![]() Rigoberto Rosas-Luis1, 2, 3
Rigoberto Rosas-Luis1, 2, 3 ![]() ,
, ![]() Nancy Cabanillas-Terán4 and
Nancy Cabanillas-Terán4 and ![]() Carmen A. Villegas-Sánchez2
Carmen A. Villegas-Sánchez2
PDF: EN XML: EN | Cite this article
Abstract
Kajikia audax, Thunnus albacares, Katsuwonus pelamis, and Auxis spp. occupy high and middle-level trophic positions in the food web. They represent important sources for fisheries in Ecuador. Despite their ecological and economic importance, studies on pelagic species in Ecuador are scarce. This study uses stable isotope analysis to assess the trophic ecology of these species, and to determine the contribution of prey to the predator tissue. Isotope data was used to test the hypothesis that medium-sized pelagic fish species have higher δ15N values than those of the prey they consumed, and that there is no overlap between their δ13C and δ15N values. Results showed higher δ15N values for K. audax, followed by T. albacares, Auxis spp. and K. pelamis, which indicates that the highest position in this food web is occupied by K. audax. The stable isotope Bayesian ellipses demonstrated that on a long time-scale, these species do not compete for food sources. Moreover, δ15N values were different between species and they decreased with a decrease in predator size.
Keywords: Ecuadorian waters, Feeding ecology, Food web, Marine ecology, Pelagic fishes.
Kajikia audax, Thunnus albacares, Katsuwonus pelamis e Auxis spp. ocupam posições tróficas intermedias e/ou elevadas nas cadeias alimentares. Estas espécies representam um importante recurso pesqueiro no Ecuador. Apesar da sua importância económica e ecológica, estudos nestas espécies pelágicas no Ecuador são raras. Este estudo usa isótopos estáveis para avaliar o seu nível trófico de modo a determinar a contribuição das suas presas para os tecidos destes predadores. Dados dos isótopos foram usados para testar a hipótese de que estas espécies de peixes pelágicos possuem valores mais elevados de δ15N do que daqueles das presas consumidas, e que não existe uma sobreposição entre os valores de δ13C e δ15N. Resultados mostram que valores mais elevados de δ15N para K. audax, seguidos por T. albacares, Auxis spp. e K. pelamis, indicam que a posição mais elevada na cadeia alimentar é ocupada por K. audax. Elipses Bayesianas de isótopos estáveis demonstram que, a uma escala de longo-termo, estas espécies de peixes não competem pelos recursos. Adicionalmente, os valores de δ15N são diferentes entre espécies de peixe estudadas e estes valores decrescem com a diminuição do tamanho do predador.
Palavras-chave: Águas Equatorianas, Ecologia alimentar, Cadeia alimentar, Ecologia marinha, Peixes pelágicos.
Introduction
The striped marlin Kajikia audax (Philippi, 1887), the skipjack tuna Katsuwonus pelamis (Linnaeus, 1758) and the yellowfin tuna Thunnus albacares (Bonnaterre, 1788) are pelagic fishes widely distributed in the oceans (Smith, Brown, 2002) and are the most important fishing sources for local and international fishing fleets in Ecuador (Schaefer et al., 2009; Martinez-Ortiz et al., 2015; Tanabe et al., 2017). The capture of these species has an economic value of approximately 73 million US dollars per year for Ecuador (Martínez-Ortiz et al., 2015). These economic gains have promoted the study and development of fisheries, mainly for T. albacares (Martinez-Ortiz et al., 2015). For K. audax, K. pelamis and the small tunas Auxis spp., there is a lack of biological and ecological knowledge for Ecuador. Hence, it is necessary to assess the trophic web to detect shifts or impacts in the ecosystem resulting from the extraction of these species and to establish relationships or differences in their trophic strategies.
The pelagic fishes K. audax, T. albacares, K. pelamis and Auxis spp. are important components in the ecosystem and facilitate energy transfer between low and top trophic levels because they are preyed on by sharks, fishes, seabirds, and marine mammals (Wang et al., 2003; Arizmendi-Rodríguez et al., 2006; Galván-Magaña et al., 2013; Rosas-Luis et al., 2016; Diop et al., 2018). They are also active predators of fishes, cephalopods, and crustaceans (Alverson, 1963; Loor-Andrade et al., 2017; Rosas-Luis et al., 2017; Varela et al., 2017). In addition, these species are efficient transfers of biomass to other areas and water depths since they are fast-moving species that perform horizontal and vertical movements (Holland et al., 1990).
The study of the trophic ecology of sympatric species in marine environments is achieved by using traditional stomach content analysis, and more recently the analysis of stable isotopes of carbon (denoted as δ13C) and nitrogen (denoted as δ15N) (Peterson, Fry, 1987). Stable isotope analysis allows the characterization of migratory movements (Wunder, 2012; Segers, Broders, 2015) and is useful for obtaining information about sympatric species (Vanderklift et al., 2006; Cabanillas-Terán et al., 2016). δ15N is an indicator of a consumer’s trophic position, as the value in consumer tissues becomes higher compared to their prey (McCutchan et al., 2003; Vanderklift, Ponsard, 2003). δ13C values can indicate primary sources in a trophic network (McCutchan et al., 2003). In marine environments, δ13C values indicate the inshore/pelagic versus offshore/benthic contribution to food intake, indicating areas with low and high primary production respectively (Hobson et al., 1994; Cherel, Hobson, 2007; Navarro et al., 2013). The stable isotopes of δ15N and δ13C have been used to study the feeding behavior of large pelagic fishes in the central and north Pacific Ocean (Graham et al., 2007; Acosta-Pachón et al., 2015; Li et al., 2016; Young et al., 2018). In Ecuadorian waters, isotope values showed that sympatric species, such as the billfish Istiophorus platypterus (Shaw, 1792), the blue marlin Makaira nigricans Lacepède, 1802, and the swordfish Xiphias gladius Linnaeus, 1758, do not compet for food sources (Rosas-Luis et al., 2017). The isotope analysis and stomach contents demonstrated that X. gladius consumed prey from deeper waters, while I. platypterus and M. nigricans fed mainly in upper waters (Rosas-Luis et al., 2017). For Thunnus albacares, Varela et al. (2017) found that stable isotope ellipses had no overlap among size classes and suggested that the prey size increases as the tuna grow. These studies allowed the understanding of the food web; however, it is necessary to include K. audax, K. pelamis, and Auxis spp. in the analysis, to better explain the trophic structure of the ecosystem.
