The fish assemblage at Itá, the most downstream reservoir of the cascade (R3), had the greatest numerical abundance and biomass of carnivorous fishes (4%; Tab. 2). Assemblages at the location immediately downstream from R3 (DR3) and the location furthest downstream (DD) had the lowest numerical abundance and biomass of fish categorized as piscivores.
Sedentary or short migratory species (S/SM) were abundant with large biomass at all eight locations (Tab. 3). Long-distance migratory fishes, including Prochilodus lineatus and Salminus brasiliensis, were absent in samples from the first reservoir (R1) and location immediately downstream (DR1) (S2). However, at Downstream R3 (DR3) and the most distant downstream location (DD) the numerical abundance and biomass of long-distance migrants were relatively high at 10% and 9%, respectively (Tab. 3).
The fish assemblage at the distant downstream location had greater numerical abundance and biomass of species with internal fertilization, but only four species were in this category. No species with internal fertilization were captured from Machadinho Reservoir (R2) or locations upstream. Functional categories with small-bodied species without parental care, stood out immediately downstream of R1, and medium-size species (without parental care – NPC), had greater numerical abundance and biomass at Barra Grande and Machadinho Reservoirs (R1 and R2). The assemblage at the location immediately downstream from Itá Reservoir (DR3) had the highest numerical abundance and biomass of medium-size species with parental care, and the most Distant Downstream location (DD) had the greatest abundance of large-size species with parental care (Tab. 3).
TABLE 3 | CPUEN and CPUEB of nine movement/reproductive/size categories within samples of fish assemblages from eight locations along Upper Uruguai River obtained between 2006–2010. Values are presented by annual mean number and percentage, S/SM = Sedentary/Short Migration, LM = Long Migration, PC = Parental Care, NPC = No Parental Care, IF = Internal Fertilization, S = small, M = medium, and L = large. Movement/reproductive/size groups: LNL (LM–NPC–L), SIM (S/SM–IF–M); SIS (S/SM–IF–S), SNL (S/SM–NPC–L), SNM (S/SM–NPC–M), SNS (S/SM–NPC–S), SPL (S/SM–PC–L), SPM (S/SM–PC–M), and SPS (S/SM–PC–S). DU (distant upstream from the most upstream dam), DD (distant downstream from the most downstream dam), R1 (Barra Grande Reservoir), R2 (Machadinho Reservoir), R3 (Itá Reservoir), DR1 (immediately downstream from Barra Grande Dam), DR2 (immediately downstream from Machadinho Dam), DR3 (immediately downstream from Itá Dam).
| Movement/reproductive/size | DU | % | R1 | % | DR1 | % | R2 | % | DR2 | % | R3 | % | DR3 | % | DD | % | |
| Abundance | SNS | 46 | 16 | 82 | 40 | 799 | 68 | 105 | 31 | 201 | 36 | 137 | 50 | 195 | 32 | 48 | 24 |
| SNM | 122 | 43 | 104 | 50 | 232 | 20 | 182 | 54 | 213 | 38 | 54 | 20 | 110 | 18 | 40 | 20 | |
| SNL | 10 | 3 | 11 | 5 | 39 | 3 | 29 | 8 | 43 | 8 | 14 | 5 | 88 | 14 | 7 | 3 | |
| SPS | 0 | <1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | <1 | 0 | 0 | 0 | <1 | |
| SPM | 89 | 32 | 3 | 1 | 46 | 4 | 18 | 5 | 68 | 12 | 26 | 10 | 191 | 32 | 31 | 16 | |
| SPL | 14 | 5 | 7 | 3 | 67 | 6 | 6 | 2 | 28 | 5 | 40 | 15 | 17 | 3 | 66 | 33 | |
| SIS | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | <1 | 0 | 0 | 0 | <1 | |
| SIM | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 5 | 3 | |
| LNL | <1 | 0 | 0 | 0 | 0 | 0 | 0 | <1 | 3 | <1 | 0 | <1 | 4 | <1 | 1 | <1 | |
| Biomass | SNS | 1038 | 4 | 1944 | 9 | 12850 | 17 | 2107 | 11 | 3027 | 5 | 2544 | 19 | 2821 | 5 | 1218 | 10 |
| SNM | 9521 | 35 | 11476 | 55 | 21260 | 28 | 8904 | 47 | 23936 | 39 | 2791 | 20 | 4734 | 8 | 2311 | 19 | |