Pelagic fishes are important and abundant components in the marine ecosystem of Ecuador (Martínez-Ortiz et al., 2015), but there is a lack of knowledge related to the trophic role of the sympatric species K. audax, K. pelamis, and Auxis spp. Therefore, this study represents an effort to identify the trophic relationships that these species have in the marine ecosystem off the coast of Ecuador, with a main objective to compare the δ15N and δ13C values of each species found in their muscle tissue. Moreover, we aim to explore the hypothesis that medium-sized pelagic fish species have higher δ15N values than those of the prey they consumed, and that the prey consumed are different for each predator. Our results represent the first attempt to study tissue samples of the fishes K. audax, K. pelamis, and Auxis spp. collected in the fishing ports of Ecuador and analyzed by isotopic analysis.
Material and methods
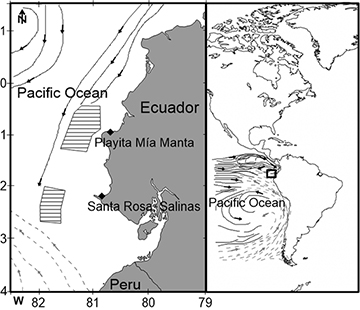
Study area. The marine environment off the coast of Ecuador is characterized by warm waters coming from the Equatorial Current System, with the influence of cold waters from the Humboldt Current System (Bendix, Bendix, 2006). High primary production areas are promoted by the convergence of the two current systems off the coast of Ecuador (Bendix, Bendix, 2006; Rincón-Martínez et al., 2010). Fisheries in Ecuador are characterized by two main groups the longline fishery, targeting large and medium pelagic fishes, and a fishery that uses gillnets to capture cephalopods and other fishes (Martínez-Ortiz et al., 2015). The longline fishery works in areas between 37 and 130 km off the Ecuadorian coast in the pelagic environment of oceanic waters and the gillnets from the shore to 130 km off the Ecuadorian coast (Rosas-Luis et al., 2017).
Samples. Kajikia audax, T. albacares, K. pelamis and the group Auxis spp. were collected from catches brought to the fishing ports of Playita Mía, Manta, Ecuador and Santa Rosa, Salinas, Ecuador during June 2014 and May 2015 (Fig. 1). The total body length (TL) was recorded to the nearest 10 mm. Auxis spp. grouped the frigate tuna, Auxis thazard (Lacepède, 1800), and the bullet tuna, Auxis rochei (Risso, 1810) since separation by morphological characteristics was not possible. Additionally, the Patagonian squid Doryteuthis gahi (d’Orbigny, 1835) and the dart squid Lolliguncula diomedae (Hoyle, 1904) were collected in the same fishing ports. A small portion of the dorsal muscle of the caudal peduncle of fishes and the mantle of squids was extracted and stored at -20°C in the laboratory of trophic ecology at the Universidad Laica Eloy Alfaro de Manabí until lipid extraction and isotopic procedures. Furthermore, prey items, collected from the stomach contents of predators reported by Rosas-Luis et al. (2017), were taken to characterize their values. Samples included complete individuals of the Peruvian anchovy Engraulis ringens Jenyns, 1842, the Peruvian hake Merluccius gayi (Guichenot, 1848), the Reinhardt’s cranch squid Liochranchia reinhardti (Steenstrup, 1856), and the pelagic octopod Japetella sp. (Tab. 1).
FIGURE 1 | Ecuadorian waters in the Pacific Ocean. Polygons indicate areas where artisanal longline fisheries operate. Black lines represent the flux of warm waters from the Equatorial Current System, and gray dotted lines represent the cold waters from the Humboldt Current System.
TABLE 1 | Mean and standard deviation (SD) of the length, δ13C and δ15N values of pelagic fishes and cephalopods sampled in Ecuadorian waters. The length obtained for squids was the dorsal mantle length, and for fishes the total length. * = isotope values were taken from Rosas-Luis et al. (2017); these samples were taken at the same time as those in the current work.
Species
| n | Length (cm) ±SD | δ13C
(°/oo) | δ15N
(°/oo) | ||
Fishes | ||||||
Kajikia
audax | 16 | 274.8 ±31.46 | -16.6 | ±0.23 | 14.9 | ±0.67 |
Thunnus
albacares | 14 | 42.4 ±2.87 | -17.2 | ±0.23 | 13.2 | ±1.06 |
Katsuwonus
pelamis | 30 | 39.4 ±3.20 | -17.3 | ±0.46 | 11.1 | ±1.52 |
Auxis
spp. | 8 | 28.1 ±0.99 | -17.9 | ±0.33 | 11.3 | ±1.20 |
Scomber
japonicus | 5* | 14.1 ±2.12 | -16.9 | ±0.06 | 11.7 | ±0.47 |
Engraulis
ringens | 1 | 15.2 | -16.3 | 12.1 | ||
Lagocephalus
lagocephalus | 1* | 32 | -17.2 | 12.3 | ||
Merluccius
gayi | 1 | 45.1 | -16.3 | 11.4 | ||
Pristigenys
serrula | 1* | 19.4 | -17.5 | 11.0 | ||
Opisthonema
libertate | 5* | 18.0 ±1.50 | -16.2 | ±0.16 | 13.3 | ±0.25 |
Cephalopods | ||||||
Dosidicus
gigas | 20* | 43.4 ±3.34 | -16.0 | ±0.54 | 13.4 | ±1.86 |
Loligunculla
diomedae | 2 | 10 | -16.6 | ±0.71 | 12.8 | ±0.3 |
Ancistrocheirus
lesueurii | 3* | 24.3 ±1.15 | -17.4 | ±0.11 | 12.4 | ± 1.08 |
Liocranchia
reindarthi | 1 | 20 | -16.9 | 12.0 | ||
Tysanoteuthis
rhombus | 1* | 45 | -16.5 | 11.5 | ||
Doryteuthis
gahi | 4 | 20.2 ±0.50 | -15.0 | ±0.15 | 11.1 | ±0.28 |
Japetella
sp. | 1 | 12 | -16.1 | 12.9 | ||
Lipid extraction and isotopic analysis. To avoid biases in the δ13C values, lipid extraction was applied to all tissue samples (Post et al., 2007). Lipids were extracted from all muscle samples with chloroform and methanol following the protocol of Bligh, Dyer (1959). All samples were then freeze-dried and powdered, with 0.3 to 0.4 mg of each sample packed into tin capsules. Isotopic analyses were performed at the Estación Biológica de Doñana, Spain. Samples were combusted at 1,020°C using a continuous flow isotope-ratio mass spectrometer (Thermo Electron) by means of a Flash HT Plus elemental analyzer interfaced with a Delta V Advantage mass spectrometer. Stable isotope ratios were expressed in the standard δ-notation (‰) relative to Vienna Pee Dee Belemnite (δ13C) and atmospheric N2 (δ15N). Based on laboratory standards, the measurement error was ±0.1‰ and ±0.2‰ for δ13C and δ15N, respectively. The standards used were EBD-23 (cow horn, internal standard), LIE-BB (whale baleen, internal standard) and LIE-PA (razorbill feathers, internal standard). These laboratory standards were previously calibrated with international standards supplied by the International Atomic Energy Agency.
Diet. The diet of K. audax was taken from Loor-Andrade et al. (2017) and for T. albacares,diet informationwas based on Varela et al. (2017). These previous reports used samples from the same area and the same sampling time of this work (Tab. 2). Unfortunately, there were no reports on the feeding habits of Auxis spp. and K. pelamis in the study area; thus, for increased clarity, fish diets from outside the study area were used for K. pelamis, based on Tanabe (2001) (Tropical Western Pacific Ocean), and for Auxis spp. based on Siraimeetan (1985) (Tuticorin coast, Gulf of Mannar).
TABLE 2 | Percentage of the index of relative importance (IRI) of pelagic fishes. Data summarized from Kajikia audax followed Loor-Andrade et al. (2017), Thunnus albacore followed Varela et al. (2017), K. pelami followed Tanabe (2001), and Auxis spp. followed Siraimeetan (1985).
%IRI | ||||
Prey/Predator | Thunnus albacares | Kajikia
audax | Auxis spp. | Katsuwonus pelamis |
Auxis spp. | 90.85 | 44.30 | ||
K.
pelamis | 1.90 | |||
Fishes | 5.04 | 37.70 | 39 | |
Cephalopods | 0.16 | 5.40 | 19 | 0.30 |
Crustaceans | 42 | 3.54 | ||
Fish larvae | 96.16 | |||
Trophic width. As a measure of trophic width (Jackson et al., 2011), we calculated the corrected standard ellipse area (SEAc) for K. audax, T. albacares, K. pelamis, and Auxis spp. This metric represents a measure of the total amount of isotopic niche exploited by a predator and is thus a proxy for the extent of the trophic niche exploited by the studied species (high values of SEAc indicate high trophic width) (Jackson et al., 2011). The corrected standard ellipse area (SEAc) based on the Bayesian ellipse area was proposed as an unbiased metric with respect to the sample size, particularly for the Bayesian method, which incorporates a robust comparison considering uncertainty with smaller sample sizes, resulting in larger ellipse areas. The SEAc was calculated by a covariance matrix of the samples. The sample variance provides an unbiased estimate of the population variance for data x and y, that defines their shape and area (Jackson et al., 2011). The SEAc was fitted using R 3.1.0 for Windows (R Development Core Team, 2017). Isotopic standard ellipse areas were calculated using the SIBER package (Jackson et al., 2011) included in the SIAR library, with R 3.1.0 for Windows (R Development Core Team, 2017). The Niche Overlap Metric was calculated as the probability that an individual from the predator species will be found within the niche of the other predator species with an alpha=0.95 using the nicheROVER routine in R (Swanson et al., 2015).
C and N contributions. Kajikia audax, T. albacares, K. pelamis, and the Auxis spp. were used as single species in the isotopic analysis because the number of tissue samples was greater than 7 for each species (Tab. 1). A sample number greater than 7 is considered adequate for posteriori statistical analysis (Jackson et al., 2011).
The Stable Isotope Analysis in R (SIAR) was used to calculate the proportion of δ13C and δ15N isotopes in the diets of the predators (Parnell et al., 2010). The prey species of K. audax and T. albacares were mixed into composite groups to obtain a high number of isotope values despite their different body size (Tab. 1). Groups were established considering that they were consumed by predators (according to previous reports for the area, Tab. 2). The fish group for K. audax and T. albacares was composed of K. pelamis, Auxis spp., O. libertate, Scomber japonicus Houttuyn, 1782, E. ringens, L. lagocephalus, M. gayi, and P. serrula. The cephalopod group for K. audax and T. albacares was composed of L. reinhardti, Japetella sp., Dosidicus gigas, T. rhombus, and A. lesueurii. Unfortunately, we had no muscle tissue of prey consumed by K. pelamis and Auxis spp., thus the contribution of δ13C and δ15N for these species was not calculated. The Trophic Discrimination Factor (TDF) used was 1.9 ± 0.4 for δ15N and 1.8 ± 0.3 for δ13C related to Pacific bluefin tuna (Madigan et al., 2012), which were the most appropriate for the species in this work.