| SNL | 2581 | 9 | 3078 | 15 | 10331 | 14 | 4381 | 23 | 6726 | 11 | 2445 | 18 | 23356 | 37 | 1854 | 16 | |
| SPS | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 13 | 0 | 0 | 0 | 3 | 0 | |
| SPM | 8866 | 32 | 443 | 2 | 4920 | 6 | 1405 | 7 | 8499 | 14 | 1858 | 14 | 21818 | 35 | 2034 | 17 | |
| SPL | 5269 | 19 | 3946 | 19 | 26981 | 35 | 2009 | 11 | 17036 | 28 | 3703 | 27 | 3390 | 5 | 3170 | 27 | |
| SIS | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 23 | 0 | |
| SIM | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 75 | 0 | 46 | 0 | 13 | 0 | 210 | 2 | |
| LNL | 160 | 1 | 0 | 0 | 0 | 0 | 32 | 0 | 2172 | 4 | 306 | 2 | 6495 | 10 | 1082 | 9 |
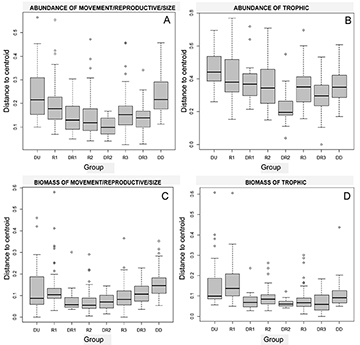
PERMDISP analysis indicated that all locations had different degrees of heterogeneity of assemblage functional groups structure (trophic numerical abundance: F(7,282) = 9.74, P < 0.0001; trophic biomass: F(7,282) = 9.06, P < 0.0001; movement/reproductive/size numerical abundance: F(7,282) = 8.95, P < 0.0001; movement/reproductive/size biomass: F(7,282) = 6.19, P < 0.0001). Overall, for both functional groups (trophic and reproductive/movement/size) and metrics (numerical abundance and biomass), pairwise differences were consistent, the locations DU and R1 had the highest heterogeneity (larger distance from centroid) compared to the others, and DR2 the lowest heterogeneity. The location DD had a higher movement/reproductive/size heterogeneity compared to locations from the middle stretch of the Uruguai River (Figs. 3A–D; Tabs. 4–5; S3).
FIGURE 3 | Analysis of multivariate homogeneity of group dispersions (PERMIDISP) by betadisper boxplot, using the Bray-Curtis index, considering the abundance (CPUEN) of movement/reproductive/size (A) and trophic categories (B), and biomass (CPUEB) of movement/reproductive/size categories (C) and trophic categories (D). The data was based on sampling sites, and presented by environment groups: [DU, DD] = Sampling points away from the dams; [R1, R2, R3] = sampling points located in the reservoirs; [DR1, DR2, DR3] = locations downstream from dams. Greater distance to spatial median indicates larger dispersion and therefore a more diverse/heterogeneous environment. Box lower and upper endpoints represent the 25th and 75th quartiles, respectively. The horizontal bar and plus symbol inside each box represent median, excluding outliers, which are presented by open circles. See Tabs. 4 and 5 for P values from environments comparisons.
TABLE 4 | Summary of PERMDISP permuted P values for comparisons of heterogeneity in movement/reproductive/size structure among locations. Above diagonal p values for numeric abundance and below diagonal for biomass movement/reproductive/size structure. Statistically significant P values (P < 0.05) are highlighted in bold.
| Location | DU | R1 | DR1 | R2 | DR2 | R3 | DR3 | DD |
| DU | 0.177 | 0.002 | 0.001 | 0.001 | 0.001 | 0.003 | 0.989 | |
| R1 | 0.801 | 0.023 | 0.003 | 0.001 | 0.028 | 0.043 | 0.128 | |
| DR1 | 0.088 | 0.043 | 0.868 | 0.039 | 0.280 | 0.758 | 0.001 | |
| R2 | 0.001 | 0.001 | 0.785 | 0.046 | 0.187 | 0.878 | 0.001 | |
| DR2 | 0.019 | 0.007 | 0.652 | 0.778 | 0.002 | 0.022 | 0.001 | |
| R3 | 0.018 | 0.004 | 0.392 | 0.070 | 0.104 | 0.442 | 0.001 | |
| DR3 | 0.453 | 0.310 | 0.136 | 0.013 | 0.010 | 0.247 | 0.001 | |
| DD | 0.293 | 0.415 | 0.001 | 0.001 | 0.001 | 0.001 | 0.019 |
TABLE 5 | Summary of PERMDISP permuted P values for comparisons of heterogeneity in trophic assemblage structure among locations. Above diagonal p values for numeric abundance and below diagonal for biomass trophic structure. Statistically significant P values (P < 0.05) are highlighted in bold.