Statistical analysis. Size, δ13C and δ15N differences between species were tested using one-way ANOVA tests, and significant differences (p≤ 0.05) between pairs of species were identified with a post hoc Tukey test. All tests were performed in the IBM SPSS statistics software v.19 (IBM, 2010).
Results
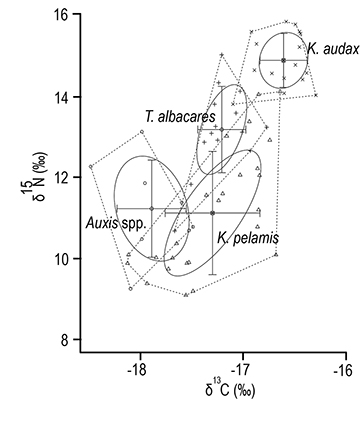
Isotope values. The mean δ13C values of K. audax, T. albacares. K. pelamis and Auxis spp. ranged between -17.9‰ and -16.6‰ (Tab. 1). The mean δ13C value of K. audax was -16.6‰, higher than those of T. albacares, K. pelamis, and Auxis spp. (F3,59 = 25.69, p < 0.05). T. albacares and K. pelamis had similar δ13C values (Tabs. 1–3). The mean δ15N values ranged between 11.1‰ and 14.9‰ (Tab. 1). Significant differences in the δ15N values were found between species (F3,59 = 35.73, p < 0.05) (Tabs. 1–3) with K. audax showing the highest values. The post hoc Tukey test showed similar δ15N values for K. pelamis and Auxis spp. (Tab. 3).
TABLE 3 | Results of the Tukey post hoc test for the comparison of δ13C and δ15N values among groups and species and overlap probability. Bold numbers are significant values.
Group | Group | p value | Overlap probability | |
δ13C | δ15N | |||
Kajikia
audax | Thunnus
albacares | 0.00 | 0.00 | 31.9 |
Katsuwonus
pelamis | 0.00 | 0.00 | 35.6 | |
Auxis spp. | 0.00 | 0.00 | 0.4 | |
Thunnus
albacares | Katsuwonus
pelamis | 0.87 | 0.00 | 71.8 |
Auxis spp. | 0.00 | 0.00 | 23.4 | |
Katsuwonus
pelamis | Auxis spp. | 0.00 | 0.99 | 46.1 |
Trophic width. The broadest SEAc was observed for K. pelamis (1.6), followed by Auxis spp. (1.4) (Fig. 2). Narrow SEAcS were recorded for K. audax (0.51) and T. albacares (0.7). A high overlap probability was found between T. albacares and K. pelamis (71.8%) (Tab. 3; Fig. 2). Moderate overlap probability was found among K. audax, T. albacares and K. pelamis, T. albacares and Auxis spp., and K. pelamis and Auxis spp. (Tab. 3; Fig. 2). Low overlap probability was found between K. audax and Auxis spp. (0.4%) (Tab. 3; Fig. 2).
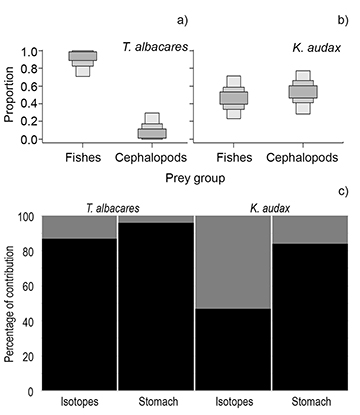
δ13C and δ15N contribution of prey groups in the diet. The results of the SIAR analysis showed thatfishes were the most important δ13C and δ15N contributors (up to 87%) for T. albacares, while cephalopods were the most important contributors for K. audax (up to 53%)(Fig. 3). The summary of the diet reports based on stomach contents indicated that fishes represent 95.8% of the diet of T. albacares and 83.9% of the diet of K. audax, with cephalopods being the second most represented group, but significantly less important (4.2 and 16.1%, respectively; Fig. 3).
FIGURE 2 | Mean and standard deviation of δ15N and δ13C values and corrected standard ellipse areas (SEAc) based on δ13C and δ15N values of pelagic fish species off the coast of Ecuador. Symbols represent the individual organisms: triangles are for Katsuwonu pelamis, circles are for Auxis spp., crosses are for Thunnus albacares and X´s are for Kajikia audax.
FIGURE 3 | Modeled proportion of prey groups in the diet of A. Thunnus albacares and B. Kajikia audax obtained using a stable isotope analysis in R. C. is the comparison between the mean proportional contribution of fish (black area) and cephalopods (gray area) to the diets of T. albacares and K. audax, left bars were based on δ15N and δ13C isotopes, and right bars were based on stomach content identification reported by Varela et al. (2017) for T. albacares, and Loor-Andrade et al. (2017) for K. audax.
Discussion
In this study, isotope analysis allowed the identification of the trophic width and overlap between Auxis spp., K. pelamis, T. albacares, and K. audax. The ellipse metrics provided quantitative and integrated information about sources and niche breadth (Boecklen et al., 2011), contributing to the ecological knowledge of pelagic and commercial species in the marine ecosystem of Ecuador. The δ13C values suggest that K. audax has a different trophic strategy, probably consuming prey from a trophic chain based in high productivity areas, while Auxis spp. may be moving to low productivity areas and consuming different prey sources. T. albacares and K. pelamis had similar δ13C values, thus indicating that they coexist in the same areas. Based on these results, the discussion is focused on explaining the trophic strategy and interactions of these sympatric species.
The highest δ13C values were recorded for K. audax, followed by T. albacares and K. pelamis. On the one hand, the highest δ13C values were related to high productivity ocean areas (France, Peters, 1997; Ménard et al., 2007; Carlisle et al., 2014), coinciding with marine areas where fishing activity in Ecuador occurs (Martinez-Ortiz et al., 2015). These species have been described as fishery sources with high abundances in waters where upwelling events favor the enrichment of primary production, such as the Humboldt Current and the Gulf of California (Stock et al., 2017). For the Ecuadorian waters, these species are usually found in catches close to the coast (Martinez-Ortiz et al., 2015). On the other hand, low values of δ13C in Auxis spp., compared to those of the other species, could be the result of feeding habits related to pelagic and open waters, affecting the signal in the muscle samples. Auxis spp. and other scombrid fishes, including T. albacares and K. pelamis, are fast-moving species in interior as well as more distant coastal waters (Holland et al., 1990; Schaefer et al., 2009), resulting in a different feeding strategy consuming small pelagic fishes, such as S. japonicus and myctophids, and pelagic crustaceans (Varela et al., 2017), that could be available in and outside of the studied area. The trophic width as indicated by the SEAc showed that the fishes Auxis spp. and K. pelamis had broader isotopic ellipse areas, and that K. audax had the narrowest area. The consumption of similar prey by predators was confirmed by the isotope values and niche overlap probability between Auxis spp. and K. pelamis (46%). Nevertheless, it is necessary to identify the potential prey of these species. If they are voracious and active predators, the results will show a wide range of prey as observed in squids of similar size (Rosas-Luis et al., 2014).
The highest δ15N values were recorded for K. audax and T. albacares, and the lowest were found in K. pelamis and Auxis spp. These values agree with the assumption that the increase in δ15N values results from prey ingestion, because the type and size of prey consumed affect the δ15N values in the predator tissue. The consumption of large prey increases the δ15N values (Post, 2002; Hussey et al., 2014). The largest predator in this study was K. audax, which consumes large prey such as K. pelamis and Auxis spp. (Loor-Andrade et al., 2017). The δ15N values allowed the comparison of K. audax with top predators and T. albacares with mid-level predators, which corresponds to the trophic position calculated for T. albacares in Ecuadorian waters (Varela et al., 2017) and for K. audax in the north Pacific Ocean (Torres-Rojas et al., 2013). The lowest level position was found in K. pelamis and Auxis spp.
As top predators, K. audax and T. albacares segregate from the other species, as suggested by the stomach contents and isotope results. They share food resources, with K. audax feeding mainly on Auxis spp. and other fishes and cephalopods, and T. albacares feeding mainly on Auxis spp. (Varela et al., 2017). For T. albacares and K. pelamis, a higher overlap probability was recorded; thus, it can be suggested that these species also share food resources in Ecuadorian waters. However, a comparison of the δ15N values indicates that T. albacares had higher δ15N values than K. pelamis. Thus, they could consume prey that are located in the same area, but of different sizes (large prey for T. albacares), as was reported for these species in the Gulf of California (Alatorre-Ramírez et al., 2017). The isotope and stomach content results are complementary because the isotope values support the evidence of prey contribution, taking into account the turnover rate of muscle tissue (several months) (Madigan et al., 2012; Vander-Zanden et al., 2015) and the estimated diet with stomach content identification, hours or days depending on the prey tissue (Olson, Boggs, 1986; Acosta-Pachón, Ortega-García, 2019). Thus, these results highlight the importance of Auxis spp. in the diets of T. albacares and K. audax, and cephalopods in the diet of K. audax. In Ecuadorian waters, K. audax and T. albacares seem to be opportunistic predators that feed on available and abundant species. The fishes Auxis spp. could be abundant in the area because they have been reported as components in the diet of top predators (Loor-Andrade et al., 2017; Rosas-Luis et al., 2017; Varela et al., 2017). More descriptions of the feeding habits of marine species are needed to corroborate the trophic relationships in the ecosystem, and other species such as Auxis spp. and K. pelamis should be included in the analysis.
In conclusion, our results suggest that the δ13C values of T. albacares and K. pelamis overlap, indicating that they share similar foraging areas or a similar trophic strategy. Their δ15N values allowed the categorization of the food web; the highest position in the food web was occupied for the large species K. audax and middle trophic positions for T. albacares, K. pelamis, and Auxis spp. confirming the hypothesis that medium-sized pelagic fish species accumulate δ15N isotopes according to the size of prey consumed (large predators consumed larger prey than medium-sized predators). In addition, the different predator size allows the use of the same habitat by partitioning in the prey consumed by each predator. Considering these results and the fact that the fishes K. audax, T. albacares and K. pelamis are important for fisheries in Ecuador, it is necessary to identify the impact that fisheries have on natural populations. The SEAc of Auxis spp. could be related to our analysis of two species as a single group, which likely do not have similar feeding habits. Unfortunately, it was not possible to segregate the two species during the morphological identification, and no stomach content samples were taken. Thus, future research will require the use of genetic and morphological methods to separate the two species and to continue trophic ecology studies of all species caught in fisheries to better understand the food web of the marine ecosystem.
Acknowledgments
The authors thank all students and fishermen for their support during the field procedures and Ricardo A. Álvarez and Susana Carrasco of the LIE Doñana for their support in the isotope analysis. Sarah Young corrected the English grammar. These results were part of project 91740000.0000.376985 “Trophic Ecology of the Large Pelagic Species of the Ecuadorian Pacific”, which was conducted from 2014 to 2015 at the Central Department of Research of the Universidad Laica Eloy Alfaro de Manabí, Ecuador.