| Location | DU | R1 | DR1 | R2 | DR2 | R3 | DR3 | DD |
| DU | 0.572 | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.023 | |
| R1 | 0.710 | 0.004 | 0.003 | 0.001 | 0.001 | 0.001 | 0.076 | |
| DR1 | 0.019 | 0.003 | 0.260 | 0.001 | 0.134 | 0.824 | 0.035 | |
| R2 | 0.001 | 0.001 | 0.366 | 0.001 | 0.828 | 0.170 | 0.286 | |
| DR2 | 0.002 | 0.001 | 0.341 | 0.026 | 0.001 | 0.003 | 0.001 | |
| R3 | 0.001 | 0.001 | 0.796 | 0.358 | 0.200 | 0.065 | 0.284 | |
| DR3 | 0.005 | 0.001 | 0.802 | 0.200 | 0.666 | 0.529 | 0.020 | |
| DD | 0.089 | 0.012 | 0.111 | 0.126 | 0.007 | 0.023 | 0.060 |
Discussion
The fish assemblage in the lowest reach (R3, DR3 and DD) and the upper portion (DU), within the dam and reservoir cascade of the Upper Uruguai River, had greater functional group diversity, and this might indicate that environmental impacts were lower compared to upstream locations in the reservoirs cascade. Although this pattern also could reflect a legacy of a natural longitudinal gradient of fish diversity during the period preceding construction of the hydroelectric dams (Lowe-McConnell, 1975; Araújo et al., 2009), the high diversity and presence of piscivorous in the upstream portion may corroborate with the reservoir cascade environmental impact. Piscivorous fishes were relatively abundant in the uppermost reach and the location downstream from the Machadinho Reservoir. Overall, fish assemblages at all locations surveyed in the Upper Uruguai River were dominated by small and medium-sized species that lack parental care. Medium-sized and large carnivorous species dominated assemblages in lower reaches between Itá Reservoir and the most Distant Downstream location, and these assemblages included diverse life history attributes, such as internal fertilization, parental care, and long-distance migratory behavior. The presence of medium-sized and large-sized fish and species with a diversity of life history attributes was associated with habitats less affected by human impacts resulting from dams, but another influence could have been the natural tendency for fish species richness to increase in the downstream direction along longitudinal river gradients (Araújo et al., 2009).
Migratory fishes were largely restricted to middle-lower reaches of the cascade system where several free-flowing tributaries, including the Ligeiro and Peixe rivers (located between DR2 and R3), join the Uruguai River (Reynalte-Tataje et al., 2012). The connectivity provided by these rivers is crucial for migratory fishes that undergo seasonal long-distance migrations (Agostinho et al., 2002; Cote et al., 2009; Silva et al., 2017). Dams in a cascade system create insurmountable barriers to migratory fish species, both in terms of upstream and downstream movement (Vörösmarty et al., 2010; Pelicice et al., 2018). The absence of large migratory fishes, such as Salminus brasiliensis from the upper stretches (DU, R1 and DR1) have the potential to alter top-down trophic dynamics structuring local fish assemblages (Taylor et al., 2015).
Habitats within reservoirs and immediately downstream from them often have hydrological regimes that vary more as a function of dam releases than seasonal precipitation and runoff (Graf, 2006; Räsänen et al., 2012). Consequently, the magnitude and timing of changes in hydrology and water quality tend to be lower and less variable and predictable compared to the natural flow regime (Vannote et al., 1980; Link et al., 2008). These conditions likely are not conducive to fish species that are seasonal spawners with recruitment dependent upon access to flooded riparian areas or other habitats formed in special hydrological conditions that serve as nurseries (Winemiller, 1989; Winemiller et al., 2008). Daily water-level fluctuations in nearshore areas of reservoirs and in tailraces are disruptive to fishes that deposit eggs in nests and guard them (Agostinho et al., 2008). It is unclear why auchenipterid catfishes with internal fertilization were absent in upper reaches of the dam/reservoir cascade. These fishes have a relatively equilibrium-type life history strategy of relatively large egg size and low fecundity (sensu Winemiller, Rose, 1992). Like cichlids and other brood-guarding fishes that also would be considered equilibrium strategists, these catfishes should be adapted for relatively stable environments (Tedesco et al., 2008).
High percentages of small and medium-sized sedentary and short-migration fishes throughout the Upper Uruguai River suggests that these species are ecological generalist in terms of feeding and habitat requirements. Neotropical fishes in these categories have been shown previously to dominate fish assemblages within reservoirs (Agostinho et al., 2007; Pelicice et al., 2018). Many of the fishes in these categories are omnivorous with broad diets that vary in response to food availability (Agostinho et al., 2007). It is notable that distributions for these fishes contrast with those of detritivorous (Agostinho et al., 2016; Dias et al., 2020) and long-distance migratory fishes (Agostinho et al., 2002; Cote et al., 2009; Silva et al., 2017) that were uncommon at locations most strongly impacted by dams and reservoirs.