References
Acosta-Pachón TA, Ortega-García S, Graham B. Stable carbon and nitrogen isotope values of dorsal spine age rings indicate temporal variation in the diet of striped marlin (Kajikia audax) in waters around Cabo San Lucas, Mexico. Rapid Commun Mass Sp. 2015; 29(18):1676–86. https://doi.org/10.1002/rcm.7271
Acosta-Pachón TA, Ortega-García S. Trophic interaction between striped marlin and swordfish using different timescales in waters around Baja California Sur, Mexico. Mar Biol Res. 2019; 15(1):97–112. https://doi.org/10.1080/17451000.2019.1578377
Alatorre-Ramírez VG, Galván-Magaña F, Torres-Rojas YE, Olson RJ. Trophic segregation of mixed schools of yellowfin tuna (Thunnus albacares) and skipjack tuna (Katsuwonus pelamis) caught in the eastern tropical Pacific Ocean. Fish Bull. 2017; 115(2):252–68. https://doi.org/10.7755/FB.115.2.11
Alverson FG. The food of yellowfin and skipjack tunas in the eastern tropical Pacific Ocean. Inter-American Tropical Tuna Commision Bulletin. 1963; 7:295–396.
Arizmendi-Rodríguez DI, Abitia-Cárdenas L, Galván-Magaña F, Trejo-Escamilla I. Food habits of sailfish Istiophorus platypterus off Mazatlán, Sinaloa, Mexico. Bull Mar Sci. 2006; 79(3):777–91.
Bendix A, Bendix J. Heavy rainfall episodes in Ecuador during El Niño events and associated regional atmospheric circulation and SST patterns. Adv Geosci. 2006; 6:43–49. https://doi.org/10.5194/adgeo-6-43-2006
Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Phys. 1959; 37:911–17.
Boecklen WJ, Yarnes CT, Cook BA, James AC. On the use of stable isotopes in trophic ecology. Annu Rev Ecol Evol S. 2011; 42:411–40. https://doi.org/10.1146/annurev-ecolsys-102209-144726
Cabanillas-Terán N, Loor-Andrade P, Rodríguez-Barreras R, Cortés J. Trophic ecology of sea urchins in coral-rocky reef systems, Ecuador. PeerJ. 2016; 4:e1578. http://dx.doi.org/10.7717/peerj.1578
Carlisle AB, Goldman KJ, Litvin SY, Madigan DJ, Bigman JS, Swithenbank AM, Kline TC, Block BA. Stable isotope analysis of vertebrae reveals ontogenetic changes in habitat in an endothermic pelagic shark. Proc R Soc Ser B-Bio. 2014; 282:20141446 http://dx.doi.org/10.1098/rspb.2014.1446
Cherel Y, Hobson KA. Geographical variation in carbon stable isotope signatures of marine predators: a tool to investigate their foraging areas in the Southern Ocean. Mar Ecol-Prog Ser. 2007; 329:281–87. http://dx.doi.org/10.3354/meps329281
Diop N, Zango L, Beard A, Ba CT, Ndiaye PI, Henry L, Clingham E, Oppel S, González-Solís J. Foraging ecology of tropicbirds breeding in two contrasting marine environments in the tropical Atlantic. Mar Ecol-Prog Ser. 2018; 607:221–36. https://doi.org/10.3354/meps12774
France RL, Peters RH. Ecosystem differences in the trophic enrichment of 13C in aquatic food webs. Can J Fish Aquat Sci. 1997; 54:1255–58.
Galván-Magaña F, Polo-Silva C, Hernández-Aguilar SB, Sandoval-Londoño A, Ochoa-Diaz MR, Aguilar-Castro N, Castañeda-Suárez D, Chávez-Costa AC, Baigorrí-Santacruz A, Torres-Rojas YE, Abítia-Cárdenas LA. Shark predation on cephalopods in the Mexican and Ecuadorian Pacific Ocean. Deep-Sea Res II. 2013; 95:52–62. https://doi.org/10.1016/j.dsr2.2013.04.002
Graham BS, Grubbs D, Holland K, Popp BN. A rapid ontogenetic shift in the diet of juvenile yellowfin tuna from Hawaii. Mar Biol. 2007; 150:647–58. https://doi.org/10.1007/s00227-006-0360-y
Hobson KA, Piatt JF, Pitocchelli J. Using stable isotopes to determine seabird trophic relationships. J Anim Ecol. 1994; 63(4):786–98. https://doi.org/10.2307/5256
Holland KN, Brill RW, Chang RKC. Horizontal and vertical movements of yellowfin and bigeye tuna associated with fish aggregating devices. Fish Bull. 1990; 88(3):493–507.