Following dam construction, the reservoir filling phase generally produces a marked increase in aquatic primary production (trophic upsurge, Baranov, 1961) and availability of food resources for fishes, especially for small omnivorous and invertivorous fishes, which in turn often leads to an eventual increase in the abundance of piscivores that exploit them as prey (Agostinho et al., 2007). The trophic upsurge and abundant prey may explain the high abundance of piscivorous fishes in the most recently created reservoir (R1). In contrast, the relatively low abundance of large piscivores within reservoirs in the lower stretch of the cascade system might account for the greater abundance of smaller carnivorous species, owing to lower predation mortality as well as competition for some of the same food resources (Petry, Schulz, 2006; Araújo et al., 2009). Detritivorous fish can play an important role in nutrient cycling in both rivers (Taylor et al., 2015) and reservoirs (Vanni et al., 2005), and therefore could influence food web structure and other trophic guilds along river longitudinal gradients. However, this trophic guild is known to be negatively impacted by creation of reservoirs (Santos et al., 2020), and was not abundant in the deep reservoirs of the Upper Uruguai River. Fish in reservoirs avoid deep areas where thermal stratification reduces dissolved oxygen concentrations, which can result in a loss of nutritional quality of detritus (Santos et al., 2020) and places much of the phytoplankton-derived detritus out of reach for detritivores (Baumgartner et al., 2020). Detritivorous fishes were most common at the Distant-Upstream and Distant-Downstream locations, the two areas with flow regimes that were least affected by dams. They also were common in river reaches downstream from dams, where they likely exploit phytoplankton-derived detritus in water released from dams (Hoeinghaus et al., 2007).
The spatial distribution of fish functional groups in the dam/reservoir cascade of the Upper Uruguai River suggests that the downstream area (R3, DR3 and DD) have retained more the natural environmental characteristics that support riverine fish diversity. The DD location lies within an extensive unimpeded reach where several free-flowing tributaries enter the river and contribute to fluvial connectivity and a more natural flow regime compared to river reaches directly downstream from reservoirs. Consequently, this location has greater fluvial connectivity and a more natural flow regime compared to upstream locations that are more impacted by dams. Dams cause profound changes in not only the taxonomic structure of river fish assemblages (Freedman et al., 2014), but also the functional groups of aquatic communities with likely effects on food-web dynamics. Our findings contribute to understanding the effects of reservoir cascades on the distribution and abundance of fish with diverse ecological characteristics. Tributaries appear to have fundamental importance for maintaining a high diversity of fish reproductive groups, and reservoir cascades create large-scale environmental heterogeneity that apparently determines the distribution of fish trophic guilds and food web structure. Future management actions should prioritize the maintenance of fluvial habitat connectivity and environment conditions to facilitate fish migration, reproduction and life cycle completion by species spanning diverse life history strategies, and food web dynamics that allow persistence of the full complement of native species.
Acknowledgments
The authors acknowledge Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Engie (Tractebel Energia), Consórcio Itá, and Consórcio Machadinho for financial support. This study was also supported by SISBIOTA – Top predator network (CNPq 563299/2010–0 and Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP 2010/52315-7). JB acknowledge scholarships provided by CAPES and PhD exchange process number 99999.004990/2014–05. JR acknowledge scholarships provided by Programa Nacional de Pós-Doutorado PNPD/CAPES. EZF thanks CNPq, grant 302860/2014–2. We are grateful to the technicians from Laboratório de Biologia e Cultivo de Peixes de Água Doce (LAPAD) for data collection and Dr. Tiago Pires for his contribution to statistical analysis. This study was financed in part by CAPES – Finance Code 001. Collecting license was provided by Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis – IBAMA (52/2007 and 145/2009) and this study did not require permission from Ethical Committee for Animal Use in Experiments. The authors also thank the anonymous reviewers who improved the manuscript.
References
Agostinho AA, Gomes LC, Fernandez DR, Suzuki HI. Efficiency of fish ladders for neotropical ichthyofauna. River Res Appl. 2002; 18(3):299–306. https://doi.org/10.1002/rra.674
Agostinho AA, Gomes LC, Pelicice FM. Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil. Maringá: EDUEM; 2007.
Agostinho AA, Pelicice FM, Gomes LC. Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz J Biol. 2008; 68(4):1119–132. https://doi.org/10.1590/S1519-69842008000500019
Agostinho AA, Gomes LC, Santos, NC, Ortega JC, Pelicice FM. Fish assemblages in Neotropical reservoirs: Colonization patterns, impacts and management. Fish Res. 2016; 173:26–36. https://doi.org/10.1016/j.fishres.2015.04.006
Allendorf FW, Luikart G, Aitken SN. Conservation and the genetics of populations. Oxford:Wiley-Blackwell Publishing; 2012.