Hussey NE, MacNeil MA, McMeans BC, Olin JA, Dudley SF, Cliff G, Wintner SP, Fennessy ST, Fisk AT. Rescaling the trophic structure of marine food webs. Ecol Lett. 2014; 17(2):239–50. https://doi.org/10.1111/ele.12226
IBM Corp. Released IBM SPSS Statistics for Windows, Version 19.0. Armonk, NY: IBM Corp. 2010. Available from: https://www.ibm.com/products
Jackson AL, Inger R, Parnell AC, Bearhop S. Comparing isotopic niche widths among and within communities: SIBER – stable isotope bayesian ellipses in R. J Anim Ecol. 2011; 80(3):595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x
Li Y, Zhang Y, Dai X. Trophic interactions among pelagic sharks and large predatory teleosts in the northeast central Pacific. J Exp Mar Biol Ecol. 2016; 483:97–103. http://dx.doi.org/10.1016%2Fj.jembe.2016.04.013
Loor-Andrade P, Pincay-Espinoza J, Carrera-Fernández M, Rosas-Luis R. Feeding habits of billfishes (Carangaria: Istiophoriformes) in the Ecuadorian Pacific Ocean. Neotropical Ichthyology. 2017; 15:e160162. https://doi.org/10.1590/1982-0224-20160162
Madigan DJ, Litvin SY, Popp BN, Carlisle AB, Farwell CJ, Block BA. Tissue turnover rates and isotopic trophic discrimination factors in the endothermic teleost, Pacific bluefin tuna (Thunnus orientalis). PLoS ONE. 2012; 7:e49220. https://doi.org/10.1371/journal.pone.0049220
Martínez-Ortiz J, Aires-da-Silva AM, Lennert-Cody CE, Maunder MN. The Ecuadorian artisanal fishery for large pelagics: species composition and spatio-temporal dynamics. PLoS ONE. 2015; 10:e0135136. https://doi.org/10.1371/journal.pone.0135136
McCutchan JH, Lewis WM, Kendall C, McGrath CC. Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulphur. Oikos. 2003; 102(2):378–90. https://doi.org/10.1034/j.1600-0706.2003.12098.x
Ménard F, Lorrain A, Poiter M, Marsac F. Isotopic evidence of distinct feeding ecologies and movement patterns in two migratory predators (yellowfin tuna and swordfish) of the western Indian Ocean. Mar Biol. 2007; 153(2):141–52. https://doi.org/10.1007/s00227-007-0789-7
Navarro J, Coll M, Somes C, Olson RJ. Trophic niche of squids: insights from isotopic data in marine systems worldwide. Deep-Sea Res Part II. 2013; 95:93–102. https://doi.org/10.1016/j.dsr2.2013.01.031
Olson RJ, Boggs CH. Apex predation by yellowfin tuna (Thunnus albacares): independent estimates from gastric evacuation and stomach contents, bioenergetics, and cesium concentrations. Can J Fish Aquat Sci. 1986; 43(9):1760–75. https://doi.org/10.1139/f86-220
Parnell AC, Inger R, Bearhop S, Jackson AL. Source partitioning using stable isotopes: coping with too much variation. PLoS ONE. 2010; 5:e9672. https://doi.org/10.1371/journal.pone.0009672
Peterson BJ, Fry B. Stable isotopes in ecosystem studies. Ann Rev Ecol S. 1987; 18:293–320.
Post DM. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology. 2002; 83(3):703–18. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2
Post DM, Layman CA, Arrington DA, Takimoto G, Quattrochi J, Montaña CG. Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia. 2007; 152:179–89. https://doi.org/10.1007/s00442-006-0630-x
R Development Core Team. R: a language and environment for statistical computing. R foundation for statistical computing, Vienna. 2017. Available from: https://www.r-project.org/
Rincón-Martínez D, Lamy F, Contreras S, Ledu G, Bard E, Saukel C, Blanz T, Mackensen A, Tiedemann R. More humid interglacials in Ecuador during the past 500 kyr linked to latitudinal shifts of the equatorial front and the intertropical convergence zone in the Eastern tropical Pacific. Paleoceanography. 2010; 25:PA2210. https://doi.org/10.1029/2009PA001868
Rosas-Luis R, Sánchez P, Portela JM, del Rio JL. Feeding habits and trophic interactions of Doryteuthis gahi, Illex argentinus and Onykia ingens in the marine ecosystem off the Patagonian Shelf. Fish Res. 2014; 152:37–44. https://doi.org/10.1016/j.fishres.2013.11.004
Rosas-Luis R, Loor-Andrade P, Carrera-Fernández M, Pincay-Espinoza JE, Vinces-Ortega C, Chompoy-Salazar L. Cephalopod species in the diet of large pelagic fish (sharks and billfishes) in Ecuadorian waters. Fish Res. 2016; 173:159–68. https://doi.org/10.1016/j.fishres.2015.07.002
Rosas-Luis R, Navarro J, Loor-Andrade P, Forero MG. Feeding ecology and trophic relationships of pelagic sharks and billfishes coexisting in the central eastern Pacific Ocean. Mar Ecol Prog Ser. 2017; 573:191–201. https://doi.org/10.3354/meps12186
Schaefer KM, Fuller DW, Block BA. Vertical movements and habitat utilization of skipjack (Katsuwonus pelamis), yellowfin (Thunnus albacares), and bigeye (Thunnus obesus) tunas in the Equatorial Eastern Pacific Ocean, ascertained through archival tag data. In: Nielsen JL, Arrizabalaga H, Fragoso N, Hobday A, Lutcavage M, Sibert J, editors. Tagging and tracking of marine animals with electronic devices. Reviews: methods and technologies in fish biology and fisheries. Vol. 9. New York (NY): Springer; 2009; p.121–44.
Segers JL, Broders HG. Carbon (δ13C) and Nitrogen (δ15N) Stable Isotope Signatures in Bat Fur Indicate Swarming Sites Have Catchment Areas for Bats from Different Summering Areas. PLoS ONE. 2015; 10(4):e0125755. https://doi.org/10.1371/journal.pone.0125755
Siraimeetan P. On the occurrence, size, distribution, morphometry and feeding habits of the juveniles of Euthynnus affinis (cantor), Auxis thazard (lacepede), and Sarda orientalis (temminck and schlegel), along the Tuticorin coast, Gulf of Mannar. Cent Mar Fish Res Institute Bull. 1985; 36:104–216.