Anderson MJ, Ellingsen KE, McArdle BH. Multivariate dispersion as a measure of beta diversity. Ecol Lett. 2006; 9:683–93. https://doi.org/10.1111/j.1461-0248.2006.00926.x
Anderson MJ. Permutational multivariate analysis of variance (PERMANOVA). Wiley StatsRef: Statistics Reference Online. 2014;1–15. https://doi.org/10.1002/9781118445112.stat07841
Anderson MJ, Walsh DCI. PERMANOVA, ANOSIM, and the Mantel test in the face of heterogeneous dispersions: What null hypothesis are you testing? Ecol monogr. 2013; 83(4):557–74. http://doi.org/10.1890/12-2010.1
Angermeier PL, Winston MR. Local vs. regional influences on local diversity in stream fish communities of Virginia. Ecology. 1998; 79(3):911–27. https://doi.org/10.2307/176589
Arantes CC, Fitzgerald DB, Hoeinghaus DJ, Winemiller KO. Impacts of hydroelectric dams on fishes and fisheries in tropical rivers through the lens of functional traits. Curr Opin Environ Sustain. 2019; 37:28–40. https://doi.org/10.1016/j.cosust.2019.04.009
Araújo ES, Marques EE, Freitas IS, Neuberger AL, Fernandes R, Pelicice FM. Changes in distance decay relationships after river regulation: similarity among fish assemblages in a large Amazonian river. Ecol Fresh Fish. 2013; 22(4):543–52. https://doi.org/10.1111/eff.12054
Araújo FG, Pinto BCT, Teixeira TP. Longitudinal patterns of fish assemblages in a large tropical river in southeastern Brazil: evaluating environmental influences and some concepts in river ecology. Hydrobiologia. 2009; 618:89–107. https://doi.org/10.1007/s10750-008-9551-5
Baranov IV. Biohydrochemical classification of the reservoirs in the European USSR. The storage lakes of the USSR and their importance for fishery. Bull State Scient Res Inst Lake River Fish. 1961; 50:1638–650.
Barbarossa V, Schmitt RJ, Huijbregts MA, Zarfl C, King H, Schipper AM. Impacts of current and future large dams on the geographic range connectivity of freshwater fish worldwide. Proc Natl Acad Sci. 2020; 117(7):3648–655. https://doi.org/10.1073/pnas.1912776117
Barbosa FAR, Padisák J, Espíndola ELG, Borics G, Rocha O. The cascading reservoir continuum concept (CRCC) and its application to the river Tietê-basin, São Paulo State, Brazil. In: Tundisi JG, Straskraba M, editors. Workshop on Theoretical Reservoir reservoir Ecologyecology and its applications. São Carlos: International Institute of Ecology, Brazilian Academy of Sciences and Backhuys Publishers; 1999; 425–37.
Baumgartner MT, Piana PA, Baumgartner G, Gomes LC. Storage or run-of-river reservoirs: exploring the ecological effects of dam operation on stability and species interactions of fish assemblages. Environ Manag. 2020; 65(2):220–31. https://doi.org/10.1007/s00267-019-01243-x
Carolsfeld J, Godinho HP, Zaniboni Filho E, Harvey BJ. Cryopreservation of sperm in Brazilian migratory fish conservation. J Fish Biol. 2003; 63(2):472–89. https://doi.org/10.1046/j.1095-8649.2003.00170.x
Cella-Ribeiro A, Costa Doria CR, Dutka-Gianelli J, Alves H, Torrente-Vilara G. Temporal fish community responses to two cascade run-of-river dams in the Madeira River, Amazon basin. Ecohydrology. 2017; 10(8):e1889. https://doi.org/10.1002/eco.1889
Cote D, Kehler DG, Bourne C, Wiersma YF. A new measure of longitudinal connectivity for stream networks. Landsc Ecol. 2009; 24(1):101–13. https://doi.org/10.1007/s10980-008-9283-y
Dias RM, Oliveira AG, Baumgartner MT, Angulo-Valencia MA, Agostinho AA. Functional erosion and trait loss in fish assemblages from Neotropical reservoirs: The man beyond the environment. Fish Fish. 2020; 22:377–90. https://doi.org/10.1111/faf.12524
Forsberg BR, Melack JM, Dunne T, Barthem RB, Goulding M, Paiva RC et al. The potential impact of new Andean dams on Amazon fluvial ecosystems. PLoS One. 2017; 12(8):e0182254. https://doi.org/10.1371/journal.pone.0182254
Freedman JA, Lorson BD, Taylor RB, Carline RF, Stauffer JR. River of the dammed: longitudinal changes in fish assemblages in response to dams. Hydrobiologia. 2014; 727(1):19–33. https://doi.org/10.1007/s10750-013-1780-6
Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: species by family/subfamily [Internet]. San Francisco: California Academy of Sciences; 2021. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Graf WL. Downstream hydrologic and geomorphic effects of large dams on American rivers. Geomorphology. 2006; 79(3–4):336–60. https://doi.org/10.1016/j.geomorph.2006.06.022
Granzotti RV, Miranda LE, Agostinho AA, Gomes LC. Downstream impacts of dams: shifts in benthic invertivorous fish assemblages. Aquat Sci. 2018; 80(3):1–14. https://doi.org/10.1007/s00027-018-0579-y
Grill G, Lehner B, Lumsdon AE, Macdonald GK, Zarfl C, Liermann CR. An index-based framework for assessing patterns and trends in river fragmentation and flow regulation by global dams at multiple scales. Environ Res Lett. 2015; 10:015001. https://doi.org/10.1088/1748-9326/10/1/015001
Gubiani EA, Gomes LC, Agostinho AA. Estimates of population parameters and consumption/ biomass ratio for fishes in reservoirs, Paraná State, Brazil. Neotrop Ichthyol. 2012; 10:177–88. https://doi.org/10.1590/S1679-62252012000100017
Hahn NS, Agostinho AA, Gomes LC, Bini LM. Estrutura trófica da ictiofauna do reservatório de Itaipu (Paraná – Brasil) nos primeiros anos de formação. Interciencia. 1998; 23(5):299–305. Available from: http://repositorio.uem.br:8080/jspui/bitstream/1/5202/1/209.pdf
Hoeinghaus DJ, Winemiller KO, Agostinho AA. Landscape-scale hydrologic characteristics differentiate patterns of carbon flow in large-river food webs. Ecosystems. 2007; 10:1019–033. https://doi.org/10.1007/s10021-007-9075-2
Hoeinghaus DJ, Agostinho AA, Gomes LC, Pelicice FM, Okada EK, Latini J et al. Effects of river impoundment on ecosystem services of large tropical rivers: embodied energy and market value of artisanal fisheries. Conserv Biol. 2009; 23(5):1222–31. https://doi.org/10.1111/j.1523-1739.2009.01248.x
Link O, Espinoza A, Stehr A, García A. Development and verification of JAZZ1D: a stream temperature model. Proceedings of the International Conference on Watershed Technology: Improving Water Quality and Environment, American Society of Agricultural and Biological Engineers, Concepción, Chile; 2008.
Lowe-McConnell RH. Fish communities in tropical freshwaters. Longman, New York; 1975.
Monaghan KA, Agostinho CS, Pelicice FM, Soares AM. The impact of a hydroelectric dam on Neotropical fish communities: A spatio-temporal analysis of the Trophic Upsurge Hypothesis. Ecol Fresh Fish. 2019; 29(2):384–97. https://doi.org/10.1111/eff.12522
Mouillot D, Graham NA, Villéger S, Mason NW, Bellwood DR. A functional approach reveals community responses to disturbances. Trends Evol Evol. 2013; 28(3):167–77. https://doi.org/10.1016/j.tree.2012.10.004
Mueller M, Pander J, Geist J. The effects of weirs on structural stream habitat and biological communities. J Appl Ecol. 2011; 48:1450–61. https://doi.org/10.1111/j.1365-2664.2011.02035.x
Pelicice FM, Pompeu PS, Agostinho AA. Large reservoirs as ecological barriers to downstream movements of Neotropical migratory fish. Fish Fish. 2015; 16(4):697–715. https://doi.org/10.1111/faf.12089
Pelicice FM, Azevedo-Santos VM, Esguícero ALH, Agostinho AA, Arcifa MS. Fish diversity in the cascade of reservoirs along the Paranapanema River, southeast Brazil. Neotrop Ichthyol. 2018; 16(2):e170150. https://doi.org/10.1590/1982-0224-20170150
Pendleton RM, Hoeinghaus DJ, Gomes LC, Agostinho AA. Loss of rare fish species from tropical floodplain food webs affects community structure and ecosystem multifunctionality in a mesocosm experiment. PLoS One. 2014; 9(1):e84568. https://doi.org/10.1371/journal.pone.0084568
Petry AC, Schulz UH. Longitudinal changes and indicator species of the fish fauna in the subtropical Sinos River, Brazil. J Fish Biol. 2006; 69:272–90. https://doi.org/10.1111/j.1095-8649.2006.01110.x
Räsänen TA, Koponen J, Lauri H, Kummu M. Downstream hydrological impacts of hydropower development in the Upper Mekong basin. Water Resour Manag. 2012; 26(12):3495–513. https://doi.org/10.1007/s11269-012-0087-0
Reynalte-Tataje DA, Zaniboni-Filho E. Biologia e identificação de ovos e larvas de peixes do alto rio Uruguai. In: Zaniboni-Filho E, Nuñer APO, editors. Reservatório de Itá: estudos ambientais, desenvolvimento de tecnologias de cultivo e conservação da ictiofauna. Florianópolis: Editora da UFSC; 2008. p.229–56.