Smith KF, Brown JH. Patterns of diversity, depth range and body size among pelagic fishes along a gradient of deep. Global Ecol Biogeogr. 2002; 11(4):313–22. http://www.jstor.org/stable/3182700
Stock CA, John JG, Rykaczewski RR, Asch RG, Cheung WW, Dunne JP, Friedland KD, Lam VW, Sarmiento JL, Watson RA. Reconciling fisheries catch and ocean productivity. PNAS. 2017; 114(8):E144–49. https://doi.org/10.1073/pnas.1610238114
Swanson HK, Lysy M, Power M, Stasco AD, Johnson JD, Reist JD. A new probabilistic method for quantifying n-dimensional ecological niches and niche overlap. Ecology. 2015; 96(2):318–24. https://doi.org/10.1890/14-0235.1
Tanabe T. Feeding habits of skipjack tuna Katsuwonus pelamis and other tuna Thunnus spp. juveniles in the tropical western Pacific. Fish Sci. 2001; 67:563–70.
Tanabe T, Kiyofuji H, Shimizu Y, Ocura M. Vertical distribution of juvenile skipjack tuna Katsuwonus pelamis in the Tropical Western Pacific Ocean. JARQ. 2017; 51(2):181–89. https://doi.org/10.6090/jarq.51.181
Torres-Rojas Y, Hernandez-Herrera A, Ortega-Garcia S, Domeier M. Stable isotope differences between blue marlin (Makaira nigricans) and the striped marlin (Kajikia audax) in the southern Gulf of California, Mexico. Bull Mar Sci. 2013; 89(2):421–36. http://dx.doi.org/10.5343/bms.2011.1105
Vander-Zanden MJ, Clayton MK, Moody EK, Solomon CT, Weidel BC. Stable isotope turnover and half-life in animal tissues: a literature synthesis. PLoS ONE. 2015; 10:e0116182. https://doi.org/10.1371/journal.pone.0116182
Vanderklift A, Ponsard S. Sources of variation in consumer-diet δ15N enrichments: a meta-analysis. Oecologia. 2003; 136:169–82.
Vanderklift MA, Kendrick GA, Smith AJ. Differences in trophic position among sympatric sea urchin species. Estuar Coast Shelf S. 2006; 66(1–2):291–97. https://doi.org/10.1016/j.ecss.2005.09.004
Varela JL, Intriago KM, Floresa JC, Lucas-Pilozo CR. Feeding habits of juvenile yellowfin tuna (Thunnus albacares) in Ecuadorian waters assessed from stomach content and stable isotope analysis. Fish Res. 2017; 194:89–98. http://dx.doi.org/10.1016/j.fishres.2017.05.017
Wang M, Walker WA, Shao K, Chou L. Feeding habits of the pantropical spotted dolphin, Stenella attenuata, off the Eastern coast of Taiwan. Zool Stud. 2003; 42:368–78.
Wunder MB. Determining geographic patterns of migration and dispersal using stable isotopes in keratins. J Mammal. 2012; 93(2):360–67. https://doi.org/10.1644/11-MAMM-S-182.1
Young T, Pincin J, Neubauer P, Ortega-García S, Jensen OP. Investigating diet patterns of highly mobile marine predators using stomach contents, stable isotope, and fatty acid analyses. ICES J Mar Sci. 2018; 75:1583–90. https://doi.org/10.1093/icesjms/fsy025
Authors
![]() Rigoberto Rosas-Luis1, 2, 3
Rigoberto Rosas-Luis1, 2, 3 ![]() ,
, ![]() Nancy Cabanillas-Terán4 and
Nancy Cabanillas-Terán4 and ![]() Carmen A. Villegas-Sánchez2
Carmen A. Villegas-Sánchez2
[1] Instituto Brasileiro de Geografia e Estatística – ReservCátedras CONACYT -Tecnológico Nacional de México/I. T. Chetumal. Av. Insurgentes, 330, Col. David Gustavo Gtz., 77013 Chetumal, Quintana Roo, Mexico. riroluis@yahoo.com.mx, rigoberto.rl@chetumal.tecnm.mx (corresponding author).
[2] Tecnológico Nacional de México/I. T. Chetumal. Av. Insurgentes, 330, Col. David Gustavo Gtz., 77013 Chetumal, Quintana Roo, Mexico. (CAVS) cavs005@gmail.com.
[3] Departamento Central de Investigación, Universidad Laica Eloy Alfaro de Manabí, Manta 130802, Ecuador.
[4] El Colegio de la Frontera Sur. Av. Centenario Km 5.5, 77014 Chetumal, Quintana Roo, Mexico. ncabanillas@ecosur.mx.
Authors Contribution 
Rigoberto Rosas-Luis: Conceptualization, Formal analysis, Investigation, Methodology, Resources, Writing-original draft, Writing-review and editing.
Nancy Cabanillas-Terán: Conceptualization, Methodology, Validation, Writing-review and editing.
Carmen A. Villegas-Sánchez: Formal analysis, Methodology, Writing-review and editing.
Ethical Statement
Not applicable.
Competing Interests
The authors declare no competing interests.
How to cite this article
Rosas-Luis R, Cabanillas-Terán N, Villegas-Sánchez CA. Stable isotope analysis reveals partitioning in prey use by Kajikia audax (Istiophoridae), Thunnus albacares, Katsuwonus pelamis, and Auxis spp. (Scombridae) in the Eastern Tropical Pacific of Ecuador. Neotrop Ichthyol. 2021; 19(4):e200015. https://doi.org/10.1590/1982-0224-2020-0015
Copyright
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Distributed under
Creative Commons CC-BY 4.0

© 2021 The Authors.
Diversity and Distributions Published by SBI
![]() Accepted September 10, 2021 by Gerson Araújo
Accepted September 10, 2021 by Gerson Araújo
![]() Submitted March 23, 2020
Submitted March 23, 2020
![]() Epub Dec 01, 2021
Epub Dec 01, 2021