Reynalte-Tataje DA, Nuñer AP, Nunes MC, Garcia V, Lopes CA, Zaniboni-Filho E. Spawning of migratory fish species between two reservoirs of the upper Uruguay River, Brazil. Neotrop Ichthyol. 2012; 10(4):829–35. https://doi.org/10.1590/S1679-62252012000400016
Rosenberg DM, Berkes F, Bodaly RA, Hecky RE, Kelly CA, Rudd JW. Large-scale impacts of hydroelectric development. Environ Rev. 1997; 5(1):27–54. https://doi.org/10.1139/a97-001
Santos NCL, Santana HS, Ortega JCG, Dias RM, Stegmann LF, Araújo IMS et al. Environmental filters predict the trait composition of fish communities in reservoir cascades. Hydrobiologia. 2017; 802(1):245–53. https://doi.org/10.1007/s10750-017-3274-4
Santos NCL, Dias RM, Alves DC, de Melo BAR, Ganassin MJM, Gomes LC et al. Trophic and limnological changes in highly fragmented rivers predict the decreasing abundance of detritivorous fish. Ecol Indic. 2020; 110:105933. https://doi.org/10.1016/j.ecolind.2019.105933
Schork G, Zaniboni-Filho E. Structure dynamics of a fish community over ten years of formation in the reservoir of the hydroelectric power plant in upper Uruguay River. Brazil J Biol. 2017; 77(4):710–23. https://doi.org/10.1590/1519-6984.17015
Schork G, Zaniboni-Filho E. Influência do gradiente espacial sobre a estrutura da ictiofauna em um reservatório subtropical – Alto Rio Uruguai. Bol Inst Pesca. 2018; 44(2):135–68. https://doi.org/10.20950/1678-2305.2018.236
Silva JC, Rosa RR, Galdioli EM, Soares CM, Domingues WM, Veríssimo S et al. Importance of dam-free stretches for fish reproduction: the last remnant in the Upper Paraná River. Acta Limnol Bras. 2017; 29. https://doi.org/10.1590/s2179-975×10216
Taylor J, Vanni M, Flecker, A. Top-down and bottom-up interactions in freshwater ecosystems: Emerging complexities. In: Hanley T, La Pierre K, editors. Top-down and bottom-up interactions in freshwater ecosystems. Cambridge: Cambridge University Press; 2015. p.55–85.
Tedesco PA, Hugueny B, Oberdorff T, Dürr HH, Merigoux S, Mérona B. River hydrological seasonality influences life history strategies of tropical riverine fishes. Oecologia. 2008; 156(3):691–702. https://doi.org/10.1007/s00442-008-1021-2
Thornton KW, Kimmel BL, Payne FE, editors. Reservoir Limnology: Ecological perspectives. JohnWiley & Sons: New York; 1990.
Vanni MJ, Arend KK, Bremigan MT, Bunnell DB, Garvey JE, González MJ et al. Linking landscapes and food webs: Effects of omnivorous fish and watersheds on reservoir ecosystems. BioScience. 2005; 55(2):155–67. https://doi.org/10.1641/0006-3568(2005)055[0155:LLAFWE]2.0.CO;2
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE. The river continuum concept. Can J Fish Aquat Sci. 1980; 37:130–37. https://doi.org/10.1139/f80-017
Vazzoler, AEAM. Biologia da reprodução de peixes teleósteos: Teoria e prática. Maringá: EDUEM; 1996.
Violle C, Navas ML, Vile D, Kazakou E, Fortunel C, Hummel I et al. Let the concept of trait be functional! Oikos. 2007; 116(5):882–92. https://doi.org/10.1111/j.0030-1299.2007.15559.x
Vitule JR, Agostinho AA, Azevedo-Santos VM, Daga VS, Darwall WR, Fitzgerald D et al. We need better understanding about functional diversity and vulnerability of tropical freshwater fishes. Biodivers Conserv. 2017; 26(3):757–62. https://doi.org/10.1007/s10531-016-1258-8
Vörösmarty CJ, McIntyre PB, Gessner MO, Dudgeon D, Prusevich A, Green P et al. Global threats to human water security and river biodiversity. Nature. 2010; 467(7315):555–61. https://doi.org/10.1038/nature09440
Ward JV, Stanford JA. The serial discontinuity concept: extending the model to floodplain rivers. Regul River. 1995; 10(2–4):159–68. https://doi.org/10.1002/rrr.3450100211
Winemiller KO. Patterns of variation in life history among South American fishes in seasonal environments. Oecologia. 1989; 81(2):225–41. Available from: https://link.springer.com/content/pdf/10.1007/BF00379810.pdf
Winemiller KO, Rose KA. Patterns of life-history diversification in North American fishes: implications for population regulation. Can J Fish Aquat Sci. 1992; 49(10):2196–218. https://doi.org/10.1139/f92-242
Winemiller KO, Agostinho AA, Caramaschi EP. Fish ecology in tropical streams. In Dudgeon D, editor. Tropical stream ecology. Amsterdam, Academic Press; 2008.
Winemiller KO, Fitzgerald DB, Bower LM, Pianka ER. Functional traits, convergent evolution, and periodic tables of niches. Ecol Lett. 2015; 18(8):737–51. https://doi.org/10.1111/ele.12462
Winemiller KO, McIntyre PB, Castello L, Fluet-Chouinard E, Giarrizzo T, Nam S et al. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science. 2016; 351(6269):128–29. https://doi.org/10.1126/science.aac7082
Zaniboni-Filho E, Schulz UH. Migratory fishes of the Uruguay River. In: Carolsfeld J, Harvey B, Ross C, Baer A, editors. Migratory fishes of South America:Biology, fisheries and conservation status. IDRC: World Fisheries Trust and World Bank; 2003. p.157–94.
Zaniboni-Filho E, Meurer S, Shibatta AO, Nuñer APO. Catálogo ilustrado de peixes do alto rio Uruguai. Florianópolis: Editora da UFSC; 2004.
Zhang C, Fujiwara M, Pawluk M, Liu H, Cao W, Gao X. Changes in taxonomic and functional diversity of fish communities after catastrophic habitat alteration caused by construction of Three Gorges Dam. Ecol Evol. 2020; 10(12):5829–39. https://doi.org/10.1002/ece3.6320
Authors
Jaqueline de Bem1 ![]()
![]() , Josiane Ribolli
, Josiane Ribolli![]() , Cristhiana Röpke2
, Cristhiana Röpke2![]() , Kirk O. Winemiller3
, Kirk O. Winemiller3![]() and Evoy Zaniboni-Filho1
and Evoy Zaniboni-Filho1![]()
[1] Laboratório de Biologia e Cultivo de Peixes de Água Doce, Departamento de Aquicultura, Universidade Federal de Santa Catarina, 88066-260 Florianópolis, SC, Brazil. (JDB) jaquedebem@gmail.com (corresponding author), (JR) josianeribolli@gmail.com, (EZF) evoyzaniboni@gmail.com.
[2] Instituto Nacional de Pesquisas da Amazônia, Coordenação em Biodiversidade, 69067-375 Manaus, AM, Brazil. crisropke@gmail.com.
[3] Department of Ecology and Conservation Biology, Texas A&M University, 77843-2258 College Station, TX, USA. k-winemiller@tamu.edu.
Authors Contribution 
Jaqueline de Bem: Conceptualization, Formal analysis, Investigation, Methodology, Writing-original draft.
Josiane Ribolli: Conceptualization, Supervision, Writing-review and editing.
Cristhiana Röpke: Formal analysis, Supervision, Writing-review and editing.
Kirk O. Winemiller: Conceptualization, Supervision, Writing-review and editing.
Evoy Zaniboni-Filho: Conceptualization, Funding acquisition, Project administration, Supervision, Writing-review and editing.
Ethical Statement
Collecting license was provided by Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis – IBAMA (52/2007 and 145/2009).
Competing Interests
The authors declare no competing interests.
How to cite this article
de Bem J, Ribolli J, Röpke C, Winemiller KO, Zaniboni-Filho E. A cascade of dams affects fish spatial distributions and functional groups of local assemblages in a subtropical river. Neotrop Ichthyol. 2021; 19(3):e200133. https://doi.org/10.1590/1982-0224-2020-0133
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted May 24, 2021 by Andrea Bialetzki
Accepted May 24, 2021 by Andrea Bialetzki
![]() Submitted November 25, 2020
Submitted November 25, 2020
![]() Epub Sept 17, 2021
Epub Sept 17, 2021